Abstract
Burkitt’s lymphoma (BL), driven by translocation and over-expression of the c-MYC gene, is an aggressive, highly proliferative lymphoma and novel therapeutic strategies are required to overcome drug resistance following conventional treatments. The importance of the pro-survival BCL-2 family member BCL-XL in BL cell survival suggests that antagonistic BH3-mimetic compounds may have therapeutic potential. Here we show that treatment of BL cell lines with ABT-737 induces caspase-3/7 activation and apoptosis with varying potency. Using selective inhibitors, we identify phosphoinositide-3-kinase (PI3K) as a pro-proliferative/survival pathway in BL cells and investigate the potential of combined pharmacological inhibition of both the BCL-2 family and PI3K signaling pathway. PI3K/AKT inhibition and ABT-737 treatment induced synergistic caspase activation, augmented BL cell apoptosis and rendered chemo-resistant cells sensitive. Targeting mTORC1/2 with PP242 was also effective, either as a monotherapy or, more generally, in combination with ABT-737. The combined use of a dual specificity PI3K/mTOR inhibitor (PI 103) with ABT-737 proved highly efficacious. PI 103 treatment of BL cells was associated with an increase in BIM/MCL-1 expression ratios and loss of c-MYC expression. Furthermore, blocking c-MYC function using the inhibitor 10058-F4 also induced apoptosis synergistically with ABT-737, suggesting that maintenance of expression of BCL-2 family members and/or c-MYC by the PI3K/AKT/mTOR pathway could contribute to BL cell survival and resistance to ABT-737. The combined use of BH3-mimetics and selective mTORC1/2 inhibitors may therefore be a useful novel therapeutic approach for the treatment of B-cell malignancy, including chemo-resistant lymphomas.
Keywords: PI3K, ABT-737, Burkitt’s Lymphoma, apoptosis, mTOR
Introduction
Burkitt’s lymphoma is an aggressive malignancy derived from germinal centre (GC) B-cells. Conventional intensive chemotherapeutic regimes are effective in eliminating disease in the majority of cases (1) however, novel treatment strategies are still required to treat patients who relapse with chemo-resistant tumours. BLs are characterised by chromosomal translocation events which deregulate the c-MYC proto-oncogene (2) resulting in rapid proliferation of the malignant cells. While BL cells proliferate rapidly, they are also sensitive to apoptotic stimuli and still maintain many of the pro-apoptotic and pro-survival signaling pathways that normally regulate their cell of origin. For example, BL cells are acutely sensitive to TGF-β-induced intrinsic apoptosis (3) which occurs via transcriptional control of members of the BCL-2 family of apoptosis regulators (4). BCL-2 family members are frequently deregulated in malignancies of GC origin (reviewed in (5)) and can be considered as valid targets for therapeutic intervention in B-cell lymphomas.
The pro-survival members of the BCL-2 family include BCL-2 itself, BCL-XL, BCL-w, MCL-1 and BOO. These proteins are regulated by other BH3-only family members that share only one region of homology with BCL-2 (the BH3-domain). These pro-apoptotic proteins (BIK, BIM, PUMA, NOXA, BAD, HRK, BID and BMF) function as naturally occurring inhibitors of the pro-survival family members. Several BCL-2 antagonists, designed to mimic the BH3-domain of the BH3-only proteins (reviewed in (6)), have been developed as potential therapeutics and have undergone testing against B-cell malignancies, either as monotherapies, or in combination with other drugs (7-12). BCL-2-antagonists have shown activity in mouse models of Myc-driven lymphomas (12-14) which suggests that BH3-mimetics may be useful in treating human disease.
However, it is currently unknown if such strategies would be effective against BL. The main targets of the BH3-mimetic ABT-737 (15) are BCL-2, BCL-XL and potentially BCL-w (13, 14). Since BL are derived from GC cells, they lack BCL-2 expression (a characteristic feature of centroblasts and centrocytes (16, 17)) but, given the important role of BCL-XL in BL cell survival (4), we hypothesised that ABT-737 might induce apoptosis of BL cells despite the lack of BCL-2 expression.
ABT-737 is the most selective of several putative BCL-2 inhibitors in that it induces BAX and BAK dependent cell death entirely through activation of the mitochondrial apoptosis pathway. The selectivity of ABT-737 for BCL-2 and BCL-XL ensures that it is least effective in cells expressing high MCL-1 levels, which are not as dependent on BCL-2 or BCL-XL for survival. In such cases, the efficacy of ABT-737 can often be enhanced by co-administration of therapeutic agents that “neutralise MCL-1 function” (13) through, for example, the induction of BIM and/or NOXA (18-20).
In this study we assess the efficacy of ABT-737 as a single agent, and identify potential therapeutic targets for synergistic activity with BH3-mimetics in BL cell lines. Cells in the GC are subject to intense selection criteria mediated by signal transduction pathways, some of which impact on BCL-2 family protein expression (reviewed in (5)). If flux through such pathways is maintained in BL cells (as in other B-cell malignancies (21)) combined use of BH3-mimetics with inhibitors of pro-proliferation and/or pro-survival signaling pathways may enhance treatment efficacy or selectivity.
Here we show that ABT-737 induces apoptosis as a single agent, and that phosphoinositide-3-kinase/AKT/mTOR (PI3K/AKT/mTOR) is required for BL cell proliferation and/or survival. Inhibition of PI3K augments ABT-737-induced apoptosis. Furthermore, we found that dual PI3K/mTOR inhibitors or active site inhibitors of mTOR (asTORi) are effective either as single agents, or synergise with ABT-737 to overcome resistance to ABT-737. These findings suggest that the combined use of BH3-mimetics with mTORC1/2 inhibitors could be a novel and effective therapeutic approach for the management of BL.
Materials and Methods
Cell Lines and reagents
Human BL cell lines were a kind gift of Chris Gregory or were obtained from the Institute of Cancer Research cell bank (Ramos). L3055 cell lines over-expressing BCL-XL or expressing BCL-2 (L3055-BCL-XL and L3055-BCL2) were derived by Chris Gregory and have been described previously (22). All lines were EBV negative and mycoplasma negative and were used within 6 months from source or following karyotyping (23). BL cell lines were maintained in RPMI-1640 (Gibco-BRL) supplemented with 5% (v/v) heat-inactivated FCS, 2mM/ml glutamine and antibiotics and were cultured for no more than 25 passages. Cells were treated as required with the BH3-mimetic ABT-737 (Selleck Chemicals), the pan-caspase inhibitor zVAD-fmk (Bachem), the pan-PI3K inhibitor, LY-294002 (Calbiochem), the isozyme selective AKT1/AKT2 inhibitor (AKT inhibitor VIII) (Merck) the dual PI3K p110α/mTOR inhibitor, PI 103, (Tocris Bioscience), inhibitors of mTOR, Rapamycin and PP242 (Sigma), or the c-MYC inhibitor 10058-F4 (5-[(4-Ethylphenyl)methylene]-2-thioxo-4-thiazolidinone) (Sigma).
Immunoblotting and antibodies
Cell protein lysates were quantified and equal amounts analysed by SDS-PAGE. Antibodies used in western blotting were mouse monoclonals against PARP (BD Biosciences), c-MYC (9E10) (Santa Cruz), actin (Sigma) and S6 ribosomal protein (Cell Signaling Technology). Rabbit polyclonal antibodies were used to detect phospho-S6 ribosomal protein (Ser235/235), phospho-4E-BP1 (ser65), 4E-BP1, phospho-AKT (ser 473), pan AKT, BCL-2, BCL-XL, BCL-w, BID, PUMA (Cell Signaling Technology), MCL-1, BIK, BAK, BAX (Santa Cruz) and BIM (Chemicon). Secondary HRP-conjugated antibodies were obtained from Dako. Bound immunocomplexes were detected by enhanced chemiluminescence (ECL; Amersham).
Analysis of cell cycle and apoptosis
Cells were fixed, labelled with propidium iodide (PI) and analysed by flow cytometry (FACSCalibur, BD Biosciences) for DNA content. Histogram plots containing a minimum of 10000 events were analysed by CellQuest Pro software (BD Pharmingen) using linear gates to determine the percentage of apoptotic cells with sub-G1 DNA content (M1) or cells in G1 (M2), S-phase (M3) or G2/M phases (M4) of the cell cycle. Intracellular caspase-3 activity was assayed by flow cytometry using PhiPhiLux G1D2, (Calbiochem) and PI as recommended by the manufacturer. Caspase 3/7 activity was also analysed using Caspase-Glo (Caspase3/7) assay reagents (Promega). Cell suspensions (50μl) treated for 24 hours were incubated for 1 hour at room temperature with an equal volume of caspase-Glo substrate. Fluorescence was then analysed using a Veritas microplate luminometer (Turner Biosystems).
Densitometry and Statistical analysis
Western blots were analysed within the linear range of enhanced chemiluminescence and the resultant films scanned and analysed using ImageJ software. Statistical analysis was performed by paired student TTEST. Significant p values of <0.05, <0.01 and <0.005 are indicated by one, two or three stars respectively. Potential synergistic effects of drug combinations were determined using CalcuSyn dose effect analysis software (Biosoft) based on the Combination Index equation and plot of Chou-Talalay (24), which analyses the doses of drug A or drug B required to give an effect (Fraction affected-Fa) in comparison with the doses of drug A and drug B used in combination to give the same effect. In our experiments the ED50, ED75 and ED90 values are compared (ie the doses of drug at which 50%, 75% and 95% maximum caspase activation is achieved). Combination index (CI) values at these points of >1 represents antagonism, =1 represents an additive effect and <1 indicates synergy.
Results
ABT-737 induces apoptosis in BL cells
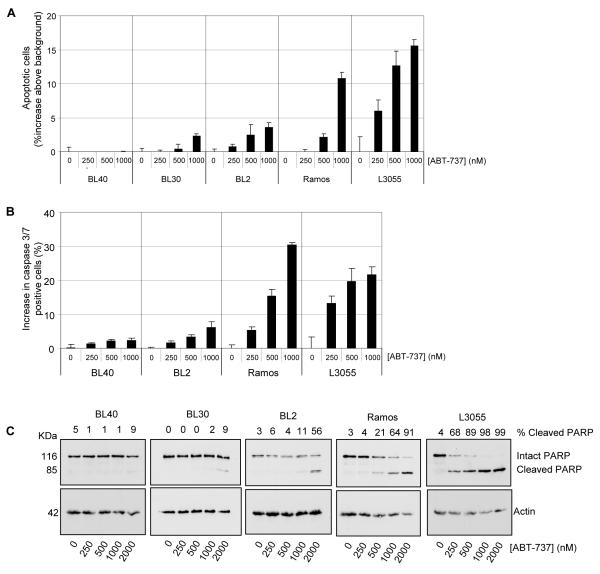
Given the critical role for BCL-XL in BL cell survival we predicted that ABT-737 may induce apoptosis in human BL cells. We assessed its’ effect on EBV-negative BL cell lines using three different assays (Figure 1). PI staining of BL cells lines treated for 48 hours demonstrated a dose-dependent increase in the percentage of cells with sub-G1 DNA content in several of the lines (Figure 1A). Induction of apoptosis by ABT-737 was confirmed by increased caspase 3/7 activity (Figure 1B and S1A), and also by cleavage of PARP (Figure 1C). ABT-737-induced apoptosis was completely ablated by pre-treatment of cells with the pan-caspase inhibitor zVAD-fmk (Figure S1B). We observed clear differences in the sensitivity of individual cell lines to ABT-737, with BL40, BL30 and BL2 being relatively resistant. Analysis of the percent of cleaved PARP (shown above each lane in Figure 1C) confirmed that BL30 and BL40 lines were largely refractory to ABT-737 treatment.
Figure 1. Sensitivity of human BL cells to apoptosis induced by ABT-737.
Cells were either left untreated or treated with a two-fold dilution series of ABT-737. (A) After 48 hours, the mean (± s.d, n=3) increase in the percentage of cells with sub-G1 DNA content above background was determined by PI staining. (B) The (mean ± s.d, n=3) percent increase in intracellular caspase 3/7 positive cells induced following 24 hours ABT-737 treatment was determined flow cytometry using PhiPhiLux. (C) Cells treated for 24 hours were lysed and analysed by western blotting for cleavage of the caspase substrate PARP. The proportion of cleaved PARP is indicated above each lane as a percentage of the total amount of PARP determined by densitometry and ImageJ software. A western blot for actin is shown as a loading control.
BCL-2 family members and sensitivity to ABT-737
Having identified differential sensitivities of cell lines to ABT-737, we attempted to correlate their responsiveness with the relative expression levels of BCL-2 family members in each line (Figure S2A). BL cell lines did not express detectable levels of two potential ABT-737 targets, BCL-2 or BCL-w and expression levels of the only other protein targeted by ABT-737, BCL-XL, did not correlate with sensitivity. In B-CLL, reduction of BIM expression renders these cells less sensitive to ABT-737, suggesting that in some lymphomas BIM could be a determinant of sensitivity (25). However, in our cell line panel, there was no correlation between sensitivity and BIM expression. Likewise we observed no correlation between the levels of MCL-1, PUMA and BID and the apoptotic response to ABT-737 (Figure S2A). Levels of both BAX and BAK were equivalent in all lines (Figure S2B), therefore the relative resistance of BL30 and BL40 cell lines to ABT-737 does not appear to be due to a “Class B” block (26) in apoptosis caused by loss of BAX or BAK (Figure S2B). CA46 BL cells which are genetically deficient in BAX and BAK expression (27) (Figure S2B), were unaffected by ABT-737 treatment as assessed by cell counting assays (Figure S2C) and are the only cell line which exhibit a complete lack of caspase activation in response to ABT-737 (Figure S2D). Cells with a Class B block in apoptosis are therefore ABT-737 resistant, but BX/BAK loss does not account for the lack of response in other lines which remain competent for intrinsic apoptosis. The analysis of individual BCL-2 family members in these cell lines therefore cannot predict the outcome of ABT-737 treatment.
In diffuse large B-cell lymphomas (26), and by in vitro BH3-profiling (25), the best predictors of sensitivity to ABT-737 are high levels of BCL-2 which is available to complex with BIM. However, all the BL cell lines, consistent with their cell of origin (16, 17), lack BCL-2 expression (Figure S2A) but are nevertheless differentially sensitive to ABT-737. Since BIM can also be sequestered by MCL-1 and BCL-XL, we assessed the ratios of BIM to MCL-1 and BCL-XL in comparison with those of PUMA and BIK. Sensitivity to ABT-737 correlated best with BIM:MCL-1 expression ratios with higher ratios evident in sensitive cells. Ratios of PUMA or BIK to MCL-1 or BCL-XL bore no correlation with sensitivity to ABT-737 (Figure S2E). Since Burkitt’s Lymphoma cell lines are refractory to transfection, we were not able to identify unequivocally which BCL-2 family members conferred sensitivity to ABT-737 using siRNA knockdown approaches however, when we compared sensitivities of the parental human cell line L3055 with BCL-XL over-expressing or BCL-2 expressing derivatives, we found that changes in pro-survival factor expression significantly altered the response of cells to ABT-737 (Figures S3A and S3B). Following treatment with ABT-737 (250nM), BCL-2 expressing cells were highly sensitised to its’ effects which concurs with studies described using Eμ-Myc/bcl-2 lymphomas (12-14). BCL-XL-over-expressing L3055-cells, however, were more resistant to ABT-737. Taken together these data suggest that the relative expression levels of pro-survival to pro-apoptosis BCL2 family members may determine the outcome of ABT-737 monotherapy.
PI3-Kinase signaling contributes to BL cell proliferation and survival
ABT-737 is often more efficacious when used in conjunction with other agents that impinge on the function of the BCL-2 family. To select potential targets for combination therapy we considered signaling pathways that might be pro-proliferative and/or pro-survival. In human B-cells, signaling through the PI3K/AKT pathway (reviewed in (5)) can promote survival. Constitutive PI3K activity is also reported to be essential for the proliferation of one of the BL cell lines used in this study (28), We therefore first tested whether constitutive PI3K signaling is operative in our BL cell line panel.
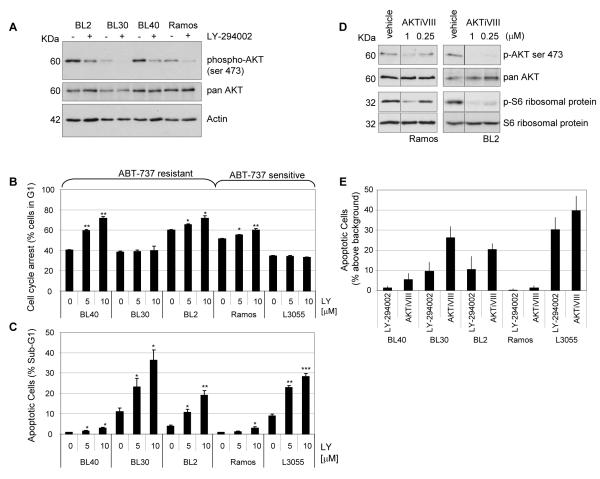
Basal PI3K signaling measured by serine 473 phosphorylation of AKT/PKB was detectable in all BL lines and could be substantially reduced by treatment with the pan PI3K inhibitor LY-294002 (Figure 2A). The effect of blocking PI3K signaling on cell proliferation and apoptosis was assessed by flow cytometry (Figures 2B and 2C respectively). PI3K inhibition had significant effects on lymphoma cell out-growth, inducing a G1 cell cycle arrest in BL40, BL2 and Ramos (Figure 2B) and apoptosis in BL30, BL2 and L3055 (Figure 2C). To determine whether the effects were mediated through inhibition of AKT, we used a selective inhibitor of AKT1/2, AKT inhibitor VIII (AKTiVIII, 1μM) which decreased AKT activation, (measured by reduced phosphorylation (Figure 2D)). Like LY-294002, AKTiVIII induced BL cell apoptosis in BL30 and BL2 and L3055 cells (Figure 2E). PI3K signaling through AKT is therefore critical for maximal BL proliferation and/or survival.
Figure 2. PI3K signaling contributes to BL cell proliferation and/or survival.
(A) BL cells were treated for 2 hours with the pan PI3K inhibitor LY-294002 (10μM), lysed and analysed by western blotting for phosphorylation of the PI3K substrate AKT/PKB (ser473). (B, C) BL cells treated for 48 hours with LY-294002 (0, 5 and 10μM) were fixed, stained with PI and analysed by flow cytometry and CellQuest Pro for G1 arrest (B), and for cell death (C). (D) Ramos and BL2 cells were treated for 2 hours with DMSO (vehicle) or with 1 or 0.25μM of the AKT1/2 inhibitor (AKTiVIII). Cell lysates were analysed by western blot using phospho-specific and total AKT and S6 ribosomal protein antibodies as indicated. (E) BL cells were treated for 48 hours with LY-294002 (5μM) or AKTiVIII (1μM) and the amount of apoptosis above background determined by PI. Shown is the mean apoptosis induction above background from at least two experiments each performed in triplicate.
mTOR inhibitors regulate BL cell proliferation and/or survival as single agents
AKT has numerous substrates implicated in cell survival pathways, including the mammalian target of rapamycin (mTOR). We investigated further whether mTOR might contribute to BL cell survival using selective inhibitors and dual PI3K/mTOR inhibitors. mTOR exists as two complexes mTORC1 and mTORC2, mTORC2 being upstream of AKT and mTORC1 regulating the activity of its’ downstream effectors p70 S6-kinase/S6 ribosomal protein and 4E-BP1/eIF4E.
PP242 is an active site, ATP competitive inhibitor of both mTORC1 and mTORC2, and rapamycin is a less effective inhibitor of mTORC1 (29). In agreement with previous studies (30-34), we were able to distinguish rapamycin sensitive and rapamycin insensitive effects of mTORC1 signaling. Phosphorylation of both S6 ribosomal protein and 4E-BP1 was inhibited using the active site inhibitor PP242, whereas rapamycin treatment efficiently inhibited phosphorylation of S6 ribosomal protein but had little effect on phosphorylation of 4E-BP1 (Figure 3A). PI 103 is a dual PI3K p110α/mTOR inhibitor and also efficiently inhibited phosphorylation of both S6 ribosomal protein and 4E-BP1. When we assessed the effect of rapamycin and PP242 in comparison with LY-294002 and PI 103 on BL cell survival, we observed that PP242 and PI 103 induced similar levels of apoptosis as single agents in BL30, BL2 and L3055 (Figure 3B). Dual inhibition of PI3K and mTOR using PI 103 was the most effective single agent therapy, inducing caspase-dependent (zVAD-fmk sensitive) cleavage of PARP (Figure 3C). Rapamycin (2nM) and LY-294002 (5μM), used at concentrations that only effectively inhibit S6 ribosomal protein phosphorylation (not phosphorylation of 4E-BP1) generally induced less apoptosis than either PP242 or PI 103, suggesting that the enhanced pro-apoptotic effects of PP242 and PI 103 may correlate with inhibition of 4E-BP1/eIF4E function. Treatments which had minimal effects on BL40 cell survival instead induced a G1 cell cycle arrest (Figure S4).
Figure 3. mTOR inhibitors as monotherapies for Burkitt’s Lymphoma.
(A) Ramos cells were treated for 2 hours with LY-294002 (LY), the dual PI3K/mTOR inhibitor PI 103 or selective inhibitors of mTOR, Rapamycin (Rap) and the active site inhibitor PP242. Equal amounts of cell lysate were analysed by western blot. (B) BL cell lines were treated for 48 hours with LY-294002 (5μM), PI 103 (1μM), Rapamycin (2nM) or PP242 (1.25μM). Cells were analysed by PI staining for percent (mean ± s.d., n=3) cell death above background. (C) BL2 cells were pretreated with the pan-caspase inhibitor ZVAD-fmk (25μM) for 1 hour prior to PI 103 (1μM) addition for the indicated time periods. Cell lysates were analysed by western blot.
PI3K inhibition augments ABT-737-induced apoptosis of BL cells
Given our observations that BL cell lines have differential sensitivities to inhibitors of either PI3K signaling and/or BCL-XL as single agents, we investigated the consequence of combined inhibition of these pathways. When used in conjunction with ABT-737, LY-294002 augmented killing of Ramos cells. LY-294002 treatment alone induced G1 arrest, but increased the amount of cell death in combination with ABT-737 (Figure S5A). Subsequent ABT-737 titrations, analysed by PARP cleavage, revealed that LY-294002 potently sensitised Ramos cells to ABT-737-induced death (Figure S5C). Importantly, combination treatment enhanced apoptosis in the relatively chemo-resistant cell line BL40 (Figures S5B and S5C). Similar results were obtained in BL2 cells (data not shown), and were confirmed by PI staining in Ramos, BL40 and L3055 (Figure S5B and data not shown). LY-294002 was also able to render the resistant BCL-XL over-expressing cell line L3055-BCL-XL sensitive to ABT-737 treatment (Figure S5D).
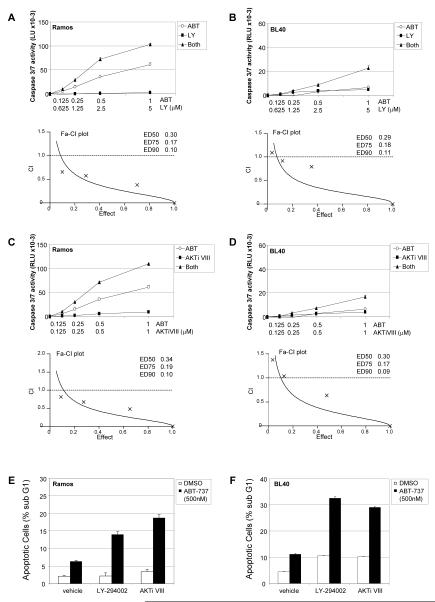
To establish whether ABT-737 and LY-294002 act synergistically, we performed dose effect analysis measuring caspase 3/7 activation induced by drug combinations. The data were analysed by the Chou-Talalay mathematical equation (24) to determine combination indices, where values of >1, =1 and <1 indicate antagonism, additive effects and synergism respectively. ABT-737 and LY-294002 acted synergistically to activate caspase 3/7 in both Ramos (Figure 4A) and BL40 cells (Figure 4B). Importantly, synergistic activity was also detected using the combination of ABT-737 and the more selective AKT inhibitor, AKTiVIII (Figures 4C and 4D). These data indicate that combining ABT-737 with inhibition of PI3K signaling via AKT has the potential to sensitise ABT-737 resistant BL cells to apoptosis, and augment apoptosis in cells already responsive to ABT-737. Our observations were confirmed by propidium iodide staining (Figure 4E and 4F), and in BL30 and BL2 cells which show apoptotic responses to AKT inhibition alone (Figures S6A, S6B, S6C and S6D).
Figure 4. Synergistic effects of PI3K/AKT inhibitors and ABT-737.
The combined effect of PI3K/AKT inhibitors and ABT-737 on apoptosis in BL cells was assessed by caspase 3/7 assay (A-D), and by PI staining (E, F). Ramos cells (A, C) or BL40 cells (B, D) were left untreated, or treated with concentrations of ABT-737 (ABT) the presence or absence of LY-294002 (LY) or AKTiVIII in equimolar ratios as indicated. Caspase activity was analysed by caspase 3/7-glo assay after 24 hours and potential synergism determined by the Chou-Talalay equation for dose effect analysis of drug combinations. Combination Indices (CI) with respect to the fraction affected (Fa-CI) plots and the values determined at ED50, ED75 and ED90 are shown directly below the relevant line graph. CI values of >1, =1 and <1 represent antagonism, additive effect and synergism respectively. (E, F) Ramos (E) and BL40 cells (F) were treated with solvent or ABT-737 (500nM) in the presence or absence of LY-294002 (5μM) or AKT inhibitor VIII (1μM). Cells were harvested after 48 hours, and the percent apoptosis (mean ± s.d., n=3) determined by PI staining.
mTORC1/2 inhibitors synergise with ABT-737 to induce BL cell apoptosis
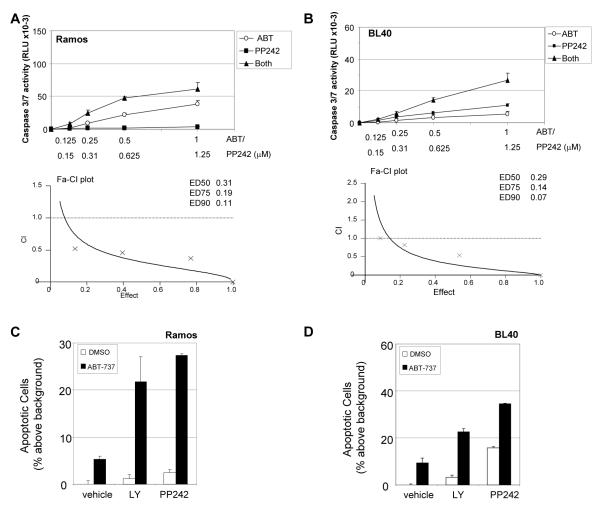
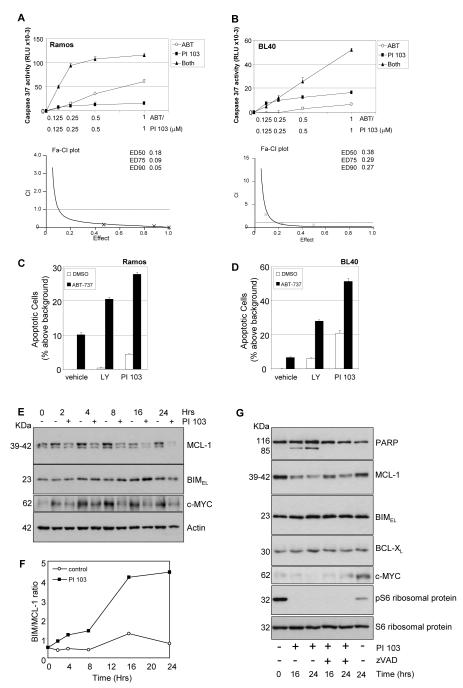
To determine if the synergy between ABT-737 and PI3K/AKT inhibition could be recapitulated when selectively targeting downstream mTORC1 and mTORC2 complexes, PP242 was used in combination with ABT-737 which resulted in the synergistic induction of caspase3/7 activity (Figures 5A and 5B), and increased apoptosis (Figures 5C and 5D). In BL30 cells that were killed by PP242 alone, the drug combination of PP242 with ABT-737 had no additional apoptotic effect (data not shown). ABT-737 in combination with PP242 was much more effective than when used in combination with doses of Rapamycin which affected only phosphorylation of S6 ribosomal proteins (data not shown). Taken together our data implicates PI3K/AKT/mTORC1/2 signaling as both a survival factor and mediator of resistance to ABT-737 in BL. This conclusion was supported by our observation of a highly synergistic apoptotic effect when combining ABT-737 with the dual PI3K p110α/mTOR inhibitor, PI 103 (Figures 6A-6D) suggesting that concomitant targeting of PI3K/mTORC1/2 along with BCL-XL is optimal for induction of BL cell death.
Figure 5. Synergistic effects of the mTOR inhibitor PP242 and ABT-737.
The combined effect of the active site mTOR inhibitor PP242 and ABT-737 on apoptosis in BL cells was assessed by caspase 3/7 activation assay (A, B), and by PI staining (C, D). Ramos (A) and BL40 cells (B) were left untreated or treated with concentrations of ABT-737 (ABT) in the presence or absence of PP242 in equimolar ratios. Increases in caspase activity were analysed by caspase 3/7-glo assay after 24 hours. Potential synergism was determined as described in Figure 4. (C) Ramos and (D) BL40 cells were pre-treated for 15 minutes with vehicle, PP242 (1.25μM) or LY-294002 (5μM), followed by addition of ABT-737 (500nM) or vehicle control. Cells were analysed by PI staining after 48 hours and the percent apoptotic cells above background (mean ± s.d., n=3) determined.
Figure 6. The dual PI3K/mTOR inhibitor PI 103 increases BIM:MCL-1 ratios and enhances ABT-737 sensitivity.
The combined effect of PI 103 and ABT-737 on apoptosis in BL cells was assessed by caspase 3/7 activation assay (A, B), and by PI staining (C, D). Ramos (A) and BL40 cells (B) were left untreated or treated with concentrations of ABT-737 in the presence or absence of PI 103 in equimolar ratios. The potential synergistic induction of caspase activity was determined as described in Figure 4. (C) Ramos and (D) BL40 cells were pre-treated for 15 minutes with vehicle, PI 103 (1μM) or LY-294002 (5μM), followed by addition of ABT-737 (500nM) or vehicle control. The percent apoptotic cells above background (mean ± s.d., n=3) was determined by PI staining after 48 hours. (E) Ramos cells were treated with PI 103 (1μM) and cell lysates analysed by western blot. (F) Western blots (representative blots shown in (E)) were analysed by densitometry and ImageJ software and the ratio of BIM to MCL-1 protein expression determined over the time course of PI 103 treatment. BIM:MCL-1 ratios determined after 24 hours for untreated versus PI 103 treated cells were 0.78±0.03 and 3.2± 1.05 respectively (n=3). (G) Western blot analysis of the indicated proteins in Ramos cells treated with PI 103 (1μM) in the presence or absence of zVAD-fmk (25μM).
Since we found that the ratio of basal levels of BIM:MCL-1 correlated best with sensitivity of BL cells to ABT-737 as a single agent (Figure S2A), we examined MCL-1 expression during a time course of PI 103 treatment of Ramos cells. PI 103 treatment caused a decrease in the expression level of MCL-1 (Figure 6E) resulting in a time-dependent increase in the ratio of BIM/MCL-1 (Figure 6F) as determined by densitometry analysis of western blots shown in Figure 6E. Similar results were observed in three independent experiments and following treatment of BL40 cells (data not shown). In addition to an effect on MCL-1 expression, we also observed an effect of PI 103 treatment on the levels of c-MYC. The loss of MCL-1 and c-MYC expression was not due to the generalised loss of proteins caused by cell death, as similar effects were observed in PI 103 treated cells in which apoptosis was blocked with zVAD (Figure 6G). PI 103 treatment did not significantly affect the level of BCL-XL, also one of the subset of proteins (like MCL-1 and c-MYC) produced through cap-dependent translation of its mRNA.
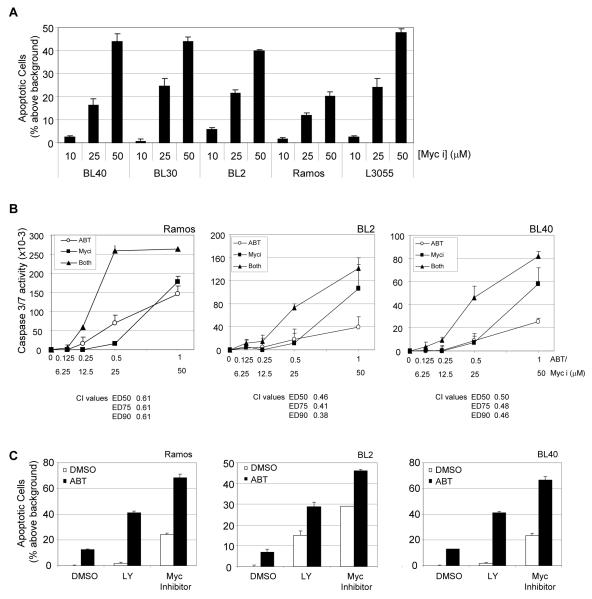
To further investigate whether the loss of c-MYC expression could account for apoptosis induction and synergistic effects of PI 103 with ABT-737 we analysed the effect of inhibition of c-MYC function on BL cells using a small molecule, selective inhibitor of MYC/MAX dimerisation and DNA binding activity, 10084-F3 (35, 36). Inhibition of c-MYC activity induced a dose dependent increase in apoptosis of all lines tested (Figure 7A) and, similar to inhibition of the PI3K/AKT/mTOR pathway, synergised with ABT-737 to increase caspase activation (Figure 7B) and enhance cell death (Figure 7C).
Figure 7.
Inhibition of c-MYC synergises with ABT-737 to induce BL cell apoptosis. (A) Cells were treated for 48 hours with a two-fold dilution series of the Myc inhibitor 10058-F4 (Myc i), fixed, stained with propidium iodide and analysed by flow cytometry. The mean (± s.d, n=3) increase in the percentage of cells with sub-G1 DNA content above background is shown. (B) The combined effect of the MYC inhibitor and ABT-737 on apoptosis in BL cells was assessed by caspase 3/7 assay. BL cells were treated with solvent as a control, or treated with concentrations of ABT-737 (ABT) the presence or absence of 10058-F4 in equimolar ratios as indicated. Caspase activity was analysed by caspase 3/7-glo assay after 24 hours and potential synergism determined by the Chou-Talalay equation for dose effect analysis of drug combinations. Combination Indices (CI) were determined at ED50, ED75 and ED90. CI values of <1 represent synergism. (C) The effect of combining ABT-737 (500nM) with the MYC inhibitor (25μM) on apoptosis of BL cells was assessed by propidium iodide staining and flow cytometry after 48 hours treatment. The drug combination of ABT and LY-294002 was included as a positive control for augmented apoptosis.
Discussion
High-dose combinations of cytotoxic drugs can be highly effective in treating Burkitt’s Lymphoma but novel therapeutic strategies may help to tackle the ongoing problem of severe side-effects and relapse. In this study we analyse apoptotic responses of BL cell lines to potential chemotherapeutic agents targeting the BCL-2 family and multiple components of the PI3K/AKT/mTOR signaling cascade. We identify the PI3K/AKT/mTOR signaling as a pro-proliferative and/or pro-survival pathway in BL cells and report on the synergistic use of BH3-mimetics combined with different pharmacological inhibitors targeting several effectors within the PI3K/mTOR pathway.
We determined that cell death in EBV-negative BL cells induced by ABT-737 as a single agent, is caspase dependent and that, in general, higher doses of ABT-737 are required to induce apoptosis in BL compared with AML (37) or B-CLL (25). This may be due to the fact that like GC B-cells, BL cells lack BCL-2 expression. Several studies in murine Eμ-myc tumour models have indicated that the efficacy of ABT-737 depends on whether cells rely primarily on its specific targets for survival (i.e. Bcl-2 or Bcl-XL). Eμ-myc lines expressing Bcl-2 (12-14), for example, are highly sensitive to ABT-737. Studies in DLBCL also suggest that “primed” BCL-2 positive cell lines are the most sensitive to ABT-737 (26) while Del Gaizo Moore et al, using a technique termed ‘BH3-profiling’ in B-CLL samples, showed that sensitivity to ABT-737 may be determined by the amount of BCL-2 able to sequester the pro-apoptotic protein BIM (25). In this case, displacement of BCL-2 by ABT-737 releases BIM to activate BAX and BAK and induce apoptosis. The fact that BCL-2/BCL-w-negative BL cells are not reliant on two of the primary targets of ABT-737 for survival could render them inherently more resistant. This hypothesis is supported by our observation that BCL-2 over-expressing BL cells (L3055-BCL-2) were highly sensitised to ABT-737. Our data also suggests that ABT-737 could act as an effective single agent therapy in a stratified patient group with aggressive ‘double-hit’ tumours which carry concurrent BCL-2 and c-MYC rearrangements (38, 39).
The BL cells exhibited differential sensitivity to ABT-737. A lack of BAX and/or BAK (as in CA46) renders BL cells completely insensitive to ABT-737, however this “Class B” block in apoptosis did not account for resistance in the other relatively insensitive lines. In the absence of reliable, efficient siRNA knockdown procedures for BL cells, the identification of the determinants of this resistance in BL is problematic. However we have identified active PI3K/mTOR signaling as contributing to the resistant state, such that inhibition of the pathway at various levels increased the effectiveness of ABT-737 treatment at lower doses. PI3K is critical for mature B-cell survival (40) and elevated signaling is also associated with chemoresistance in diffuse large B-cell lymphoma (DLBCL) (41). Since many B-cell malignancies retain constitutive signaling through the BCR/Syk/PI3K/AKT pathway (including follicular lymphoma, DLBCL and B-CLL (reviewed in (42)) our finding that combination treatments utilising inhibitors of PI3K/mTOR augment apoptosis, may suggest effective therapies for other malignancies of B-cell origin. The EBV-positive status of some BL tumours may be a cofounding factor for the use of ABT-737, since viral genes have a number of effects on expression and function of BCL-2 family members (reviewed in (43)) but further studies will be required to address this issue.
Having established that inhibition of the PI3K/AKT pathway sensitises BL cells to ABT-737, we investigated the possible downstream effectors involved. There are numerous potential mechanisms of AKT-dependent apoptosis suppression (reviewed in (44)) however the use of PP242 (which is a selective active site mTOR inhibitor lacking activity against PI3K/AKT) establishes mTOR activity as a key component for BL survival and chemoresistance. In this study we used rapamycin at doses which only affected phosphorylation of S6 ribosomal protein without inhibiting phosphorylation of 4EBP-1. Both PP242 and PI 103 (which inhibit both S6 ribosomal protein and 4EBP-1 phosphorylation) were more effective in combination with ABT-737 than rapamycin. Blocking both rapamycin sensitive and insensitive functions of mTORC1 in this way indicated that inhibition of cap-dependent translation regulated by 4E-BP1/eIF4E may be necessary to overcome resistance to BH3-mimetics.
The hypothesis that 4E-BP1/eIF4E-dependent protein translation is important for maintaining cell survival and chemoresistance is supported by the observation that the expression levels of MCL-1 decrease following PI 103 treatment of BL cells. The effect on MCL-1 concurs with recent studies in DLBCL which demonstrated that phosphorylation of 4EBP-1 is essential for MCL-1 synthesis and that inhibition of mTORC1 activity can overcome MCL-1-mediated resistance to ABT-737 (45). We also found that resistance to ABT-737 correlated with the lowest ratio of BIM to MCL-1 expression. ABT-737 resistance determined by the amount of BIM sequestered by MCL-1 (which cannot be targeted by ABT-737) has been reported in both AML and murine lymphoma models (13, 14, 25). Treatment of BL cells with PI 103 increased the BIM/MCL-1 ratio, which could explain the increased sensitivity of PI 103 treated cells to ABT-737.
PI 103 treatment, however, also decreased expression of c-MYC. The loss of c-MYC protein as a result of mTOR inhibition has been reported previously (46). C-MYC is the oncogenic driver of BL and is bound as a MYC/MAX transcription factor complex to approximately 15% of gene promoters in BL cells (47). Both c-MYC and MCL-1 (48) are regulated at the level of protein stability since their half-life is short. The half-life for c-MYC, for example, is 18 minutes in Ramos cells (49). Recently proteomic approaches identified c-MYC as the key upstream regulator for the majority of proteins whose expression levels are affected by inhibition of cap-dependent translation (46). The similar synergistic apoptotic effects of either the MYC inhibitor 10058-F4 or active site mTOR inhibitors in combination with ABT-737 implies that continuous synthesis of c-MYC is required for BL survival and chemoresistance. It is possible that the c-MYC inhibitor may also have an indirect effect on the stability of pro-survival factors (MCL-1 or others) through inhibition of c-MYC-mediated regulation of genes involved in protein biosynthesis (47). Further studies are required to determine which c-MYC regulated genes are important in BL cell survival.
Although our data suggests that inhibitors of mTOR or c-MYC could work as effective single agents in BL, they were much more effective treatments when used in combination with ABT-737, across a broader range of cells and at lower doses. Combination therapy may therefore reduce any requirement for patient selection, and lower drug doses may be tolerated better.
Reports that PP242 is an effective treatment in combination with ABT-737, but simultaneously shows greater tolerability in models of leukemia (34, 50) are promising. A number of other mTOR inhibitors are currently in clinical trials therefore increasing the likelihood that suitable combination therapy strategies based on our observations could be tested clinically for BL.
Supplementary Material
Acknowledgments
Financial support: This study was supported by Cancer Research UK, Dundee University and the Association for International Cancer Research.
Footnotes
Disclosures of Conflicts of Interest: The authors declare no conflict of interest.
References
- 1.Bishop PC, Rao VK, Wilson WH. Burkitt’s lymphoma: molecular pathogenesis and treatment. Cancer Invest. 2000;18(6):574–83. doi: 10.3109/07357900009012197. [DOI] [PubMed] [Google Scholar]
- 2.Dalla-Favera R, Bregni M, Erikson J, Patterson D, Gallo RC, Croce CM. Human c-myc onc gene is located on the region of chromosome 8 that is translocated in Burkitt lymphoma cells. Proc Natl Acad Sci U S A. 1982 Dec;79(24):7824–7. doi: 10.1073/pnas.79.24.7824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Inman GJ, Allday MJ. Apoptosis induced by TGF-beta 1 in Burkitt’s lymphoma cells is caspase 8 dependent but is death receptor independent. J Immunol. 2000 Sep 1;165(5):2500–10. doi: 10.4049/jimmunol.165.5.2500. [DOI] [PubMed] [Google Scholar]
- 4.Spender LC, O’Brien DI, Simpson D, Dutt D, Gregory CD, Allday MJ, et al. TGF-beta induces apoptosis in human B cells by transcriptional regulation of BIK and BCL-XL. Cell Death Differ. 2009 Apr;16(4):593–602. doi: 10.1038/cdd.2008.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Spender LC, Inman GJ. Targeting the BCL-2 family in malignancies of germinal centre origin. Expert Opin Ther Targets. 2009 Dec;13(12):1459–72. doi: 10.1517/14728220903379565. [DOI] [PubMed] [Google Scholar]
- 6.Chonghaile TN, Letai A. Mimicking the BH3 domain to kill cancer cells. Oncogene. 2008 Dec;27(Suppl 1):S149–57. doi: 10.1038/onc.2009.52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kline MP, Rajkumar SV, Timm MM, Kimlinger TK, Haug JL, Lust JA, et al. ABT-737, an inhibitor of Bcl-2 family proteins, is a potent inducer of apoptosis in multiple myeloma cells. Leukemia. 2007 Jul;21(7):1549–60. doi: 10.1038/sj.leu.2404719. [DOI] [PubMed] [Google Scholar]
- 8.Perez-Galan P, Roue G, Lopez-Guerra M, Nguyen M, Villamor N, Montserrat E, et al. BCL-2 phosphorylation modulates sensitivity to the BH3 mimetic GX15-070 (Obatoclax) and reduces its synergistic interaction with bortezomib in chronic lymphocytic leukemia cells. Leukemia. 2008 Sep;22(9):1712–20. doi: 10.1038/leu.2008.175. [DOI] [PubMed] [Google Scholar]
- 9.Ackler S, Xiao Y, Mitten MJ, Foster K, Oleksijew A, Refici M, et al. ABT-263 and rapamycin act cooperatively to kill lymphoma cells in vitro and in vivo. Mol Cancer Ther. 2008 Oct;7(10):3265–74. doi: 10.1158/1535-7163.MCT-08-0268. [DOI] [PubMed] [Google Scholar]
- 10.Paoluzzi L, Gonen M, Bhagat G, Furman RR, Gardner JR, Scotto L, et al. The BH3-only mimetic ABT-737 synergizes the antineoplastic activity of proteasome inhibitors in lymphoid malignancies. Blood. 2008 Oct 1;112(7):2906–16. doi: 10.1182/blood-2007-12-130781. [DOI] [PubMed] [Google Scholar]
- 11.Paoluzzi L, Gonen M, Gardner JR, Mastrella J, Yang D, Holmlund J, et al. Targeting Bcl-2 family members with the BH3 mimetic AT-101 markedly enhances the therapeutic effects of chemotherapeutic agents in in vitro and in vivo models of B-cell lymphoma. Blood. 2008 Jun 1;111(11):5350–8. doi: 10.1182/blood-2007-12-129833. [DOI] [PubMed] [Google Scholar]
- 12.Mason KD, Vandenberg CJ, Scott CL, Wei AH, Cory S, Huang DC, et al. In vivo efficacy of the Bcl-2 antagonist ABT-737 against aggressive Myc-driven lymphomas. Proc Natl Acad Sci U S A. 2008 Nov 18;105(46):17961–6. doi: 10.1073/pnas.0809957105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.van Delft MF, Wei AH, Mason KD, Vandenberg CJ, Chen L, Czabotar PE, et al. The BH3 mimetic ABT-737 targets selective Bcl-2 proteins and efficiently induces apoptosis via Bak/Bax if Mcl-1 is neutralized. Cancer Cell. 2006 Nov;10(5):389–99. doi: 10.1016/j.ccr.2006.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Whitecross KF, Alsop AE, Cluse LA, Wiegmans A, Banks KM, Coomans C, et al. Defining the target specificity of ABT-737 and synergistic antitumor activities in combination with histone deacetylase inhibitors. Blood. 2009 Feb 26;113(9):1982–91. doi: 10.1182/blood-2008-05-156851. [DOI] [PubMed] [Google Scholar]
- 15.Oltersdorf T, Elmore SW, Shoemaker AR, Armstrong RC, Augeri DJ, Belli BA, et al. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature. 2005 Jun 2;435(7042):677–81. doi: 10.1038/nature03579. [DOI] [PubMed] [Google Scholar]
- 16.Gormley RP, Madan R, Dulau AE, Xu D, Tamas EF, Bhattacharyya PK, et al. Germinal center and activated b-cell profiles separate Burkitt lymphoma and diffuse large B-cell lymphoma in AIDS and non-AIDS cases. Am J Clin Pathol. 2005 Nov;124(5):790–8. doi: 10.1309/7CEA-WV0D-NLLU-WQTF. [DOI] [PubMed] [Google Scholar]
- 17.Frost M, Newell J, Lones MA, Tripp SR, Cairo MS, Perkins SL. Comparative immunohistochemical analysis of pediatric Burkitt lymphoma and diffuse large B-cell lymphoma. Am J Clin Pathol. 2004 Mar;121(3):384–92. doi: 10.1309/8WYN-VUTG-V9RP-HUQH. [DOI] [PubMed] [Google Scholar]
- 18.Inoue S, Riley J, Gant TW, Dyer MJ, Cohen GM. Apoptosis induced by histone deacetylase inhibitors in leukemic cells is mediated by Bim and Noxa. Leukemia. 2007 Aug;21(8):1773–82. doi: 10.1038/sj.leu.2404760. [DOI] [PubMed] [Google Scholar]
- 19.Cragg MS, Kuroda J, Puthalakath H, Huang DC, Strasser A. Gefitinib-induced killing of NSCLC cell lines expressing mutant EGFR requires BIM and can be enhanced by BH3 mimetics. PLoS Med. 2007 Oct;4(10):1681–89. doi: 10.1371/journal.pmed.0040316. discussion 90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Okumura K, Huang S, Sinicrope FA. Induction of Noxa sensitizes human colorectal cancer cells expressing Mcl-1 to the small-molecule Bcl-2/Bcl-xL inhibitor, ABT-737. Clin Cancer Res. 2008 Dec 15;14(24):8132–42. doi: 10.1158/1078-0432.CCR-08-1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Davis RE, Ngo VN, Lenz G, Tolar P, Young RM, Romesser PB, et al. Chronic active B-cell-receptor signalling in diffuse large B-cell lymphoma. Nature. Jan 7;463(7277):88–92. doi: 10.1038/nature08638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gordon J, Challa A, Levens JM, Gregory CD, Williams JM, Armitage RJ, et al. CD40 ligand, Bcl-2, and Bcl-xL spare group I Burkitt lymphoma cells from CD77-directed killing via Verotoxin-1 B chain but fail to protect against the holotoxin. Cell Death Differ. 2000 Sep;7(9):785–94. doi: 10.1038/sj.cdd.4400710. [DOI] [PubMed] [Google Scholar]
- 23.Sobol H, Benziane A, Kerangueven F, Yin L, Noguchi T, Pauly S, et al. Genome-wide search for loss of heterozygosity in Burkitt lymphoma cell lines. Genes Chromosomes Cancer. 2002 Feb;33(2):217–24. doi: 10.1002/gcc.10022. [DOI] [PubMed] [Google Scholar]
- 24.Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984;22:27–55. doi: 10.1016/0065-2571(84)90007-4. [DOI] [PubMed] [Google Scholar]
- 25.Del Gaizo Moore V, Brown JR, Certo M, Love TM, Novina CD, Letai A. Chronic lymphocytic leukemia requires BCL2 to sequester prodeath BIM, explaining sensitivity to BCL2 antagonist ABT-737. J Clin Invest. 2007 Jan;117(1):112–21. doi: 10.1172/JCI28281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Deng J, Carlson N, Takeyama K, Dal Cin P, Shipp M, Letai A. BH3 profiling identifies three distinct classes of apoptotic blocks to predict response to ABT-737 and conventional chemotherapeutic agents. Cancer Cell. 2007 Aug;12(2):171–85. doi: 10.1016/j.ccr.2007.07.001. [DOI] [PubMed] [Google Scholar]
- 27.Doucet JP, Hussain A, Al-Rasheed M, Gaidano G, Gutierrez MI, Magrath I, et al. Differences in the expression of apoptotic proteins in Burkitt’s lymphoma cell lines: potential models for screening apoptosis-inducing agents. Leuk Lymphoma. 2004 Feb;45(2):357–62. doi: 10.1080/10428190310001595713. [DOI] [PubMed] [Google Scholar]
- 28.Curnock AP, Knox KA. LY294002-mediated inhibition of phosphatidylinositol 3-kinase activity triggers growth inhibition and apoptosis in CD40-triggered Ramos-Burkitt lymphoma B cells. Cell Immunol. 1998 Aug 1;187(2):77–87. doi: 10.1006/cimm.1998.1335. [DOI] [PubMed] [Google Scholar]
- 29.Thoreen CC, Kang SA, Chang JW, Liu Q, Zhang J, Gao Y, et al. An ATP-competitive mammalian target of rapamycin inhibitor reveals rapamycin-resistant functions of mTORC1. J Biol Chem. 2009 Mar 20;284(12):8023–32. doi: 10.1074/jbc.M900301200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hsieh AC, Costa M, Zollo O, Davis C, Feldman ME, Testa JR, et al. Genetic dissection of the oncogenic mTOR pathway reveals druggable addiction to translational control via 4EBP-eIF4E. Cancer Cell. Mar 16;17(3):249–61. doi: 10.1016/j.ccr.2010.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dowling RJ, Topisirovic I, Alain T, Bidinosti M, Fonseca BD, Petroulakis E, et al. mTORC1-mediated cell proliferation, but not cell growth, controlled by the 4E-BPs. Science. May 28;328(5982):1172–6. doi: 10.1126/science.1187532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Feldman ME, Apsel B, Uotila A, Loewith R, Knight ZA, Ruggero D, et al. Active-site inhibitors of mTOR target rapamycin-resistant outputs of mTORC1 and mTORC2. PLoS Biol. 2009 Feb 10;7(2):e38. doi: 10.1371/journal.pbio.1000038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Thoreen CC, Sabatini DM. Rapamycin inhibits mTORC1, but not completely. Autophagy. 2009 Jul;5(5):725–6. doi: 10.4161/auto.5.5.8504. [DOI] [PubMed] [Google Scholar]
- 34.Janes MR, Limon JJ, So L, Chen J, Lim RJ, Chavez MA, et al. Effective and selective targeting of leukemia cells using a TORC1/2 kinase inhibitor. Nat Med. 2010 Feb;16(2):205–13. doi: 10.1038/nm.2091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yin X, Giap C, Lazo JS, Prochownik EV. Low molecular weight inhibitors of Myc-Max interaction and function. Oncogene. 2003 Sep 18;22(40):6151–9. doi: 10.1038/sj.onc.1206641. [DOI] [PubMed] [Google Scholar]
- 36.Huang MJ, Cheng YC, Liu CR, Lin S, Liu HE. A small-molecule c-Myc inhibitor, 10058-F4, induces cell-cycle arrest, apoptosis, and myeloid differentiation of human acute myeloid leukemia. Exp Hematol. 2006 Nov;34(11):1480–9. doi: 10.1016/j.exphem.2006.06.019. [DOI] [PubMed] [Google Scholar]
- 37.Konopleva M, Watt J, Contractor R, Tsao T, Harris D, Estrov Z, et al. Mechanisms of antileukemic activity of the novel Bcl-2 homology domain-3 mimetic GX15-070 (obatoclax) Cancer Res. 2008 May 1;68(9):3413–20. doi: 10.1158/0008-5472.CAN-07-1919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wessendorf S, Schwaenen C, Kohlhammer H, Kienle D, Wrobel G, Barth TF, et al. Hidden gene amplifications in aggressive B-cell non-Hodgkin lymphomas detected by microarray-based comparative genomic hybridization. Oncogene. 2003 Mar 6;22(9):1425–9. doi: 10.1038/sj.onc.1206297. [DOI] [PubMed] [Google Scholar]
- 39.Johnson NA, Savage KJ, Ludkovski O, Ben-Neriah S, Woods R, Steidl C, et al. Lymphomas with concurrent BCL2 and MYC translocations: the critical factors associated with survival. Blood. 2009 Sep 10;114(11):2273–9. doi: 10.1182/blood-2009-03-212191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Srinivasan L, Sasaki Y, Calado DP, Zhang B, Paik JH, DePinho RA, et al. PI3 kinase signals BCR-dependent mature B cell survival. Cell. 2009 Oct 30;139(3):573–86. doi: 10.1016/j.cell.2009.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hasselblom S, Hansson U, Olsson M, Toren L, Bergstrom A, Nilsson-Ehle H, et al. High immunohistochemical expression of p-AKT predicts inferior survival in patients with diffuse large B-cell lymphoma treated with immunochemotherapy. Br J Haematol. 2010 May;149(4):560–8. doi: 10.1111/j.1365-2141.2010.08123.x. [DOI] [PubMed] [Google Scholar]
- 42.Kuppers R. Mechanisms of B-cell lymphoma pathogenesis. Nat Rev Cancer. 2005 Apr;5(4):251–62. doi: 10.1038/nrc1589. [DOI] [PubMed] [Google Scholar]
- 43.Spender LC, Inman GJ. Inhibition of Germinal Centre Apoptotic Programmes by Epstein-Barr Virus. Advances in Hematology. 2011 doi: 10.1155/2011/829525. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Franke TF, Hornik CP, Segev L, Shostak GA, Sugimoto C. PI3K/Akt and apoptosis: size matters. Oncogene. 2003 Dec 8;22(56):8983–98. doi: 10.1038/sj.onc.1207115. [DOI] [PubMed] [Google Scholar]
- 45.Coloff JL, Macintyre AN, Nichols AG, Liu T, Gallo CA, Plas DR, et al. Akt-Dependent Glucose Metabolism Promotes Mcl-1 Synthesis to Maintain Cell Survival and Resistance to Bcl-2 Inhibition. Cancer Res. Jun 13; doi: 10.1158/0008-5472.CAN-10-4531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mazzoletti M, Bortolin F, Brunelli L, Pastorelli R, Di Giandomenico S, Erba E, et al. Combination of PI3K/mTOR inhibitors: antitumor activity and molecular correlates. Cancer Res. Jul 1;71(13):4573–84. doi: 10.1158/0008-5472.CAN-10-4322. [DOI] [PubMed] [Google Scholar]
- 47.Li Z, Van Calcar S, Qu C, Cavenee WK, Zhang MQ, Ren B. A global transcriptional regulatory role for c-Myc in Burkitt’s lymphoma cells. Proc Natl Acad Sci U S A. 2003 Jul 8;100(14):8164–9. doi: 10.1073/pnas.1332764100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mills JR, Hippo Y, Robert F, Chen SM, Malina A, Lin CJ, et al. mTORC1 promotes survival through translational control of Mcl-1. Proc Natl Acad Sci U S A. 2008 Aug 5;105(31):10853–8. doi: 10.1073/pnas.0804821105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gregory MA, Hann SR. c-Myc proteolysis by the ubiquitin-proteasome pathway: stabilization of c-Myc in Burkitt’s lymphoma cells. Mol Cell Biol. 2000 Apr;20(7):2423–35. doi: 10.1128/mcb.20.7.2423-2435.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Vu C, Fruman DA. Targeting TOR signaling in leukemia and lymphoma. Clin Cancer Res. 2010 Sep 8; doi: 10.1158/1078-0432.CCR-10-0480. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.