Abstract
Background
The c-Met signaling pathway regulates a variety of biological processes, including proliferation, survival and migration. Deregulated c-Met activation has been implicated in the pathogenesis and prognosis of many human malignancies. We studied the function and prognostic significance of c-Met and hepatocyte growth factor protein expression in patients with classical Hodgkin’s lymphoma.
Design and Methods
Expression of c-Met and its ligand, hepatocyte growth factor, were determined by immunohistochemistry. Prognostic values were defined in cohorts of German and Dutch patients with classical Hodgkin’s lymphoma. Functional studies were performed on Hodgkin’s lymphoma cell lines.
Results
Expression of c-Met was detected in the tumor cells of 52% (80/153) of the patients and expression of its ligand, hepatocyte growth factor, in 8% (10/121) of the patients. c-Met expression correlated with a 5-year freedom from tumor progression of 94%, whereas lack of expression correlated with a 5-year freedom from tumor progression of 73% (P<0.001) in the combined cohort. In multivariate analysis both c-Met (hazard ratio 5.0, 95% confidence interval 1.9–13.3, P<0.001) and stage (hazard ratio 2.8, 95% confidence interval 1.2–6.4, P=0.014) were independent predictors for freedom from tumor progression. In functional studies activation with hepatocyte growth factor did not affect cell growth, while the c-Met inhibitor SU11274 suppressed cell growth by inducing G2/M cell cycle arrest.
Conclusions
Although functional studies showed an oncogenic role of the hepatocyte growth factor/c-Met signaling pathway in cell cycle progression, expression of c-Met in tumor cells from patients with classical Hodgkin’s lymphoma strongly correlated with a favorable prognosis in two independent cohorts.
Keywords: c-Met, classical Hodgkin’s lymphoma, hepatocyte growth factor, prognosis
Introduction
Hodgkin’s lymphoma is a B-cell neoplasm characterized by a minority of neoplastic cells within an extensive infiltrate of reactive cells. There is a classical form and a nodular lymphocyte predominant variant. The tumor cells in classical Hodgkin’s lymphoma (cHL), the so-called Hodgkin and Reed-Sternberg (HRS) cells, are derived from pre-apoptotic germinal center B cells that acquire crippling immunoglobulin gene mutations and/or lose their capacity to express a high affinity B-cell receptor.1 Epstein-Barr virus (EBV) infection, constitutive activation of nuclear factor κB and aberrant activation of receptor tyrosine kinases contribute to the survival and proliferation of HRS cells.2,3
The prognosis of cHL patients improved greatly over the last three decades and current treatment regimens have reduced treatment failure to less than 20% even in advanced stages.4 However, long-term toxicities and especially secondary malignancies are major concerns and the main challenge in cHL is to avoid overtreatment in individual patients. The Ann Arbor staging system is used to classify cHL into early or advanced stage disease and treatment is tailored accordingly. In addition, several other clinicopathological factors are used for risk stratification.5 Although some prognostic tissue markers, such as number of tumor-infiltrating macrophages,6 HLA class II expression by tumor cells7 and tumor cell EBV status,8 have been published, there is currently no pre-treatment cHL prognostic factor that is applied for prediction of treatment response in a clinical setting.
The hepatocyte growth factor (HGF)/c-Met signaling pathway regulates a variety of biological processes, including proliferation, survival and migration.9 Deregulated c-Met activation, caused by gene amplification, translocation, mutation or autocrine/paracrine HGF signaling, has been implicated in the pathogenesis of many human cancers.10 Furthermore, c-Met is of prognostic significance in numerous malignancies, such as diffuse large B-cell lymphoma,11 bladder cancer,12 breast cancer,13 colorectal cancer14 and ovarian cancer.15 Inhibitors have been developed to target this signaling pathway and one of these, SU11274,16 shows effective inhibition of the c-Met signaling pathway, thereby affecting the survival and growth of lung cancer,17 mesothelioma,18 and melanoma19 cell lines.
c-Met expression has been reported in HRS cells in tissue samples from 33% (6/18)20 and 100% (n=57)21 of cHL patients. HGF expression was observed in infiltrating cells, especially in dendritic cells, but not in tumor cells.21 Additionally, both c-Met and HGF are expressed in some cHL cell lines.22,23 We studied the function of the HGF/c-Met pathway in cHL cell lines and determined the prognostic value of c-Met expression in two independent cohorts of patients.
Design and Methods
Patients and tissue data
Primary cHL tissues were retrieved from the Department of Pathology, University Medical Center Groningen, the Netherlands (n=62, collected from 1993 up to 2009) and from the Institute of Pathology, University of Ulm, Germany (n=91, collected from 1994 up to 2009).
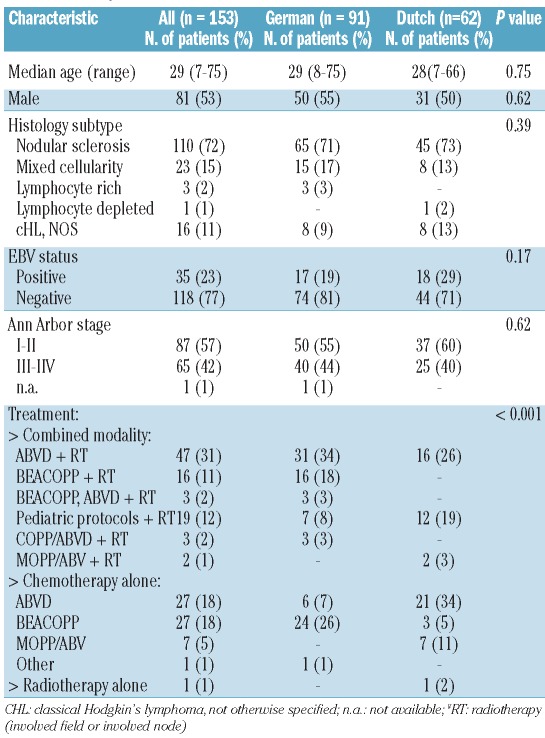
Almost all patients were treated and staged according to pediatric or adult clinical trial protocols (EORTC or GHSG). The basic characteristics of the German and the Dutch patients are presented in Table 1. Information on the presence of B symptoms was not available for two patients and data were lacking on the presence of bulky disease for four patients; these patients were excluded from the respective analyses. The International Prognostic Score (IPS), which incorporates information on age, stage, sex, hemoglobin and albumin levels and leukocyte and lymphocyte counts, was applied only in patients with advanced stage (stages III and IV). The IPS was unknown for four patients and these patients were excluded from the comparisons between c-Met expression and IPS. No significant differences regarding age at diagnosis, sex, histological subtype, stage of disease and EBV status were observed between the cohorts. The median follow-up was 61 months for the German cohort (interquartile range, 39–87 months) and 44 months for the Dutch cohort (interquartile range, 22–83 months). The German cohort differed significantly from the Dutch cohort by treatment modality (more German patients received the BEA-COPP regimen and combined modality treatment was more often used) (Table 1). The study protocol was consistent with international ethical and professional guidelines (the Declaration of Helsinki and the International Conference on Harmonization Guidelines for Good Clinical Practice).
Table 1.
Basic characteristics of the German, Dutch and total combined cohorts of patients. The P value refers to the difference between the German and the Dutch patients.
Immunohistochemistry
Immunohistochemistry (IHC) was performed with antibodies against c-Met (C-28) (Santa Cruz Biotechnology, Santa Cruz, CA, USA), HGF (R&D Systems, Minneapolis, MN, USA) and CD30 (Dako, Glostrup, Denmark) on paraffin-embedded tissue sections after antigen retrieval (pH 9). Staining was visualized using horse-radish peroxidase-labeled secondary antibodies and 3,3′-diaminobenzidine (Sigma Aldrich, St Louis, MO, USA). Appropriate positive and negative controls were performed for each staining. The Dutch cases were stained and scored on a tissue micro-array (TMA) or on whole tissue sections and the German cases were scored on whole tissue sections. For c-Met all 153 cases were stained and for HGF all German cases and the Dutch TMA were stained (121 cases). Each case in the TMA was represented by two tissue cores and cases were scored only if at least ten tumor cells were present in both cores. CD30 expression was used to identify the tumor cells. The median number of tumor cells for each case was 30. Only staining of the tumor cells was scored. The distribution of percentages of c-Met-positive tumor cells showed a distinction at 30% (20% or less: 72/153 and 30% or more: 80/153). We hypothesized that this distinction reflects two biologically different subsets and a cut-off of ≥ 30% positive HRS cells was chosen. The percentage of HGF-positive cells for most patients was 10% or less (94/121) and a cut-off of ≥ 20% of positive HRS cells was chosen.
Cell lines
The cHL cell lines L428, L1236, KMH224 and U-HO125 were cultured in RPMI-1640 medium supplemented with ultraglutamine-1, 100 U/mL penicillin/streptomycin and 10% fetal calf serum (5% for the L428 cell line) (Lonza Walkersville, Walkersville, MD, USA).
Enzyme-linked immunosorbent assay on the supernatant of cultures of classical Hodgkin’s lymphoma cell lines
Levels of HGF protein were measured in cell culture supernatants by an enzyme-linked immunosorbent assay according to the protocol provided by the manufacturer (R&D Systems).
Quantitative reverse transcriptase polymerase chain reaction
RNA was extracted using Trizol® (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s protocol. cDNA was made from 500 ng of total RNA in 20 μL reactions using Superscript II and random primers (Invitrogen). Two nanograms of cDNA were used in the quantitative reverse transcriptase polymerase chain reaction (qRT-PCR) in triplicate using Sybergreen (for HGF) and probe (for U6), as described by the manufacturer (Applied Biosystems, Foster City, CA, USA). The primer sequences used for the amplification were as follows: U6 forward primer: 5’-ttcg-gcagcacatatactaa-3’ and reverse primer 5’-aatatggaacgcttcacgaa-3’; U6 probe: 5’-ccctgcgcaaggatgaca-3‘, HGF forward primer (exon 5): 5’-caatccagaggtacgctacgaa-3’ and reverse primer (exon 6) 5’-actctc-cccattgcaggtcat-3’. U6 was used for normalization (ΔCt = CtHGF -CtU6). Relative expression levels of HGF were determined using the formula 2-ΔCt.
Western blot
Cell lysates were separated on polyacrylamide gels and electroblotted onto nitrocellulose membranes using standard protocols. Blots were incubated with primary antibodies, c-Met (C12, Santa Cruz), p-Met (Tyr1234/1235), p-p44/42 MAPK (Thr202/Tyr204) (20G11), and p-Akt (Ser473) (D9E) (Cell Signaling Technology, Boston MA, USA), at 4ºC overnight. Immunostaining was amplified by incubation with horseradish peroxidase-conjugated antibodies and chemiluminescence was detected with ECL (Pierce, Rockford, USA).
MTT assay
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Sigma Aldrich) was added to cells and incubated for 4 h at 37°C. The cells were resolved in dimethylsulfoxide (Sigma Aldrich) and absorption was measured at 540 nm.
Cell cycle analysis
Hypotonic DNA staining buffer (0.1% sodium citrate; 0.3% Triton–X 100; 0.01% propidium iodide, 0.002% ribonuclease A) was added to the cells and mixed well. Data were acquired with a flow cytometer (Calibur, BD Biosciences, San Jose, CA, USA).
Definition of endpoints
Freedom from tumor progression (FFTP), and overall survival were the end-points for this study. FFTP was calculated from the date of diagnosis until the date of a disease-related event. Disease-related events were defined as progression during treatment, death during treatment with unknown disease status, less than a complete remission after treatment and relapse after treatment, including death due to lymphoma progression. In case none of these events occurred, patients were censored at the date of last follow-up or death. Overall survival was defined as the date of diagnosis until date of death. In the case of no events, patients were censored at the date of last follow-up.
Statistical analysis
Fisher’s exact test was used for the analysis of differences in patients’ characteristics between the German and Dutch cohorts of patients and between the c-Met-positive and c-Met-negative patients. Survival curves were computed using the method of Kaplan and Meier. Differences between the curves were calculated using the log-rank statistic. Univariate and multivariate Cox logistic regression models were applied to identify prognostic factors for survival. SPSS statistical software version 18.0 (SPSS Inc., Chicago, IL, USA) was used.
Results
c-Met and hepatocyte growth factor expression in chronic Hodgkin’s lymphoma tissue
Cytoplasmic and membranous c-Met expression in more than 30% of the tumor cells was observed in 49 out of 91 German patients (54%) and 31 out of 62 Dutch patients (50%) (Figure 1A and Online Supplementary Table S1). Expression was not significantly different between the two cohorts. Although c-Met expression could be detected in the reactive cellular background, no specific pattern of c-Met expression was observed between tumor cells and infiltrating cells.
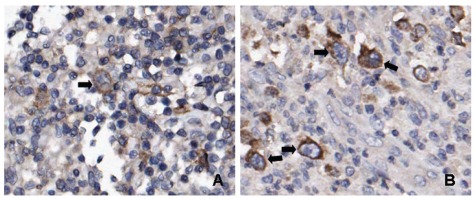
Figure 1.
c-Met and HGF expression in cHL tissue. (A) Representative cHL case showing expression of c-Met in HRS cells (arrow). Expression of c-Met in the reactive cells can also be observed. (B) Representative cHL case showing expression of HGF in HRS cells (arrows). Reactive cells were also positive for HGF.
HGF staining in more than 20% of the tumor cells was detected in 6 out 91 German patients (7%) and 4 out of 30 Dutch patients (13%) (Figure 1B and Online Supplementary Table S1). Expression was not significantly different between the two cohorts. Co-expression of c-Met and HGF in tumor cells was found in only 4 of 121 cHL cases (3%) (Online Supplementary Table S2). Reactive cells stained positive in all cHL cases, but no HGF staining was observed in the lymphocytes directly surrounding the tumor cells.
Expression of c-Met (52% positive in the total cohort) did not correlate with age, sex, histological subtype, stage of disease, EBV status (Online Supplementary Table S2) or other known clinical prognostic factors (erythrocyte sedimentation rate, number of nodal sites, presence of B-symptoms, bulky disease and IPS).
Prognostic significance of c-Met expression
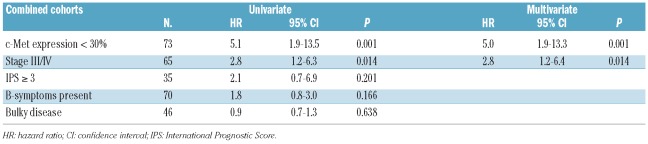
In both the German and the Dutch cohorts, c-Met-positive patients had a significantly more favorable 5-year FFTP compared to c-Met-negative patients (Figure 2A,B). Univariate Cox regression analysis in the German cohort showed that lack of c-Met expression correlated with a hazard ratio (HR) of 4.2 (P=0.013, 95% CI 1.4–13.2), and stage with a HR of 4.6 (P=0.008, 95% CI 1.5–14.4). In multivariate analysis both stage (HR 4.5, P=0.010, 95% CI 1.4–13.9) and c-Met (HR 3.9, P=0.018, 95% CI 1.3–12.3) remained independent predictors for FFTP. In the Dutch cohort only lack of c-Met expression was significant in the univariate analysis (HR 8.7, P=0.041, 95% CI 1.1–69.9) (Online Supplementary Table S3). In the combined cohort, c-Met expression correlated with a 5-year FFTP of 94%, whereas no expression correlated with a 5-year FFTP of 73% (log rank P<0.001, Figure 2C). Univariate Cox regression analysis showed that lack of c-Met expression correlated with a HR of 5.1 (P<0.001, 95% CI 1.9–13.5) compared to positive expression (Table 2). Stage was the only other factor (HR 2.8, P=0.012) that could significantly predict FFTP. In multivariate analysis both c-Met (HR 5.0, P<0.001) and stage (HR 2.8, P=0.014) remained independent predictors for FFTP. Especially in patients with advanced disease, lack of c-Met expression correlated with a very poor 5-year FFTP rate (57% versus 91% in c-Met-positive patients; Figure 2E, F).
Figure 2.
Kaplan-Meier estimates for freedom from tumor progression (FFTP) and overall survival (OS) according to c-Met expression. c-Met positivity of HRS cells correlated with very favorable FFTP in both the German (A) and the Dutch (B) cohorts. In the combined cohort, c-Met expression again correlated with favorable FFTP (C) and OS (D) and was independent of disease stage (E and F). Especially among patients with advanced stage disease (F), absence of c-Met correlated with a low 5-year FFTP rate (57%) compared to the rate in patients who were c-Met-positive (91%).
Table 2.
Univariate and multivariate Cox regression analyses for freedom from tumor progression for c-Met expression and other clinical prognostic factors in the combined cohort.
In the combined cohort, 5-year overall survival rates were 98% and 84% for patients expressing or not expressing c-Met, respectively (log rank P=0.006, Figure 2D). For HGF, no significant differences were observed in FFTP and overall survival.
The c-Met/hepatocyte growth factor signaling pathway in classical Hodgkin’s lymphoma cell lines
High c-Met expression was only observed in the L428 cell line (Figure 3A). HGF protein levels were high in the cell culture supernatant of KMH2, with corresponding levels of HGF mRNA (Figure 3B, C).
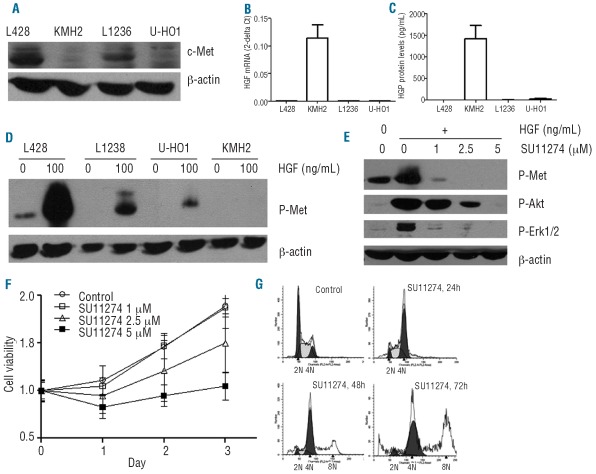
Figure 3.
c-Met and HGF expression and signaling in cHL cell lines. (A) Baseline expression of c-Met was determined in cHL cell lines L428, KMH2, L1236 and U-HO1. The c-Met level was high in L428, moderate in L1236 and very low to negative in KMH2 and U-HO1. (B) HGF mRNA levels were measured in cHL cell lines by qRT-PCR. The HGF mRNA level was high in KMH2 and very low in the other three cHL cell lines. (C) HGF protein levels in the supernatant of cHL cell lines determined by ELISA. The HGF protein level was high in the supernatant of KMH2 and low to negative in the other three cHL cell lines. (D) p-Met level was compared with or without HGF(R&D systems) stimulation (100 ng/mL) in cHL cell lines L428, KMH2, L1236 and U-HO1. Without HGF stimulation, constitutive p-Met was only detected in L428. Upon HGF stimulation, p-Met was strongly up-regulated in L428 and weak to negative in the other three cHL cell lines. (E) Up-regulation of p-Met, p-Akt and p-Erk1/2 with HGF stimulation could be blocked by the c-Met kinase inhibitor SU11274 (Calbiochem, San Diego, CA, USA) in L428 cells. (F) L428 cells were grown in the presence of increasing concentrations of SU11274 (0, 1, 2.5, 5 μM) for 3 days, and the effect on cell growth was assayed by MTT. Cell growth at 2.5 μM and 5 μM SU11274 compared with control growth was significantly decreased (P<0.001). (G) Cell cycle distribution of SU11274 (2.5 μM)-treated L428 cells. Flow cytometry histograms are given with fitted analysis model underlying, in which dark gray areas are calculated areas for cells in G0/G1 and G2 phase and the shaded area is the calculated area for cells in S phase. G2/M cell cycle arrest is shown to be an effect of c-Met inhibition. Tetraploid cells with 8N DNA content can be observed after 48 h and 72 h incubation with SU11274.
Phosphorylated c-Met (p-Met) was present only in the L428 cell line and stimulation by HGF strongly increased p-Met levels in this cell line. Low levels of p-Met were induced in L1236 and U-HO1 (Figure 3D). Analysis of p-Met downstream substrates in the L428 cell line showed up-regulation of p-Akt and p-Erk1/2 upon HGF stimulation and this up-regulation was effectively blocked by SU11274 in a dose-dependent manner (Figure 3E). HGF stimulation did not affect cell growth (data not shown), whereas SU11274 significantly suppressed cell growth in a dose-dependent manner (Figure 3F). This suppression of cell growth was not caused by an induction of apoptosis (data not shown). Cell cycle analysis showed that the proportion of cells in the G2/M phase increased from 15% in control cells to 52% in cells treated with SU11274 (2.5 μM) (P<0.05) (Figure 3G). This effect was greater at a higher concentration (5 μM) (data not shown) or after longer incubation (48 h or 72 h) (Figure 3G), indicating dose- and time-dependent effects. After 48 h and 72 h a population of tetraploid cells appeared (Figure 3G).
Discussion
Deregulation of HGF/c-Met signaling has been implicated in the pathogenesis of many malignancies by disrupting fundamental biological processes including cell cycle, survival, adhesion and migration.20,26 In this study, we demonstrated that c-Met was present in HRS cells in half of the patients with cHL. In contrast to findings in other malignancies,11–15 positivity of c-Met in the HRS cells was associated with favorable survival in two different cohorts of patients. Especially in advanced stage disease, c-Met-positive patients had a strikingly good FFTP rate (91%) compared to c-Met-negative patients (57%). This effect on prognosis appeared to be stronger than the effects that have been observed for other prognostic tissue markers such as CD68 (tumor infiltrating macrophages),6 HLA class II7 and EBV status.8
Expression of c-Met and HGF has been studied previously in cHL patients and cell lines.20–23 One study showed a correlation between c-Met expression and EBV-positivity in a small cohort,20 whereas in a larger study c-Met was detected in HRS cells of all cHL cases independently of EBV status.21 We found c-Met expression in more than 30% of HRS cells in 52% of cHL cases and observed no correlation with EBV status. The lower percentage of c-Met-positive cHL cases can be explained by our strict scoring criteria compared to those used in previous studies. HGF expression was found in infiltrating cells in cHL tissues, and was higher in the serum of patients at diagnosis and at relapse than in the serum of healthy donors and patients in remission.21 In our study, HGF expression was detected in HRS cells in a low percentage of cHL cases and in a variable percentage of the infiltrating cells in all cases. Co-expression of HGF and c-Met in HRS cells was found in only 3% of cHL patients. Paracrine activation of c-Met by HGF might be involved in the pathogenesis of cHL in c-Met-positive cases.
In contrast to our findings, in solid malignancies expression of c-Met correlates with an unfavorable prognosis.12–15 However, this involves overexpression rather than normal physiological expression and we found no evidence of overexpression of c-Met in Hodgkin’s lymphoma. Although a favorable prognostic impact of c-Met expression has been shown in a small study on breast cancer27 and in diffuse large B-cell lymphoma,28 other studies showed opposite effects for both types of tumors.11,13
To explain the unexpected favorable prognostic impact of c-Met expression, we studied the functionality of the c-Met/HGF signaling pathway in the cHL cell line L428. Although p-Met, p-Akt and p-Erk1/2 are strongly up-regulated upon stimulation with HGF, no effect was observed on cell growth in L428 cells. Nevertheless we did observe an inhibitory effect on cell growth with c-Met inhibitor SU11274, which blocked the constitutive phosphorylation of c-Met and downstream kinases and induced G2/M cell cycle arrest.
The stimulating effects of c-Met on cell cycle progression in L428 cells are inconsistent with the favorable prognostic value of c-Met positivity in HRS cells in cHL patients. The unique histological phenotype of cHL might be a key factor in this unexpected positive prognostic effect. The proportion of tumor cells in cHL is small, typically less than 1%, and these cells are located within an extensive infiltrate of reactive cells. In comparison, the proportion of tumor cells in diffuse large B-cell lymphoma and solid malignancies is generally between 40% and 80%. When focusing on the infiltrating cells several known effects of c-Met might explain the favorable effect of c-Met in Hodgkin’s lymphoma. In a rat model of cirrhosis c-Met strongly reduced liver fibrosis and decreased transforming growth factor-β (TGF-β) levels.29 Since TGF-β plays an important role in suppression of an effective immune response in cHL,30 the activation of c-Met in HRS cells could result in decreased TGF-β production and, thereby, in enhanced anti-tumor responses. In monocytes, activation of c-Met induced up-regulation of pro-inflammatory cytokines31 and a pro-inflammatory response of the HGF/c-Met pathway was also found in experimental autoimmune encephalomyelitis.32 Overall, it can be speculated that activation of c-Met in HRS cells might alter the immunosuppressive properties of the reactive cells by secretion of certain factors that shape the microenvironment in a way that is less beneficial for survival and growth of the HRS cells.
In conclusion, despite its oncogenic function in cHL cell lines and other malignancies, c-Met expression by tumor cells in cHL patients predicts a very favorable outcome, independently of disease stage. Future studies should show whether c-Met can be used in clinical practice as a pre-treatment marker to select suitable patients for less toxic treatment regimens.
Supplementary Material
Footnotes
Funding: CX is a recipient of a Bernoulli bursary. Part of this study was supported by a grant from the JK de Cock foundation.
The online version of this article has a Supplementary Appendix.
Authorship and Disclosures
The information provided by the authors about contributions from persons listed as authors and in acknowledgments is available with the full text of this paper at www.haematologica.org.
Financial and other disclosures provided by the authors using the ICMJE (www.icmje.org) Uniform Format for Disclosure of Competing Interests are also available at www.haematologica.org.
References
- 1.Kanzler H, Küppers R, Hansmann ML, Rajewski K. Hodgkin and Reed-Sternberg cells in Hodgkin’s disease represent the outgrowth of a dominant tumor clone derived from (crippled) germinal center B cells. J Exp Med. 1996;184(4):1495–505. doi: 10.1084/jem.184.4.1495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Küppers R. The biology of Hodgkin’s lymphoma. Nat Rev Cancer. 2009;9(1):15–27. doi: 10.1038/nrc2542. [DOI] [PubMed] [Google Scholar]
- 3.Renné C, Willenbrock K, Küppers R, Hansmann ML, Bräuninger A. Autocrine and paracrine activated receptor tyrosine kinases in classical Hodgkin lymphoma. Blood. 2005;105(10):4051–9. doi: 10.1182/blood-2004-10-4008. [DOI] [PubMed] [Google Scholar]
- 4.Klimm B, Engert A. Combined modality treatment of Hodgkin’s lymphoma. Cancer J. 2009;15(2):143–9. doi: 10.1097/PPO.0b013e31819e31ba. [DOI] [PubMed] [Google Scholar]
- 5.Hasenclever D, Diehl V. A Prognostic score for advanced Hodgkin’s disease. N Engl J Med. 1998;339(21):1506–14. doi: 10.1056/NEJM199811193392104. [DOI] [PubMed] [Google Scholar]
- 6.Steidl C, Lee T, Shah SP, Farinha P, Han G, Nayar T, et al. Tumor-associated macrophages and survival in classic Hodgkin’s lymphoma. N Engl J Med. 2010;362(10):875–85. doi: 10.1056/NEJMoa0905680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Diepstra A, van Imhoff GW, Karim-Kos HE, van den Berg A, te Meerman GJ, Niens M, et al. HLA class II expression by Hodgkin Reed-Sternberg cells is an independent prognostic factor in classical Hodgkin’s lymphoma. J Clin Oncol. 2007;25(21):3101–8. doi: 10.1200/JCO.2006.10.0917. [DOI] [PubMed] [Google Scholar]
- 8.Diepstra A, van Imhoff GW, Schaapveld M, Karim-Kos H, van den Berg A, Vellenga E, et al. Latent Epstein-Barr virus infection of tumor cells in classical Hodgkin’s lymphoma predicts adverse outcome in older adult patients. J Clin Oncol. 2009;27(23):3815–21. doi: 10.1200/JCO.2008.20.5138. [DOI] [PubMed] [Google Scholar]
- 9.Ma PC, Maulik G, Christensen J, Salgia R. c-Met: structure, functions and potential for therapeutic inhibition. Cancer Metastasis Rev. 2003;22(4):309–25. doi: 10.1023/a:1023768811842. [DOI] [PubMed] [Google Scholar]
- 10.Comoglio PM, Giordano S, Trusolino L. Drug development of MET inhibitors: targeting oncogene addiction and expedience. Nat Rev Drug Discov. 2008;7(22):504–16. doi: 10.1038/nrd2530. [DOI] [PubMed] [Google Scholar]
- 11.Kawano R, Ohsima K, Karube K, Yamaguchi T, Kohno S, Suzumiya J, et al. Prognostic significance of hepatocyte growth factor and c-MET expression in patients with diffuse large B-cell lymphoma. Br J Haematol. 2004;127(3):305–7. doi: 10.1111/j.1365-2141.2004.05197.x. [DOI] [PubMed] [Google Scholar]
- 12.Cheng HL, Trink B, Tzai TS, Liu HS, Chan SH, Ho CL, et al. Overexpression of c-met as a Prognostic indicator for transitional cell carcinoma of the urinary bladder: a comparison with p53 nuclear accumulation. J Clin Oncol. 2002;20(6):1544–50. doi: 10.1200/JCO.2002.20.6.1544. [DOI] [PubMed] [Google Scholar]
- 13.Lengyel E, Prechtel D, Resau JH, Gauger K, Welk A, Lindemann K, et al. C-Met overexpression in node-positive breast cancer identifies patients with poor clinical outcome independent of Her2/neu. Int J Cancer. 2005;113(4):678–82. doi: 10.1002/ijc.20598. [DOI] [PubMed] [Google Scholar]
- 14.Lee CT, Chow N, Su PF, Lin SC, Lin PC, Lee JC. The prognostic significance of RON and MET receptor coexpression in patients with colorectal cancer. Dis Colon Rectum. 2008;51(8):1268–74. doi: 10.1007/s10350-008-9297-1. [DOI] [PubMed] [Google Scholar]
- 15.Ayhan A, Ertunc D, Tok EC, Ayhan A. Expression of the c-Met in advanced epithelial ovarian cancer and its prognostic significance. Int J Gynecol Cancer. 2005;15(4):618–23. doi: 10.1111/j.1525-1438.2005.00117.x. [DOI] [PubMed] [Google Scholar]
- 16.Sattler M, Pride YP, Ma P, Gramlich JL, Chu SC, Quinnan LA, et al. A novel small molecule met inhibitor induces apoptosis in cells transformed by the oncogenic TPR-MET tyrosine kinase. Cancer Res. 2003;63(20):5462–9. [PubMed] [Google Scholar]
- 17.Ma PC, Jagadeeswaran R, Jagadeesh S, Tretiakova MS, Nallasura V, Fox EA, et al. Functional expression and mutations of c-Met and its therapeutic inhibition with SU11274 and small interfering RNA in non-small cell lung cancer. Cancer Res. 2005;65(4):1479–88. doi: 10.1158/0008-5472.CAN-04-2650. [DOI] [PubMed] [Google Scholar]
- 18.Jagadeeswaran R, Ma PC, Seiwert TY, Jagadeeswaran S, Zumba O, Nallasura V, et al. Functional analysis of c-Met/hepatocyte growth factor pathway in malignant pleural mesothelioma. Cancer Res. 2006;66(1):352–61. doi: 10.1158/0008-5472.CAN-04-4567. [DOI] [PubMed] [Google Scholar]
- 19.Puri N, Ahmed S, Janamanchi V, Tretiakova M, Zumba O, Krausz T, et al. c-Met is a potentially new therapeutic target for treatment of human melanoma. Clin Cancer Res. 2007;13(8):2246–53. doi: 10.1158/1078-0432.CCR-06-0776. [DOI] [PubMed] [Google Scholar]
- 20.Weimar IS, de Jong D, Muller EJ, Nakamura T, van Gorp JM, de Gast GC, et al. Hepatocyte growth factor/scatter factor promotes adhesion of lymphoma cells to extra-cellular matrix molecules via alpha 4beta 1 and alpha 5beta 1 Integrins. Blood. 1997;89(3):990–1000. [PubMed] [Google Scholar]
- 21.Teofili L, di Febo AL, Pierconti F, Maggiano N, Bendandi M, Rutella S, et al. Expression of the c-met proto-oncogene and its ligand, hepatocyte growth factor, in Hodgkin disease. Blood. 2001;97(4):1063–9. doi: 10.1182/blood.v97.4.1063. [DOI] [PubMed] [Google Scholar]
- 22.Jücker M, Günther A, Gradl G, Fonatsch C, Krüger G, Diehl V, et al. The Met/hepatocyte growth factor receptor (HGFR) gene is overexpressed in some cases of human leukemia and lymphoma. Leuk Res. 1994;18(1):7–16. doi: 10.1016/0145-2126(94)90003-5. [DOI] [PubMed] [Google Scholar]
- 23.Pons E, Uphoff CC, Drexler HG. Expression of hepatocyte growth factor and its receptor c-met in human leukemia-lymphoma cell lines. Leukemia Res. 1998;22(9):797–804. doi: 10.1016/s0145-2126(98)00071-x. [DOI] [PubMed] [Google Scholar]
- 24.Drexler HG. Recent results on the biology of Hodgkin and Reed-Sternberg cells. II. Continuous cell lines. Leuk Lymphoma. 1993;9(1–2):1–25. doi: 10.3109/10428199309148499. [DOI] [PubMed] [Google Scholar]
- 25.Mader A, Bruderlein S, Wegener S, Melzner I, Popov S, Muller-Hermelink HK, et al. U-HO1, a new cell line derived from a primary refractory classical Hodgkin lymphoma. Cytogenet Genome Res. 2007;119(3–4):204–10. doi: 10.1159/000112062. [DOI] [PubMed] [Google Scholar]
- 26.Birchmeier C, Birchmeier W, Gherardi E, Vande Woude GF. Met, metastasis, motility and more. Nat Rev Mol Cell Biol. 2003;4(12):915–25. doi: 10.1038/nrm1261. [DOI] [PubMed] [Google Scholar]
- 27.Nakopoulou L, Gakiopoulou H, Kerampoulos A, Giannopoulou I, Athanassiadou P, Mavrommatis J, et al. c-Met tyrosine kinase receptor expression is associated with abnormal β-catenin expression and favourable prognostic factors in invasive breast carcinoma. Histopathol. 2000;36(4):313–25. doi: 10.1046/j.1365-2559.2000.00847.x. [DOI] [PubMed] [Google Scholar]
- 28.Uddin S, Hussain AR, Ahmed M, Al-Dayel F, Bu R, Bavi P, et al. Inhibition of c-Met is a potential therapeutic strategy for treatment of diffuse large B-cell lymphoma. Lab Invest. 2010;90(9):1346–56. doi: 10.1038/labinvest.2010.108. [DOI] [PubMed] [Google Scholar]
- 29.Ueki T, Kaneda Y, Tsutsui H, Nakanishi K, Sawa Y, Morishita R, et al. Hepatocyte growth factor gene therapy of liver cirrhosis in rats. Nat Med. 1999;5(2):226–30. doi: 10.1038/5593. [DOI] [PubMed] [Google Scholar]
- 30.Poppema S. Immunobiology and pathophysiology of Hodgkin lymphomas. Am Soc Hematol Educ Program. 2005:231–8. doi: 10.1182/asheducation-2005.1.231. [DOI] [PubMed] [Google Scholar]
- 31.Beilmann M, Vande Woude GF, Dienes HP, Schirmacher P. Hepatocyte growth factor-stimulated invasiveness of monocytes. Blood. 2000;95(12):3964–9. [PubMed] [Google Scholar]
- 32.Moransard M, Sawitzky M, Fontana A, Suter T. Expression of the HGF receptor c-Met by macrophages in experimental autoimmune encephalomyelitis. Glia. 2010;58(5):559–71. doi: 10.1002/glia.20945. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.