Abstract
The existence of allogeneic cells within an individual has been demonstrated in multiple fields such as hematopoietic stem cell or solid organ transplantation, non-depleted blood transfusions and the most common form which is bidirectional maternal-fetal cell trafficking, whereby cells from the fetus pass through the placental barrier. In order to graphically illustrate this early natural phenomenon that initiates the journey of a child’s cells within the mother’s blood and other tissues, we used a new procedure in microscopy imaging generating Large Scale Panoramic Pictures (LSPP). This technique can also be extended to explore a broad diversity of experimental models.
Keywords: fetal-maternal interface, fish, microchimerism, placenta, tolerance
Introduction
Chimerism is the presence in a host of genetically distinct cells that originated from another individual.1 Its existence has clearly been demonstrated in multiple fields such as hematopoietic stem cell2 or solid organ transplantation,3 non-depleted blood transfusions4 and the most common form which is bidirectional maternal-fetal cell trafficking whereby cells from the fetus pass through the placenta and establish durable cell lineages within the mother. By delving a little deeper into this last topic, one can notice that normal fetal development directly correlates with previous and concomitant growth of the placenta. Placental development starts with trophoblast differentiation from trophoectoderm, a key factor for implantation (which begins from the sixth day after fertilization).5
Extravillous cytotrophoblastic cells literally invade the decidua basalis to form the fetal-maternal interface. Trophoblastic-derived cells enzymatically digest the maternal endometrium and even myometrium in a hypoxia to normoxia gradient,5 leading to constrained bleeding that forms numerous blood-filled lacunae. Extravillous infiltrating trophoblasts actively remodel maternal spiral afferent arteries resulting in a significant increase of vessel diameter (up to 10-fold) and a reduction of blood flow pressure, in order to assure enough blood provision for ongoing fetal requirements.6 Villous trophoblasts are responsible for chorionic villi formation, which augment the surface of contact allowing for an extensive and controlled nutrient and gas exchange between oxygen’s high-avidity fetal erythrocytes and maternal blood in the intervillous space. Taking into account all the above-mentioned characteristics, it is reasonable to conclude that allogeneic cells are in very close contact during the fetal development process. Many of the immunological hypo-reactivity mechanisms orchestrated in part by trophoblast and its derived cells have been described elsewhere,7,8 even though this tolerant state must live with a certain degree of inflammatory responses, thus enabling both blood flow increase and trophoblast invasion. The maintenance of this acquired tolerance in both the mother and the progeny has been studied, and persistent microchimerism is common9 in the context of autoimmune diseases10,11 and even in tissue regeneration.12
Our previous experimental background on the use of different stem cell sources13,14 has focused on studying immunological reactivity of newly implanted allogeneic cells used to regenerate cardiac damaged tissue. In order to graphically illustrate an early natural allogeneic contact that initiates the journey of a child’s cells (in this case a son’s cells) within the mother’s blood and other tissues, we used a new procedure in microscopy imaging generating large scale panoramic pictures (LSPP). Unique aspects of the LSPP technique are that it provides high definition pictures covering large areas and permits a global contextualization of targeted zones or cells.
Results and Discussion
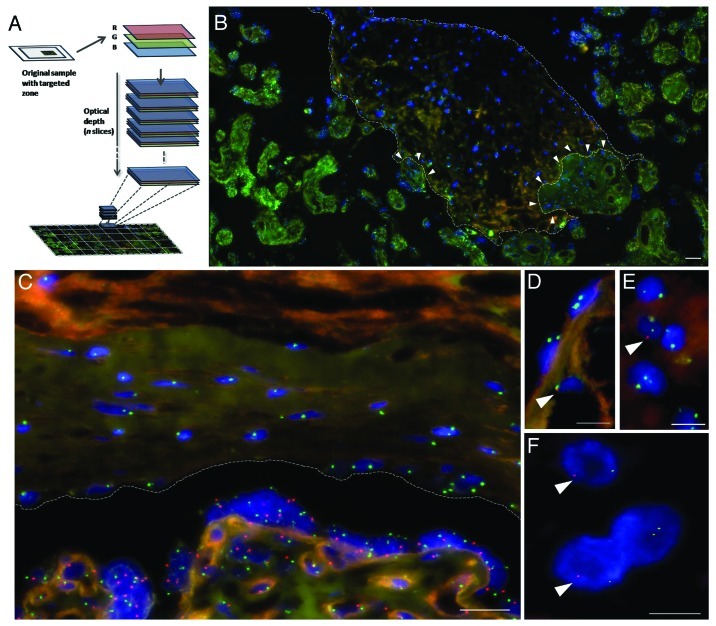
Using this novel imaging procedure, we composed an LSPP (Fig. 1A) covering a macroscopic area (rounding 1 mm2) of the maternal decidua basalis surrounded by invasive fetal villi (Fig. 1B). Surprisingly, autofluorescence of extra-cellular matrix components within red and green channels helped in imaging global tissue structures without interfering with expected FISH probes fluorescence. As expected, the decidua contained maternal cells organized within the endometrial tissue (XX, nuclei showing two green dots) surrounded by fetal structures (cells containing one green “X” dot and one red “Y” dot within the same nucleus in Fig. 1B and C), suggesting a maternal micro-septum (Fig. 1B). It is also frequent to find in the placenta, fetal-derived structures infiltrating maternal tissues as shown with white arrows in Figure 1B, and even at the cell level as shown in Figure 1D and E. Figure 1F illustrates the presence of two distinct adjacent allogeneic cells from the mother and the baby at the fetal-maternal interface. After observing the exposed LSPP, it is reasonable to consider that fetal cells can easily be transferred and circulate within maternal blood either because cells can migrate from villi or simply due to infiltrating cells. The question of how immunogenic processes of alloresponse have been altered can consequently be addressed. In other words, a deeper understanding of this “nonaggression pact” at the fetal-maternal interface, in which two genetically distinct cell lineages coexist, may improve the options to mimic this procedure in transplantation, autoimmunity and regenerative medicine using allogeneic sources.
Figure 1. (A) LSPP were constructed using MosaiX (authorizing 10% of overlapping), Zstacking with focus correction for each quadrant, Stitching frames algorithm and Extended Focus functions from AxioVision v4.8.2 software. (B) Example of a resulting LSPP of the decidua basalis of a human placenta covering an area of 0.78 mm2 with a resolution of 30.5 megapixels. Decidua basalis was parallel to the sectioning surface. The fetal-maternal interface is labeled with PNA-X (Spectrum Green, green) and PNA-Y (Spectrum Orange, red) chromosome-specific probes. FISH was used for sex-typing and Dapi for nuclear staining (blue). The dotted line-delimited zone is the maternal decidua. White arrows show direct invasion of fetal structures into maternal tissue. (C) Common natural maternal cell (XX) distribution within tissues of the decidua. (D and E) Dispersed infiltrating fetal-derived cells (XY) neighboring maternal cells. (F) Two adjacent cells of maternal and fetal origin. Scale bars: (A) 50 µm; (C) 20 µm; (D–F) 10 µm.
In summary, here we visualize the classic concept of chimerism by generating newly panoramic images of human placental sections following simple dual fluorescence in situ hybridization labeling to co-localize male fetal and maternal cells. Since this process generates vast amounts of data requiring advanced computer technology, we believe that generation of LSPP may be advantageous to map and explore large extensions of tissue. Thus, this novel technique may be extended to a broad diversity of experimental models, such as the visualization of large post-infarct border zones following implantation of regenerative stem-cell embedded patches.
Methods
Placenta collection
Written consent was obtained from each participant after an explanation of the study, which was previously approved by the local ethics committee. All participants had no previous pregnancy to ensure no confounding due to cells from previous gestations. Approaching term but prior to the onset of labor, placentas of male sons were collected in sterile conditions during routine C-sections. Vaginal birth was not chosen for study to obviate the risk of contamination due to passage within the vaginal tract. All enrolled donors had no complications during pregnancy or C-section.
FISH staining
Initial material was obtained from a transversal section of 10 mm2 quadrant base of a caesarean placenta initiated from the chorionic plate down to decidua materna, thus including amnios, chorionic plate and vessels, fetal villi expansions and decidua materna. Segments were fixed with 10% formalin, then included in paraffin-embedded blocks and 4 µm sections were used for fluorescence in situ hybridization (FISH) using specific probes (Dako) labeled with Spectrum Green and Spectrum Orange for X and Y chromosome respectively, according to manufacturer’s recommendation. Tissue sections were also counterstained with 4',6-diamidino-2phenylindole (DAPI) (Vysis).
Image capture and processing
All FISH stained samples were analyzed under an Axio Observer Z1 fluorescence microscope (Zeiss). LSPP methodology included first, area localization by MosaiX module and subsequent focus correction for each determined quadrant with a 10% of image overlapping. For each quadrant, the number of depth slices was defined using Zstacking module. All quadrants were then joined with Stitching overlapping algorithm. Finally, obtained frames were consecutively organized in one single picture with the Extended Focus module. All modules used are contained in Zeiss AxioVision v4.8.2 software.
Acknowledgments
The authors are grateful to the members of the Gynecology and Obstetrics Department of the University Hospital Germans Trias i Pujol and V. Guirao for the priceless contributions in fluorescence microscopy imaging. We also thank the invaluable advice received during revision. A.B.-G. has full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Footnotes
Previously published online: www.landesbioscience.com/journals/chimerism/article/19439
References
- 1.Starzl TE, Zinkernagel RM. Antigen localization and migration in immunity and tolerance. N Engl J Med. 1998;339:1905–13. doi: 10.1056/NEJM199812243392607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ildstad ST, Sachs DH. Reconstitution with syngeneic plus allogeneic or xenogeneic bone marrow leads to specific acceptance of allografts or xenografts. Nature. 1984;307:168–70. doi: 10.1038/307168a0. [DOI] [PubMed] [Google Scholar]
- 3.Pujal JM. Should microchimerism turn into rejection prophylactics? Expert Rev Mol Diagn. 2010;10:107–18. doi: 10.1586/erm.09.79. [DOI] [PubMed] [Google Scholar]
- 4.Reed W, Lee TH, Norris PJ, Utter GH, Busch MP. Transfusion-associated microchimerism: a new complication of blood transfusions in severely injured patients. Semin Hematol. 2007;44:24–31. doi: 10.1053/j.seminhematol.2006.09.012. [DOI] [PubMed] [Google Scholar]
- 5.Red-Horse K, Zhou Y, Genbacev O, Prakobphol A, Foulk R, McMaster M, et al. Trophoblast differentiation during embryo implantation and formation of the maternal-fetal interface. J Clin Invest. 2004;114:744–54. doi: 10.1172/JCI22991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Whitley GS, Cartwright JE. Trophoblast-mediated spiral artery remodelling: a role for apoptosis. J Anat. 2009;215:21–6. doi: 10.1111/j.1469-7580.2008.01039.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Scherjon S, Lashley L, van der Hoorn ML, Claas F. Fetus specific T cell modulation during fertilization, implantation and pregnancy. Placenta. 2011;32(Suppl 4):S291–7. doi: 10.1016/j.placenta.2011.03.014. [DOI] [PubMed] [Google Scholar]
- 8.Warning JC, McCracken SA, Morris JM. A balancing act: mechanisms by which the fetus avoids rejection by the maternal immune system. Reproduction. 2011;141:715–24. doi: 10.1530/REP-10-0360. [DOI] [PubMed] [Google Scholar]
- 9.Drexler C, Wagner T. Blood group chimerism. Curr Opin Hematol. 2006;13:484–9. doi: 10.1097/01.moh.0000245690.54956.f3. [DOI] [PubMed] [Google Scholar]
- 10.Adams KM, Nelson JL. Microchimerism: an investigative frontier in autoimmunity and transplantation. JAMA. 2004;291:1127–31. doi: 10.1001/jama.291.9.1127. [DOI] [PubMed] [Google Scholar]
- 11.Sarkar K, Miller FW. Possible roles and determinants of microchimerism in autoimmune and other disorders. Autoimmun Rev. 2004;3:454–63. doi: 10.1016/j.autrev.2004.06.004. [DOI] [PubMed] [Google Scholar]
- 12.Bayes-Genis A, Roura S, Prat-Vidal C, Farre´ J, Soler-Botija C, de Luna AB, et al. Chimerism and microchimerism of the human heart: evidence for cardiac regeneration. Nat Clin Pract Cardiovasc Med. 2007;4(Suppl 1):S40–5. doi: 10.1038/ncpcardio0748. [DOI] [PubMed] [Google Scholar]
- 13.Roura S, Farre´ J, Hove-Madsen L, Prat-Vidal C, Soler-Botija C, G´lvez-Monto´n C, et al. Exposure to cardiomyogenic stimuli fails to transdifferentiate human umbilical cord blood-derived mesenchymal stem cells. Basic Res Cardiol. 2010;105:419–30. doi: 10.1007/s00395-009-0081-8. [DOI] [PubMed] [Google Scholar]
- 14.Bayes-Genis A, Soler-Botija C, Farre´ J, Sepu´lveda P, Raya A, Roura S, et al. Human progenitor cells derived from cardiac adipose tissue ameliorate myocardial infarction in rodents. J Mol Cell Cardiol. 2010;49:771–80. doi: 10.1016/j.yjmcc.2010.08.010. [DOI] [PubMed] [Google Scholar]