Abstract
Low folate intake is associated with vascular disease. Causality has been attributed to hyperhomocysteinemia. However, human intervention trials have failed to show the benefit of homocysteine-lowering therapies. Alternatively, low folate may promote vascular disease by deregulating DNA methylation. We investigated whether folate could alter DNA methylation and atherosclerosis in ApoE null mice. Mice were fed one of six diets (n = 20 per group) for 16 weeks. Basal diets were either control (C; 4% lard) or high fat (HF; 21% lard and cholesterol, 0.15%) with different B-vitamin compositions: (1) folic acid and B-vitamin replete, (2) folic acid deficient (−F), (3) folic acid, B6 and B12 deficient (−F−B). −F diets decreased plasma (up to 85%; P < 0.05), whole blood (up to 70%; P < 0.05), and liver folate (up to 65%; P < 0.05) and hepatic SAM/SAH (up to 80%; P < 0.05). −F−B diets reduced plasma (up to 76%; P < 0.05), whole blood (up to 72%; P < 0.05), and liver B12 (up to 39%; P < 0.05) and hepatic SAM/SAH (up to 90%; P < 0.05). −F increased homocysteine 2-fold, while −F−B increased homocysteine 3.6- and 6.8-fold in the C and HF groups (P < 0.05). Plaque formation was increased 2-fold (P < 0.0001) in mice fed a HF diet. Feeding a HF–F diet increased lesion formation by 17% (P < 0.05). There was no change in 5-methyldeoxycytidine in liver or vascular tissue (aorta, periadventitial tissue and heart). These data suggest that atherogenesis is not associated with genome-wide epigenetic changes in this animal model.
Keywords: Folic acid and B-vitamin deficiency in ApoE null mice, Atherosclerosis, Aortic plaques, Hyperhomocysteinemia, Genome-wide DNA methylation, Vascular tissue
Introduction
Suboptimal B-vitamin intake is strongly associated with predisposition to vascular disease in humans (Boushey et al. 1995; Verhaar et al. 2002; McNulty et al. 2008). Low intracellular folate and vitamin B12 induce hyperhomocysteinemia by retarding methionine remethylation. Not only is homocysteine cardiotoxic, it causes endoplasmic reticular stress, lipid oxidation and impaired thrombolysis and vasodilation (Werstruck et al. 2001; Castro et al. 2006). Additionally, it induces vascular smooth muscle cell (SMC) proliferation and monocyte chemotaxis (Splaver et al. 2004). Most human observational studies support a positive association between homocysteine and heart disease (Splaver et al. 2004; Homocysteine Studies Collaboration 2002). One early meta-analysis of studies reported a significant increase in CVD risk (60% for men and 80% for women) for each 5-μmol/L increase in circulating homocysteine (Boushey et al. 1995). Subsequent meta-examination of prospective studies described a reduced risk of ischemic heart disease (11%) and stroke (19%) associated with a 25% drop in plasma homocysteine (Homocysteine Studies Collaboration 2002). In contrast, recent large-scale intervention trials have failed to show any benefit of homocysteine-lowering therapies against death from cardiovascular causes in patients either with pre-existing vascular disease or diabetes despite baseline homocysteine predicting subsequent vascular events (Homocysteine Studies Collaboration 2002; HOPE 2 2006; Wierzbicki 2007). Consequently, there is considerable controversy as to whether homocysteine is causal for vascular disease (Splaver et al. 2004). An alternative viewpoint is that homocysteine serves merely as a functional indicator of low folate status. Two prospective studies of men without CVD observed that high serum folate was associated with a significantly reduced risk of subsequent coronary events (Voutilainen et al. 2000, 2004). Homocysteine was not associated with risk (Voutilainen et al. 2004). Moreover, intervention with folic acid positively influences surrogate endpoints of vascular function (abnormal exercise ECG tests and flow-mediated dilatation) in patients with CVD, supporting an independent role for folate in vascular health (Vermeulen et al. 2000; Doshi et al. 2002; Woo et al. 2002).
Folate is critical in generating S-adenosylmethionine (SAM) for DNA methylation. Genes methylated at specific sites (e.g. upstream of a promoter) are either not transcribed or transcribed at a reduced rate. Epigenetic DNA methylation therefore contributes to the control of gene and ultimately protein expression (Costello and Plass 2001). Folate deficiency, by inhibiting normal DNA methylation, may disrupt this function.
It is accepted that aberrant DNA methylation is involved in human cancer. The role of abnormal DNA methylation in the pathogenesis of vascular disease is largely unknown. Altered DNA methylation, as a consequence of suboptimal folate status, may be a common mechanism linking malignant transformation and vascular dysfunction. The atherosclerotic plaque and the tumour are both monoclonal in origin with aberrant DNA methylation and the expression of genes regulating cell proliferation providing a growth advantage for selected clones. During atherogenesis, SMCs transform from a contractile to a synthetic phenotype and migrate and proliferate into the intima. Over-expression of growth factors and pro-proliferative oncogenes such as c-myc and p53 drive SMC invasiveness. In addition, over-expression of genes regulating lipid metabolism, connective tissue formation, calcification and inflammation accelerates plaque progression (Dong et al. 2002; Zaina et al. 2005; Turunen et al. 2009).
Determining whether abnormal DNA methylation causes plaque development in vivo is extremely difficult due to the intimate metabolic relationship between folate, homocysteine and cytosine methylation. We have recently developed a transgenic ApoE knockout mouse model demonstrating either mild or moderate hyperhomocysteinemia. Here, we describe how folate deficiency affects genomic DNA methylation in vascular tissue and aortic plaque formation in these mice, largely independently of increasing plasma homocysteine.
Methods
Animals and diets
Male ApoE-/- mice (n = 120, aged 4 weeks), congenic with C57BL/6J mice, were obtained from Charles River, UK (Margate, UK) and housed individually on grid floors to prevent coprophagy. All the mice were fed standard laboratory chow ad libitum for 1 week acclimatisation before being assigned (by weight) to experimental diets (n = 20 animals per group). Six diets were prepared based on AIN 93M recommendations but with different levels of fat/cholesterol and B vitamins. Two basal diets were designated either as control (C; 4% w/w lard) or high fat (HF; 21% w/w lard and cholesterol, 0.15% w/w). Different B-vitamin compositions were applied to each of these diets: (1) folic acid and B-vitamin replete, (2) folic acid deficient (−F) or (3) folic acid, B6 and B12 deficient (−F−B). Dietary composition is shown in Tables 1 and 2. Diets were prepared by Special Diets Services (UK). Powdered diet was reconstituted by adding glycerol (10% w/w) and distilled H2O (36% w/w control-fat diet or 18% w/w high-fat diet), frozen, lyophilised and stored at −20°C. Mice were fed ad libitum and allowed free access to water throughout the experiment. Animals were weighed three times weekly. Food intake was monitored at weeks 1, 8 and 16. All procedures were carried out in accordance with the requirements of UK Animals (Scientific Procedures) Act 1986.
Table 1.
Composition of experimental diets (grams per kilogram)
| Diet | Control fat (C) | High fat (HF) |
|---|---|---|
| Casein | 140 | 140 |
| Cellulose | 50 | 50 |
| Corn starch | 466 | 296 |
| Cholesterol | 0 | 1.5 |
| Choline bitartrate | 2.5 | 2.5 |
| Cystine | 1.8 | 1.8 |
| Lard | 0 | 170 |
| Maltodextrin | 155 | 155 |
| Sucrose | 100 | 100 |
| Soya bean oil | 40 | 40 |
| TBHQ antioxidant | 0.008 | 0.008 |
| Mineral mix (AIN-93M-Mx) | 35 | 35 |
| Vitamin mix (AIN-93-VX) | 10 | 10 |
Table 2.
Composition of vitamin mix (grams per kilogram)
| C/HF | −F | −F−B | |
|---|---|---|---|
| Folic acid | 0.2 | 0 | 0 |
| Vitamin B6 | 0.7 | 0.7 | 0 |
| Vitamin B12 | 2.5 | 2.5 | 0 |
Blood and tissue collection
After 16 weeks on experimental diet, mice (fasted for 4–6 h) were killed by i.p. injection of a lethal dose of sodium pentobarbital and exsanguination under terminal anaesthesia. Whole blood was immediately collected from the vena cava into a heparinised syringe for analysis of folate and for plasma preparation (see below). Residual blood was flushed from the tissues by injection of DMEM (5 ml) into the left ventricle of the heart. The liver was collected and snap frozen for analysis of folate, B12 and DNA methylation or in perchloric acid (PCA) for analyses of SAM and S-Adenosyl-L-homocysteine (SAH) (Duthie et al. 2010). The aorta and heart were removed carefully from the chest, as described previously, and placed in a Petri dish containing DMEM (Beattie et al. 2009). The aorta was separated from the heart, flushed with DMEM using a 25-gauge needle and syringe and microdissected (using a dissecting microscope) of all tunica adventitia and adipose tissue. The aorta was cut at the interface between the thoracic arch and abdominal section, and the arch was placed in 10% formalin. The abdominal section of the aorta, the adventitia and a small portion of heart were snap frozen in liquid nitrogen, for DNA methylation analysis. After 24 h, the fixed aortic arches were removed from formalin, blotted dry, snap frozen and stored at −80°C for plaque analysis.
Atherosclerotic plaque analysis
Atherosclerotic lesions in the aortic arch were measured as described in detail previously (Beattie et al. 2009). Aortic lesions are quantified by staining plaques in situ with the neutral lipid-targeting lysochrome Oil Red O (ORO; Sigma, Poole, UK) that targets fatty deposits in the arterial plaques and solubilising and quantifying spectrophotometrically the dye retained by the aorta (Beattie et al. 2009). Solubilised ORO as a biomarker of atherosclerotic plaque volume was corrected for tissue weight.
B vitamin, total plasma homocysteine and SAM/SAH status
Whole blood (100 μl) was incubated with ascorbic acid (0.9 ml, 1% w/v) for 1.5 h in the dark, snap frozen in liquid nitrogen and stored at −80°C for folate and B12 analysis (Duthie et al. 2010). The remaining blood was centrifuged at 2,400×g for 15 min at 4°C, and the plasma aliquoted, snap frozen and stored as above. Whole blood, plasma folate and plasma B12 were determined by radioassay (Duthie et al. 2010). Plasma homocysteine was measured by gas chromatography (Duthie et al. 2010). Liver SAM and SAH were measured in PCA-treated samples by HPLC (Wang et al. 2001).
Genome-wide DNA methylation in vascular tissue and liver
DNA was isolated from mouse aorta, adventitia, heart and liver using a Nucleospin C&T kit (Abgene Ltd, Epsom, UK). 5-Methyldeoxycytosine levels were quantified by LC MS/MS (Duthie et al. 2010; Friso et al. 2002).
Statistical analysis
Data are presented as means ± SEM with the number of animals in parenthesis. Data were analysed by two-way ANOVA for the effect of fat intake, vitamin composition and interaction between these factors. Significant differences between all groups were detected using the least significant difference (LSD) post-hoc method for P < 0.05. Data not sharing superscript letters differ significantly. Analyses were carried out using SPSS (version 17).
Results
B vitamin and methyl donor status
Mice fed the HF Westernised diet ate approximately 30% less than animals fed the C fat diet (data not shown). Folate deficiency alone had no effect on body weight in either the C or HF groups. Conversely, animals fed the −F−B-deficient diet gained body weight at a slower rate compared with animals fed a −F diet (P < 0.05; Fig. 1). This effect was exacerbated by HF (P < 0.05; Fig. 1). No behavioural or pathological abnormalities were observed in any of the animals (data not shown).
Fig. 1.
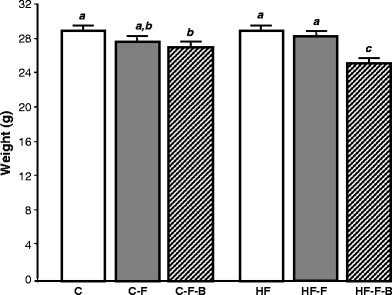
The effect of folic acid, B vitamins and fat intake on growth. Results are mean body weight ± SEM after 16 weeks on experimental diet for n = 17–20 animals per group. Data not sharing superscript letters differ by P < 0.05
Plasma, whole blood and liver folate concentrations were profoundly dependent on dietary folic acid and were influenced to a limited extent by fat intake (Table 3). Plasma (but not whole blood or liver) folate was significantly higher in mice fed the HF vitamin-replete diet for 16 weeks compared with animals fed C fat (P < 0.05; Table 3). Plasma folate declined 82% and 85% in mice fed the C−F and HF−F diets, respectively (P < 0.05; Table 3); whole blood folate decreased approx. 67% and 70% (P < 0.05; Table 3), and liver folate was depleted by 55% and 64% in these groups (Table 3). Feeding a −F−B diet did not deplete plasma, whole blood or liver folate more than folate deficiency alone (Table 3).
Table 3.
Indices of methyl donor status in blood and tissues from ApoE mice fed a control (C) or high fat (HF) diet depleted of folic acid (F−) or folic acid and vitamins B6 and B12 (F−B−) for 16 weeks
| Control fat | High fat | |||||
|---|---|---|---|---|---|---|
| C | C−F | C−F−B | HF | HF−F | HF−F−B | |
| Plasma folate (ng/ml) | 87.8 ± 4.8a | 16.2 ± 1.5b | 14.9 ± 0.9b | 104.9 ± 5.5c | 15.5 ± 1.7b | 15.0 ± 1.4b |
| Whole blood folate (ng/ml) | 498.9 ± 42.9a | 163.5 ± 8.9b | 184.7 ± 6.8b | 507.9 ± 26.0a | 149.6 ± 9.7b | 193.3 ± 18.7b |
| Liver folate (ng/mg protein) | 106.0 ± 8.0a | 47.1 ± 2.8b | 50.2 ± 3.6b | 117.1 ± 8.3a | 42.0 ± 2.3b | 41.1 ± 1.6b |
| Plasma B12 (ng/mg) | 24.8 ± 2.3a | 15.3 ± 1.0b | 6.8 ± 0.1c | 25.6 ± 3.0a | 12.5 ± 0.5d | 6.1 ± 0.5c |
| Whole blood B12 (ng/ml) | 22.5 ± 1.0a,c | 22.0 ± 0.6a,c | 7.2 ± 0.7b | 24.9 ± 1.6a | 20.5 ± 1.1c | 6.9 ± 1.0b |
| Liver B12 (ng/g protein) | 1.49 ± 0.05a | 1.43 ± 0.11a | 0.96 ± 0.04b | 1.38 ± 0.09a | 1.18 ± 0.09c | 0.84 ± 0.04b |
| Liver SAM (nmol/g tissue) | 100.2 ± 9.2a | 50.7 ± 4.0b | 43.7 ± 2.6c | 84.1 ± 4.7d | 47.6 ± 6.7b | 36.1 ± 1.8c |
| Liver SAH (nmol/g tissue) | 50.4 ± 2.3a | 71.6 ± 3.1b | 86.1 ± 4.3c | 47.6 ± 3.1a | 68.1 ± 3.8b | 89.7 ± 4.4c |
Values are means ± SEM for n = 10 mice sampled per group. Data not sharing superscript letters differ by P < 0.05
Circulating B12 was affected differentially by −F or −F−B deficiency. Plasma B12 was depleted by 73% in the C group and by 76% in the HF group by feeding a −F−B diet (P < 0.0001; Table 3). Feeding a −F diet alone depleted plasma B12 38% and 51% respectively in these groups (P < 0.05; Table 3). HF exacerbated the effect of −F and −F−B deficiency on plasma B12 (P < 0.0001; Table 3). Feeding a −F diet alone did not influence whole blood B12 in mice fed C fat but did induce a significant drop in B12 in animals fed a HF diet (P < 0.05; Table 3). Feeding a −F−B diet decreased liver B12 by 35% and 39% in the C and HF groups, respectively (P < 0.001; Table 3). Feeding a −F−B diet induced a more severe hepatic B12 depletion irrespective of fat intake (P > 0.05; Table 3).
The ratio of hepatic SAM to SAH was significantly decreased in mice fed a −F diet for 16 weeks (approx. 75% in the C fat group and 80% in the HF group; P < 0.0001; Table 3). Combined −F−B deficiency reduced the SAM/SAH ratio even further (P < 0.05) (Fig. 2).
Fig. 2.
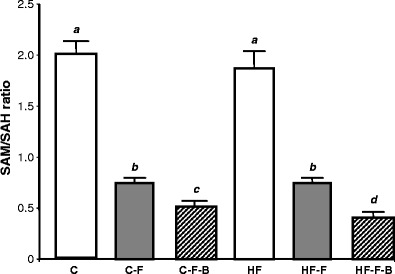
The effect of folic acid, B vitamins and fat intake on the ratio of hepatic SAM to SAH. Results are means ± SEM for n = 10 animals per group. Data not sharing superscript letters differ by P < 0.05
Folate deficiency increased plasma homocysteine 2.3-fold in the C−F and 2.1-fold in the HF−F diet. Feeding a combined −F−B diet induced hyperhomocysteinemia to a greater extent (3.6- and 6.8-fold in the C and HF groups, respectively). This augmentation was significantly dependent on fat intake (P < 0.05 for all data; Fig. 3).
Fig. 3.
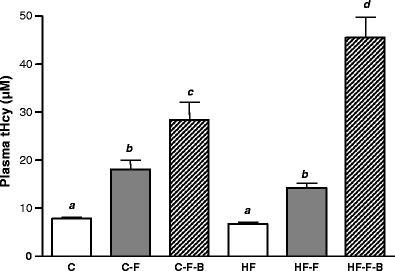
The effect of folic acid, B vitamins and fat intake on plasma homocysteine. Results are means ± SEM for n = 10 animals per group. Data not sharing superscript letters differ by P < 0.05
Plaque formation
Plaque development was increased (approximately 2.0-fold; P < 0.0001) in mice fed a Westernised HF diet compared with animals fed a C fat diet (Fig. 4). Folate deficiency increased lesion formation in the HF−F group (17%; P < 0.05). Feeding on a HF−F−B diet did not accelerate plaque formation further (Fig. 4).
Fig. 4.
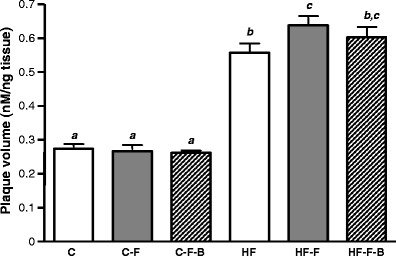
The effect of folic acid, B vitamins and fat intake on aortic plaque development. Results are means ± SEM for n = 20 animals per group. Data not sharing superscript letters differ by P < 0.05
DNA methylation
Genome-wide DNA methylation was measured in the cleaned aorta, tunica adventitia, heart and liver from −F and −F−B-deficient animals fed a C or HF diet. 5-methyldeoxycytidine concentrations and percentage methylated DNA (calculated as a percentage of total cytosines in the DNA) were similar for all treatment groups (P > 0.05; Table 4).
Table 4.
Global DNA methylation in tissues from ApoE mice fed a control (C) or high fat (HF) diet depleted of folic acid (−F) or folic acid and vitamins B6 and B12 (−F−B) for 16 weeks
| Global DNA methylation | Control fat | High fat | |||||
|---|---|---|---|---|---|---|---|
| C | C−F | C−F−B | HF | HF−F | HF−F−B | ||
| Aorta | 5-methyldeoxycytidine (ng/μg DNA) | 125.5 ± 8.2 (6) | 135.7 ± 7.7 (6) | 134.4 ± 22. (6) | 116.6 ± 11.5 (6) | 123.7 ± 7.8 (6) | 133.2 ± 15.7 (6) |
| DNA methylation (%) | 3.72 ± 0.06 (6) | 3.61 ± 0.05 (6) | 3.62 ± 0.07 (6) | 3.60 ± 0.05 (6) | 3.57 ± 0.04 (6) | 3.58 ± 0.07 (6) | |
| Heart | 5-methyldeoxycytidine (ng/μg DNA) | 158.9 ± 10.9 (6) | 135.3 ± 4.8 (6) | 130.8 ± 3.7 (6) | 134.7 ± 6.1 (6) | 157.0 ± 13.4 (6) | 148.8 ± 9.8 (6) |
| DNA methylation (%) | 3.8 ± 0.08 (6) | 3.68 ± 0.11 (6) | 3.75 ± 0.04 (6) | 3.79 ± 0.05 (6) | 3.85 ± 0.12 (6) | 3.81 ± 0.05 (6) | |
| Aorta tunica adventitia | 5-methyldeoxycytidine (ng/μg DNA) | 137.9 ± 12.2 (9) | 141.0 ± 8.6 (10) | 184.9 ± 22.3 (10) | 171.5 ± 13.0 (8) | 149.7 ± 20.1 (9) | 157.8 ± 16.2 (7) |
| DNA methylation (%) | 4.05 ± 0.11 (10) | 3.94 ± 0.15 (10) | 4.25 ± 0.10 (10) | 4.31 ± 0.12 (8) | 3.99 ± 0.10 (9) | 3.98 ± 0.13 (7) | |
| Liver | 5-methyldeoxycytidine (ng/μg DNA) | 68.1 ± 5.5 (10) | 66.6 ± 4.7 (10) | 71.4 ± 5.3 (10) | 71.5 ± 5.9 (10) | 68.4 ± 6.5 (10) | 67.6 ± 6.6 (10) |
| DNA methylation (%) | 3.19 ± 0.04 (10) | 3.20 ± 0.04 (10) | 3.21 ± 0.05 (10) | 3.21 ± 0.05 (10) | 3.20 ± 0.06 (10) | 3.21 ± 0.05 (10) | |
Values are means ± SEM with the number of mice sampled per group in parentheses
Discussion
Low folate intake and low folate blood concentrations are associated with an increased risk of vascular disease (Boushey et al. 1995; Verhaar et al. 2002; McNulty et al. 2008). Historically, causality has been attributed to high circulating homocysteine, which is cardiotoxic and deregulates vascular cell integrity and function (Werstruck et al. 2001; Castro et al. 2006). Low folate may also promote or accelerate vascular disease by deregulating DNA methylation, gene expression and cellular metabolism (Dong et al. 2002; Zaina et al. 2005; Turunen et al. 2009). 5-methyltetrahydrofolate remethylates homocysteine to methionine, which is further metabolised to S-adenosylmethionine (SAM), the principal 1-C donor in the methylation of cytosine. Under conditions of low dietary folate, SAM is depleted causing hypomethylation of DNA and abnormal gene expression. Aberrant cytosine methylation and proto-oncogene activation may be causal in vascular dysfunction.
The primary aims of this study were to determine whether folate status, independently of homocysteine, would increase vascular disease in vivo and to further establish whether abnormal genome-wide DNA methylation in the vasculature was causal. We developed a nutritional model of mild and moderate hyperhomocysteinemia in order to try to determine the differential effects of folate deficiency and homocysteine on plaque formation. Feeding ApoE null mice a −F diet for 16 weeks caused moderate intracellular folate deficiency. As expected, blood and cellular B12 were significantly decreased in animals fed the combined −F−B-deficient diet. Additionally, folate deficiency alone induced a significant reduction in B12 status (approx. 40–50% depending on fat intake; Table 3). Prolonged folate deficiency has been found previously to induce serum B12 depletion in rats (Hsu et al. 1962). This appears to be due to altered B12 transport, metabolism and utilisation, rather than via impaired intestinal absorption (Hsu et al. 1962). Folate deficiency may similarly disrupt B12 absorption, metabolism and excretion in humans (Van Royen et al. 1978; Cattan et al. 1982; Dawson et al. 1984).
A similar pattern of effect was measured in hepatic SAM and SAH in response to −F or −F−B-vitamin depletion. However, the extent of hyperhomocysteinemia in response either to −F or −F−B deficiency was different.
Critically, plaque volume was associated primarily with folic acid deficiency and not with circulating plasma homocysteine concentrations. Folate deficiency alone significantly increased plaques in ApoE null mice fed a HF−F diet. While plaque volume was higher in mice fed a HF−F−B diet compared with mice fed HF, this was not significantly different. Mice on this diet grew more slowly (14%) than mice fed the HF−F diet. This may have retarded plaque progression in this group. Atherosclerotic plaque volume was also measured in mice fed either a C−F or a C−F−B diet to establish whether these treatments could influence early plaque formation. Plaque volume was similar in all groups fed the low fat diet.
In light of the diverse effect that diet had on absolute circulating homocysteine concentrations in this study, increased plaque formation is probably not primarily caused by elevated homocysteine. The effect of hyperhomocysteinemia on aortic plaque formation in rodents has been investigated previously. However, the data are highly inconsistent, and the magnitude of hyperhomocysteinemia is profoundly dependent on the dietary regime (Hoffman et al. 2001; Zhou et al. 2001, 2003). Homocysteine increased 19-fold (from 2.5 μM to 47.0 μM) in ApoE null mice fed a combined high methionine (HM) and −F−B diet for 8 weeks (Hoffman et al. 2001). This was associated with a 2-fold increase in the atherosclerotic plaque area. Critically, plaque development was unchanged in mice fed a −F−B diet containing adequate methionine, and homocysteine concentrations were surprisingly comparable between treatment and control animals (2.7 μM versus 2.5 μM). Similar results were described for ApoE null mice fed either methionine or homocysteine (Zhou et al. 2001). Feeding animals a HM diet for 10 weeks increased plaques (approx. 2-fold) but without inducing hyperhomocysteinemia (5.1 μM versus 4.6 μM in chow fed mice; Troen et al. 2003). Conversely, plaque area was unchanged in mice fed diets that did induce hyperhomocysteinemia (Troen et al. 2003). Total plasma homocysteine was increased 17-fold (to 86.7 μM) in mice fed a HM−F−B diet and 48-fold (to 243.7 μm) in animals fed only a −F−B vitamin-deficient diet (Troen et al. 2003). Why homocysteine increased so markedly in animals fed a −F−B vitamin-depleted diet relative to animals fed a combined HF−F−B diet remains unexplained. Moreover, feeding ApoE mice a HM diet has been shown to induce hyperhomocysteinemia (4-fold and 6-fold after 4 or 18 weeks, respectively) and plaque formation (60% after 18 weeks) in subsequent studies (Zhou et al. 2004). While data describing the effects of homocysteine and plaque formation are highly variable and contradictory, in general, hyperhomocysteinemia is associated with an increase in aortic plaque area, but B-vitamin deficiency is not (Hoffman et al. 2001; Zhou et al. 2001, 2003; Troen et al. 2003). In contrast, we have shown in this study that low folate, and not primarily plasma homocysteine concentration, is linked with atherogenesis in ApoE null mice fed a HF diet. We believe that the magnitude of hyperhomocysteinemia induced here may be more physiologically relevant to what occurs in humans than described previously. However, it should be noted that tissue homocysteine concentrations were not measured, and the potential detrimental effect of homocysteine accumulation in the vasculature cannot be excluded.
We went on to determine whether aortic plaque formation, in response to folate or B-vitamin depletion, was associated with changes in key intermediates in the methionine cycle and genome-wide DNA methylation in the vascular tissue of these mice. Genomic cytosine hypomethylation is observed in cultured vascular cells and diseased vascular tissue. 5-methylcytosine levels drop 65% in rabbit aorta SMC explants, while genomic DNA is hypomethylated in advanced lesions from ApoE knockout mice, denuded New Zealand White rabbit aorta and human atherosclerotic lesions compared with normal arteries or arteries with fatty streaks (Hiltunen et al. 2002). In this study, global DNA methylation in the liver, aorta, tunica adventitia and heart tissue was measured by LC MS/MS. Perivascular adventitial adipose is a metabolically active tissue that secretes vasoresponsive factors and regulates vascular function (Lohn et al. 2002). Growth factors produced by rat perivascular adipose tissue stimulate human vascular SMC growth in culture (Barandier et al. 2005). We found a weak positive association (P = 0.08) between plasma folate and liver DNA methylation irrespective of treatment. However, despite a considerable reduction in blood and tissue folate and B12 and a significant decrease in the ratio of hepatic SAM to SAH, neither folate nor B-vitamin depletion significantly altered genome-wide cytosine methylation either in the liver or in any of the vascular tissues examined. Fat intake and homocysteine may influence DNA methylation. Cytosine DNA methylation was reported to be lower in atherosclerotic lesions from ApoE null mice fed a HF Westernised diet compared with mice fed regular chow (Lund et al. 2004). Maternal hypercholesterolemia accelerates cardiovascular disease in the offspring of rodents and rabbits (reviewed in Turunen et al. 2009), although the mechanism regulating risk remains to be established. In our study, genomic DNA methylation in the vasculature of ApoE null mice was unaffected by feeding on saturated fat and cholesterol from weaning through to adulthood. Elevated homocysteine is associated with DNA hypomethylation in some MTHFR-deficient mice tissue, but interestingly, not in the vasculature (Chen et al. 2005). Genomic DNA is also hypomethylated in blood cells from hyperhomocysteinemic patients compared with matched controls (Yi et al. 2000; Ingrosso et al. 2003). Currently, there is no data that folate itself independently alters global cytosine methylation in relation to atherosclerosis.
Only genome-wide DNA methylation was measured in this study. Locus-specific DNA hypomethylation may promote atherogenicity by upregulating genes involved in SMC proliferation, lipid deposition and inflammation. Gene-specific hypomethylation in human atherosclerotic lesions is associated with increased expression of PDGF, 15-lipoxygenase, IFN-g, MMP-2, MMP-7, TIMP-3, p53 and EC-SOD (Hiltunen and Yla-Herttuala 2003). Moreover, altered DNA methylation and dysregulated gene expression in vascular endothelial cells (rather than SMC) may be critical in maintaining vascular function (Matouk and Marsden 2008). It remains to be established whether folate deficiency alone influences gene-specific cytosine methylation in vascular tissue in vivo.
As described earlier, folate status is independently linked to risk of CVD. Despite this, several recent large-scale placebo-controlled and randomised folic acid and B-vitamin human intervention studies (calculated to lower plasma homocysteine) have failed to demonstrate significant benefit (Homocysteine Studies Collaboration 2002; Splaver et al. 2004; HOPE 2 2006). If aberrant DNA methylation is causal in deregulating vascular function, supplementing with B vitamins would equally be expected to positively influence CVD outcomes in these trials. However, these studies were designed to investigate the effects of homocysteine lowering in people with pre-existing vascular disease, diabetes, stroke or renal failure. It remains to be established whether B-vitamin supplementation may be effective in primary prevention.
In conclusion, moderate but prolonged folate deficiency significantly increased plaque formation in the aorta of ApoE null mice fed a high fat diet, indicating a positive role for folate in modulating vascular disease in vivo. This was not associated with changes in intermediates in the methionine cycle or with global DNA methylation status in the vasculature or liver of these animals. Precisely how folate maintains vascular function remains to be established. Our findings do not support the theory that genome-wide epigenetic changes in the vasculature are causal for plaque development.
Acknowledgements
The Scottish Government Rural and Environment Research and Analysis Directorate (RERAD) funded this work.
Conflicts of interest
None.
References
- Barandier C, Montani J-P, Yang Z. Mature adipocytes and perivascular adipose tissue stimulate vascular smooth muscle cell proliferation: effects of ageing and obesity. Am J Physiol Heart Circ Physiol. 2005;289:H1807–H1813. doi: 10.1152/ajpheart.01259.2004. [DOI] [PubMed] [Google Scholar]
- Beattie JH, Duthie SJ, Kwun I-S, Ha T-Y, Gordon M-J. Rapid quantification of aortic lesions in ApoE -/- mice. J Vasc Res. 2009;46:347–352. doi: 10.1159/000189795. [DOI] [PubMed] [Google Scholar]
- Boushey CJ, Beresford SAA, Omenn GS, Motulsky AGA. A quantitative assessment of plasma homocysteine as a risk factor for vascular disease: probable benefits of increasing folic acid intakes. JAMA. 1995;274:1049–1057. doi: 10.1001/jama.274.13.1049. [DOI] [PubMed] [Google Scholar]
- Castro R, Rivera I, Blom HJ, Jakobs C, Tavares de Almeida I. Homocysteine metabolism, hyperhomocysteinemia and vascular disease: an overview. J Inherit Metab Dis. 2006;29:3–20. doi: 10.1007/s10545-006-0106-5. [DOI] [PubMed] [Google Scholar]
- Cattan D, Belaiche J, Zittoun J, Yvart J. Effect of folate deficiency on vitamin B12 absorption. Ann Nutr Metab. 1982;26:367–373. doi: 10.1159/000176587. [DOI] [PubMed] [Google Scholar]
- Chen Z, Karaplis AC, Ackerman SL, Pogribny IP, Melnyk S, Lussier-Cacan S. Mice deficient in methylenetetrahydrofolate reductase exhibit hyperhomocysteinemia and decreased methylation capacity within neuropathology and aortic lipid deposition. Hum Mol Genet. 2005;10:433–443. doi: 10.1093/hmg/10.5.433. [DOI] [PubMed] [Google Scholar]
- Costello JF, Plass C. Methylation matters. J Med Genet. 2001;38:285–303. doi: 10.1136/jmg.38.5.285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawson DW, Sawers AH, Sharma RK. Malabsorption of protein bound vitamin B12. BMJ. 1984;288:675–678. doi: 10.1136/bmj.288.6418.675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong C, Yoon W, Goldschmidt-Clermont PJ. DNA methylation and atherosclerosis. J Nutr. 2002;132:2406S–2409S. doi: 10.1093/jn/132.8.2406S. [DOI] [PubMed] [Google Scholar]
- Doshi SN, McDowell IFW, Moat SJ, Payne N, Durrant HJ, Lewis M. Folic acid improves endothelial function in coronary artery disease via mechanisms largely independent of homocysteine lowering. Circulation. 2002;105:22–26. doi: 10.1161/hc0102.101388. [DOI] [PubMed] [Google Scholar]
- Duthie SJ, Grant G, Pirie LP, Watson AJ, Margison GP. Folate deficiency alters hepatic and colon MGMT and OGG-1 DNA repair protein expression in rats but has no effect on genome-wide DNA methylation. Cancer Prev Res. 2010;3:92–100. doi: 10.1158/1940-6207.CAPR-09-0231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friso S, Choi SW, Girelli D. A common mutation in the 5, 10-methylenetetrahydrofolate reductase gene affects genomic DNA methylation through an interaction with folate status. PNAS. 2002;99:5606–5611. doi: 10.1073/pnas.062066299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- The Heart Outcomes Prevention Evaluation (HOPE) 2 Investigators Homocysteine lowering with folic acid and B vitamins in vascular disease. NEJM. 2006;354:1567–1577. doi: 10.1056/NEJMoa060900. [DOI] [PubMed] [Google Scholar]
- Hiltunen MO, Yla-Herttuala S. DNA methylation, smooth muscle cells and atherogenesis. Arterioscler Thromb Vasc Biol. 2003;23:1750–1753. doi: 10.1161/01.ATV.0000092871.30563.41. [DOI] [PubMed] [Google Scholar]
- Hiltunen MO, Turunen MP, Hakkinen TP, Rutanen J, Hedman M, Makinen K. DNA hypomethylation and methyltransferase expression in atherosclerotic lesions. Vasc Med. 2002;7:5–11. doi: 10.1191/1358863x02vm418oa. [DOI] [PubMed] [Google Scholar]
- Hoffman MA, Lalla E, Lu Y, Gleason MR, Wolf BM, Tanji N. Hyperhomocysteinemia enhances vascular inflammation and accelerates atherosclerosis in a murine model. J Clin Invest. 2001;107:675–683. doi: 10.1172/JCI10588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homocysteine Studies Collaboration Homocysteine and risk of ischemic heart disease and stroke: a meta-analysis. JAMA. 2002;288:2015–2022. doi: 10.1001/jama.288.16.2015. [DOI] [PubMed] [Google Scholar]
- Hsu JM, Tantengco V, Chow BF. Effect of folic acid deficiency on absorption of Co60-labeled vitamin B12 by rats. J Clin Invest. 1962;41:532–536. doi: 10.1172/JCI104506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ingrosso D, Cimmino A, Perna AF, Masella L, Santo NG, Bonis ML. Folate treatment and unbalanced methylation and changes of allelic expression induced by hyperhomocysteinemia in patients with uraemia. Blood. 2003;361:1693–1699. doi: 10.1016/S0140-6736(03)13372-7. [DOI] [PubMed] [Google Scholar]
- Lohn M, Dubrovska G, Lauterbach B, Luft FC, Gollasch M, Sharma AM. Periadventitial fat releases a vascular relaxing factor. FASEB J. 2002;16:1057–1063. doi: 10.1096/fj.02-0024com. [DOI] [PubMed] [Google Scholar]
- Lund G, Andersson L, Lauria M, Lindholm M, Fraga MF, Villar-Garea A. DNA methylation polymorphisms precede any histological sign of atherosclerosis in mice lacking Apolipoprotein E. J Biol Chem. 2004;279:29147–29154. doi: 10.1074/jbc.M403618200. [DOI] [PubMed] [Google Scholar]
- Matouk CC, Marsden PA. Epigenetic regulation of vascular endothelial cell gene expression. Circ Res. 2008;102:873–887. doi: 10.1161/CIRCRESAHA.107.171025. [DOI] [PubMed] [Google Scholar]
- McNulty H, Pentieva K, Hoey L, Ward M. Homocysteine, B vitamins and CVD. Proc Nutr Soc. 2008;67:232–237. doi: 10.1017/S0029665108007076. [DOI] [PubMed] [Google Scholar]
- Splaver A, Lamas GA, Hennekens CH. Homocysteine and cardiovascular disease: biological mechanisms, observational epidemiology and the need for randomised trials. Am Heart J. 2004;148:34–40. doi: 10.1016/j.ahj.2004.02.004. [DOI] [PubMed] [Google Scholar]
- Troen AM, Lutgens E, Smith DE, Rosenberg IH, Selhub J. The atherogenic effect of excess methionine intake. PNAS. 2003;100:15089–15094. doi: 10.1073/pnas.2436385100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turunen MP, Aavik E, Yla-Herttula S. Epigenetics and atherosclerosis. Biochim Biophys Acta. 2009;1790:886–891. doi: 10.1016/j.bbagen.2009.02.008. [DOI] [PubMed] [Google Scholar]
- Royen E, Blockx P, Molter F. The influence of serum folate on urinary excretion of vitamin B12. Eur J Nucl Med. 1978;3:175–178. doi: 10.1007/BF00256639. [DOI] [PubMed] [Google Scholar]
- Verhaar MC, Stroes E, Rabelink TJ. Folates and cardiovascular disease. Arterioscler Thromb Vasc Biol. 2002;22:6–13. doi: 10.1161/hq0102.102190. [DOI] [PubMed] [Google Scholar]
- Vermeulen AGJ, Stehouwer CDA, Twisk JWR, Berg M, Mackaay ALC, Campen CMC. Effect of homocysteine-lowering therapy with folic acid plus vitamin B6 on progression of subclinical atherosclerosis: a randomized placebo-controlled trial. Lancet. 2000;355:517–522. doi: 10.1016/S0140-6736(99)07391-2. [DOI] [PubMed] [Google Scholar]
- Voutilainen S, Lakka TA, Porkkala-Sarataho E, Rissanen T, Kaplan GA, Salonen JT. Low serum folate concentrations are associated with an excess incidence of acute coronary events: the Kuopio Ischaemic Heart Disease Risk Factor Study. Eur J Clin Nutr. 2000;54:424–428. doi: 10.1038/sj.ejcn.1600991. [DOI] [PubMed] [Google Scholar]
- Voutilainen S, Virtanen JK, Rissanen TH, Alfthan G, Laukkanen J, Nyyssonen K. Serum folate and homocysteine and the incidence of acute coronary events: the Kuopio Ischaemic Heart Disease Risk Factor Study. Am J Clin Nutr. 2004;80:317–323. doi: 10.1093/ajcn/80.2.317. [DOI] [PubMed] [Google Scholar]
- Wang W, Kramer PM, Yang S, Pereira MA, Tao L. Reversed-phase high performance liquid chromatography procedure for the simultaneous determination of S-adenosyl-L-methionine and S-adenosyl-L-homocysteine in mouse liver and the effect of methionine on their concentrations. J Chromatogr B. 2001;762:59–65. doi: 10.1016/S0378-4347(01)00341-3. [DOI] [PubMed] [Google Scholar]
- Werstruck GH, Lentz RS, Dayal S, Hossain GS, Sood SK, Shi YY. Homocysteine-induced endoplasmic reticulum stress causes dysregulation of the cholesterol and triglyceride biosynthetic pathways. J Clin Invest. 2001;107:1263–1273. doi: 10.1172/JCI11596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wierzbicki AS. Homocysteine and cardiovascular disease: a review of the evidence. Diab Vasc Dis Res. 2007;4:143–149. doi: 10.3132/dvdr.2007.033. [DOI] [PubMed] [Google Scholar]
- Woo KS, Chook P, Chan LL, Cheung AS, Fung WH, Qiao M. Long term improvement in homocysteine levels and arterial endothelial function after 1-year folic acid supplementation. Am J Med. 2002;112:535–539. doi: 10.1016/S0002-9343(02)01075-6. [DOI] [PubMed] [Google Scholar]
- Yi P, Melnyk S, Pogribna M, Pogribny IP, Hine RJ, James SJ. Increase in plasma homocysteine associated with parallel increase in plasma S-adenosyl homocysteine and lymphocyte DNA hypomethylation. J Biol Chem. 2000;275:29318–29323. doi: 10.1074/jbc.M002725200. [DOI] [PubMed] [Google Scholar]
- Zaina S, Lindholm MW, Lund G. Nutrition and aberrant DNA methylation patterns in atherosclerosis: more than just hyperhomocysteinemia? J Nutr. 2005;153:5–8. doi: 10.1093/jn/135.1.5. [DOI] [PubMed] [Google Scholar]
- Zhou J, Moller J, Danielsen CC, Bentzon J, Ravn HB, Austin RC. Dietary supplementation with methionine and homocysteine promotes early atherosclerosis but not plaque rupture in ApoE-deficient mice. Arterioscler Thromb Vasc Biol. 2001;21:1470–1476. doi: 10.1161/hq0901.096582. [DOI] [PubMed] [Google Scholar]
- Zhou J, Moller J, Ritskes-Hoitinga M, Larsen ML, Austin RC, Falk E. Effects of vitamin supplementation and hyperhomocysteinemia on atherosclerosis in ApoE-deficient mice. Atherosclerosis. 2003;168:255–262. doi: 10.1016/S0021-9150(03)00138-2. [DOI] [PubMed] [Google Scholar]
- Zhou J, Werstruck GH, Lhotak S, Koning L, Sood SK, Hossain GS. Association of multiple cellular stress pathways with accelerated atherosclerosis in hyperhomocysteinemic apolipoprotein E-deficient mice. Circulation. 2004;110:207–213. doi: 10.1161/01.CIR.0000134487.51510.97. [DOI] [PubMed] [Google Scholar]


