Background: Ligation of the glucocorticoid-induced tumor necrosis (TNF) receptor (GITR) regulates T suppressor cell activity.
Results: Regulation of JNK phosphorylation following GITR ligation plays a central role in the suppressive activity of T regulatory cells.
Conclusion: Inhibition of JNK phosphorylation modulates T regulatory effector cell function in vitro and in vivo.
Significance: Identification of JNK phosphorylation as a regulator of T suppressor cell function.
Keywords: Asthma, Cell Signaling, Cellular Immune Response, Jun N-terminal Kinase (JNK), MAP Kinases (MAPKs), CD4+CD25+Foxp3+ T Regulatory Cells, GITR, SP600125
Abstract
Naturally occurring Foxp3+CD4+CD25+ T regulatory cell (nTreg)-mediated suppression of lung allergic responses is abrogated following ligation of glucocorticoid-induced tumor necrosis receptor (GITR) family-related protein. In vitro stimulation of nTregs with GITR ligand increased phosphorylation of c-Jun N-terminal kinase (JNK) but not extracellular signal-regulated protein kinase (ERK) or p38 MAPK. SP600125, a known JNK inhibitor, prevented GITR-mediated phosphorylation of JNK. Activation of JNK was associated with increases in the upstream mitogen-activated protein kinase kinase 7 (MKK7) and the downstream transcription factor NF-κβ. Phosphorylated c-Jun (p-c-Jun), indicative of the activation of JNK, was detected in the immunoprecipitates of nTregs from wild-type but not JNK- or GITR-deficient mice. Treatment with an inhibitor of JNK phosphorylation resulted in complete reversal of all GITR-induced changes in nTreg phenotype and function, with full restoration of suppression of in vivo lung allergic responses and in vitro proliferation of activated CD4+CD25− T cells. Thus, regulation of JNK phosphorylation plays a central role in T regulatory cell function with therapeutic implications for the treatment of asthma and autoimmune diseases.
Introduction
Glucocorticoid-induced tumor necrosis factor receptor (GITR)2 family-related protein, also know as TNFRSF18, was first described in mice as a dexamethasone-inducible molecule in T cells (1), and subsequently the human equivalent was defined (2). GITR is a type 1 transmembrane protein with an extracellular N terminus and cytoplasmic C terminus, and it shares significant homology in the C-terminal domain with other members of the TNF receptor family, including 4-1BB, CD27, CD40, and OX40 (1, 2). Signaling cascades triggered through members of the TNF receptor superfamily influence many physiologic and pathologic immune responses by differentially regulating proliferation, differentiation, survival, and functions of cells in both the innate and adaptive immune systems (3). Specifically in the lung, GITR was shown to play a role in acute inflammation (4) and airway hyper-responsiveness (AHR) (5). Constitutive expression and function of GITR in CD4+CD25+ T regulatory cells (Tregs) have been described (6, 7). Stimulation of GITR on T regulatory cells (nTregs) by either GITR ligand (GITRL) or an agonistic antibody (DTA-1) abolished suppression of allergen-induced lung allergic responses (8), in contrast to rendering effector cells resistant to the suppressive activities of Tregs (9–11).
In both humans and experimental animal models, allergic asthma is an inflammatory disease of the airways characterized by an increase in AHR, airway inflammation, Th2 cytokine skewing, goblet cell metaplasia, excessive mucus production, elevated antigen-specific IgE, and structural remodeling of the airways. Increasingly, nTregs have been shown to be important in modifying the development and outcome of lung allergic diseases (12, 13) through both antigen-specific (14) and antigen-nonspecific mechanisms (15). However, the phenotypic and functional stability of nTregs has been shown to depend on a number of factors, including the expression of the key transcription factor, Foxp3 (16). Mutations of Foxp3 are associated with multiorgan autoimmune disease in Scurfy mice (17) and immune dysregulation, polyendocrinopathy, enteropathy, and X-linked syndrome in humans (18). Cytokines such as IL-6 (19) and the expression of CD8 have also been shown to alter nTreg function (13, 15). Importantly, in the absence and/or interference with MHC-I/CD8 interactions, regulatory functions were subverted, resulting in the loss of suppression. Indeed, under these conditions nTregs were converted into pathogenic effector cells, enhancing lung allergic responses in sensitized and challenged mice (8). Furthermore, the maintenance of suppressive activities in peripheral tissues and the regulation of endogenous IL-6 production by nTregs were shown to be dependent on the presence of CD8+ T cells (19). Conversion of Tregs to pathogenic effector cells has now been described in other animal models (20, 21).
In contrast to earlier studies (6, 7), host GITRL was demonstrated to effectively abrogate suppression in vivo, associated with the pathogenic conversion of nTregs (8). Moreover, enhancement of lung allergic responses was prevented by in vivo administration of anti-GITRL antibody prior to the transfer of nTregs, identifying the importance of GITRL-GITR signaling in controlling the phenotype of these cells (8). Although various signaling cascades through GITR have been described in T cells (22–24) and macrophages (25), there are little or no data linking GITR signal transduction pathways to the loss of suppression and phenotypic conversion of nTregs.
In this study, we defined the signaling pathways activated through GITR in nTregs in vitro and in vivo and determined the impact of interfering with activation on the expression of surface receptors, production of immunosuppressive cytokines, suppression of proliferation of activated T cells, and the consequences in the airways of allergen-sensitized and challenged mice.
EXPERIMENTAL PROCEDURES
Animals
Pathogen-free, 6–8-week-old female C57BL/6 and JNK−/− mice were obtained from The Jackson Laboratory (Bar Harbor, ME). GITR−/− mice were kindly provided by Dr. Carlo Riccardi (Perugia, Italy). All mice were maintained on an OVA-free diet, and all protocols were approved by the Institutional Animal Care and Use Committee of National Jewish Health.
Sensitization and Challenge
Sensitization was carried out by intraperitoneal injection of 20 μg of OVA (Sigma) emulsified in 2.25 mg of alum hydroxide (AlumImject; Pierce) in a total volume of 100 μl on days 1 and 14. Sensitized and challenged mice, denoted OVA/OVA, and nonsensitized but challenged littermates (PBS/OVA) received aerosol challenges for 20 min each day on 3 consecutive days (days 26, 27, and 28) with 1% OVA in PBS using an ultrasonic nebulizer (Omron, Vernon Hills, IL) (10).
Measurement of Airway Responsiveness
Airway responsiveness was assessed 48 h following the last challenge as a change in airway function to increasing concentrations of aerosolized methacholine administered for 10 s (60 breaths/min, 500 μl of tidal volume). Lung resistance was continuously computed (Labview, National Instruments, TX) by fitting flow, volume, and pressure to an equation of motion. Maximum values of lung resistance were taken and expressed as the percent change from base line following PBS aerosol.
Bronchoalveolar Lavage
Immediately following measurement of AHR, lungs were lavaged. Total leukocyte numbers were counted (Coulter Counter, Coulter Corp., Hialeah, FL), and differential cell counts were performed in a blinded manner under light microscopy by counting at least 200 cells on cytocentrifuged preparations (Cytospin 2; Cytospin, Shandon Ltd., Runcorn, Cheshire, UK) stained with Leukostat (Fisher Diagnostics, Middletown, VA) and differentiated by standard hematological procedures.
Cell Preparation and Culture
CD4+CD25+ and CD4+ CD25− T cells from naive C57BL/6, JNK−/−, and GITR−/− donors were isolated by collagenase digestion from lungs and enriched using nylon wool columns as described previously (10). Lymphocytes were further purified by CD4+CD25+ regulatory T cell MACS beads (Miltenyi Biotec, Bergisch-Gladbach, Germany), resulting in a purity of >95% CD4+CD25+ cells and by sorting on MoFlo (DakoCytomation, Fort Collins, CO) >99% of CD4+CD25+ T cells.
Cells were washed, counted, and resuspended to a final concentration of 4 × 106 cells per ml in RPMI 1640 (Mediatech Celgro, Manassas, VA) tissue culture medium, containing heat-inactivated fetal calf serum (FCS 10%; Sigma), l-glutamine (5 mm), β-mercaptoethanol (2 mm), Hepes buffer (15 mm), penicillin (100 units/ml), and streptomycin (100 μg/ml) (all from Invitrogen).
In Vitro Treatment and Adoptive Transfer
Isolated CD4+CD25+ T cells were pretreated with JNKi (SP600125) or inactive JNKi alone (50 μm, all from Calbiochem) and prior to stimulation with GITRL (1 μg/ml, R&D Systems, Minneapolis, MN) in complete medium for assessing the in vitro production of cytokines in the supernatants, protein extraction for quantifying kinase phosphorylation by Western blotting, and intratracheal adoptive transfer of 5 × 105 nTregs in 50 μl of PBS into sensitized recipients prior to allergen challenge.
Measurement of Cytokine Levels
Cytokine levels in the BAL fluid and supernatants of in vitro cultured lung cells were measured by ELISA (IL-4, IL-5, IL-10, IL-13, and IFN-γ (eBioscience, San Diego)), and TGF-β (Pharmingen). ELISAs were performed according to the manufacturers' directions. The limits of detection were 4 pg/ml for IL-4 and IL-5, 10 pg/ml for IL-10 and IFN-γ, 8 pg/ml for IL-13, and 6 pg/ml for TGF-β.
FACS Analysis
Isolated cells, following preincubation with naive mouse serum in staining buffer (PBS, 2% FCS, 0.2% sodium azide), were labeled with the following conjugated antibodies purchased from Pharmingen: anti-CD3 FITC, PE, PerCP, and allophycocyanin (17A2); anti-CD4 FITC, PE, PerCP, and APC (L3T4); anti-CD25 FITC (7D4) and PE (PC61); anti-GITR FITC, PE, and APC (DTA-1); CD152 FITC and PE APC (9H10). For intracellular staining, cells were stimulated with phorbol 12-myristate 13-acetate (100 ng/ml) and ionomycin (2 μg/ml, Sigma) in complete medium overnight and for 6 h in the presence of brefeldin A (10 μg/ml, Sigma). Cells were fixed with 4% formaldehyde in PBS, permeabilized in 0.5% saponin, and stained with anti-IL-10 PE and APC (JES5-16E3); IFN-γ PE and APC (XMG1.2); and Foxp3 PE and TGF-β (A75-3.1) (eBioscience). Fluorochrome (FITC, PE, PerCPAPC)-labeled isotype-matched control antibodies were used for background fluorescence staining. Staining was analyzed on a FACSCalibur flow cytometer (Pharmingen) using CellQuest Pro software. Fluorescence intensity was compared with cells stained with the corresponding labeled isotype-matched controls.
Real Time PCR
Total RNA was isolated using the Qiagen RNeasy mini kit (Valencia, CA) according to the manufacturer's instructions. Total RNA was reverse-transcribed using Bio-Rad iScript cDNA synthesis kit. TaqMan primers and probes for mouse Foxp3 and GAPDH were obtained from Applied Biosystems (Carlsbad, CA). Real time PCR was performed using ABI Prism 7000 sequence detection system. Relative amounts of cDNA were normalized by subtracting the threshold cycle (Ct) of internal GAPDH reference gene to target gene generating a ΔCt value. Relative mRNA expression of specific gene was obtained by using the formula 2^ (−ΔCt).
Western Blotting
Cells (5 × 106) were lysed with RIPA buffer containing protease inhibitors and phosphatase inhibitors (Sigma) on ice. Lysates were resolved by SDS-PAGE and transferred to nitrocellulose membranes. Blotted membranes were blocked in TBST buffer (25 mm Tris, pH 8.0, 125 mm NaCl, 0.025% Tween 20) containing 2% BSA (Promega, Madison, WI) and then incubated overnight at 4 °C with TBST buffer containing 2% BSA with anti-phospho-ERK1/2, anti-phospho-p38 MAPK, anti-phospho-JNK (Cell Signaling), anti-ERK, anti-p38 MAPK, anti-JNK, anti-MAPK kinases 4 and 7, anti-c-Jun, anti-p-c-Jun, anti-p50, anti-p65, anti-c-Rel mAbs, or anti-IκB-α (Santa Cruz Biotechnology, Santa Cruz, CA or Cell Signaling). Specific proteins were detected by a chemiluminescence method using a horseradish peroxidase-conjugated anti-rabbit, anti-goat, or anti mouse IgG Ab (Amersham Biosciences). Immunoreactive bands were quantified by densitometric quantification of autoradiographs using ImageJ (National Institutes of Health, Bethesda) and expressed as relative phosphorylated kinase to total kinase or β-actin normalized to the unstimulated, untreated cells.
JNK Activation
The assay for JNK activity was performed according to the manufacturer's directions (SAPK/JNK kinase assay, Cell Signaling, Danvers, MA). Briefly, similar amounts of cell lysate from the different cell groups were incubated with the recommended amount of immobilized c-Jun fusion protein overnight. Mixtures were washed extensively with 1× cell lysis buffer and the kinase buffer. SAPK phosphorylation of c-Jun substrate was carried out for 30 min at 30 °C by adding the same volume of 1× kinase buffer supplemented with 200 μm ATP. The reaction was terminated with 25 μl of 3× SDS. The supernatants were then prepared for standard Western immunoblotting probing for p-c-Jun.
Proliferation Assays
Isolated WT CD4+CD25− (2.5 × 104) cells were first activated with bound anti-CD3 (2 μg/ml, 17A2) and soluble anti-CD28 (1 μg/ml, 37.51; both from eBioscience) cultured with or without CD4+CD25+ T cells treated with active (SP600125) or inactive JNKi (25 μm, both from Calbiochem) and then stimulated with GITRL (1 μg/ml, R&D Systems) at a 1:1 ratio in complete medium. GITR-stimulated CD4+CD25+ T cells were carboxyfluorescein diacetate succinimidyl ester-labeled for dilution assay according to the manufacturer's instructions (eBioscience). Cultures were pulsed during the final 6 h with 1 μCi/well thymidine, and cell proliferation was determined as counts/min.
Histochemistry
Lungs were fixed by inflation (1 ml) and immersion in 10% formalin. Cells containing eosinophilic major basic protein were identified by immunohistochemical staining as described previously using rabbit anti-mouse major basic protein (kindly provided by Dr. J. J. Lee, Mayo Clinic, Scottsdale, AZ). The slides were examined in a blinded fashion with a Nikon light microscope. Numbers of eosinophils in the tissues were evaluated with ImageJ counting 6–8 different fields per animal. For detection of mucus-containing cells in formalin-fixed airway tissue, sections were stained with periodic acid-Schiff.
Statistical Analysis
Analysis of variance was used to determine statistical significance. Comparisons for all pairs were performed by the Tukey-Kramer highest significant difference test. The p values for significance were set to 0.05. Values for all measurements were expressed as the means ± S.E.
RESULTS
GITR Stimulation Activates c-Jun N-terminal Kinase
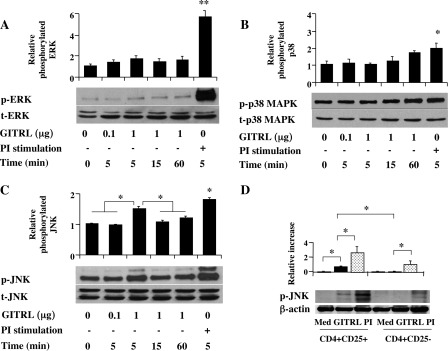
In mammalian cells, three main families of the MAPKs, including extracellular signal-regulated kinase (ERK), c-Jun N-terminal kinase (JNK), and p38, have been implicated in developing immune responses, and individual MAPKs are activated by different stimuli (26). Although p38 (27), ERK (28), and TAK1 (29) have been implicated in the development and function of Tregs, the involvement of MAPK in GITR-mediated loss of suppression and phenotypic conversion has not been described. We investigated the activation of the MAPK pathways in nTregs following GITR stimulation with different concentrations of GITRL (Fig. 1, A–C). Compared with nonstimulated cells, GITRL significantly increased the phosphorylation of JNK without evidence for activation of ERK or p38, as determined by densitometric quantification of immunoreactive bands on autoradiographs. Total JNK, ERK, and p38 were unchanged under all conditions. In contrast to CD4+CD25+ T cells, CD4+CD25− T cells, which express lower levels of GITR (8), demonstrated no increases in GITRL-stimulated increases in p-JNK (Fig. 1D).
FIGURE 1.
Effect of GITR ligation by GITRL on MAPK phosphorylation in nTregs. nTregs were isolated from the lungs of C57BL/6 mice and incubated with different concentrations of GITRL for varying lengths of time. Data illustrated are derived from densitometric quantification of autoradiographs using ImageJ. Levels of relative phosphorylation are derived from base-line values (nonstimulated, time 0) of the ratio of p-ERK to total ERK. A–C, dose and time course of GITR-mediated phosphorylation of JNK but not ERK or p38. D, GITR-mediated phosphorylation of JNK in CD4+CD25− T cells. PI, phorbol/ionomycin. Med, medium. Results are from three independent experiments carried out in duplicate. **, p < 0.01; *, p < 0.05.
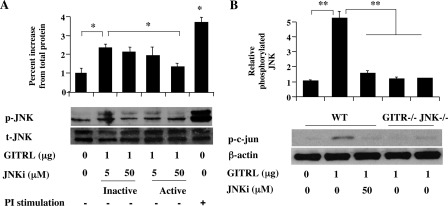
Similar to other MAPKs, full activation of JNK requires dual phosphorylation on Thr-183 and Tyr-184 of the tripeptide motif (Thr-Xaa-Tyr) (30), and specific activity of JNK can be blocked by SP600125, an anthrapyrazolone inhibitor (31). In an animal model, treatment with SP600125 was shown to reduce airway inflammation and levels of OVA-IgE in the serum of sensitized and challenged mice (32). The effect of treatment with SP600125 on the phosphorylation of JNK following GITR stimulation was investigated. Pretreatment of nTregs with the active but not inactive JNK inhibitor (JNKi) decreased GITRL-induced phosphorylation of JNK (Fig. 2A).
FIGURE 2.
Effect of JNKi (SP600125) on phosphorylation of JNK and JNK activity in immunoprecipitates. JNKi was added 1–2 h prior to GITRL. A, dose effect of JNKi on GITR-induced phosphorylation of JNK. B, JNK activity is detected in immunoprecipitates of GITR-stimulated, untreated but not JNKi-treated, nTregs from WT, and GITR−/−, or JNK−/− mice. Results are from three independent experiments carried out in duplicate. **, p < 0.01; *, p < 0.05. PI, phorbol/ionomycin.
The specificity of activation of JNK following GITR ligation was confirmed by comparing nTregs from wild-type (WT) with nTregs from JNK−/− or GITR−/− mice. JNK activity detected as phosphorylation of c-Jun (p-c-Jun) was only detected in the immunoprecipitates from WT but not the deficient (JNK−/− or GITR−/−) or JNKi-treated nTregs (Fig. 2B).
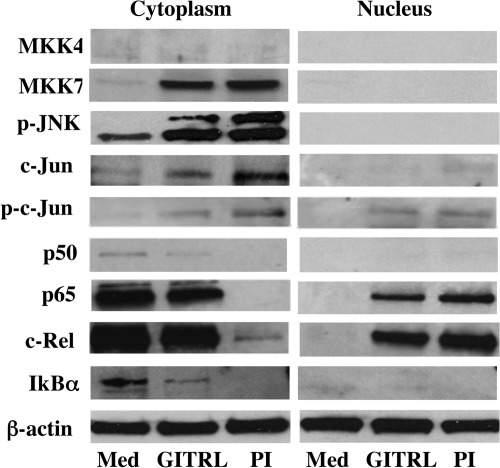
We next investigated which of the upstream MAPK kinases (MKK) were involved in GITR-mediated activation of JNK at the time point associated with changes in levels of cytokines in the supernatants of in vitro cultured cells. To date, only MKK4 (SEK1) and MKK7 have been implicated in the activation of JNK (26, 33, 34). Following the in vitro addition of GITRL, increases in protein levels of MKK7 but not MKK4 were detected (Fig. 3).
FIGURE 3.
MAPK cascade in JNK activation. GITR ligation (1 μg) for 72 h increases levels of MKK7, c-Jun, p-c-Jun, and subunits of NF-κB in cytoplasmic and nuclear cell extracts, and decreases levels of NF-κB inhibitor (IκB) in cytoplasmic extracts. Data are representative of three independent and similar experiments. PI, phorbol/ionomycin; Med, medium.
Upon activation, JNK phosphorylates a number of downstream targets, including c-Jun, which heterodimerizes with Fos to form the transcription factor activator protein 1 (35). To further define the role of JNK in the signaling cascade through GITR, total protein and phosphorylation of c-Jun were determined. Both total protein and phosphorylation of c-Jun increased following GITR stimulation, consistent with the results detected in immunoprecipitates. Correspondingly, increased c-Jun was detected in the nuclear extracts of stimulated compared with nonstimulated nTregs.
Consistent with a previous report (9), increases in activation of subunits of NF-κB, including c-Rel, p65, and to a lesser degree p50, were detected following GITR stimulation, indicated by the increases in nuclear translocation of these subunit proteins (Fig. 3). Activation of NF-κB can also be determined by assessing the levels of the protein inhibitor of NF-κB (Iκβ) (22). In contrast to nonstimulated cells, GITR-stimulated cells had less Iκβ protein in the cytoplasm.
GITRL Stimulation Induces Changes in the Phenotype, Function, and Cytokine Production of nTregs and Are Prevented by Inhibition of JNK Activation
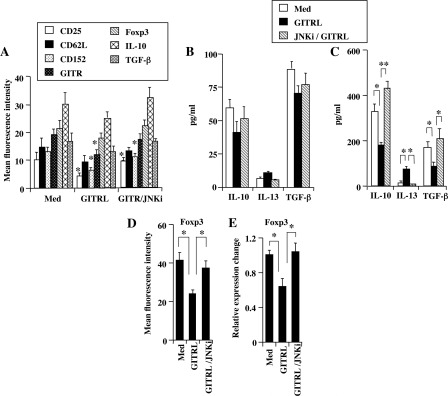
Naturally occurring CD4+ CD25+ nTregs have been characterized by the expression of CD25, CTLA-4, and GITR (6, 7, 12, 36), and under certain experimental conditions, decreased expression of these receptors was shown to associate with loss or attenuation of suppressive activity (19–21, 36). The effects of GITR ligation on the expression of surface markers were assessed by flow cytometry in the presence or absence of the JNKi. Compared with nonstimulated cells, nTregs treated with GITRL expressed lower mean fluorescence intensities of CD25, GITR, and CTLA-4 (Fig. 4A). In contrast, treatment of nTregs with the active JNKi prevented the down-regulation of expression of these receptors, retaining the regulatory phenotype.
FIGURE 4.
Effect of GITR ligation on expression of surface markers, production of cytokines at different time course in the presence and absence of JNKi (50 μm), and expression of Foxp3. A, FACS analysis of nTregs following overnight GITR ligation; *, p < 0.05 compared with expression in medium control. B, cytokine levels in supernatants (overnight); C, cytokine levels in supernatants (72 h). D, expression of Foxp3 protein following GITRL stimulation (72 h). E, expression of Foxp3 mRNA following GITRL stimulation. Results are from three independent experiments carried out in duplicate. Med, medium. **, p < 0.01; *, p < 0.05.
Previously, nTregs isolated from naive BALB/c and C57BL/6 mice were shown to produce the immunosuppressive cytokines IL-10 and TGF-β (12, 13, 15). We determined the consequences of sustained GITR ligation over 72 h on cytokine production and the impact of adding the JNKi. Despite the phenotypic changes induced following the shorter incubation, intracellular staining of Foxp3 and immunocytokines (Fig. 4A), similar to levels detected in the supernatants of in vitro cultures (Fig. 4B), did not change to any appreciable degree. In contrast, significant changes in cytokine profile were detected following the longer stimulation. Compared with nonstimulated nTregs, GITRL-stimulated nTregs released lower amounts of IL-10 and TGF-β in the culture supernatants (Fig. 4C), similar to GITRL-treated cells in the presence of the inactive inhibitor (data not shown). In contrast, treatment with the active JNKi prevented these decreases, maintaining the levels seen in untreated cells. In parallel, similar to previous results showing increased IL-13 production when nTregs were shown to have reduced IL-10 and TGF-β production (8), levels of IL-13 were increased in cultures of GITRL-treated nTregs but not those that included the active JNKi. Furthermore, decreases in Foxp3 protein (Fig. 4D) and mRNA (Fig. 4E) expression were associated with the changes in cytokine production (Fig. 4C).
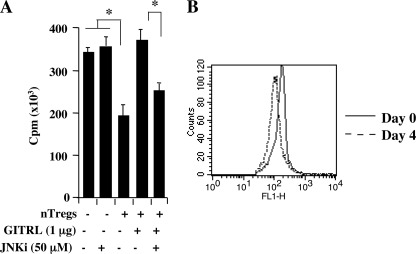
Consistent with others (6, 7), we reported that the proliferation of CD4+CD25− T cells activated with anti-CD3 and anti-CD28 was inhibited by nTregs (19) and that treatment of nTregs with either the GITRL or agonistic anti-GITR antibody (DTA-1) abolished their suppressive activity. Here, we confirmed that in vitro stimulation of nTregs with GITRL abolished the suppression of CD4+CD25− T cell proliferation triggered by anti-CD3/anti-CD28 (Fig. 5A). The loss of suppression, in contrast to a previous report (40), was not associated with GITR-induced proliferation of nTregs (Fig. 5B). However, suppression was maintained in the presence of the active, but not inactive, JNKi. This was not due to any toxic effects of the inhibitor, as washing out of the inhibitor after a similar incubation period was associated with full proliferative activity of the cells (data not shown).
FIGURE 5.
Effect of GITR ligation on suppression of in vitro proliferation of activated CD4CD25− T cell in the presence and absence of JNKi (50 μm). A, GITRL stimulation abrogates suppression of CD4+CD25− T cell proliferation, and treatment of nTregs with JNKi restores suppression. B, absence of carboxyfluorescein diacetate succinimidyl ester dilution in GITR-stimulated CD4+CD25+ T cells. The means ± S.E. from three independent experiments were carried out in triplicate. *, p < 0.05.
Taken together, these in vitro studies demonstrated that ligation of GITR had profound effects on kinase phosphorylation, surface receptor expression, suppressive activity, and cytokine production, and all of these effects were reversed in the presence of the active JNKi.
Prevention of JNK Activation in Vivo Is Associated with Maintenance of Suppression in an Experimental Model of Asthma
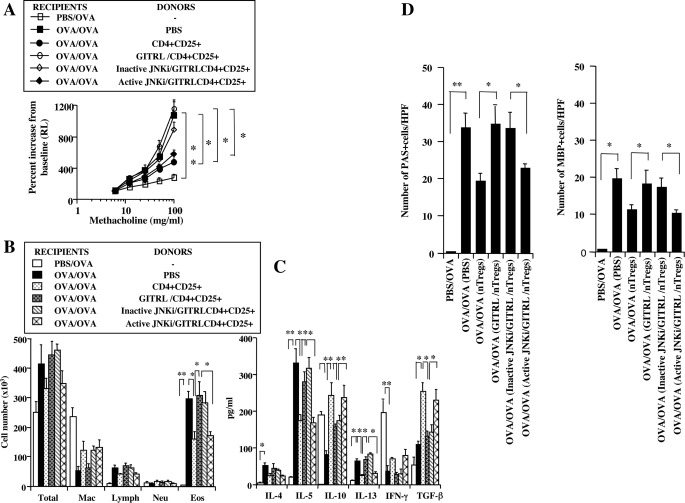
Following adoptive transfer, nTregs suppressed the development of allergen-induced lung allergic responses in sensitized and challenged mice (8, 12, 13, 15, 19). This suppression mediated by nTregs was abolished by the in vitro treatment of nTregs with GITRL or agonistic antibody when transferred after sensitization but prior to challenge (8). We determined whether prevention of JNK activation affected the activity of nTregs in sensitized and challenged mice. Prior to transfer, cell viability of GITRL- and JNK-treated cells exceeded 85–90%. Transfer of nTregs treated with GITRL, as compared with nontreated cells, failed to suppress AHR (Fig. 6A). In contrast, treatment of nTregs with the active but not inactive JNKi restored the suppression of AHR. Associated with the increases in AHR, the numbers of airway eosinophils were significantly increased in sensitized and challenged mice as shown in Fig. 6B. Airway eosinophilia was significantly reduced in recipients of nonstimulated nTregs and GITRL-stimulated nTregs treated with the active inhibitor but was maintained in the GITRL-stimulated nTregs treated with the inactive inhibitor. Similar effects of treatment with the JNKi were also observed in BALB/c mice (data not shown).
FIGURE 6.
Effect of transfer of nTregs from C57BL/6 donors into sensitized and challenged C57BL/6 recipients. Sensitized C57BL/6 recipients received unstimulated/untreated, GITRL-stimulated, and active or inactive GITRL-stimulated/JNKi-treated nTregs (0.5 × 106) cells intratracheally, and airway responses were assessed 48 h later prior to challenge. A, AHR, B, BAL fluid inflammatory cell composition. Mac, macrophages; Lymph, lymphocytes; Neu, neutrophils; Eos, eosinophils. C, BAL cytokine levels. D, quantitation of MBP+ and PAS+ cells. Shown are the means ± S.E. from three independent experiments. n = 12. **, p < 0.01; *, p < 0.05.
Following sensitization and challenge with allergen, Th2 cytokine levels were increased in BAL fluid. Significant increases in levels of IL-4, IL-5, and IL-13 were detected, and levels of IL-10 and IFN-γ were decreased; levels of TGF-β were modestly increased (Fig. 6C). Intratracheal transfer of nonligated nTregs decreased the levels of IL-5 and IL-13, whereas IL-10 and TGF-β levels were increased. Little change was seen in levels of IL-4 and IFN-γ. Transfer of GITRL-stimulated nTregs, untreated or treated with the inactive JNKi, failed to significantly reduce the levels of IL-5 and IL-13, and the levels of IL-4, IL-10, TGF-β, and IFN-γ were similar to the levels in sensitized and challenged mice that did not receive transferred cells (Fig. 6C). In contrast, treatment of GITRL-stimulated nTregs with the active JNKi reversed the effects of GITR stimulation, with restoration of levels of IL-10 and TGF-β and decreased levels of IL-5 and IL-13.
Lung histopathology paralleled the changes in inflammatory cell composition and numbers and levels of cytokines in BAL fluid. The lung tissue from sensitized and challenged mice that received GITRL-stimulated nTregs, untreated or treated with inactive JNKi, similar to mice given PBS, showed a significant accumulation of eosinophils, mucus hyperproduction, and goblet cell metaplasia (Fig. 6D). In contrast, mice that received non-GITRL-stimulated or nTregs that were GITRL-stimulated but treated with active JNKi exhibited significantly less lung histopathology.
DISCUSSION
Naturally occurring CD4+CD25+Foxp3+ T cells (nTregs), although comprising only a small subset of T cells derived in the thymus (37), appear essential to the maintenance of immunological tolerance. In the absence of functional Foxp3, the key transcription regulator for Treg development and function (38), severe and life-threatening autoimmune and allergic diseases develop (17, 18). These regulatory T cells have also been implicated in the maintenance of immune homeostasis in the airways as they have been shown to regulate allergen-induced AHR and inflammation, at least in part, through the actions of IL-10 and TGF-β (12), and in an antigen-nonspecific manner (15). In the lungs of both BALB/c and C57BL/6 mice, the activation and expression of this suppressive phenotype appeared dependent on the interaction of MHC class I on nTregs with CD8; inhibition or interference with this interaction led to the loss of nTreg suppressive activity (13, 15). A role for CD8 in maintaining suppressive activity in the periphery and regulating the production of endogenous IL-6 in Tregs was recently demonstrated (19). Similar to other reports (6, 7), in the absence of CD8-expressing cells, activation of nTregs through GITR in the lungs of recipient mice was shown to be sufficient to abrogate suppression and convert these cells to pathogenic effector cells resulting in the enhancement of lung allergic responses (8). Significantly, GITRL expression in recipients appeared to provide the necessary signals to attenuate suppression, as administration of anti-GITRL antibody prevented rather than enhanced lung allergic responses in association with little increase in levels of IL-13 and maintenance of basal levels of IL-10 and TGF-β known to be essential for suppressive activity (12, 39).
In this study, we investigated the molecular events following stimulation of nTregs through GITR. Although the roles of p38 (27), ERK (28), and TAK1 (29) have been implicated in the development and function of Tregs, their involvement in the stimulatory activity of GITR in Tregs has not been defined. In this study, we defined a signaling pathway following ligation of GITR, a cascade that was associated with the loss of suppressive activity and phenotypic conversion. In vitro stimulation of nTregs with GITRL increased phosphorylation of JNK but not ERK or p38. The activation of JNK was linked to changes in the phenotype, capacity to produce immunosuppressive cytokines, and in vitro and in vivo suppressive activities. The expression levels of the surface receptors CD25, CD152 (CTLA-4), and GITR were decreased following GITR stimulation, rendering nTregs with an effector phenotype. Loss of suppression was associated with a decrease in expression of these surface molecules and levels of Foxp3 mRNA and protein. Corresponding with the effector phenotype, lower levels of IL-10 and TGF-β and higher levels of IL-13 were detected in the supernatants of in vitro cultures. Similar to previous studies (6, 7, 38), following GITR ligation nTregs failed to suppress the in vitro proliferation of CD4+CD25− T cells activated with anti-CD3 and anti-CD28. In the absence of additional stimulation and unlike a previous study (40), the loss of suppression was not associated with GITR-induced proliferation of nTregs.
The importance of JNK activation in the spectrum of GITR-mediated effects on nTregs was confirmed using SP600125, a known inhibitor of JNK (31), and genetically deficient mice. Phosphorylation of JNK in GITRL-treated nTregs was prevented by the inhibitor. The link between JNK activation and GITR stimulation was also demonstrated by increases in the phosphorylation of JNK and corresponding activity, as indicated by levels of p-c-Jun, detected only in the immunoprecipitates from ligated nTregs from WT but not JNK−/− or GITR−/− mice. Furthermore, treatment with the inhibitor abolished GITRL-mediated phosphorylation and activity of JNK in the immunoprecipitates of nTregs from WT mice. The GITR signaling cascade was further defined both upstream and downstream of JNK phosphorylation. Upstream, activation of MKK7 but not MKK4 was demonstrated. Both MKK7 and MKK4 have been reported to activate JNK (26, 33, 34), but only MKK4 has additional activity phosphorylating p38 MAPK in vitro (9, 26). Our data are consistent with this notion because little or no increase in the activation of p38 following GITR stimulation was detected and only MKK7 was identified in the activation of nTregs through GITR. Downstream, phosphorylation of JNK was associated with increases in total c-Jun and p-c-Jun, a known downstream substrate of JNK (30, 31). Corresponding with the increase in p-c-Jun, greater amounts of c-Jun were also detected in the nucleus.
The loss of nTreg-mediated suppression following GITR ligation has been shown to be dependent on the activation of NF-κB (9). We confirmed that GITR ligation increased the activation of subunits of NF-κB, including c-rel, p65, and to a lesser extent p50 in cytoplasmic and nuclear cell extracts. Interestingly, the activation of NF-κB coincided with divergent effects on the levels of proinflammatory and immunosuppressive cytokines. Multiple factors may be involved in these transcriptional responses, including selective phosphorylation and subsequent degradation of NF-κB inhibitors or the result of differential subunit distribution, affinity, or number of binding sites engaged in these reactions.
The importance of JNK activation in mediating the abrogation of nTreg suppressive activity through GITR was also demonstrated in vivo in sensitized and challenged mice. As in previous studies (12, 13, 15, 19), following sensitization and allergen challenge, mice developed a significant increase in airway responsiveness to inhaled methacholine, eosinophilic inflammation, and Th2 skewing in cytokine production. These findings were accompanied by changes in lung pathology, especially eosinophil accumulation in the lung parenchyma and goblet cell metaplasia and mucus hyperproduction. Consistent with our previous report (8), GITR ligation of nTregs prior to transfer into sensitized and challenged mice eliminated the suppression of lung allergic responses. However, the full suppression of all lung allergic responses and lung pathology could be restored when the GITR-ligated nTregs were treated with the JNKi prior to transfer into sensitized recipients prior to airway allergen challenge.
Taken together, the results demonstrate that stimulation of nTregs through GITR initiates a signaling cascade that involves MKK7, JNK phosphorylation, c-Jun activation, and the activation of NF-κB. Associated with the phosphorylation of JNK were changes in nTreg surface marker expression, conversion of the cytokine profile from IL-10/TGF-β to IL-13 production, attenuation of in vitro suppressive activities, and in an experimental model of allergic asthma, elimination of the nTreg regulatory activities on development of altered airway responsiveness, eosinophilic airway inflammation, and Th2 cytokine production. Incubation of the GITRL-stimulated nTregs with an inhibitor of JNK activation resulted in the restoration of nTreg activities and phenotype in vitro and in vivo. These findings demonstrate that targeting JNK phosphorylation can have a major influence on nTreg suppressive function.
Acknowledgments
We are indebted to Drs. Joseph Lucas, Roberta Pelanda, Raul Torres, and Laurent Gapin for critical review of the manuscript; to Dr. Carlo Riccardi for providing the GITR-deficient mice, and Diana Nabighian for assistance in preparation of this manuscript.
This work was supported, in whole or in part, by National Institutes of Health Grants AI-77609, HL-36577, and HL-61005.
- GITR
- glucocorticoid-induced tumor necrosis receptor
- AHR
- airway hyper-responsiveness
- Treg
- T regulatory cell
- nTreg
- naturally occurring Treg
- GITRL
- GITR ligand
- OVA
- ovalbumin
- PE
- phycoerythrin
- BAL
- bronchoalveolar lavage.
REFERENCES
- 1. Nocentini G., Giunchi L., Ronchetti S., Krausz L. T., Bartoli A., Moraca R., Migliorati G., Riccardi C. (1997) A new member of the tumor necrosis factor/nerve growth factor receptor family inhibits T cell-receptor-induced apoptosis. Proc. Natl. Acad. Sci. U.S.A. 94, 6216–6221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Kwon B., Yu K. Y., Ni J., Yu G. L., Jang I. K., Kim Y. J., Xing L., Liu D., Wang S. X., Kwon B. S. (1999) Identification of a novel activation-inducible protein of the tumor necrosis factor receptor superfamily and its ligand. J. Biol. Chem. 274, 6056–6061 [DOI] [PubMed] [Google Scholar]
- 3. Watts T. H. (2005) TNF/TNFR family members in costimulation of T cell responses. Annu. Rev. Immunol. 23, 23–68 [DOI] [PubMed] [Google Scholar]
- 4. Cuzzocrea S., Nocentini G., Di Paola R., Agostini M., Mazzon E., Ronchetti S., Crisafulli C., Esposito E., Caputi A. P., Riccardi C. (2006) Proinflammatory role of glucocorticoid-induced TNF receptor-related gene in acute lung inflammation. J. Immunol. 177, 631–641 [DOI] [PubMed] [Google Scholar]
- 5. Motta A. C., Vissers J. L., Gras R., Van Esch B. C., Van Oosterhout A. J., Nawijn M. C. (2009) GITR signaling potentiates airway hyper-responsiveness by enhancing Th2 cell activity in a mouse model of asthma. Respir. Res. 10, 93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Shimizu J., Yamazaki S., Takahashi T., Ishida Y., Sakaguchi S. (2002) Stimulation of CD25+CD4+ regulatory T cells through GITR breaks immunological self-tolerance. Nat. Immunol. 3, 135–142 [DOI] [PubMed] [Google Scholar]
- 7. McHugh R. S., Whitters M. J., Piccirillo C. A., Young D. A., Shevach E. M., Collins M., Byrne M. C. (2002) CD4+CD25+ immunoregulatory T cells. Gene expression analysis reveals a functional role for the glucocorticoid-induced TNF receptor. Immunity 16, 311–323 [DOI] [PubMed] [Google Scholar]
- 8. Joetham A., Matsubara S., Okamoto M., Takeda K., Miyahara N., Dakhama A., Gelfand E. W. (2008) Plasticity of regulatory T cells. Subversion of suppressive function and conversion to enhancement of lung allergic responses. J. Immunol. 180, 7117–7124 [DOI] [PubMed] [Google Scholar]
- 9. Ji H. B., Liao G., Faubion W. A., Abadía-Molina A. C., Cozzo C., Laroux F. S., Caton A., Terhorst C. (2004) Cutting Edge. The natural ligand for glucocorticoid-induced TNF receptor-related protein abrogates regulatory T cell suppression. J. Immunol. 172, 5823–5827 [DOI] [PubMed] [Google Scholar]
- 10. Stephens G. L., McHugh R. S., Whitters M. J., Young D. A., Luxenberg D., Carreno B. M., Collins M., Shevach E. M. (2004) Engagement of glucocorticoid-induced TNFR family-related receptor on effector T cells by its ligand mediates resistance to suppression by CD4+CD25+ T cells. J. Immunol. 173, 5008–5020 [DOI] [PubMed] [Google Scholar]
- 11. Cardona I. D., Goleva E., Ou L. S., Leung D. Y. (2006) Staphylococcal enterotoxin B inhibits regulatory T cells by inducing glucocorticoid-induced TNF receptor-related protein ligand on monocytes. J. Allergy Clin. Immunol. 117, 688–695 [DOI] [PubMed] [Google Scholar]
- 12. Joetham A., Takeda K., Takada K., Taube C., Miyahara N., Matsubara S., Matsubara S., Koya T., Rha Y. H., Dakhama A., Gelfand E. W. (2007) Naturally occurring lung CD4+CD25+ T-cell regulation of airway allergic responses depends on IL-10 induction of TGF-β. J. Immunol. 178, 1433–1442 [DOI] [PubMed] [Google Scholar]
- 13. Joetham A., Takeda K., Miyahara N., Matsubara S., Ohnishi H., Koya T., Dakhama A., Gelfand E. W. (2007) Functional activation of naturally occurring lung CD4+CD25+ regulatory cells on lung allergic responses requires CD8 and MHC I interaction. Proc. Natl. Acad. Sci. 104, 15057–15062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Akbari O., Freeman G. J., Meyer E. H., Greenfield E. A., Chang T. T., Sharpe A. H., Berry G., DeKruyff R. H., Umetsu D. T. (2002) Antigen-specific regulatory T cells develop via the ICOS-ICOSSL pathway and inhibit allergen-induced airway hyper-reactivity. Nat. Med. 8, 1024–1032 [DOI] [PubMed] [Google Scholar]
- 15. Joetham A., Takeda K., Okamoto M., Taube C., Matsuda H., Dakhama A., Gelfand E. W. (2009) Antigen specificity is not required for modulation of lung allergic responses by naturally occurring regulatory T cells. J. Immunol. 183, 1821–1827 [DOI] [PubMed] [Google Scholar]
- 16. Fontenot J. D., Rasmussen J. P., Williams L. M., Dooley J. L., Farr A. G., Rudensky A. Y. (2005) Regulatory T cell lineage specification by the forkhead transcription factor Foxp3. Immunity 22, 329–341 [DOI] [PubMed] [Google Scholar]
- 17. Clark L. B., Appleby M. W., Brunkow M. E., Wilkinson J. E., Ziegler S. F., Ramsdell F. (1999) Cellular and molecular characterization of the scurfy mouse mutant. J. Immunol. 162, 2546–2554 [PubMed] [Google Scholar]
- 18. Bennett C. L., Christie J., Ramsdell F., Brunkow M. E., Ferguson P. J., Whitesell L., Kelly T. E., Saulsbury F. T., Chance P. F., Ochs H. D. (2001) The immune dysregulation, polyendocrinopathy, enteropathy, and X-linked syndrome are caused by mutations of Foxp3. Nat. Genet. 27, 20–21 [DOI] [PubMed] [Google Scholar]
- 19. Joetham A., Okamoto M., Takeda K., Schedel M., Ohnishi H., Dakhama A., Gelfand E. W. (2011) CD8 regulates the endogenous production of IL-6 in naturally occurring T regulatory cells and maintains their suppressive phenotype in allergic lung disease. J. Immunol. 186, 113–120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Wan Y. Y., Flavell R. A. (2007) Regulatory T-cell functions are subverted and converted owing to attenuated Foxp3 expression. Nature 445, 766–770 [DOI] [PubMed] [Google Scholar]
- 21. Zhou X., Bailey-Bucktrout S. L., Jeker L. T., Penaranda C., Martínez-Llordella M., Ashby M., Nakayama M., Rosenthal W., Bluestone J. A. (2009) Instability of the transcription factor Foxp3 leads to generation of pathogenic memory T cells in vivo. Nat. Immunol. 10, 1000–1007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Esparza E. M., Arch R. H. (2005) Glucocorticoid-induced TNF receptor, a costimulatory receptor on naive and activated T cells, uses TNF receptor-associated factor 2 in a novel fashion as an inhibitor of NF-κB activation. J. Immunol. 174, 7875–7882 [DOI] [PubMed] [Google Scholar]
- 23. Kanamaru F., Youngnak P., Hashiguchi M., Nishioka T., Takahashi T., Sakaguchi S., Ishikawa I., Azuma M. (2004) Costimulation via glucocorticoid-induced TNF receptor in both conventional and CD25+ regulatory CD4+ T cells. J. Immunol. 172, 7306–7314 [DOI] [PubMed] [Google Scholar]
- 24. Ronchetti S., Nocentini G., Bianchini R., Krausz L. T., Migliorati G., Riccardi C. (2007) Glucocorticoid-induced TNFR-related protein lowers the threshold of CD28 costimulation in CD8+ T cells. J. Immunol. 179, 5916–5926 [DOI] [PubMed] [Google Scholar]
- 25. Bae E. M., Kim W. J., Suk K., Kang Y. M., Park J. E., Kim W. Y., Choi E. M., Choi B. K., Kwon B. S., Lee W. H. (2008) Reverse signaling initiated from GITRL induces NF-κB activation through ERK in the inflammatory activation of macrophages. Mol. Immunol. 45, 523–533 [DOI] [PubMed] [Google Scholar]
- 26. Dong C., Davis R. J., Flavell R. A. (2002) MAPKs in the immune response. Annu. Rev. Immunol. 20, 55–72 [DOI] [PubMed] [Google Scholar]
- 27. Adler H. S., Kubsch S., Graulich E., Ludwig S., Knop J., Steinbrink K. (2007) Activation of MAP kinase p38 is critical for the cell cycle-controlled suppressor function of regulatory T cells. Blood 109, 4351–4359 [DOI] [PubMed] [Google Scholar]
- 28. Luo X., Zhang Q., Liu V., Xia Z., Pothoven K. L., Lee C. (2008) Cutting Edge. TGF-b-induced expression of Foxp3 in T cells is mediated through inactivation of ERK. J. Immunol. 180, 2757–2761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Sato S., Sanjo H., Tsujimura T., Ninomiya-Tsuji J., Yamamoto M., Kawai T., Takeuchi O., Akira S. (2006) TAK1 is indispensable for development of T cells and prevention of colitis by the generation of regulatory T cells. Int. Immunol. 18, 1405–1411 [DOI] [PubMed] [Google Scholar]
- 30. Dérijard B., Hibi M., Wu I. H., Barrett T., Su B., Deng T., Karin M., Davis R. J. (1994) JNK1. A protein kinase stimulated by UV light and Ha-ras that binds and phosphorylates the c-Jun activation domain. Cell 76, 1025–1037 [DOI] [PubMed] [Google Scholar]
- 31. Bennett B. L., Sasaki D. T., Murray B. W., O'Leary E. C., Sakata S. T., Xu W., Leisten J. C., Motiwala A., Pierce S., Satoh Y., Bhagwat S. S., Manning A. M., Anderson D. W. (2001) SP600125, an anthrapyrazolone inhibitor of Jun N-terminal kinase. Proc. Natl. Acad. Sci. U.S.A. 98, 13681–13686 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Chialda L., Zhang M., Brune K., Pahl A. (2005) Inhibitors of mitogen-activated protein kinases differentially regulate costimulated T cells cytokine production and mouse airway eosinophilia. Respir. Res. 6, 36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Tournier C., Whitmarsh A. J., Cavanagh J., Barrett T., Davis R. J. (1997) Mitogen-activated protein kinase kinase 7 is an activator of the c-Jun N-terminal kinase. Proc. Natl. Acad. Sci. U.S.A. 94, 7337–7342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Sánchez I., Hughes R. T., Mayer B. J., Yee K., Woodgett J. R., Avruch J., Kyriakis J. M., Zon L. I. (1994) Role of SAPK/ERK kinase-1 in the stress-activated pathway regulating transcription factor c-Jun. Nature 372, 794–798 [DOI] [PubMed] [Google Scholar]
- 35. Whitmarsh A. J., Davis R. J. (1996) Transcription factor AP-1 regulation by mitogen-activated protein kinase signal transduction pathways. J. Mol. Med. 17, 2360–2371 [DOI] [PubMed] [Google Scholar]
- 36. Wing K., Onishi Y., Prieto-Martin P., Yamaguchi T., Miyara M., Fehervari Z., Nomura T., Sakaguchi S. (2008) CTLA-4 control over Foxp3+ regulatory T cell function. Science 322, 271–275 [DOI] [PubMed] [Google Scholar]
- 37. Sakaguchi S., Yamaguchi T., Nomura T., Ono M. (2008) Regulatory T cells and immune tolerance. Cell 133, 775–787 [DOI] [PubMed] [Google Scholar]
- 38. Lin A., Minden A., Martinetto H., Claret F. X., Lange-Carter C., Mercurio F., Johnson G. L., Karin M. (1995) Identification of a dual specificity kinase that activates the Jun kinases and p38-Mpk2. Science 268, 286–290 [DOI] [PubMed] [Google Scholar]
- 39. Presser K., Schwinge D., Wegmann M., Huber S., Schmitt S., Quaas A., Maxeiner J. H., Finotto S., Lohse A. W., Blessing M., Schramm C. (2008) Coexpression of TGF-β1 and IL-10 enables regulatory T cells to completely suppress airway hyper-reactivity. J. Immunol. 181, 7751–7758 [DOI] [PubMed] [Google Scholar]
- 40. Liao G., Nayak S., Regueiro J. R., Berger S. B., Detre C., Romero X., de Waal Malefyt R., Chatila T. A., Herzog R. W., Terhorst C. (2010) GITR engagement preferentially enhances proliferation of functionally competent CD4+CD25+ Foxp3 regulatory cells. Int. Immunol. 22, 259–270 [DOI] [PMC free article] [PubMed] [Google Scholar]