Abstract
Objective
Late-preterm birth (LPB: 34–36 weeks) has been associated with an increased risk of attention problems in childhood relative to full-term birth (FTB: ≥ 37 weeks), but little is known about factors contributing to this risk. We investigated the contributions of clinical circumstances surrounding delivery using follow-up data from the Pregnancy Outcomes and Community Health (POUCH) Study.
Methods
Women who delivered late-preterm or full-term and completed the sex- and age-referenced Conners’ Parent Rating Scales-Short Form: Revised (CPRS-R:S) were included in the present analysis (N=762: children’s ages: 3–9 years). The CPRS-R:S measures dimensions of behavior linked to attention problems, including: oppositionality, inattention, hyperactivity, and a global attention problem index. Using general linear models, we evaluated whether LPB subtype (medically indicated (MI) or spontaneous) was associated with these dimensions relative to FTB.
Results
After adjustment for parity, socio-demographics, child age, and maternal symptoms of depression and serious mental illness during pregnancy and at the child survey, only MI LPB was associated with higher hyperactivity and global index scores (mean difference from FTB=3.8 [95%CI 0.5,7.0] and 3.1 [95%CI 0.0, 6.2]). These findings were largely driven by children between 6 and 9 years. Removal of women with hypertensive disorders during pregnancy (N=85) or placental findings related to hypertensive conditions (obstruction, decreased maternal spiral artery conversion; N=134) reduced the differences below significance thresholds.
Conclusion
Among LPBs, only MI LPB was associated with higher levels of parent-reported childhood attention problems, suggesting that complications motivating medical intervention during the late-preterm period mark increased risk for such problems. Hypertensive disorders appear to play a role in these associations.
Introduction
From 1998–2008, rates of preterm delivery (< 37 weeks of gestation) increased by 20% in the United States, a trend largely attributed to the rise in deliveries between 34–36 weeks (late-preterm birth (LPB)).1,2 Although many secular trends accompany this pattern (e.g., multifetal gestations), nearly 75% of all singleton preterm deliveries can be accounted for by LPB.1,3 Combined with mounting evidence suggesting that children born late-preterm are at increased risk for neonatal mortality and a range of neonatal morbidities,4–6 the antecedents and sequelae associated with LPB have received much attention from the medical and public health communities.
LPB has also been associated with long-term cognitive and behavioral problems, including cerebral palsy, reading impairments, lower IQ scores, and higher levels of internalizing and attention-related problems at school-age.7–12 Retrospective population-based investigations from Europe suggest that adults born late-preterm exhibit elevated risks for psychiatric diagnoses as well as socio-economic hardship,13,14 providing further evidence of long-term impairment among LPBs.13,14 Although little is known about factors contributing to such risks, there is increasing evidence that perinatal health may play a role. For example, children born late-preterm who were admitted to the neonatal intensive care unit (NICU) were more likely to exhibit deficits in non-verbal processing in relation to their full-term peers and late-preterm children not admitted to the NICU; no differences between the full-terms and non-NICU admitted LPBs were observed.15 Conversely, while LPB was unrelated to cognitive functioning, behavioral problems, and academic achievement in a longitudinal study spanning childhood and adolescence, analyses excluded pregnancies marked by perinatal health risks (e.g., maternal infection, substance use).16 Delivery circumstance (i.e., medically indicated or spontaneous) may also shed light on LPB associations with long-term child development outcomes,7 but this hypothesis has not been subjected to empirical investigation. Doing so may help identify etiological pathways that may be involved in any associations observed, given that delivery circumstance has been linked to specific indices of pre- and or postnatal complications.17
To address this issue, we used follow-up data from the Pregnancy Outcomes and Community Health (POUCH) Study, a prospective investigation of the etiological pathways leading to preterm delivery. Mothers provided reports of attention problems for the child resulting from the POUCH pregnancy using a norm-referenced instrument. To account for any long-term biological effects related to maternal mental health during pregnancy as well as reporting bias due to such characteristics at the time of the child survey, adjusted analyses incorporated measures of maternal psychosocial functioning obtained at each of these time periods. Our goals were to: 1) replicate previous findings linking LPB to higher levels of attention problems in childhood, and 2) examine whether delivery circumstances surrounding LPB (i.e., MI, spontaneous) were associated with these problems. In the event that significant associations were observed, we utilized additional medical record information and placental histology findings to investigate pathways that might be involved.
Methods
Participants
The POUCH Study, designed to examine etiological pathways leading to preterm delivery, enrolled 3019 women (15–27 weeks of gestation) from 52 prenatal clinics in five Michigan communities from 1998–2004.18 Eligibility criteria included English proficiency and a singleton pregnancy with no known birth defects, chromosomal anomalies, or preexisting diabetes. All interested, eligible women with maternal serum alpha-fetoprotein (MSAFP) ≥ 2.0 multiples of the median (MoM) and a race-stratified sample of women with MSAFP < 2.0 MoM were included in the POUCH cohort. For a subset oversampled for African-American race, high MSAFP, and preterm delivery (i.e., subcohort: N=1371), in-depth medical record abstraction was performed and placental samples were collected. This sampling scheme was employed to maximize resources when investigating at-risk subgroups,18 but we excluded early preterm deliveries (< 34 weeks, N=68) here given the topic at hand. This study was approved by the Institutional Review Boards of Michigan State University and participating medical centers.
In 2007–2008, women were invited to complete a survey about their health and that of the child born during the POUCH Study (i.e., POUCHchild Survey). Exclusion criteria included women who: 1) declined participation in future studies (N=70), 2) did not have legal custody of the POUCH child (N=10), or 3) were deceased or whose child was deceased (N=12), yielding 1211 subcohort women eligible for participation. Of these, 780 (64%) completed the POUCHchild Survey.
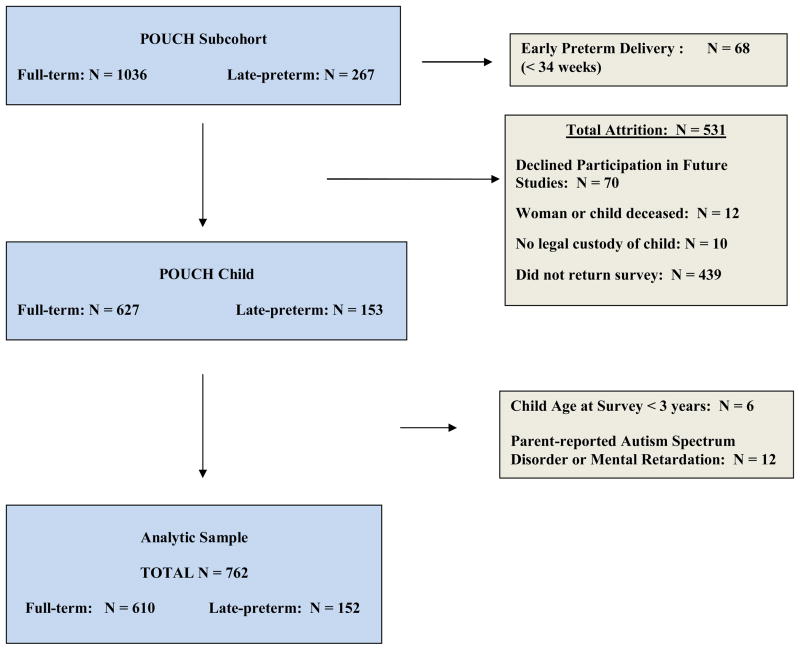
To preserve the fidelity of attention problem measurement, children whose mothers reported that they were diagnosed with mental retardation or an autism spectrum disorder (N=12), or were less than 3 years of age at the time of the survey (N=6) were excluded prior to analysis. Thus, the sample size for the present study is 762 with children’s ages ranging between 3 and 9 years (Late-preterm (34–36 weeks): N=152; Full-term (≥ 37 weeks): N=610) (Figure 1).
Figure 1.
Analytic Sample Derivation
Measures
Pregnancy Outcomes
Gestational age was estimated using last menstrual period unless it was unavailable or differed from the ultrasound estimate (at < 25 weeks) by more than two weeks. In these cases (20% of the subcohort; 15% of the analytic sample), the ultrasound-based estimate was employed. Medical records were independently abstracted by a physician and labor and delivery nurse, and two categories of preterm delivery circumstances were described: 1) spontaneous: women with regular contractions that led to cervical changes (≥ 2 cm of dilatation) or rupture of membranes before or simultaneously with the onset of contractions, and 2) medically indicated: women induced or given caesarean sections before either preterm labor or rupture of membranes. The source of gestational age estimation was unrelated to pregnancy outcome (data not shown).
Using gestational age, sex, and birthweight information abstracted from medical records, children were identified as small for gestational-age (SGA) using estimates corresponding to the 10th percentile.19
Attention Problems
Mothers completed the Conners’ Parent Rating Scales-Revised: Short Form (CPRS-R:S), an instrument yielding sex- and age-referenced T-scores for three dimensions of behavior associated with attention deficit/hyperactivity disorder (ADHD), including: oppositionality (irritability, anger-proneness, and defiance), cognitive problems/inattention (difficulty concentrating, planning, or maintaining attention on tasks requiring sustained mental effort), and hyperactivity (impulsivity and restlessness).20 The CPRS-R:S also yields a global ADHD Index, which measures elements of both inattention and hyperactivity. T-scores were normally distributed and not transformed prior to analysis. The CPRS -R:S is normed for 3–17 years olds and exhibits discriminate validity. That is, children diagnosed attention deficit hyperactivity disorder (ADHD) score significantly higher on each of the CPRS-R:S scales compared to non-clinical samples as well as children exhibiting other types of psychopathologies (e.g., anxiety disorders).20
Maternal Psychosocial Functioning
At POUCH Study enrollment, women completed the Center for Epidemiologic Studies Depression Scale (CES-D).21 Women were grouped according to whether their total scores were equal to or above 24, a threshold that has been associated with adverse perinatal outcomes.22 At POUCHchild Survey completion, women completed the Kessler Scales,23 a validated instrument designed to screen for serious mental illnesses (SMIs) in the general population. SMIs are mental, behavioral, and emotional disorders associated with functional impairment in daily activities. Using thresholds described in previous research,23 total scores equal to or above 19 were defined as high.
Hypertensive Disorders
Evidence of hypertensive disorders (i.e., chronic hypertension (CH), gestational hypertension (GH), preeclampsia (PE)) was abstracted from medical charts. Diagnostic categories included: CH (diastolic blood pressure (DBP) > 90 or systolic blood pressure (SBP) > 140 on at least two occasions prior to 20 weeks, medical record diagnosis, or use of anti-hypertensive medication), GH (no CH, DBP > 90 or SBP > 140 on at least two occasions after 20 weeks), and PE (same criteria as GH plus evidence of proteinuria).24–26
Placental Pathology
Placentas were obtained from 88% of the subcohort and, to date, are available for 85% of women included in the present analysis (N=649). Nine samples per placenta (placental disc (5), membrane roll (2), umbilical cord (2)) were examined by the study pathologist blinded to all clinical circumstances surrounding delivery. Microscopic evidence of vascular pathology was evaluated and grouped into five constructs, detailed elsewhere.27 Here, we focus on two constructs that have been linked to the presence of hypertensive disorders during pregnancy: Maternal Vascular–Obstructive (MV-O) (e.g., infarcts, decidual vessel atherosis), and Maternal Vascular–Developmental (MV-D) (e.g., abnormal/incomplete conversion of the uterine spiral arteries).28,29 To maximize power in data analysis, women were grouped according to whether they exhibited high levels of pathology in either the MV-O or MV-D construct.27
Covariates
Maternal self-reports of race (non-Hispanic white/other, African-American), education (≤ 12, 12 years), age (<25, ≥ 25 years), parity (primiparous, multiparous), and tobacco use (none, any) were obtained at POUCH Study enrollment. Child age was obtained at the time of the POUCHchild Survey.
Statistical Analysis
All analyses, both unadjusted and adjusted, were weighted for the over-sampling of women with high MSAFP into the cohort and high MSAFP, African-American race, and preterm delivery into the subcohort (see Participants). That is, these subsets of participants were given less weight in analyses because they were oversampled into the POUCH cohort and subcohort. All covariates noted above were included in the adjusted models along with indices of maternal psychosocial functioning.
Associations between LPB and attention problems were evaluated in three steps. First, to better contextualize any findings observed, general linear models (GLMs) comparing characteristics of the POUCH subcohort and the analytic sample were performed. In an attempt to replicate previous research findings, a second set of GLMs evaluated whether LPB, relative to FTB, was associated with higher levels of attention problems. Finally, GLMs were used to evaluate whether LPB circumstance was associated with attention problems relative to FTB and whether child sex or maternal symptoms of SMIs modified any observed findings. Due to concerns regarding the validity of assessments of attention problems at preschool-age,30 this final set of analyses were stratified by child age at survey (3 to < 6 years: N = 484; 6–9 years: N =278) to better contextualize any findings observed.
Because MI LPB was significantly associated with child attention problems (see Results/Table 3) and hypertensive conditions have been implicated in MI LPBs,3,28,31 the final models were repeated after removing women with indicators linked to hypertensive disorders during pregnancy (e.g., high MV-O/MV-D or SGA birth) to see if such factors partially or fully accounted for the findings. Because indices of inflammatory conditions and bleeding have also been associated with LPB,32,33 we repeated analyses following the removal of women with severe histologic chorioamnionitis (HCA) or placental abruption to further probe the specificity of any observed associations. Small cell sizes precluded the removal of these subgroups in age-stratified analyses.
Table 3.
Unadjusted and adjusted associations between pregnancy outcome and parent-reported attention problemsa,b
| Full Term (referent) N = 610 |
Late-Preterm N = 152 |
Late-Preterm Circumstance (N=152)
|
||||||
|---|---|---|---|---|---|---|---|---|
| Medically Indicated N=38 |
Spontaneous N = 114 |
|||||||
| Mean | [95%CI] | Mean | [95%CI] | Mean | [95%CI] | Mean | [95%CI] | |
| Oppositionality | ||||||||
| unadjusted | 51.5 | [50.5, 52.5] | 52.6 | [50.8, 54.4] | 53.6 | [50.0, 57.6] | 52.2 | [50.1, 54.3] |
| adjusted | 57.7 | [55.5, 60.1] | 58.6 | [55.7, 61.6] | 60.7 | [55.8, 66.0] | 57.9 | [54.9, 61.0] |
| Inattention | ||||||||
| unadjusted | 51.5 | [50.7, 52.4] | 52.0 | [50.4, 53.5] | 52.4 | [49.6, 55.4] | 51.8 | [50.0, 53.7] |
| adjusted | 56.1 | [54.0, 58.3] | 56.6 | [54.1, 59.2] | 57.0 | [53.2, 61.0] | 56.5 | [53.8, 59.3] |
| Hyperactivity | ||||||||
| unadjusted | 54.9 | [53.9, 56.0] | 55.6 | [53.8, 57.3] | 58.8* | [55.5, 62.0] | 54.5 | [52.4, 56.5] |
| adjusted | 62.5 | [60.0, 65.0] | 62.7 | [59.9, 65.6] | 66.3* | [62.3, 70.3] | 61.5 | [58.6, 64.5] |
| ADHD Index | ||||||||
| unadjusted | 53.2 | [52.3, 54.1] | 53.1 | [51.5, 54.8] | 56.2 | [53.1, 59.4] | 52.1 | [50.2, 53.9] |
| adjusted | 59.5 | [57.4, 61.5] | 59.3 | [56.9, 61.8] | 62.6* | [59.0, 66.2] | 58.2 | [55.6, 60.8] |
Weighting applied so that the analytic sample reflects the POUCH Subcohort.
Adjusted for: maternal race, education, marital status, parity, maternal age, tobacco use, and depressive symptoms during pregnancy, symptoms of serious mental illness at time of child survey, child sex, and child age at survey
p < 0.05 relative to full-term group
Results
Relative to the subcohort, participation in the follow-up study and inclusion in the analytic sample was significantly associated with older maternal age, higher levels of maternal education, and married marital status at POUCH Study enrollment and older child age at POUCHChild Study participation (Table 1). Pregnancy outcomes and their association with maternal, child, and placental characteristics in the analytic sample are summarized in Table 2. Compared to FTB, MI LPB was associated with African-American race, diagnosis of hypertensive disorders during pregnancy, and SGA birth. No covariates were associated with spontaneous LPB.
Table 1.
Maternal, child, and placental characteristics of the POUCH subcohort and the analytic samplea
| POUCH Subcohort (N = 1303)b |
Analytic Sample (N = 762)
|
|||
|---|---|---|---|---|
| N | (Wt %) | N | (Wt %) | |
| Maternal Characteristics | ||||
| Age (years)* | ||||
| < 25 | 622 | (43) | 322 | (39) |
| ≥ 25 | 681 | (57) | 440 | (61) |
| Education (years)* | ||||
| ≤ 12 | 672 | (46) | 329 | (41) |
| > 12 | 631 | (54) | 433 | (59) |
| Race | ||||
| White/Other | 754 | (76) | 504 | (76) |
| African-American | 549 | (24) | 258 | (24) |
| Parity | ||||
| Primiparous | 544 | (41) | 325 | (42) |
| Multiparous | 758 | (59) | 437 | (58) |
| Marital Status* | ||||
| Unmarried | 750 | (48) | 385 | (46) |
| Married | 549 | (52) | 377 | (54) |
| Tobacco Use | ||||
| No | 1054 | (82) | 637 | (84) |
| Yes | 249 | (18) | 125 | (16) |
| Hypertensive Disorders | ||||
| No | 1168 | (91) | 677 | (90) |
| Yes | 135 | (9) | 85 | (10) |
| Depressive Symptoms during Pregnancy | ||||
| CES-D < 24 | 1049 | (84) | 641 | (86) |
| CES-D ≥ 24 | 249 | (16) | 119 | (14) |
| Serious Mental Illness Symptoms: POUCHChild Survey | ||||
| K6 < 19 | 681 | (90) | ||
| K6 ≥ 19 | 81 | (10) | ||
| Child Characteristics | ||||
| Small for Gestational Age | ||||
| No | 1158 | (91) | 689 | (91) |
| Yes | 144 | (9) | 73 | (9) |
| Child Sex | ||||
| Male | 668 | (49) | 386 | (48) |
| Female | 635 | (51) | 376 | (52) |
| Child Age at Survey* | ||||
| 3 to < 6 years | 1019 | (71) | 484 | (61) |
| 6 to 9 years | 284 | (29) | 278 | (39) |
| Placental Characteristics | ||||
| High MV-O or MV-D | N = 1064 | N = 649 | ||
| No | 836 | (80) | 515 | (80) |
| Yes | 228 | (20) | 134 | (20) |
Abbreviations: CES-D (Center for Epidemiologic Studies Depression Scale); K6 (Kessler Screening Instrument for Serious Mental Illnesses); MV-D (Maternal Vascular Pathology: Developmental); MV-O (Maternal Vascular Pathology: Obstructive)
Note. Missing data from POUCH subcohort: Parity (N=1), Marital Status (N=4), Depressive Symptom during Pregnancy (N=5), Small for Gestational Age (N=1). Missing data from analytic sample: Depressive Symptoms during Pregnancy (N=2).
Weighted percents reflect the removal of early preterm deliveries (< 34 weeks) from the POUCH cohort (and thus, the subcohort) prior to adjustment for the POUCH Study sampling scheme.
Sample size reflects removal of early preterm deliveries (N=68)
p < 0.05
Table 2.
Maternal, child, and placental characteristics in relation to pregnancy outcome (N=762) a
| Full Term N=610 |
Late-Preterm Circumstance (N=152)
|
|||||
|---|---|---|---|---|---|---|
| Medically Indicated N = 38 |
Spontaneous N = 114 |
|||||
| N | (Wt %) | N | (Wt %) | N | (Wt %) | |
| Maternal Characteristics | ||||||
| Age (years) | ||||||
| < 25 | 256 | (36) | 14 | (37) | 52 | (45) |
| ≥ 25 | 354 | (64) | 24 | (63) | 62 | (55) |
| Education (years) | ||||||
| ≤ 12 | 259 | (38) | 15 | (38) | 55 | (48) |
| > 12 | 351 | (62) | 23 | (62) | 59 | (52) |
| Race* | ||||||
| White/Other | 390 | (83) | 28 | (73) | 86 | (77) |
| African-American | 220 | (17) | 10 | (27) | 28 | (23) |
| Parity | ||||||
| Primiparous | 250 | (42) | 17 | (46) | 58 | (51) |
| Multiparous | 360 | (58) | 21 | (54) | 56 | (49) |
| Marital Status | ||||||
| Unmarried | 308 | (41) | 19 | (49) | 58 | (50) |
| Married | 302 | (59) | 19 | (51) | 56 | (50) |
| Tobacco Use | ||||||
| No | 510 | (85) | 36 | (95) | 91 | (81) |
| Yes | 100 | (15) | 2 | (5) | 23 | (19) |
| Hypertensive Disorders* | ||||||
| No | 551 | (90) | 21 | (54) | 105 | (92) |
| Yes | 59 | (10) | 17 | (45) | 9 | (8) |
| Depressive Symptoms during Pregnancy | ||||||
| CES-D < 24 | 511 | (87) | 36 | (95) | 94 | (82) |
| CES-D ≥ 24 | 97 | (13) | 2 | (5) | 20 | (18) |
| Serious Mental Illness Symptoms: POUCHChild Survey | ||||||
| K6 < 19 | 545 | (90) | 33 | (87) | 103 | (90) |
| K6 ≥ 19 | 65 | (10) | 5 | (13) | 11 | (10) |
| Child Characteristics | ||||||
| Small for Gestational Age* | ||||||
| No | 552 | (92) | 29 | (77) | 108 | (94) |
| Yes | 58 | (8) | 9 | (23) | 6 | (6) |
| Child Sex | ||||||
| Male | 305 | (47) | 16 | (42) | 65 | (55) |
| Female | 305 | (53) | 22 | (58) | 49 | (45) |
| Child Age at Survey | ||||||
| 3 to < 6 years | 388 | (60) | 22 | (57) | 74 | (64) |
| 6 to 9 years | 222 | (40) | 16 | (43) | 40 | (36) |
| Placental Characteristics | ||||||
| High MV-O or MV-D | N = 510 | N = 36 | N = 103 | |||
| No | 407 | (81) | 25 | (70) | 83 | (80) |
| Yes | 103 | (19) | 11 | (30) | 20 | (20) |
Abbreviations: CES-D (Center for Epidemiologic Studies Depression Scale); K6 (Kessler Screening Instrument for Serious Mental Illnesses); MV-D (Maternal Vascular Pathology: Developmental); MV-O (Maternal Vascular Pathology: Obstructive)
Note. Missing data: Depressive Symptoms during Pregnancy (N=2)
Analytic sample weighted to reflect the POUCH Subcohort
p < 0.05 relative to full-term group
In both the unadjusted and adjusted analyses, LPB collapsed across delivery circumstance was not associated with any aspect of parent-reported attention problems relative to FTB (unadjusted mean differences: oppositionality: 1.1 [95%CI −1.3,3.5]; inattention: 0.5 [95% CI −1.5,2.5]; hyperactivity: 0.7 [95%CI −1.5,2.9]; ADHD Index: −.10 [95%CI −0.3,0.1]) (Table 3).
When grouped by delivery circumstance, MI LPB was associated with higher levels of hyperactivity and tended to be associated with higher ADHD Index scores relative to FTB in unadjusted analyses (mean differences: 3.9 [95%CI .5,7.3] and 3.0 [95%CI −0.3,6.3], respectively) (Table 3). These findings were replicated following adjustment for potential confounders, except that the association between MI LPB and the ADHD Index exceeded significance thresholds (mean differences: 3.8 [95%CI 0.6,7.2] and 3.1 [95%CI 0.0,6.1]); findings corresponded to effect sizes (Cohen’s d) between .3 and .4, a range often interpreted as moderate in magnitude.34 We found no evidence that associations were modified by child sex or maternal symptoms of SMIs at the time of the child survey. Findings were similarly unaffected when birth weight z-scores, maternal pre-pregnancy body mass index, or multiparity between POUCH Study enrollment and POUCHChild Survey participation were included in the multivariate models (data not shown). With the exception of the inattention subscale, MI LPB was not associated with any CPRS dimension among the preschool-aged children. However, MI LPB was significantly associated with the inattention, hyperactivity, and the ADHD Index among the 6–9 year olds (Table 4); the size of these effects was considerable, with d’s ranging from .5–.6. No outliers were detected in these age-stratified analyses.
Table 4.
Adjusted associations between pregnancy outcome and parent-reported attention problems stratified by child age (3 to <6 years: N = 484; 6–9 years: N = 278) a,b
| Full Term (referent) | Late-Preterm | Late-Preterm Circumstance
|
||||||
|---|---|---|---|---|---|---|---|---|
| Medically Indicated | Spontaneous | |||||||
| 3 to < 6 years: N = 388 6 – 9 years: N = 222 |
3 to < 6 years: N = 96 6 – 9 years: N = 56 |
3 to < 6 years: N = 22 6 – 9 years: N = 16 |
3 to < 6 years: N = 74 6 – 9 years: N = 40 |
|||||
| Mean | [95%CI] | Mean | [95%CI] | Mean | [95%CI] | Mean | [95%CI] | |
| Oppositionality | ||||||||
| 3 to < 6 years | 57.3 | [54.7, 59.9] | 57.4 | [54.7, 59.9] | 57.2 | [52.2, 62.7] | 57.5 | [53.9, 61.4] |
| 6 to 9 years | 58.1 | [53.8, 62.7] | 60.4 | [55.1, 66.3] | 64.8 | [55.4, 75.8] | 58.8 | [53.6, 64.5] |
| Inattention | ||||||||
| 3 to < 6 years | 56.8 | [54.4, 59.3] | 57.1 | [54.2, 60.1] | 53.2* | [50.1, 56.5] | 58.3 | [55.0, 66.8] |
| 6 to 9 years | 55.4 | [51.3, 59.8] | 57.5 | [52.6, 63.0] | 63.4* | [55.9, 71.9] | 55.4 | [50.2, 61.1] |
| Hyperactivity | ||||||||
| 3 to < 6 years | 62.5 | [59.7, 65.3] | 62.6 | [59.3, 65.9] | 63.8 | [59.1, 68.6] | 62.1 | [58.6, 65.8] |
| 6 to 9 years | 62.9 | [58.4, 67.4] | 64.4 | [59.3, 69.5] | 70.3* | [63.8, 76.8] | 62.2 | [56.7, 67.4] |
| ADHD Index | ||||||||
| 3 to < 6 years | 58.9 | [56.7, 61.1] | 58.3 | [55.5, 61.1] | 58.3 | [54.4, 61.2] | 58.8 | [55.2, 61.4] |
| 6 to 9 years | 60.2 | [56.3, 64.1] | 61.8 | [57.2, 66.4] | 68.1* | [62.2, 74.0] | 59.4 | [54.5, 64.3] |
Weighting applied so that the analytic sample reflects the POUCH Subcohort.
Adjusted for: maternal race, education, marital status, parity, maternal age, tobacco use, and depressive symptoms during pregnancy, symptoms of serious mental illness at time of child survey, and child sex.
p < 0.05 relative to full-term group
Because hypertensive conditions have been linked to MI LPB,27,31 analyses were repeated excluding women with clinical diagnoses of hypertensive disorders (N=85) or placental findings linked to these conditions (N=134), and were performed on the entire analytic sample to maximize cell sizes (see Methods). In each of the analyses (Table 5), the unadjusted and adjusted associations of MI LPB with hyperactivity and ADHD Index scores were no longer significant; effect sizes also decreased (d=.06–.25). Removal of SGAs reduced associations between MI LPB and ADHD Index scores to non-significance and attenuated the associations between MI LPB and hyperactivity to trend levels (unadjusted mean difference: 3.6 [95%CI −0.3,7.3]). In contrast, associations of MI LPB with hyperactivity and ADHD Index scores were maintained when women exhibiting severe levels of histologic chorioamnionitis35 (HCA) (N=63) or placental abruption36 (N=16) were removed from analysis (data not shown).
Table 5.
Unadjusted and adjusted associations between pregnancy outcome and attention problems following the removal of women with hypertensive disorders, high placental MV-O or MV-D, or small for gestational age births a,b
| Full Term (referent) N = 610 |
Late-Preterm Circumstance (N=152)
|
|||||
|---|---|---|---|---|---|---|
| Medically Indicated N = 38 |
Spontaneous N = 114 |
|||||
| Mean | [95%CI] | Mean | [95%CI] | Mean | [95%CI] | |
| No Hypertensive Disorders | N = 551 | N = 21 | N = 105 | |||
| Oppositionality | ||||||
| unadjusted | 51.6 | [50.5, 52.7] | 49.5 | [46.5, 52.7] | 52.3 | [50.1, 54.5] |
| adjusted | 57.2 | [54.8, 59.6] | 54.7 | [50.5, 59.2] | 57.5 | [54.5, 60.8] |
| Inattention | ||||||
| unadjusted | 51.6 | [50.7, 52.6] | 51.5 | [47.3, 55.9] | 52.1 | [50.1, 54.1] |
| adjusted | 56.2 | [54.0, 58.5] | 55.8 | [50.7, 61.4] | 56.8 | [53.9, 59.8] |
| Hyperactivity | ||||||
| unadjusted | 55.1 | [53.9, 56.2] | 56.0 | [51.8, 60.2] | 54.6 | [52.4, 56.9] |
| adjusted | 62.4 | [59.8, 64.9] | 62.7 | [58.2, 67.3] | 61.5 | [58.4, 64.6] |
| ADHD Index | ||||||
| unadjusted | 53.2 | [52.3, 54.2] | 53.6 | [48.9, 58.3] | 52.5 | [50.5, 54.5] |
| adjusted | 59.3 | [57.2, 61.4] | 59.5 | [54.4, 64.5] | 58.4 | [55.7, 61.1] |
| No SGAs | N = 552 | N = 29 | N = 108 | |||
| Oppositionality | ||||||
| unadjusted | 51.4 | [50.4, 52.5] | 53.6 | [49.6, 57.9] | 51.8 | [49.8, 54.0] |
| adjusted | 57.3 | [54.9, 59.7] | 60.1 | [54.9, 65.8] | 56.7 | [53.7, 59.8] |
| Inattention | ||||||
| unadjusted | 51.5 | [50.6, 52.4] | 51.9 | [48.8, 55.3] | 51.4 | [49.6, 53.3] |
| adjusted | 55.3 | [53.0, 57.6] | 55.5 | [51.4, 59.9] | 55.2 | [52.5, 58.0] |
| Hyperactivity | ||||||
| unadjusted | 54.7 | [53.6, 55.9 | 58.3 | [54.6, 61.9] | 54.0 | [51.9, 56.1] |
| adjusted | 61.6 | [58.9, 64.3 | 64.8 | [60.5, 69.2] | 60.1 | [57.1, 63.1] |
| ADHD Index | ||||||
| unadjusted | 53.0 | [52.1, 54.0] | 55.7 | [52.1, 59.4] | 51.6 | [49.8, 53.5] |
| adjusted | 58.6 | [56.3, 60.8] | 61.1 | [57.1, 65.2] | 56.8 | [54.2, 59.5] |
| No High MV-O or MV-D | N = 407 | N = 25 | N = 83 | |||
| Oppositionality | ||||||
| unadjusted | 51.3 | [50.1, 52.5] | 53.5 | [48.7, 58.7] | 53.2 | [50.5, 56.0] |
| adjusted | 57.4 | [54.4, 60.5] | 60.1 | [53.7, 67.2] | 58.7 | [54.9, 62.8] |
| Inattention | ||||||
| unadjusted | 52.3 | [51.2, 53.4] | 51.4 | [48.4, 54.6] | 53.4 | [51.0, 55.9] |
| adjusted | 57.6 | [54.9, 60.5] | 56.3 | [52.2, 60.8] | 58.7 | [55.1, 62.5] |
| Hyperactivity | ||||||
| unadjusted | 54.7 | [53.5, 56.0] | 58.1 | [53.7, 62.6] | 55.4 | [53.0, 57.8] |
| adjusted | 63.0 | [60.1, 65.9] | 65.5 | [60.3, 70.7] | 63.4 | [59.9, 66.8] |
| ADHD Index | ||||||
| unadjusted | 53.6 | [52.5, 54.7] | 54.5 | [50.4, 58.6] | 53.5 | [51.0, 55.9] |
| adjusted | 61.0 | [58.6, 63.5] | 61.6 | [57.2, 66.1] | 60.7 | [57.4, 64.1] |
Abbreviations: MV-D (Maternal Vascular Pathology: Developmental); MV-O (Maternal Vascular Pathology: Obstructive); SGA (small for gestational age)
Weighting applied so that the analytic sample reflects the POUCH Subcohort.
Adjusted for: maternal race, education, marital status, parity, maternal smoking and depressive symptoms during pregnancy, symptoms of serious mental illness at time of child survey, child sex, and child age at survey
All mean differences were non-significant.
Discussion
On average, children born late-preterm did not exhibit higher levels of parent-reported attention problems compared to those born full-term. However, when LPB was grouped by delivery circumstance, MI LPB was associated with higher levels of hyperactivity and higher scores on a composite marking inattentive and hyperactivity symptoms. Following removal of women with hypertensive disorders or placental findings associated with hypertensive conditions, these findings were attenuated and no longer significant, suggesting that hypertensive disorders may play a role in the associations.
Delivery during the late-preterm period has received considerable epidemiologic attention. To date, conditions linked to LPB include infections (e.g., HCA)33 as well as hypertensive disorders, fetal growth restriction, and placental abruption;37–39 this latter group of conditions has been linked to deliveries prompted by medical intervention.37,40 Despite the range of factors associated with LPB, we provide evidence suggesting that diagnoses of hypertensive conditions as well as placental findings implicated in these conditions contribute to the associations reported here. Indeed, removal of women with placental abruption or who exhibit severe HCA did not affect any of our findings. Although removal of SGA births did slightly attenuate links between MI LPB and hyperactivity symptoms, this group likely includes children who are constitutionally small and may not represent the most sensitive approach to identifying fetal growth restriction. Hypertensive conditions during pregnancy, as well as the placental vascular findings investigated here, may mark disturbances in placental perfusion which in turn impede gas, waste, and nutrient exchange between mother and fetus;29 links between hypertensive disorders and alterations in placental hormone secretion have also been observed (e.g., corticotropin-releasing hormone).41–43 The extent to which such biological alterations affect brain development, particularly circuits implicated in the development of attention problems (e.g., frontal-striatal and cerebellar networks), require further elucidation.44–47
Interestingly, studies investigating links between hypertensive conditions and the development of attention problems have generally yielded null results.48–50 There may be several factors contributing to this apparent contradiction to our findings, including a relative lack of socio-economic and racial/ethnic diversity in the study samples,49 and more importantly, grouping preterms and terms together when evaluating these associations.50 To the extent that the threshold motivating medical intervention changes across gestational age, such approaches may obscure associations that are due to symptom severity and may be revealed by testing for effect modification by gestational age at delivery. Many studies also employ composite indices of perinatal risk that represent a range of obstetric and neonatal complications,49,51–54 thus obscuring associations between any given condition and the outcome of interest.
Nonetheless, our study findings may be relevant to outstanding issues in the LPB outcome literature. Although delivery prior to 34 weeks is more strongly associated with a range of developmental outcomes, including long-term behavioral problems,55,56 we provide evidence suggesting that links between MI LPB and attention problems may be clinically-meaningful (0.3<d<0.4). Additionally, previous investigations suggest that only a subset of children born late-preterm appear to be at-risk for later attention problems.7,15 Our study extends these findings by identifying maternal hypertensive disorders as one potential source/marker of such heterogeneity.
In contrast to other investigations, we did not observe an association between LPB and attention problems when collapsing across delivery circumstance. There may be several reasons for this. First, the majority of children in our study were younger than those in studies documenting associations between LPB and attention-related problems; to the extent that main effects of LPB are more readily observed at older ages, especially with respect to attention problems (next paragraph), this might have interfered with our ability to detect such differences across the LPB subgroups. Additionally, other studies documenting associations between LPB and attention problems might reflect the characteristics of the samples from which they were drawn. For example, links between LPB and attention problems may be more readily apparent in samples enriched with children who were growth restricted at birth or experienced neonatal complications.7,15 Providing some support for this hypothesis, analyses utilizing NICHD Early Child Care data found no differences between LPB and FTB in terms of behavioral problems,16 but children from complicated pregnancies were excluded from study.
There are several caveats to consider when interpreting our findings. First, although we employed a norm-referenced measure of attention problems, the main outcome of this study was parent-reported. We therefore cannot rule out that maternal knowledge of children’s PTB status influenced their reports of attention problems. However, because our findings were limited to MI LPB, it is unlikely this issue explains the study findings. Additionally, longitudinal investigations of parenting quality suggest that while there may be alterations in maternal behavior during the first year of life as a function of PTB status, such differences are not observed in toddlerhood when measured using the Strange Situation, a lab-based observation considered to be the gold standard for characterizing parent-child relationship quality at that age.57–59 Second, measures of socio-economic and demographic factors used in this report were obtained at POUCH Study enrollment. Changes in family structure and/or income over time may contribute to individual differences in behavioral problems in children,60–63 and we were unable to investigate such effects or otherwise contextualize findings with contemporaneous socio-economic indicators. Third, although our analyses were drawn from a community sample, some were limited in power due to the relatively small number of MI LPBs. This is particularly true with respect to the age-stratified analyses as well as those involving the removal of subgroups based upon placental or medical record information. Given these concerns, we chose not to adjust for multiple comparisons. While we believe the consistency of findings across analyses helps assuage concerns regarding Type I error rate, replication of our findings is warranted and encouraged. Our analyses also assumed that non-missing data from a particular weighted sampling stratum were representative of that stratum. To address this assumption, we performed unweighted analyses and observed no alterations to the pattern of findings reported here. Caution should nonetheless be employed when considering the generalizability of the study findings, particularly given the follow-up rate for eligible LPBs (54%). Combined with evidence suggesting that POUCHChild Survey participation was associated with higher socio-economic status indicators (see Results), the current analysis may not reflect the full-spectrum of LPBs. Finally, it is important to note that MI LPB is a risk factor for subsequent neonatal morbidity,64 and as such, this study is unable to identify whether prenatal, postnatal, or some combination of these factors contribute to the outcomes observed here.
Despite these caveats, there are notable strengths of this investigation, including the prospective design and demographic breadth from which the analytic sample was drawn. Additionally, the availability of detailed medical record information and placental findings allowed for an investigation of factors that may underlie the associations reported here. Information regarding maternal psychosocial functioning during pregnancy and at child survey completion helped address concerns that co-occurrences between maternal and child psychopathology contributed to the study findings.65 Because there is no indication that women with attention problems are more likely to exhibit MI LPBs, we do not believe that other factors contributing to such co-occurrence fully account for the findings here (e.g., heritability).
This study provides many directions for future investigation. In addition to replicating the current findings, the extent to which associations between hypertension-associated LPB and attention problems persist across development requires elucidation. Additionally, LPB has been associated with other types of behavioral problems in childhood besides attention problems (e.g., anxious/depressive symptoms),7,13 but at present, we have very little understanding of what factors contribute to the different types of outcomes observed among these children. This latter issue underscores the need to better understand the mechanisms involved in associations reported here and elsewhere.
In sum, we provide evidence suggesting that MI LPB is associated with higher levels of attention problems in childhood. Given that previous investigations point to marked heterogeneity in outcomes of children born late-preterm, studies such as ours may help identify children at highest risk for later difficulties while at the same time, uncover the mechanisms involved.
Acknowledgments
This study was supported by the Perinatal Epidemiological Research Initiative Program [Grant 20-FY04-37] from the March of Dimes Foundation, the National Institute of Child Health and Human Development and the National Institute of Nursing Research [Grant R01 HD34543], the Thrasher Research Foundation [Grant 02816-7], and the Centers for Disease Control and Prevention [Grant U01 DP000143-01] to Claudia Holzman. The principal investigator of the POUCH Study (Claudia Holzman) had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis
ABBREVIATIONS
- ADHD
attention deficit hyperactivity disorder
- CES-D
Center for Epidemiologic Studies Depression Scale
- CPRS-R: S
Conners’ Parent Report-Revised: Short Form
- GLM
general linear model
- HCA
histologic chorioamnionitis
- IQ
intelligence quotient
- LPB
late preterm birth
- MI
medically indicated
- MoM
multiples of the median
- MSAFP
maternal serum alpha fetoprotein
- MV-D
maternal vascular-developmental
- MV-O
maternal vascular-obstructive
- NICHD
National Institute of Child Health and Human Development
- NICU
neonatal intensive care unit
- PE
preeclampsia
- POUCH Study
Pregnancy Outcome and Community Health Study
- PTD
preterm delivery
- SGA
small for gestational age
- SMIs
Serious Mental Illnesses
Footnotes
CONFLICT OF INTEREST: The authors have no conflicts of interest to report.
FINANCIAL DISCLOSURE: This study was supported by the Perinatal Epidemiological Research Initiative Program [Grant 20-FY04-37] from the March of Dimes Foundation, the National Institute of Child Health and Human Development and the National Institute of Nursing Research [Grant R01 HD34543], the Thrasher Research Foundation [Grant 02816-7], and the Centers for Disease Control and Prevention [Grant U01 DP000143-01] to Claudia Holzman.
References
- 1.Martin JA, Kirmeyer S, Osterman M, Shepherd RA. Born a bit too early: recent trends in late preterm births. NCHS Data Brief. 2009 Nov;(24):1–8. [PubMed] [Google Scholar]
- 2.Martin JA, Hamilton BE, Sutton PD, Ventura SJ, Menacker F, Munson ML. Births: final data for 2003. Natl Vital Stat Rep. 2005 Sep 8;54(2):1–116. [PubMed] [Google Scholar]
- 3.Davidoff MJ, Dias T, Damus K, et al. Changes in the gestational age distribution among U.S. singleton births: impact on rates of late preterm birth, 1992 to 2002. Semin Perinatol. 2006 Feb;30(1):8–15. doi: 10.1053/j.semperi.2006.01.009. [DOI] [PubMed] [Google Scholar]
- 4.Kramer MS. Late preterm birth: appreciable risks, rising incidence. J Pediatr. 2009 Feb;154(2):159–160. doi: 10.1016/j.jpeds.2008.09.048. [DOI] [PubMed] [Google Scholar]
- 5.Shapiro-Mendoza CK, Tomashek KM, Kotelchuck M, et al. Effect of late-preterm birth and maternal medical conditions on newborn morbidity risk. Pediatrics. 2008 Feb;121(2):e223–232. doi: 10.1542/peds.2006-3629. [DOI] [PubMed] [Google Scholar]
- 6.Tomashek KM, Shapiro-Mendoza CK, Davidoff MJ, Petrini JR. Differences in mortality between late-preterm and term singleton infants in the United States, 1995–2002. J Pediatr. 2007 Nov;151(5):450–456. 456, e451. doi: 10.1016/j.jpeds.2007.05.002. [DOI] [PubMed] [Google Scholar]
- 7.Talge NM, Holzman C, Wang J, Lucia V, Gardiner J, Breslau N. Late-preterm birth and its association with cognitive and socioemotional outcomes at 6 years of age. Pediatrics. 2010 Dec;126(6):1124–1131. doi: 10.1542/peds.2010-1536. [DOI] [PubMed] [Google Scholar]
- 8.Nomura Y, Rajendran K, Brooks-Gunn J, Newcorn JH. Roles of perinatal problems on adolescent antisocial behaviors among children born after 33 completed weeks: a prospective investigation. J Child Psychol Psychiatry. 2008 Oct;49(10):1108–1117. doi: 10.1111/j.1469-7610.2008.01939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nomura Y, Halperin JM, Newcorn JH, et al. The risk for impaired learning-related abilities in childhood and educational attainment among adults born near-term. J Pediatr Psychol. 2009 May;34(4):406–418. doi: 10.1093/jpepsy/jsn092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Petrini JR, Dias T, McCormick MC, Massolo ML, Green NS, Escobar GJ. Increased risk of adverse neurological development for late preterm infants. J Pediatr. 2009 Feb;154(2):169–176. doi: 10.1016/j.jpeds.2008.08.020. [DOI] [PubMed] [Google Scholar]
- 11.Linnet KM, Wisborg K, Agerbo E, Secher NJ, Thomsen PH, Henriksen TB. Gestational age, birth weight, and the risk of hyperkinetic disorder. Arch Dis Child. 2006 Aug;91(8):655–660. doi: 10.1136/adc.2005.088872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Morse SB, Zheng H, Tang Y, Roth J. Early school-age outcomes of late preterm infants. Pediatrics. 2009 Apr;123(4):e622–629. doi: 10.1542/peds.2008-1405. [DOI] [PubMed] [Google Scholar]
- 13.Lindstrom K, Lindblad F, Hjern A. Psychiatric morbidity in adolescents and young adults born preterm: a Swedish national cohort study. Pediatrics. 2009 Jan;123(1):e47–53. doi: 10.1542/peds.2008-1654. [DOI] [PubMed] [Google Scholar]
- 14.Moster D, Lie RT, Markestad T. Long-term medical and social consequences of preterm birth. N Engl J Med. 2008 Jul 17;359(3):262–273. doi: 10.1056/NEJMoa0706475. [DOI] [PubMed] [Google Scholar]
- 15.Baron IS, Erickson K, Ahronovich MD, Baker R, Litman FR. Cognitive deficit in preschoolers born late-preterm. Early Hum Dev. 2011 Feb;87(2):115–119. doi: 10.1016/j.earlhumdev.2010.11.010. [DOI] [PubMed] [Google Scholar]
- 16.Gurka MJ, LoCasale-Crouch J, Blackman JA. Long-term cognition, achievement, socioemotional, and behavioral development of healthy late-preterm infants. Arch Pediatr Adolesc Med. 2010 Jun;164(6):525–532. doi: 10.1001/archpediatrics.2010.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Berkowitz GS, Blackmore-Prince C, Lapinski RH, Savitz DA. Risk factors for preterm birth subtypes. Epidemiology. 1998 May;9(3):279–285. [PubMed] [Google Scholar]
- 18.Holzman C, Bullen B, Fisher R, Paneth N, Reuss L. Pregnancy outcomes and community health: The POUCH study of preterm delivery. Paediatr Perinat Ep. 2001;15:136–158. doi: 10.1046/j.1365-3016.2001.00014.x. [DOI] [PubMed] [Google Scholar]
- 19.Alexander GR, Himes JH, Kaufman RB, Mor J, Kogan M. A United States national reference for fetal growth. Obstet Gynecol. 1996;87:163–168. doi: 10.1016/0029-7844(95)00386-X. [DOI] [PubMed] [Google Scholar]
- 20.Conners CK. Conners’ rating scales-revised (CRS-R) technical manual. Tonawanda, N.Y: Multihealth Systems; 2001. [Google Scholar]
- 21.Radloff L. The CES-D Scale: A self-report depression scale for research in the general population. Applied Psychological Measurement. 1977;1(3):385–401. [Google Scholar]
- 22.Gavin AR, Holzman C, Siefert K, Tian Y. Maternal depressive symptoms, depression, and psychiatric medication use in relation to risk of preterm delivery. Womens Health Issues. 2009 Sep-Oct;19(5):325–334. doi: 10.1016/j.whi.2009.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kessler RC, Andrews G, Colpe LJ, et al. Short Screening Scales to Monitor Population Prevalences and Trends in Non-specific Psychological Distress. Psychol Med. 2002;32:959–976. doi: 10.1017/s0033291702006074. [DOI] [PubMed] [Google Scholar]
- 24.Levine RJ, Hauth JC, Curet LB, et al. Trial of calcium to prevent pre-eclampsia. New Eng J Med. 1997;337(2):69–77. doi: 10.1056/NEJM199707103370201. [DOI] [PubMed] [Google Scholar]
- 25.Zambrowski NHBPEP report on high blood pressure in pregnancy: A summary for family physicians. Am Fam Physician. 2001;64(2):264. [PubMed] [Google Scholar]
- 26.Report of the National High Blood Pressure Education Program Working Group on high blood pressure in pregnancy. Am J Obstet Gynecol. 2000;183(1):S1–S22. [PubMed] [Google Scholar]
- 27.Kelly R, Holzman C, Senagore P, et al. Placental vascular pathology findings and pathways to preterm delivery. Am J Epidemiol. 2009 Jul 15;170(2):148–158. doi: 10.1093/aje/kwp131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Salafia CM, Ernst LM, Pezzullo JC, Wolf EJ, Rosenkrantz TS, Vintzileos AM. The very low birthweight infant: maternal complications leading to preterm birth, placental lesions, and intrauterine growth. Am J Perinatol. 1995 Mar;12(2):106–110. doi: 10.1055/s-2007-994417. [DOI] [PubMed] [Google Scholar]
- 29.Baergen RN. Manual of Benirschke and Kaufmann’s Pathology of the Human Placenta. New York: Springer; 2005. [Google Scholar]
- 30.Wolraich M, Brown L, Brown RT, et al. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2011 Nov;128(5):1007–1022. doi: 10.1542/peds.2011-2654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Carreno CA, Costantine MM, Holland MG, Ramin SM, Saade GR, Blackwell SC. Approximately one-third of medically indicated late preterm births are complicated by fetal growth restriction. Am J Obstet Gynecol. Jan 12; doi: 10.1016/j.ajog.2010.12.004. in press. [DOI] [PubMed] [Google Scholar]
- 32.McIntire DD, Leveno KJ. Neonatal mortality and morbidity rates in late preterm births compared with births at term. Obstet Gynecol. 2008 Jan;111(1):35–41. doi: 10.1097/01.AOG.0000297311.33046.73. [DOI] [PubMed] [Google Scholar]
- 33.Khashu M, Narayanan M, Bhargava S, Osiovich H. Perinatal outcomes associated with preterm birth at 33 to 36 weeks’ gestation: a population-based cohort study. Pediatrics. 2009 Jan;123(1):109–113. doi: 10.1542/peds.2007-3743. [DOI] [PubMed] [Google Scholar]
- 34.Cohen J. Statistical power analysis for the behavioral sciences. 2. Hillsdale Lawrence Erlbaum; 1988. [Google Scholar]
- 35.Holzman C, Lin X, Senagore P, Chung H. Histologic chorioamnionitis and preterm delivery. Am J Epidemiol. 2007 Oct 1;166(7):786–794. doi: 10.1093/aje/kwm168. [DOI] [PubMed] [Google Scholar]
- 36.Gargano JW, Holzman CB, Senagore PK, et al. Polymorphisms in thrombophilia and renin-angiotensin system pathways, preterm delivery, and evidence of placental hemorrhage. Am J Obstet Gynecol. 2009 Sep;201(3):317, e311–319. doi: 10.1016/j.ajog.2009.05.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fuchs K, Gyamfi C. The influence of obstetric practices on late prematurity. Clin Perinatol. 2008 Jun;35(2):343–360. vi. doi: 10.1016/j.clp.2008.03.004. [DOI] [PubMed] [Google Scholar]
- 38.Dobak WJ, Gardner MO. Late preterm gestation: physiology of labor and implications for delivery. Clin Perinatol. 2006 Dec;33(4):765–776. abstract vii. doi: 10.1016/j.clp.2006.09.001. [DOI] [PubMed] [Google Scholar]
- 39.Sibai BM. Preeclampsia as a cause of preterm and late preterm (near-term) births. Semin Perinatol. 2006 Feb;30(1):16–19. doi: 10.1053/j.semperi.2006.01.008. [DOI] [PubMed] [Google Scholar]
- 40.Holland MG, Refuerzo JS, Ramin SM, Saade GR, Blackwell SC. Late preterm birth: how often is it avoidable? Am J Obstet Gynecol. 2009 Oct;201(4):404, e401–404. doi: 10.1016/j.ajog.2009.06.066. [DOI] [PubMed] [Google Scholar]
- 41.Goland RS, Conwell IM, Jozak S. The effect of pre-eclampsia on human placental corticotrophin-releasing hormone content and processing. Placenta. 1995 Jun;16(4):375–382. doi: 10.1016/0143-4004(95)90095-0. [DOI] [PubMed] [Google Scholar]
- 42.Ahmed I, Glynn BP, Perkins AV, et al. Processing of procorticotropin-releasing hormone (pro-CRH): molecular forms of CRH in normal and preeclamptic pregnancy. J Clin Endocrinol Metab. 2000 Feb;85(2):755–764. doi: 10.1210/jcem.85.2.6351. [DOI] [PubMed] [Google Scholar]
- 43.Liapi CA, Tsakalia DE, Panitsa-Faflia CC, Antsaklis AI, Aravantinos DI, Batrinos ML. Corticotropin-releasing-hormone levels in pregnancy-induced hypertension. Eur J Obstet Gynecol Reprod Biol. 1996 Sep;68(1–2):109–114. doi: 10.1016/0301-2115(96)02508-0. [DOI] [PubMed] [Google Scholar]
- 44.Konrad K, Eickhoff SB. Is the ADHD brain wired differently? A review on structural and functional connectivity in attention deficit hyperactivity disorder. Hum Brain Mapp. 2010 Jun;31(6):904–916. doi: 10.1002/hbm.21058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tian L, Jiang T, Wang Y, et al. Altered resting-state functional connectivity patterns of anterior cingulate cortex in adolescents with attention deficit hyperactivity disorder. Neurosci Lett. 2006 May 29;400(1–2):39–43. doi: 10.1016/j.neulet.2006.02.022. [DOI] [PubMed] [Google Scholar]
- 46.Castellanos FX, Margulies DS, Kelly C, et al. Cingulate-precuneus interactions: a new locus of dysfunction in adult attention-deficit/hyperactivity disorder. Biol Psychiatry. 2008 Feb 1;63(3):332–337. doi: 10.1016/j.biopsych.2007.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rubia K, Halari R, Cubillo A, Mohammad AM, Brammer M, Taylor E. Methylphenidate normalises activation and functional connectivity deficits in attention and motivation networks in medication-naive children with ADHD during a rewarded continuous performance task. Neuropharmacology. 2009 Dec;57(7–8):640–652. doi: 10.1016/j.neuropharm.2009.08.013. [DOI] [PubMed] [Google Scholar]
- 48.Nigg JT, Nikolas MB, SA Measured gene-by-environment interaction in relation to attention deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2010;49:863–873. doi: 10.1016/j.jaac.2010.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wagner AI, Schmidt NL, Lemery-Chalfant K, Leavitt LA, Goldsmith HH. The limited effects of obstetrical and neonatal complications on conduct and attention-deficit hyperactivity disorder symptoms in middle childhood. J Dev Behav Pediatr. 2009 Jun;30(3):217–225. doi: 10.1097/DBP.0b013e3181a7ee98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gustafsson P, Kallen K. Perinatal, maternal, and fetal characteristics of children diagnosed with attention-deficit-hyperactivity disorder: results from a population-based study utilizing the Swedish Medical Birth Register. Dev Med Child Neurol. 2010 Mar;53(3):263–268. doi: 10.1111/j.1469-8749.2010.03820.x. [DOI] [PubMed] [Google Scholar]
- 51.Curtis WJ, Lindeke LL, Georgieff MK, Nelson CA. Neurobehavioural functioning in neonatal intensive care unit graduates in late childhood and early adolescence. Brain. 2002 Jul;125(Pt 7):1646–1659. doi: 10.1093/brain/awf159. [DOI] [PubMed] [Google Scholar]
- 52.Luciana M, Lindeke L, Georgieff M, Mills M, Nelson CA. Neurobehavioral evidence for working-memory deficits in school-aged children with histories of prematurity. Dev Med Child Neurol. 1999 Aug;41(8):521–533. doi: 10.1017/s0012162299001140. [DOI] [PubMed] [Google Scholar]
- 53.Ben Amor L, Grizenko N, Schwartz G, et al. Perinatal complications in children with attention-deficit hyperactivity disorder and their unaffected siblings. J Psychiatry Neurosci. 2005 Mar;30(2):120–126. [PMC free article] [PubMed] [Google Scholar]
- 54.Allen NB, Lewinsohn PM, Seeley JR. Prenatal and perinatal influences on risk for psychopathology in childhood and adolescence. Dev Psychopathol. 1998 Summer;10(3):513–529. doi: 10.1017/s0954579498001722. [DOI] [PubMed] [Google Scholar]
- 55.Lindstrom K, Lindblad F, Hjern A. Preterm birth and attention-deficit/hyperactivity disorder in schoolchildren. Pediatrics. 2011 May;127(5):858–865. doi: 10.1542/peds.2010-1279. [DOI] [PubMed] [Google Scholar]
- 56.Schothorst PF, van Engeland H. Long-term behavioral sequelae of prematurity. J Am Acad Child Adolesc Psychiatry. 1996 Feb;35(2):175–183. doi: 10.1097/00004583-199602000-00011. [DOI] [PubMed] [Google Scholar]
- 57.Frodi A, Thompson R. Infants’ affective responses in the strange situation: effects of prematurity and of quality of attachment. Child Dev. 1985 Oct;56(5):1280–1290. doi: 10.1111/j.1467-8624.1985.tb00196.x. [DOI] [PubMed] [Google Scholar]
- 58.Miles MS, Holditch-Davis D. Parenting the prematurely born child: pathways of influence. Semin Perinatol. 1997 Jun;21(3):254–266. doi: 10.1016/s0146-0005(97)80067-5. [DOI] [PubMed] [Google Scholar]
- 59.Easterbrooks MA. Quality of attachment to mother and to father: Effects of perinatal risk status. Child Dev. 1989;60:825–830. [Google Scholar]
- 60.Weinfield NS, Whaley GJ, Egeland B. Continuity, discontinuity, and coherence in attachment from infancy to late adolescence: sequelae of organization and disorganization. Attach Hum Dev. 2004 Mar;6(1):73–97. doi: 10.1080/14616730310001659566. [DOI] [PubMed] [Google Scholar]
- 61.Weinfield NS, Sroufe LA, Egeland B. Attachment from infancy to early adulthood in a high-risk sample: continuity, discontinuity, and their correlates. Child Dev. 2000 May-Jun;71(3):695–702. doi: 10.1111/1467-8624.00178. [DOI] [PubMed] [Google Scholar]
- 62.Sroufe LA. Attachment and development: a prospective, longitudinal study from birth to adulthood. Attach Hum Dev. 2005 Dec;7(4):349–367. doi: 10.1080/14616730500365928. [DOI] [PubMed] [Google Scholar]
- 63.Urban J, Carlson E, Egeland B, Sroufe LA. Patterns of individual adaptation across childhood. Dev Psychopathol. 1991;3:445–460. [Google Scholar]
- 64.Gouyon JB, Vintejoux A, Sagot P, Burguet A, Quantin C, Ferdynus C. Neonatal outcome associated with singleton birth at 34–41 weeks of gestation. Int J Epidemiol. 2010 Jun;39(3):769–776. doi: 10.1093/ije/dyq037. [DOI] [PubMed] [Google Scholar]
- 65.Chilcoat HD, Breslau N. Does psychiatric history bias mothers’ reports? An application of a new analytic approach. J Am Acad Child Adolesc Psychiatry. 1997 Jul;36(7):971–979. doi: 10.1097/00004583-199707000-00020. [DOI] [PubMed] [Google Scholar]