Abstract
Purpose.
The purpose of this study was to investigate the presence of progenitor cells in the uninjured, adult rat lacrimal gland (LG).
Methods.
The presence of progenitor cells was examined in LG sections from male rats using antibodies against selected stem cell markers and α-smooth muscle actin (SMA), which marks myoepithelial cells (MECs), by immunofluorescence microscopy (IF). Small, immature cells were isolated after digestion of LG with collagenase and culture in RPMI 1640 for 2 weeks. Immature cells were examined for expression of stem cell markers by IF. Immature cell were grown in neuronal, epithelial, and myoepithelial cell media, and examined by light morphology and IF using antibodies to markers of different cell lineages.
Results.
In the intact LGs, MECs expressed the stem cell markers nestin, Musashi 1, ABCG2, Pax6, Chx 10, ΔN p63, and Sox 2. All markers colocalized with SMA. Isolated immature cells contained Ki-67, nestin, Musashi 1, Pax 6, and CHX 10. In neuronal media, immature cells differentiated and assumed a neuronal cell morphology expressing neurofilament 200. In media for human corneal endothelial cells, immature cells differentiated, assumed cobblestone morphology, and labeled with the epithelial marker AE1/AE3. In RPMI media immature cells differentiated into cells with MEC-like morphology, and expressed the MEC markers SMA, α-actinin, adenylate cyclase II, and vimentin.
Conclusions.
We conclude that uninjured, adult LG contains progenitor cells that may be MECs, which can be isolated and differentiated into multiple lineages.
Progenitor cells were isolated from unwounded, adult rat lacrimal glands.
Introduction
The lacrimal gland (LG) is an exocrine, tubloacinar gland that is responsible for most of the aqueous portion of the tear film. The LG is composed of three major cell types: acinar, ductal, and myoepithelial cells. Acinar cells, which comprise about 80% of the gland, synthesize and secrete proteins, as well as secrete water and electrolytes into the acinar lumen that empties into the duct system. Smaller ducts coalesce into larger ducts, eventually leading to the main excretory duct, which terminates on the ocular surface. Ductal cells line the ducts and modify the secretory fluid by secreting electrolytes and water.1
Myoepithelial cells (MECs) have a characteristic stellate shape with thin processes, which surround the acinar cells. Based on the presence of α-smooth muscle actin (SMA) in these cells, it is believed that they contract to squeeze the secretory products out of the acinar cells and into the ducts.2 In the LG, investigations have demonstrated that MECs contain M3 muscarinic receptors and the VIP receptors, known as VPAC1 and VPAC2.3,4 Muscarinic receptors are functional, as cholinergic agonists have been shown to stimulate contraction of MECs.5 Our laboratory recently has been able to culture MECs from LG, and have demonstrated that the purinergic receptors P2X7, P2Y1, P2Y11, and P2Y13 also are present.5 The purinergic receptors are active, as addition of purinergic agonists increase intracellular [Ca2+]i.5,6
Dysfunction of the LG has been demonstrated in a variety of conditions, including immune-mediated processes, such as autoimmune diseases, viral infections, organ transplantation, and injury.7 The mechanisms of the destruction of the LG are not well understood.8 Regardless of the cause, LG dysfunction results in aqueous deficient dry eye, which in severe cases can lead to eye infections, impaired wound healing, and scarring of the cornea. Thus, repair and/or regeneration of the lacrimal gland would be advantageous to ocular surface health and function.
There is a large body of evidence for the capacity of exocrine glands, such as pancreas, salivary, and mammary glands, to regenerate.9–11 In the parotid, submandibular, and sublingual glands, ligation of the excretory ducts leads to atrophy. When the ligation is removed, the glands regenerate through proliferation of acinar, ductal, and myoepithelial cells.10,12,13 In the LG, Zoukhri et al. demonstrated that a single injection of the pro-inflammatory cytokine IL-1 into the murine LG led to a severe inflammatory response, impaired release of secretory protein, decreased tear output, and extensive acinar cell death.14 Within 3–7 days, the LG regenerated and cell function returned. During this time of repair, acinar and myoepithelial cells proliferated, restoring lacrimal gland function. Zoukhri et al. also demonstrated that progenitor cells that were positive for nestin were proliferating, and a small population of these cells were identified as MECs.15 These data suggest that the LG is capable of self-repair and contains a population of stem/progenitor cells that proliferate to repair the LG. Recently, You et al. isolated the progenitor cells from the injured murine LG and identified them as mesenchymal stem cells.16 They were not able to isolate progenitor cells from the uninjured murine LG.16
We recently developed a method to culture MECs from uninjured, adult rat LGs.5 MECs typically take 3–4 weeks to differentiate. During this time, a number of actively dividing immature cells were observed growing over the underlying monolayer. The morphology of these immature cells was similar to that of stem cells reported in other tissues.17,18 The purpose of our study was to investigate if progenitor cells were present in normal, uninjured LGs, and if they could be isolated, proliferated, and differentiated into other cell types.
Materials and Methods
Materials
Primary antibodies used in this research were anti-mouse α-SMA (Diagnostic Biosystems, Pleasanton, CA); anti-mouse nestin (Developmental Studies Hybridoma Bank, University of Iowa, Iowa City, IA); anti-rabbit Musashi 1 and neurofilament-200 (Millipore, Billerica, MA); and anti-goat ΔN p63, anti-goat SOX 2, anti-goat PAX 6, anti-goat CHX 10, anti-rabbit ABCG2, anti-rabbit adenylate cyclase II, anti-mouse α-actinin, normal goat IgG, normal mouse IgG2a and normal rabbit IgG (Santa Cruz Biotechnology, Inc., Santa Cruz, CA), as well as an antibody against AE1/AE3, a pan-cytokeratin marker (BD Biosciences, San Jose, CA), an antibody against vimentin (Sigma Aldrich, St. Louis, MO) and anti-mouse Ki-67 (Vector Laboratories, Burlingame, CA). We also used secondary antibodies Cy2 anti-mouse, Cy3 anti-rabbit, and Cy3 anti-goat (Jackson Immunoresearch Labs, Inc., West Grove, PA); RPMI-1640, X-VIVO 15, KBM cell culture media and KBM supplements (Lonza, Walkersville, MD); and corneal endothelial cell medium (a kind gift from Dr Nancy Joyce, Schepens Eye Research Institute). In addition, we used fetal bovine serum (Hyclone Laboratories, Logan, UT), EGF (Peprotech, Rocky Hill, NJ), The neuronal media supplements N2 supplement B (Stem Cell Technologies, Tukwila, WA) and B27 (Invitrogen, Carlsbad, CA). Tissue culture plastics and pipettes were from VWR International (Radnor, PA) and Fisher Scientific (Pittsburgh, PA). All reagents, unless otherwise specified were from Sigma-Aldrich Corp. (St. Louis, MO).
Animals
Male Sprague-Dawley rats weighing between 125 to 150 g (approximately 6–8 weeks of age) were obtained (Taconic Farms, Germantown, NY). All procedures used to obtain LGs were in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. These protocols were approved by the Schepens Eye Research Institute Animal Care and Use Committee. Rats were anesthetized with CO2 for 1 minute and decapitated, and both LGs were excised.
Immunofluorescence Microscopy (IF) for Tissue Sections
LGs were fixed in 4% paraformaldehyde in PBS (145 mM NaCl, 7.3 mM Na2HPO4, and 2.7 mM NaH2PO4 at pH 7.2) and cryopreserved in 30% sucrose in PBS. Glands were embedded in optimal cutting temperature embedding compound, and 6 μm sections were cut and placed on slides. Frozen sections of rat LGs were rinsed with PBS and blocked for 2 hours in buffer containing PBS with 0.2% Triton X-100 and 2% BSA. Sections were incubated for 2 hours at room temperature in a humidified chamber or overnight at 4°C with primary antibodies specific for progenitor cells in combination with α-SMA, which identifies myoepithelial cells. All primary antibodies were used at a dilution of 1:100 in PBS containing 0.2% BSA. Isotype negative controls consisted of normal goat IgG, normal mouse IgG2a, and normal rabbit IgG at dilutions of 1:100. Sections then were rinsed three times with PBS, after which secondary antibodies (1:300 dilution) were added for 1 hour at room temperature. Sections were rinsed three times in PBS and coverslips were mounted on the slides using polyvinyl alcohol (PVA) with 1,4 diazabicyclo[2.2.2]octane (DABCO) to prevent photobleaching. Sections were analyzed for the expression of progenitor cell markers using a Leica TSC-SPC upright laser scanning confocal microscope.
Isolation and Growth of Lacrimal Gland Cells
Total gland digestion.
LGs were removed and rinsed in PBS containing antibiotics, finely minced, and subjected to four 20-minute cycles of digestion in Type I collagenase at 37°C. Following each 20-minute digestion, liberated cells were collected, forced through a 0.4 μm mesh, pelleted and resuspended in 3 mL of RPMI 1640 culture medium supplemented with 10% fetal bovine serum, 2 mM L-glutamine and 100 μg/mL penicillin-streptomycin (complete RPMI). The cells were divided evenly into each well of a 6-well culture plate. Cells were re-fed with complete RPMI every two days.
Isolation of acini.
LGs were minced and incubated at 37°C with collagenase CLSIII (100 U/mL) in Krebs-Ringer bicarbonate buffer with HEPES (KRB-HEPES, 119 mM NaCl, 4.8 mM KCl, 1.0 mM CaCl2, 1.2 mM MgSO4, 1.2 mM KH2PO4, 25 MM NaHCO3, 10 mM HEPES, and 5.5 mM glucose, pH 7.45) plus 0.5% BSA. After incubation, fragments were triturated, filtered through nylon mesh (150-μm pore size), and centrifuged at 50 g for 2 minutes. The pellet was washed twice through KRB-HEPES containing 4% BSA. Following isolation of acini, they were placed in 6-well culture dishes (5 mg protein per well) and re-fed with complete RPMI medium every two days.
Growth of Cultured Immature Cells
Cells from the digestion of the entire LG and from acini were grown in culture initially in complete RPMI medium with 10% FBS. As soon as round immature cells appeared (approximately 2–4 weeks), RPMI was diluted gradually with serum-free KBM medium containing the following supplements: insulin 5 μg/ml; hydrocortisone 0.5 μg/ml; EGF 10 ng/mL and bovine pituitary extract 25 μg/ml. Cells were grown first in a mixture of 75% RPMI:25% KBM for two days, then 50% RPMI:50% KBM for two days, followed by 25% RPMI:75% KBM for two days and finally in 100% KBM, which maintained the immature cells in an undifferentiated state.
Immunocytochemistry of Immature Cells
Undifferentiated, immature cells, either single cells or cells that had formed spheres, were evaluated by immunofluorescence microscopy for their expression of various progenitor cell markers. We evaluated cells grown either in the culture dish following their appearance or on glass coverslips that were seeded at the time the LG cells were digested. The same protocol described above for IF of tissue sections was used in these experiments. Cells and spheres were incubated with primary antibodies at concentrations of 1:100 in PBS for 2 hours at room temperature followed by incubation with the appropriate Cy3 secondary antibodies at a dilution of 1:300 in PBS. An Olympus fluorescence microscope was used to determine expression of progenitor cell markers in these cells.
Determination of Immature Cell Plasticity
Contaminating cell types were scraped away from the immature cells and nodules that appeared following lacrimal gland digestion. Cells/nodules then were trypsinized and placed in three specific medias to examine their plasticity. The first media was a neuronal medium, which consisted of X-Vivo medium supplemented with 10% FBS, 20 ng/mL EGF, 20 ng/mL bFGF, 100 μg/mL N2 neural supplement and 50 μg/mL B27 neural supplement. The second media consisted of OptMEM-I supplemented with 8% FBS, 5 ng/mL epidermal growth factor, 20 ng/nL nerve growth factor, 100 μg/mL pituitary extract, 20 μg/mL ascorbic acid, 200 μg/mL calcium chloride, 0.08% chondroitin sulfate, and antibiotic/antimycotic solution diluted 1/100. This media has been demonstrated to promote corneal endothelial cell growth.19,20 The third media was RPMI 1640 media supplemented with 10% FBS, 2 mM l-glutamine and 100 μg/mL streptomycin-penicillin, which promotes epithelial cell growth.21,22 Cultures were re-fed every second day and morphology was monitored using an Olympus inverted microscope. IF was performed on cultures following two weeks of growth in their respective medias. Cells were evaluated for the neuronal marker neurofilament-200 at a dilution of 1:1000 in PBS, the epithelial cell markers AE1/AE3, a pan-cytokeratin marker, and cytokeratin 7 at a dilution of 1:100 in PBS. The secondary antibody was Cy3 at a dilution of 1:300 in PBS. Expression of markers was visualized using an Olympus inverted fluorescence microscope.
Results
Detection of Progenitor Cells in Normal Rat Lacrimal Gland
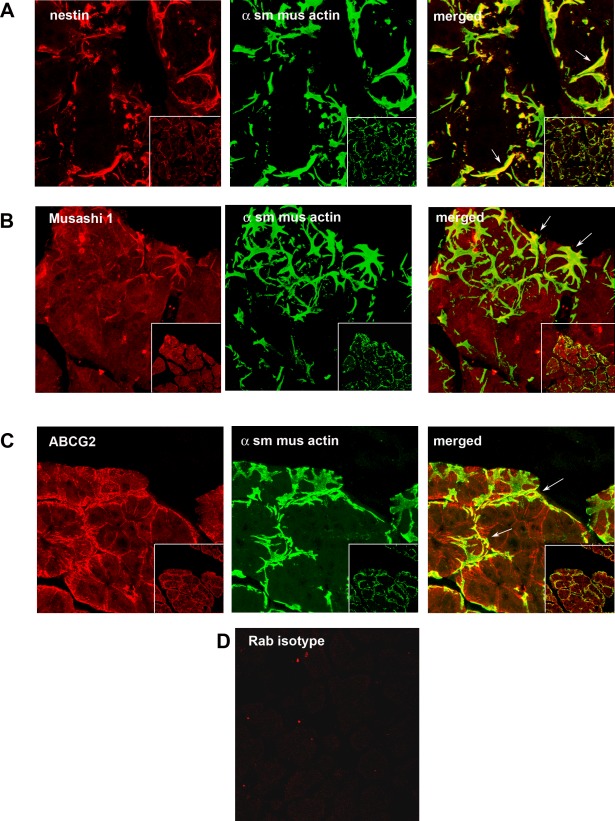
Recent reports indicate that stem/progenitor cells appear transiently in the IL-1 injected mouse LGs until the injury is resolved.14,15 To determine if progenitor cells are present in healthy, uninjured LGs, adult rat LGs were examined by confocal microscopy for proteins that are expressed in stem cells. One such protein, nestin is a type VI intermediate filament, which is expressed in dividing cells during early stages of development in the central nervous system.23 Nestin was not present in the acinar cells (Fig. 1A). Sections also were double labeled with an antibody directed against α-SMA as there is evidence to suggest that myoepithelial cells (which express α-SMA) may be the source of progenitor cells in the mammary and lacrimal glands and MECs surround acini.15,24 In the LG, nestin was colocalized with α-SMA (Fig. 1A).
Figure 1.
Presence of progenitor cell markers in LG. Rat LG sections were analyzed by confocal IF using the progenitor cell markers nestin (A), Musashi 1 (B), and ABCG2 (C), all shown in red in left column. Sections also were double labeled with the myoepithelial cell marker α-SMA (α sm mus actin), shown in green in the middle column. Merged images showing colocalization of two markers is shown in the right column. The rabbit (rab) isotype control is shown in (D). Arrows indicate myoepithelial cells. Magnification 900×, inset 400×.
Musashi 1 is an RNA-binding protein that is expressed in proliferating multipotent neural precursor cells and disappears in differentiated cells.25 In the uninjured, healthy LG, Musashi 1 appeared throughout the acinar cells in a punctate pattern (Fig. 1B). In addition, Musashi 1 colocalized with α-SMA (Fig. 1B), indicating that it is present in MECs.
ABCG2 is a member of the ATP binding cassette transporters, which transport molecules across extra- and intracellular membranes, and is present in a variety of pluripotent stem cells from various adult sources.26 In the normal, untreated LG, ABCG2 was present in the lateral membranes of the acinar cells (Fig. 1C). ABCG2 also is present in MECs as it colocalized with α-SMA (Fig. 1C). The isotype, negative control is shown in Figure 1D.
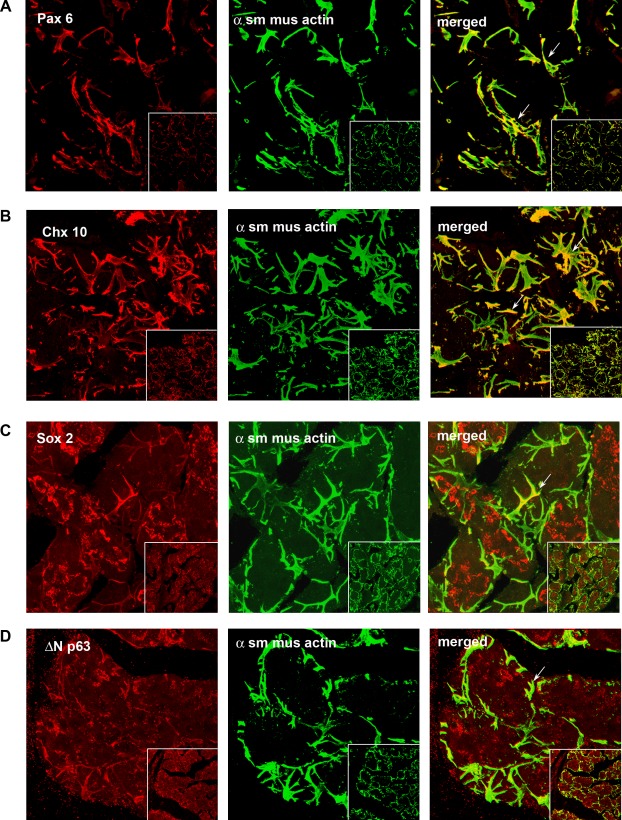
Additional proteins expressed early in development and in progenitor cells include the transcription factors Pax 6, Chx 10, Sox 2, and ΔN p63.27–30 We examined the expression of these proteins in uninjured, adult LGs. As shown in Figure 2A, Pax 6 was not seen in acinar cells, but appeared to be exclusively in MECs, as demonstrated by co-localization with α-SMA. Similar localization was seen with Chx 10 (Fig. 2B). Sox 2 was localized to the cytoplasm of acinar cells and colocalized with α-SMA (Fig. 2C). ΔN p63 appeared in a punctate pattern in acinar cells and also was localized with MECs (Fig. 2D).
Figure 2.
Presence of progenitor cell markers in LG. Rat LG sections were analyzed by confocal IF using the progenitor cell markers Pax 6 (A), Chx 10 (B), Sox 2 (C) and ΔN p63 (D), all shown in red in left column. Sections also were double labeled with the myoepithelial cell marker α-SMA (α sm mus actin), shown in green in the middle column. Merged images showing colocalization of two markers is shown in the right column. Arrows indicate myoepithelial cells. Magnification 900×, inset 400×.
The uninjured adult LG appears to contain progenitor cells that are MECs.
Growth of Immature Cells Isolated from LGs
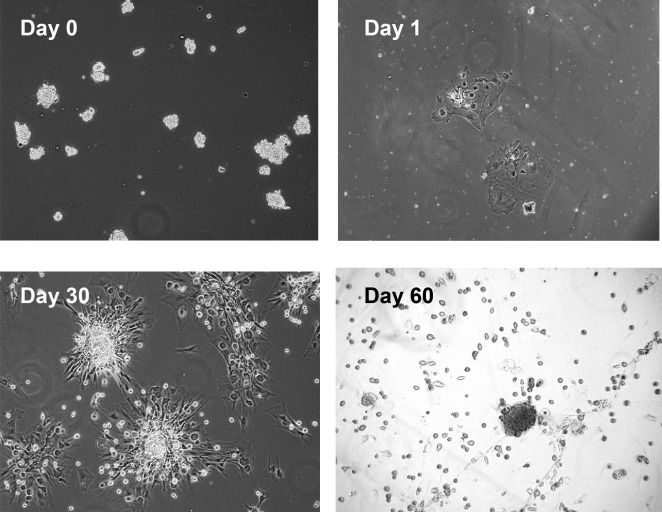
Most of the progenitor cell markers used in the present study colocalized with α-SMA, suggesting an MEC location. Since previously we developed a method to culture rat LG MECs,5 we determined if progenitor cells could be propagated from the normal LG using methods used to grow MECs. Cells isolated from the lacrimal gland or acini were placed in culture as described in the Methods and cell growth was followed by light microscopy. Immature cells were isolated consistently using both methods, and the two methods yielded identical growth of cells. As shown in Figure 3, small clumps of cells were plated on day 0. On day 1, cells had attached. After 7–14 days, small, round immature cells appeared on the surface of the underlying monolayer (data not shown). By day 30, the small cells had aggregated to form sphere-like structures. By day 60 the small cells were released from the aggregates.
Figure 3.
Growth of immature cells from rat LG. Rat LG was digested with collagenase and placed into culture. Light micrographs documenting the appearance of immature cells at Days 0, 1, 30, and 60. Magnification 100×.
Detection of Progenitor Cell Markers in Immature Cells Isolated from Normal LG
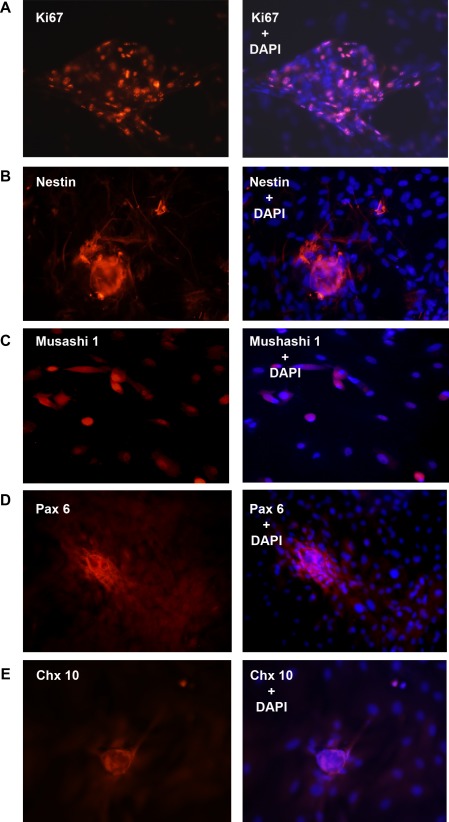
To determine if immature cells in culture are proliferating, the presence of Ki-67, a nuclear protein present during all stages of cell division, was determined. As shown in Figure 4A, cells within the sphere or cells growing from the sphere expressed Ki-67, indicating these cells are proliferating, a characteristic of progenitor cells in culture.
Figure 4.
Presence of immature cell markers in rat LG progenitor cells. Immature cells from rat LG were isolated and analyzed by IF using progenitor cell markers Ki67 (A), nestin (B), Musashi 1 (C), Pax 6 (D), and Chx 10 (E), all shown in red in the left column. Cells also were double labeled with the nuclear marker DAPI, shown in blue. Colocalization of two antibodies is shown in right column. Magnification 200×.
Immature cells and spheres of undifferentiated cells also were analyzed for progenitor cell markers by IF. The progenitor cell markers, nestin, Musashi 1, Pax 6, and Chx 10, which were present in MECs in LG sections, also were present in undifferentiated spheres (Figs. 4B–E).
Plasticity of Immature Cells
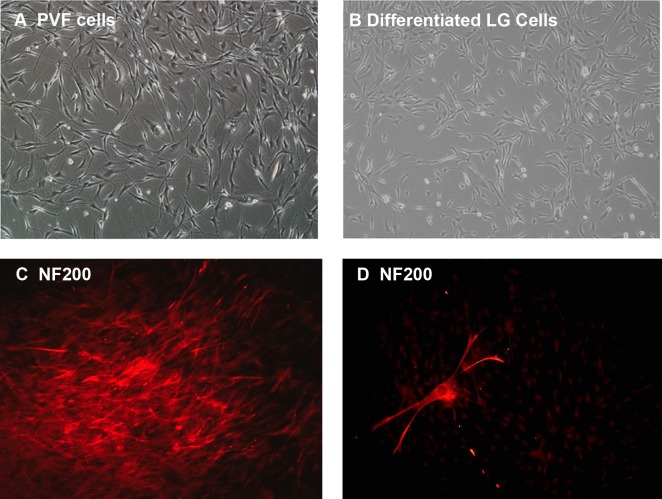
We next examined whether the immature cells in culture exhibited plasticity, a known characteristic of progenitor cells. Immature cells were isolated and placed in neuronal media. Cells were fed every other day and monitored for morphological changes. Within one week, the immature cells began to assume the morphology of persistent fetal vascular (PFV) cells, a neuro-retinal cell line. Light micrograph photographs of PFV cells are shown in Figure 5A, while LG differentiated cells grown in neuronal media for 2 weeks are shown in Figure 5B. The morphology of the LG differentiated cells was similar to that of the PFV cells. IF of LG progenitor cells in this media was performed to determine whether these cells expressed any markers of differentiated neural cells, including neurofilament 200 (NF200) and microtubule-associated protein 5 (MAP5) as well the astrocyte marker glial fibrillary acidic protein (GFAP). Many cells expressed NF200 (Fig. 5C) and some cells developed the distinct morphology of neural cells (Fig. 5D). An occasional cell expressed MAP5 (data not shown), but no cells expressed GFAP.
Figure 5.
Differentiation of rat LG immature cells in neuronal media. Progenitor cells from retinal cell line (PVF, [A]) and immature cells from LG (B) were grown in neuronal media for two weeks. Differentiated LG cells were then stained with the neuronal marker neurofilament 200 (NF200). A sphere of differentiated cells is shown in (C), while an individual cell is shown in (D). Magnification 100× (A, B), 200× (C), and 400× (D).
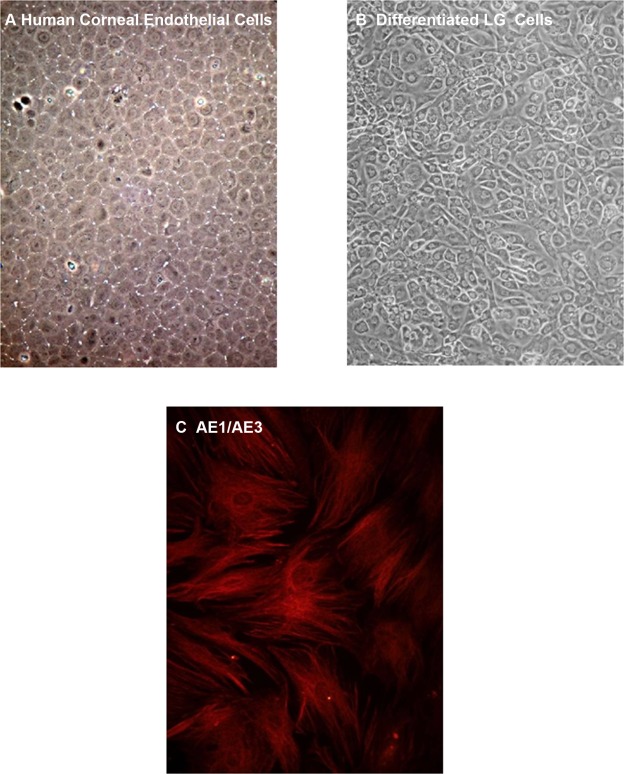
LG immature cells also were placed in media known to promote growth of corneal endothelial cells.19,20 As a control, corneal endothelial cells grown in this media assumed the typical cobblestone morphology (Fig. 6A).16,17 After 2–3 weeks in culture, LG differentiated cells assumed a cobblestone morphology similar to corneal endothelial cells (Fig. 6B). The LG differentiated cells stained positively for the pan-cytokeratin marker AE1/AE3, which also is expressed in corneal endothelial cells (Fig. 6C).31
Figure 6.
Differentiation of rat LG immature cells in corneal endothelial cell media. Human corneal endothelial cells (A) and immature cells from LG (B) were grown in media developed for corneal endothelial cells for two weeks. Differentiated LG immature cells then were stained with the epithelial cell marker cytokeratin AE1/AE3 (C). Magnification 100× (A, B) and 200× (C).
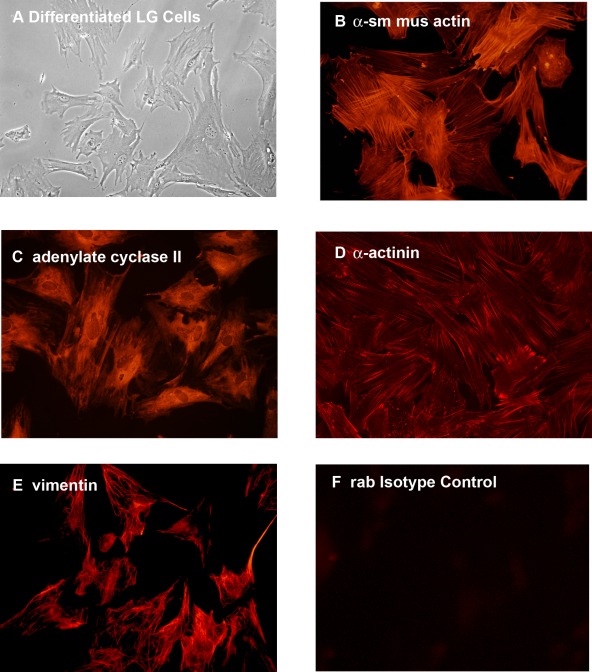
Finally, LG immature cells were grown in complete RPMI, a media that promotes the growth of epithelial cells. Cells that differentiated in this media were elongated and many had ruffled edges, characteristics of epithelial cells (Fig. 7A). In addition, these cells expressed proteins that are present in LG MECs, including the α-SMA (Fig. 7B), adenylate cyclase II (Fig. 7C), α-actinin (Fig. 7D), and vimentin (Fig. 7E).5 Incubation of cells with the isotype control showed no staining (Fig. 7F).
Figure 7.
Differentiation of rat LG immature cells in epithelial cell media. Immature cells from LG (A) were grown in RPMI media, which selects for epithelial cells for two weeks. Differentiated LG cells then were stained with the markers for LG myoepithelial cells α-SMA (α sm mus actin, [B]), adenylate cyclase II (C), α-actinin (D), and vimentin (E). Rabbit (rab) isotype control is shown in (F). Magnification 400×.
Immature cells isolated from uninjured, adult rat LGs appear to be progenitor cells, as they express progenitor cell markers and differentiate into multiple cell lineages.
Discussion
Tissue regeneration and self-repair is inherent to most tissues to replace defective cells so that healing and normal function can occur. A rapidly evolving area of research has described the presence of stem/progenitor cells in non-diseased, uninjured adult tissues. Progenitor cells from adult tissues are capable of self-renewal through asymmetric division, and simultaneously can generate committed progenitor cells and transient amplifying cells. The latter continue to differentiate into tissue-specific cell types that maintain the tissue population of cells. The adult progenitor cells also can function in disease or injury to replace damaged or dead cells.
In the present study we demonstrated that adult progenitor cells can be isolated and propagated from adult, non-diseased, uninjured lacrimal gland. Using a battery of stem cell markers and transcription factors, we identified the MECs as the progenitor cells. Markers of progenitor cells labeled MECs in LG sections as well as immature cells in culture. Our study is in agreement with those of Zoukhri et al., who also found that MECs could be a progenitor cell in the mouse lacrimal gland.14 The progenitor cells isolated by Zoukhri et al. also could be stained with the same markers used in the present study.14 In contrast, however, in mouse lacrimal gland Zoukhri et al. were unable to isolate progenitor cells from uninjured LG, but needed to inject IL-1, which caused massive cell loss in the LG.14 There are two potential reasons for the difference between the two studies, use of different culture media and use of different species. First, Zoukhri et al. used DMEM to culture the LG cells,14 whereas we used RPMI and then gradually changed the media to KBM. Second, Zoukhri et al. used mouse instead of rat lacrimal gland.14 Either of these explanations is possible.
The isolation of progenitor cells was reliable and robust. Progenitor cells were isolated from collagenase-digested glands as well as isolated acini. In addition progenitor cells were obtained from every gland used.
In the present study, we found that most myoepithelial cells were identified by the progenitor cell markers used. Stem cells are expected to represent a small population of cells. It is possible that we identified a population of cells that were more differentiated than stem cells, but still capable of plasticity, and thus were more prevalent. In that case, the more immature cells may be in a niche provided by the MECs and not the MECs themselves. In this context You et al. found that a population of mesenchymal cells functioned as stem cells to repair the LG after injury.16 A second possibility for the large population of progenitor cells could be the rapid time-course of repair demonstrated by the IL-1 injected murine LG. One injection of IL-1 caused a massive destruction of the LG that repaired in 2–3 days.14 In contrast in the ligated duct model of salivary gland repair, it takes 2–3 weeks for damage to injure the gland and repair to occur.32–34 A large population of progenitor cells would be needed for the lacrimal gland repair. No data are available to discriminate between these possibilities.
Stem cells have been identified in the other exocrine tissues. In parotid, submandibular and sublingual salivary glands, ductal ligation damages the gland, which is repaired by a population of cells that reside in the ductal compartment.10,12,13 These stem cells repair the three main cell types of the salivary gland, the acinar, ductal, and myoepithelial cells. Sialospheres (analogous to neurospheres), composed of salivary gland stem cells, also can repair the damaged gland.35 In the mouse mammary gland duct (luminal) cells also appear to function as stem cells.36 The duct cells are able to give rise to three different cell types, acinar, ductal, and myoepithelial, whereas myoepithelial cells only give rise to myoepithelial cells. Thus, to date, the LG appears to be different than the salivary and mammary glands, as either MECs (the present study) or mesenchymal cells (You et al.16) function as the progenitor cells in the LG.
One important characteristic of progenitor cells is that they exhibit plasticity and can differentiate into multiple cell types. In the present study, we cultured the LG progenitor cells in three different types of media. The cells differentiated into MECs in RPMI, epithelial cells in corneal endothelial cell medium, and neuronal cells in neuronal media supplemented X-Vivo. In the study by You et al., the mouse LG progenitor cells differentiated into one other cell type, adipocytes, when cultured in adipogenic media.16 Thus, isolated LG progenitor cells are plastic.
In conclusion, the uninjured, non-diseased, adult rat LG contains progenitor cells that can be isolated and propagated, and these cells can differentiate into three different cell types. The progenitor cells could be MECs or the MECs could provide a progenitor cell niche.
Footnotes
Supported by NIH Grants EY06177 and EY020992.
Disclosure: M.A. Shatos, None; L. Haugaard-Kedstrom, None; R.R. Hodges, None; D.A. Dartt, None
References
- 1. Hodges RR, Dartt DA. Regulatory pathways in lacrimal gland epithelium. Int Rev Cytol. 2003;231:129–196 [DOI] [PubMed] [Google Scholar]
- 2. Deugnier MA, Teulière J, Faraldo MM, Thiery JP, Glukhova MA. The importance of being a myoepithelial cell. Breast Cancer Res. 2002;4:224–230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lemullois M, Rossignol B, Mauduit P. Immunolocalization of myoepithelial cells in isolated acini of rat exorbital lacrimal gland: cellular distribution of muscarinic receptors. Biol Cell. 1996;86:175–181 [DOI] [PubMed] [Google Scholar]
- 4. Hodges RR, Zoukhri D, Sergheraert C, Zieske JD, Dartt DA. Identification of vasoactive intestinal peptide receptor subtypes in the lacrimal gland and their signal-transducing components. Invest Ophthalmol Vis Sci. 1997;38:610–619 [PubMed] [Google Scholar]
- 5. Ohtomo K, Shatos MA, Vrouvlianis J, Li D, Hodges RR, Dartt DA. Increase of intracellular Ca2+ by purinergic receptors in cultured rat lacrimal gland myoepithelial cells. Invest Ophthalmol Vis Sci. 2011;52:9503–9515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Satoh Y, Sano K, Habara Y, Kanno T. Effects of carbachol and catecholamines on ultrastructure and intracellular calcium-ion dynamics of acinar and myoepithelial cells of lacrimal glands. Cell Tissue Res. 1997;289:473–485 [DOI] [PubMed] [Google Scholar]
- 7. Zoukhri D. Effect of inflammation on lacrimal gland function. Exp Eye Res. 2006;82:885–898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Zoukhri D. Mechanisms involved in injury and repair of the murine lacrimal gland: role of programmed cell death and mesenchymal stem cells. Ocul Surf. 2010;8:60–69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Scoggins CR, Meszoely IM, Wada M, Means AL, Yang L, Leach SD. p53-dependent acinar cell apoptosis triggers epithelial proliferation in duct-ligated murine pancreas. Am J Physiol Gastrointest Liver Physiol. 2000;279:G827–836 [DOI] [PubMed] [Google Scholar]
- 10. Burford-Mason AP, Cummins MM, Brown DH, MacKay AJ, Dardick I. Immunohistochemical analysis of the proliferative capacity of duct and acinar cells during ligation-induced atrophy and subsequent regeneration of rat parotid gland. J Oral Pathol Med. 1993;22:440–446 [DOI] [PubMed] [Google Scholar]
- 11. Walker NI, Bennett RE, Kerr JF. Cell death by apoptosis during involution of the lactating breast in mice and rats. Am J Anat. 1989;185:19–32 [DOI] [PubMed] [Google Scholar]
- 12. Takahashi S, Nakamura S, Suzuki R, et al. Apoptosis and mitosis of parenchymal cells in the duct-ligated rat submandibular gland. Tissue Cell. 2000;32:457–463 [DOI] [PubMed] [Google Scholar]
- 13. Takahashi S, Shinzato K, Nakamura S, Domon T, Yamamoto T, Wakita M. Cell death and cell proliferation in the regeneration of atrophied rat submandibular glands after duct ligation. J Oral Pathol Med. 2004;33:23–29 [DOI] [PubMed] [Google Scholar]
- 14. Zoukhri D, Macari E, Kublin CL. A single injection of interleukin-1 induces reversible aqueous-tear deficiency, lacrimal gland inflammation, and acinar and ductal cell proliferation. Exp Eye Res. 2007;84:894–904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Zoukhri D, Fix A, Alroy J, Kublin CL. Mechanisms of murine lacrimal gland repair after experimentally induced inflammation. Invest Ophthalmol Vis Sci. 2008;49:4399–4406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. You S, Kublin CL, Avidan O, Miyasaki D, Zoukhri D. Isolation and propagation of mesenchymal stem cells from the lacrimal gland. Invest Ophthalmol Vis Sci. 2011;52:2087–2094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Feng Z, Li C, Jiao S, Hu B, Zhao L. In vitro differentiation of rat bone marrow mesenchymal stem cells into hepatocytes. Hepatogastroenterology. 2011;58:2081–2086 [DOI] [PubMed] [Google Scholar]
- 18. Kumar A, Gao H, Xu J, Reuben J, Yu D, Mehta K. Evidence that aberrant expression of tissue transglutaminase promotes stem cell characteristics in mammary epithelial cells. PLoS One. 2011;6:e20701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Chen KH, Azar D, Joyce NC. Transplantation of adult human corneal endothelium ex vivo: a morphologic study. Cornea. 2001;20:731–737 [DOI] [PubMed] [Google Scholar]
- 20. Joyce NC, Harris DL. Decreasing expression of the G1-phase inhibitors, p21Cip1 and p16INK4a, promotes division of corneal endothelial cells from older donors. Mol Vis. 2010;16:897–906 [PMC free article] [PubMed] [Google Scholar]
- 21. Shatos MA, Rios JD, Horikawa Y, et al. Isolation and characterization of cultured human conjunctival goblet cells. Invest Ophthalmol Vis Sci. 2003;44:2477–2486 [DOI] [PubMed] [Google Scholar]
- 22. Shatos MA, Rios JD, Tepavcevic V, Kano H, Hodges R, Dartt DA. Isolation, characterization, and propagation of rat conjunctival goblet cells in vitro. Invest Ophthalmol Vis Sci. 2001;42:1455–1464 [PubMed] [Google Scholar]
- 23. Reynolds BA, Weiss S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science. 1992;255:1707–1710 [DOI] [PubMed] [Google Scholar]
- 24. Van Keymeulen A, Rocha AS, Ousset M, et al. Distinct stem cells contribute to mammary gland development and maintenance. Nature. 2011;479:189–193 [DOI] [PubMed] [Google Scholar]
- 25. Sakakibara S, Imai T, Hamaguchi K, et al. Mouse-Musashi-1, a neural RNA-binding protein highly enriched in the mammalian CNS stem cell. Dev Biol. 1996;176:230–242 [DOI] [PubMed] [Google Scholar]
- 26. Ding XW, Wu JH, Jiang CP. ABCG2: a potential marker of stem cells and novel target in stem cell and cancer therapy. Life Sci. 2010;86:631–637 [DOI] [PubMed] [Google Scholar]
- 27. Osumi N, Shinohara H, Numayama-Tsuruta K, Maekawa M. Concise review: Pax6 transcription factor contributes to both embryonic and adult neurogenesis as a multifunctional regulator. Stem Cells. 2008;26:1663–1672 [DOI] [PubMed] [Google Scholar]
- 28. Fischer AJ, Reh TA. Identification of a proliferating marginal zone of retinal progenitors in postnatal chickens. Dev Biol. 2000;220:197–210 [DOI] [PubMed] [Google Scholar]
- 29. Violini S, Gorni C, Pisani LF, Ramelli P, Caniatti M, Mariani P. Isolation and differentiation potential of an equine amnion-derived stromal cell line. Cytotechnology. 2011;64:1–7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Hernandez Galindo EE Theiss C Steuhl KP Meller D. Expression of Delta Np63 in response to phorbol ester in human limbal epithelial cells expanded on intact human amniotic membrane. Invest Ophthalmol Vis Sci. 2003;44:2959–2965 [DOI] [PubMed] [Google Scholar]
- 31. Hirst LW, Bancroft J, Yamauchi K, Green WR. Immunohistochemical pathology of the corneal endothelium in iridocorneal endothelial syndrome. Invest Ophthalmol Vis Sci. 1995;36:820–827 [PubMed] [Google Scholar]
- 32. Hisatomi Y, Okumura K, Nakamura K, et al. Flow cytometric isolation of endodermal progenitors from mouse salivary gland differentiate into hepatic and pancreatic lineages. Hepatology. 2004;39:667–675 [DOI] [PubMed] [Google Scholar]
- 33. Okumura K, Nakamura K, Hisatomi Y, et al. Salivary gland progenitor cells induced by duct ligation differentiate into hepatic and pancreatic lineages. Hepatology. 2003;38:104–113 [DOI] [PubMed] [Google Scholar]
- 34. Zhang YQ, Kritzik M, Sarvetnick N. Identification and expansion of pancreatic stem/progenitor cells. J Cell Mol Med. 2005;9:331–344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Lombaert IM, Brunsting JF, Wierenga PK, et al. Rescue of salivary gland function after stem cell transplantation in irradiated glands. PLoS One. 2008;3:e2063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Regan J, Smalley M. Prospective isolation and functional analysis of stem and differentiated cells from the mouse mammary gland. Stem Cell Rev. 2007;3:124–136 [DOI] [PubMed] [Google Scholar]