Abstract
Diabetes can be controlled with insulin injections, but a curative approach that restores the number of insulin-producing β-cells is still needed. Using a zebrafish model of diabetes, we screened ~7000 small molecules to identify enhancers of β-cell regeneration. The compounds we identified converge on the adenosine signaling pathway and include exogenous agonists and compounds that inhibit degradation of endogenously produced adenosine. The most potent enhancer of β-cell regeneration was the adenosine agonist 5′-N-Ethylcarboxamidoadenosine (NECA), which acting through the adenosine receptor A2aa increased β-cell proliferation and accelerated restoration of normoglycemia in zebrafish. Despite markedly stimulating β-cell proliferation during regeneration, NECA had only a modest effect during development. The proliferative and glucose-lowering effect of NECA was confirmed in diabetic mice, suggesting an evolutionarily conserved role for adenosine in β-cell regeneration. With this whole-organism screen, we identified components of the adenosine pathway that could be therapeutically targeted for the treatment of diabetes.
Introduction
Diabetes is characterized by elevated blood glucose levels, a consequence of insufficient insulin supply and/or insulin resistance. Although diabetes can be treated with injections of insulin, a curative approach that increases life expectancy and reduces morbidity is an unmet medical need. Despite mechanistic differences, both type 1 and late-stage type 2 diabetes feature depletion of β-cells, the insulin-producing cells of the pancreas. Experimental ablation of β-cells by chemical treatment or partial pancreatectomy in rodents is followed by significant recovery of the β-cell mass, indicating that the adult pancreas has the capacity to regenerate (reviewed by Bonner-Weir et al., 2010). This regenerative capacity could potentially be exploited therapeutically—if the underlying mechanisms were better understood. Indeed, although the transcriptional cascade that regulates β-cell formation is fairly well characterized (reviewed by Pan and Wright, 2011), the extrinsic signals that regulate β-cell regeneration remain unclear (reviewed by Halban et al., 2010).
Several signals can increase β-cell formation, by stimulating β-cell neogenesis and/or proliferation. For example, hepatocyte growth factor, insulin-like growth factor I, and placental lactogen can increase β-cell proliferation (reviewed by Ackermann and Gannon, 2007), while exendin-4 (a synthetic analog of glucagon-like peptide 1)(Xu et al., 1999; Lavine and Attie, 2010) or a combination of epidermal growth factor and gastrin (Krakowski et al., 1999; Rooman and Bouwens, 2004) has been reported to increase both the proliferation of β-cells and their formation from ductal progenitors. However, recent findings in mice suggest that ductal progenitors do not contribute significantly to β-cell neogenesis in adult organisms, and that combined epidermal growth factor and gastrin are not in fact efficient promoters of β-cell neogenesis (Solar et al., 2009). In addition, most of these factors are peptide hormones, which affect a multitude of cellular processes and need to be administered by injection. There is therefore a need for small-molecule therapeutics that can specifically promote β-cell regeneration and ideally be taken orally.
Studies of factors affecting β-cell regeneration in vivo have traditionally been based on hypothesis-driven selection of candidate factors or on preliminary findings from in vitro screens. However, the candidate approach is biased, and in vitro screens cannot replicate the endogenous micro- and macro-environments of a living organism—such as communication between different cell types and tissues, the existence of different types of endocrine progenitors, and physiological responses to β-cell depletion. Unbiased screens performed in vivo are therefore warranted. The zebrafish model is particularly good for studying pancreatic development in vivo. First, the simplicity of its organ structures, e.g. the zebrafish embryo has only one pancreatic islet during the first week of development, allows rapid analysis of cellular changes (reviewed by Kinkel and Prince, 2009). Second, zebrafish embryos are amenable to efficient transgenesis and drug delivery. The zebrafish model is thus ideal for the efficient transgenic-based and cost-effective screening of small-molecule libraries in vivo (reviewed by Zon and Peterson, 2005). Indeed, several chemical screens in zebrafish have identified drugs that are now in clinical development (North et al., 2007; Yu et al., 2008).
Here, we screened ~7000 compounds in a transgenic zebrafish model of β-cell regeneration in which a conditionally toxic protein is expressed in β-cells (Curado et al., 2007; Pisharath et al., 2007). This screening approach allowed us to discover compounds that promote β-cell regeneration regardless of cellular mechanism, e.g. proliferation (Dor et al., 2004), neogenesis (Inada et al., 2008; Xu et al., 2008), or transdifferentiation (Zhou et al., 2008; Thorel et al., 2010), and thus does not restrict the discovery of hit-compounds. We then used secondary assays to pinpoint the precise mechanisms of action. Finally, we tested the most promising hit-compound in a mouse model of diabetes to assess the relevance of the findings to β-cell regeneration in adult mammals. Using this three-pronged approach, we found that the most potent enhancers of β-cell regeneration activate the adenosine signaling pathway and increase β-cell regeneration by promoting β-cell proliferation.
Results
In vivo chemical screen for β-cell regeneration
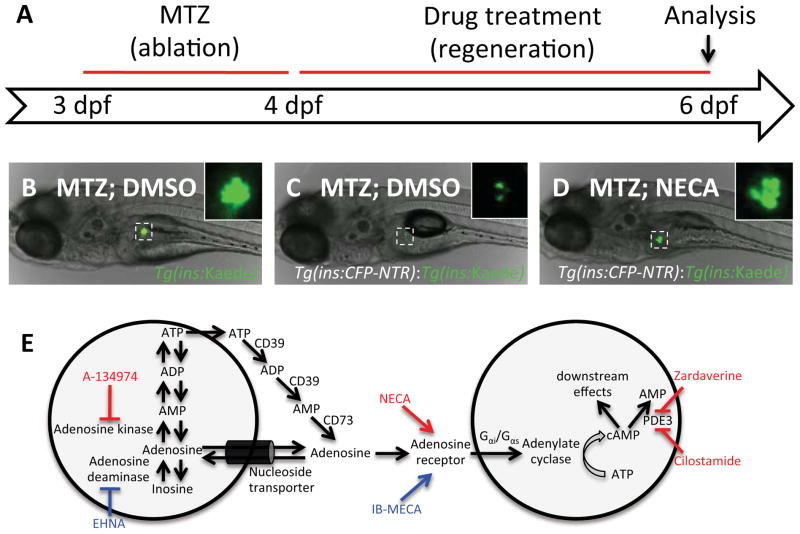
We performed a chemical screen for enhancers of β-cell regeneration by using a zebrafish model in which β-cells are ablated by nitroreductase (NTR), an enzyme that converts the chemical metronidazole (MTZ) to a toxic product that induces β-cell apoptosis (Curado et al., 2007; Pisharath et al., 2007). Importantly, β-cell mass recovers once MTZ is removed from the water, and the zebrafish survive to adulthood. Using this system, we ablated the β-cells 3–4 days post fertilization (dpf) (at which stage each larva has ~35 β-cells) and then treated the larvae with the compounds from 4–6 dpf, when all the structures of the mature pancreas are present (Figure 1A). The transgenic larvae we used for this screen express a fusion protein of cyan fluorescent protein (CFP) and NTR under the control of the insulin promoter, Tg(ins:CFP-NTR). Because CFP is only weakly fluorescent, we crossed the Tg(ins:CFP-NTR) line with another transgenic line that expresses the brightly fluorescent protein Kaede under the control of the insulin promoter, Tg(ins:Kaede). By treating these double-transgenic Tg(ins:CFP-NTR);Tg(ins:Kaede) larvae with MTZ from 3–4 days post fertilization (dpf), we ablated essentially all β-cells and could easily follow the ensuing β-cell regeneration. At 4 dpf, we washed out MTZ and transferred the larvae to 96-well plates for treatment with the compounds. After allowing β-cells to regenerate for two days in the presence of chemicals from the libraries, we analyzed β-cell regeneration by evaluating the number of Tg(ins:Kaede)-positive β-cells. At 6 dpf, control larvae carrying only the Tg(ins:Kaede) transgene were not affected by MTZ and had a normal number of β-cells (Figure 1B), whereas MTZ-treated Tg(ins:CFP-NTR);Tg(ins:Kaede) larvae had only a few β-cells (Figure 1C). Compounds found to increase the β-cell regenerative response in MTZ-treated Tg(ins:CFP-NTR);Tg(ins:Kaede) larvae (see example in Figure 1D) were re-tested and, if warranted, further analyzed at a range of concentrations. Following this schema, we used ~100,000 larvae to identify enhancers of β-cell regeneration by screening 7186 compounds, including FDA-approved drugs, natural products, and uncharacterized compounds (Table S1).
Figure 1. A chemical screen for β-cell regeneration.
(A) Schema for the β-cell regeneration screen. The pancreatic β-cells are conditionally targeted for ablation from 3–4 dpf by using β-cell specific expression of nitroreductase [Tg(ins:CFP-NTR)], which converts Metronidazole (MTZ) into a cytotoxic product. After washing away the MTZ, the larvae are placed in 96-well plates, and exposed to 2–50μM of the compounds in 1% DMSO. After 2 days of recovery, from 4–6 dpf, β-cell regeneration can be easily quantified in double-transgenic larvae, Tg(ins:CFP-NTR);Tg(ins:Kaede), because Kaede labels the β-cells with bright fluorescence.
(B) Picture of a control Tg(ins:Kaede) larva at 6 dpf that was not affected by the MTZ treatment (because it does not express NTR) and therefore displays a typical number of β-cells, as visualized with the microscope used for screening. The inset displays a magnified view of the pancreatic islet (outlined by the dashed square).
(C) 6 dpf Tg(ins:CFP-NTR);Tg(ins:Kaede) larva following β-cell ablation with MTZ from 3–4 dpf and vehicle treatment from 4–6 dpf. Typically, these control larvae have 3–7 β-cells at this stage.
(D) Tg(ins:CFP-NTR);Tg(ins:Kaede) larva following β-cell ablation with MTZ from 3–4 dpf and treatment with the hit-compound NECA from 4–6 dpf. This particular larva contains too many β-cells to count without the use of confocal microscopy.
(E) The hit-compounds converge on adenosine signaling/metabolism by targeting adenosine kinase (A-134974), adenosine receptors (NECA), and phosphodiesterase 3/4 (Cilostamide and Zardaverine). Compounds that increased β-cell regeneration more than two fold after two days of treatment are labeled in red. After re-screening all activators of adenosine signaling contained in the Sigma LOPAC library, we found that an adenosine deaminase inhibitor (EHNA) and another non-selective adenosine agonist (IB-MECA) (labeled in blue) could also increase β-cell regeneration, although less than two-fold. See also Figure S1.
The most potent enhancers of β-cell regeneration converge on the adenosine pathway
In our screen, we defined the threshold for a hit-compound as a doubling of the number of β-cells after two days of regeneration, i.e. from ~5 β-cells in vehicle-treated larvae to ≥10 β-cells in chemical-treated larvae. We identified five compounds that could double the number of β-cells after two days of treatment. Interestingly, four of these five compounds converge on the adenosine signaling pathway and are predicted to augment the signaling output of the pathway (Figure 1E): (1) 5′-N-Ethylcarboxamidoadenosine (NECA), a non-specific adenosine agonist that activates adenosine G protein-coupled receptor (GPCR) signaling; (2) A-134974, an adenosine kinase inhibitor that blocks degradation of adenosine and thereby increases the level of endogenous adenosine; (3) Cilostamide, which can affect adenosine signaling by inhibiting phosphodiesterase (PDE3) and thereby decrease degradation of the intracellular mediator cAMP; and (4) Zardaverine, which can affect adenosine signaling by inhibiting PDE3 and PDE4. The fifth hit, CID661578, has no known mechanism of action. Hits with different mechanisms of action were tested in various combinations and a synergistic effect was observed between NECA and Cilostamide (Figure S1A), indicating that NECA on its own does not activate the full potential of the cAMP pathway.
The convergence of hits on the adenosine pathway prompted us to re-screen other modulators of this pathway. In doing so, we found that the adenosine agonist, IB-MECA, and the adenosine deaminase inhibitor, EHNA, also increased β-cell regeneration, though they were not as potent as the original hits, i.e. IB-MECA increased the number of β-cells from 5.4 to 9.0 (± 0.55, n = 21 vehicle-treated larvae; ± 0.65, n = 22 IB-MECA-treated larvae; ± SEM, P < 0.001), and EHNA from 5.5 to 8.3 (± 0.35 n = 22 vehicle-treated larvae; ± 0.58, n = 19 EHNA-treated larvae; ± SEM, P < 0.001). Together, these findings confirm that the adenosine pathway enhances β-cell regeneration in zebrafish and identify several components of this pathway that could be therapeutically targeted to increase β-cell regeneration.
The hit-compounds increase regeneration, not survival, of β-cells
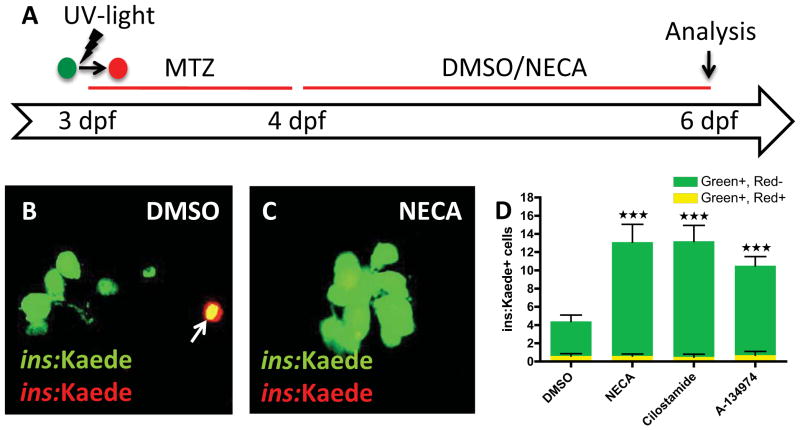
To determine whether the hit-compounds increase the number of β-cells by promoting β-cell survival rather than β-cell regeneration, we followed the fate of β-cells during ablation and regeneration via cell labeling. Using Tg(ins:CFP-NTR);Tg(ins:Kaede) larvae, we converted the fluorescence of the Kaede protein from green to red by exposing the larvae to UV light (Ando et al., 2002) at 3 dpf; this conversion permanently marked all β-cells that were present before the ablation step. We then treated the larvae from 3–4 dpf with MTZ to ablate the β-cells, and from 4–6 dpf with the hit-compounds (Figure 2A). In this set-up, β-cells that survive the ablation will co-express red- and green-fluorescent Kaede (visualized as yellow overlap), whereas newly formed β-cells will express only green-fluorescent Kaede. Confocal analysis at 6 dpf showed that, on average, less than one β-cell per larva survived the ablation in vehicle-treated larvae (Figure 2B–D), and that none of the hit-compounds increased this survival rate (Figure 2D). By contrast, we found many β-cells that were labeled green only, indicating that they were newly formed. The number of green-only β-cells was significantly greater in larvae treated with the hit-compounds than in those treated with vehicle, i.e. the number of green-only β-cells increased from 3.8 in vehicle-treated larvae to 12.7 in NECA-, 12.5 in Cilostamide-, and 9.8 in A-134974-treated larvae. A few red-only β-cells remained, but their number did not significantly change with the different treatments (Figure S2A–C). The ins:Kaede transgene was no longer actively transcribed in the red-only cells, suggesting that these β cells were critically damaged by MTZ. To further test whether these compounds might act by increasing survival of newly formed β-cells, we introduced a transitional day between β-cell ablation and drug treatment. This time lag allowed the MTZ-induced ablation to conclude and MTZ to be fully cleared before the compounds were added. NECA, Cilostamide, and A-134974 all increased β-cell regeneration equally potently with or without the transitional day (Figure S2D–F). Together, these findings show that the hit-compounds increase regeneration rather than survival of β-cells. For the rest of the study, we focused on the most potent enhancer of β-cell regeneration, the adenosine agonist NECA.
Figure 2. The hit-compounds increase regeneration, not survival, of β-cells.
(A) Schematic diagram for cell-labeling and assessment of β-cell survival/regeneration. To examine β-cell survival, we made use of the photo-convertible property of the fluorescent protein Kaede. At 3 dpf, before ablating the β-cells with MTZ from 3–4 dpf, we converted Tg(ins:Kaede)-expressing β-cells from green to red by exposing them to UV-light. After two days of regeneration (6 dpf), the surviving β-cells are red and green (yellow overlap), whereas the newly formed β-cells are green-only.
(B & C) Confocal images of DMSO- and NECA-treated larvae with Tg(ins:Kaede)-expressing β-cells at 6 dpf. Note that there is one β-cell that survived the ablation in this particular DMSO-treated larva (arrow in B), whereas there are no β-cells that survived the ablation in this NECA-treated larva (C).
(D) Quantification of β-cell regeneration (green bars) and β-cell survival (yellow bars) per larva at 6 dpf, following treatment with DMSO, NECA, Cilostamide, or A-134974 from 4–6 dpf. P < 0.0001; n = 10 larvae for each group. Error bars represent SEM. See also Figure S2.
NECA potently increases regeneration of β-cells by promoting their proliferation
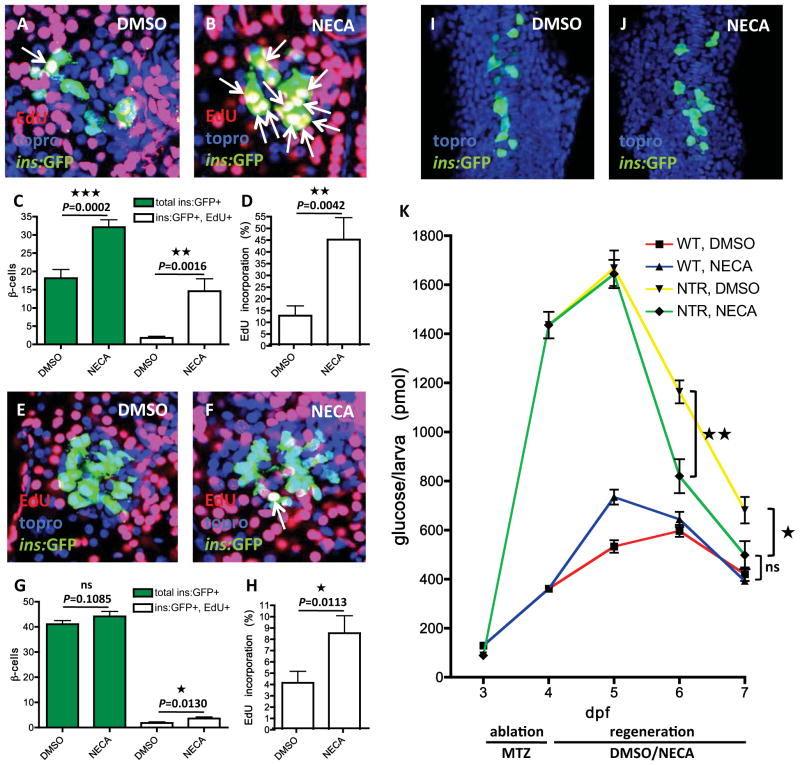
The NECA-induced increase in β-cell number could result from neogenesis, proliferation, or both. To determine effects on proliferation, we examined the incorporation of EdU as an indicator of DNA replication. We exposed Tg(ins:CFP-NTR);Tg(ins:GFP) larvae to MTZ from 3–4 dpf and then incubated them with EdU in the presence of vehicle or NECA from 4–6 dpf (Figure 3A–B). Vehicle-treated larvae had an average of 18 regenerated β-cells, of which only 2 had incorporated EdU, whereas NECA-treated larvae had an average of 32 β-cells, of which 15 had incorporated EdU (Figure 3C). Thus, the rate of EdU incorporation in β-cells increased from 13% in vehicle-treated larvae to 45% in NECA-treated larvae (Figure 3D). These data indicate that, although proliferation also contributes, β-cell neogenesis is the major endogenous mechanism underlying β-cell regeneration at this stage in control larvae, and that NECA enhances regeneration by markedly increasing the proportion of new β-cells that proliferate. A longer treatment with NECA, i.e. from 4–8 dpf after β-cell ablation from 3–4 dpf, showed that NECA does not lead to an overshoot in β-cell number and that the endogenous regeneration in vehicle-treated larvae catches up with that in NECA-treated larvae (at 8 dpf, 31 ± 3.3 β-cells for vehicle, n = 10 larvae; 42 ± 3.2 β-cells for NECA, n = 10 larvae; ± SEM, P = 0.0152). These findings indicate that NECA promotes β-cell proliferation mainly during the most active period of β-cell regeneration.
Figure 3. NECA has a modest effect on normal β-cell development, but strongly accelerates β-cell regeneration.
(A–D) Tg(ins:GFP);Tg(ins:CFP-NTR) larvae were treated with MTZ from 3–4 dpf to ablate the β-cells, and subsequently treated with DMSO/NECA and EdU during regeneration from 4–6 dpf. EdU was developed in red and nuclei were counterstained with topro (in blue). (A) Confocal image of a 6 dpf DMSO-treated larva displaying one β-cell that had incorporated EdU (arrow) during regeneration from 4–6 dpf. (B) Confocal image of a 6 dpf NECA-treated larva displaying 10 β-cells that had incorporated EdU (arrows) during regeneration from 4–6 dpf. (C) Quantification of the total number of β-cells (green bars) and β-cells that had incorporated EdU (white bars) per larva during DMSO or NECA treatment from 4–6 dpf during β-cell regeneration. (D) Rate of EdU incorporation, displayed as the percentage of β-cells that incorporated EdU. n = 18 larvae for each group.
(E–H) Tg(ins:GFP) larvae were treated with DMSO/NECA and EdU from 4–6 dpf to assess EdU incorporation in β-cells during normal development. EdU was developed in red and nuclei were counterstained with topro (in blue). (E) Confocal image of a 6 dpf DMSO-treated larva showing no EdU incorporation in its β-cells between 4–6 dpf of normal development. (F) Confocal image of a 6 dpf NECA-treated larva displaying one β-cell that had incorporated EdU between 4–6 dpf of normal development. (G) Quantification of the total number of β-cells (green bars) and β-cells that had incorporated EdU (white bars) per larva during DMSO or NECA treatment from 4–6 dpf of normal development. (H) Rate of EdU incorporation, displayed as the percentage of β-cells that incorporated EdU. n = 25 larvae for DMSO-treated, and n = 32 larvae for NECA-treated.
(I–J) During normal development of the dorsal pancreatic bud, Tg(ins:GFP) embryos were treated with DMSO or NECA from 6–18 hpf. Nuclei were counterstained with topro (blue). (I) Confocal projection of a DMSO-treated Tg(ins:GFP) embryo at 18 hpf. (J) Confocal projection of a NECA-treated Tg(ins:GFP) embryo at 18 hpf. We observed no significant difference in the number of β-cells.
(K) Free-glucose levels during β-cell regeneration in the presence of DMSO or NECA. After being treated with MTZ from 3–4 dpf, the larvae either had an islet depleted of β-cells owing to the expression of Tg(ins:CFP-NTR) (NTR), or a normal islet because they were wild-type (WT). The larvae were subsequently treated with DMSO or NECA from 4–7 dpf. Free-glucose levels were significantly lower in NTR larvae treated with NECA (green line) than in NTR larvae treated with DMSO (yellow line), after treatment for 2 days (from 1164 to 820 pmol/larva, P = 0.0031) and 3 days (from 681 to 498 pmol/larva, P = 0.0292) days. n = 40 larvae (4 pools of 10 larvae) per data point. Error bars represent SEM. See also Figure S3.
NECA increases β-cell formation only modestly during normal development
We next assessed the effect of NECA on β-cell formation during normal development, rather than regeneration, of the pancreas. To examine the total number of β-cells, as well as their proliferative potential, we exposed Tg(ins:GFP)-expressing larvae to EdU in the presence of vehicle or NECA from 4–6 dpf. Although NECA did not significantly increase the total number of β-cells (41 β-cells in vehicle-treated larvae and 44 in NECA-treated larvae), it caused a small but significant increase in the number of β-cells that incorporated EdU (Figure 3E–G). An average of 1.8 β-cell per larva incorporated EdU in vehicle-treated larvae, whereas an average of 3.6 β-cells per larva incorporated EdU in NECA-treated larvae, translating to a doubling of the rate of EdU incorporation (Figure 3H).
Because the pancreas forms sequentially from two different buds (Field et al., 2003), we also examined the effect of NECA on the formation of β-cells at the earliest stage of pancreas development. The dorsal pancreatic bud generates β-cells from 14–18 hours post fertilization (hpf), whereas the ventral pancreatic bud generates β-cells from 33 hpf onwards (Field et al., 2003). When we treated Tg(ins:GFP)-expressing embryos with vehicle or NECA from 6–18 hpf, we found no difference in the number of dorsal bud-derived β-cells (Figure 3I–J), (25.1 ± 1.3 β-cells for vehicle, n = 19 embryos; 26.6 ± 1.4 β-cells for NECA, n = 20 embryos; ± SEM, P = 0.2256). NECA’s ability to increase proliferation substantially during β-cell regeneration (Figure 3A–C), but only modestly during normal development (Figure 3E–G), was independent of glucose levels and β-cell age (Figure S3). It may be that NECA’s differential effect is important for restoring an optimal number of β-cells.
NECA accelerates restoration of a normal free-glucose level
We examined the functionality of the regenerated β-cells by testing the ability of NECA to restore normoglycemia in our β-cell regeneration model. By measuring free glucose, i.e., glucose that has not been phosphorylated intracellularly by hexokinases, we get an estimate of glycemia. Using this assay, we found that the total amount of free glucose increases as the larva grows from 3–6 dpf; after this time, the yolk dwindles and the amount of free glucose starts to decrease. Tg(ins:CFP-NTR) and wild-type larvae were exposed to MTZ from 3–4 dpf and then allowed to recover from 4–7 dpf in the presence of vehicle or NECA. Free glucose was measured from pooled clutches every 24 hours throughout the experiment (Figure 3K). We found that free-glucose levels were 4 times higher after β-cell ablation, and that both the vehicle-treated and the NECA-treated larvae recovered their glucose control to a great extent from 5–7 dpf. However, normal levels of free glucose were restored significantly faster in the NECA-treated larvae than in the vehicle-treated controls. Furthermore, at the end of the experiment (i.e., after 3 days of regeneration), there was no significant difference in free-glucose levels between larvae depleted of β-cells and then treated with NECA, and larvae that had not been depleted of β-cells. In contrast, larvae depleted of β-cells and then treated with vehicle still had increased levels of free glucose at this stage. Thus, the NECA-induced increase in β-cells generates a functional response that accelerates restoration of normal free-glucose levels.
NECA enhances proliferation of β-cells, and not cell proliferation in general
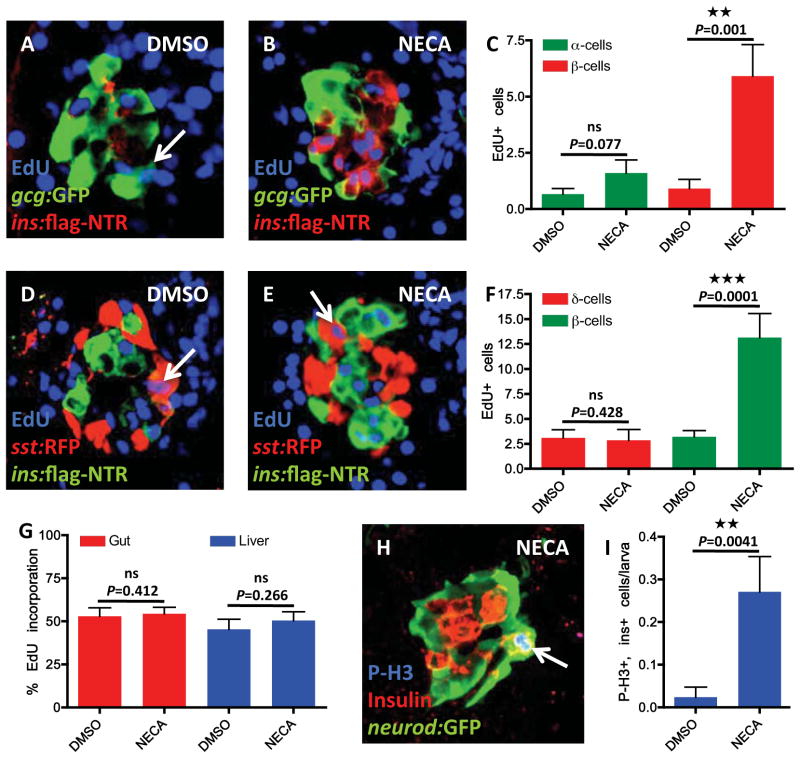
To determine whether NECA increases proliferation of β-cells specifically, or whether it elicits a general increase in cell proliferation, we examined its effect on several other endocrine cells and tissues. To examine the effect of NECA on glucagon-producing α-cells during regeneration of β-cells, we exposed Tg(ins:flag-NTR);Tg(gcg:GFP) larvae to MTZ from 3–4 dpf and then treated them with EdU in the presence of vehicle or NECA from 4–6 dpf. NECA did not significantly increase EdU incorporation in α-cells (Figure 4A–C). The α-cells that did incorporate EdU were often located in the periphery of the islet and had low Tg(gcg:GFP)-expression (Figure 4A), indicating that they were newly differentiated. Importantly, in the same set of larvae, NECA significantly increased EdU incorporation in β-cells (Figure 4C). We also examined whether NECA increased proliferation of somatostatin-producing δ-cells during regeneration of β-cells. By exposing Tg(ins:flag-NTR);Tg(sst:RFP) larvae to MTZ from 3–4 dpf and then incubating them with EdU in the presence of vehicle or NECA from 4–6 dpf, we found that NECA markedly increased EdU incorporation in β-cells (Figure 4F) but did not increase EdU incorporation in δ-cells (Figure 4D–F). These findings indicate that, during β-cell regeneration, NECA increases β-cell proliferation without inducing a general increase in proliferation of other pancreatic endocrine cell types.
Figure 4. NECA enhances proliferation of β-cells, and not cell proliferation in general.
(A–C) Proliferation of α-cells was assessed by EdU incorporation in Tg(gcg:GFP)-expressing α-cells. Tg(gcg:GFP);Tg(ins:flag-NTR) larvae were treated with MTZ from 3–4 dpf for ablation of the β-cells, and subsequently treated with DMSO/NECA and EdU during β-cell regeneration from 4–6 dpf. (A) Confocal image of a DMSO-treated larva displaying one α-cell that had incorporated EdU (arrow). (B) Confocal image of a NECA-treated larva that does not show any EdU incorporation in α-cells, but substantial EdU incorporation in β-cells (positive for Tg(ins:flag-NTR) expression). (C) Quantification of EdU incorporation in α-cells (green bars) and β-cells (red bars) in the presence of DMSO or NECA. Note that the number of α-cells that incorporated EdU during treatment with NECA did not significantly increase, although incorporation of EdU in Tg(ins:flag-NTR)-expressing β-cells increased markedly in the same larvae. n = 11 larvae for each group.
(D–F) Proliferation of δ-cells was assessed by EdU incorporation in Tg(sst:RFP)-expressing δ-cells. Tg(sst:RFP);Tg(ins:flag-NTR) larvae were treated with MTZ from 3–4 dpf for ablation of the β-cells, and subsequently treated with DMSO/NECA and EdU during β-cell regeneration from 4–6 dpf. (D) Confocal image of a DMSO-treated larva displaying one δ-cell that had incorporated EdU (arrow). (E) Confocal image of a NECA-treated larva displaying one δ-cell that had incorporated EdU, and substantial EdU incorporation in β-cells (positive for Tg(ins:flag-NTR) expression). (F) Quantification of EdU incorporation in δ-cells (red bars) and β-cells (green bars) in the presence of DMSO or NECA. n = 10 larvae for each group.
(G) Quantification of proliferation in the gut and liver in Tg(ins:CFP-NTR) larvae following β-cell ablation from 3–4 dpf and subsequent treatment with DMSO/NECA from 4–6 dpf. The percentage of cells that incorporated EdU between 4–6 dpf during β-cell regeneration did not change with NECA treatment when compared to DMSO-treated controls. n = 10 larvae for each group. (H) The mitosis marker Phospho-Histone H3 (P-H3) co-labels a β-cell that expresses both insulin and Tg(neurod:GFP) (arrow) following β-cell ablation from 3–4 dpf and subsequent treatment with NECA from 4–6 dpf. Note that the β-cell that is positive for P-H3 is located in the periphery of the islet.
(I) Quantification of β-cell proliferation, as marked by co-localization of insulin and P-H3, following β-cell ablation from 3–4 dpf and subsequent treatment with DMSO/NECA from 4–6 dpf. n = 42 larvae for DMSO-treated; n = 48 larvae for NECA-treated. Error bars represent SEM. See also Figures S4 & 5.
To examine proliferation of other tissues, we assessed EdU incorporation in the gut, liver, and neural compartments after β-cell ablation from 3–4 dpf and subsequent vehicle or NECA treatment from 4–6 dpf. There was no significant difference between vehicle- and NECA-treated larvae at 6 dpf in the number of cells that had incorporated EdU in the liver or gut (Figure 4G), or in neural compartments such as the diencephalon, cerebellum, vagal ganglia and posterior lateral line ganglia (Figure S4). Thus, NECA increases β-cell proliferation during β-cell regeneration, but does not induce a general increase in proliferation of other cell types and tissues.
We also assessed pan-endocrine proliferation, as well as principal and secondary islet morphology, by studying the Tg(neurod:GFP) line during normal development. Treating developing Tg(neurod:GFP) larvae with NECA from 4–6 dpf did not significantly affect the diameter of the principal islet, the number of secondary islets, or the proliferation of endocrine cells in the principal islet (Figure S5).
Because EdU incorporation does not distinguish between β-cells that are actively proliferating and those that proliferated as β-cell progenitors, we assessed phosphorylation of Histone H3 (P-H3) as a marker of mitosis. Examining 42 vehicle-treated larvae two days into regeneration, we found only one β-cell that was P-H3+ (on average 0.02 β-cell per larva; Figure 4I), and 9 endocrine cells labeled by the pan-endocrine marker Tg(neurod:GFP) that were P-H3+. In contrast, examining 48 NECA-treated larvae two days into regeneration, we found 13 β-cells that were P-H3+ (on average 0.27 β-cell per larva; the NECA-treated larvae all had 0–2 P-H3+ β-cells; Figure 4I). These proliferating β-cells were most often located in the periphery of the islet (Figure 4H). A similar number of cells was double-positive for the pan-endocrine marker Tg(neurod:GFP) and P-H3, indicating that other endocrine cells do not proliferate to a great extent in response to NECA treatment (15 Tg(neurod:GFP)-positive endocrine cells in 45 larvae). Thus, β-cells normally proliferate very slowly at this stage of regeneration, but can proliferate rapidly in the presence of NECA. Altogether, these data show that NECA-activated adenosine signaling promotes β-cell regeneration by inducing β-cell proliferation.
NECA mediates its proliferative effect via the adenosine receptor A2aa
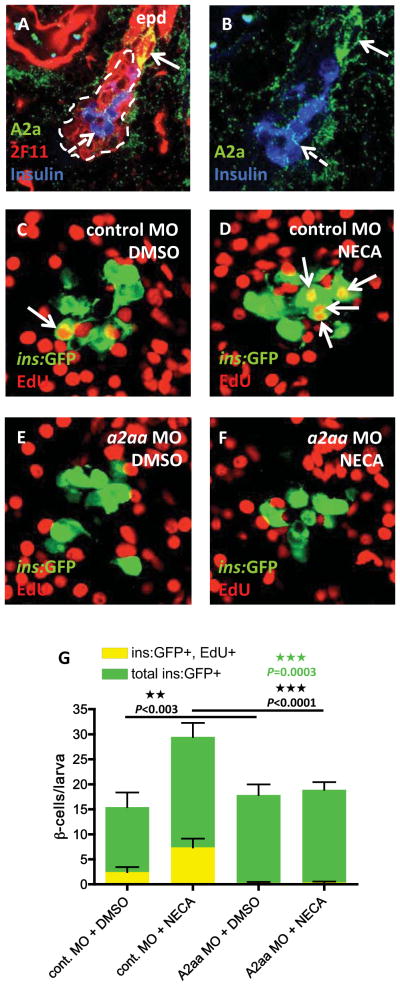
There are four adenosine receptors, A1, A2a, A2b, and A3. A1 and A3 couple primarily to Gαi and thus decrease intra-cellular levels of cAMP, whereas A2a and A2b couple primarily to Gαs and thus increase levels of cAMP. Because we also identified two hit-compounds with phosphodiesterase inhibitory activity, which increases cAMP levels, we focused on A2a and A2b as candidate receptors that boost β-cell regeneration. We examined their expression in the pancreas by immunohistochemical analysis of 5 dpf larvae. We found that the A2a receptor is highly expressed in cells budding off from the tip of the extra-pancreatic duct, which has been shown to contain endocrine progenitors (Dong et al., 2007) (Figure 5A–B). The A2a receptor was also expressed, albeit at lower levels, in β-cells and in cells scattered throughout the exocrine pancreas. This expression pattern correlates with, and could allow for, the effect of NECA on β-cell proliferation. To test this hypothesis directly, we targeted the A2a receptor with an antisense morpholino (MO). The a2a receptor gene is duplicated in zebrafish (a2aa and a2ab), but we could detect only the a2aa transcript in the pancreas (data not shown), and therefore designed a splice MO against this transcript. We confirmed that the MO inhibits a2aa mRNA splicing (Figure S6), and then tested whether the A2aa receptor mediates the effect of NECA on β-cell regeneration by injecting the a2aa MO into one-cell stage Tg(ins:CFP-NTR);Tg(ins:GFP) embryos, exposing them to MTZ from 3–4 dpf, and incubating them with EdU in the presence of vehicle or NECA from 4–6 dpf. NECA-treated larvae injected with a control MO had a greater number of regenerated β-cells at 6 dpf than NECA-treated larvae injected with the a2aa MO (Figure 5D,F,G). Importantly, the a2aa MO abolished NECA’s proliferative effect during β-cell regeneration, leading to a decrease in the number of β-cells that incorporated EdU (Figure 5C–G). The a2aa MO also significantly decreased the number of β-cells that incorporated EdU in vehicle-treated controls (from 2.5 to 0.3), suggesting that adenosine constitutes an endogenous signal that promotes β-cell proliferation during regeneration (Figure 5C,E,G). Together, these data indicate that NECA signals via the A2aa receptor to mediate its proliferative effect during β-cell regeneration in zebrafish.
Figure 5. The adenosine receptor A2aa mediates the regenerative effect of NECA.
(A) Confocal image of the expression of the A2a adenosine receptors in a 5 dpf larva. The 2F11 antibody marks the extra-pancreatic duct (epd) as well as the endocrine islet (outlined by the dashed line). High expression of A2a is found in cells budding off the epd (arrow), and low expression is found in insulin-expressing β-cells (dashed arrow) and cells scattered throughout the exocrine pancreas. (B) For clarity, a magnified view of (A), without the red color, is displayed.
(C–G) Tg(ins:GFP);Tg(ins:CFP-NTR) embryos were injected with a control MO or an a2aa MO at the one-cell stage, and subsequently treated with MTZ from 3–4 dpf to ablate the β-cells, and DMSO/NECA and EdU during β-cell regeneration from 4–6 dpf. (C) Confocal image of a DMSO-treated control MO-injected larva displaying one β-cell that had incorporated EdU (arrow). (D) Confocal image of a NECA-treated control MO-injected larva displaying 4 β-cells that had incorporated EdU (arrows). (E) Confocal image of a DMSO-treated a2aa MO-injected larva where no β-cells had incorporated EdU. (F) Confocal image of a NECA-treated a2aa MO-injected larva where no β-cells had incorporated EdU. (G) Quantification of the total number of β-cells and the number of β-cells that incorporated EdU per larva during DMSO or NECA treatment of control MO-injected or a2aa MO-injected embryos. P-values in black refers to Tg(ins:GFP)+, EdU+ cells, whereas the P-value in green refers to total number of Tg(ins:GFP)+ cells. n = 11–25 larvae per group. Error bars represent SEM. See also Figure S6.
NECA stimulates β-cell proliferation and regeneration in mice
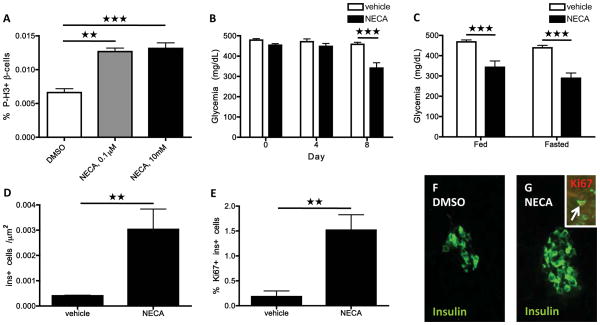
To determine whether NECA’s effect is conserved across species, we assessed NECA’s ability to increase β-cell proliferation in isolated mouse islets. After four days in culture, twice as many β-cells were labeled by P-H3 in the NECA-treated islets than in the vehicle-treated ones (Figure 6A), with NECA treatment increasing the percentage of P-H3-positive β-cells even at a low concentration.
Figure 6. NECA enhances murine β-cell proliferation and glucose control after STZ-induced diabetes.
(A) Proliferation rate of β-cells, displayed as percentage of P-H3-labeled β-cells, in isolated mouse islets following four days of vehicle or NECA treatment. P < 0.01 for 0.1 μM NECA; P < 0.001 for 10 μM NECA.
(B) Blood glucose levels after STZ-induced diabetes and subsequent treatment with vehicle or NECA for 8 days. The blood glucose levels were determined in fed mice. P < 0.001; n = 21 mice for vehicle-treated; n = 18 mice for NECA-treated.
(C) Blood glucose levels in fed and fasted state of STZ-injected mice after 15 days of treatment with vehicle or NECA. P < 0.001; n = 14 mice for vehicle-treated; n = 10 mice for NECA-treated.
(D) β-cell mass as determined by the number of insulin-positive cells per unit area (Ins+ cell per μm2) in STZ-injected mice after 15 days of control or NECA treatment. P = 0.0058; n = 5 mice per group.
(E) Proliferation of insulin-expressing cells in STZ-injected mice following vehicle or NECA treatment. Quantification of the percent of insulin-positive cells that co-labeled with Ki67. P = 0.0019; n = 5 mice per group.
(F–G) Representative images of islets in STZ-injected mice after 15 days of vehicle (F) or NECA treatment (G). A Ki67+ Ins+ cell (arrow) in a NECA-treated mouse is shown in the inset (G). Error bars represent SEM.
We also investigated whether NECA could increase β-cell regeneration in a mouse model of diabetes. To do so, we injected mice with streptozotocin (STZ) to deplete their β-cells and then treated the mice with daily injections of NECA or vehicle. Fed blood glucose levels were determined at 0, 4, and 8 days after the start of NECA treatment. By day 8, blood glucose levels were 30% lower in NECA-treated mice than vehicle-treated ones (Figure 6B). They were also significantly decreased after 15 days of NECA treatment (Figure 6C), at which time both fed and fasted blood glucose levels were decreased to an equal extent in NECA-treated compared to vehicle-treated mice. Importantly, after 15 days of treatment, the β-cell mass was 8 times larger in NECA-treated than in vehicle-treated mice (Figure 6D,F,G). Moreover, the β-cells in NECA-treated mice were also 8 times more likely to be Ki67+, indicating that they were proliferating at a higher rate (Figure 6E,G). There was no difference in the weight of the mice based on treatment, neither at the start nor at the end of the experiments (data not shown), indicating that the mice were not generally affected by NECA treatment. Together, these data indicate that NECA can improve management of β-cell trauma by enhancing β-cell regeneration via proliferation - not only in zebrafish larvae, but also in adult mice.
Discussion
In this study we identified five compounds that increase β-cell regeneration in zebrafish. Interestingly, despite using a large-scale, unbiased screening approach, we found that four of these compounds converge on the activation of the adenosine signaling pathway. Findings from complementary assays showed that the adenosine pathway promotes β-cell regeneration by increasing β-cell proliferation. Because the screen was performed in larval zebrafish, it was important to confirm these findings in a mammalian model. Using an adult model was also important because β-cell formation in mice is distinct during development, when there is active neogenesis from ductal progenitors, and postnatal life (Solar et al., 2009). Therefore, the effect of the most potent hit-compound, NECA, was examined in mice and found to increase both β-cell proliferation ex vivo and β-cell regeneration in vivo following STZ-induced β-cell death. These results indicate that adenosine GPCR signaling plays a previously unappreciated role in promoting β-cell regeneration.
Although our data suggest that adenosine signaling promotes β-cell regeneration by increasing β-cell proliferation, NECA may also simultaneously stimulate β-cell neogenesis. However, we did not observe any obvious sign of enhanced β-cell neogenesis such as increased formation of endocrine progenitors or β-cells along the ductal structures. Thus, NECA appears to increase β-cell proliferation without any overt upregulation of β-cell neogenesis, although one cannot completely rule out that NECA also stimulates β-cell neogenesis because it could potentially increase specifically within the islet. Adenosine is a cytoprotective and anti-inflammatory agent that can promote repair in a variety of tissues, most notably by stimulating angiogenesis during wound healing (Fredholm, 2007). It is a known stress signal that can be generated from the degradation of ATP released from dying cells (Elliott et al., 2009) and may therefore constitute an endogenous signal that increases β-cell proliferation during the regenerative response to β-cell death. In support of this hypothesis, we found that knock-down of the adenosine receptor A2aa decreased β-cell proliferation in regenerating control larvae. Moreover, we saw only a marginal effect of adenosine signaling during β-cell development but a major effect during their regeneration. Furthermore, adenosine stimulation of β-cell regeneration appeared to fade once normoglycemia was approached. This point is important because a regenerative therapy should not result in an overshoot in β-cell formation and hypoglycemia.
β-cells proliferate in response to a high physiological demand for insulin, such as in pregnancy and obesity. It is tempting to speculate that adenosine also participates in expanding β-cell mass under these conditions, because insulin is co-secreted with ATP (Jacques-Silva et al., 2010), which is in turn dephosphorylated to adenosine. ATP participates in a positive feedback loop that stimulates insulin secretion (Jacques-Silva et al., 2010), but it could also potentially serve as a source of adenosine and thereby increase β-cell proliferation. Interestingly, homeostatic control of β-cell mass is regulated by glucose metabolism in β-cells (Porat et al., 2011). Specifically, glucose metabolism increases intracellular ATP levels, which close KATP channels and depolarize β-cells, leading to increased β-cell proliferation. Our results raise the possibility that an increase in intracellular levels of ATP and in β-cell depolarization, leading to an increased secretion of insulin and ATP, enhances β-cell proliferation by increasing extracellular levels of adenosine. This model would be consistent with a paracrine action of adenosine stimulating adenosine receptors. In this context it is notable that adenosine has recently been shown to constitute a homeostatic signal that regulates proliferation of hematopoietic progenitors in Drosophila (Mondal et al., 2011), which, in light of our findings, suggests that adenosine has a more widespread role in maintaining tissue homeostasis than previously appreciated.
We identified inhibitors of PDE 3/4 as enhancers of β-cell regeneration, suggesting that signaling via GPCRs coupled to Gαs (which stimulates cAMP production) promotes β-cell regeneration, in agreement with previous studies (reviewed by Ackermann and Gannon, 2007). Of the four adenosine receptors, A2a and A2b are the ones that couple primarily to Gαs. In zebrafish, we detected high expression of the A2a receptor in cells budding off the extra-pancreatic duct, a known source of endocrine progenitors (Dong et al., 2007), and low expression in β-cells. This expression pattern correlates with that in the rat pancreas (Novak et al., 2008) and could explain NECA’s effect. In support of this notion, injection of a morpholino targeting the A2aa receptor abolished NECA’s ability to stimulate regeneration of β-cells. Therefore, it will be important to examine whether NECA or specific A2a receptor agonists are the most efficient promoters of β-cell regeneration and proliferation in mammalian models of diabetes.
Interestingly, NECA has been tested as a potential immunomodulator in mouse models of type 1 diabetes, in which it improved glucose homeostasis (Nemeth et al., 2007). β-cell proliferation was not assessed in that study and the data were interpreted as showing that NECA signals via the A2b receptor to increase β-cell survival by modulating the immune response. Moreover, adenosine and agonists of the A2a receptor improved glucose homeostasis after transplantation of islets to diabetic mice (Chhabra et al., 2010; Nitta et al., 2009), an effect that has been attributed to increased survival of the islet grafts (in the absence of data on β-cell regeneration or proliferation). Another report suggests that the A2b receptor mediates the immunomodulatory effects of adenosine that can potentially regulate β-cell survival, on the basis of a link between SNPs in the A2b locus and proinflammatory markers in diabetics (Figler et al., 2011). However, we found that NECA stimulates proliferation of mouse β-cells both ex vivo and in vivo following STZ-induced diabetes, supporting a direct effect on β-cells and not a relay mechanism via the immune system. Because our data show that the potent regenerative effect of NECA occurs via an A2a-dependent proliferative effect, and not via survival, we suggest that the A2a and A2b receptors have distinct functions that might both be favorable in the event of β-cell damage.
A recent report has shown that inhibition of adenosine kinase promotes rodent and porcine β-cell proliferation (Annes et al., 2012). We also identified an adenosine kinase inhibitor in our screen, but chose to focus our investigation on the adenosine agonist NECA because it was the most potent enhancer of β-cell regeneration in our assay. Because NECA is a receptor agonist and knock-down of the adenosine receptor A2aa abolishes NECA’s ability to stimulate β-cell proliferation, we propose that adenosine signals via receptors at the cell surface to generate a response that increases β-cell proliferation. However, Annes et al propose a nuclear role for adenosine kinase in promoting β-cell proliferation. It is thus possible that adenosine signaling has acquired several independent mechanisms by which it can promote β-cell proliferation.
The zebrafish model has been successfully used to conduct large-scale chemical screens for rapid identification of relevant signaling pathways in a variety of biological contexts. By using zebrafish for chemical screening in a regeneration context, we identified compounds that activate the adenosine pathway as enhancers of β-cell regeneration, findings that we subsequently confirmed in a mouse model of diabetes. The next step is to translate these findings, with the overarching goal of developing novel therapies for diabetes.
Experimental Procedures
Chemical screening
The β-cells were ablated in Tg(ins:CFP-NTR);Tg(ins:Kaede) larvae through treatment with 10 mM MTZ (Sigma) dissolved in eggwater supplemented with 1% DMSO (Sigma) and 0.2 mM 1-phenyl-2-thiourea (to inhibit pigment formation; TCI America) from 3–4 dpf. The larvae were subsequently transferred to 96-well plates for two days of chemical treatment, i.e. 4 larvae per well in a volume of 400 μL with 2–50 μM of the compounds from the chemical libraries. At 6 dpf, the larvae were anesthetized with Tricaine, and the number of β-cells counted using a wide-field Zeiss Z.1 inverted microscope.
Zebrafish experiments
Immunohistochemistry was performed according to standard procedures and analyzed using a Zeiss LSM5 Pascal confocal microscope. The whole endocrine portion of the pancreas was scanned through in every examined larva. Confocal stacks were analyzed using the ImageJ64 software. Proliferation was assessed by adding 10mM EdU to the eggwater, and using the Click-iT EdU imaging kit (Invitrogen). Free glucose was determined by grinding larvae in groups of 10 and using a glucose assay kit (BioVision). Knock-down of a2aa was performed by injection of 5 ng of a morpholino targeting the first coding exon-intron splice junction, (5′-3′) AGAAACACCCTTCACTCACCTAAGC (Gene Tools), at the one-cell stage. The standard control morpholino, (5′-3′) CCTCTTACCTCAGTTACAATTTATA (Gene Tools), was used as a reference.
Mouse experiments
For the ex vivo proliferation assay, picked islets were randomly dispersed in 6-well plates and treated with 0, 0.1 or 10 μM NECA for 4 days, then fixed and stained with anti-insulin and anti-P-H3. To induce diabetes, mice were injected intraperitoneally with 150 mg/kg STZ after a four hour fast once per day for two consecutive days. Tail vein blood was measured with a portable glucometer (FreeStyle Lite blood glucose monitoring system, Abbott Diabetes Care). Only mice that became diabetic based on fed blood glucose values of >300 mg glucose/dl were used. Mice were given daily intraperitoneal injections of PBS (vehicle) or 0.003 mg/kg NECA. 10 μm thick cryosections were made from the whole pancreas, and every 40th section was stained for Insulin and Ki67 for assessment of β-cell mass and proliferation.
Statistical analysis
Statistical analyses were carried out by one-tailed t-tests when two groups were analyzed, Anova when more than two groups were analyzed, and displayed as ± SEM. The glucose values in Figure 6 were analyzed with a Mann-Whitney test due to non-normal distribution of values, because a few blood glucose values reached the maximum level that the glucometer could record and were therefore assigned 500mg/dL.
Supplementary Material
Highlights.
An in vivo screen identifies adenosine signaling as a promoter of β-cell regeneration
The adenosine agonist NECA stimulates proliferation of β-cells in zebrafish and mouse
NECA boosts β-cell regeneration, but only weakly affects β-cell development
Adenosine receptor A2aa mediates NECA-enhanced regeneration of β-cells in zebrafish
Acknowledgments
We thank Vinh Nguyen and Katherine Yang for technical assistance with the mouse experiments, Alethia Villasenor, Dan Hesselson, Jennifer Hampton, and Tamsin Lindström for critical reading of the manuscript, and Matthias Hebrok for feedback during the project. We thank the SMDC at UCSF for advice as well as for the Iconix and the Microsource Spectrum small molecule libraries, and Tao Zhong for the Vanderbilt Collection. O.A. thanks the Wenner-Gren Fellows program for support. R.M.A. was supported by an advanced postdoctoral fellowship from the JDRF. This work was supported in part by grants from the NIH (β-cell biology consortium, U01DK089541, principal investigator M.S.G.; R01DK075032 to D.Y.R.S.), the JDRF (16-2007-428 to M.S.G.; 26-2008-636 to D.Y.R.S.), and the Packard Foundation to D.Y.R.S.
Footnotes
The authors declare no conflict of interests.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ackermann AM, Gannon M. Molecular regulation of pancreatic beta-cell mass development, maintenance, and expansion. J Mol Endocrinol. 2007;38:193–206. doi: 10.1677/JME-06-0053. [DOI] [PubMed] [Google Scholar]
- Ando R, Hama H, Yamamoto-Hino M, Mizuno H, Miyawaki A. An optical marker based on the UV-induced green-to-red photoconversion of a fluorescent protein. Proc Natl Acad Sci U S A. 2002;99:12651–12656. doi: 10.1073/pnas.202320599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Annes JP, Ryu JH, Lam K, Carolan PJ, Utz K, Hollister-Lock J, Arvanites AC, Rubin LL, Weir G, Melton DA. Adenosine kinase inhibition selectively promotes rodent and porcine islet beta-cell replication. Proceedings of the National Academy of Sciences of the United States of America. 2012;109:3915–3920. doi: 10.1073/pnas.1201149109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonner-Weir S, Li WC, Ouziel-Yahalom L, Guo L, Weir GC, Sharma A. Beta-cell growth and regeneration: replication is only part of the story. Diabetes. 2010;59:2340–2348. doi: 10.2337/db10-0084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chhabra P, Wang K, Zeng Q, Jecmenica M, Langman L, Linden J, Ketchum RJ, Brayman KL. Adenosine A(2A) agonist administration improves islet transplant outcome: Evidence for the role of innate immunity in islet graft rejection. Cell Transplant. 2010;19:597–612. doi: 10.3727/096368910X491806. [DOI] [PubMed] [Google Scholar]
- Curado S, Anderson RM, Jungblut B, Mumm J, Schroeter E, Stainier DY. Conditional targeted cell ablation in zebrafish: a new tool for regeneration studies. Dev Dyn. 2007;236:1025–1035. doi: 10.1002/dvdy.21100. [DOI] [PubMed] [Google Scholar]
- Dong PD, Munson CA, Norton W, Crosnier C, Pan X, Gong Z, Neumann CJ, Stainier DY. Fgf10 regulates hepatopancreatic ductal system patterning and differentiation. Nat Genet. 2007;39:397–402. doi: 10.1038/ng1961. [DOI] [PubMed] [Google Scholar]
- Dor Y, Brown J, Martinez OI, Melton DA. Adult pancreatic beta-cells are formed by self-duplication rather than stem-cell differentiation. Nature. 2004;429:41–46. doi: 10.1038/nature02520. [DOI] [PubMed] [Google Scholar]
- Elliott MR, Chekeni FB, Trampont PC, Lazarowski ER, Kadl A, Walk SF, Park D, Woodson RI, Ostankovich M, Sharma P, et al. Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature. 2009;461:282–286. doi: 10.1038/nature08296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Field HA, Dong PD, Beis D, Stainier DY. Formation of the digestive system in zebrafish. II. Pancreas morphogenesis. Dev Biol. 2003;261:197–208. doi: 10.1016/s0012-1606(03)00308-7. [DOI] [PubMed] [Google Scholar]
- Figler RA, Wang G, Srinivasan S, Jung DY, Zhang Z, Pankow JS, Ravid K, Fredholm B, Hedrick CC, Rich SS, et al. Links between insulin resistance, adenosine A2B receptors, and inflammatory markers in mice and humans. Diabetes. 2011;60:669–679. doi: 10.2337/db10-1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fredholm BB. Adenosine, an endogenous distress signal, modulates tissue damage and repair. Cell Death Differ. 2007;14:1315–1323. doi: 10.1038/sj.cdd.4402132. [DOI] [PubMed] [Google Scholar]
- Halban PA, German MS, Kahn SE, Weir GC. Current status of islet cell replacement and regeneration therapy. J Clin Endocrinol Metab. 2010;95:1034–1043. doi: 10.1210/jc.2009-1819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inada A, Nienaber C, Katsuta H, Fujitani Y, Levine J, Morita R, Sharma A, Bonner-Weir S. Carbonic anhydrase II-positive pancreatic cells are progenitors for both endocrine and exocrine pancreas after birth. Proc Natl Acad Sci U S A. 2008;105:19915–19919. doi: 10.1073/pnas.0805803105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacques-Silva MC, Correa-Medina M, Cabrera O, Rodriguez-Diaz R, Makeeva N, Fachado A, Diez J, Berman DM, Kenyon NS, Ricordi C, et al. ATP-gated P2X3 receptors constitute a positive autocrine signal for insulin release in the human pancreatic beta cell. Proc Natl Acad Sci U S A. 2010;107:6465–6470. doi: 10.1073/pnas.0908935107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinkel MD, Prince VE. On the diabetic menu: zebrafish as a model for pancreas development and function. Bioessays. 2009;31:139–152. doi: 10.1002/bies.200800123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krakowski ML, Kritzik MR, Jones EM, Krahl T, Lee J, Arnush M, Gu D, Mroczkowski B, Sarvetnick N. Transgenic expression of epidermal growth factor and keratinocyte growth factor in beta-cells results in substantial morphological changes. J Endocrinol. 1999;162:167–175. doi: 10.1677/joe.0.1620167. [DOI] [PubMed] [Google Scholar]
- Lavine JA, Attie AD. Gastrointestinal hormones and the regulation of beta-cell mass. Ann N Y Acad Sci. 2010;1212:41–58. doi: 10.1111/j.1749-6632.2010.05802.x. [DOI] [PubMed] [Google Scholar]
- Mondal BC, Mukherjee T, Mandal L, Evans CJ, Sinenko SA, Martinez-Agosto JA, Banerjee U. Interaction between differentiating cell- and niche-derived signals in hematopoietic progenitor maintenance. Cell. 2011;147:1589–1600. doi: 10.1016/j.cell.2011.11.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nemeth ZH, Bleich D, Csoka B, Pacher P, Mabley JG, Himer L, Vizi ES, Deitch EA, Szabo C, Cronstein BN, et al. Adenosine receptor activation ameliorates type 1 diabetes. Faseb J. 2007;21:2379–2388. doi: 10.1096/fj.07-8213com. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nitta T, Itoh T, Matsuoka N, Mera T, Kojima D, Nakano M, Yamashita Y, Yasunami Y. Prevention of early loss of transplanted islets in the liver of mice by adenosine. Transplantation. 2009;88:49–56. doi: 10.1097/TP.0b013e3181aa6c9b. [DOI] [PubMed] [Google Scholar]
- North TE, Goessling W, Walkley CR, Lengerke C, Kopani KR, Lord AM, Weber GJ, Bowman TV, Jang IH, Grosser T, et al. Prostaglandin E2 regulates vertebrate haematopoietic stem cell homeostasis. Nature. 2007;447:1007–1011. doi: 10.1038/nature05883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novak I, Hede SE, Hansen MR. Adenosine receptors in rat and human pancreatic ducts stimulate chloride transport. Pflugers Arch. 2008;456:437–447. doi: 10.1007/s00424-007-0403-3. [DOI] [PubMed] [Google Scholar]
- Pan FC, Wright C. Pancreas organogenesis: from bud to plexus to gland. Dev Dyn. 2011;240:530–565. doi: 10.1002/dvdy.22584. [DOI] [PubMed] [Google Scholar]
- Pisharath H, Rhee JM, Swanson MA, Leach SD, Parsons MJ. Targeted ablation of beta cells in the embryonic zebrafish pancreas using E. coli nitroreductase. Mech Dev. 2007;124:218–229. doi: 10.1016/j.mod.2006.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porat S, Weinberg-Corem N, Tornovsky-Babaey S, Schyr-Ben-Haroush R, Hija A, Stolovich-Rain M, Dadon D, Granot Z, Ben-Hur V, White P, et al. Control of pancreatic beta cell regeneration by glucose metabolism. Cell Metab. 2011;13:440–449. doi: 10.1016/j.cmet.2011.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rooman I, Bouwens L. Combined gastrin and epidermal growth factor treatment induces islet regeneration and restores normoglycaemia in C57Bl6/J mice treated with alloxan. Diabetologia. 2004;47:259–265. doi: 10.1007/s00125-003-1287-1. [DOI] [PubMed] [Google Scholar]
- Solar M, Cardalda C, Houbracken I, Martin M, Maestro MA, De Medts N, Xu X, Grau V, Heimberg H, Bouwens L, et al. Pancreatic exocrine duct cells give rise to insulin-producing beta cells during embryogenesis but not after birth. Developmental cell. 2009;17:849–860. doi: 10.1016/j.devcel.2009.11.003. [DOI] [PubMed] [Google Scholar]
- Thorel F, Nepote V, Avril I, Kohno K, Desgraz R, Chera S, Herrera PL. Conversion of adult pancreatic alpha-cells to beta-cells after extreme beta-cell loss. Nature. 2010;464:1149–1154. doi: 10.1038/nature08894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu G, Stoffers DA, Habener JF, Bonner-Weir S. Exendin-4 stimulates both beta-cell replication and neogenesis, resulting in increased beta-cell mass and improved glucose tolerance in diabetic rats. Diabetes. 1999;48:2270–2276. doi: 10.2337/diabetes.48.12.2270. [DOI] [PubMed] [Google Scholar]
- Xu X, D’Hoker J, Stange G, Bonne S, De Leu N, Xiao X, Van de Casteele M, Mellitzer G, Ling Z, Pipeleers D, et al. Beta cells can be generated from endogenous progenitors in injured adult mouse pancreas. Cell. 2008;132:197–207. doi: 10.1016/j.cell.2007.12.015. [DOI] [PubMed] [Google Scholar]
- Yu PB, Hong CC, Sachidanandan C, Babitt JL, Deng DY, Hoyng SA, Lin HY, Bloch KD, Peterson RT. Dorsomorphin inhibits BMP signals required for embryogenesis and iron metabolism. Nat Chem Biol. 2008;4:33–41. doi: 10.1038/nchembio.2007.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature. 2008;455:627–632. doi: 10.1038/nature07314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zon LI, Peterson RT. In vivo drug discovery in the zebrafish. Nat Rev Drug Discov. 2005;4:35–44. doi: 10.1038/nrd1606. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.