Abstract
Background
Despite advances in the treatment of hepatocellular carcinoma (HCC), a great proportion of patients are eligible only for palliative therapy for reasons of advanced-stage disease or poor hepatic reserve. The use of transarterial chemoembolization (TACE) in the palliation of non-resectable HCC has shown a survival benefit in European and Asian populations. The aim of this study was to assess the efficacy of TACE by analysing overall 5-year survival, interval changes of tumour size and serum alpha-fetoprotein (AFP) levels in a prospective North American cohort.
Methods
From September 2005 to December 2010, 46 candidates for TACE were enrolled in the study. Collectively, they underwent 102 TACE treatments. Data on tumour response, serum AFP and survival were prospectively collected.
Results
In compensated cirrhotic patients, serial treatment with TACE had a stabilizing effect on tumour size and reduced serum AFP levels during the first 12 months. Overall survival rates at 1, 2 and 3 years were 69%, 58% and 20%, respectively. Younger individuals and patients with a lower body mass index, affected by early-stage HCC with involvement of a single lobe, had better survival in univariate analysis. After adjustment for risk factors, early tumour stage (T1 and T2 vs. T3 and T4) at diagnosis was the only statistically significant predictor for survival.
Conclusions
In compensated cirrhotic patients, TACE is an effective palliative intervention and HCC stage at diagnosis seems to be the most important predictor of longterm outcomes.
Keywords: hepatocellular carcinoma, transarterial chemoembolization, TACE, survival, predictors, multivariate Cox regression survival analysis, body mass index, MELD
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer in the world and the third most frequent cause of cancer-related mortality.1 Although HCC is more common in Asia and Africa, its incidence in the Western world is increasing.2 According to the Surveillance and Epidemiology End Results (SEER) registries in the USA, the age-adjusted incidence of HCC increased from 1.3 per 100 000 in 1978–1980 to 6.6 per 100 000 in 2002.3 Hepatic resection or liver transplantation are the only curative interventions for HCC, but the vast majority of patients are not surgical candidates for reasons of advanced tumour stage or underlying liver disease. Therefore, palliative modalities such as local ablation and transarterial chemoembolization (TACE) are the only possible therapies.4
TACE can be performed in patients who are unable to undergo surgery and in patients on waiting lists for liver transplants in order to prevent disease progression.5–7 The process combines the injection of antineoplastic agents with the selective obstruction of arteries feeding the tumour. This results in tumour cell necrosis with preservation of normal liver parenchyma reducing the risk for liver failure.8 During the last decade, TACE has also found a role as a neoadjuvant treatment in resectable patients,9 with a partial response observed in 17.0–61.9%,10 although a complete response is extremely rare.9
TACE had been used for many years to treat patients with HCC worldwide, but controversy about its efficacy was not completely resolved until two randomized controlled trials (RCTs) showed it to have survival benefit compared with supportive therapy.11,12 The literature on predictors of longterm survival in patients undergoing TACE for primary hepatic tumours in Western countries remains limited. Therefore, the objectives of this study were to analyse overall survival and tumour response in patients undergoing sequential TACE treatments for non-resectable HCC in a tertiary university hospital in North America and to explore potential prognostic factors.
Materials and methods
Patient population
The study was approved by the local institutional ethics review board (ERB) (Queen Elizabeth II Health Sciences Centre, Halifax, NS, Canada). All patients diagnosed with non-resectable and non-ablatable HCC were potential candidates. Patients affected by HCC were triaged to their therapeutic algorithm according to Barcelona Clinic Liver Cancer (BCLC) staging criteria.13 Cases of HCC were considered non-resectable when at least one of these conditions was present: a severe comorbidity prohibiting the administration of general anaesthesia; liver dysfunction, and/or portal hypertension contraindicating parenchyma loss during radical tumour resection. Ablation therapy was not considered when the maximum diameter of the tumour was >5 cm, when the tumour was in proximity to major vascular or biliary structures, or if the patient was affected by multifocal disease. All participants were recruited from hepatology, oncology and surgical clinics and underwent serial TACE sessions at intervals of 3–4 months even when radiological cross-sectional studies showed stable disease or partial response, according to the study protocol submitted to the ERB.
Study design
From September 2005 until December 2010, 46 consecutive patients satisfied the inclusion criteria and underwent a total of 102 TACE sessions. All participants were presented at multidisciplinary rounds during which medical oncologists, hepatologists, surgeons and interventional radiologists reached a consensus on the best treatment strategy for each patient. Data on patients' clinical characteristics and survival were recorded prospectively.
Patients included in this study were all adults affected by non-resectable and non-ablatable HCC in the presence of compensated liver function (Child–Pugh class A or B),14 with Eastern Cooperative Oncology Group (ECOG) performance status of 0–2,15 and without radiological evidence of extrahepatic disease according to the 2005 guidelines of the American Association for the Study of Liver Diseases (AASLD).16
Patients were excluded if they were aged <18 years, were affected by advanced liver dysfunction (Child–Pugh class C) or renal impairment (defined as serum creatinine of >180 µmol/l), were allergic to i.v. dye, were diagnosed with extrahepatic disease, had main portal vein or main branch portal vein thrombosis, demonstrated total serum bilirubin >30 µmol/l, or were affected by neutropenia (neutrophil count ≤1000/ml) or thrombocytopenia (platelet count ≤50 000/ml).
Multifocal tumours were recorded if multiple lesions were identified in several segments of the same hepatic lobe; bilobar disease was recorded when both lobes were involved according to the Brisbane terminology of liver anatomy.17
In the presence of bilobar disease, chemoembolization of the lobe with the greatest tumour burden was performed first, followed by treatment of the contralateral lobe after 3–4 months. Physical examination, radiological abdominal studies with i.v. contrast injection, chest radiographs and haematological and blood chemistry tests were obtained as per the study protocol at the time of diagnosis and then prior to all TACE procedures or more often if necessary.
Transarterial chemoembolization procedures were performed every 3–4 months by dedicated interventional radiologists unless extrahepatic disease, liver decompensation or significant side-effects occurred. Patients who had transient liver decompensation or who developed systemic infection or bone marrow suppression during the duration of this study were re-evaluated every 3–4 months and were considered as candidates for repeat TACE if they satisfied the original inclusion criteria.
All data were prospectively collected and entered into a digital database with secure access.
TACE procedures
All patients underwent TACE administered by dedicated interventional radiologists following standard protocols. Abdominal contrast computerized tomography (CT) or magnetic resonance imaging (MRI) studies were obtained in the 4 weeks prior to each therapeutic intervention. Intravenous fluid hydration and parenteral antibiotic prophylaxis with 1 g cefazolin (or 1 g vancomycin in patients allergic to cephalosporin) and 500 mg metronidazole (or 600 mg clindamycin in patients allergic to metronidazole) were administered before arterial groin catheterization. Post-TACE, all patients received 4–8 mg i.v. ondasentron to prevent nausea or vomiting. Patients affected by viral hepatitis B received lamivudine (100 mg/day orally) 1 week prior to and continuing after TACE to prevent hepatitis flare-ups. A selective 4- or 5-French catheter was used to cannulate the common femoral artery and visceral angiography was carried out to assess the arterial blood supply to the liver and to confirm patency of the portal vein. Depending on the size, location and arterial supply of the tumour, the tip of the catheter was then advanced into the right or left hepatic artery or superselectively when possible using microcatheters measuring 2.5–3.0-French [Renegade® (Boston Scientific Corp., Natick, MA, USA) or Cantata® (Cook Medical, Inc., Bloomington, IN, USA)]. Chemoembolization of only one lobe of the liver was carried out in each TACE session after mixing doxorubicin hydrochloride (75 mg/m2 body surface area) with 10 ml of lipiodol (ethiodized poppy seed oil; E-Z-EM Canada, Inc., Montreal, QC, Canada). Polyvinyl alcohol particles (Contour® PVA; Boston Scientific Corp.) of 45–700 µm in size were then injected if the chemoembolized artery territory did not show stagnant flow after the infusion of the chemotoxic agent mixed with lipiodol. During 2009 and 2010, doxorubicin eluting beads of 100–700 µm in size were used to perform TACE in eight patients affected by HCC who were randomized in an international multicentre controlled trial comparing chemoembolization delivered using drug-eluting beads with conventional TACE. All the patients who were treated with doxorubicin eluting beads satisfied the initial inclusion criteria for this study and signed a separate consent form after their recruitment. Before discharge, all patients underwent baseline post-treatment CT without parenteral contrast injection to assess the distribution of lipiodol in the tumour or, in patients treated with drug-eluting beads who did not require the use of lipiodol, with i.v. dye injection to assess the vascularity of neoplastic lesions.
Aims of the study
The primary aims of the study were to assess the overall survival of the cohort and to establish whether the presence of bilobar disease and tumour size (T-stage) were predicting factors for the overall survival of patients undergoing serial TACE sessions for non-resectable HCC. The secondary aims of the study were to assess tumour response according to RECIST (response evaluation criteria in solid tumours) criteria10 and interval changes in serum alpha-fetoprotein (AFP) as a biological marker of response to the therapy.
Diagnosis of primary hepatic tumours
HCC was diagnosed when one cross-sectional study (triphasic contrast abdominal CT scan or MRI) was suggestive for HCC in the presence of elevated serum AFP (≥100 ng/ml). Alternatively, HCC was diagnosed when two contrast-enhanced cross-sectional studies concurred with the identification of hypervascular tumours with portal vein washout. When diagnosis was not established by non-invasive modalities, a percutaneous liver biopsy was obtained according to the guidelines of the AASLD.16
Tumour staging
The largest tumour nodule identified by contrast cross-sectional imaging was sized for each patient at the time of diagnosis and before each TACE by one of the primary investigators (KME) and measured according to the RECIST criteria.10 All the measurements were performed using an electronic scale in millimetres provided by the software used by the radiology department at our institution (IMPAX Web1000™; Agfa HealthCare Corp., Greenville, SC, USA). Tumour–node–metastasis (TNM) classification [American Joint Committee on Cancer (AJCC), 7th edition]18 was used to stratify tumours according to their largest diameter.
Survival
Overall survival was defined as the time interval between the date of the first TACE and the date when patients expired or were censored. Patients were censored if they were alive at the time of the closure of this study, when they had undergone liver transplantation, or if they were lost to follow-up. For the latest group, the time of censoring was defined as the last date of documented follow-up at our institution or by the patient's general practitioner. Missing data were minimized by contacting patients, their families or their primary physicians by telephone or by letter. A patient's date of demise was confirmed by any of the following criteria: death certificate; data from the prospective provincial tumour registry, or contact with the patient's primary doctor or next of kin. Longterm data were available for 43 participants; three subjects were lost to follow-up.
Follow-up investigations
All patients were evaluated with physical examinations, chest radiographs and serial radiological cross-sectional studies (CT or MRI) with parenteral contrast infusion in addition to haematological and biochemical tests including serum AFP every 3–4 months until death or liver transplantation.
Sample size calculation
This study was powered to detect a minimal hazard ratio of 2.5, assuming that the median survival of patients with T4 or bilobar HCC undergoing TACE was 6–8 months, with an accrual period of 48 months and a minimum of 24 months follow-up for the entire cohort of patients. A total of 40 subjects were needed for a two-tailed study with an alpha level of 0.05 and a beta level of 0.8.
Statistical and survival analysis
Summary statistics were constructed for the baseline values, using frequencies and proportions for categorical data, and medians and ranges (minimum–maximum values) for continuous variables. Categorical outcomes were analysed using the chi-squared test or Fisher's exact test as appropriate. Continuous variables were compared using Mann–Whitney or Kruskal–Wallis tests. All statistical tests were two-tailed and a P-value of <0.05 was considered significant. For time-to-event outcomes, the distributions of time to the first event were compared using the log-rank test. The Kaplan–Meier method was used to estimate the absolute risk for each event in each group, and hazard ratios and 95% confidence intervals (CIs) were estimated using the Cox proportional hazard model. To identify the baseline and clinical variables associated with the overall survival time, multivariable analysis was performed using the Cox proportional hazard model with a stepwise selection procedure set at thresholds of 0.10 for inclusion and 0.05 for exclusion. All statistical analyses were performed using spss Version 19.0 (SPSS, Inc., Chicago, IL, USA).
Results
Patient characteristics
Among all the participants, six patients (13%) underwent cadaveric liver transplants after at least one TACE session and were censored at the time of surgery, six patients (13%) had at least one previous ablation and were recruited after developing recurrent disease not treatable with locoregional therapy, and none received any chemotherapy agent other than doxorubicin delivered by selective or superselective hepatic arterial catheterization.
Tables 1 and 2 outline the demographic and clinical characteristics of the study population and variables related to tumour location and stage, in addition to serum AFP and biochemical variables used to measure Model of End-stage Liver Disease (MELD) scores in order to stratify each patient's hepatic function.
Table 1.
Study population characteristics
| Variable | |
|---|---|
| Age, years, median (range) | 51 (44–83) |
| Gender, male, n | 41 |
| Risk factors for primary hepatic tumour, n | |
| Viral hepatitis C | 17 |
| Viral hepatitis B | 2 |
| Non-alcoholic steatohepatitis | 4 |
| Alcohol | 16 |
| Other | 7 |
| Body mass index, median (range) | 27.3 (20.1–39.0) |
| Body surface area, m2, median (range) | 1.91 (1.56–2.51) |
| Candidates for liver transplantation, n | 21 |
| Number of patients transplanted, n | 6 |
| Prior interventions for hepatic tumour, n | |
| Radiofrequency ablation | 6 |
| Histological diagnosis, n | 9 |
| Radiological diagnosis, n | 37 |
| Total number of TACE procedures | 102 |
| Number of TACE procedures/patient, median (range) | 1.5 (1–6) |
| Number of tumours/patient, median (range) | 1.0 (1–6) |
| Total number of TACE procedures, n | |
| Left lobe | 2 |
| Right lobe | 28 |
| Both lobes | 16 |
| Follow-up, months, median (range) | 17.1 (2–48) |
| Interval between TACE 1 and TACE 2, weeks, median (range) | 17 (9–40) |
| Interval between TACE 2 and TACE 3, weeks, median (range) | 22 (17–32) |
| Interval between TACE 3 and TACE 4, weeks, median (range) | 13 (12–14) |
TACE, transarterial chemoembolization.
Table 2.
Tumour and laboratory variables at diagnosis
| Tumour and laboratory variables at diagnosis | |
|---|---|
| Tumour characteristics, n | |
| Single tumour | 25 |
| Multiple tumours | 21 |
| Size of the largest tumour, cm, median (range) | 4.4 (1–14) |
| Location of tumour, n | |
| Left lobe | 2 |
| Right lobe | 28 |
| Bilobar | 16 |
| Radiological vascular tumour invasion, n | |
| Present | 5 |
| Absent | 41 |
| TNM classification (AJCC, 7th Edition), n | |
| T1 | 9 |
| T2 | 15 |
| T3 | 22 |
| White blood cell count, ×109/l, median (range) | 5.9 (2.7–11.8) |
| Red blood cells, ×10120/l, median (range) | 4.0 (2.7–5.2) |
| Platelets, ×109/l, median (range) | 112 (49–462) |
| International normalized ratio, median (range) | 1.1 (1.0–2.4) |
| Potassium, mmol/l, median (range) | 4.1 (3.4–5.3) |
| Sodium, mmol/l, median (range) | 136 (127–143) |
| Creatinine, µmol/l, median (range) | 73.5 (55–153) |
| Total bilirubin, µmol/l, median (range) | 18 (4–88) |
| Albumin, g/l, median (range) | 33 (22–42) |
| MELD scorea, median (range) | 7.5 (4–16) |
| Patients with MELD scores ≥10, n | 14 |
| Serum alpha-fetoprotein level, median (range) | 123 (1–206 006) |
| Patients with serum AFP ≥100 ng/ml, n | 24 |
MELD scores at diagnosis were calculated without adjusting for extra points for the presence of hepatocellular carcinoma.36
TNM, tumour–node–metastasis; AJCC, American Joint Committee on Cancer; MELD, Model for End-stage Liver Disease; AFP, alpha-fetoprotein.
Tumour response
A disease-stabilizing effect on tumour diameter was observed after initiation of the treatment protocol, along with a concomitant reduction in serum AFP. The period of reduction in these two variables persisted for almost 1 year, corresponding to TACE treatment 4, when both tumour size and serum AFP increased, although this increase did not reach statistical significance (Figs 1 and 2).
Figure 1.
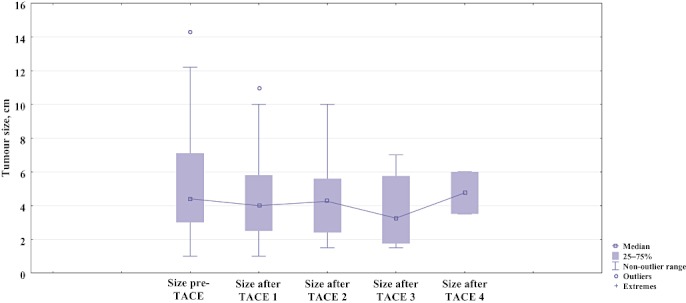
Boxplot showing changes in diameter of the largest tumour nodule prior to and after initiation of the therapeutic protocol (P= 0.761, not significant). TACE, transarterial chemoembolization
Figure 2.
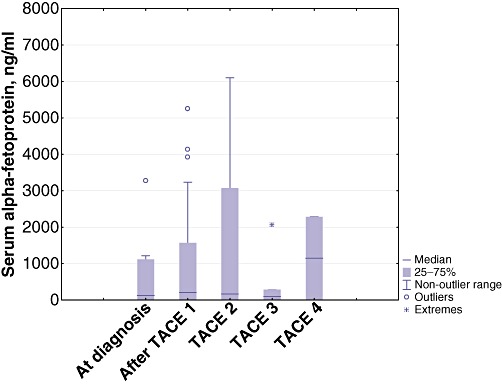
Boxplot showing changes in mean serum alpha-fetoprotein at diagnosis and after initiation of the therapeutic protocol (P= 0.490). TACE, transarterial chemoembolization
Survival
Median survival in this cohort was 18.2 months (95% CI 12.0–23.2). Overall survival probabilities at 1, 2 and 3 years were 69%, 58% and 20%, respectively (Fig. 3).
Figure 3.
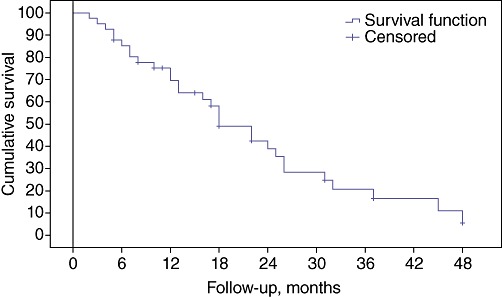
Kaplan–Meier curve showing survival function of the entire cohort of patients affected by hepatocellular carcinoma treated with at least one session of transarterial chemoembolization. Patients at risk: at 6 months, n= 34; at 12 months, n= 26; at 18 months, n= 23; at 24 months, n= 11; at 30 months, n= 8; at 36 months, n= 5; at 42 months, n= 3; at 48 months, n= 1
In univariate analysis, early tumour stage and involvement of only one hepatic lobe were predictors for better survival. Other variables that emerged as positive predictors were: younger age; lower body mass index (BMI); involvement of only one segment of the liver, and the presence of three or fewer tumours (Table 3).
Table 3.
Univariate analysis of survival predictors
| Variable | Median | P-value | ||
|---|---|---|---|---|
| Estimate | SE | 95% CI | ||
| Age, years | ||||
| <60 | 24.000 | 2.726 | 18.657–29.343 | 0.092 |
| ≥60 | 18.000 | 0.663 | 16.700–19.300 | |
| Gender | ||||
| Female | 32.000 | 11.429 | 9.600–54.400 | 0.393 |
| Male | 22.000 | 2.498 | 17.103–26.897 | |
| Body mass index | ||||
| <30 | 18.000 | 3.350 | 11.434–24.566 | 0.015 |
| >30 | 48.000 | 12.565 | 23.374–72.626 | |
| Tumour stage | ||||
| T1 | 48.000 | 0.000 | 0.025 | |
| T2 | 26.000 | 10.241 | 5.928–46.072 | |
| T3 | 18.000 | 3.318 | 11.496–24.504 | |
| Multifocality | ||||
| No | 32.000 | 6.480 | 19.299–44.701 | 0.013 |
| Yes | 17.000 | 3.536 | 10.070–23.930 | |
| Vascular invasion by radiological characteristics | ||||
| No | 23.000 | 3.026 | 17.069–28.931 | 0.679 |
| Yes | 12.000 | 8.500 | 0.000–28.660 | |
| Presence of ≥3 tumours | ||||
| No | 32.000 | 7.433 | 17.431–46.569 | 0.047 |
| Yes | 18.000 | 4.629 | 8.927–27.073 | |
| Single lobe vs. bilobar disease | ||||
| Single lobe disease | 32.000 | 4.849 | 22.497–41.503 | 0.030 |
| Bilobar | 17.000 | 3.118 | 10.889–23.111 | |
| Model for End-stage Liver Disease (MELD) score ≥10 | ||||
| No | 22.000 | 2.676 | 16.754–27.246 | 0.228 |
| Yes | 13.000 | 5.233 | 2.743–23.257 | |
| Serum alpha-fetoprotein (AFP) ≥100 mg/ml | ||||
| No | 24.000 | 8.039 | 8.243–39.757 | 0.250 |
| Yes | 22.000 | 3.262 | 15.607–28.393 | |
SE, standard error; 95% CI, 95% confidence interval.
Multivariate Cox proportional hazard regression analysis revealed that none of these variables reached statistical significance after adjusting for tumour stage at the time of diagnosis (Table 4).
Table 4.
Multivariate Cox survival regression analysis
| Variable | B | SE | Wald | d.f. | P-value | Exp(B) | 95.0% CI for Exp(B)a |
|---|---|---|---|---|---|---|---|
| Age (cut-off value: 60 years) | −0.283 | 0.479 | 0.347 | 1 | 0.556 | 0.754 | 0.295–1.929 |
| Body mass index (cut-off value: 30) | −0.041 | 0.915 | 0.002 | 1 | 0.964 | 0.959 | 0.160–5.765 |
| Number of tumours (cut-off value: 3) | −11.176 | 238.273 | 0.002 | 1 | 0.963 | 0.000 | 0.000–1.800 (E202) |
| Presence of bilobar disease | 4.649 | 124.401 | 0.001 | 1 | 0.970 | 104.495 | 0.000–2.390 (E107) |
| Largest tumour diameter (cut-off value: 5 cm) | 0.880 | 0.654 | 1.808 | 1 | 0.179 | 2.410 | 0.669–8.688 |
| Venous tumour invasion | 6.104 | 213.578 | 0.001 | 1 | 0.970 | 447.535 | 0.000–2.810 (E184) |
| MELD score not adjusted for HCC (cut-off value: 10) | −0.098 | 0.905 | 0.012 | 1 | 0.914 | 0.907 | 0.154–5.341 |
| Serum AFP level (cut-off value: 100 ng/ml) | −0.207 | 0.534 | 0.150 | 1 | 0.699 | 0.813 | 0.285–2.318 |
Exponential terms.
SE, standard error; 95% CI, 95% confidence interval; MELD, Model for End-stage Liver Disease; HCC, hepatocellular carcinoma; AFP, alpha-fetoprotein.
Discussion
One of the relative contraindications to TACE for patients with HCC and who are not candidates for resection or ablation is the presence of moderate to severe liver dysfunction. In North America, this is a common event because the majority of patients affected by HCC have cirrhosis caused by hepatitis C virus (HCV) infection, alcohol or non-alcoholic steatohepatitis (NASH). In these patients, treatment modalities are more likely to be dictated by their liver function than by tumour stage. Quite often, clinicians managing cirrhotic patients with HCC must make a difficult decision on whether the treatment is worth the potential risk for liver decompensation or, in the worst case scenario, irreversible organ failure and premature death. Therefore, it is important to validate the impact of TACE on overall survival in patients treated in geographical areas in which the incidence of HCC is low and cirrhosis is often present at diagnosis.
The body of literature on outcomes in Asian patients undergoing treatment for HCC is much greater than that for patients living in the West, where risk factors and the degree of liver disease differ from those in other geographical areas.11,19–21 Although arterial embolization with or without the addition of chemotherapy has been used for several decades in Asia,22 only in 2002 did two RCTs report a significant survival advantage in non-resectable HCC treated by TACE in comparison with best supportive care.11,12 Llovet et al.11 reported 2-year survival rates of 63% and 27% in patients undergoing TACE and patients receiving best supportive care, and Lo et al.12 reported 3-year survival rates of 26% and 3%, respectively. The survival advantage of TACE was also supported by a recent meta-analysis that confirmed the benefit of chemoembolization in selected patients.23 Since then, TACE has become the standard of care for this group of patients, as suggested in practice guidelines endorsed by the AASLD first in 2005 and then in 2011.16,24
The current study validates the benefit of TACE in a cohort of Western patients, all of whom had cirrhosis at the time of diagnosis of HCC and were at high risk for liver decompensation. With strict application of the BCLC triaging algorithm, patients were selected and treated according to the most recent AASLD practice guidelines with sequential sessions of TACE. Stabilization of the tumour burden and reduction of serum AFP were observed during the first year post-initiation of the treatment protocol. Although the interval changes over time in tumour diameter and serum tumour marker did not achieve significance, the median survival and survival probability observed were comparable with the results reported by other investigators.25,26 Overall survival in the cohort of patients enrolled at our centre is comparable with the survival results reported in the two earlier RCTs11,12 and confirmed by other groups in both Asian and Western countries.7,19,20,26
In the current cohort, patients tolerated TACE well and were discharged from hospital after only 1 or 2 days during which they were treated for hydration or for abdominal pain arising from the glissonian inflammation caused by the embolization of the tumour. The most common immediate side-effect after TACE was the development of post-embolization syndrome, which occurred in 50% of patients. Severe side-effects included the development of a hepatic abscess (one patient) and transient liver decompensation with ascites responsive to diuretics (five patients). These findings were similar to previously reported data.19
Previous European and Asian studies have examined prognostic factors associated with improved outcomes after TACE. A reduction in serum AFP after the intervention,27 the number of TACE procedures,20,28 low MELD score,7 the absence of diffuse disease26,29 and small tumour size30 have been shown to correlate with better survival. In the current study, univariate analysis showed survival to be significantly better in younger individuals and in patients with higher BMI, with early-stage tumours and with single lobe involvement. However, in multivariate analysis the only significant variable associated with improved survival was the presence of early-stage tumours (T1 and T2 vs. T3 and T4).
Previous studies have shown that the introduction of screening for HCC is cost-effective, represents the only method for the early detection of HCC and subsequently increases the percentage of patients able to undergo curative interventions.31–33 The findings of this study would also suggest that the use of screening programmes could be beneficial even when patients are not candidates for surgery because tumour size is a prognostic factor for response to TACE and overall survival. As HCC screening has become the standard of care for cirrhotic patients, approximately 60% of subjects in the current cohort were enrolled in a screening or surveillance programme although their compliance was suboptimal. The remaining patients had been diagnosed with HCC after presenting with symptoms or signs of liver decompensation. This group of patients had more advanced tumours and none of them fulfilled the Milan criteria for liver transplantation.34
One of the limitations of this study is the relatively small number of patients and the fact that not all subjects were evaluated for histological proof of HCC. Therefore, there is the potential risk that some patients treated with TACE were not affected by primary malignant lesions of the liver, but, rather, by benign conditions such as adenomas, focal nodular hyperplasia, and regenerative or dysplastic nodules that have radiological features similar to those of HCC. The risk for misdiagnosis was minimized by the fact that our protocol applied the criteria suggested by the AASLD for the diagnosis of HCC, for which liver biopsy is not always necessary in the presence of confirming cross-sectional contrast-enhanced radiological studies and elevation of serum tumour marker levels.16
Another limitation is that, during 2009 and 2010, doxorubicin eluting beads were used to perform TACE in eight patients who were enrolled in an international multicentre RCT comparing chemoembolization delivered by drug-eluting beads with conventional TACE.35 Although these patients satisfied the same inclusion criteria as the rest of the cohort, and received the same amount of doxorubicin per square metre of body surface area, their therapy deviated from conventional TACE. As drug-eluting beads enhance selective tumour drug delivery,35 an internal analysis of possible different outcomes in patients treated with drug-eluting beads vs. conventional therapy would have been interesting, but was not feasible given the small number of patients. Nevertheless, the results of a large multicentre study (PRECISION V) did not confirm survival differences between the two modalities although patients treated with drug-eluting beads tolerated the intervention better.35
One of the strengths of this study was its prospective design and the low attrition rate in the cohort, in which follow-up data were available for 93% of patients. In addition, the management of patients recruited in this study was based on well-established staging and therapeutic algorithms such as the BCLC staging criteria13 and clinical guidelines endorsed by the AASLD.16,24 Therefore, the results should be generalizable to other populations of cirrhotic patients with similar selection and treatment protocols.
This study would suggest that even in compensated cirrhotic patients and patients with advanced tumour stage, bilobar involvement and obesity, TACE provides a survival benefit in comparison with supportive care. In addition, when properly selected, liver decompensation leading to severe side-effects or death after TACE is very infrequent, even in cirrhotic patients, in whom tumour stage at the time of diagnosis appears to be the most significant prognostic factor for survival.
Conflicts of interest
None declared.
References
- 1.Kamangar F, Dores GM, Anderson WF. Patterns of cancer incidence, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24:2137–2150. doi: 10.1200/JCO.2005.05.2308. [DOI] [PubMed] [Google Scholar]
- 2.Parkin DM, Bray F, Ferlay J, Pisani P. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001;94:153–156. doi: 10.1002/ijc.1440. [DOI] [PubMed] [Google Scholar]
- 3.National Cancer Institute. Surveillance, Epidemiology and End Results (SEER) Registry. 2007. http://seer.cancer.gov/. [Accessed 3 October 2011.
- 4.Wood TF, Rose DM, Chung M, Allegra DP, Foshag LJ, Bilchik AJ. Radiofrequency ablation of 231 unresectable hepatic tumours: indications, limitations, and complications. Ann Surg Oncol. 2000;7:593–600. doi: 10.1007/BF02725339. [DOI] [PubMed] [Google Scholar]
- 5.Schaudt A, Kriener S, Schwarz W, Wullstein C, Zangos S, Vogl T, et al. Role of transarterial chemoembolization for hepatocellular carcinoma before liver transplantation with special consideration of tumour necrosis. Clin Transplant. 2009;23(Suppl. 21):61–67. doi: 10.1111/j.1399-0012.2009.01111.x. [DOI] [PubMed] [Google Scholar]
- 6.Shi XJ, Jin X, Wang MQ, Wei LX, Ye HY, Liang YR, et al. Outcomes of loco-regional therapy for down-staging of hepatocellular carcinoma prior to liver transplantation. Hepatobiliary Pancreat Dis Int. 2011;10:143–150. doi: 10.1016/s1499-3872(11)60023-4. [DOI] [PubMed] [Google Scholar]
- 7.Dhanasekaran R, Kooby DA, Staley CA, Kauh JS, Khanna V, Kim HS. Prognostic factors for survival in patients with unresectable hepatocellular carcinoma undergoing chemoembolization with doxorubicin drug-eluting beads: a preliminary study. HPB (Oxford) 2010;12:174–180. doi: 10.1111/j.1477-2574.2009.00138.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Verslype C, Van Cutsem E, Dicato M, Arber N, Berlin JD, Cunningham D, et al. The management of hepatocellular carcinoma. Current expert opinion and recommendations derived from the 10th World Congress on Gastrointestinal Cancer, Barcelona, 2008. Ann Oncol. 2009;20(Suppl. 7):1–6. doi: 10.1093/annonc/mdp281. [DOI] [PubMed] [Google Scholar]
- 9.Lau WY, Lai EC. Hepatocellular carcinoma: current management and recent advances. Hepatobiliary Pancreat Dis Int. 2008;7:237–257. [PubMed] [Google Scholar]
- 10.Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1) Eur J Cancer. 2009;45:228–247. doi: 10.1016/j.ejca.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 11.Llovet JM, Real MI, Montana X, Planas R, Coll S, Aponte J, et al. Arterial embolization or chemoembolization versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomized controlled trial. Lancet. 2002;359:1734–1739. doi: 10.1016/S0140-6736(02)08649-X. [DOI] [PubMed] [Google Scholar]
- 12.Lo CM, Ngan H, Tso WK, Liu CL, Lam CM, Poon RT, et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology. 2002;35:1164–1171. doi: 10.1053/jhep.2002.33156. [DOI] [PubMed] [Google Scholar]
- 13.Pons F, Varela M, Llovet JM. Staging systems in hepatocellular carcinoma. HPB (Oxford) 2005;7:35–41. doi: 10.1080/13651820410024058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Child CG, Zuidema GD. Experimental surgery of the portal vein, hepatic artery and hepatic veins. Major Probl Clin Surg. 1964;1:189–224. [PubMed] [Google Scholar]
- 15.Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–655. [PubMed] [Google Scholar]
- 16.Bruix J, Sherman M. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208–1236. doi: 10.1002/hep.20933. [DOI] [PubMed] [Google Scholar]
- 17.Pang YY. The Brisbane 2000 terminology of liver anatomy and resections. HPB (Oxford) 2000;2:333–339. doi: 10.1080/136518202760378489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.American Joint Committee on Cancer. AJCC Cancer Staging Handbook. 7th. New York, NY: Springer-Verlag; 2010. [Google Scholar]
- 19.Molinari M, Kachura JR, Dixon E, Rajan DK, Hayeems EB, Asch MR, et al. Transarterial chemoembolization for advanced hepatocellular carcinoma: results from a North American cancer centre. Clin Oncol (R Coll Radiol) 2006;18:684–692. doi: 10.1016/j.clon.2006.07.012. [DOI] [PubMed] [Google Scholar]
- 20.Farinati F, De Maria N, Marafin C, Herszenyi L, Del Prato S, Rinaldi M, et al. Unresectable hepatocellular carcinoma in cirrhosis: survival, prognostic factors, and unexpected side-effects after transcatheter arterial chemoembolization. Dig Dis Sci. 1996;41:2332–2339. doi: 10.1007/BF02100123. [DOI] [PubMed] [Google Scholar]
- 21.Bosch FX, Ribes J, Borras J. Epidemiology of primary liver cancer. Semin Liver Dis. 1999;19:271–285. doi: 10.1055/s-2007-1007117. [DOI] [PubMed] [Google Scholar]
- 22.Kato T, Nemoto R, Mori H, Takahashi M, Tamakawa Y, Harada M. Arterial chemoembolization with microencapsulated anticancer drug. An approach to selective cancer chemotherapy with sustained effects. JAMA. 1981;245:1123–1127. [PubMed] [Google Scholar]
- 23.Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: chemoembolization improves survival. Hepatology. 2003;37:429–442. doi: 10.1053/jhep.2003.50047. [DOI] [PubMed] [Google Scholar]
- 24.Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–1022. doi: 10.1002/hep.24199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Takayasu K, Arii S, Ikai I, Omata M, Okita K, Ichida T, et al. Prospective cohort study of transarterial chemoembolization for unresectable hepatocellular carcinoma in 8510 patients. Gastroenterology. 2006;131:461–469. doi: 10.1053/j.gastro.2006.05.021. [DOI] [PubMed] [Google Scholar]
- 26.Ji SK, Cho YK, Ahn YS, Kim MY, Park YO, Kim JK, et al. Multivariate analysis of the predictors of survival for patients with hepatocellular carcinoma undergoing transarterial chemoembolization: focusing on superselective chemoembolization. Korean J Radiol. 2008;9:534–540. doi: 10.3348/kjr.2008.9.6.534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Berger DH, Carrasco CH, Hohn DC, Curley SA. Hepatic artery chemoembolization or embolization for primary and metastatic liver tumours: post-treatment management and complications. J Surg Oncol. 1995;60:116–121. doi: 10.1002/jso.2930600210. [DOI] [PubMed] [Google Scholar]
- 28.Okuda K, Obata H, Nakajima Y, Ohtsuki T, Okazaki N, Ohnishi K. Prognosis of primary hepatocellular carcinoma. Hepatology. 1984;4(Suppl.):3–6. doi: 10.1002/hep.1840040703. [DOI] [PubMed] [Google Scholar]
- 29.Lopez RR, Jr, Pan SH, Hoffman AL, Ramirez C, Rojter SE, Ramos H, et al. Comparison of transarterial chemoembolization in patients with unresectable, diffuse vs. focal hepatocellular carcinoma. Arch Surg. 2002;137:653–657. doi: 10.1001/archsurg.137.6.653. discussion 657–658. [DOI] [PubMed] [Google Scholar]
- 30.Savastano S, Miotto D, Casarrubea G, Teso S, Chiesura-Corona M, Feltrin GP. Transcatheter arterial chemoembolization for hepatocellular carcinoma in patients with Child's grade A or B cirrhosis: a multivariate analysis of prognostic factors. J Clin Gastroenterol. 1999;28:334–340. doi: 10.1097/00004836-199906000-00010. [DOI] [PubMed] [Google Scholar]
- 31.Sarasin FP, Giostra E, Hadengue A. Cost-effectiveness of screening for detection of small hepatocellular carcinoma in Western patients with Child–Pugh class A cirrhosis. Am J Med. 1996;101:422–434. doi: 10.1016/S0002-9343(96)00197-0. [DOI] [PubMed] [Google Scholar]
- 32.Tseng PL, Wang JH, Tung HD, Hung CH, Kee KM, Chen CH, et al. Optimal treatment increased survival of hepatocellular carcinoma patients detected with community-based screening. J Gastroenterol Hepatol. 2010;25:1426–1434. doi: 10.1111/j.1440-1746.2010.06285.x. [DOI] [PubMed] [Google Scholar]
- 33.Qian MY, Yuwei JR, Angus P, Schelleman T, Johnson L, Gow P. Efficacy and cost of a hepatocellular carcinoma screening programme at an Australian teaching hospital. J Gastroenterol Hepatol. 2010;25:951–956. doi: 10.1111/j.1440-1746.2009.06203.x. [DOI] [PubMed] [Google Scholar]
- 34.Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, Bozzetti F, et al. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med. 1996;334:693–699. doi: 10.1056/NEJM199603143341104. [DOI] [PubMed] [Google Scholar]
- 35.Lammer J, Malagari K, Vogl T, Pilleul F, Denys A, Watkinson A, et al. Prospective randomized study of doxorubicin-eluting bead embolization in the treatment of hepatocellular carcinoma: results of the PRECISION V study. Cardiovasc Intervent Radiol. 2010;33:41–52. doi: 10.1007/s00270-009-9711-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sheth M, Riggs M, Patel T. Utility of the Mayo End-Stage Liver Disease (MELD) score in assessing prognosis of patients with alcoholic hepatitis. BMC Gastroenterol. 2002;2:2. doi: 10.1186/1471-230X-2-2. [DOI] [PMC free article] [PubMed] [Google Scholar]


