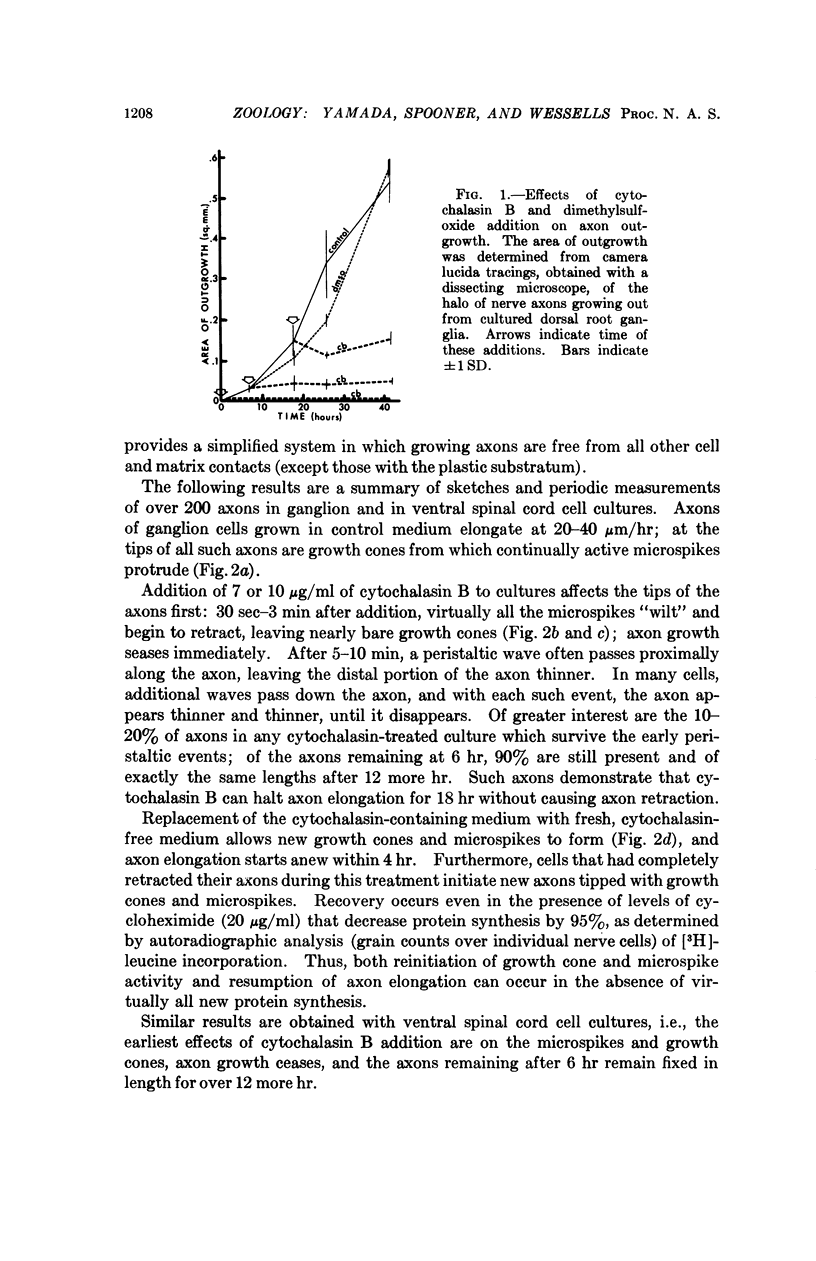
Abstract
The motile tips of elongating axons consist of growth cones from which microspikes protrude. Cytochalasin B causes retraction of microspikes, rounding-up of growth cones, and cessation of axon elongation. Drug withdrawal is followed by resumption of growth cone and microspike activity and of axon elongation. In contrast, colchicine causes shortening and retraction of axons, but it does not initially affect the tips. Growth cones and microspikes of elongating axons contain a network of 50 Å microfilaments, the pattern of which is altered by cytochalasin treatment. These experiments indicate that both structural integrity of the axon and continuing function of its motile tip are essential elements in axonal elongation.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Carter S. B. Effects of cytochalasins on mammalian cells. Nature. 1967 Jan 21;213(5073):261–264. doi: 10.1038/213261a0. [DOI] [PubMed] [Google Scholar]
- Cohen S. PURIFICATION OF A NERVE-GROWTH PROMOTING PROTEIN FROM THE MOUSE SALIVARY GLAND AND ITS NEURO-CYTOTOXIC ANTISERUM. Proc Natl Acad Sci U S A. 1960 Mar;46(3):302–311. doi: 10.1073/pnas.46.3.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GROBSTEIN C. Trans-filter induction of tubules in mouse metanephrogenic mesenchyme. Exp Cell Res. 1956 Apr;10(2):424–440. doi: 10.1016/0014-4827(56)90016-7. [DOI] [PubMed] [Google Scholar]
- HUGHES A. The growth of embryonic neurites; a study of cultures of chick neural tissues. J Anat. 1953 Apr;87(2):150–162. [PMC free article] [PubMed] [Google Scholar]
- Inoué S., Sato H. Cell motility by labile association of molecules. The nature of mitotic spindle fibers and their role in chromosome movement. J Gen Physiol. 1967 Jul;50(6 Suppl):259–292. [PMC free article] [PubMed] [Google Scholar]
- Kreutzberg G. W. Neuronal dynamics and axonal flow. IV. Blockage of intra-axonal enzyme transport by colchicine. Proc Natl Acad Sci U S A. 1969 Mar;62(3):722–728. doi: 10.1073/pnas.62.3.722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levi-Montalcini R., Cohen S. IN VITRO AND IN VIVO EFFECTS OF A NERVE GROWTH-STIMULATING AGENT ISOLATED FROM SNAKE VENOM. Proc Natl Acad Sci U S A. 1956 Sep;42(9):695–699. doi: 10.1073/pnas.42.9.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmiter R. D., Christensen A. K., Schimke R. T. Organization of polysomes from pre-existing ribosomes in chick oviduct by a secondary administration of either estradiol or progesterone. J Biol Chem. 1970 Feb 25;245(4):833–845. [PubMed] [Google Scholar]
- Peters A., Vaughn J. E. Microtubules and filaments in the axons and astrocytes of early postnatal rat optic nerves. J Cell Biol. 1967 Jan;32(1):113–119. doi: 10.1083/jcb.32.1.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott B. S., Engelbert V. E., Fisher K. C. Morphological and electrophysiological characteristics of dissociated chick embryonic spinal ganglion cells in culture. Exp Neurol. 1969 Feb;23(2):230–248. doi: 10.1016/0014-4886(69)90060-0. [DOI] [PubMed] [Google Scholar]
- Spooner B. S. The expression of differentiation by chick embryo thyroid in cell culture. I. Functional and fine structural stability in mass and clonal culture. J Cell Physiol. 1970 Feb;75(1):33–47. doi: 10.1002/jcp.1040750105. [DOI] [PubMed] [Google Scholar]
- Spooner B. S., Wessells N. K. Effects of cytochalasin B upon microfilaments involved in morphogenesis of salivary epithelium. Proc Natl Acad Sci U S A. 1970 Jun;66(2):360–361. doi: 10.1073/pnas.66.2.360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weisenberg R. C., Borisy G. G., Taylor E. W. The colchicine-binding protein of mammalian brain and its relation to microtubules. Biochemistry. 1968 Dec;7(12):4466–4479. doi: 10.1021/bi00852a043. [DOI] [PubMed] [Google Scholar]
- Wrenn J. T., Wessells N. K. An ultrastructural study of lens invagination in the mouse. J Exp Zool. 1969 Jul;171(3):359–367. doi: 10.1002/jez.1401710310. [DOI] [PubMed] [Google Scholar]
- Wrenn J. T., Wessells N. K. Cytochalasin B: effects upon microfilaments involved in morphogenesis of estrogen-induced glands of oviduct. Proc Natl Acad Sci U S A. 1970 Jul;66(3):904–908. doi: 10.1073/pnas.66.3.904. [DOI] [PMC free article] [PubMed] [Google Scholar]




