Abstract
Inflammation, interstitial fibrosis (IF), and tubular atrophy (TA) precede chronic transplant dysfunction, which is a major cause of renal allograft loss. There is an association between IF/TA and loss of peritubular capillaries (PTCs) in advanced renal disease, but whether PTC loss occurs in an early stage of chronic transplant dysfunction is unknown. Here, we studied PTC number, IF/TA, inflammation, and renal function in 48 patients who underwent protocol biopsies. Compared with before transplantation, there was a statistically significant loss of PTCs by 3 months after transplantation. Fewer PTCs in the 3-month biopsy correlated with high IF/TA and inflammation scores and predicted lower renal function at 1 year. Predictors of PTC loss during the first 3 months after transplantation included donor type, rejection, donor age, and the number of PTCs at the time of implantation. In conclusion, PTC loss occurs during the first 3 months after renal transplantation, associates with increased IF and TA, and predicts reduced renal function.
Peritubular capillary (PTC) loss correlates with renal interstitial inflammation and fibrosis in animal models1–5 and in patients with native kidney disease6–8 as well as late chronic transplant dysfunction (CTD).9 CTD is a major cause of renal graft loss and is related to immunological and nonimmunological factors such as rejection, ischemia/reperfusion (I/R) injury, and toxicity of immunosuppressive therapy.10 We questioned whether early PTC loss is related with chronic renal failure by studying protocol transplant biopsies. Inflammation and interstitial fibrosis (IF)/tubular atrophy (TA) in protocol biopsies predict long-term renal graft function.11,12 We hypothesized that PTC loss occurs in an early stage of CTD and precedes the development of IF/TA. Therefore, we analyzed whether PTC loss in the first 3 months is associated with higher IF/TA score at 12 months in the renal biopsy and reduced renal graft function. Second, we studied whether early PTC loss is associated with specific donor and/or transplant characteristics.
Table 1 shows the baseline demographic data and clinical characteristics of our study population in this retrospective consecutive cohort study. We studied PTC numbers in biopsies taken 0, 3, and 12 months after transplantation. Because I/R damage is associated with PTC loss in animal studies,1 we specifically compared grafts from living donors (LDs) to grafts from postmortal donors. In the latter group, we distinguished between donation after brain death (DBD) and donation after cardiac death (DCD) grafts because DCD kidneys have more extensive warm ischemic damage.13 As expected from previous studies,9,14 statistically significant differences between the donor groups were found in preoperative ischemia time and incidence of delayed graft function (Table 1). The incidence of rejection and subclinical rejection (SCR) was not significantly different between the three donor groups, as has been shown previously.15
Table 1.
Baseline demographics and clinical characteristics of study population overall (total) and by donor type (LD, DBD, DCD)
| Parameter | Total (n = 48) | LD (n = 17) | DBD (n = 12) | DCD (n = 19) | P, ANOVA |
|---|---|---|---|---|---|
| Demographics | |||||
| recipient age (years) | 52.5 ± 13.5 | 50.6 ± 13.5 | 50.4 ± 13.5 | 55.5 ± 13.7 | 0.475 |
| recipient male/female | 31/17 | 10/7 | 6/6 | 15/4 | 0.205 |
| donor age (years) | 48.6 ± 14.2 | 51.8 ± 13.8 | 49.2 ± 11.3 | 45.3 ± 16.1 | 0.388 |
| donor male/female | 28/20 | 8/9 | 8/4 | 12/7 | 0.494 |
| Peritransplantation events | |||||
| first warm ischemia time (min) | 11.0 ± 16.3 | — | 0.3 ± 1.2 | 27.5 ± 14.8 | <0.001c |
| range | 0 to 73 | — | 0 to 4 | 10 to 73 | |
| cold ischemia time (hours) | 13.9 ± 9.8 | 2.5 ± 0.7 | 18.1 ± 5.3 | 21.7 ± 5.6 | <0.001a,b |
| range | 1.5 to 34.3 | 1.5 to 4.0 | 10.1 to 30.3 | 14 to 34.3 | |
| delayed graft function (yes/no) | 23/25 | 2/15 | 4/8 | 17/2 | <0.001a,c |
| Immunological events | |||||
| total (yes/no) | 11/37 | 2/15 | 4/8 | 5/14 | 0.336 |
| rejection (yes/no) | 6/42 | 1/16 | 3/9 | 2/17 | 0.320 |
| SCR (yes/no) | 9/39 | 2/15 | 3/9 | 4/15 | 0.619 |
| Renal function after 1 year | |||||
| MDRD at 12 months | 48.5 ± 17.6 | 53.5 ± 18.4 | 49.7 ± 21.5 | 43.1 ± 13.1 | 0.204 |
| proteinuria/creatinine 12 months | 0.038 ± 0.06 | 0.021 ± 0.01 | 0.051 ± 0.09 | 0.044 ± 0.07 | 0.526 |
| range | 0.006 to 0.325 | 0.008 to 0.047 | 0.012 to 0.325 | 0.006 to 0.304 | — |
Dummy regression analysis results with significant differences in first warm ischemia time, cold ischemia time, and delayed graft function, but not in rejection or SCR between aLD versus DCD,
bLD versus DBD, or
cDCD versus DBD.
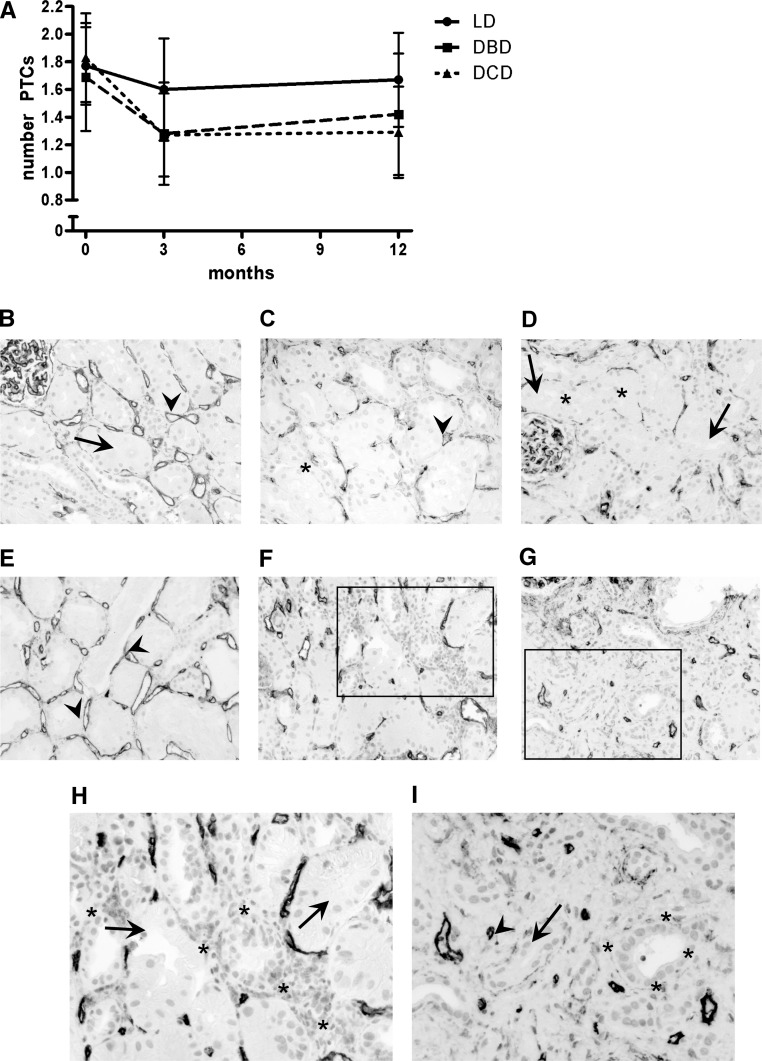
Figure 1 shows representative examples of CD31/CD34 staining in protocol graft biopsies. In the total study population, there was a significant loss of PTCs in the 3 months after transplantation (t = 6.070, P < 0.001, paired t test, Figure 1A). In our data, PTC loss did not occur between 3 and 12 months (t = −1.499, P = 0.141). There is a statistically significant difference in PTC loss over the three donor groups (repeated-measures ANOVA; F = 3.95 by 2 and 45 degrees of freedom, P = 0.026), with statistically significant more PTC loss in the DCD group than in the LD group (dummy regression analysis; B = −0.386, P = 0.007). When corrected for donor age and PTC number at implantation, a statistically significant loss of PTCs was observed in the DCD and DBD groups as compared with the LD group (analysis of covariance; F = 6.97 by 2 and 43 degrees of freedom, P = 0.002; respective B = −0.391, P = 0.001; B = −0.319, P = 0.012). In literature, long-term graft survival and kidney function are quite similar for DCD and DBD allografts, but they are worse as compared with LD allografts.13,15 It is thought that extensive I/R damage—as a result of prolonged ischemia time—is one of the major factors in the multifactorial pathway leading toward decreased survival of postmortal renal allografts.13 Our data show significantly more PTC loss in DCD and DBD allografts as compared with LD allografts, which may correspond with animal studies on PTC loss after I/R damage.1,4 Although DCD allografts have more I/R damage because of an extra-warm ischemia time, our data show no statistically significant difference in PTC loss between DCD and DBD allografts. However, by study design it was not possible to include patients with early graft loss. It is well known that DCD donor kidneys have a higher incidence of primary nonfunction as compared with DBD and LD donor kidneys.15,16 Thus, further studies, in which kidneys that fail within the first year after transplantation are included, are needed to firmly assess the effect of postmortal donor type on PTC loss.
Figure 1.
Loss of PTCs occurs within the first 3 months after transplantation (P < 0.001, paired samples t test). DCD and DBD renal allografts show a significant loss of PTCs within the first 3 months after transplantation (respective P < 0.001, n = 19; P = 0.004, n = 12). LD renal allografts showed a trend toward PTC loss in the first 3 months (P = 0.061, n = 17) (paired samples t test per group). Immunohistochemical quantification of a (B through D) LD and (E through G) DCD kidney. Panels H and I are enlargements of respective figures F and G. In preimplantation biopsies, PTCs are evenly distributed (B, E), whereas after (C, F) 3 and (D, G) 12 months, areas can occur without PTCs (asterisks) even when no IF/TA is present (D). In areas with inflammation (F, H), and with IF/TA (G, I), a lower number of PTCs is observed. Arrowhead denotes PTC; arrow denotes tubule.
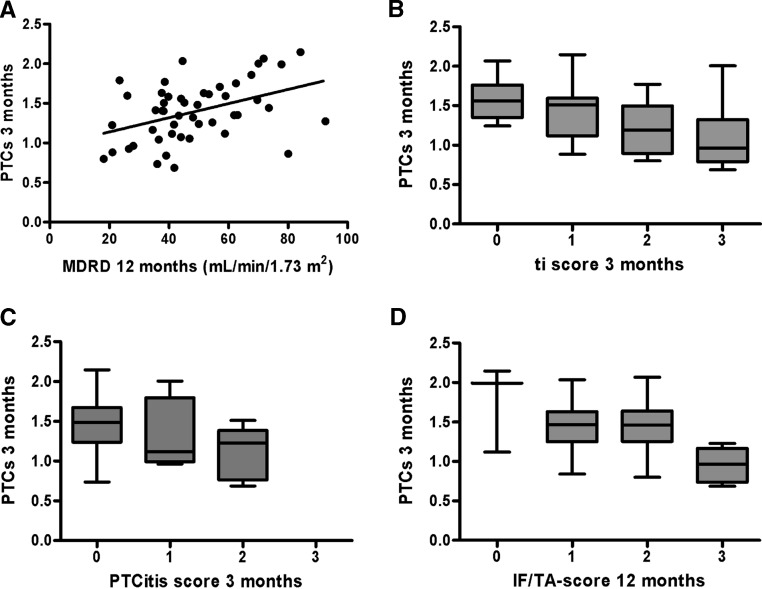
Because inflammation and IF/TA in protocol biopsies are surrogate end points for CTD,15,10–12 correlation of PTC number with renal function was studied, as well as correlations with inflammation and IF/TA scores in the biopsy. Figure 2 shows that PTC number at 3 months correlated significantly with MDRD. Subsequently, we studied in a multivariate analysis if PTC loss during the first 3 months (change in PTC number [ΔPTC] 0 to 3 months) predicts a worse Modification of Diet in Renal Disease (MDRD) score than donor and transplant characteristics. Significant predictors associated with a worse MDRD score were ΔPTC (0 to 3 months), a long first warm ischemia time, and higher donor age (Table 2, model 1; r2 = 0.379, P < 0.001). PTC number at 3 months was associated with the total inflammation score at 3 months (Figure 2B) and the IF/TA score at 12 months (Figure 2D). Furthermore, at 3 months, nine biopsies were positive for peritubular capillaritis (PTCitis). We found a strong correlation between PTCitis at 3 months and immunological events (ρ = 0.483, P = 0.001). PTCitis at 3 months was associated with lower PTC numbers at 3 months (ρ = −0.315, P = 0.033, Figure 2C) and more IF/TA at 12 months (ρ = 0.293, P = 0.049). This is in line with studies emphasizing the significance of PTCitis as a predictor of graft dysfunction.17
Figure 2.
Relationship between number of PTCs at 3 months and (A) MDRD and (D) IF/TA after 12 months and with (B) inflammation and (C) PTCitis after 3 months. A low number of PTCs at 3 months is significantly correlated with a low MDRD score (r = 0.424, P = 0.003). Number of PTCs at 3 months is significantly negatively rank correlated with (B) total inflammation (ρ = −0.469, P = 0.001) and (C) PTCitis score (ρ = −0.469, P = 0.001) at 3 months and with (D) IF/TA score at 12 months (ρ = −0.377, P = 0.008).
Table 2.
Multivariate analysis of clinical, baseline, and histological parameters for MDRD score at 12 months, IF/TA score at 12 months, and PTC loss from 0 to 3 months (dummy regression analysis)
| Model (Outcome) | Predictord | B | SEM | β | t | P | 95% Confidence Interval |
|
|---|---|---|---|---|---|---|---|---|
| Lower | Upper | |||||||
| 1 (MDRD 12 months)a | Donor age | −0.577 | 0.152 | −0.466 | −3.806 | <0.001 | −0.882 | −0.271 |
| WIT 1 | −0.329 | 0.135 | −0.306 | −2.442 | 0.019 | −0.601 | −0.057 | |
| ΔPTC 0 to 3 mo | −9.897 | 4.977 | −0.246 | −1.989 | 0.053 | −19.928 | 0.133 | |
| Constant | 83.881 | 7.944 | 10.560 | <0.001 | 67.871 | 99.890 | ||
| 2 (IF/TA 12 months)b | ΔPTC 0 to 3 mo | 0.733 | 0.254 | 0.392 | 2.892 | 0.006 | 0.223 | 1.244 |
| Constant | 1.323 | 0.147 | — | 9.018 | <0.001 | 1.028 | 1.618 | |
| 3 (ΔPTC 0 to 3 months)c | DCD versus LD | −0.253 | 0.119 | −0.253 | −2.125 | 0.040 | −0.494 | −0.013 |
| DBD versus LD | −0.344 | 0.106 | −0.389 | −3.248 | 0.002 | −0.558 | −0.130 | |
| Immunological events | −0.261 | 0.111 | −0.253 | −2.349 | 0.024 | −0.485 | −0.037 | |
| PTC number at 0 months | −0.734 | 0.142 | −0.545 | −5.162 | <0.001 | −1.021 | −0.447 | |
| Donor age | −0.006 | 0.003 | −0.209 | −1.946 | 0.058 | −0.013 | <0.001 | |
| Constant | 1.494 | 0.033 | — | 4.489 | <0.001 | 0.822 | 2.165 | |
aModel 1, outcome MDRD at 12 months, final model: r2 = 0.379, P < 0.001.
bModel 2, outcome IF/TA at 12 months, final model: r2 = 0.154, P = 0.006.
cModel 3, outcome ΔPTC 0 to 3 months, final model: r2 =0.567, P < 0.001.
dDCD versus DBD: B = −0.091, β = −0.102, P = 0.434, 95% confidence interval = −0.323; 0.141.
In a multivariate analysis, we studied if PTC loss during the first 3 months (ΔPTC 0 to 3 months) after transplantation was associated with more IF/TA at 12 months next to donor and transplant characteristics. The final model showed only ΔPTC (0 to 3 months) as a significant predictor of IF/TA at 12 months (Table 2, model 2; r2 = 0.154, P = 0.006). On the basis of these results, we hypothesize that loss of PTCs early after transplantation is associated with later IF/TA development and decreased renal function. This further substantiates the hypothesis that PTC loss may be an important factor in the development of IF/TA and chronic renal failure. In this retrospective study, we cannot establish whether PTC loss precedes or results from IF/TA in patients. However, experimental studies suggest that PTC loss is associated with inflammation and precedes IF/TA.2,4 Further studies using larger study populations and experimental models are needed to study the interactions among inflammation, PTC loss, and IF/TA. The extent of PTC loss is causally related to the balance between angiogenic and angiostatic factors,4,5 with inflammatory cells mediating decreased vascular endothelial growth factor production and subsequent capillary rarefaction.3 Decreased expression of vascular endothelial growth factor in renal biopsies of patients with capillary loss suggests a similar pathway in humans.7,8 In our study, we show that total inflammation correlates negatively with PTC number, which suggests that there may be a disturbed interplay between inflammation and endothelial repair.
Subsequently, we studied which donor and transplant characteristics predict PTC loss during the first 3 months after transplantation (ΔPTC 0 to 3 months). No relation was found for ΔPTC (0 to 3 months) and donor gender, recipient age, or recipient gender. In Table 2, the final multiple regression model is shown for ΔPTC from 0 to 3 months (r2 = 0.567, P < 0.001). Significant factors associated with PTC loss in the first 3 months were donor type (with DCD and DBD grafts losing more PTCs than LD grafts), immunological events, PTC number at implantation (patients with low PTC number at implantation have more PTC loss), and donor age.
Our data show a role for immunological events in PTC loss, which is in line with findings by others that clinical acute cellular rejection and SCR predict IF/TA in protocol biopsies.10,11 Because there were only six patients with a clinical rejection in our cohort, we could not discern whether capillary loss in the 3-month biopsy is different for patients with treated clinical cellular rejection versus untreated SCR.
Renal allografts with a low number of PTCs at implantation show a low PTC number at 3 months and more PTC loss in the first 3 months. PTC number at implantation is therefore an important determinant of PTC loss. In the study presented here, which includes only renal allografts functioning at 1 year, PTC number in the implantation biopsy was not associated with IF/TA score in the same biopsy (ρ = −0.100, P = 0.391), nor with total inflammation or IF/TA scores in the 3-month (ρ = 0.073, P = 0.526) and 12-month biopsies (ρ = −0.036, P = 0.753). The assessment of a preimplantation biopsy is indispensable for the measurement of PTC loss after transplantation. This is in line with recent studies showing that IF/TA and glomerulosclerosis in preimplantation biopsies can predict CTD, especially in marginal donors.18,19
Finally, we show that donor age affects the extent of PTC loss in the first months after transplantation. Animal studies show impaired angiogenesis, with less PTCs and more IF/TA in the aging kidney.2 Others have reported that kidneys from older donors have decreased graft survival.18,20 Older donor age may be associated with impaired angiogenesis (i.e., less regenerative capacity or increased apoptosis of endothelial cells) after transplantation in human kidneys.
In summary, this study shows that PTC loss mainly occurs in the first 3 months after transplantation and that it is associated with higher rates of inflammation and fibrosis and a lower MDRD score at 1 year. Predictive clinical factors for early PTC loss are donor type, (subclinical) rejection, PTC number at implantation, and donor age. These findings indicate that early monitoring of PTC number may become a valuable predictive marker for CTD. The preservation of capillary endothelium can play an important role in preservation of long-term renal graft function.
CONCISE METHODS
Patients
Forty-eight consecutive patients transplanted at the Maastricht University Medical Centre in The Netherlands between March 2003 and June 2006 with representative renal biopsies taken at implantation (0) and 3 and 12 months after transplantation were included in this study. Our study sample of 48 patients appears to be representative for the total group of transplanted patients in that period with a functioning graft the first year after transplantation. Representative biopsies had at least five glomeruli, one interlobular artery, and ten cortical fields for computerized image analysis of PTCs. Patients received a kidney from LD, DBD, or DCD donors.
This retrospective cohort study was approved by the Medical Ethical Committee of the Maastricht University Medical Centre (number 094002). Collection, storage, and use of tissue and patient data have been performed in agreement with the Code for Proper Secondary Use of Human Tissue put forward by the Federation of Dutch Medical Scientific Societies (http://www.federa.org).
All patients received a triple tacrolimus-based immunosuppressive regimen without induction and with early steroid minimization. The target range of tacrolimus was 15 to 20 ng/ml in weeks 1 and 2, 10 to 15 ng/ml in weeks 3 and 4, and 5 to 7 ng/ml thereafter. All patients received 125 mg of methylprednisolon at day 0 and day 1 and 10 mg of prednisone from day 2 onward. In patients with high immunological risk (defined as >85% panel-reactive HLA antibodies in the previous 6 months and/or a previous graft survival of <1 year because of immunological reasons), prednisone was tapered to 5 mg/d at month 2 and continued at this dose during the first year. In all other patients, steroids were stopped at day 10, but in the case of rejection, steroids were reintroduced at a dose of 10 mg/d for 30 days and thereafter tapered to 5 mg/d in the following 2 months.21 A Banff 1A or 1B rejection was treated with three pulse doses of 1 g methylprednisolon. Banff 2A or higher rejections were treated with a 10-day course of rabbit anti-thymocyte globulin (Thymoglobulin, Fresenius).
Definitions
SCR was based on one of two histologic categories in the 3-month biopsy: borderline SCR (tubulitis score [t] = 1, interstitial inflammation score [i] > 0) and SCR (t = 2, i ≥ 0), as described by Scholten et al.,22 in the absence of clinical renal function deterioration. Protocol biopsies with a Banff score higher than IB rejection (t = 3; i ≥ 1) were considered and treated as “rejection” biopsies with a 10-day course of anti-thymocyte globulin (see above). Immunological events were subdivided into clinical rejection within the first 3 months and SCR in the 3-month protocol biopsy. Estimated GFR was calculated by the abbreviated MDRD formula.23 Proteinuria was expressed as the proteinuria/creatinine ratio (in g/mmol) in a morning spot urine. Delayed graft function was defined as a need for temporary dialysis treatment starting within the first week after grafting.
Histology, Immunohistochemistry, and Morphometry
Paraffin tissue sections for light microscopy were stained by hematoxylin and eosin, periodic acid–Schiff, and periodic acid methenamine silver methods according to standard protocols. Histopathologic analysis was performed by two blinded pathologists (C.P. and R.vS) according to Banff 97.24 IF, TA, interstitial inflammation, tubulitis, and IF/TA were scored as an ordinal four-point scale on periodic acid–Schiff- and silver-stained sections. Additionally, biopsies were scored for total inflammation and PTCitis.12 In the case of a >1 point difference in score of a biopsy between both pathologists, the eventual score was solved by conference.
To assess the number of PTCs, a CD31/CD34 co-staining was used as described previously in a study of microvessel density in human melanomas.25 Briefly, renal tissue was fixed in 4% buffered formalin and embedded in paraffin. Paraffin sections (3 μm) were cut onto Starfrost adhesive microscope slides. Monoclonal antibodies were used to stain paraffin-embedded tissue sections for endothelial markers using CD31/CD34 co-staining. (CD31: DAKO Cytomation; The Netherlands; clone JC70A) (CD34: Neomarkers; Freemont, CA; clone QBEnd/10). Sections were incubated with primary antibodies for 45 minutes, after which they were incubated with the Powervision polymer (Immunologic; Duiven, The Netherlands) for 30 minutes. Furthermore, 3,3-diaminobenzidine (DAKO Cytomation, Glostrup, Denmark) was used as chromogen, and slides were counterstained with hematoxylin (Klinipath, Duiven, The Netherlands).
The number of PTCs and tubuli were assessed in 0-, 3-, and 12-month biopsies stained with CD31/CD34 co-staining using an image processing and analysis system (QWin, Leica's Windows-based image analysis tool kit, Leica, Cambridge, United Kingdom) linked to a Leica DML3000 light microscope (Leica Quantimet, Germany). Images were obtained and analyzed with a 40× objective by one blinded observer (F.S.). Per slide, ten images of the cortex were obtained in a serpentine manner. The medulla and the subcapsular cortex with a width of 0.5 mm were excluded when images were acquired. Initially, the software was set to differentiate positively from negatively stained areas. PTCs were identified by positive CD31/CD34 staining. Per field, the number of PTCs and the number of tubuli were counted. Finally, PTC number was corrected for the number of tubuli, as described previously by Kaukinen et al.7
Statistical Analysis
Univariate data are presented as means and SD. To test for normality of distribution, the Kolmogorov–Smirnov test is used. If data are non-normally distributed, the lowest and highest scores are also given. Univariate classified nominal and ordinal data are presented as frequencies and percentages. Overall baseline demographical, clinical, and histologic data are also presented as broken down to donor type: LD, DBD, and DCD. In normally distributed metric data, differences between these groups are tested by one-way ANOVA using Bonferroni-corrected t tests between pairs of groups. For non-normally distributed metric data, the Kruskal–Wallis test is used with Mann–Whitney tests between pairs of groups. Dichotomic categories such as gender and immunological events are analyzed by cross tabulations and log-likelihood χ2 tests.
Data analysis focused upon differences in PTCs found at implantation (i.e., at baseline) and 3 and 12 months after transplantation. For PTC loss, differences are tested by paired t test or (broken down for donor type) by repeated-measures analysis of covariance.
Data analysis on number of PTCs at 0, 3, and 12 months was done by multiple (dummy) regression analysis using baseline data and possible intervening immunological posttransplant events as predictors, confounders, or baseline conditions. For this purpose, the categorical trichotomy of donor type is broken down into two dummy 0 to 1 dichotomies indicating contrasts of DBD(1) with LD(0) and of DCD(1) with LD(0), which are used as a pair in regression analysis. Three groups of predictors were used in dummy regression analysis on the basis of outcome variables “PTC loss the first 3 months,” “MDRD score at 12 months,” and “IF/TA score at 12 months”: (1) baseline demographic and clinical parameters consisting of donor type, gender of donor and recipient, and age of donor and recipient; (2) baseline histologic parameters (PTC number, PTC loss, and IF/TA score at implantation); and (3) clinical parameters within the first 3 months, consisting of immunological events and warm and cold ischemia time. Delayed graft function was not used as a clinical parameter for statistical analysis because it is too highly related to DCD donors as compared with DBD and LD donors. Searching for the final best-fitting model within regression analysis was done by means of a combination of forward selection and backward elimination.
The univariate relationship between PTC number at implantation and other baseline factors and variables was analyzed by respectively one-way ANOVA and (rank) correlation coefficients (Spearman's ρ). All potential predictors were chosen based on their known relationship with CTD and renal allograft survival.10,12
P < 0.05 was considered as statistically significant. All data analyses were performed using the SPSS statistical software package (version 16.0).
DISCLOSURES
None.
Acknowledgments
The authors thank Drs. S. Heeneman (Department of Pathology, Maastricht, The Netherlands) and M.G. Snoeijs (Department of Surgery) for critical and stimulating discussions and Mrs. M. Mullens (Department of Internal Medicine) for her assistance in organizing the protocol biopsies.
Footnotes
Published online ahead of print. Publication date available at www.jasn.org.
REFERENCES
- 1. Basile DP: The endothelial cell in ischemic acute kidney injury: Implications for acute and chronic function. Kidney Int 72: 151–156, 2007 [DOI] [PubMed] [Google Scholar]
- 2. Kang DH, Anderson S, Kim YG, Mazzalli M, Suga S, Jefferson JA, Gordon KL, Oyama TT, Hughes J, Hugo C, Kerjaschki D, Schreiner GF, Johnson RJ: Impaired angiogenesis in the aging kidney: Vascular endothelial growth factor and thrombospondin-1 in renal disease. Am J Kidney Dis 37: 601–611, 2001 [DOI] [PubMed] [Google Scholar]
- 3. Kang DH, Kanellis J, Hugo C, Truong L, Anderson S, Kerjaschki D, Schreiner GF, Johnson RJ: Role of the microvascular endothelium in progressive renal disease. J Am Soc Nephrol 13: 806–816, 2002 [DOI] [PubMed] [Google Scholar]
- 4. Ohashi R, Shimizu A, Masuda Y, Kitamura H, Ishizaki M, Sugisaki Y, Yamanaka N: Peritubular capillary regression during the progression of experimental obstructive nephropathy. J Am Soc Nephrol 13: 1795–1805, 2002 [DOI] [PubMed] [Google Scholar]
- 5. Yuan HT, Li XZ, Pitera JE, Long DA, Woolf AS: Peritubular capillary loss after mouse acute nephrotoxicity correlates with down-regulation of vascular endothelial growth factor-A and hypoxia-inducible factor-1 alpha. Am J Pathol 163: 2289–2301, 2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Bohle A, Mackensen-Haen S, Wehrmann M: Significance of postglomerular capillaries in the pathogenesis of chronic renal failure. Kidney Blood Press Res 19: 191–195, 1996 [DOI] [PubMed] [Google Scholar]
- 7. Kaukinen A, Lautenschlager I, Helin H, Karikoski R, Jalanko H: Peritubular capillaries are rarefied in congenital nephrotic syndrome of the Finnish type. Kidney Int 75: 1099–1108, 2009 [DOI] [PubMed] [Google Scholar]
- 8. Lindenmeyer MT, Kretzler M, Boucherot A, Berra S, Yasuda Y, Henger A, Eichinger F, Gaiser S, Schmid H, Rastaldi MP, Schrier RW, Schlondorff D, Cohen CD: Interstitial vascular rarefaction and reduced VEGF-A expression in human diabetic nephropathy. J Am Soc Nephrol 18: 1765–1776, 2007 [DOI] [PubMed] [Google Scholar]
- 9. Ishii Y, Sawada T, Kubota K, Fuchinoue S, Teraoka S, Shimizu A: Injury and progressive loss of peritubular capillaries in the development of chronic allograft nephropathy. Kidney Int 67: 321–332, 2005 [DOI] [PubMed] [Google Scholar]
- 10. Nankivell BJ, Borrows RJ, Fung CL, O'Connell PJ, Allen RD, Chapman JR: The natural history of chronic allograft nephropathy. N Engl J Med 349: 2326–2333, 2003 [DOI] [PubMed] [Google Scholar]
- 11. Seron D, Moreso F: Protocol biopsies in renal transplantation: Prognostic value of structural monitoring. Kidney Int 72: 690–697, 2007 [DOI] [PubMed] [Google Scholar]
- 12. Solez K, Colvin RB, Racusen LC, Haas M, Sis B, Mengel M, Halloran PF, Baldwin W, Banfi G, Collins AB, Cosio F, David DS, Drachenberg C, Einecke G, Fogo AB, Gibson IW, Glotz D, Iskandar SS, Kraus E, Lerut E, Mannon RB, Mihatsch M, Nankivell BJ, Nickeleit V, Papadimitriou JC, Randhawa P, Regele H, Renaudin K, Roberts I, Seron D, Smith RN, Valente M: Banff 07 classification of renal allograft pathology: Updates and future directions. Am J Transplant 8: 753–760, 2008 [DOI] [PubMed] [Google Scholar]
- 13. Wijnen RM, Booster MH, Stubenitsky BM, de Boer J, Heineman E, Kootstra G: Outcome of transplantation of non-heart-beating donor kidneys. Lancet 345: 1067–1070, 1995 [PubMed] [Google Scholar]
- 14. Moers C, Smits JM, Maathuis MH, Treckmann J, van Gelder F, Napieralski BP, van Kasterop-Kutz M, van der Heide JJ, Squifflet JP, van Heurn E, Kirste GR, Rahmel A, Leuvenink HG, Paul A, Pirenne J, Ploeg RJ: Machine perfusion or cold storage in deceased-donor kidney transplantation. N Engl J Med 360: 7–19, 2009 [DOI] [PubMed] [Google Scholar]
- 15. Brook NR, Waller JR, Nicholson ML: Nonheart-beating kidney donation: Current practice and future developments. Kidney Int 63: 1516–1529, 2003 [DOI] [PubMed] [Google Scholar]
- 16. Keizer KM, de Fijter JW, Haase-Kromwijk BJ, Weimar W: Non-heart-beating donor kidneys in the Netherlands: Allocation and outcome of transplantation. Transplantation 79: 1195–1199, 2005 [DOI] [PubMed] [Google Scholar]
- 17. Lerut E, Naesens M, Kuypers DR, Vanrenterghem Y, Van Damme B: Subclinical peritubular capillaritis at 3 months is associated with chronic rejection at 1 year. Transplantation 83: 1416–1422, 2007 [DOI] [PubMed] [Google Scholar]
- 18. Snoeijs MG, Buurman WA, Christiaans MH, van Hooff JP, Goldschmeding R, van Suylen RJ, Peutz-Kootstra CJ, van Heurn LW: Histological assessment of preimplantation biopsies may improve selection of kidneys from old donors after cardiac death. Am J Transplant 8: 1844–1851, 2008 [DOI] [PubMed] [Google Scholar]
- 19. Anglicheau D, Loupy A, Lefaucheur C, Pessione F, Letourneau I, Cote I, Gaha K, Noel LH, Patey N, Droz D, Martinez F, Zuber J, Glotz D, Thervet E, Legendre C: A simple clinico-histopathological composite scoring system is highly predictive of graft outcomes in marginal donors. Am J Transplant 8: 2325–2334, 2008 [DOI] [PubMed] [Google Scholar]
- 20. de Fijter JW, Mallat MJ, Doxiadis II, Ringers J, Rosendaal FR, Claas FH, Paul LC: Increased immunogenicity and cause of graft loss of old donor kidneys. J Am Soc Nephrol 12: 1538–1546, 2001 [DOI] [PubMed] [Google Scholar]
- 21. Gelens MA, Christiaans MH, van Heurn EL, van den Berg-Loonen EP, Peutz-Kootstra CJ, van Hooff JP: High rejection rate during calcineurin inhibitor-free and early steroid withdrawal immunosuppression in renal transplantation. Transplantation 82: 1221–1223, 2006 [DOI] [PubMed] [Google Scholar]
- 22. Scholten EM, Rowshani AT, Cremers S, Bemelman FJ, Eikmans M, van Kan E, Mallat MJ, Florquin S, Surachno J, ten Berge IJ, Bajema IM, de Fijter JW: Untreated rejection in 6-month protocol biopsies is not associated with fibrosis in serial biopsies or with loss of graft function. J Am Soc Nephrol 17: 2622–2632, 2006 [DOI] [PubMed] [Google Scholar]
- 23. Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D: A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 130: 461–470, 1999 [DOI] [PubMed] [Google Scholar]
- 24. Racusen LC, Solez K, Colvin RB, Bonsib SM, Castro MC, Cavallo T, Croker BP, Demetris AJ, Drachenberg CB, Fogo AB, Furness P, Gaber LW, Gibson IW, Glotz D, Goldberg JC, Grande J, Halloran PF, Hansen HE, Hartley B, Hayry PJ, Hill CM, Hoffman EO, Hunsicker LG, Lindblad AS, Yamaguchi Y, et al. : The Banff 97 working classification of renal allograft pathology. Kidney Int 55: 713–723, 1999 [DOI] [PubMed] [Google Scholar]
- 25. Hillen F, van de Winkel A, Creytens D, Vermeulen AH, Griffioen AW: Proliferating endothelial cells, but not microvessel density, are a prognostic parameter in human cutaneous melanoma. Melanoma Res 16: 453–457, 2006 [DOI] [PubMed] [Google Scholar]