Abstract
Expression of CCL2 (CC chemokine ligand 2) (or monocyte chemoattractant protein-1) regulates inflammatory cell infiltration in the liver and adipose tissue, favouring steatosis. However, its role in the pathogenesis of steatohepatitis is still uncertain. In the present study, we investigated the development of non-alcoholic steatohepatitis induced by an MCD diet (methionine/choline-deficient diet) in mice lacking the CCL2 gene on two different genetic backgrounds, namely Balb/C and C57/Bl6J. WT (wild-type) and CCL2-KO (knockout) mice were fed on a lipid-enriched MCD diet or a control diet for 8 weeks. In Balb/C mice fed on the MCD diet, a lack of CCL2 was associated with lower ALT (alanine transaminase) levels and reduced infiltration of inflammatory cells, together with a lower generation of oxidative-stress-related products. Sirius Red staining demonstrated pericellular fibrosis in zone 3, and image analysis showed a significantly lower matrix accumulation in CCL2-KO mice. This was associated with reduced hepatic expression of TGF-β (transforming growth factor-β), type I procollagen, TIMP-1 (tissue inhibitor of metalloproteinases-1) and α-smooth muscle actin. In contrast, in mice on a C57Bl/6 background, neither ALT levels nor inflammation or fibrosis were significantly different comparing WT and CCL2-KO animals fed on an MCD diet. In agreement, genes related to fibrogenesis were expressed to comparable levels in the two groups of animals. Comparison of the expression of several genes involved in inflammation and repair demonstrated that IL (interleukin)-4 and the M2 marker MGL-1 (macrophage galactose-type C-type lectin 1) were differentially expressed in Balb/C and C57Bl/6 mice. No significant differences in the degree of steatosis were observed in all groups of mice fed on the MCD diet. We conclude that, in experimental murine steatohepatitis, the effects of CCL2 deficiency are markedly dependent on the genetic background.
Keywords: CC chemokine ligand 2, chemokine, cytokine, liver fibrosis, non-alcoholic steatohepatitis
Abbreviations: ALT, alanine transaminase; AST, aspartate aminotransferase; CCL2, CC chemokine ligand 2; CCR2, CC chemokine receptor 2; FAM, 6-carboxyfluorescein; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; HNE, 4-hydroxynonenal, IFN-γ, interferon-γ; IL, interleukin; iNOS, inducible NO synthase; KO, knockout; MCD diet, methionine/choline-deficient diet; MGL1, macrophage galactose-type C-type lectin 1; NAFLD, non-alcoholic fatty liver disease; NASH, non-alcoholic steatohepatitis; ROS, reactive oxygen species; RT, real-time; TGF-β, transforming growth factor-β; TIMP-1, tissue inhibitor of metalloproteinases-1; TNFα, tumour necrosis factor α; WT, wild-type
INTRODUCTION
NAFLD (non-alcoholic fatty liver disease) is the most common cause of chronic liver disease in the Western world [1]. A fraction of patients with NAFLD shows findings of NASH (non-alcoholic steatohepatitis), which is characterized by hepatocellular injury, inflammation and fibrosis [2]. In this case, parenchymal injury and inflammation are associated with generation of oxidative-stress-related products, which have a deleterious impact on disease progression [2]. More importantly, NASH potentially leads to fibrosis, cirrhosis and its complications.
Understanding the cellular and molecular mechanisms that are responsible for hepatic damage and fibrogenesis is of key relevance in the field of fatty liver. Several lines of evidence suggest that cytokines represent relevant mediators in this context. In particular, different pro-inflammatory cytokines are up-regulated in the liver of patients with NAFLD, and their expression correlates with parameters of inflammation and/or fibrogenesis [3]. Among cytokines, attention has been recently directed to the chemokine family of pro-inflammatory factors. Chemokines are a class of small cytokines that are involved in the regulation of leukocyte recruitment to different tissues, including the liver and orchestrate inflammatory responses [4].
CCL2 (CC chemokine ligand 2), also known as MCP-1 (monocyte chemoattractant protein-1), is an inflammatory chemokine that targets predominantly monocytes and T-lymphocytes [5]. In addition, CCL2 has been implicated in the process that leads to polarization of lymphocytes towards a Th1 or Th2 phenotype [6]. CCR2 (CC chemokine receptor 2) represents the major receptor for CCL2, although some actions of this chemokine are independent of CCR2 binding [7,8]. In the liver, increased CCL2 expression has been shown in patients with acute and chronic inflammatory diseases and in numerous rodent models of injury [9,10]. Recently, the CCL2/CCR2 axis has been suggested to be a mediator of adipose tissue inflammation during obesity, favouring hepatic steatosis [11,12]. Moreover, CCL2 expression is increased in the liver of patients with NAFLD, where it is correlated with fat accumulation [13,14]. However, the contribution of this pathway to the development of steatohepatitis is still uncertain. Recent studies by Kassel et al. [15] have shown that CCL2 deficiency in C57Bl/6 mice did not affect aminotransferase levels or inflammation in mice fed on an MCD diet (methionine/choline-deficient diet) for 4 weeks, whereas it down-modulated pro-fibrogenic gene expression. As these results were in contrast with findings in humans [14] and in other experimental models of liver injury [9,10], and fibrosis is generally modest in mice after 4 weeks on the MCD diet, we have studied CCL2-KO (knockout) mice fed on the MCD diet for 8 weeks, a time point when fibrosis is well detectable. In addition, we have conducted the experiments in mice with two different genetic backgounds. Our results indicate that a lack of CCL2 reduces inflammation, oxidative stress and fibrogenesis in Balb/C but not in C57Bl/6 mice, indicating that the effects of CCL2 deficiency are markedly dependent on the mouse genetic background.
MATERIALS AND METHODS
Reagents
Unless otherwise indicated all chemicals were purchased from Sigma.
Animals and experimental protocol
CCL2-KO mice (initial colony kindly provided by Dr Barrett Rollins, Department of Medical Oncology, Harvard University, Boston, MA, U.S.A.), backcrossed with WT (wild-type) Balb/C mice for greater than ten generations, have been described previously [16]. CCL2-KO on a C57Bl/6J genetic background (referred to as C57Bl/6) were obtained from Jackson Laboratories, and a colony established. WT Balb/C or C57Bl/6 mice were obtained from Harlan Nossan. In all experiments, 8-week-old male mice were used, and six to eight animals were included for each experimental point. All mice were fed on a lipid-enriched MCD diet or the same diet supplemented with the two amino acids (control diet) for 8 weeks, as described previously [17].
Body weight was recorded weekly. At the end of the study protocol, mice were anaesthetized and killed by exsanguination, blood being collected from the inferior vena cava. Blood was centrifuged and serum stored for further analysis. Livers were rapidly dissected out, weighed, snap-frozen in liquid nitrogen and kept at −80°C for RNA and protein extraction. A portion of the liver was immediately fixed in formalin for histological analyses, and another portion was processed for determination of oxidative-stress-related products (see below).
All animals were treated with humane care, and experimental protocols were performed according to established international guidelines (Guide for the Care and Use of Laboratory Animals, NIH publication no. 86-23) after approval by the local regulatory authorities.
Aminotransferase activity and measurement of oxidative-stress-related products
These determinations were carried out exactly as described previously [16,17]. In particular, determination of hepatic levels of ROS (reactive oxygen species) and HNE (4-hydroxynonenal) were carried out on cytosolic fractions obtained from the fresh liver tissue immediately after the animals were killed.
Liver histology
Fixation and staining of liver tissue were conducted as described previously [17]. Liver histology was evaluated by an experienced hepatopathologist blinded to the type of treatment received by the animals. Scoring of slides was performed using a semi-quantitative method, as reported previously [17]. Quantification of fibrosis was carried out by histomorphometric analysis using a digital camera (DFC 320; Leica) coupled to a bright-field microscope (DM4000; Leica), essentially as described elsewhere [17].
Immunohistochemistry for MoMa2
Intrahepatic levels of MoMa2, a specific marker for monocyte/macrophages, were determined by indirect immunofluorescence performed on liver cryostat sections (4 μm thick) as described previously [18]. Primary rat monoclonal antibodies (1:50 dilution) were from Abcam. Immune positivity was revealed by the appropriate secondary Cy3 (indocarbocyanine)-conjugated antibody (1:1000 dilution; Amersham Pharmacia Biotech). Nuclei were stained using DAPI (4′,6-diamidino-2-phenylindole) and slides were examined with an Olympus Fluoview 300 confocal laser scanning microscope.
Quantitative RT (real-time)-PCR
These experiments were carried out as described previously [16,17]. The FAM (6-carboxyfluorescein)-labelled probes and primers for the specific genes or the housekeeping gene GAPDH (glyceraldehyde-3-phosphate dehydrogenase) are shown in Table 1. All reagents were purchased from Applied Biosystems (Foster City). Relative gene expression was calculated as 2−ΔCt (ΔCt=Ct of the target−Ct of GAPDH).
Table 1. FAM-labelled probes and primers (Applied Biosystems) for specific genes or for the housekeeping gene, GAPDH, used in the RT-PCR assays.
| Gene | Primer ID |
|---|---|
| CD11b (Itgam) | Mm00434455-m1 |
| TNF | Mm00443258-m1 |
| TGF-β1 | Mm00441724-m1 |
| TIMP-1 | Mm00441818-m1 |
| Type-I procollagen | Mm00801666-g1 |
| α-Smooth muscle actin | Mm01546133_m1 |
| MGL1 | Mm00546124_m1 |
| IFN-γ | Mm00801778_m1 |
| IL-4 | Mm00445259_m1 |
| iNOS | Mm00440485_m1 |
| CCL5 | Mm01302427_m1 |
| GAPDH | Mm99999915-g1 |
Isolation and culture of mouse hepatic stellate cells
Hepatic stellate cells were isolated from three Balb/C and three C57Bl/6 mice in parallel. Procedures for isolation have been described in detail elsewhere [19]. After isolation, cells were cultured in a complete medium until confluent, deprived of serum for 16 h, and utilized for a chemotactic assay in response to CCL2, as described previously [8].
Statistical analysis
Results are expressed as means ± S.D. Comparisons of animal groups were analysed by Student's t test or by non-parametric tests, as appropriate. P values ≤0.05 were considered significant.
RESULTS
Lack of CCL2 protects Balb/C mice from injury and inflammation
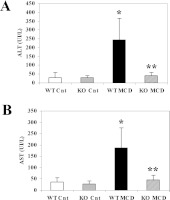
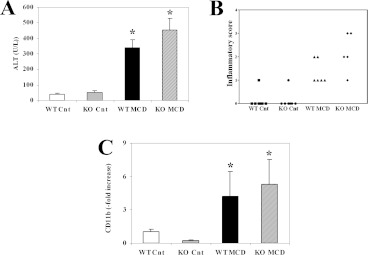
We first analysed the effects of an MCD diet given for 8 weeks to WT and CCL2-KO Balb/C mice. At the end of the treatment period, mice fed on the MCD diet showed a marked and significant decrease in body weight (Table 2). However, no differences were observed comparing WT and CCL2-KO mice with either diet regimen. The liver/body weight ratio tended to be higher in mice fed on the MCD diet (Table 2), but no significant differences were found comparing the different diets or the different mouse genotypes. In WT mice fed on the MCD diet, the levels of ALT and AST (aspartate aminotransferase) were increased more than 5-fold in comparison with mice treated with the control diet (Figure 1). In contrast, in mice lacking CCL2, aminotransferase levels were significantly lower than in WT animals fed on the MCD diet. No differences were observed comparing WT and CCL2-KO mice fed on the control diet.
Table 2. Body weight and liver/body weight in WT and CCL2-KO Balb/C mice subjected to different dietary regimens.
*P<0.05 compared with WT animals fed on the control diet.
| Diet | Body weight (g) | Liver/body weight (×100) |
|---|---|---|
| Control diet | ||
| WT | 22.56±2.00 | 4.33±0.24 |
| CCL2-KO | 21.88±1.21 | 4.61±0.41 |
| MCD | ||
| WT | 14.48±7.69* | 4.79±0.47 |
| CCL2-KO | 12.59±1.22* | 5.31±0.94 |
Figure 1. Lack of CCL2 reduces aminotransferase levels in Balb/C mice.
WT or CCL2-KO mice were fed for 8 weeks on the control diet or on the MCD diet. WT Cnt, WT animals fed on the control diet; KO Cnt, CCL2-KO animals fed on the control diet; WT MCD, WT animals fed on the MCD diet; KO MCD, CCL2-KO animals fed on the MCD diet. At the end of the study protocol, animals were killed and serum ALT (A) and AST (B) were assayed as described in Materials and methods section. *P<0.01 compared with WT animals fed on the control diet; **P<0.01 compared with WT animals fed on the MCD diet. UI, international units.
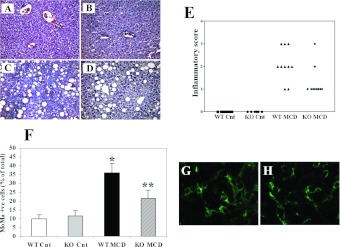
Feeding with an MCD diet is associated with steatosis, hepatocellular injury and inflammation, which recapitulate the histological picture observed in patients with NASH. In MCD-diet-fed WT mice, the appearance of macrovescicular steatosis was accompanied by infiltration with inflammatory cells and degenerative changes in hepatocytes, including lobular necrosis (Figures 2A–2D). In addition, aggregates of inflammatory cells surrounding fat-laden hepatocytes were observed, forming lipogranulomas. In CCL2-KO animals, no differences in steatosis were evident, whereas inflammatory changes were less marked, with a lower number of inflammatory cell aggregates and less dense infiltrates (Figures 2A–2D). When fed on a control diet, the livers of CCL2-KO mice had a histological appearance indistinguishable from that of WT mice. Blinded evaluation of necro-inflammatory changes observed in the different genotypes demonstrated an increased score in WT mice fed on the MCD diet (Figure 2E). Necro-inflammation was less severe in CCL2-KO mice fed on an MCD diet, with an overall score significantly lower than that of WT animals (Figure 2E). Scoring of steatosis confirmed that no significant differences were present comparing CCL2-KO and WT mice (results not shown).
Figure 2. Effects of CCL2 deficiency on liver histology and inflammation following administration of an MCD diet in Balb/C mice.
WT or CCL2-KO mice were fed for 8 weeks on the control diet or the MCD diet. (A–D) At the end of the study protocol, animals were killed and liver sections were stained with haematoxylin and eosin. (A) WT animals fed on the control diet (WT-Cnt); (B) CCL2-KO animals fed on the control diet (KO Cnt); (C) WT animals fed on the MCD diet (WT MCD); (D) CCL2-KO animals fed on the MCD diet (KO MCD). (E) Individual necro-inflammatory scores in the four groups of mice are indicated. (F) Cells positive for the monocyte/macrophage marker MoMa2 were counted in slides from each group of mice indicated as above. *P<0.01 compared with WT animals fed on the control diet; **P<0.01 compared with WT animals fed on the MCD diet. (G,H) Representative immunofluorescence staining for MoMa2-positive cells (green) in WT animals fed on the MCD diet (G) and CCL2-KO animals fed on the MCD diet (H).
CCL2 is one of the major chemoattactrants for mononuclear cells, including monocytes. Thus we immunostained the liver of different groups of mice with MoMa2, which is an antibody recognizing an intracellular antigen expressed by macrophages and infiltrating monocytes (Figures 2F–2H). WT mice fed on the MCD diet had an approximate 4-fold increase in the percentage of MoMa2-positive cells compared with those fed on the control diet (Figure 2F). A lack of CCL2 was associated with a significantly lower abundance of monocytes/macrophages, which were approximately half as many as in WT mice. In both genotypes of mice fed on the control diet the MoMa2 antibodies recognized Kupffer cells, and no differences were observed (Figure 2F).
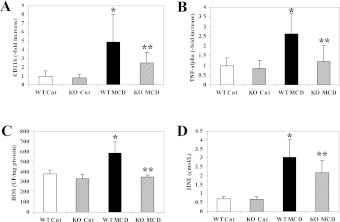
To provide further evidence for the protective effects of CCL2 deficiency on inflammation, we measured the gene expression of CD11b, a marker of inflammatory cells. A 5-fold significant increase in CD11b expression was observed in WT mice fed on the MCD diet in comparison with those fed on the control diet (Figure 3A). Expression of CD11b was significantly reduced in MCD-diet-fed CCL2-KO mice, confirming reduced inflammation and in agreement with the results of the histological examination. TNFα (tumour necrosis factor α) is a major inflammatory cytokine and has been implicated in the pathogenesis of NASH-associated hepatocyte damage [3]. Transcripts encoding the TNFα gene were markedly more abundant after feeding WT mice with the MCD diet (Figure 3B), and also in this case CCL2 deficiency reduced the expression of this cytokine.
Figure 3. Lack of CCL2 reduces the expression of proinflammatory genes and limits generation of oxidative stress induced by an MCD diet in Balb/C mice.
WT or CCL2-KO mice were fed for 8 weeks on the control diet or the MCD diet. Groups are as in the legend to Figure 1. (A,B) At the end of the study protocol, RNA was isolated from liver tissue and expression of CD11b (A) or of TNFα (B) was measured by RT-PCR. (C,D) A sample of fresh liver tissue was harvested, and levels of ROS (C) and of HNE (D) were assayed as described in the Materials and methods sections. *P<0.01 compared with WT animals fed on the control diet; **P<0.01 compared with WT animals fed on the MCD diet. UF, units of fluorescence.
CCL2 is involved in the generation of oxidative stress in Balb/C mice
Increased generation of oxidative-stress-related products is a hallmark of NASH and is reproduced by the MCD model in rodents [20]. In Balb/C mice fed on the MCD diet for 8 weeks, we analysed intrahepatic levels of ROS and HNE, which represent the major oxidant and non-oxidant products of oxidative stress respectively. Both these compounds were significantly more abundant in WT mice fed on the MCD diet than in those fed on the control diet (Figures 3C and 3D). A lack of CCL2 resulted significantly in lower levels of both ROS and HNE in comparison with WT mice. These results demonstrate that CCL2 contributes to the generation of oxidative stress associated with the MCD diet in Balb/C mice.
Fibrogenesis induced by the MCD diet is modulated by the absence of CCL2 in Balb/C mice
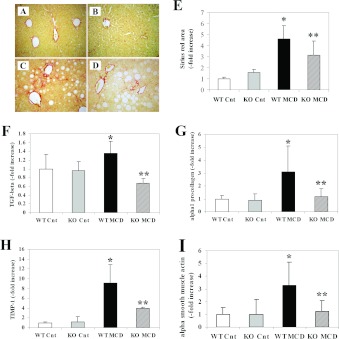
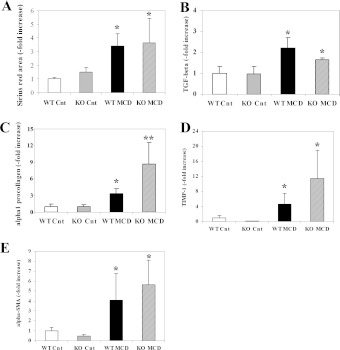
As in other forms of chronic liver damage, the MCD diet triggers the initiation of a fibrogenic process that, in mice, leads to accumulation of fibrillar matrix after at least 8 weeks. To determine whether protection from necro-inflammatory damage is associated with changes in fibrogenesis, sections from mice fed on the control or MCD diets were stained with Sirius Red (Figures 4A–4D). As already described, administration of the MCD diet for 8 weeks induced the matrix accumulation predominantly in zone 3 of the hepatic acinus, with the appearance of a ‘chickenwire’ pattern. Fibrosis was also detectable in mice defective for CCL2, but in this case fibrotic bundles appeared to be less thick and more sparse compared with WT Balb/C mice. Computerized image analysis of the slides stained with Sirius Red demonstrated that in CCL2-KO mice accumulation of fibrillar collagen was significantly reduced compared with WT animals (Figure 4E).
Figure 4. Effects of CCL2 deficiency on the development of fibrosis induced by an MCD diet and expression of pro-fibrogenic genes in Balb/C mice.
WT or CCL2-KO mice were fed for 8 weeks on the control diet or the MCD diet. Groups are as in the legend to Figure 1. (A–D) At the end of the study protocol, animals were killed and liver sections stained with Sirius Red. (A) WT animals fed on the control diet (WT-Cnt); (B) CCL2-KO animals fed on the control diet (KO Cnt); (C) WT animals fed on the MCD diet (WT MCD); (D) CCL2-KO animals fed on the MCD diet (KO MCD). (E) Sirius-Red-stained slides were subjected to image analysis, and the area occupied by fibrotic tissue was measured in WT and CCL2-KO mice fed on the control diet or the MCD diet. (F–I) RNA was isolated from liver tissue and the expression of TGF-β (F), type I procollagen (G), TIMP-1 (H) and α-smooth muscle actin (I) were measured by RT-PCR. *P<0.01 compared with WT animals fed on the control diet; **P<0.01 compared with WT animals fed on the MCD diet.
To obtain further insight into the modulation of fibrogenesis by CCL2 in this model, transcript levels of genes implicated in hepatic wound healing and fibrosis were evaluated. The epression of TGF-β (transforming growth factor-β), a major pro-fibrogenic cytokine, was significantly up-regulated by feeding the MCD diet in WT Balb/C mice. This effect was significantly reduced in CCL2-KO animals (Figure 4F). In a similar fashion, up-regulated gene expression of type I procollagen, and of TIMP-1 (tissue inhibitor of metalloproteinases-1), were significantly reduced in mice lacking CCL2 (Figures 4G and 4H). In addition, the expression of α-smooth muscle actin, which is a marker of hepatic myofibroblasts, was markedly increased by the MCD diet and significantly reduced in the absence of CCL2 (Figure 4I). No significant changes in the expression of fibrogenic genes were observed when comparing WT and CCL2-KO mice fed on the control diet. Taken together, these results indicate that, in Balb/C mice, CCL2 is involved in the fibrogenic progression of experimental steatohepatitis induced by the MCD diet.
Injury, inflammation and fibrosis are not affected by CCL2 deficiency in C57Bl/6 mice
Recently, Kassel et al. [15] investigated the development of MCD-induced steatohepatitis in CCL2-KO mice raised on a C57Bl/6 background, without detecting significant differences in hepatic injury and inflammation compared with WT mice. Since our findings obtained in Balb/C mice demonstrate that CCL2 deficiency protects from necro-inflammation and fibrosis, we also analysed the effects of MCD feeding for 8 weeks and CCL2 deficiency in C57Bl/6 mice. In these mice, ALT levels were elevated to a higher extent than in Balb/C mice (approximately 7-fold). However, CCL2 deficiency did not result in any significant changes in ALT levels (Figure 5A). In agreement with this observation, steatosis (results not shown) and necro-inflammation (Figure 5B) evaluated by histological scoring were not significantly different when comparing the two genotypes. Moreover, intrahepatic expression of the inflammatory marker CD11b was increased to a similar extent in mice fed on the MCD diet irrespective of the absence of CCL2 (Figure 5C).
Figure 5. CCL2 deficiency does not affect aminotransferase levels and inflammation following administration of an MCD diet in C57Bl/6 mice.
WT or CCL2-KO mice were fed for 8 weeks on the control diet or the MCD diet. Groups are as in the legend to Figure 1. (A) Serum ALT was assayed as described in Materials and methods section. (B) Individual necro-inflammatory scores in the four groups of mice. (C) RNA was isolated from liver tissue, and expression of CD11b was measured by RT-PCR. U, units.
We next evaluated whether a similar inflammatory response to the MCD diet was associated with a similar fibrogenic response in the absence of CCL2 in C57Bl/6 mice. Image analysis of the area stained with Sirius Red demonstrated a marked and significant increase in mice fed on the MCD diet compared with control animals (Figure 6A). However, no differences comparing WT mice and mice lacking CCL2 were observed. In agreement with this observation, intrahepatic expression of genes involved in fibrogenesis was not reduced in the absence of CCL2 in C57Bl/6 mice fed on the MCD diet, and the expression levels of α1-procollagen were even higher (Figures 6C–6E). Accordingly, α-smooth muscle actin expression, a marker of activated myofibroblasts, was similar irrespective of the presence of CCL2 (Figure 6E). These results demonstrate that the effects of CCL2 on hepatic necro-inflammation and fibrosis induced by the MCD diet are dependent on the mouse genetic background.
Figure 6. CCL2 deficiency does not affect the development of fibrosis or the expression of pro-fibrogenic genes following administration of an MCD diet in C57Bl/6 mice.
WT or CCL2-KO mice were fed for 8 weeks on the control diet or the MCD diet. Groups are as in the legend to Figure 1. (A) Sirius-Red-stained slides were subjected to image analysis, and the area occupied by fibrotic tissue was measured in WT and CCL2-KO mice fed on a control diet or an MCD diet. (B–E) RNA was isolated from liver tissue and the expression of TGF-β (B), type I procollagen (C), TIMP-1 (D) and α-smooth muscle actin (E) were measured by RT-PCR. *P<0.01 compared with WT animals fed on the control diet; **P<0.01 compared with WT animals fed on the MCD diet.
Comparison of cytokine expression and of macrophage polarization markers in mice with different genetic backgrounds
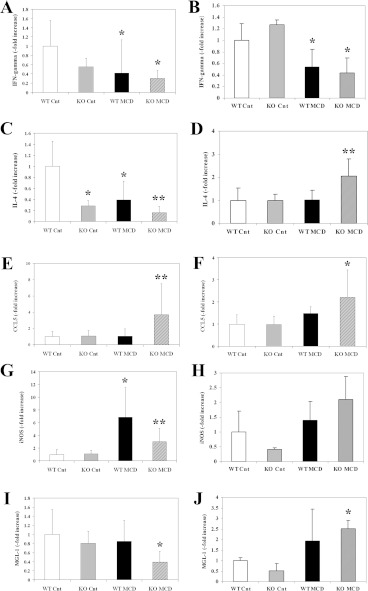
To investigate the mechanisms underlying the different inflammatory and fibrogenic response observed in the two strains of mice upon CCL2 deletion, we first investigated a group of cytokines involved in immune response and inflammation. The levels of IFN-γ (interferon-γ), which promotes inflammation and is associated with a lower fibrogenic response, were reduced by the MCD diet in both Balb/C and C57Bl/6 mice, and no effects of CCL2 deletion were observed (Figures 7A and 7B). In contrast, the expression of IL (interleukin)-4, a cytokine related to Th2 polarization, was reduced in Balb/C mice fed on the MCD diet, and the reduction was more evident in the absence of CCL2 (Figure 7C). In contrast, in C57Bl/6 mice, the levels of IL-4 were higher in animals lacking CCL2 when fed on the MCD diet (Figure 7D).
Figure 7. Differential expression of genes involved in inflammation and immune response in mice with a different genetic background fed on the MCD diet.
WT or CCL2-KO mice on a Balb/C (A, C, E, G, I) or C57Bl/6 (B, D, F, H, J) genetic background were fed for 8 weeks on the control diet or on the MCD diet. At the end of the study protocol, RNA was isolated from liver tissue and the expression of IFN-γ (A,B), IL-4 (C,D), CCL5 (E,F), iNOS (G,H) or MGL1 (I,J) was assayed by RT-PCR. *P<0.01 compared with WT animals fed on the control diet; **P<0.01 compared with WT animals fed on the MCD diet.
To explore whether another member of the chemokine family could compensate for the function of CCL2, we analysed the expression of CCL5, a chemokine that targets inflammatory cells and activated stellate cells [21]. Expression of CCL5 was only modestly changed by the MCD diet in WT mice on both genetic backgrounds (Figures 7E and 7F). In the absence of CCL2, a significant increase in CCL5 expression was found to a similar extent in both Balb/C and C57Bl/6 mice.
We finally analysed the expression of iNOS (inducible NO synthase) and of MGL1 (macrophage galactose-type C-type lectin 1), markers of classically-activated (M1) or alternatively-activated (M2) macrophages respectively. In Balb/C mice, expression of iNOS was markedly increased in response to the MCD diet, and lack of CCL2 reduced the expression levels (Figure 7G), in agreement with the milder inflammatory phenotype of these mice (see Figures 2 and 3). iNOS was only modestly increased in C57Bl/6 mice, and no effects of CCL2 deficiency were observed. A different behaviour comparing the two genetic backgrounds was observed when MGL1 levels were measured. In fact, this M2 polarization marker was expressed at lower levels in MCD-diet-treated Balb/C mice lacking CCL2 (Figure 7I), and an opposite behaviour was found in C57Bl/6 mice, where MGL1 levels increased in the CCL2-deficient animals receiving the steatogenic diet (Figure 7J). We also explored whether the chemotactic effects of CCL2 on hepatic stellate cells could be different in the two mouse strains. However, hepatic stellate cells isolated from Balb/C or C57Bl/6 mice showed a similar chemotactic response to CCL2 (results not shown). Taken together, these results indicate that the different effects of CCL2 deficiency on inflammation and fibrogenesis caused by the MCD diet are associated with differential expression of immune response cytokines and macrophage polarization markers.
DISCUSSION
Hepatocellular damage, inflammation and fibrogenic progression are the pivotal characteristics of steatohepatitis. Inflammatory chemokines, such as CCL2, are expressed at low levels in normal conditions, whereas they are up-regulated following inflammatory damage such as in NASH [13,22,23]. In the present study, we provide evidence that in mice with experimental steatohepatitis, the effects of deletion of CCL2 on hepatic necro-inflammation and fibrogenesis are dependent on the genetic background of the mice. In Balb/C mice, modulation of hepatocellular injury and inflammation by CCL2 is supported by several lines of evidence, including a significant reduction of aminotransferase levels and of histological necro-inflammatory score. In addition, lack of CCL2 was associated with reduced expression of several genes implicated in fibrogenesis, and the net deposition of fibrillar matrix was significantly limited. Moreover, accumulation of activated hepatic stellate cells, as assessed by α-smooth muscle actin expression, was decreased. Taken together, these data indicate that in this genetic background, CCL2 plays a relevant role in the pathogenesis of the changes that characterize experimental steatohepatitis, including fibrosis.
Another important and novel finding of the experiments conducted in Balb/C mice, is the interference of CCL2 with oxidative stress. Generation of oxidative stress-related molecules has been suggested to be one of the ‘multiple hits’ that are responsible for progression of fatty liver to NASH [2,24]. Using state-of-the-art techniques, we show that intra-hepatic generation of ROS and reactive aldehydes such as HNE is significantly lower when CCL2 is deleted in Balb/C mice. These data become even more relevant in light of the recent report demonstrating that antioxidants may provide a clinical benefit to patients with NASH [25].
Very recently, Kassel et al. [15] have reported the effects of CCL2 deficiency in C57Bl/6 mice challenged with an MCD diet for 4 weeks. No differences in inflammation and liver injury were observed, whereas a lower expression of genes involved in fibrogenesis was reported. However, in the MCD diet model, fibrogenesis is barely evident at 4 weeks, and time points longer than 8 weeks are necessary to appropriately establish the fibrogenic phenotype. For this reason, we investigated in detail the inflammatory and fibrogenic phenotype induced by the lack of CCL2 in C57Bl/6 mice on an MCD diet for 8 weeks, when most of the animals have unequivocal evidence of fibrosis. We confirmed that, in this strain of mice, no differences in hepatic injury and inflammation were associated with CCL2 deficiency, as indicated by histological scoring and CD11b expression. Surprisingly, we found that even fibrosis was not affected by CCL2 deficiency, as shown by image analysis of Sirius Red staining, expression of pro-fibrogenic genes, and of genes indicating accumulation of activated stellate cells.
Interestingly, both our study and the one of Kassel et al. [15] demonstrate the lack of effect of CCL2 deficiency on steatosis, irrespective of the inhibition of inflammation. These findings are probably dependent on the mechanisms that lead to steatosis in the MCD diet model, where impaired VLDL release is a major determinant of triacylglycerol (triglyceride) accumulation. This pathogenic mechanism is probably to be poorly sensitive to changes in inflammation possibly associated with CCL2 deficiency.
The different inflammatory and fibrogenic phenotype associated with CCL2 deficiency in the two mouse strains is a very intriguing finding. For this reason, we conducted additional experiments to elucidate possible mechanisms of the diverging phenotypes observed in the absence of CCL2. We first tested the possibility that hepatic stellate cells isolated from the two mouse strains could be differentially responsive to CCL2 in vitro. However, a similar chemotactic index was found incubating with CCL2 stellate cells from Balb/C or C57Bl/6 mice, ruling out the possibility of a lack of response to CCL2 as a mechanism for defective protection from fibrosis in C57Bl/6 mice. Next, we measured the expression of different cytokines implicated in inflammation and immune response and of markers of cytokine polarization. Analysis of the expression of cytokines implicated in the polarization toward a Th1 or Th2 phenotype, revealed a partially divergent response in the two strains of mice. Specifically, while the expression of IFN-γ was substantially similar, expression of IL -4, a Th2 cytokine, was significantly increased in MCD diet-treated, CCL2-deficient mice on a C57Bl/6 background, but not in the corresponding group on the Balb/C background. It is well established that a Th2-polarized response is associated with more marked fibrogenesis during injury in many tissues, including the liver [26–28]. In addition, IL-4 has been found to directly modulate the fibrogenic properties of hepatic myofibroblasts, resulting in increased expression of extracellular matrix [29,30]. Thus, it is likely that increased IL-4 expression and possibly a general skewing towards a Th2 response contribute to the maintenance of fibrogenesis despite the lack of CCL2.
Another interesting finding revealed by cytokine expression analysis in the two genetic backgrounds is the increased expression of MGL1, a marker related to alternative macrophage activation, in C57Bl/6 mice on an MCD diet. In recent years, classically activated (M1) macrophages, mostly involved in tissue injury and inflammation, and M2, or alternatively activated macrophages, implicated in the process of tissue repair, have been described and characterized [31]. It should be kept in mind that M1 or M2 macrophages represent extremes of a continuum in a universe of activation states that are still incompletely characterized [32]. Nonetheless, the finding that a lack of CCL2 in C57Bl/6 mice fed on the MCD diet might show a greater M2 polarization suggests a possible additional mechanism responsible for the maintenance of a fibrogenic stimulus. In fact, M2 polarization is generally associated with Th2 lymphocyte polarization and with expression of pro-fibrogenic factors, such as TGF-β [31]. In addition, several conditions characterized by chronic tissue injury and a fibrogenic evolution are associated with an infiltrate that comprises M2 macrophages.
Besides the conserved fibrogenesis, CCL2-deficient, MCD diet-treated C57Bl/6 mice also have comparable inflammation with respect to the Balb/C counterpart. We hypothesized that, in C57Bl/6 mice, CCL2 deficiency in mice fed on the MCD diet could be compensated by an increase in other chemokines involved in the recruitment of inflammatory cells. Thus we tested the expression of CCL5, a chemokine that targets both inflammatory and fibrogenic cells, and found a marked increase in comparison with WT mice on the same genetic background. However, a similar behaviour was observed in Balb/C mice, indicating that CCL5 is not responsible for the lack of protection from inflammation observed in C57Bl/6. Interestingly, in some systems, including the adipose tissue, infiltration with M2-polarized macrophages has been associated with a marked infiltration of M1-polarized cells [33]. These results indicate that the general concept that M2 macrophages are anti-inflammatory may not be applied to all situations and allows us to speculate that increased M2 macrophages in C57Bl/6 mice could contribute to the maintenance of inflammation. Clearly, additional studies, including a wide-spectrum analysis of cytokine expression and inflammatory cell phenotypes are required to provide detailed information on the mechanisms underlying the differences between the two genetic backgrounds. Although the mechanisms underlying the different inflammatory response observed comparing C57Bl/6 and Balb/C mice fed on the MCD diet remain incompletely resolved, these results suggest that genetic differences may be responsible for a different inflammatory response in patients with steatohepatitis. Along these lines, the severity of NASH progression has been shown to change according to the different genetic backgrounds of the patients [34], and the possibility that variations in the inflammatory response contributes to these observations warrants further studies.
In principle, CCL2 appears to be an appealing target in human steatohepatitis. Besides its well-established role on inflammation, CCL2 has been shown to directly modulate the biology of fibrogenic cells, suggesting the possibility that interference with this chemokine directly blocks fibrosis [8,35]. This is supported by elegant studies in chimaeric mice, where deletion of CCR2, the receptor for CCL2, in liver resident cells, including hepatic stellate cells, was sufficient to inhibit the fibrogenic response [36]. Moreover, CCL2 has been shown to play a detrimental role in adipose tissue inflammation [11,12], favouring the release of non-esterified ‘free’ fatty acids and leading to adipokine imbalance. Very recently, a pharmacological inhibitor of CCL2 was found to block inflammation and steatosis, but not fibrosis, in the MCD diet model of steatohepatitis in C57Bl/6 mice [37]. The reasons for the discrepancy with the results of our present study and that of Kassel et al. [15] are unclear, but may be based on the fact that we utilized CCL2-KO mice in which compensatory mechanisms may have taken place. Results provided herein, highlighting the different response of mice with different genetic backgrounds, make the translation of these proof-of-concept studies more difficult. It can be speculated that interfering with CCL2 may prove to be beneficial only in a group of patients, although genetic differences in rodents do not necessarily parallel the genetic diversity of humans. Although additional work in experimental models is warranted to shed further light on the role of this chemokine in mouse steatohepatitis, there is still a strong rationale to investigate approaches that lead to diminished effects of CCL2 in patients with NASH, considering that chemokines and their receptors are also the focus of considerable attention as drug targets [38].
AUTHOR CONTRIBUTION
Sara Galastri, Elena Zamara, Erica Novo, Angela Provenzano, Wanda Delogu, Francesco Vizzutti, Salvatore Sutti, Irene Locatelli, Nadia Navari, Elisa Vivoli and Alessandra Caligiuri treated the animals, collected the samples, performed the experiments, analysed the data and revised the paper. Stefano Milani planned the experiments, scored the histological slides, analysed the data and revised the paper. Massimo Pinzani, Emanuele Albano and Maurizio Parola planned the experiments, analysed the data and revised the paper. Fabio Marra planned the experiments, analysed the data and wrote the paper.
ACKNOWLEDGEMENTS
We are indebted to Dr Barrett Rollins for the gift of the CCL2-KO mice.
FUNDING
This work was supported by the Ministero dell'Istruzione, dell'Università e della Ricerca Scientifica (MIUR) (PRIN project); the University of Florence and the European Community's Seventh Framework Programme [FP7/2007-2013 under the grant number HEALTH-F2-2009-241762 for the project FLIP (to F.M.)]; and the Regione Piemonte (Progetti Ricerca Sanitaria Finalizzata) (grants to M. Parola and E.A.).
References
- 1.Angulo P. Nonalcoholic fatty liver disease. N Engl. J. Med. 2002;346:1221–1231. doi: 10.1056/NEJMra011775. [DOI] [PubMed] [Google Scholar]
- 2.Marra F., Gastaldelli A., Svegliati Baroni G., Tell G., Tiribelli C. Molecular basis and mechanisms of progression of non-alcoholic steatohepatitis. Trends Mol. Med. 2008;14:72–81. doi: 10.1016/j.molmed.2007.12.003. [DOI] [PubMed] [Google Scholar]
- 3.Tilg H., Moschen A. R. Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology. 2010;52:1836–1846. doi: 10.1002/hep.24001. [DOI] [PubMed] [Google Scholar]
- 4.Wasmuth H. E., Tacke F., Trautwein C. Chemokines in liver inflammation and fibrosis. Semin. Liver Dis. 2010;30:215–225. doi: 10.1055/s-0030-1255351. [DOI] [PubMed] [Google Scholar]
- 5.Deshmane S. L., Kremlev S., Amini S., Sawaya B. E. Monocyte chemoattractant protein-1 (MCP-1): an overview. J. Interferon Cytokine Res. 2009;29:313–326. doi: 10.1089/jir.2008.0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gu L., Tseng S., Horner R. M., Tam C., Loda M., Rollins B. J. Control of TH2 polarization by the chemokine monocyte chemoattractant protein-1. Nature. 2000;404:407–411. doi: 10.1038/35006097. [DOI] [PubMed] [Google Scholar]
- 7.Schecter A. D., Berman A. B., Yi L., Ma H., Daly C. M., Soejima K., Rollins B. J., Charo I. F., Taubman M. B. MCP-1-dependent signaling in CCR2−/− aortic smooth muscle cells. J. Leukocyte Biol. 2004;75:1079–1085. doi: 10.1189/jlb.0903421. [DOI] [PubMed] [Google Scholar]
- 8.Marra F., Romanelli R. G., Giannini C., Failli P., Pastacaldi S., Arrighi M. C., Pinzani M., Laffi G., Montalto P., Gentilini P. Monocyte chemotactic protein-1 as a chemoattractant for human hepatic stellate cells. Hepatology. 1999;29:140–148. doi: 10.1002/hep.510290107. [DOI] [PubMed] [Google Scholar]
- 9.Marra F. Chemokines in liver inflammation and fibrosis. Front. Biosci. 2002;7:d1899-1914. doi: 10.2741/A887. [DOI] [PubMed] [Google Scholar]
- 10.Berres M. L., Nellen A., Wasmuth H. E. Chemokines as immune mediators of liver diseases related to the metabolic syndrome. Dig. Dis. 2010;28:192–196. doi: 10.1159/000282085. [DOI] [PubMed] [Google Scholar]
- 11.Kanda H., Tateya S., Tamori Y., Kotani K., Hiasa K.-i., Kitazawa R., Kitazawa S., Miyachi H., Maeda S., Egashira K., Kasuga M. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Invest. 2006;116:1494–1505. doi: 10.1172/JCI26498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Weisberg S. P., Hunter D., Huber R., Lemieux J., Slaymaker S., Vaddi K., Charo I., Leibel R. L., Ferrante A.W., Jr CCR2 modulates inflammatory and metabolic effects of high-fat feeding. J. Clin. Invest. 2006;116:115–124. doi: 10.1172/JCI24335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Westerbacka J., Kolak M., Kiviluoto T., Arkkila P., Siren J., Hamsten A., Fisher R. M., Yki-Jarvinen H. Genes involved in fatty acid partitioning and binding, lipolysis, monocyte/macrophage recruitment, and inflammation are overexpressed in the human fatty liver of insulin-resistant subjects. Diabetes. 2007;56:2759–2765. doi: 10.2337/db07-0156. [DOI] [PubMed] [Google Scholar]
- 14.Bertola A., Bonnafous S., Anty R., Patouraux S., Saint-Paul M. C., Iannelli A., Gugenheim J., Barr J., Mato J. M., Le Marchand-Brustel Y., et al. Hepatic expression patterns of inflammatory and immune response genes associated with obesity and NASH in morbidly obese patients. PLoS ONE. 2010;5:e13577. doi: 10.1371/journal.pone.0013577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kassel K. M., Guo G. L., Tawfik O., Luyendyk J. P. Monocyte chemoattractant protein-1 deficiency does not affect steatosis or inflammation in livers of mice fed a methionine-choline-deficient diet. Lab. Invest. 2010;90:1794–1804. doi: 10.1038/labinvest.2010.143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zamara E., Galastri S., Aleffi S., Petrai I., Aragno M., Mastrocola R., Novo E., Bertolani C., Milani S., Vizzutti F., et al. Prevention of severe toxic liver injury and oxidative stress in MCP-1-deficient mice. J. Hepatol. 2007;46:230–238. doi: 10.1016/j.jhep.2006.09.007. [DOI] [PubMed] [Google Scholar]
- 17.Vizzutti F., Provenzano A., Galastri S., Milani S., Delogu W., Novo E., Caligiuri A., Zamara E., Arena U., Laffi G., et al. Curcumin limits the fibrogenic evolution of experimental steatohepatitis. Lab. Invest. 2010;90:104–115. doi: 10.1038/labinvest.2009.112. [DOI] [PubMed] [Google Scholar]
- 18.Novo E., Busletta C., Bonzo L. V., Povero D., Paternostro C., Mareschi K., Ferrero I., David E., Bertolani C., Caligiuri A., et al. Intracellular reactive oxygen species are required for directional migration of resident and bone marrow-derived hepatic pro-fibrogenic cells. J. Hepatol. 2011;54:964–974. doi: 10.1016/j.jhep.2010.09.022. [DOI] [PubMed] [Google Scholar]
- 19.Brun P., Castagliuolo I., Pinzani M., Palu G., Martines D. Exposure to bacterial cell wall products triggers an inflammatory phenotype in hepatic stellate cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2005;289:G571–G578. doi: 10.1152/ajpgi.00537.2004. [DOI] [PubMed] [Google Scholar]
- 20.Leclercq I. A., Farrell G. C., Field J., Bell D. R., Gonzalez F. J., Robertson G. R. CYP2E1 and CYP4A as microsomal catalysts of lipid peroxides in murine nonalcoholic steatohepatitis. J. Clin. Invest. 2000;105:1067–1075. doi: 10.1172/JCI8814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sahin H., Trautwein C., Wasmuth H. E. Functional role of chemokines in liver disease models. Nat. Rev. Gastroenterol. Hepatol. 2010;7:682–690. doi: 10.1038/nrgastro.2010.168. [DOI] [PubMed] [Google Scholar]
- 22.Haukeland J. W., Damas J. K., Konopski Z., Loberg E. M., Haaland T., Goverud I., Torjesen P. A., Birkeland K., Bjoro K., Aukrust P. Systemic inflammation in nonalcoholic fatty liver disease is characterized by elevated levels of CCL2. J. Hepatol. 2006;44:1167–1174. doi: 10.1016/j.jhep.2006.02.011. [DOI] [PubMed] [Google Scholar]
- 23.Leclercq I. A., Farrell G. C., Sempoux C., dela Pena A., Horsmans Y. Curcumin inhibits NF-κB activation and reduces the severity of experimental steatohepatitis in mice. J. Hepatol. 2004;41:926–934. doi: 10.1016/j.jhep.2004.08.010. [DOI] [PubMed] [Google Scholar]
- 24.Feldstein A. E., Bailey S. M. Emerging role of redox dysregulation in alcoholic and nonalcoholic fatty liver disease. Antioxid. Redox Signaling. 2011;15:421–424. doi: 10.1089/ars.2011.3897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sanyal A. J., Chalasani N., Kowdley K. V., McCullough A., Diehl A. M., Bass N. M., Neuschwander-Tetri B. A., Lavine J. E., Tonascia J., Unalp A., et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N. Engl. J. Med. 2010;362:1675–1685. doi: 10.1056/NEJMoa0907929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shi Z., Wakil A. E., Rockey D. C. Strain-specific differences in mouse hepatic wound healing are mediated by divergent T helper cytokine responses. Proc. Natl. Acad. Sci. U.S.A. 1997;94:10663–10668. doi: 10.1073/pnas.94.20.10663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Marra F., Aleffi S., Galastri S., Provenzano A. Mononuclear cells in liver fibrosis. Semin Immunopathol. 2009;31:345–358. doi: 10.1007/s00281-009-0169-0. [DOI] [PubMed] [Google Scholar]
- 28.Barron L., Wynn T. A. Fibrosis is regulated by Th2 and Th17 responses and by dynamic interactions between fibroblasts and macrophages. Am. J. Physiol. Gastrointest. Liver Physiol. 2011;300:G723–G728. doi: 10.1152/ajpgi.00414.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Jin Z., Sun R., Wei H., Gao X., Chen Y., Tian Z. Accelerated liver fibrosis in hepatitis B virus transgenic mice: involvement of natural killer T cells. Hepatology. 2011;53:219–229. doi: 10.1002/hep.23983. [DOI] [PubMed] [Google Scholar]
- 30.Aoudjehane L., Pissaia A., Jr, Scatton O., Podevin P., Massault P. P., Chouzenoux S., Soubrane O., Calmus Y., Conti F. Interleukin-4 induces the activation and collagen production of cultured human intrahepatic fibroblasts via the STAT-6 pathway. Lab. Invest. 2008;88:973–985. doi: 10.1038/labinvest.2008.61. [DOI] [PubMed] [Google Scholar]
- 31.Laskin D. L., Sunil V. R., Gardner C. R., Laskin J. D. Macrophages and tissue injury: agents of defense or destruction? Annu. Rev. Pharmacol. Toxicol. 2010;51:267–288. doi: 10.1146/annurev.pharmtox.010909.105812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sica A., Mantovani A. Macrophage plasticity and polarization: in vivo veritas. J. Clin. Invest. 2012;122:787–795. doi: 10.1172/JCI59643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Westcott D. J., Delproposto J. B., Geletka L. M., Wang T., Singer K., Saltiel A. R., Lumeng C. N. MGL1 promotes adipose tissue inflammation and insulin resistance by regulating 7/4hi monocytes in obesity. J. Exp. Med. 2009;206:3143–3156. doi: 10.1084/jem.20091333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lomonaco R., Ortiz-Lopez C., Orsak B., Finch J., Webb A., Bril F., Louden C., Tio F., Cusi K. Role of ethnicity in overweight and obese subjects with nonalcoholic steatohepatitis (NASH) Hepatology. 2011;54:837–845. doi: 10.1002/hep.24483. [DOI] [PubMed] [Google Scholar]
- 35.Kruglov E. A., Nathanson R. A., Nguyen T., Dranoff J. A. Secretion of MCP-1/CCL2 by bile duct epithelia induces myofibroblastic transdifferentiation of portal fibroblasts. Am. J. Physiol. Gastrointest. Liver Physiol. 2006;290:G765–G771. doi: 10.1152/ajpgi.00308.2005. [DOI] [PubMed] [Google Scholar]
- 36.Seki E., de Minicis S., Inokuchi S., Taura K., Miyai K., van Rooijen N., Schwabe R. F., Brenner D. A. CCR2 promotes hepatic fibrosis in mice. Hepatology. 2009;50:189–197. doi: 10.1002/hep.22952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Baeck C., Wehr A., Karlmark K. R., Heymann F., Vucur M., Gassler N., Huss S., Klussmann S., Eulberg D., Luedde T., et al. Pharmacological inhibition of the chemokine CCL2 (MCP-1) diminishes liver macrophage infiltration and steatohepatitis in chronic hepatic injury. Gut. 2012;61:416–426. doi: 10.1136/gutjnl-2011-300304. [DOI] [PubMed] [Google Scholar]
- 38.Berres M. L., Koenen R. R., Rueland A., Zaldivar M. M., Heinrichs D., Sahin H., Schmitz P., Streetz K. L., Berg T., Gassler N., et al. Antagonism of the chemokine Ccl5 ameliorates experimental liver fibrosis in mice. J. Clin. Invest. 2010;120:4129–4140. doi: 10.1172/JCI41732. [DOI] [PMC free article] [PubMed] [Google Scholar]