Abstract
Recent studies have shown that a small subpopulation of stem-like cancer cells within most solid tumors are responsible for the malignancy of aggressive cancer cells, including tumorigenicity and relapse of solid tumors. These tumor cells may be enriched and maintained in vitro in the presence of growth factors (GFs), including epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF), without serum in almost all solid tumor cell lines. In the present study, however, we found that the enrichment of cancer stem-like cells (CSCs) from established cell lines using sphere cultures was achieved efficiently without GFs. Enhanced sphere-forming activity was detected in GF-free cultures compared with GF-containing cultures in MCF7, A549 and U87 cells. Analysis of conditioned media (CM) demonstrated an elevated level of EGF secretion in GF-free CM compared with GF-containing CM in MCF7 cells. By contrast, cells in GF-free CM exhibited almost no secretion of bFGF, whereas cells in GF-containing CM secreted a high level of bFGF. The addition of EGF enhanced sphere formation, whereas bFGF suppressed sphere formation under GF-free conditions in MCF7 cells. Conversely, the addition of bFGF suppressed sphere formation in these cells. Notably, bFGF markedly suppressed EGF receptor (EGFR) expression and EGF secretion in MCF7 and A549 cells. Consistent with this result, EGFR blockade with pharmacological inhibitors significantly suppressed sphere formation in MCF7 and A549 cells under GF-free conditions. Furthermore, the neutralization of EGF also inhibited sphere formation, whereas bFGF neutralization still enhanced sphere formation under these conditions. Together, CSCs may be maintained in a serum-free culture condition without GFs, possibly through autocrine secretion of GFs such as EGF, and the addition of bFGF may not be sufficient for the enrichment of stem-like cancer cells.
Keywords: cancer stem-like cells, epidermal growth factor, basic fibroblast growth factor, epidermal growth factor receptor
Introduction
A growing body of evidence has shown that a small subpopulation of stem-like cancer cells within the tumor bed may be the major cause of tumor progression and relapse following extreme therapeutic treatment (1,2). These cells are known as cancer stem-like cells (CSCs), cancer-initiating cells or tumor-propagating cells, since they form the small group of tumor cells capable of inducing tumors and self-renewal in the same manner as normal stem cells. After first being identified in leukemia (3), CSCs have also been identified in almost all solid tumors, including brain (4), breast (5), colon (6), lung (7) and other tissues (8). These cells are regarded as being responsible for resistance to chemo- and radiotherapy, tumor angiogenesis and tumor metastasis (2). CSCs have been reported to be enriched by treatment with chemotherapeutic agents and irradiation (9,10). Therefore, targeting CSCs may be a useful therapeutic strategy for reducing the risk of tumor relapse following therapy. However, a critical approach to eradicate these cells has not been developed.
CSCs also persist in established cell lines. In serum-free media with growth factors (GFs), such as epidermal growth factor (EGF), basic fibroblast growth factor (bFGF) and platelet-derived growth factor (PDGF), the CSC population may be enriched from serum-cultured cells as a side population or sphere type (11,12). The standard culture condition for maintaining CSCs in vitro uses media (DMEM/F12 with B27 and/or N2 supplements and growth factors such as EGF and bFGF) in the absence of serum, although the subtle ingredients vary depending on the cell type. EGF signaling has been reported to act through the EGF receptor (EGFR) and is crucial for the maintenance of stemness in glioma stem cells (13). In contrast, bFGF is known to be a major mitogen for neural stem cells (NSCs) (14). In the C6 glioma cell line model, both PDGF and bFGF are required to increase the side population and form tumor spheres (15). However, glioblastoma stem cells may be grown without the exogenous addition of GFs by autocrine factors (16,17).
In the present study, we aimed to determine whether CSCs from established cell lines may be enriched as tumor spheres more efficiently without the GFs, EGF and bFGF, as compared with the GFs.
Materials and methods
Reagents
Gefitinib and AG1478 were obtained from Calbiochem (La Jolla, CA, USA). Antibodies against EGFR were obtained from Cell Signaling Biotechnology (Denvers, MA, USA). Neutralizing antibodies against EGF and bFGF were purchased from Upstate (Lake Placid, NY, USA). Recombinant EGF and bFGF were obtained from Peprotech (Rocky Hill, NJ, USA).
Cell culture and sphere-forming assay
Human breast cancer cell line (MCF7), glioma cell line (U87) and non-small cell lung cancer cell line (A549) were cultured in Dulbecco's modified Eagle's medium (DMEM) and RPMI-1640, respectively, supplemented with 10% fetal bovine serum (FBS; JR Scientific, Inc., Woodland, CA, USA) and 1% penicillin/streptomycin and were maintained at 37˚C in a 5% CO2 incubator.
For the sphere-forming assay, a single cell suspension from trypsinization was cultured in ultra low cluster 6-well plates (Corning, Corning, NY, USA) with DMEM/F12 (Cellgro, Manassas, VA, USA) in the presence or absence of GFs (10 ng/ml each of EGF and bFGF) without serum at a density of 1×103 cells/ml. After 10 days, spheres were attached by adding FBS (10%), stained with Diff-Quick solution (Sysmex, Kobe, Japan), and counted.
Antibody array
An analysis of conditioned media (CM) using an antibody array kit was conducted. CM was collected from the MCF7 cells cultured in the presence or absence of FBS with or without GFs for 48 h and was concentrated with Centrifugal Filter Units (Millipore Corporation, Billerica, MA, USA). Five micrograms of each sample were subjected to antibody array for the detection of secreted proteins using the human angiogenesis antibody array kit (R&D Systems, Minneapolis, MN, USA). Antibody array membranes were visualized by enhanced chemiluminescence (Amersham, Arlington Heights, IL, USA) according to the manufacturer's instructions.
Western blot analysis
Cells were lysed in TNN buffer [50 mM Tris-HCl (pH 7.4), 100 mM NaCl, 5 mM EDTA, 0.5% Nonidet P-40 and protease inhibitor cocktail tablet (Roche, Indianapolis, IN, USA)] and protein content was determined by Bio-Rad protein assay (Bio-Rad, Hercules, CA, USA). An aliquot (30–50 μg protein/lane) of the total protein was separated by SDS-PAGE and electrotransferred to the nitrocellulose membrane (Millipore Corporation) for 2 h at 80 volts. The membrane was blocked with 5% skimmed milk in TBST [20 mmol/l Tris-HCl (pH 7.6), 137 mmol/l NaCl, and 0.01% Tween-20] for 1 h at room temperature followed by incubation with the primary antibody overnight at 4˚C. After extensive washing with TBST the membrane was probed with a secondary antibody conjugated with horseradish peroxidase for 1 h at room temperature. After washing five times with TBST, membranes were visualized by enhanced chemiluminescence (Amersham, Arlington Heights, IL, USA) according to the manufacturer's instructions.
Statistical analysis
Statistical analysis was performed using an independent samples t-test. Differences were considered statistically significant at p<0.05.
Results
GF-free culture is more efficient for the formation of spheres in cancer cell lines than GF-containing culture
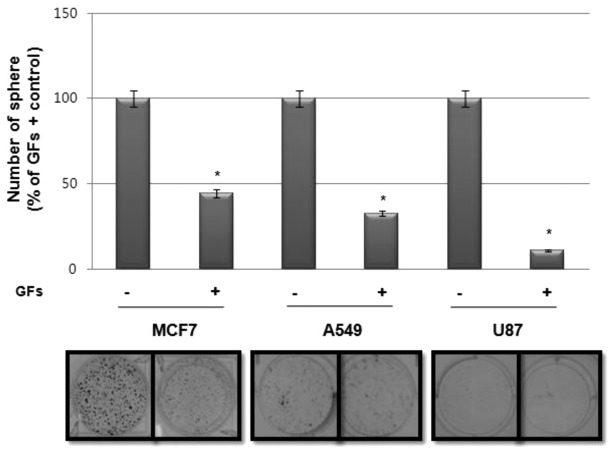
Since sphere-forming activity is regarded as a surrogate marker for the self-renewal activity of CSCs in vitro, we investigated the effect of GFs, particularly EGF and bFGF, on sphere formation in various tumor cell lines. Notably, as shown in Fig. 1, sphere formation in cancer cell lines, including MCF7, A549 and U87, was much higher in cultures without GFs than those with GFs.
Figure 1.
Effect of growth factors (GFs) on sphere-forming activity in various cancer cell lines. Sphere-forming assay of MCF7, A549 and U87 cell lines. Cells (1×103 cells/ml) were cultured in serum-free DMEM/F12 medium with GFs [10 ng/ml each of epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF)] or without GFs for 10 days and spheres were counted as described in Materials and methods. Representative images of sphere formation (lower panel) and quantification of the assay (upper panel). Data are presented as the means ± SD. *p<0.01, sphere-forming activity compared with GF-free culture.
GF-free culture enhances EGF secretion and EGFR expression in MCF7 and A549 cells
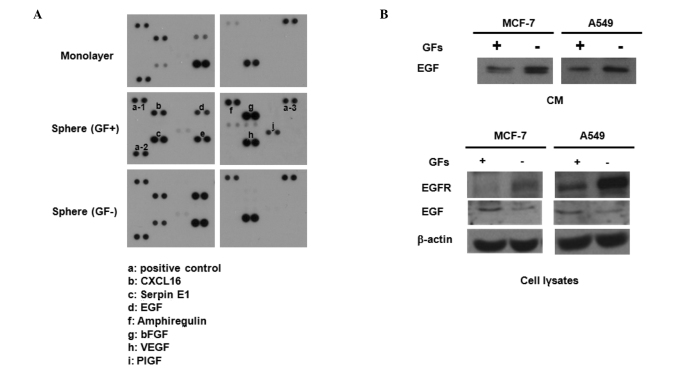
To investigate the possible mechanism involved in these events, we collected CM from monolayer cultures, GF-containing sphere cultures and GF-free sphere cultures of MCF7 cells, and then obtained blots using an antibody array kit. As shown in Fig. 2A, sphere-cultured CM revealed a high secretion of cytokines, including serpin E1, amphiregulin, EGF and placental growth factor (PIGF). Notably, EGF secretion was higher in GF-free CM than in GF-containing CM. Western blot analysis also confirmed the elevated secretion of EGF in CM from GF-free cultures compared with that of GF-containing cultures of MCF7 and A549 cell lines (Fig. 2B, upper panel). Notably, EGFR expression was also greatly enhanced in the cell lysates of GF-free cultures compared with those from GF-containing cultures of MCF7 and A549 cells (Fig. 2B, lower panel). Taken together, the higher sphere-forming activity in cell lines cultured under GF-free conditions may be due to the high secretion of EGF and expression of EGFR followed by autocrine activation of the EGF/EGFR signaling cascade.
Figure 2.
Effect of growth factors (GFs) on the secretion of epidermal growth factor (EGF) and expression of epidermal growth factor receptor (EGFR). (A) Cytokine array of conditioned media (CM) from MCF7 cells cultured with serum (upper panel), with GFs without serum (middle panel), and without GFs and serum (lower panel). (B) Western blot analysis of CM (upper panel) and cell lysates (lower panel) from MCF7 and A549 cells cultured with or without GFs. CM and cell lysates were collected after 48 h of incubation.
Blockade of EGFR with pharmacological inhibitors and neutralization of EGF suppresses sphere-forming activity in MCF7 and A549 cells
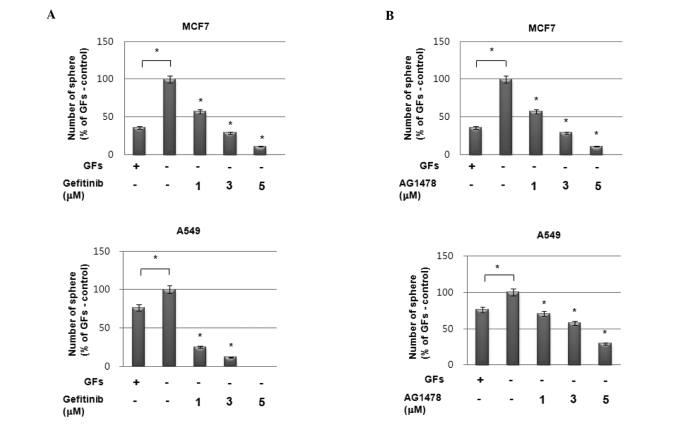
To investigate the possible role of enhanced EGF secretion followed by EGFR activation on increased sphere formation under GF-free conditions, we treated cells with the EGFR-targeted inhibitors gefitinib and AG1478. As shown in Fig. 3A and B, pretreatment with EGFR-specific inhibitors significantly suppressed sphere formation by MCF7 and A549 cells in a dose-dependent manner under GF-free sphere culture conditions. Additionally, treatment with a neutralizing antibody against EGF also demonstrated the same pattern of results as EGFR inhibitors in these cells (Fig. 3C). By contrast, treatment with a neutralizing antibody against bFGF even enhanced sphere formation in these cells (Fig. 3D). Therefore, these data strongly suggest that the high sphere-forming activity of the cell lines under GF-free culture conditions is caused by the enhanced activation of the EGF/EGFR signaling cascade.
Figure 3.
Role of epidermal growth factor (EGF)/epidermal growth factor receptor (EGFR) signaling in sphere-forming activity of MCF7 cells. (A and B) Sphere-forming assay of MCF7 cells (upper panels) and A549 (lower panels) with or without GFs in the presence or absence of EGFR inhibitors (A) gefitinib and (B) AG1478. (C and D) Sphere-forming activity of MCF7 cells (upper panels) and A549 cells (lower panels) without GFs with or without treatment with neutralizing antibodies anti-EGF (C) and anti-bFGF (D). Data are presented as the means ± SD. *p<0.01, sphere-forming activity compared with untreated control.
bFGF suppresses EGFR expression in MCF7 and A549 cells
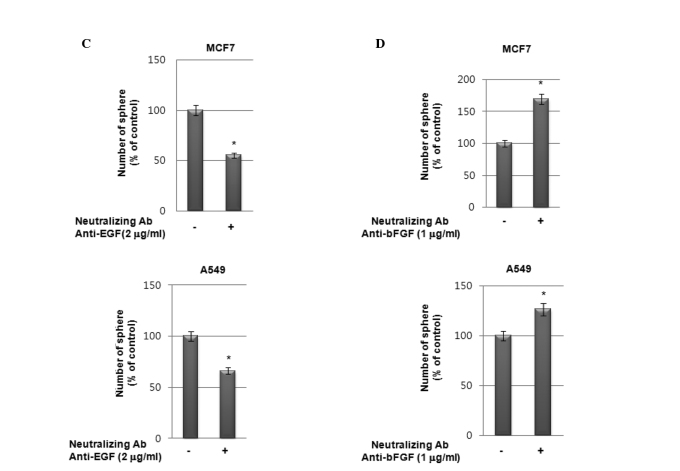
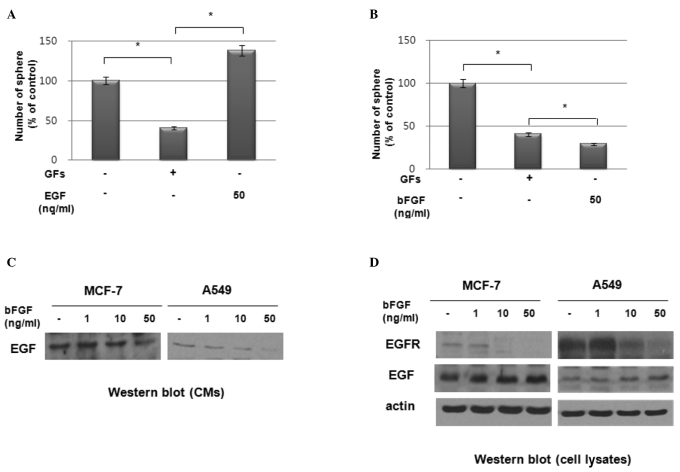
We investigated the effect of exogenous EGF and bFGF on sphere formation by these cell lines. As shown in Fig. 4A, the exogenous addition of EGF stimulated sphere formation in a dose-dependent manner in MCF7 cells. However, the addition of exogenous bFGF suppressed sphere formation in these cells (Fig. 4B). To determine the possible role of bFGF in suppressing sphere formation in these cell lines, we treated MCF7 and A549 cells with bFGF and examined the expression of EGF and EGFR. Notably, western blot analysis revealed that bFGF inhibited the secretion of EGF (Fig. 4C) and the expression of EGFR (Fig. 4D) in these cells. By contrast, EGF expression in cell lysates was increased by bFGF treatment in a dose-dependent manner (Fig. 4D). These data therefore suggest that the increased sphere formation under GF-free culture conditions was due to the elimination of the effect of bFGF on the suppression of EGFR expression under these culture conditions.
Figure 4.
Effect of epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF) on sphere-forming activity in A549 cells. (A and B) Sphere-forming activity in A549 cells with or without treatment with EGF (50 ng/ml, A) and bFGF (50 ng/ml, B). Data are presented as the means ± SD (*p<0.01). (C and D) Western blot analysis of conditioned media (CM) (C) and cell lysates (D) from A549 cells treated with bFGF at the concentrations indicated.
Discussion
Stem-like cancer cells are believed to be preserved in culture with serum, and transition of these cultures into serum-free media with GFs such as EGF and bFGF enriches these cells. In this study, however, we found that the sphere formation by cancer cells in CSC media was much higher under GF-free culture conditions than under GF-containing culture conditions. The increased sphere formation of GF-free cultured cells may be due to the increased autocrine secretion of EGF and the enhanced expression of EGFR compared with the GF-containing cultured cells.
It has been reported that EGF-mediated EGFR signaling, but not bFGF-mediated signaling, is crucial for the maintenance of brain tumor stem cells (13). Flow cytometry has shown that the CD133-positive population increased by EGF in a concentration-dependent manner (13). In a similar manner, Kelly et al indicated that in a model of brain tumor stem cell culture without the exogenous addition of GFs, the blockade of EGFR signaling reduced exogenous mitogen-independent sphere formation (16). Our data also suggest that EGF/EGFR signaling is critical for the maintenance of CSCs in tumor cells. Since the addition of EGF further increased sphere formation, we speculated that bFGF suppresses EGF expression followed by secretion outside of the cells. bFGF was found to suppress the secretion of EGF in epithelial tumor cell lines. Of note, bFGF also inhibited the expression of EGFR in a dose-dependent manner. Thus, the increase in CSCs from epithelial tumors through sphere cultures is much more efficient under GF-free culture conditions due to the inhibitory effect of bFGF on EGF secretion and EGFR expression.
In conclusion, our data suggested that CSCs from cell lines are enriched more efficiently without GFs than with GFs, since the addition of bFGF suppressed EGF/EGFR signaling. The results also showed that the autocrine secretion of EGF is more prominent in GF-free CM than in GF-containing CM. The blockade of EGF action with neutralizing antibody EGF, or the inhibition of EGFR by pharmacological inhibitors significantly reduced tumor sphere formation, whereas bFGF neutralizing antibody enhanced sphere formation. The exogenous addition of EGF further stimulated sphere formation, whereas bFGF suppressed this event. bFGF also suppressed EGFR expression and EGF secretion. Our findings suggest the that autocrine secretion of GFs, including EGF, may sustain CSCs effectively, and that bFGF may have a negative effect on tumor sphere formation by suppressing EGFR expression in sphere culture conditions. Therefore, GF-free culture promotes tumor sphere formation more than GF-containing culture in epithelial tumor cell lines. However, the critical mechanism of action of bFGF should be defined through further investigation.
Acknowledgements
This study was supported by a grant from the Nuclear Research and Development Program of the Korea Science and Engineering Foundation funded by the Korean government (MEST).
References
- 1.Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–111. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- 2.Jordan CT, Guzman ML, Noble M. Cancer stem cells. N Engl J Med. 2006;355:1253–1261. doi: 10.1056/NEJMra061808. [DOI] [PubMed] [Google Scholar]
- 3.Lapidot T, Sirard C, Vormoor J, Murdoch B, Hong T, Caceres-Cortes J, Minden M, Paterson B, Caligiuri MA, Dick JE. A cell initiating human acute myeloid leukemia after transplantation into SCID mice. Nature. 1994;367:645–648. doi: 10.1038/367645a0. [DOI] [PubMed] [Google Scholar]
- 4.Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, Henkelman RM, Cusimano MD, Dirks PB. Identification of human brain tumour initiating cells. Nature. 2004;432:396–401. doi: 10.1038/nature03128. [DOI] [PubMed] [Google Scholar]
- 5.Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumorigenic breast cancer cells. Proc Natl Acad Sci USA. 2003;100:3983–3988. doi: 10.1073/pnas.0530291100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ricci-Vitiani L, Lombardi DG, Pilozzi E, Biffoni M, Todaro M, Peschle C, De Maria R. Identification and expansion of human colon-cancer-initiating cells. Nature. 2007;445:111–115. doi: 10.1038/nature05384. [DOI] [PubMed] [Google Scholar]
- 7.Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni A, Di Virgilio A, Conticello C, Ruco L, Peschle C, De Maria R. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ. 2008;15:504–514. doi: 10.1038/sj.cdd.4402283. [DOI] [PubMed] [Google Scholar]
- 8.Visvader JE, Lindeman GJ. Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer. 2008;8:755–768. doi: 10.1038/nrc2499. [DOI] [PubMed] [Google Scholar]
- 9.Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, Dewhirst MW, Bigner DD, Rich JN. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature. 2006;444:756–760. doi: 10.1038/nature05236. [DOI] [PubMed] [Google Scholar]
- 10.Lee HE, Kim JH, Kim YJ, Choi SY, Kim SW, Kang E, Chung IY, Kim IA, Kim EJ, Choi Y, Ryu HS, Park SY. An increase in cancer stem cell population after primary systemic therapy is a poor prognostic factor in breast cancer. Br J Cancer. 2011;104:1730–1738. doi: 10.1038/bjc.2011.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Setoguchi T, Taga T, Kondo T. Cancer stem cells persist in many cancer cell lines. Cell Cycle. 2004;3:414–415. doi: 10.4161/cc.3.4.799. [DOI] [PubMed] [Google Scholar]
- 12.Patrawala L, Calhoun T, Schneider-Broussard R, Zhou J, Claypool K, Tang DG. Side population is enriched in tumorigenic, stem-like cancer cells, whereas ABCG2+ and ABCG2- cancer cells are similarly tumorigenic. Cancer Res. 2005;65:6207–6219. doi: 10.1158/0008-5472.CAN-05-0592. [DOI] [PubMed] [Google Scholar]
- 13.Soeda A, Inagaki A, Oka N, Ikegame Y, Aoki H, Yoshimura S, Nakashima S, Kunisada T, Iwama T. Epidermal growth factor plays a crucial role in mitogenic regulation of human brain tumor stem cells. J Biol Chem. 2008;283:10958–10966. doi: 10.1074/jbc.M704205200. [DOI] [PubMed] [Google Scholar]
- 14.Kitchens DL, Snyder EY, Gottlieb DI. FGF and EGF are mitogens for immortalized neural progenitors. J Neurobiol. 1994;25:797–807. doi: 10.1002/neu.480250705. [DOI] [PubMed] [Google Scholar]
- 15.Kondo T, Setoguchi T, Taga T. Persistence of a small subpopulation of cancer stem-like cells in the C6 glioma cell line. Proc Natl Acad Sci USA. 2004;101:781–786. doi: 10.1073/pnas.0307618100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kelly JJ, Stechishin O, Chojnacki A, Lun X, Sun B, Senger DL, Forsyth P, Auer RN, Dunn JF, Cairncross JG, Parney IF, Weiss S. Proliferation of human glioblastoma stem cells occurs independently of exogenous mitogens. Stem Cells. 2009;27:1722–1733. doi: 10.1002/stem.98. [DOI] [PubMed] [Google Scholar]
- 17.Li G, Chen Z, Hu YD, Wei H, Li D, Ji H, Wang DL. Autocrine factors sustain glioblastoma stem cell self-renewal. Oncol Rep. 2009;21:419–424. [PubMed] [Google Scholar]