Abstract
Stromal cells are essential components of the bone marrow (BM) microenvironment that regulate and support the survival of different tumors, including chronic lymphocytic leukemia (CLL). In this study, we investigated the role of Notch signaling in the promotion of survival and chemoresistance of human CLL cells in coculture with human BM-mesenchymal stromal cells (hBM-MSCs) of both autologous and allogeneic origin. The presence of BM-MSCs rescued CLL cells from apoptosis both spontaneously and following induction with various drugs, including Fludarabine, Cyclophosphamide, Bendamustine, Prednisone and Hydrocortisone. The treatment with a combination of anti-Notch-1, Notch-2 and Notch-4 antibodies or γ-secretase inhibitor XII (GSI XII) reverted this protective effect by day 3, even in presence of the above-mentioned drugs. Overall, our findings show that stromal cell-mediated Notch-1, Notch-2 and Notch-4 signaling has a role in CLL survival and resistance to chemotherapy. Therefore, its blocking could be an additional tool to overcome drug resistance and improve the therapeutic strategies for CLL.
Keywords: Notch signaling, human bone marrow mesenchymal stromal cells, hBM-MSCs, chronic lymphocytic leukemia, CLL, drug resistance
Introduction
Chronic lymphocytic leukemia (CLL) is the most common lymphoproliferative disorder characterized by a variable clinical course according to well-defined prognostic factors, such as mutation status of V genes, CD38 and ZAP-70 expression and specific gene profiles. CLL is characterized by the clonal expansion of mature, antigen-stimulated CD5+/CD23+ cells in blood, secondary lymphoid tissues and bone marrow (BM), in close contact with stromal microenvironment.1, 2 The complex interactions among growth factors, extracellular matrix components and stromal cells provide extrinsic signals that regulate growth, differentiation and survival of normal B-cell precursors and neoplastic B cells, such as CLL cells and B lymphoblasts.1, 2, 3, 4, 5 Stromal cell integrins are involved in regulating leukemia cell survival through the interaction with the extracellular matrix;6 when CLL cells are removed from the in vivo stromal microenvironment and placed in culture without supportive stroma, they undergo spontaneous apoptosis.7 CLL cell coculture with different adherent cell types, collectively referred to as stromal cells, leads to leukemia cell survival, migration and resistance to drugs such as Fludarabine,4, 5, 8, 9, 10, 11, 12, 13, 14 thus highlighting the importance of external signals by accessory cells. Supportive microenvironmental cells include BM-mesenchymal stromal cells (BM-MSCs),8, 10 CD68+ nurse-like cells derived from monocytes,10, 11, 12, 13 and follicular dendritic cells.14, 15 BM-MSCs are multilineage non-hematopoietic progenitor cells that have a key role in supporting lympho-hematopoiesis, and give rise to different stromal cell lineages, including BM fibroblasts, adipocytes, osteocytes and chondrocytes, as shown in vitro and partially in vivo.16, 17 Stromal cell cocultures are used to mimic the in vivo microenvironment and to develop strategies to overcome stroma-derived drug resistance.18 Previous studies reported that the adhesion to stroma of hematopoietic malignant cell lines, including myeloma, acute and chronic myeloid leukemia and CLL, confers resistance to a variety of cytotoxic drugs, as well as Fas-mediated cell death.8, 19, 20
Notch signaling is an evolutionarily conserved molecular pathway that has a pivotal role in cell fate and differentiation in many tissues during both embryonic and postnatal development.21, 22 Four mammalian Notch receptors have been identified and designated as Notch1–4.21, 22 The interactions of Notch receptors with membrane-bound ligands of Delta and Jagged families, that is, delta-like ligand (DLL)-1/-3/-4, Jagged-1 and -2, are critical for Notch signaling.21, 22 Ligand binding induces γ-secretase-mediated cleavage and translocation of Notch intracellular domain into the nucleus, where it interacts with the DNA-binding protein RBP-J to induce the expression of numerous downstream target genes, such as Hes-1 and Deltex-1.22 Jagged-1/-2 and DLL-1, commonly named as Delta/Serrate/LAG-2 (DSL) proteins, are ligands for Notch 1–4;22, 23 Delta-4 can bind and activate Notch-1 and -4 receptors,22, 23, 24 whereas Delta-3 can bind and activate Notch-1 or similar Notch receptors.22, 25
Human BM-MSCs (hBM-MSCs) express a number of Notch ligands26, 27 that are still present in MSC progeny; in addition, MSC-derived osteoblasts regulate hematopoietic stem cell niche through Jagged-1/Notch-1 signaling.27 Notch system is crucial during hematopoiesis and embryonic development27, 28 and its dysregulation is associated with several human disorders, including cancer.28, 29, 30 A pivotal role for Notch signaling has been clearly shown in promoting survival and apoptosis resistance in T/B-cell acute lymphoblastic leukemia and CLL.3, 31, 32, 33 However, a few data are available on both the expression of different Notch molecules by CLL cells33 and the link between Notch signaling and the stromal cell-mediated molecular mechanisms involved in this phenomenon. Therefore, we evaluated the contribution of different Notch molecules to survival and chemoresistance of human primary CLL cells in coculture with autologous and allogeneic BM-MSCs.
Materials and methods
Cell collection
CLL cells were obtained after informed consent from peripheral blood samples of 12 patients with newly diagnosed CLL, according to the Institutional guidelines. Cells were isolated by Ficoll–Hypaque centrifugation (Pharmacia, Uppsala, Sweden) and characterized by immunophenotyping. Most cells were CD19+/CD5+/CD23+ CLL cells, as assessed by flow cytometry (median: 97.0% range: 95.9–99.0). Human CD73+/CD45− BM-MSCs were obtained after informed consent from BM samples collected from both five healthy donors and five CLL patients (hBM-MSCs*) to carry out both allogenic and autologous coculture experiments. CLL patients characteristics are described in Supplementary Table 1. As deletions in p53 gene have been reported to increase significantly CLL cell survival as compared with CLL with intact p53 signaling, we selected all cases with wild-type p53 (including the CLL7 case, displaying del17 aberration) to avoid any additional factor that could influence the role of Notch signaling in the experiments with MSCs.
Expansion and immunophenotype characterization of MSCs
Both hBM-MSCs and hBM-MSCs* were seeded at a density of 3.1 × 104 cells/cm2 and cultured in Dulbecco's modified Eagle medium with 20% fetal bovine serum, 1% ℒ-glutamine, and penicillin–streptomycin (Gibco, Milan, Italy), at 37 °C in 5% CO2 and humidified atmosphere, as previously described.21, 29 hBM-MSCs and hBM-MSCs* at passage 2 or 3 displayed homogeneous mesenchymal immunophenotype (including CD105, CD44, CD73, CD146 marker expression) and multipotent differentiation potential (into osteoblastic, adipocytic and chondrocytic lineages), and were used for the coculture experiments.26, 31, 34
Coculture of CLL cells with MSCs
CLL cells were cultured alone or cocultured with either hBM-MSCs or hBM-MSCs* at 10/1 ratio for 3, 7 and 28 days to study the capability of MSCs of supporting CLL cell survival. The expression of Notch molecules on both cell types was evaluated at days 3 and 7; most of the other experiments were performed after 3 days of culture.
Experiments with or without inhibitors or blocking antibodies against Notch molecules were carried out in 96-well plates with a confluent monolayer of MSCs: 105 CLL cells were cultured in 200 μl RPMI 1640 medium supplemented with 10% fetal bovine serum, 1% ℒ-glutamine, and 1% penicillin–streptomycine, with or without 104 adherent hBM-MSCs or hBM-MSCs*. Media collected from the 1-day MSC culture was added to CLL cell culture to study the effects of the soluble factors produced by MSCs. To address whether these soluble factors are Notch-related, CLL cells were cocultured either with hBM-MSCs or hBM-MSCs* at 10/1 ratio in Transwell conditions for 3 days with or without either γ-secretase inhibitor GSI-XII (X-IL-CHO) or blocking antibodies against Notch molecules. Other inhibitors, such as GSI-I (Z-LLNle-CHO) and GSI-IX (DAPT) did not lead to the complete block of Notch signaling pathway and therefore were not employed (data not shown). Cocultured CLL cells were separated from hBM-MSCs or hBM-MSCs* monolayer by careful pipetting with ice-cold phosphate-buffered saline. Viable cells were identified by using the Trypan blue dye exclusion method and counted in a hemocytometer.
Coculture of CLL cells with MSCs in presence of GSI XII and chemotherapeutic agents
Stock solutions of GSI XII, Fludarabine and Cyclophosphamide were purchased from Calbiochem (La Jolla, CA, USA), and Bendamustine, Prednisone and Hydrocortisone from Sigma-Aldrich (Saint Louis, MO, USA). GSI XII and Fludarabine were resuspended in dimethyl sulfoxide as a vehicle. Cyclophosphamide, Bendamustine, prednisone and hydrocortisone were diluted in 0.9% Sodium Chloride (NaCl); all reagents were diluted to appropriate concentrations with culture medium.
GSI XII, Notch-specific neutralizing antibodies and chemotherapeutic agents were titrated in subsequent experiments to determine the lowest efficient dose leading to specific Notch inhibition and induction of apoptosis. The effective concentration killing 50% of CLL cells (EC50) derived from the equations that best fit the linear range of the dose-response curve. The specific relative sensitivity of CLL cells to GSI XII was performed by culturing alone or coculturing CLL cells with hBM-MSCs for 3 days in presence of increasing concentrations of GSI XII. The effects of increasing concentrations of GSI XII on hBM-MSCs-induced survival of CLL cells were evaluated after 3 days of co-culture at 10/1 and 1/1 ratios. CLL cells were then cultured without or with hBM-MSCs at 10/1 ratio in absence or presence of chemotherapeutic agents, and without or with blockade of Notch pathway for 3 days. Percentage of specific apoptosis was calculated according to the following formula: (test (inhibitor-induced apoptosis)–control (spontaneous apoptosis)) × 100/(100–control).35
Flow cytometric analysis of Notch molecules expression, treatment procedure with inhibitors and neutralizing anti-Notch molecule antibodies, assays with recombinant Notch ligands and interleukin (IL)-6/-7, as well as annexinV/7AAD apoptosis assay, cell cycle analysis, measurement of caspase activity, immunofluorescence staining, and western blot analysis were performed as previously described.27 Monoclonal antibodies were used to detect Jagged-2 (R&D Systems, Minneapolis, MN, USA), DLL-1 (R&D Systems), Notch-1 (BD Biosciences, San Jose, CA, USA), Bcl-2 (clone 124–Dakocytomation, Carpinteria, CA, USA), p53 (BD Biosciences), VEGFR2 (KDR) (R&D Systems), caspase-3 Cell Signaling Technology, Danvers, MA, USA, poly(ADP-ribose) polymerase (PARP) (Cell Signaling Technology), CD127 (IL-7R, Immunotech, Marseille, France), NF-kB (Cell Signaling Technology), protein 2 (c-IAP2) (R&D Systems), Jagged-1 (Cell Signaling Technology), Notch-2 (LifeSpan Biosciences, Seattle, WA, USA), Notch-3 (Abcam plc, Cambridge, MA, USA), Notch-4/DLL-3 (Santa Cruz Biotechnology, Santa Cruz, CA, USA), DLL-4 (Abcam plc), all with IgG isotype controls. Directly conjugated monoclonal antibodies anti-CD19, CD23 and CD5 were from Becton Dickinson (San Jose, CA, USA). Cells displaying Notch molecules were expressed as either percentage of positive cells, in case of bi-modal fluorescence distribution, or mean fluorescence intensity (MFI), after electronic gating of CLL cells, hBM-MSCs or hBM-MSCs*. In case of uni-modal fluorescence distribution, MFI was calculated as the geometric mean of fluorescent intensity of hBM-MSCs, hBM-MSCs* or CLL cells expressing Notch molecules.
Neutralizing antibodies were used at the following concentrations: anti-human Notch-1 (Genetex, Irvine, CA, USA), -2 and -3 (R&D Systems) 10 μg/ml; goat anti-human Notch-4 (Santa Cruz Biotechnology) 5 μg/ml; goat anti-human Jagged-1/-2, DLL-1/-3 and -4 (R&D Systems) 10 μg/ml. Blocking antibodies against Notch molecules and appropriate isotype controls were added to CLL cells or MSCs before their mixing in coculture.
Statistical analysis
Statistical analysis was performed by using one-way analysis of variance test to compare multiple groups with Holm–Sidak test used for internal comparison between multiple groups. P value ⩽0.05 was considered statistically significant. Results were expressed as the mean±SD of 12 independent experiments from different human CLL donors. All statistical calculations were performed using STATA, Version 10.0 (StataCorp, College Station, TX, USA).
Results
Expression of Notch receptors and ligands by CLL cells and MSCs in culture and coculture conditions
Flow cytometry analysis of the expression of Notch molecules by CLL cells and hBM-MSCs in culture and coculture at day 3 and 7 is shown in Table 1. Freshly isolated CLL cells expressed high levels of all Notch receptors and ligands except DLL-1, which was detected only at low levels. No significant differences in the expression of Notch receptors and ligands by CLL cells were observed according to Zap-70 expression or mutational status of V genes (data not shown). hBM-MSCs* showed the same pattern of expression as hBM-MSCs, except Notch-1 that was expressed 3.4-fold higher than hBM-MSCs at basal levels. Following CLL cell and hBM-MSC coculture (3 or 7 days), upregulation of Notch-1 and -4 by CLL cells was observed. Similarly, Notch-2 expression was induced in hBM-MSCs at day 7. By contrast, Notch-3 was significantly downregulated at day 3. Furthermore, Notch3 expression was significantly reduced in CLL cells cultured with BM-MSCs* at both timepoints examined and with hBM-MSCs for 7 days. Notch-1 expression by hBM-MSCs cultured alone was similar to that observed during the coculture, whereas Notch-4 was significantly downregulated. DLL-3 was markedly upregulated by CLL cells but not by hBM-MSCs, whereas DLL-4 was significantly downregulated following coculture for 3 days. In addition, DLL-1 expression by hBM-MSCs was not observed during coculture, and the expression of Jagged-1 and -2 did not significantly change in hBM-MSCs after 3-day coculture, as well as the expression of DLL-3 after 7-day coculture. The expression of the different Notch molecules by CLL cells in coculture with hBM-MSCs or hBM-MSCs* is shown in Table 1.
Table 1. Notch receptor and ligand expression by CLL cells and MSCs.
|
Notch-1 |
Notch-2 |
Notch-3 |
Notch-4 |
|||||
|---|---|---|---|---|---|---|---|---|
| Day 3 | Day 7 | Day 3 | Day 7 | Day 3 | Day 7 | Day 3 | Day 7 | |
| Expression by CLL cells | ||||||||
| a. Alone | 2.6±0.8 | 1.6±0.5 | 17.6±4.3 | 40.4±8.3 | 23.1±4.6 | 28.3±5.6 | 11.1±2.3 | 6.3±1.8 |
| b.+hBM-MSCs | 3.8±1.3 | 3.7±0.9 | 21.7±5.6 | 44.1±7.1 | 18.3±5.4 | 23.1±5.2 | 21.5±4.4 | 14.8±3.3 |
| c.+hBM-MSCs* | 3.2±0.9 | 2.8±1.1 | 16.6±4.5 | 41.4±9.1 | 12.2±2.6 | 16.8±4.8 | 12.2±3.2 | 14.1±2.5 |
| a. versus b. | NS | P<0.05 | NS | NS | NS | NS | P<0.05 | P<0.05 |
| b. versus c. | NS | NS | NS | NS | NS | NS | P<0.05 | NS |
| a. versus c. | NS | NS | NS | NS | P<0.05 | P<0.05 | NS | P<0.05 |
| Expression by MSCs | ||||||||
| a. Alone | 4.0±1.5 | 3.7±1.1 | 7.1±1.8 | 7.8±1.6 | 13.1±3.1 | 8.7±2.7 | 9.9±2.1 | 9.7±2.8 |
| b.+hBM-MSCs | 3.9±1.4 | 2.6±1.3 | 5.6±1.6 | 14.5±4.3 | 5.3±2.4 | 9.9±3.9 | 5.3±1.6 | 5.5±1.2 |
| c.+hBM-MSCs* | 4.6±1.7 | 3.3±1.4 | 6.8±1.9 | 11.6±3.2 | 8.8±3.1 | 10.5±3.3 | 7.0±2.3 | 6.2±2.1 |
| a. versus b. | NS | NS | NS | P<0.05 | P<0.05 | NS | P<0.05 | P<0.05 |
| b. versus c. | NS | NS | NS | NS | NS | NS | NS | NS |
|
Jagged-1 |
Jagged-2 |
DLL-1 |
DLL-3 |
DLL-4 |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Day 3 | Day 7 | Day 3 | Day 7 | Day 3 | Day 7 | Day 3 | Day 7 | Day 3 | Day 7 | |
| Expression by CLL cells | ||||||||||
| a. Alone | 10.0±2.5 | 6.8±2.8 | 10.4±2.5 | 12.1±3.0 | 9.2±3.5 | 3.2±1.5 | 11.0±3.2 | 15.2±4.0 | 13.9±4.8 | 13.2±3.7 |
| b.+hBM-MSCs | 18.4±3.1 | 1.5±0.3 | 13.1±4.5 | 16.6±4.6 | 1.0±0.5 | 2.4±0.7 | 19.7±4.1 | 20.0±5.2 | 20.3±6.4 | 22.0 ±7.5 |
| c.+hBM-MSCs* | 7.5±2.8 | 1.3±0.1 | 6.1±2.1 | 12.7±5.7 | 3.0±0.7 | 2.6±0.9 | 20.6±4.7 | 21.0±6.8 | 20.6±5.7 | 23.0±4.9 |
| a. versus b. | P<0.05 | P<0.05 | NS | NS | P<0.05 | NS | P<0.05 | NS | NS | NS |
| b. versus c. | P<0.05 | NS | P<0.05 | NS | NS | NS | NS | NS | NS | NS |
| a. versus c. | NS | P<0.05 | P<0.05 | NS | P<0.05 | NS | P<0.05 | NS | P<0.05 | P<0.05 |
| Expression by MSCs | ||||||||||
| a. Alone | 7.4±1.6 | 7.2±1.3 | 1.5±0.0 | 1.5±0.0 | 1.9±0.7 | 1.8±0.1 | 9.7±2.3 | 9.0±3.8 | 8.5±2.3 | 8.3±2.2 |
| b.+hBM-MSCs | 4.7±1.1 | 1.7±0.6 | 1.4±0.1 | 7.5±1.2 | 1.7±0.4 | 2.0±0.3 | 5.2±1.4 | 5.3±1.6 | 4.3±1.2 | 3.4±1.0 |
| c.+hBM-MSCs* | 3.9±0.8 | 1.6±0.8 | 1.3±0.3 | 2.6±0.4 | 1.5±0.6 | 3.0±1.1 | 5.7±0.4 | 5.9±1.9 | 6.1±1.9 | 4.8±1.3 |
| a. versus b. | P<0.05 | P<0.01 | NS | P<0.01 | NS | NS | P<0.05 | NS | P<0.05 | P<0.01 |
| b. versus c. | NS | NS | NS | P<0.01 | NS | NS | NS | NS | NS | NS |
Abbreviations: CLL, chronic lymphocytic leukemia; DLL, delta-like ligand; MFI, mean fluorescence intensity; NS, not significant.
Mean values±SD of MFI (n=12); DLL. The results are expressed as mean of geometric MFI values of either CD73+CD45− hBM-MSC or CD19+CD5+CD45+ CLL cell population expressing Notch ligands and receptors. ANOVA test: *P<0.05, **P<0.01, ***P<0.001 versus IgG-treated control were considered statistically significant.
Effects of Notch pathway inhibition and drugs on CLL cell survival and MSC functions
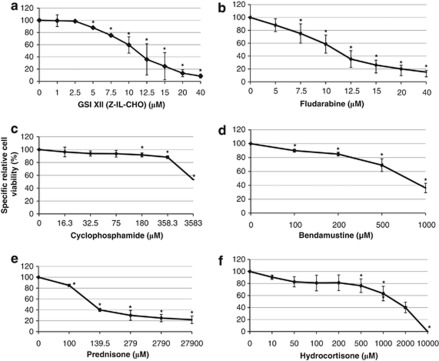
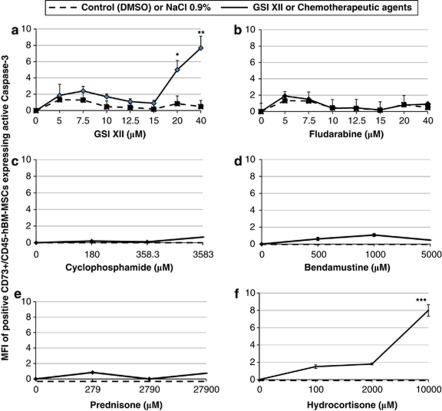
Dose-response curve showing the effects of increasing concentrations of GSI XII and drugs on CLL cells cultured alone for 1 day is shown in Figure 1. Treatment with increasing concentrations of Fludarabine, Cyclophosphamide, Bendamustine, Prednisone and Hydrocortisone resulted in a dose-dependent decrease in CLL cell viability, with EC50 >7.5, >100.0, >200.0, >100.0 and >100.0 μℳ, respectively. GSI XII did not kill CLL cells at concentrations below 2.5 μℳ; however, higher concentrations promoted specific apoptosis of CLL cells, with EC50 >10.0 μℳ. On the other hand, apoptosis or morphological changes of hBM-MSCs in culture for 3 days in presence of GSI XII, Fludarabine, Cyclophosphamide, Bendamustine, Prednisone and Hydrocortisone were observed up to 15.0, 40.0, 358.3, 500.0, 279.0 and 200.0 μℳ concentrations, respectively. Apoptosis signs or clear morphological changes were observed in hBM-MSCs treated with GSI XII and Hydrocortisone at concentrations of 40 and 10 000 μℳ, respectively (data not shown). Active caspase-3 expression was not induced in hBM-MSCs cultured alone for 3 days in presence of GSI XII, Fludarabine, Cyclophosphamide, Bendamustine, Prednisone and Hydrocortisone at concentrations up to 15.0, 40.0, 358.3, 500.0, 279.0 and 200.0 μℳ, respectively (Figure 2). As activation of caspase-3 is a hallmark of death receptor-mediated apoptosis, these results demonstrate that inhibition of signaling pathways with small-molecule inhibitors may promote death receptor-mediated apoptosis, as shown by heat-shock proteins inhibition.36 In addition, GSI XII and drugs did not significantly affect mesenchymal marker expression (data not shown).
Figure 1.
The specific relative cell viability (%) of CLL cells cultured alone with increasing concentrations of chemotherapeutic agents for 1 day. CLL cells were cultured alone with increasing concentrations of GSI XII (a), Fludarabine (b), Cyclophosphamide (c), Bendamustine (d), Prednisone (e) and Hydrocortisone (f) for 1 day. Viable CLL cells were quantified by flow cytometry as percentage of Annexin V−/7-AAD− (lower left quadrant) after electronic gating on CD5+CD19+CD45+ CLL cells. The percentage of specific viable CLL cells was calculated according to the following formula: 100-((test (inhibitor-induced apoptosis)–control (spontaneous apoptosis)) × 100/(100–control)).30 Statistical analysis was carried out by using one-way analysis of variance, Holm–Sidak test. *P<0.05 was considered statistically significant. Data were represented as mean±SD of twelve independent experiments.
Figure 2.
Effects of GSI XII, Fludarabine, Cyclophosphamide, Bendamustine, Prednisone and Hydrocortisone on the active caspase-3 expression by hBM-MSCs. MFI of active caspase-3 expression by CD73+CD45-hBM-MSCs after culture for 3 days with increasing concentrations of GSI XII (a), Fludarabine (b), Cyclophosphamide (c), Bendamustine (d), Prednisone (e) and Hydrocortisone (f). Statistical analysis was carried out by using analysis of variance test, Holm–Sidak test. Data were represented as mean±SD of 12 independent experiments. *P<0.05, **P<0.01, ***P<0.001 versus IgG control were considered statistically significant. GSI XII and Fludarabine were resuspended in dimethyl sulfoxide (DMSO). Cyclophosphamide, Bendamustine, Prednisone and Hydrocortisone were resuspended in NaCl (0.9%). DMSO and NaCl (0.9%) were used as controls for vehicle effects.
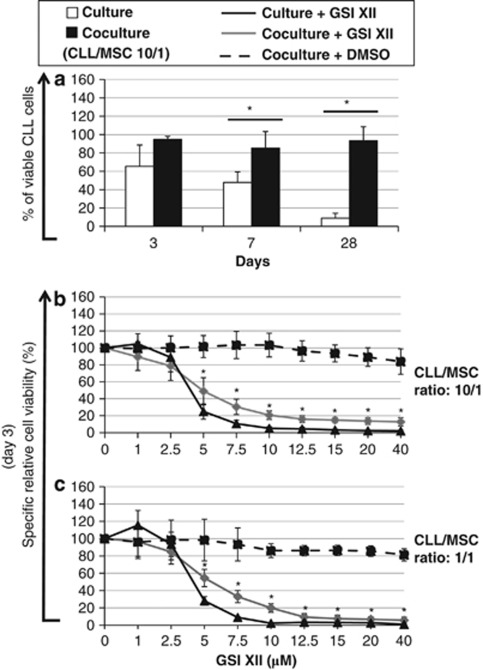
Effects of GSI XII on MSC-mediated survival of CLL cells
Coculture of CLL cells with hBM-MSCs significantly increased the number of surviving CLL cells, as compared with what obtained by culturing CLL cells alone in similar conditions7, 8, 9 (Figure 3a). At low concentrations, GSI XII had no detectable specific cytotoxic effects on CLL cells either cultured alone or cocultured for 3 days with hBM-MSCs at 10/1 and 1/1 ratios (Figures 3b and c). GSI XII was capable of significantly inhibiting specific apoptosis of CLL cells cultured alone and cocultured with hBM-MSCs (1/1 ratio) at concentrations below 2.1 μℳ and 0.8 μℳ, respectively. However, higher concentrations of GSI XII promoted specific apoptosis of CLL cells in both culture conditions. Thus, the reciprocal interactions among CLL cells via Notch signaling appear to have an important role in cell survival.
Figure 3.
Percentage of viable CLL cells and specific relative cell viability (%) of CLL cells cultured alone or cocultured with hBM-MSCs in presence of increasing concentrations of GSI XII. (a) Percentage of viable CLL cells cultured alone or cocultured with hBM-MSCs at 10/1 ratio for 3, 7 and 28 days. (b, c) Specific relative cell viability (%) of CLL cells was evaluated by culturing alone or coculturing CLL cells with hBM-MSCs at 10/1 and 1/1 ratios in presence of increasing concentrations of GSI XII for 3 days. Statistical analysis was carried out by using one-way analysis of variance, Holm–Sidak test. *P<0.05 versus control was considered statistically significant. Data were represented as the mean±SD of 12 independent experiments. GSI XII was resuspended in dimethyl sulfoxide (DMSO) and DMSO was used as control for vehicle effects.
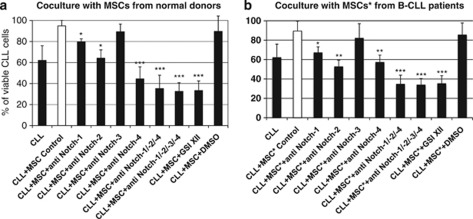
Notch-1, Notch-2 and Notch-4 are involved in MSC-mediated survival of CLL cells
A dramatic increase of the apoptotic rate of CLL cells, both in culture alone and in coculture with either hBM-MSCs or hBM-MSCs* was achieved by blocking each Notch receptor (Table 2, Figures 4a and b). Interestingly, hBM-MSCs supported CLL cell survival as efficiently as hBM-MSCs* after 3 days of coculture. Notch-1, -2 and -4 appeared to be mostly involved in both hBM-MSCs and hBM-MSCs*-mediated CLL cell survival, as previously shown,33, 37 whereas Notch-3 had only a low impact in the same conditions. Blockade of Notch-2 significantly increased the percentage of apoptotic CLL cells in coculture with hBM-MSCs*, whereas blockade of Notch-4 was more effective on CLL cells in coculture with hBM-MSCs. In presence of anti-Notch-1 or anti-Notch-3-blocking antibodies, no significant differences in the decrease of viable CLL cells were observed after 3 days of coculture with either hBM-MSCs or hBM-MSCs*.
Table 2. CLL cell survival following blockade of Notch receptors and ligands.
| Treatment | + hBM-MSCs (% of viable cells) | + hBM-MSCs* (% of viable cells) | ANOVA test (a. versus b.) | CLL alone (% of viable cells) |
|---|---|---|---|---|
| No treatment | 94.8 (5.3) | 89.5 (10.1) | NS | 62.3 (14.4) |
| Anti-Notch-1 | 79.6 (8.5)* | 67.0 (6.2)* | NS | 37.2 (7.9)* |
| Anti-Notch-2 | 64.2 (7.9)* | 52.6 (6.8)** | * | 26.6 (8.7)** |
| Anti-Notch-3 | 89.3 (7.3) | 82.0 (15.1) | NS | 53.08 (15.6) |
| Anti-Notch-4 | 44.4 (11.5)*** | 57.1 (7.5)** | * | 27.2 (8.2)** |
| Anti-Notch-1/-2/-4 | 35.3 (9.6)*** | 34.6 (9.3)*** | NS | 14.6 (7.5)*** |
| Anti-Notch-1/-2/-3/-4 | 32.6 (8.2)*** | 33.8 (6.7)*** | NS | 12.8 (2.0)*** |
| Anti-Jagged-1 | 69.4 (11.4)* | 69.4 (8.3)* | NS | 35.6 (8.8)* |
| Anti-Jagged-2 | 67.8 (11.9)* | 68.8 (7.5)* | NS | 35.8 (7.2)* |
| Anti-DLL-1 | 87.6 (12.2) | 88.8 (10.3) | NS | 65.5 (16.8) |
| Anti-DLL-3 | 72.3 (10.7)* | 63.9 (6.2)* | NS | 35.3 (4.8)* |
| Anti-DLL-4 | 70.3 (9.3)* | 64.1 (8.5)* | NS | 33.5 (8.1)* |
| Anti-Jagged-1/-2, DLL-3/-4 | 38.1 (8.5)*** | 37.3 (9.3)*** | NS | 15.7 (3.8)*** |
| Anti-Jagged-1/-2, DLL-1/-3/-4 | 36.8 (9.4)*** | 34.3 (8.6)*** | NS | 13.5 (2.1)*** |
| GSI XII | 33.3 (9.2)*** | 35.1 (8.3)*** | NS | 14.4 (3.8)*** |
| DMSO | 89.6 (14.6) | 85.5 (12.3) | NS | 63.1 (23.4) |
Abbreviations: ANOVA, analysis of variance; Anti-, blocking antibody; CLL, chronic lymphocytic leukemia; DLL, delta-like ligand; NS, not significant; DMSO, dimethyl sulfoxide.
Values are percentages of overall viable human CLL cells (mean±SD of 12 independent experiments); DMSO is the vehicle control for GSI XII, 7.5 μM. Data were represented as mean±SD of 12 independent experiments. Statistical analysis was done using ANOVA test. *P<0.05, **P<0.01, ***P<0.001 versus IgG-treated control was considered statistically significant.
Figure 4.
Viable CLL cells cultured alone and cocultured with hBM-MSCs or hBM-MSCs* in absence or presence of either neutralizing antibodies anti-Notch receptors or GSI XII. B-CLL cells were cultured alone or cocultured with either hBM-MSCs (a) or hBM-MSCs* (b) (10/1 ratio), in absence or presence of anti-Notch-1, -2, -3, -4 antibodies, or GSI XII for 3 days. Percentage of viable CLL cells (Annexin V−/7-AAD−, lower left quadrant) was measured by flow cytometry after electronic gating on CD5+CD19+CD45+ CLL cells. Statistical analysis was carried out by using one-way analysis of variance, Holm–Sidak test. *P<0.05, **P<0.01, ***P<0.001 versus IgG-treated control were considered statistically significant. GSI XII was resuspended in dimethyl sulfoxide (DMSO) and DMSO was used as control for vehicle effects.
Jagged-1, Jagged-2, DLL-3 and DLL-4 are involved in MSC-mediated survival enhancement of B-CLL cells
By blocking Jagged-1, -2, DLL-3 or -4 a significant reduction of CLL cell survival was achieved in both culture conditions (Table 2). By contrast, blockade of DLL-1 had little effect. The combination of anti-Jagged-1, -2, DLL-3 and -4 was more efficient in promoting apoptosis of CLL cells cultured in both conditions. The addition of anti-DLL-1 to the mixture of anti-Jagged-1, -2, DLL-3 and -4 did not modify CLL cell survival, thus suggesting the major role of the loop among Notch-1, -2 and -4 and their ligands Jagged-1, -2 and DLL-3, -4. In addition, these results suggest that Jagged-1, -2, DLL-3 and -4 or Notch-1, -2 and -4 have different downstream targets involved in the prevention of CLL cell apoptosis. To further confirm the role of the Notch pathway in CLL cell survival, Notch receptors were stimulated directly by adding exogenously their recombinant Notch ligands. We observed that biologically active Jagged-1, -2 and DLL-3, -4 significantly enhanced the survival of CLL cells when cultured alone, whereas DLL-1 did not (Supplementary Figure 1). Blockade of Jagged-1, -2, DLL-3 and -4 or Notch-1, -2 and -4 only on either hBM-MSCs or hBM-MSCs* significantly decreased the survival of CLL cells in coculture conditions (data not shown). The survival of CLL cells previously incubated with antibodies against Jagged-1, -2, DLL-3 and -4 or Notch-1, -2 and -4 also significantly decreased in coculture with either hBM-MSCs or hBM-MSCs* (data not shown). Thus, Jagged-1, -2, DLL-3 ligands as well as Notch-1, -2 and -4 receptors expressed by both cell types resulted involved in CLL cell survival.
Molecular pathways involved in Notch-1, Notch-2 and Notch-4-dependent antiapoptotic effect of MSCs on CLL cells
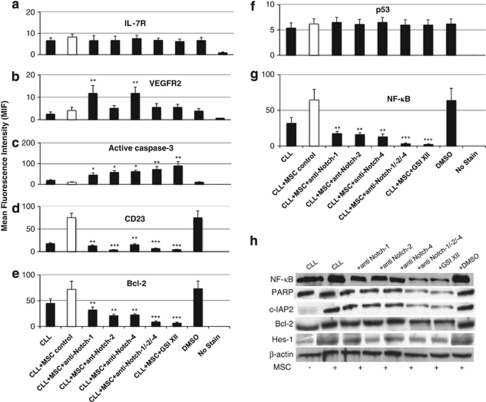
To identify the downstream targets of Notch signaling pathway involved in the prevention of CLL cell apoptosis after direct contact with either hBM-MSCs or hBM-MSCs*, we assessed by flow cytometry the expression of IL-7R, VEGFR2, active caspase-3, CD23, Bcl-2, p53 and NF-κB by CLL cells cocultured for 3 days (Figures 5a–g). We observed that VEGFR2 and active caspase-3 were weakly expressed by CLL cells at basal conditions, whereas IL-7R, CD23, Bcl-2, p53 and NF-κB were highly expressed, as previously shown.6, 38 Active caspase-3 downregulation and IL-7R, CD23, Bcl-2, p53 and NF-κB overexpression by CLL cells were even more evident following coculture of CLL cells with hBM-MSCs. Accordingly, active caspase-3 downregulation and IL-7R, CD23, Bcl-2, NF-κB upregulation were reverted by adding either the mixture of anti-Notch-1+anti Notch-2+anti-Notch-4 antibodies or GSI XII, whereas p53 expression remained unchanged, as previously shown for Notch-1.39 The addition of blocking anti-Notch-3 had little effect on the expression of active IL-7R, VEGFR2, active caspase-3, CD23, Bcl-2 and NF-κB by CLL cells in coculture (data not shown).
Figure 5.
Expression of IL-7R, VEGFR2, active caspase-3, Bcl-2, CD23, c-IAP protein, NF-κB and Hes-1 in CLL cells cocultured with hBM-MSCs. The expression of IL-7 R (a), VEGFR2 (b), active caspase-3 (c), CD23 (d), Bcl-2 (e), p53 (f) and NF-κB (g) by CD5+CD19+CD45+ CLL cells was analyzed by flow cytometry following coculture with hBM-MSCs (10/1 ratio), in absence or presence of anti-Notch-1,-2 or -4 antibodies or GSI XII for 3 days. Western blot analysis was performed to assess the expression of c-IAP2, Hes-1, PARP, NF-κB and Bcl-2 by CLL cells cocultured with hBM-MSCs in the same conditions (h). Hes-1 expression was used as functional read-out for Notch activation. NF-κB and Bcl-2 expression was evaluated to confirm CLL cell activation following coculture with hBM-MSCs in the same conditions. β-actin was included as loading control. Statistical analysis was carried out by using one-way analysis of variance, Holm–Sidak test. *P<0.05, **P<0.01, ***P<0.001 versus IgG-treated control were considered statistically significant. GSI XII was resuspended in dimethyl sulfoxide (DMSO) and DMSO was used as control for vehicle effects.
Western blot analysis was performed to verify the targets inhibited by the Notch pathway. As shown in Figure 5h, the treatment of CLL cells with either the mixture of anti-Notch-1+Notch-2+Notch-4 antibodies or GSI XII resulted in the loss of protein 2 (c-IAP2), Bcl-2, NF-κB and Hes-1 protein expression followed by a remarkable increase in PARP degradation. These data confirmed the specific inhibition of the Notch pathway in CLL cells following the treatment with inhibitors. Moreover, the addition of recombinant IL-7 significantly increased the survival of CLL cells cocultured with hBM-MSCs or hBM-MSCs* (data not shown), whereas IL-6 did not, as previously reported.6
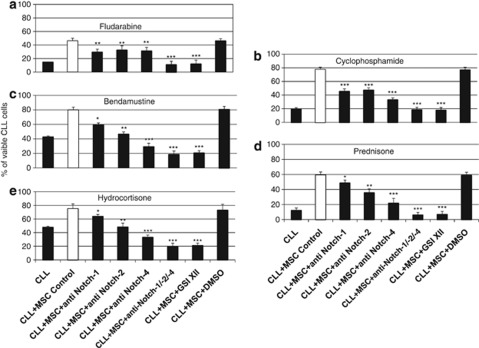
Notch-1, Notch-2 and Notch-4 promote the chemo-resistance of CLL cells in contact with MSCs
CLL cells were cultured with or without hBM-MSCs or hBM-MSCs*, in absence or presence of drugs, by adding either the mixture of anti-Notch-1, -2, -4 or GSI XII for 3 days. Drugs promoted apoptosis of CLL cells when cultured alone, whereas a consistent increase in the overall number of viable CLL cells was observed in coculture with MSCs, as previously shown.3, 4, 39, 40, 41, 42, 43 Interestingly, blockade of Notch-1, -2 and -4 or all Notch signaling by GSI XII in presence of Fludarabine, Cyclophosphamide, Bendamustine, Prednisone and Hydrocortisone dramatically decreased the total number of viable CLL cells even when cocultured with MSCs (Supplementary Table 2, Figure 6). To verify whether there was a direct functional link between Notch pathway and drug-induced apoptosis, we assessed by flow cytometry the effect of the blockade of Notch-1, -2 and -4 or whole Notch signaling on the expression of active caspase-3, Bcl-2, NF-κB, p53 in CLL cells in culture and coculture conditions in presence of drugs for 3 days. Active caspase-3 was overexpressed by CLL cells when cultured alone in presence of drugs, whereas Bcl-2, NF-κB and p53 were only weakly expressed (Supplementary Figure 2). Active caspase-3 downregulation and Bcl-2, NF-κB, and p53 overexpression by CLL cells were further observed following coculture of CLL cells with hBM-MSCs in presence of drugs. By contrast, active caspase-3 downregulation and Bcl-2 and NF-κB overexpression were reverted by adding either the mixture of anti-Notch-1+anti-Notch-2+anti-Notch-4 antibodies or GSI XII in presence of drugs, whereas p53 expression remained unchanged.
Figure 6.
Percentage of viable CLL cells cocultured with hBM-MSCs in absence or presence of chemotherapeutic agents and neutralizing Notch receptor antibodies and GSI XII.Percentage of viable CLL cells (Annexin V−/7-AAD−, lower left quadrant) cultured alone in absence or presence of Fludarabine (5.0 μℳ) (a), Cyclophosphamide (100 μℳ) (b), Bendamustine (200.0 μℳ) (c), Prednisone (100.0 μℳ) (d) and Hydrocortisone (100.0 μℳ) (e), and cocultured with hBM-MSCs (10/1 ratio) in presence or absence of neutralizing Notch receptor antibodies or GSI XII for 3 days. CLL cells were analyzed by flow cytometry after electronic gating on CD5+CD19+CD45+ CLL cells. GSI XII was resuspended in dimethyl sulfoxide (DMSO) and DMSO was used as control for vehicle effects. One-way analysis of variance, Holm–Sidak test was used to compare the different groups. *P<0.05, **P<0.01, ***P<0.001 were considered statistically significant.
In the same conditions, no proliferation of CLL cells in coculture with either hBM-MSCs* or hBM-MSCs was observed. In all samples, following drugs addition, we found a significant decrease in the percentage of CLL cells in S-phase and a significant increase of those in G0G1-phase, with a very low proportion of CLL cells in G2/M phases, with no significant differences between hBM-MSCs and hBM-MSCs* (Supplmentary Table 3).
MSC-mediated survival of CLL cells requires both cell contact and soluble factors
Coculture of CLL cells with hBM-MSCs in Transwell conditions resulted in loss of the protective effect of MSCs (data not shown), according to previous data.6, 8 However, the supernatant obtained from 1-day culture of hBM-MSCs significantly increased the survival of CLL cells as compared with CLL cells cultured alone for 5 days (47.1±10.5% (culture+RPMI) versus 63.7±5.2% (culture+supernatant), P<0.05). Furthermore, similar results were obtained by coculturing CLL cells with hBM-MSCs* in the same experimental conditions (data not shown). Blocking antibodies significantly decreased CLL cell survival in Transwell coculture conditions, even by coculturing CLL cells with hBM-MSCs* (data not shown). Thus, both soluble factors and adherence of CLL cells to MSCs are required to obtain protection from apoptosis, as previously shown.6, 8
Regulatory properties of MSCs from CLL patients
We asked whether hBM-MSCs from CLL patients had the same immune regulatory properties of hBM-MSCs from normal donors. T and NK cell proliferation was similarly inhibited by hBM-MSCs* and hBM-MSCs, and the differences were not statistically significant (data not shown). By contrast, we observed that both hBM-MSCs* and hBM-MSCs significantly increased the number of surviving unstimulated B cells, as compared with that obtained by culturing B cells alone (data not shown).32 The reduction in apoptosis was not associated with B-cell proliferation, according to previous data,44 as the proliferation index at day 3 were 1.01 (coculture with hBM-MSCs*) versus 1.03 (coculture with hBM-MSCs). When grown on a layer of hBM-MSCs* or hBM-MSCs, the proportion of B cells in the G0G1-phase increased substantially, with a concomitant decrease in the proportion of cells in the S and G2/M phases, as compared with what obtained by culturing B cells alone (data not shown).
Discussion
It is well known that BM-MSCs promote the growth and accumulation of normal B cells and CLL cells7, 8, 9, 10, 11, 12, 13 and normally express molecules of the Notch family that are strictly related to some of their functions.33, 45, 46 However, it is unknown whether Notch signaling pathway has a role in promoting chemoresistance upon contact with BM stromal cells during the leukemogenic process.
In this study, we found that Notch-1, -2 and -4 were involved in stroma-dependent CLL cell resistance to Fludarabine, Cyclophosphamide, Bendamustine, Prednisone and Hydrocortisone. CLL cells cultured alone underwent progressive and time-dependent apoptosis, as previously reported,9 whereas MSCs dramatically increased the survival of CLL cells, as previously shown.8, 9, 10, 11, 12, 13 Autologous BM-MSCs obtained from CLL patients supported the survival of both normal B cells and CLL cells in a similar manner than allogeneic BM-MSCs from normal donors. This effect was reverted by inhibiting the entire Notch signaling pathway with GSI XII, thus suggesting that some molecules of the Notch pathway were involved in the phenomenon. This evidence supports the concept that Notch signaling has a direct role in CLL cell survival via the reciprocal interactions among leukemia cells, independently of the interaction with stromal cells; nevertheless, the presence of stromal cells significantly enhanced this phenomenon. In fact, CLL cells and MSC coculture led to the significant overexpression of DLL-3, Notch-1 and -4 by CLL cells, as well as of Notch-4 only by hBM-BMSCs, but not hBM-MSCs*. Blocking and stimulating experiments demonstrated that Jagged-1/-2 and DLL-3/-4 ligands synergistically interact with Notch-1, -2 and -4 and are important components of the stromal cell-mediated antiapoptotic effect observed in cocultured CLL cells. Blocking antibodies against all Notch receptors and ligands but Notch-3 or DLL-1 decreased the survival of CLL cells even when cultured alone. Thus, the reciprocal interactions among CLL cells via Notch signaling also have an important role in cell survival. Coculture experiments of CLL cells with hBM-MSCs in Transwell conditions showed that the enhancement of CLL cell survival is not only due to the direct contact of CLL cells with MSCs, but also to soluble factors released by MSCs, such as IL-7 and VEGF, as previously shown.6, 7, 8 Furthermore, Notch-1, -2 and -4-dependent pro-survival effect by hBM-MSCs was associated to the maintenance of IL-7R, CD23, Bcl-2, NF-κB, protein 2 (c-IAP2) and Hes-1 expression by CLL cells, together with the downregulation of active caspase-3, without affecting p53 expression. These findings are in agreement with previous data describing the role of Bcl-2 and NF-κB signaling in the persistence of CLL residual clones following chemotherapy 47, 48 and demonstrating that BM-MSCs may protect CLL cells from drug-induced apoptosis.1, 4, 49 In addition, our data suggest that Notch-1, -2 and -4 are involved in the chemo-resistance of CLL cells in direct contact with hBM-MSC following drugs exposure. We found that Notch-1, -2 and -4 signaling upregulates Bcl-2 and NF-κB and downregulates active caspase-3 expression by CLL cells cocultured with hBM-MSCs or hBM-MSCs*, in absence or presence of drugs. Previous studies have shown that VLA-4 engagement by fibronectin induces in vitro resistance of CLL cells to Fludarabine.50 Therefore, the ability of Notch-1, Notch-2, Notch-4 signaling to confer resistance towards drugs to CLL cells in direct contact with MSCs, by modulating Bcl-2, NF-κB and p53 expression,51, 52, 53 supports the concept that this effect may occur at least in part through Bcl-2-, NF-κB- and p53-dependent mechanisms. In addition, the involvement of p53 in VLA-4 pathway has been reported to mediate resistance of CLL cells to drugs.43, 49
Overall, our data show that Notch pathway normally has an important role in MSC-dependent promotion of the survival and resistance of CLL cells to drugs. Therefore, the onset of dysregulated Notch pathway, that is, as a consequence of mutations occurring in the Notch1 gene, could even enhance the prosurvival signals that the normal pathway already confers to CLL cells interacting reciprocally or with BM stromal cells. Thus, Notch inactivation by specific inhibitors, as suggested for acute lymphoblastic leukemia,3, 32 could represent an additional tool to overcome drug resistance and ameliorate the therapeutic strategies for CLL in both aggressive and indolent phases of the disease.
Acknowledgments
This work was partially supported by the Italian Ministry of University and Scientific Research (PRIN 2005, PRIN 2007), and Fondazione CARIVERONA, Bando 2003 (‘Microambiente e progressione neoplastica: il network di regolazione indotto da cellule stromali. Biologia e applicazioni terapeutiche') and Bando 2008 (‘Utilizzo delle capacità rigenerative ed immunoregolatorie delle cellule staminali mesenchimali per il trattamento di patologie ossee, neurologiche e cardiologiche'); AIRC, Fondazioni CARIVERONA and CARIPARO (An integrated approach to Chronic Lymphoproliferative disorders: B-CLL and virus-related disorders).
Armel Hervé Nwabo Kamdje designed and performed research, analyzed data and wrote the paper; Giulio Bassi, Luciano Pacelli, Giorgio Malpeli, Eliana Amati and Ilaria Nichele performed research and Giovanni Pizzolo contributed to paper writing; Mauro Krampera designed research, analyzed data and wrote the paper.
The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on Blood Cancer Journal website (http://www.nature.com/bcj)
Supplementary Material
References
- Chiorazzi N, Rai KR, Ferrarini M. Chronic lymphocytic leukemia. N Engl J Med. 2005;352:804–815. doi: 10.1056/NEJMra041720. [DOI] [PubMed] [Google Scholar]
- Nagasawa T. Microenvironmental niches in the bone marrow required for B-cell development. Nat Rev Immunol. 2006;6:107–116. doi: 10.1038/nri1780. [DOI] [PubMed] [Google Scholar]
- Nwabo Kamdje AH, Krampera M. Notch signaling in acute lymphoblastic leukemia: any role for stromal microenvironment. Blood. 2011;118:6506–6514. doi: 10.1182/blood-2011-08-376061. [DOI] [PubMed] [Google Scholar]
- Burger M, Hartmann T, Krome M, Rawluk J, Tamamura H, Fujii N, et al. Small peptide inhibitors of the CXCR4 chemokine receptor (CD184) antagonize the activation, migration, and antiapoptotic responses of CXCL12 in chronic lymphocytic leukemia B cells. Blood. 2005;106:1824–1830. doi: 10.1182/blood-2004-12-4918. [DOI] [PubMed] [Google Scholar]
- Balakrishnan K, Burger JA, Wierda WG, Gandhi V. AT-101 induces apoptosis in CLL B cells and overcomes stromal cell-mediated Mcl-1 induction and drug resistance. Blood. 2009;113:149–153. doi: 10.1182/blood-2008-02-138560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertrand FE, Spengemen JD, Shelton JG, McCubrey JA. Inhibition of PI3K, mTOR and MEK signaling pathways promotes rapid apoptosis in B-lineage ALL in presence of stromal cell support. Leukemia. 2005;19:98–102. doi: 10.1038/sj.leu.2403560. [DOI] [PubMed] [Google Scholar]
- Long BW, Witte PL, Abraham GN, Gregory SA, Plate JM. Apoptosis and interleukin 7gene expression in chronic B-lymphocytic leukemia cells. Proc Natl Acad Sci USA. 1995;92:1416–1420. doi: 10.1073/pnas.92.5.1416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lagneaux L, Delforge A, Bron D, De Bruyn C, Stryckmans P. Chronic lymphocytic leukemic B cells but not normal B cells are rescued from apoptosis by contact with normal bone marrow stromal cells. Blood. 1998;91:2387–2396. [PubMed] [Google Scholar]
- Panayiotidis P, Jones D, Ganeshaguru K, Foroni L, Hoffbrand AV. Human bone marrow stromal cells prevent apoptosis and support the survival of chronic lymphocytic leukaemia cells in vitro. Br J Haematol. 1996;92:97–103. doi: 10.1046/j.1365-2141.1996.00305.x. [DOI] [PubMed] [Google Scholar]
- Burger JA, Tsukada N, Burger M, Zvaifler NJ, Dell′Aquila M, Kipps TJ. Blood-derived nurse-like cells protect chronic lymphocytic leukemia B cells from spontaneous apoptosis through stromal cell-derived factor-1. Blood. 2000;96:2655–2663. [PubMed] [Google Scholar]
- Nishio M, Endo T, Tsukada N, Ohata J, Kitada S, Reed JC, et al. Nurselike cells express BAFF and APRIL, which can promote survival of chronic lymphocytic leukemia cells via a paracrine pathway distinct from that of SDF-1alpha. Blood. 2005;106:1012–1020. doi: 10.1182/blood-2004-03-0889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deaglio S, Vaisitti T, Bergui L, Bonello L, Horenstein AL, Tamagnone L, et al. CD38 and CD100 lead a network of surface receptors relaying positive signals for B-CLL growth and survival. Blood. 2005;105:3042–3050. doi: 10.1182/blood-2004-10-3873. [DOI] [PubMed] [Google Scholar]
- Richardson SJ, Matthews C, Catherwood MA, Alexander HD, Carey BS, Farrugia J, et al. ZAP-70 expression is associated with enhanced ability to respond to migratory and survival signals in B-cell chronic lymphocytic leukemia (B-CLL) Blood. 2006;107:3584–3592. doi: 10.1182/blood-2005-04-1718. [DOI] [PubMed] [Google Scholar]
- Pedersen IM, Kitada S, Leoni LM, Zapata JM, Karras JG, Tsukada N, et al. Protection of CLL B cells by a follicular dendritic cell line is dependent on induction of Mcl-1. Blood. 2005;100:1795–1801. [PubMed] [Google Scholar]
- Ruan J, Hyjek E, Kermani P, Christos PJ, Hooper AT, Coleman M, et al. Magnitude of stromal hemangiogenesis correlates with histologic subtype of non-Hodgkin′s lymphoma. Clin Cancer Res. 2006;12:5622–5631. doi: 10.1158/1078-0432.CCR-06-1204. [DOI] [PubMed] [Google Scholar]
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- Phinney DG, Prockop DJ. Concise review: mesenchymal stem/multipotent stromal cells: the state of transdifferentiation and models of tissue repair - current views. Stem Cells. 2007;25:2896–2902. doi: 10.1634/stemcells.2007-0637. [DOI] [PubMed] [Google Scholar]
- Burger JA, Gandhi V, et al. The lymphatic tissue microenvironments in chronic lymphocytic leukemia: in vitro models and the significance of CD40-CD154 interactions. Blood. 2009;114:2560–2561. doi: 10.1182/blood-2009-06-228981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shain KH, Landowski TH, Dalton WS. Adhesion-mediated intracellular redistribution of c-Fas-associated death domain-like IL-1-converting enzyme-like inhibitory protein-long confers resistance to CD95-induced apoptosis in hematopoietic cancer cell lines. J Immunol. 2002;168:2544–2553. doi: 10.4049/jimmunol.168.5.2544. [DOI] [PubMed] [Google Scholar]
- Vogler M, Butterworth M, Majid A, Walewska RJ, Sun XM, Dyer MJ, et al. Concurrent up-regulation of BCL-XL and BCL2A1 induces approximately 1000-fold resistance to ABT-737 in chronic lymphocytic leukemia. Blood. 2009;113:4403–4413. doi: 10.1182/blood-2008-08-173310. [DOI] [PubMed] [Google Scholar]
- Artavanis-Tsakonas S, Rand MD, Lake RJ. Notch signaling: cell fate control and signal integration in development. Science. 1999;284:770–776. doi: 10.1126/science.284.5415.770. [DOI] [PubMed] [Google Scholar]
- Radtke F, Fasnacht N, MacDonald HR. Notch signaling in the immune system. Immunity. 2010;32:14–27. doi: 10.1016/j.immuni.2010.01.004. [DOI] [PubMed] [Google Scholar]
- Apelqvist A, Li H, Sommer L, Beatus P, et al. Notch signaling controls pancreatic cell differentiation. Nature. 1999;400:877–881. doi: 10.1038/23716. [DOI] [PubMed] [Google Scholar]
- Lobov IB, Renard RA, Papadopoulos N, et al. Delta-like ligand 4 (Dll4) is induced by VEGF as a negative regulator of angiogenic sprouting. PNAS. 2007;104:3219–3224. doi: 10.1073/pnas.0611206104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loomes KM, Stevens SA, O′Brien ML, et al. Dll3 and Notch1 genetic interactions model axial segmental and craniofacial malformations of human birth defects. Dev Dyn. 2007;236:2943–2951. doi: 10.1002/dvdy.21296. [DOI] [PubMed] [Google Scholar]
- Liotta F, Angeli R, Cosmi L, Filì L, Manuelli C, Frosali F, et al. TLR3 and TLR4 are expressed by human bone marrowderived mesenchymal stem cells and can inhibit their T-cell modulatory activity by impairing Notch signalling. Stem Cells. 2008;26:279–289. doi: 10.1634/stemcells.2007-0454. [DOI] [PubMed] [Google Scholar]
- Calvi LM, Adams GB, Weibrecht KW, Weber JM, Olson DP, Knight MC, et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature. 2003;425:841–848. doi: 10.1038/nature02040. [DOI] [PubMed] [Google Scholar]
- Lobov IB, Renard RA, Papadopoulos N, Gale NW, Thurston G, Yancopoulos GD, et al. Delta-like ligand 4 (Dll4) is induced by VEGF as a negative regulator of angiogenic sprouting. PNAS. 2007;104:3219–3224. doi: 10.1073/pnas.0611206104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puente XS, Pinyol M, Quesada V, Conde L, Ordóñez GR, Villamor N, et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukemia. Nature. 2011;475:101–105. doi: 10.1038/nature10113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fabbri G, Rasi S, Rossi D, Trifonov V, Khiabanian H, Ma J, et al. Analysis of the chronic lymphocytic leukemia coding genome: role of Notch1 mutational activation. J Exp Med. 2011;208:1389–1401. doi: 10.1084/jem.20110921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zweidler-McKay PA, He Y, Xu L, Rodriguez CG, Karnell FG, Carpenter AC, et al. Notch signaling is a potent inducer of growth arrest and apoptosis in a wide range of B-cell malignancies. Blood. 2005;106:3898–3906. doi: 10.1182/blood-2005-01-0355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nwabo Kamdje AH, Mosna F, Bifari F, Lisi V, Bassi G, Malpeli G, et al. Notch-3 and Notch-4 signaling rescues from apoptosis Human B-lineage acute lymphoblastic leukemia cells in contact with human bone marrow-derived mesenchymal stromal cells. Blood. 2011;118:380–389. doi: 10.1182/blood-2010-12-326694. [DOI] [PubMed] [Google Scholar]
- Rosati E, Sabatini R, Rampino G, Tabilio A, Di Ianni M, Fettucciari K, et al. Constitutively activated Notch signaling is involved in survival and apoptosis resistance of B-CLL cells. Blood. 2009;113:856–865. doi: 10.1182/blood-2008-02-139725. [DOI] [PubMed] [Google Scholar]
- Krampera M, Pasini A, Rigo A, Scupoli MT, Tecchio C, Malpeli G, et al. HB-EGF/HER-1 signalling in bone marrow mesenchymal stem cells: inducing cell expansion and reversibly preventing multi-lineage differentiation. Blood. 2005;106:59–66. doi: 10.1182/blood-2004-09-3645. [DOI] [PubMed] [Google Scholar]
- Tabe Y, Jin L, Tsutsumi-Ishii Y, Xu Y, McQueen T, Priebe W, et al. Activation of Integrin-linked kinase is a critical prosurvival pathway induced in leukemic cells by bone marrow-derived stromal cells. Cancer Res. 2007;67:684–694. doi: 10.1158/0008-5472.CAN-06-3166. [DOI] [PubMed] [Google Scholar]
- Creagh EM, Sheehan D, Cotter TG. Heat shock proteins − modulators of apoptosis in tumour cells. Leukemia. 2000;14:1161–1173. doi: 10.1038/sj.leu.2401841. [DOI] [PubMed] [Google Scholar]
- Hubmann R, Schwarzmeier JD, Shehata M, Hilgarth M, Duechler M, Dettke M, et al. Notch2 is involved in the over-expression of CD23 in B-cell chronic lymphocytic leukemia. Blood. 2002;99:3742–3747. doi: 10.1182/blood.v99.10.3742. [DOI] [PubMed] [Google Scholar]
- Secchiero P, Melloni E, di Iasio MG, Tiribelli M, Rimondi E, Corallini F, et al. Nutlin-3 up-regulates the expression of Notch1 in both myeloid and lymphoid leukemic cells, as part of a negative feedback antiapoptotic mechanism. Blood. 2009;113:4300–4308. doi: 10.1182/blood-2008-11-187708. [DOI] [PubMed] [Google Scholar]
- Kitada S, Andersen J, Akar S, Zapata JM, Takayama S, Krajewski S, et al. Expression of apoptosis-regulating proteins in chronic lymphocytic leukemia: correlations with In vitro and In vivo chemoresponses. Blood. 1998;91:3379–3389. [PubMed] [Google Scholar]
- Robertson LE, Chubb S, Meyn RE, Story M, Ford R, Hittelman WN, et al. Induction of apoptosis cell death in chronic lymphocytic leukemia by 2-chloro-2′-deoxyadenosine and 9-β-D-arabinosyl-2-fluoroadenine. Blood. 1993;81:143–150. [PubMed] [Google Scholar]
- Schwänen C, Hecker T, Hübinger G, Wölfle M, Rittgen W, Bergmann L, et al. In vitro evaluation of bendamustine induced apoptosis in B-chronic lymphocytic leukemia. Leukemia. 2002;16:2096–2105. doi: 10.1038/sj.leu.2402651. [DOI] [PubMed] [Google Scholar]
- Hoellenriegel J, Meadows SA, Sivina M, Wierda WG, Kantarjian H, Keating MJ, et al. The phosphoinositide 3′-kinase delta inhibitor, CAL-101, inhibits B-cell receptor signaling and chemokine networks in chronic lymphocytic leukemia. Blood. 2011;118:133603–133612. doi: 10.1182/blood-2011-05-352492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuente MT, Casanova B, Moyano JV, Mercedes GG, Laura S, José JM, et al. Engagement of α4β1 integrin by fibronectin induces in vitro resistance of B chronic lymphocytic leukemia cells to fludarabine. J Leuk Biol. 2002;71:495–502. [PubMed] [Google Scholar]
- Corcione A, Benvenuto F, Ferretti E, Giunti D, Cappiello V, Cazzanti F, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006;107:367–372. doi: 10.1182/blood-2005-07-2657. [DOI] [PubMed] [Google Scholar]
- Hajdu M, Sebestyén A, Barna G, Reiniger L, Jánosi J, Sréter L, et al. Activity of the notch-signalling pathway in circulating human chronic lymphocytic leukaemia cells. Scand J Immunol. 2007;65:271–275. doi: 10.1111/j.1365-3083.2006.01897.x. [DOI] [PubMed] [Google Scholar]
- Apelqvist A, Li H, Sommer L, Beatus P, Anderson DJ, Honjo T, et al. Notch signaling controls pancreatic cell differentiation. Nature. 1999;400:877–881. doi: 10.1038/23716. [DOI] [PubMed] [Google Scholar]
- Hallaert DYH, Jaspers A, van Noesel CJ, van Oers MHJ, Kater AP, Eldering E. c-Abl kinase inhibitors overcome CD40-mediated drug resistance in CLL: implications for therapeutic targeting of chemo-resistant niches. Blood. 2008;112:5141–5149. doi: 10.1182/blood-2008-03-146704. [DOI] [PubMed] [Google Scholar]
- Hanada M, Delia D, Aiello A, Stadtmauer E, Reed JC. bcl-2 gene hypomethylation and high-level expression in B-cell chronic lymphocytic leukemia. Blood. 1993;82:1820–1828. [PubMed] [Google Scholar]
- Kurtova AV, Balakrishnan K, Chen R, Ding W, Schnabl S, Quiroga MP, et al. Diverse marrow stromal cells protect CLL cells from spontaneous and drug-induced apoptosis: development of a reliable and reproducible system to assess stromal cell adhesion-mediated drug resistance. Blood. 2009;114:4441–4450. doi: 10.1182/blood-2009-07-233718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuente MT, Casanova B, Cantero E, Cerro MH, Garcia-Marco J, Silva A. Involvement of p53 in α4β1 integrin-mediated resistance of B-CLL cells to fludarabine. Biochem Biophys Res Communs. 2003;311:708–712. doi: 10.1016/j.bbrc.2003.10.054. [DOI] [PubMed] [Google Scholar]
- Damiano JS, Dalton WS. Integrin-Mediated Drug Resistance in Multiple Myeloma. Leuk Lymphoma. 2000;38:71–81. doi: 10.3109/10428190009060320. [DOI] [PubMed] [Google Scholar]
- Sturm I, Bosanquet AG, Hermann S, Guner D, Dorken B, Daniel PT. Mutation of p53 and consecutive selective drug resistance in B-CLL occurs as a consequence of prior DNA-damaging chemotherapy. Cell Death Differ. 2003;10:477–484. doi: 10.1038/sj.cdd.4401194. [DOI] [PubMed] [Google Scholar]
- Wickremasinghe RG, Prentice AG, Steele AJ. p53 and Notch signaling in chronic lymphocytic leukemia: clues to identifying novel therapeutic strategies. Leukemia. 2011;25:1400–1407. doi: 10.1038/leu.2011.103. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.