Abstract
Introduction
This prospective magnetic resonance imaging (MRI) study in chronic low-back pain (CLBP) patients evaluated the natural course of degenerative lumbar spine changes in relation to Modic 1 type changes (M1) within 1 year.
Materials and methods
From 3,811 consecutive CLBP patients referred to lumbar spine MRI 54 patients with a large M1 were selected using strict exclusion criteria to exclude specific back disorders. Follow-up MRI was obtained within 11–18 months.
Results
At baseline M1 was associated with an adjacent endplate lesion in 96% of the cases. In follow-up, an unstable M1 was associated both with an increase of endplate lesions, decrease of disc height and change in disc signal intensity, most found at L4/5 or L5/S1. In disc spaces without M1, progression of degenerative changes was rare.
Conclusion
Endplate deformation, decreasing disc height and change of disc signal intensity appear essential features of accelerated degenerative process associated with M1.
Keywords: Magnetic resonance imaging, Intervertebral disc, Modic change, Endplate lesion, Degenerative disc disease
Introduction
The importance of Modic changes has been pointed out in many studies due to their association with chronic low-back pain (CLBP) [1–3]. They are quite uncommon among asymptomatic people [4]. Three types of subchondral signal abnormalities in vertebral body marrow were originally described by Modic et al. [5]. Modic 1 type change (M1) was hypointense on T1-weighted magnetic resonance imaging (MRI) and hyperintense on T2-weighted MRI. Fibrovascular tissue was found in such lesions. Modic 2 type change (M2), hyperintense on both T1-weighted image and T2-weighted image was detected in lesions with fatty degeneration. Modic 3 type change (M3) was hypointense on both T1-weighted image and T2-weighted image and detected in lesions with sclerosis. Modic changes are closely related to the degenerative process affecting the disc, including disc herniation [6–8]; their prevalence increases with age [9].
M1 that is associated with fibrovascular tissue has been noted to convert to M2 reflecting fatty degeneration [5]. The conversion may be slow since M1s have been found to enlarge even during several years’ follow-up [10, 11]. Reconversion from M2 to M1 has also been reported [7].
The etiology and pathomechanism of Modic changes and their role in the process of disc degeneration is poorly understood so far. Therefore, studies on various subgroups of patients with Modic changes are needed.
In previous studies an association between M1 and bony endplate lesion and an accelerated progress of disc degeneration was found [11, 12]. The patient sample was, however, small and the span of follow-up period quite variable (18–72 months). A shorter and more homogeneous follow-up interval is needed to study the velocity of progress and interval of appearance of degenerative disc changes.
The aim of this study was to evaluate the association of M1 in the lumbar spine with degenerative disc changes and bony endplate lesions and the short term-evolution of these variables.
Materials and methods
Patients
The patients were selected from 3,811 consecutive CLBP patients referred during six consecutive years to a standard lumbar spine magnetic resonance imaging (MRI) study at a university hospital. Patients with nonspecific CLBP of at least 3 months’ duration and a definite M1 or a mixed Modic change (M1/M2 or M1/M3) occupying an area of at least 15% of that of the vertebral body in sagittal MR (T1- and T2-weighted) images were included. The slice with the largest Modic change in T1-weighted image was chosen for measurement of the lesion. The strict exclusion criteria were: age older than 65 years, any specific back disease such as fracture, neoplasm, infectious or rheumatic spine disease, spondylolisthesis (4 mm or more), spinal stenosis, disc extrusion or any other finding with neural compression. In addition, patients with any major or a recent (<6 months before) minor spine operation were excluded. The mean age of the eligible 54 patients (45 females, 9 males) was 43.6 years (range 24–65 years). Eight of them had undergone an extirpation of a disc extrusion 3–7 years prior to the MRI, and all of them had been symptomless for at least 1-year period after operation. All patients gave a written informed consent to use their clinical data for the study purposes. The study protocol was approved at the research ethics review committee of the university hospital.
MRI and image analysis
The baseline MRI studies were performed prior to referral to the study clinic in four district hospitals. Standard imaging protocol was used in all hospitals with T1- and T2-weighted conventional turbo spin-echo and fast spin-echo sequences in sagittal and axial slices. The used MRI units were: two Gyroscan units (1.0 T, Philips, Eindhoven, The Netherlands), Signa (1.5 T, GE, Milwaukee, WI, USA), Sonata and Symphony (1.5 T, Siemens, Erlangen, Germany). All follow-up MRI studies (T1- and T2-weighted spin-echo sequences, sagittal slices) were performed with the 1.0 T Gyroscan unit—except for two claustrophobic patients with large M1s who underwent MRI in an open 0.35 T unit (Toshiba Opart, Toshiba America, San Francisco, USA). Standard hospital protocol for lumbar spine was used in each MRI unit. Due to different MRI units there was variation in imaging parameters. Typically TE/TR was 11–18/600–740 in T1-weighted sagittal and axial images and 90–120/3,395–4,500 in T2-weighted sagittal and axial images. FOV was 28–32 cm in sagittal images and 20 × 20 cm in axial images. Slice thickness 4 mm and matrix was 512 × 512 in T1- and T2-weighted sagittal images. An average interval from baseline to follow-up was 13.2 [11–18] months. As digital archiving systems were not available at the beginning of the study all imaging data were for uniformity assessed from hard copies.
The MRI analysis was modified from the classification criteria used in previous studies [11–14]. All images were analyzed together by two experienced radiologists—a neuroradiologist and a musculoskeletal radiologist with a 25 and 13 years’ clinical experience, respectively. They were blinded for the patients’ clinical data. The final conclusions were reached by consensus: in borderline cases the less severe grade of degeneration was reported. The repeatability of the MRI variables has been tested earlier [11]. First, all baseline images were evaluated to assure by location that the same Modic lesion was followed, then the progression of changes was assessed by visual comparison of the baseline and follow-up images. Findings in each discovertebral unit were analyzed separately at each disc level including changes in intervertebral disc and in its upper and lower endplate and adjacent bony endplates and in subchondral bone adjacent to the corresponding endplates.
At baseline the size of the M1 adjacent to the upper and lower endplate of the disc in each disc space (n = 270) was evaluated on sagittal T1- and T2-weighted images; the slice with the largest Modic change in T1-weighted image was chosen for the measurement of the lesion. The maximal area of M1, M2 and M3 was compared with that of the sagittal area of the vertebral body and was presented as a percentage.
Endplate lesions were assessed in each endplate and adjacent subchondral bone (n = 540) and classified as absent, a smooth depression, a dark spot (a focal subchondral hypointensity) (Fig. 1), multiple dark spots, a Schmorl lesion type focal defect, general irregularity and a Schmorl lesion type focal defect with general irregularity. Separate analysis of bony changes and endplate changes at the border between disc and vertebra was not attempted.
Fig. 1.
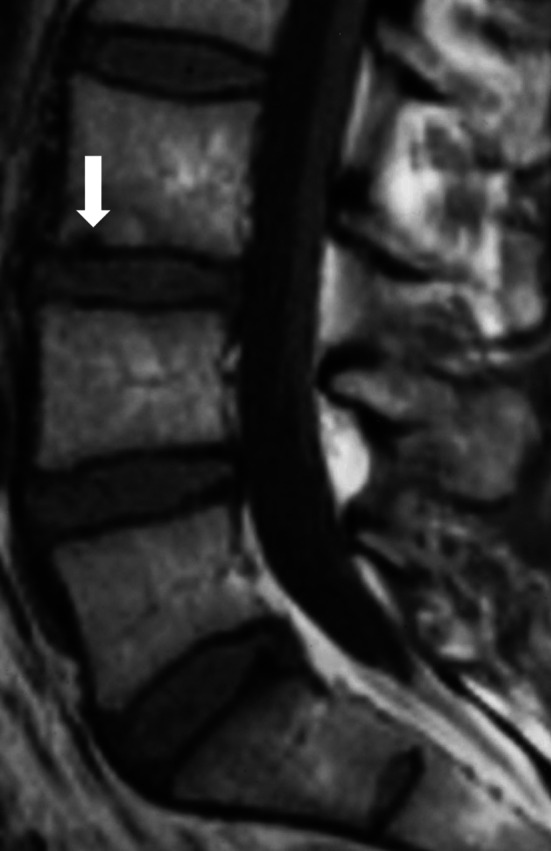
On T1-weighted image, a “dark spot” type focal subchondral hypointensity (arrow) surrounded by a mixed type (M1/M2) signal abnormality adjacent to the upper endplate of L3/4 disc
The presence of anterior and posterior bulging or protrusion reaching definitely beyond the intervertebral disc space was assessed at baseline on axial and sagittal images and recorded in a dichotomous manner as present or absent.
Disc height, the distance between the upper and the lower endplate of the adjacent vertebrae, was visually graded as normal (higher than or as high as the upper normal disc space), slightly decreased (33% lower than the upper, normal disc space), decreased (34–66% lower than the upper, normal disc space) or severely decreased (>66% lower than the upper, normal disc space) at levels L1/2–L4/5. Since L5/S1 is normally lower than upper disc space different criteria were applied to L5/S1 disc; normal bright signal intensity of nucleus pulposus was regarded always as normal disc height and degenerated disc was visually graded as slightly decreased, decreased or severely decreased.
Signal intensity of the nucleus pulposus (disc signal intensity) was estimated in each disc (n = 270) as normal (bright), fair (slightly decreased), dark (clearly decreased), black (annulus fibrosus and/or nucleus pulposus region similar to that of cortical bone) and black/white disc (dark annulus fibrosus but bright signal in nucleus pulposus in an otherwise severely degenerated disc with decreased disc height).
Comparative variables were created for the final analysis by comparing the size and signal intensity of the original lesion with that of the corresponding lesion in the follow-up image. The presence and change in size of M1 type lesion during the follow-up was classified as absent, constant, decreased, disappeared, increased and appearance of a new M1. The development of the M1 lesion into M2 or M3 type was classified accordingly. The presence and stability of endplate lesions was classified as absent, constant, decreased or disappeared, increased and appearance of a new finding. Disc height and its change were classified as constantly normal, constantly decreased (unchanged) and further decreased. A visually estimated decrease of disc height at least 20% between baseline and follow-up image was accepted. The change of disc signal intensity was evaluated as follows: constantly normal, constantly decreased (unchanged), further decreased, and increased (change from a dark or black to a bright nucleus pulposus or an increase of the bright area in the nucleus pulposus region in a black/white disc). The presence and change in size of the disc bulge (bulging or protrusion) was assessed on the sagittal image as absent, constant, decreased, disappeared and appeared.
Statistical analysis
The associations between baseline subchondral signal abnormalities and degenerative variables were evaluated by cross-tabulating. The association between the development of M1s and other degenerative changes was evaluated by cross-tabulating the comparative variables created to classify changes in M1, disc signal intensity, disc height and endplate lesions. Chi-square tests were run to detect significant differences in cell frequencies. Due to multiple observations per patient, intracluster correlation between these changes was computed [15]. Intracluster correlation coefficient compares the within-group variance (in this case, the progression of M1 changes within a single individual) with the between-group variance (the corresponding changes between patients) [16]. As the intracluster correlation was negative (ρ = −0.06), the conversion coefficient for Chi-square statistics was smaller than 1. The resulting adjusted Chi-square test statistic would be larger than the crude one and the corresponding adjusted p-values (not given) even smaller than the crude values (p < 0.001).
Results
Baseline
At baseline, altogether 110 M1 lesions in 540 endplates (20%) were detected in the study cohort (n = 54); 53% of the lesions were uniform M1s and 47% mixed (M1/M2) (Fig. 1). In addition, 29 uniform M2s and 20 mixed M3s were detected. Most M1s (89%) were located at the two lowermost lumbar disc spaces as were also most M2s (78%) and M3s (75%).
Endplate lesions was detected in 203/540 (38%) endplates; 51% of them in upper and 49% in lower endplates. Almost all (96%) of 110 M1s had an adjacent endplate lesion (Fig. 2). Dark spots were observed in 16/203 (8% of all endplate lesions), both with M1 or M2 and without a detectable Modic lesion (Fig. 1). Smooth depression was found in 16 (8%), Schmorl-type lesion in 39 (19%), irregularities in 69 (34%), multiple dark spots in 17 (8%) and Schmorl-type lesion and irregularities in 23 (11%) (Fig. 2). Of the 39 Schmorl-type endplate lesions, 33% had an adjacent M1, 33% an M2 and 33% had no Modic change. The majority of the most severely deformed endplate lesions were observed at the two lowermost disc spaces (Fig. 2) like most M1s.
Fig. 2.

On T1-weighted image, irregularity of the both endplates and defects in the adjacent subchondral bone surrounded by M1 type hypointensities in L5/S1 disc with decreased disc height and posterior bulge (arrow)
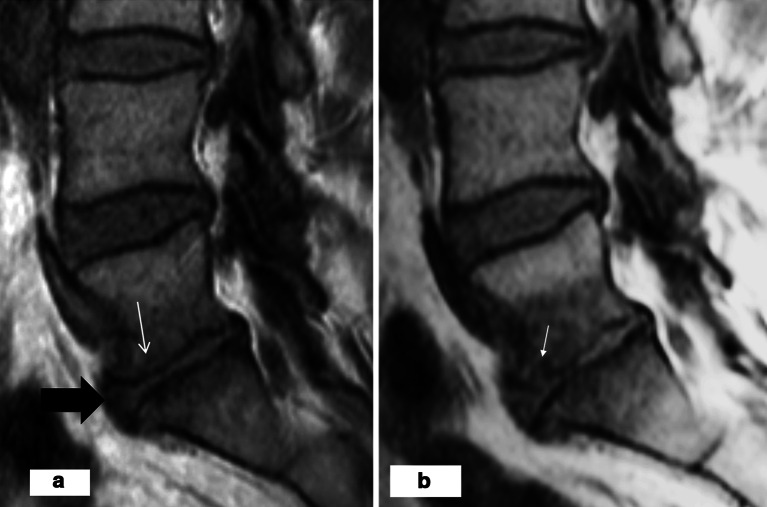
Posterior and anterior bulging/protrusion was observed in 100 (37%) and 93 (34%) of the 270 disc spaces, respectively. Fifty-one (51%) of the posterior and 47 (51%) of the anterior bulges were found in discs with an adjacent M1 (Fig. 3a).
Fig. 3.
a At baseline on T1-weighted image, the L5/S1 disc has a decreased disc height and an anterior bulge (broad arrow). There is an M1 type hypointensity adjacent to the both endplates and only slight depression in the upper endplate of L5/S1 disc (thin arrow). b After 1-year follow-up, on T1-weighted image, M1 has enlarged and there is still an anterior bulge. A focal defect has appeared in the upper endplate of L5/S1 disc anteriorly (arrow)
Disc height was normal in 169 (63%) of discs, slightly decreased in 58 (21%), decreased in 25 (9%), and severely decreased in 18(7%). Only 3 (0.6%) of M1s were found adjacent to a disc with a normal disc height.
Disc signal intensity was normal in 17%, slightly decreased in 35%, decreased in 35%, severely decreased in 3% and black/white in 10% of the 270 lumbar discs. None of the discs with a normal or only slightly decreased signal intensity had an adjacent M1 while 29% of discs with a decreased disc signal intensity and 93% of the 28 black/white discs had.
Follow-up
During the 1-year follow-up most of the M1s changed regarding their signal intensity, shape and size. 10 (9%) M1s remained constant, 44 (40%) increased, 49 (45%) decreased, seven (6%) disappeared, and 10 new M1s appeared. Sixty-four (58%) M1s converted partly and five (6%) totally to M2s, larger M1s more likely than small ones. 5/81 (6%) of M2s reconverted to M1s. Signal intensity did not change at the same pace in all regions and heterogeneous, mixed signal abnormalities were observed adjacent to most disc spaces after follow-up. In disc spaces with an absent or constant adjacent M1 a slighter progress of degenerative changes was noted than in those with a changing or appearing M1 (Tables 1, 2, 3, 4).
Table 1.
Modic 1 type change in relation to the change in endplate lesion after 1-year follow-up
| Presence/change of M1 | Presence/change of endplate lesions | |||
|---|---|---|---|---|
| no EPL | EPL constant | EPL increased/appeared | Total (n) | |
| no M1 | 329 | 83 | 8 | 420 |
| M1 constant | 0 | 5 | 5 | 10 |
| M1 increased/appeared | 4 | 16 | 34 | 54 |
| M1 decreased/disappeared | 4 | 24 | 28 | 56 |
| Total (n) | 337 | 128 | 75 | 540 |
EPL endplate lesion, M1 Modic 1 type change
p < 0.001 Pearson chi-sq
Table 2.
Modic 1 type change in the upper endplate in relation to the change of posterior bulge after 1-year follow-up
| Presence/change of M1 | Presence/change of posterior bulge | ||||
|---|---|---|---|---|---|
| no | Constant | Decreased | Increased/appeared | Total (n) | |
| no M1 | 166 | 39 | 2 | 2 | 209 |
| M1 constant | 0 | 2 | 1 | 2 | 5 |
| M1 increased/appeared | 0 | 22 | 2 | 4 | 28 |
| M1 decreased/disappeared | 2 | 22 | 0 | 4 | 28 |
| Total (n) | 168 | 85 | 5 | 12 | 270 |
M1 Modic 1 type change
Table 3.
Modic 1 type change in the upper endplate in relation to the change of disc height after 1-year follow-up
| Presence/change of M1 | Presence/change of disc height | |||
|---|---|---|---|---|
| Constantly normal | Constantly decreased | Further decreased | Total (n) | |
| no M1 | 157 | 39 | 13 | 209 |
| M1 constant | 0 | 1 | 4 | 5 |
| M1 increased/appeared | 1 | 9 | 18 | 28 |
| M1 decreased/disappeared | 1 | 15 | 12 | 28 |
| Total (n) | 159 | 64 | 47 | 270 |
M1 Modic 1 type change
p < 0.001 Pearson chi-sq
Table 4.
M1 in the upper endplate in relation to the change of disc signal intensity after 1-year follow-up
| Presence/change of M1 | Presence/change of disc signal intensity | ||||
|---|---|---|---|---|---|
| Constantly normal | Constantly decreased | Further decreased | Increased | Total (n) | |
| no M1 | 44 | 163 | 1 | 1 | 209 |
| M1 constant | 0 | 4 | 0 | 1 | 5 |
| M1 increased/appeared | 0 | 18 | 1 | 9 | 28 |
| M1 decreased/disappeared | 0 | 12 | 8 | 8 | 28 |
| Total (n) | 44 | 197 | 10 | 19 | 270 |
M1 Modic 1 type change
p < 0.001 Pearson chi-sq
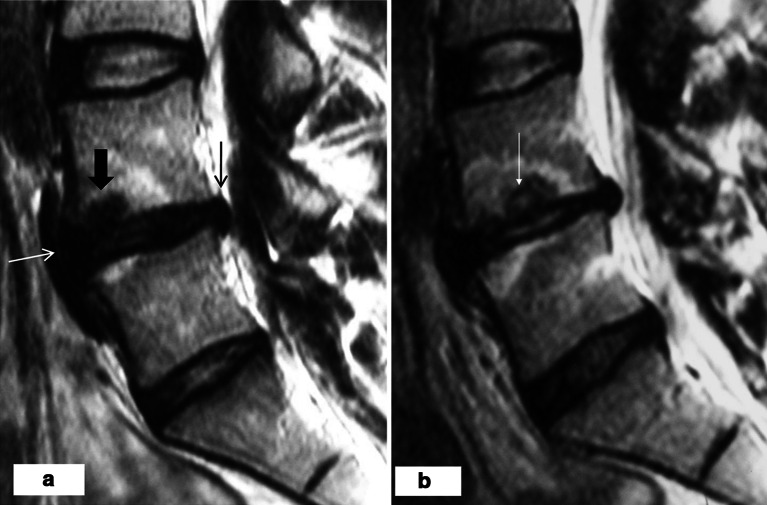
Endplate lesions increased in 70/203 (34%) endplates. In five normal endplates a new endplate lesion appeared. The unstable M1 was associated with an increasing or new endplate lesion (p < 0.001) (Table 1; Fig. 3b). Both an increase (Fig. 4) and a decrease (Fig. 5) of bulging or protrusion were found in discs with an adjacent unstable M1. The relation of the change in posterior disc bulging with that of M1 adjacent to the upper endplate is represented in Table 2. The results regarding anterior bulges and lower endplates were similar.
Fig. 4.
a At baseline, there is M1 adjacent to the both endplates and a bony endplate defect (broad arrow) adjacent to the upper endplate of L4/L5 disc on T2-weighted image. The L4/L5 disc has a posterior (thin black arrow) and anterior (thin white arrow) bulge and a decreased disc signal intensity. b After 1 year, both M1s and the endplate defect have enlarged (thin arrow). Disc height has decreased, anterior and posterior bulge have increased and the disc signal intensity has increased (turned to black/white)
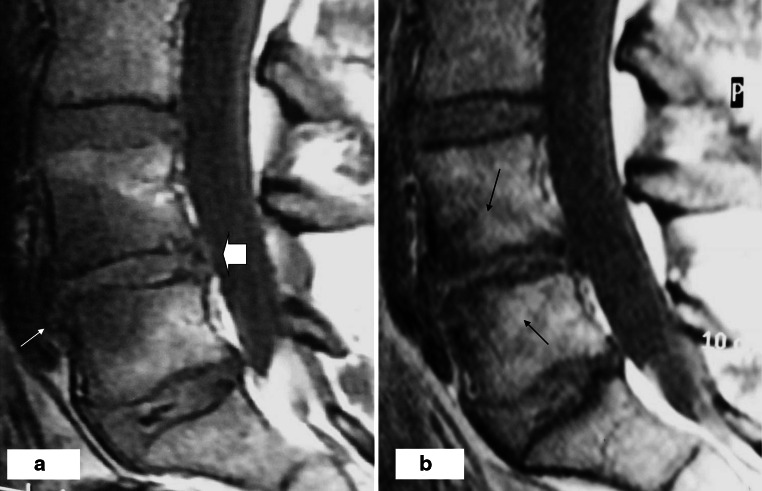
Fig. 5.
a On T1-weighted image at baseline, L4/L5 disc has an anterior (thin arrow) and posterior (thick arrow) extrusion and a large hypointensity (M1) in the subchondral bone adjacent to the both endplates with irregularities posteriorly. b After follow-up, M1 type hypointensity adjacent to the both endplates has changed into a mixed type (M1/M2) subchondral hypo- and hyperintensity (small arrows). Disc height and extrusions have decreased and irregularities of the endplates have increased
The change of M1 was associated with the decrease of disc height (Table 3, p < 0.001). 55% of unstable M1s were found adjacent to discs with a further decrease of disc height, whereas only 8% of discs with an absent or constant M1s were observed to have further decreased height. Newly appearing or increasing M1 was slightly more common than disappearing or decreasing M1 in disc spaces with a decreasing disc height (Figs. 3, 4, 5; Table 3), although the difference was not statistically significant.
Both an increase (Fig. 4b) and a decrease of disc signal intensity were detected more frequently with an M1 alteration than with an absent or constant M1 (Table 4, p < 0.001). Only dark or black discs converted to the black/white-type. Black/white discs persisted in 50% of the cases whereas in 25% the bright area in nucleus pulposus region enlarged and in 25% the disc signal intensity decreased. Increase of signal intensity was slightly more common than decrease in discs with an adjacent increasing or appearing M1 (Table 4; Fig. 4b) but the number of cases was too small to find a statistically significant difference.
Discussion
During a 1-year follow-up the progress of disc degeneration was associated with the presence and size alterations of M1. This study strengthens the view that M1 is a sign of a fast progressing and deforming “pathologic” disc degeneration, in contrast to the more slowly progressing partly genetically determined and partly age-dependent degeneration with different accompanying morphologic features.
Increase of disc signal intensity and endplate lesions were rare in disc spaces without M1. Such findings have not been included in MRI-based classification of disc degeneration. These features seem to be found only in disc spaces where a fast progressing degenerative process is found in association with M1.
The importance of the endplate for disc integrity is widely accepted [17–19]. The current study strengthens the earlier findings of bony endplate changes [11] being essential features of the degenerative process associated with Modic changes. When a Schmorl-type defect occurred simultaneously with an M1 most of them increased in line with an increase of M1. Half of the decreasing M1s were associated with increasing endplate lesions, too. It seems that any change of M1 is important in relation to increasing endplate lesions. However, one-third of the Schmorl lesions at baseline were observed without a M1. It is possible that they have a different etiology than those found with coexisting Modic lesions. Schmorl lesions are observed already in young people. The endplate lesions found at baseline without adjacent M1 in this study, may represent Schmorl lesions that have appeared far earlier. Modic et al. [5] described an increased number of osteoblasts and osteoclasts in subchondral bone of vertebrae with M1. Their activity could explain the progressing deforming changes in endplates and subchondral bone in association of M1.
Our study is in accordance with the previous ones in which Modic changes are detected in association with intervertebral disc degeneration, including disc herniations [7, 12, 20]. Most M1s at baseline were found in disc spaces with a decreased or slightly decreased disc height, decreased disc signal intensity and disc with bulging or protrusion.
In line with a previous study [12], we observed that an increase in disc signal intensity was related with changes in M1. A bright signal in nucleus pulposus of discs with advanced degeneration has been shown previously [14]. According to our study, an increase of disc signal intensity may be a relatively early change associated with the increase of M1 while a decrease of disc signal intensity may be more common when M1 decreases and turns to M2. Disc height decrease was found more often with an M1 increase than decrease suggesting that disc height begins to decrease in the early phase of the degenerative process related with M1, as was observed regarding increase of disc signal intensity, as well. Disc degeneration has been found to be associated with degenerative changes in facet joints and claimed to precede them. The decrease of disc height inevitably decreases also the height of the corresponding intervertebral foramens narrowing them. Instability, subluxation and deforming changes in the facet joints are suggested to follow [21] and subsequently lead to deformities in the nerve root foramen.
Our study cohort was selected from a large patient population with strict exclusion criteria. The composition of our sample was exceptional with the percentage of females being exceedingly high (83%). The reason for the observed female predominance remains unclear. Earlier studies have also shown a high prevalence of females in LBP patients with M1 [10] and the female gender as a potential predictor for Modic changes [6]. Therefore, it is possible that hormonal factors may also play some as yet unknown role in the development of Modic changes.
There are limitations in this study that need to be mentioned. Our baseline studies were performed with variable equipments and field strength. However, the similar MRI sequences were used in all participating hospitals and follow-up MRI studies were performed with a single 1.0 T equipment and with uniform protocol. Signal intensities were compared with those in other discs and vertebrae in the same study in addition to comparison between studies. The evaluation of the L5/S1 disc height was problematic due to the fact that L4/5 is normally the highest lumbar disc and the decrease could not be evaluated similarly to other disc spaces. Consensus reading was used to accept only definite changes, agreed by both readers, as positive findings. Blinded reading was not used although in comparing the baseline and follow-up images the expectation of changes may add the number of positive findings. However, in blinded reading the location of the heterogeneous signal changes and endplate changes to be followed may be difficult to determine because more than one lesion may be found in the endplate. Therefore, consensus reading comparing baseline and follow-up images was used for this study.
Despite the use of variable imagers at baseline MRI, we believe that differences in the imaging equipment did not significantly affect our results. If so, they would be likely to cause random error in results, to increase statistical “noise” and thus weaken the p values. Our study also lacked control subjects but the relationship between M1 and degenerative disc findings were analyzed using the disc spaces without M1 as internal controls. Due to different clinical presentations and imaging capacity, the interval between the appearance of the CLBP symptoms and the baseline MRI study was variable.
We think that it is important to recognize the signs of the process leading to disc disruption. The knowledge of the precise sequence of biologic events leading to deforming degenerative disc changes could offer some opportunity for intervention to suppress the progression of that process. The current study shows that the accelerated process of degeneration in discs with an adjacent M1 may lead to deforming changes in the discovertebral unit in a much shorter time than would be expected with age-dependent degeneration.
Conclusions
The present study indicates that even during 1-year follow-up unstable M1 coexists with an accelerated process of adjacent disc degeneration with decrease of the disc height, alteration of the signal intensity of the nucleus pulposus and deformation of the bony endplates. In contrast, degeneration in the absence of M1 seems to advance slowly, possibly according to the normal aging process. It may be useful to include subchondral signal changes and bony endplate changes in the MRI-classification of disc degeneration to differentiate between these two types of degenerative process.
Acknowledgments
This study was financially supported by Finska Läkaresällskapet.
Conflict of interest
None.
References
- 1.Braithwaite I, White J, Saifuddin A, Renton P, Taylor BA. Vertebral end-plate (Modic) changes on lumbar spine MRI: correlation with pain reproduction at lumbar discography. Eur Spine J. 1998;7:363–368. doi: 10.1007/s005860050091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Weishaupt D, Zanetti M, Hodler J, Min K, Fuchs B, Pfirrmann CW, Boos N. Painful lumbar disk derangement: relevance of endplate abnormalities at MR imaging. Radiology. 2001;218:420–427. doi: 10.1148/radiology.218.2.r01fe15420. [DOI] [PubMed] [Google Scholar]
- 3.Kuisma M, Karppinen J, Niinimäki J, Ojala R, Haapea M, Heliövaara M, Korpelainen R, Taimela S, Natri A, Tervonen O. Modic changes in endplates of lumbar vertebral bodies. Prevalence and association with low back and sciatic pain among middle-aged male workers. Spine. 2007;32:1116–1122. doi: 10.1097/01.brs.0000261561.12944.ff. [DOI] [PubMed] [Google Scholar]
- 4.Weishaupt D, Zanetti M, Hodler J, Boos N. MR imaging of the lumbar spine: prevalence of intervertebral disk extrusion and sequestration, nerve root compression, end plate abnormalities, and osteoarthritis of the facet joints in asymptomatic volunteers. Radiology. 1998;209:661–666. doi: 10.1148/radiology.209.3.9844656. [DOI] [PubMed] [Google Scholar]
- 5.Modic MT, Steinberg PM, Ross JS, Masaryk TJ, Carter JR. Degenerative disk disease: assessment of changes in vertebral body marrow with MR imaging. Radiology. 1988;166:193–199. doi: 10.1148/radiology.166.1.3336678. [DOI] [PubMed] [Google Scholar]
- 6.Jensen TS, Kjaer P, Korsholm L, Bendix T, Sorensen JS, Manniche C, Leboeuf-Yde C. Predictors of new vertebral endplate signal (Modic) changes in the general population. Eur Spine J. 2010;19:129–135. doi: 10.1007/s00586-009-1184-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kuisma M, Karppinen J, Niinimäki J, Kurunlahti M, Haapea M, Vanharanta H, Tervonen O. A three-year follow-up of lumbar spine endplate (Modic) changes. Spine. 2006;31:1714–1718. doi: 10.1097/01.brs.0000224167.18483.14. [DOI] [PubMed] [Google Scholar]
- 8.Albert HB, Manniche C. Modic changes following lumbar disc herniation. Eur Spine J. 2007;16:977–982. doi: 10.1007/s00586-007-0336-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Boos N, Weissbach S, Rohrbach H, Weiler C, Spratt KF, Nerlich AG. Classification of age-related changes in lumbar intervertebral discs: 2002 Volvo Award in basic science. Spine. 2002;27:2631–2644. doi: 10.1097/00007632-200212010-00002. [DOI] [PubMed] [Google Scholar]
- 10.Mitra D, Cassar-Pullicino VN, McCall IW. Longitudinal study of vertebral type-1 end-plate changes on MR of the lumbar spine. Eur Radiol. 2004;14:1574–1581. doi: 10.1007/s00330-004-2314-4. [DOI] [PubMed] [Google Scholar]
- 11.Luoma K, Vehmas T, Grönblad M, Kerttula L, Kääpä E. MRI follow-up of subchondral signal abnormalities in a selected group of chronic low back pain patients. Eur Spine J. 2008;17:1300–1308. doi: 10.1007/s00586-008-0716-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Luoma K, Vehmas T, Grönblad M, Kerttula L, Kääpä E. Relationship of Modic type 1 change with disc degeneration: a prospective MRI study. Skeletal Radiol. 2009;38:237–244. doi: 10.1007/s00256-008-0611-8. [DOI] [PubMed] [Google Scholar]
- 13.Pfirrmann CW, Metzdorf A, Zanetti M, Hodler J, Boos N. Magnetic resonance classification of lumbar intervertabral disc degeneration. Spine. 2001;26:1873–1878. doi: 10.1097/00007632-200109010-00011. [DOI] [PubMed] [Google Scholar]
- 14.Yu S, Haughton VM, Ho PS, Sether LA, Wagner M, Ho KC. Progressive and regressive changes in the nucleus pulposus. Part II: the adult. Radiology. 1988;169:93–97. doi: 10.1148/radiology.169.1.3420285. [DOI] [PubMed] [Google Scholar]
- 15.Gönen M, Panageas KS, Larson SM. Statistical issues in analysis of diagnostic imaging experiments with multiple observations per patient. Radiology. 2001;221:763–767. doi: 10.1148/radiol.2212010280. [DOI] [PubMed] [Google Scholar]
- 16.Killip S, Mahfoud Z, Pearce K. What is an intracluster correlation coefficient? Crucial concepts for primary care researchers. Ann Fam Med. 2004;2:204–208. doi: 10.1370/afm.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Adams MA, Freeman BJ, Morrison HP, Nelson IW, Dolan P. Mechanical initiation of intervertebral disc degeneration. Spine. 2000;25:1625–1636. doi: 10.1097/00007632-200007010-00005. [DOI] [PubMed] [Google Scholar]
- 18.Moore RJ. The vertebral end-plate: what do we know? Eur Spine J. 2000;9:92–96. doi: 10.1007/s005860050217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rajasekaran S, Babu JN, Arun R, Armstrong BR, Shetty AP, Murugan S. A study of diffusion in human lumbar discs: a serial magnetic resonance imaging study documenting the influence of the endplate on diffusion in normal and degenerate discs. Spine. 2004;29:2654–2667. doi: 10.1097/01.brs.0000148014.15210.64. [DOI] [PubMed] [Google Scholar]
- 20.Modic MT, Masaryk TJ, Ross JS, Carter JR. Imaging of degenerative disk disease. Radiology. 1988;168:177–186. doi: 10.1148/radiology.168.1.3289089. [DOI] [PubMed] [Google Scholar]
- 21.Jinkins JR. Acquired degenerative changes of the intervertebral segments at and suprajacent to the lumbosacral junction. A radioanatomic analysis of the nondiscal structures of the spinal column and perispinal soft tissues. Eur J Radiol. 2004;50:134–158. doi: 10.1016/j.ejrad.2003.10.014. [DOI] [PubMed] [Google Scholar]