Abstract
Nitric oxide (NO) production by the vascular endothelium maintains an essential antiinflammatory, cytoprotective influence on the blood vessel wall. A key component of this activity is attributed to prevention of leukocyte–endothelial cell interactions, yet the underlying mechanisms remain unclear. The NO receptor, soluble guanylate cyclase (sGC), is expressed in endothelial cells but fulfils an unknown function. Therefore, we used intravital microscopy in mesenteric postcapillary venules from WT and endothelial nitric oxide synthase (eNOS) knockout (eNOS–/–) mice, and an sGC activator (BAY 41–2272), to investigate a potential role for sGC in the regulation of adhesion molecule expression and leukocyte recruitment. Leukocyte rolling and adhesion was 6-fold greater in eNOS–/– than WT animals. BAY 41–2272 and the NO-donor, diethylamine-NONOate, reduced leukocyte rolling and adhesion in eNOS–/– mice to levels observed in WT animals. These effects were blocked by the sGC inhibitor ODQ [1H-(1,2,4)oxadiazolo(4,3-a)quinoxalin-1-one], which itself caused a 6-fold increase in leukocyte rolling and adhesion in WT mice. Increased leukocyte rolling and adhesion in IL-1β-treated mice was also inhibited by BAY 41–2272. Fluorescence-activated cell sorting analysis in vitro and a specific P-selectin neutralizing antibody in vivo revealed that selective down-regulation of P-selectin expression accounted for the antiadhesive effects of sGC activation. These data demonstrate that sGC plays a key antiinflammatory role by inhibiting P-selectin expression and leukocyte recruitment.
Keywords: endothelium, intravital microscopy
The recruitment of immune cells to sites of tissue injury is an important facet of an inflammatory response and is thought to represent a multistage process involving leukocyte rolling, adhesion, and emigration. Each stage of this response is triggered by the expression of specific adhesion molecules on the surface of leukocytes, platelets, and the vascular endothelium (see ref. 1). The regulation of this process therefore plays a key role in the development of an inflammatory response; moreover, it is clear that the active suppression of leukocyte/platelet activation under physiological conditions is paramount for maintenance of blood vessel integrity and patency. Nitric oxide (NO) production by the vascular endothelium exerts an important cytoprotective, antithrombotic influence on the blood vessel wall by preventing the activation and adherence of circulating cells and platelets (2–4). Loss of this inhibitory effect of NO, and other endothelium-derived mediators including prostacyclin (PGI2), results in a change in the endothelial cell to a prothrombotic, proinflammatory phenotype. It is thought that such changes contribute to diseases such as sepsis and atherosclerosis, and restenosis after balloon angioplasty. Hence, therapeutic intervention to prevent leukocyte/platelet activation may be beneficial in numerous inf lammatory cardiovascular pathologies.
The mechanism underlying the antiplatelet activity of NO has been well characterized and shown to involve cGMP and down-regulation of P-selectin and glycoprotein IIb/IIIa expression (5, 6). In contrast, the mechanism(s) by which NO modulates leukocyte adherence and recruitment, in terms of the putative second messenger systems and adhesion molecules involved, remains controversial. Investigations with specific nitric oxide synthase (NOS) knockout (KO) mice suggest that the endothelial (eNOS) and neuronal (nNOS) enzymes, but not the inducible (iNOS) isoform, are important in modulating adhesion molecule expression and leukocyte adherence (7–9). However, it is not clear whether the antileukocyte effects of NO are dependent on the activation of soluble guanylate cyclase (sGC) and subsequent formation of cGMP. Soluble GC is found in abundance in vascular endothelial cells (10), but its function remains undefined. Identification of a physiological role for sGC in endothelial cells would therefore characterize a previously unknown aspect of NO-sGC-cGMP signaling. The specific adhesion molecule(s) that might be targeted by cGMP to bring about a cytoprotective effect is also uncertain; indeed, several candidates including intercellular adhesion molecule 1 (ICAM-1), P-selectin, and β2-integrin (11–13) have been proposed. To address these uncertainties, we have used intravital microscopy to compare the antiadhesive activities of NO and a non-NO-based sGC activator, BAY 41–2272 (14), on leukocyte rolling and adherence to mesenteric postcapillary venules in vivo, in WT and eNOS KO (eNOS–/–) mice, and in WT animals exposed to an inf lammatory stimulus (IL-1β). Moreover, f luorescence-activated cell sorting (FACS) has been used to assess adhesion molecule expression on endothelial cells in culture and leukocytes/platelets in whole blood to determine the mechanism of the antiadherent effects of sGC activation.
Methods
Intravital Microscopy. All experiments were conducted according to the Animals (Scientific Procedures) Act of 1986 (United Kingdom).
Male WT and eNOS–/– mice (10–15 g, in-house colony) were anesthetized with diazepam (6 mg/kg, s.c.) and Hypnorm (0.7 mg/kg fentanyl citrate and 20 mg/kg fluanisone, i.m.). Cautery incisions were made along the abdominal region, and the mesenteric vascular bed was exteriorized and placed on a viewing Plexiglas stage. The preparation was mounted on a Zeiss Axioskop FS with a water immersion objective lens (magnification ×40), and an eyepiece (magnification ×10) was used to observe the microcirculation. The preparation was transilluminated with a 12-V, 100-W halogen light source. A Hitachi charge-coupled device color camera (model KPC571) acquired images that were displayed on a Sony Trinitron color video monitor (model PVM 1440QM) and recorded on a Sony superVHS video cassette recorder (model SVO-9500 MDP) for subsequent offline analysis. A video time–date generator (FOR.A video timer, model VTG-33) projected the time, date, and stopwatch function onto the monitor. Mesenteries were superfused with bicarbonate-buffered solution at 37°C [in g/liter: NaCl, 7.71; KCl, 0.25; MgSO4, 0.14; NaHCO3, 1.51; and CaCl2, 0.22 (pH 7.4); gassed with 5% CO2/95% N2] at a rate of 2 ml/min. The temperature of the stage was maintained at 37°C. This procedure has no effect on rectal temperature or blood pressure (data not shown). Red blood cell (rbc) velocity was measured in venules by using an optical Doppler velocimeter (Microcirculation Research Institute, Texas A&M University, College Station). Venular blood flow was calculated from the product of mean rbc velocity (Vmean = centerline velocity/1.6) and microvascular cross-sectional area, assuming a cylindrical geometry. Wall shear rate was calculated by the Newtonian definition: shear rate = 8,000 × (Vmean/diameter) (15). One to three randomly selected postcapillary venules (diameter between 20 and 40 μm, length at least 100 μm) were observed for each mouse; measurements were taken 5–10 min after exposure of the chosen vessels.
Murine recombinant IL-1β (PeproTech, Rocky Hill, NJ) was used to induce leukocyte rolling and adhesion. Mice received either IL-1β (5 ng i.p. in 0.25 ml of sterile saline) or vehicle alone, and the mesenteric vascular bed was prepared for microscopic observation 90 min or 3 h later, as described (16). The extent of the inflammatory response elicited by IL-1β was analyzed by measuring white blood cell velocity (Vwbc) in mm/s. Cell adhesion was quantified by counting, for each vessel, the number of adherent leukocytes in a 100-μm length.
The involvement of sGC in the cellular response to IL-1β was evaluated by using the non-NO-based sGC activator BAY 41–2272 (kind gift of Bayer, Wuppertal, Germany) or the sGC inhibitor ODQ [1H-(1,2,4)oxadiazolo(4,3-a)quinoxalin-1-one; synthesized by D. Madge, Medicinal Chemistry Department, Wolfson Institute for Biomedical Research]; these drugs were superfused over the exposed tissue at the time of microscopic observation. The concentrations of BAY 41–2272 and ODQ were titrated such that they produced effects on leukocyte adherence but did not alter vascular tone/blood flow.
To evaluate the importance of P-selectin in regulating leukocyte recruitment and mediating the effects of sGC/cGMP activation, after assessment of leukocyte rolling and adhesion in eNOS–/– animals and in WT animals treated with IL-1β (5 ng), a selective neutralizing monoclonal antibody (1 mg/kg, clone RB40.34, BD Biosciences) was injected into the tail vein, and rolling and adhesion were assessed 1 min later.
Cell Culture and Treatment. Human umbilical vein endothelial cells (HUVECs; BioWhittaker) were grown in EGM-II (BioWhittaker) supplemented with an endothelial cell bullet pack (3 mg/ml bovine brain extract/10 μg/ml hEGF/2% FCS, Clontech). Cells were harvested in trypsin/EDTA (BioWhittaker) and subcultured in six-well plates (Falcon) for use at passage 3. Confluent HUVECs were treated with human IL-1β (200 ng/ml) for 30 min to 5 h. Certain samples were treated with BAY 41–2272 (10 μM) at the time of stimulation. Cells were then washed in 2 ml of Hanks' balanced salt solution (HBSS) (BioWhittaker) for 1 min. Mouse anti-human monoclonal antibodies against adhesion molecules were diluted to 4 μg/ml with EGM-II containing 0.1% BSA and incubated in the dark at 37°C for 30 min with HUVECs (500 μl per well). HUVEC dissociation was performed by treating HUVECs with trypsin/EDTA (500 μl per well) at 37°C for 2 min. Trypsin activity was blocked by the addition of 500 μl of Trypsin Neutralizing Solution (BioWhittaker), and FACS analysis was performed on collected cells.
Blood Samples. Blood (3 ml) was taken from normal healthy subjects who had taken no medication for at least two weeks. Blood was collected into a 10-ml vacutainer tube containing 1.5 ml of acid-citrate-dextrose anticoagulant (ACD, Vacutainer, Becton Dickinson). Blood (5 μl) was placed in 40 μl of PBS-Hepes-BSA buffer (pH 7.4) in polypropylene tubes (ICN). Samples were then treated with either tumor necrosis factor (TNF)-α (20 ng/ml for 30 min) to stimulate adhesion molecule expression on white blood cells or thrombin (1 units/ml for 10 min) to stimulate CD62P expression on platelets. In some samples, BAY 41–2272 (10 μM) was administered at the time of stimulation. FITC-labeled antibodies were added after stimulation for 30 min at room temperature and were fixed with 0.5% paraformaldehyde (PFA).
Flow Cytometry. Flow cytometry was conducted on a FACSCalibur with a 15-mW, 488-nm, air-cooled argon-ion laser and analyzed by using cellquest software (Becton Dickinson). Adhesion molecule expression was determined as the percentage of cells emitting a fluorescent signal in either the FL1 or FL2 channel, depending on the fluorophore used (FL1 for FITC-conjugated antibodies and FL2 for RPE-conjugated antibodies). The boundary between cells that stained positive and negative for the adhesion molecules was determined according to the fluorescence distribution of positively stained relative to unstained samples. For HUVEC analysis, live cells were gated by using forward and side scatter characteristics, and only the fluorescence emissions of these cells were subsequently measured.
Platelets were identified from whole blood samples according to light scatter parameters, and a live gate was drawn around the platelet cloud for data acquisition. Anti-CD42b-stained samples demonstrated that >98% of events collected within this gate were platelets (data not shown).
FITC-conjugated CD62L, CD62E, CD62P, CD11b, and CD54 were purchased from Serotec. RPE-conjugated CD16 (a leukocyte-specific marker) was purchased from DAKO. RPE-conjugated CD62P (for endothelial cell study) was purchased from Serotec.
Statistical Analysis. All statistical analyses were performed by using prism (GraphPad, San Diego). All data are plotted graphically as mean values with vertical bars representing SEM. Tests of significance were conducted by using two-way ANOVA for multiple comparisons (intravital studies) or a Student's t test for differences between two data groups (FACS analysis). A P value of <0.05 was taken as an appropriate level of significance. The n values quoted similarly indicate the number of experiments and animals used.
Results
Leukocyte–Endothelial Cell Interactions in WT and eNOS KO Mice. In control (WT) animals injected with saline, leukocytes passed over the endothelium at high speed (VWBC = 54.9 ± 6.9 mm/s, n = 7), and this was accompanied by very low levels of basal rolling and adhesion (Figs. 1 and 2). In eNOS KO (eNOS–/–) mice, the leukocyte velocity (VWBC = 12.4 ± 2.2 mm/s, n = 7) was considerably slower, and basal rolling and adhesion were significantly increased (≈6-fold, Figs. 1 and 2). There was no significant difference between erythrocyte velocity and wall shear rates in vessels from WT and eNOS–/– animals or after treatment with any of the reagents used (Table 1), suggesting that any changes observed were not the result of alterations in vessel diameter or blood flow.
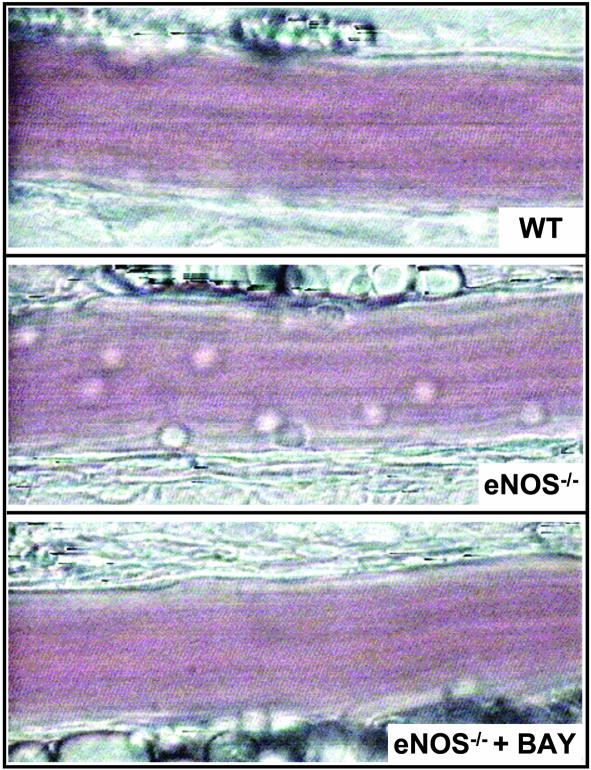
Fig. 1.
Video-microscopy images of leukocyte trafficking responses in mouse mesenteric postcapillary venules in vivo under basal conditions in WT mice (Top), eNOS–/– mice (Middle), and eNOS–/– mice in the presence of BAY 41–2272 (1 μM) (Bottom). The images are representative of at least five separate experiments.
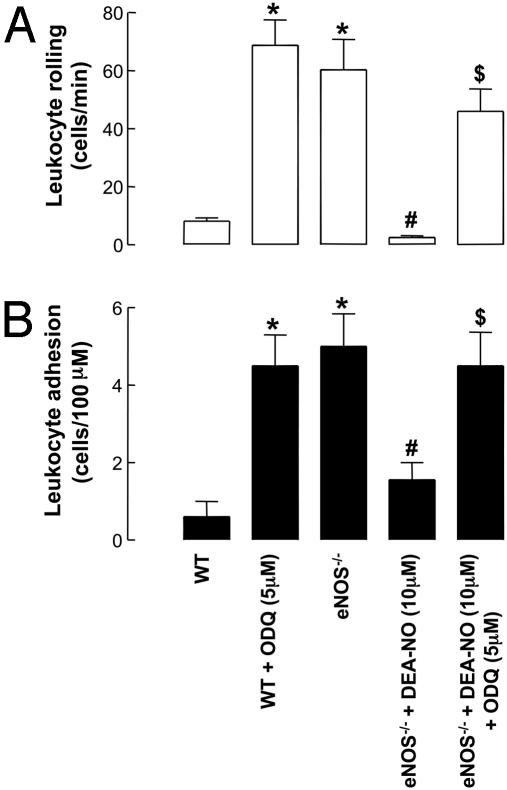
Fig. 2.
Leukocyte–endothelial cell interactions [rolling (A) and adhesion (B)] in mouse mesenteric postcapillary venules in vivo in WT and eNOS–/– mice in the absence and presence of DEA-NO (10 μM) and ODQ (5 μM). *, P < 0.05, significantly greater than WT animals; #, P < 0.05, significantly lower than eNOS–/– mice; $, P < 0.05, significantly greater than eNOS–/– mice with DEA-NO alone; n > 5.
Table 1. Hemodynamic parameters in the isolated postcapillary venules of WT and eNOS-/- mice.
| Venule diameter, μm | Centerline velocity, mm/s | Calculated wall shear rate, per s | |
|---|---|---|---|
| WT | 30.1 ± 2.6 | 2.2 ± 0.2 | 396.1 ± 50.8 |
| WT + ODQ | 33.7 ± 2.6 | 2.9 ± 0.4 | 428.2 ± 52.5 |
| eNOS-/- | 30.1 ± 2.1 | 2.4 ± 0.2 | 423.0 ± 53.1 |
| eNOS-/- + BAY (0.3 μM) | 29.7 ± 1.9 | 2.4 ± 0.2 | 374.0 ± 38.5 |
| eNOS-/- + BAY (1 μM) | 33.7 ± 1.9 | 2.5 ± 0.3 | 393.9 ± 44.5 |
| eNOS-/- + BAY + ODQ | 33.9 ± 2.2 | 2.4 ± 0.4 | 343.5 ± 35.0 |
| eNOS-/- + DEA-NO (1 μM) | 32.7 ± 2.5 | 2.5 ± 0.3 | 401.1 ± 37.8 |
| IL-1β (90 min) | 29.5 ± 3.7 | 1.9 ± 0.2 | 403.6 ± 23.1 |
| IL-1β (90 min) + BAY (1 μM) | 27.2 ± 1.2 | 1.9 ± 0.1 | 363.4 ± 22.5 |
| IL-1β (180 min) | 25.7 ± 2.1 | 1.9 ± 0.2 | 381.9 ± 31.2 |
| IL-1β (180 min) + BAY (1 μM) | 25.0 ± 2.0 | 1.9 ± 0.2 | 382.0 ± 27.4 |
n ≥ 7; P > 0.05 for all compared to WT control.
Effect of NO and cGMP on Leukocyte–Endothelial Cell Interactions. The NO-donor DEA-NO (1 μM) produced a significant inhibition of leukocyte rolling and adhesion in eNOS–/– animals (Fig. 2) that was reversed in the presence of ODQ (5 μM, Fig. 2). ODQ (5 μM) alone had no effect on leukocyte rolling and attachment in eNOS–/– animals (data not shown); however, in WT animals in the presence of ODQ (5 μM) the leukocyte rolling and adhesion were increased to levels equivalent to those observed in eNOS–/– mice (Fig. 2).
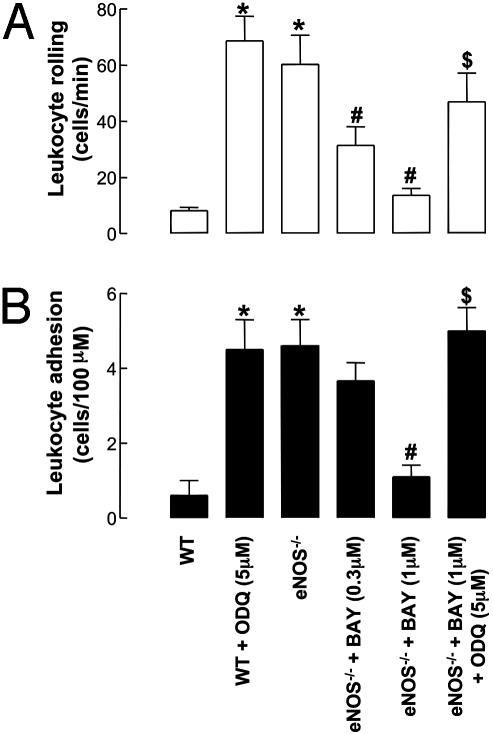
BAY 41–2272 (0.3–1 μM) decreased both leukocyte rolling and adhesion in eNOS–/– animals in a concentration-dependent manner, such that at 1 μM BAY 41–2272 the extent of each was equivalent to that observed in WT animals [Figs. 1 and 3; BAY 41–2272 had no significant effect on blood flow per se at these concentrations, as indicated by equivalent Vrbc values (data not shown)]. Moreover, in the presence of the sGC inhibitor ODQ (5 μM), the inhibitory effects of BAY 41–2272 on leukocyte rolling and attachment were reversed to levels comparable to untreated eNOS–/– mice (Fig. 3).
Fig. 3.
Leukocyte–endothelial cell interactions [rolling (A) and adhesion (B)] in mouse mesenteric postcapillary venules in vivo in WT and eNOS–/– mice in the absence and presence of BAY 41–2272 (0.3–1 μM) and ODQ (5 μM). *, P < 0.05, significantly greater than WT animals; #, P < 0.05, significantly lower than eNOS–/– mice; $, P < 0.05, significantly greater than eNOS–/– mice with BAY 41–2272 alone; n > 5.
Effect of NO and cGMP on IL-1β-Induced Leukocyte–Endothelial Cell Interactions. Treatment of WT animals with IL-1β (5 ng, i.p.) resulted in a significant increase in leukocyte rolling and adhesion at both an early (90 min) and late (180 min) time point after challenge compared to saline-injected controls (Fig. 4).
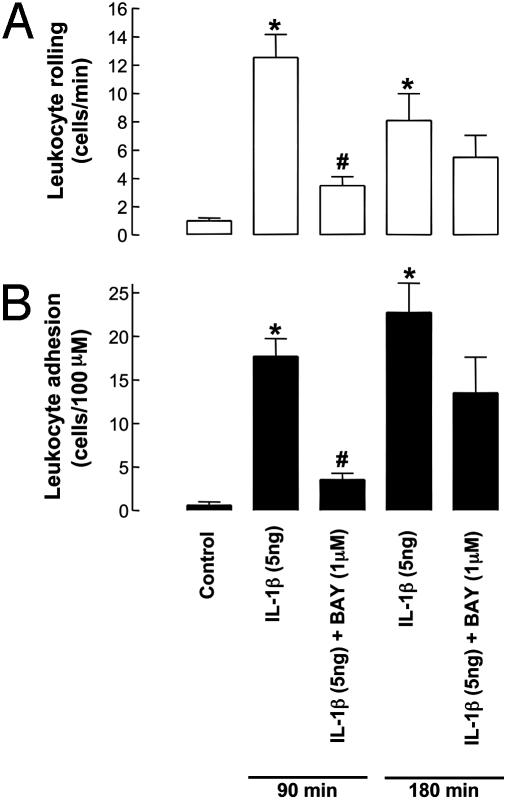
Fig. 4.
Leukocyte–endothelial cell interactions [rolling (A) and adhesion (B)] in mouse mesenteric postcapillary venules in vivo in WT mice in response to IL-1β (5 ng, 90- or 180-min exposure) in the absence and presence of BAY 41–2272 (1 μM). *, P < 0.05, significantly greater than control (saline-treated) animals; #, P < 0.05, significantly lower than IL-1β alone; n > 5.
After tissue superfusion of BAY 41–2272 (1 μM), the increased leukocyte rolling and adhesion in response to IL-1β was significantly reduced at 90 min after challenge (Fig. 4). However, leukocyte rolling and adhesion were unaffected by BAY 41–2272 3 h after IL-1β administration (Fig. 4).
Effect of cGMP on Adhesion Molecule Expression. To ascertain which adhesion molecule(s) cGMP might be influencing to bring about changes in leukocyte rolling and attachment, FACS analysis was conducted on HUVECs in culture activated with IL-1β (200 ng/ml) or histamine (100 μM) and leukocytes/platelets in whole blood exposed to thrombin (1 unit/ml) or TNF-α (20 ng/ml). Expression of P-selectin (CD62P), E-selectin (CD62E), L-selectin (CD62L), ICAM-1 (CD54), and β2 integrin (CD11b) was evaluated in the presence and absence of BAY 41–2272 (1 μM).
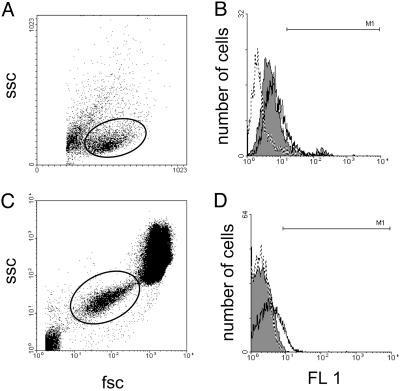
In the HUVECs, basal P-selectin expression was negligible, indicating that processing of the cells had not inadvertently caused activation (see refs. 17 and 18 for comparison). IL-1β (200 ng/ml) caused a significant increase in the expression of E-selectin, P-selectin, and ICAM-1 90 min after challenge (Fig. 5 and Tables 2 and 3). In the presence of BAY 41–2272 (1 μM), E-selectin and ICAM-1 expression remained unchanged; however, the expression of P-selectin was dramatically attenuated (Fig. 5 and Tables 2 and 3). In further experimentation, P-selectin expression was assessed in HUVECs after activation with histamine (100 μM), because the in vivo effects of IL-1β (used in the intravital studies) are mediated in part via mast cell degranulation and, consequently, histamine release (19, 20). In this case, histamine (100 μM) caused a dramatic increase in P-selectin expression that was significantly reversed in the presence of BAY 41–2272 (1 μM, Tables 2 and 3). A similar pattern was observed in whole blood. Expression of P-selectin was increased on platelets exposed to thrombin (1 unit/ml), and this was reversed in the presence of BAY 41–2272 (1 μM, Tables 2 and 3). TNF-α (20 ng/ml) induced a marked increase in leukocyte expression of L-selectin and CD11b; however, this was not altered in the presence of BAY 41–2272 (1 μM; L-selectin-positive cells, TNF-α alone 8.52 ± 0.41%, TNF-α + BAY 41–2272 9.21 ± 1.16%; CD11b-positive cells, TNF-α alone 46.15 ± 0.99, TNF-α + BAY 41–2272 47.08 ± 2.22; n ≥ 5, P > 0.05 for both).
Fig. 5.
FACS analysis of P-selectin expression on HUVECs and platelets. (A and C) Dot plot analysis of the scatter characteristics of HUVECs grown in culture (A) and whole blood (C). Solid lines represent gating on live cells (A) and platelets (C). (B and D) P-selectin expression on gated HUVECs (B) and platelets (D) as demonstrated by fluorescence histograms. Broken outline, control cells; solid outline, cells stimulated with IL-1β (200 ng/ml); filled outline, cells stimulated with IL-1β (200 ng/ml) and treated with BAY 41–2272 (1 μM).
Table 2. Percentage of HUVECs positive for specified adhesion molecule expression under control conditions and after exposure to an inflammatory stimulus in the presence and absence of BAY 41-2272 (1 μM).
| Adhesion molecule expressed | Control | + IL-1β (200 ng/ml) | + IL-1β (200 ng/ml) + BAY 41-2272 (1 μM) | + Histamine (100 μM) | + Histamine (100 μM) + BAY 41-2272 (1 μM) |
|---|---|---|---|---|---|
| P-selectin | 0.39 ± 0.15 | 3.90 ± 0.93* | 1.77 ± 0.17** | 12.28 ± 1.63* | 4.34 ± 0.95** |
| E-selectin | 5.89 ± 0.13 | 7.11 ± 0.32* | 6.69 ± 0.41 | ||
| ICAM-1 | 0.19 ± 0.17 | 90.09 ± 0.47* | 89.78 ± 1.92 |
, P < 0.05, significantly greater than control; **, P < 0.05, significantly lower than inflammatory stimulus alone; n > 5.
Table 3. Percentage of platelets positive for specified adhesion molecule expression under control conditions and after exposure to an inflammatory stimulus in the presence and absence of BAY 41-2272 (1 μM).
| Adhesion molecule expressed | Control | + Thrombin (1 unit/ml) | + Thrombin (1 unit/ml) + BAY 41-2272 (1 μM) |
|---|---|---|---|
| P-selectin | 0.28 ± 0.12 | 4.89 ± 0.28* | 1.19 ± 0.14** |
, P < 0.05, significantly greater than control; **, P < 0.05, significantly lower than inflammatory stimulus alone; n > 5.
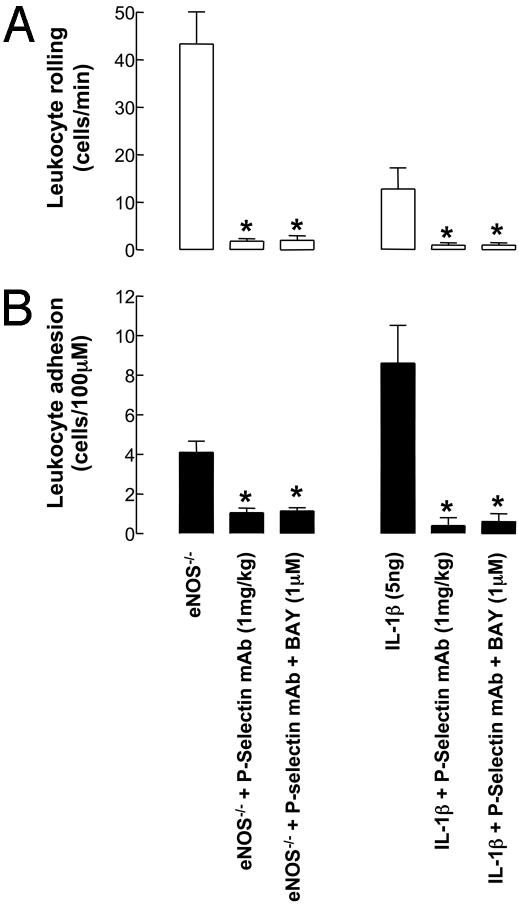
Effect of a P-Selectin mAb on Leukocyte–Endothelial Cell Interactions. To ascertain the importance of P-selectin in the process of leukocyte recruitment in mouse mesenteric postcapillary venules, and to link the in vitro findings (i.e., FACS analysis) with in vivo function, a neutralizing P-selectin monoclonal antibody was injected i.v. during microscopic observation. The increased rolling and adhesion observed in eNOS–/– mice (Fig. 2), and after administration of IL-1β (5 ng, i.p., 90 min), were significantly reversed in the presence of this antibody (Fig. 6), indicating that in vivo P-selectin plays a pivotal role in the process of leukocyte recruitment. Moreover, administration of BAY 41–2272 (1 μM) concomitantly with the P-selectin mAb did not produce an additional attenuation of leukocyte rolling or adhesion (Fig. 6); such an observation confirms that the antileukocyte effects of BAY 41–2272 in vivo require the presence of functional P-selectin expression.
Fig. 6.
Leukocyte–endothelial cell interactions [rolling (A) and adhesion (B)] in mouse mesenteric postcapillary venules in vivo in eNOS–/– mice and in WT mice in response to IL-1β (5 ng, 90-min exposure) in the absence and presence of a P-selectin neutralizing antibody (RB40.34, 1 mg/kg) and BAY 41–2272 (1 μM). *, P < 0.05, significantly different from control; n ≥ 4.
Discussion
NO exerts an essential physiological cytoprotective/antithrombotic influence on the blood vessel wall by preventing the adherence of immune cells and platelets. The mechanism underlying the antiplatelet activity of NO has been well characterized (6, 21). However, despite considerable attention, the mechanism by which NO modulates leukocyte adherence to the vascular endothelium remains obscure; in particular, it is not clear whether this process is cGMP-dependent or indeed which adhesion molecule(s) are involved in the altered adhesive characteristics. Studies in specific NOS KO animals have demonstrated the importance of NO as a leukocyte (7) and platelet (22) modulator and confirmed eNOS to be the primary source, although neuronal NOS (nNOS)-derived NO also appears to influence leukocyte reactivity under some circumstances (7, 8). In addition, immunohistological data suggest that P-selectin may be involved, at least in part, in the antileukocyte effect of NO (7). However, recent evidence suggests that P-selectin expression is reduced in eNOS–/– animals (23), and functional data oppose the view that this adhesion molecule is involved in the antileukocyte effects of NO (12). In the vasculature, activation of sGC underlies the majority of physiological actions of NO, mediating smooth muscle relaxation, increased capillary permeability, and inhibition of platelet aggregation and smooth muscle proliferation (24). The presence of sGC in the vascular endothelium has been reported (10) but has no defined role. In the present study, we used the non-NO-based sGC activator BAY 41–2272 (14, 25) to assess whether the antileukocyte and antiplatelet effects of NO are sGC-dependent and, if so, which adhesion molecule(s) are modulated by cGMP to bring about this effect. Moreover, we investigated whether sGC activation could reverse the leukocyte recruitment during an inflammatory episode in response to IL-1β.
To assess the role of cGMP in regulating leukocyte rolling and adhesion, these parameters were compared in WT and eNOS–/– mice. In animals deficient in eNOS, basal leukocyte rolling and adhesion were increased 6-fold compared to their WT counterparts, confirming an important antithrombotic/antiadhesive role for endothelial-derived NO (7). In accord with these observations [and previous publications (2, 7)], the NO-donor diethylamine-NONOate (DEA-NO) also reversed the proadhesive environment in vessels of eNOS–/– animals to a level equivalent to that observed in WT animals. The effects of NO were abolished in the presence of the sGC inhibitor ODQ. Importantly however, in the presence of ODQ, leukocyte rolling and adhesion were increased in WT mice to the elevated basal levels observed in eNOS–/– animals. This observation provides the first indication that the antileukocyte effects of NO are mediated predominantly via activation of sGC and, therefore, production of cGMP. This thesis was confirmed by observations with BAY 41–2272, which caused a concentration-dependent reversal of the increased leukocyte rolling and adhesion observed in eNOS–/– mice, such that in the presence of 1 μM BAY 41–2272 these parameters were identical to those observed in WT mice. Furthermore, the antileukocyte effect of BAY 41–2272 was absent in the presence of ODQ. Both of these findings suggest that sGC activation (and subsequent cGMP synthesis) accounts entirely for the antiadhesive actions of NO and assigns a previously uncharacterized antiinf lammatory role to this enzyme.
After the demonstration that sGC activation results in a profound antileukocyte activity, we investigated whether BAY 41–2272 could also prevent the leukocyte recruitment (in terms of rolling and adhesion) induced by an inflammatory stimulus, namely IL-1β. In WT animals, administration of IL-1β induced a significant increase in leukocyte rolling and adhesion 90 and 180 min after challenge. Administration of BAY 41–2272 elicited a marked inhibition of IL-1β-induced leukocyte rolling and adhesion at 90 min after IL-1β but not at the later time point. This differential activity may be explained by the temporal expression of different adhesion molecules during the inflammatory episode. Initial leukocyte–endothelial cell interactions are mediated by the selectin family of adhesion molecules. L-selectin is expressed constitutively on the leukocyte surface and is believed to play a pivotal role in the initial rolling of leukocytes along the endothelial layer; P- and E-selectins are expressed subsequently in a matter of minutes and hours, respectively, to aid this process (26–28). Firm adhesion of leukocytes to the endothelium depends predominantly on the expression of β2-integrin (CD11b/CD18) on the leukocyte surface and ICAM-1 on the endothelial cells (26, 29, 30). Therefore, the present findings that BAY 41–2272 inhibits leukocyte rolling and adhesion only at earlier time points after IL-1β administration, and that acutely both BAY 41–2272 and NO inhibit leukocyte rolling and adhesion to a similar extent, suggest that the mechanism underlying NO- and cGMP-mediated suppression of leukocyte adherence involves modulation of adhesion molecules expressed in the early stages on leukocyte recruitment, such as L- or P-selectin, and that the inhibition of leukocyte adhesion observed is simply a consequence of reduced rolling. In addition, the antileukocyte effects of BAY 41–2272 occur immediately after administration. Thus, the mechanism of action cannot be the result of decreased selectin mRNA and protein expression but rather a rapid reversal of the proadhesive activity of the relevant adhesion molecule. The mechanism(s) underlying this effect remains unclear and merits further attention.
To determine which adhesion molecule(s) may be important in the regulation of leukocyte adherence by cGMP, we used FACS analysis to assess expression of key adhesion molecules (as mentioned above) in the absence and presence of BAY 41–2272. These studies revealed that sGC activation induces a specific down-regulation of the expression of P-selectin (on both the endothelial cell surface and platelets), with no effect on any of the other adhesion molecules studied. To reconcile the observations made by intravital microscopy with the cell-based FACS analysis, a specific P-selectin neutralizing monoclonal antibody was used in vivo. P-selectin blockade using this reagent reversed almost entirely the increased leukocyte rolling and adhesion apparent in eNOS–/– and WT mice treated with IL-1β. Significantly, BAY 41–2272 was unable to cause an additional decrease in leukocyte rolling and adhesion when administered concomitantly with the P-selectin mAb. These observations confirm that P-selectin is up-regulated in vivo after blockade of endogenous NO synthesis and after exposure to the proinflammatory cytokine IL-1β, and is the predominant adhesion molecule responsible for the enhanced leukocyte recruitment under these conditions. Moreover, BAY 41–2272 does not exert an antileukocyte effect in vivo after P-selectin blockade, indicating that this compound (and therefore sGC/cGMP activation) prevents leukocyte rolling and adhesion via modulation of P-selectin expression. These observations fit with the ability of BAY 41–2272 to alter initial leukocyte rolling (and consequently adhesion), which depends on the expression of L- and P-selectin, but not at later time points after IL-1β challenge. At this later stage, rolling of cells progresses to firm attachment, which is no longer dependent on P-selectin but rather β2 integrin and ICAM-1. Moreover, the fact that BAY 41–2272 inhibits P-selectin expression on platelets (5) suggests that cGMP will also prevent platelet–endothelial cell and platelet–leukocyte interactions in the circulation, representing further important steps in the inflammatory process. Soluble GC activation also leads to a reversal of both IL-1β- and histamine-induced P-selectin expression. The in vivo actions of IL-1β are mediated in part via mast cell degranulation and histamine release (19, 20). Thus, the ability of BAY 41–2272 to reduce P-selectin expression in response to both inflammatory mediators provides further evidence that this is a common antiinflammatory mechanism governed by sGC.
The ability of BAY 41–2272 to inhibit P-selectin expression on platelets and endothelial cells indicates a previously uncharacterized role for sGC in regulating expression of adhesion molecules. Perhaps more importantly, it suggests that non-NO-based sGC activators may represent potent antiinflammatory agents that may be of considerable therapeutic benefit in the treatment of inflammatory cardiovascular disorders. The clinical use of NO-donor (or nitrovasodilator) drugs is problematic because of development of tolerance and the nonspecific interaction of NO with other biological molecules; these reactions are difficult to control because of the lipophilic, radical nature of NO (31). Thus, agents that activate sGC selectively, in a NO-independent manner, will offer a considerable therapeutic advance. In the cardiovascular system, the multifaceted actions of cGMP intimate that BAY 41–2272 and analogous compounds will potently reduce blood pressure, attenuate leukocyte and platelet activation and adherence, and inhibit muscle proliferation. Each of these actions represents a powerful antiatherogenic influence and in combination, as a result of the administration of non-NO-based sGC activators, may be of considerable benefit to individuals with atherosclerosis and other inflammatory vascular disorders.
In summary, the present study demonstrates that the antiadhesive properties of NO are mediated predominantly by the activation of sGC and the production of the second messenger cGMP. In turn, cGMP causes a specific down-regulation of the expression of P-selectin on endothelial cells and platelets to prevent leukocyte rolling, which has a knock-on effect on adhesion and emigration. The study therefore assigns a previously uncharacterized role to sGC/cGMP signaling in endothelial cells in regulating the activation and attachment of immune cells and promotes the concept that non-NO-based sGC activators are potent antiinflammatory agents and may represent a new treatment for inflammatory cardiovascular diseases.
Acknowledgments
This work was supported, in part, by an equipment grant from the Wellcome Trust. A.J.H. is the recipient of a Wellcome Trust Senior Fellowship in Basic Biomedical Sciences, P.F. is supported by The Special Trustees of St. Barts Hospital Joint Research Board, and M.P. is supported by the Arthritis Research Campaign.
This paper was submitted directly (Track II) to the PNAS office.
Abbreviations: eNOS, endothelial NOS; FACS, fluorescence-activated cell sorting; ICAM-1, intercellular adhesion molecule 1; iNOS, inducible NOS; KO, knockout; NOS, nitric oxide synthase; ODQ, 1H-(1,2,4)oxadiazolo(4,3-a)quinoxalin-1-one; sGC, soluble guanylate cyclase; TNF, tumor necrosis factor.
References
- 1.Granger, D. N. & Kubes, P. (1994) J. Leukocyte Biol. 55, 662–675. [PubMed] [Google Scholar]
- 2.Kubes, P., Suzuki, M. & Granger, D. N. (1991) Proc. Natl. Acad. Sci. USA 88, 4651–4655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gaboury, J., Woodman, R. C., Granger, D. N., Reinhardt, P. & Kubes, P. (1993) Am. J. Physiol. 265, H862–H867. [DOI] [PubMed] [Google Scholar]
- 4.Radomski, M. W., Palmer, R. M. & Moncada, S. (1987) Lancet 2, 1057–1058. [DOI] [PubMed] [Google Scholar]
- 5.Salas, E., Langford, E. J., Marrinan, M. T., Martin, J. F., Moncada, S. & de Belder, A. J. (1998) Heart 80, 146–150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Radomski, M. W., Palmer, R. M. & Moncada, S. (1987) Biochem. Biophys. Res. Commun. 148, 1482–1489. [DOI] [PubMed] [Google Scholar]
- 7.Lefer, D. J., Jones, S. P., Girod, W. G., Baines, A., Grisham, M. B., Cockrell, A. S., Huang, P. L. & Scalia, R. (1999) Am. J. Physiol. 276, H1943–H1950. [DOI] [PubMed] [Google Scholar]
- 8.Sanz, M. J., Hickey, M. J., Johnston, B., McCafferty, D. M., Raharjo, E., Huang, P. L. & Kubes, P. (2001) Br. J. Pharmacol. 134, 305–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hickey, M. J., Granger, D. N. & Kubes, P. (2001) Acta Physiol. Scand. 173, 119–126. [DOI] [PubMed] [Google Scholar]
- 10.Zabel, U., Kleinschnitz, C., Oh, P., Nedvetsky, P., Smolenski, A., Muller, H., Kronich, P., Kugler, P., Walter, U., Schnitzer, J. E., et al. (2002) Nat. Cell Biol. 4, 307–311. [DOI] [PubMed] [Google Scholar]
- 11.Liu, P., Xu, B., Hock, C. E., Nagele, R., Sun, F. F. & Wong, P. Y. (1998) Am. J. Physiol. 275, H2191–H2198. [DOI] [PubMed] [Google Scholar]
- 12.Kubes, P., Kurose, I. & Granger, D. N. (1994) Am. J. Physiol. 267, H931–H937. [DOI] [PubMed] [Google Scholar]
- 13.Armstead, V. E., Minchenko, A. G., Schuhl, R. A., Hayward, R., Nossuli, T. O. & Lefer, A. M. (1997) Am. J. Physiol. 273, H740–H746. [DOI] [PubMed] [Google Scholar]
- 14.Stasch, J. P., Becker, E. M., Alonso-Alija, C., Apeler, H., Dembowsky, K., Feurer, A., Gerzer, R., Minuth, T., Perzborn, E., Pleiss, U., et al. (2001) Nature 410, 212–215. [DOI] [PubMed] [Google Scholar]
- 15.Bienvenu, K. & Granger, D. N. (1993) Am. J. Physiol. 264, H1504–H1508. [DOI] [PubMed] [Google Scholar]
- 16.Lim, L. H., Solito, E., Russo-Marie, F., Flower, R. J. & Perretti, M. (1998) Proc. Natl. Acad. Sci. USA 95, 14535–14539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Torres, R., de Castellarnau, C., Ferrer, L. L., Puigdemont, A., Santamaria, L. F. & de Mora, F. (2002) Immunol. Cell Biol. 80, 170–177. [DOI] [PubMed] [Google Scholar]
- 18.Easton, A. S. & Dorovini-Zis, K. (2001) Microvasc. Res. 62, 335–345. [DOI] [PubMed] [Google Scholar]
- 19.Subramanian, N. & Bray, M. A. (1987) J. Immunol. 138, 271–275. [PubMed] [Google Scholar]
- 20.McLean, P. G., Ahluwalia, A. & Perretti, M. (2000) J. Exp. Med. 192, 367–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Langford, E. J., Brown, A. S., Wainwright, R. J., de Belder, A. J., Thomas, M. R., Smith, R. E., Radomski, M. W., Martin, J. F. & Moncada, S. (1994) Lancet 344, 1458–1460. [DOI] [PubMed] [Google Scholar]
- 22.Freedman, J. E., Sauter, R., Battinelli, E. M., Ault, K., Knowles, C., Huang, P. L. & Loscalzo, J. (1999) Circ. Res. 84, 1416–1421. [DOI] [PubMed] [Google Scholar]
- 23.Prorock, A. J., Hafezi-Moghadam, A., Laubach, V. E., Liao, J. K. & Ley, K. (2003) Am. J. Physiol. 284, H133–H140. [DOI] [PubMed] [Google Scholar]
- 24.Hobbs, A. J. (1997) Trends Pharmacol. Sci. 18, 484–491. [DOI] [PubMed] [Google Scholar]
- 25.Becker, E. M., Alonso-Alija, C., Apeler, H., Gerzer, R., Minuth, T., Pleibeta, U., Schmidt, P., Schramm, M., Schroder, H., Schroeder, W., et al. (2001) BMC Pharmacol. 1, 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bevilacqua, M. P. & Nelson, R. M. (1993) Thromb. Haemostasis 70, 152–154. [PubMed] [Google Scholar]
- 27.Springer, T. A. (1994) Cell 76, 301–314. [DOI] [PubMed] [Google Scholar]
- 28.Tedder, T. F., Steeber, D. A., Chen, A. & Engel, P. (1995) FASEB J. 9, 866–873. [PubMed] [Google Scholar]
- 29.Kubes, P. & Granger, D. N. (1996) Cardiovasc. Res. 32, 699–708. [PubMed] [Google Scholar]
- 30.von Andrian, U. H., Chambers, J. D., McEvoy, L. M., Bargatze, R. F., Arfors, K. E. & Butcher, E. C. (1991) Proc. Natl. Acad. Sci. USA 88, 7538–7542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hobbs, A. J. (2002) Br. J. Pharmacol. 136, 637–640. [DOI] [PMC free article] [PubMed] [Google Scholar]