Abstract
Reactive oxygen species (ROS) are well known to contribute to the pathophysiology of Parkinson’s disease (PD). Clinical trials of antioxidants are currently underway in PD patients, however, antioxidant research has been hindered by a lack of peripheral biomarkers.
METHODS
Twenty-two patients with PD elected to have a novel antioxidant assessment (Functional Intracellular Assay (FIA), Spectra Cell Lab, Houston, TX) performed between 2004–2008. Each PD case was compared to four age- and gender- matched controls (n=88) in four separate, random iterations using laboratory data submitted during the same time period. Logistic regression was used to determine the odds of functional deficiency in antioxidant nutrients (i.e., glutathione, coenzyme Q10, selenium, vitamin E and alpha-lpoic acid) by case-control status. The proportion of cases with functional deficiency was also compared to that for controls by chi2 test.
RESULTS
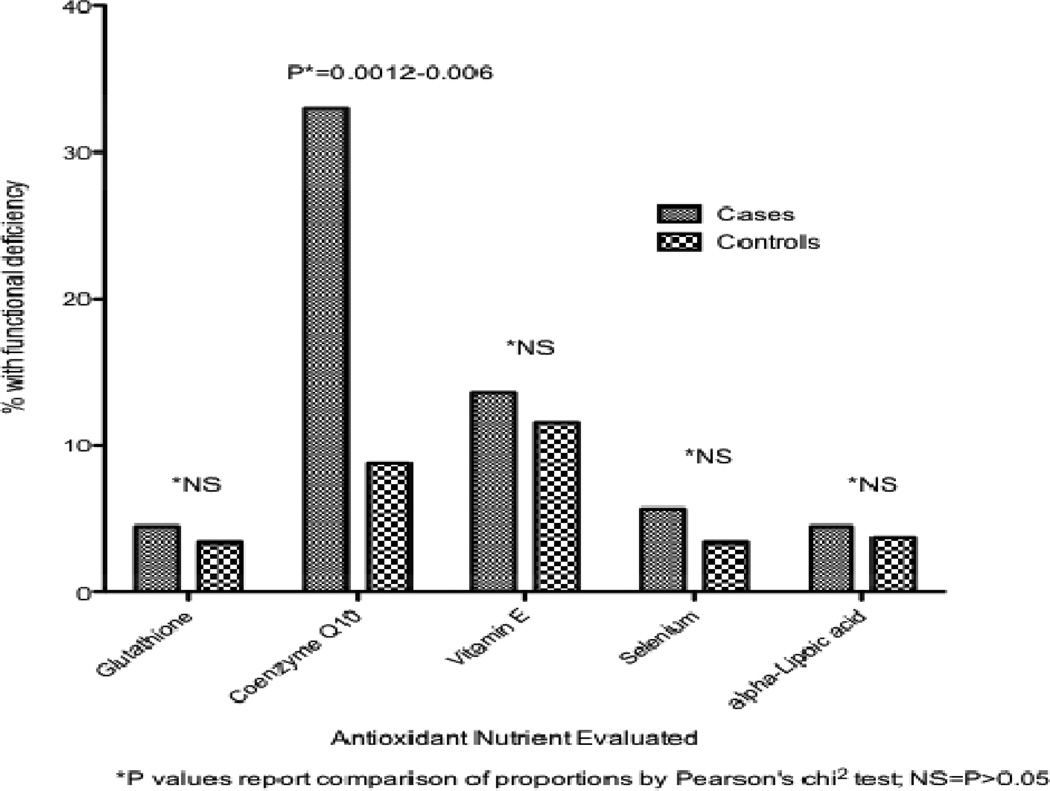
Compared to cases, PD patients had a significantly greater odds of deficiency in coenzyme Q10 status (OR: 4.7–5.4; 95% CI: 1.5–17.7; P = 0.003–0.009) based on FIA results, but not of vitamin E, selenium, lipoic acid, or glutathione (all P>0.05). The proportion of cases with coenzyme Q10 deficiency was also significantly greater in cases than in controls (32–36% vs. 8–9%; P=0.0012–0.006).
CONCLUSIONS
Deficiency of coenzyme Q10 assessed via FIA should be explored as a potential peripheral biomarker of antioxidant status in PD.
Background and Significance
Excessive production of ROS is a hallmark of disease progression in PD pathophysiology(1, 2), however attempts at antioxidant intervention strategies have been largely unsuccessful (3–5). Intervention studies using antioxidants have been hindered by the lack of established biomarkers for antioxidant status in PD (6). Identification of biomarkers of antioxidant depletion has the potential to identify subgroups of patients with deficiency, as well as monitor change in antioxidant status associated with repletion. Most attempts to identify perturbations in peripheral levels of antioxidants fail to show an association between PD and antioxidant status, and those that do show associations are not clinically available(7–11).
Coenzyme Q10 is an especially relevant antioxidant in PD research, because it also facilitates function of the mitochondrial transport chain (MTC). In vitro (12) and in vivo studies (13, 14) suggest a defect of mitochondrial complex 1 activity in PD, resulting in disruption of redox equilibrium. While exogenously administered CoQ10 has been shown to restore electron transport chain activity in in vitro studies using fibroblasts from PD patients (15), cultured fibroblasts from PD patients and controls were shown to have similar CoQ10 levels(16). Evidence of CoQ10 deficiency has been reported in tissue homogenates of the cerebral cortex, but not in the substantia nigra, striatum, or cerebellum (17).
As a lipophilic antioxidant, CoQ10 is capable of scavenging radicals within membranes and in the cytosol and plasma when bound to lipoproteins(8). Its ubiquitous presence throughout cytosol, membranes, and plasma earned it the name ubiquinone (L- ubique, ‘everywhere’). Preliminary data from a phase I study suggested that that exogenously administered CoQ10 may retard disease progression in PD(18). Platelet CoQ10 redox ratios have been shown to be significantly decreased in PD patients(8), but this test has not been developed for clinical use. A clinically available peripheral biomarker capable of identifying diminished coenzyme Q10 activity has the potential to accelerate research and enhance clinical outcomes in PD.
Materials and Methods
Patients with PD were identified upon presentation to a private outpatient, integrative medicine clinic that specializes in complementary and alternative medicine (CAM) neurology. A total of 26 patients with a diagnosis of PD elected to have their blood analyzed for FIA status. The study was approved by the Institutional Review Board (IRB) of Bastyr University, Kenmore, WA and conducted at a private practice naturopathic medical clinic in Seattle, WA (Seattle Integrative Medicine). The charts of these 26 individuals were reviewed and case report forms (CRFs) were completed to de-identify the subjects.
The functional assessment of intracellular deficiencies of micronutrients was performed by the lymphocyte transformation assay developed by Shive et al(19–24). In this procedure, blood is collected in two 10ml cell preparation tubes (Becton Dickinson) containing sodium citrate and ficoll hypaque. Following overnight shipment to the laboratory in Houston, TX, the blood specimens were centrifuged and the peripheral blood mononuclear cells were isolated. The cells were washed to remove platelets and other cells, and the final preparation is diluted to a concentration of 150,000 cells/ ml. Aliquots of the cell suspension (25,000 cells) are added to individual wells of a 96 well microtiter plate containing multiple variations of the Clayton Foundation Biochemical Institute (CFBI) 1000 media.
The culture media is chemically defined, serum-free, and contains optimal concentrations of each essential nutrient required for short-term growth of lymphocytes in culture. Individual variations of the growth medium include growth assays in media in which each micronutrient is deleted, one at a time. Growth rates were assessed after stimulating the lymphocyte proliferation by the addition of phytohemagglutinin (PHA).
Cells were then incubated at 37 degrees C in a 5% CO2 incubator and tritiated thymidine (H3) is added after three days of incubation. Cell cultures were grown for an additional 24 hours. At the conclusion of the incubation period, the cells from each well of the microtiter plate were harvested using a Perkin Elmer Filter Mate cell harvester, retaining H3 labeled cells on a filter mat. Proliferation or growth was measured by determining the radioactive content of each well using a Perkin Elmer NXT Scintillation and Luminescence Counter. Each analyte was determined in triplicate and growth rates were reported as a percentage of growth compared to the growth rate in optimal media.
Individual antioxidants (vitamins E, glutathione, selenium and lipoic acid) were measured by pre-incubation of aliquots of lymphocytes in a solution of individual antioxidants for 24 hours. For Coenzyme Q10, the lymphocytes were incubated in a solution of ubiquinone-10 at a concentration of 1 microgram/ml for 72 hours. The lymphocytes were retested by the antioxidant function test to determine if additional individual antioxidant to the cells improved the antioxidant function. If improvements are determined, the lymphocytes were deficient in the individual antioxidants associated with pre-incubation media. The results are reported as a percentage of growth compared to cells in pre-incubation media.
Reference ranges (mean and standard deviation (SD)) for individual micronutrients were established by testing a minimum of 500 healthy subjects. All values outside of 3 SD are deleted from the data and the mean value and standard deviation determined. A test result lower that the mean value minus 2 SD were considered to be deficient.
Control data were provided from the 34,522 available samples submitted to Spectra Cell after providing them with age and gender characteristics of the case sample; the laboratory was not provided with data on the frequency of functional deficiency in the case cohort, nor were they aware of the main study objectives. The laboratory selected control samples per case, matched for age and gender (i.e., because loss of coenzyme Q10 may be a normal consequence of aging (25)). Although the laboratory provided a data set that included approximately 4 controls per case, in four cases the laboratory failed to provide sufficient numbers of controls for each case. Therefore, the total cases for which chart data was abstracted (n=26) was reduced to twenty-two (n=22) for the final analysis in order to maintain a 4:1 control:case ratio. Because there was more than one case that could have been excluded to meet the age- and gender- matching criteria (e.g., there were two cases aged 64 of male gender that could have been excluded to age and gender match the control data provided by the laboratory), we performed four random iterations of exclusions, analyzed each iteration separately, and report the results of all iterations below. For each iteration, the ratio of controls was confirmed to be four to one per case, and the age and gender distributions were confirmed to be identical. Analyses were repeated including all original cases (n=26) and all original controls (n=92) to ensure that elimination of random cases to correct the matching ratio did not impact the overall results of the analyses.
Both comparisons of odds of deficiency and the proportion deficient for each cohort were compared for case versus controls. For logistic regression, a binary variable was created to denote nutrient deficiency, and the odds of deficiency for each nutrient was calculated for cases compared to controls. For comparisons of proportions, the proportion deficient in cases and controls was also calculated and compared using Pearson’s chi2 test of proportion Analyses were repeated for each of the four random iterations of case/control matching performed. P-values <0.05 were considered statistically significant for this pilot study. All analyses were performed using Stata 11.2 (StataCorp LP; College Station, TX).
Results
The results of both analyses were consistent. The proportions of deficient in each nutrient were not significantly different for cases compared to controls for all antioxidant nutrients, except for coenzyme Q10, which ranged from 32–36% in cases compared to 8–9% in controls (P=0.0012–0.006) (Figure 1).
Figure 1.
Frequency of Antioxidant Deficiency by Case-Control Status
The odds of deficiency status were also comparable amongst cases and controls, except for coenzyme Q10, in which Parkinson’s disease cases showed significantly greater odds of functional deficiency (Table 1).
Table 1.
Odds of Antioxidant Nutrient Deficiency in Parkinson’s Disease Cases
| Nutrient | Odds Ratio | 95% CI | P value* |
|---|---|---|---|
| Glutathione (GSH) | |||
| Iteration 1 | 1.3 | 0.13, 13.6 | 0.80 |
| Iteration 2 | 1.3 | 0.13, 13.6 | 0.80 |
| Iteration 3 | 1.3 | 0.13, 13.6 | 0.80 |
| Iteration 4 | 1.3 | 0.13, 13.6 | 0.80 |
| Coenzyme Q10 | |||
| Iteration 1 | 5.4 | 1.7, 17.6 | 0.005 |
| Iteration 2 | 4.7 | 1.5, 14.8 | 0.009 |
| Iteration 3 | 5.7 | 1.8, 17.7 | 0.003 |
| Iteration 4 | 4.7 | 1.5, 14.8 | 0.009 |
| Vitamin E | |||
| Iteration 1 | 1.1 | 0.28, 4.4 | 0.80 |
| Iteration 2 | 1.1 | 0.28, 4.4 | 0.90 |
| Iteration 3 | 1.1 | 0.28, 4.4 | 0.89 |
| Iteration 4 | 1.1 | 0.28, 4.4 | 0.89 |
| Selenium | |||
| Iteration 1 | 1.3 | 0.13, 13.6 | 0.80 |
| Iteration 2 | 1.3 | 0.13, 13.6 | 0.80 |
| Iteration 3 | 2.8 | 0.44, 18.1 | 0.27 |
| Iteration 4 | 1.3 | 0.13, 13.6 | 0.80 |
| Alpha-lipoic Acid | |||
| Iteration 1 | 4.1 | 0.25, 69.0 | 0.32 |
| Iteration 2 | 2.0 | 0.18, 23.7 | 0.57 |
| Iteration 3 | 2.0 | 0.18, 23.7 | 0.57 |
| Iteration 4 | 2.0 | 0.18, 23.7 | 0.57 |
For point estimate in odds
Repeating the analyses using all original cases and controls had limited impact on the results overall, and did not effect interpretation of the results, i.e., ORCoQ10 = 5.6; 95% CI: 1.9–16.5, P=0.002 with 34.6% of cases deficient compared to 7.7% of controls, P=0.0009.
Study Limitations
The samples submitted to Spectra Cell, used for controls, do not likely reflect a healthy population (selection patient bias). The test is not typically covered by insurance and incurs an out-of-pocket expense (~$160). Thus, the controls provided by the lab are presumably comprised of individuals who can afford the test and are motivated to invest in their health, i.e. symptoms, disease, or family history of disease (self-selection bias). One would expect a small percentage of these samples to include individuals with PD; population studies suggest that the prevalence of PD is approximately 1%. Presence of illness is likely to result in a greater incidence of deficiency compared to controls and it is possible that the data would be stronger if PD patients were compared to healthy controls.
All of the PD data is based on individuals living in the Pacific Northwest, whereas controls are from a nationwide sample. Several patients in the PD group were taking supplements, including coenzyme Q10. No conclusions about the impact of supplementation can be drawn given the small sample size and the variations in supplement brands and doses used.
Regarding laboratory methods, Q10 is incubated differently than the other antioxidants evaluated in this study. Future studies should explore whether varying the incubation technique alters the test results, and these values should be compared to serum levels of reduced/oxidized Q10.
Discussion
The nature of the Spectra Cell FIA methodology is unique in that it is a functional test. The test demonstrates that ex vivo, the cells of those affected with PD were more functional with exogenous supplementation of coenzyme Q10 than without it. This test may be of clinical utility in identifying a subgroup of patients with PD who may benefit from Q10 supplementation. Q10 supplementation is a popular complementary medicine therapy among individuals with PD following preliminary research from UCLA suggesting that supplementation in high doses slows PD progression.(26)
It is likely that Q10 status influences disease states other than PD. Unsuccessful attempts have been made to correlate a serum level deficiency of Q10 with long-term statin therapy(27) and Huntington’s disease(28, 29), two conditions theorized to be affected by Q10 depletion; it is possible that reassessment of Q10 status using a FIA analysis may generate different results.
We propose coenzyme Q10 is a conditionally essential nutrient, required only by some individuals or under certain circumstances. It is likely that PD is a condition where the requirements of Q10 increase, making individuals, or a subset of individuals, dependent on this nutrient. These data are the first to demonstrate improved cellular function ex vivo with Q10 fortification. Follow-up studies should prospectively seek to determine whether supplementation improves FIA status in a dose-dependent fashion, and whether FIA CoQ10 status correlates with clinical disease status or progression.
Acknowledgments
Funding
NIH NCCAM/ Bernard Osher K01 Training Award provided personnel (PI). Bastyr University provided the IRB. Spectra Cell provided support for a research assistant to gather data.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosure of potential conflicts of interest:
Spectra Cell was not involved in the data analysis or manuscript preparation.
References
- 1.Chinta SJ, Andersen JK. Redox imbalance in Parkinson's disease. Biochim Biophys Acta. 2008 Nov;1780(11):1362–1367. doi: 10.1016/j.bbagen.2008.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Miller RL, James-Kracke M, Sun GY, Sun AY. Oxidative and inflammatory pathways in Parkinson's disease. Neurochem Res. 2009 Jan;34(1):55–65. doi: 10.1007/s11064-008-9656-2. [DOI] [PubMed] [Google Scholar]
- 3.Storch A, Jost WH, Vieregge P, Spiegel J, Greulich W, Durner J, et al. Randomized, double-blind, placebo-controlled trial on symptomatic effects of coenzyme Q(10) in Parkinson disease. Arch Neurol. 2007 Jul;64(7):938–944. doi: 10.1001/archneur.64.7.nct60005. [DOI] [PubMed] [Google Scholar]
- 4.Medeiros CA, Carvalhedo de Bruin PF, Lopes LA, Magalhaes MC, de Lourdes Seabra M, de Bruin VM. Effect of exogenous melatonin on sleep and motor dysfunction in Parkinson's disease. A randomized, double blind, placebo-controlled study. J Neurol. 2007 Apr;254(4):459–464. doi: 10.1007/s00415-006-0390-x. [DOI] [PubMed] [Google Scholar]
- 5.Shoulson I. DATATOP: a decade of neuroprotective inquiry. Parkinson Study Group. Deprenyl And Tocopherol Antioxidative Therapy Of Parkinsonism. Ann Neurol. 1998 Sep;44(3) Suppl 1:S160–S166. [PubMed] [Google Scholar]
- 6.O'Keeffe GC, Michell AW, Barker RA. Biomarkers in Huntington's and Parkinson's Disease. Ann N Y Acad Sci. 2009 Oct;1180:97–110. doi: 10.1111/j.1749-6632.2009.04943.x. [DOI] [PubMed] [Google Scholar]
- 7.Ebadi M, Govitrapong P, Sharma S, Muralikrishnan D, Shavali S, Pellett L, et al. Ubiquinone (coenzyme q10) and mitochondria in oxidative stress of parkinson's disease. Biol Signals Recept. 2001 May-Aug;10(3–4):224–253. doi: 10.1159/000046889. [DOI] [PubMed] [Google Scholar]
- 8.Gotz ME, Gerstner A, Harth R, Dirr A, Janetzky B, Kuhn W, et al. Altered redox state of platelet coenzyme Q10 in Parkinson's disease. J Neural Transm. 2000;107(1):41–48. doi: 10.1007/s007020050003. [DOI] [PubMed] [Google Scholar]
- 9.Jimenez-Jimenez FJ, Molina JA, de Bustos F, Garcia-Redondo A, Gomez-Escalonilla C, Martinez-Salio A, et al. Serum levels of coenzyme Q10 in patients with Parkinson's disease. J Neural Transm. 2000;107(2):177–181. doi: 10.1007/s007020050015. [DOI] [PubMed] [Google Scholar]
- 10.Lagendijk J, Ubbink JB, Vermaak WJ. Measurement of the ratio between the reduced and oxidized forms of coenzyme Q10 in human plasma as a possible marker of oxidative stress. J Lipid Res. 1996 Jan;37(1):67–75. [PubMed] [Google Scholar]
- 11.Sohmiya M, Tanaka M, Tak NW, Yanagisawa M, Tanino Y, Suzuki Y, et al. Redox status of plasma coenzyme Q10 indicates elevated systemic oxidative stress in Parkinson's disease. J Neurol Sci. 2004 Aug 30;223(2):161–166. doi: 10.1016/j.jns.2004.05.007. [DOI] [PubMed] [Google Scholar]
- 12.Tatton WG, Chalmers-Redman RM. Mitochondria in neurodegenerative apoptosis: an opportunity for therapy? Ann Neurol. 1998 Sep;44(3) Suppl 1:S134–S141. doi: 10.1002/ana.410440720. [DOI] [PubMed] [Google Scholar]
- 13.Haas RH, Nasirian F, Nakano K, Ward D, Pay M, Hill R, et al. Low platelet mitochondrial complex I and complex II/III activity in early untreated Parkinson's disease. Ann Neurol. 1995 Jun;37(6):714–722. doi: 10.1002/ana.410370604. [DOI] [PubMed] [Google Scholar]
- 14.Krige D, Carroll MT, Cooper JM, Marsden CD, Schapira AH. Platelet mitochondrial function in Parkinson's disease. The Royal Kings and Queens Parkinson Disease Research Group. Ann Neurol. 1992 Dec;32(6):782–788. doi: 10.1002/ana.410320612. [DOI] [PubMed] [Google Scholar]
- 15.Winkler-Stuck K, Wiedemann FR, Wallesch CW, Kunz WS. Effect of coenzyme Q10 on the mitochondrial function of skin fibroblasts from Parkinson patients. J Neurol Sci. 2004 May 15;220(1–2):41–48. doi: 10.1016/j.jns.2004.02.003. [DOI] [PubMed] [Google Scholar]
- 16.del Hoyo P, Garcia-Redondo A, de Bustos F, Molina JA, Sayed Y, Alonso-Navarro H, et al. Oxidative stress in skin fibroblasts cultures from patients with Parkinson's disease. BMC Neurol. 2010;10:95. doi: 10.1186/1471-2377-10-95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hargreaves IP, Lane A, Sleiman PM. The coenzyme Q10 status of the brain regions of Parkinson's disease patients. Neurosci Lett. 2008 Dec 5;447(1):17–19. doi: 10.1016/j.neulet.2008.09.069. [DOI] [PubMed] [Google Scholar]
- 18.Shults CW, Flint Beal M, Song D, Fontaine D. Pilot trial of high dosages of coenzyme Q10 in patients with Parkinson's disease. Exp Neurol. 2004 Aug;188(2):491–494. doi: 10.1016/j.expneurol.2004.05.003. [DOI] [PubMed] [Google Scholar]
- 19.Shive W, Pinkerton F, Humphreys J, Johnson MM, Hamilton WG, Matthews KS. Development of a chemically defined serum- and protein-free medium for growth of human peripheral lymphocytes. Proc Natl Acad Sci U S A. 1986 Jan;83(1):9–13. doi: 10.1073/pnas.83.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shive W, Matthews KS. Nutritional requirements for growth of human lymphocytes. Annu Rev Nutr. 1988;8:81–97. doi: 10.1146/annurev.nu.08.070188.000501. [DOI] [PubMed] [Google Scholar]
- 21.Matthews KS, Mrowczynski E, Matthews R. Dietary deprivation of B-vitamins reflected in murine splenocyte proliferation in vitro. Biochem Biophys Res Commun. 1994 Jan 28;198(2):451–458. doi: 10.1006/bbrc.1994.1066. [DOI] [PubMed] [Google Scholar]
- 22.Boerner P. Functional Intracellular Analysis of Nutritional and Antioxidant Status. JANA. 2001;4:27–40. [Google Scholar]
- 23.Baum SJea. Health Personnel Antioxidant Study (HPAS): Effects of Antioxidant Supplementation on Functional Antioxidant Capacity. J Am Neutra Assoc. 2004;7:25–31. [Google Scholar]
- 24.Crawford JFea. Normalization of homocysteine in dialysis patients by directed repletion with apparent reduction of access thrombosis. Dialysis and Transplantation. 2001;30:512–521. [Google Scholar]
- 25.Wada H, Goto H, Hagiwara S, Yamamoto Y. Redox status of coenzyme Q10 is associated with chronological age. J Am Geriatr Soc. 2007 Jul;55(7):1141–1142. doi: 10.1111/j.1532-5415.2007.01209.x. [DOI] [PubMed] [Google Scholar]
- 26.Shults CW, Oakes D, Kieburtz K, Beal MF, Haas R, Plumb S, et al. Effects of coenzyme Q10 in early Parkinson disease: evidence of slowing of the functional decline. Arch Neurol. 2002 Oct;59(10):1541–1550. doi: 10.1001/archneur.59.10.1541. [DOI] [PubMed] [Google Scholar]
- 27.Young JMS, Florkowski C, Reinheimer A, Scott R, George P. Coenzyme Q10 levels and vascular function in patients on long-term statin therapy for familial hypercholesterolemia. Atherosclerosis Supplement. 2009;10(2):S4–S11. [Google Scholar]
- 28.del Hoyo P, Garcia-Redondo A, de Bustos F, Molina JA, Sayed Y, Alonso-Navarro H, et al. Oxidative stress in skin fibroblasts cultures of patients with Huntington's disease. Neurochem Res. 2006 Sep;31(9):1103–1109. doi: 10.1007/s11064-006-9110-2. [DOI] [PubMed] [Google Scholar]
- 29.Andrich J, Saft C, Gerlach M, Schneider B, Arz A, Kuhn W, et al. Coenzyme Q10 serum levels in Huntington's disease. J Neural Transm Suppl. 2004;(68):111–116. doi: 10.1007/978-3-7091-0579-5_13. [DOI] [PubMed] [Google Scholar]