Abstract
Acute SSRI (selective serotonin reuptake inhibitor) treatment has been shown to attenuate the abuse-related effects of cocaine; however, SSRIs have had limited success in clinical trials for cocaine abuse, possibly due to neurobiological changes that occur during chronic administration. In order to better understand the role of serotonin (5HT) in cocaine abuse and treatment, we examined the effects of chronic treatment with the SSRI fluoxetine at clinically relevant serum concentrations on cocaine-related neurobiology and behavior. Rhesus macaques self-administering cocaine underwent a 6-week dosing regimen with fluoxetine designed to approximate serum concentrations observed in humans. Self-administration and reinstatement were monitored throughout the treatment and washout period. In vivo microdiaylsis was used to assess changes in dopaminergic and serotonergic neurochemistry. Positron emission tomography was used to assess changes in the 5HT transporter and 2A receptor binding potential (BP). Functional output of the 5HT system was assessed using prolactin levels. Cocaine-primed reinstatement and cocaine-elicited dopamine overflow were significantly suppressed following chronic fluoxetine treatment. 5HT2A receptor BP was increased in the frontal cortex following treatment while prolactin release was blunted, suggesting desensitization of the 5HT2A receptor. These effects persisted after a 6-week washout period. Measures of pre-synaptic serotonergic function and cocaine self-administration were unaffected. These data demonstrate that acute and chronic fluoxetine treatments exert different effects on cocaine-related behavior. Furthermore, chronic fluoxetine treatment causes alterations in 5HT2A receptors in the frontal cortex that may selectively disrupt cocaine-primed reinstatement. Fluoxetine may not be useful for treatment of ongoing cocaine abuse but may be useful in relapse prevention.
Keywords: fluoxetine, cocaine, self-administration, reinstatement, nonhuman primate, 5HT2A
INTRODUCTION
Cocaine abuse is a serious problem, with 1.1 million abusers reported in 2009 (Substance Abuse and Mental Health Services Administration, 2010), yet there is currently no FDA-approved effective pharmacotherapy for cocaine addiction. While substitute agonist therapies are being considered (Howell and Wilcox, 2001a), these medications often have high abuse potential. Different pharmacological targets should be considered; the serotonin (5HT) system, which modulates dopamine (DA), is one such target. Key brain areas involved in drug abuse, including the ventral tegemental area, the nucleus accumbens, and the prefrontal and frontal cortex, receive sertoneregic innervation. The serotonin 2A and 2C receptors have been implicated as likely candidates for mediating the influence of 5HT on DA neurotransmission (Bubar and Cunningham, 2008).
Selective serotonin reuptake inhibitors (SSRIs) bind and block the serotonin transporter (SERT) and are currently approved to treat depression and mood disorders. Preclinically, they have been shown to attenuate the reinforcing (Carroll et al, 1990; Czoty et al, 2002) and reinstating (Baker et al, 2001; Ruedi-Bettschen et al, 2009) effects of cocaine in rodents and primates, although they may potentiate the discriminative-stimulus effects of low doses (Cunningham and Callahan, 1991; Schama et al, 1997). In human laboratory studies, pretreatment with fluoxetine decreased ratings of cocaine's positive subjective effects (Walsh et al, 1994). SSRIs also caused an acute increase in extracellular 5HT in rats (Kreiss and Lucki, 1995; Marek et al, 2005; Qu et al, 2009) and attenuated cocaine-induced increases in extracellular DA in nonhuman primates (Czoty et al, 2002).
However, clinical trials testing SSRIs failed to show reductions in cocaine abuse (Grabowski et al, 1995; Schmitz et al, 2001; Winstanley et al, 2011), although recently there has also been some success (Moeller et al, 2007; Oliveto et al, 2012). This may be due in part to the different dosing regimens and end points employed by the various preclinical and clinical studies; preclinical studies generally employ single-dose treatments and evaluate end points related to cocaine pharmacology, whereas clinical studies administer the drug chronically and generally focus on reductions in current cocaine use. Furthermore, the therapeutic effects of SSRIs for depression and mood disorders do not emerge until 3–4 weeks after beginning treatment, and are believed to rely on neurobiological changes (Wong et al, 1995). For example, 5HT levels are not elevated during chronic treatment (Smith et al, 2000), ostensibly due to downregulation of the SERT (Benmansour et al, 1999; Kugaya et al, 2003), and desensitization of the 5HT1A autoreceptor (Ceglia et al, 2004; Riad et al, 2008), which do not occur during acute treatment. Neurobiological changes due to prolonged SSRI treatment may alter the effects of the SSRI on cocaine-related behavior and neurobiology. Additionally, the presence of cocaine-induced neurobiological changes, such as the sensitization of both DA and 5HT release in key areas of the mesolimbic system (Parsons and Justice, 1993) and upregulation of SERT (Banks et al, 2008; Mash et al, 2000), could alter the effects of prolonged SSRI treatment.
We examined the effects of chronic treatment with the SSRI, fluoxetine, at serum concentrations comparable to clinically observed concentrations, on cocaine self-administration behavior and neurochemistry, as well as on 5HT neurobiology and functional integrity in rhesus macaques in order to better understand the neurobiological effects of SSRI treatment in the context of cocaine use, and their relationship to cocaine-related behavior. Such knowledge could lead to more appropriate and effective use of SSRIs in both preclinical and clinical applications, and implicate particular receptors and neurobiological changes in the success or failure of serotonergic compounds in the treatment of cocaine abuse. Fluoxetine was chosen since it is one of the most well-characterized SSRIs (Wong et al, 1995), as well as the drug of choice for several clinical trials that examined SSRIs' effects on cocaine abuse (Grabowski et al, 1995; Schmitz et al, 2001; Winstanley et al, 2011).
MATERIALS AND METHODS
Subjects
Adult rhesus macaques (macaca mulatta; n=9) served as subjects. Five females weighing 6–9 kg participated in experiments 1 and 2; four males weighing 12–16 kg participated in experiment 3 (Supplementary Information). All subjects were individually housed within a primate colony and fed Purina monkey chow supplemented with fruits and vegetables. Water was freely available. Each animal was implanted with a chronic indwelling subcutaneous access port (Access Technologies, Skokie, IL, USA) and venous catheter as described previously (Howell and Wilcox, 2001b) and had experience self-administering cocaine. All procedures and studies followed the National Institutes of Health Guide for the Care and Use of Laboratory Animals (Publication No. 85--23, revised 1985), and were approved by the Institutional Animal Care and Use Committee of Emory University.
Drugs
Cocaine HCl (National Institute on Drug Abuse, Rockville, MD, USA) was dissolved in 0.9% saline and diluted to the desired unit dose according to each individual's body weight. Fenfluramine, a 5HT releaser, was purchased from Sigma-Aldrich (St Louis, MO, USA) and dissolved in 0.9% saline. Fluoxetine HCl was purchased from Spectrum Chemicals and Laboratory Products (New Jersey, USA) and administered orally (10 mg/kg/day) for 42 days. Each animal's dose was individually weighed and mixed with peanut butter or other palatable substances.
Behavioral Paradigms
Self-administration
Subjects (n=9) were trained to self-administer cocaine on a fixed ratio (FR) 20 response schedule of i.v. drug administration using an operant panel equipped with response levers and lights and controlled by a computer program (MedPC, MedAssociates, St Albans, VT, USA). Drug infusions, delivered by an automated syringe pump, were paired with brief illumination of a red light and followed with a 30-s timeout. Sessions occurred once a day and lasted until either 30 infusions had been earned or an hour had elapsed, whichever occurred first. Response rate served as a measure of reinforcing effects.
Reinstatement
Subjects underwent extinction training in which saline was substituted for cocaine in the syringe and the brief paired stimuli (lights) omitted until responding fell below 20% of previous response rate. At this point, reinstatement sessions were conducted by restoring the brief paired stimuli and priming the animal with a noncontingent dose of cocaine immediately before the session. Only saline was available during reinstatement sessions. Two doses of cocaine (0.01 and 0.03 mg/kg) and saline were tested in reinstatement and responding was extinguished between reinstatement sessions. Response rate during reinstatement sessions was normalized to the individual's cocaine self-administration response rate before extinction.
Positron Emission Tomography
Subjects were anesthetized and scanned using [11C]M100907 (Ito et al, 1998) and [18F]FEmZIENT (Stehouwer et al, 2008) to determine 5HT2A and SERT binding potential (BP), respectively, on separate occasions. Nondisplaceable BPs, BPND, were calculated using the simplified reference tissue model (Lammertsma and Hume, 1996) and cerebellum reference region (Hinz et al, 2007; Kish et al, 2005) for the frontal cortex, midbrain/brainstem, and caudate nuclei (Supplementary Figure S1). It is important to note that the cerebellum contains a low level of 5HT2A expression and thus low BPs reflect similar levels, as opposed to the absence of 5HT2A binding. Procedural details are reported in Supplementary Materials and Methods.
In Vivo Microdialysis and Prolactin Response
Subjects were surgically prepared with guide cannulae targeting the caudate nucleus before the start of any experimental measures but after cocaine self-administration training had been completed. Awake subjects underwent in vivo microdialysis sessions with cocaine (1.0 mg/kg, i.v.) and fenfluramine (3.0 mg/kg, i.v.) on separate occasions to determine the effects of treatment on the DA response to cocaine and the pre-synaptic function of the 5HT system, respectively. During fenfluramine-challenge sessions, animals also underwent blood sample collection; samples were frozen and later processed for prolactin, to assess the post-synaptic function of the 5HT system. Procedural details are reported in Supplementary Materials and Methods.
Experiment 1: Chronic Fluoxetine Treatment
Subjects (female; n=5) underwent chronic fluoxetine treatment designed to approximate serum concentrations observed in the human clinical setting as described elsewhere (Sawyer and Howell, 2011). Briefly, fluoxetine (10 mg/kg/day, p.o.) was administered for 6 weeks, which has been previously demonstrated to achieve serum concentrations comparable to those reported in the human clinical condition (200–1000 ng/ml). This dosing regimen was chosen to maximize the translational relevance of the study, since the relationship between serum concentration and drug effects, particularly therapeutic effects for depression, are not well understood for SSRIs (Baumann, 1996).
Serum concentrations were monitored via weekly blood draws to ensure maintenance in the target range (200–1000 ng/ml; see Supplementary Materials and Methods for additional details). Each animal had a minimum of 1 year of experience self-administering cocaine before beginning experimental measures.
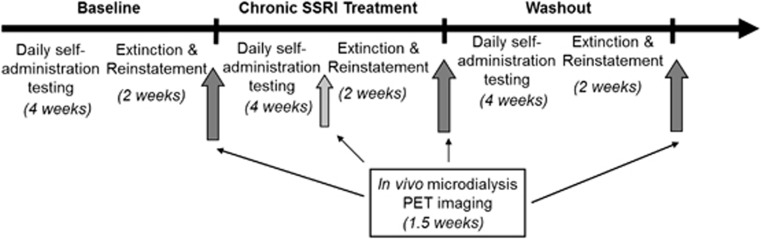
Cocaine dose-effect curves were determined in each individual by substituting different concentrations of cocaine in the syringe. The dose that engendered the highest response rates, or the peak dose, was selected for the following experiments (0.01 mg/kg/infusion for all subjects). Subjects were then stabilized on this dose (<20% variability in response rate across 5 days) before beginning SSRI treatment. This served as the baseline time point response rate for self-administration. All neurobiological and behavioral baseline measures were collected after determination of the dose-response curve and before beginning fluoxetine treatment. Fluoxetine treatment was administered in a block design (Figure 1) and lasted for 6 weeks (42 days), followed by a 6-week washout period. Each 6-week block consisted of 4 weeks of cocaine self-administration sessions, followed by a 2-week period of extinction and reinstatement testing. Average response rate across 5 days served as an indicator of reinforcing effects and was derived from the 5 days of week 4 in each block. At the end of each block, animals resumed daily self-administration testing, and 5HT and DA neurochemistry were assessed using in vivo microdialysis; 5HT function assessed using prolactin response; and SERT and 5HT2A availability assessed using PET imaging. All neurobiological assessments were conducted a minimum of 3–4 days following the end of fluoxetine treatment to ensure that observed effects would reflect alterations in neurobiology, not the direct effects of fluoxetine (Sawyer and Howell, 2011). An additional microdialysis session with a cocaine challenge was conducted during week 4 of chronic fluoxetine treatment to directly assess the effects of ongoing treatment on the response to cocaine. Details of data analysis are reported in Supplementary Materials and Methods.
Figure 1.
Experimental design for experiment 1, consisting of three 6-week blocks of cocaine self-administration and reinstatement testing, followed by neurochemical and neurobiological assessments (dark gray arrows). The light gray arrow indicates a single in vivo microdialysis session with cocaine challenge during fluoxetine treatment; all other testing at dark gray arrows is timed such that no SSRI would be present in the system. Total time for all three blocks is 18 weeks.
RESULTS
Experiment 1: Chronic Fluoxetine Treatment
Cocaine-related behavior
Daily administration of 10 mg/kg fluoxetine, p.o., sustained serum concentrations of active drug within the clinically reported range (200–1000 ng/ml; Supplementary Figure S2) (Sawyer and Howell, 2011). Serum concentrations were consistently between 250–450 ng/ml during weeks 3–6 of treatment when all behavioral measures were determined.
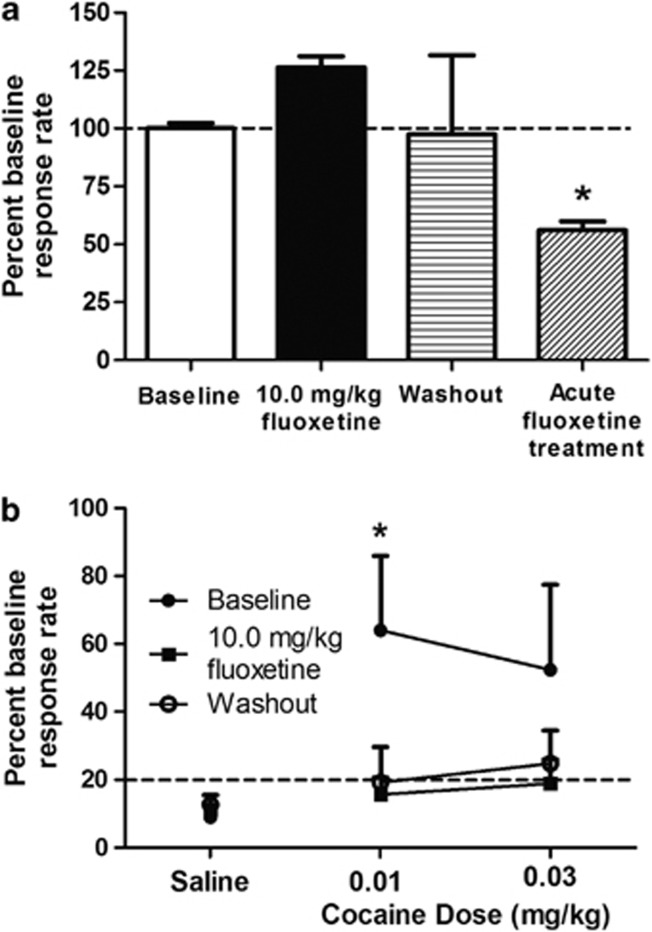
Cocaine self-administration rates varied between individuals (average: 1.01±0.46 presses/s; see Supplementary Figure S7); thus, rate data were normalized to the stable baseline average for each subject. All five subjects participated in the self-administration portion of the experiment; however, one animal failed to maintain a stable baseline rate and thus was not included in the behavioral analyses. No significant differences were seen between baseline, chronic fluoxetine treatment, and washout conditions (Figure 2a).
Figure 2.
Cocaine self-administration and reinstatement before, during, and after washout of chronic fluoxetine treatment. (a) Average cocaine self-administration rates from week 4, normalized to baseline, for each phase of experiment 1 (n=4), and rates following acute fluoxetine treatment resulting in similar blood levels (Experiment 2, Supplementary Information) (n=3). (b) Cocaine-primed reinstatement for each phase of experiment 1 (n=4). Rates are depicted as percent of previous cocaine self-administration rate before extinction. *Indicates significance at p<0.05.
At the baseline time point, a dose of 0.01 mg/kg cocaine resulted in significant reinstatement of responding as compared with a saline prime (F(2,3)=4.94, p=0.05; Dunnet's multiple comparison p<0.05; Figure 2b) and 0.03 mg/kg resulted in responding above extinction levels. Both drug and nondrug primes were accompanied by restoring the brief paired stimuli; saline plus brief paired stimuli did not result in responding above extinction levels. Following chronic fluoxetine treatment, significant reinstatement was not observed and response rate remained at extinction levels for all doses of cocaine tested. This effect persisted after the 6-week washout period.
Cocaine-induced DA overflow
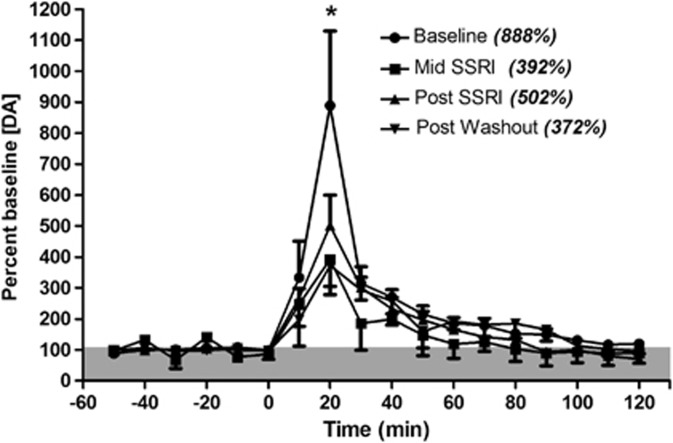
Two of the five animals implanted lost the cannula preparation before starting experiments; thus only three animals participated in the microdialysis studies in experiment 1. There was no difference between baseline DA dialysate levels (Supplementary Table S1) within individuals across the different study points, thus each data set was normalized to its respective baseline. All sessions demonstrated an increase in DA in response to high potassium challenge (average: baseline, 4235±3495% mid-SSRI, 1754±1197% post-SSRI, 2403±982% post-washout, 3684±1884%), confirming site viability. Cocaine induced a significant increase in DA overflow in the caudate nucleus (main effect of time, F(17,34)=29.26, p<0.001). There was no main effect of fluoxetine treatment, but there was a significant interaction between time and treatment (F(51,102)=1.65, p=0.016). Post hoc testing indicated that the DA overflow was significantly attenuated at 20 min post-injection (peak effect) during and immediately after fluoxetine treatment, and after the 6-week washout period (p<0.05; Figure 3), indicating that chronic fluoxetine treatment resulted in a lasting attenuation of cocaine-induced increases in DA.
Figure 3.
Dopamine levels in the caudate nucleus following cocaine challenge (1.0 mg/kg, i.v., at time 0; n=3). Peak overflow, at 20 min, is given in parentheses. Cocaine administration resulted in a significant increase in DA overflow for each stage, but peak levels were significantly reduced compared with baseline during fluoxetine treatment, immediately post-treatment, and remained suppressed following washout. *Indicates significance at p<0.05.
Serotonergic function
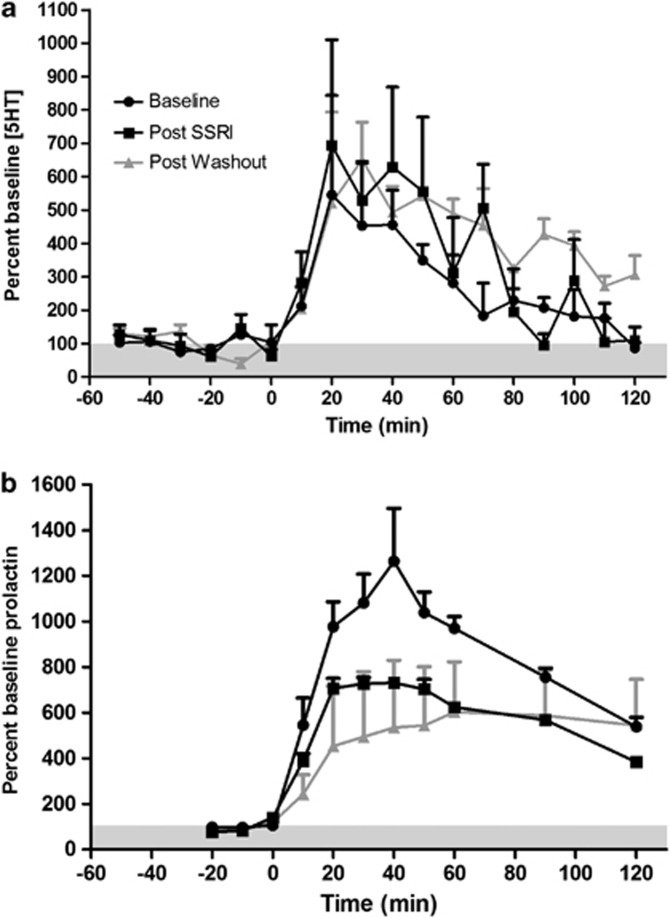
There was no difference between baseline 5HT dialysate levels between testing days (Supplementary Table S1), thus each data set was normalized to its respective baseline. All sessions demonstrated an increase in 5HT in response to high potassium challenge (average: baseline, 692±103% post-SSRI, 2785±1553% post-washout, 1402±117%), confirming site viability. Fenfluramine induced significant increases in 5HT at all treatment stages as indicated by a main effect of time (F(17,85)=7.39, p<0.0001; Figure 4a); however, chronic fluoxetine treatment had no effect on fenfluramine-induced 5HT overflow as measured by treatment main effect, peak effect, or area under the curve.
Figure 4.
Serotonin (a) and prolactin (b) response to fenfluramine challenge (3.0 mg/kg, i.v. at time 0; n=3). There were no significant differences in 5HT response to fenfluramine challenge but prolactin was significantly blunted.
Baseline levels of prolactin did not differ between testing days (Supplementary Table S1), thus each data set was normalized to its respective baseline. Fenfluramine administration resulted in significant increases in serum prolactin levels at all treatment stages with a main effect for time (F(10,20)=19.36, p<0.001; see Figure 4b), a significant interaction with treatment (F(20,40)=2.29, p=0.013), and a trend toward a main effect of treatment (F(2,4)=4.563, p=0.09), indicating that prolactin release was blunted following fluoxetine treatment. Post hoc tests did not reveal significant pairwise comparisons at any time point. However, this lack of a statistically strong treatment effect may be due to high variability in the washout condition, during which one animal's prolactin response recovered and the other two did not.
5HT2A and SERT BPs
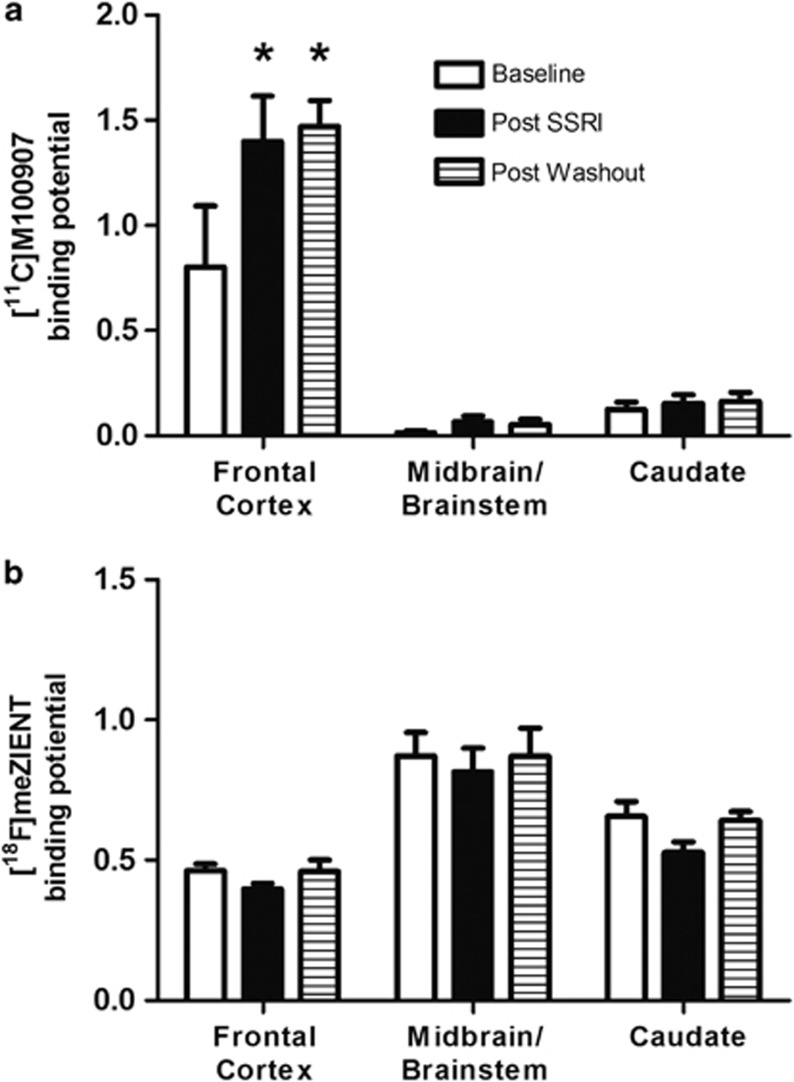
[11C]M100907 demonstrated uptake in all regions of interest (Supplementary Figures S3 and S4). [11C]M100907 uptake values in the cerebellum, the reference region, did not significantly vary between scans. BPs for [11C]M100907 are reported in Supplementary Table S2. Following chronic fluoxetine treatment, 5HT2A binding was unchanged in the caudate and midbrain/brainstem, but significantly increased in the frontal cortex (main effect for treatment, F(2,8)=6.50, p=0.021; ROI, F(2,8)=41.49, p<0.001; interaction effect, F(4,16)=6.95, p=0.002; Tukey's critical difference p<0.05; Figure 5a).
Figure 5.
Binding potentials for 5HT2A and SERT during experiment 1. (a) 5HT2A binding is significantly increased in the frontal cortex following chronic fluoxetine administration and remains elevated after 6 weeks of washout (n=5). (b) SERT binding is unchanged following chronic fluoxetine treatment (n=5). *p<0.05 with respect to ROI baseline.
[18F]FEmZIENT demonstrated uptake in all regions of interest (Supplementary Figures S5 and S6). Cerebellar uptake significantly differed across treatments (F(2,8)=5.501, p=0.031). Post hoc testing revealed that mean uptake in the post-SSRI condition significantly differed from post-washout, but post hoc testing revealed no significant differences during quasi-equilibrium, thus this difference is unlikely to affect calculations of BP. BPs for [18F]FEmZIENT are reported in Supplementary Table S2. Chronic fluoxetine treatment had no effect on SERT BPs in the frontal cortex, midbrain/brainstem, or caudate (significant main effect for ROI, F(2,8)=21.01, p<0.001; no significant treatment or interaction effect; Figure 5b).
DISCUSSION
This is the first study to examine the neurobiological changes underlying the effects of chronic fluoxetine treatment at clinically relevant concentrations in an animal model of cocaine abuse. Chronic fluoxetine treatment attenuated cocaine-primed reinstatement and cocaine-induced DA overflow, possibly through desensitization of 5HT2A receptors. Furthermore, these effects persist up to 6 weeks after the conclusion of fluoxetine treatment. However, chronic fluoxetine treatment did not disrupt ongoing cocaine self-administration behavior, and there was no effect on SERT availability or serotonin overflow.
Behavior and Neurochemistry
Previous preclinical work has demonstrated that acute administration of SSRIs attenuates cocaine self-administration (Carroll et al, 1990; Czoty et al, 2002), reinstatement (Baker et al, 2001; Ruedi-Bettschen et al, 2009), and neurochemical effects (Czoty et al, 2002). In the present study, chronic fluoxetine treatment suppressed cocaine-primed reinstatement, in agreement with the results of previous studies using acute pretreatments. Cocaine-induced DA overflow was also significantly attenuated during chronic treatment. However, this attenuation of cocaine's neurochemical effects did not affect ongoing cocaine self-administration, which is in contrast to previous preclinical studies, but in concordance with clinical studies reporting no effect of fluoxetine treatment in human cocaine users (Grabowski et al, 1995; Schmitz et al, 2001; Winstanley et al, 2011). The difference in effects between the current study and previous preclinical studies is unlikely to be due to differences in serum concentrations achieved by acute vs chronic dosing, since when chronic serum concentrations were matched using a single injection, fluoxetine significantly attenuated cocaine self-administration (see Supplementary Materials and Methods, experiment 2). Rather, it suggests the effects of chronic SSRI treatment in altering cocaine use may be mediated in part by neurobiological changes, as is hypothesized for the therapeutic effects of SSRIs in depression (Wong et al, 1995). This is further supported by the observation that these effects persisted for 6 weeks following the conclusion of fluoxetine treatment.
Given the observed reductions in cocaine-induced DA overflow, it is somewhat surprising that cocaine self-administration behavior was not affected. It is possible that the diminished DA response to cocaine was not due to fluoxetine treatment but rather the effects of long-term cocaine self-administration, as has been previously reported (Kirkland Henry et al, 2009); however, the decrease in cocaine-elicited DA reported in that study was observed in animals following the acquisition of cocaine self-administration, whereas the animals in the present study self-administered cocaine for at least 1 year before beginning fluoxetine treatment. Furthermore, Kirkland Henry and colleagues reported no differences between limited- and extended-access to cocaine, suggesting that the changes in cocaine-elicited DA overflow were due to the initiation of cocaine self-administration and not due to long-term chronic use. A second possibility is that ongoing self-administration was more robust and difficult to disrupt compared with cocaine-induced reinstatement. A likely explanation is that the repeated dosing that occurs during self-administration sessions makes it possible to overcome a decrease in the reinforcing potency of a single injection, while the single, noncontingent dose that precedes reinstatement sessions does not.
Neurobiological Mechanisms
Given the prolonged changes in cocaine-related neurochemistry and behavior following chronic fluoxetine administration, we evaluated potential mechanisms underlying these changes by examining pre- and post-synaptic markers and function. There was no change in SERT BP in any of the brain areas examined immediately following chronic treatment or the 6-week washout period, and there was a corresponding lack of change in the magnitude and time course of 5HT overflow elicited by fenfluramine. However, fenfluramine-induced prolactin release, measured concurrently, was blunted following chronic treatment. This has traditionally been used as a measure of integrated 5HT function or integrity (Newman et al, 1998) and although it may be influenced by menstrual cycle (O'Keane et al, 1991; Tanner et al, 2011), individual data showed orderly decreases across study time points. Together, these data indicate that, while measures of pre-synaptic function were unchanged, there were altered downstream, post-synaptic effects.
The 5HT2A receptor has been extensively implicated in the reinstatement and discriminative-stimulus effects of cocaine (Filip et al, 2006; Fletcher et al, 2002) as well as in the antidepressant effects of SSRIs (Carr and Lucki, 2011). Increased 5HT2A availability was observed in the frontal cortex following chronic treatment and 6 weeks post-treatment. This is unlikely to be due to changes in extracellular 5HT levels, which may compete with the radiotracer, as no changes in regulation of extracellular 5HT were observed. However, PET ligands are generally unable to detect desensitization of a receptor, and thus it is impossible to determine the functional status of a receptor solely with a PET scan. The 5HT2A receptor is involved in both mediating the prolactin response (Chaiseha et al, 2010; Jorgensen, 2007) and facilitating DA release (Bubar and Cunningham, 2008); therefore an increase in the prolactin response as well as in DA overflow would be expected. However, both the prolactin response and cocaine-induced DA overflow decreased with no change in the amount of 5HT overflow, suggesting that these receptors may in fact be desensitized. Agonist treatment is known to cause desensitization of 5HT2A receptors (Van Oekelen et al, 2003), and this is further supported by data demonstrating reduced G-protein signaling in cells (Brink et al, 2004), and reductions in 5HT2A-mediated corticosterone release in rats (Yamauchi et al, 2006) following chronic SSRI treatment. Therefore, it seems plausible that the decrease in prolactin response, cocaine-induced DA overflow, and thus, suppressed cocaine-induced reinstatement, may be due to desensitization of the 5HT2A receptor.
The continued suppression of cocaine-primed reinstatement and DA overflow even after 6 weeks of washout from the SSRI is a surprising and exciting finding. Although we hypothesize these effects to rely on the putative desensitization of the 5HT2A receptor, it is possible there is another underlying mechanism. Furthermore, it is unclear why the effects persist. The time course of recovery from desensitization for 5HT2A receptors is not known. Further investigations are needed to clarify the underlying mechanisms behind the maintenance of these effects.
SERT and 5HT2A Regulation
It is well accepted that chronic administration of SSRIs induces neurobiological changes but these have not been previously evaluated in the context of cocaine abuse, which also induces neurobiological changes (Koob and Volkow, 2010). Our data demonstrate that, in contrast to previous reports of SERT downregulation by SSRIs (Benmansour et al, 1999; Kugaya et al, 2003), prolonged fluoxetine treatment has no effect on SERT BPs in the context of ongoing cocaine use. Chronic exposure to cocaine has been shown to induce increases in SERT in primates (Banks et al, 2008; Mash et al, 2000), and thus may oppose the effects of fluoxetine on SERT regulation. Since both compounds are reuptake inhibitors, it is not clear why they affect SERT regulation differently, but it may be due to different binding kinetics or actions at other serotonergic targets.
There is less of a consensus on regulation of the 5HT2A receptor, which has been reported to both upregulate and downregulate in response to SSRIs (Audenaert et al, 2006; Meyer et al, 2001). We report a significant increase in 5HT2A receptors in the frontal cortex following chronic fluoxetine treatment in the context of ongoing cocaine use, and furthermore, a similar increase following extended cocaine exposure alone (see Experiment 3, Supplementary Table S3 and Supplementary Materials and Methods for details and additional discussion). The effects of chronic cocaine and chronic SSRI treatment were not examined in the same animals, so it is not possible to say whether the increases may be additive. Furthermore, it is possible that since experiment 1 (fluoxetine) was conducted in females while experiment 3 (cocaine) was conducted in males, the findings may not generalize to females.
Clinical Implications
This study is the first to evaluate the effects of long-term, clinically relevant serum concentrations of fluoxetine on cocaine-related neurobiology and behavior and highlights the importance of evaluating potential treatments in the context of the target population. Although previous studies have demonstrated downregulation of SERT availability in noncocaine-using populations (Kugaya et al, 2003), the results of our study suggest that this may not be the case for cocaine users and caution against the use of nondrug-using populations to predict results in drug-using populations. Furthermore, the lack of effect on ongoing self-administration behavior aligns with the results of clinical, rather than preclinical, studies, suggesting that the discrepancy in the results of these previous studies is due to differences in SSRI administration, underscoring the need to appropriately design preclinical studies to mirror the potential clinical usage of a treatment. Importantly, cocaine-primed reinstatement was suppressed, suggesting that while fluoxetine may not be effective as an intervention in ongoing cocaine abuse, it may be useful as an adjunct treatment to prevent relapse during initial or continuing abstinence, especially given the striking persistence of the effects after the conclusion of treatment. Indeed, to date only one study has examined the effects of SSRIs in preventing relapse in abstinent patients and found that treatment with sertraline delayed time to relapse in depressed cocaine-dependent patients (Oliveto et al, 2012). Our results strongly suggest additional studies would be warranted.
Acknowledgments
We acknowledge Lisa Neidert, Juliet Brown, and Paul Chen for expert technical assistance. We also thank Dr James Ritchie and Bailey Glover for providing the MS/MS analyses of fluoxetine concentrations and the Yerkes BioMarkers Core for the prolactin analyses. A portion of this work was submitted in partial fulfillment of the dissertation requirements of Emory University.
The authors declare that, except for income received from primary employers, no financial support or compensation has been received from any individual or corporate entity over the past 3 years for research or professional service and there are no personal financial holdings that could be perceived as constituting a potential conflict of interest. This work was supported by DA10344 (LLH), DA000517 (LLH), T-32 DA015040 (EKS), and RR00065 (YNPRC). A portion of this work was also supported by the Intramural Research Programs of the National Institute on Drug Abuse and the National Institute on Alcohol Abuse and Alcoholism.
Footnotes
Supplementary Information accompanies the paper on the Neuropsychopharmacology website (http://www.nature.com/npp)
Supplementary Material
References
- Audenaert K, Peremans K, Goethals I, van Heeringen C. Functional imaging, serotonin and the suicidal brain. Acta Neurol Belg. 2006;106:125–131. [PubMed] [Google Scholar]
- Baker DA, Tran-Nguyen TL, Fuchs RA, Neisewander JL. Influence of individual differences and chronic fluoxetine treatment on cocaine-seeking behavior in rats. Psychopharmacology (Berl) 2001;155:18–26. doi: 10.1007/s002130000676. [DOI] [PubMed] [Google Scholar]
- Banks ML, Czoty PW, Gage HD, Bounds MC, Garg PK, Garg S, et al. Effects of cocaine and MDMA self-administration on serotonin transporter availability in monkeys. Neuropsychopharmacology. 2008;33:219–225. doi: 10.1038/sj.npp.1301420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumann P. Pharmacokinetic-pharmacodynamic relationship of the selective serotonin reuptake inhibitors. Clin Pharmacokinet. 1996;31:444–469. doi: 10.2165/00003088-199631060-00004. [DOI] [PubMed] [Google Scholar]
- Benmansour S, Cecchi M, Morilak DA, Gerhardt GA, Javors MA, Gould GG, et al. Effects of chronic antidepressant treatments on serotonin transporter function, density, and mRNA level. J Neurosci. 1999;19:10494–10501. doi: 10.1523/JNEUROSCI.19-23-10494.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brink CB, Viljoen SL, de Kock SE, Stein DJ, Harvey BH. Effects of myo-inositol versus fluoxetine and imipramine pretreatments on serotonin 5HT2A and muscarinic acetylcholine receptors in human neuroblastoma cells. Metab Brain Dis. 2004;19:51–70. doi: 10.1023/b:mebr.0000027417.74156.5f. [DOI] [PubMed] [Google Scholar]
- Bubar MJ, Cunningham KA. Prospects for serotonin 5-HT2R pharmacotherapy in psychostimulant abuse. Prog Brain Res. 2008;172:319–346. doi: 10.1016/S0079-6123(08)00916-3. [DOI] [PubMed] [Google Scholar]
- Carr GV, Lucki I. The role of serotonin receptor subtypes in treating depression: a review of animal studies. Psychopharmacology (Berl) 2011;213:265–287. doi: 10.1007/s00213-010-2097-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carroll ME, Lac ST, Asencio M, Kragh R. Fluoxetine reduces intravenous cocaine self-administration in rats. Pharmacol Biochem Behav. 1990;35:237–244. doi: 10.1016/0091-3057(90)90232-7. [DOI] [PubMed] [Google Scholar]
- Ceglia I, Acconcia S, Fracasso C, Colovic M, Caccia S, Invernizzi RW. Effects of chronic treatment with escitalopram or citalopram on extracellular 5-HT in the prefrontal cortex of rats: role of 5-HT1A receptors. Br J Pharmacol. 2004;142:469–478. doi: 10.1038/sj.bjp.0705800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaiseha Y, Kang SW, Leclerc B, Kosonsiriluk S, Sartsoongnoen N, El Halawani ME. Serotonin receptor subtypes influence prolactin secretion in the turkey. Gen Comp Endocrinol. 2010;165:170–175. doi: 10.1016/j.ygcen.2009.06.018. [DOI] [PubMed] [Google Scholar]
- Cunningham KA, Callahan PM. Monoamine reuptake inhibitors enhance the discriminative state induced by cocaine in the rat. Psychopharmacology (Berl) 1991;104:177–180. doi: 10.1007/BF02244175. [DOI] [PubMed] [Google Scholar]
- Czoty PW, Ginsburg BC, Howell LL. Serotonergic attenuation of the reinforcing and neurochemical effects of cocaine in squirrel monkeys. J Pharmacol Exp Ther. 2002;300:831–837. doi: 10.1124/jpet.300.3.831. [DOI] [PubMed] [Google Scholar]
- Filip M, Bubar MJ, Cunningham KA. Contribution of serotonin (5-HT) 5-HT2 receptor subtypes to the discriminative stimulus effects of cocaine in rats. Psychopharmacology (Berl) 2006;183:482–489. doi: 10.1007/s00213-005-0197-y. [DOI] [PubMed] [Google Scholar]
- Fletcher PJ, Grottick AJ, Higgins GA. Differential effects of the 5-HT(2A) receptor antagonist M100907 and the 5-HT(2C) receptor antagonist SB242084 on cocaine-induced locomotor activity, cocaine self-administration and cocaine-induced reinstatement of responding. Neuropsychopharmacology. 2002;27:576–586. doi: 10.1016/S0893-133X(02)00342-1. [DOI] [PubMed] [Google Scholar]
- Grabowski J, Rhoades H, Elk R, Schmitz J, Davis C, Creson D, et al. Fluoxetine is ineffective for treatment of cocaine dependence or concurrent opiate and cocaine dependence: two placebo-controlled double-blind trials. J Clin Psychopharmacol. 1995;15:163–174. doi: 10.1097/00004714-199506000-00004. [DOI] [PubMed] [Google Scholar]
- Hinz R, Bhagwagar Z, Cowen PJ, Cunningham VJ, Grasby PM. Validation of a tracer kinetic model for the quantification of 5-HT(2A) receptors in human brain with [(11)C]MDL 100,907. J Cereb Blood Flow Metab. 2007;27:161–172. doi: 10.1038/sj.jcbfm.9600323. [DOI] [PubMed] [Google Scholar]
- Howell LL, Wilcox KM. The dopamine transporter and cocaine medication development: drug self-administration in nonhuman primates. J Pharmacol Exp Ther. 2001a;298:1–6. [PubMed] [Google Scholar]
- Howell LL, Wilcox KM.2001bIntravenous drug self-administration in nonhuman primatesIn: Buccafusco JJ (ed).Methods of Behavior Analysis in Neuroscience CRC Press: Boca Raton, FL; 91–110. [Google Scholar]
- Ito H, Nyberg S, Halldin C, Lundkvist C, Farde L. PET imaging of central 5-HT2A receptors with carbon-11-MDL 100,907. J Nucl Med. 1998;39:208–214. [PubMed] [Google Scholar]
- Jorgensen HS. Studies on the neuroendocrine role of serotonin. Dan Med Bull. 2007;54:266–288. [PubMed] [Google Scholar]
- Kirkland Henry P, Davis M, Howell LL. Effects of cocaine self-administration history under limited and extended access conditions on in vivo striatal dopamine neurochemistry and acoustic startle in rhesus monkeys. Psychopharmacology (Berl) 2009;205:237–247. doi: 10.1007/s00213-009-1534-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kish SJ, Furukawa Y, Chang LJ, Tong J, Ginovart N, Wilson A, et al. Regional distribution of serotonin transporter protein in postmortem human brain: is the cerebellum a SERT-free brain region. Nucl Med Biol. 2005;32:123–128. doi: 10.1016/j.nucmedbio.2004.10.001. [DOI] [PubMed] [Google Scholar]
- Koob GF, Volkow ND. Neurocircuitry of addiction. Neuropsychopharmacology. 2010;35:217–238. doi: 10.1038/npp.2009.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreiss DS, Lucki I. Effects of acute and repeated administration of antidepressant drugs on extracellular levels of 5-hydroxytryptamine measured in vivo. J Pharmacol Exp Ther. 1995;274:866–876. [PubMed] [Google Scholar]
- Kugaya A, Seneca NM, Snyder PJ, Williams SA, Malison RT, Baldwin RM, et al. Changes in human in vivo serotonin and dopamine transporter availabilities during chronic antidepressant administration. Neuropsychopharmacology. 2003;28:413–420. doi: 10.1038/sj.npp.1300036. [DOI] [PubMed] [Google Scholar]
- Lammertsma AA, Hume SP. Simplified reference tissue model for PET receptor studies. Neuroimage. 1996;4 (3 Part 1:153–158. doi: 10.1006/nimg.1996.0066. [DOI] [PubMed] [Google Scholar]
- Marek GJ, Martin-Ruiz R, Abo A, Artigas F. The selective 5-HT2A receptor antagonist M100907 enhances antidepressant-like behavioral effects of the SSRI fluoxetine. Neuropsychopharmacology. 2005;30:2205–2215. doi: 10.1038/sj.npp.1300762. [DOI] [PubMed] [Google Scholar]
- Mash DC, Staley JK, Izenwasser S, Basile M, Ruttenber AJ. Serotonin transporters upregulate with chronic cocaine use. J Chem Neuroanatomy. 2000;20:271–280. doi: 10.1016/s0891-0618(00)00102-2. [DOI] [PubMed] [Google Scholar]
- Meyer JH, Kapur S, Eisfeld B, Brown GM, Houle S, DaSilva J, et al. The effect of paroxetine on 5-HT(2A) receptors in depression: an [(18)F]setoperone PET imaging study. Am J Psychiatry. 2001;158:78–85. doi: 10.1176/appi.ajp.158.1.78. [DOI] [PubMed] [Google Scholar]
- Moeller FG, Schmitz JM, Steinberg JL, Green CM, Reist C, Lai LY, et al. Citalopram combined with behavioral therapy reduces cocaine use: a double-blind, placebo-controlled trial. Am J Drug Alcohol Abuse. 2007;33:367–378. doi: 10.1080/00952990701313686. [DOI] [PubMed] [Google Scholar]
- Newman ME, Shapira B, Lerer B. Evaluation of central serotonergic function in affective and related disorders by the fenfluramine challenge test: a critical review. Int J Neuropsychopharmacol. 1998;1:49–69. doi: 10.1017/S1461145798001072. [DOI] [PubMed] [Google Scholar]
- O'Keane V, O'Hanlon M, Webb M, Dinan T. d-fenfluramine/prolactin response throughout the menstrual cycle: evidence for an oestrogen-induced alteration. Clin Endocrinol (Oxf) 1991;34:289–292. doi: 10.1111/j.1365-2265.1991.tb03768.x. [DOI] [PubMed] [Google Scholar]
- Oliveto A, Poling J, Mancino MJ, Williams DK, Thostenson J, Pruzinsky R, et al. Sertraline delays relapse in recently abstinent cocaine-dependent patients with depressive symptoms (1) Addiction. 2012;107:131–141. doi: 10.1111/j.1360-0443.2011.03552.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons LH, Justice JB., Jr Serotonin and dopamine sensitization in the nucleus accumbens, ventral tegmental area, and dorsal raphe nucleus following repeated cocaine administration. J Neurochem. 1993;61:1611–1619. doi: 10.1111/j.1471-4159.1993.tb09794.x. [DOI] [PubMed] [Google Scholar]
- Qu Y, Aluisio L, Lord B, Boggs J, Hoey K, Mazur C, et al. Pharmacokinetics and pharmacodynamics of norfluoxetine in rats: Increasing extracellular serotonin level in the frontal cortex. Pharmacol Biochem Behav. 2009;92:469–473. doi: 10.1016/j.pbb.2009.01.023. [DOI] [PubMed] [Google Scholar]
- Riad M, Rbah L, Verdurand M, Aznavour N, Zimmer L, Descarries L. Unchanged density of 5-HT(1A) autoreceptors on the plasma membrane of nucleus raphe dorsalis neurons in rats chronically treated with fluoxetine. Neuroscience. 2008;151:692–700. doi: 10.1016/j.neuroscience.2007.11.024. [DOI] [PubMed] [Google Scholar]
- Ruedi-Bettschen D, Rowlett JK, Spealman RD, Platt DM. Attenuation of cocaine-induced reinstatement of drug seeking in squirrel monkeys: kappa opioid and serotonergic mechanisms. Psychopharmacology (Berl) 2009;210:169–177. doi: 10.1007/s00213-009-1705-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawyer EK, Howell LL. Pharmacokinetics of fluoxetine in rhesus macaques following multiple routes of administration. Pharmacology. 2011;88:44–49. doi: 10.1159/000329417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schama KF, Howell LL, Byrd LD. Serotonergic modulation of the discriminative-stimulus effects of cocaine in squirrel monkeys. Psychopharmacology (Berl) 1997;132:27–34. doi: 10.1007/s002130050316. [DOI] [PubMed] [Google Scholar]
- Schmitz JM, Averill P, Stotts AL, Moeller FG, Rhoades HM, Grabowski J. Fluoxetine treatment of cocaine-dependent patients with major depressive disorder. Drug Alcohol Depend. 2001;63:207–214. doi: 10.1016/s0376-8716(00)00208-8. [DOI] [PubMed] [Google Scholar]
- Smith TD, Kuczenski R, George-Friedman K, Malley JD, Foote SL. In vivo microdialysis assessment of extracellular serotonin and dopamine levels in awake monkeys during sustained fluoxetine administration. Synapse. 2000;38:460–470. doi: 10.1002/1098-2396(20001215)38:4<460::AID-SYN11>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- Stehouwer JS, Jarkas N, Zeng F, Voll RJ, Williams L, Camp VM, et al. Synthesis, radiosynthesis, and biological evaluation of fluorine-18-labeled 2beta-carbo(fluoroalkoxy)-3beta-(3′-((Z)-2-haloethenyl)phenyl)nortropanes: candidate radioligands for in vivo imaging of the serotonin transporter with positron emission tomography. J Med Chem. 2008;51:7788–7799. doi: 10.1021/jm800781a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Substance Abuse and Mental Health Services Administration 2010Results from the 2009 National Survey on Drug Use and Health: Volume I Summary of National Findings(Office of Applied Studies, NSDUH Series H-38A, HHS Publication No SMA 10-4586 Findings) Rockville, MD.
- Tanner MJ, Hadlow NC, Wardrop R. Variation of female prolactin levels with menopausal status and phase of menstrual cycle. Aust N Z J Obstet Gynaecol. 2011;51:321–324. doi: 10.1111/j.1479-828X.2011.01321.x. [DOI] [PubMed] [Google Scholar]
- Van Oekelen D, Luyten WH, Leysen JE. 5-HT2A and 5-HT2C receptors and their atypical regulation properties. Life Sci. 2003;72:2429–2449. doi: 10.1016/s0024-3205(03)00141-3. [DOI] [PubMed] [Google Scholar]
- Walsh SL, Preston KL, Sullivan JT, Fromme R, Bigelow GE. Fluoxetine alters the effects of intravenous cocaine in humans. J Clin Psychopharmacol. 1994;14:396–407. [PubMed] [Google Scholar]
- Winstanley EL, Bigelow GE, Silverman K, Johnson RE, Strain EC. A randomized controlled trial of fluoxetine in the treatment of cocaine dependence among methadone-maintained patients. J Subst Abuse Treat. 2011;40:255–264. doi: 10.1016/j.jsat.2010.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong DT, Bymaster FP, Engleman EA. Prozac (fluoxetine, Lilly 110140), the first selective serotonin uptake inhibitor and an antidepressant drug: twenty years since its first publication. Life Sci. 1995;57:411–441. doi: 10.1016/0024-3205(95)00209-o. [DOI] [PubMed] [Google Scholar]
- Yamauchi M, Miyara T, Matsushima T, Imanishi T. Desensitization of 5-HT2A receptor function by chronic administration of selective serotonin reuptake inhibitors. Brain Res. 2006;1067:164–169. doi: 10.1016/j.brainres.2005.10.075. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.