Abstract
Metastatic non-small-cell lung cancer (nsclc) is the leading cause of cancer mortality in Canada. Although treatment outcomes in advanced disease remain modest, with paradigm shifts in the approach to treatment, they are steadily improving. Customizing treatment based on histology and molecular typing has become the standard of care. EGFR genotyping and pathology subtyping should be considered routine in new diagnoses of metastatic nsclc. Treatment options for those with somatic EGFR activating mutations include gefitinib until progression, followed by standard chemotherapy. For patients with wild-type EGFR, or in patients whose EGFR genotype is unknown, platinum-based chemotherapy remains the first-line standard, with single-agent chemotherapy as an option for older patients and those who are unfit for platinum-doublet therapy. Patients with nonsquamous histology may receive treatment regimens incorporating pemetrexed or bevacizumab. In patients with squamous cell carcinoma, the latter agents should be avoided because of concerns about enhanced toxicity or decreased efficacy. Second-line chemotherapy is offered to a selected subgroup of patients upon progression and may include pemetrexed in non-squamous histology and docetaxel or erlotinib (or both) in all histologies. Currently, only erlotinib is offered as a third-line option in unselected nsclc patients after failure of first- and second-line chemotherapy. Maintenance therapy is emerging as a new option for patients, as are targeted therapies for particular molecular subtypes of nsclc, such as crizotinib in tumours harbouring the EML4–ALK gene rearrangement.
Keywords: Metastatic non-small-cell lung cancer, nsclc, advanced, systemic therapy, chemotherapy
1. INTRODUCTION
At more than 25,000 new cases, lung cancer remains the most commonly diagnosed cancer among Canadians (excluding non-melanoma skin cancer); it also causes the greatest number of cancer-related deaths (more than 20,000 deaths per year) 1. As well, more than half the Canadians diagnosed with lung cancer present with metastatic disease. Most lung cancer patients, approximately 85%, have non-small-cell lung cancer (nsclc). Of those, approximately 40%–60% have adenocarcinoma histology; 10%–15%, squamous histology; 5%, neuroendocrine histology; and the rest, “not otherwise specified” 2,3.
Survival is short in the setting of advanced nsclc, a median of 4–6 months, and more than 80% patients have multiple severe cancer-related symptoms 4–6. Systemic therapy, the mainstay of treatment in advanced nsclc, can improve survival by up to 8–12 months in selected patients and can improve symptom control and quality of life in 60%–70% despite treatment toxicity 4,6–8.
Since the year 2000, exciting developments have occurred in the treatment of lung cancer, resulting in a paradigm shift. The approach to lung cancer has evolved from a general treatment approach based on ability to withstand the toxicities of chemotherapy, to one that incorporates important predictive markers of benefit from specific agents, including pathology subtype (histology) and molecular genotype [such as the presence of epidermal growth factor receptor (EGFR) activating mutations in tumour tissue]. This paradigm shift has allowed for improvements in response rates, quality of life, toxicity of treatment, and progression-free and overall survival in patients with metastatic nsclc 9–14.
2. ESSENTIAL DIAGNOSTIC ELEMENTS IN METASTATIC NSCLC
With the number of attractive treatment options for patients with non-squamous nsclc increasing, the pathologist’s contribution to the patient’s treatment plan has become key. In addition to confirmation of a diagnosis of cancer, the clinician relies on the pathologist to determine that the cancer is of pulmonary origin; to define the pathology subtype, if possible (that is, adenocarcinoma, squamous cell carcinoma, or neuroendocrine tumour); and to perform molecular testing. Testing for the presence of EGFR mutation and, in the near future, an ALK-rearranged gene or protein product are also essential to optimal treatment planning for the advanced nsclc patient 15–18.
To address the paradigm shift in lung cancer treatment, the International Association for the Study of Lung Cancer published a new international multidisciplinary classification of lung adenocarcinoma, the subtype in which advances in therapy have occurred 15. The recommendations support pathologic subtyping (whenever possible) and EGFR mutation testing, with guidelines for pathologists processing small biopsies and cytology samples. Canadian consensus guidelines for biomarkers in lung cancer have also been developed, as have helpful criteria from a Canadian perspective for immunohistochemical testing to establish pulmonary origin and pathology subtype, and for molecular testing 16,18.
Currently, as many as 30%–50% of Canadian patients with advanced nsclc do not have a pathology subtype reported, although the proportion is significantly lower in academic centres, in the presence of larger diagnostic samples, and in centres that routinely use immunohistochemistry as part of routine lung cancer diagnosis 2,3,19. In the Canadian EGFR testing program initiated in 2010, it was estimated that EGFR testing was requested in approximately 37% of potentially eligible patients (that is, nonsquamous, incurable nsclc) 20. Mutation testing did not proceed in 12% of cases, because samples were either inadequate or were not submitted to 1 of 5 designated testing laboratories across the country. The median time from initiation of testing to final result was 18 ± 9.7 days (range: 15–26 days), including a median of 7 days for the sample to arrive at the testing centre and 11 days for actual testing. Testing is reported to take longer in provinces or centres that do not perform EGFR testing, with time to retrieve and ship samples being a major cause of delay. Thus, for many patients, neither pathology subtype nor EGFR genotype will be adequately defined in time for first-line treatment decision-making. Ensuring sufficient lung cancer tissue at the time of diagnosis and systematically reporting pathology subtype and EGFR genotype are key issues to be addressed.
3. FIRST-LINE THERAPY IN METASTATIC NSCLC
Current standards for advanced nsclc include first-line platinum-based doublet therapy, second-line chemotherapy, and erlotinib [an egfr tyrosine kinase inhibitor (tki)] in all patients after chemotherapy failure 4,11,12,21–23. However, treatment may be further customized by EGFR genotype and pathology subtype or histology, as described next.
3.1. Treatment Options by EGFR Status: EGFR Mutation–Positive NSCLC
EGFR activating mutations are detected in 12%–17% of nsclc cases diagnosed in North America, predominantly in adenocarcinoma or non-squamous histology 20,24,25. These mutations are an important predictive factor for first-line treatment with egfr-tki therapy. Several studies have now demonstrated dramatic improvement in response rates, quality of life, symptoms, and median progression-free survival (by 2–5 months) with first-line egfr-tki therapy compared with standard platinum-doublet chemotherapy in patients with EGFR mutation–positive nsclc 9,10,26–29. Treatment with an egfr-tki is also associated with a more favourable toxicity profile than is standard chemotherapy; it also has the greater convenience of oral administration. In 2012, gefitinib is the only Health Canada–approved egfr-tki treatment for advanced nsclc. Similar data for erlotinib are available, and pending studies suggest that more egfr-tkis with similar results may become available in the near future.
Initial egfr-tki treatment in patients with EGFR mutation–positive advanced nsclc yields superior response rates of 58%–85% in all trials, compared with 15%–47% with chemotherapy 9,10,26–29. Treatment with egfr-tki is associated with greater symptom improvement and quality of life 9,27 and superior progression-free survival. Hazard ratios for progression-free survival in the relevant studies range from 0.16 to 0.61, statistically significant in all but one trial (and that trial trended toward significance at p = 0.08) 27. A significant difference in overall survival has not been demonstrated in those studies, but that endpoint is confounded by high rates of crossover to egfr-tki, with subsequent benefit in the patients initially randomized to chemotherapy.
An important lesson learned from the trials is that patients with EGFR wild-type disease fare better on first-line platinum-based chemotherapy than on egfr-tkis, in terms of greater response rates, better quality of life, and improvement in symptoms and progression-free survival 9,27. Accordingly, patients should receive an egfr-tki instead of chemotherapy in the first-line setting only if they have documented EGFR mutation–positive nsclc, so as to avoid potential harm in those without EGFR mutations.
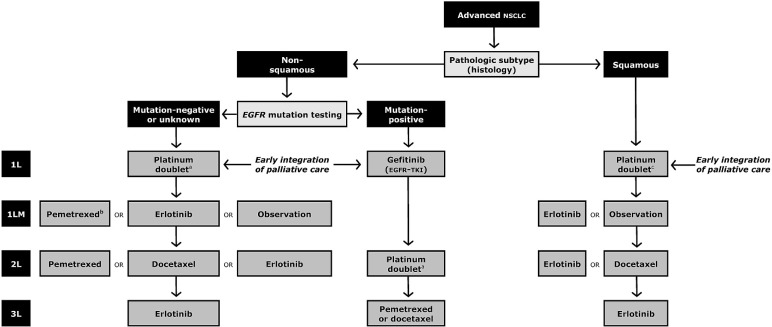
Thus, first-line treatment with gefitinib until progression in patients with incurable stage iiib/iv EGFR mutation–positive nsclc is associated with better outcomes during initial therapy and with less toxicity than that seen with standard platinum-based doublet chemotherapy. Although this approach does not introduce a new line of treatment for this subgroup (who previously would have received erlotinib after chemotherapy failure), it optimizes the sequence of treatment. Figure 1 outlines current standards for EGFR mutation–positive lung cancer, which would include first-line egfr-tki until progression, followed by platinum-based and subsequent chemotherapy. But these patients may also be treated with the traditional sequence of first-line platinum-doublet chemotherapy followed by second-line chemotherapy or erlotinib. Those strategies are associated with similar survival, although lesser progression-free survival, symptom improvement, and greater toxicity than are seen in the first-line setting with egfr-tki treatment.
FIGURE 1.
Algorithm for first- [1L, 1LM (maintenance)], second- (2L), and third-line (3L) therapy in advanced non-small-cell lung cancer (nsclc). Subsequent lines of treatment assume no previous exposure to the agent about to be used. EGFR = epidermal growth factor receptor gene. egfr-tki = epidermal growth factor receptor tyrosine kinase inhibitor. a Includes pemetrexed-based doublet. b As maintenance after non-pemetrexed-containing doublets only. c Excludes pemetrexed doublets.
3.2. Treatment Options in Wild-Type EGFR or Unknown EGFR Genotype
Most advanced nsclc patients in Canada have EGFR wild-type nsclc or unknown EGFR status because testing has not been performed or is unavailable at the time of treatment decision-making. First-line therapy with platinum doublets for 4–6 cycles is the standard treatment for those who have good performance status (Eastern Cooperative Oncology Group 0 or 1, and selected patients with performance status 2) 7. Platinum doublets have similar outcomes, with response rates ranging from 20% to 30%, median time to progression of 3.5–5 months, and median overall survival of 8–12 months in selected patients 12,21. Differences between trials are often related to different patient populations: for example, greater inclusion of those with stage iiib disease (more favourable outcomes) or exclusion of those with performance status 2 (less favourable outcomes). Gemcitabine with either cisplatin or carboplatin is one of the most commonly used regimens, followed by paclitaxel–carboplatin, vinorelbine–platinum, and docetaxel–platinum combinations 30,31. Cisplatin has minor superiority over carboplatin, with a higher response rate (30% vs. 24%) and a median survival time approximately 1 month longer 32,33. Cisplatin has less potential to cause alopecia and thrombocytopenia, but it causes greater emesis and renal and ototoxicity. For older patients and those not fit enough for platinum combination therapy, single-agent therapy such as vinorelbine, gemcitabine, or taxanes are offered 7. Some centres also offer non-platinum-based doublets that have been shown to have similar efficacy in clinical trials, such as gemcitabine–taxane combinations or gemcitabine–vinorelbine 34,35.
When tailoring treatment by histology, those with non-squamous nsclc have more therapeutic options than do their counterparts with squamous carcinoma of the lung. They are also more likely to have tumours that harbour EGFR mutations or ALK rearrangements, with newer highly active treatment options available 36,37. The introduction of pemetrexed into the platinum doublet may improve outcomes for patients with non-squamous nsclc (predominantly adenocarcinoma). A randomized trial of pemetrexed–cisplatin compared with gemcitabine–cisplatin demonstrated similar outcomes in unselected nsclc in terms of response and progression-free and overall survival 12. But a preplanned analysis of outcomes by histology revealed an approximate 1.5-month median survival gain with pemetrexed–cisplatin in non-squamous nsclc patients, with response rates and progression-free survival similar to those seen with gemcitabine–cisplatin. Patients with squamous histology appeared to derive a similar magnitude of median survival benefit from gemcitabine–cisplatin as from pemetrexed–cisplatin therapy, allowing for further refinements in the selection of first-line therapy to improve outcomes in both pathologic subtypes. Given the incremental cost of pemetrexed, this newer regimen is funded only in a select number of provinces as part of first-line therapy in non-squamous nsclc. Thus, many provinces continue to use gemcitabine–platinum as their standard, regardless of histology. The other platinum doublets are indicated in all nsclc subtypes, with no evidence to support differential efficacy between histologies with the taxane- or vinorelbine-based platinum combinations 31,38,39.
The addition of selected novel agents to the platinum doublet may also improve outcomes. Bevacizumab is currently approved by Health Canada in combination with paclitaxel–carboplatin as first-line therapy in advanced non-squamous nsclc. Squamous histology became a major exclusion criterion for bevacizumab trials in advanced nsclc after a randomized phase ii study of paclitaxel, carboplatin, and bevacizumab revealed a clear association between squamous cell carcinoma and an increased risk of major hemoptysis 40. In the Eastern Cooperative Oncology Group 4599 trial, bevacizumab added to paclitaxel–carboplatin improved median survival by 2 months in non-squamous patients, with higher response rates and longer progression-free survival. In a subgroup analysis of patients with adenocarcinoma, survival was improved from a median of 10 months with the platinum doublet to 14 months with the addition of bevacizumab 14,41. A similar study using gemcitabine–cisplatin-based chemotherapy did not demonstrate a survival advantage despite higher response rates and longer progression-free survival 42. Although bevacizumab has become an important component of advanced non-squamous nsclc treatment in the United States, it is not funded through Canadian provincial health care systems because of the high incremental cost and the negative results of the confirmatory trial.
By contrast, cetuximab added to platinum-based chemotherapy has not been approved by Health Canada. One phase iii study (in all pathology subtypes) demonstrated a median survival benefit of 1 month with the addition of cetuximab compared with vinorelbine–cisplatin chemotherapy alone, and another demonstrated better response rates and progression-free survival with cetuximab added to paclitaxel–carboplatin, but no improvement in overall survival was seen 43,44.
3.3. Duration of First-Line Therapy
More than 4–6 cycles of platinum-based therapy does not significantly improve patient outcomes, but does lead to increased toxicity 7,45. The option of switching to non-cross-resistant therapy or of maintaining at least a single agent from first-line combination therapy may yield better outcomes. Pemetrexed in non-squamous nsclc patients with stable or better disease after a non-pemetrexed-containing doublet, and erlotinib in all nsclc patients with stable disease after platinum chemotherapy are options approved by Health Canada for maintenance treatment 46,47. Those options are further discussed elsewhere in this supplement.
4. SECOND- AND THIRD-LINE THERAPY IN ADVANCED NSCLC
Approximately 30% of patients that receive first-line therapy are candidates for subsequent treatment. Second-line chemotherapy with docetaxel has been shown to improve survival by a median of 3 months (despite response rates of less than 10%) and to delay deterioration of quality of life in patients with a good performance status 22,48. It has also been demonstrated to be cost-effective and to reduce requirements for transfusions, radiotherapy, and pain medications 49.
Docetaxel is approved as second-line therapy by Health Canada in all nsclc histologies, but pemetrexed is approved for use in non-squamous nsclc only and has emerged as the preferred option in this subgroup because of its favourable toxicity profile. In a randomized comparison, outcomes were similar with pemetrexed and docetaxel, but a subgroup analysis revealed significantly superior outcomes with pemetrexed in non-squamous patients and inferior outcomes in squamous cell disease (median of 9.3 months vs. 6.2 months respectively; response rate: 11.5% vs. 2.8%) 11,13. Docetaxel demonstrated similar activity in squamous and non-squamous patients. While not approved for second-line therapy in Canada, topotecan, vinflunine, and gefitinib have also demonstrated outcomes similar to those with docetaxel in randomized trials 50–52.
After chemotherapy failure, second- or third-line erlotinib has been shown to improve survival and symptoms; it was also marginally cost-effective in a placebo-controlled trial in unselected advanced nsclc patients 23,53,54. Erlotinib is the only third-line therapy approved by Health Canada in advanced nsclc, being indicated for use in both squamous and non-squamous histology, and in both EGFR wild-type and mutated nsclc. In the ncic Clinical Trials Group br.21 trial, adenocarcinoma emerged as a predictor of survival in the multivariate analysis, although patients of all histologies appeared to derive benefit. EGFR mutation predicted response to erlotinib, although it did not predict a differential survival benefit in that trial, perhaps because of small numbers of mutation-positive patients 24.
5. BEYOND SYSTEMIC THERAPY: EARLY REFERRAL TO PALLIATIVE CARE
Although systemic therapy remains the mainstay of treatment in advanced nsclc, a recent randomized trial highlighted the importance of supportive care in lung cancer. At a major U.S. centre, Temel and colleagues randomized 151 patients to early palliative care integrated with their ongoing oncology care or to standard oncology care alone 55. Patients assigned to receive early palliative care had a better quality of life and less depression; they also were less likely to receive futile chemotherapy and aggressive care at the end of life (33% vs. 54% in the standard arm, p = 0.05). Despite less treatment at the end of life, the patients receiving palliative care lived longer, achieving a median survival of 11.6 months compared with 8.9 months in the standard arm (p = 0.02). This gain in median survival is similar in magnitude to the gain offered by many chemotherapy advances. Thus, Canadians with advanced nsclc should be offered palliative care referral and supportive services early in the course of their disease.
6. PERSONALIZED THERAPY—FUTURE DIRECTIONS
Current research is focused not only on developing new agents in lung cancer, but also on identifying potential predictors of treatment benefit, with earlier integration of molecular hypotheses into lung cancer trials. An excellent example is the discovery of a subgroup of nsclc patients whose tumours contain the EML4–ALK fusion gene (approximately 4% of adenocarcinomas) and potentially another subgroup with ROS1 rearrangements 37,56. In those patients, who tend to be younger, never-smokers or light smokers, with EGFR wild-type tumours, therapy with crizotinib, an oral inhibitor of Alk and Met kinases, has produced dramatic and prolonged responses 17. In the next year, as crizotinib is integrated into this rapidly evolving treatment paradigm, Canadian oncologists and pathologists will need to add ALK testing to their list of essential diagnostic elements in lung cancer.
Although overall progress has been slow and steady, the last decade has witnessed multiple major breakthroughs in the understanding of lung cancer pathogenesis (especially in never-smokers) and major treatment advances. Histologic, clinical, and molecular selection is helping clinicians to identify the patients who may benefit most from these novel therapies.
7. CONFLICT OF INTEREST DISCLOSURES
NL has no personal or financial conflicts of interest to declare. The University Health Network received clinical trial funding from AstraZeneca (2003) and Roche (2005) for investigator-initiated trials.
8. REFERENCES
- 1.Canadian Cancer Society’s Steering Committee on Cancer Statistics . Canadian Cancer Statistics 2011. Toronto, ON: Canadian Cancer Society; 2011. [Google Scholar]
- 2.Sulpher JA, Owen SP, Hon H, et al. Factors influencing a specific histologic diagnosis of non-small cell lung cancer [abstract 7541] J Clin Oncol. 2011. p. 29. [Available online at: http://www.asco.org/ASCOv2/Meetings/Abstracts?&vmview=abst_detail_view&confID=102&abstractID=81096; cited April 21, 2012] [DOI] [PubMed]
- 3.Nadjafi M, Santos GDC, Le L, Hwang D, Tsao MS, Leighl NB. Diagnostic patterns of nsclc at Princess Margaret Hospital [abstract e18027] J Clin Oncol. 2011;29 [Available online at: http://www.asco.org/ascov2/Meetings/Abstracts?&vmview=abst_detail_view&confID=102&abstractID=82739; cited April 23, 2012] [Google Scholar]
- 4.Chemotherapy for non-small cell lung cancer Non-small Cell Lung Cancer Collaborative Group. Cochrane Database Syst Rev. 2000:CD002139. doi: 10.1002/14651858.CD002139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hollen PJ, Gralla RJ, Kris MG, Eberly SW, Cox C. Normative data and trends in quality of life from the Lung Cancer Symptom Scale (lcss) Support Care Cancer. 1999;7:140–8. doi: 10.1007/s005200050244. [DOI] [PubMed] [Google Scholar]
- 6.Sörenson S, Glimelius B, Nygren P, on behalf of the sbu-Group Swedish Council of Technology Assessment in Health Care. A systematic overview of chemotherapy effects in non-small cell lung cancer. Acta Oncol. 2001;40:327–39. doi: 10.1080/02841860151116402. [DOI] [PubMed] [Google Scholar]
- 7.Goffin J, Lacchetti C, Ellis PM, et al. First-line systemic chemotherapy in the treatment of advanced non-small cell lung cancer: a systematic review. J Thorac Oncol. 2010;5:260–74. doi: 10.1097/JTO.0b013e3181c6f035. [DOI] [PubMed] [Google Scholar]
- 8.Paesmans M. Benefits of chemotherapy for quality of life in patients with advanced non-small-cell lung cancer. Curr Opin Oncol. 2002;14:389–93. doi: 10.1097/00001622-200207000-00003. [DOI] [PubMed] [Google Scholar]
- 9.Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin–paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–57. doi: 10.1056/NEJMoa0810699. [DOI] [PubMed] [Google Scholar]
- 10.Maemondo M, Inoue A, Kobayashi K, et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362:2380–8. doi: 10.1056/NEJMoa0909530. [DOI] [PubMed] [Google Scholar]
- 11.Hanna N, Shepherd FA, Fossella FV, et al. Randomized phase iii trial of pemetrexed versus docetaxel in patients with non-small-cell lung cancer previously treated with chemotherapy. J Clin Oncol. 2004;22:1589–97. doi: 10.1200/JCO.2004.08.163. [DOI] [PubMed] [Google Scholar]
- 12.Scagliotti GV, Parikh P, von Pawel J, et al. Phase iii study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol. 2008;26:3543–51. doi: 10.1200/JCO.2007.15.0375. [DOI] [PubMed] [Google Scholar]
- 13.Scagliotti G, Brodowicz T, Shepherd FA, et al. Treatment-by-histology interaction analyses in three phase iii trials show superiority of pemetrexed in nonsquamous non-small cell lung cancer. J Thorac Oncol. 2011;6:64–70. doi: 10.1097/JTO.0b013e3181f7c6d4. [DOI] [PubMed] [Google Scholar]
- 14.Sandler A, Gray R, Perry MC, et al. Paclitaxel–carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. 2006;355:2542–50. doi: 10.1056/NEJMoa061884. [DOI] [PubMed] [Google Scholar]
- 15.Travis WD, Brambilla E, Noguchi M, et al. on behalf of the American Thoracic Society International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society international multidisciplinary classification of lung adenocarcinoma. Proc Am Thorac Soc. 2011;8:381–5. doi: 10.1513/pats.201107-042ST. [DOI] [PubMed] [Google Scholar]
- 16.Dubinski W, Leighl NB, Tsao MS, Hwang DM. Ancillary testing in lung cancer diagnosis. Pulm Med. 2012;2012:249082. doi: 10.1155/2012/249082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kwak EL, Bang YJ, Camidge DR, et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med. 2010;363:1693–703. doi: 10.1056/NEJMoa1006448. [Erratum in: N Engl J Med 2011;364:588] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ellis PM, Blais N, Soulieres D, et al. A systematic review and Canadian consensus recommendations on the use of biomarkers in the treatment of non-small cell lung cancer. J Thorac Oncol. 2011;6:1379–91. doi: 10.1097/JTO.0b013e318220cb8e. [DOI] [PubMed] [Google Scholar]
- 19.Canadian Partnership Against Cancer (cpac) Lung Cancer in Canada: A Supplemental System Performance Report. Toronto, ON: CPAC; May, 2011. [Available online at: http://www.cancerview.ca/idc/groups/public/documents/webcontent/cpac015706.pdf; cited April 29, 2012] [Google Scholar]
- 20.Ellis PM, Verma S, Sehdev S, Younas J, Leighl N. Implementation of a national EGFR testing strategy in a publicly funded health system [abstract P4.199] J Thorac Oncol. 2011;6(suppl 2):S1513. doi: 10.1097/JTO.0b013e31829f6a43. [DOI] [PubMed] [Google Scholar]
- 21.Schiller JH, Harrington D, Belani CP, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med. 2002;346:92–8. doi: 10.1056/NEJMoa011954. [DOI] [PubMed] [Google Scholar]
- 22.Shepherd FA, Dancey J, Ramlau R, et al. Prospective randomized trial of docetaxel versus best supportive care in patients with non-small cell lung cancer previously treated with platinum-based chemotherapy. J Clin Oncol. 2000;18:2095–103. doi: 10.1200/JCO.2000.18.10.2095. [DOI] [PubMed] [Google Scholar]
- 23.Shepherd FA, Rodrigues Pereira J, Ciuleanu T, et al. on behalf of the National Cancer Institute of Canada Clinical Trials Group Erlotinib in previously treated non-small-cell lung cancer. N Engl J Med. 2005;353:123–32. doi: 10.1056/NEJMoa050753. [DOI] [PubMed] [Google Scholar]
- 24.Zhu CQ, da Cunha Santos G, Ding K, et al. Role of KRAS and EGFR as biomarkers of response to erlotinib in National Cancer Institute of Canada Clinical Trials Group Study br.21. J Clin Oncol. 2008;26:4268–75. doi: 10.1200/JCO.2007.14.8924. [DOI] [PubMed] [Google Scholar]
- 25.Bell DW, Lynch TJ, Haserlat SM, et al. Epidermal growth factor receptor mutations and gene amplification in non-small cell lung cancer: molecular analysis of the ideal/intact gefitinib trials. J Clin Oncol. 2005;23:8081–92. doi: 10.1200/JCO.2005.02.7078. [DOI] [PubMed] [Google Scholar]
- 26.Mitsudomi T, Morita S, Yatabe Y, et al. on behalf of the West Japan Oncology Group Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (wjtog 3405): an open label, randomised phase 3 trial. Lancet Oncol. 2010;11:121–8. doi: 10.1016/S1470-2045(09)70364-X. [DOI] [PubMed] [Google Scholar]
- 27.Han JY, Park K, Kim SW, et al. First-signal: first-line single-agent Iressa versus gemcitabine and cisplatin trial in never-smokers with adenocarcinoma of the lung. J Clin Oncol. 2012;30:1122–8. doi: 10.1200/JCO.2011.36.8456. [DOI] [PubMed] [Google Scholar]
- 28.Zhou C, Wu YL, Chen G, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (optimal, ctong-0802): a multicentre, open-label, randomized, phase 3 study. Lancet Oncol. 2011;12:735–42. doi: 10.1016/S1470-2045(11)70184-X. [DOI] [PubMed] [Google Scholar]
- 29.Rosell R, Carcereny E, Gervais R, et al. on behalf of the Spanish Lung Cancer Group in collaboration with Groupe Français de Pneumo-Cancérologie and Associazione Italiana Oncologia Toracica Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (eurtac): a multicentre, open-label, randomized phase 3 trial. Lancet Oncol. 2012;13:239–46. doi: 10.1016/S1470-2045(11)70393-X. [DOI] [PubMed] [Google Scholar]
- 30.Kelly K, Crowley J, Bunn PA, Jr, et al. Randomized phase iii trial of paclitaxel plus carboplatin versus vinorelbine plus cisplatin in the treatment of advanced non-small-cell lung cancer: a Southwest Oncology Group trial. J Clin Oncol. 2001;19:3210–18. doi: 10.1200/JCO.2001.19.13.3210. [DOI] [PubMed] [Google Scholar]
- 31.Fossella F, Pereira JR, von Pawel J, et al. Randomized, multinational phase iii study of docetaxel plus platinum combinations versus vinorelbine plus cisplatin for advanced non-small-cell lung cancer: the tax 326 study group. J Clin Oncol. 2003;21:3016–22. doi: 10.1200/JCO.2003.12.046. [DOI] [PubMed] [Google Scholar]
- 32.Hotta K, Matsuo K, Ueoka H, Kiura K, Tabata M, Tanimoto M. Meta-analysis of randomized clinical trials comparing cisplatin to carboplatin in patients with advanced non-small-cell lung cancer. J Clin Oncol. 2004;22:3852–9. doi: 10.1200/JCO.2004.02.109. [DOI] [PubMed] [Google Scholar]
- 33.Ardizzoni A, Boni L, Tiseo M, et al. on behalf of cisca (Cisplatin versus Carboplatin) Meta-analysis Group Cisplatinversus carboplatin-based chemotherapy in first-line treatment of advanced non-small-cell lung cancer: an individual patient data meta-analysis. J Natl Cancer Inst. 2007;99:847–57. doi: 10.1093/jnci/djk196. [DOI] [PubMed] [Google Scholar]
- 34.Pujol JL, Barlesi F, Daurès JP. Should chemotherapy combinations for advanced non-small cell lung cancer be platinumbased? A meta-analysis of phase iii randomized trials. Lung Cancer. 2006;51:335–45. doi: 10.1016/j.lungcan.2005.11.001. [DOI] [PubMed] [Google Scholar]
- 35.D’Addario G, Pintilie M, Leighl NB, Feld R, Cerny T, Shepherd FA. Platinum-based versus non-platinum-based chemotherapy in advanced non-small-cell lung cancer: a meta-analysis of the published literature. J Clin Oncol. 2005;23:2926–36. doi: 10.1200/JCO.2005.03.045. [DOI] [PubMed] [Google Scholar]
- 36.Rekhtman N, Paik PK, Arcila ME, et al. Clarifying the spectrum of driver oncogene mutations in biomarker-verified squamous carcinoma of lung: lack of EGFR/KRAS and presence of PIK3CA/AKT1 mutations. Clin Cancer Res. 2012;18:1167–76. doi: 10.1158/1078-0432.CCR-11-2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shaw AT, Yeap BY, Mino–Kenudson M, et al. Clinical features and outcome of patients with non-small cell lung cancer who harbor EML4–ALK. J Clin Oncol. 2009;27:4247–53. doi: 10.1200/JCO.2009.22.6993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Leighl NB, Paz–Ares L, Douillard JY, et al. Randomized phase iii study of matrix metalloproteinase inhibitor BMS-275291 in combination with paclitaxel and carboplatin in advanced non-small-cell lung cancer: National Cancer Institute of Canada-Clinical Trials Group Study br.18. J Clin Oncol. 2005;23:2831–9. doi: 10.1200/JCO.2005.04.044. [DOI] [PubMed] [Google Scholar]
- 39.Bennouna J, Senellart H, Hiret S, Vaissiere N, Douillard JY. Impact of histology on survival of resected non-small cell lung cancer (nsclc) receiving adjuvant chemotherapy: subgroup analysis of the adjuvant vinorelbine (nvb) cisplatin (cddp) versus observation in the anita trial. Lung Cancer. 2011;74:30–4. doi: 10.1016/j.lungcan.2011.02.004. [DOI] [PubMed] [Google Scholar]
- 40.Johnson DH, Fehrenbacher L, Novotny WF, et al. Randomized phase ii trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol. 2004;22:2184–91. doi: 10.1200/JCO.2004.11.022. [DOI] [PubMed] [Google Scholar]
- 41.Sandler A, Yi J, Dahlberg S, et al. Treatment outcomes by tumor histology in Eastern Cooperative Group Study E4599 of bevacizumab with paclitaxel/carboplatin for advanced non-small cell lung cancer. J Thorac Oncol. 2010;5:1416–23. doi: 10.1097/JTO.0b013e3181da36f4. [DOI] [PubMed] [Google Scholar]
- 42.Reck M, von Pawel J, Zatloukal P, et al. Phase iii trial of cisplatin plus gemcitabine with either placebo or bevacizumab as first-line therapy for nonsquamous non-small-cell lung cancer: avail. J Clin Oncol. 2009;27:1227–34. doi: 10.1200/JCO.2007.14.5466. [DOI] [PubMed] [Google Scholar]
- 43.Pirker R, Pereira JR, Szczesna A, et al. Cetuximab plus chemotherapy in patients with advanced non-small-cell lung cancer (flex): an open-label randomised phase iii trial. Lancet. 2009;373:1525–31. doi: 10.1016/S0140-6736(09)60569-9. [DOI] [PubMed] [Google Scholar]
- 44.Lynch TJ, Patel T, Dreisbach L, et al. Cetuximab and first-line taxane/carboplatin chemotherapy in advanced non-small-cell lung cancer: results of the randomized multicenter phase iii trial BMS099. J Clin Oncol. 2010;28:911–17. doi: 10.1200/JCO.2009.21.9618. [DOI] [PubMed] [Google Scholar]
- 45.Soon YY, Stockler MR, Askie LM, Boyer MJ. Duration of chemotherapy for advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized trials. J Clin Oncol. 2009;27:3277–83. doi: 10.1200/JCO.2008.19.4522. [DOI] [PubMed] [Google Scholar]
- 46.Ciuleanu T, Brodowicz T, Zielinski C, et al. Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: a randomised, double-blind, phase 3 study. Lancet. 2009;374:1432–40. doi: 10.1016/S0140-6736(09)61497-5. [DOI] [PubMed] [Google Scholar]
- 47.Cappuzzo F, Ciuleanu T, Stelmakh L, et al. on behalf of saturn investigators Erlotinib as maintenance treatment in advanced non-small-cell lung cancer: a multicentre, randomized, placebo-controlled phase 3 study. Lancet Oncol. 2010;11:521–9. doi: 10.1016/S1470-2045(10)70112-1. [DOI] [PubMed] [Google Scholar]
- 48.Dancey J, Shepherd FA, Gralla RJ, Kim YS. Quality of life assessment of second-line docetaxel versus best supportive care in patients with non-small-cell lung cancer previously treated with platinum-based chemotherapy: results of a prospective, randomized phase iii trial. Lung Cancer. 2004;43:183–94. doi: 10.1016/j.lungcan.2003.09.001. [DOI] [PubMed] [Google Scholar]
- 49.Leighl NB, Shepherd FA, Kwong R, Burkes RL, Feld R, Goodwin PJ. Economic analysis of the tax 317 trial: docetaxel versus best supportive care as second-line therapy of advanced non-small-cell lung cancer. J Clin Oncol. 2002;20:1344–52. doi: 10.1200/JCO.20.5.1344. [DOI] [PubMed] [Google Scholar]
- 50.Ramlau R, Gervais R, Krzakowski M, et al. Phase iii study comparing oral topotecan to intravenous docetaxel in patients with pretreated advanced non-small-cell lung cancer. J Clin Oncol. 2006;24:2800–7. doi: 10.1200/JCO.2005.03.6491. [DOI] [PubMed] [Google Scholar]
- 51.Krzakowski M, Ramlau R, Jassem J, et al. Phase iii trial comparing vinflunine with docetaxel in second-line advanced non-small-cell lung cancer previously treated with platinum-containing chemotherapy. J Clin Oncol. 2010;28:2167–73. doi: 10.1200/JCO.2009.23.4146. [DOI] [PubMed] [Google Scholar]
- 52.Kim ES, Hirsh V, Mok T, et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (interest): a randomized phase iii trial. Lancet. 2008;372:1809–18. doi: 10.1016/S0140-6736(08)61758-4. [DOI] [PubMed] [Google Scholar]
- 53.Bezjak A, Tu D, Seymour L, et al. Symptom improvement in lung cancer patients treated with erlotinib: quality of life analysis of the National Cancer Institute of Canada Clinical Trials Group Study br.21. J Clin Oncol. 2006;24:3831–7. doi: 10.1200/JCO.2006.05.8073. [DOI] [PubMed] [Google Scholar]
- 54.Bradbury PA, Tu D, Seymour L, et al. on behalf of the ncic Clinical Trials Group Working Group on Economic Analysis Economic analysis: randomized placebo-controlled clinical trial of erlotinib in advanced non-small cell lung cancer. J Natl Cancer Inst. 2010;102:298–306. doi: 10.1093/jnci/djp518. [DOI] [PubMed] [Google Scholar]
- 55.Temel JS, Greer JA, Muzikansky A, et al. Early palliative care for patients with metastatic non-small-cell lung cancer. N Engl J Med. 2010;363:733–42. doi: 10.1056/NEJMoa1000678. [DOI] [PubMed] [Google Scholar]
- 56.Bergethon K, Shaw AT, Ou SH, et al. ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol. 2012;30:863–70. doi: 10.1200/JCO.2011.35.6345. [DOI] [PMC free article] [PubMed] [Google Scholar]