Abstract
In an era of disease-modifying treatments, the non-fluent/agrammatic variant of primary progressive aphasia (naPPA) may help screen for a specific cause of neurodegenerative disease. However, there are controversies surrounding the identification of naPPA. This review describes the characteristic features associated with this discrete, young-onset neurodegenerative condition. Patients with naPPA have a distinct limitation in language emphasizingtheir poor grammatical comprehension and expression, as well as a disorder of speech sound production. Imaging studies associate an impairment of this uniquely human language capacity with disruption of a large-scale neural network centered in left inferior frontal and anterior-superior temporal regions. This corresponds to thepathologic burden of disease anatomically focused in left inferior frontal and anterior-superior temporal regions. A review of the histopathology underlying naPPA relates this condition to frontotemporal lobar degeneration spectrum pathology involving the microtubule-associated protein tau in a majority of cases. While much work remains to be done, these observations point to unique clinical-pathological correlations that can advance care for an important class of diseases while supplementing our knowledge of human cognitive neuroscience.
INTRODUCTION
The central feature of primary progressive aphasia (PPA) is declining language. Pick first described a woman withgradually worsening speech who eventually became mute in the context of a progressive social disorder characterized by disinhibited, socially inappropriate behavior1. The first report of progressive difficulty limited to language was provided one year later2. This case had declining speech fluency without difficulty in memory, social, or visuospatial domains. In the modern literature, Mesulam described five cases of declining speech fluency under the name of Slowly Progressive Aphasia3.
PPA, first defined by Mesulam4, refers to an aphasic disorder, that is, an impairment of language comprehension and expression without peripheral sensory and motor deficits that may mimic aphasia. Moreover, the language impairment must be insidiously progressive in nature. This is in order to rule out non-neurodegenerative etiologies such as stroke or head trauma. Finally, the language disorder must be the primary deficit that is present for about two years.This eliminates other progressive neurodegenerative conditions associated with a variety of underlying pathologies, including:behavioral variant frontotemporal degeneration (bvFTD) characterized a social disorder;typicalAlzheimer’s disease (AD) associated with memory difficulty; posterior cortical atrophy that often has visuospatial deficits;corticobasal syndrome (CBS), progressive supranuclear palsy syndrome (PSPS), and parkinsonian syndromes like Lewy body disease that are associated with cognitive deficits and an extrapyramidal movement disorder; and amyotrophic lateral sclerosis (ALS) that can have cognitive difficulties associated with weakness.
PPA represents a spectrum of selective language disorders5. These are important to recognize because each syndrome potentially screens for a distinct underlying pathology. Following clinical screening, more definitive diagnosis may be supplemented by biofluid biomarkers6. In this review, we focus on one form of PPA known as the non-fluent/agrammatic variant (naPPA) or “progressive non-fluent aphasia.” This variant of PPA has proven controversial because of difficulty defining the characteristic language deficits, heterogeneity in the anatomic distribution of disease, and thevariety ofunderlying pathologies. This review is timely because of recently published clinical consensus criteria for naPPA7, important advances in understanding the spectrum of pathology contributing to PPA8, and the emergence of disease-modifying medication trials focused on tau. The typicalclinical characteristics of naPPA,the anatomic distribution of disease defined by imaging studies, the genetic associations of this condition, and the common histopathologic underpinnings of naPPA are described below.
There are two other common variants of PPA. One is semantic variant PPA (svPPA) or “semantic dementia”9. This features a disorder of word and object meaning. The second is logopenic variant PPA (lvPPA) or “logopenic progressive aphasia”10. This ismarked by impaired word-finding and repetition difficulty. Recently published clinical recommendations for identifying these common forms of PPA7 are summarized in Table 1.
TABLE 1.
CLINICAL CHARACTERISTICS OF PRIMARY PROGRESSIVE APHASIA
| Variant | Clinical Features1 | Cortical Atrophy |
Pathology2 | Alternative Nomenclature |
|---|---|---|---|---|
| naPPA |
|
Left inferior frontal and insula |
|
|
| svPPA |
|
Anterior and ventral temporal lobe |
|
|
| lvPPA |
|
Left posterior- superior temporal and inferior parietal |
|
|
Based on expert consensus 7.
From a literature survey of confirmed pathology in patients with PPA recruited without a priori bias 5.
Abbreviations: AD, Alzheimer’s disease; FTLD-tau, frontotemporal lobar degeneration with tau-positive pathology; FTLD-TDP, frontotemporal lobar degeneration with ubiquitin- and TDP-43-positive pathology.
Search Strategy and Selection Criteria
A search in pubmed and medline for English language publications for the years 1981-2012 using the key words “progressive non-fluent aphasia” and “primary progressive aphasia” was supplemented by reviews of relevant bibliographies to select papers representing a balanced view of perspectives on this condition. Because of the recent, narrowed definition of naPPA, some earlier work referring to “progressive non-fluent aphasia” should be interpreted cautiously.
CLINICAL CHARACTERISTICS OF NONFLUENT/AGRAMMATIC PRIMARY PROGRESSIVE APHASIA
Demographic Features of naPPA
We can estimate the frequency of naPPA by considering that the etiology is often in the spectrum of pathology related to frontotemporal lobar degeneration (FTLD). Estimates of the prevalence of FTLD are 2.7-15 per 100,00011-13, while estimates of the incidence are 2.2-3.5 per 100,00014, 15. In autopsy studies examining unselected clinical cases thought to be associated with FTLD spectrum pathology, up to 45% of FTLD cases have PPA, and a little less than half of these PPA patients have naPPA5, 16-23. Based on an estimate that 20% of autopsied FTLD cases have naPPA, the prevalence naPPA due to FTLD pathology is 0.5-3.0 per 100,000 and incidence is 0.4-0.7 per 100,000. If up to 30% of naPPA cases are due to AD, then the approximate prevalence of naPPA due to any cause is 0.65-3.9 per 100,000 and incidence is 0.5-0.9 per 100,000.
naPPA is a young-onset condition with an average age of onset at about 6024. The age of onset is quite broad,ranging from the 30’s to the early 80’s, and this differs from conditions like AD because of its poisson distribution around the average onset age rather than a skewed distribution that increases with age. Survival is about 7 years, although this too is quite variable17, 25-29. There is no gender bias. There are no known environmental risk factors30. We are highly dependent on language in day-to-day functioning, and a language disorder limits self-care activities and independence in daily living. There is a significant reduction in quality of life. Poor communicative efficacy in naPPA has profound consequences for psychological integrity and can be associated with depression31.
Speech and Language Deficits in naPPA
The clinical hallmark ofnaPPA is effortful, non-fluent speech32, 33. This is best quantified by a semi-structured protocol that is long enough to reflectthe variety of utterances that can occur in spontaneous speech, yet is standardized enough so that all participants have an opportunity to produce speech prompted by the same content. Our lab asks patients to review and then narrate a wordless children’s picture story34. Based on this technique, therate of speech in naPPAaverages45 words per minute,less than one-third the speech rate of healthy adults35. This is a robust marker of naPPA, andhas been confirmed using a briefer speech sample36. Speech does not emerge as a slow but steady flow. Instead, speech is interrupted by lengthy pauses within and between utterances.Even when controlling for pauses, naPPA patients produce fewer words per minute than controls35. Another clinical characteristic of effortful, non-fluent speech is the distortion of prosody. Prosody is the pattern of pitch contours spanning words and sentences that helps provide emphasis, is critical for marking questions, and reflects the emotional content of speech. Prosody loses its normal contours in naPPA.
Several factors may contribute to effortful, non-fluent speech in naPPA37. The most prominent factoris difficulty processing grammatical aspects of speech. A transcription of a speech sample from a naPPA patient is provided in Figure 1. A detailed analysis of a lengthy, semi-structured speech sample in naPPA reveals significant simplification of grammatical forms. There is also a significantly greater number of grammatical errors and omissions compared to controls and other PPA patients35-37. These characteristics results ina significantly abbreviatedmean length of utterance. Verbs play a critical role in structuring a sentence, and naPPA patients use fewer verbs in their speech35. They also have difficulty with verbs on other tasks that assess comprehension38 and naming39.
FIGURE 1. SEMISTRUCTURED SPEECH SAMPLE OF naPPA.
1. Speech sample of a patient with naPPA narrating the children’s wordless picture story Frog, Where Are You?34. The narrative accompanying each of the three pictures is below the corresponding picture. While description of the content is accurate, the grammatical structure of the utterances is distorted and simplified, containing many errors. There are also speech sound errors. Pauses longer than 2 seconds are noted in parentheses in the transcript.
Another potential cause of slowed, effortful speech may be a motor speech disorder known as apraxia of speech (AoS). From this perspective, speech is slowed because the complex coordination of muscle groups underlying the motor speech apparatus has been compromised. There are at least two sourcesfor the large number of speech sound errors in patients with naPPA: While some of these errors are due to a disturbance of the motor system responsible for coordinating and articulating speech sounds known as AoS, other speech sound errors are due to disturbance of the linguistic system of phonology that is responsible for the abstract representations of speech sounds and the rules governing their use in a speaker’s language. Several reports describe an increased frequency of AoS in patients with naPPA40-42. AoS is most prominent in conditions with co-occurring involuntary limb movements and poor limb motor control such as progressive supranuclear palsy syndrome (PSPS) and corticobasal syndrome (CBS), although AoS can occur without an accompanying extrapyramidal disorder, and AoS can occur as an isolated entity without other evidence of the language impairments found in naPPA43, 44. An important problem has been difficulty quantifying AoS. Ash and her colleagues (2010) proposed on epotential method for quantifying AoS. Specifically, they identified speech errors that are not part of the corpus of speech sounds in the speaker’s native language.This is because“phonetic” speech errors of this sort are more likely to emerge when an impaired motor coordination system produces sounds due to misplaced articulators. This contrasts with“phonologic” speech sound errors that violate the abstract rules for representing and ordering phonemes in the speaker’s native language. While this method rigorously characterizes the nature of the speech errors, this captures only some examples of AoS. With this caveat in mind, these investigators document many speech errors in naPPA, but phonetic errors consistent with AoS characterize only 21% of the speech sound errors of these patients45.
One strategy to assess grammatical processing while minimizing confounds associated with a motor speech disorderinvolves assessment of grammatical comprehension32. naPPA patients thus have difficulty pointing to one of two pictures based on a sentence, where selecting the correct picture depends on appreciating the sentence’s grammatical structure46. Another measure uses an anagram task to determine whether patients can order words printed on cards into a grammatically complex utterance describing a picture47. Yet another task is entirely language-based and probes brief sentences varying in grammatical complexity with a simple question about “who did what to whom.” In the sentence “Boys that girls kick are unfriendly,” for example, naPPApatients oftenerr when asked: “Who did the kicking?”48. Grammatical difficulties such as this can be used to distinguish naPPA from healthy seniors and from other PPA variants48, 49. Another mark of the “central” grammatical deficitin naPPA is the presence of parallel grammatical impairments in writing and reading32.
It is important to keep in mind that these measures of grammatical processing are performed “off-line” and may not fully reflect day-to-day natural language use. Thus, these tasks areunder the control of an executive resource system that is making conscious, deliberative interpretations and decisions about sentence meaning, and as noted below, naPPA patients have limited executive resources and working memorythat can confound interpretation of performance onthese language tasks. A handful of studies have probed grammatical processing in an “on-line” manner that minimizes executive control during task performance. One study examinednaPPA patients whowere impaired in their comprehension of grammatically complex sentences using a traditional measure. The investigators reported that naPPA patients also have slowed processing of grammatically complex sentences in an on-line measure, suggestingthat grammatically relevant information may degrade in working memory during the course of sentence processing50. Another on-line study found that naPPA patients are selectively insensitive to grammatical violations in a sentence,although they are normal in their sensitivity to semantic violations51.
Several aspects of language are relatively preserved in naPPA. Oral production of over-learned sequences such as counting and repetition of phrasesismore fluent than spontaneous speech. The ability to name orally and write a pictured object’s nameto confrontation is performed relatively well,although there may be difficulty naming with verbs39. There is relatively good comprehension of single words presented orally and in writing, although there may be difficulty with verb comprehension38. Spelling is largely preserved and there is little difficulty reading written words aloud.
naPPA is a progressive disorder of language, but there have been few studies examining the longitudinal course of this condition. These patients appear to develop broad-based decline in their language functioning52, and a validated algorithm has been developed to quantify disease severity in naPPA53. Two studies have documented progressive decline in grammatical comprehension in naPPA54, 55, and their speech becomes progressively effortful and non-fluent to the point where speech consists of single words. While performance worsensin other domains of language such as naming, reading and spelling43, 56, and while these patients deteriorate in their performance on measures of working memory and executive control56, effortful speech, grammatical deficits, and speech sound errors remain relatively more impaired throughout the course of the disease.
In sum, patients with naPPA appear to have profoundly slowed and effortful speech that is often related to an impairment of grammatical comprehension and production as well as poor motor speech control, but many other aspects of language do not appear to decline as much.
Other Cognitive Deficits in naPPA
Patients with naPPAmay develop other neuropsychological deficits. For example, a limitation of executive resources often can be found. This includes difficulty with working memory, mental planning and dual-tasking57. Thus, impairments can be seen on measures such as reverse digit span, where a sequence of digits is repeated in its reverse order. There is also difficulty on letter-guided naming fluency (e.g. providing as many words as possible beginning with a letter like “F”).Patients decline in their performance on these measures over time as well56.
In contrast to performance on measures of executive functioning, patients with naPPAtypically have relatively preserved episodic memory57. Visuospatial functioning also is relatively preserved, although there are some exceptions such as naPPA presenting in the context of CBS58. It is uncommon to observe a disorder of social functioning and personality early in the course ofnaPPA,although socially inappropriate behaviors seen in patients with behavioral variant FTD (bvFTD) can emerge over time, includingapathy, disinhibition or repetitive behaviors withlittle empathy and poor self-insight59, 60. The non-language spectrum of cognitive deficits in naPPA thus appears to involve limitations in working memory and executive control, but may implicate other cognitive and social domains particularly as the condition progresses.
Elementary Neurologic Deficits in naPPA
Many patients with naPPA have a normal elementary neurologic exam, but neurologic abnormalities can be seen in some patients. These may provide hints about the histopathologic basis for the patient’s disorder. Abnormalities on neurologic exam, when present,mayinvolve an extrapyramidal disorder. This includes features of CBS such as unilateral rigidity, dystonia, myoclonus and limb apraxia58, 61. There may also be a disorder of vertical gaze and axial rigidity associated with PSPS40, 62. As noted above, some of these patients have prominent AoS.
naPPA also can be seen in the context of a pyramidal motor system disorder. Thisclinical picture suggests amyotrophic lateral sclerosis (ALS), with bulbar and limb weakness, muscle wasting, fasciculations, abnormal myotactic reflexes, and an extensor great toe63-65. Patients presenting with naPPA thus should be assessed neurologically since the presence of CBS or ALS may have a significant impact on prognosis.
THE NEUROANATOMIC BASIS FOR NONFLUENT/AGRAMMATIC PRIMARY PROGRESSIVE APHASIA
Structural and Functional Imaging Studies of naPPA
The term “primary” in “primary progressive aphasia” is intended to emphasize the absence of obvious structural abnormalities that can explain the impairment in these patients. This includes vascular disease,space-occupying lesions, infections, inflammatory conditions, head trauma, hydrocephalus, and other disorders.
Nevertheless, there is extensive imaging evidence suggesting focal CNS disease in naPPA. This clinical imaging marker of naPPAiscentered in the left frontal lobe. Several imaging techniques have been used to help specify the anatomic distribution of disease associated with naPPA. Structural MRI studies of the brain emphasize gray matter atrophyin the inferior frontal region of the left hemisphere48, 66, 67 (Figure 2). This typically involves adjacent frontal operculum and anterior insula, and may extend more dorsally into left prefrontal regions and ventrally into superior portions of the left anterior temporal lobe55. This may correspond to the anatomic distribution of atrophy in patients with known tau pathology, a frequent cause of naPPA, as discussed below68. In patients with prominent AoS, imaging changes reported in premotor and supplementary motor areas somewhat more dorsally in the left hemisphere40, 45. These structural findings are confirmed by functional imaging with single photon emission computed tomography, positron emission tomography (PET), and arterial spin labeling MRI. For example, PET glucose hypometabolism is seen in the left inferior frontal lobe, including the frontal operculum and the anterior insula69.
FIGURE 2. GRAY MATTER ATROPHY AND WHITE MATTER DISEASE IN naPPA.
1. Patients (n=12) meeting published criteria for naPPA 7 with CSF totaltau: amyloid-beta ratio <0.34 consistent with FTD spectrum pathology 6. UPPER PANEL: Red areas show significant gray matter atrophy at p<0.0025 (FDRcorrected) that is most evident in the left frontal lobe, including inferior, opercular, dorsolateral regions, extending into insula and anterior-superior temporal regions. There is also involvement of the right frontal lobe. LOWER PANEL: Solid areas show significantly reduced fractional anisotropy at p<0.01 (FDRcorrected) that is more prominent on the left than the right. This includes left inferior frontal-occipital fasciculus as it courses through the external capsule (solid green), bilateral inferior frontal-occipital fasciculus, arcuate fasciculus and anterior thalamic radiations coursing through the anterior corona radiata and most anterior portion of the internal capsule bilaterally (solid red), and interhemispheric fibers of the corpus callosum (solid blue).blue=corpus callosum, red=internal capsule, green=external/extreme capsule, orange=fornix.
Disease in naPPA also compromises white matter, as illustrated in Figure 2. This reflects histopathologic evidence for extensive white matter disease in these patients70. Recent work using diffusion tensor imaging (DTI) demonstrates reduced fractional anisotropy (FA) that reflectschanges in white matter integrity in projections related to the inferior frontal lobe71, 72.
Imaging studies also can contribute to understanding the cause of naPPA. This is important because, as noted below, most naPPA patients have a tauopathy but up to 30% of patients with naPPA may have underlying AD pathology. Reduced parietal glucose metabolism thus is seen in PET scans of patients with pathologically-confirmed naPPA due to AD compared to naPPA patients with non-AD pathology73. Likewise, MRI gray matter atrophy extending into the left inferior parietal regionin naPPAis associated with biomarker and autopsy evidence for AD compared to naPPA patients with FTLD spectrum pathology74.
PET also can be used for radioligand imaging with Pittsburgh compound B (PiB). PiB can help determine the pathologic basis for naPPAmore directly because PiB tags amyloid, a component of AD pathology that is not associated with FTLD spectrum pathology. PiB studies of PPA thusmay help distinguish between naPPA due to AD and non-AD pathology75.
MR spectroscopy also can be informative byobtaining a chemical profileof the brain. In a study of naPPA occurring in the context of CBS76, a reduction in the ratio of n-acetyl aspartate/creatine (NAA/Cr) relative to phosphocreatine was found in the left hemisphere. This is significant because NAA is thought to reflect neuronal integrity. Likewise, patients with a mutation of microtubule-associated protein - tau (MAPT) that codes for tau, a common pathology associated with naPPA, may have an abnormal reduction of NAA/Cr77.
Structure-Function Relationships in naPPA
Additional work has begun to demonstrate how disease intheleft inferior frontal and anterior-superior temporal regionsas well as the associated white matter tracts interrupts a large-scale neural network contributing to the language disorder evident in this syndrome. This has been addressed with several approaches. The most straightforward methodinvolves a regression analysis directly relating quantitative gray matter atrophy to a measure of language functioning. A cardinal characteristic of naPPA is reduced speech fluency, and quantification of this deficit in a semi-structured speech sample is related to atrophy in inferior frontal and anterior superior temporal regions of the left hemisphere35, 36, 78. Reduced speech fluency in turn may be related to three factors, including grammatical difficulty, AoS, and executive dysfunction on measures like letter-guided naming fluency. In one study, only difficulty associated with grammatical processing overlaps areas of cortical atrophy related to non-fluent speech, and this is evident in left inferior frontal and anterior superior temporal regions37. Another study associates AoS with left inferior frontal atrophy 41, although the operational definition of AoS included both phonetic errors consistent with AoS and phonologic errors resulting from an impairment of the linguistic system responsible for processing the abstract rules governing speech sounds.
Functional MRI also hasbeen used to assess the role of left inferior frontal disease in naPPA. In one study, healthy controls, patients with naPPA, and patients with bvFTD silently read sentences featuring a complex grammatical structure and containing a prepositional phrase that stresses working memory79. Controls activate both ventral portions of the left frontal lobeassociated with grammatical processing and dorsal left frontal regions associated with working memory. By comparison, naPPA patients activate only dorsal portions of the left frontal lobe, but did not activate the ventral region associated with grammatical processing. Non-aphasic patients with bvFTD activate the ventral frontal region but not the dorsal area, in keeping with their prominent working memory deficit. Another fMRI studyshowed grammatically simple sentences and grammatically complex sentences to naPPA patients and healthy controls46. Controls activate left inferior frontal regions during grammatically complex sentences more than simple sentences, while naPPA patients do not show adifference in left inferior frontal activation for these types of sentences. These findings emphasize the crucial contribution of left inferior frontal disease to the naPPA syndrome.
Recent DTI work such as that illustrated in Figure 2, when combined with areas of gray matter atrophy,emphasizes the breakdown of a large-scale neural network80 that is responsible in part for the language deficits seen in naPPA. A ventral stream and a dorsal stream support connectivity within this peri-Sylvian language system. In Figure 2, interruption of the ventral stream may play a critical role in grammatical and lexical processing deficits related to major grammatical category found in naPPA. In Figure 2, ventral fiber tracts projecting posteriorly via the inferior frontal-occipital fasciculus that have reduced FA are green. Likewise, projections through the so-called dorsal stream appear to play an important role in the long-distance syntactic dependencies that contribute to the grammatical deficits of naPPA. Dorsal fiber tracts contributing to the arcuate fasciculus that have significantly reduced FA are red.
GENETIC ASSOCIATIONS OF NONFLUENT/AGRAMMATIC PRIMARY PROGRESSIVE APHASIA
A strongly positive family history approaches 30% in FTLD81, 82. However, there is little evidence suggesting that the clinical syndrome of naPPA is inherited in an autosomal dominant manner. The gene first associated with FTLD – MAPT – is found on chromosome 1783. Another common mutation associated with FTLD – progranulin (PGRN) – also is found on chromosome 1784, 85. Recently, a common cause of familial FTLD and ALS was associated with a hexanucleotide repeat expansionin a non-coding region on chromosome 9 (C9ORF72)86, 87. Each of these genetic mutations is associated with a specific histopathologic abnormality commonly associated with naPPA, as summarized in Table 2 and discussed in greater detail below. Other less common chromosomal mutations include VCP on chromosome 988, CHMP2B on chromosome 389, and a mutation of TARDBP on chromosome 190.
TABLE 2.
GENETIC-PATHOLOGICAL ASSOCIATIONS FOUND IN PATIENTS WITH NONFLUENT/AGRAMMATIC PRIMARY PROGRESSIVE APHASIA
| Genetic Mutation |
Pathology | |
|---|---|---|
| FTLD-tau | FTLD-TDP | |
| MAPT | X | |
| PGRN | X | |
| C9ORF72 | X | |
| TARDBP | X | |
While FTLD is frequently inherited, well-documented families with naPPA appear to be rare. Hereditary dysphasic dementia (HDDD), for example, appears to be related to the FTLD spectrum of disease91, 92. More recently, HDDD-2 was associated with ubiquitin-positive, tau-negative pathology due to a PGRN mutation93. Two familieswithnaPPA have been described, and this was associated with a PGRN mutation94, 95. Other reports have described families with naPPA and a behavioral disorder due to a PGRN mutation96, 97. While mutations of PGRN clearly can result in naPPA98-100, PGRN mutations do not appear to be predisposed to causing a naPPA phenotype. It is important to note in this context that clinical FTLD syndromes can be highly variable even in members of a family with the identicalPGRN mutation100, 101. Indeed, the presence of a PGRN mutation and a PPA phenotype do not appear to be highly correlated. One study, for example, reported that 60% of 25 patients with a PGRN mutation have reduced speech production for a variety of reasons, only half of these patients had speech difficulty at presentation, and only three of these patients appeared to have a naPPA phenotype at presentation without an accompanying social disorder98. In another series, PPA appeared to be relatively uncommon at presentation, although a language disorder did eventually emerge in many individuals102.
Mutations of MAPT are associated with a tauopathy, and these also have been related at times tonaPPA. For example, one sister of a positive sib-pair with a MAPT mutation had reduced speech fluency and was impaired on the Token test assessment of grammatical comprehension103. The H1/H1 haplotype that is linkedto the region on chromosome 17 coding for tau appears to be associated with PPA104. While mutations of MAPT may be related tonaPPA105, a mutation of MAPT does not result in a disproportionately high frequency of naPPA. As with PGRN, the phenotype associated with a specific MAPT mutation within a family may be highly variable106.
Recently, a hexanucleotide repeat expansion on chromosome 9 in a non-coding open reading frame location of unclear function (C9ORF72) was reported in families with FTD as well as ALS86, 87.As in ALS-FTD, occasional patients with a C9ORF72 repeat expansion may have a naPPA phenotype107, 108. Although other genetic mechanisms of disease remain to be elucidated, chromosomal mutations associated with an autosomal dominant disorder do not appear to be disproportionately associated with naPPA.
PATHOLOGY ASSOCIATED WITH NONFLUENT/AGRAMMATIC PRIMARY PROGRESSIVE APHASIA
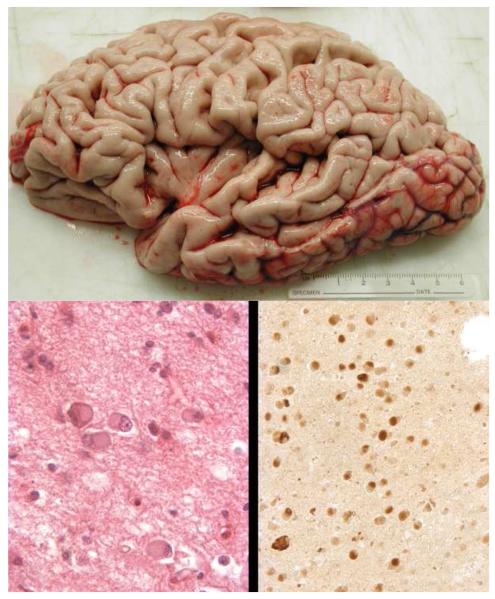
Gross pathology in naPPA shows focal atrophy centered in the left inferior frontal and anterior-superior temporal region of the left hemisphere (Figure 3). This corresponds to the core area of disease seen in imaging studies during life. It has been proposed that the class of von Economo neurons found only in higher-order primates may be diseased in patients with FTLD109.These neurons are found in inferior frontal regions that support uniquely human capacities like grammar, although this cannot account for the full spectrum of disease seen in naPPA.
FIGURE 3. PATHOLOGY IN naPPA.
1. Upper panel shows gross specimen of a patient with naPPA, illustrating significant inferior frontal and anterior-superior temporal atrophy. Lower panel left shows H & E preparation of histopathology demonstrating neuronal cytoplasmic inclusions consistent with Pick bodies; lower panel right shows tauimmunoreactive staining of neuronal inclusions.
Several different pathologies can result in naPPA. Microscopic examination of naPPA at autopsy often reveals frontotemporal lobar degeneration associated with a tauopathy (FTLD-tau). Someclinical-pathological series have associated naPPA only with tau-positive pathologies. In a comprehensive assessment of naPPA and AoS, cases with either naPPA or AoS clinical features have tau-positive forms of pathology40. This included patients with progressive supranuclear palsy (PSP), although this was also seen in corticobasal degeneration (CBD) and dementia with Pick bodies (PiD). In another series, the clinical diagnosis of naPPA was associated only with PiD pathology110.
In other studies, the pathology associated with naPPA has been overwhelmingly associated withFTLD with transactive-response DNA-binding protein of ~43 kD (FTLD-TDP). For example, all but one of the naPPA patients in a large series had FTLD-TDP pathology111. Four different forms of TDP-43 histopathology have been described112, 113, and the variant with frequent dystrophic neurites and neuronal cytoplasmic inclusions known as Type A was found to be particularly prominent in naPPA18, 114.
In most series, however, the pathology underlying naPPAhas been mixed, with a predominance of FTLD-tau pathology. Hodges noted mostly tau-immunoreactive pathology associated with naPPA20. In follow-up studies, a variety of pathologies have been catalogued in their 23 naPPA patients, including FTLD-tau in 11 patients, AD in seven patients, dementia lacking distinctive histopathology in one patient, and “motor neuron disease inclusion dementia” (presumably FTLD-TDP) in four patients115-117. Among 10 cases with naPPA, another British series reported tau pathology in 7 cases, equally divided between PiD and CBD, and TDP-43 Type Apathology in 3 cases23. Kertesz and his colleagues followed 20 naPPA patients longitudinally. In addition to 9 cases of tau-related pathology, they found AD pathology in 9 cases, and “motor neuron disease type inclusions” (presumably FTLD-TDP) in 2 patients17. Mesulamfound mostly tau pathology associated with naPPA,but one of his of his 6cases had FTLD-TDP pathology118. In another series of 9 naPPA cases followed longitudinally, FTLD-tau was found in 6 cases and AD pathology in 3 patients119. Among Knopman’s naPPA cases, most had tau-immunoreactive pathology but2had pathology consistent with FTLD-U and one of these cases had additional AD pathology21. Another series from the same institution found PiD, FTLD-U, PSP, and CBD pathologies in cases of naPPA120. Two cases of dementia with Lewy bodies (DLB) have been associated with naPPA5, 17. Two summaries of clinical-pathological series such as these have suggested that about 70% of naPPA patients have tau pathology, andmany of the remainder have AD pathology5, 22. While not definitive, the naPPA phenotype thus appears to be biased towards FTLD-tau pathology. In an era of disease-modifying treatments, naPPA thus may be avaluable screening tool for identifying patients likely to have FTLD-tau pathology who would then be eligible for additional biomarker studies defining the underlying cause of their disorder such as cerebrospinal fluid.
RESOLVING SOURCES OF CONFUSION IN NONFLUENT/AGRAMMATIC PRIMARY PROGRESSIVE APHASIA
Clinical identification
A major source of controversy has been clinical identification of patients with naPPA. The speech characteristic of naPPA involves grammatical processing difficulty, including simplified syntactic structures and grammatical errors. This accompanied by difficulty understanding sentences that depend on grammatical relations. This may be accompanied by a specific pattern of errors in speech sound production that mark AoS. Effortful, non-fluent speech characteristic of naPPA is not the gradual diminution of speech initiation to the point of muteness that can be seen in bvFTD patients with apathy.
Likewise, effortful speech in naPPA is not a reduction of speech fluency due to lengthy, word-finding pauses that can be seen in lvPPA and even svPPA. Cases of lvPPA may be particularly difficult to distinguish from patients with naPPA because lvPPA also may manifest impaired sentence processing because of auditory-verbal short-term memory deficits. Thiscan interfere with processing lengthy sentences and thus resemble the grammatical deficits found in naPPA. Written sentence materials can be used to help distinguish between the grammatical deficit of naPPA compared to the auditory-verbal limitations of lvPPA. Other features of lvPPA that can help distinguishthis syndrome from naPPA include difficulty with multi-syllabic word and sentence repetition, and poor performance on neuropsychological measures like repeating sequences of digits 10, 121. While both lvPPA and naPPA may have gray matter atrophy in a left anterior peri-Sylvian distribution, cortical disease extends into the posterior peri-Sylvian region in lvPPA73, 74. Finally, since many patients with lvPPA can evolve to typical clinical Alzheimer’s disease and often have underlying AD pathology5, 118, 122, 123, cerebrospinal fluid tau:amyloid-beta ratio74 and PiB PET imaging75 studies can be helpful in identifying patients more likely to have lvPPA rather than naPPA.
Imaging abnormalities
There has been some controversy identifying clinical imaging markers of naPPA. The focus of disease detected by MRI and PET appears to be in the left inferior frontal lobe and adjacent frontal operculum and anterior insula. This frequently extends dorsally and anteriorly to involve additional frontal regions, and inferiorly to involve anterior-superior portions of the left temporal lobe. Recent diffusion-weighted imaging studies emphasize that there is extensive involvement of white matter that limits connectivity between these frontal regions and other language and cognitive areas in the cerebrum. Thus, an important consequence of this anatomic distribution of disease is the disruption of the large-scale peri-Sylvian neural network essential for sentence processing.
Pathologic basis for disease
The histopathologic abnormalities found in naPPA are not homogeneous. However, there appears to be a clear bias in a majority of clinical-pathological series toward finding a tauopathy in naPPA. The syndrome of naPPA thus may be a relatively inexpensive and non-invasive way to screen for underlying tau pathology. Follow-up studies with imaging and biofluid biomarkers thus may be used to rule out other pathologies and confirm the presence of tau-immunoreactive pathology. Imaging studies using agents such as Pittsburgh Compound B can help identify some patients who may have the histopathologic features of AD75, and cerebrospinal fluid analyses can be used to rule out AD as well as patients who may have underlying TDP-43 pathology124.
SUMMARY
naPPA is a progressive neurodegenerative condition that is important to recognize because it may help identify patients with underlying tau pathology. The sentinel clinical feature of naPPA is effortful, non-fluent speech. This can be quantified by reduced words per minute,and confirmed by grammatical simplifications and errors in grammatical comprehension and expression, and the presence of AoS. Limited executive resourcesalso may be present, and other aspects of language and cognition may become compromised over time. There are many other characteristics of impaired language in naPPA that remain to be quantified, such as phonologic processing of speech sounds during reception, the role of executive resources in sentence processing,and the nature of disorders of prosody. Additional longitudinal work would be very valuable in optimizing our syndromic classification of the PPAs.
Many imaging modalities associate naPPA with disease centered in the inferior portions of the left frontal lobe. This is a useful clinical marker, but the language disorders characteristic of this syndrome appear to be compromised by disruption of a large-scale peri-Sylvian language network. A recent advance underlining this approach is the demonstration of significant white matter disease in diffusion-weighted imaging studies of naPPA. Future contributions can focus on defining the specific roles of disease in dorsal and ventral white matter tracts that are compromised in the large-scale neural networks of naPPA patients. The mechanism underlying these white matter imaging changes also remains to be elucidated, such as distinguishing between Wallerian degeneration associated with cortical disease compared todirect white matter pathology. This work will lead to important advances in the cognitive neuroscience of language.
Perhaps the most pressing clinical issue for the field is improving our ability to identify tauopathies. naPPA is most commonly associated with a form of FTLD-tau, and thus identifying the presence of this syndrome may be a useful clinical screen for tauopathy. With the emergence of disease-modifying substances, it becomes essential to recognize conditions like naPPA that are often caused by tauopathies because of the potential benefit that these patients may derive from these treatments. Additional biomarker work is needed to help identify a set of features that can identify a tauopathy with greater reliability.
Acknowledgments
This work was supported in part by National Institutes of Health (AG017586, AG015116, AG032953, NS044266 and NS053488) and the Wyncote Foundation. I participate in clinical trials sponsored by Allon Pharmaceuticals, Forest Laboratories, and Bristol-Myer-Squibb, and consult Allon and Bristol-Myer-Squibb for treatment trials unrelated to this condition. I receive travel funds directly from academic institutions to support speaking at Grand Rounds periodically.
I would like to express my appreciation to many outstanding collaborators, to patients and their families who taught me about naPPA, and to Sherry Ash who provided valuable comments on an earlier version of this manuscript.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
CONFLICT OF INTEREST STATEMENT
There are no conflicts of interest.
BIBLIOGRAPHY
- 1.Pick A. Uber die Beziehungen der senilen Hirnatrophie zue Aphasie. Prager Medicinische Wochenschrift. 1892;17:165–7. [Google Scholar]
- 2.Serieux P. Sur un cas de surdite verbale pure. Revue Medicale. 1893;13:733–50. [Google Scholar]
- 3.Mesulam MM. Slowly progressive aphasia without generalized dementia. Annals of Neurology. 1982;11:592–8. doi: 10.1002/ana.410110607. [DOI] [PubMed] [Google Scholar]
- 4.Mesulam MM. Primary progressive aphasia. Annals of Neurology. 2001;49:425–32. [PubMed] [Google Scholar]
- 5.Grossman M. Primary progressive aphasia: Clinical-pathological correlations. Nature Reviews Neurology. 2010;6:88–97. doi: 10.1038/nrneurol.2009.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Irwin DJ, McMillan C, Toledo JB, Arnold SE, Shaw LM, Wang L-S, et al. Comparison of cerebrospinal fluid levels of tau and Abeta1-42 in Alzheimer’s disease and frontotemporal degeneration using two analytical platforms. Archives of Neurology. 2012 doi: 10.1001/archneurol.2012.26. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gorno-Tempini ML, Hillis AE, Weintraub S, Kertesz A, Mendez M, Cappa SF, et al. Classification of primary progressive aphasia and its variants. Neurology. 2011;76(11):1006–14. doi: 10.1212/WNL.0b013e31821103e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mackenzie IR, Neumann M, Bigio E, Cairns NJ, Alafuzoff I, Kril JJ, et al. Nomenclature and nosology for neuropathologic subtypes of frontotemporal lobar degeneration: an update. Acta Neuropathologica. 2010;119(1):1–4. doi: 10.1007/s00401-009-0612-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hodges JR, Patterson K. Semantic dementia: A unique clinicopathological syndrome. Lancet Neurology. 2007;6:1004–14. doi: 10.1016/S1474-4422(07)70266-1. [DOI] [PubMed] [Google Scholar]
- 10.Gorno-Tempini ML, Brambati SM, Ginex V, Ogar J, Dronkers NF, Marcone A, et al. The logopenic/phonological variant of primary progressive aphasia. Neurology. 2008;71(16):1227–34. doi: 10.1212/01.wnl.0000320506.79811.da. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ratnavalli E, Brayne C, Dawson K, Hodges JR. The prevalence of frontotemporal dementia. Neurology. 2002;58:1615–21. doi: 10.1212/wnl.58.11.1615. [DOI] [PubMed] [Google Scholar]
- 12.Harvey RJ, Skelton-Robinson M, Rossor MN. The prevalence and causes of dementia in people under the age of 65 years. J Neurol Neurosurg Psychiatry. 2003;74(9):1206–9. doi: 10.1136/jnnp.74.9.1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ikejima C, Yasuno F, Mizukami K, Sasaki M, Tanimukai S, Asada T. Prevalence and causes of early-onset dementia in Japan: A population-based study. Stroke. 2009;40(8):2709–14. doi: 10.1161/STROKEAHA.108.542308. [DOI] [PubMed] [Google Scholar]
- 14.Knopman DS, Petersen RC, Edland SD, Cha RH, Rocca WA. The incidence of frontotemporal lobar degeneration in Rochester, Minnesota, 1990 through 1994. Neurology. 2004;62:506–8. doi: 10.1212/01.wnl.0000106827.39764.7e. [DOI] [PubMed] [Google Scholar]
- 15.Mercy L, Hodges JR, Dawson K, Barker RA, Brayne C. Incidence of early-onset dementias in Cambridgeshire, United Kingdom. Neurology. 2008;71(19):1496–9. doi: 10.1212/01.wnl.0000334277.16896.fa. [DOI] [PubMed] [Google Scholar]
- 16.Forman MS, Farmer J, Johnson JK, Clark CM, Arnold SE, Coslett HB, et al. Frontotemporal dementia: Clinicopathological correlations. Annals of Neurology. 2006;59:952–62. doi: 10.1002/ana.20873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kertesz A, McMonagle P, Blair M, Davidson W, Munoz DG. The evolution and pathology of frontotemporal dementia. Brain. 2005;128:1996–2005. doi: 10.1093/brain/awh598. [DOI] [PubMed] [Google Scholar]
- 18.Snowden JS, Thompson JC, Stopford CL, Richardson AMT, Gerhard A, Neary D, et al. The clinical diagnosis of early-onset dementias: diagnostic accuracy and clinicopathological relationships. Brain. 2011;134(9):2478–92. doi: 10.1093/brain/awr189. [DOI] [PubMed] [Google Scholar]
- 19.Shi J, Shaw C, Plessis D, Richardson A, Bailey K, Julien C, et al. Histopathological changes underlying frontotemporal lobar degeneration with clinicopathological correlation. Acta Neuropathologica. 2005;110(5):501–12. doi: 10.1007/s00401-005-1079-4. [DOI] [PubMed] [Google Scholar]
- 20.Hodges JR, Davies RR, Xuereb J, Casey BJ, Broe M, Bak T, et al. Clinicopathological correlates in frontotemporal dementia. Annals of Neurology. 2004;56:399–406. doi: 10.1002/ana.20203. [DOI] [PubMed] [Google Scholar]
- 21.Knopman DS, Boeve BF, Parisi JE, Dickson DW, Smith GE, Ivnik RJ, et al. Antemortem diagnosis of frontotemporal lobar degeneration. Annals of Neurology. 2005;57:480–8. doi: 10.1002/ana.20425. [DOI] [PubMed] [Google Scholar]
- 22.Josephs KA, Hodges JR, Snowden JS, Mackenzie IR, Neumann M, Mann D, et al. Neuropathological background of phenotypical variability in frontotemporal dementia. Acta Neuropathologica. 2011;122(2):137–53. doi: 10.1007/s00401-011-0839-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rohrer JD, Lashley T, Schott JM, Warren JE, Mead S, Isaacs AM, et al. Clinical and neuroanatomical signatures of tissue pathology in frontotemporal lobar degeneration. Brain. 2011;134(9):2565–81. doi: 10.1093/brain/awr198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Johnson JK, Diehl J, Mendez MF, Neuhaus J, Shapira JS, Forman MS, et al. Frontotemporal lobar degeneration: Demographic characteristics of 353 patients. Archives of Neurology. 2005;62:925–30. doi: 10.1001/archneur.62.6.925. [DOI] [PubMed] [Google Scholar]
- 25.Roberson ED, Hesse JH, Rose KD, Slama H, Johnson JK, Yaffe K, et al. Frontotemporal dementia progresses to death faster than Alzheimer disease. Neurology. 2005;65:719–25. doi: 10.1212/01.wnl.0000173837.82820.9f. [DOI] [PubMed] [Google Scholar]
- 26.Rascovsky K, Salmon DP, Lipton AM, Leverenz JB, DeCarli C, Jagust WJ, et al. Rate of progression differs in frontotemporal dementia and Alzheimer disease. Neurology. 2005;65:397–403. doi: 10.1212/01.wnl.0000171343.43314.6e. [DOI] [PubMed] [Google Scholar]
- 27.Hodges JR, Davies R, Xuereb J, Kril JJ, Halliday GM. Survival in frontotemporal dementia. Neurology. 2003;61:349–54. doi: 10.1212/01.wnl.0000078928.20107.52. [DOI] [PubMed] [Google Scholar]
- 28.Josephs KA, Knopman DS, Whitwell JL, Boeve BF, Parisi JE, Petersen RC, et al. Survival in two variants of tau-negative frontotemporal lobar degeneration: FTLD-U vs FTLD-MND. Neurology. 2005;65:645–7. doi: 10.1212/01.wnl.0000173178.67986.7f. [DOI] [PubMed] [Google Scholar]
- 29.Xie SX, Forman MS, Farmer J, Moore P, Wang Y, Wang X, et al. Factors associated with survival probability in autopsy-proven frontotemporal lobar degeneration. J Neurol Neurosurg Psychiatry. 2008;79(2):126–9. doi: 10.1136/jnnp.2006.110288. [DOI] [PubMed] [Google Scholar]
- 30.Rosso SM, Landweer EJ, Houterman M, Donker Kaat L, van Duijn CM, van Swieten JC. Medical and environmental risk factors for sporadic frontotemporal dementia: a retrospective case-control study. Journal of Neurology, Neurosurgery, and Psychiatry. 2003;74:1574–6. doi: 10.1136/jnnp.74.11.1574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Medina J, Weintraub S. Depression in Primary Progressive Aphasia. Journal of Geriatric Psychiatry and Neurology. 2007;20(3):153–60. doi: 10.1177/0891988707303603. [DOI] [PubMed] [Google Scholar]
- 32.Grossman M, Mickanin J, Onishi K, Hughes E, D’Esposito M, Ding XS, et al. Progressive non-fluent aphasia: Language, cognitive and PET measures contrasted with probable Alzheimer’s disease. Journal of Cognitive Neuroscience. 1996;8:135–54. doi: 10.1162/jocn.1996.8.2.135. [DOI] [PubMed] [Google Scholar]
- 33.Snowden JS, Neary D, Mann DM. Fronto-temporal Lobar Degeneration: Fronto-temporal Dementia, Progressive Aphasia, Semantic Dementia. Churchill Livingstone; New York: 1996. [Google Scholar]
- 34.Mayer M. Frog, Where Are You? Penguin Books; New York: 1969. [Google Scholar]
- 35.Ash S, Moore P, Vesely L, Gunawardena D, McMillan C, Anderson C, et al. Non-fluent speech in frontotemporal lobar degeneration. Journal of Neurolinguistics. 2009;22:370–83. doi: 10.1016/j.jneuroling.2008.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wilson SM, Henry ML, Besbris M, Ogar JM, Dronkers NF, Jarrold W, et al. Connected speech production in three variants of primary progressive aphasia. Brain. 2010;133(7):2069–88. doi: 10.1093/brain/awq129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gunawardena D, Ash S, McMillan C, Avants B, Gee J, Grossman M. Why are patients with progressive nonfluent aphasia nonfluent? Neurology. 2010;75(7):588–94. doi: 10.1212/WNL.0b013e3181ed9c7d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bak TH, Hodges JR. Kissing and dancing--a test to distinguish the lexical and conceptual contributions to noun/verb and action/object dissociation. Preliminary results in patients with frontotemporal dementia. Journal of Neurolinguistics. 2003;16:169–81. [Google Scholar]
- 39.Hillis AE, Oh S, Ken L. Deterioration of naming nouns versus verbs in primary progressive aphasia. Annals of Neurology. 2004;55:268–75. doi: 10.1002/ana.10812. [DOI] [PubMed] [Google Scholar]
- 40.Josephs KA, Duffy JR, Strand EA, Whitwell JL, Layton KF, Parisi JE, et al. Clinicopathological and imaging correlates of progressive aphasia and apraxia of speech. Brain. 2006;129:1385–98. doi: 10.1093/brain/awl078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rohrer J, Rossor M, Warren J. Apraxia in progressive nonfluent aphasia. Journal of Neurology. 2010;257(4):569–74. doi: 10.1007/s00415-009-5371-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Knibb JA, Woollams AM, Hodges JR, Patterson K. Making sense of progressive non-fluent aphasia: an analysis of conversational speech. Brain. 2009 doi: 10.1093/brain/awp207. awp207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rohrer JD, Rossor MN, Warren JD. Syndromes of nonfluent primary progressive aphasia: A clinical and neurolinguistic analysis. Neurology. 2010;75(7):603–10. doi: 10.1212/WNL.0b013e3181ed9c6b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Josephs KA, Duffy JR, Strand EA, Machulda MM, Senjem ML, Master AV, et al. Characterizing a neurodegenerative syndrome: primary progressive apraxia of speech. Brain. 2012 doi: 10.1093/brain/aws032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ash S, McMillan C, Gunawardena D, Avants B, Morgan B, Khan A, et al. Speech errors in progressive non-fluent aphasia. Brain and Language. 2010;113:13–20. doi: 10.1016/j.bandl.2009.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wilson SM, Dronkers NF, Ogar JM, Jang J, Growdon ME, Agosta F, et al. Neural correlates of syntactic processing in the nonfluent variant of primary progressive aphasia. The Journal of Neuroscience. 2010;30(50):16845–54. doi: 10.1523/JNEUROSCI.2547-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Weintraub S, Mesulam MM, Wieneke C, Rademaker A, Rogalski EJ, Thompson CK. The Northwestern Anagram Test: Measuring sentence production in primary progressive aphasia. American Journal of Alzheimer’s Disease and Other Dementias. 2009;24(5):408–16. doi: 10.1177/1533317509343104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Peelle JE, Troiani V, Gee JC, Moore P, McMillan CT, Vesely L, et al. Sentence comprehension and voxel-based morphometry in progressive nonfluent aphasia, semantic dementia, and nonaphasic frontotemporal dementia. Journal of Neurolinguistics. 2008;21(5):418–32. doi: 10.1016/j.jneuroling.2008.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Mesulam MM, Wieneke C, Rogalski E, Cobia D, Thompson CK, Weintraub S. Quantitative template for subtyping primary progressive aphasia. Arch Neurol. 2009;66(12):1545–51. doi: 10.1001/archneurol.2009.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Grossman M, Rhee J, Antiquena P. Sentence processing in frontotemporal dementia. Cortex. 2005;41:764–77. doi: 10.1016/s0010-9452(08)70295-8. [DOI] [PubMed] [Google Scholar]
- 51.Peelle JE, Cooke A, Moore P, Vesely L, Grossman M. Syntactic and thematic components of sentence processing in progressive nonfluent aphasia and nonaphasic frontotemporal dementia. Journal of Neurolinguistics. 2007;20:482–94. doi: 10.1016/j.jneuroling.2007.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Blair M, marczinski CA, Davis-Faroque N, Kertesz A. A longitudinal study of language decline in Alzheimer’s disease and frontotemporal dementia. Journal of the International Neuropsychological Society. 2007;13:237–45. doi: 10.1017/S1355617707070269. [DOI] [PubMed] [Google Scholar]
- 53.Sapolsky D, Bakkour A, Negreira A, Nalipinski P, Weintraub S, Mesulam M-M, et al. Cortical neuroanatomic correlates of symptom severity in primary progressive aphasia. Neurology. 2010;75(4):358–66. doi: 10.1212/WNL.0b013e3181ea15e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Grossman M, Moore P. A longitudinal study of sentence comprehension difficulty in primary progressive aphasia. Journal of Neurology, Neurosurgery, and Psychiatry. 2005;76:644–9. doi: 10.1136/jnnp.2004.039966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rogalski E, Cobia D, Harrison TM, Wieneke C, Weintraub S, Mesulam M-M. Progression of language decline and cortical atrophy in subtypes of primary progressive aphasia. Neurology. 2011;76(21):1804–10. doi: 10.1212/WNL.0b013e31821ccd3c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Libon DJ, Xie S, Wang X, Massimo L, Moore P, Vesely L, et al. Neuropsychological decline in frontotemporal lobar degeneration: A longitudinal analysis. Neuropsychology. 2008;23:337–46. doi: 10.1037/a0014995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Libon DJ, Xie SX, Moore P, Farmer J, Antani S, McCawley G, et al. Patterns of neuropsychological impairment in frontotemporal dementia. Neurology. 2007;68:369–75. doi: 10.1212/01.wnl.0000252820.81313.9b. [DOI] [PubMed] [Google Scholar]
- 58.Murray R, Neumann M, Forman MS, Farmer J, Massimo L, Rice A, et al. Cognitive and motor assessment in autopsy-proven corticobasal degeneration. Neurology. 2007;68:1274–83. doi: 10.1212/01.wnl.0000259519.78480.c3. [DOI] [PubMed] [Google Scholar]
- 59.Banks SJ, Weintraub S. Neuropsychiatric symptoms in behavioral variant frontotemporal dementia and primary progressive aphasia. Journal of Geriatric Psychiatry and Neurology. 2008;21(2):133–41. doi: 10.1177/0891988708316856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rohrer JD, Warren JD. Phenomenology and anatomy of abnormal behaviours in primary progressive aphasia. Journal of the Neurological Sciences. 2010;293(1-2):35–8. doi: 10.1016/j.jns.2010.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gorno-Tempini ML, Murray RC, Rankin KP, Weiner MW, Miller BL. Clinical, cognitive and anatomical evolution from nonfluent progressive aphasia to corticobasal syndrome: A case report. Neurocase. 2004;10:426–36. doi: 10.1080/13554790490894011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Deramecourt V, Lebert F, Debachy B, Mackowiak-Cordoliani MA, Bombois S, Kerdraon O, et al. Prediction of pathology in primary progressive language and speech disorders. Neurology. 2010;74(1):42–9. doi: 10.1212/WNL.0b013e3181c7198e. [DOI] [PubMed] [Google Scholar]
- 63.Heidler-Gary J, Hillis AE. Distinctions between the dementia in Amyotrophic Lateral Sclerosis with Frontotemporal Dementia and the dementia of Alzheimer’s disease. Amyotrophic Lateral Sclerosis. 2007;8(5):276–82. doi: 10.1080/17482960701381911. [DOI] [PubMed] [Google Scholar]
- 64.Hu WT, Seelaar H, Josephs KA, Knopman DS, Boeve BF, Sorenson EJ, et al. Survival profiles of patients With frontotemporal dementia and motor neuron disease. Archives of Neurology. 2009;66(11):1359–64. doi: 10.1001/archneurol.2009.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lomen-Hoerth C, Anderson T, Miller BL. The overlap of amyotrophic lateral sclerosis and frontotemporal dementia. Neurology. 2002;59:1077–9. doi: 10.1212/wnl.59.7.1077. [DOI] [PubMed] [Google Scholar]
- 66.Gorno-Tempini ML, Dronkers NF, Rankin KP, Ogar JM, Phengrasamy L, Rosen HJ, et al. Cognition and anatomy in three variants of primary progressive aphasia. Annals of Neurology. 2004;55:335–46. doi: 10.1002/ana.10825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sonty SP, Mesulam MM, Thompson CK, Johnson N, Weintraub S, Parrish TB, et al. Primary progressive aphasia: PPA and the language network. Annals of Neurology. 2003;53:35–49. doi: 10.1002/ana.10390. [DOI] [PubMed] [Google Scholar]
- 68.Rohrer JD, Warren JD, Modat M, Ridgway GR, Douiri A, Rossor MN, et al. Patterns of cortical thinning in the language variants of frontotemporal lobar degeneration. Neurology. 2009;72(18):1562–9. doi: 10.1212/WNL.0b013e3181a4124e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Nestor PJ, Graham NL, Fryer TD, Williams GB, Patterson K, Hodges JR. Progressive non-fluent aphasia is associated with hypometabolism centred on the left anterior insula. Brain. 2003;126:2406–18. doi: 10.1093/brain/awg240. [DOI] [PubMed] [Google Scholar]
- 70.Forman MS, Zhukareva V, Bergeron CB, Chin SSM, Grossman M, Clark C, et al. Signature tau neuropathology in gray and white matter of corticobasal degeneration. American Journal of Pathology. 2002;160:2045–53. doi: 10.1016/S0002-9440(10)61154-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Galantucci S, Tartaglia MC, Wilson SM, Henry ML, Filippi M, Agosta F, et al. White matter damage in primary progressive aphasias: a diffusion tensor tractography study. Brain. 2011 doi: 10.1093/brain/awr099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Duda JT, Avants B, Asmuth JA, Zhang H, Grossman M, Gee JC. A fiber tractography-based examination of neurodegeneration on language-network neuroanatomy. Medical Imaging Computation and Computer-Assisted Analysis: MICCAI. 2008:191–8. [Google Scholar]
- 73.Nestor PJ, Balan K, Cheow HK, Fryer TD, Knibb JA, Xuereb JH, et al. Nuclear imaging can predict pathologic diagnosis in progressive nonfluent aphasia. Neurology. 2007;68(3):238–9. doi: 10.1212/01.wnl.0000251309.54320.9f. [DOI] [PubMed] [Google Scholar]
- 74.Hu WT, McMillan C, Libon D, Leight S, Forman M, Lee VM-Y, et al. Multimodal predictors for Alzheimer disease in nonfluent primary progressive aphasia. Neurology. 2010;75(7):595–602. doi: 10.1212/WNL.0b013e3181ed9c52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Rabinovici GD, Jagust WJ, Furst AJ, Ogar JM, Racine CA, Mormino EC, et al. Abeta amyloid and glucose metabolism in three variants of primary progressive aphasia. Annals of Neurology. 2008;64(4):388–401. doi: 10.1002/ana.21451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Vion-Dury J, Rochefort N, Michotey P, Planche D, Ceccaldi M. Proton magnetic resonance neurospectroscopy and EEG cartography in corticobasal degeneration: correlations with neuropsychological signs. Journal of Neurology, Neurosurgery & Psychiatry. 2004;75(9):1352–5. doi: 10.1136/jnnp.2003.018903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kantarci K, Boeve BF, Wszolek ZK, Rademakers R, Whitwell JL, Baker MC, et al. MRS in presymptomatic MAPT mutation carriers. Neurology. 2010;75(9):771–8. doi: 10.1212/WNL.0b013e3181f073c7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Rogalski E, Cobia D, Harrison TM, Wieneke C, Thompson CK, Weintraub S, et al. Anatomy of Language Impairments in Primary Progressive Aphasia. The Journal of Neuroscience. 2011;31(9):3344–50. doi: 10.1523/JNEUROSCI.5544-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Cooke A, DeVita C, Gee JC, Alsop D, Detre J, Chen W, et al. Neural basis for sentence comprehension deficits in frontotemporal dementia. Brain and Language. 2003;85:211–21. doi: 10.1016/s0093-934x(02)00562-x. [DOI] [PubMed] [Google Scholar]
- 80.Friederici AD. The brain basis of language processing: From structure to function. Physiological Reviews. 2011;91(4):1357–92. doi: 10.1152/physrev.00006.2011. [DOI] [PubMed] [Google Scholar]
- 81.Seelaar H, Kamphorst W, Rosso SM, Azmani A, Masdjedi R, de Koning I, et al. Distinct genetic forms of frontotemporal dementia. Neurology. 2008;71:1220–6. doi: 10.1212/01.wnl.0000319702.37497.72. [DOI] [PubMed] [Google Scholar]
- 82.Goldman JS, Rademakers R, Huey ED, Boxer AL, Mayeux R, Miller BL, et al. An algorithm for genetic testing of frontotemporal lobar degeneration. Neurology. 2011;76(5):475–83. doi: 10.1212/WNL.0b013e31820a0d13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Hutton M, Lendon CL, Rizzu P, Baker M, Froelich S, Houlden H, et al. Association of missense and 5′-splice-site mutations in tau with the inherited dementia FTDP-17. Nature. 1998;393:702–5. doi: 10.1038/31508. [DOI] [PubMed] [Google Scholar]
- 84.Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, et al. Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17. Nature. 2006;442:916–9. doi: 10.1038/nature05016. [DOI] [PubMed] [Google Scholar]
- 85.Gass J, Cannon A, Mackenzie IR, Boeve B, Baker M, Adamson J, et al. Mutations in progranulin are a major cause of ubiquitin-positive frontotemporal lobar degeneration. Human Molecular Genetics. 2006;15:2988–3001. doi: 10.1093/hmg/ddl241. [DOI] [PubMed] [Google Scholar]
- 86.DeJesus-Hernandez M, Mackenzie IR, Boeve BF, Boxer AL, Baker M, Rutherford NJ, et al. Expanded GGGGCC Hexanucleotide Repeat in Noncoding Region of C9ORF72 Causes Chromosome 9p-Linked FTD and ALS. Neuron. 2011;72(2):245–56. doi: 10.1016/j.neuron.2011.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Renton AE, Majounie E, Waite A, SimÛn-Snchez J, Rollinson S, Gibbs JR, et al. A Hexanucleotide Repeat Expansion in C9ORF72 Is the Cause of Chromosome 9p21-Linked ALS-FTD. Neuron. 2011;72(2):257–68. doi: 10.1016/j.neuron.2011.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Watts GDJ, Wymer J, Kovach MJ, Mehta SG, Mumm S, Darvish D, et al. Inclusion body myopathy associated with Paget disease of bone and frontotemporal dementia is caused by mutant valosin-containing protein. Nature Genetics. 2004;36(4):377–81. doi: 10.1038/ng1332. [DOI] [PubMed] [Google Scholar]
- 89.Skibinski G, Parkinson NI, Brown JM, Chakrabarti L, Lloyd SL, Hummerich H, et al. Mutations in the endosomal ESCRTIII-complex subunit CHMP2B in frontotemporal dementia. Nature Genetics. 2005;37:806–9. doi: 10.1038/ng1609. [DOI] [PubMed] [Google Scholar]
- 90.Chio A, Calvo A, Moglia C, Restagno G, Ossola I, Brunetti M, et al. Amyotrophic Lateral Sclerosis-Frontotemporal Lobar Dementia in 3 Families With p.Ala382Thr TARDBP Mutations. Arch Neurol. 2010;67(8):1002–9. doi: 10.1001/archneurol.2010.173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lendon CL, Shears S, Busfield F. Molecular genetics of hereditary dysphasic dementia. Neurobiology of Aging. 1994;15:S128. [Google Scholar]
- 92.Morris JC, Cole M, Banker BQ, Wright D. Hereditary dysphasic dementia and the Pick-Alzheimer spectrum. Annals of Neurology. 1984;16:455–66. doi: 10.1002/ana.410160407. [DOI] [PubMed] [Google Scholar]
- 93.Mukherjee O, Pastor P, Cairns NJ, Chakraverty C, Kauwe JSK, Shears S, et al. HDDD2 is a familial frontotemporal lobar degeneration with ubiquitin-positive, tau-negative inclusions caused by a missense mutation in the signal peptide of progranulin. Annals of Neurology. 2006;60(3):314–22. doi: 10.1002/ana.20963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Mesulam MM, Johnson N, Krefft TA, Gass JM, Cannon AD, Adamson JL, et al. Progranulin mutations in primary progressive aphasia: The PPA1 and PPA3 families. Archives of Neurology. 2007;64:43–7. doi: 10.1001/archneur.64.1.43. [DOI] [PubMed] [Google Scholar]
- 95.Davion S, Johnson N, Weintraub S, Mesulam M-M, Engberg A, Mishra M, et al. Clinicopathologic correlation in PGRN mutations. Neurology. 2007;69(11):1113–21. doi: 10.1212/01.wnl.0000267701.58488.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Snowden JS, Pickering-Brown SM, Mackenzie IR, Richardson AMT, Varma A, Neary D, et al. Progranulin gene mutations associated with frontotemporal dementia and progressive non-fluent aphasia. Brain. 2006;129(11):3091–102. doi: 10.1093/brain/awl267. [DOI] [PubMed] [Google Scholar]
- 97.Rohrer JD, Geser F, Zhou J, Gennatas ED, Sidhu M, Trojanowski JQ, et al. TDP-43 subtypes are associated with distinct atrophy patterns in frontotemporal dementia. Neurology. 2010;75(24):2204–11. doi: 10.1212/WNL.0b013e318202038c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Beck J, Rohrer JD, Campbell T, Isaacs A, Morrison KE, Goodall EF, et al. A distinct clinical, neuropsychological and radiological phenotype is associated with progranulin gene mutations in a large UK series. Brain. 2008;131(3):706–20. doi: 10.1093/brain/awm320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kelley BJ, Haidar W, Boeve BF, Baker M, Graff-Radford NR, Krefft T, et al. Prominent phenotypic variability associated with mutations in Progranulin. Neurobiology of Aging. 2009;30(5):739–51. doi: 10.1016/j.neurobiolaging.2007.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Le Ber I, Camuzat A, Hannequin D, Pasquier F, Guedj E, Rovelet-Lecrux A, et al. Phenotype variability in progranulin mutation carriers: a clinical, neuropsychological, imaging and genetic study. Brain. 2008;131(3):732–46. doi: 10.1093/brain/awn012. [DOI] [PubMed] [Google Scholar]
- 101.Leverenz JB, Yu CE, Montine TJ, Steinbart E, Bekris LM, Zabetian C, et al. A novel progranulin mutation associated with variable clinical presentation and tau, TDP43 and alpha-synuclein pathology. Brain. 2007;130(5):1360–74. doi: 10.1093/brain/awm069. [DOI] [PubMed] [Google Scholar]
- 102.Rademakers R, Baker M, Gass J, Adamson J, Huey ED, Momeni P, et al. Phenotypic variability associated with progranulin haploinsufficiency in patients with the common 1477C T (Arg493X) mutation: an international initiative. Lancet Neurology. 2007;6:857–68. doi: 10.1016/S1474-4422(07)70221-1. [DOI] [PubMed] [Google Scholar]
- 103.Boeve BF, Tremont-Lukats IW, Waclawik AJ, Murrell JR, Hermann B, Jack CR, Jr., et al. Longitudinal characterization of two siblings with frontotemporal dementia and parkinsonism linked to chromosome 17 associated with the S305N tau mutation. Brain. 2005;128:752–72. doi: 10.1093/brain/awh356. [DOI] [PubMed] [Google Scholar]
- 104.Sobrido MJ, Abu-Khalil A, Weintraub S, Johnson N, Quinn B, Cummings J, et al. Possible association of the tau H1/H1 genotype with primary progressive aphasia. Neurology. 2003;60:862–4. doi: 10.1212/01.wnl.0000049473.36612.f2. [DOI] [PubMed] [Google Scholar]
- 105.Kaivorinne A-L, Kruger J, Kuivaniemi K, Tuominen H, Moilanen V, Majamaa K, et al. Role of MAPT mutations and haplotype in frontotemporal lobar degeneration in Northern Finland. BMC Neurology. 2008;8(1):48. doi: 10.1186/1471-2377-8-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Bird TD, Nochlin D, Poorkaj P, Cherrier M, Kaye J, Payami H, et al. A clinical pathological comparison of three families with frontotemporal dementia and identical mutations in the tau gene (P301L) Brain. 1999;122(4):741–56. doi: 10.1093/brain/122.4.741. [DOI] [PubMed] [Google Scholar]
- 107.Snowden JS, Rollinson S, Thompson JC, Harris JM, Stopford CL, Richardson AMT, et al. Distinct clinical and pathological characteristics of frontotemporal dementia associated with C9ORF72 mutations. Brain. 2012;135(3):693–708. doi: 10.1093/brain/awr355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Mahoney CJ, Beck J, Rohrer JD, Lashley T, Mok K, Shakespeare T, et al. Frontotemporal dementia with the C9ORF72 hexanucleotide repeat expansion: clinical, neuroanatomical and neuropathological features. Brain. 2012;135(3):736–50. doi: 10.1093/brain/awr361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Seeley WW, Carlin DA, Allman JM, Macedo MN, Bush C, Miller BL, et al. Early frontotemporal dementia targets neurons unique to apes and humans. Annals of Neurology. 2006;60(6):660–7. doi: 10.1002/ana.21055. [DOI] [PubMed] [Google Scholar]
- 110.Yokota O, Tsuchiya K, Arai T, Yagishita S, Matsubara O, Mochizuki A, et al. Clinicopathological characterization of Pick’s disease versus frontotemporal lobar degeneration with ubiquitin/TDP-43-positive inclusions. Acta Neuropathologica. 2009;117(4):429–44. doi: 10.1007/s00401-009-0493-4. [DOI] [PubMed] [Google Scholar]
- 111.Snowden JS, Neary D, Mann D. Frontotemporal lobar degeneration: clinical and pathological relationships. Acta Neuropathologica. 2007;114(1):31–8. doi: 10.1007/s00401-007-0236-3. [DOI] [PubMed] [Google Scholar]
- 112.Sampathu DM, Neumann M, Kwong LK, Chou TT, Micsenyi M, Truax AC, et al. Pathological heterogeneity of frontotemporal lobar degeneration with ubiquitin-positive inclusions delineated by ubiquitin immunohistochemistry and novel monoclonal antibodies. American Journal of Pathology. 2006;169:1343–52. doi: 10.2353/ajpath.2006.060438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Mackenzie IR, Baborie A, Pickering-Brown S, Plessis D, Jaros E, Perry R, et al. Heterogeneity of ubiquitin pathology in frontotemporal lobar degeneration: classification and relation to clinical phenotype. Acta Neuropathologica. 2006;112(5):539–49. doi: 10.1007/s00401-006-0138-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Josephs KA, Stroh A, Dugger B, Dickson D. Evaluation of subcortical pathology and clinical correlations in FTLD-U subtypes. Acta Neuropathologica. 2009;118(3):349–58. doi: 10.1007/s00401-009-0547-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Alladi S, Xuereb J, Bak T, Nestor P, Knibb JA, Patterson K, et al. Focal cortical presentations of Alzheimer’s disease. Brain. 2007;130(10):2636–45. doi: 10.1093/brain/awm213. [DOI] [PubMed] [Google Scholar]
- 116.Galton CJ, Patterson K, Xuereb J, Hodges JR. Atypical and typical presentations of Alzheimer’s disease: A clinical, neuropsychological, neuroimaging, and pathological study of 13 cases. Brain. 2000;123:484–98. doi: 10.1093/brain/123.3.484. [DOI] [PubMed] [Google Scholar]
- 117.Knibb JA, Xuereb J, Patterson K, Hodges JR. Clinical and pathological characterization of progressive aphasia. Annals of Neurology. 2006;59(1):156–65. doi: 10.1002/ana.20700. [DOI] [PubMed] [Google Scholar]
- 118.Mesulam MM, Wicklund A, Johnson N, Rogalski E, Leger GC, Rademaker A, et al. Alzheimer and frontotemporal pathology in subsets of primary progressive aphasia. Annals of Neurology. 2008;63(6):709–19. doi: 10.1002/ana.21388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Grossman M, Xie SX, Libon DJ, Wang X, Massimo L, Moore P, et al. Longitudinal decline in autopsy-defined frontotemporal lobar degeneration. Neurology. 2008;70(22):2036–45. doi: 10.1212/01.wnl.0000303816.25065.bc. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Josephs KA, Petersen RC, Knopman DS, Boeve BF, Whitwell JL, Duffy JR, et al. Clinicopathologic analysis of frontotemporal and corticobasal degenerations and PSP. Neurology. 2006;66:41–8. doi: 10.1212/01.wnl.0000191307.69661.c3. [DOI] [PubMed] [Google Scholar]
- 121.Rohrer JD, Ridgway GR, Crutch SJ, Hailstone J, Goll JC, Clarkson MJ, et al. Progressive logopenic/phonological aphasia: Erosion of the language network. Neuroimage. 2009;49(1):984–93. doi: 10.1016/j.neuroimage.2009.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Josephs KA, Whitwell JL, Duffy JR, Vanvoorst WA, Strand EA, Hu WT, et al. Progressive aphasia secondary to Alzheimer disease vs FTLD pathology. Neurology. 2008;70(1):25–34. doi: 10.1212/01.wnl.0000287073.12737.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Rohrer JD, Rossor MN, Warren JD. Alzheimer’s pathology in primary progressive aphasia. Neurobiology of Aging. 2010 doi: 10.1016/j.neurobiolaging.2010.05.020. In Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Hu WT, Chen-Plotkin A, Grossman M, Arnold SA, Clark C, Shaw LM, et al. Novel CSF biomarkers for frontotemporal lobar degenerations. Neurology. 2010;75:2079–86. doi: 10.1212/WNL.0b013e318200d78d. [DOI] [PMC free article] [PubMed] [Google Scholar]