Abstract
We recently found that allogeneic intrabone marrow–bone marrow transplantation (IBM–BMT) plus adult thymus transplantation (ATT) from the same donor is effective in mice bearing solid tumors. In the current study, we examined the effects of this strategy on the survival of mice with leukemia. One week after intravenous injection of 1×106 leukemic cells (EL-4, H-2b) into 8-week-old B6 (H-2b) mice, the mice were 8 Gy irradiated and transplanted with 1×107 bone marrow cells (BMCs) from 8-week-old BALB/c mice (H-2d) by IBM–BMT with or without donor lymphocyte infusion (DLI) or ATT. All the mice without treatment died within 70 days after injection of EL-4. About 40% of those treated with IBM–BMT alone died within 100 days due to tumor relapse. In contrast, those treated with IBM–BMT+DLI or ATT showed the longest survival rate without relapse of leukemia. In addition, the former showed less graft versus host disease (GVHD) than the latter. The mice treated with IBM–BMT+ATT also showed an intermediate percentage of effector memory (EM) and central memory (CM) cells between those treated with BMT alone and those treated with IBM–BMT+DLI. The numbers and functions of T cells increased in those treated with IBM–BMT+ATT with interleukin-2 and interferon-γ production. These results suggest that IBM–BMT+ATT is effective in the treatment of leukemia with strong graft versus leukemia without increased risk of GVHD.
Introduction
Allogeneic bone marrow transplantation (allo-BMT) has been used for the radical treatment of leukemia. However, allo-BMT has some side effects. Graft versus host disease (GVHD) occurs if anti-host reaction in donor T cells is too strong, whereas relapse occurs if it is too weak [1]. In addition, a failure of bone marrow cell (BMC) engraftment in the early phase of transplantation may induce immunodeficiency, which, in turn, leads to severe infection [2]. Although donor lymphocyte infusion (DLI) is sometimes used to enhance engraftment and/or graft versus leukemia (GVL) activity [3], this is associated with an increased risk of GVHD [4]. Therefore, new cellular-based methods are required.
We recently developed a new BMT method, intrabone marrow–bone marrow transplantation (IBM–BMT), in which BMCs are directly injected into the bone marrow cavity [5]. IBM–BMT results in a reduced incidence of GVHD and greater engraftment of donor cells, including mesenchymal stem cells (MSCs) than the conventional intravenous method [6,7].
We have also developed a BMT method that is combined with thymus transplantation (TT), which includes the transplantation of adult thymus transplantation (ATT), newborn thymus, and fetal thymus. The combination of BMT+TT is effective in restoring donor-derived T cell function even in aged, chimeric-resistant, supralethally irradiated, and low-dose irradiated mice, mice with metabolic diseases, and also in mice injected with a small number of BMCs [8–12]. Further, we demonstrated that IBM–BMT+TT is effective for graft versus tumor (GVT) and long-term survival with a low risk of GVHD [13,14].
In the current study, we examined the BMT+ATT method in mice with leukemia. We also performed BMT alone and BMT+DLI in these mice and compared the survival rate, degree of GVHD, and T-cell functions.
Materials and Methods
Mice
Female 6- to 8-week-old C57BL/6 (B6) (H-2b) and BALB/c (H-2d) mice were obtained from Shimizu Laboratory Supplies (Shizuoka, Japan) and maintained until use in our animal facilities under specific pathogen-free conditions. All protocols for these animal experiments were performed in accordance with the Guidelines for Animal Experimentation, Kansai Medical University, and received approval from the Committee on Animal Experiments. EL-4 cells (H-2b) were derived from thymoma in B6 mice. Cells were maintained in RPMI 1640 medium supplemented with 10% fetal calf serum with antibiotics. These cells were intravenously transferred to the recipients (B6 mice).
IBM–BMT and ATT
Recipient B6 mice with tumors were irradiated (8 Gy) by using a 137Cs irradiator (Gammacell 40 Exactor; MDS Nordion International, Ottawa, ON, Canada) 7 days after transfer of the EL-4 cells. The next day, BMCs were flushed from the shafts of donor femora and tibiae, and single-cell suspensions were prepared. Next, 1×107 BMCs were directly injected into the bone marrow cavity of the recipient's tibia, as previously described for the IBM–BMT method [7]. Briefly, the knee was flexed to 90°, and the proximal side of the tibia was drawn to the anterior. A 26-gauge needle was inserted into the joint surface of the tibia through the patellar tendon and then inserted into the bone cavity. Simultaneously, one quarter of the AT was grafted under the renal capsule of the left kidney in some mice.
Histology
Histological studies were performed in the liver, intestine (for evaluation of GVHD), and engrafted tumors from the recipients 4 weeks after the BMT. The tissues were fixed in 10% formaldehyde and embedded in paraffin. Serial tissue sections (4 μm thick) were prepared and stained by using hematoxylin and eosin. The degree of GVHD was evaluated by using a semiquantitative scoring system for abnormalities known to be associated with GVHD, as previously described [13].
Analysis of surface marker antigens and intracellular FoxP3 and cytokines by flow cytometry
Surface markers on lymphocytes from the spleen were analyzed by 3-color fluorescence staining by using a FACScan system (Becton Dickinson, Franklin Lakes, NJ). Fluorescein isothiocyanate (FITC)-conjugated anti-H-2Kb (Pharmingen, San Diego, CA) mAbs and phycoerythrin (PE)-conjugated anti-H-2Kd mAbs were used to determine chimerism. FITC, PE, or biotin-conjugated CD4, CD8, B220, CD44, or CD62L (Pharmingen) were used to analyze spleen cell subsets. Avidin-Cy5 (Dako, Kyoto, Japan) was used as the third color in the avidin/biotin system. Intracytoplasmic FoxP3 staining was performed by using an eBioscience FITC-anti mouse/rat FoxP3 staining kit in accordance with the manufacturer's instructions (eBioscience, San Diego, CA). Intracellular cytokines [interleukin (IL)-2, IL-4, IL-10, IL-17, interferon (IFN)-γ, and tumor necrosis factor] were detected by using an Intracellular Cytokine Staining Kit in accordance with the manufacturer's instructions (Becton Dickinson).
Mitogen responses
To analyze lymphocyte function, mitogen responses were examined in chimeric mice 2 months after transplantation. For mitogen response, a total of 2×105 splenocytes collected from chimeric mice and untreated B6 and BALB/c mice as responders were plated in 96-well flat-bottomed plates (Corning Glass Works, Corning, NY) containing 200 μL of RPMI 1640 medium (Nissui Seiyaku, Tokyo, Japan) supplemented with 2 μL of glutamine (Wako Pure Chemicals, Tokyo, Japan), penicillin (100 U/mL), streptomycin (100 μg/mL), and 10% heat-inactivated fetal calf serum. For mitogen responses, responder cells were incubated with 2.5 μg/mL of Concanavalin A (ConA) (Calbiochem, San Diego, CA) or 25 μg/mL of lipopolysaccharide (LPS) (Difco Laboratories, Franklin Lakes, NJ) for 48 or 72 h. During the last 18 h of the culture period, 20 mL of 0.5 μCi 3H-thymidine (3H-TdR; New England Nuclear, Cambridge, MA) was introduced. Incorporation of 3H-TdR was measured by using Microbeta TriLux (PerkinElmer, Wellesley, MA). The stimulation index was calculated as the average of 3H-TdR incorporation in triplicate samples of responding cells with mitogen/3H-TdR incorporation of responding cells in medium alone.
Statistical analyses and nonparametric analyses (Mann–Whitney U-test and log rank-test) were performed by using StatView software (Abacus Concepts, Berkley, CA). In all analyses, P<0.05 was taken to indicate statistical significance.
Results
Survival rate and body weight
First, we examined the effects of BMT alone, BMT+ATT, or BMT+DLI on the survival rate of mice transplanted with EL-4 (Fig. 1A). All the untreated mice transplanted with EL-4 died within 70 days due to tumor growth (Fig. 2B). Those treated with BMT alone showed a survival rate of about 60% 6 months after BMT. The remaining 40% of the mice died due to tumor growth. Interestingly, those treated with BMT+ATT or BMT+DLI showed the longest survival rate. Next, we investigated the weight of these mice. The mice not treated with EL-4 showed a gradual increase in weight (Fig. 1B), which was due to growth of the tumor (Fig. 2B). Those treated with IBM–BMT alone and IBM–BMT+ATT surviving for a long time showed a stable weight, and those treated with IBM–BMT+DLI showed a gradual weight loss.
FIG. 1.
Survival rate and body weight in mice with leukemia treated with BMT+TT. Survival rate (A) and weight (B) of mice with advanced tumors are shown. *P< 0.0001 compared with those treated with BMT alone, BMT+ATT, or BMT+DLI. **P<0.05 compared with those treated with BMT+ATT or BMT+DLI. #P<0.05 compared with those treated with BMT alone, BMT+ATT, or BMT+DLI. ##P<0.05 compared with those treated with BMT+ATT or BMT+DLI. Data are shown as means±SE. Untreated controls (n=12), those treated with BMT alone (n=12), BMT+ATT (n=12), or BMT+DLI (n=15). ATT, adult thymus transplantation; BMT, bone marrow transplantation; DLI, donor lymphocyte infusion; TT, thymus transplantation.
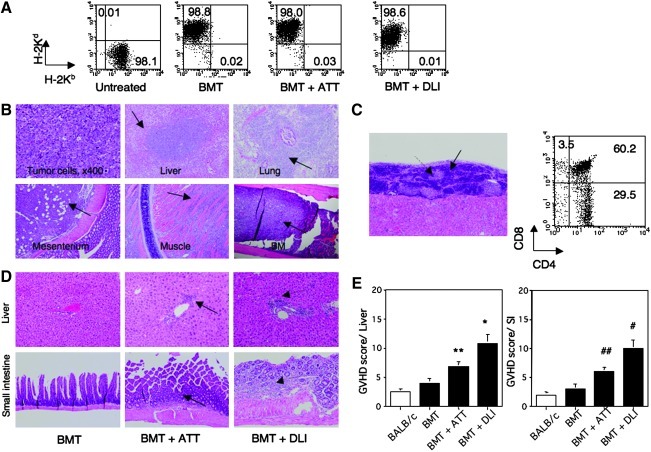
FIG. 2.
Chimerism, histology, transplanted thymus, and GVHD in mice with leukemia treated with BMT+TT. Chimerism in experimental mice (A), histology in the liver, lung, mesenterium, muscle, and BM from mice with leukemia (B), histology in transplanted thymus and the FACS profile in (C), histology for GVHD in chimeric mice (D) and the GVHD scores in the mice (E) are shown. Chimerism was analyzed in the spleens from mice transplanted with leukemia and those treated with BMT alone, BMT+ATT, or BMT+DLI (A). The numbers in the profiles show the percentage. Tumor cells (upper, left) infiltrated the liver, lung, mesenterium, muscle, and BM in the mice transplanted with leukemia (arrows) (B). Histological findings (left) and FACS profile (right) of thymocytes in the transplanted thymus from the mice treated with BMT and ATT (C). Plain arrow, cortex; dotted arrow, medulla. The numbers in the profiles show the percentage. Representative data from 4 experiments are shown. The histology of GVHD is shown in the liver (upper) and small intestine (lower) (D). Some lymphocytes infiltrated the liver and small intestine in BMT+ATT (arrows) and destroyed the tissue in BMT+DLI (dotted arrow). GVHD scores are shown in the liver (left) and small intestine (right) (E). The GVHD score was calculated as described in Materials and Methods. *P<0.05 compared with normal BALB/c mice and mice treated with BMT alone and BMT+ATT. **P<0.05 compared with normal BALB/c mice and the mice treated with BMT alone. #P<0.05 compared with normal BALB/c mice and mice treated with BMT alone and BMT+ATT. ##P<0.05 compared with normal BALB/c mice and mice treated with BMT alone. The mice transplanted with leukemia cells (EL-4) were analyzed 5 weeks after transplantation and those treated with BMT alone, BMT+ATT, or BMT+DLI were analyzed 8 weeks after treatment. Data are shown as means±SE. Normal BALB/c (n=4), BMT (n=4), BMT+ATT (n=4), BMT+DLI (n=4). GVHD, graft versus host disease.
Chimerism and histology
All mice treated with BMT showed donor-derived chimerism (H-2Kd), whereas untreated controls showed host-derived chimerism (H-2Kb) (Fig. 2A). The untreated mice showed massive infiltration of tumor cells throughout the whole body, including the liver, lung, mesenterium, muscle, and bone (Fig. 2B). All mice that died treated with BMT alone showed such tumor growth (as just mentioned). In contrast, most of those treated with BMT+ATT or BMT+DLI showed little tumor growth and long-term survival. The engrafted thymus showed a normal structure with cortical and medullar areas under the renal capsule (Fig. 2C). Normal T-cell differentiation was also observed in the thymus. In addition, those treated with BMT+ATT and BMT+DLI showed mild and moderate infiltrations of lymphocytes in the liver and small intestine, and the latter also showed some fibrosis with tissue destruction (Fig. 2D). Since the chimerism was of the donor type, this suggested the occurrence of GVHD. The GVHD scores are summarized in Fig. 2E. Those treated with BMT alone showed little GVHD, whereas those treated with BMT+ATT showed mild GVHD, and those treated with BMT+DLI showed moderate GVHD.
Lymphocyte subsets
We next analyzed the lymphocyte subsets in the spleen 4 weeks after transplantation. The number of CD4+ T cells was significantly greater in the mice treated with BMT+ATT compared with those treated with BMT alone and those with BMT+DLI, in which the levels were comparable to those in normal BALB/c mice (Fig. 3). This was followed by those treated with BMT alone, followed by those treated with BMT+DLI, which showed the lowest levels. The percentage of FoxP3+ regulatory T cells, which suppress immune responses, including GVH reactions [15,16], among CD4+ T cells was the highest in the mice treated with BMT alone, the percentage being comparable to that in BALB/c mice. This was followed by those treated with BMT+ATT, whereas those treated with BMT+DLI showed the lowest percentage. The results for CD8+ T cells were similar to those for CD4+ T cells, although all values for mice treated with BMT were lower than those of BALB/c mice. The number of B220+ T cells was lowest in those treated with BMT+DLI.
FIG. 3.
Numbers of lymphocytes in the spleen from leukemia-bearing mice treated with BMT+ATT. Numbers of CD4+ T cells (A), percentage of FoxP3+ cells in CD4+ T cells (B), numbers of CD8+ T cells (C), and B220+ B cells (D) in the spleen were evaluated in normal BALB/c mice, leukemia-bearing mice treated with BMT alone, BMT+ATT, or BMT+DLI. The experiments were performed 5 weeks after BMT. *P<0.05 compared with BMT alone and BMT+DLI. **P<0.01 compared with BMT+DLI. #P<0.05 compared with BMT+ATT and BMT+DLI. ##P<0.05 compared with BMT+DLI. §P<0.01 compared with BMT, BMT+ATT, and BMT+DLI. §§P<0.05 compared with BMT and BMT+DLI. §§§P<0.05 compared with BMT+DLI. ‡P<0.01 compared with BMT+DLI. Data are shown as means±SD. Normal BALB/c (n=5), BMT (n=5), BMT+ATT (n=5), and BMT+DLI (n=5).
Effector memory, central memory, and naïve T cell subsets
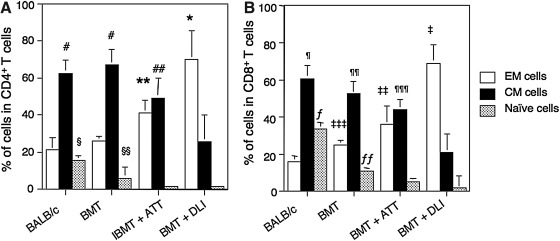
T cells can be functionally divided into CD62L-CD44- naïve, CD62L+CD44+ central memory (CM), and CD62L-CD44+ effector memory (EM) cells from prestimulation to terminal differentiation [17,18]. Therefore, we examined the proportions of these cells in both CD4+ and CD8+ subsets of T cells in the spleen. The percentage of EM among CD4+ T cells was the highest in the mice treated with BMT+DLI followed by those treated with BMT+ATT (Fig. 4A). The lowest percentage of EM among CD4+ T cells was seen in those treated with BMT alone, being comparable to that in BALB/c mice. Conversely, the percentage of CM was the highest in the mice treated with BMT alone and BALB/c mice, followed by those treated with BMT+ATT, and the lowest percentage of CM was seen in those treated with BMT+DLI. The percentage of naïve T cells was similar to that of CM cells, although the highest percentage was seen only in the BALB mice, and there were no significant differences between those treated with BMT+ATT and those treated with BMT+DLI. The results for CD8+ T cells were similar to those for CD4+ T cells (Fig. 4B).
FIG. 4.
Proportions of EM, CM, and naïve T cells from leukemia-bearing mice treated with BMT+ATT. Percentages of CD62L-CD44+ EM, CD62L+CD44+ CM, and CD62L-CD44- naïve cells in CD4+ (A) and CD8+ (B) T cell subsets were analyzed in the spleens from normal BALB/c mice and leukemia-bearing mice treated with BMT alone, BMT+ATT, and BMT+DLI. Analyses were performed at the same time as for the experiment in Fig. 3. *P<0.01 compared with BALB/c, BMT, and BMT+ATT. **P<0.01 compared with BALB/c and BMT. #P<0.05 compared with BMT+ATT and BMT+DLI. ##P< 0.01 compared with BMT+DLI. §P<0.01 compared with BMT, BMT+ATT, and BMT+DLI. §§P<0.01 compared with BMT+ATT and BMT+DLI. ‡P<0.01 compared with BALB/c, BMT, and BMT+ATT. ‡‡P<0.01 compared with BALB/c and BMT. ‡‡‡P<0.01 compared with BALB/c. ¶P<0.05 compared with BMT, BMT+ATT, and BMT+DLI. ¶¶P<0.05 compared with BMT+ATT and BMT+DLI. ¶¶¶P<0.01 compared with BMT+DLI. fP<0.01 compared with BMT, BMT+ATT, and BMT+DLI. ffP<0.05 compared with BMT+ATT and BMT+DLI. Data are shown as means±SD. Normal BALB/c (n=5), BMT (n=5), BMT+ATT (n=5), and BMT+DLI (n=5). CM, central memory; EM, effector memory.
Mitogen responses and cytokine production
Finally, we examined lymphocyte functions by monitoring mitogen responses (Con A for T cells and LPS for B cells) and cytokine production. The mice treated with BMT+ATT showed significantly increased Con A response compared with those treated with BMT alone, and the level was comparable to that in BALB/c mice (Fig. 5A). Those treated with BMT+DLI showed the lowest response. In contrast, the LPS response was almost the same in those treated with BMT alone, BMT+ATT, and BALB/c mice, and the lowest response was seen in BMT+DLI. With regard to cytokine production, those treated with BMT+ATT showed significant increases in IL-2 production compared with those treated with BMT alone and BMT+DLI, the level being comparable to that in BALB/c mice (Fig. 5B). In contrast, those treated with BMT+DLI showed significantly higher levels of IFN-γ production than those treated with BMT+ATT and BMT alone, although they did not reach the level of BALB/c mice. The production of IFN-γ was also elevated in those treated with BMT+TT, and was higher than in those treated with BMT alone.
FIG. 5.
Mitogen responses and percentages of cytokine-producing cells in the spleens from leukemia-bearing mice treated with BMT and ATT Mitogen responses: Con A and LPS (A) and percentages of cytokine-producing cells (B) in the spleen were evaluated in the spleens from normal BALB/c mice and leukemia-bearing mice treated with BMT alone, BMT+ATT, and BMT+DLI. Analyses were performed at the same time as for the experiment in Fig. 3. *P<0.05 compared with BMT and BMT+DLI. **P<0.05 compared with BMT+DLI. #P<0.05 compared with BMT+DLI. §P<0.05 compared with BMT and BMT+DLI. ‡P<0.05 compared with BMT, BMT+ATT, and BMT+DLI. ‡‡P<0.05 compared with BMT and BMT+ATT. ‡‡‡P<0.05 compared with BMT. Data are shown as means±SD. Normal BALB/c (n=4), BMT (n=4), BMT+ATT (n=4), and BMT+DLI (n=4). LPS, lipopolysaccharide.
Discussion
The current study was performed to examine the effects of BMT+ATT on leukemia in mice. The mice treated with BMT+ATT showed a longer survival than those treated with BMT alone, and milder GVHD than those treated with BMT+DLI. Leukemia showed little growth in BMT+ATT mice comparable to those treated with BMT+DLI. Those treated with BMT+ATT showed higher numbers of both CD4+ and CD8+ T cell subsets than those treated with BMT alone or with BMT+DLI. Interestingly, the percentages of FoxP3+ regulatory T cells, CM, and EM T cells in those treated with BMT+ATT were intermediate between those treated with BMT alone and those treated with BMT+DLI. T-cell functions with production levels of some cytokines were also elevated in those treated with BMT+ATT. These findings suggest that the BMT+ATT method is more effective in the treatment of leukemia than previous methods.
First, we examined the survival rates in association with GVH and GVL effects. All mice with the development of leukemia died early, whereas those without leukemia showed long-term survival, with or without GVHD, thus indicating that the presence of leukemia is the factor with the greatest influence on mortality. However, we did not examine the further long-term effects of GVHD, and chronic GVHD may also lead to death in the long term [19]. Therefore, these observations suggest that BMT+ATT is superior to BMT alone and BMT+DLI.
We next investigated the mechanism of these effects. The numbers of both T-cell subsets significantly increased in mice treated with BMT+ATT compared with those treated with BMT alone and BMT+DLI. The low numbers of T-cell subsets as well as B cells in those treated with BMT+DLI may have resulted in GVHD [20]. Interestingly, those treated with BMT+ATT showed a lower percentage of regulatory T cells than those treated with BMT alone, whereas they showed a higher percentage of regulatory T cells than BMT+DLI. Since regulatory T cells suppressed GVHD, preserving GVT and GVL [21,22] the intermediate percentage of these cells also played an important role in the mechanism underlying our results. The elevation of regulatory T-cell numbers compared with BMT alone suggested that some regulatory T cells in BMT+ATT may be produced from the TT [13].
The percentages of EM T cells were highest in the mice treated with BMT+DLI, followed by those treated with BMT+ATT, and lowest in those treated with BMT alone. In contrast, CM and naïve T cells were lowest in the BMT+DLI group, followed by the BMT+ATT group, and highest in those treated with BMT alone. Since freshly isolated CM and naïve T cells in DLI induce GVHD and GVL [23,24], the observation of the lowest numbers of these cells in BMT+DLI suggested that these cells in DLI had differentiated into EM T cells with activation and/or were consumed [25].
The mice treated with BMT+ATT showed sufficient mitogen responses to both T and B cells, whereas those treated with BMT alone showed low T-cell response, and those treated with BMT+DLI showed low responses to both T and B cells. Such immunodeficiency in BMT+DLI may be induced by GVHD [20]. These findings indicated that BMT+TT is the best method compared with BMT alone or BMT+DLI. Although the mechanism underlying the long-term survival in the BMT+DLI group is unclear, the SPF condition may have prevented severe infection. Alternatively, the IBM–BMT method itself may suppress lethal GVHD by DLI [6].
Analysis of cytokines indicated significantly elevated IL-2 production in the mice treated with BMT+ATT compared with those treated with BMT alone or with BMT+DLI, whereas IFN-γ production was significantly higher in those treated with BMT+DLI. Although several cytokines play a role in GVHD, IL-2 is effective in inducing strong GVL while avoiding GVHD [26]. In addition, the highest degree of elevation of IFN-γ production may contribute to GVHD with lymphoid hypoplasia (immunodeficiency) in BMT+DLI [27]. Therefore, the cytokine patterns may also be at least partly associated with the pathogenesis.
We thus found that donor-derived T cells play an important role in the treatment of leukemia. However, further gene analyses are needed for a more comprehensive understanding. For example, cytokine profiles at the transcriptional level should be analyzed by using DNA microarry in T cells [28]. In addition, differences in T-cell clones may become evident between GVL and GVH, or between host and donor-thymus derived by next-generation sequencing and/or other methods [29]. These findings would help detail the mechanism of BMT+TT.
We examined means of treating several intractable diseases and/or serious complications by BMT+TT [8–14]. In most studies, BMT+TT showed better results than BMT alone or BMT+DLI. The results of the current study were also compatible with these previous findings. The thymus is an organ involved in the maintenance of homeostasis itself and regulates the production of not only T cells but also several cytokines and hormones in a feedback mechanism [30]. Thus, TT is different from DLI, which supplies mature lymphocytes in one direction. Hence, TT is a type of functional organ transplantation, and may represent an approach that significantly regulates the immune function of T cells in vivo for the benefit of the host.
Overall, we found that allogeneic IBM–BMT+ATT induces strong GVL effects with mild GVHD. Although it may be both ethically and technically difficult to obtain adequate thymus tissue in clinical cases (including the question of donor age), grafts could be obtained from patients with congenital heart diseases or from aborted fetuses, as previously utilized [31,32]. We have recently found that even if the thymus donor is different from the BMC donor, then the effect is comparable to that seen with transplantation from the same donor using triple chimeric mice [33]. In addition, a method of regenerating the thymus has also been developed [34–37]. Therefore, IBM–BMT+ATT could become a viable strategy for the treatment of malignant hematological tumors in humans.
Acknowledgments
This work was supported by a grant from Haiteku Research Center of the Ministry of Education; a grant from the Millennium Program of the Ministry of Education, Culture, Sports, Science, and Technology; a grant from the Science Frontier Program of the Ministry of Education, Culture, Sports, Science, and Technology; a grant from The 21st Century Center of Excellence (COE) Program of the Ministry of Education, Culture, Sports, Science, and Technology; a Research Grant B from Kansai Medical University, Health and Labor Sciences (Research on Human Genome, Tissue Engineering Food Biotechnology); a grant from the Department of Transplantation for Regeneration Therapy (sponsored by Otsuka Pharmaceutical Co., Ltd.); a grant from the Molecular Medical Science Institute (Otsuka Pharmaceutical Co., Ltd.); and a grant from Japan Immunoresearch Laboratories Co., Ltd. (JIMRO). The project was supported by Guangdong Natural Science Foundation. The authors wish to thank Ms. Y. Tokuyama and Ms. A. Kitajima for their technical assistance, and Mr. Hilary Eastwick-Field and Ms. K. Ando for their help in the preparation of this article.
Author Disclosure Statement
The authors disclose no commercial association that might create a conflict in connection with the submitted article. No competing financial interests exist.
References
- 1.Chao NJ. Graft-versus-host disease. The view point from the donor T cell. Biol Blood Marrow Transplant. 1997;3:1–10. [PubMed] [Google Scholar]
- 2.Watson JG. Problems of infection after bone marrow transplantation. J Clin Pathol. 1983;36:683–692. doi: 10.1136/jcp.36.6.683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Russell NH. Byrne JL. Faulkner RD. Gilyead M. Das-Gupta EP. Haynes AP. Donor lymphocyte infusions can result in sustained remissions in patients with residual or relapsed lymphoid malignancy following allogeneic haemopoietic stem cell transplantation. Bone Marrow Transplant. 2005;36:437–441. doi: 10.1038/sj.bmt.1705074. [DOI] [PubMed] [Google Scholar]
- 4.Deol A. Lum LG. Role of donor lymphocyte infusions in relapsed hematological malignancies after stem cell transplantation revisited. Cancer Treat Rev. 2010;36:528–538. doi: 10.1016/j.ctrv.2010.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kushida T. Inaba M. Hisha H. Ichioka N. Esumi T. Ogawa R. Iida H. Ikehara S. Intra-bone marrow injection of allogeneic bone marrow cells: a powerful new strategy for treatment of intractable autoimmune diseases in MRL/lpr mice. Blood. 2001;97:3292–3299. doi: 10.1182/blood.v97.10.3292. [DOI] [PubMed] [Google Scholar]
- 6.Ikehara S. A novel method of bone marrow transplantation (BMT) for intractable autoimmune diseases. J Autoimmun. 2008;30:108–113. doi: 10.1016/j.jaut.2007.12.011. [DOI] [PubMed] [Google Scholar]
- 7.Nakamura K. Inaba M. Sugiura K. Yoshimura T. Kwon AH. Kamiyama Y. Ikehara S. Enhancement of allogeneic hematopoietic stem cell engraftment and prevention of graft-versus-host diseases (GvHD) by intra-bone marrow-bone marrow transplantation plus donor lymphocyte infusion. Stem Cells. 2004;22:125–134. doi: 10.1634/stemcells.22-2-125. [DOI] [PubMed] [Google Scholar]
- 8.Hosaka N. Nose M. Kyogoku M. Nagata N. Miyashima S. Good RA. Ikehara S. Thymus transplantation, a critical factor for correction of autoimmune disease in aging MRL/+mice. Proc Natl Acad Sci U S A. 1996;93:8558–8562. doi: 10.1073/pnas.93.16.8558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hosaka N. Ryu T. Miyake T. Cui W. Nishida T. Takaki T. Inaba M. Ikehara S. Treatment of autoimmune diseases in MRL/lpr mice by allogeneic bone marrow transplantation plus adult thymus transplantation. Clin Exp Immunol. 2007;147:555–663. doi: 10.1111/j.1365-2249.2006.03310.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ryu T. Hosaka N. Miyake T. Cui W. Nishida T. Takaki T. Li M. Kawamoto K. Ikehara S. Transplantation of newborn thymus plus hematopoietic stem cells can rescue supralethal irradiated mice. Bone Marrow Transplant. 2008;41:659–666. doi: 10.1038/sj.bmt.1705957. [DOI] [PubMed] [Google Scholar]
- 11.Li M. Abraham NG. Vanella L. Zhang Y. Inaba M. Hosaka N. Hoshino S. Shi M. Ambrosini YM. Gershwin ME. Ikehara S. Successful modulation of mice with intra-bone marrow—bone marrow transplantation plus concurrent thymic transplantation. J Autoimmun. 2010;35:414–423. doi: 10.1016/j.jaut.2010.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nishida T. Hosaka N. Takaki T. Miyake T. Cui W. Inaba M. Kinoshita H. Matsuda T. Ikehara S. Allogeneic intra-BM-BMT plus adult thymus transplantation from same donor has benefits for long-term survival even after sublethal irradiation or low-dose BM cell injection. Bone Marrow Transplant. 2009;43:829–837. doi: 10.1038/bmt.2008.396. [DOI] [PubMed] [Google Scholar]
- 13.Miyake T. Hosaka N. Cui W. Nishida T. Takaki T. Inaba M. Kamiyama Y. Ikehara S. Adult thymus transplantation with allogeneic intra-bone marrow-bone marrow transplantation from same donor induces high thymopoiesis, mild graft-versus-host reaction and strong graft-versus-tumour effects. Immunology. 2009;126:552–564. doi: 10.1111/j.1365-2567.2008.02920.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hosaka N. Cui W. Zhang Y. Takaki T. Inaba M. Ikehara S. Prolonged survival in mice with advanced tumors treated with syngeneic or allogeneic intra-bone marrow-bone marrow transplantation plus fetal thymus transplantation. Cancer Immunol Immunother. 2010;59:1121–1130. doi: 10.1007/s00262-010-0840-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sakaguchi S. Ono M. Setoguchi R. Setoguchi R. Yagi H. Hori S. Fehervari Z. Shimizu J. Takahashi T. Nomura T. Foxp3+ CD25+ CD4+ natural regulatory T cells in dominant self-tolerance and autoimmune disease. Immunol Rev. 2006;212:8–27. doi: 10.1111/j.0105-2896.2006.00427.x. [DOI] [PubMed] [Google Scholar]
- 16.Hoffmann P. Ermann J. Edinger M. Fathman CG. Strober S. Donor-type CD4(+)CD25(+) regulatory T cells suppress lethal acute graft-versus-host disease after allogeneic bone marrow transplantation. J Exp Med. 2002;196:389–399. doi: 10.1084/jem.20020399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sallusto F. Lenig D. Förster R. Lipp M. Lanzavecchia A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature. 1999;401:708–712. doi: 10.1038/44385. [DOI] [PubMed] [Google Scholar]
- 18.Blander JM. Sant'Angelo DB. Metz D. Kim SW. Flavell RA. Bottomly K. Janeway CA., Jr A pool of central memory-like CD4 T cells contains effector memory precursors. J Immunol. 2003;17:2940–2948. doi: 10.4049/jimmunol.170.6.2940. [DOI] [PubMed] [Google Scholar]
- 19.Wingard JR. Piantadosi S. Vogelsang GB. Farmer ER. Jabs DA. Levin LS. Beschorner WE. Cahill RA. Miller DF. Harrison D. Predictors of death from chronic graft-versus-host disease after bone marrow transplantation. Blood. 1989;74:1428–1435. [PubMed] [Google Scholar]
- 20.Graze PR. Gale RP. Chronic graft versus host disease: a syndrome of disordered immunity. Am J Med. 1979;66:611–620. doi: 10.1016/0002-9343(79)91171-9. [DOI] [PubMed] [Google Scholar]
- 21.Edinger M. Hoffmann P. Ermann J. Drago K. Fathman CG. Strober S. Negrin RS. CD4+CD25+ regulatory T cells preserve graft-versus-tumor activity while inhibiting graft-versus-host disease after bone marrow transplantation. Nat Med. 2003;9:1144–1150. doi: 10.1038/nm915. [DOI] [PubMed] [Google Scholar]
- 22.Trenado A. Charlotte F. Fisson S. Yagello M. Klatzmann D. Salomon BL. Cohen JL. Recipient-type specific CD4+CD25+ regulatory T cells favor immune reconstitution and control graft-versus-host disease while maintaining graft-versus-leukemia. J Clin Invest. 2003;112:1688–1696. doi: 10.1172/JCI17702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dutt S. Baker J. Kohrt HE. Kambham N. Sanyal M. Negrin RS. Strober S. CD8+ CD44hi but not CD4+ CD44hi memory T cells mediate potent graft anti-lymphoma activity without GVHD. Blood. 2011;117:3230–3239. doi: 10.1182/blood-2010-10-312751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zheng H. Matte-Martone C. Jain D. McNiff J. Shlomchik WD. Central memory CD8+ T cells induce graft-versus-host disease and mediate graft-versus-leukemia. J Immunol. 2009;182:5938–5948. doi: 10.4049/jimmunol.0802212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhang Y. Joe G. Hexner E. Zhu J. Emerson SG. Alloreactive memory T cells are responsible for the persistence of graft-versus-host disease. J Immunol. 2005;174:3051–3058. doi: 10.4049/jimmunol.174.5.3051. [DOI] [PubMed] [Google Scholar]
- 26.Weiss L. Lubin I. Factorowich I. Lapidot Z. Reich S. Reisner Y. Slavin S. Effective graft-versus-leukemia effects independent of graft-versus-host disease after T cell-depleted allogeneic bone marrow transplantation in a murine model of B cell leukemia/lymphoma. Role of cell therapy and recombinant IL-2. J Immunol. 1994;153:2562–2567. [PubMed] [Google Scholar]
- 27.Klimpel GR. Annable CR. Cleveland MG. Jerrells TR. Patterson JC. Immunosuppression and lymphoid hypoplasia associated with chronic graft versus host disease is dependent upon IFN-gamma production. J Immunol. 1990;144:84–93. [PubMed] [Google Scholar]
- 28.Tosolini M. Kirilovsky A. Mlecnik B. Fredriksen T. Mauger S. Bindea G. Berger A. Bruneval P. Fridman WH. Pagès F. Galon J. Clinical impact of different classes of infiltrating T cytotoxic and helper cells (Th1, th2, treg, th17) in patients with colorectal cancer. Cancer Res. 2011;71:1263–1271. doi: 10.1158/0008-5472.CAN-10-2907. [DOI] [PubMed] [Google Scholar]
- 29.Baum PD. Young JJ. McCune JM. Measurement of absolute T cell receptor rearrangement diversity. J Immunol Methods. 2011;368:45–53. doi: 10.1016/j.jim.2011.03.001. [DOI] [PubMed] [Google Scholar]
- 30.Mehr R. Perelson AS. Fridkis-Hareli M. Globerson A. Regulatory feedback pathways in the thymus. Immunol Today. 1997;18:581–585. doi: 10.1016/s0167-5699(97)01150-x. [DOI] [PubMed] [Google Scholar]
- 31.Markert ML. Boeck A. Hale LP. Kloster AL. McLaughlin TM. Transplantation of thymus tissue in complete DiGeorge syndrome. N Engl J Med. 1999;341:1180–1189. doi: 10.1056/NEJM199910143411603. [DOI] [PubMed] [Google Scholar]
- 32.Markert ML. Hicks CB. Bartlett JA. Harmon JL. Hale Effect of highly active antiretroviral therapy and thymic transplantation on immunoreconstitution in HIV infection. AIDS Res Hum Retroviruses. 2000;16:403–413. doi: 10.1089/088922200309061. [DOI] [PubMed] [Google Scholar]
- 33.Cui W. Hosaka N. Miyake T. Wang X. Guo K. Cui Y. Li Q. Song C. Feng W et al. Analysis of tolerance induction using triple chimeric mice: MHC-disparate thymus, hemopoietic cells and microenvironment. Transplantation. 2008;85:1151–1158. doi: 10.1097/TP.0b013e31816a8f1f. [DOI] [PubMed] [Google Scholar]
- 34.Zhang L. Sun L. Zhao Y. Thymic epithelial progenitor cells and thymus regeneration: an update. Cell Res. 2007;17:50–55. doi: 10.1038/sj.cr.7310114. [DOI] [PubMed] [Google Scholar]
- 35.Lai L. Jin J. Generation of thymic epithelial cell progenitors by mouse embryonic stem cells. Stem Cells. 2009;27:3012–3020. doi: 10.1002/stem.238. [DOI] [PubMed] [Google Scholar]
- 36.Takaki T. Hosaka N. Miyake T. Cui W. Nishida T. Inaba M. Ikehara S. Presence of donor-derived thymic epithelial cells in [B6—>MRL/lpr] mice after allogeneic intra-bone marrow-bone marrow transplantation (IBM-BMT) J Autoimmun. 2008;31:408–415. doi: 10.1016/j.jaut.2008.09.003. [DOI] [PubMed] [Google Scholar]
- 37.Rossi SW. Jenkinson WE. Anderson G. Jenkinson EJ. Clonal analysis reveals a common progenitor for thymic cortical and medullary epithelium. Nature. 2006;441:988–991. doi: 10.1038/nature04813. [DOI] [PubMed] [Google Scholar]