Abstract
Objective
The aim of this study was to compare the outcomes of enucleation versus resection in patients with small pancreatic, ampullary, and duodenal neuroendocrine tumors (NETs).
Methods
Multi-institutional retrospective review identified all patients with pancreatic and peri-pancreatic NETs who underwent surgery from January 1990 to October 2008. Patients with tumors ≤3 cm and without nodal or metastatic disease were included.
Results
Of the 271 patients identified, 122 (45%) met the inclusion criteria and had either an enucleation (n=37) and/or a resection (n=87). Enucleated tumors were more likely to be in the pancreatic head (P=0.003) or functioning (P<0.0001) and, when applicable, less likely to result in splenectomy (P=0.0003). The rate of pancreatic fistula formation was higher after enucleation (P<0.01), but the fistula severity tended to be worse following resection (P=0.07). The enucleation and resection patients had similar operative times, blood loss, overall morbidity, mortality, hospital stay, and 5-year survival. However, for pancreatic head tumors, enucleation resulted in decreased blood loss, operative time, and length of stay compared to pancreaticoduodenectomy (P<0.05).
Conclusion
These data suggest that most outcomes of enucleation and resection for small pancreatic and peri-pancreatic NETs are comparable. However, enucleation has better outcomes than pancreaticoduodenectomy for head lesions and the advantage of preserving splenic function for tail lesions.
Keywords: Neuroendocrine tumor, Islet cell tumor, Pancreas neoplasm, Ampulla of Vater, Duodenum
Introduction
Pancreatic islet cell tumors were first described in 1902 by Nicholls et al. and are rare, indolent neoplasms that can be either “benign” or malignant.1 In the last 10 years, the nomenclature of these lesions has evolved to pancreatic neuroendocrine tumor (NET) with a stress on the degree of tumor differentiation.2–5 These NETs of the pancreas, ampulla, and duodenum are usually sporadic and classified according to their ability to secrete hormones—functioning or non-functioning. Functioning tumors are frequently diagnosed earlier than their non-functioning counterparts because of the development of hormonal symptoms. As a result, non-functioning NETs present later in the disease course and are adversely associated with survival.6–8 Indications for surgery in patients with pancreatic and peri-pancreatic NETs include systemic symptoms due to hormone release, local compressive symptoms, and prevention of malignant transformation and/or dissemination.9 However, the optimal surgical management for pancreatic, ampullary, and duodenal NETs is controversial.
The first successful operation on a “benign” NET was an enucleation of a functioning pancreatic insulinoma in 1929.10 Subsequently, surgeons were classically taught to enucleate such lesions. Over the last 40 years, however, the morbidity and mortality of pancreatic resection has diminished from nearly 25% to less than 5% in certain “centers of excellence”.11–13 As a result, the proportion of patients undergoing pancreatic resection has increased (5% in the last 15 years), and pancreatectomy has become the standard therapy in many institutions, even for small lesions.13 However, data comparing these two surgical approaches for small pancreatic and peri-pancreatic NETs are lacking. Therefore, the purpose of this study was to document the morbidity, mortality, and outcomes of enucleation versus resection for small pancreatic, ampullary, and duodenal NETs at low risk for malignant transformation.
Methods
Multi-institutional retrospective review identified 271 patients with pancreatic, ampullary, and duodenal NETs who were operated on at four institutions or partner hospitals between January 1990 and October 2008. The participating institutions were Indiana University (IU), University of Wisconsin (UW), Northwestern University (NU), and the Medical College of Wisconsin (MCW). The IU, UW, NU, and MCW Institutional Review Boards each granted approval for the study. Electronic medical records, clinic charts, pathology reports, and tumor registries were used to determine patient demographics, pathology, treatment, and outcome data. The enucleation group included patients who underwent enucleation (Fig. 1), duodenal wall excision, or transduodenal ampullary tumor excision, while the resection group was comprised of patients treated by pancreaticoduodenectomy; distal, central, or total pancreatectomy; or partial pancreatectomy not otherwise specified (NOS). The transduodenal ampullary and local duodenal wall excisions were included in the enucleation group because formal pancreatic resection was not performed. The decision to perform an enucleation or resection was at the discretion of the attending surgeon. Prior to enucleation, the absence of liver metastases and peri-pancreatic lymphadenopathy was confirmed.
Figure 1.
Operative photograph depicting the enucleation of a pancreatic neuroendocrine tumor.
All specimens were reviewed by the pathologists at each institution and determined to be pancreatic or peri-pancreatic (ampullary or duodenal) NETs. Those tumors that came to surgery and were less than or equal to 3 cm by final pathology were included in this study. The 3-cm cutoff was chosen in order to create comparable groups since enucleation is not indicated for patients with large tumors and/or nodal or distant metastases. In addition, LaRosa and colleagues recently classified pancreatic NETs into stepwise groups of increasing malignant potential, and found that among well to moderately differentiated tumors the best overall discriminative power for size was at a cutoff of 3 cm.8 For the present investigation, malignant tumors were defined as having positive locoregional lymph nodes or the presence of distant metastatic disease and were excluded from the analysis. All tumors were stained for a variety of hormones including gastrin, glucagon, insulin, somatostatin, and vasoactive intestinal peptide (VIP) as well as neuroendocrine markers such as chromogranin A and synaptophysin. An NET was considered functional if symptoms from hormone release were present and/or the surgical specimen stained strongly for a specific hormone.
Morbidity was defined as any complication that occurred as a direct result of the enucleation or resection. Only complications that increased the hospital stay, required readmission, or necessitated invasive intervention were included. Postoperative pancreatic fistula was graded (A–C) as defined by the International Study Group on Pancreatic Fistula (ISGPF).14 Retrospective chart review was required in each case to grade the fistulas. Mortality was characterized as death within 30 days of surgery. Blood loss and operative time were obtained from operative notes and anesthesia records. Follow-up and survival data were obtained on all patients from hospital records, clinic notes, and the Social Security Death Index database (SSDI; http://ssdi.rootsweb.ancestry.com/cgi-bin/ssdi.cgi). Survival was calculated from the date of surgery to the date of death, last known follow-up, or last SSDI update (February 17, 2009; last accessed March 9, 2009).
Data are presented as mean±standard error of the mean (SEM) except where otherwise specified. Statistical analyses were performed by two-sided independent t test and chi-square analysis for continuous and categorical variables, respectively, with statistical significance achieved at P<0.05. For analysis of fistula severity, the proportion of grade A fistulas (less severe) was compared by chi-square to the proportion of grade B and C (more severe) fistulas. Survival rates were analyzed by the Kaplan–Meier actuarial method, with statistical significance determined by the log-rank statistic using SPSS statistical software version 10.0 (SPSS Inc., Chicago, IL, USA).15
Results
One hundred twenty-two (45%) of 271 patients with pancreatic, ampullary, and duodenal NETs met all study criteria and were included in this investigation. One hundred nineteen patients were excluded on the basis of positive lymph nodes and/or metastatic disease, and 30 additional patients who had no evidence of nodal or metastatic disease were excluded for size >3 cm. All 30 of these patients underwent resection; thus, no enucleation patients were excluded based on size. A total of 124 operations were performed and divided into two groups: enucleation (n=37) and resection (n=87). Two patients underwent both an enucleation and a distal pancreatectomy (DP) during the same operation. The median age of the patients was 53 years (range 23–90 years). The two groups were similar with respect to mean age and gender (Table 1).
Table 1.
Patient Demographics
| Variable | Enucleation | Resection | Total | P value |
|---|---|---|---|---|
| N, patients | 36 | 86 | 122 | |
| N, operations | 37 | 87 | 124 | |
| Age (years) | 56±2 | 52±1 | 53±1 | 0.14 |
| %, Female | 54 | 56 | 55 | 0.83 |
Data are presented as mean±standard error of the mean
Several different surgical procedures were performed in these patients and are summarized in Table 2. In the enucleation group, two of the 32 (6%) procedures were completed laparoscopically; whereas in the resection group, eight of 56 (14%) patients had a laparoscopic distal pancreatectomy. No splenectomies (0%) were required in any of the enucleated patients who had pancreatic tail tumors (n=9). Additionally, 16 of the 50 (32%) patients in the resection group with pancreatic tail tumors had spleen-preserving distal pancreatectomies. Therefore, when applicable, patients with tail lesions underwent significantly more splenectomies compared to the enucleation patients (P=0.0003). However, seven of the eight patients who had laparoscopic distal pancreatectomies had splenic-preserving procedures.
Table 2.
Surgical Management
| Operative details | Enucleation (n=37) | Resection (n=87) | Total (n=124) |
|---|---|---|---|
| Enucleation (%) | 32 (87) | 32 (26) | |
| Distal pancreatectomy (%) | 56 (64) | 56 (45) | |
| Pancreaticoduodenectomy (%) | 26 (30) | 26 (21) | |
| Central pancreatectomy (%) | 3 (4) | 3 (2) | |
| Transduodenal ampullary excision (%) | 3 (8) | 3 (2) | |
| Duodenal wall excision (%) | 2 (5) | 2 (2) | |
| Partial pancreatectomy, NOS (%) | 1 (1) | 1 (1) | |
| Total pancreatectomy (%) | 1 (1) | 1 (1) | |
| Splenectomy (%) | 0 (0)* | 34 (68) | 34 (27) |
NOS not otherwise specified
P=0.0003 vs. resection (only for tumors located in the tail of the pancreas)
A total of 128 NETs were enucleated (n=39) or resected (n=89) during the 124 procedures (Table 3). Overall, 39% of the tumors were located in the head of the pancreas, ampulla, or the duodenum (Table 3). Tumors that were enucleated were significantly more likely to be in the head of the pancreas when compared to tumors that were resected (P=0.003). The mean and median size of the lesions was similar between the enucleation and resection patients (Table 3). Functional status was able to be determined for 91 of 128 (71%) tumors (Table 3). Patients who underwent enucleation had a smaller proportion of non-functioning tumors compared to the patients who had resections (P<0.0001). The histologic subtypes seen on pathology are shown in Table 3.
Table 3.
Tumor Pathology
| Enucleation (n=37) | Resection (n=87) | Total (n=124) | |
|---|---|---|---|
| Location | |||
| Head/ampulla/duodenum (%) | 23 (59)* | 27 (30) | 50 (39) |
| Body/tail (%) | 16 (41) | 62 (70) | 78 (61) |
| Mean size (cm) | 1.8±0.1 | 1.7±0.1 | 1.7±0.1 |
| Median size (cm) | 1.7 | 1.6 | 1.7 |
| Pathology | |||
| Insulinoma (%) | 22 (63) | 11 (20) | 33 (32) |
| Non-functioning (%) | 8 (23)** | 42 (75) | 50 (55) |
| Gastrinoma (%) | 3 (9) | 3 (5) | 6 (7) |
| Glucagonoma (%) | 2 (6) | 0 | 2 (2) |
P=0.003 vs. resection,
P<0.0001 vs. functioning tumors
Examination of patient intraoperative and hospital data revealed that the enucleated and resected patients had comparable blood loss (P=0.11, Table 4). The mean operative time and length of stay between the two groups also was similar (P=0.11 and P=0.50, respectively; Table 4). However, when patients with tumors in the head of the pancreas were analyzed separately, both blood loss and operative time were greater after pancreaticoduodenectomy when compared to enucleation (blood loss=874±264 vs. 286±81 ml, P=0.04; operative time=334±30 vs. 229±34 min, P=0.03, respectively). In addition, the patients who underwent pancreaticoduodenectomy had a longer length of stay than patients who had enucleation (9.3±0.6 vs. 6.9±0.9 days, P=0.03).
Table 4.
Outcome Data
| Enucleation (n=37) | Resection (n=87) | Total (n=124) | |
|---|---|---|---|
| Estimated blood loss (ml) | 365±70 | 690±135 | 596±99 |
| Operative time (min) | 216±22 | 250±13 | 240±11 |
| Length of stay (days) | 8.7±1.2 | 10.2±1.3 | 9.7±1.0 |
| Complications (%) | 18 (49) | 38 (44) | 56 (45) |
| Complication type | |||
| Infectious (%) | 3 (8) | 17 (20) | 20 (16) |
| Fistula (%) | 14 (38)* | 13 (15) | 27 (20) |
| A | 8 (57) | 3 (23) | 11 (41) |
| B | 6 (43) | 8 (62) | 14 (52) |
| C | 0 | 2 (15) | 2 (7) |
| 30-day mortality (%) | 0 (0) | 1 (1.1) | 1 (0.8) |
| 5-year survival | 35 (94) | 78 (91) | 113 (92) |
| Mean follow-up (months) | 49.7±6.6 | 50.3±4.7 | 50.1±3.8 |
| Median follow-up (months) | 42 | 41 | 41 |
P<0.01 vs. resection
We also analyzed the overall morbidity experienced by the patients in this study which showed a similar rate of complications after enucleation and resection (P=0.69, Table 4). Patients who underwent enucleation experienced pancreatic fistula formation more frequently than resected patients (P<0.01, Table 4). However, when the fistulas were graded on an A, B, C scale according to the ISGPF classification, the majority of fistulas in enucleated patients was grade A, and the remainder was grade B (Table 4).14 No grade C fistulas developed after an enucleation. Conversely, fistulas that formed after resection were mostly grade B, and 15% were grade C (Table 4). Comparison of the proportion of grade A (less severe) fistulas to the proportion of grade B and C (more severe) fistulas revealed that the fistulas tended to be worse in patients who underwent resections, though this difference did not reach statistical significance (P=0.07). On the other hand, the percentage of infectious complications in the two groups was similar (P=0.18, Table 4). Small bowel obstruction, ileus, or delayed gastric emptying occurred after one (2.7%) enucleation as opposed to ten (11.5%) resections (P=0.17). The only operative death in the series occurred after a distal pancreatectomy, and the 30-day mortality rates were similar between enucleated and resected groups (P=1.00, Table 4).
In addition to examining complications, we measured survival and recurrence. Follow-up ranged from 1 to 161 months (Table 4). The 5-year survival of the patients in this study was 91.9%, which is consistent with the low malignant potential of small, node-negative tumors without evidence of metastatic disease. No difference in 5-year survival was detected between enucleated and resected patients (Fig. 2, Table 4). While nodal and distant metastases were absent in all patients at initial surgery, five patients who underwent resections experienced systemic recurrence of their disease. The incidence of systemic disease recurrence was comparable between patients who had enucleations versus resections (0% vs. 5.7%, P=0.32). No local recurrences were observed during the follow-up period.
Figure 2.
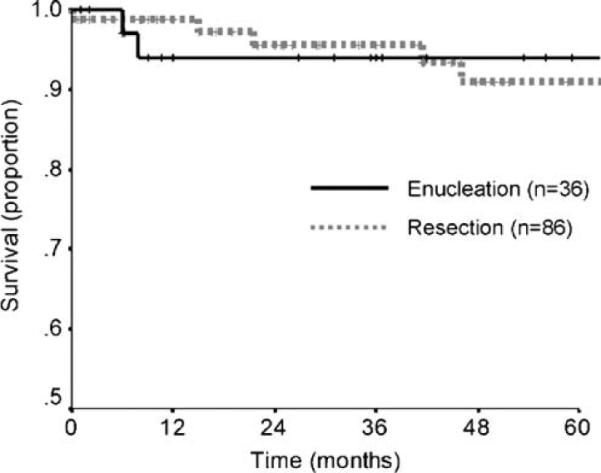
Kaplan–Meier actuarial survival curve comparing patients who underwent enucleation (n=36) versus those who underwent resection (n=86) (P=0.50 by log-rank test).
Discussion
In this series, we analyzed 122 patients with small (≤3 cm) pancreatic, ampullary, and duodenal NETs based upon the type of surgical treatment (enucleation vs. resection) received over an 18-year period at four institutions. Patients undergoing enucleation were more likely to have functional tumors in the head of the pancreas and less likely to have a splenectomy. The estimated blood loss, operative time, length of stay, overall morbidity, and all-cause mortality were similar between the enucleations and resections. While the pancreatic fistula rate was higher after enucleation, the fistulas tended to be less severe compared to those that occurred following resection. For patients with NETs in the head of the pancreas, enucleation was associated with decreased blood loss, operative time, and length of stay compared to pancreaticoduodenectomy.
The type of procedure performed for NETs of the pancreas, ampulla, and duodenum is important because surgical resection is considered to be the only curative modality.16 Even for small tumors, the risk of malignant transformation is present. This risk is highlighted by the 4% overall recurrence rate in this study of patients with 3 cm or less tumors who were node negative and metastasis free. The operative strategy regarding these NETs has focused on the relative advantages and disadvantages of local, less invasive procedures versus a formal pancreatic resection. As the morbidity and mortality of pancreatic resection at high-volume centers has decreased, distal pancreatectomy of small pancreatic tail lesions has become the norm.13 Similarly, pancreaticoduodenectomy, although more invasive than distal pancreatectomy, has grown to be an acceptable treatment option for small tumors of the pancreatic head, especially when in close proximity to the pancreatic duct.13,17,18 Central or middle segment pancreatectomy is also being employed in patients with pancreatic neck lesions.19,20 Reports of safe and effective laparoscopic resections have added to the types of surgical resections performed.21–23 However, risks associated with formal pancreatic resection include loss of healthy pancreatic tissue (with possible endocrine or exocrine insufficiency), the potential for splenectomy with distal resections, and a variety of complications related to bowel anastomoses or dysfunction of the stomach. Our study confirms that the rate of splenectomy is higher in patients undergoing resection. In addition, small bowel obstruction, ileus, and delayed gastric emptying occurred more frequently after resection, though this difference was not statistically significant.
As an alternative to resection, enucleation has remained an important part of the surgical armamentarium for pancreatic, ampullary, and duodenal NETs. The guiding principles for enucleation are the size of the tumor, absence of evidence of malignancy, and proximity to the pancreatic duct.18,24–26 Previous reported benefits of enucleation include reduced blood loss and operative time compared to resection, but not decreased length of stay.22,25–28 Like resection, enucleations can be performed laparoscopically with reduced blood loss and operative time when compared to resection.22,29,30 In this investigation, operative blood loss and time were statistically similar when all patients undergoing enucleation or resection were compared (P=0.11). However, when enucleation was evaluated against pancreaticoduodenectomy, the blood loss and operative time were greater after pancreaticoduodenectomy. Comparison of these same two procedures performed laparoscopically also supports this conclusion.22 The length of hospital stay for our enucleated and resected patients was similar which confirms prior findings.27 But our analysis indicated that hospital stay is significantly longer following pancreaticoduodenectomy than enucleation. In addition, enucleation has been shown to preserve pancreatic tissue.18
This investigation focused on small pancreatic and peripancreatic NETs with a relatively equal overall distribution of functional (45%) and non-functional (55%) lesions. A retrospective review of 125 patients with pancreatic NETs by Phan et al. revealed a similar proportion of functional hormone expression (52%).31 The distribution of functional tumor types in their study showed that insulinomas were the most common followed by gastrinomas, VIPomas, and glucagonomas.31 In the current series, the majority of functional tumors also were insulinomas, and the dispersion was similar, though no VIPomas were seen (Table 3). Thus, the functional classification of NETs in our study is comparable to previously published data.9,31–33 We also found that enucleated tumors were more likely to be functioning and in the head of the pancreas. These findings may be the result of surgical preference. Non-functioning tumors were resected more often, likely because non-functional status is a known adverse prognostic factor for survival.6–8 In addition, distal pancreatectomy is often the procedure of choice for pancreatic tail lesions.
In this study, we also examined the morbidity, mortality, and survival of enucleations compared to resections. The overall complication rate of 45% is comparable to rates observed in other studies that range from 14% to 50%.18,27,28,30,31,34–37 Our data reveal that overall morbidity does not differ significantly between patients undergoing enucleation (49%) versus resection (44%). Enucleation has previously been shown to have similar morbidity to resection while preserving pancreatic tissue.18 The 30-day mortality rate in this series (0.8%) also was comparable between the patients studied and was not different from previously reported rates for these operations.11,12,36 While the overall morbidity and mortality were similar, pancreatic fistula development occurred more commonly following enucleation. After enucleation, 38% of patients developed a pancreatic fistula which is within the previously reported range for enucleated patients—16% to 38%.22,27,30,31 In patients who were treated with resections, 15% formed fistulas which also is similar to other studies (range 9–26%).22,27,30,31 Retrospective chart review in each case showed that the leaks following enucleation were ISGPF grade A or B pancreatic fistulas which, by definition, are not associated with other complications or prolonged hospitalizations. Comparison of grade A versus grade B and C pancreatic fistulas in the two groups revealed that those fistulas diagnosed in the resected patients tended to be worse suggesting that the overall leak rate should be examined in the context of fistula grade. In terms of survival, when compared to tumors of other cell types, the prognosis of patients with pancreatic and peri-pancreatic NETs is very good and is excellent when only patients with “benign” or localized disease are evaluated.7,8,33 With a mean follow-up of 50 months, the survival in our study was no different between the surgical groups. In addition, the overall mortality for the resection group is in line with other reports of formal pancreatic resections.7,8,11,12
The present study is limited by the non-randomized retrospective design and inherent selection bias. Thus, resection may have been performed more often in patients with more aggressive disease. The resection group did have more systemic recurrences and a larger proportion of non-functioning tumors. Because enucleation is not indicated for patients with large tumors, lesions in close proximity to the pancreatic duct, or in the known presence of nodal or metastatic disease, a size limitation was essential to creating comparable groups. In recent years, laparoscopic approaches to NETs have been reported with increasing frequency.22,29,30 Therefore, in the future, open enucleation will need to be compared to laparoscopic enucleation. An analysis of the associated costs of these procedures also might enhance forthcoming studies. Due to the rarity of pancreatic and peri-pancreatic NETs, multi-institutional studies and larger population-based data sets also will be important to analyze in order to advance future practices.
In conclusion, this multi-institutional retrospective review of 122 patients compared enucleation to resection for small pancreatic, ampullary, and duodenal NETs. The overall effectiveness of enucleation and resection for these NETs is comparable, with similar morbidity, mortality, and survival.9,31 The surgical procedures also were similar with respect to estimated blood loss, operative time, and length of hospital stay. However, enucleation resulted in decreased blood loss, operative time, and duration of stay compared to pancreaticoduodenectomy when just patients with NETs in the head of the pancreas were considered. Furthermore, enucleation was associated with a significantly lower rate of splenectomy compared to all distal pancreatectomies. While enucleated patients had a higher incidence of pancreatic fistula formation compared to the resection group, the fistulas that formed after resection were mostly grade B and C, clinically significant fistulas. Therefore, enucleation of small pancreatic and peri-pancreatic NETs is safe and does not compromise long-term survival. This analysis further confirms that enucleation of small NETs with low malignant potential remains a viable operative approach. The procedure of choice in these patients with smaller NETs may be enucleation for lesions in the pancreatic head and distal pancreatectomy with splenic preservation for lesions in the pancreatic tail.
Acknowledgements
This work was supported by the American College of Surgeons Resident Research Scholarship and NIH Grant T32 CA009614 Physician Scientist Training in Cancer Medicine.
American College of Surgeons Resident Research Scholarship, NIH Grant T32 CA009614 Physician Scientist Training in Cancer Medicine.
References
- 1.Nicholls AG. Simple adenoma of the pancreas arising from an island of Langerhans. J Med Res. 1902;8:385–395. [PMC free article] [PubMed] [Google Scholar]
- 2.Capella C, Heitz PU, Hoefler H, et al. Revised classification of neuroendocrine tumors of the lung, pancreas, and gut. Virchows Arch. 1995;425:547–560. doi: 10.1007/BF00199342. [DOI] [PubMed] [Google Scholar]
- 3.Klöppel G, Perren A, Heitz PU. The gastroenteropancreatic neuroendocrine cell system and its tumors: the WHO classification. Ann NY Acad Sci. 2004;1014:13–27. doi: 10.1196/annals.1294.002. [DOI] [PubMed] [Google Scholar]
- 4.Klöppel G, Coulevard A, Perren A, et al. ENETs guidelines for the standards of care in patients with neuroendocrine tumors: towards a standardized approach to the diagnosis of gastroenteropancreatic neuroendocrine tumors and their prognostic stratification. Neuroendocrinology. doi: 10.1159/000182196. In press. Epub ahead of print on December 5, 2008. [DOI] [PubMed] [Google Scholar]
- 5.Rindi G, Klöppel G, Alhman H, et al. TNM staging of foregut (neuro)endocrine tumors: a consensus proposal including grading system. Virchows Arch. 2006;449:395–401. doi: 10.1007/s00428-006-0250-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bilimoria KY, Talamonti MS, Tomlinson JS, et al. Prognostic score predicting survival after resection of pancreatic neuroendocrine tumors: analysis of 3851 patients. Ann Surg. 2008;247(3):490–500. doi: 10.1097/SLA.0b013e31815b9cae. [DOI] [PubMed] [Google Scholar]
- 7.Halfdanarson TR, Rabe KG, Rubin J, Peterson GM. Pancreatic neuroendocrine tumors (PNETs): incidence in prognosis and recent trend toward improved survival. Ann Onc. 2008;19(10):1727–1733. doi: 10.1093/annonc/mdn351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.LaRosa S, Klersy C, Uccella S, et al. Improved histologic and clinicopathologic criteria for evaluation of pancreatic endocrine tumors. Human Pathol. 2009;40:30–40. doi: 10.1016/j.humpath.2008.06.005. [DOI] [PubMed] [Google Scholar]
- 9.Yeo CJ, Wang BH, Anthone GJ, Cameron JL. Surgical experience with pancreatic islet-cell tumors. Arch Surg. 1993;128:1143–1148. doi: 10.1001/archsurg.1993.01420220063008. [DOI] [PubMed] [Google Scholar]
- 10.Howland G, Campbell WR, Maltby EJ, Robinson WL. Dysinsulinism: convulsions and coma due to islet cell tumor of the pancreas, with operation and cure. JAMA. 1929;93:674–679. [Google Scholar]
- 11.Lillemoe KD, Kaushal S, Cameron JL, Sohn TA, Pitt HA, Yeo CJ. Distal pancreatectomy: indications and outcomes in 235 patients. Ann Surg. 1999;229:693–698. doi: 10.1097/00000658-199905000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yeo CJ, Cameron JL, Sohn TA, Lillemoe KD, Pitt HA, Talamini MA, Hruban RH, Ord SE, Sauter PK, Coleman J, Zahurak ML, Grochow LB, Abrams RA. Six hundred fifty consecutive pancreaticoduodenectomies in the 1990s: pathology, complications, and outcomes. Ann Surg. 1997;226:248–257. doi: 10.1097/00000658-199709000-00004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bilimoria KY, Tomlinson JS, Merkow RP, et al. Clinicopathologic features and treatment trends in pancreatic neuroendocrine tumors: analysis of 9,821 patients. J Gastrointest Surg. 2007;11:1460–1467. doi: 10.1007/s11605-007-0263-3. discussion 1467–1469. [DOI] [PubMed] [Google Scholar]
- 14.Bassi C, Dervenis C, Butturini G, et al. Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery. 2005;138(1):8–13. doi: 10.1016/j.surg.2005.05.001. [DOI] [PubMed] [Google Scholar]
- 15.Kaplan E, Meier P. Non parametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–481. [Google Scholar]
- 16.Kouvaraki MA, Solorzano CC, Shapiro SE, Yao JC, Perrier ND, Lee JE, Evans DB. Surgical treatment of non-functioning pancreatic islet cell tumors. J Surg Oncol. 2005;89:170–185. doi: 10.1002/jso.20178. [DOI] [PubMed] [Google Scholar]
- 17.Phan GQ, Yeo CJ, Cameron JL, Maher MM, Hruban RH, Udelsman R. Pancreaticoduodenectomy for selected periampullary neuroendocrine tumors: fifty patients. Surgery. 1997;122:989–996. doi: 10.1016/s0039-6060(97)90200-2. [DOI] [PubMed] [Google Scholar]
- 18.Park BJ, Alexander HR, Libutti SK, Huang J, Royalty D, Skarulis MC, Jensen RT, Gorden P, Doppman JL, Shawker TH, Fraker DL, Norton JA, Bartlett DL. Operative management of islet-cell tumors arising in the head of the pancreas. Surgery. 1998;124:1056–1061. doi: 10.1067/msy.1998.92171. [DOI] [PubMed] [Google Scholar]
- 19.Christein JD, Smoot RL, Farnell MB. Central pancreatectomy: a technique for the resection of pancreatic neck lesions. Arch Surg. 2006;141(3):293–299. doi: 10.1001/archsurg.141.3.293. [DOI] [PubMed] [Google Scholar]
- 20.Lavu H, Knuth JL, Baker MS, et al. Middle segment pancreatectomy can be safely incorporated into a pancreatic surgeon's clinical practice. HPB (Oxford) 2008;10(6):491–497. doi: 10.1080/13651820802356580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Assalia A, Gagner M. Laparoscopic pancreatic surgery for islet cell tumors of the pancreas. World J Surg. 2004;28(12):1239–1247. doi: 10.1007/s00268-004-7617-8. [DOI] [PubMed] [Google Scholar]
- 22.Fernández-Cruz L, Blanco L, Cosa R, Rendón H. Is laparoscopic resection adequate in patients with neuroendocrine pancreatic tumors. World J Surg. 2008;32(5):904–917. doi: 10.1007/s00268-008-9467-2. [DOI] [PubMed] [Google Scholar]
- 23.Luo Y, Liu R, Hu MG, Mu YM, An LC, Huang ZQ. Laparoscopic surgery for pancreatic insulinomas: a single-institution experience of 29 cases. J Gastrointest Surg. 2009 May;13(5):945–950. doi: 10.1007/s11605-009-0830-x. [DOI] [PubMed] [Google Scholar]
- 24.Sarmiento JM, Farnell MB, Que FG, Nagorney DM. Pancreaticoduodenectomy for islet cell tumors of the head of the pancreas: long-term survival analysis. World J Surg. 2002;26:1267–1271. doi: 10.1007/s00268-002-6714-9. [DOI] [PubMed] [Google Scholar]
- 25.Balzano G, Zerbi A, Veronesi P, Cristallo M, Di Carlo V. Surgical treatment of benign and borderline neoplasms of the pancreatic body. Dig Surg. 2003;20:506–510. doi: 10.1159/000073646. [DOI] [PubMed] [Google Scholar]
- 26.Dralle H, Krohn SL, Karges W, Boehm BO, Brauckhoff M, Gimm O. Surgery of resectable nonfunctioning neuroendocrine pancreatic tumors. World J Surg. 2004;28:1248–1260. doi: 10.1007/s00268-004-7609-8. [DOI] [PubMed] [Google Scholar]
- 27.Kiely JM, Nakeeb A, Komorowski RA, Wilson SD, Pitt HA. Cystic pancreatic neoplasms: enucleate or resect. J Gastrointest Surg. 2003;7:890–897. doi: 10.1007/s11605-003-0035-7. [DOI] [PubMed] [Google Scholar]
- 28.Talamini MA, Moesinger R, Yeo CJ, Poulose B, Hruban RH, Cameron JL, Pitt HA. Cystadenomas of the pancreas: is enucleation an adequate operation. Ann Surg. 1998;227:896–903. doi: 10.1097/00000658-199806000-00013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shimizu S, Tanaka M, Konomi H, Mizumoto K, Yamaguchi K. Laparoscopic pancreatic surgery: current indications and surgical results. Surg Endosc. 2004;18:402–406. doi: 10.1007/s00464-003-8164-3. [DOI] [PubMed] [Google Scholar]
- 30.Kooby DA, Gillespie T, Bentrem D, et al. Left-sided pancreatectomy: a multicenter comparison of laparoscopic and open approaches. Ann Surg. 2008;248(3):438–446. doi: 10.1097/SLA.0b013e318185a990. [DOI] [PubMed] [Google Scholar]
- 31.Phan GQ, Yeo CJ, Hruban RH, Lillemoe KD, Pitt HA, Cameron JL. Surgical experience with pancreatic and peripancreatic neuroendocrine tumors: review of 125 patients. J Gastrointest Surg. 1998;2:472–482. [PubMed] [Google Scholar]
- 32.Touzios JG, Kiely JM, Pitt SC, Rilling WS, Quebbeman EJ, Wilson SD, Pitt HA. Neuroendocrine hepatic metastases: does aggressive management improve survival. Ann Surg. 2005;241:776–783. doi: 10.1097/01.sla.0000161981.58631.ab. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wiedenmann B, Jensen RT, Mignon M, Modlin CI, Skogseid B, Doherty G, Oberg K. Preoperative diagnosis and surgical management of neuroendocrine gastroenteropancreatic tumors: general recommendations by a consensus workshop. World J Surg. 1998;22:309–318. doi: 10.1007/s002689900387. [DOI] [PubMed] [Google Scholar]
- 34.Ahrendt SA, Komorowski RA, Demeure MJ, Wilson SD, Pitt HA. Cystic pancreatic neuroendocrine tumors: is preoperative diagnosis possible. J Gastrointest Surg. 2002;6:66–74. doi: 10.1016/s1091-255x(01)00020-8. [DOI] [PubMed] [Google Scholar]
- 35.Baker MS, Knuth JL, DeWitt J, et al. Pancreatic cystic neuroendocrine tumors: preoperative diagnosis with endoscopic ultrasound and fine-needle immunocytology. J Gastrointest Surg. 2008;12(3):450–456. doi: 10.1007/s11605-007-0219-7. [DOI] [PubMed] [Google Scholar]
- 36.King J, Kazanijian K, Matsumoto J, et al. Distal pancreatectomy: incidence of post-operative diabetes mellitus. J Gastrointest Surg. 2008;12(9):1548–1553. doi: 10.1007/s11605-008-0560-5. [DOI] [PubMed] [Google Scholar]
- 37.Guo KJ, Liao HH, Tian YL, Guo RX, He SG, Shen K. Surgical treatment of nonfunctioning islet cell tumor: report of 41 cases. Hepatobiliary Pancreat Dis Int. 2004;3:469–472. [PubMed] [Google Scholar]


