Abstract
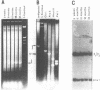
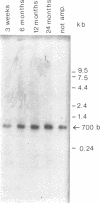
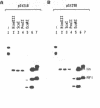
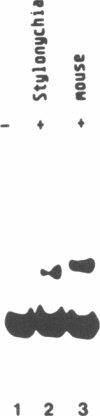
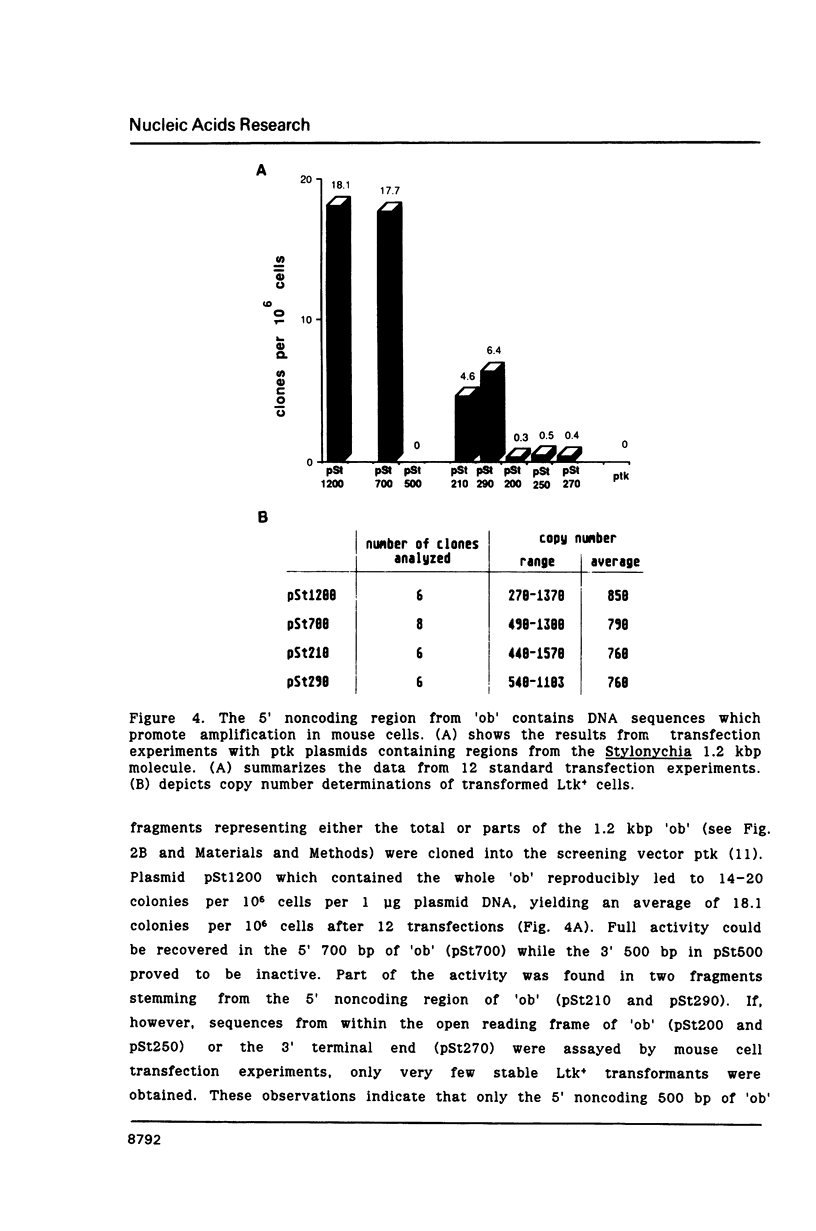
The macronucleus of the hypotrichous ciliate Stylonychia lemnae contains a 1218 bp long DNA molecule which becomes highly amplified during vegetative growth due to a continuous overreplication over a long time range. The region which is located upstream the open reading frame of the overamplified 1.2kbp Stylonychia DNA molecule enabled plasmids containing an inefficiently transcribed thymidine kinase gene to persist and amplify upon transfection into mouse L fibroblasts under selective conditions. This region contains long AT-rich stretches. The AT-rich sequences interact with a previously characterized HMG-I like protein from mouse Ehrlich ascites tumour cells. A binding activity for AT-rich stretches could also be identified in macronuclear extracts from Stylonychia lemnae. We suggest a common mechanism for overamplification in Stylonychia macronuclei during vegetative growth and amplification of plasmid DNA in heterologous mouse cells under the influence of a common element.
Full text
PDF


















Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ammermann D. Morphology and development of the macronuclei of the ciliates Stylonychia mytilus and Euplotes aediculatus. Chromosoma. 1971;33(2):209–238. doi: 10.1007/BF00285634. [DOI] [PubMed] [Google Scholar]
- Ammermann D., Steinbrück G., von Berger L., Hennig W. The development of the macronucleus in the ciliated protozoan Stylonychia mytilus. Chromosoma. 1974 May 10;45(4):401–429. doi: 10.1007/BF00283386. [DOI] [PubMed] [Google Scholar]
- Broach J. R., Li Y. Y., Feldman J., Jayaram M., Abraham J., Nasmyth K. A., Hicks J. B. Localization and sequence analysis of yeast origins of DNA replication. Cold Spring Harb Symp Quant Biol. 1983;47(Pt 2):1165–1173. doi: 10.1101/sqb.1983.047.01.132. [DOI] [PubMed] [Google Scholar]
- Caron F., Meyer E. Does Paramecium primaurelia use a different genetic code in its macronucleus? Nature. 1985 Mar 14;314(6007):185–188. doi: 10.1038/314185a0. [DOI] [PubMed] [Google Scholar]
- Cech T. R., Brehm S. L. Replication of the extrachromosomal ribosomal RNA genes of Tetrahymena thermophilia. Nucleic Acids Res. 1981 Jul 24;9(14):3531–3543. doi: 10.1093/nar/9.14.3531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conzelmann K. K., Helftenbein E. Nucleotide sequence and expression of two beta-tubulin genes in Stylonychia lemnae. J Mol Biol. 1987 Dec 20;198(4):643–653. doi: 10.1016/0022-2836(87)90207-5. [DOI] [PubMed] [Google Scholar]
- Dignam J. D., Lebovitz R. M., Roeder R. G. Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucleic Acids Res. 1983 Mar 11;11(5):1475–1489. doi: 10.1093/nar/11.5.1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilbert D., Cohen S. N. Autonomous replication in mouse cells: a correction. Cell. 1989 Jan 27;56(2):143–144. doi: 10.1016/0092-8674(89)90885-4. [DOI] [PubMed] [Google Scholar]
- Glisin V., Crkvenjakov R., Byus C. Ribonucleic acid isolated by cesium chloride centrifugation. Biochemistry. 1974 Jun 4;13(12):2633–2637. doi: 10.1021/bi00709a025. [DOI] [PubMed] [Google Scholar]
- Greslin A. F., Loukin S. H., Oka Y., Prescott D. M. An analysis of the macronuclear actin genes of Oxytricha. DNA. 1988 Oct;7(8):529–536. doi: 10.1089/dna.1.1988.7.529. [DOI] [PubMed] [Google Scholar]
- Hager D. A., Burgess R. R. Elution of proteins from sodium dodecyl sulfate-polyacrylamide gels, removal of sodium dodecyl sulfate, and renaturation of enzymatic activity: results with sigma subunit of Escherichia coli RNA polymerase, wheat germ DNA topoisomerase, and other enzymes. Anal Biochem. 1980 Nov 15;109(1):76–86. doi: 10.1016/0003-2697(80)90013-5. [DOI] [PubMed] [Google Scholar]
- Helftenbein E., Müller E. Both alpha-tubulin genes are transcriptionally active in Stylonychia lemnae. Curr Genet. 1988 May;13(5):425–432. doi: 10.1007/BF00365664. [DOI] [PubMed] [Google Scholar]
- Helftenbein E. Nucleotide sequence of a macronuclear DNA molecule coding for alpha-tubulin from the ciliate Stylonychia lemnae. Special codon usage: TAA is not a translation termination codon. Nucleic Acids Res. 1985 Jan 25;13(2):415–433. doi: 10.1093/nar/13.2.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrick G., Hunter D., Williams K., Kotter K. Alternative processing during development of a macronuclear chromosome family in Oxytricha fallax. Genes Dev. 1987 Dec;1(10):1047–1058. doi: 10.1101/gad.1.10.1047. [DOI] [PubMed] [Google Scholar]
- Holst A., Müller F., Zastrow G., Zentgraf H., Schwender S., Dinkl E., Grummt F. Murine genomic DNA sequences replicating autonomously in mouse L cells. Cell. 1988 Feb 12;52(3):355–365. doi: 10.1016/s0092-8674(88)80028-x. [DOI] [PubMed] [Google Scholar]
- Jahn C. L., Nilles L. A., Krikau M. F. Organization of the Euplotes crassus micronuclear genome. J Protozool. 1988 Nov;35(4):590–601. doi: 10.1111/j.1550-7408.1988.tb04157.x. [DOI] [PubMed] [Google Scholar]
- James C. D., Leffak M. Polarity of DNA replication through the avian alpha-globin locus. Mol Cell Biol. 1986 Apr;6(4):976–984. doi: 10.1128/mcb.6.4.976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaine B. P., Spear B. B. Nucleotide sequence of a macronuclear gene for actin in Oxytricha fallax. Nature. 1982 Feb 4;295(5848):430–432. doi: 10.1038/295430a0. [DOI] [PubMed] [Google Scholar]
- Kiss G. B., Amin A. A., Pearlman R. E. Two separate regions of the extrachromosomal ribosomal deoxyribonucleic acid of Tetrahymena thermophila enable autonomous replication of plasmids in Saccharomyces cerevisiae. Mol Cell Biol. 1981 Jun;1(6):535–543. doi: 10.1128/mcb.1.6.535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klobutcher L. A., Jahn C. L., Prescott D. M. Internal sequences are eliminated from genes during macronuclear development in the ciliated protozoan Oxytricha nova. Cell. 1984 Apr;36(4):1045–1055. doi: 10.1016/0092-8674(84)90054-0. [DOI] [PubMed] [Google Scholar]
- Linskens M. H., Huberman J. A. Organization of replication of ribosomal DNA in Saccharomyces cerevisiae. Mol Cell Biol. 1988 Nov;8(11):4927–4935. doi: 10.1128/mcb.8.11.4927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipps H. J., Erhardt P. DNase I hypersensitivity of the terminal inverted repeat DNA sequences in the macronucleus of the ciliate Stylonychia mytilus. FEBS Lett. 1981 Apr 20;126(2):219–222. doi: 10.1016/0014-5793(81)80246-3. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. A new method for sequencing DNA. Proc Natl Acad Sci U S A. 1977 Feb;74(2):560–564. doi: 10.1073/pnas.74.2.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKnight S. L., Kingsbury R. C., Spence A., Smith M. The distal transcription signals of the herpesvirus tk gene share a common hexanucleotide control sequence. Cell. 1984 May;37(1):253–262. doi: 10.1016/0092-8674(84)90321-0. [DOI] [PubMed] [Google Scholar]
- Mukherjee S., Patel I., Bastia D. Conformational changes in a replication origin induced by an initiator protein. Cell. 1985 Nov;43(1):189–197. doi: 10.1016/0092-8674(85)90023-6. [DOI] [PubMed] [Google Scholar]
- Murti K. G., Prescott D. M. Replication forms of the gene-sized DNA molecules of hypotrichous ciliates. Mol Cell Biol. 1983 Sep;3(9):1562–1566. doi: 10.1128/mcb.3.9.1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka Y., Honjo T. Common terminal repeats of the macronuclear DNA are absent from the micronuclear DNA in hypotrichous ciliate, Stylonychia pustulata. Nucleic Acids Res. 1983 Jul 11;11(13):4325–4333. doi: 10.1093/nar/11.13.4325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preer J. R., Jr, Preer L. B., Rudman B. M., Barnett A. J. Deviation from the universal code shown by the gene for surface protein 51A in Paramecium. Nature. 1985 Mar 14;314(6007):188–190. doi: 10.1038/314188a0. [DOI] [PubMed] [Google Scholar]
- Steinbrück G., Haas I., Hellmer K. H., Ammermann D. Characterization of macronuclear DNA in five species of ciliates. Chromosoma. 1981;83(2):199–208. doi: 10.1007/BF00286789. [DOI] [PubMed] [Google Scholar]
- Wigler M., Silverstein S., Lee L. S., Pellicer A., Cheng Y. c., Axel R. Transfer of purified herpes virus thymidine kinase gene to cultured mouse cells. Cell. 1977 May;11(1):223–232. doi: 10.1016/0092-8674(77)90333-6. [DOI] [PubMed] [Google Scholar]
- Wilkie N. M., Clements J. B., Boll W., Mantei N., Lonsdale D., Weissmann C. Hybrid plasmids containing an active thymidine kinase gene of Herpes simplex virus 1. Nucleic Acids Res. 1979 Oct 25;7(4):859–877. doi: 10.1093/nar/7.4.859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu H. M., Crothers D. M. The locus of sequence-directed and protein-induced DNA bending. Nature. 1984 Apr 5;308(5959):509–513. doi: 10.1038/308509a0. [DOI] [PubMed] [Google Scholar]
- Wünning I. U., Lipps H. J. A transformation system for the hypotrichous ciliate Stylonychia mytilus. EMBO J. 1983;2(10):1753–1757. doi: 10.1002/j.1460-2075.1983.tb01653.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zastrow G., Koehler U., Müller F., Klavinius A., Wegner M., Wienberg J., Weidle U. H., Grummt F. Distinct mouse DNA sequences enable establishment and persistence of plasmid DNA polymers in mouse cells. Nucleic Acids Res. 1989 Mar 11;17(5):1867–1879. doi: 10.1093/nar/17.5.1867. [DOI] [PMC free article] [PubMed] [Google Scholar]