Abstract
Directed specification and prospective isolation of chondrogenic paraxial mesoderm progeny from human pluripotent stem (PS) cells have not yet been achieved. Here we report the successful generation of KDR−PDGFRα+ progeny expressing paraxial mesoderm genes and the mesendoderm reporter MIXL1-GFP in a chemically defined medium containing the canonical WNT signaling activator, BMP-inhibitor, and the Nodal/Activin/TGFβ signaling controller. Isolated (GFP+)KDR−PDGFRα+ mesoderm cells were sensitive to sequential addition of the three chondrogenic factors PDGF, TGFβ and BMP. Under these conditions, the cells showed robust chondrogenic activity in micromass culture, and generated a hyaline-like translucent cartilage particle in serum-free medium. In contrast, both STRO1+ mesenchymal stem/stromal cells from adult human marrow and mesenchymal cells spontaneously arising from hPS cells showed a relatively weaker chondrogenic response in vitro, and formed more of the fibrotic cartilage particles. Thus, hPS cell-derived KDR−PDGFRα+ paraxial mesoderm-like cells have potential in engineered cartilage formation and cartilage repair.
Human joint cartilage lacks the intrinsic capacity to repair itself through regeneration by the resident chondrogenic progenitors of the articular cartilage (hyaline cartilage) that confers high resistance to repetitive compressive and shear forces. One promising treatment for repairing damaged cartilage is the transplantation of chondroprogenitor cells, such as the dedifferentiated chondrocytes and mesenchymal stem/stromal cells (MSCs), which are typically isolated from bone marrow. However, because of their limited supply, these adult stem/progenitor cells require ex vivo expansion to be used in current cellular therapies. This expansion reduces the cells' differentiation potential, increases the risk of oncogenic transformation of the cells1, and results in the production of fibrocartilage with poor biomechanical properties2.
The pluripotent embryonic stem (ES) cells and the recently established induced pluripotent stem (iPS) cells (designated collectively, pluripotent stem cells or PS cells) have considerable advantages over adult stem/progenitor cells, due to their capacities of unlimited proliferation and multi-lineage differentiation. Chondrogenesis occurs predominantly during embryogenesis, and chondrocytes are mainly derived from the precursors of the following three different embryonic cell lineages: sclerotome (somitic paraxial mesoderm), limb mesenchyme (lateral plate mesoderm), and ectomesenchyme (cranial neural crest). Because mouse (m)ES cell differentiation can mimic early embryogenesis3,4, we have postulated that human (h)PS cells can also be directed to differentiate into one of those embryonic chondrocyte precursor types. This differentiation would allow a large number of robustly chondrogenic cells to be obtained without expansion prior to transplantation or other applications.
Chondrocyte differentiation from hES cells has already been reported, but most of the research involves spontaneous differentiation culture that includes undefined medium components and/or long-term differentiation culture in the presence of other cell types; both types of culture obscure the underlying mechanisms and would produce artifacts5. Reports have revealed that mesodermal genes are expressed in parallel during the generation of chondrocytes or their precursors6,7. However, no report has confirmed this mesodermal origin by lineage tracing using genetic means and/or by fluorescence-activated cell sorting (FACS), and none has compared the chondrogenic activity between the progeny and the gold standard MSCs.
The specification of mesoderm from the pluripotent epiblast is tightly regulated by Wnt, bone morphogenetic protein (BMP) and Nodal/Activin/transforming growth factor-β (TGFβ) signaling during early embryogenesis. We previously reported that the activation of Wnt signaling and inhibition of BMP signaling lead to the effective specification from mES cells of chondrogenic somitic and/or rostral presomitic mesoderm that express platelet-derived growth factor receptor-α (PDGFRα) but not vascular endothelial growth factor receptor-2 (FLK1 or KDR)8. In this study, we report the signaling requirements for hPS cells to generate paraxial mesoderm in a chemically defined medium (CDM) and the cell surface markers that are effective in prospectively isolating paraxial mesoderm by FACS. From the investigation of the signaling requirements for the isolated mesoderm to generate cartilage particles in a serum-free medium, we also report that hPS cell-derived mesoderm has a potentially greater capacity to produce hyaline cartilage-like particles in culture than either adult bone marrow MSCs or the PS cell-derived mesenchymal progeny prepared using conventional methods.
Results
Specification of paraxial mesoderm from mouse and human PS cells by canonical WNT signaling without BMP signaling
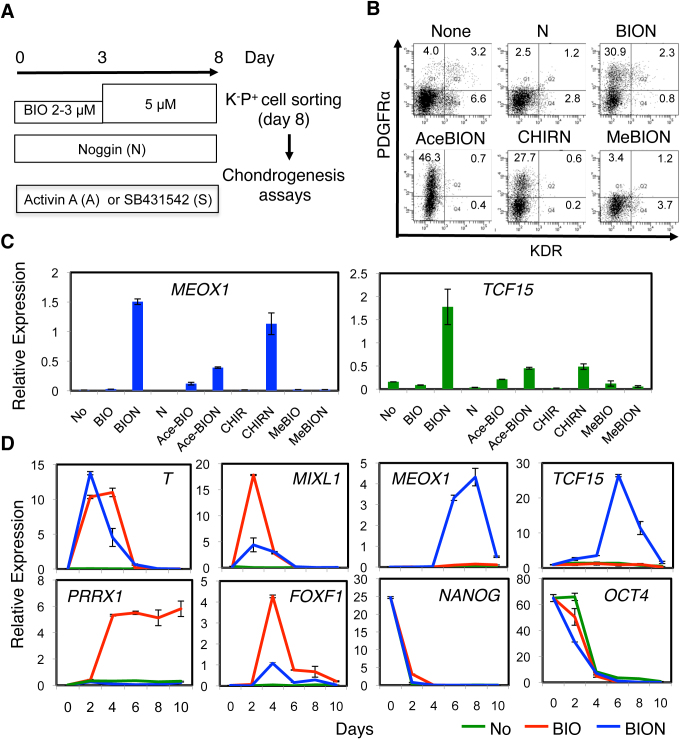
The specification of paraxial mesoderm from hES cells by the manipulation of signaling was monitored by the expression of the specific transcripts for MEOX1, MEOX2, PARAXIS (TCF15) and MESP2 and the emergence of the KDR−PDGFRα+ progeny. Mouse ES cells that were differentiated in CDM in the presence of WNT3a and Noggin gave rise to FLK1/KDR−PDGFRα+ paraxial mesoderm cells with robust chondrogenic activity8. However, H9 (Supplementary Fig. 1A) and Mixl1-GFP hES cells that differentiated in the presence of WNT3a+Noggin or WNT5a+Noggin in CDM generated few KDR−PDGFRα+ progeny and only poorly specified paraxial mesoderm as determined by the expression of MEOX1 and TCF15. WNT primarily exerts its biological effects through β-catenin-mediated transcription (i.e., the canonical pathway), which can also be activated by the inhibition of glycogen synthase kinase (GSK)3β, which triggers the degradation of β-catenin9. Therefore, we instead initiated the differentiation of H9 hES cell in the presence of a small-molecule GSK3 inhibitor (BIO) and Noggin (hereafter designated as BIO + Noggin or BION) (Fig. 1A). As a consequence, the proportion of KDR−PDGFRα+ cells (BION, Fig. 1B) and the levels of MEOX1 and TCF15 transcripts (BION, Fig. 1C, Supplementary Fig. 1A) by day 8 increased significantly. In the presence of Noggin, the GSK3 inhibitors Acetoxime-BIO (AceBIO) and CHIR99021 (CHIR) were also effective, but the inactive analogue of BIO, 1-methyl-BIO (MeBIO), was not effective (Fig. 1B, C). These results suggest that paraxial mesoderm specification during hES cell differentiation is achieved by the activation of canonical WNT signaling and the inhibition of BMP signaling.
Figure 1. Directed specification of paraxial mesoderm in a chemically defined medium.
(A) Differentiation protocol for hPS cells. (B, C) GSK3 inhibitor and Noggin-dependent specification of paraxial mesoderm and associated changes in the progenitor profile after 8 days of differentiation of H9 hES cells. (B) FACS analysis. (C) RT-PCR with MEOX1 and TCF15 primers. No, None; N, Noggin; AceBIO/Ace-BIO, Acetoxime-BIO; CHIR, CHIR99021; MeBIO, Methyl-BIO. (D) Time-dependent differentiation of H9 hES cells in the presence of no factors (No), BIO, and BIO + Noggin (BION). Specification of mesoderm was monitored by RT-PCR.
The emergence of the KDR−PDGFRα+ progeny from H9 hES cells was apparent from day 4 and reached a peak at around day 6 when differentiated in the presence of BIO + Noggin (Supplementary Fig. 1B). Consistently, the early mesendodermal markers T and MIXL1 showed a pattern of transient expression that peaked around day 2 to 3, while expression of MEOX1 and TCF15 increased from day 6 (blue, Fig. 1D). The removal of Noggin had no effect on the expression of T and elevated the expression of MIXL1 (red). In contrast, the removal of Noggin strongly induced the expression of the extraembryonic and/or lateral plate mesoderm genes FOXF1 and PRRX1 from around day 4 and suppressed that of MEOX1 and TCF15. In either case, the pluripotent stem cell marker genes NANOG and OCT4 were downregulated during differentiation.
Fine tuning of Nodal/Activin/TGFβ signaling for efficient specification of paraxial mesoderm from mouse and human PS cells
In addition to WNT and BMP signaling, Nodal signaling is involved in gastrulation and germ layer specification in a dose-dependent manner10. To further improve the specification of paraxial mesoderm, we adjusted the level of Nodal signaling during differentiation by titrating Activin A, a Nodal mimic, or SB431542, a small-molecule inhibitor of the Nodal/Activin/TGFβ receptor, against BIO + Noggin.
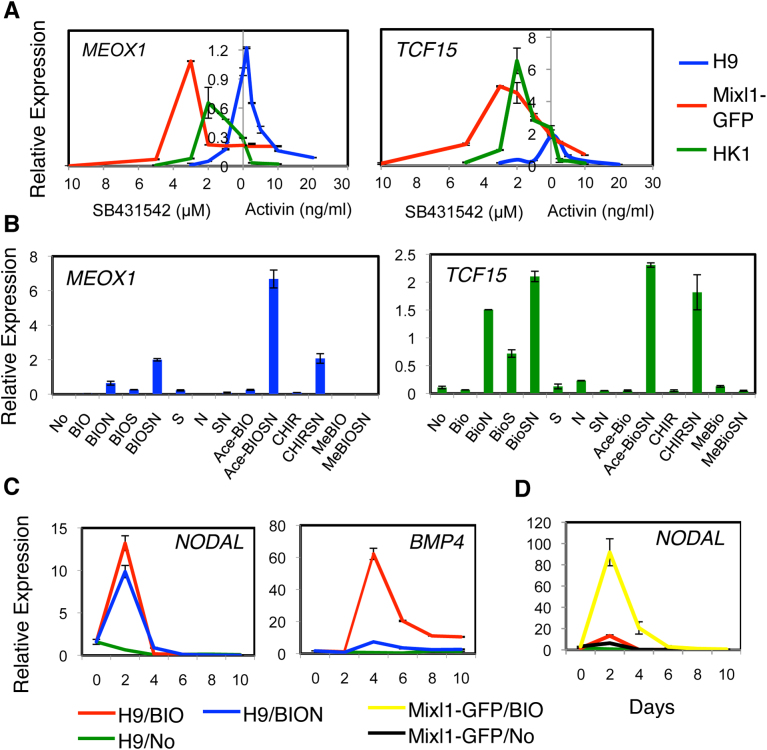
We first tested H9 and Mixl1-GFP hES cells11. When H9 hES cells were differentiated under varying concentrations of Activin A and SB431542 in the presence of BIO + Noggin, the expression profile of MEOX1 and TCF15 displayed a parabolic distribution with a peak of approximately 0 ng/ml Activin A/0 μM SB431542 (blue Fig. 2A). However, for the Mixl1-GFP hES cell progeny (red), the peak was reached at 2–3 μM SB431542 in the presence of BIO + Noggin (the condition hereafter referred to BIO + SB + Noggin or BIOSN). The BIO + SB (BIOS) or BIO + Noggin (BION) condition showed weaker enhancing effects on MEOX1 and TCF15 expression than did the BIOSN condition (Fig. 2B). Similarly, AceBIO and CHIR also induced MEOX1 and TCF15 expression in the presence of SB + Noggin (AceBioSN, CHIRSN).
Figure 2. Fine-tuning Nodal/Activin/TGFβ signaling for efficient specification of paraxial mesoderm.
(A) Activin A/SB431542 dose-dependency. H9 and Mixl1-GFP hES cells and HK1 hiPS cells were differentiated in the presence of BIO, Noggin, and various concentrations of Activin A or SB431542, and the MEOX1 and TCF15 transcripts were quantified by RT-PCR. (B) GSK3 inhibitor, Noggin and SB431542-dependent specification of paraxial mesoderm from Mixl1-GFP hES cells. RT-PCR was performed with MEOX1 and TCF15 primers. No, None; N, Noggin; AceBIO/Ace-BIO, Acetoxime-BIO; CHIR, CHIR99021; MeBIO, Methyl-BIO; S, SB431542. (C, D) Time-dependent expression of NODAL and BMP4. The H9 (C, D) and Mixl1-GFP (D) hES cells were differentiated with no factor (No), BIO or BIO + Noggin (BION), and subjected on the day indicated to RT-PCR.
The requirement to modulate Nodal/Activin/TGFβ signaling for the maximal specification of paraxial mesoderm in the presence of BIO + Noggin seems to apply to both mouse and human PS cells. The HK1 hiPS cells required SB431542 at 1–2 μM (i.e., BIO + SB + Noggin) (green, Fig. 2A). In contrast, the Bry-GFP mES cells required Activin A at 2–5 ng/ml (i.e., BIO + Activin + Noggin) (Supplementary Fig. 2A). The MEL1 hES cells were similar to the H9 hES cells because they did not require exogenous Activin or SB431542 for the specification of paraxial mesoderm (i.e., BIO + Noggin) (Supplementary Fig. 2B).
As in the case of mES cell differentiation8, the canonical WNT signaling activated by BIO induced the expression of NODAL and BMP4 during hPS cell differentiation (Fig. 2C, D). Although the BIO-induction of BMP4 was dependent on endogenous BMP activity as demonstrated by reduced expression in the presence of Noggin, the induction of NODAL was independent of such BMP activity as shown by the lack of effect of Noggin (BIO-red vs. BION-blue, Fig. 2C). However, the level of induced NODAL varied considerably among hPS cell lines. This variation appeared to correlate with the requirement for either Activin or SB432542 for the maximal specification of MEOX1-expressing paraxial mesoderm (Fig. 2D). For example, the Mixl1-GFP hES cells (yellow) required SB432542 for paraxial mesoderm specification and induced the NODAL transcript more than the H9 hES cells (red). Therefore, the level of WNT-induced NODAL expression may determine the requirement of exogenous Activin A or SB431542 for the maximal specification of paraxial mesoderm from hPS cells by canonical WNT signaling.
The KDR−PDGFRα+ progeny developed under BIO + Noggin +/− SB431542/Activin are mesendoderm derivatives
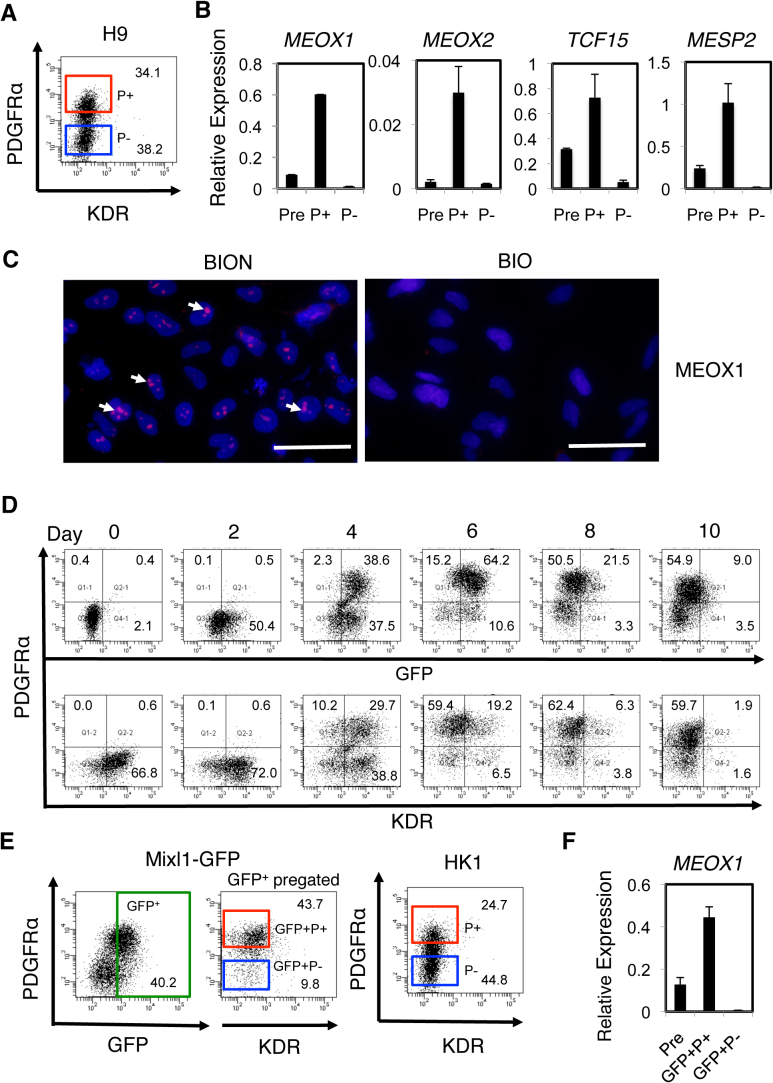
As for mES cells8, the KDR−PDGFRα+ progeny generated from hPS cells under conditions in which expression of MEOX1 and TCF15 is optimized, i.e. BIO + Noggin (BION) or BIO + SB + Noggin (BIOSN), may be enriched in paraxial mesoderm. In support, when KDR−PDGFRα+ and KDR−PDGFRα− progeny generated from H9 hES cells under BION conditions were isolated by FACS (Fig. 3A), the paraxial mesoderm transcripts, e.g., MEOX1, MEOX2, TCF15 and MESP2, were found to be enriched in the KDR−PDGFRα+ cell fraction (P+, Fig. 3B). Furthermore, after 3–5 days in culture, cells appeared that expressed MEOX1 protein at the correct intracellular locus of the nucleolus (BION, pink spots, Fig. 3C), suggesting that, analogous to mouse rostral paraxial mesoderm (FLK1/KDR−PDGFRα+)8, human paraxial mesoderm is also KDR−PDGFRα+. In addition, lateral plate/extraembryonic mesoderm was specified under BIO but not BION conditions (Fig. 1D), and MEOX1 protein was not detected in cells generated under BIO conditions (Fig. 3C). These observations also suggest that paraxial mesoderm but not lateral plate/extraembryonic mesoderm is responsible for the mesodermal activities of the KDR−PDGFRα+ progeny generated under BION or BIOSN conditions.
Figure 3. Prospective isolation of paraxial mesoderm and demonstration of their chondrogenic activity.
(A, B) Enrichment of paraxial mesoderm generated from H9 hES cells. H9 hES cells were differentiated under BION conditions, and total progeny (Pre) and the KDR−PDGFRα+ (P+, red gate in A) and KDR−PDGFRα− (P-, blue gate in A) progeny were isolated by FACS and subjected to RT-PCR (B). (C) MEOX1 protein expression. Mesodermal progeny, derived from H9 hES cells in the presence of BIO + Noggin (BION) or BIO alone (BIO) and cultured on a chamber slide for 5 day, were immunostained with the anti-MEOX1 antibody. The nucleoli staining pink within a blue nucleus, some of which are indicated with white arrows, are detectable in progeny generated under the BION condition. Isotype controls are shown in Supplementary Fig. 1C. Scale Bars, 50 μm. (D) Time-course of FACS analysis for MIXL1-GFP (GFP), KDR and PDGFRα expression during Mixl1-GFP hES cell differentiation induced in the presence of BIO + SB + Noggin (BIOSN). (E, F) Enrichment of paraxial mesoderm derived from other hPS cells. Mixl1-GFP hES cells and HK1 hiPS cells were differentiated under BIOSN conditions. From Mixl1-GFP hES cells, total EB cells (Pre) and the GFP+KDR−PDGFRα+ (GFP+P+ red gate) and GFP+KDR−PDGFRα− (GFP+P− blue gate) progeny were isolated by FACS (E) and subjected to RT-PCR using the MEOX1 primers (F). From HK1 hiPS cells, only the KDR−PDGFRα+ (P+) and KDR−PDGFRα− (P−) progeny were accumulated (E) as in the case of H9 hES cells.
The MIXL1-expressing early mesendoderm cells generated at the primitive streak are common precursors of mesoderm and definitive endoderm12. Later MIXL1 expression stays in the endoderm. The GFP (green fluorescence protein)+ EB cells from Mixl1-GFP hES cells were thus considered to be mesendoderm and/or mesendoderm derivatives such as early progenitors committed to mesoderm or endoderm, and definitive endoderm. FACS analyses showed that the conditions supporting paraxial mesoderm specification, i.e., BIOSN, AceBIOSN, or CHIRSN, resulted in the development of KDR−PDGFRα+ cells that were also GFP+ by 6–8 days of differentiation (Fig. 3D, Supplementary Fig. 3A). The GFP+ and PDGFRα+ progeny were first detected on day 2 and day 4, respectively, during differentiation under BIOSN conditions, and their relative proportions increased thereafter. Over 95% of the PDGFRα+ progeny generated were GFP+ on day 4, at which time 75% of them were also KDR+. The GFP level in the PDGFRα+ cell fraction diminished as the MIXL1 transcript level declined (Supplementary Fig. 3B) as did the KDR level, suggesting the presence of mesendoderm derivatives in the GFP+KDR−PDGFRα+ fraction at day 8. The overall kinetics of mesodermal gene expression and the concomitant loss of pluripotency transcripts, and the Noggin requirement for paraxial mesoderm gene expression were similar to those of the H9 hES cells (Supplementary Fig. 3B).
Furthermore, the GFP+KDR−PDGFRα+ mesendodermal cell fraction isolated from day 8 Mixl1-GFP EBs formed under BIOSN conditions (GFP+P+, Fig. 3E), was enriched in the MEOX1 transcript (Fig. 3F), as was seen with H9 hES cells. There was no sign of specification of lateral plate/extraembryonic mesoderm from the Mixl1-GFP hES cells under BIOSN conditions (Supplementary Fig. 3B). Therefore, the KDR−PDGFRα+ progeny that were also MIXL1-GFP+ were enriched in paraxial mesoderm.
These results indicate that the KDR−PDGFRα+ progeny induced from hES cells under the optimal condition for expression of MEOX1 and TCF15 (BION, BIOSN, etc) are mesendoderm derivatives enriched in paraxial mesoderm but not lateral plate/extraembryonic mesoderm.
Factor-dependent chondrogenesis from the isolated KDR-PDGFRα+ paraxial mesoderm
Depending on which extracellular signaling factors are present, chondrogenesis from the mesenchymal precursor cells proceeds through multiple developmental stages: mesenchymal condensation, chondroprogenitor cell development and proliferation, and chondrocyte development and maturation13,14. The KDR−PDGFRα+ hES cell-derived paraxial mesodermal cells were, therefore, isolated by FACS and subjected to 2D micromass culture in the presence of various protein factors.
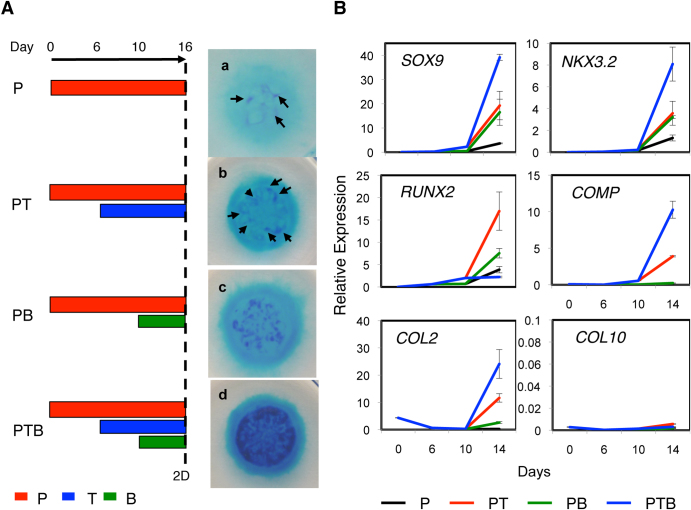
The KDR−PDGFRα+ progeny from H9 hES cells survived and proliferated in the presence of PDGF (“P”, Fig. 4A), as did mES cell-derived paraxial mesoderm15. In the presence of PDGF, however, the KDR−PDGFRα+ cells produced few, if any, sulfated proteoglycan (PG)-rich cartilage nodules in the micromass formed in 16 days as assessed by dark blue staining with acid Alcian Blue (a, Fig. 4A). The addition of TGFβ3 on days 3–6 (day 6 was the standard, “PT”, Fig. 4A) but not days 0–3 enhanced cartilage nodule formation, seemingly by supporting the growth of the KDR−PDGFRα+ progeny to the level needed for condensation and nodule formation (b, Fig. 4A). Furthermore, when BMP4 was added on day 10, 4 days after TGFβ, conditions under which chondrogenesis from mES cell-derived paraxial mesoderm was enhanced15 (“PTB”, Fig. 4A), there was a synergistic enhancement of cartilage nodule formation in micromass culture (d, Fig. 4A). The addition of BMP4 alone (“PB”, Fig. 4A) slightly increased the number of detectable cartilage nodules (c, Fig. 4A). Conversely, the sorted KDR−PDGFRα− cells survived in the presence of PDGF but never produced cartilaginous nodules in the presence of TGFβ3 and/or BMP4 (data not shown).
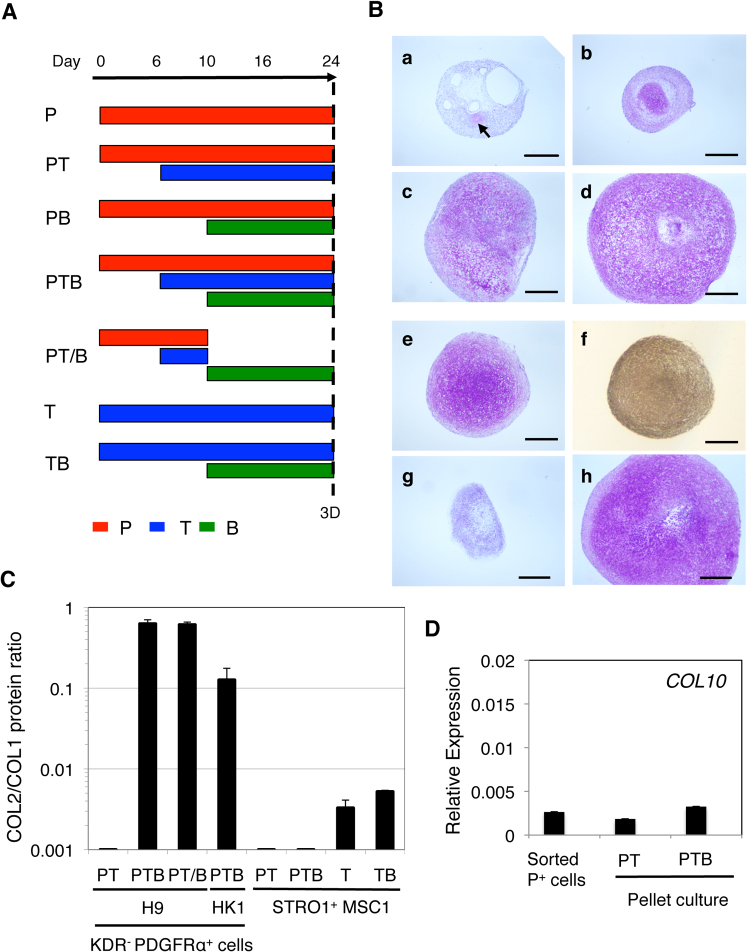
Figure 4. Factor-dependent chondrogenesis from the KDR−PDGFRα+ paraxial mesoderm-like cells from various hPS cell lines.
(A) Chondrogenesis from the H9 hES cell-derived KDR−PDGFRα+ paraxial mesoderm performed under P (a), PT (c), PB (d), and PTB (d) conditions in 2D micromass culture for 16 days. (Left) Period of treatment with PDGF (P, red), TGFβ3 (T, blue) and/or BMP4 (B, green) is indicated as a bar. (Right) The resulting micromasses were stained with acid Alcian Blue. Arrows indicate small cartilage nodules. Scale bars, 500 μm. (B) Expression of chondrogenesis-specific genes during 2D micromass culture of the H9 hES cell-derived KDR−PDGFRα− paraxial mesodermal cells performed under P, PT, PB or PTB conditions. The resulting micromasses were subjected to RT-PCR on the day indicated with primers for the genes indicated.
The kinetics of chondrogenesis from the isolated KDR−PDGFRα+ paraxial mesodermal cells under P, PT, PB and PTB conditions were monitored by expression of the chondroprogenitor genes NKX3.2, SOX9 and RUNX2 and the chondrocyte/cartilage matrix genes COL2 and COMP. PTB conditions were always the best for the expression of NKX3.2, SOX9, COL2 and COMP, which increased from day 10 (blue, Fig. 4A). However, RUNX2 expression was the highest under PT conditions (red), suggesting the suppression of RUNX2 expression and the promotion of SOX9 and NKX3.2 expression by BMP4. The type X collagen (COL10) transcript was not detected under the culture conditions tested. The isolated KDR−PDGFRα+ paraxial mesodermal cells thus appear to be directed toward undifferentiated /prehypertrophic chondrocyte formation under PTB conditions in 16 days of micromass culture. This is a similar result to that reported in chick somite explant culture16,17.
These observations were reproduced with 3D pellet culture (Fig. 5). In the absence of PDGF, the KDR−PDGFRα+ cell pellet grew poorly and failed to form a particle by 24 days of culture (data not shown). In the presence of PDGF (“P” Fig. 5A), the cells produced a small particle containing few, if any, sulfated PG-rich cartilage nodules as assessed by purple to pink staining (metachromasia) with Toluidine Blue (a, Fig. 5B). The addition of TGFβ3 on day 6 (“PT”, Fig. 5A) enhanced cartilage nodule formation (b, Fig. 5B) and, when followed by BMP4 on day 10 (“PTB”, Fig. 5A), resulted in marked enhancement in pellet growth and the accumulation of intercellular matrix that was metachromatically stained with Toluidine blue (d, Fig. 5). Although addition of BMP4 (“PB”, Fig. 3A) resulted in a cartilage particle significantly larger than that formed in the presence of PDGF alone (c, Fig. 5B), the sequential addition of TGFβ3 and BMP4 reproducibly gave rise to a much larger, translucent particle filled with a cartilaginous matrix.
Figure 5. Quantitative monitoring of chondrogenesis from hPS cell-derived paraxial mesoderm and adult hMSCs.
(A) 3D pellet chondrogenesis culture conditions for the sorted hPS cell progeny and the STRO1+ hMSCs. (B) Cartilage particle formation from the KDR−PDGFRα+ paraxial mesoderm-derived from H9 and Mix1-GFP hES cells and HK1 hiPS cells. Chondrogenesis from the KDR−PDGFRα+ progeny (P+ cells, Fig. 3A) from H9 hES cells was performed under P (a), PT (b), PB (c), and PTB (d–h) conditions in 3D pellet culture for 24 days. The GFP+KDR−PDGFRα+ progeny (e, f) and GFP+KDR−PDGFRα− progeny (g) from Mixl1-GFP hES cells (GFP+P+ and GFP+P− cells, respectively, Fig. 3E) and the KDR−PDGFRα+ progeny (P+ cells, Fig. 3E) from HK1 hiPS cells (h) were also pellet-cultured under PTB conditions. The resulting cartilage particles were paraffin-sectioned, and stained with Toluidine blue (a–e, g, h) or immunostained with the anti-COL2 antibody (f). The arrow indicates a small cartilage nodule. Scale bars, 500 μm. (C) The COL2/COL1 protein ratio. The conditioned media of the cartilage particles formed under PT, PTB, PT/B, T, or TB conditions as indicated, using the KDR−PDGFRα+ paraxial mesoderm from H9 hES cells and HK1 hiPS cells, and the STRO1+ hMSCs from the bone marrow of donor 1 (STRO1+ MSC1), were subjected to COL1 and COL2 ELISA. The ratio ng COL2/ng COL1 was calculated, averaged and plotted (shown as a bar). The SD is shown as a thin vertical line. (D) Change in the level of COL10 transcript in the H9 hES cell-derived KDR−PDGFRα+ paraxial mesoderm before (Sorted P+) and after 3D pellet culture performed under PT and PTB conditions.
Chondroprogenitors can be generated from non-mesodermal cells, such as cranial neural crest cells18. To confirm that the mesodermal progeny in the KDR−PDGFRα+ cell fraction contributed to the cartilage particles, GFP+KDR−PDGFRα+ paraxial mesoderm cells derived from day 8 Mixl1-GFP EBs formed under BIOSN were sorted (GFP+P+, Fig. 3E) and subjected to 3D pellet cultures (Fig. 5B). Under PTB conditions, a cartilage particle that was filled with cartilaginous matrix formed and was found to stain metachromatically with Toluidine Blue (e, Fig. 5B) and to immunostain uniformly with a COL2 antibody (f). However, other fractions, such as GFP+KDR−PDGFRα− cells, did not form cartilaginous particles (g), demonstrating that the chondrogenic activity was restricted to the KDR−PDGFRα+ paraxial mesodermal cells. Cartilage formation under PTB conditions was also demonstrated with the KDR−PDGFRα+ paraxial mesoderm progeny derived from HK1 hiPS cells (P+, Fig. 3E) differentiated under BIOSN conditions (h, Fig. 5B). Thus, the PTB conditions facilitated chondrocyte formation from paraxial mesoderm-like cells generated from various hPS cell lines.
Hyaline-like cartilage formation from the isolated KDR−PDGFRα+ paraxial mesoderm
Cartilage is classified according to the composition of the extracellular matrix. The translucent joint articular cartilage is hyaline cartilage; it is rich in COL2 and PG but is poor in COL1, i.e., PG+COL2+COL1− 19. In contrast, the fibrocartilage of the meniscus contains COL2 and a significant level of COL1 but it is poor in PG, i.e., PGlowCOL2+COL1+. In the growth plate, chondrocytes mature into hypertrophic chondrocytes that express COL10 instead of COL213,14. TGFβ facilitates the initial mesenchymal condensation but inhibits the hypertrophic differentiation of chondrocytes, whereas BMP signaling plays additional roles in the proliferation, maintenance and maturation of chondrocytes20.
The degree of chondrogenesis during 3D pellet culture was monitored by ELISA quantification of the amount of COL2 protein (from chondrocytes) and COL1 protein (from undifferentiated mesenchymal cells or chondrocytes in fibrocartilage) secreted into the culture medium. As shown in Fig. 5B, under PT conditions (b, Fig. 5B), 3D pellet culture with the KDR−PDGFRα+ paraxial mesoderm from H9 hES cells yielded a small particle with few small cartilage nodules. However, under PTB conditions (d), the cartilage particle that formed was bigger and was filled with cartilaginous matrix. When we determined the ratio of COL2 protein levels and COL1 protein levels (COL2/COL1), it was greater in the conditioned media of particles formed under the PTB and PT/B (condition where PDGF and TGFβ3 were replaced with BMP4 on day 10, Fig. 5A) conditions than under the PT conditions (Fig. 5C). Interestingly, there was no sign of upregulation of COL10 expression during the 24-day 3D pellet culture under either the PT or PTB conditions (Fig. 5D). Therefore, similar to the results from the 2D micromass culture (Fig. 4), the chondrocytes in the cartilage particle that formed seemed not to undergo hypertrophic differentiation under the conditions tested.
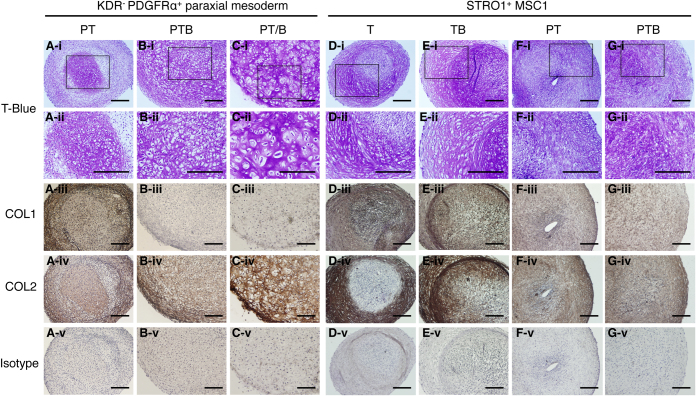
To confirm the quantitative ELISA data, the cartilage particles generated were subjected to immunohistochemical analyses (Fig. 6A–C). The cartilage particle formed under PT conditions consisted of heterogeneous areas: a small cartilaginous area strongly stained and peripheral mesenchymal cells weakly stained metachromatically (i.e. pink to purple) with Toluidine Blue, all of which were immunostained positively for both COL1 and COL2, i.e., COL2+COL1+ (Fig. 6A). The larger, translucent cartilage particle formed under PTB conditions consisted of a uniform cartilaginous area that was stained metachromatically with Toluidine Blue and immunostained strongly with COL2 (Fig. 6B-iv) but not COL1, i.e., PG+COL2+COL1− (Fig. 6B). The PT/B conditions yielded a similar PG+COL2+COL1− cartilage particle consisting of larger round chondrocytes with abundant cytoplasms (Fig. 6C). Therefore, these semi-quantitative immunohistochemical analyses on the cartilage particles correlated well with the quantitative ELISA analysis on the pellet culture supernatants.
Figure 6. Analysis of comparative cartilage-formation by hES cell-derived paraxial mesoderm-like cells and adult bone marrow-derived hMSCs.
The H9 hES cell-derived KDR−PDGFRα+ paraxial mesoderm (A–C) and human bone marrow-derived STRO1+ MSCs from donor 1 (p4, STRO1+ MSC1) (D–G) were subjected to 3D pellet chondrogenesis culture under PT (A, F), PTB (B, G), PT/B (C), T (D), and TB (E) conditions. Four contiguous sections were stained with Toluidine Blue (T-Blue) (i, ii), or immunostained for COL1 (iii), COL2 (iv), or isotype control (v). (ii) shows an enlarged view of the square in (i). Scale bars, 200 μm.
Comparative analysis of chondrogenesis from adult MSCs and hES cell-derived non-mesodermal mesenchymal cells
Next, we compared the chondrogenic capacity of the KDR−PDGFRα+ human paraxial mesoderm with that of the STRO1+ adult hMSCs using 3D pellet culture. Under standard chondrogenic differentiation conditions designed for bone marrow stromal cells (TGFβ3 without BMP4 and PDGF, i.e., “T”, Fig. 5A)21, the STRO1+ hMSCs gave rise to small particles consisting of heterogeneous areas: a cartilaginous area strongly stained and mesenchymal cell areas weakly stained metachromatically (i.e. pink to purple) with Toluidine Blue, which were immunostained with COL2 and COL1, i.e., PG+/lowCOL2+COL1+ (Fig. 6D, Supplementary Fig. 4A). The results obtained with the STRO1+ hMSCs from a different donor are shown in Supplementary Fig. 4. The later addition of BMP4 on day 10 (“TB”) led to the enlargement of both the particle size and the cartilaginous area (purple), which was restricted to the periphery of the particle. However, COL1 staining was still significant, i.e., PG+COL2+COL1+ (Fig. 6E, Supplementary Fig. 4B). The PT conditions resulted in a larger particle with less PG and COL2 as judged by the lack of metachromatic staining with Toluidine Blue (i.e. many blue stained nuclei/cells but only a very low level of pink to purple stained intercellular matrices found) and faint COL2 immunostaining. A significant level of COL1 staining was observed, i.e., PGlow/−COL2lowCOL1+ (Fig. 6F, Supplementary Fig. 4C). Even the PTB conditions failed to support the production of a large translucent particle, and the matrix profile remained in PG+/lowCOL2lowCOL1+ (Fig. 6G, Supplementary Fig. 4D). Note that there was a slight increase in metachromatic staining with Toluidine blue compared with that in the particle formed under PT conditions (Fig. 6F).
Consistent with the immunohistological results, the ELISA analysis demonstrated that the COL2/COL1 protein ratio obtained during pellet culture of STRO1+ hMSCs under the T and TB conditions was significant, but much lower than those obtained during hyaline-like cartilage particle formation from the H9 hES cell- and HK1 hiPS cell-derived paraxial mesoderm (i.e. under the PTB and PT/B conditions, Fig. 5C). In contrast, the value obtained with the STRO1+ hMSCs during pellet culture under the PT and PTB conditions was negligible (Fig. 5C). The same results were obtained with the STRO1+ hMSCs from a different donor (Supplementary Fig. 5). Thus, these results suggest that TGFβ and BMP are crucial for the chondrogenic differentiation of STRO1+ hMSCs, and that PDGF prevents the cells from forming COL2-positive COL1-poor cartilage particles but promotes the enlargement of the particles.
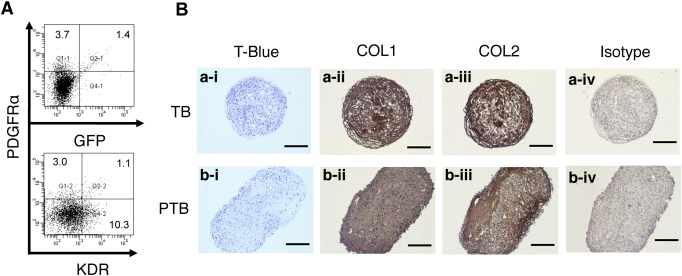
It has been reported that MSC-like mesenchymal progeny can be generated spontaneously from hES cells5. To compare the chondrogenic capacity of the KDR−PDGFRα+ paraxial mesodermal cells with such mesenchymal cells, H9 and Mixl1-GFP hES cells were differentiated, and mesenchymal cells were prepared as EB-outgrowth cells using one of the reported methods22 with slight modifications. This protocol utilizes no exogenous factors during the first 8 days of EB formation (i.e., lineage specification) culture in CDM and the following 10 days of EB-outgrowth (i.e., mesenchymal cell generation/expansion) culture in the KnockOut Serum Replacement (KSR)-based serum-free medium. When Mixl1-GFP hES cells were used, there were zero to a very small number of GFP+ or PDGFRα+ progeny generated (Fig. 7A), demonstrating that mesodermal specification did not occur. Mesenchymal cells generated in the same way from H9 hES cells were subjected to 3D pellet chondrogenesis culture under two conditions: TB (originally developed for MSCs) and PTB (developed for paraxial mesoderm) (Fig. 7B). Although PDGF caused an increase in size, the cartilaginous particles formed under either condition accumulated very low levels of PG, as judged by to the lack of, or very weak, metachromatic staining with Toluidine Blue. However, significant levels of both COL2 and COL1 were detected by immunostaining, i.e., PG−COL2+COL1+. These results suggest the presence of fibrotic cartilage particle formation.
Figure 7. Chondrogenic activity of non-mesodermal mesenchymal cells arising from hES cells.
Mixl1-GFP (A) and H9 (B) hES cells were differentiated in the absence of exogenous factors, and subjected to FACS analysis (A) and to the EB-outgrowth culture (B), respectively. The outgrowth cells (p4) were then subjected to 3D pellet culture under TB (a) and PTB (b) conditions. Four contiguous sections were stained with Toluidine Blue (T-Blue) (i), or immunostained for COL1 (ii), COL2 (iii), or isotype control (iv). Scale bars, 200 μm.
Discussion
In this report, we have demonstrated the following: the suppression of GSK3β activity and BMP signaling and the appropriate level of Nodal/Activin/TGFβ signaling effectively specify paraxial mesoderm from hPS cells; FACS-isolated (Mixl1-GFP+)KDR−PDGFRα+ progeny readily form a hyaline-like cartilage particle in a serum-free medium via sequential functions of PDGF, TGFβ and BMP; and the gold standard STRO1+ hMSCs from adult bone marrow and non-mesodermal mesenchymal cells from hES cells respond to these factors differently to form particles poor in PG that express both COL2 and COL1.
Although FLK1−PDGFRα+ rostral paraxial mesoderm is readily induced from mES cells with WNT3a and Noggin in CDM8, the derivation of paraxial mesoderm from hPS cells required the replacement of WNT3a with the GSK3β inhibitor BIO. Interestingly, a lower concentration of BIO, such as 0.5–1 μM, resulted in a similar progeny profile to that obtained using WNT3a at 100 ng/ml; however, the hemoangiogenic KDR+PDGFRαlo population23,24 was not generated, and the inclusion of Noggin suppressed the accumulation of all PDGFRα+ progeny (i.e. only very low levels of KDR−PDGFRα+ progeny remained) and failed to induce the MEOX1 transcript (data not shown). Therefore, although suppression of GSK3β will affect many signalling pathways in addition to the WNT pathway, it is possible that the efficient specification of paraxial mesoderm requires a strong canonical WNT signal, and that WNT3a at 100 ng/ml fails to satisfy such a signalling requirement in hPS cells.
The benefit of combining TGFβ and BMP for robust chondrogenesis in vitro has been demonstrated with the mES cell-derived mesodermal progeny15 and the MSCs25. However, the advantages of the PDGF and TGFβ combination (“PT”) and later treatment with BMP (“PTB”) or later transition from PDGF + TGFβ to BMP (“PT/B”) had only been clearly demonstrated with the mES cell-derived mesoderm15. In this respect, the hPS cell-derived paraxial mesoderm, which shows efficient chondrogenesis under PTB conditions, is strikingly similar to the mES cell-derived mesoderm. In contrast, the adult marrow hMSCs failed to show equivalent levels of chondrogenesis in the presence of PDGF, which stimulated the growth of the pellets formed. These results are partly consistent with the previous observations that PDGF signaling through PDGFRα is required for efficient somitic chondrogenesis and limb chondrogenesis in vivo and in vitro26,27, while such signaling has little effect on adult articular chondrocytes in vivo and causes only a small increase in PG synthesis in vitro during a cartilage explant culture28,29.
Minor bleeding, which is inevitable at the site of chondroprogenitor transplantation, leads to the formation of a fibrin clot. At the clot, accumulated platelets are activated to release various bioactive factors, including PDGF. In such a PDGF-rich environment, the use of hPS cell-derived chondrogenic mesoderm may produce a better clinical outcome than the use of bone marrow hMSCs.
The mesenchymal cells generated from hES cells without exogenous factors are known to display a limited chondrogenic activity in regular 3D pellet culture, producing fibrotic cartilage particles poor in PG but expressing both COL2 and COL15. We have demonstrated the non-mes(end)odermal identity of such mesenchymal cells (Fig. 7), and have reproduced their tendency to form fibrotic cartilage particles in vitro under the conditions in which the hPS cell-derived paraxial mesodermal cells formed hyaline-like cartilage particles (Fig. 6). The neural origin of the early bone marrow stromal cells has been demonstrated in the mouse30,31. The ‘memory’ of the developmental origin of mesenchymal cells may dictate the type of cartilage formed during chondrogenesis. However, it is also possible that the necessary expansion of the hMSCs and the hEB-outgrowth cells before use may not only lead to the reduction of chondrogenic activity as seen in other cultured mesenchymal cells32 (Supplementary Fig. 6E–H), but also determine the type of cartilage the cells will tend to form.
In summary, these data indicate that the KDR−PDGFRα+ paraxial mesodermal cells from hPS cells are a promising alternative to adult MSCs for the future in vitro generation of engineered hyaline cartilage constructs and cartilage regenerative therapy for joint disorders.
Methods
Cells, factors, and antibodies
H9 (WiCell Research Institute, Madison, WI) and MEL1 (Stem Cell Sciences, Melbourne Australia) hES cells were maintained on mouse embryonic fibroblasts (MEFs) as bulk culture in the KnockOut Serum Replacement (KSR, Invitrogen, Carlsbad, CA)-based hES cell medium containing 4 ng/ml fibroblast growth factor-2 (FGF2, R&D Systems Inc., Minneapolis, MN) as previously described23,24. The Mixl1-GFP hES cells were maintained on MEFs in KSR-based hES cell medium with 10 ng/ml FGF2 and expanded using a trypsin-based cell dissociation method as previously described11. However, the cells were adapted to the same maintenance conditions as those for H9 and MEL1 from 4 weeks before differentiation. The human keratinocyte-derived iPS cell line HK1, established by the retroviral transduction of the four Yamanaka-factors and NANOG to human primary hair follicle cells as previously described33, was kindly provided by Dr. Brian Davis (Houston TX). The HK1 cells were maintained in the same manner as the hES cells except that the concentration of FGF2 was higher (40 ng/ml). The Bry-GFP mES cells were obtained from Dr. Gordon Keller (Toronto, Canada) and cultured as previously described15,34. Human bone marrow stromal cells were obtained at the University of Adelaide according to the University of Adelaide Human Research Ethics Committees. The preparation of mononuclear cells and purification of MSCs by magnetic cell sorting (Miltenyi Biotec, Auburn CA) with the STRO1 antibody were performed as previously described35. The isolated MSCs were cultured in α-modified Minimal Essential Medium, 2 mM GlutaMax, 1 mM Na-pyruvate (all from Invitrogen), and they were supplemented with 20% (v/v) fetal bovine serum (HyClone, Logan, UT). The antibodies for FACS, cytokines, media and supplements, buffers and specialized tissue culture plates were generally sourced as described23,24.
Mesodermal differentiation of PS cells
Human PS cells (H9, MEL1, Mixl1-GFP and HK1) were differentiated 3-dimensionally using the embryoid body (EB)-forming culture. The culture was initiated in suspension in the CDM: Iscove's modified Dulbecco's medium (Sigma-Aldrich, St. Louis, MO): Ham's F12 (Invitrogen) (1:1), 5 mg/ml fatty acid-free bovine serum albumin (Sigma), 2% (v/v) chemically defined lipid concentrate (Invitrogen), 2 mM GlutaMax (Invitrogen), 100 μg/ml human holo-transferrin (Sigma), 10 μg/ml bovine insulin (Sigma), 0.45 mM monothioglycerol (MTG, Sigma), 0.17 mM ascorbic acid-2-phosphate (AA2P, Sigma) with the factors indicated, and they were maintained at 37°C under 5% CO2 and 5% O28. The EBs that developed in 3 days were transferred to a semi-solid CDM containing 0.9% (w/v) methylcellulose (Stem Cell Technology, Vancouver, Canada) with the factors indicated. The EBs were collected on day 8, and single EB cells were obtained with a brief (3 min) treatment in TrypLE Select (Invitrogen). The Bry-GFP mES cells were differentiated using the EB-forming culture in semi-solid CDM (without AA2P) with the factors added on day 2 of differentiation, and single EB cells were obtained non-enzymatically with 1 mM EDTA treatment as previously described8. The concentration ranges of factors used was as follows: Noggin (mNoggin-Fc, R&D) at 100–300 ng/ml, WNT3a and WNT5a (mouse proteins, Millipore/Chemicon, Billerica, MA) at 100 ng/ml, BMP4 (hBMP4, R&D) at 10 ng/ml, Activin (hActivin A, PeproTech, Rocky Hill, NJ) at 1–20 ng/ml, BIO (6-bromoindirubin-3′-oxime), Acetoxime-BIO (AceBIO), 1-methyl-BIO (MeBIO) (all from Calbiochem, San Diego, CA) and CHIR99021 (6-{2-[4-(2,4-Dichloro-phenyl)-5-(4-methyl-1H-imidazol-2-yl)-pyrimidin-2-ylamino]-ethylamino}-nicotinonitrile, BioVision, Mountain View, CA) at 2–5 µM, and SB431542 (4-(5-Benzol[1,3]dioxol-5-yl-4-pyrldin-2-yl-1H-imidazol-2-yl)-benzamide, Tocris Bioscience, Ellisville, MO) at 1–10 µM. BIO served as the major mesoderm inducer in this study. During the first 3 days of EB culture, BIO was added at 2 μM for the H9 hES cells and at 3 μM for the other hPS cells (Mixl1-GFP, MEL1 and HK1) in the presence of 100 ng/ml Noggin. When the EBs were transferred to the semi-solid medium on day 3, BIO was supplied at 5 μM with Noggin (Fig. 1A).
EB-outgrowth culture
The mesenchymal cells were developed spontaneously from hPS cells using one of the established EB-outgrowth methods22 with slight modifications. Briefly, the hPS cells were differentiated using the EB-forming culture in CDM for 8 days in the absence of exogenous factors at 37°C under conditions of 5% CO2 and 5% O2. The resulting EBs were transferred to a tissue culture plate and cultured in the hES cell medium without FGF2 for 10 days to allow for attachment of the EBs to the plate and slow outgrowth of mesenchymal cells. Next, the mesenchymal cells were purified by selective expansion (for 3 or more passages) in Dulbecco's modified Eagle's medium (DMEM, Invitrogen) supplemented with 10% (v/v) fetal bovine serum (HyClone) and the repetitive filtering method as described22.
Flow cytometry
Single-cell suspensions from human EBs were stained with the mouse anti-human PDGFRα and anti-human KDR monoclonal antibodies as previously described23,24. The FACS analysis was performed on LSRII (BD Biosciences, San Jose, CA), and cell sorting was performed with Aria II (BD). Viable single cells were gated using DAPI (4′,6-diamidino-2-phenylindole, Sigma). The purity of the sorted cells ranged from 92 to 96%.
Immunofluorescence detection of paraxial mesoderm
The H9 hES cells were differentiated in CDM in the presence of BIO + Noggin or BIO alone to generate PDGFRα+ mesenchymal progeny, which were further cultured in an 8-welll chamber slide (Nunc, Rochester, NY) coated with human fibronectin (Sigma, at 10 μg/ml for 24 h at 4°C) for 3–5 days in CDM containing 10 ng/ml PDGF (hPDGF-BB, R&D), 100 ng/ml Noggin, 0.1 μM CHIR99021, 0.1 μM PD173074 (Tocris), and 1 μM SB431542 at 37°C under 5% CO2 and 5% O2. Cultures were stopped and fixed with 10% (v/v) buffered Zn-Formalin (Z-fix, Anatech, Battle Creek, MI) for 20 min at r.t., then immunostained with the rabbit anti- hMEOX1 polyclonal antibody (Prestige antibody, Sigma), followed by biotinylated anti-rabbit IgG (Vector Labs, Burlingame, CA), and, finally, AlexaFluor 594-conjugated streptavidin (Invitrogen). The stained slides were inspected with a BX61 fluorescence microscope (Olympus, Center Valley, PA) one day after being mounted with a DAPI-containing solution (Invitrogen).
Chondrogenesis assays
Two-dimensional (2D) micromass culture was performed with FACS-purified mesodermal progeny (1.5×105 cells) transferred in 5 μl to a fibronectin-coated 24-well plate (BD) and cultured in serum-free chondrogenic medium: DMEM: F12 (1:1) (Invitrogen), 1% (v/v) ITS+ mix (BD), 0.17 mM AA2P, 0.35 mM Proline (Sigma), 0.1 μM dexamethasone (Sigma), 0.15% (v/v) glucose (Sigma), 1 mM Na-pyruvate (Invitrogen), 2 mM GlutaMax, 0.05 mM MTG8,15 and supplemented with 40 ng/ml PDGF and 1% (v/v) fetal bovine serum (Hyclone). TGFβ3 (hTGFβ3, R&D) was subsequently added at 10 ng/ml on day 3 to 6. As indicated, BMP4 was added to a concentration of 50 ng/ml on day 10. The micromass cultures were maintained at 37°C under 5% CO2 and 5% O2 for 16 days, fixed with Z-Fix, and stained with 1% (w/v) Alcian Blue (Sigma) pH 1.0 (i.e. acid Alcian Blue). For 3-dimensional (3D) pellet culture, 3.0×105 FACS-purified EB cells, EB-outgrowth cells at passage 4 to 5, and adult MSCs at passage 4 to 8 were centrifuged and cultured as a pellet in 0.5 ml of the serum-free chondrogenic media8,15 that was supplemented with the defined factors added in the same sequence as in the micromass culture. The pellet cultures were maintained at 37°C under 5% CO2 for 24 days, fixed with Gendre's Fluid, paraffin-embedded, sectioned, and stained with 0.1% (w/v) Toluidine Blue (Sigma) or immunostained with anti-type I (COL1) and type II (COL2) collagen antibodies, followed by color development in the presence of horseradish peroxidase and 3, 3′-diaminobenzidine as described8,15.
Quantitative comparison of collagen production
During 3D pellet culture, media conditioned with cartilage particles for 3 days were collected on day 24, and the levels of COL1 and COL2 proteins were quantified with ELISA kits (MD Biosciences, St. Paul, MN) according to the manufacturer's recommendation. The proportion of COL2 to COL1 production (ng COL2/ng COL1) was calculated from duplicate or triplicate reactions and averaged to obtain a measure of the extent of chondrogenesis by the undifferentiated mesenchymal cells used for 3D pellet culture.
Gene-expression profiling
Total RNA from the whole differentiated progeny or FACS-purified progeny was purified and reverse transcribed (RT). Real-time polymerase chain reaction (PCR) was then performed using the Taqman Gene Expression Assay and ABI7900 (Applied Biosystems, Foster City, CA) as previously described23. The individual gene expression levels from duplicate or triplicate reactions were normalized against the EEF1A1 transcript and averaged to obtain relative expression data. The real-time RT-PCR results are presented as average relative expression levels in a bar or line graph. The standard deviation (SD) is shown as a thin vertical line.
Author Contributions
K.U. performed research, analysed data and wrote the paper, J.Z. performed research, P.S., E.S. and A.E. contributed to preparation of human cells and cell lines and generation of new reagents and tools, and N.N. designed and performed research, analysed data and wrote the paper.
Supplementary Material
Supplementary Information
Acknowledgments
First, we would like to acknowledge B. Davis and A. Crane for providing the HK1 hiPS cell line prior to publication. We also thank D. Havilland for cell sorting, and M.M. Jiang for histological analysis. This work was supported by Brown Foundation Institute of Molecular Medicine UTHSC (Startup Fund, NN), and Rolanette and Berdon Lawrence Bone Disease Program of Texas Research Award (NN).
References
- Prockop D. J. Repair of tissues by adult stem/progenitor cells (MSCs): controversies, myths, and changing paradigms. Mol. Ther. 17, 939–946 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinert A. F. et al. Major biological obstacles for persistent cell-based regeneration of articular cartilage. Arthritis Res Ther 9, 213 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishikawa S.-I., Jakt L. M. & Era T. Embryonic stem-cell culture as a tool for developmental cell biology. Nat. Rev. Mol. Cell Biol. 8, 502–507 (2007). [DOI] [PubMed] [Google Scholar]
- Murry C. E. & Keller G. Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell 132, 661–680 (2008). [DOI] [PubMed] [Google Scholar]
- Nakayama N. & Umeda K. in Embryonic Stem Cells: The Hormonal Regulation of Pluripotency and Embryogenesis. (ed. Atwood C.) 621–648 (INTECH, Wien; 2011).
- Mahmood A., Harkness L., Schroder H. D., Abdallah B. M. & Kassem M. Enhanced differentiation of human embryonic stem cells to mesenchymal progenitors by inhibition of TGF-beta/activin/nodal signaling using SB-431542. J. Bone Miner. Res. 25, 1216–1233 (2010). [DOI] [PubMed] [Google Scholar]
- Oldershaw R. A. et al. Directed differentiation of human embryonic stem cells toward chondrocytes. Nat. Biotechnol. 28, 1187–1194 (2010). [DOI] [PubMed] [Google Scholar]
- Tanaka M. et al. BMP inhibition stimulates WNT-dependent generation of chondrogenic mesoderm from embryonic stem cells. Stem Cell Res. 3, 126–141 (2009). [DOI] [PubMed] [Google Scholar]
- MacDonald B. T., Tamai K. & He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17, 9–26 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schier A. F. Nodal morphogens. Cold Spring Harb Perspect Biol 1, a003459 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis R. P. et al. Targeting a GFP reporter gene to the MIXL1 locus of human embryonic stem cells identifies human primitive streak-like cells and enables isolation of primitive hematopoietic precursors. Blood 111, 1876–1884 (2008). [DOI] [PubMed] [Google Scholar]
- Hart A. H. et al. Mixl1 is required for axial mesendoderm morphogenesis and patterning in the murine embryo. Development 129, 3597–3608 (2002). [DOI] [PubMed] [Google Scholar]
- Goldring M. B., Tsuchimochi K. & Ijiri K. The control of chondrogenesis. J. Cell. Biochem. 97, 33–44 (2006). [DOI] [PubMed] [Google Scholar]
- Karsenty G., Kronenberg H. M. & Settembre C. Genetic control of bone formation. Annu. Rev. Cell Dev. Biol. 25, 629–648 (2009). [DOI] [PubMed] [Google Scholar]
- Nakayama N., Duryea D., Manoukian R., Chow G. & Han C. Y. Macroscopic cartilage formation with embryonic stem-cell-derived mesodermal progenitor cells. J. Cell Sci. 116, 2015–2028 (2003). [DOI] [PubMed] [Google Scholar]
- Provot S. et al. Nkx3.2/Bapx1 acts as a negative regulator of chondrocyte maturation. Development 133, 651–662 (2006). [DOI] [PubMed] [Google Scholar]
- Kempf H., Ionescu A., Udager A. M. & Lassar A. B. Prochondrogenic signals induce a competence for Runx2 to activate hypertrophic chondrocyte gene expression. Dev Dyn 236, 1954–1962 (2007). [DOI] [PubMed] [Google Scholar]
- Abzhanov A., Tzahor E., Lassar A. B. & Tabin C. J. Dissimilar regulation of cell differentiation in mesencephalic (cranial) and sacral (trunk) neural crest cells in vitro. Development 130, 4567–4579 (2003). [DOI] [PubMed] [Google Scholar]
- Onyekwelu I., Goldring M. B. & Hidaka C. Chondrogenesis, joint formation, and articular cartilage regeneration. J. Cell. Biochem. (2009). [DOI] [PubMed] [Google Scholar]
- Song B., Estrada K. D. & Lyons K. M. Smad signaling in skeletal development and regeneration. Cytokine Growth Factor Rev 20, 379–388 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo J. U. et al. The chondrogenic potential of human bone-marrow-derived mesenchymal progenitor cells. J Bone Joint Surg Am 80, 1745–1757 (1998). [DOI] [PubMed] [Google Scholar]
- Hwang N. S., Varghese S., Zhang Z. & Elisseeff J. Chondrogenic differentiation of human embryonic stem cell-derived cells in arginine-glycine-aspartate-modified hydrogels. Tissue Eng 12, 2695–2706 (2006). [DOI] [PubMed] [Google Scholar]
- Wang Y. & Nakayama N. WNT and BMP signaling are both required for hematopoietic cell development from human ES cells. Stem Cell Res. 3, 113–125 (2009). [DOI] [PubMed] [Google Scholar]
- Wang Y., Umeda K. & Nakayama N. Collaboration between WNT and BMP signaling promotes hemoangiogenic cell development from human fibroblast-derived iPS cells. Stem Cell Res. 4, 223–231 (2010). [DOI] [PubMed] [Google Scholar]
- Indrawattana N. et al. Growth factor combination for chondrogenic induction from human mesenchymal stem cell. Biochem Biophys Res Commun 320, 914–919 (2004). [DOI] [PubMed] [Google Scholar]
- Andrae J., Gallini R. & Betsholtz C. Role of platelet-derived growth factors in physiology and medicine. Genes Dev. 22, 1276–1312 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoch R. V. & Soriano P. Roles of PDGF in animal development. Development 130, 4769–4784 (2003). [DOI] [PubMed] [Google Scholar]
- Hulth A. et al. Effect of transforming growth factor-beta and platelet-derived growth factor-BB on articular cartilage in rats. J Orthop Res 14, 547–553 (1996). [DOI] [PubMed] [Google Scholar]
- Schafer S. J., Luyten F. P., Yanagishita M. & Reddi A. H. Proteoglycan metabolism is age related and modulated by isoforms of platelet-derived growth factor in bovine articular cartilage explant cultures. Arch Biochem Biophys 302, 431–438 (1993). [DOI] [PubMed] [Google Scholar]
- Morikawa S. et al. Development of mesenchymal stem cells partially originate from the neural crest. Biochem Biophys Res Commun 379, 1114–1119 (2009). [DOI] [PubMed] [Google Scholar]
- Takashima Y. et al. Neuroepithelial cells supply an initial transient wave of MSC differentiation. Cell 129, 1377–1388 (2007). [DOI] [PubMed] [Google Scholar]
- Neuhuber B., Swanger S. A., Howard L., Mackay A. & Fischer I. Effects of plating density and culture time on bone marrow stromal cell characteristics. Exp. Hematol. 36, 1176–1185 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aasen T. & Belmonte J. C. Isolation and cultivation of human keratinocytes from skin or plucked hair for the generation of induced pluripotent stem cells. Nat Protoc 5, 371–382 (2010). [DOI] [PubMed] [Google Scholar]
- Nakayama N., Fang I. & Elliott G. Natural killer and B-lymphoid potential in CD34+ cells derived from embryonic stem cells differentiated in the presence of vascular endothelial growth factor. Blood 91, 2283–2295 (1998). [PubMed] [Google Scholar]
- Gronthos S. et al. Molecular and cellular characterisation of highly purified stromal stem cells derived from human bone marrow. J. Cell Sci. 116, 1827–1835 (2003). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Information