Abstract
Little is known about the impact of indoor residual spraying (IRS) in areas with intense malaria transmission such as sub-Saharan Africa. In Malawi, IRS with lambda-cyhalothrin has been applied annually in an area of intense year-long transmission since 2007. We evaluated the impact of IRS on parasitemia and anemia prevalence in children less than five years of age by using a cross-sectional household survey conducted in 2009, six months after the second IRS spray round. We measured malaria parasitemia and anemia (hemoglobin level < 11 g/dL) in 899 children less than five years of age and used binomial regression to assess the impact of IRS by comparing children living in a household sprayed with IRS (direct IRS) with those in a household not sprayed with IRS, but in an IRS area (indirect IRS) and those living in a household not sprayed with IRS and not in an IRS area (no IRS). In the IRS area, 77% of households reported receiving IRS. Adjusting for bed net use, house construction, and socioeconomic status, we found that receiving direct IRS and indirect IRS were significantly associated with a 33% (95% confidence interval [CI] = 1–54%) and 46% (95% CI = 20–64%) reduction in parasitemia and a 21% (95% CI = 4–34%) and 30% (95% CI = 12–45%) reduction in anemia prevalence, respectively.
Introduction
Vector control interventions such as insecticide-treated nets (ITNs) and indoor residual spraying (IRS) are key malaria control tools.1,2 Both of these strategies depend on the action of insecticide either sprayed onto the wall or applied to the netting material to kill adult female anopheline mosquitoes either before or after a blood meal and reduce both the size and longevity of the adult mosquito population. In addition, some insecticides, including the synthetic pyrethroids, also repel mosquitoes from households, leading to reduced human-mosquito contact and increased mortality of adult mosquitos.3 Lastly, bed nets provide a physical barrier to human–mosquito contact. Both ITNs and IRS are most effective for endophagic and endophilic mosquito vectors, where they provide a community-wide effect; thus persons who do not receive personal protection can still benefit from these interventions in their communities.3–5
Insecticide-treated nets have proven effective in a large number of field trials and have been shown to reduce mortality in children 1–59 months of age, asexual malaria parasitemia, and anemia prevalence, as well as uncomplicated malaria and severe malaria incidence.6 In recent years, because of their proven efficacy and relative ease of implementation, ITNs have become the predominant vector control strategy in many malaria-endemic areas of the world.2
Evidence of IRS efficacy in reducing or interrupting malaria transmission has been available since the 1940s, and many historical examples of successful IRS programs exist from the Global Malaria Eradication Program era in the 1950s to 1970s.7 However, IRS was not a major component of malaria control as part of the Global Malaria Eradication Program or afterwards in areas with intense, year-round transmission such as most of sub-Saharan Africa. Much of our early data on IRS efficacy in areas with intense, year-long malaria transmission comes from relatively small scale demonstration projects and cluster randomized trials.8–12 More recently, IRS use has increased in sub-Saharan Africa and has had demonstrated impact in a number of different transmission settings.13–21 In 2006, the World Health Organization has revised its recommendation for the use of IRS and endorsed IRS use in all malaria transmission zones, including much of sub-Saharan Africa.1 Although some historical examples of IRS use in high transmission settings exist,9,10,22 there is a paucity of recent data on IRS use in areas of intense, year-long malaria transmission.
Although IRS and ITNs have been shown to be effective at reducing malaria transmission,6,11 there are unanswered questions about their relative efficacy8 and cost effectiveness,23–25 as well as the additional protection afforded by the combined use of IRS and ITNs.11,26 There are few head-to-head comparisons of ITN and IRS efficacy, and the data thus far are mixed. A review of six previously conducted studies across sub-Saharan Africa and South Asia concluded that IRS appeared more protective in one trial conducted in a stable malaria transmission area, but that ITNs seemed to be more protective than IRS in unstable transmission areas.11 In addition, there is relatively little information to guide program managers regarding the relative cost effectiveness of the two interventions given mixed results from previous studies.23–25 Moreover, we do not understand if there are additional benefits of combining IRS and ITNs. Thus far, field studies and mathematical models are far from conclusive and suggest variable effects dependent on epidemiologic conditions as well as choice of insecticide.11,26–28 Lastly, we do not fully understand the impact of insecticide resistance on IRS and ITNs and their relative efficacy in the face of resistance as well as their use as insecticide resistance management tools.29 Although randomized trials to address the role of ITNs versus IRS are being planned, observational data might provide some insights into the relative merits of each intervention individually versus their use in combination, especially in areas with intense, year-round transmission.
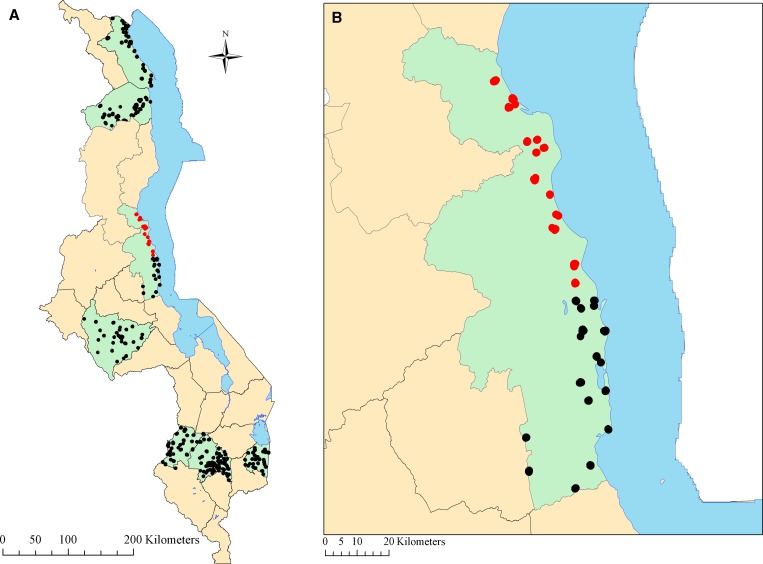
In Malawi, the National Malaria Control Program with funding from the U.S. President's Malaria Initiative initiated an IRS program administered by Research Triangle International (Research Triangle Park, NC) by using 10% lambda-cyhalothrin slow-release capsule suspension (ICON 10 CS(R); Syngenta AG, Basel, Switzerland) in the northern half of Nkhotakota District, an area of intense year-round transmission, in December 2007 (Figure 1).30 Nkhotakota District was chosen for IRS on the basis of intensity of malaria transmission and burden of disease, as well as the potential applicability to other rural areas in Malawi. In addition, the district was previously used for vector assessments, has distinct boundaries including Lake Malawi on the east and the national forest on the west, and had included IRS as a proposed activity in its District Implementation Plan. The first round of IRS occurred in December 2007 (28,227 structures) in a limited geographic area and the second round of IRS occurred in October–November of 2008 (42,044 structures) in an expanded geographic area; both IRS rounds were conducted only in the northern half of Nkhotakota.30 We assessed the impact of IRS on malaria parasitemia and anemia prevalence in children less than five years of age by using a cross-sectional household survey in the April 2009, approximately six months after the second IRS spray round.
Figure 1.
Districts and census enumeration areas included in the household survey in eight districts, Malawi 2009 (A) with a close-up of Nkhotakota District (B). Districts included in the household survey are shaded in green and sampled census enumeration areas with indoor residual spraying (IRS) for malaria control are indicated by red circles and those without IRS are indicated by black circles.
Materials and Methods
Study site.
Nkhotakota District is in the Central Region of Malawi and includes a main port on Lake Malawi. As of 2008, the district had a population of 303,659.31 Nkhotakota lies at an elevation of 1,548 feet (472 meters) above sea level and has year-round malaria transmission with a seasonal peak from December to June, after the main rainy season. Ninety-eight percent of malaria infections in Malawi are caused by Plasmodium falciparum. Anopheles gambiae complex and An. funestus are the primary malaria vectors.32 Malaria infection burden is high in Nkhotakota District; according to a pre-IRS household survey conducted in 2007, 72% of children 6–30 months of age had malaria parasitemia (Mathanga D, unpublished data). No published data on malaria endemicity in Nkhotakota District exist, but distribution and seasonality geospatial models suggest similar climate suitability to sustain endemic malaria transmission in the northern and southern halves of Nkhotkota District.33
Household survey.
Full details of data collection procedures have been described.34 In brief, we conducted a cross-sectional household survey during April 16–30, 2009, at the end of the long rains and in the middle of the high malaria transmission season in urban and rural communities of Nkhotakota District. We used a two-stage cluster sampling design. The first stage was composed of selecting 30 enumeration areas (EAs) by using systematic random sampling with selection probability proportional to size using the 1998 census. In the second stage, we divided the EA into segments of approximately 30–60 households and randomly selected a segment by using a personal digital assistant (PDA, Dell Axim X50s; Dell Inc., Austin, TX) with a specially designed program for random segment selection developed by the Centers for Disease Control and Prevention. On the basis of the estimated number of households in the EA or selected segment, all households or a randomly selected subset of households were invited to participate in the survey. Informed consent was obtained from the head of household to participate in the survey. All household members were asked to participate and those who agreed were asked standardized questions about themselves, as well as other members of the household. If no household members were at home, the household was revisited at the end of the day. If no one was available after two visits, the household was dropped from the survey and not replaced. Data were collected electronically by using a questionnaire designed and programmed into PDAs using Visual CE 11.0 (Syware Inc., Cambridge, MA).35 The questionnaire was designed in English and translated into the three main languages spoken in Malawi: Chichewa, Chitumbuka, and Chiyao. All questions were closed-ended, but the choices were not read to respondents. Responses were coded as other if the respondent's answer did not match any of the anticipated categories. Skip patterns, internal logic checks, and informational pop-up screens were programmed into the PDA-based survey to improve the ease and accuracy of data collection.
Laboratory procedures and treatment.
Parent or guardian consent was obtained for a finger prick blood sample for all children less than five years of age. A thick blood film was prepared and hemoglobin concentration was measured by using the Hemocue Hb 201+ Analyzer (Hemocue Inc., Cypress, CA). The thick blood smears were stained with Field's stain A and B (azure dye and eosin). Blood smears were considered negative if no parasites were found after counting 200 fields. Thick blood films were examined at central laboratory facilities located in each region. For quality control purposes, 10% of slides were re-examined by an expert microscopist at a reference laboratory in Blantyre, Malawi.
Hemoglobin results were shared with the parent or guardian at the time of the household visit. Children with hemoglobin levels < 8 g/dL were provided written results, given artemether-lumefantrine, albendazole (if > 24 months of age), an age appropriate two-week dosage of daily iron, and referred to a health facility. Children with a history of fever received immediate presumptive treatment of malaria with artemether-lumefantrine, according to Malawi national treatment guidelines. Children who had been treated with artemether-lumefantrine within the past two weeks, but remained febrile at the time of the survey, were treated with quinine. Children who were found to be seriously ill, as determined by the survey nurses, were provided transportation to the nearest health facility.
Definitions.
Given the known community-wide effect of IRS, we classified exposure to IRS in three categories: living in a household that had been sprayed with IRS as determined by self-report (direct IRS); living in a household that had not received IRS, but was in an IRS area (indirect IRS); and living in a household that had not received IRS and was not in an IRS area (no IRS). An ITN was defined as any long-lasting insecticide treated bed net, any bed net factory-treated with insecticide and obtained less than 12 months ago, or any bed net treated with insecticide less than 12 months ago. Bed net and ITN use were defined as reportedly sleeping under a bed net or ITN the previous night, respectively. In children less than five years of age, any anemia was defined as a hemoglobin level < 11 g/dL and moderate-to-severe anemia was defined as a hemoglobin level < 8 g/dL. Parasitemia was defined as presence of asexual P. falciparum parasites on a thick blood film.
Data analysis.
All responses were entered directly into a PDA database in the field. Data were downloaded into a relational database by using Access 2000 software (Microsoft Inc., Redmond, WA). Analyses were performed by using SAS version 9.2 (SAS Institute, Cary, NC) using the proc survey procedures, which uses the Taylor expansion method to account for cluster sampling and unequal selection probabilities. Binomial regression was performed using proc genmod, which uses generalized estimating equations to account for cluster sampling. Analyses were weighted, and weights equaled the inverse of the exact probability of selection. Percentages reported in this report reflect this weighting unless otherwise noted. Statistical significance was defined as a P value < 0.05.
As described,34 a relative index of household socioeconomic status was derived on the basis of 19 categorical variables using principal components analysis.36,37 The variables included were a combination of utilities (sources of water, light, toilet type), use of domestic workers, and ownership of assets (land, bicycle, motorcycle, car, oxcart, lamp, radio, television, telephone, cell phone, refrigerator, bed, sofa, table). Of note, house construction variables (floor, walls, and roofs) were not used because they might be independently associated with outcomes of interest such as parasitemia or anemia in children less than five years of age.
Using a binomial regression model, we assessed the impact of exposure to IRS (direct IRS and indirect IRS), ITN, and untreated bed net use, and the additional benefit of exposure to direct IRS and ITN use (an interaction term of direct IRS and ITN use) on asexual parasitemia and any anemia in children less than five years of age, adjusting for potential confounders including house construction variables (wall and roof) and socioeconomic status by wealth quintile. House construction variables and socioeconomic status were identified as potential confounders because they were significantly associated with the outcomes of interest and differentially distributed between IRS and non-IRS areas.
Ethics statement.
This study was approved by the University of Malawi College of Medicine (Blantyre, Malawi) ethical committee and the Centers for Disease Control and Prevention (Atlanta, GA) Institutional Review Board. Written informed consent was obtained from all adult participants and all parents or guardians of children.
Results
Characteristics of households and children less than five years of age.
We surveyed 884 households that contained 899 children less than five years of age in Nkhotakota District; 46% of households were located in an IRS area (Table 1 and Figure 1). Household characteristics such as ITN ownership were similar between households in IRS and non-IRS areas, except for house construction variables (wall or roof) and socioeconomic status. Households in non-IRS areas were significantly more likely to be in poorest quintile, and reside in a house with mud walls and a roof made of natural materials. Within the IRS area, 77% of households reported receiving IRS during the last spray season.
Table 1.
Characteristics of households in an IRS area and a non-IRS area in Nkhotakota District, Malawi, 2009 (n = 884)*
| Characteristic | Total (n = 884), no. (%, 95% CI) | IRS area (n = 410), no. (%, 95% CI) | Non-IRS area (n = 474), no. (%, 95% CI) | P† |
|---|---|---|---|---|
| Mean household size (95% CI) | 4.4 (4.1–4.7) | 4.6 (4.2–5.1) | 4.2 (3.8–4.5) | 0.08 |
| Mean no. sleeping spaces per household (95% CI) | 2.1 (2.0–2.2) | 2.2 (2.0–2.4) | 2.0 (1. 9–2.2) | 0.16 |
| Any bed net ownership | 601 (68, 63–74) | 287 (71, 64–77) | 314 (66, 58–74) | 0.34 |
| Insecticide-treated bed net ownership | 437 (50, 45–54) | 212 (52, 44–60) | 225 (47, 43–52) | 0.31 |
| IRS coverage | 313 (35, 20–51) | 313 (77, 68–86) | 0 | NA |
| Wall construction | 0.01 | |||
| Mud | 388 (43, 33–53) | 130 (31, 14–48) | 258 (54, 44–65) | |
| Brick/stone | 487 (56, 46–65) | 280 (69, 54–85) | 216 (46, 36–56) | |
| Roof construction | 0.02 | |||
| Natural materials | 678 (77, 65–89) | 255 (62, 38–86) | 423 (90, 82–97) | |
| Iron sheets | 206 (23, 11–36) | 155 (38, 14–62) | 51 (10, 3–18) | |
| Socioeconomic status by wealth quintile | 0.03 | |||
| Poorest | 266 (30, 22–38) | 79 (19, 9–29) | 187 (39, 31–48) | |
| Second | 188 (21, 16–26) | 72 (17, 11–23) | 116 (25, 18–31) | |
| Third | 157 (18, 14–21) | 78 (19, 13–24) | 79 (17, 13–21) | |
| Fourth | 143 (16, 13–20) | 82 (21, 17–25) | 61 (13, 9–16) | |
| Least poor | 130 (15, 6–24) | 99 (25, 9–40) | 31 (7, 3–13) |
IRS = indoor residual spraying; CI = confidence interval; NA = not applicable.
Statistical testing comparing IRS area and non-IRS-area using t-test or Wald chi-square test as appropriate.
Surveyed children less than five years of age residing in IRS and non-IRS areas had similar bed net and ITN use, but children residing in non-IRS areas were significantly more likely to be in poorest quintile, and reside in a house with mud walls and a roof made of natural materials (Table 2). Although the prevalence of fever in the past two weeks was similar among children less than five years of age residing in IRS and non-IRS areas (50% versus 50%; P = 0.99), the prevalence of malaria parasitemia (22% versus 42%; P < 0.001), any anemia (53% versus 72%; P = 0.002), and moderate-to-severe anemia (6% versus 11%; P = 0.05) were significantly lower in IRS versus non-IRS areas, respectively.
Table 2.
Characteristics of children less than five years of age who reside in an IRS area and a non-IRS area in Nkhotakota District, Malawi, 2009 (n = 899)*
| Characteristic | Total (n = 899), no. (%, 95% CI) | IRS area (n = 392), no. (%; 95% CI) | Non-IRS area (n = 507), no. (%; 95% CI) | P† |
|---|---|---|---|---|
| Resides in a home that received IRS | 298 (33, 18–49) | 298 (77, 67–86) | 0 | NA |
| Bed net use | ||||
| Slept under insecticide treated net (ITN) | 446 (49, 45–53) | 196 (51, 44–55) | 250 (49, 43–54) | 0.84 |
| Slept under untreated net | 177 (20, 15–25) | 71 (19, 13–25) | 106 (21, 13–29) | 0.67 |
| Resides in a home that received IRS and slept under an ITN | 146 (16, 8–24) | 146 (37, 31–44) | 0 | NA |
| Resides in a house with walls made of | 0.02 | |||
| Mud | 397 (44, 34–53) | 129 (31, 17–46) | 268 (53, 43–63) | |
| Brick/stone | 502 (56, 47–66) | 263 (69, 54–83) | 239 (47, 37–57) | |
| Resides in a house with roof made of | 0.02 | |||
| Natural materials | 719 (80, 69–91) | 256 (65, 43–86) | 463 (92, 87–97) | |
| Iron sheets | 180 (20, 9–31) | 136 (35, 14–57) | 44 (8, 3–13) | |
| Socioeconomic status by wealth quintile | 0.03 | |||
| Poorest | 286 (31, 24–39) | 80 (20, 9–30) | 206 (40, 32–48) | |
| Second | 197 (22, 17–27) | 72 (18, 11–25) | 125 (25, 18–32) | |
| Third | 161 (18, 14–22) | 72 (18, 11–26) | 89 (18, 13–22) | |
| Fourth | 135 (15, 11–19) | 73 (19, 13–26) | 62 (12, 8–16) | |
| Least poor | 120 (13, 5–22) | 95 (25, 8–41) | 25 (5, 1–9) | |
| Fever in past two weeks | 449 (50, 44–57) | 194 (50, 40–61) | 255 (50, 41–59) | 0.99 |
| Any parasitemia | 282 (33, 27–40) | 81 (22, 16–28) | 201 (42, 34–50) | < 0.001 |
| Any anemia (Hb level < 11 g/dL) | 530 (64, 57–70) | 200 (53, 46–61) | 330 (72, 64–79) | 0.002 |
| Any moderate-to severe-anemia (Hb level < 8 g/dL) | 69 (8, 6–11) | 21 (6, 3–8) | 48 (10, 7–14) | 0.05 |
IRS = indoor residual spraying; CI = confidence interval; NA = not applicable; ITN = insecticide-treated net; Hb = hemoglobin.
Statistical testing comparing IRS area and non-IRS-area using t-test or Wald chi-square test as appropriate.
Factors associated with parasitemia and anemia in children less than five years of age.
We assessed the association between living in a household that had received IRS (direct IRS), living in a household in an IRS area that had not received IRS (indirect IRS), and living in a non-IRS area with malaria parasitemia and any anemia in children less than five years of age in Nkhotakota District. Children less than five years of age who resided in households with either direct (adjusted prevalence ratio [aPR] = 0.67, 95% confidence interval [CI] = 0.46–0.99, P = 0.04) or indirect (aPR = 0.54, 95% CI = 0.36–0.80, P = 0.002) IRS were less likely to have malaria parasitemia, after adjusting for ITN and untreated bed net use, socioeconomic status, and house construction (Table 3). Untreated bed net and ITN use were not significantly associated with malaria parasitemia. In addition, children less than five years of age who resided in households with either direct (aPR = 0.79, 95% CI = 0.66–0.96, P = 0.02) or indirect (aPR = 0.70, 95% CI = 0.55–0.88, P = 0.003) IRS were less likely to have any anemia after adjusting for ITN and untreated bed net use, socioeconomic status, and house construction (Table 4). Insecticide-treated net use was not significantly associated with reduced anemia prevalence, but untreated bed net use had a modest protective effect (aPR = 0.88, 95% CI = 0.79–0.98, P = 0.02). In addition we explored the additional benefit of combining IRS and ITNs and found no significant association between combined IRS and ITN use and reduced parasitemia or anemia prevalence (Tables 3 and 4).
Table 3.
Factors associated with parasitemia in children less than five years of age in Nkhotakota District, Malawi, 2009 (n = 849)*
| Factor | Proportion with parasitemia, n/N (%) | Adjusted prevalence ratio (95% confidence interval) | P |
|---|---|---|---|
| IRS | |||
| Resides in a home that received IRS | 61/281 (22) | 0.67 (0.46–0.99) | 0.04 |
| Did not receive IRS but resides in an IRS area | 20/87 (22) | 0.54 (0.36–0.80) | 0.002 |
| No IRS | 201/280 (42) | Referent | Referent |
| Bed net use | |||
| Child slept under an ITN | 140/424 (32) | 0.98 (0.75–1.27) | 0.86 |
| Child slept under untreated bed net | 50/162 (31) | 0.97 (0.76–1.24) | 0.80 |
| Child did not sleep under any bed net | 92/263 (36) | Referent | Referent |
| Resides in a home that received IRS and slept under an ITN† | 27/140 (19) | 0.81 (0.56–1.19) | 0.29 |
| Resides in a house with walls made of | |||
| Mud | 151/380 (40) | 1.17 (0.95–1.44) | 0.14 |
| Brick/stone | 131/469 (28) | Referent | Referent |
| Resides in a house with roof made of | |||
| Natural materials | 250/685 (37) | 1.04 (0.73–1.46) | 0.84 |
| Iron sheets | 32/164 (19) | Referent | Referent |
| Socioeconomic status by wealth quintile | |||
| Poorest | 105/269 (39) | 1.47 (0.98–2.20) | 0.06 |
| Second | 71/192 (37) | 1.52 (1.01–2.29) | 0.04 |
| Third | 58/152 (39) | 1.72 (1.09–2.70) | 0.02 |
| Fourth | 30/123 (24) | 1.19 (0.71–2.00) | 0.52 |
| Least poor | 18/113 (15) | Referent | Referent |
IRS = indoor residual spraying; ITN = insecticide-treated net.
Interaction term of resides in a home that received IRS × slept under an ITN.
Table 4.
Factors associated with any anemia (hemoglobin level < 11 g/dL) in children less than five years of age in Nkhotakota District, Malawi, 2009 (n = 835)*
| Variable | Proportion with any anemia, no. (%) | Adjusted prevalence ratio (95% confidence interval) | P |
|---|---|---|---|
| IRS | |||
| Resides in a home that received IRS | 156/284 (55) | 0.79 (0.66–0.96) | 0.02 |
| Did not receive IRS but resides in an IRS area | 44/89 (48) | 0.70 (0.55–0.88) | 0.003 |
| No IRS | 330/462 (72) | Referent | Referent |
| Bed net use | |||
| Child slept under an ITN | 269/416 (64) | 0.98 (0.87–1.10) | 0.69 |
| Child slept under untreated bed net | 93/163 (57) | 0.88 (0.79–0.98) | 0.02 |
| Child did not sleep under any bed net | 168/256 (66) | Referent | Referent |
| Resides in a home that received IRS and slept under an ITN† | 81/139 (58) | 1.10 (0.90–1.36) | 0.35 |
| Resides in a house with walls made of | |||
| Mud | 245/368 (67) | 0.95 (0.84–1.07) | 0.38 |
| Brick/stone | 285/467 (61) | Referent | Referent |
| Resides in a house with roof made of | |||
| Natural materials | 450/672 (67) | 1.09 (0.93–1.28) | 0.31 |
| Iron sheets | 80/163 (48) | Referent | Referent |
| Socioeconomic status by wealth quintile | |||
| Poorest | 185/266 (70) | 1.30 (1.03–1.64) | 0.03 |
| Second | 133/184 (72) | 1.38 (1.12–1.71) | 0.003 |
| Third | 93/148 (63) | 1.27 (1.00–1.62) | 0.05 |
| Fourth | 68/124 (55) | 1.14 (0.91–1.43) | 0.25 |
| Least poor | 51/113 (43) | Referent | Referent |
IRS = indoor residual spraying; ITN = insecticide-treated net.
Interaction term of resides in a home that received IRS × slept under an ITN.
Discussion
Indoor residual spraying is increasingly being adopted as a vector control strategy in areas of intense, year-round transmission in sub-Saharan Africa. We present the direct and indirect impact of an IRS program using lambda-cyhalothrin in an area of intense malaria transmission in Malawi. Compared with areas of the same district with similar transmission potential, children who lived in an IRS area (regardless of whether the house that they lived in was sprayed) had lower prevalence of malaria parasitemia and anemia, even after adjusting for bed net use, socioeconomic status, and house construction. These findings suggest that one round of IRS per year might reduce malaria infection burden and anemia in areas with potential for endemic malaria transmission.
However, the impact of yearly IRS in this area needs to be interpreted with caution because the survey was conducted six months after the second spray round. Indoor residual spraying in Nkhotakota District reduced An. gambiae complex and An. funestus populations as measured by window exit traps and pyrethrum spray catches (Mzilahowa T, unpublished data). In addition, residual insecticide activity was noted for at least six months after the spray round using wall cone bioassays with susceptible Kisumu strain An. gambiae s.s. Given the maximum expected residual half-life on sprayed surfaces of lambda-cyhalothrin of approximately six months, we would expect to have some effect of IRS on vector control at the time of the survey, approximately six months after the second spray round. However, given the timing of the survey, we cannot determine if a single round of IRS was sufficient to maintain adequate vector control for the whole year.
In this study, we were able to detect a community-wide effect of IRS; all children who resided in the IRS area were less likely to have malaria parasitemia or anemia regardless of whether they lived in a household that had received IRS. This finding is consistent with our understanding of how IRS works and suggests that a community-wide effect can be detected even with IRS coverage of only 77% of households in a community. Whether this degree of coverage is adequate in all settings needs to be studied further.
In our analysis, we did not find an association between ITN or untreated bed net use and either reduced malaria parasitemia or anemia prevalence in children less than five years of age. We might not detect an impact of bed net use on disease burden because of our relatively small sample size. Of note, in a larger analysis using data from eight districts in Malawi, we found a significant association between ITN use and reduced parasitemia and anemia prevalence.34 Although we explored the potential impact of the combined use of IRS and ITNs using an interaction term in our model (received IRS × slept under an ITN), we did not have a sufficiently large sample size to detect any additional benefit. This finding is an area of increased interest to the global malaria control community and deserves further study using rigorous field trials.
The U.S. President's Malaria Initiative expanded support for IRS to all of Nkhotakota District (74,772 structures) in November 2009 and to all of Nkhotakota and adjacent Salima Districts in November 2010 (approximately 163,000 structures).30 In addition, the National Malaria Control Program with the use of Government of Malawi funds has initiated an IRS program in five additional districts in the lakeside and Shire Valley, including Nkhata Bay, Nsanje, Mangochi, Karonga, and Chikwawa Districts, which will cover an additional 650,000 houses and approximately 2.7 million persons.30 Although results of this analysis suggest a substantial impact of IRS using lambda-cyhalothrin on parasitemia and anemia prevalence, recent reports of pyrethroid resistance in A. funestus in Nkhotakota District (Mzilahowa T, unpublished data) and Likoma Island, Malawi,38 phenotypically similar to pyrethroid-resistant An. funestus in other parts of southern Africa,39–43 are concerning and could undermine the efficacy of IRS with pyrethroids and ITNs. In response to these reports, the IRS program in Nkhotakota and Salima Districts has replaced pyrethroids with pirimiphos-methyl, an organophosphate. Close monitoring of the IRS program in Malawi will be needed to assess the impact of insecticide resistance on IRS efficacy and to guide the choice of insecticide for future spray rounds.
Given the cross-sectional nature of this household survey, we were unable to control or even measure all known confounders between the IRS and non-IRS areas of Nkhotakota District. There were some differences between northern and southern Nkhotakota in socioeconomic status, which could potentially affect the prevalence of malaria in each area. However, the northern and southern parts of the district have similar climate suitability to maintain malaria transmission and had similar levels of malaria parasitemia and anemia prevalence in 2007 before the introduction of IRS. To mitigate potential differences between the two study areas, we used an adjusted analysis to control for socioeconomic status, house construction, and the use of malaria control interventions such as ITNs. In addition, we were hampered by a relatively small sample size and thus unable to further explore the impact of bed nets either alone or in combination with IRS on parasitemia and anemia in children less than five years of age.
In this cross-sectional household survey, IRS was associated with reduced malaria parasitemia and anemia prevalence in children less than five years of age. This study supports the use of IRS in areas with intense transmission such as much of sub-Saharan Africa, but should be interpreted with caution. The impact of IRS is dependent on the use of an effective insecticide, adequate coverage, and a sufficient number of spray rounds per year to adequately reduce vector populations. The study found no impact of ITNs, regardless of whether a child lived in an area covered by IRS. However, previous work in Malawi has shown a clear protective effect from ITNs34 and a larger sample size may be required to address this specific question. As Malawi expands its IRS program and faces the threat of the spread pyrethroid-resistant An. funestus, close monitoring is needed to ensure that IRS continues to be effective.
ACKNOWLEDGMENTS
We thank the many investigators, data collectors, and field supervisors who contributed to the collection and analyses of these data, and the community members who participated in or otherwise contributed to these studies.
Disclaimer: The findings and conclusions presented in this manuscript are those of the authors and do not necessarily reflect the official position of the U.S. President's Malaria Initiative, United States Agency for International Development, or U.S. Centers for Disease Control and Prevention.
Footnotes
Financial support: This study was supported by the United States President's Malaria Initiative, U.S. Agency for International Development, under the terms of an Interagency Agreement with the Centers for Disease Control and Prevention (CDC) and through a Cooperative Agreement (Number 5 U01 CI000189) between the CDC and the Malaria Alert Centre, College of Medicine. The sponsor of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all of the data in the study and had final responsibility for the decision to submit for publication.
Disclosure: The authors declare that they have no competing interests.
Authors' addresses: Jacek Skarbinski, Adam Wolkon, Alison Smith, John Gimnig, and Carl Campbell, Malaria Branch, Centers for Disease Control and Prevention, Atlanta, GA, E-mails: jskarbinski@cdc.gov, aow5@cdc.gov, alison.smith01@gmail.com, hzg1@cdc.gov, and ccamp@uga.edu. Dyson Mwandama, Madalitso Luka, James Jafali, Themba Mzilahowa, and Don P. Mathanga, Malaria Alert Centre, Chichiri, Blantyre, Malawi, E-mails: dmwandama@mac.medcol.mw, mluka@mac.medcol.mw, jjafali@gmail.com, tmzilahowa@mlw.medcol.mw, and dmathang@mac.medcol.mw. John Chiphwanya and Doreen Ali, National Malaria Control Programme, Ministry of Health, Lilongwe, Malawi, E-mails: johnchiphwanya@yahoo.co.uk and alidoreen@yahoo.com.
References
- 1.World Health Organization, Global Malaria Programme . Indoor Residual Spraying: Use of Indoor Residual Spraying for Scaling Up Global Malaria Control and Elimination. Geneva: World Health Organization; 2006. [Google Scholar]
- 2.World Health Organization, Global Malaria Programme . Insecticide-Treated Mosquito Nets: A Position Statement. Geneva: World Health Organization; 2007. [Google Scholar]
- 3.Killeen GF, Smith TA, Ferguson HM, Mshinda H, Abdulla S, Lengeler C, Kachur SP. Preventing childhood malaria in Africa by protecting adults from mosquitoes with insecticide-treated nets. PLoS Med. 2007;4:e229. doi: 10.1371/journal.pmed.0040229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Binka FN, Indome F, Smith T. Impact of spatial distribution of permethrin-impregnated bed nets on child mortality in rural northern Ghana. Am J Trop Med Hyg. 1998;59:80–85. doi: 10.4269/ajtmh.1998.59.80. [DOI] [PubMed] [Google Scholar]
- 5.Hawley WA, Phillips-Howard PA, ter Kuile FO, Terlouw DJ, Vulule JM, Ombok M, Nahlen BL, Gimnig JE, Kariuki SK, Kolczak MS, Hightower AW. Community-wide effects of permethrin-treated bed nets on child mortality and malaria morbidity in western Kenya. Am J Trop Med Hyg. 2003;68:121–127. [PubMed] [Google Scholar]
- 6.Lengeler C. Insecticide-treated bed nets and curtains for preventing malaria. Cochrane Database Syst Rev. 2004 doi: 10.1002/14651858.CD000363.pub2. CD000363. [DOI] [PubMed] [Google Scholar]
- 7.Najera JA, Gonzalez-Silva M, Alonso PL. Some lessons for the future from the Global Malaria Eradication Programme (1955–1969) PLoS Med. 2011;8:e1000412. doi: 10.1371/journal.pmed.1000412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Curtis CF, Mnzava AE. Comparison of house spraying and insecticide-treated nets for malaria control. Bull World Health Organ. 2000;78:1389–1400. [PMC free article] [PubMed] [Google Scholar]
- 9.Molineaux L, Gramiccia G. The Garki Project. Research on the Epidemiology and Control of Malaria in the Sudan Savanna of West Africa. Geneva: World Health Organization; 1980. [Google Scholar]
- 10.Matola YG, Magayuka SA. Malaria in the Pare area of Tanzania. V. Malaria 20 years after the end of residual insecticide spraying. Trans R Soc Trop Med Hyg. 1981;75:811–813. doi: 10.1016/0035-9203(81)90418-1. [DOI] [PubMed] [Google Scholar]
- 11.Pluess B, Tanser FC, Lengeler C, Sharp BL. Indoor residual spraying for preventing malaria. Cochrane Database Syst Rev. 2010 doi: 10.1002/14651858.CD006657.pub2. CD006657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Curtis CF, Maxwell CA, Finch RJ, Njunwa KJ. A comparison of use of a pyrethroid either for house spraying or for bednet treatment against malaria vectors. Trop Med Int Health. 1998;3:619–631. doi: 10.1046/j.1365-3156.1998.00281.x. [DOI] [PubMed] [Google Scholar]
- 13.Protopopoff N, Van Bortel W, Marcotty T, Van Herp M, Maes P, Baza D, D'Alessandro U, Coosemans M. Spatial targeted vector control in the highlands of Burundi and its impact on malaria transmission. Malar J. 2007;6:158. doi: 10.1186/1475-2875-6-158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Protopopoff N, Van Bortel W, Marcotty T, Van Herp M, Maes P, Baza D, D'Alessandro U, Coosemans M. Spatial targeted vector control is able to reduce malaria prevalence in the highlands of Burundi. Am J Trop Med Hyg. 2008;79:12–18. [PubMed] [Google Scholar]
- 15.Protopopoff N, Van Herp M, Maes P, Reid T, Baza D, D'Alessandro U, Van Bortel W, Coosemans M. Vector control in a malaria epidemic occurring within a complex emergency situation in Burundi: a case study. Malar J. 2007;6:93. doi: 10.1186/1475-2875-6-93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kleinschmidt I, Sharp B, Benavente LE, Schwabe C, Torrez M, Kuklinski J, Morris N, Raman J, Carter J. Reduction in infection with Plasmodium falciparum one year after the introduction of malaria control interventions on Bioko Island, Equatorial Guinea. Am J Trop Med Hyg. 2006;74:972–978. [PubMed] [Google Scholar]
- 17.Sharp B, van Wyk P, Sikasote JB, Banda P, Kleinschmidt I. Malaria control by residual insecticide spraying in Chingola and Chililabombwe, Copperbelt Province, Zambia. Trop Med Int Health. 2002;7:732–736. doi: 10.1046/j.1365-3156.2002.00928.x. [DOI] [PubMed] [Google Scholar]
- 18.Sharp BL, Kleinschmidt I, Streat E, Maharaj R, Barnes KI, Durrheim DN, Ridl FC, Morris N, Seocharan I, Kunene S, La Grange JJ, Mthembu JD, Maartens F, Martin CL, Barreto A. Seven years of regional malaria control collaboration–Mozambique, South Africa, and Swaziland. Am J Trop Med Hyg. 2007;76:42–47. [PMC free article] [PubMed] [Google Scholar]
- 19.Sharp BL, Ridl FC, Govender D, Kuklinski J, Kleinschmidt I. Malaria vector control by indoor residual insecticide spraying on the tropical island of Bioko, Equatorial Guinea. Malar J. 2007;6:52. doi: 10.1186/1475-2875-6-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Beier JC. Malaria control in the highlands of Burundi: an important success story. Am J Trop Med Hyg. 2008;79:1–2. [PMC free article] [PubMed] [Google Scholar]
- 21.Nyarango PM, Gebremeskel T, Mebrahtu G, Mufunda J, Abdulmumini U, Ogbamariam A, Kosia A, Gebremichael A, Gunawardena D, Ghebrat Y, Okbaldet Y. A steep decline of malaria morbidity and mortality trends in Eritrea between 2000 and 2004: the effect of combination of control methods. Malar J. 2006;5:33. doi: 10.1186/1475-2875-5-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kolaczinski K, Kolaczinski J, Kilian A, Meek S. Extension of indoor residual spraying for malaria control into high transmission settings in Africa. Trans R Soc Trop Med Hyg. 2007;101:852–853. doi: 10.1016/j.trstmh.2007.04.003. [DOI] [PubMed] [Google Scholar]
- 23.Yukich JO, Lengeler C, Tediosi F, Brown N, Mulligan JA, Chavasse D, Stevens W, Justino J, Conteh L, Maharaj R, Erskine M, Mueller DH, Wiseman V, Ghebremeskel T, Zerom M, Goodman C, McGuire D, Urrutia JM, Sakho F, Hanson K, Sharp B. Costs and consequences of large-scale vector control for malaria. Malar J. 2008;7:258. doi: 10.1186/1475-2875-7-258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guyatt HL, Kinnear J, Burini M, Snow RW. A comparative cost analysis of insecticide-treated nets and indoor residual spraying in highland Kenya. Health Policy Plan. 2002;17:144–153. doi: 10.1093/heapol/17.2.144. [DOI] [PubMed] [Google Scholar]
- 25.Goodman CA, Mnzava AE, Dlamini SS, Sharp BL, Mthembu DJ, Gumede JK. Comparison of the cost and cost-effectiveness of insecticide-treated bednets and residual house-spraying in KwaZulu-Natal, South Africa. Trop Med Int Health. 2001;6:280–295. doi: 10.1046/j.1365-3156.2001.00700.x. [DOI] [PubMed] [Google Scholar]
- 26.Kleinschmidt I, Schwabe C, Shiva M, Segura JL, Sima V, Mabunda SJ, Coleman M. Combining indoor residual spraying and insecticide-treated net interventions. Am J Trop Med Hyg. 2009;81:519–524. [PMC free article] [PubMed] [Google Scholar]
- 27.Yakob L, Dunning R, Yan G. Indoor residual spray and insecticide-treated bednets for malaria control: theoretical synergisms and antagonisms. J R Soc Interface. 2011;8:799–806. doi: 10.1098/rsif.2010.0537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chitnis N, Schapira A, Smith T, Steketee R. Comparing the effectiveness of malaria vector-control interventions through a mathematical model. Am J Trop Med Hyg. 2010;83:230–240. doi: 10.4269/ajtmh.2010.09-0179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ranson H, N'Guessan R, Lines J, Moiroux N, Nkuni Z, Corbel V. Pyrethroid resistance in African anopheline mosquitoes: what are the implications for malaria control? Trends Parasitol. 2011;27:91–98. doi: 10.1016/j.pt.2010.08.004. [DOI] [PubMed] [Google Scholar]
- 30.President's Malaria Initiative President's Malaria Initiative Malaria Operational Plan Malawi FY 2011. 2011. http://www.fightingmalaria.gov/countries/mops/fy11/malawi_mop-fy11.pdf Available at: Accessed April 4, 2011.
- 31.National Statistical Office (NSO) Malawi . 2008 Population and Housing Census. Zomba, Malawi: National Statistical Office (NSO) Malawi; 2009. [Google Scholar]
- 32.Mathanga DP, Walker ED, Wilson ML, Ali D, Taylor TE, Laufer MK. Malaria control in Malawi: current status and directions for the future. Acta Trop. 2012;121:212–217. doi: 10.1016/j.actatropica.2011.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mapping Malaria Risk in Africa (MAP MARA) Map of the Distribution of Endemic Malaria and Duration of Transmission Season in Malawi. http://www.mara.org.za/ Available at. Accessed October 6, 2011.
- 34.Skarbinski J, Mwandama D, Luka M, Jafali J, Wolkon A, Townes D, Campbell C, Zoya J, Ali D, Mathanga DP. Impact of health facility-based insecticide treated bednet distribution in Malawi: progress and challenges towards achieving universal coverage. PLoS ONE. 2011;6:e21995. doi: 10.1371/journal.pone.0021995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vanden Eng JL, Wolkon A, Frolov AS, Terlouw DJ, Eliades MJ, Morgah K, Takpa V, Dare A, Sodahlon YK, Doumanou Y, Hawley WA, Hightower AW. Use of handheld computers with global positioning systems for probability sampling and data entry in household surveys. Am J Trop Med Hyg. 2007;77:393–399. [PubMed] [Google Scholar]
- 36.Filmer D, Pritchett LH. Estimating wealth effects without expenditure data–or tears: an application to educational enrollments in states of India. Demography. 2001;38:115–132. doi: 10.1353/dem.2001.0003. [DOI] [PubMed] [Google Scholar]
- 37.McKenzie DJ. Measuring inequality with asset indicators. J Popul Econ. 2005;18:229–260. [Google Scholar]
- 38.Hunt R, Edwardes M, Coetzee M. Pyrethroid resistance in southern African Anopheles funestus extends to Likoma Island in Lake Malawi. Parasit Vectors. 2010;3:122. doi: 10.1186/1756-3305-3-122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Brooke BD, Kloke G, Hunt RH, Koekemoer LL, Temu EA, Taylor ME, Small G, Hemingway J, Coetzee M. Bioassay and biochemical analyses of insecticide resistance in southern African Anopheles funestus (Diptera: Culicidae) Bull Entomol Res. 2001;91:265–272. doi: 10.1079/ber2001108. [DOI] [PubMed] [Google Scholar]
- 40.Casimiro S, Coleman M, Mohloai P, Hemingway J, Sharp B. Insecticide resistance in Anopheles funestus (Diptera: Culicidae) from Mozambique. J Med Entomol. 2006;43:267–275. doi: 10.1603/0022-2585(2006)043[0267:iriafd]2.0.co;2. [DOI] [PubMed] [Google Scholar]
- 41.Casimiro SL, Hemingway J, Sharp BL, Coleman M. Monitoring the operational impact of insecticide usage for malaria control on Anopheles funestus from Mozambique. Malar J. 2007;6:142. doi: 10.1186/1475-2875-6-142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cuamba N, Morgan JC, Irving H, Steven A, Wondji CS. High level of pyrethroid resistance in an Anopheles funestus population of the Chokwe District in Mozambique. PLoS ONE. 2010;5:e11010. doi: 10.1371/journal.pone.0011010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hargreaves K, Koekemoer LL, Brooke BD, Hunt RH, Mthembu J, Coetzee M. Anopheles funestus resistant to pyrethroid insecticides in South Africa. Med Vet Entomol. 2000;14:181–189. doi: 10.1046/j.1365-2915.2000.00234.x. [DOI] [PubMed] [Google Scholar]