Abstract
Background. The link between endogenous estrogen, coronary artery disease (CAD), and death in postmenopausal women is uncertain. We analyzed the association between death and blood levels of estrone in postmenopausal women with known coronary artery disease (CAD) or with a high-risk factor score for CAD. Methods. 251 postmenopausal women age 50–90 years not on estrogen therapy. Fasting blood for estrone and heart disease risk factors were collected at baseline. Women were grouped according to their estrone levels (<15 and ≥15 pg/mL). Fatal events were recorded after 5.8 ± 1.4 years of followup. Results. The Kaplan-Meier survival curve showed a significant trend (P = 0.039) of greater all-cause mortality in women with low estrone levels (<15 pg/mL). Cox multivariate regression analysis model adjusted for body mass index, diabetes, dyslipidemia, family history, and estrone showed estrone (OR = 0.45; P = 0.038) as the only independent variable for all-cause mortality. Multivariate regression model adjusted for age, body mass index, hypertension, diabetes, dyslipidemia, family history, and estrone showed that only age (OR = 1.06; P = 0.017) was an independent predictor of all-cause mortality. Conclusions. Postmenopausal women with known CAD or with a high-risk factor score for CAD and low estrone levels (<15 pg/mL) had increased all-cause mortality.
1. Background
Endogenous estrogens are associated with greater vascular protection in premenopausal women [1]. Estradiol is the main and most active representative of endogenous estrogens in premenopausal women. This protection occurs through genomic and nongenomic actions [2]. The antiatherogenic and antithrombogenic effects of estradiol slow the process of atherosclerosis, and therefore cardiovascular events occur later in women compared to men [3]. In postmenopausal women, a significant reduction in estradiol production occurs due to ovarian failure. The natural protection ceases to exist, and at this stage, a gradual and progressive increase begins in cardiovascular events in women. However, hormone replacement therapy with estrogen and progesterone was not effective in primary or secondary prevention of cardiovascular events in postmenopausal women [4, 5]. Several hypotheses have been considered to justify these results, among them, the characteristics of exogenous estrogens, the high dose used, the association of progestins, and time since menopause [6]. It is known, however, that in postmenopausal women production of endogenous estrogen is very low. In this period, estrone is the main representative of the endogenous estrogens and is produced by several tissues, especially adipose tissue. Estrone is the result of the process of aromatization of androstenedione that occurs in fat cells [7]. Obese women have higher plasma concentrations of estrone [8]. However, the influence of estrone on cardiovascular protection in postmenopausal women is unknown. A previous study showed higher plasma levels of estrone in obese patients with coronary artery disease (CAD) or at high risk for coronary heart disease [9]. The same study also showed that increased plasma levels of estrone were associated with a nonsignificant trend toward a lower incidence of cardiovascular events. However, the follow-up period was only 2 years. The current study analyzed, in the same population, the effects of serum estrone in death from all causes in a long-term followup.
2. Methods
2.1. Study Populations
Participants were 251 postmenopausal women aged 50 to 90 years who had known CAD or were at high risk for CAD based on a risk factor score of 4 or more points, using the score developed for use in raloxifene trials [10]. Patients using postmenopausal hormone therapy and with advanced stage or clinically limiting disease were not included in the study. Women were patients in the ambulatory care clinic of a tertiary cardiology hospital, who were followed from March 2004 to December 2010. Primary end-point was death of any cause. Follow-up data were obtained through telephone calls and validated if given by a family member. Five patients were missed.
2.2. Clinical Characteristics
Baseline clinical characteristics included body mass index (BMI), blood pressure, medication use, and history of cardiac procedures and cardiac events. Laboratory tests for biologic CAD risk factors included fasting venous blood glucose, triglycerides, total cholesterol, HDL-cholesterol, LDL-cholesterol, C-reactive protein, and estrone. Risk factors for CAD were diabetes mellitus (fasting blood glucose ≥126 mg/dL [11]); hypertension (diastolic blood pressure ≥90 mm Hg [12]); dyslipidemia (triglycerides ≥200 mg/dL and/or LDL cholesterol ≥130 mg/dL [13]); family history (CAD occurring in parents before age 55 for men, 65 for women and siblings); at least 6 months of daily smoking of any kind or quantity of tobacco [14]; obesity status based on BMI (kg/m2) defined by the National Institutes of Health and the World Health Organization as normal (≥18.5 to <25), overweight (≥25 to <30), and obese (≥30) [15].
2.3. Laboratory Measurements
Morning blood was collected after a requested 12-hour fast. Estrone samples were measured on frozen plasma by a manual processing radioimmunoassay method using DSL 8700 (Diagnostic Systems Laboratories, Inc., Webster, TX, USA) with reference values 14.1–102.6 pg/mL for postmenopausal women. Based on the lower limit of the reference value in this laboratory, low estrone levels were defined as <15 pg/mL (43 patients) versus ≥15 pg/mL (208 patients). High sensitivity C-reactive protein was measured using an immunoturbidimetric ultrasensitive detection method, (Roche Diagnostics GmbH, D-68298 Mannheim, Germany), with a normal reference value <0.5 mg/dL.
The ethics committee of the Heart Institute (InCor) approved the study design, and all participants provided written informed consent.
2.4. Statistical Analysis
A sample size of at least 200 patients, with 100 subjects per treatment group, was determined to give 80% power to detect a 20% difference in the incidence of cardiovascular events with a 5% significance level.
The chi-square test was used for analysis of categorical variables, and Student's t test or 2-way ANOVA analysis was used for continuous variables. Mortality analyses used Kaplan-Meier estimation curves stratified by low or normal estrone levels (<15 and ≥15 pg/mL, resp.). Three models of Cox multivariate regression analysis were done. Model 1 was adjusted for age, hypertension, body mass index, diabetes, dyslipidemia, and estrone; model 2 was adjusted for hypertension, body mass index, diabetes, dyslipidemia, and estrone; model 3 was adjusted for body mass index, diabetes, dyslipidemia, and estrone. The statistical software used was SAS 9.2 (SAS Institute Inc, Cary, NC, USA).
3. Results
Baseline clinical and laboratory characteristics of the patients and group comparisons of estrone levels of <15 and ≥15 pg/mL are shown in Table 1. Estrone histogram is shown in Figure 1. Mean estrone levels were, respectively, 10.8 ± 2.1 and 27.6 ± 12.9 pg/mL (P < 0.001). Women with a low estrone level were older (72 ± 7.5 versus 69.2 ± 6.7 years; P = 0.016), thinner (27.9 ± 4.6 versus 30 ± 5.4 kg/m2; P = 0.020), and had less hypertension (95% versus 88%; P = 0.036), diabetes (37% versus 62%; P = 0.003), and lower triglyceride (148.9 ± 63.1 versus 178.5 ± 123.8 mg/dL; P = 0.023), glucose (107.5 ± 32.8 versus 137.3 ± 59.5 mg/dL; P = 0.002), and C-reactive protein levels (0.45 ± 0.55 versus 0.70 ± 1.31 mg/dL; P = 0.045). Women with BMI ≥30 kg/m2 were younger (70.5 ± 7.4 versus 68.7 ± 6.3; P = 0.046) and had more hypertension (92% versus 99%; P = 0.009), less diabetes (52% versus 31%; P = 0.001), and lower HDL (54.3 ± 13.2 versus 50.9 ± 11.3 mg/dL; P = 0.033) and LDL (117 ± 35.8 versus 107 ± 35.6 mg/dL; P = 0.029). Estrone was greater in patients with BMI ≥30 kg/m2 (22.4 ± 9.9 versus 28.2 ± 16.4; P = 0.001) (Table 2). Death was associated with higher age (69.2 ± 6.9 versus 72.6 ± 7.2 years; P = 0.010) and almost significant higher prevalence of hypertension (96% versus 88%; P = 0.051) and LDL plasma levels (111.2 ± 37.1 versus 121.3 ± 25.2 mg/dL; P = 0.054) (Table 3). There were 32 deaths, 27 (84%) attributed to cardiovascular disease (23 to CAD and 4 to stroke), 3 to pneumonia, and 2 to cancer (1 lung and 1 bone). The Kaplan-Meier survival curve showed a significant trend (P = 0.039) of greater all-cause mortality in women with low estrone levels (<15 pg/mL) (Figure 2). Three models of Cox regression analysis were done: model 1 was adjusted for age, body mass index, hypertension, diabetes, dyslipidemia, family history, and estrone; model 2 was adjusted for body mass index, hypertension, diabetes, dyslipidemia, family history, and estrone; model 3 was adjusted for body mass index, diabetes, dyslipidemia, family history, and estrone. In model 1, age (OR = 1.05 [95%CI: 1.00–1.11]; P = 0.016) was the only independent predictor of all-cause mortality; in model 2, hypertension (OR = 2.44 [95%CI: 0.83–7.20]; P = 0.030); in model 3, estrone (OR = 0.45 [95%CI: 0.21–0.95]; P = 0.038) (Table 4).
Table 1.
Baseline clinical characteristics and comparison of estrone <15 pg/mL and estrone ≥15 pg/mL in 251 patients.
| Baseline N = 251 | Estrone <15 pg/mL, N = 43 (17%) | Estrone ≥15 pg/mL, N = 208 (83%) | P | |
|---|---|---|---|---|
| Age (years) | 69.7 ± 7.0 | 72 ± 7.5 | 69.2 ± 6.7 | 0.016 |
| Body mass index (kg/m2) | 29.7 ± 5.3 | 27.9 ± 4.6 | 30 ± 5.4 | 0.020 |
| Hypertension | 238 (95) | 38 (88) | 200 (96) | 0.036 |
| Diabetes | 144 (57) | 16 (37) | 128 (62) | 0.003 |
| Dyslipidemia | 233 (93) | 40 (93) | 193 (93) | 0.956 |
| Family history | 170 (68) | 31 (72) | 139 (67) | 0.641 |
| Coronary artery disease | 189 (75) | 35 (81) | 154 (74) | 0.308 |
| Smoking | 25 (10) | 4 (10) | 21 (10) | 0.837 |
| SBP (mm Hg) | 141.8 ± 18.4 | 137.7 ± 21.2 | 142.6 ± 17.7 | 0.110 |
| DBP (mm Hg) | 83 ± 9.9 | 80.7 ± 10.8 | 83.5 ± 9.6 | 0.092 |
| Triglycerides (mg/dL) | 173.4 ± 116.1 | 148.9 ± 63.1 | 178.5 ± 123.8 | 0.023 |
| Total cholesterol (mg/dL) | 199.3 ± 41.1 | 200.7 ± 40.7 | 199 ± 41.2 | 0.808 |
| HDL cholesterol (mg/dL) | 52.8 ± 12.5 | 54.8 ± 13.1 | 52.4 ± 12.3 | 0.247 |
| LDL cholesterol (mg/dL) | 112.7 ± 36 | 116.1 ± 37.8 | 112 ± 35.7 | 0.496 |
| Glucose (mg/dL) | 132.2 ± 56.9 | 107.5 ± 32.8 | 137.3 ± 59.5 | 0.002 |
| C reactive protein (mg/dL) | 0.65 ± 1.22 | 0.45 ± 0.55 | 0.70 ± 1.31 | 0.045 |
| Estrone (pg/mL) | 24.9 ± 13.4 | 10.8 ± 2.1 | 27.6 ± 12.9 | <0.001 |
SBP: systolic blood pressure; DBP: diastolic blood pressure.
Figure 1.
Histogram of estrone plasma levels.
Table 2.
Baseline clinical characteristics and comparison of body mass index (BMI) <30 kg/m2 and ≥30 kg/m2 in 246 patients.
| IMC <30 kg/m2 (N = 141) | IMC ≥30 kg/m2 (N = 105) | P | |
|---|---|---|---|
| Age (years) | 70.5 ± 7.4 | 68.7 ± 6.3 | 0.046 |
| Hypertension | 129 (92) | 104 (99) | 0.009 |
| Diabetes | 73 (52) | 33 (31) | 0.001 |
| Dyslipidemia | 131 (93) | 97 (92) | 0.667 |
| Family history | 94 (67) | 76 (72) | 0.337 |
| Coronary artery disease | 36 (26) | 24 (23) | 0.629 |
| Smoking | 14 (10) | 11 (10) | 0.888 |
| SBP (mm Hg) | 139.6 ± 19.8 | 144.6 ± 15.9 | 0.026 |
| DBP (mm Hg) | 81.8 ± 10.4 | 84.67 ± 8.9 | 0.020 |
| Triglycerides (mg/dL) | 160.2 ± 81.0 | 190.9 ± 149.3 | 0.055 |
| Total cholesterol (mg/dL) | 202.9 ± 41.7 | 194.7 ± 39.9 | 0.116 |
| HDL cholesterol (mg/dL) | 54.3 ± 13.2 | 50.9 ± 11.3 | 0.033 |
| LDL cholesterol (mg/dL) | 117.0 ± 35.8 | 107.0 ± 35.6 | 0.029 |
| Glucose (mg/dL) | 128.6 ± 57.2 | 136.9 ± 56.3 | 0.249 |
| C reactive protein (mg/dL) | 0.25 ± 0.13 | 0.63 ± 0.57 | 0.779 |
| Estrone (pg/mL) | 22.4 ± 9.9 | 28.2 ± 16.4 | 0.001 |
SBP: systolic blood pressure; DBP: diastolic blood pressure.
Table 3.
Baseline clinical characteristics and comparison of 246 patients by survival status (32 deaths).
| Alive (N = 214) | Death (N = 32) | P | |
|---|---|---|---|
| Age (years) | 69.2 ± 6.9 | 72.6 ± 7.2 | 0.010 |
| Body mass index | 29.7 ± 5.2 | 28.3 ± 5.7 | 0.157 |
| Hypertension | 205 (96) | 28 (88) | 0.051 |
| Diabetes | 93 (43) | 13 (41) | 0.763 |
| Dyslipidemia | 200 (93) | 28 (88) | 0.227 |
| Coronary artery disease | 54 (25) | 6 (19) | 0.426 |
| Family history | 151 (71) | 19 (59) | 0.202 |
| Smoking | 23 (11) | 2 (6) | 0.432 |
| SBP (mm Hg) | 141.4 ± 18.2 | 143.1 ± 19.7 | 0.612 |
| DBP (mm Hg) | 82.9 ± 9.8 | 82.8 ± 10.2 | 0.938 |
| Triglycerides (mg/dL) | 172.2 ± 119 | 170.6 ± 101 | 0.908 |
| Total cholesterol (mg/dL) | 198.4 ± 41.9 | 204 ± 33.9 | 0.468 |
| HDL cholesterol (mg/dL) | 53.3 ± 12.8 | 50.6 ± 10.7 | 0.251 |
| LDL cholesterol (mg/dL) | 111.2 ± 37.1 | 121.3 ± 25.2 | 0.054 |
| Glucose (mg/dL) | 132.6 ± 56.4 | 131.3 ± 64.5 | 0.904 |
| C reactive protein (mg/dL) | 0.62 ± 1.04 | 0.85 ± 2.12 | 0.548 |
| Estrone (pg/mL) | 25.1 ± 13.6 | 21.6 ± 11.5 | 0.166 |
SBP: systolic blood pressure; DBP: diastolic blood pressure.
Figure 2.
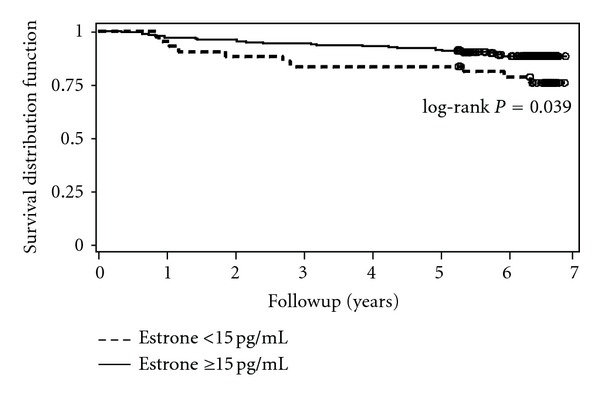
Kaplan-Meier curve for all causes of deaths according to estrone levels (<15 pg/mL versus ≥15 pg/mL).
Table 4.
Cox regression multivariate analysis for different models of variable adjustments.
| Model | Variable | Hazard ratio | 95% Confidence interval | P |
|---|---|---|---|---|
| 1 | Age | 1.05 | 1.00–1.11 | 0.016 |
| 2 | Hypertension | 2.44 | 0.83–7.20 | 0.030 |
| 3 | Estrone | 0.45 | 0.21–0.95 | 0.038 |
Model 1: adjusted for age, body mass index, hypertension, diabetes, dyslipidemia, family history, and estrone; 2: adjusted for body mass index, hypertension, diabetes, dyslipidemia, family history, and estrone; 3: adjusted for body mass index, diabetes, dyslipidemia, family history, and estrone.
4. Discussion
This study of long-term followup showed that serum estrone level <15 pg/mL was an independent risk factor for increased mortality from all causes in postmenopausal women with CHD or at high risk for CAD. Almost all deaths (84%) were from cardiovascular origin and from myocardial infarction. The current study confirmed the possible beneficial effects of estrone suggested by our previous study of short-term followup in the same population [9]. However, the association of estrone and CAD is unknown. Few studies have analyzed this association, and they have not shown any correlation between estrone plasma levels and CAD [16–18]. In a recent nested case-control study, estrone levels did not correlate with the incidence of CAD [19].
Estrone is the main endogenous estrogen in postmenopausal women. Nevertheless, estrone effects on the vascular system are significantly lower than estradiol mainly present in the premenopausal period [20]. Appropriate physiological levels of estrone in postmenopausal women may therefore bring additional benefits in cardiovascular protection. In postmenopausal women, a reduction occurs in the number and activity of estrogen receptors in tissues, and therefore more potent endogenous or exogenous estrogens are not necessary in this period. Likewise, the doses of exogenous estrogens are not physiological and are generally above the baseline needs. Estrogen therapy significantly increases the serum levels of estrone far above the levels observed in postmenopausal women. Estrone is also the main precursor of the small amount of estradiol still present in postmenopausal women. Estrone can then be the most suitable endogenous estrogen during the postmenopausal period for maintenance of basic physiological needs at this time. The estrone in postmenopausal women is produced mainly in adipose tissue, and obese women have higher serum estrone. However, obesity is associated with metabolic syndrome characterized by alterations in glucose metabolism, lipids, and presence of hypertension [21]. Inflammatory markers, such as CRP and leptin, are also increased in obese individuals. CRP is also increased in postmenopausal women who use hormone replacement therapy. In our study, we observed increased serum levels of CRP in the group of women with estrone ≥15 pg/mL. However, this increase may be due to the higher prevalence of hypertension, diabetes, serum triglycerides, and glucose in this group, suggesting a pattern of metabolic syndrome that is known to increase CRP. Being estrone protective of the cardiovascular system, the increased production of estrone in obese women may be a feedback mechanism for protecting the cardiovascular system in this population. In our study, estrone ≥15 pg/mL may be a factor to explain the lower mortality despite the high risk profile for cardiovascular patients. Higher estrone levels remained an independent variable even after adjustment for body mass index and prevalence of diabetes and dyslipidemia. The action of estrone in the metabolism of glucose and lipid is still controversial [22–25], but direct and indirect beneficial effects similar to those observed for estradiol, but of lesser intensity, may also occur with estrone in these 2 pathways. The prevalence of risk factors for CAD was significantly higher in our population. Diabetes was more prevalent in women with estrone levels ≥15 pg/mL (62% versus 37%, P = 0.003), but even with a worse cardiovascular risk profile [26], this was the group that had fewer deaths. In conclusion, serum estrone levels ≥15 pg/mL were associated with lower mortality from all causes in postmenopausal women with CHD or at high risk for CAD. Therefore, we hypothesized that the better prognosis was due to the possible protective effects of increased physiological levels of estrone. Nevertheless, this hypothesis should be validated by a large prospective study.
Authors' Contributions
A. P. Mansur, T. C. B. F. Silva, J. Y. Takada, and S. D. Avakian made substantial contributions to conception, design, analysis and interpretation of data. C. M. C. Strunz, L. A. M. César, J. M. Aldrighi and J. A. F. Ramires made substantial contributions to the design of the study and drafted the first version of the paper. All authors were involved in revising the drafts critically for important intellectual content and gave final approval of the version to be published.
Acknowledgment
The authors acknowledge the financial support provided by “Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP)” number 03/127152.
References
- 1.Xing D, Nozell S, Chen YF, Hage F, Oparil S. Estrogen and mechanisms of vascular protection. Arteriosclerosis, Thrombosis, and Vascular Biology. 2009;29(3):289–295. doi: 10.1161/ATVBAHA.108.182279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nilsson S, Mäkelä S, Treuter E, et al. Mechanisms of estrogen action. Physiological Reviews. 2001;81(4):1535–1565. doi: 10.1152/physrev.2001.81.4.1535. [DOI] [PubMed] [Google Scholar]
- 3.Murphy E, Steenbergen C. Gender-based differences in mechanisms of protection in myocardial ischemia-reperfusion injury. Cardiovascular Research. 2007;75(3):478–486. doi: 10.1016/j.cardiores.2007.03.025. [DOI] [PubMed] [Google Scholar]
- 4.Hulley S, Grady D, Bush T, et al. Randomized trial of estrogen plus progestin for secondary prevention of coronary heart disease in postmenopausal women. Journal of the American Medical Association. 1998;280(7):605–613. doi: 10.1001/jama.280.7.605. [DOI] [PubMed] [Google Scholar]
- 5.Group for the Women's Health Initiative Investigators Writing. Risks and benefits of estrogen plus progestin in healthy postmenopausal women. Journal of the American Medical Association. 2002;288:321–333. doi: 10.1001/jama.288.3.321. [DOI] [PubMed] [Google Scholar]
- 6.Harman SM, Vittinghoff E, Brinton EA, et al. Timing and duration of menopausal hormone treatment may affect cardiovascular outcomes. American Journal of Medicine. 2011;124(3):199–205. doi: 10.1016/j.amjmed.2010.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gruber CJ, Tschugguel W, Schneeberger C, Huber JC. Mechanisms of disease: Production and actions of estrogens. New England Journal of Medicine. 2002;346(5):340–352. doi: 10.1056/NEJMra000471. [DOI] [PubMed] [Google Scholar]
- 8.Lukanova A, Lundin E, Zeleniuch-Jacquotte A, et al. Body mass index, circulating levels of sex-steroid hormones, IGF-I and IGF-binding protein-3: a cross-sectional study in healthy women. European Journal of Endocrinology. 2004;150(2):161–171. doi: 10.1530/eje.0.1500161. [DOI] [PubMed] [Google Scholar]
- 9.Silva TCBF, Barrett-Connor E, Ramires JAF, Mansur AP. Obesity, estrone, and coronary artery disease in postmenopausal women. Maturitas. 2008;59(3):242–248. doi: 10.1016/j.maturitas.2008.01.008. [DOI] [PubMed] [Google Scholar]
- 10.Mosca L, Barrett-Connor E, Wenger NK, et al. Design and methods of the Raloxifene Use for The Heart (RUTH) study. American Journal of Cardiology. 2001;88(4):392–395. doi: 10.1016/s0002-9149(01)01685-x. [DOI] [PubMed] [Google Scholar]
- 11.Diabetes Association American. Diagnosis and classification of diabetes mellitus 2006. Diabetes Care. 2006;29(Suppl S)):S43–S48. [PubMed] [Google Scholar]
- 12.The Seventh Report of the Joint National Committee on Prevention, Detection, evaluation and treatment of high blood pressure. Journal of the American Medical Association. 2003;289:2560–2572. doi: 10.1001/jama.289.19.2560. [DOI] [PubMed] [Google Scholar]
- 13.III Diretrizes Brasileiras Sobre Dislipidemias e Diretriz de Prevenção da Aterosclerose do Departamento de Aterosclerose da Sociedade Brasileira de Cardiologia. Arquivos Brasileiros de Cardiologia. 2001;77:4–8. [PubMed] [Google Scholar]
- 14.Health Organization World. Guidelines for the conduct of the tobacco smoking surveys of the general population: report of a meeting. Geneva, Switzerland: WHO; 1992. [Google Scholar]
- 15.Obesity: Preventing and managing the global epidemic. Report of a WHO Consultation. Geneva, Switzerland: World Health Organization; 2000. (Technical Report Series, no. 894). [PubMed] [Google Scholar]
- 16.Cauley JA, Gutai JP, Glynn NW, Paternostro-Bayles M, Cottington E, Kuller LH. Serum estrone concentrations and coronary artery disease in postmenopausal women. Arteriosclerosis and Thrombosis. 1994;14(1):14–18. doi: 10.1161/01.atv.14.1.14. [DOI] [PubMed] [Google Scholar]
- 17.Barrett-Connor E, Goodman-Gruen D. Prospective study of endogenous sex hormones and fatal cardiovascular disease in postmenopausal women. British Medical Journal. 1995;311(7014):1193–1196. doi: 10.1136/bmj.311.7014.1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rexrode KM, Manson JE, Lee IM, et al. Sex hormone levels and risk of cardiovascular events in postmenopausal women. Circulation. 2003;108(14):1688–1693. doi: 10.1161/01.CIR.0000091114.36254.F3. [DOI] [PubMed] [Google Scholar]
- 19.Chen Y, Zeleniuch-Jacquotte A, Arslan AA, et al. Endogenous hormones and coronary heart disease in postmenopausal women. Atherosclerosis. 2011;216(2):414–419. doi: 10.1016/j.atherosclerosis.2011.01.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kuiper GGJM, Carlsson B, Grandien K, et al. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors and α and β . Endocrinology. 1997;138(3):863–870. doi: 10.1210/endo.138.3.4979. [DOI] [PubMed] [Google Scholar]
- 21.Grundy SM, Hansen B, Smith SC, Cleeman JI, Kahn RA. Clinical management of metabolic syndrome: report of the American Heart Association/National Heart, Lung, and Blood Institute/American Diabetes Association conference on scientific issues related to management. Arteriosclerosis, Thrombosis, and Vascular Biology. 2004;24(2):e19–e24. doi: 10.1161/01.ATV.0000112379.88385.67. [DOI] [PubMed] [Google Scholar]
- 22.Knopp RH, Zhu X, Bonet B. Effects of estrogens on lipoprotein metabolism and cardiovascular disease in women. Atherosclerosis. 1994;110:S83–S91. doi: 10.1016/0021-9150(94)05379-w. [DOI] [PubMed] [Google Scholar]
- 23.Ding EL, Song Y, Malik VS, Liu S. Sex differences of endogenous sex hormones and risk of type 2 diabetes: A systematic review and meta-analysis. Journal of the American Medical Association. 2006;295(11):1288–1299. doi: 10.1001/jama.295.11.1288. [DOI] [PubMed] [Google Scholar]
- 24.Lambrinoudaki I, Christodoulakos G, Rizos D, et al. Endogenous sex hormones and risk factors atherosclerosis in healthy Greek postmenopausal women. European Journal of Endocrinology. 2006;154(6):907–916. doi: 10.1530/eje.1.02167. [DOI] [PubMed] [Google Scholar]
- 25.Hsia J, Otvos JD, Rossouw JE, et al. Lipoprotein particle concentrations may explain the absence of coronary protection in the women’s health initiative hormone trials. Arteriosclerosis, Thrombosis, and Vascular Biology. 2008;28:1666–1671. doi: 10.1161/ATVBAHA.108.170431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Walton C, Godsland IF, Proudler AJ, Wynn V, Stevenson JC. The effects of the menopause on insulin sensitivity, secretion and elimination in non-obese, healthy women. European Journal of Clinical Investigation. 1993;23(8):466–473. doi: 10.1111/j.1365-2362.1993.tb00792.x. [DOI] [PubMed] [Google Scholar]


