Abstract
Three different mRNA analogues (28 to 34 nucleotides long) were prepared by T7 transcription from synthetic DNA templates. Each message contained the sequence ACC-GCG (coding for threonine and alanine, respectively), together with a single thio-U residue located at a variable position on the 5'-side of these coding triplets. A photo-reactive group was introduced by substitution of the thio-U with 4-azidophenacyl bromide. The messages were bound to E. coli 70S ribosomes in the presence of the appropriate tRNA-Thr or tRNA-Ala, and the azidophenyl group was photoactivated. Cross-linking was found to occur exclusively within the 30S subunit, with the 32P-label in the cross-linked mRNA being divided roughly equally between 30S ribosomal proteins and 16S RNA. Immunological analysis of the cross-linked proteins showed that, in the presence of either tRNA species, protein S7 was the primary target, whereas in the absence of tRNA only small amounts of protein S21 were cross-linked. The cross-link site to 16S RNA lay in all cases very close to its extreme 3'-terminus. These data indicate that the outgoing message leaves the cleft of the 30S subunit in a "northerly" direction.
Full text
PDF








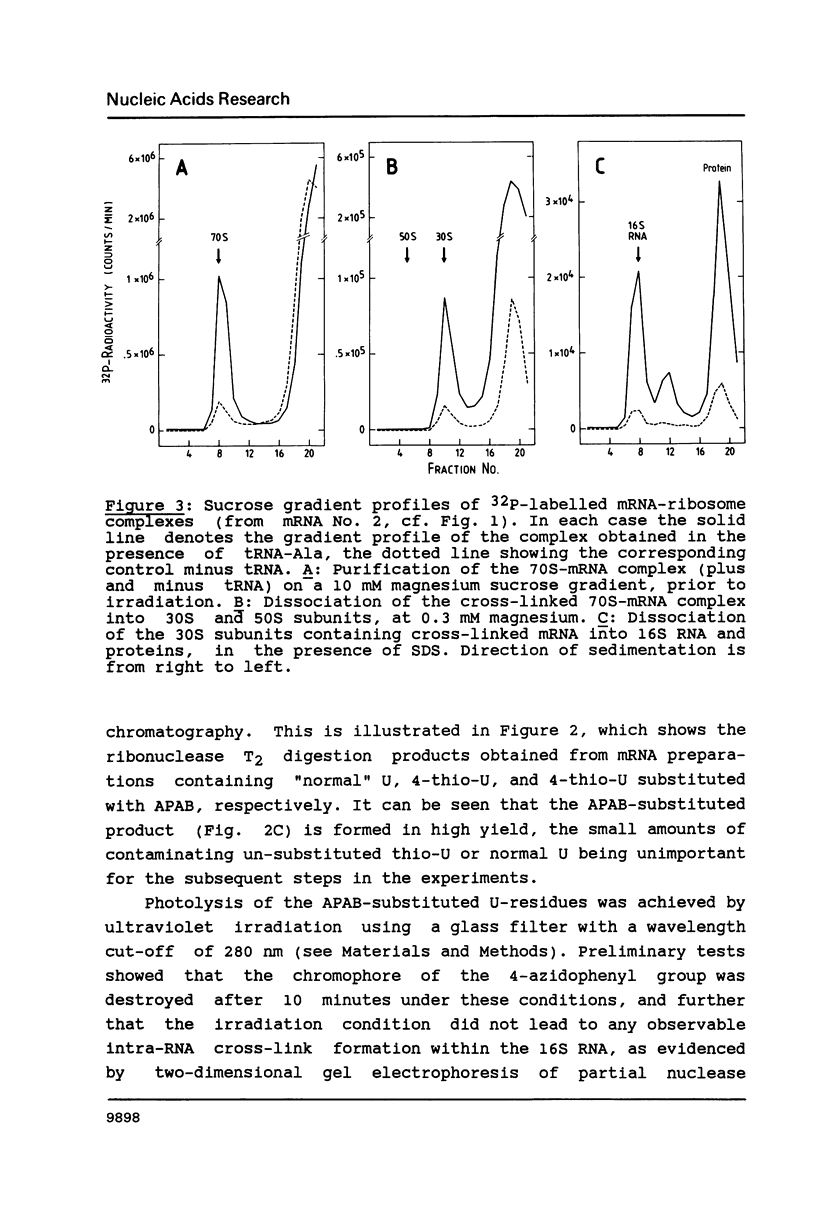

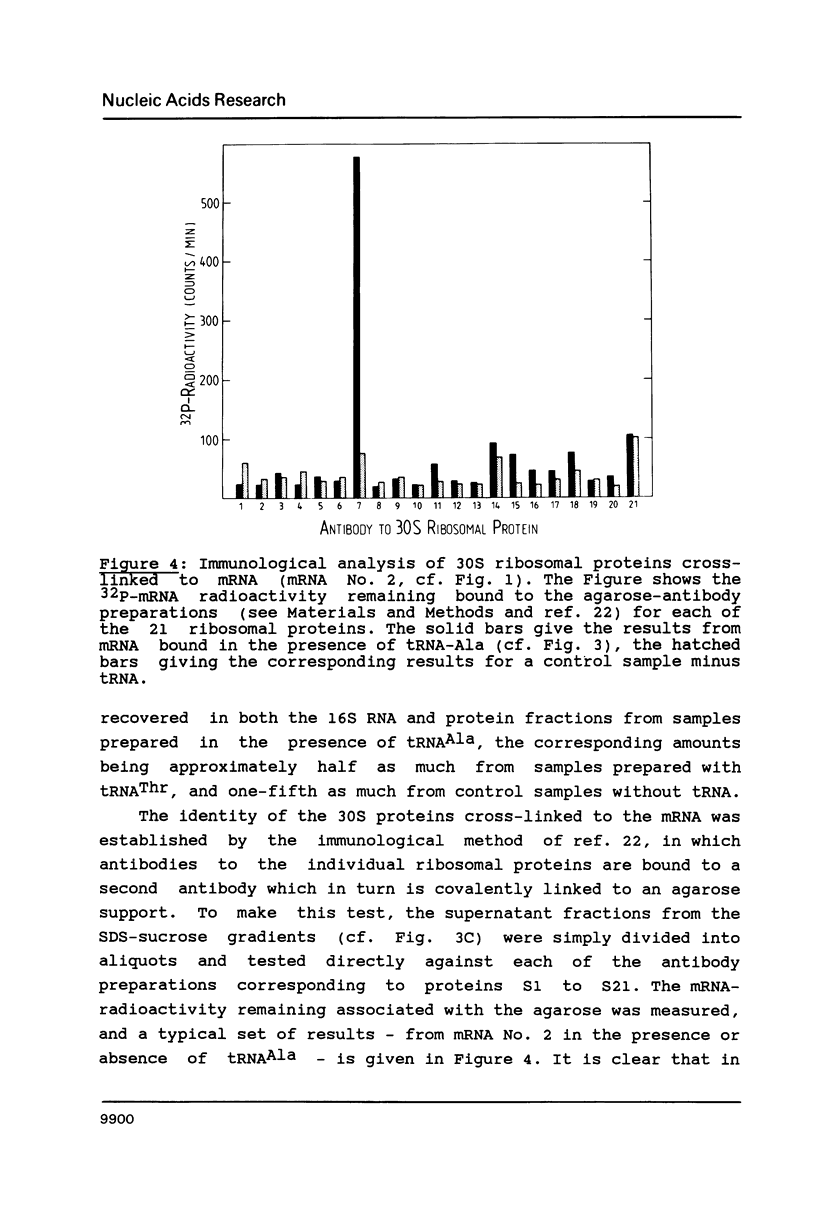








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Babkina G. T., Veniaminova A. G., Vladimirov S. N., Karpova G. G., Yamkovoy V. I., Berzin V. A., Gren E. J., Cielens I. E. Affinity labelling of Escherichia coli ribosomes with a benzylidene derivative of AUGU6 within initiation and pretranslocational complexes. FEBS Lett. 1986 Jul 7;202(2):340–344. doi: 10.1016/0014-5793(86)80714-1. [DOI] [PubMed] [Google Scholar]
- Brimacombe R., Atmadja J., Stiege W., Schüler D. A detailed model of the three-dimensional structure of Escherichia coli 16 S ribosomal RNA in situ in the 30 S subunit. J Mol Biol. 1988 Jan 5;199(1):115–136. doi: 10.1016/0022-2836(88)90383-x. [DOI] [PubMed] [Google Scholar]
- Brimacombe R., Stiege W., Kyriatsoulis A., Maly P. Intra-RNA and RNA-protein cross-linking techniques in Escherichia coli ribosomes. Methods Enzymol. 1988;164:287–309. doi: 10.1016/s0076-6879(88)64050-x. [DOI] [PubMed] [Google Scholar]
- Brimacombe R. The emerging three-dimensional structure and function of 16S ribosomal RNA. Biochemistry. 1988 Jun 14;27(12):4207–4214. doi: 10.1021/bi00412a001. [DOI] [PubMed] [Google Scholar]
- Calogero R. A., Pon C. L., Canonaco M. A., Gualerzi C. O. Selection of the mRNA translation initiation region by Escherichia coli ribosomes. Proc Natl Acad Sci U S A. 1988 Sep;85(17):6427–6431. doi: 10.1073/pnas.85.17.6427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evstafieva A. G., Shatsky I. N., Bogdanov A. A., Semenkov Y. P., Vasiliev V. D. Localization of 5' and 3' ends of the ribosome-bound segment of template polynucleotides by immune electron microscopy. EMBO J. 1983;2(5):799–804. doi: 10.1002/j.1460-2075.1983.tb01503.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Expert-Bezançon A., Wollenzien P. L. Three-dimensional arrangement of the Escherichia coli 16 S ribosomal RNA. J Mol Biol. 1985 Jul 5;184(1):53–66. doi: 10.1016/0022-2836(85)90043-9. [DOI] [PubMed] [Google Scholar]
- Gnirke A., Geigenmüller U., Rheinberger H. J., Nierhaus L. H. The allosteric three-site model for the ribosomal elongation cycle. Analysis with a heteropolymeric mRNA. J Biol Chem. 1989 May 5;264(13):7291–7301. [PubMed] [Google Scholar]
- Gornicki P., Nurse K., Hellmann W., Boublik M., Ofengand J. High resolution localization of the tRNA anticodon interaction site on the Escherichia coli 30 S ribosomal subunit. J Biol Chem. 1984 Aug 25;259(16):10493–10498. [PubMed] [Google Scholar]
- Gulle H., Hoppe E., Osswald M., Greuer B., Brimacombe R., Stöffler G. RNA-protein cross-linking in Escherichia coli 50S ribosomal subunits; determination of sites on 23S RNA that are cross-linked to proteins L2, L4, L24 and L27 by treatment with 2-iminothiolane. Nucleic Acids Res. 1988 Feb 11;16(3):815–832. doi: 10.1093/nar/16.3.815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hixson S. H., Hixson S. S. P-Azidophenacyl bromide, a versatile photolabile bifunctional reagent. Reaction with glyceraldehyde-3-phosphate dehydrogenase. Biochemistry. 1975 Sep 23;14(19):4251–4254. doi: 10.1021/bi00690a016. [DOI] [PubMed] [Google Scholar]
- Kang C. W., Cantor C. R. Structure of ribosome-bound messenger RNA as revealed by enzymatic accessibility studies. J Mol Biol. 1985 Jan 20;181(2):241–251. doi: 10.1016/0022-2836(85)90088-9. [DOI] [PubMed] [Google Scholar]
- Milligan J. F., Groebe D. R., Witherell G. W., Uhlenbeck O. C. Oligoribonucleotide synthesis using T7 RNA polymerase and synthetic DNA templates. Nucleic Acids Res. 1987 Nov 11;15(21):8783–8798. doi: 10.1093/nar/15.21.8783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moazed D., Stern S., Noller H. F. Rapid chemical probing of conformation in 16 S ribosomal RNA and 30 S ribosomal subunits using primer extension. J Mol Biol. 1986 Feb 5;187(3):399–416. doi: 10.1016/0022-2836(86)90441-9. [DOI] [PubMed] [Google Scholar]
- Olson H. M., Lasater L. S., Cann P. A., Glitz D. G. Messenger RNA orientation on the ribosome. Placement by electron microscopy of antibody-complementary oligodeoxynucleotide complexes. J Biol Chem. 1988 Oct 15;263(29):15196–15204. [PubMed] [Google Scholar]
- Prince J. B., Taylor B. H., Thurlow D. L., Ofengand J., Zimmermann R. A. Covalent crosslinking of tRNA1Val to 16S RNA at the ribosomal P site: identification of crosslinked residues. Proc Natl Acad Sci U S A. 1982 Sep;79(18):5450–5454. doi: 10.1073/pnas.79.18.5450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RATLIFF R. L., WEAVER R. H., LARDY H. A., KUBY S. A. NUCLEOSIDE TRIPHOSPHATE-NUCLEOSIDE DIPHOSPHATE TRANSPHOSPHORYLASE (NUCLEOSIDE DIPHOSPHOKINASE). I. ISOLATION OF THE CRYSTALLINE ENZYME FROM BREWERS' YEAST. J Biol Chem. 1964 Jan;239:301–309. [PubMed] [Google Scholar]
- Rheinberger H. J., Geigenmüller U., Wedde M., Nierhaus K. H. Parameters for the preparation of Escherichia coli ribosomes and ribosomal subunits active in tRNA binding. Methods Enzymol. 1988;164:658–670. doi: 10.1016/s0076-6879(88)64076-6. [DOI] [PubMed] [Google Scholar]
- Schüler D., Brimacombe R. The Escherichia coli 30S ribosomal subunit; an optimized three-dimensional fit between the ribosomal proteins and the 16S RNA. EMBO J. 1988 May;7(5):1509–1513. doi: 10.1002/j.1460-2075.1988.tb02970.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern S., Weiser B., Noller H. F. Model for the three-dimensional folding of 16 S ribosomal RNA. J Mol Biol. 1988 Nov 20;204(2):447–481. doi: 10.1016/0022-2836(88)90588-8. [DOI] [PubMed] [Google Scholar]
- Stiege W., Stade K., Schüler D., Brimacombe R. Covalent cross-linking of poly(A) to Escherichia coli ribosomes, and localization of the cross-link site within the 16S RNA. Nucleic Acids Res. 1988 Mar 25;16(6):2369–2388. doi: 10.1093/nar/16.6.2369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volckaert G., Fiers W. A micromethod for base analysis of 32P-labeled oligoribonulcleotides. Anal Biochem. 1977 Nov;83(1):222–227. doi: 10.1016/0003-2697(77)90530-9. [DOI] [PubMed] [Google Scholar]
- Weiss R. B., Dunn D. M., Dahlberg A. E., Atkins J. F., Gesteland R. F. Reading frame switch caused by base-pair formation between the 3' end of 16S rRNA and the mRNA during elongation of protein synthesis in Escherichia coli. EMBO J. 1988 May;7(5):1503–1507. doi: 10.1002/j.1460-2075.1988.tb02969.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zwieb C., Brimacombe R. Max-Planck-Institut für Molekulare Genetik, Abteilung Wittmann, Berlin-Dahlem, GFR. Nucleic Acids Res. 1979;6(5):1775–1790. doi: 10.1093/nar/6.5.1775. [DOI] [PMC free article] [PubMed] [Google Scholar]






