Abstract
In this paper we report the design, testing and use of a scannerless probe specifically for minimally invasive imaging of deep tissue in vivo with an epi-fluorescence modality. The probe images a 500 μm diameter field of view through a 710 μm outer diameter probe with a maximum tissue penetration depth of 15 mm specifically configured for eGFP imaging. Example results are given from imaging the pituitary gland of rats and zebrafish hearts with lateral resolution of 2.5 μm.
OCIS codes: (180.2520) Fluorescence microscopy, (170.2150) Endoscopic imaging
1. Introduction
With the advent of genetic manipulation and specifically the introduction of green fluorescent protein into routine biological investigation the requirement for in vivo imaging has grown. In addition, the ability to express luciferase into an animal means that bioluminescence can be used for observation, reducing the requirement for excitation light [1]. Through the use of in vivo imaging, the life science researcher is able to watch one sample for an extended period of time rather than sacrifice several animals at different time periods. Not only does this reduce the number of animals used, it also improves the observations, as a full time series can be followed on a single subject. However, this transfers the onus onto the optical expert to design instruments that are small enough for minimally invasive procedures, and in the long term suitable for placement into the animal to enable imaging of freely moving animals. Previous work in this area has centered upon beam scanning systems and there are numerous variations in the literature, frequently based upon beam scanned implementations of confocal or multiphoton microscopy or OCT [2–5] Limitations on the sizes of the miniature dichroic optics and the beam scanners tend to produce a probe with an outer diameter approximately 5–7 times larger than the field of view of the device. For a probe with a wide field of view such as the 425 μm of Bao [3] this leads to an outer diameter of the probe of 3.3 mm, which is very large for deep tissue imaging such as our pituitary study. Micro-endoscopes have been produced with a comparable outside diameter to ours [4] but their field of view is limited to perhaps 200 μm. Such point scanning systems, at least in the short to medium term, are all going to require at least a fiber optic connection to a femtosecond laser and power for the scanning system and it is also hard to imagine packaging the laser source and photon counting detectors required for such a scanned process into an implantable, wireless package.
For deep tissue imaging it is essential to maintain a small probe diameter in order to minimize invasive damage. We present a system that delivers a field of view comparable to the probe’s diameter. To achieve this our system employs a GRIN relay lens for both the delivery of fluorescence excitation light and the recovery of fluorescence emission light. Previously reported systems designed for deep tissue penetration employ a GRIN objective lens that is a short, 0.25 pitch optic with high NA, to form a pupil within the subject. This lens is then combined further optics to reform an image outside the subject. These further optics may be either another 0.25 pitch lens with a significantly longer focal length and lower NA [6], or with a GRIN relay lens—that is, a long, 1.0 pitch optic with low NA, followed by an imaging lens [7,8]. In these systems, the low NA of the second lens acts to reduce the field of view significantly compared to the diameter of the probe. In our system we utilize the relay lens as an image plane relay for the collection of emitted fluorescence light, rather than as a pupil plane relay. In this configuration, the necessarily low NA of the relay lens results in a low NA for the collection of fluorescence light, instead of a greatly reduced field of view as in other systems.
Our system has been designed with the long-term goal of implanting in the animal for extended periods of time using our GRIN probe, an LED illumination source, micro-optics and a miniature CMOS camera. Miniaturized designs for the imaging component of such a system have been demonstrated [9]. The optical sectioning capability has been removed but this may be possible in the longer term through the use of structured illumination based upon micro-LED array technology [10]. The initial system described in this paper utilizes a stereo-tactic system for the placement of the probe and currently uses a large camera for studies of the pituitary gland for extended studies of prolactin expression [11]. To our knowledge this is the first reported use of extended pitch GRIN optics for in vivo widefield imaging with sub-cellular resolution and a novel method of utilizing the GRIN lens for the illumination and imaging is presented.
2. Instrumentation
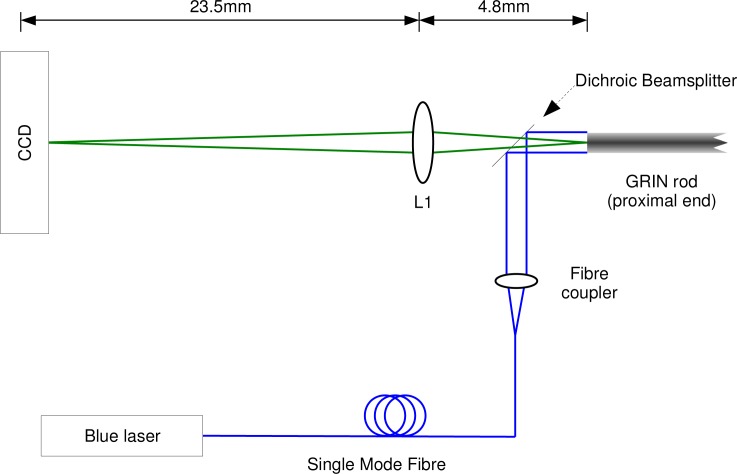
The basic optical configuration is shown in Fig. 1 . Light from a frequency-doubled diode pumped laser operating at 473 nm (Iforlight Ltd, UK) was coupled into a single mode optical fiber (Thorlabs P1-405A-FC-2). The output from the fiber was then collimated and directed onto the GRIN lens using a dichroic beamsplitter (Thorlabs MD498, cut to 3 mm square). The light then enters the single pitch GRIN relay rod, 0.5 mm in diameter and 30 mm long (GoFoton LRL-050-P100-0-2-0-2). The excitation light then exits the “distal” end of the rod through a 125 μm thick coverslip, which seals the end of the assembly against moisture ingress. This is placed in contact with the tissue to be imaged. For the emission path, the fluorescence image reformed on the proximal end of the probe is reimaged onto a CCD sensor by an aspheric lens, diameter 6.5 mm and focal length 6.24 mm AR coated for 400-600 nm (Thorlabs 352110-A). The lens was placed 4.8 mm from the remote end of the GRIN rod, casting an image onto a CCD sensor (QImaging Retiga 1300) 23.5 mm beyond the lens with a magnification of 3.0 times giving a calculated image scale of 2.2 μm/pixel. By bringing the GRIN optic closer to L1 it is possible to focus beyond the end of the GRIN optic, which is necessary to focus beyond the sealing coverslip, and can optionally allow imaging through some depth of tissue. Prior to surgical use the position of the GRIN rod was refocused experimentally.
Fig. 1.
Optical path of the instrument. 405 nm light is delivered to the probe by a single mode fiber. This light is expanded into a collimated beam by a fiber coupler. The central 0.5 mm of this beam is incident upon the proximal end of the GRIN rod. This light emerges as a collimated beam at the distal end (not shown), where it causes fluorescence of biomarkers. The GRIN rod reimages this fluorescent light to its proximal end. The proximal end of the rod is reimaged onto a detector by lens L1. A dichroic beamsplitter separates excitation and emission light.
The large ratio of pitch length to diameter, characteristic of a relay rod, leads to a low NA of approximately 0.1 limiting the quantity of emission light that we may collect through the rod, and placing constraints upon the delivery of widefield fluorescence illumination. The simplest way to ensure uniform illumination across the full field of view at the distal end of the probe was to use the GRIN rod as a pupil plane relay—that is, the remote face is illuminated with a collimated laser beam, which comes to a focus in the center of the rod and is then re-collimated upon the distal face. In this novel configuration, the GRIN lens was used as a pupil plane relay for excitation light and an image plane relay for emission light. The output from the fiber coupler was measured at 380 μW, with 34 μW being measured from the GRIN rod. We attribute the majority of the losses due to the coupler having to produce a beam with significantly larger diameter than the GRIN rod, as this helped with system alignment and mechanical stability.
Considering the small size of the optics and the spacing between them a custom mechanical design was used to house them. The rod was assembled inside a 0.71 mm outer diameter surgical grade stainless steel needle for mechanical protection with a 125 μm thick cover glass over the distal end of the probe as water and body fluid ingression is known to damage GRIN optics. For mechanical protection against bending, all but the final surgically inserted 15 mm of the assembly were encased in a second stainless steel tube, 1.0 mm OD. This was primarily to prevent accidental bending of the probe, which may snap the GRIN rod. 15 mm of protected GRIN rod protrudes from the mechanical mount, setting the maximum permissible tissue penetration depth.
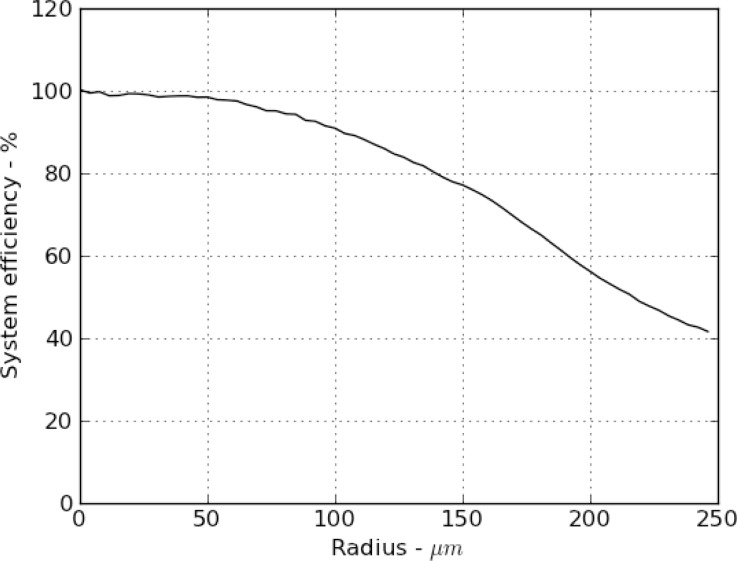
Fluorescence detection response of the probe (field flatness) was calibrated by illuminating a uniform, thin strip of fluorescent plastic at the distal end of the probe. This sample was illuminated via the GRIN rod, and excitation light collected and imaged through the probe. Figure 2 shows a radial cross section of the measured intensity profile. This curve has been normalized by the laser intensity profile measured at the entrance aperture of the GRIN rod, as the fiber collimator delivers a Gaussian beam profile. Insufficient data on the relay lens was available to perform a detailed ray-tracing simulation for comparison. The efficiency drops to 50% at a radius of approximately 225 μm due to vignetting, equivalent to a circle encompassing 80% of the imaging area.
Fig. 2.
Measured efficiency of the widefield probe as a function of radius from the center of the field of view. This is the combined effect of vignetting of the GRIN rod on the pupil-plane relay of illumination light and image-plane relay of collected fluorescence light.
3. Experiments
The initial measurements were taken on pollen grains (cultivated Rye grass pollen, Sigma-Aldrich P9145-1G) using their broadband autofluorescence, Fig. 3(a) , to ensure that the system was operating correctly before the use of animals. Following the procedure of Zucker [12] and Inoue [13] using a single grain as an extended emitter with a sharp edge a lateral intensity profile was recorded and differentiated to give a full width half maximum lateral resolution of 3 μm. This is in line with the manufacturers quoted specification of 200 line pairs/mm for the GRIN optic. The system is not designed for optically sectioned imaging and the quality of extended depth fluorescent samples was similar to that seen from a conventional wide field microscope with a similar NA objective lens. The sample we were imaging in vivo was not expected to have a great depth of fluorescing cells and thus optical sectioning was not crucial for this application.
Fig. 3.
Example images taken with the epi-fluorescence GRIN probe. (a) Pollen grains on a test sample visible through auto-fluorescence (b) Ex vivo image showing GFP expression in the myocardium of an adult Tg(cmlc2:GFP) transgenic zebrafish heart. (c) In vivo image of individual cells expressing EGFP as a reporter gene for prolactin on the surface of the pituitary gland in a rat model. It appears that the probe has been placed towards the edge of the gland, covering approximately one quadrant of the field of view.
The probe was tested in vivo with PRL-d2EGFP transgenic rat1 that expresses destabilized EGFP under the control of the human prolactin promoter in the pituitary gland. This study was undertaken at the Queen's Medical Research Institute, Edinburgh, under a UK Home Office License, following review by the local ethics committee. All rats were housed individually, given free access to water and standard commercial rat chow (Special Diet Service, Witham), and maintained under controlled conditions of temperature (21°C) and humidity, under a 12 h light/dark cycle.
Adult female rats weighting approximately 200 g were anaesthetized with Isoflurane (Iso) (Merial, Essex) (induction 4%, surgery 2–2.5% and imaging 1.75%), with the body temperature being measured using a rectal probe. The top of the head was shaved and the rat was placed in a stereotaxis apparatus [14]. The surface of the skull was exposed by incising and retracting the overlaying skin and fascia. A surgical opening was then drilled through the skull at coordinates X = −1.0 mm and Y = −5.8 mm, that localize the target pituitary according to the rat’s brain atlas [15]. The probe was then lowered through the brain towards the target depth (Z = −9.8 mm). Images from the camera were observed, with out-of-focus light from the pituitary cells becoming visible approximately 1 mm above the gland. The probe was then gradually lowered, bringing the images into sharp focus. An example image of the fluorescing cells in the pituitary gland can be seen in Fig. 3(c). A cluster of excited cells can be seen in the top part of the image. The camera exposure was 2s. The expressing cells have previously been shown to been found only near the surface of the gland [1] and this is in line with our images. Figure 3(b) shows an image of the heart of a Tg(cmlc2:GFP) transgenic zebrafish that express green fluorescent protein (GFP) exclusively in the myocardium [16]. This image illustrates the use of the probe on an ex vivo sample.
4. Discussion
The imaging probe developed has been used to obtain in vivo images of GFP expression within the pituitary gland of a transgenic rat. Very low levels of excitation illumination were used to minimize any potential photoxic damage deeply within the animal. In this system the probe was suitable for reuse. With this proof of concept demonstration system we have shown that in vivo imaging is possible using low excitation levels with a reasonable field of view with no scanning. Recent advances in camera miniaturization, for example sub-mm sized sensors from Awaiba GmbH, which when integrated with micro LED illumination and miniature diamond turned optics mean that a fully in vivo camera based imaging system is now possible with wireless connection to a receiving station either being made via optical or RF communication systems, the former having the advantage of potentially lower power consumption. We believe that this approach to in vivo imaging adds a further tool to the life scientist wishing to undertake in vivo experimentation with the aim of reducing animal usage.
Acknowledgments
This work was supported by a British Heart Foundation Research Excellence award to Edinburgh University and a Durham Biophysical Sciences Institute summer bursery.
References and links
- 1.Semprini S. S., Friedrichsen S., Harper C. V., McNeilly J. R., Adamson A. D., Spiller D. G., Kotelevtseva N., Brooker G., Brownstein D. G., McNeilly A. S., White M. R., Davis J. R., Mullins J. J., “Real-time visualization of human prolactin alternate promoter usage in vivo using a double-transgenic rat model,” Mol. Endocrinol. 23(4), 529–538 (2009). 10.1210/me.2008-0399 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jung J. C., Schnitzer M. J., “Multiphoton endoscopy,” Opt. Lett. 28(11), 902–904 (2003). 10.1364/OL.28.000902 [DOI] [PubMed] [Google Scholar]
- 3.Bao H., Allen J., Pattie R., Vance R., Gu M., “Fast handheld two-photon fluorescence microendoscope with a 475 μm × 475 μm field of view for in vivo imaging,” Opt. Lett. 33(12), 1333–1335 (2008). 10.1364/OL.33.001333 [DOI] [PubMed] [Google Scholar]
- 4.Engelbrecht C. J., Johnston R. S., Seibel E. J., Helmchen F., “Ultra-compact fiber-optic two-photon microscope for functional fluorescence imaging in vivo,” Opt. Express 16(8), 5556–5564 (2008). 10.1364/OE.16.005556 [DOI] [PubMed] [Google Scholar]
- 5.Sun J., Guo S., Wu L., Liu L., Choe S. W., Sorg B. S., Xie H., “3D in vivo optical coherence tomography based on a low-voltage, large-scan-range 2D MEMS mirror,” Opt. Express 18(12), 12065–12075 (2010). 10.1364/OE.18.012065 [DOI] [PubMed] [Google Scholar]
- 6.Knittel J., Schnieder L., Buess G., Messerschmidt B., Possner T., “Endoscope-compatible confocal microscope using a gradient index-lens system,” Opt. Commun. 188(5–6), 267–273 (2001). 10.1016/S0030-4018(00)01164-0 [DOI] [Google Scholar]
- 7.Levene M. J., Dombeck D. A., Kasischke K. A., Molloy R. P., Webb W. W., “In vivo multiphoton microscopy of deep brain tissue,” J. Neurophysiol. 91(4), 1908–1912 (2004). 10.1152/jn.01007.2003 [DOI] [PubMed] [Google Scholar]
- 8.Li X., Yu W., “Deep tissue microscopic imaging of the kidney with a gradient-index lens system,” Opt. Commun. 281(7), 1833–1840 (2008). 10.1016/j.optcom.2007.08.074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ghosh K. K., Burns L. D., Cocker E. D., Nimmerjahn A., Ziv Y., Gamal A. E., Schnitzer M. J., “Miniaturized integration of a fluorescence microscope,” Nat. Methods 8(10), 871–878 (2011). 10.1038/nmeth.1694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Poher V., Zhang H. X., Kennedy G. T., Griffin C., Oddos S., Gu E., Elson D. S., Girkin M., French P. M. W., Dawson M. D., Neil M. A. A., “Optical sectioning microscopes with no moving parts using a micro-stripe array light emitting diode,” Opt. Express 15(18), 11196–11206 (2007). 10.1364/OE.15.011196 [DOI] [PubMed] [Google Scholar]
- 11.Harper C. V., Featherstone K., Semprini S., Friedrichsen S., McNeilly J., Paszek P., Spiller D. G., McNeilly A. S., Mullins J. J., Davis J. R., White M. R., “Dynamic organisation of prolactin gene expression in living pituitary tissue,” J. Cell Sci. 123(3), 424–430 (2010). 10.1242/jcs.060434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zucker R. M., Price O., “Evaluation of confocal microscopy system performance,” Cytometry 44(4), 273–294 (2001). [DOI] [PubMed] [Google Scholar]
- 13.S. Inoue and K. Spring, Video Microscopy: The Fundamentals (Kluwer Academic/Plenum, New York, 1997). [Google Scholar]
- 14.Gardiner T. W., Toth L. A., “Stereotactic surgery and long-term maintenance of cranial implants in research animals,” Contemp. Top. Lab. Anim. Sci. 38(1), 56–63 (1999). [PubMed] [Google Scholar]
- 15.G. Paxinos and C. Watson, The Rat Brain in Stereotaxic Coordinates, (Academic, Sydney, 1986). [Google Scholar]
- 16.Burns C. G., Milan D. J., Grande E. J., Rottbauer W., MacRae C. A., Fishman M. C., “High-throughput assay for small molecules that modulate zebrafish embryonic heart rate,” Nat. Chem. Biol. 1(5), 263–264 (2005). 10.1038/nchembio732 [DOI] [PubMed] [Google Scholar]