Abstract
The role of lipid metabolism has gained particular interest in prostate cancer research. A large body of literature has outlined the unique upregulation of de novo lipid synthesis in prostate cancer. Concordant with this lipogenic phenotype is a metabolic shift, in which cancer cells use alternative enzymes and pathways to facilitate the production of fatty acids. These newly synthesized lipids may support a number of cellular processes to promote cancer cell proliferation and survival. Hence, de novo lipogenesis is under intense investigation as a therapeutic target. Epidemiologic studies suggest dietary fat may also contribute to prostate cancer; however, whether dietary lipids and de novo synthesized lipids are differentially metabolized remains unclear. Here, we highlight the lipogenic nature of prostate cancer, especially the promotion of de novo lipid synthesis, and the significance of various dietary lipids in prostate cancer development and progression.
Keywords: Prostate cancer, de novo lipogenesis, dietary fat
Introduction
Prostate cancer is the second most common occurring cancer in men worldwide [1] and the leading site for cancer incidence in men of the United States [2]. It is estimated that every one in six men will develop prostate cancer in their lifetime [2]. With sharply increasing statistics in incidence, it is imperative that we identify major contributors to prostate cancer development and progression to recommend guidelines for prevention and treatment. One common theme in a growing body of literature is the interplay between lipid metabolism and prostate cancer. The metabolic shift from catabolic to anabolic metabolism is a hallmark of cancer cells, and many cancers, including that of the prostate, appear to require the de novo synthesis of fatty acids. Besides fatty acids, other lipids derived from the mevalonic acid pathway, such as cholesterol and androgens, are also strongly implicated in prostate cancer. Whether these fatty acids must be produced de novo, or whether they can be supplied by dietary means remains unclear; however, epidemiological studies suggest dietary fat consumption also contributes to risk of prostate cancer development and progression. In this review, we describe the lipogenic phenotype of prostate cancer, whereby cancer cells metabolically reprogram to promote lipid synthesis, and the contribution of various dietary fats in prostate cancer.
De Novo Lipogenesis in Prostate Cancer
Cancer cells are characterized by a large number of metabolic alterations. One major change from their normal counterpart is a shift from catabolic to anabolic metabolism. Otto Warburg first described this metabolic shift nearly a century ago [3]. The proclaimed Warburg Effect describes the unique phenomenon of aerobic glycolysis in a cancer cell, whereby cancer cells consume a large quantity of glucose, metabolize it via glycolysis, and release the majority as lactic acid into the extracellular space – most peculiarly, this occurs under normal oxygen conditions. Normally, each molecule of glucose consumed by a cell is metabolized through glycolysis to two molecules of pyruvate. Pyruvate is then converted to acetyl-CoA in the mitochondria where it enters the tricarboxylic acid (TCA) cycle to produce redox substrates for oxidative phosphorylation, the cell’s major energy-producing, catabolic pathway. Interestingly, cancer cells seem to consume an excessive quantity of glucose, and utilize it both catabolically, as just described, and anabolically, whereby the carbons are used as a source to synthesize and fulfill the macromolecular demand of their proliferative phenotype. Although the production of amino acids and nucleic acids is certainly an integral characteristic of the cancer anabolic phenotype, here we describe the cancer cell’s requirement for de novo fatty acids, and the specific use of alternative enzymes, metabolic pathways, and common oncogenic pathways to promote the synthesis of fatty acids and other lipids in prostate cancer cells.
Fatty Acid Synthesis and Prostate Cancer
Fatty acid synthesis occurs in the cytosol and begins with the production of acetyl-CoA from citrate by ATP citrate lyase (ACLY) (Figure 1). Acetyl-CoA is then converted to malonyl-CoA by acetyl-CoA carboxylase (ACACA, commonly referred as ACC1); this is the first committed and rate-limiting step of fatty acid synthesis. Fatty acid synthase (FASN) then processes one acetyl-CoA and seven malonyl-CoA molecules through a series of catalytic domains to produce palmitate, a saturated 16-carbon fatty acid. Palmitate is the primary product of FASN, representing about 80-90% of total fatty acids produced; FASN also produces myristate (14:0) and stearate (18:0) [4, 5]. Further modification may occur by other desaturase enzymes, such as stearoyl-CoA desaturase (SCD) or elongase enzymes, to insert double bonds or increase the carbon chain length, respectively, before the fatty acids are ultimately utilized for energy, protein modification, or incorporation into complex lipid structures for cellular signaling and membrane integrity.
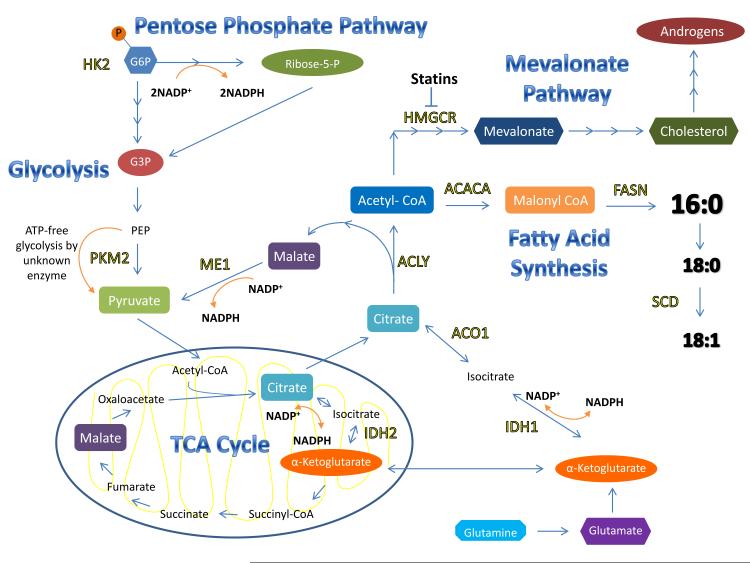
Figure 1. Cancer Cell Metabolism Promotes Fatty Acid Synthesis.
Glucose entering the cell is immediately phosphorylated by Hexokinase 2 (HK2), producing glucose-6-phosphate (G6P). G6P can enter both glycolysis and the pentose phosphate pathway (PPP). Expression of pyruvate kinase M2 (PKM2) promotes the ATP-free conversion of phosphoenolpyruvate (PEP) to pyruvate by a currently unknown enzyme, which may prevent ATP-mediated inhibition of glycolysis. Citrate is exported from the mitochondrial TCA cycle to fuel the fatty acid synthesis pathway by conversion to acetyl-CoA by ATP citrate lyase (ACLY). Acetyl-CoA carboxylase 1 (ACACA) initiates the first committed step to fatty acid synthesis to produce malonyl-CoA. Seven malonyl-CoA molecules are added to acetyl-CoA by fatty acid synthase (FASN) to produce palmitic acid (16:0). Palmitic acid can be further elongated to stearic acid (18:0) and desaturated by stearoyl-CoA desaturase (SCD) to produce oleic acid (18:1). Citrate may also be recycled to produce NADPH for fatty acid synthesis by malic enzyme (ME1), which produces pyruvate, or by cytosolic aconitase (ACO1) followed by cytosolic isocitrate dehydrogenase (IDH1) to produce α-ketoglutarate. Glutamine can be converted to α-ketoglutarate upon entering the cell, to replenish TCA cycle intermediates or fuel fatty acid synthesis. The reduced form of nicotinamide adenine dinucleotide phosphate (NADPH), a critical reductant for fatty acid synthase (FASN), is produced from the activity of the PPP, ME1, and IDH.
With the exception of brain, liver, adipose and lung tissues, fatty acid synthesis in normal, differentiated adult cells is relatively low [5, 6]. Outside of these tissue types, cells may utilize circulating fatty acids derived from dietary sources to fulfill their fatty acid requirements. However, it was recognized over 50 years ago that cancer cells exhibit increased production of de novo fatty acids [7]. Upregulation of fatty acid synthesis in prostate cancer can arise as a consequence of a variety of events. Increased gene copy number of FASN in prostate adenocarcinoma coordinates with overexpression of FASN protein [8]. Increased transcriptional activation of FASN has been well described and is very common in prostate cancer [9-12]. Stabilization of FASN protein by ubiquitin-specific protease-2a (USP2a) has been shown to increase fatty acid synthesis in prostate cancer [13]. Loss of tumor suppressor proteins, such as PTEN and LKB1, results in prostate neoplasia and activation of AKT [14, 15], a well-described activator of FASN expression [16]. Finally, FASN itself has been postulated as an oncogene, as its overexpression in mouse prostate leads to prostatic intraepithelial neoplasia [17]. Collectively, these cellular perturbations demonstrate that fatty acid synthesis is a unique characteristic of cancer and, hence, an attractive therapeutic target for prostate cancer.
Prostate Cancer Cells Require Fatty Acid Synthesis
In line with an altered metabolism, cancer cells shunt glucose-derived carbons from the mitochondrial TCA cycle to the cytosol to feed fatty acid synthesis. This increased influx of carbon toward fatty acid synthesis is accompanied by upregulation of the enzymes in the fatty acid synthesis pathway. The major transcriptional regulator of enzymes in this pathway is the sterol response element binding protein-1c (SREBP-1c). SREBP-1c is a lipogenic transcription factor that regulates the expression of glycolytic enzymes and fatty acid synthesis enzymes, including ACLY, ACACA, FASN, and SCD-1 [18]. Expression of SREBP-1c can be stimulated by androgens and epidermal growth factor (EGF) in prostate cancer cells [9, 10], and in turn, expression of the androgen receptor is regulated by SREBP-1c [19], creating a positive feedback loop for the expression of lipogenic enzymes. Overexpression of SREBP-1 is sufficient to increase tumorigenicity and invasion of prostate cancer cells [19]. Not surprisingly, increased expression of FASN and the lipogenic phenotype has been documented in various cancer types, including prostate [12], breast [20], ovarian [21], colorectal [22], and others (as reviewed in [23-25]). Pharmacological or small-interfering RNA-mediated inhibition of enzymes in the de novo fatty acid synthesis pathway, namely ACLY, FASN, or SCD-1 inhibits prostate tumor growth [26-28]. Additionally, inhibition of ACACA, FASN, or SCD-1 induces cancer cell death [11, 26-31], demonstrating the requirement for de novo fatty acids for prostate cancer survival.
Utility of de novo Fatty Acids in Prostate Cancer
Unlike their normal counterparts, cancer cells require fatty acid synthesis for growth and survival; hence, they likely utilize these de novo synthesized fatty acids to fulfill specific needs. Because cancer cells divide rapidly, one major purpose for increased fatty acid synthesis is to supply the heavy demand for new membrane biogenesis. In fact, Swinnen et al. [32] showed that de novo fatty acids used for membrane biogenesis also preferentially localize in the membrane’s signaling raft domains. Membrane composition has been shown to affect the conformation and activation of membrane-bound proteins [33]; thus, de novo fatty acids may play a significant role in propagating oncogenic signals from the plasma membrane. Indeed, FASN regulates HER2 expression and activation of AKT [34]. Moreover, enrichment of saturated fatty acids by FASN in tumor cell membranes protects cells from lipid peroxidation and oxidative stress-induced cell death [35]. Overexpression of FASN in prostate cancer supports palmitoylation and activation of oncogenic proteins [36]. Additionally, lipid signaling ligands can be derived specifically from the de novo fatty acid pool [37], suggesting an absolute requirement of fatty acid synthesis to activate specific signaling pathways. Although lacking evidence of a direct role for fatty acids, FASN and ACACA are also required for cell cycle progression [31, 38]. Overall, prostate cancer cells may utilize de novo fatty acids for a variety of uses, all of which facilitate proliferation and survival of cancer cells.
Prostate Cancer Metabolism Favors Fatty Acid Synthesis
Fatty acid synthesis is an extremely demanding process; it requires 8 citrate, 8 ATP, and 14 NADPH molecules to produce just a single palmitate molecule. Excessive fatty acid synthesis appears to be a requirement for prostate cancer growth and survival; thus, cancer cells have adapted the use of alternative metabolic pathways and enzymes to provide the necessary substrates for lipogenesis and prevent its inhibition by upstream signaling mediators. These alternative pathways include isoform switching, truncation of the TCA cycle and the use of TCA cycle intermediates for fatty acid synthesis, and the increased use of other non-canonical metabolic enzymes, such as the pentose phosphate pathway and cytosolic TCA cycle enzymes (Figure 1).
Metabolism for fatty acid synthesis ultimately begins with cellular uptake of glucose. Cancer cells consume a large quantity of glucose as their major energy source. Upon entering a cell, glucose is phosphorylated by hexokinase (HK) to glucose-6-phosphate (G6P). Humans possess four isozymes of HK1-4 and most cell types express HK1 as the housekeeping enzyme for normal catabolic metabolism of glucose [39]. However, cancer cells seem to preferentially express HK2, which is thought to support anabolic metabolism [40]. Prostate cancer cells strongly upregulate HK2 and lipid synthesis in response to androgen [41]. G6P, produced by HK2, may take one of two fates: metabolism by the pentose phosphate pathway (PPP) or glycolysis. Cancer cells can shunt G6P through the PPP to produce NADPH, which is a critical reductant for fatty acid synthase. PPP intermediates can also return to glycolysis, producing pyruvate, which may be used later for fatty acid synthesis.
Tumor cells preferentially express the embryonic isoform of pyruvate kinase (PKM2), the terminal enzyme of glycolysis. Compared to PKM1, the splice variant found in most adult tissues, PKM2 is characterized by a weaker substrate affinity and decreased activity [42, 43]. Insights to the advantages of which PKM2 imposes on cancer cells have surfaced only recently. Studies by Cantley and colleagues have shown PKM2 promotes tumor growth and survival by maintaining a high glycolytic rate, low oxidative phosphorylation, and redox homeostasis [44, 45]. Expression of PKM2 in prostate cancer was also shown to be associated with an alternative mechanism of producing pyruvate in the absence of ATP production [46]. This suggests expression of PKM2 in prostate cancer cells promotes anabolic metabolism, such as fatty acid synthesis, by allowing high glucose consumption as a carbon source for fatty acids, but without inducing ATP-mediated inhibition of glycolysis [46].
Typically, glucose-derived citrate is processed through the mitochondrial TCA cycle to produce intermediates for oxidative phosphorylation. In contrast, cancer cells shunt citrate from the TCA cycle to the cytoplasm and use alternative methods to metabolize citrate that promote fatty acid synthesis. Cytosolic citrate can be converted to isocitrate by cytosolic aconitase (ACO1), and subsequently to α-ketoglutarate by cytosolic isocitrate dehydrogenase (IDH1); the latter reaction produces additional NADPH for fatty acid synthesis. Overexpression of IDH1 in male mice leads to significantly increased epididymal fat weight and adipogenesis, coinciding with hyperlipidemia and obesity [47]. Interestingly, oncogenic mutations in IDH have been characterized in several cancer types [48, 49]. These findings suggest IDH1 plays a significant role promoting fatty acid synthesis and lipid metabolism in cancer.
Cytosolic citrate, in addition to being metabolized outside of the TCA cycle to produce NADPH, can also be converted to acetyl-CoA by ACLY, and can be used for both fatty acid and sterol synthesis. A byproduct of this reaction is oxaloacetate, which is converted to malate by malate dehydrogenase. Cytosolic malate may be converted back to pyruvate by cytosolic malic enzyme (ME1) to produce yet more NADPH, or it can be recycled back to the mitochondria by the citrate transport protein in the mitochondrial membrane, which coordinates the influx of cytosolic malate with the efflux of mitochondrial citrate [50], completing a loop for driving mitochondrial carbon sources toward fatty acid synthesis. Prostate tissue is unique in its transport system of citrate in that normal prostate cells produce large amounts of intracellular citrate and release it into the prostatic fluid [51], where it can act as an energy source for sperm [52]. Prostate cancer cells, however, have significantly decreased intracellular citrate levels. This is accompanied by increased c-ACNT expression and activity [53] and increased ACLY [27]. Collectively, these findings suggest that prostate cancer cells rapidly utilize citrate for lipogenesis, either to produce NADPH or shunt as a carbon source for fatty acid production.
Although de novo synthesized fatty acids are typically derived from glucose, some cancer cells rely on glutamine metabolism. Because glucose carbons are shunted from the mitochondria for fatty acid synthesis, prostate cancer cells convert glutamine to α-ketoglutarate and shuttle it into the mitochondria to replenish TCA cycle intermediates for anaplerotic reactions [54, 55]. Additionally, cancer cells experiencing selective pressure by hypoxia, or a defective electron transport chain, use glutamine, rather than glucose, as the major lipid precursor. In such a scenario, stabilized HIF-1α inhibits the conversion of pyruvate to acetyl-CoA, and IDH1 and ACO1 work in reverse to convert α-ketoglutarate to citrate, which is then metabolized by ACLY for lipid synthesis [56, 57] (Figure 1).
Oncogenic Signaling Pathways Promote Fatty Acid Synthesis
Further demonstrating the importance of fatty acid synthesis in prostate cancer, some of the major disease-driving signaling pathways regulate the expression of lipogenic enzymes. As a lipid phosphatase, PTEN opposes the action of PI3K by removing a phosphate group from phosphatidylinositol-3,4,5-triphosphate, an essential activator of AKT [58]. Loss of PTEN, is one of the most common events observed in prostate cancer, occurring in approximately 40% of primary cancers and over 70% of castration resistant and metastatic prostate cancers [59-62]. Pten deletion leads to invasive prostatic adenocarcinoma in mice [14], and induces expression of FASN [63]. Treatment with a PI3K inhibitor or reintroduction of PTEN in PTEN-null prostate cancer cells reduces FASN expression [63]. Conversely, inhibition of FASN reduces expression and activation of AKT [64], which was recently shown to occur by increased degradation of AKT and its downstream effectors [65]. Additionally, cell growth inhibition by inhibiting FASN can be overcome by hyperactivation of AKT [65]. Collectively, these studies strongly suggest coordinate feedback between lipogenesis and PTEN/PI3K/AKT oncogenic signaling to promote cancer growth and tumor progression.
HER2 is a well-recognized oncogene and diagnostic marker in multiple cancers [66]. Overexpression of HER2 in the absence of amplification has been identified in approximately 20% of prostate cancers, and significantly associates with high Gleason grade, proliferation, and tumor recurrence [67]. It activates fatty acid synthesis via activation of the PI3K/AKT signaling pathway [68]. Surprisingly, HER2 regulation of fatty acid synthesis does not occur transcriptionally via SREBP-1c, but rather, translationally through activation of mTOR and increased FASN protein [69]. Inhibition of FASN attenuates HER2 oncogenic signaling and downregulates activation of AKT and ERK1/2 [34].
A number of studies have linked oxidative stress to prostate cancer development and progression [70-73]. Hypoxia, a common feature of tumors characterized by lack of blood supply, and consequently, nutrients and oxygen to the growing tumor, can lead to oxidative stress. Furuta and colleagues [74] demonstrate that a hypoxic environment or treatment with hydrogen peroxide induces AKT/HIF-1α-mediated activation of SREBP-1, which increases transcription of FASN. SREBP-1 can also increase expression of NOX5 [19], a prominent producer of ROS and regulator of prostate cancer cell growth [75], suggesting another feed forward mechanism inducing lipogenesis and prostate cancer growth.
Androgen signaling is a major driver of prostate cancer, and androgen ablation is a primary therapeutic strategy for prostate cancer patients. Activation of the androgen receptor (AR) by androgen increases expression of lipogenic enzymes in a SREBP-1c-dependent manner [9, 10], and a positive feedback loop promotes this signaling pathway since binding sites for SREBP-1 transcription factors are also found in the AR gene of prostate cancer cells [19]. AR is regulated by β2-microglobulin (β2-M), a component of the housekeeping major histocompatibility complex class I molecule found on prostate cancer cells, in a MAPK/SREBP-1-dependent manner. Inhibition of β2-M by a selective antibody decreases the interaction between SREBP-1 and its binding site in the AR promoter region, resulting in decreased AR expression and lipogenesis by FASN [76]. A new AR splice variant, AR8, was recently identified and is strongly expressed in castration resistant prostate cancer (CRPC). AR8 is found at the plasma membrane where it associates with EGFR and induces nuclear translocation of AR in response to EGF [77]. Because we know AR activates FASN [10], this may partly explain how EGF activates FASN [9], and provides a mechanism for activation of AR signaling in CRPC. AR8 also requires FASN for functionality, as palmitoylation appears to tether it to the membrane [77]. Whether AR8 activates SREBP-1 is unknown. Interestingly, inhibition of SREBP-1, ACACA, FASN, or SCD-1 results in decreased AR expression and activity [19, 78, 79]. Concomitant overexpression of AR and FASN in prostate is sufficient to induce adenocarcinoma in mice [17]. Overall, these results suggest androgen signaling is strongly associated with activation of fatty acid synthesis and conversely, fatty acid synthesis promotes androgen signaling. Together these signaling pathways may work in concert with other major oncogenic signaling pathways to promote prostate cancer development and progression.
Cholesterol and Steroid Synthesis in Prostate Cancer
De novo steroids are produced from cholesterol, which is synthesized by the mevalonic acid pathway (Figure 1). Transcriptional regulation of the enzymes in this pathway occurs through activation of the sterol response element binding protein-2 transcription factor (SREBP-2) [80]. SREBP-2, a regulator of androgen synthesis, is also itself regulated by androgens [80], demonstrating a direct feedback circuit for regulation of androgen production. Interestingly, SREBP-2 expression increases during disease progression and is significantly higher after castration [81]. The transcription factor also lacks its feedback inhibition in prostate cancer cells [82], overall, implicating a role for cholesterol and androgen synthesis in prostate cancer.
Major biological functions of the lipid molecules produced by the mevalonic acid pathway include post-translational modification, lipid raft formation, and steroid synthesis, all of which influence prostate cancer development and progression [83, 84]. Statin drugs were designed to inhibit HMG-CoA reductase, the rate-limiting enzyme of cholesterol synthesis, and were originally indicated for the treatment of cardiovascular disease; however, long term use of statin drugs has shown beneficial effects in reducing prostate cancer risk [85, 86]. Therefore, much research has focused on possible mechanisms of statin-mediated inhibition of prostate cancer (reviewed in [87-90].
During the synthesis of cholesterol, the mevalonate pathway produces several metabolic intermediates used as protein modifiers, such as isoprenoids, geranylgeranyl pyrophosphate, and farnesyl pyrophosphate [83]. These protein modifiers can prenylate oncogenic proteins such as Ras and Rho, which can regulate proliferation, migration, and invasion of prostate cancer cells [88]. Cholesterol itself can post-translationally modify cancer promoting proteins, such as sonic hedgehog [91], promoting dedifferentiation and facilitating cancer growth and metastasis [92]. Cholesterol is also a major component of plasma membrane lipid raft microdomains, which mediate formation of oncogenic protein signaling complexes [93], including that of the PI3K/AKT pathway. The cholesterol binding protein caveolin-1 has been implicated as a promoter of prostate cancer, as its deletion in the TRAMP prostate cancer mouse model decreased tumor growth and slowed histopathological progression [94]. Hence, inhibition of the mevalonic acid pathway may reduce prostate cancer growth and progression through several lipid signaling mechanisms. Although, statin-mediated inhibition of prostate cancer has also been suggested to occur through other mechanisms, such as anti-angiogenic and anti-inflammatory effects [95].
A common therapy for advance prostate cancer patients involves deprivation of androgens by surgical or medical castration, resulting in decreased circulating androgens and prostate tumor shrinkage. However, despite these initial responses, essentially all patients experience relapse and progression to castration resistant disease and metastasis. Formerly described as androgen independent, it is now well recognized that castration resistant prostate cancer (CRPC), including bone metastases, express AR and maintain an active androgen signaling pathway [96, 97]. New evidence has emerged to suggest prostate cancer cells produce androgen themselves, thereby propagating the androgen signaling pathway, even after androgen deprivation therapy [98, 99]. Increased cholesterol concentration was identified in prostate cancer bone metastases [100], along with increased expression of enzymes involved in steroidogenesis [101, 102]. Studies suggest these intratumoral androgens may be derived from de novo synthesized cholesterol of the mevalonic acid pathway [102], as well as circulating cholesterol from the diet [103]. Recall that androgens, via activation of AR, stimulate activation of the de novo fatty acid synthesis pathway [10]. Therefore, after androgen deprivation therapy prostate cancers likely increase their steroid synthesis from circulating and de novo synthesized cholesterol as a means to amplify activation of fatty acid synthesis and encourage progression to castration resistance.
Clinical Implications for Lipogenesis and Prostate Cancer
Expression and activation of the fatty acid synthesis and mevalonic acid pathways is clearly an integral component of prostate cancer as a disease. Hence, a number of studies have attempted to exploit the lipogenic phenotype, by chemical inhibition of lipogenic enzymes as a means to treat cancer [26, 27, 31, 104-109]. Because cancer cells depend on FASN for tumor growth and survival, pharmacological inhibition of FASN is an attractive approach, especially considering that inhibition of FASN does not appear to affect normal prostate cells [32]. Unfortunately, such attempts have not yet yielded fruitful results, as inhibitors are either too insoluble and have low bioavailability due to the hydrophobic properties of the FASN active site [28, 110], or alter eating habits in mice by inactivation of hypothalamic AMPK, ultimately leading to rapid weight loss and anorexia [111].
Although FASN is an ideal target due to the numerous metabolic alterations that converge on this enzyme, inhibition of other enzymes in the fatty acid synthesis pathway may be an alternative approach. Soraphen A, an inhibitor of ACACA, possesses anti-lipogenic and anti-tumorigenic properties; however, similar to FASN inhibitors [112], it is non-selective and activates fatty acid oxidation, while concomitantly inhibiting fatty acid synthesis [31]. Other drugs, such as SB-204990 and BZ36, which target ACLY and SCD-1, respectively, have demonstrated efficacy in animal models [26, 31], but also have not advanced to clinical trials.
Studies suggest dysregulation of glucose and lipid metabolism, components of a disease described as metabolic syndrome, strongly associate with cancer incidence and aggressiveness [113, 114]. Even the transcriptional signatures and molecular properties of cancer and metabolic syndrome display significant overlap [115]. Hence, it should be no surprise that common drug treatments for metabolic disease, especially cholesterol lowering statin drugs, have shown efficacy in regulating cancer [85, 90, 116]. In fact, a search for active studies on “statin” intervention in “cancer” conditions identifies 36 different clinical trials investigating the adjuvant or prophylactic therapeutic value of statin drug use in various cancers (clinicaltrials.gov). Another well described culprit of metabolic disease is the 5′-AMP-activated protein kinase (AMPK) [117-119]. AMPK acts as an energy sensor by monitoring the ratio of AMP to ATP. At high concentrations of AMP, AMPK is activated by phosphorylation and begins to shut down ATP consuming processes, including fatty acid synthesis and sterol synthesis, and activates energy producing pathways to maintain energy homeostasis. AMPK directly inhibits ACACA, HMG-CoA reductase, and SREBP-1, making it an attractive target for inhibiting lipogenesis in prostate cancer (reviewed in [120]). Several small molecules and natural products have been shown to either directly or indirectly activate AMPK, including Metformin, a well-recognized and approved treatment for type II diabetes. Metformin and other natural products known to activate AMPK are currently being tested in clinical trials to determine their efficacy in treating various cancers (reviewed in [121]).
Despite complications of inhibitor development, lipogenesis in prostate cancer can be exploited for prostate cancer diagnosis as well. Detection of FASN in prostate is an early diagnostic marker and indicator of disease progression [12, 122]. Both SREBP-1c and FASN staining correlate with high Gleason grade [12, 16, 122]. Expression of FASN may complement the current AMACR staining to distinguish questionable cases during diagnosis [123]. Radiolabeled substrates of fatty acid precursors, such as 1-11C-acetate and 4-18F-fluoro-L-glutamic acid, also carry diagnostic value for cancer patients [124, 125]. Radiolabeled 11C-acetate displayed less background noise and proved more efficient over the standard 18F-fluorodeoxyglucose in diagnostic PET imaging for prostate cancer patients [125-127]; although, a very recent study reported complications in distinguishing benign hyperplasia and prostate cancer [128]. A clinical trial for 4-18F-fluoro-L-glutamic acid as a metabolic substrate was also recently completed (NCT00961831); results are pending.
Dietary Lipids and Prostate Cancer
Epidemiological studies show that prostate cancer is associated with obesity and increased body mass index [129-131], and research suggests that a diet low in fat decreases risk of prostate cancer development and progression [132-138]; however, some studies argue dietary fat is not associated with cancer risk [139, 140]. Diets around the globe vary in their total fat content, as well as their enrichment for specific lipids, which may contain varying quantities of saturated fatty acid (SAFA), monounsaturated fatty acid (MUFA), and polyunsaturated fatty acid (PUFA). These epidemiological studies correlating regional diets and risk of prostate cancer development have initiated a surge of research investigating the role of various types of dietary lipids in prostate cancer. Unfortunately, a large majority of epidemiology studies rely on patient questionnaires to assess the implications of diet in prostate cancer, and can often inaccurately reflect the patient’s true diet [141]. Many preclinical studies have attempted to ameliorate this inaccuracy and identify the dietary components affecting prostate cancer development and progression by formulating controlled diets in animal models; however, even preclinical studies remain controversial in their findings. A confounding factor between many preclinical studies is the variance in dietary formulation, specifically the source of dietary fat and the quantity of specific types of SAFA, MUFA, and PUFA, and especially their ratio to the respective control diet. Additionally, it can be difficult to distinguish whether the type of dietary fat or simply the quantity of fat consumed is the underlying culprit of disease. While the significance of diet was proposed long ago, only recently has literature attempted to discriminate molecular mechanisms and specific types of dietary fat that may contribute to prostate tumorigenesis and progression.
Saturated Fatty Acids
Common dietary sources rich in SAFA include animal meat, milk, eggs, palm oil, and coconut oil. The most prevalent species of dietary SAFA are palmitic acid (16:0) and stearic acid (18:0), although some foods, such as coconut oil, are rich in short chain SAFA such as lauric acid (12:0) and myristic acid (14:0) [142]. Several epidemiological studies have suggested increased consumption of SAFA correlates with increased risk of prostate cancer and reduced progression-free survival [143-145]; however others report no significant association [146, 147].
Although a prospective study in men suggests both short- and long-chain SAFA have similar negative effects on prostate cancer risk [148], preclinical data suggests short-chain SAFA may have a beneficial effect on prostate cancer [149]. A study in rats showed that coconut oil, which is rich in lauric acid, compared to sunflower oil, which is rich in linoleic acid (18:2, n-6), can prevent prostate hyperplasia [149]. As previously mentioned, a major difficulty in assessing dietary effects of fat is identifying a reasonable control. Arguably, this effect of a coconut oil diet may not necessarily be associated with the increased short-chain SAFA consumption, but could be due to the decreased consumption of linoleic acid.
Only a few preclinical animal studies have investigated the role of dietary saturated fat in prostate cancer, and results are controversial. Some studies suggest a diet high in saturated fat does not increase prostate tumor growth or survival in mice [140]; however, others argue the opposite. Escobar et al [150] elegantly demonstrated that the quality of dietary fat, rather than quantity, could regulate prostate growth and proliferation. When comparing isocaloric diets of only 7% fat, rats on a lard-derived diet displayed significantly increased prostate weight, testosterone, cell proliferation, and expression of androgen receptor compared to rats on a linseed oil-derived diet, which is rich in α-linolenic acid (18:3, n-3) [150]. These results suggest lard, which is rich in palmitic acid and oleic acid, may have cancer-promoting effects compared to an omega-3 PUFA enriched diet.
A study by Tamura et al. [151] demonstrated a role for a novel elongase of very long chain SAFA, ELOVL7, whose expression was found to be upregulated in prostate cancer. Overexpression of ELOVL7 supplemented with a high fat diet composed of beef fat increased tumor growth and testosterone production in mouse prostate tumor xenografts. Conversely, knockdown of ELOVL7 significantly decreased 20:0 and 22:0 SAFA, but not long chain MUFA or PUFA, in phospholipids and cholesterol esters in prostate cancer cells. These data suggest prostate cancer cells use SAFA, the most abundant fatty acid found in animal fat, as substrates for membrane and androgen production to drive tumor growth. Surprisingly, a study characterizing the kinetics and substrate specificity of purified ELOVL7 showed preferential activity toward carbon chains of 16-20 in length, regardless of the presence of double bonds, with the greatest activity for 18:3 (n-3) and 18:3 (n-6) fatty acids [152]. These results confound the data observed in prostate cancer cells, suggesting perhaps ELOVL7 knockdown was compensated by another ELOVL enzyme. If so, ELOVL5 may be suspect as it is the only other ELOVL enzyme previously implicated in prostate cancer and is strongly increased in response to androgen [153].
It is important to note that saturated fatty acids may be acquired through diet and produced de novo by fatty acid synthesis. Several studies have demonstrated that cell death induced by FASN inhibition in cell culture can be rescued by addition of palmitic acid, the primary product of FASN [17, 29, 30]. To date, no study has described a phenotypic rescue of FASN inhibition by dietary fatty acids in a cancer animal model. However, several non-cancer models have shown that a high-fat diet is unable to rescue the effects of FASN inhibition in liver and brain [37, 154, 155]. It is possible that the cellular pools of dietary and de novo fatty acids distinctly supply different cellular processes, or perhaps that prostate cancer cells, which require fatty acid synthesis for survival, are fulfilling a high demand for fatty acids that simply cannot be met by the diet. Considering the appeal of fatty acid synthesis inhibition as a cancer therapeutic strategy, it would be interesting to determine whether a high-fat diet could negate this therapeutic approach.
Monounsaturated Fatty Acids
Early epidemiological studies have highlighted a lower incidence of certain cancers in Mediterranean regions [156]. A plausible cause of such a statistic is the olive oil-enriched diet of Mediterranean dwellers. The main fatty acid constituent of olive oil is oleic acid, an 18-carbon, delta-9 MUFA (18:1, n-9). Other foods rich in oleic acid include almond oil, hazelnut oil, and safflower oil [142]. Despite these early findings, effects of dietary MUFA, particularly oleic acid, on prostate cancer risk remain especially controversial. Some studies suggest a protective effect of MUFA in cancer development [157], while others describe no association between MUFA and prostate cancer [144, 147]. What is more, some studies have described a cancer promoting effect of MUFA in humans and mice [132, 158]. Because the Mediterranean diet consists of other nutrients suspected to affect cancer risk, such as tomatoes and lycopene, resveratrol, high fish and low red meat consumption, all of which may influence the development of prostate cancer [159], it is difficult to distinguish whether one or a combination of dietary factors are contributing to the observed decrease in cancer incidence. Further investigation is required to determine a more precise role, if any, for dietary MUFA in prostate cancer.
Similar to the conundrum between dietary SAFA and de novo SAFA, dietary MUFA and de novo MUFA may also have distinct roles in prostate cancer. While the effects of dietary MUFA remain quite a mystery, two studies have highlighted a role for SCD-1, the enzyme responsible for de novo MUFA synthesis, in the promotion of prostate cancer [26, 78]. While these studies describe the role of MUFA in the de novo lipogenesis pathway of cancer, no study has investigated the interplay between dietary fat and SCD-1 in prostate cancer.
Polyunsaturated Fatty Acids
In contrast to SAFA and MUFA, the two main types of PUFA, omega-6 (n-6) and omega-3 (n-3), cannot be synthesized de novo in mammals and are therefore acquired through the diet. Linoleic acid (LA, 18:2, n-6) is the most common dietary omega-6 PUFA and can be converted to arachidonic acid (AA, 20:4, n-6), which is a precursor to many different eicosanoids [160]. LA is enriched in certain oils such as that of corn, sunflower, and safflower [142]. α-Linolenic acid (α-LNA, 18:3, n-3) is the major precursor to long-chain omega-3 PUFA but its conversion is limited [161]. Therefore, omega-3 derived eicosanoids are primarily derived from dietary eicosapentaenoic acid (EPA, 20:5, n-3) and docosanoids from docosahexaenoic acid (DHA, 22:6, n-3). Although largely unclear, multiple mechanisms by which PUFA exert their effects in prostate cancer have been postulated. These include processes such as eicosanoid synthesis, angiogenesis, immune cell regulation, and membrane structure and function (reviewed [160, 162]).
It is generally understood that omega-6 PUFA increase prostate cancer risk and disease progression while omega-3 PUFA show a unique, protective effect (reviewed in [160, 163]). A notable exception to this general rule is dihomo-γ-linolenic acid (DGLA 20:3, n-6). DGLA is a metabolite produced from desaturation and elongation of LA. DGLA has been shown to possess anti-tumorigenic affects due to its conversion to the anti-inflammatory 1-series and 3-series of eicosanoids by cyclooxygenase and lipoxygenase enzymes, respectively (reviewed in [164]). Perhaps because the conversion of DGLA to AA is a relatively inefficient reaction [165], DGLA does not produce the same pro-tumorigenic results as AA. A recent publication suggests the anti-tumorigenic effects of DGLA are due to its ability to directly inhibit FASN. Work by Zhang et al. [166] depicted the crystal structure of DGLA bound to the thioesterase domain of FASN, consequently inhibiting fatty acid synthesis and inducing cancer cell death.
While the subject of good versus bad PUFA is still debated, no study has refuted the benefits of a balanced PUFA diet. In support of epidemiological data [167], several animal studies have described an anticancer benefit from diet containing a low omega-6 to omega-3 ratio [137, 168-170], which is in contrast to the high 30:1 ratio of the standard Western diet. Overall, literature seems to suggest dietary PUFA likely modulate prostate cancer biology; however, its molecular mechanisms warrant further investigations.
Conclusions
Both de novo and dietary lipids seem to be important contributors to prostate cancer growth and development. Cancer cells themselves have adjusted their metabolic priorities toward the synthesis of macromolecules, especially lipids for membrane biogenesis and signaling molecules, as an apparent means to fulfill their demanding proliferation rate. Even more peculiar, however, is their absolute dependence on fatty acid synthesis for survival, highlighting an attractive metabolic addiction that may be exploited at multiple molecular targets by cancer therapeutics. It remains uncertain whether cancer cells differentially utilize de novo lipids and dietary lipids. Although significant evidence from epidemiology and preclinical animal studies suggests dietary fat consumption affects prostate tumorigenesis and cancer progression, no study has effectively determined whether diet could potentially negate therapeutic efforts targeting cancer cell lipogenesis. Overall, a diet balanced in fat consumption may reduce the risk of prostate cancer development and prevent disease progression.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- [1].Baade PD, Youlden DR, Krnjacki LJ. International epidemiology of prostate cancer: geographical distribution and secular trends. Mol Nutr Food Res. 2009;53(2):171–84. doi: 10.1002/mnfr.200700511. [DOI] [PubMed] [Google Scholar]
- [2].Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61(4):212–36. doi: 10.3322/caac.20121. [DOI] [PubMed] [Google Scholar]
- [3].Warburg O, Wind F, Negelein E. The Metabolism of Tumors in the Body. J Gen Physiol. 1927;8(6):519–30. doi: 10.1085/jgp.8.6.519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Kuhajda FP, Jenner K, Wood FD, Hennigar RA, Jacobs LB, Dick JD, et al. Fatty acid synthesis: a potential selective target for antineoplastic therapy. Proc Natl Acad Sci U S A. 1994;91(14):6379–83. doi: 10.1073/pnas.91.14.6379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Jayakumar A, Tai MH, Huang WY, al-Feel W, Hsu M, Abu-Elheiga L, et al. Human fatty acid synthase: properties and molecular cloning. Proc Natl Acad Sci U S A. 1995;92(19):8695–9. doi: 10.1073/pnas.92.19.8695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Semenkovich CF, Coleman T, Fiedorek FT., Jr Human fatty acid synthase mRNA: tissue distribution, genetic mapping, and kinetics of decay after glucose deprivation. J Lipid Res. 1995;36(7):1507–21. [PubMed] [Google Scholar]
- [7].Medes G, Thomas A, Weinhouse S. Metabolism of neoplastic tissue. IV. A study of lipid synthesis in neoplastic tissue slices in vitro. Cancer Res. 1953;13(1):27–9. [PubMed] [Google Scholar]
- [8].Shah US, Dhir R, Gollin SM, Chandran UR, Lewis D, Acquafondata M, et al. Fatty acid synthase gene overexpression and copy number gain in prostate adenocarcinoma. Hum Pathol. 2006;37(4):401–9. doi: 10.1016/j.humpath.2005.11.022. [DOI] [PubMed] [Google Scholar]
- [9].Swinnen JV, Heemers H, Deboel L, Foufelle F, Heyns W, Verhoeven G. Stimulation of tumor-associated fatty acid synthase expression by growth factor activation of the sterol regulatory element-binding protein pathway. Oncogene. 2000;19(45):5173–81. doi: 10.1038/sj.onc.1203889. [DOI] [PubMed] [Google Scholar]
- [10].Swinnen JV, Esquenet M, Goossens K, Heyns W, Verhoeven G. Androgens stimulate fatty acid synthase in the human prostate cancer cell line LNCaP. Cancer Res. 1997;57(6):1086–90. [PubMed] [Google Scholar]
- [11].Swinnen JV, Vanderhoydonc F, Elgamal AA, Eelen M, Vercaeren I, Joniau S, et al. Selective activation of the fatty acid synthesis pathway in human prostate cancer. Int J Cancer. 2000;88(2):176–9. doi: 10.1002/1097-0215(20001015)88:2<176::aid-ijc5>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- [12].Swinnen JV, Roskams T, Joniau S, Van Poppel H, Oyen R, Baert L, et al. Overexpression of fatty acid synthase is an early and common event in the development of prostate cancer. Int J Cancer. 2002;98(1):19–22. doi: 10.1002/ijc.10127. [DOI] [PubMed] [Google Scholar]
- [13].Graner E, Tang D, Rossi S, Baron A, Migita T, Weinstein LJ, et al. The isopeptidase USP2a regulates the stability of fatty acid synthase in prostate cancer. Cancer Cell. 2004;5(3):253–61. doi: 10.1016/s1535-6108(04)00055-8. [DOI] [PubMed] [Google Scholar]
- [14].Wang S, Gao J, Lei Q, Rozengurt N, Pritchard C, Jiao J, et al. Prostate-specific deletion of the murine Pten tumor suppressor gene leads to metastatic prostate cancer. Cancer Cell. 2003;4(3):209–21. doi: 10.1016/s1535-6108(03)00215-0. [DOI] [PubMed] [Google Scholar]
- [15].Pearson HB, McCarthy A, Collins CM, Ashworth A, Clarke AR. Lkb1 deficiency causes prostate neoplasia in the mouse. Cancer Res. 2008;68(7):2223–32. doi: 10.1158/0008-5472.CAN-07-5169. [DOI] [PubMed] [Google Scholar]
- [16].Van de Sande T, Roskams T, Lerut E, Joniau S, Van Poppel H, Verhoeven G, et al. High-level expression of fatty acid synthase in human prostate cancer tissues is linked to activation and nuclear localization of Akt/PKB. J Pathol. 2005;206(2):214–9. doi: 10.1002/path.1760. [DOI] [PubMed] [Google Scholar]
- [17].Migita T, Ruiz S, Fornari A, Fiorentino M, Priolo C, Zadra G, et al. Fatty acid synthase: a metabolic enzyme and candidate oncogene in prostate cancer. J Natl Cancer Inst. 2009;101(7):519–32. doi: 10.1093/jnci/djp030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Liang G, Yang J, Horton JD, Hammer RE, Goldstein JL, Brown MS. Diminished hepatic response to fasting/refeeding and liver X receptor agonists in mice with selective deficiency of sterol regulatory element-binding protein-1c. J Biol Chem. 2002;277(11):9520–8. doi: 10.1074/jbc.M111421200. [DOI] [PubMed] [Google Scholar]
- [19].Huang WC, Li X, Liu J, Lin J, Chung LW. Activation of Androgen Receptor, Lipogenesis, and Oxidative Stress Converged by SREBP-1 Is Responsible for Regulating Growth and Progression of Prostate Cancer Cells. Mol Cancer Res. 2012;10(1):133–42. doi: 10.1158/1541-7786.MCR-11-0206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Milgraum LZ, Witters LA, Pasternack GR, Kuhajda FP. Enzymes of the fatty acid synthesis pathway are highly expressed in in situ breast carcinoma. Clin Cancer Res. 1997;3(11):2115–20. [PubMed] [Google Scholar]
- [21].Gansler TS, Hardman W, 3rd, Hunt DA, Schaffel S, Hennigar RA. Increased expression of fatty acid synthase (OA-519) in ovarian neoplasms predicts shorter survival. Hum Pathol. 1997;28(6):686–92. doi: 10.1016/s0046-8177(97)90177-5. [DOI] [PubMed] [Google Scholar]
- [22].Rashid A, Pizer ES, Moga M, Milgraum LZ, Zahurak M, Pasternack GR, et al. Elevated expression of fatty acid synthase and fatty acid synthetic activity in colorectal neoplasia. Am J Pathol. 1997;150(1):201–8. [PMC free article] [PubMed] [Google Scholar]
- [23].Menendez JA, Lupu R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat Rev Cancer. 2007;7(10):763–77. doi: 10.1038/nrc2222. [DOI] [PubMed] [Google Scholar]
- [24].Kuhajda FP. Fatty-acid synthase and human cancer: new perspectives on its role in tumor biology. Nutrition. 2000;16(3):202–8. doi: 10.1016/s0899-9007(99)00266-x. [DOI] [PubMed] [Google Scholar]
- [25].Swinnen JV, Brusselmans K, Verhoeven G. Increased lipogenesis in cancer cells: new players, novel targets. Curr Opin Clin Nutr Metab Care. 2006;9(4):358–65. doi: 10.1097/01.mco.0000232894.28674.30. [DOI] [PubMed] [Google Scholar]
- [26].Fritz V, Benfodda Z, Rodier G, Henriquet C, Iborra F, Avances C, et al. Abrogation of de novo lipogenesis by stearoyl-CoA desaturase 1 inhibition interferes with oncogenic signaling and blocks prostate cancer progression in mice. Mol Cancer Ther. 2010;9(6):1740–54. doi: 10.1158/1535-7163.MCT-09-1064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Hatzivassiliou G, Zhao F, Bauer DE, Andreadis C, Shaw AN, Dhanak D, et al. ATP citrate lyase inhibition can suppress tumor cell growth. Cancer Cell. 2005;8(4):311–21. doi: 10.1016/j.ccr.2005.09.008. [DOI] [PubMed] [Google Scholar]
- [28].Kridel SJ, Axelrod F, Rozenkrantz N, Smith JW. Orlistat is a novel inhibitor of fatty acid synthase with antitumor activity. Cancer Res. 2004;64(6):2070–5. doi: 10.1158/0008-5472.can-03-3645. [DOI] [PubMed] [Google Scholar]
- [29].Brusselmans K, De Schrijver E, Verhoeven G, Swinnen JV. RNA interference-mediated silencing of the acetyl-CoA-carboxylase-alpha gene induces growth inhibition and apoptosis of prostate cancer cells. Cancer Res. 2005;65(15):6719–25. doi: 10.1158/0008-5472.CAN-05-0571. [DOI] [PubMed] [Google Scholar]
- [30].De Schrijver E, Brusselmans K, Heyns W, Verhoeven G, Swinnen JV. RNA interference-mediated silencing of the fatty acid synthase gene attenuates growth and induces morphological changes and apoptosis of LNCaP prostate cancer cells. Cancer Res. 2003;63(13):3799–804. [PubMed] [Google Scholar]
- [31].Beckers A, Organe S, Timmermans L, Scheys K, Peeters A, Brusselmans K, et al. Chemical inhibition of acetyl-CoA carboxylase induces growth arrest and cytotoxicity selectively in cancer cells. Cancer Res. 2007;67(17):8180–7. doi: 10.1158/0008-5472.CAN-07-0389. [DOI] [PubMed] [Google Scholar]
- [32].Swinnen JV, Van Veldhoven PP, Timmermans L, De Schrijver E, Brusselmans K, Vanderhoydonc F, et al. Fatty acid synthase drives the synthesis of phospholipids partitioning into detergent-resistant membrane microdomains. Biochem Biophys Res Commun. 2003;302(4):898–903. doi: 10.1016/s0006-291x(03)00265-1. [DOI] [PubMed] [Google Scholar]
- [33].Zheng H, Liu W, Anderson LY, Jiang QX. Lipid-dependent gating of a voltage-gated potassium channel. Nat Commun. 2011;2:250. doi: 10.1038/ncomms1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Alli PM, Pinn ML, Jaffee EM, McFadden JM, Kuhajda FP. Fatty acid synthase inhibitors are chemopreventive for mammary cancer in neu-N transgenic mice. Oncogene. 2005;24(1):39–46. doi: 10.1038/sj.onc.1208174. [DOI] [PubMed] [Google Scholar]
- [35].Rysman E, Brusselmans K, Scheys K, Timmermans L, Derua R, Munck S, et al. De novo lipogenesis protects cancer cells from free radicals and chemotherapeutics by promoting membrane lipid saturation. Cancer Res. 2011;70(20):8117–26. doi: 10.1158/0008-5472.CAN-09-3871. [DOI] [PubMed] [Google Scholar]
- [36].Fiorentino M, Zadra G, Palescandolo E, Fedele G, Bailey D, Fiore C, et al. Overexpression of fatty acid synthase is associated with palmitoylation of Wnt1 and cytoplasmic stabilization of beta-catenin in prostate cancer. Lab Invest. 2008;88(12):1340–8. doi: 10.1038/labinvest.2008.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Chakravarthy MV, Lodhi IJ, Yin L, Malapaka RR, Xu HE, Turk J, et al. Identification of a physiologically relevant endogenous ligand for PPARalpha in liver. Cell. 2009;138(3):476–88. doi: 10.1016/j.cell.2009.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Zhou W, Simpson PJ, McFadden JM, Townsend CA, Medghalchi SM, Vadlamudi A, et al. Fatty acid synthase inhibition triggers apoptosis during S phase in human cancer cells. Cancer Res. 2003;63(21):7330–7. [PubMed] [Google Scholar]
- [39].Wilson JE. Isozymes of mammalian hexokinase: structure, subcellular localization and metabolic function. J Exp Biol. 2003;206(Pt 12):2049–57. doi: 10.1242/jeb.00241. [DOI] [PubMed] [Google Scholar]
- [40].Mathupala SP, Ko YH, Pedersen PL. Hexokinase II: cancer’s double-edged sword acting as both facilitator and gatekeeper of malignancy when bound to mitochondria. Oncogene. 2006;25(34):4777–86. doi: 10.1038/sj.onc.1209603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Moon JS, Jin WJ, Kwak JH, Kim HJ, Yun MJ, Kim JW, et al. Androgen stimulates glycolysis for de novo lipid synthesis by increasing the activities of hexokinase 2 and 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 2 in prostate cancer cells. Biochem J. 2011;433(1):225–33. doi: 10.1042/BJ20101104. [DOI] [PubMed] [Google Scholar]
- [42].Mazurek S, Boschek CB, Hugo F, Eigenbrodt E. Pyruvate kinase type M2 and its role in tumor growth and spreading. Semin Cancer Biol. 2005;15(4):300–8. doi: 10.1016/j.semcancer.2005.04.009. [DOI] [PubMed] [Google Scholar]
- [43].Eigenbrodt E, Reinacher M, Scheefers-Borchel U, Scheefers H, Friis R. Double role for pyruvate kinase type M2 in the expansion of phosphometabolite pools found in tumor cells. Crit Rev Oncog. 1992;3(1-2):91–115. [PubMed] [Google Scholar]
- [44].Christofk HR, Vander Heiden MG, Harris MH, Ramanathan A, Gerszten RE, Wei R, et al. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature. 2008;452(7184):230–3. doi: 10.1038/nature06734. [DOI] [PubMed] [Google Scholar]
- [45].Anastasiou D, Poulogiannis G, Asara JM, Boxer MB, Jiang JK, Shen M, et al. Inhibition of pyruvate kinase M2 by reactive oxygen species contributes to cellular antioxidant responses. Science. 2011;334(6060):1278–83. doi: 10.1126/science.1211485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Vander Heiden MG, Locasale JW, Swanson KD, Sharfi H, Heffron GJ, Amador-Noguez D, et al. Evidence for an alternative glycolytic pathway in rapidly proliferating cells. Science. 2010;329(5998):1492–9. doi: 10.1126/science.1188015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Koh HJ, Lee SM, Son BG, Lee SH, Ryoo ZY, Chang KT, et al. Cytosolic NADP+-dependent isocitrate dehydrogenase plays a key role in lipid metabolism. J Biol Chem. 2004;279(38):39968–74. doi: 10.1074/jbc.M402260200. [DOI] [PubMed] [Google Scholar]
- [48].Reitman ZJ, Yan H. Isocitrate dehydrogenase 1 and 2 mutations in cancer: alterations at a crossroads of cellular metabolism. J Natl Cancer Inst. 2010;102(13):932–41. doi: 10.1093/jnci/djq187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Murugan AK, Bojdani E, Xing M. Identification and functional characterization of isocitrate dehydrogenase 1 (IDH1) mutations in thyroid cancer. Biochem Biophys Res Commun. 2010;393(3):555–9. doi: 10.1016/j.bbrc.2010.02.095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Mycielska ME, Patel A, Rizaner N, Mazurek MP, Keun H, Ganapathy V, et al. Citrate transport and metabolism in mammalian cells: prostate epithelial cells and prostate cancer. Bioessays. 2009;31(1):10–20. doi: 10.1002/bies.080137. [DOI] [PubMed] [Google Scholar]
- [51].Kavanagh JP. Isocitric and citric acid in human prostatic and seminal fluid: implications for prostatic metabolism and secretion. Prostate. 1994;24(3):139–42. doi: 10.1002/pros.2990240307. [DOI] [PubMed] [Google Scholar]
- [52].Medrano A, Fernandez-Novell JM, Ramio L, Alvarez J, Goldberg E, Rivera M Montserrat, et al. Utilization of citrate and lactate through a lactate dehydrogenase and ATP-regulated pathway in boar spermatozoa. Mol Reprod Dev. 2006;73(3):369–78. doi: 10.1002/mrd.20414. [DOI] [PubMed] [Google Scholar]
- [53].Mycielska ME, Broke-Smith TP, Palmer CP, Beckerman R, Nastos T, Erguler K, et al. Citrate enhances in vitro metastatic behaviours of PC-3M human prostate cancer cells: status of endogenous citrate and dependence on aconitase and fatty acid synthase. Int J Biochem Cell Biol. 2006;38(10):1766–77. doi: 10.1016/j.biocel.2006.04.008. [DOI] [PubMed] [Google Scholar]
- [54].DeBerardinis RJ, Mancuso A, Daikhin E, Nissim I, Yudkoff M, Wehrli S, et al. Beyond aerobic glycolysis: transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc Natl Acad Sci U S A. 2007;104(49):19345–50. doi: 10.1073/pnas.0709747104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Liu X, Fu YM, Meadows GG. Differential effects of specific amino acid restriction on glucose metabolism, reduction/oxidation status and mitochondrial damage in DU145 and PC3 prostate cancer cells. Oncol Lett. 2011;2(2):349–55. doi: 10.3892/ol.2011.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Mullen AR, Wheaton WW, Jin ES, Chen PH, Sullivan LB, Cheng T, et al. Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature. 2012;481(7381):385–8. doi: 10.1038/nature10642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Metallo CM, Gameiro PA, Bell EL, Mattaini KR, Yang J, Hiller K, et al. Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia. Nature. 2012;481(7381):380–4. doi: 10.1038/nature10602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [58].Manning BD, Cantley LC. AKT/PKB signaling: navigating downstream. Cell. 2007;129(7):1261–74. doi: 10.1016/j.cell.2007.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [59].Gray IC, Stewart LM, Phillips SM, Hamilton JA, Gray NE, Watson GJ, et al. Mutation and expression analysis of the putative prostate tumour-suppressor gene PTEN. Br J Cancer. 1998;78(10):1296–300. doi: 10.1038/bjc.1998.674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [60].Li Y, Su J, DingZhang X, Zhang J, Yoshimoto M, Liu S, et al. PTEN deletion and heme oxygenase-1 overexpression cooperate in prostate cancer progression and are associated with adverse clinical outcome. J Pathol. 2011;224(1):90–100. doi: 10.1002/path.2855. [DOI] [PubMed] [Google Scholar]
- [61].Friedlander TW, Roy R, Tomlins SA, Ngo VT, Kobayashi Y, Azameera A, et al. Common structural and epigenetic changes in the genome of castration-resistant prostate cancer. Cancer Res. 2012;72(3):616–25. doi: 10.1158/0008-5472.CAN-11-2079. [DOI] [PubMed] [Google Scholar]
- [62].Chalhoub N, Baker SJ. PTEN and the PI3-kinase pathway in cancer. Annu Rev Pathol. 2009;4:127–50. doi: 10.1146/annurev.pathol.4.110807.092311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Van de Sande T, De Schrijver E, Heyns W, Verhoeven G, Swinnen JV. Role of the phosphatidylinositol 3′-kinase/PTEN/Akt kinase pathway in the overexpression of fatty acid synthase in LNCaP prostate cancer cells. Cancer Res. 2002;62(3):642–6. [PubMed] [Google Scholar]
- [64].Wang HQ, Altomare DA, Skele KL, Poulikakos PI, Kuhajda FP, Di Cristofano A, et al. Positive feedback regulation between AKT activation and fatty acid synthase expression in ovarian carcinoma cells. Oncogene. 2005;24(22):3574–82. doi: 10.1038/sj.onc.1208463. [DOI] [PubMed] [Google Scholar]
- [65].Tomek K, Wagner R, Varga F, Singer CF, Karlic H, Grunt TW. Blockade of fatty acid synthase induces ubiquitination and degradation of phosphoinositide-3-kinase signaling proteins in ovarian cancer. Mol Cancer Res. 2011;9(12):1767–79. doi: 10.1158/1541-7786.MCR-10-0467. [DOI] [PubMed] [Google Scholar]
- [66].Higgins MJ, Baselga J. Targeted therapies for breast cancer. J Clin Invest. 2011;121(10):3797–803. doi: 10.1172/JCI57152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [67].Minner S, Jessen B, Stiedenroth L, Burandt E, Kollermann J, Mirlacher M, et al. Low level HER2 overexpression is associated with rapid tumor cell proliferation and poor prognosis in prostate cancer. Clin Cancer Res. 2010;16(5):1553–60. doi: 10.1158/1078-0432.CCR-09-2546. [DOI] [PubMed] [Google Scholar]
- [68].Kumar-Sinha C, Ignatoski KW, Lippman ME, Ethier SP, Chinnaiyan AM. Transcriptome analysis of HER2 reveals a molecular connection to fatty acid synthesis. Cancer Res. 2003;63(1):132–9. [PubMed] [Google Scholar]
- [69].Yoon S, Lee MY, Park SW, Moon JS, Koh YK, Ahn YH, et al. Up-regulation of acetyl-CoA carboxylase alpha and fatty acid synthase by human epidermal growth factor receptor 2 at the translational level in breast cancer cells. J Biol Chem. 2007;282(36):26122–31. doi: 10.1074/jbc.M702854200. [DOI] [PubMed] [Google Scholar]
- [70].Kumar B, Koul S, Khandrika L, Meacham RB, Koul HK. Oxidative stress is inherent in prostate cancer cells and is required for aggressive phenotype. Cancer Res. 2008;68(6):1777–85. doi: 10.1158/0008-5472.CAN-07-5259. [DOI] [PubMed] [Google Scholar]
- [71].Shiota M, Yokomizo A, Tada Y, Inokuchi J, Kashiwagi E, Masubuchi D, et al. Castration resistance of prostate cancer cells caused by castration-induced oxidative stress through Twist1 and androgen receptor overexpression. Oncogene. 2010;29(2):237–50. doi: 10.1038/onc.2009.322. [DOI] [PubMed] [Google Scholar]
- [72].Shiota M, Yokomizo A, Naito S. Oxidative stress and androgen receptor signaling in the development and progression of castration-resistant prostate cancer. Free Radic Biol Med. 2011;51(7):1320–8. doi: 10.1016/j.freeradbiomed.2011.07.011. [DOI] [PubMed] [Google Scholar]
- [73].Clerkin JS, Naughton R, Quiney C, Cotter TG. Mechanisms of ROS modulated cell survival during carcinogenesis. Cancer Lett. 2008;266(1):30–6. doi: 10.1016/j.canlet.2008.02.029. [DOI] [PubMed] [Google Scholar]
- [74].Furuta E, Pai SK, Zhan R, Bandyopadhyay S, Watabe M, Mo YY, et al. Fatty acid synthase gene is up-regulated by hypoxia via activation of Akt and sterol regulatory element binding protein-1. Cancer Res. 2008;68(4):1003–11. doi: 10.1158/0008-5472.CAN-07-2489. [DOI] [PubMed] [Google Scholar]
- [75].Brar SS, Corbin Z, Kennedy TP, Hemendinger R, Thornton L, Bommarius B, et al. NOX5 NAD(P)H oxidase regulates growth and apoptosis in DU 145 prostate cancer cells. Am J Physiol Cell Physiol. 2003;285(2):C353–69. doi: 10.1152/ajpcell.00525.2002. [DOI] [PubMed] [Google Scholar]
- [76].Huang WC, Zhau HE, Chung LW. Androgen receptor survival signaling is blocked by anti-beta2-microglobulin monoclonal antibody via a MAPK/lipogenic pathway in human prostate cancer cells. J Biol Chem. 2010;285(11):7947–56. doi: 10.1074/jbc.M109.092759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [77].Yang X, Guo Z, Sun F, Li W, Alfano A, Shimelis H, et al. Novel membrane-associated androgen receptor splice variant potentiates proliferative and survival responses in prostate cancer cells. J Biol Chem. 2011;286(41):36152–60. doi: 10.1074/jbc.M111.265124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [78].Kim SJ, Choi H, Park SS, Chang C, Kim E. Stearoyl CoA desaturase (SCD) facilitates proliferation of prostate cancer cells through enhancement of androgen receptor transactivation. Mol Cells. 2011;31(4):371–7. doi: 10.1007/s10059-011-0043-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [79].Guseva NV, Rokhlin OW, Glover RA, Cohen MB. TOFA (5-tetradecyl-oxy-2-furoic acid) reduces fatty acid synthesis, inhibits expression of AR, neuropilin-1 and Mcl-1 and kills prostate cancer cells independent of p53 status. Cancer Biol Ther. 2011;12(1):80–5. doi: 10.4161/cbt.12.1.15721. [DOI] [PubMed] [Google Scholar]
- [80].Eberle D, Hegarty B, Bossard P, Ferre P, Foufelle F. SREBP transcription factors: master regulators of lipid homeostasis. Biochimie. 2004;86(11):839–48. doi: 10.1016/j.biochi.2004.09.018. [DOI] [PubMed] [Google Scholar]
- [81].Ettinger SL, Sobel R, Whitmore TG, Akbari M, Bradley DR, Gleave ME, et al. Dysregulation of sterol response element-binding proteins and downstream effectors in prostate cancer during progression to androgen independence. Cancer Res. 2004;64(6):2212–21. doi: 10.1158/0008-5472.can-2148-2. [DOI] [PubMed] [Google Scholar]
- [82].Chen Y, Hughes-Fulford M. Human prostate cancer cells lack feedback regulation of low-density lipoprotein receptor and its regulator, SREBP2. Int J Cancer. 2001;91(1):41–5. doi: 10.1002/1097-0215(20010101)91:1<41::aid-ijc1009>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- [83].Freeman MR, Solomon KR. Cholesterol and prostate cancer. J Cell Biochem. 2004;91(1):54–69. doi: 10.1002/jcb.10724. [DOI] [PubMed] [Google Scholar]
- [84].Hager MH, Solomon KR, Freeman MR. The role of cholesterol in prostate cancer. Curr Opin Clin Nutr Metab Care. 2006;9(4):379–85. doi: 10.1097/01.mco.0000232896.66791.62. [DOI] [PubMed] [Google Scholar]
- [85].Boudreau DM, Yu O, Johnson J. Statin use and cancer risk: a comprehensive review. Expert Opin Drug Saf. 2010;9(4):603–21. doi: 10.1517/14740331003662620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [86].Shannon J, Tewoderos S, Garzotto M, Beer TM, Derenick R, Palma A, et al. Statins and prostate cancer risk: a case-control study. Am J Epidemiol. 2005;162(4):318–25. doi: 10.1093/aje/kwi203. [DOI] [PubMed] [Google Scholar]
- [87].Solomon KR, Freeman MR. Do the cholesterol-lowering properties of statins affect cancer risk? Trends Endocrinol Metab. 2008;19(4):113–21. doi: 10.1016/j.tem.2007.12.004. [DOI] [PubMed] [Google Scholar]
- [88].Roy M, Kung HJ, Ghosh PM. Statins and prostate cancer: role of cholesterol inhibition vs. prevention of small GTP-binding proteins. Am J Cancer Res. 2011;1(4):542–61. [PMC free article] [PubMed] [Google Scholar]
- [89].Mener DJ. Prostate specific antigen reduction following statin therapy: Mechanism of action and review of the literature. IUBMB Life. 2010;62(8):584–90. doi: 10.1002/iub.355. [DOI] [PubMed] [Google Scholar]
- [90].Clendening JW, Penn LZ. Targeting tumor cell metabolism with statins. Oncogene. doi: 10.1038/onc.2012.6. advanced online publication, 6 February 2012; doi:10.1038/onc.2012.6, PMID: 22310279. [DOI] [PubMed] [Google Scholar]
- [91].Grover VK, Valadez JG, Bowman AB, Cooper MK. Lipid modifications of Sonic hedgehog ligand dictate cellular reception and signal response. PLoS One. 2011;6(7):e21353. doi: 10.1371/journal.pone.0021353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [92].Bailey JM, Singh PK, Hollingsworth MA. Cancer metastasis facilitated by developmental pathways: Sonic hedgehog, Notch, and bone morphogenic proteins. J Cell Biochem. 2007;102(4):829–39. doi: 10.1002/jcb.21509. [DOI] [PubMed] [Google Scholar]
- [93].Staubach S, Hanisch FG. Lipid rafts: signaling and sorting platforms of cells and their roles in cancer. Expert Rev Proteomics. 2011;8(2):263–77. doi: 10.1586/epr.11.2. [DOI] [PubMed] [Google Scholar]
- [94].Williams TM, Hassan GS, Li J, Cohen AW, Medina F, Frank PG, et al. Caveolin-1 promotes tumor progression in an autochthonous mouse model of prostate cancer: genetic ablation of Cav-1 delays advanced prostate tumor development in tramp mice. J Biol Chem. 2005;280(26):25134–45. doi: 10.1074/jbc.M501186200. [DOI] [PubMed] [Google Scholar]
- [95].Papadopoulos G, Delakas D, Nakopoulou L, Kassimatis T. Statins and prostate cancer: molecular and clinical aspects. Eur J Cancer. 2011;47(6):819–30. doi: 10.1016/j.ejca.2011.01.005. [DOI] [PubMed] [Google Scholar]
- [96].Crnalic S, Hornberg E, Wikstrom P, Lerner UH, Tieva A, Svensson O, et al. Nuclear androgen receptor staining in bone metastases is related to a poor outcome in prostate cancer patients. Endocr Relat Cancer. 2010;17(4):885–95. doi: 10.1677/ERC-10-0059. [DOI] [PubMed] [Google Scholar]
- [97].Mostaghel EA, Page ST, Lin DW, Fazli L, Coleman IM, True LD, et al. Intraprostatic androgens and androgen-regulated gene expression persist after testosterone suppression: therapeutic implications for castration-resistant prostate cancer. Cancer Res. 2007;67(10):5033–41. doi: 10.1158/0008-5472.CAN-06-3332. [DOI] [PubMed] [Google Scholar]
- [98].Cai C, Chen S, Ng P, Bubley GJ, Nelson PS, Mostaghel EA, et al. Intratumoral de novo steroid synthesis activates androgen receptor in castration-resistant prostate cancer and is upregulated by treatment with CYP17A1 inhibitors. Cancer Res. 2011;71(20):6503–13. doi: 10.1158/0008-5472.CAN-11-0532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [99].Cai C, Balk SP. Intratumoral androgen biosynthesis in prostate cancer pathogenesis and response to therapy. Endocr Relat Cancer. 2011;18(5):R175–82. doi: 10.1530/ERC-10-0339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [100].Thysell E, Surowiec I, Hornberg E, Crnalic S, Widmark A, Johansson AI, et al. Metabolomic characterization of human prostate cancer bone metastases reveals increased levels of cholesterol. PLoS One. 2010;5(12):e14175. doi: 10.1371/journal.pone.0014175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [101].Montgomery RB, Mostaghel EA, Vessella R, Hess DL, Kalhorn TF, Higano CS, et al. Maintenance of intratumoral androgens in metastatic prostate cancer: a mechanism for castration-resistant tumor growth. Cancer Res. 2008;68(11):4447–54. doi: 10.1158/0008-5472.CAN-08-0249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [102].Locke JA, Guns ES, Lubik AA, Adomat HH, Hendy SC, Wood CA, et al. Androgen levels increase by intratumoral de novo steroidogenesis during progression of castration-resistant prostate cancer. Cancer Res. 2008;68(15):6407–15. doi: 10.1158/0008-5472.CAN-07-5997. [DOI] [PubMed] [Google Scholar]
- [103].Mostaghel EA, Solomon KR, Pelton K, Freeman MR, Montgomery RB. Impact of circulating cholesterol levels on growth and intratumoral androgen concentration of prostate tumors. PLoS One. 2012;7(1):e30062. doi: 10.1371/journal.pone.0030062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [104].Brusselmans K, De Schrijver E, Heyns W, Verhoeven G, Swinnen JV. Epigallocatechin-3-gallate is a potent natural inhibitor of fatty acid synthase in intact cells and selectively induces apoptosis in prostate cancer cells. Int J Cancer. 2003;106(6):856–62. doi: 10.1002/ijc.11317. [DOI] [PubMed] [Google Scholar]
- [105].Orita H, Coulter J, Lemmon C, Tully E, Vadlamudi A, Medghalchi SM, et al. Selective inhibition of fatty acid synthase for lung cancer treatment. Clin Cancer Res. 2007;13(23):7139–45. doi: 10.1158/1078-0432.CCR-07-1186. [DOI] [PubMed] [Google Scholar]
- [106].McFadden JM, Medghalchi SM, Thupari JN, Pinn ML, Vadlamudi A, Miller KI, et al. Application of a flexible synthesis of (5R)-thiolactomycin to develop new inhibitors of type I fatty acid synthase. J Med Chem. 2005;48(4):946–61. doi: 10.1021/jm049389h. [DOI] [PubMed] [Google Scholar]
- [107].Loftus TM, Jaworsky DE, Frehywot GL, Townsend CA, Ronnett GV, Lane MD, et al. Reduced food intake and body weight in mice treated with fatty acid synthase inhibitors. Science. 2000;288(5475):2379–81. doi: 10.1126/science.288.5475.2379. [DOI] [PubMed] [Google Scholar]
- [108].Kuhajda FP, Pizer ES, Li JN, Mani NS, Frehywot GL, Townsend CA. Synthesis and antitumor activity of an inhibitor of fatty acid synthase. Proc Natl Acad Sci U S A. 2000;97(7):3450–4. doi: 10.1073/pnas.050582897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [109].Vazquez MJ, Leavens W, Liu R, Rodriguez B, Read M, Richards S, et al. Discovery of GSK837149A, an inhibitor of human fatty acid synthase targeting the beta-ketoacyl reductase reaction. FEBS J. 2008;275(7):1556–67. doi: 10.1111/j.1742-4658.2008.06314.x. [DOI] [PubMed] [Google Scholar]
- [110].Maier T, Leibundgut M, Ban N. The crystal structure of a mammalian fatty acid synthase. Science. 2008;321(5894):1315–22. doi: 10.1126/science.1161269. [DOI] [PubMed] [Google Scholar]
- [111].Kim EK, Miller I, Aja S, Landree LE, Pinn M, McFadden J, et al. C75, a fatty acid synthase inhibitor, reduces food intake via hypothalamic AMP-activated protein kinase. J Biol Chem. 2004;279(19):19970–6. doi: 10.1074/jbc.M402165200. [DOI] [PubMed] [Google Scholar]
- [112].Landree LE, Hanlon AL, Strong DW, Rumbaugh G, Miller IM, Thupari JN, et al. C75, a fatty acid synthase inhibitor, modulates AMP-activated protein kinase to alter neuronal energy metabolism. J Biol Chem. 2004;279(5):3817–27. doi: 10.1074/jbc.M310991200. [DOI] [PubMed] [Google Scholar]
- [113].Braun S, Bitton-Worms K, LeRoith D. The link between the metabolic syndrome and cancer. Int J Biol Sci. 2011;7(7):1003–15. doi: 10.7150/ijbs.7.1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [114].Faulds MH, Dahlman-Wright K. Metabolic diseases and cancer risk. Curr Opin Oncol. 2012;24(1):58–61. doi: 10.1097/CCO.0b013e32834e0582. [DOI] [PubMed] [Google Scholar]
- [115].Hirsch HA, Iliopoulos D, Joshi A, Zhang Y, Jaeger SA, Bulyk M, et al. A transcriptional signature and common gene networks link cancer with lipid metabolism and diverse human diseases. Cancer Cell. 2010;17(4):348–61. doi: 10.1016/j.ccr.2010.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [116].Jiralerspong S, Palla SL, Giordano SH, Meric-Bernstam F, Liedtke C, Barnett CM, et al. Metformin and pathologic complete responses to neoadjuvant chemotherapy in diabetic patients with breast cancer. J Clin Oncol. 2009;27(20):3297–302. doi: 10.1200/JCO.2009.19.6410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [117].Ruderman N, Prentki M. AMP kinase and malonyl-CoA: targets for therapy of the metabolic syndrome. Nat Rev Drug Discov. 2004;3(4):340–51. doi: 10.1038/nrd1344. [DOI] [PubMed] [Google Scholar]
- [118].Luo Z, Saha AK, Xiang X, Ruderman NB. AMPK, the metabolic syndrome and cancer. Trends Pharmacol Sci. 2005;26(2):69–76. doi: 10.1016/j.tips.2004.12.011. [DOI] [PubMed] [Google Scholar]
- [119].Viollet B, Andreelli F, Jorgensen SB, Perrin C, Flamez D, Mu J, et al. Physiological role of AMP-activated protein kinase (AMPK): insights from knockout mouse models. Biochem Soc Trans. 2003;31(Pt 1):216–9. doi: 10.1042/bst0310216. [DOI] [PubMed] [Google Scholar]
- [120].Zadra G, Priolo C, Patnaik A, Loda M. New strategies in prostate cancer: targeting lipogenic pathways and the energy sensor AMPK. Clin Cancer Res. 2010;16(13):3322–8. doi: 10.1158/1078-0432.CCR-09-1955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [121].Flavin R, Zadra G, Loda M. Metabolic alterations and targeted therapies in prostate cancer. J Pathol. 2011;223:283–94. doi: 10.1002/path.2809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [122].Pflug BR, Pecher SM, Brink AW, Nelson JB, Foster BA. Increased fatty acid synthase expression and activity during progression of prostate cancer in the TRAMP model. Prostate. 2003;57(3):245–54. doi: 10.1002/pros.10297. [DOI] [PubMed] [Google Scholar]
- [123].Wu X, Zayzafoon M, Zhang X, Hameed O. Is there a role for fatty acid synthase in the diagnosis of prostatic adenocarcinoma?: A comparison with AMACR. Am J Clin Pathol. 2011;136(2):239–46. doi: 10.1309/AJCP0Y5QWWYDKCJE. [DOI] [PubMed] [Google Scholar]
- [124].Krasikova RN, Kuznetsova OF, Fedorova OS, Belokon YN, Maleev VI, Mu L, et al. 4-[18F]fluoroglutamic acid (BAY 85-8050), a new amino acid radiotracer for PET imaging of tumors: synthesis and in vitro characterization. J Med Chem. 2011;54(1):406–10. doi: 10.1021/jm101068q. [DOI] [PubMed] [Google Scholar]
- [125].Oyama N, Akino H, Kanamaru H, Suzuki Y, Muramoto S, Yonekura Y, et al. 11C-acetate PET imaging of prostate cancer. J Nucl Med. 2002;43(2):181–6. [PubMed] [Google Scholar]
- [126].Fricke E, Machtens S, Hofmann M, van den Hoff J, Bergh S, Brunkhorst T, et al. Positron emission tomography with 11C-acetate and 18F-FDG in prostate cancer patients. Eur J Nucl Med Mol Imaging. 2003;30(4):607–11. doi: 10.1007/s00259-002-1104-y. [DOI] [PubMed] [Google Scholar]
- [127].Vavere AL, Kridel SJ, Wheeler FB, Lewis JS. 1-11C-acetate as a PET radiopharmaceutical for imaging fatty acid synthase expression in prostate cancer. J Nucl Med. 2008;49(2):327–34. doi: 10.2967/jnumed.107.046672. [DOI] [PubMed] [Google Scholar]
- [128].Mena E, Turkbey B, Mani H, Adler S, Valera VA, Bernardo M, et al. 11C-Acetate PET/CT in Localized Prostate Cancer: A Study with MRI and Histopathologic Correlation. J Nucl Med. 2012;53(4):1–8. doi: 10.2967/jnumed.111.096032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [129].Rodriguez C, Freedland SJ, Deka A, Jacobs EJ, McCullough ML, Patel AV, et al. Body mass index, weight change, and risk of prostate cancer in the Cancer Prevention Study II Nutrition Cohort. Cancer Epidemiol Biomarkers Prev. 2007;16(1):63–9. doi: 10.1158/1055-9965.EPI-06-0754. [DOI] [PubMed] [Google Scholar]
- [130].Giovannucci E, Liu Y, Platz EA, Stampfer MJ, Willett WC. Risk factors for prostate cancer incidence and progression in the health professionals follow-up study. Int J Cancer. 2007;121(7):1571–8. doi: 10.1002/ijc.22788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [131].Freedland SJ. Obesity and prostate cancer: a growing problem. Clin Cancer Res. 2005;11(19 Pt 1):6763–6. doi: 10.1158/1078-0432.CCR-05-1305. [DOI] [PubMed] [Google Scholar]
- [132].Narita S, Tsuchiya N, Saito M, Inoue T, Kumazawa T, Yuasa T, et al. Candidate genes involved in enhanced growth of human prostate cancer under high fat feeding identified by microarray analysis. Prostate. 2008;68(3):321–35. doi: 10.1002/pros.20681. [DOI] [PubMed] [Google Scholar]
- [133].Mukherjee P, Sotnikov AV, Mangian HJ, Zhou JR, Visek WJ, Clinton SK. Energy intake and prostate tumor growth, angiogenesis, and vascular endothelial growth factor expression. J Natl Cancer Inst. 1999;91(6):512–23. doi: 10.1093/jnci/91.6.512. [DOI] [PubMed] [Google Scholar]
- [134].Aronson WJ, Barnard RJ, Freedland SJ, Henning S, Elashoff D, Jardack PM, et al. Growth inhibitory effect of low fat diet on prostate cancer cells: results of a prospective, randomized dietary intervention trial in men with prostate cancer. J Urol. 2010;183(1):345–50. doi: 10.1016/j.juro.2009.08.104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [135].Salem S, Salahi M, Mohseni M, Ahmadi H, Mehrsai A, Jahani Y, et al. Major dietary factors and prostate cancer risk: a prospective multicenter case-control study. Nutr Cancer. 2011;63(1):21–7. doi: 10.1080/01635581.2010.516875. [DOI] [PubMed] [Google Scholar]
- [136].Llaverias G, Danilo C, Wang Y, Witkiewicz AK, Daumer K, Lisanti MP, et al. A Western-type diet accelerates tumor progression in an autochthonous mouse model of prostate cancer. Am J Pathol. 2010;177(6):3180–91. doi: 10.2353/ajpath.2010.100568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [137].Kobayashi N, Barnard RJ, Said J, Hong-Gonzalez J, Corman DM, Ku M, et al. Effect of low-fat diet on development of prostate cancer and Akt phosphorylation in the Hi-Myc transgenic mouse model. Cancer Res. 2008;68(8):3066–73. doi: 10.1158/0008-5472.CAN-07-5616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [138].Lophatananon A, Archer J, Easton D, Pocock R, Dearnaley D, Guy M, et al. Dietary fat and early-onset prostate cancer risk. Br J Nutr. 2010;103(9):1375–80. doi: 10.1017/S0007114509993291. [DOI] [PubMed] [Google Scholar]
- [139].Freedland SJ, Mavropoulos J, Wang A, Darshan M, Demark-Wahnefried W, Aronson WJ, et al. Carbohydrate restriction, prostate cancer growth, and the insulin-like growth factor axis. Prostate. 2008;68(1):11–9. doi: 10.1002/pros.20683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [140].Lloyd JC, Antonelli JA, Phillips TE, Masko EM, Thomas JA, Poulton SH, et al. Effect of isocaloric low fat diet on prostate cancer xenograft progression in a hormone deprivation model. J Urol. 2010;183(4):1619–24. doi: 10.1016/j.juro.2009.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [141].Dennis LK, Snetselaar LG, Smith BJ, Stewart RE, Robbins ME. Problems with the assessment of dietary fat in prostate cancer studies. Am J Epidemiol. 2004;160(5):436–44. doi: 10.1093/aje/kwh243. [DOI] [PubMed] [Google Scholar]
- [142].Beare-Rogers J, Dieffenbacher A, J.V. H. Lexicon of Lipid Nutrition. Pure Applied Chemistry. 2001;73(4):685–744. [Google Scholar]
- [143].Strom SS, Yamamura Y, Forman MR, Pettaway CA, Barrera SL, DiGiovanni J. Saturated fat intake predicts biochemical failure after prostatectomy. Int J Cancer. 2008;122(11):2581–5. doi: 10.1002/ijc.23414. [DOI] [PubMed] [Google Scholar]
- [144].Park SY, Wilkens LR, Henning SM, Le Marchand L, Gao K, Goodman MT, et al. Circulating fatty acids and prostate cancer risk in a nested case-control study: the Multiethnic Cohort. Cancer Causes Control. 2009;20(2):211–23. doi: 10.1007/s10552-008-9236-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [145].Whittemore AS, Kolonel LN, Wu AH, John EM, Gallagher RP, Howe GR, et al. Prostate cancer in relation to diet, physical activity, and body size in blacks, whites, and Asians in the United States and Canada. J Natl Cancer Inst. 1995;87(9):652–61. doi: 10.1093/jnci/87.9.652. [DOI] [PubMed] [Google Scholar]
- [146].Jackson MD, Walker SP, Simpson-Smith CM, Lindsay CM, Smith G, McFarlane-Anderson N, et al. Associations of whole-blood fatty acids and dietary intakes with prostate cancer in Jamaica. Cancer Causes Control. 2012;23(1):23–33. doi: 10.1007/s10552-011-9850-4. [DOI] [PubMed] [Google Scholar]
- [147].Crowe FL, Key TJ, Appleby PN, Travis RC, Overvad K, Jakobsen MU, et al. Dietary fat intake and risk of prostate cancer in the European Prospective Investigation into Cancer and Nutrition. Am J Clin Nutr. 2008;87(5):1405–13. doi: 10.1093/ajcn/87.5.1405. [DOI] [PubMed] [Google Scholar]
- [148].Kurahashi N, Inoue M, Iwasaki M, Sasazuki S, Tsugane AS. Dairy product, saturated fatty acid, and calcium intake and prostate cancer in a prospective cohort of Japanese men. Cancer Epidemiol Biomarkers Prev. 2008;17(4):930–7. doi: 10.1158/1055-9965.EPI-07-2681. [DOI] [PubMed] [Google Scholar]
- [149].de Lourdes Arruzazabala M, Molina V, Mas R, Carbajal D, Marrero D, Gonzalez V, et al. Effects of coconut oil on testosterone-induced prostatic hyperplasia in Sprague-Dawley rats. J Pharm Pharmacol. 2007;59(7):995–9. doi: 10.1211/jpp.59.7.0012. [DOI] [PubMed] [Google Scholar]
- [150].Escobar EL, Gomes-Marcondes MC, Carvalho HF. Dietary fatty acid quality affects AR and PPARgamma levels and prostate growth. Prostate. 2009;69(5):548–58. doi: 10.1002/pros.20905. [DOI] [PubMed] [Google Scholar]
- [151].Tamura K, Makino A, Hullin-Matsuda F, Kobayashi T, Furihata M, Chung S, et al. Novel lipogenic enzyme ELOVL7 is involved in prostate cancer growth through saturated long-chain fatty acid metabolism. Cancer Res. 2009;69(20):8133–40. doi: 10.1158/0008-5472.CAN-09-0775. [DOI] [PubMed] [Google Scholar]
- [152].Naganuma T, Sato Y, Sassa T, Ohno Y, Kihara A. Biochemical characterization of the very long-chain fatty acid elongase ELOVL7. FEBS Lett. 2011;585(20):3337–41. doi: 10.1016/j.febslet.2011.09.024. [DOI] [PubMed] [Google Scholar]
- [153].Romanuik TL, Ueda T, Le N, Haile S, Yong TM, Thomson T, et al. Novel biomarkers for prostate cancer including noncoding transcripts. Am J Pathol. 2009;175(6):2264–76. doi: 10.2353/ajpath.2009.080868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [154].Chakravarthy MV, Zhu Y, Lopez M, Yin L, Wozniak DF, Coleman T, et al. Brain fatty acid synthase activates PPARalpha to maintain energy homeostasis. J Clin Invest. 2007;117(9):2539–52. doi: 10.1172/JCI31183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [155].Chakravarthy MV, Pan Z, Zhu Y, Tordjman K, Schneider JG, Coleman T, et al. “New” hepatic fat activates PPARalpha to maintain glucose, lipid, and cholesterol homeostasis. Cell Metab. 2005;1(5):309–22. doi: 10.1016/j.cmet.2005.04.002. [DOI] [PubMed] [Google Scholar]
- [156].Trichopoulou A, Lagiou P, Kuper H, Trichopoulos D. Cancer and Mediterranean dietary traditions. Cancer Epidemiol Biomarkers Prev. 2000;9(9):869–73. [PubMed] [Google Scholar]
- [157].Lopez-Miranda J, Perez-Jimenez F, Ros E, De Caterina R, Badimon L, Covas MI, et al. Olive oil and health: summary of the II international conference on olive oil and health consensus report, Jaen and Cordoba (Spain) 2008. Nutr Metab Cardiovasc Dis. 2010;20(4):284–94. doi: 10.1016/j.numecd.2009.12.007. [DOI] [PubMed] [Google Scholar]
- [158].Bidoli E, Talamini R, Bosetti C, Negri E, Maruzzi D, Montella M, et al. Macronutrients, fatty acids, cholesterol and prostate cancer risk. Ann Oncol. 2005;16(1):152–7. doi: 10.1093/annonc/mdi010. [DOI] [PubMed] [Google Scholar]
- [159].Venkateswaran V, Klotz LH. Diet and prostate cancer: mechanisms of action and implications for chemoprevention. Nat Rev Urol. 2010;7(8):442–53. doi: 10.1038/nrurol.2010.102. [DOI] [PubMed] [Google Scholar]
- [160].Berquin IM, Edwards IJ, Kridel SJ, Chen YQ. Polyunsaturated fatty acid metabolism in prostate cancer. Cancer Metastasis Rev. 2011;30(3-4):295–309. doi: 10.1007/s10555-011-9299-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [161].Burdge GC, Calder PC. Conversion of alpha-linolenic acid to longer-chain polyunsaturated fatty acids in human adults. Reprod Nutr Dev. 2005;45(5):581–97. doi: 10.1051/rnd:2005047. [DOI] [PubMed] [Google Scholar]
- [162].Berquin IM, Edwards IJ, Chen YQ. Multi-targeted therapy of cancer by omega-3 fatty acids. Cancer Lett. 2008;269(2):363–77. doi: 10.1016/j.canlet.2008.03.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [163].Chen YQ, Edwards IJ, Kridel SJ, Thornburg T, Berquin IM. Dietary fat-gene interactions in cancer. Cancer Metastasis Rev. 2007;26(3-4):535–51. doi: 10.1007/s10555-007-9075-x. [DOI] [PubMed] [Google Scholar]
- [164].Wang X, Lin H, Gu Y. Multiple roles of dihomo-gamma-linolenic acid against proliferation diseases. Lipids Health Dis. 2012;11(1):25. doi: 10.1186/1476-511X-11-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [165].Bernert JT, Jr., Sprecher H. Studies to determine the role rates of chain elongation and desaturation play in regulating the unsaturated fatty acid composition of rat liver lipids. Biochim Biophys Acta. 1975;398(3):354–63. doi: 10.1016/0005-2760(75)90186-1. [DOI] [PubMed] [Google Scholar]
- [166].Zhang W, Chakravarty B, Zheng F, Gu Z, Wu H, Mao J, et al. Crystal structure of FAS thioesterase domain with polyunsaturated fatty acyl adduct and inhibition by dihomo-gamma-linolenic acid. Proc Natl Acad Sci U S A. 2011;108(38):15757–62. doi: 10.1073/pnas.1112334108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [167].Williams CD, Whitley BM, Hoyo C, Grant DJ, Iraggi JD, Newman KA, et al. A high ratio of dietary n-6/n-3 polyunsaturated fatty acids is associated with increased risk of prostate cancer. Nutr Res. 2011;31(1):1–8. doi: 10.1016/j.nutres.2011.01.002. [DOI] [PubMed] [Google Scholar]
- [168].Berquin IM, Min Y, Wu R, Wu J, Perry D, Cline JM, et al. Modulation of prostate cancer genetic risk by omega-3 and omega-6 fatty acids. J Clin Invest. 2007;117(7):1866–75. doi: 10.1172/JCI31494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [169].Kobayashi N, Barnard RJ, Henning SM, Elashoff D, Reddy ST, Cohen P, et al. Effect of altering dietary omega-6/omega-3 fatty acid ratios on prostate cancer membrane composition, cyclooxygenase-2, and prostaglandin E2. Clin Cancer Res. 2006;12(15):4662–70. doi: 10.1158/1078-0432.CCR-06-0459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [170].Akinsete JA, Ion G, Witte TR, Hardman WE. Consumption of high omega-3 fatty acid diet suppressed prostate tumorigenesis in C3(1) Tag mice. Carcinogenesis. 2012;33(1):140–8. doi: 10.1093/carcin/bgr238. [DOI] [PMC free article] [PubMed] [Google Scholar]