Executive Summary
Objective
The objective of this review was to assess the safety and effectiveness of metal on metal (MOM) hip resurfacing arthroplasty for young patients compared with that of total hip replacement (THR) in the same population.
Clinical Need
Total hip replacement has proved to be very effective for late middle-aged and elderly patients with severe degenerative diseases of the hips. As indications for THR began to include younger patients and those with a more active life style, the longevity of the implant became a concern. Evidence suggests that these patients experience relatively higher rates of early implant failure and the need for revision. The Swedish hip registry, for example, has demonstrated a survival rate in excess of 80% at 20 years for those aged over 65 years, whereas this figure was 33% by 16 years in those aged under 55 years.
Hip resurfacing arthroplasty is a bone-conserving alternative to THR that restores normal joint biomechanics and load transfer. The technique has been used around the world for more than 10 years, specifically in the United Kingdom and other European countries.
The Technology
Metal-on-metal hip resurfacing arthroplasty is an alternative procedure to conventional THR in younger patients. Hip resurfacing arthroplasty is less invasive than THR and addresses the problem of preserving femoral bone stock at the initial operation. This means that future hip revisions are possible with THR if the initial MOM arthroplasty becomes less effective with time in these younger patients. The procedure involves the removal and replacement of the surface of the femoral head with a hollow metal hemisphere, which fits into a metal acetabular cup.
Hip resurfacing arthroplasty is a technically more demanding procedure than is conventional THR. In hip resurfacing, the femoral head is retained, which makes it much more difficult to access the acetabular cup. However, hip resurfacing arthroplasty has several advantages over a conventional THR with a small (28 mm) ball. First, the large femoral head reduces the chance of dislocation, so that rates of dislocation are less than those with conventional THR. Second, the range of motion with hip resurfacing arthroplasty is higher than that achieved with conventional THR.
A variety of MOM hip resurfacing implants are used in clinical practice. Six MOM hip resurfacing implants have been issued licences in Canada.
Review Strategy
A search of electronic bibliographies (OVID Medline, Medline In-Process and Other Non-Indexed Citations, Embase, Cochrane CENTRAL and DSR, INAHTA) was undertaken to identify evidence published from Jan 1, 1997 to October 27, 2005. The search was limited to English-language articles and human studies. The literature search yielded 245 citations. Of these, 11 met inclusion criteria (9 for effectiveness, 2 for safety).
The result of the only reported randomized controlled trial on MOM hip resurfacing arthroplasty could not be included in this assessment, because it used a cemented acetabular component, whereas in the new generation of implants, a cementless acetabular component is used. After omitting this publication, only case series remained.
Summary of Findings
Health Outcomes
The Harris hip score and SF-12 are 2 measures commonly used to report health outcomes in MOM hip resurfacing arthroplasty studies. Other scales used are the Oxford hip score and the University of California Los Angeles hip score.
The case series showed that the mean revision rate of MOM hip resurfacing arthroplasty is 1.5% and the incidence of femoral neck fracture is 0.67%. Across all studies, 2 cases of osteonecrosis were reported. Four studies reported improvement in Harris hip scores. However, only 1 study reported a statistically significant improvement. Three studies reported improvement in SF-12 scores, of which 2 reported a significant improvement. One study reported significant improvement in UCLA hip score. Two studies reported postoperative Oxford hip scores, but no preoperative values were reported.
None of the reviewed studies reported procedure-related deaths. Four studies reported implant survival rates ranging from 94.4% to 99.7% for a follow-up period of 2.8 to 3.5 years. Three studies reported on the range of motion. One reported improvement in all motions including flexion, extension, abduction-adduction, and rotation, and another reported improvement in flexion. Yet another reported improvement in range of motion for flexion abduction-adduction and rotation arc. However, the author reported a decrease in the range of motion in the arc of flexion in patients with Brooker class III or IV heterotopic bone (all patients were men).
Safety of Metal-on-Metal Hip Resurfacing Arthroplasty
There is a concern about metal wear debris and its systemic distribution throughout the body. Detectable metal concentrations in the serum and urine of patients with metal hip implants have been described as early as the 1970s, and this issue is still controversial after 35 years.
Several studies have reported high concentration of cobalt and chromium in serum and/or urine of the patients with metal hip implants. Potential toxicological effects of the elevated metal ions have heightened concerns about safety of MOM bearings. This is of particular concern in young and active patients in whom life expectancy after implantation is long.
Since 1997, 15 studies, including 1 randomized clinical trial, have reported high levels of metal ions after THR with metal implants. Some of these studies have reported higher metal levels in patients with loose implants.
Adverse Biological Effects of Cobalt and Chromium
Because patients who receive a MOM hip arthroplasty are shown to be exposed to high concentrations of metallic ions, the Medical Advisory Secretariat searched the literature for reports of adverse biological effects of cobalt and chromium. Cobalt and chromium make up the major part of the metal articulations; therefore, they are a focus of concern.
Risk of Cancer
To date, only one study has examined the incidence of cancer after MOM and polyethylene on metal total hip arthroplasties. The results were compared to that of general population in Finland. The mean duration of follow-up for MOM arthroplasty was 15.7 years; for polyethylene arthroplasty, it was 12.5 years. The standardized incidence ratio for all cancers in the MOM group was 0.95 (95% CI, 0.79–1.13). In the polyethylene on metal group it was 0.76 (95% CI, 0.68–0.86). The combined standardized incidence ratio for lymphoma and leukemia in the patients who had MOM THR was 1.59 (95% CI, 0.82–2.77). It was 0.59 (95% CI, 0.29–1.05) for the patients who had polyethylene on metal THR. Patients with MOM THR had a significantly higher risk of leukemia. All patients who had leukemia were aged over than 60 years.
Cobalt Cardiotoxicity
Epidemiological Studies of Myocardiopathy of Beer Drinkers
An unusual type of myocardiopathy, characterized by pericardial effusion, elevated hemoglobin concentrations, and congestive heart failure, occurred as an epidemic affecting 48 habitual beer drinkers in Quebec City between 1965 and 1966. This epidemic was directly related the consumption of a popular beer containing cobalt sulfate. The epidemic appeared 1 month after cobalt sulfate was added to the specific brewery, and no further cases were seen a month after this specific chemical was no longer used in making this beer. A beer of the same name is made in Montreal, and the only difference at that time was that the Quebec brand of beer contained about 10 times more cobalt sulphate. Cobalt has been added to some Canadian beers since 1965 to improve the stability of the foam but it has been added in larger breweries only to draught beer. However, in small breweries, such as those in Quebec City, separate batches were not brewed for bottle and draught beer; therefore, cobalt was added to all of the beer processed in this brewery.
In March 1966, a committee was appointed under the chairmanship of the Deputy Minister of Health for Quebec that included members of the department of forensic medicine of Quebec’s Ministry of Justice, epidemiologists, members of Food and Drug Directorate of Ottawa, toxicologists, biomedical researchers, pathologists, and members of provincial police. Epidemiological studies were carried out by the Provincial Ministry of Health and the Quebec City Health Department.
The association between the development of myocardiopathy and the consumption of the particular brand of beer was proven. The mortality rate of this epidemic was 46.1% and those who survived were desperately ill, and recovered only after a struggle for their lives.
Similar cases were seen in Omaha (Nebraska). The epidemic started after a cobalt additive was used in 1 of the beers marketed in Nebraska. Sixty-four patients with the clinical diagnosis of alcoholic myocardiopathy were seen during an 18-month period (1964–1965). Thirty of these patients died. The first patient became ill within 1 month after cobalt was added to the beer, and the last patient was seen within 1 month of withdrawal of cobalt.
A similar epidemic occurred in Minneapolis, Minnesota. Between 1964 and 1967, 42 patients with acute heart failure were admitted to a hospital in Minneapolis, Minnesota. Twenty of these patients were drinking 6 to 30 bottles per day of a particular brand of beer exclusively. The other 14 patients also drank the same brand of beer, but not exclusively. The mortality rate from the acute illness was 18%, but late deaths accounted for a total mortality rate of 43%. Examination of the tissue from these patients revealed markedly abnormal changes in myofibrils (heart muscles), mitochondria, and sarcoplasmic reticulum.
In Belgium, a similar epidemic was reported in 1966, in which, cobalt was used in some Belgian beers. There was a difference in mortality between the Canadian or American epidemic and this series. Only 1 of 24 patients died, 1.5 years after the diagnosis. In March 1965, at an international meeting in Brussels, a new heart disease in chronic beer drinkers was described. This disease consists of massive pericardial effusion, low cardiac output, raised venous pressure, and polycythemia in some cases. This syndrome was thought to be different from the 2 other forms of alcoholic heart disease (beriberi and a form characterized by myocardial fibrosis).
The mystery of the above epidemics as stated by investigators is that the amount of cobalt added to the beer was below the therapeutic doses used for anemia. For example, 24 pints of Quebec brand of beer in Quebec would contain 8 mg of cobalt chloride, whereas an intake of 50 to 100 mg of cobalt as an antianemic agent has been well tolerated. Thus, greater cobalt intake alone does not explain the occurrence of myocardiopathy. It seems that there are individual differences in cobalt toxicity. Other features, like subclinical alcoholic heart disease, deficient diet, and electrolyte imbalance could have been precipitating factors that made these patients susceptible to cobalt’s toxic effects.
In the Omaha epidemic, 60% of the patients had weight loss, anorexia, and occasional vomiting and diarrhea 2 to 6 months before the onset of cardiac symptoms. In the Quebec epidemic, patients lost their appetite 3 to 6 months before the diagnosis of myocardiopathy and developed nausea in the weeks before hospital admission. In the Belgium epidemic, anorexia was one of the most predominant symptoms at the time of diagnosis, and the quality and quantity of food intake was poor. Alcohol has been shown to increase the uptake of intracoronary injected cobalt by 47%. When cobalt enters the cells, calcium exits; this shifts the cobalt to calcium ratio. The increased uptake of cobalt in alcoholic patients may explain the high incidence of cardiomyopathies in beer drinkers’ epidemics.
As all of the above suggest, it may be that prior chronic exposure to alcohol and/or a nutritionally deficient diet may have a marked synergistic effect with the cardiotoxicity of cobalt.
Conclusions
MOM hip resurfacing arthroplasty has been shown to be an effective arthroplasty procedure as tested in younger patients.
However, evidence for effectiveness is based only on 7 case series with short duration of follow-up (2.8–3.5 years). There are no RCTs or other well-controlled studies that compare MOM hip resurfacing with THR.
Revision rates reported in the MOM studies using implants currently licensed in Canada (hybrid systems, uncemented acetabular, and cemented femoral) range from 0.3% to 3.6% for a mean follow-up ranging from 2.8 to 3.5 years.
Fracture of femoral neck is not very common; it occurs in 0.4% to 2.2% of cases (as observed in a short follow-up period).
All the studies that measured health outcomes have reported improvement in Harris Hip and SF-12 scores; 1 study reported significant reduction in pain and improvement in function, and 2 studies reported significant improvement in SF-12 scores. One study reported significant improvement in UCLA Hip scores.
Concerns remain on the potential adverse effects of metal ions. Longer-term follow-up data will help to resolve the inconsistency of findings on adverse effects, including toxicity and carcinogenicity.
Ontario-Based Economic Analysis
The device cost for MOM ranges from $4,300 to $6,000 (Cdn). Traditional hip replacement devices cost about $2,000 (Cdn). Using Ontario Case Costing Initiative data, the total estimated costs for hip resurfacing surgery including physician fees, device fees, follow-up consultation, and postsurgery rehabilitation is about $15,000 (Cdn).
Figure 1: Cost of Total Hip Replacement Surgery in Ontario.
| Hip Resurfacing Surgery (Based on 2005/06 inflationary-adjusted rates) | ||
|---|---|---|
| Weighted Average (Academic Community! al-inclusive hospitalization cost of surgery | $ | 6.767.18 |
| Device Cost | $ | 5,400.00 |
| Additional Medication | $ | 330.75 |
| Follow-up Consultation | $ | 190.30 |
| Rehab (per outpatient CCAC case) | $ | 1,500.00 |
| OH P costs (average) | $ | 1,068.60 |
| Total Estimated Cost | $ | 15,256.83 |
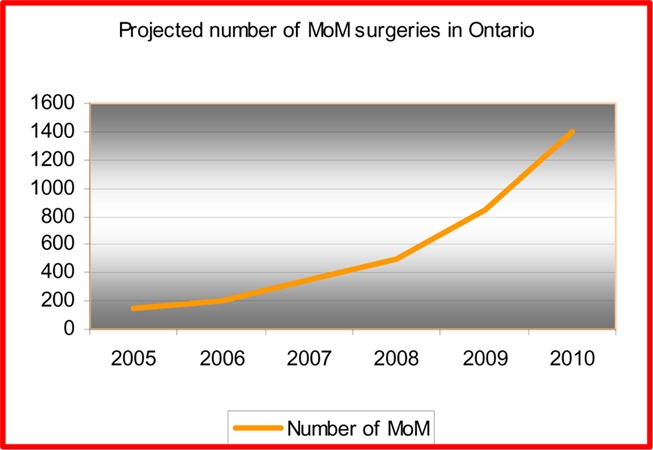
MOM hip arthroplasty is generally recommended for patients aged under 55 years because its bone-conserving advantage enables patients to “buy time” and hence helps THRs to last over the lifetime of the patient. In 2004/2005, 15.9% of patients who received THRs were aged 55 years and younger. It is estimated that there are from 600 to 1,000 annual MOM hip arthroplasty surgeries in Canada with an estimated 100 to 150 surgeries in Ontario. Given the increased public awareness of this device, it is forecasted that demand for MOM hip arthroplasty will steadily increase with a conservative estimate of demand rising to 1,400 cases by 2010 (Figure 10). The net budget impact over a 5-year period could be $500,000 to $4.7 million, mainly because of the increasing cost of the device.
Figure 2: Projected Number of Metal-on-Metal Hip Arthroplasty Surgeries in Ontario: to 2010.
Objective
The objective of this review was to assess the safety and effectiveness of metal on metal (MOM) hip resurfacing arthroplasty for young patients compared with that of total hip replacement (THR) in the same population.
Background
Clinical Need: Target Population and Condition
Total hip replacement (THR) has proved to be very effective for late middle-aged and elderly patients with severe degenerative diseases of the hips. As indications for THR began to include younger patients and those with a more active life style, the longevity of the implant became a concern. Evidence suggests that these patients experience relatively higher rates of early implant failure and the need for revision. The Swedish hip registry, for example, has demonstrated a survival rate in excess of 80% at 20 years for those aged over 65 years, whereas this figure was 33% by 16 years in those aged under 55 years. (1)
Hip resurfacing arthroplasty is a bone-conserving alternative to THR that restores normal joint biomechanics and load transfer. The technique has been used around the world for more than 10 years, specifically in the United Kingdom and other European countries. (2)
Degenerative joint disease results in the deterioration of articular cartilage that supports weight-bearing joints. The main underlying causes of degenerative hip disease are osteoarthritis and rheumatoid arthritis. Other types of degenerative hip disease include avascular necrosis of the femoral head, congenital (or developmental) dysplasia, Paget’s disease, ankylosing spondylitis, and traumatic arthritis.
Osteoarthritis
Osteoarthritis is the most common type of arthritis and is caused by “wear and tear” on the joint. The hip and knee joints are the most commonly affected. With the progression of osteoarthritis, the cartilage softens, the joint surface becomes thinner, and the integrity of the joint surface is breached. Over time, complete loss of the articular cartilage can occur, resulting in formation of rough bony surfaces (osteophytes). Ultimately, the cartilage may wear away entirely and the opposing bony surfaces articulate against each other. This results in stiffness of the joint, pain, and disability. Osteoarthritis of the weight-bearing joints develops slowly and often involves both articulating surfaces.
There is a reported negative correlation between OA of the hip (coxarthrosis) and osteoporosis of the hip. (3;4) The absence of coxarthrosis in patients with femoral neck fractures was initially reported 3 decades ago. (4) The femoral head in patients with coxarthrosis shows higher bone mineral density, stiffness, and the ability to absorb force. Increased cancellous bone mass and connectivity seen in cases of coxarthrosis probably explain, in part, the ability of patients with coxarthrosis to resist fractures of the femoral neck following a fall. (5)
Rheumatoid Arthritis
Rheumatoid arthritis is an autoimmune disease that has the potential to cause joint destruction and functional disability. It affects the lining of the joints and causes inflammatory responses, which destroy the articular cartilage and the tissues around the joints, causing joint deformity. Because it can affect multiple other organs of the body, rheumatoid arthritis is regarded as a systemic disease.
Avascular Necrosis
Avascular necrosis (osteonecrosis) of the femoral head is a debilitating disease that usually leads to destruction of the hip joint in patients in the third to fifth decades. (6) The average age of patients who have a THR for avascular necrosis is 38 years, with only 20% being more than 50 years old at the time of replacement. (6) The prevalence is unknown; however, avascular necrosis has been estimated to develop in 10,000 to 20,000 new patients a year in the United States (6) and is responsible for about 10% of the THR performed in this country. (7)
The results of THR for the treatment of avascular necrosis reported in the literature are less than optimal. THR for the treatment of this condition failed in 10 (45%) of 22 hips in the study by Dorr et al. (8) and in 11 (39%) of 28 hips in the study by Cornell et al. (9) Long life expectancy of patients, poor quality of the femoral bone, and possible persistent defects in the bone mineral metabolism associated with the use of corticosteroids are contributing factors for a high rate of THR failure. (10) Resurfacing only the femoral side of the hip joint (hemi-resurfacing) has shown favourable results after short-term follow-ups. (11)
Existing Technology Other Than That Being Reviewed
Total Hip Replacement in Young Patients
There is no cure for degenerative hip disease. Treatment options include pain management, reducing joint mobility and stiffness, exercise, and weight reduction. Surgery is indicated when conservative medical treatment has failed. In THR, both surfaces of the hip joint (the femoral head and the acetabulum) are replaced with prostheses. A variety of prostheses with different materials and different methods of fixation are available.
The outcomes of THR are generally good in older people. However, the outcomes of THR in younger active patients are less impressive. Younger patients place increased demands on their joints, because they are more active and because they have a longer life expectancy. A variety of implant options for young patients are available. This includes cemented, hybrid, proximally coated, and extensively coated implants. Different bearing surfaces such as metal on polyethylene, ceramic on ceramic, and metal-on-metal can be used.
The Swedish National Hip Replacement Register provides data for THRs in patients in 4 different age groups. (See Figures 1–4.)
Figure 1: Total Hip Replacement in People Aged Under 50 Years.
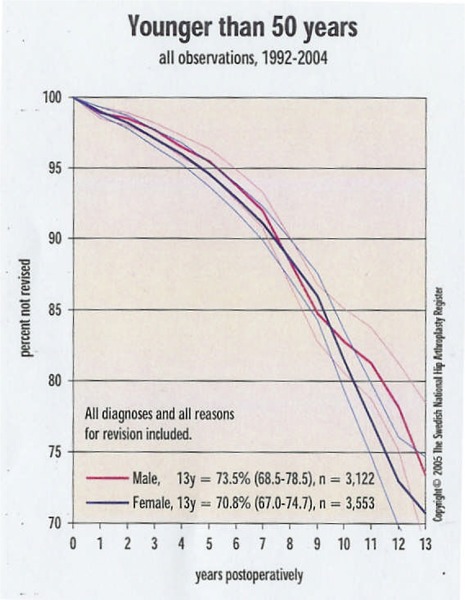
Reproduced with permission from Swedish Total Hip Replacement Register; Annual report 2004: www.jru.orthop.gu.se
Figure 4: Total Hip Replacement in People Aged Over 75 Years.
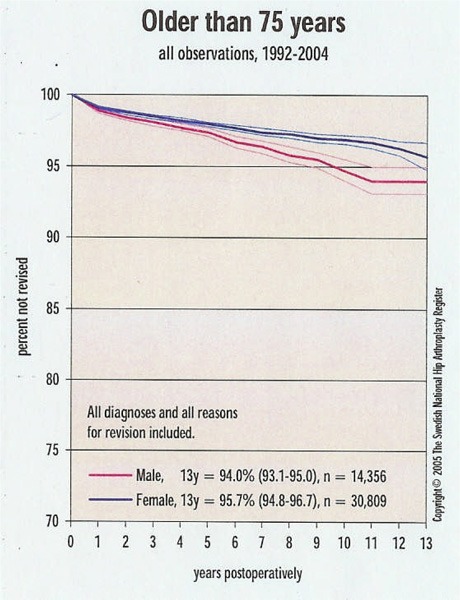
Reproduced with permission from Swedish Total Hip Replacement Register; Annual report 2004: www.jru.orthop.gu.se
Figure 2: Total Hip Replacement in People Aged 50 to 59 Years.
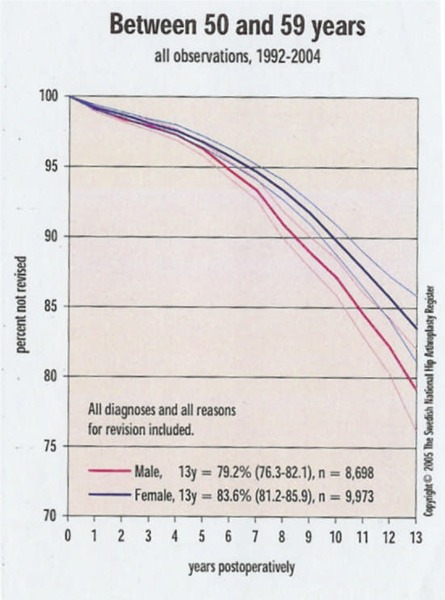
Reproduced with permission from Swedish Total Hip Replacement Register; Annual report 2004: www.jru.orthop.gu.se
Figure 3: Total Hip Replacement in People Aged 60 to 75 Years.
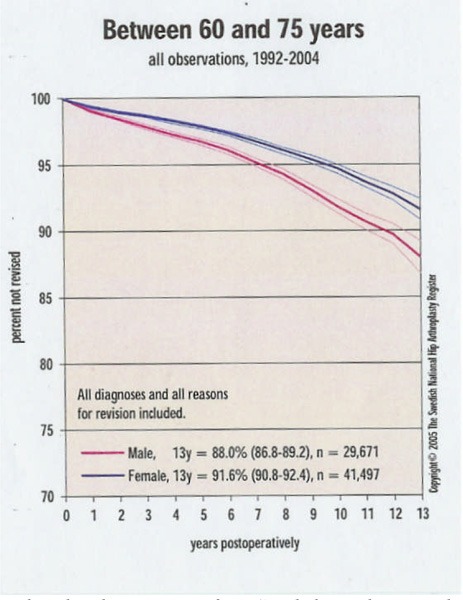
Reproduced with permission from Swedish Total Hip Replacement Register; Annual report 2004: www.jru.orthop.gu.se
Hip Resurfacing Methods
Hip resurfacing has emerged as a bone-conserving alternative to THR for younger patients with the goal to delay the need for primary THR and to reduce the need for future revision surgery. This method provides pain relief, restores function, and preserves the bone stock. The proposed benefits of hip resurfacing compared with THR are the lower rate of wear, dislocation, and loosening.
Hip resurfacing requires having good bone stock; therefore, diseases that reduce the viability of the femoral head are contraindications for this operation. Osteoporosis has been considered as a contraindication to total hip resurfacing since it predisposes the femoral neck to fracture following resurfacing. Destructive arthritis caused by long-term use of anti-inflammatory drugs has also been considered as contraindication for this method.
Complications of Hip Arthroplasty
Osteolysis and Wear Debris
Osteolysis (inflammatory bone resorption) is defined as the progressive destruction of the bony tissues around prosthetic devices because of wear particles in the joint space. It is demonstrated by radiolucent lines or cavitations at the implant-bone or cement-bone interface. Several retrieval studies (12) have shown that osteolysis is responsible for most failures.
However, osteolysis occurs less often with metal-on-metal (MOM) implants than it does with polyethylene implants. (12) Dorr et al. (13) reported a polyethylene wear at least 0.2 mm per year or volumetric wear at least 150 cubic mm per year. The rationale for the use of a MOM articulation is that it produces fewer wear particles than a polyethylene or metal on polyethylene articulation. Schmalzried et al. (14) estimated that the wear of the acetabular and femoral components of McKee-Farrar MOM implants retrieved at 20 years was 4.2 micrometers per year.
Ceramic and titanium against polyethylene have also been used for THR. These materials produce less debris, but all have potential problems. A ceramic femoral shell can break and produce large sharp fragments within the hip. A titanium surface can fragment and wear rapidly. (15)
Dislocation
Dislocation is one of the most common complications of THR. This complication has multifactorial causes including demographic, operative, and implant design variables. (16) Femoral head diameter is one of the most important operative variables that might affect dislocation for the following reasons: a larger femoral head improves the head-to-neck ratio, which increases the range of motion of the prosthetic components without prosthetic impingement; because a larger femoral head is better contained by the surrounding soft-tissue envelope, there may be greater soft-tissue restraints to dislocation; and a greater amount of translation of the femoral head is required before dislocation occurs. Berry et al. (16) showed that, of the femoral head diameters that were investigated, the largest was associated with the lowest risk of dislocation. The dislocation risk was highest for the 22-mmdiameter heads, intermediate for the 28-mmdiameter heads, and lowest for the 32-mmdiameter heads.
Osteonecrosis
Secondary avascular necrosis can occur after resurfacing of the hip if the blood supply to the femoral head and neck is disturbed. However, the incidence of this complication is low. (17) Fracture of the femoral neck may result in avascular necrosis of the femoral head. Recently, Little et al. (18) reported an incidence of femoral neck fracture in about 2% of the cases of hip resurfacing. The fractured necks displayed established osteonecrosis although, at the time of surgery, there was no histological evidence of this pathology.
Steffen et al. (19) inserted an electrode up to the femoral neck into the femoral head of 10 patients undergoing an MOM hip resurfacing arthroplasty and measured the oxygen concentration during the operation. In every patient, the blood flow was compromised during surgery, but the extent varied. In 3 patients, the oxygen concentration was zero at the end of the procedure. The surgical approach caused a mean 60% drop (P < .005) in oxygen concentration, whereas component insertion led to a further 20% drop (P < .04). The oxygen concentration did not improve significantly after the wound was closed. This study shows that during hip resurfacing arthroplasty, patients experience some compromise to their femoral head blood supply and some have complete disruption.
Femoral Neck Fracture
Another concern regarding resurfacing is the incidence of postoperative femoral neck fracture. Steffen et al. (19) reported that fractures of the femoral neck occurred from 0% to 12% of patients. Amstutz et al. (20) reported an incidence of 0.83% in their series of 600 MOM hip resurfacing arthroplasties.
Adverse Effects of Metal Particles and Ions
Metallic implants are composed of 3 groups of alloys: cobalt-chrome, stainless steel, and titanium. (15) MOM hip resurfacing implants are made of Co-Cr alloy. There is a concern about the spread of metal debris through the body as small particles or metal ions following implantation of MOM implants. (21;22) Potential sources of ion release include wear of the bearing surfaces and corrosion. The possibility of systemic toxicity and a concern about late carcinogenesis, particularly because these implants are used for young people for a very long period, is a matter of continuing debate. (22)
Corrosion is the gradual destruction of the metal or alloy by electrochemical reaction. Movement between components of the same metal causes corrosion. This phenomenon is not only a function of the implant material, but also of the movement imposed around it. The surface of the implant oxidizes, and it is the stability of the oxide layer that determines how resistant the implant will be to corrosion. The most stable oxide layer is titanium and the least is stainless steel, which is highly likely to corrode, particularly when in contact with other metals, such as wires or screw used for bone fixation. (15)
The safety standards for cobalt and chromium are derived primarily from inhalation exposure to these elements rather than the direct route via the blood stream. (23) Chromium VI has been considered as human carcinogen. Long-term occupational inhalation exposure to chromium levels 100 to 1,000 times higher than those found in the natural environment have been associated with respiratory cancers. (23)
End-stage chronic renal disease has been considered as a contraindication for the use of MOM articulation. Brodner et al. (24) demonstrated a markedly elevated serum cobalt level (119.2 μ/L) in a patient with end-stage renal failure.
Another potential adverse effect is that people with metal implants may become somehow sensitized to metal particles. Metal ions released from the metal implant can combine with proteins and activate the immune system as antigens and elicit hypersensitivity reactions. (25) Nickel is the most common metal sentitizer in humans, followed by cobalt and chromium. (25) Willert et al. (26) performed histological examinations on periprosthetic tissues retrieved during revision surgeries and found immunological response and lymphocytic infiltration in the tissues around the implant. Hallab et al. (27) have shown a link between metal exposure and lymphocyte reactivity. The incidence of hypersensitivity is about 2 per 10,000. (25)
The amount of the wear debris from MOM articulation is much less than that produced by polyethylene on metal or ceramic on polyethylene articulations. (28) The metal ions produced by MOM implants, unlike polyethylene particles, can be transported to the blood and other body fluids. The measurement of cobalt and chromium in the blood can, therefore, be used to estimate the bearing couple wear. (28)
New Generation of Metal-on-Metal Hip Prostheses
Improvements in manufacturing procedures have enhanced the metallurgy and design of MOM hip implants. (29) Improvement in manufacturing parameters such as clearance, tolerance, and sphericity, has allowed the implant to be made with more precision. (2)
The human acetabulum is considerably less than a hemisphere and it is necessary to mimic this in the acetabular component in resurfacing. Irregularities in the manufacturing process could cause the metal head and the cup to grab each other (impingement). this feature was realized by Freeman et al. (15) and incorporated into a number of designs. However, testing of the implants after revision of some recent designs shows that the problem of impingement between the femoral neck and the edge of the prosthetic socket has not been fully eliminated. (15)
Incidence and Prevalence of Hip Replacements
About 10% of hip replacements are done for patients under the age of 55. (1) Data from Canadian Joint Replacement Registry (CJRR) show the following statistics for number of hip arthroplasties for different age groups (Table 1). (Personal communication)
Table 1: Number of Hip Arthroplasties Reported to the Canadian Joint Replacement Registry in 2002/2003.
| Age Group | Rate per 100,000 | |
|---|---|---|
| Males | Females | |
| <45 | 5.7 | 5.5 |
| 45–54 | 35.9 | 42.9 |
| 55–64 | 131.0 | 131.8 |
| 65–74 | 295.5 | 335.8 |
| 75–84 | 400.2 | 434.0 |
New Technology Being Reviewed
Metal-on-Metal Hip Resurfacing Arthroplasty
Metal-on-metal hip resurfacing arthroplasty is an alternative procedure to conventional THR for younger patients. The technique preserves femoral bone stock at the initial operation (30) and future hip revisions are possible with THR if the initial MOM arthroplasty becomes less effective over time in these younger patients. The procedure involves the removal and replacement of the surface of the femoral head with a hollow metal hemisphere, which fits into a metal acetabular cup. Hip resurfacing has the advantages of preservation of proximal femoral bone stock at the time of surgery. However, this technique conserves only the femoral bone and not on the acetabular side. The MOM implant has a low wear-producing articulation. Wear particles from MOM bearings are nanometers in linear dimension, substantially smaller than polyethylene wear particles. (25) In addition, the large diameter of the articulation offers increased instability and a greater range of motion.
The first hip resurfacing arthroplasty was developed by Charnley (31) in the early 1950s. This was Teflon-on-Teflon bearing, which had a high failure rate. Loosening of both components due to rapid wear and an intense tissue reaction resulted in clinical failure and abandonment of the procedure. In the mid 1960s, Muller and Boltzy (30) used a MOM hip resurfacing system. In the 1970s and 1980s, a number of surface replacement implants made of polyethylene were used. The results of hip resurfacing in 1970s and 1980s were disappointing because of excessive wear, osteolysis, bone loss, and early failure, (29) and the procedure was largely abandoned in the mid-1980s.
Hip resurfacing arthroplasty is an old concept that has undergone a resurgence of interest. Two European designs (Wagner and McMinn) were the first to reintroduce MOM hip resurfacing arthroplasty. Over the past 15 years, with increased understanding of wear products and its role in the cause of aseptic loosening, there has been increased interest in the use of MOM bearings. Some studies (32) have shown that in MOM hip resurfacing, formation of lubricating film separates the bearing surfaces and reduces the amount of wear; therefore, this procedure may provide an opportunity to reduce the amount of wear. Two parameters that largely influence the lubrication are the diameter of the femoral head and the clearance of the articulation. (32).
Hip resurfacing arthroplasty is a technically more demanding procedure than is conventional THR. (2) In hip resurfacing, the femoral head is retained, which makes it much more difficult to access the acetabular cup. However, hip resurfacing arthroplasty has several advantages over a conventional THR with a small (28 mm) ball. First, the large femoral head reduces the chance of dislocation, so that rates of dislocation are less than those with conventional THR. Second, the range of motion with hip resurfacing arthroplasty is higher than that achieved with conventional THR.
Regulatory Status
A variety of MOM hip resurfacing implants are used in clinical practice. Six MOM hip resurfacing implants have been issued licences in Canada (Table 2).
Table 2: Hip Resurfacing Prostheses Licensed by Health Canada.
| Prosthesis | Manufacturer | Introduced | Licensed in Canada | License number | Class |
|---|---|---|---|---|---|
| Conserve Plus | Wright Medical Technology | 1996 | 2003 (Femoral resurfacing system was approved in 1999) |
61779 | 3 |
| Birmingham Hip | Smith & Nephew | 1997 | 2002 | 60640 | 3 |
| Resurfacing (BHR) | Orthopedics | ||||
| Cormet | Corin Medical Limited | 1997 | 2005 | 70449 | 3 |
| Durum | Zimmer | 2001 | 2005 | 68144 | 3 |
| ASR (Articulating Surface Replacement) | DePuy Orthopedics | 2003 | 2005 | 69823 | 3 |
| ReCap Femoral resurfacing component) | Biomet | 2004 | 2004 | 72082 | 3 |
Literature Review on Effectiveness
Objective
The objective of this review was to assess the safety and effectiveness of MOM hip resurfacing arthroplasty for young patients compared with that of THR in the same population.
Questions Asked
How do long-term outcomes of MOM hip resurfacing arthroplasty compare with THR, specifically the need for revisions?
Is there any risk associated with metal-on-metal hip arthroplasty?
Methods
Inclusion Criteria
Studies comparing the clinical outcomes of MOM hip resurfacing arthroplasty with THR or other procedures for the treatment of degenerative hip diseases in young patients
Studies reporting on the effectiveness of MOM hip resurfacing arthroplasty in young patients
Studies reporting on the safety of implant materials
Exclusion Criteria
Studies reporting on the technical aspect of hip resurfacing
Studies that did not contain patient data
Measures of Effectiveness
Revision rate
Pain relief and improvement in function
Measures of Safety
Safety of hip resurfacing arthroplasty
Safety of the implant material
-
Complications
Osteolysis
Dislocation
Osteonecrosis
Femoral neck fracture
Literature Search
A search of electronic bibliographies (OVID Medline, Medline In-Process and Other Non-Indexed Citations, EMBASE, Cochrane CENTRAL and DSR, INAHTA) was undertaken to identify evidence published from Jan 1, 1997 to October 27, 2005. The search was limited to English-language articles and human studies.
Results of Literature Search
Health Technology Assessments
The search identified a systematic review from United Kingdom published in 2002, and a Technote from Alberta Heritage Foundation for Medical Research published in 2002.
It also identified 245 citations, of which11 met the inclusion criteria (Table 3).
Table 3: Number of Identified Studies on Metal-on-Metal Hip Resurfacing Arthroplasty.
| Type of Study | Number |
|---|---|
| Randomized controlled trial | 1 |
| Observational study | 0 |
| Case series (effectiveness) | 8 |
| Case series (safety) | 2 |
Grading the Body of Evidence
To evaluate the strength of the body of evidence, we applied the GRADE system formulated by the GRADE working group and adopted by the Medical Advisory Secretariat. This system classifies quality of evidence as high (Grade A), moderate (Grade B), or low (Grade C) according to the 4 key elements: study design, study quality, consistency across studies, and directness. (See Appendix 3)
Randomized Controlled Trials
Howie et al. (33) examined the results of hip resurfacing arthroplasty in a randomized controlled trial of patients 55 years of age or younger. The investigators compared MOM cementing hip resurfacing with cemented THR. The hip resurfacing arthroplasty implant comprised a low profile McMinn acetabular component and a mini-stemmed McMinn femoral resurfacing component, both manufactured with high carbon cast cobalt-chrome. The nonarticular surface of the acetabular component had recesses to aid cement fixation. The THR comprised an Exeter polished stainless steel femoral stem and an Exeter ultrahigh molecular weight polyethylene acetabular component. The trial hypothesis was that when 80% of THR hips require second femoral revision, fewer than 50% of resurfacing hips require a second femoral revision. Based on 80% power, the sample size was calculated as 47 patients in each group. Table 4 shows the study characteristics.
Table 4: Randomized Controlled Trial on Metal-on-Metal Hip Resurfacing Arthroplasty: Study Characteristics*.
| Study Design and Period |
Duration of Follow-up (Years) Median (Range) |
Groups | Number of patients (hips) |
Mean age Years (range) |
Preop Diagnosis (%) |
Prosthesis | Patients Available for Follow-Up |
|---|---|---|---|---|---|---|---|
| RCT (block randomization) 1993–1995 | 8.5 (8–10) | Hip resurfacing | 11 | 46 (16–55) | OA (64) | McMinn | All |
| THR | 13 | 50 (22–54) | OA (62) | Exeter | All |
Howie et al.; (33) The trial was stopped after 2 years of recruitment because of a high incidence of failure of the cemented hip resurfacing arthroplasty. OA indicates osteoarthritis.
This trial was stopped because of a high incidence of failure of the cemented hip resurfacing arthroplasty. Eight (73%) of 11 hips were converted to THR. The reasons for failure were femoral neck fractures in 2 patients, femoral component loosening at the prosthesis-cement and cement-bone interface in 1 patient who had had a previous femoral neck surgery, and loosening of the cemented metal-backed acetabular component in 5 patients. Polishing wear of the matt surface of the non-articulating metal back of the acetabular component was seen at the time of retrieval. This confirmed the movement of acetabular component within the cement. Osteolysis was evident at the time of revision in 3 hips.
In THR group, 2 cemented acetabular components in 2 patients (15%) were revised. One case had a loosening at the prosthesis-cement and cement-bone interfaces and was revised after 5 years. The second case had cement fracture and loosening at the prosthesis–cement interface and was revised after 9 years. In addition, 1 patient had a radiographically loose acetabular component and was in the waiting list for revision.
In both groups, Harris hip scores (HHS) and Harris pain scores improved. Table 5 shows details of postoperative outcomes of hip resurfacing arthroplasty and THR.
Table 5: Clinical Outcomes of Hip Resurfacing Arthroplasty and Total Hip Arthroplasty: Randomized Clinical Trial*.
| Study | Groups | Revision/ Conversion Rate |
Evidence of Loosening | Evidence of Fracture of Femur | Evidence of Osteolysis | Harris Hip Score (Median) | Harris Pain Score (Median) |
|---|---|---|---|---|---|---|---|
| Number (%) | Number (%) | Number (%) | Number (%) | ||||
| Howie et al. 2005(33) | Hip resurfacing | Conversion to THR: 8/11 (73) | 6/11 (56) | 2/11 (18) | 3 (27) were found at the time of revision | Preoperative: 43/100 At 2-years follow-up: 89/100 |
Preoperative: 10/44 At 2-years follow-up: 40/44 |
| THR | 2/13 (15) | 3/13 (23) | 0 (0) | NR | Preoperative: 46/100 At 2-years follow-up: 93/100 |
Preoperative: 20/44 At 2-years follow-up: 44/44 |
(See appendix 1 for Harris hip score); NR indicates not reported.
Seven of the 8 revisions of MOM occurred within 5 years and 1 occurred at 9.5 years postoperatively. In the THR group, 1 revision occurred at 5 years, and 1 occurred at 9 years postoperatively.
The 2 early femoral neck fractures in a 53-year-old patient and a 45-year-old patient and 1 case of femoral loosening with a history of previous femoral neck surgery reinforced the opinion that MOM hip arthroplasty should be confined to young patients who have good bone integrity and no previous femoral neck surgery.
There was no difference in the range of flexion or rotation between MOM and THR. For abduction and adduction range of motion, there was no difference in the medians or ranges of values between groups.
Loosening of the acetabular component raised concern about the design of the prosthesis. Subsequent new designs of MOM have a porous, coated, cementless acetabular component that eliminates the potential for loosening of the acetabular component in the cement. (34;35).
The result of this randomized controlled trial (RCT) on MOM hip resurfacing arthroplasty could not be used for this assessment because in the new generation of implants, a cementless acetabular component is used. Therefore, the results of case series were reviewed to establish evidence of effectiveness and complication rates.
Case Series
Eight case series reported clinical outcomes of hip resurfacing arthroplasty. (34-41) Seven of these used prostheses with uncemented acetabular components (34-40) One (41) used the McMinn cemented version. The characteristics of patients in these case series are shown in Table 6 (next page).
Table 6: Metal-on-Metal Hip Resurfacing Arthroplasty: Clinical Outcomes of Case Series*.
| Study | Study Period | Mean Duration of Follow-up, Years | Number of Patients (Hips) | Mean Age, Years (Range) | Preoperative Diagnosis Number (%) | Prosthesis | Number of Patients Available for Follow-up |
|---|---|---|---|---|---|---|---|
| De Smet et al. 2005 (36) | 1998–2004 | 2.8 (2–5.0) | 252 (268) | 49.7 (16–75) | OA: 203 (80.6) RA: 9 (3.57) Necrosis: 22 (7.26) Congenital dislocation: 12 (4.76) Traumatic: 3 (1.19) Neurometabolic: 1 (0.4) Other: 2 (0.79) |
BHR | All except 3 patients who died |
| Back et al. 2005 (37) | 1999–2001 | 3 (2–4.4) | 230 | 52.1 (18–82) | OA: 203 (88.3) RA: 3 (1.3) Avascular necrosis: 12 (5.2) Neurometabolic: 2 (0.9) Other: 10 (4.3) |
BHR | All |
| Treacy et al. 2005 (38) | 1997–1998 | 130 (144) | 52.1 (17–76) | OA: 125 (87) RA: 2 (1) Avascular necrosis: 10 (7) Developmental dysplasia: 3 (2) Other: 4 (3) |
BHR | At 5 years: 107 (76) hips | |
| Lilikakis et al. 2005 (39) | 2002–2002 | 2.8 (2–3.2) | 66 (70) | 51.5 (23.3–72.7) | OA: 64 (97) Osteonecrosis: 1 (1) Chondrolysis: 1 (1) |
Cormet 2000: femoral and acetabular components were coated with hydroxyapatite | 59/66 radiographs were available 60/66 patients returned the questionnaire at the 2-year follow-up |
| Amstutz et al. 2004 (34) | 1996–2000 | 3.5 (2.26.2) | 355 (400 hips) | 48.2 (1577) | OA: 262 (66) Osteonecrosis: 36 (9) Developmental dysplasia: 43 (10.8) Posttraumatic c arthritis: 31 (7.8) Leg-Calve-Perthes disease: 10 (2.5) Slipped capital femoral epiphysis: 7 (1.8) Ankylosing spondylitis: 4 (1) Juvenile rheumatoid arthritis: 3 (0.8) Rheumatoid arthritis: 3 (0.8) Melorheostosis: 1 (0.3) Previous operations: 25 (6.3) |
Conserve Plus | 352 |
| Daniel et al. 2004 (35) | 1994–2001 (but not 1996)* | 3.3 (1.1-8.2) | 384 (446 hips) | 48.3 (26.8-54.9) | Primary OA | McMinn (43 hips, 1994–1995) 43 prostheses BHR (403 hips 1997-2001) 403 prostheses |
All |
| Beaule et al. 2004 (40) | NR | 3 (2–5) | 83 (94) | 34.2 (15–40) | OA: (24.4) Trauma: (18.1) Osteonecrosis: (18.1) Developmental dysplasia of the hip: (19.1) Rheumatoid diseases: (6.4) Slipped capital femoral epiphysis: (4.3) Leg-Calve-Perthes disease: (6.4) Ankylosing spondylitis: (3.2) |
Conserve Plus | 81 |
| Beaule et al. 2004 (41) | 1993–1996 | 8.7 (7.2–10) | 39 (42 hips) | 47.5 (22–69) | OA: 23 (55) Osteonecrosis: 7 (16.7) Hip dysplasia: 4 (9.5) Arthrokatadysis: 3 (7) RA: 2 (4.8) Slipped capital femoral epiphysis: 2 (4.8) Legg-Calve-Perthes: 1 (2.4) |
McMinn femoral components: All were cemented. acetabular component: Was cemented into the native acetabulum in 19 hips. 16 hips were reconstructed by cementing the McMinn acetabular component into the cemented into a cementless cobalt chromium alloy, porous-coated surface replacement socket due to absence of a locking mechanism. 7 hips were hydroxyapatite coated. |
All |
186 patients operated on in 1996 were excluded from the study because a unique pattern of failure, (believed to be due to the manufacture) occurred in the implants used; OA indicates osteoarthritis; RA, rheumatoid arthritis; AN, avascular necrosis; BHR, Birmingham Hip Resurfacing; NR, not reported.
Clinical Outcomes
Clinical outcomes of MOM hip resurfacing arthroplasty are summarized in Tables 7 to 10.
Table 7: Metal-on-Metal Hip Resurfacing Arthroplasty: Clinical Outcomes of Case Series*.
| Study | Revision/Conversion Rate | Femoral Neck Fracture | Avascular Necrosis | Osteolysis | Heterotopic Bone | Other Complications |
|---|---|---|---|---|---|---|
| Number (%) | Number (%) | Number (%) | Number (%) | Number (%) | Number (%) | |
| Reasons | Reasons | Reasons | ||||
| De Smet 2005 (36) | 3 (1.2) 1 femoral neck fracture at 3 weeks 1 avascular necrosis at 2 years 1 low grade infection at 2 years |
1 at 3 weeks (0.4) | 1 (0.4) | 2 (0.8) (Seen in revision cases) | 4 (1.6) | 1 deep venous thrombosis 1 pulmonary embolism 1 infection 2 (0.8) sciatic nerve palsy 1 dislocation |
| Back et al. 2005 (37) | 1 (0.4) at 18 month revised to THR Loose acetabular component |
1 (2.2) at 6 weeks 4 stress fractures |
0 | 0 | 59.56% of the hips (3 underwent excision of the heterotopic bone at a mean of 1 year after surgery) | Notched femoral neck: 5 Nerve palsy: 5 Vascular injury: 3 Superficial infection: 11 Urinary tract infection: 9 Deep venous thrombosis: 11 Pulmonary embolism: 2 Sinus tachycardia: 5 Hypotension: 14 Pressure sores: 4 |
| Treacy et al. 2005 (38) | 3 (2.3) within 2 years 2 Loosening due to deep infection 1 Subcapital fracture due to deep infection |
1 (0.7) Deep infection | 1 (0.7) at 9 months Local emboli | 0 (0) | 30 (28) | |
| Lilikakis et al. 2005 (39) | 2 (3) 1 for aseptic loosening at 15 months 1 for infection |
0 | 0 | 0 | 1 | Intraoperative notching: 16 (the hips with notching had significantly higher degrees of valgus placement [13.4 vs. 9.9, P.08]) Displacement of the cup: 1 Pulmonary embolism: 1 Superficial wound infection: 1 Wound hematoma: 1 |
| Amstutzet al. 2005 (34) | 12 (3.4) were converted to THR 7 for loosening of femoral component 3 for femoral neck fracture 1 for recurrent subluxations 1 for a late hematogenous infection (at 36 months) |
3 (0.85) 2 within the first 6 weeks 1 at 20 months |
0 | 0 | 106 26 (7) All were men |
Dislocation: 3 (0.75) 4 hips required reoperation (1 for cup exchange because of component mismatch 2 hips in 1 patient required removal of heterotopic bone 1 hip with trochanteric bursitis required wire removal |
| Daniel et al. 2004 (35) | 1 (0.3) was revised to THR with a ceramic-on-polyethylene implant after 8 months Avascular necrosis |
0 | 0 | 0 | 0 | 1 pulmonary embolism |
| Beaule et al. 2004 (40) | 3 (3.6) were converted to THR at a mean of 27 (2–50) months 1 for component loosening at 29 months 1 femoral neck fracture at 2 months 1 for subluxation at 50 months |
1 (1.2) | 0 | (2.1) | NR | 1 subluxation 1 patient had a socket exchange because of component size mismatch during index surgery. 1 patient required surgery for trochanteric bursitis and developed trochanteric nonunion |
| Beaule et al. 2004 (41) | 14/42 (35.9) hip were revised at an average 52.4 months (9.7-95.5) (11 to THR and 3 to MOM arthroplasty) Aseptic loosening (9 due to loosening of a cemented acetabular socket, 1 was cementless acetabular socket, 3 were femoral failures, 1 due to a late hematogenous sepsis) |
1 (2.6) | 0 | 1 (2.6) | NR | NR |
NR indicates not reported; THR, total hip replacement.
Table 10: Implant Survival, Patient Survival, and Improvement in Motion*.
| Study | Survival Rate % |
Improvement in Range of Motion Mean (range) |
Death |
|---|---|---|---|
| De Smet 2005 (36) | 3 (were lost to further follow-up) | ||
| Back et al. 2005 (37) | NR | Improved in all patients The mean flexion improved from 91.52° (25°-130°) to 110.41° (80°-130°) | 1 (unrelated causes) |
| Treacy et al. 2005 (38) | 98 (95% CI, 92-100) at the start of 6 year excluding the 2 septic failures | NR | 4 (unrelated causes) |
| Lilikakis et al. 2005 (39) | 97.1, SD 0.02 | NR | 1 (unrelated causes) |
| Amstutz et al. 2004 (34) | 94.4 (95% CI, 91-98) at 4 years | Improved Flexion: 85.5 to 122 Abduction-adduction measured in extension: 30.5 to 69.8 Rotation arc measured in extension: 18.5 to 73.7 Patients with Brooker class III or IV heterotopic bone (all were men) had a decreased range of motion in the arc of flexion (mean 109.5°; range 55°-140° compared with that for the rest of male patients (121.9°; range 85°-155°; P = .001]. All of the patients had functional arcs of rotation and abduction-adduction |
2 (unrelated causes) |
| Daniel et al. 2004 (35) | 99.7 | NR | 6 (unrelated causes) |
| Beaule et al. 2004 (40) | NR | Range of motion improved Flexion and extension: 79.5 to 116.2 abduction and adduction measured in extension: 28.9 to 70 Rotation measured in extension. 20.2 to 74.5 |
0 |
| Beaule et al. 2004 (41) | At 7 years: 79 (95% CI, 65-92) Conversion to THR as the end point (excluding 1 case revised for sepsis): 87 (95% CI, 76-98) |
NR | 1 patient died |
| Cemented socket fixation did significantly worse than cementless fixation: 66 (95% CI, 44-88 vs. 95 (95 CI, 85-100); P = .014 |
NR indicates not reported.
The results of the study (41) that used a cemented acetabular implant (McMinn prosthesis) were not included in our analysis since all companies are now manufacturing cementless acetabular implants. However, we have presented the data in the Table 8 for further discussion. The revision rate of MOM hip resurfacing arthroplasty was calculated as 1.5% and the incidence of femoral neck fracture as 0.67%. Overall, 2 cases of osteonecrosis were reported across all studies. Important clinical outcomes are summarized in Table 8.
Table 8: Summary of Clinical and Radiological Findings*.
| Study | Number of Patients (Hips) |
Mean Duration of Follow-up, Years (Range) |
Prosthesis | Revision Rate Number (%) |
Femoral Fracture Number (%) |
Osteonecrosis Number (%) |
Osteolysis Number (%) |
|---|---|---|---|---|---|---|---|
| De Smet et al. 2005 (36) | 252 (268) | 2.8 (2.0-5.0) | BHR | 3 (1.2) | 1 (0.4) | 1 (0.4) | 2 (0.8) |
| Back et al. 2005 (37) | (230) | 3 (2.0-4.4) | BHR | 1 (0.4) | 5 (2.2) | 0 | 0 (0.0) |
| Treacy et al. 2005 (38) | 130 (144) | NR | BHR | 3 (2.3) | 1 (0.7) | 1 (0.7) | 0 (0.0) |
| Lilikakis et al. 2005 (39) | 66 (70) | 2.8 (2.0-3.2) | Cormet 2000 | 2 (3.0) | 0 (0.0) | 0 | 0 (0.0) |
| Amstutz et al. 2004 (34) | 355 (400) | 3.5 (2.2-6.2) | Conserve Plus | 12 (3.4) | 3 (.85) | 0 | 0 (0.0) |
| Daniel et al. 2004 (35) | 384 (446) | 3.3 (1.1-8.2) | BHR: 403 McMinn: 43 | 1 (0.3) | 0 (0.0) | 0 | 0 (0.0) |
| Beaule et al. 2004 (40) | 83 (94) | 3 (2.0-5.0) | Conserve Plus | 3 (3.6) | 1 (1.2) | 0 | 2 (2.0) |
| Beaule et al. 2004*(41) | 39 (42) | 8.7 (7.2-10) | McMinn | 14 (35.9) | 1 (2.6) | 0 | 1 (2.6) |
The results of the study that used implants with a cemented acetabular component are highlighted. This study was not included in this analysis based on the reasons mentioned above; NR indicates not reported; BHR, Birmingham Hip Resurfacing.
In Beaule’s study (41), 50% (9 out of 18) of the cemented acetabular components failed. According to the author, this result justified discontinuing use of cement fixation on the acetabular side. The authors noted that failures with the cemented sockets occurred at the prosthesis-cement interface where the smooth finish of the backside of these components was inadequate for this type of fixation. The result for the cemented femoral component was better than that of the first generation of metal-on-polyethylene surface arthroplasty. The cemented femoral design continues to be used. However, De Smet et al. (36) did not advise cementing of the stem because stress distribution by the stem is not desirable, and the first signs of femoral loosening, avascular necrosis, infection, and wear, which are often seen around the stem of the resurfacing implant may be masked.
Health Outcome Measures
The Harris Hip Score (HHS) and SF-12 are 2 measures that have been used to report health outcomes in MOM hip resurfacing arthroplasty studies.
The HHS measures pain, functional capacity, range of motion, and deformity. Pain and reduced function are the indications for surgery in the vast majority of patients with hip problems. The HHS is a point scale with a maximum of 100 points was created. The maximum possible scores for pain and for function are 44 and 47 respectively. (42) (See Appendix 2.)
The SF-12 provides 2 summary measures: physical and mental. The SF-12 was developed in the United States to provider a shorter alternative to the SF-36 measure. The SF-12 contains a subset of 12 items, including 1 or 2 items from each of the 8 SF-36 scales. The Oxford hip score, a disease-specific measure that was developed to assess the outcomes of hip replacement, is also used. This scale is a 12-item questionnaire, which assesses pain and function of the hip in relation to activities of daily living. (43)
Reported Harris hip, SF-12, Oxford hip, and University of California Los Angeles (UCLA) hip scores are shown in Table 9.
Table 9: Changes in Harris Hip Scores, SF-12 Scores, UCLA Hip Scores, and Other Scores*.
| Study | Survival Rate of the Implant/ Components % |
Change in Harris Hip Score |
Change in SF-12 Scores |
Change in Oxford Hip Score |
Change in UCLA Hip Score |
|---|---|---|---|---|---|
| De Smet 2005 (36) | NR | Preoperative: all < 60 At follow-up: 97.24 (41–100) |
NR | NR | NR |
| Back et al. 2005 (37) | NR | Charnley category A*: 63.9-97.7 Charnley category B: 56.2-99.4 Charnley category C: 64.8-85.5 (C vs. A or B, P < .001) |
SF-12 Physical Charnley category A: 31.1-54.1 Charnley category B: 30.3-54.1 Charnley category C: 31.5-48.2 SF-12 Mental Charnley category A: 58.6-56.9 Charnley category B: 60.5-57.7 Charnley category C: 52.2-55.9 |
NR | NR |
| Treacy et al. 2005 (38) | 98 (95% CI, 92-100) |
NR | NR | At follow-up only: Median 2.1, interquartile range: 0-10.4 | NR |
| Lilikakis et al. 2005 (39) | 97.1, SD 0.02 | Pain: 12 (0–30) to 39.3 (0–44) (P < .0001) Function: 28.3 (3–42) to 43.1 (9–47) (P < .0001) |
NR | NR | NR |
| Daniel et al. 2004 (35) | 99.7 | NR | NR | Mean score of surviving hips: 13.5 | UCLA Activity Level: All patients who responded (97.6%) had a score of 5 or more indicating an active life style |
| Amstutz et al. 2004 (34) | 94.4 (95% CI, 91-98) |
Overall postoperative: 93.5 (41–100) Charnley category A: 95.2 (61–100) Charnley category B 93.3 (66–100) (B vs. A P.008) Charnley category C 80.7 (41–100) C vs. A or B, P.001) (C vs. A or B, P < .001) |
SF-12 physical: 31.2 (16.8-54.8) to 50 (17.6-62.7) P < .0001 Mental: 46.8 (4–68.5) to 53.1 (10.5-67.1) P < .0001 |
NR | NR |
| Beaule et al. 2004 (40) | NR | NR |
Physical: 29.9-47.7 (P =. 00) Mental: 44.8-1.5 (P =. 00) |
NR |
UCLA hip score Pain: 3.1-9.1 Walking: 4.4-9.2 Function: 5.8-9.1 Activity: 5.5-7.1 (P =. 00) |
| Beaule et al. 2004 (41) | 79 (95% CI, 65-92) | NR | NR | NR |
UCLA hip score: Pain: 3.9-9.3 (P < .05) Walking: 6.2-9.2 Function: 5.8-8.5 Activity: 4.6-6.6 |
Charnley class A (affected one hip); Charnley class B and C (affected in both hips/other conditions directly impeding mobility); NR indicates not reported.
Four studies reported improvement in Harris hip scores. (34;36;37;39) However, only 1 study (39) reported a statistically significant improvement. Three studies reported improvement in SF-12 scores, (34;37;40) of which 2 reported significant improvement. (34;40) One study (40) reported significant improvement in UCLA hip score. Two studies (35;38) reported postoperative Oxford hip scores, but no preoperative values were reported.
Implant survival rate, number of deaths, and improvement in motion are summarized in Table 10.
None of the reviewed studies reported procedure-related deaths. Four studies reported implant survival rates (34;35;38;39) ranging from 94.4% to 99.7% for a follow-up period of 2.8 to 3.5 years. Three studies reported on the range of motion. (34;37;40) Beaule et al. (40) reported improvement in all motions including flexion, extension, abduction-adduction, and rotation, and Back et al. (37) reported improvement in flexion. Amstutz et al. (34) reported improvement in range of motion for flexion abduction-adduction and rotation arc. However, the author reported a decrease in the range of motion in the arc of flexion in patients with Brooker class III or IV heterotopic bone (all patients were men).
Only 2 studies reported length of hospital stay. (36;37) De Smet et al. (36) reported an average of 5 days and Back et al (37) reported a mean stay of 7.25 (range, 3–14) days in hospital.
Some authors have indicated that the quality of bone at the time of implantation is an important factor in success of hip resurfacing arthroplasty. The presence of a bone cyst may put the hip at risk of fracture or over time to the loosening. Appearance of radiolucencies during the follow-up is a cause for concern. Amstutz et al. (34) found radiolucency score of greater than or equal to 7 in 4.2% of the hips that had not been revised. The mean time to the first appearance of these radiolucencies occurred at 23 months (range, 10–50) postoperatively. However, the mean pain and activity scores of the group with a radiolucency score of greater than or equal to 7 were not significantly different from those of the rest of the cohort. The mean time to the first observation of the metaphyseal radiolucency and the mean time to the occurrence of the first symptoms was 20 months (range, 12.5—36.0) and 27 months (range, 16–51). The mean time to the revision was 35 months (range, 23–61). In 5 of the patients who had a revision to THR due to the loosening of the femoral component, large areas of cystic degeneration of the head and osseous defects were seen that diminished the surface area available for fixation.
Surface Arthroplasty Risk Index
The Chandler Index (44) has been used to identify patients at risk of failure following THR. Beaule et al. (40) have developed a surface arthroplasty risk index (SARI) for surface arthroplasty to optimize patient selection and to predict implant survivorship. With the SARI, higher scores indicate higher risk of failure. The initial Chandler index included 6 factors: absence of collagen disease, avascular necrosis, unilateral hip disease, previous surgery, weight, and activity level. One point was assigned to each factor. Beaule et al. (40) developed SARI through a multivariate logistic regression analysis. With the SARI, 2 points were given to femoral head cyst of greater than 1 cm; 2 points for a weight of less than 82 kg, 1 point for previous surgery, and 1 point for an activity level of greater than or equal to 7, according to the UCLA rating system.
The Chandler Index, does not assign points for a femoral cyst, whereas in SARI, 2 points were assigned to the femoral head cysts because femoral cysts have an impact on femoral fixation, which is different in hip resurfacing than in THR. In SARI, no point was assigned to the unilateral hip disease, as was the case in Chandler index. Unilateral hip disease is not considered in SARI because a significant correlation has been shown between unilateral hip disease and UCLA activity score greater than 6. Using this score, Beaule et al. (40) and Amstutz et al. (34) showed that increased height and reduced weight are risk factors for early failure. Beaule et al. showed that weight less than 82 kg was associated with early radiological changes, a smaller femoral component size, and a smaller fixation area. Several studies (45) also show that smaller femoral components survive less than the larger size components.
In the study by Beaule et al (40), 13 patients who had hip problems after hip resurfacing arthroplasty had a significantly higher SARI scores than the remaining patients (P = .001). Patients with SARI scores greater than 3 were 12 times more likely to have early problems than patients who had a SARI score of less than or equal to 3.
Amstutz et al. (34) reported that patients with metaphyseal stem radiolucencies had higher SARI scores (3.77 ± 1.45 points) compared with those who did not have these radiolucencies (2.66±1.53 points) (P = .0004). These authors also reported that SARI scores of greater than 3 points were significantly associated with an earlier time to revision (P = .004). The rate of survival of the implant at 4 years for patients with low SARI scores (≤ 3 points) was 97% (95% CI, 94–100) compared with 88.8% (95% CI, 80– 97.6) for patients with a high SARI score (>3 points). A Cox multivariate proportional hazard model showed that in women, large femoral cysts (P = .029), lesser height (P = .032), being female (P = .005), and SARI greater than 3 (P = .001) were significantly associated with stem radiolucencies and component loosening. In males, smaller component size was significantly associated with stem radiolucencies and component loosening (P = .005).
Other Factors Influencing the Success of Hip Resurfacing Arthroplasty
Varus placement of the femoral component has been implicated in femoral loosening. Amstutz et al. (34) found that the component stem-shaft angle in the hips that were revised because of femoral loosening was significantly lower (more varus) than that in the rest of the cohort (129° [range, 110°–148°] compared with 136° [range, 111° –163°] [P=.025]). De Smet et al. (36) have argued that long-term follow-up is needed to see whether varus placement has an adverse effect on the fixation and loosening of the head.
Lilikakis et al. (39) have indicated that neck thinning predisposes to femoral fracture and loosening in the future. Possible causes of neck thinning are avascular necrosis caused by femoral preparation at surgery and stress shielding owing to the alteration of the biomechanical parameters of the hip. (39)
De Smet et al. (36) have described surgical factors that could attribute to the development of femoral neck fracture. (36) This include notching of the neck during surgery, malpositioning of the central guide pin, varus positioning of the head component, incomplete coverage of the reamed bone, removing soft tissues around the femoral neck, removing too much bone from femoral neck, and incorrect impacting technique.
Amstutz et al. (34) reported the development of heterotopic bone in 36% of the male and 12% of the female patients. The authors also observed high rate of Brooker class III or IV in 10% of the male patients. The range of motion in the arc of flexion was significantly less in these patients compared with the rest of the male patients (P = .001).
Safety of the Metal-on-Metal Hip Resurfacing Arthroplasty
Concentration of Metal Ions in Body Fluids
There is a concern about metal wear debris and its systemic distribution throughout the body. Detectable metal concentrations in the serum and urine of patients with metal hip implants have been described as early as the 1970s, and this issue is still controversial after 35 years.
Several studies have reported high concentration of cobalt and chromium in serum and/or urine of the patients with metal hip implants. Potential toxicological effects of the elevated metal ions have heightened concerns about safety of MOM bearings. This is of particular concern in young and active patients in whom life expectancy after implantation is long.
Since 1997, 15 studies, (24;27;28;46-58) including 1 randomized clinical trial, (50) have reported high levels of metal ions after THR with metal implants. Some of these studies have reported higher metal levels in patients with loose implants.
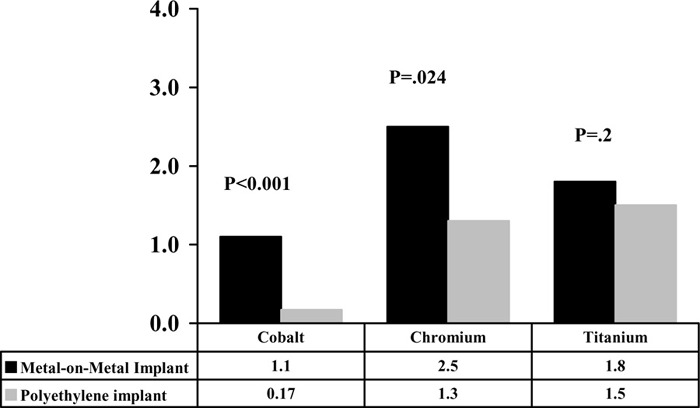
MacDonald et al. (50) conducted a prospective randomized, blinded clinical trial to evaluate the outcomes of MOM versus polyethylene surfaces in total hip arthroplasty. Forty-one patients were randomized to receive either a MOM (23 patients) or a polyethylene (18 patients) hip prostheses. Patients were assessed using a variety of outcome measures including the Western Ontario/McMaster University Score, Harris hip score, SF-12, as well as erythrocyte and urine analysis for metal ions (cobalt, chromium, and titanium). Patients were followed up for a minimum of 2 years (mean, 3.2 years; range, 2.2–3.9 years). This study showed no significant differences in radiographical or clinical outcome measures between the 2 groups. An important finding of this study was that patients who received a MOM implant had significantly higher levels of erythrocyte and urine metal ions than those who received a polyethylene implant. Intergroup comparisons showed no significant differences in the preoperative values for erythrocyte cobalt and chromium concentration. At 2-year evaluation, there was a significant difference in median erythrocyte cobalt and chromium concentrations between the 2 groups (P < .001 and P = .024 respectively). No significant differences were found in the median erythrocyte titanium concentrations in either preoperative or at 2-year follow-up between the 2 groups (Figure 5).
Figure 5: Erythrocyte Cobalt, Chromium, and Titanium Concentrations in Patients with Metal-on-Metal and Polyethylene Implants at 2-Year Follow-up.
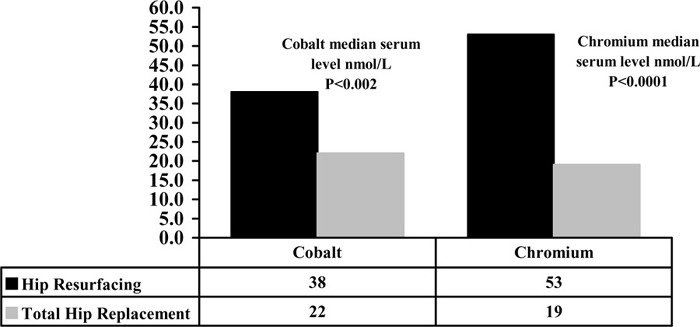
Two studies (59;60) reported on serum and urine levels of cobalt and chromium after MOM hip resurfacing arthroplasty. Clarke et al. (59) measured the serum levels of cobalt and chromium in 22 patients who had undergone MOM hip resurfacing arthroplasty and compared the results with a matched group of 22 patients (matched for date after surgery, activity level, and body mass) who had undergone MOM total hip arthroplasty. The patients in the resurfacing group were significantly younger by a mean of 7.9 years (P = .016), which reflects the indications for resurfacing in younger patients. Since the serum levels had not been determined before surgery, a conservative threshold level of 5 nmol/L for both cobalt and chromium was considered as normal value. This was based on the review of the literature on the values in normal individuals.
This study found the median cobalt level to be 7.6 times higher than normal after MOM hip resurfacing arthroplasty compared with levels 4.4 times higher than normal after MOM THR (P = .0021). The median chromium level was 10.6 times higher than normal after MOM hip resurfacing arthroplasty compared with levels 3.8 times higher than normal after MOM THR (P < .0001). (See Figure 6.)
Figure 6: Levels of Metal Ions After Metal-on-Metal Hip Resurfacing Arthroplasty and Total Hip Replacement (59).
Skipor et al. (60) measured the concentration of chromium in serum and urine and the concentration of cobalt in serum of 25 patients with modern MOM hip resurfacing arthroplasty. The results showed that after surgery, the mean serum chromium levels increased 22-fold, 23-fold, and 21-fold after 3, 6, and 12 months respectively compared with the preoperative values. The mean urine chromium levels increased 9-fold, 10-fold, and 14-fold at 3, 6, and 12 months postoperatively compared with the preoperative values. The mean serum cobalt increased 8-fold, 7-fold, and 6-fold compared with the preoperative values at 3, 6, and 12 months after surgery (Figure 7).
Figure 7: Serum and Urine Metal Concentrations in Patients With MOM Hip Resurfacing Arthroplasty (Mean ± 95% Confidence Interval).
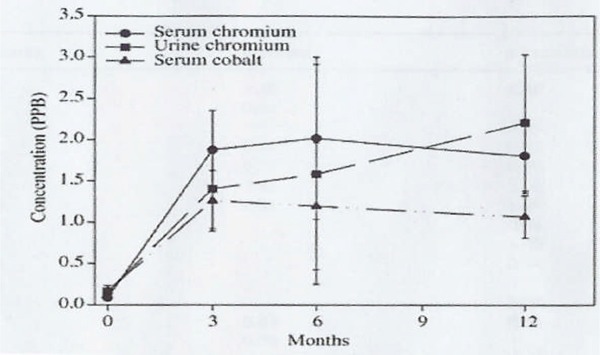
Reproduced with kind permission from Springer Science and Business Media, and the author; Source: Journal of Material Science; Materials in Medicine; 2002; 13(12):1227-1234; Skipor AK, Campbell PA, Patterson LM, Anstutz HC, Schmalzried TP, Jacobs JJ.. Serum and urine metal levels in patients with metal on metal surface arthroplast, Figure2
Adverse Biological Effects of Cobalt and Chromium
Because patients with MOM hip arthroplasties are shown to be exposed to high concentrations of metallic ions, the literature was searched for reports of adverse biological effects of cobalt and chromium.
Carcinogenicity
Cobalt and chromium make up the major part of the metal articulations and are therefore, a focus of concern. Cobalt is a cofactor in vitamin B12, and chromium is an essential cofactor in the interaction between insulin and its receptor. These 2 elements are toxic in high concentrations (57) Toxicological studies (61;62) examining occupational and environmental exposure have defined adverse biological effects to cobalt and chromium with long-term exposure to these elements. Chromium III (trivalent chromium) is less toxic than chromium VI (hexavalent chromium). Chromium VI has been shown to be carcinogenic in human and animal models and it was characterized as a class I human carcinogen by the International Agency for Research on Cancer (IARC). (63)
Below are the conclusions of IARC regarding orthopedic metal implants. (64) The IARC summary includes a variety of implant materials. However, only sections that were related to metal implants are shown below.
Evaluation Conducted by IARC (64)
“There is inadequate evidence in humans for the carcinogenicity of metallic implants and metallic foreign bodies.”
“There is inadequate evidence in humans for the carcinogenicity of orthopedic implants of complex composition and of cardiac pacemakers.”
“There is sufficient evidence in experimental animals for the carcinogenicity of implants of metallic cobalt, metallic nickel, and for nickel alloy powder containing about 66% to 67% nickel, 13% to 16% chromium, and 7% iron.”
“There is limited evidence in experimental animals for the carcinogenicity of implants of alloys containing cobalt and alloys containing nickel, other than the specific aforementioned alloy.”
“There is inadequate evidence in experimental animals for the carcinogenicity of implants of chromium metal, stainless steel, titanium metal, titanium-based alloys and depleted uranium.”
“There is inadequate evidence in dogs for the carcinogenicity of metallic implants and metallic and non-metallic foreign bodies.”
IARC’s overall evaluation of implanted foreign bodies was as follows: (64)
“Implanted foreign bodies of metallic cobalt, metallic nickel, and an alloy powder containing 66% to 67% nickel, 13% to 16% chromium, and 7% iron are possibly carcinogenic to humans.”
“Implanted foreign bodies of metallic chromium or titanium and of cobalt-based, chromium-based and titanium-based alloys, stainless steel, and depleted uranium are not classifiable as to their carcinogenicity to humans.”
The IARC has reported that “There is sufficient evidence in experimental animals for the carcinogenicity of implants of metallic cobalt, metallic nickel and for nickel alloy powder containing about 66% to 67% nickel, 13% to 16% chromium and 7% iron.”
As cited in the IARC monograph, the epidemiologic evidence for increased risk of cancers of the lung and sinonasal cavity is limited to conditions of high exposure, as encountered in the chromate production, chromate pigment production, and chromium plating industries. With regard to the mutagenicity and carcinogenicity of the metal alloys, the IARC has stated that the release of 1 type of metal ion can be strongly influenced by the identity of other metals in the alloy, and there is potential for the release of chemical species of known mutagenicity and carcinogenicity.
To date, only 1 study (65) examined the incidence of cancer after MOM (N = 698) and polyethylene on metal total hip arthroplasties (N = 1,831). The results were compared with cancer rates in the general population in Finland. The mean follow-up for MOM arthroplasty was 15.7 years and for polyethylene arthroplasty, it was 12.5 years. The standardized incidence ratio for all cancers of the MOM group was 0.95 (95% CI, 0.79–1.13) and for the polyethylene-on-metal group it was 0.76 (95% CI, 0.68–0.86). The combined standardized incidence ratio for lymphoma and leukemia in the patients who had MOM THR was 1.59 (95% CI, 0.82–2.77) and 0.59 (95% CI, 0.29–1.05) for the patients who had polyethylene on metal THR. Patients with MOM THR had significantly increased risk of leukemia. All patients who had leukemia were older than 60 years of age.
Another study (66) investigated the incidence of cancer in a large cohort of patients who had received THR between 1974 and 1988. Codes for primary THR, hemiarthroplasty, and revision hip arthroplasty were included, and 12,329 individuals were identified. In this study, the numbers of different types of prostheses implanted is not known, but as the authors have indicated, the original Charnley stainless-steel was the most commonly used prosthesis. John Charnley designed this implant in England in the 1960s. Charnley’s design used a one-piece metal stem with a 22-mm diameter head that was cemented into the proximal femur. The acetabular component was made entirely of polyethylene and cemented into the acetabulum. A cohort of 10,785 individuals (68.7% were women) who had had hip replacements was followed from the date of operation to the first malignant tumour, to death, or to the end of 1989. The following groups of patients were excluded from the analysis: patients who had revision arthroplasty at their first registration (n = 620), patients who had been treated for cancer before their first hip operation (n = 902), patients who died during the initial hospital admission (n = 9), 8 patients, mostly with juvenile arthritis who had multiple operations and who were 20 years or younger at the time of operation, and 5 patients without a complete personal identification. Therefore, a total follow-up of 58,437 person-years at risk, contributed by 10,785 patients, was available for the main analysis. The information was linked to the National Cancer Registry and the National Cause-of-Death Register. At the end of the study, 8, 408 people (78%) were alive; 2,377 (22%) had died. Person-years at risk was calculated for follow-up for less than 1 year, 1 to 2 years, 2 to 5 years, 5 to 10 years, and over 10 years.
Malignant tumours had developed in 881 patients (8.2%) during the observation period. The calculated number of expected cancers was 917.7; therefore, the standardized morbidity ratio was 0.96 (95% CI, 0.9 –1.03). Observed and expected malignancies at different points in the follow-up period from the date of primary hip replacement to the first cancer, to death, or to the end of 1989 are shown in Table 11.
Table 11: Observed and Expected Malignancies at Different Follow-ups Followed From the Date of Primary Hip Replacement to the First Cancer, to Death, or to the End of 1989.
| Cancer Site | Standardized Mortality Ratio (95% Confidence Interval) | ||
|---|---|---|---|
| < 1 year | 1 Year to < 2 years | By the Study End Point* |
|
| Upper gastrointestinal | 2.07 (1.10–3.54) | 0.59 (0.32–0.99) | 0.88 (0.68–1.11) |
| Colorectal | 1.94 (1.17–3.04) | 0.56 (0.35–0.86) | 0.95 (0.78–1.14) |
| Bile/liver/pancreas | 2.28 (1.27–3.75) | 0.44 (0.22–0.79) | 0.94 (0.74–1.17) |
| Lung | 1.75 (0.87–3.12) | 0.52 (0.27–0.90) | 0.76 (0.57–0.98) |
| Respiratory system, lung excluded | 3.72 (1.01–9.51) | 0.26 (0.01–1.43) | 0.98 (0.51–1.71) |
| Breast | 2.51 (1.64–3.68) | 0.37 (0.20–0.61) | 0.85 (0.69–1.03) |
| Female reproductive system | 1.30 (0.56–2.57) | 0.68 (0.38–1.13) | 1.17 (0.93–1.46) |
| Male reproductive system | 2.75 (1.68–4.25) | 0.32 (0.14–0.60) | 1.18 (0.97–1.42) |
| Kidney/urinary system | 0.65 (0.18–1.65) | 0.30 (0.12–0.63) | 0.84 (0.65–1.08) |
| Malignant melanoma/skin | 3.03 (1.51–5.42) | 0.49 (0.20–1.00) | 1.15 (0.87–1.50) |
| Eye/nervous system | 1.01 (0.12–3.66) | 0.83 (0.31–1.82) | 0.88 (0.54–1.36) |
| Thyroid/endocrine gland | 0.63 (0.02–3.50) | 0.35 (0.04–1.26) | 1.28 (0.81–1.92) |
| Bone/connective tissue | 2.09 (.05–11.64) | 0.00 (0.00–2.03) | 0.85 (0.28–1.98) |
| Other/unspecified | 0.99 (0.12–3.59) | 0.13 (0.00–0.71) | 1.08 (0.72–1.56) |
| Lymphoma/leukemia | 2.66 (1.49–4.39) | 0.42 (0.19–0.80) | 0.89 (0.68–1.14) |
| All sites | 2.02 (1.71–2.37) | 0.46 (0.38–0.54) | 0.96 (0.90–1.03) |
From the date of primary hip replacement to the first cancer, to death, or to the end of 1989
During the first year of observation, the incidence of several malignancies was higher than expected. The incidence of leukemia/lymphoma, respiratory system, lung, colorectal, upper gastrointestinal, bile/liver/pancreas, breast, female reproductive system, male reproductive system, malignant melanoma, and bone/connective tissue were significantly higher than expected. The incidence of all these cancers was reduced during the second year of observation. Seven out of 15 leukemia/lymphoma cases had been reported within first 30 days after discharge from the hospital, implying they may have been detected in routine testing before operation. The authors indicated that considering the long period of latency for exposure-associated cancers, the length of follow-up in this study is short to draw any conclusion about the association of between hip arthroplasty and an increase in the incidence of cancer.
Cobalt Cardiotoxicity
Epidemiological Studies of Myocardiopathy of Beer Drinkers
The Quebec Beer Drinkers’ Epidemic
An unusual type of myocardiopathy, characterized by pericardial effusion, elevated hemoglobin concentrations, and congestive heart failure, occurred as an epidemic affecting 48 habitual beer drinkers in Quebec City between 1965 and 1966. (67-80) This epidemic was directly related the consumption of a popular beer containing cobalt sulfate. The epidemic appeared 1 month after cobalt sulfate was added to the specific brewery, and no further cases were seen a month after this specific chemical was no longer used in making this beer. A beer of the same name is made in Montreal, and the only difference at that time was that the Quebec brand of beer contained about 10 times more cobalt sulphate. Cobalt has been added to some Canadian beers since 1965 to improve the stability of the foam but it has been added in larger breweries only to draught beer. However, in small breweries, such as those in Quebec City, separate batches were not brewed for bottle and draught beer; therefore, cobalt was added to all of the beer processed in this brewery.
In March 1966, a committee was appointed under the chairmanship of the Deputy Minister of Health for Quebec and included members of the department of forensic medicine of Quebec’s Ministry of Justice, epidemiologists, members of Food and Drug Directorate of Ottawa, toxicologists, biomedical researchers, pathologists, and members of provincial police. Epidemiological studies were carried out by the Provincial Ministry of Health and the Quebec City Health Department. Virological and immunological studies were negative. A search for toxic substances such as arsenic or lead was negative. The series of cases were typical of an epidemic. All cases occurred within a limited period of time and in the same region. Most of the patients lived in the Lower Town of Quebec City. Patients were in good health from 3 months up to 2 weeks before admission.
Shortness of breath was the most frequent symptom followed by abdominal pain, localized in the epigastrium. Other symptoms included orthopnea, cough, leg edema, asthenia, anorexia, nausea and vomiting, thoracic pain, hematemesis, peripheral pain, weight loss, melena, and diarrhea. The most frequent sign recorded was tachycardia, a gallop rhythm of the heart, tachypnea, cyanosis, hepatomegaly, jugular veins distension, and ankle edema. A significant increase in liver enzyme levels and hyperbilirubinemia was seen. On physical examination, the most remarkable signs were related to the cardiovascular system and progressive signs of heart failure were evident.
The incidence of this unusual myocardiopathy was directly related to the consumption of this particular brand of beer and to the amounts consumed daily and the duration of beer consumption. Higher incidence was associated with the duration of consumption and larger amounts of beer consumed (Figures 8 and 9). All these patients were unusually heavy beer drinkers, and 36 had been drinking heavily for at least 20 years. The mean alcoholic intake was 24 pints daily.
Figures 8 and 9: Distribution of Cases According to Daily Quantity of Beer Consumed and Duration of Consumption.
The association between the development of myocardiopathy and the consumption of the particular brand of beer was proven. The mortality rate of this epidemic was 46.1% and those who survived were desperately ill, and recovered only after a struggle for their lives.
The Omaha Beer Drinkers’ Epidemic
Similar cases were seen in Omaha (Nebraska). (81-83) The epidemic started after a cobalt additive was used in 1 of the beers marketed in Nebraska. Sixty-four patients with the clinical diagnosis of alcoholic myocardiopathy were seen during an 18-month period (1964–1965). Thirty of these patients died. The first patient became ill within 1 month after cobalt was added to the beer, and the last patient was seen within 1 month of withdrawal of cobalt.
Patients were presented with dyspnea, abdominal pain, and edema, all of relatively brief duration of 1 to 2 weeks. Blood pressure and pulse pressure were low, and tachycardia with regular rhythm and a protodiastolic gallop were present. Extreme cardiomegaly was universally present and a peripheral cyanosis was often noted. Venous distension and hepatic enlargement were present in some cases.
The investigators (84) measured the concentration of cobalt in the cardiac muscle of patients who died due to myocardiopathy associated with the consumption of beer containing cobaltous salt and compared with that of the patients who died from causes other than cardiac disease malnutrition or from those with a history of alcoholism. Results were also compared with the normal values reported by Tipton et al. (85) The significant finding of this study was the presence of cobalt in the heart muscles of patients who died of cobalt-associated myocardiopathy. This level was about 10 times greater than that found in the control tissue and normal values reported by Tipton et al. (mean concentration of cobalt in heart muscles was: 0.48 μg per gram in study cases; 0.04 μg per gram in study controls, and between 0.02 and 0.03 in Tipton’s controls.
The Minnesota Beer Drinkers’ Epidemic
Between 1964 and 1967, 42 patients with acute heart failure were admitted to a hospital in Minneapolis, Minnesota. (86;87) Twenty of these patients were drinking 6 to 30 bottles per day of a particular brand of beer exclusively. The other 14 patients also drank the same brand of beer, but not exclusively. The mortality rate from the acute illness was 18%, but late deaths accounted for a total mortality rate of 43%. Examination of the tissue from these patients revealed markedly abnormal changes in myofibrils (heart muscles), mitochondria, and sarcoplasmic reticulum.
The Belgium Beer Drinkers’ Epidemic
In Belgium, a similar epidemic was reported in 1966, (88) in which, cobalt was used in some Belgian beers. There was a difference in mortality between the Canadian or American epidemic and this series. Only 1 of 24 patients died, 1.5 years after the diagnosis. In March 1965, at an international meeting in Brussels, a new heart disease in chronic beer drinkers was described. This disease consists of massive pericardial effusion, low cardiac output, raised venous pressure, and polycythemia in some cases. This syndrome was thought to be different from the 2 other forms of alcoholic heart disease (beriberi and a form characterized by myocardial fibrosis).
The mystery of the above epidemics as stated by investigators is that the amount of cobalt added to the beer was below the therapeutic doses used for anemia. For example, 24 pints of Quebec brand of beer in Quebec would contain 8 mg of cobalt chloride, whereas an intake of 50 to 100 mg of cobalt as an antianemic agent has been well tolerated. (79) Thus, greater cobalt intake alone does not explain the occurrence of myocardiopathy. It seems that there are individual differences in cobalt toxicity. Other features, like subclinical alcoholic heart disease, deficient diet, and electrolyte imbalance could have been precipitating factors that made these patients susceptible to cobalt’s toxic effects. In the Omaha epidemic, 60% of the patients had weight loss, anorexia, and occasional vomiting and diarrhea 2 to 6 months before the onset of cardiac symptoms. (81) In the Quebec epidemic, patients lost their appetite 3 to 6 months before the diagnosis of myocardiopathy and developed nausea in the weeks before hospital admission. (76) In the Belgium epidemic, anorexia was one of the most predominant symptoms at the time of diagnosis, and the quality and quantity of food intake was poor. Alcohol has been shown to increase the uptake of intracoronary injected cobalt by 47%. (69) When cobalt enters the cells, calcium exits; this shifts the cobalt to calcium ratio. The increased uptake of cobalt in alcoholic patients may explain the high incidence of cardiomyopathies in beer drinkers’ epidemics. (89)
Experimental Studies
An animal model involving 120 male guinea pigs was used to reproduce the Quebec epidemic. (90) Guinea pigs were divided among 6 groups (groups received normal diet, cobalt or alcohol or both, cobalt and sucrose, or sucrose alone). Cardiac lesions of the pericardium, the myocardium, and the endocardium were produced in groups receiving 20 mg per kg per day of cobalt. Light and electron microscopic features and electrocardiographic findings were strikingly similar to those observed in Quebec beer drinkers’ myocardiopathy. The frequency and severity of the lesions were enhanced in those animals that were given alcohol plus cobalt. Given the fact that alcohol was given for a relatively short period, it is not expected that this agent have greatly modified the course of the disease in the guinea pigs.
Case Reports of Cobalt Cardiotoxicity
It has been suggested that cobalt myocardiopathy should be considered in the differential diagnosis of cardiac disease. Data from National Occupational Hazard Survey developed at National Institute of Occupational Safety and Health (1979–1987) show that white women and men listing occupations such as grinders, filers, fitters, polishers, buffers, and tool sharpeners in industries with potential cobalt exposure had a statistically significant increase in proportional mortality ratios for ischemic heart disease and acute myocardial infarction. (91) Nevada Public Health Authorities have confirmed 2 cases of myocardiopathy caused by occupational exposure to cobalt. (91)
A case report from Quebec (92) described a patient who had been exposed for 4 years to cobalt and developed cobalt myocardiopathy. Clinical features, short clinical course with a fatal outcome, clinical signs of cardiac insufficiency, and electrocardiographic findings resembled those of beer drinkers’ myocardiopathy. Postmortem chemical analysis showed massive accumulation of cobalt in tissues. Pathological findings also corresponded with the pathological findings in cases of beer drinkers. In this case, no other contributory factors were specified. There was no indication of inadequate diet or excessive alcohol usage. Because of the lack of any other causal factor, the case was presented as an example of cobalt myocardiopathy secondary to an industrial exposure to this element.
Another report from Australia (93) described a 48-year-old man who went into cardiac shock under anesthesia during an operation for a duodenal ulcer. He was considered a satisfactory patient for surgery, and no unusual problems were expected. Early in the operation, the patient became cyanosed and the pulse became impalpable. The patient developed physical signs of shock and hypoxia, and 12 days after operation, he died of cardiac failure. Subsequent investigations established that he had been exposed to cobalt in his work for 4 years, which involved handling powdered metal. A sample of myocardium showed presence of cobalt with the concentration of 7 μg/g.
There are other reports of occupational exposure to cobalt and development of cardiac disease. Reports from Russia (94) showed the development of myocardiopathy in workers who had occupational exposure to cobalt. From 4 cases reported by Vermel et al., (94) 3 also had alcoholism.
As all of the above suggest, it may be that prior chronic exposure to alcohol and/or a nutritionally deficient diet may have a marked synergistic effect with the cardiotoxicity of cobalt.
Conclusions
MOM hip resurfacing arthroplasty has been shown to be an effective arthroplasty procedure as tested in younger patients.
However, evidence for effectiveness is based only on 7 case series with short duration of follow-up (2.8–3.5 years). There are no RCTs or other well-controlled studies that compare MOM hip resurfacing with THR.
Revision rates reported in the MOM studies using implants currently licensed in Canada (hybrid systems, uncemented acetabular, and cemented femoral) range from 0.3% to 3.6% for a mean follow-up ranging from 2.8 to 3.5 years.
Fracture of femoral neck is not very common; it occurs in 0.4% to 2.2% of cases (as observed in a short follow-up period).
All the studies that measured health outcomes have reported improvement in Harris Hip and SF-12 scores; 1 study reported significant reduction in pain and improvement in function, and 2 studies reported significant improvement in SF-12 scores. One study reported significant improvement in UCLA Hip scores.
Concerns remain on the potential adverse effects of metal ions. Longer-term follow-up data will help to resolve the inconsistency of findings on adverse effects, including toxicity and carcinogenicity.
Economic Analysis
Results of Literature Review on Economics
Due to the extensive follow-up necessary for evaluation of MOM resurfacing arthroplasty, there is only 1 comprehensive cost-utility analysis, which was conducted by McKenzie et al. (95) in the United Kingdom in 2003. The study used Markov modeling with data from a systematic review of evidence to explore the use of MOM as the initial hip replacement procedure for patients with advanced hip disease. This was compared with watchful waiting followed by THR where watchful waiting included patient monitoring, drug-based treatments, and physiotherapy. The study found that MOM dominated the case of watchful waiting following by THR (Table 12), but long-term data were lacking to identify accurately the revision rates for MOM.
Table 12: Cost Effective Study on Metal on Metal Hip Replacement Arthroplasty.
| Cost effective study | Results |
| MOM hip resurfacing vs watchful waiting followed by THR | MOM dominates |
| MOM hip resurfacing vs THR | THR dominates |
Ontario-Based Economic Analysis
Notes & Disclaimer
The Medical Advisory Secretariat uses a standardized costing methodology for all of its economic analyses of technologies. The main cost categories and the associated methodology from the province’s perspective are as follows:
Hospital: Ontario Case Costing Initiative (OCCI) cost data is used for all program costs when there are 10 or more hospital separations, or one-third or more of hospital separations in the ministry’s data warehouse are for the designated International Classification of Diseases-10 diagnosis codes and Canadian Classification of Health Interventions procedure codes. Where appropriate, costs are adjusted for hospital-specific or peer-specific effects. In cases where the technology under review falls outside the hospitals that report to the OCCI, PAC-10 weights converted into monetary units are used. Adjustments may need to be made to ensure the relevant case mix group is reflective of the diagnosis and procedures under consideration. Due to the difficulties of estimating indirect costs in hospitals associated with a particular diagnosis or procedure, the Medical Advisory Secretariat normally defaults to considering direct treatment costs only. Historical costs have been adjusted upward by 3% per annum, representing a 5% inflation rate assumption less a 2% implicit expectation of efficiency gains by hospitals.
Non-Hospital: These include physician services costs obtained from the Provider Services Branch of the Ontario Ministry of Health and Long-Term Care, device costs from the perspective of local health care institutions, and drug costs from the Ontario Drug Benefit formulary list price.
Discounting: For all cost-effective analyses, discount rates of 5% and 3% are used as per the Canadian Coordinating Office for Health Technology Assessment and the Washington Panel of Cost-Effectiveness, respectively.
Downstream cost savings: All cost avoidance and cost savings are based on assumptions of utilization, care patterns, funding, and other factors. These may or may not be realized by the system or individual institutions. In cases where a deviation from this standard is used, an explanation has been given as to the reasons, the assumptions and the revised approach.
The economic analysis represents an estimate only, based on assumptions and costing methods that have been explicitly stated above. These estimates will change if different assumptions and costing methods are applied for the purpose of developing implementation plans for the technology.
Up-front Budget Impact Analysis
The device cost for MOM ranges from $4,300 to $6,000 (Cdn). Traditional hip replacement devices cost about $2,000 (Cdn). Using Ontario Case Costing Initiative data, the total estimated costs for hip resurfacing surgery including physician fees, device fees, follow-up consultation, and postsurgery rehabilitation is about $15,000 (Cdn).
Table 13: Estimated Costs of Hip Resurfacing Surgery.
| Hip Resurfacing Surgery (Based on 2005/06 inflationary-adjusted rates) | ||
|---|---|---|
| Weighted Average (Academic/Community) all-inclusive hospitalization cost of surgery | $ | 0,707.18 |
| Device Cost | $ | 5,400.00 |
| Additional Medication | $ | 330.75 |
| Follow-up Consultation | $ | 190.30 |
| Rehab (per outpatient CCAC case) | $ | 1,500.00 |
| OH P costs (average) | $ | 1,060.60 |
| Total Estimated Cost | $ | 15,256.83 |
MOM hip arthroplasty is generally recommended for patients aged under 55 years because its bone-conserving advantage enables patients to “buy time” and hence helps THRs to last over the lifetime of the patient. In 2004/2005, 15.9% of patients who received THRs were aged 55 years and younger. It is estimated that there are from 600 to 1,000 annual MOM hip arthroplasty surgeries in Canada with an estimated 100 to 150 surgeries in Ontario. Given the increased public awareness of this device, it is forecasted that demand for MOM hip arthroplasty will steadily increase with a conservative estimate of demand rising to 1,400 cases by 2010 (Figure 10). The net budget impact over a 5-year period could be $500,000 to $4.7 million, mainly because of the increasing cost of the device.
Figure 10: Projected Number of Metal-on-Metal Hip Arthroplasty Surgeries in Ontario: to 2010.
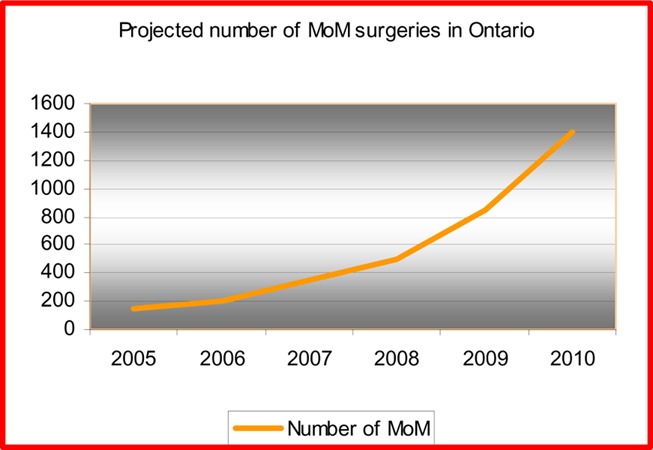
Appraisal/Policy Development
Policy Considerations
There is increasing interest among orthopedic surgeons and demand from patients for hip resurfacing arthroplasty. Young patients are generally active, hold jobs, and have a life expectancy that makes additional operations probable. Hip resurfacing arthroplasty is less invasive than THR and addresses the problem of preserving femoral bone stock at the initial operation. It has been claimed that MOM hip resurfacing arthroplasty is the ideal hip arthroplasty for younger patients because it has better bearing surfaces that enhance the implant survival compared to THR.
The following advantages have been claimed for MOM hip resurfacing arthroplasty:
It is less invasive and preserves bone stock.
The large femoral head reduces the chance of dislocation; therefore, the rate of dislocation is less than that for conventional THR.
The range of motion with hip resurfacing arthroplasty is higher than that achieved with conventional THR.
Revision of a hip resurfacing is much easier than revision of THR.
MOM articulation is wear resistant; therefore, it has lower risk of wear-induced osteolysis compared with the alternative materials.
It is advisable to delay the need for THR by treating the hip condition in younger patients with less invasive procedures. However, no RCTs have been conducted to compare MOM hip resurfacing arthroplasty with THR for lower rates of osteolysis, dislocation, osteonecrosis, and consequent revision.
Several criteria are important in assessing the clinical effectiveness of MOM hip arthroplasty. These include pain relief and improved function. However, the key criterion is the proportion of primary resurfacing arthroplasties that require revision surgery. The results of this assessment shows that revision rate of MOM hip resurfacing arthroplasty is 1.5% for a follow-up period ranging from 2.8 to 3.5 years. The incidence of femoral neck fracture and osteolysis were very low. However, this duration of follow-up is too short to reach a definite conclusion.
A number of factors influence the overall success of MOM hip resurfacing arthroplasty. These include the patient’s factors, surgeon’s skills, and postoperative care. The SARI is a guide, used by some surgeons, to appropriately select patients for this procedure. Surgeons’ experience and skills are critical to the success of this procedure. For example, inappropriate component stem-shaft angle and varus/valgus positioning may result in implant failure. Therefore, only orthopedic surgeons who have received training specifically for this procedure perform MOM hip resurfacing arthroplasty.
Patient Outcomes
Hip resurfacing arthroplasty is usually performed in patients under 65 years of age. However, 55 to 65 can be considered as “the gray zone” (personal communication). Several designs of hip resurfacing implants have been manufactured. The new generation of hip resurfacing implants have produced short-term and medium-term results that are much better than those of the earlier designs. The introduction of a hybrid system with a cementless acetabulum has led to a low incidence of early acetabular failure.
The diffusion of the technology requires ongoing collection of data on clinical effectiveness and revision, which can be used as a guide in the clinical decision making process.
The greatest concern about MOM hip arthroplasty is the generation and distribution of metal particles throughout the body fluids and tissues. Despite many toxicological studies about occupational or incidental exposure to environmental metal ions and long-standing recognition of the toxicity of these ions, human studies on endogenous exposure to these elements are rare. This issue needs further investigation to prevent adverse health effects of metal hip implants.
Measurement of Cobalt in the Blood
Periodic evaluation of metal ions in biological fluids and cells (e.g., whole blood, serum, erythrocytes, and urine samples) in patients after MOM hip arthroplasty to monitor the patient’s ion levels and compare them to the reference levels would be useful, but currently it is not practical because it is expensive.(expert opinion).
Issue of Hypersensitivity
The development of hypersensitivity reactions in patients with metal implants may require early intervention. The prevalence of hypersensitivity following insertion of metal implants appears to be low, but patients should be informed about the risk of becoming sensitive to the MOM implant.
Bioengineering Developments
It has been shown that the design and engineering parameters can affect the wear behaviour of MOM hip implants and therefore, control the concentration of ions levels in body fluids. The quality of the implants has clearly improved over time. Manufacturers are aiming to improve the bearing geometry, in particular an optimal head-cup clearance (i.e., a good fit between the head and the socket for the perfect positioning of the implant) and sphericity, as well as obtaining the best quality control to find a solution for wear problem (expert opinion).
Ethics
A clear discussion of the risks and benefits of MOM hip resurfacing arthroplasty must be presented to the patient when discussing the surgical options available. Issues such as elevated metal ion levels, specifically cobalt and chromium in the blood and urine of patients with metal bearings, must be discussed.
Existing Guidelines
The National Institute for Clinical Excellence (NICE) has issued guidance recommending the selective use of the technique. (96) NICE recommends that MOM hip resurfacing arthroplasty be considered as an option for people with advanced hip disease who would otherwise receive a conventional primary THR and who are likely to live longer than the device is likely to last.
Strength of the Body of Evidence
According to the GRADE system formulated by the GRADE working group, the score for the body of evidence on MOM hip resurfacing arthroplasty is low. (See Appendices 3 and 4.)
Glossary
- Clearance
A good fit between the head and the socket for a perfect positioning of the implant.
- Dislocation
Displacement of the bone.
- Heterotopic ossification
Unwanted bone growth around an implant that causes pain and reduces range of motion.
- Metaphyseal
Pertaining to the metaphysic (the extremity of a long bone).
- Mutagenicity
The property of being able to induce genetic mutation.
- Osteoarthritis
A noninflammatory degenerative joint disease seen mainly in older people or people with significant joint injury. It is the most common type of arthritis and is caused by “wear and tear” on the joint. The hip and knee joints are the most commonly affected.
- Osteolysis
Destruction of bony tissues; applied especially to the removal or loss of the calcium of bone.
- Osteonecrosis
Necrosis (death) of bone due to obstruction of its blood supply.
- Osteophyte
Unwanted bone growth.
- Osteoporosis
Reduction in the amount of bone mass, leading to fractures after minimal trauma.
- Radiolucency
Being radiolucent (permitting the passage of x-rays or other forms of radiant energy with little attenuation.
- Rheumatoid arthritis
A chronic, systemic disease that affects the lining of peripheral joints. It causes inflammatory responses, which destroy the articular cartilage and the tissues around the joints, causing joint deformity.
- Valgus
Bent or twisted outward.
- Varus
Bent or twisted inward.
Appendices
Appendix 1: Harris Hip Scores
| Pain | 44 |
None 44 Slight 40 Mild 30 Moderate 20 Marked 10 Disabled 0 |
|||||||||
| Function | 47 | Daily activities |
|
||||||||
| Gait |
|
||||||||||
| Range of motion | 5 |
|
|||||||||
| Absence of deformity | 4 |
|
|||||||||
| Total | 100 | ||||||||||
Appendix 2: SF-12 Scores
Significant correlation with other summary measure.
Items in boxes were selected for SF-12.
Appendix 3: GRADE Scores for the Body of Evidence on Metal-on-Metal Total Hip Resurfacing Arthroplasty*
| Number of Studies | Study Design | Quality of Studies | Consistency | Directness | Other Modifying Factors |
|---|---|---|---|---|---|
| N | RCT = high Observational = low Any other evidence = very low |
Serious limitation (-1) Very serious limitation (-2) |
Important inconsistency (-1) | Same uncertainty (-1) Major uncertainty (-2) |
|
Grade Working Group (97)
GRADE System Applied to the Studies.
| Number of Studies | Study Design | Quality of Studies | Consistency | Directness | Other Modifying Factors | Overall Quality of Evidence |
|---|---|---|---|---|---|---|
| 8 | Case series = low | High | Yes | Yes | Not applicable | Low |
Appendix 4: GRADE System for Recommendations of the American College of Chest Physicians*
| Grade of Recommendation | Benefit Versus Risk and Burdens | Methodological Strength of Supporting Evidence | Implications |
|---|---|---|---|
| Strong recommendation, High quality evidence 1 A |
Benefits clearly outweigh risk and burdens, or vice versa | RCTs without important limitations or overwhelming evidence from observational studies | Strong recommendation, can apply to most patients in most circumstances without reservation |
| Strong recommendation, Moderate quality evidence 1 B |
Benefits clearly outweigh risk and burdens, or vice versa | RCTs with important limitations (inconsistent results, methodological flaws, indirect, or imprecise) or exceptionally strong evidence from observational studies | |
| Strong recommendation, Low or very low quality evidence 1 C |
Benefits clearly outweigh risk and burdens, or vice versa | Observational studies or case series | Strong recommendation but may change when higher quality evidence becomes available |
| Weak recommendation, High quality evidence 2 A |
Benefits closely balanced with risks and burden | RCTs without important limitations or overwhelming evidence from observational studies | Weak recommendation, best action may differ depending on circumstances or patients’ or societal values |
| Weak recommendation, Moderate quality evidence 2 B |
Benefits closely balanced with risks and burden | RCTs with important limitations (inconsistent results, methodological flaws, indirect, or imprecise) or exceptionally strong evidence from observational studies | |
| Weak recommendation, Low or very low quality evidence 2 C |
Uncertainty in the estimates of benefits, risks, and burden; benefits, risk and burden may be closely balanced | Observational studies or case series | Very weak recommendations; other alternatives may be equally reasonable |
Developed by a task force convened in 2005: Guyatt G, Gutterman D, Baumann MH. Grading Strength of Recommendations and Quality of Evidence in Clinical Guidelines. Report from an American College of Chest Physicians Task Force. (98)
Suggested Citation
This report should be cited as follows:
Medical Advisory Secretariat. Metal-on-metal total hip resurfacing arthroplasty: an evidence-based analysis. Ontario Health Technology Assessment Series 2006; 6(4)
Permission Requests
All inquiries regarding permission to reproduce any content in the Ontario Health Technology Assessment Series should be directed to MASinfo@moh.gov.on.ca
How to Obtain Issues in the Ontario Health Technology Assessment Series
All reports in the Ontario Health Technology Assessment Series are freely available in PDF format at the following URL: www.health.gov.on.ca/ohtas
Print copies can be obtained by contacting MASinfo@moh.gov.on.ca
Conflict of Interest Statement
All analyses in the Ontario Health Technology Assessment Series are impartial and subject to a systematic evidence-based assessment process. There are no competing interests or conflicts of interest to declare.
Peer Review
All Medical Advisory Secretariat analyses are subject to external expert peer review. Additionally, the public consultation process is also available to individuals wishing to comment on an analysis prior to finalization. For more information, please visit http://www.health.gov.on.ca/english/providers/program/ohtac/public_engage_overview.html
Contact Information
The Medical Advisory Secretariat
Ministry of Health and Long-Term Care
20 Dundas Street West, 10th floor
Toronto, Ontario
CANADA
M5G 2N6
Email: MASinfo@moh.gov.on.ca
Telephone: 416-314-1092
ISSN 1915-7398
ISBN 1-4249-1970-3
About the Medical Advisory Secretariat
The Medical Advisory Secretariat is part of the Ontario Ministry of Health and Long-Term Care. The mandate of the Medical Advisory Secretariat is to provide evidence-based policy advice on the coordinated uptake of health services and new health technologies in Ontario to the Ministry of Health and Long-Term Care and to the healthcare system. The aim is to ensure that residents of Ontario have access to the best available new health technologies that will improve patient outcomes.
The Medical Advisory Secretariat also provides a secretariat function and evidence-based health technology policy analysis for review by the Ontario Health Technology Advisory Committee (OHTAC).
The Medical Advisory Secretariat conducts systematic reviews of scientific evidence and consultations with experts in the health care services community to produce the Ontario Health Technology Assessment Series.
About the Ontario Health Technology Assessment Series
To conduct its comprehensive analyses, the Medical Advisory Secretariat systematically reviews available scientific literature, collaborates with partners across relevant government branches, and consults with clinical and other external experts and manufacturers, and solicits any necessary advice to gather information. The Medical Advisory Secretariat makes every effort to ensure that all relevant research, nationally and internationally, is included in the systematic literature reviews conducted.
The information gathered is the foundation of the evidence to determine if a technology is effective and safe for use in a particular clinical population or setting. Information is collected to understand how a new technology fits within current practice and treatment alternatives. Details of the technology’s diffusion into current practice and information from practicing medical experts and industry, adds important information to the review of the provision and delivery of the health technology in Ontario. Information concerning the health benefits; economic and human resources; and ethical, regulatory, social and legal issues relating to the technology assist policy makers to make timely and relevant decisions to maximize patient outcomes.
If you are aware of any current additional evidence to inform an existing Evidence-Based Analysis, please contact the Medical Advisory Secretariat: MASInfo@moh.gov.on.ca. The public consultation process is also available to individuals wishing to comment on an analysis prior to publication. For more information, please visit http://www.health.gov.on.ca/english/providers/program/ohtac/public_engage_overview.html
Disclaimer
This evidence-based analysis was prepared by the Medical Advisory Secretariat, Ontario Ministry of Health and Long-Term Care, for the Ontario Health Technology Advisory Committee and developed from analysis, interpretation and comparison of scientific research and/or technology assessments conducted by other organizations. It also incorporates, when available, Ontario data, and information provided by experts and applicants to the Medical Advisory Secretariat to inform the analysis. While every effort has been made to do so, this document may not fully reflect all scientific research available. Additionally, other relevant scientific findings may have been reported since completion of the review. This evidence-based analysis is current to the date of publication. This analysis may be superceded by an updated publication on the same topic. Please check the Medical Advisory Secretariat Website for a list of all evidence-based analyses: http://www.health.gov.on.ca/ohtas
Abbreviations
- IARC
International Agency for Research on Cancer
- CI
Confidence interval
- MOM
Metal-on-metal
- OA
Osteoarthritis
- NR
Not reported
- RA
Rheumatoid arthritis
- SARI
Surface arthroplasty risk index
- THR
Total hip replacement
References
- 1.Roberts J, Meek RM, Roberts P, Grigoris P. Metal-on-metal hip resurfacing. Scott Med J. 2005;50(1):10–12. doi: 10.1177/003693300505000103. [DOI] [PubMed] [Google Scholar]
- 2.Mont MA. Michael Mont on metal-on-metal hip resurfacing arthroplasty. Orthopedics. 2004;27(10):1047–1048. doi: 10.3928/0147-7447-20041001-12. [DOI] [PubMed] [Google Scholar]
- 3.Cooper C, Cook PL, Osmond C, Fisher L, Cawley MI. Osteoarthritis of the hip and osteoporosis of the proximal femur. Ann Rheum Dis. 1991;50(8):540–542. doi: 10.1136/ard.50.8.540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Foss MV, Byers PD. Bone density, osteoarthrosis of the hip and fracture of the upper end of the femur. Ann Rheum Dis. 1972;31(4):259–264. doi: 10.1136/ard.31.4.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jordan GR, Loveridge N, Bell KL, Power J, Dickson GR, Vedi S, et al. Increased femoral neck cancellous bone and connectivity in coxarthrosis (hip osteoarthritis) Bone. 2003;32(1):86–95. doi: 10.1016/s8756-3282(02)00920-1. [DOI] [PubMed] [Google Scholar]
- 6.Mont MA, Hungerford DS. Non-traumatic avascular necrosis of the femoral head. J Bone Joint Surg Am. 1995;77(3):459–474. doi: 10.2106/00004623-199503000-00018. [DOI] [PubMed] [Google Scholar]
- 7.Mont MA, Rajadhyaksha AD, Hungerford DS. Outcomes of limited femoral resurfacing arthroplasty compared with total hip arthroplasty for osteonecrosis of the femoral head. J Arthroplasty. 2001;16(8 SUPPL. 1):134–139. doi: 10.1054/arth.2001.28722. [DOI] [PubMed] [Google Scholar]
- 8.Dorr LD, Takei GK, Conaty JP. Total hip arthroplasties in patients less than forty-five years old. J Bone Joint Surg Am. 1983;65(4):474–479. [PubMed] [Google Scholar]
- 9.Cornell CN, Salvati EA, Pellicci PM. Long-term follow-up of total hip replacement in patients with osteonecrosis. Orthop Clin North Am. 1985;16(4):757–769. [PubMed] [Google Scholar]
- 10.Hungerford MW, Mont MA, Scott R, Fiore C, Hungerford DS, Krackow KA. Surface replacement hemiarthroplasty for the treatment of osteonecrosis of the femoral head. J Bone Joint Surg Am. 1998;80(11):1656–1664. doi: 10.2106/00004623-199811000-00013. [DOI] [PubMed] [Google Scholar]
- 11.Krackow KA, Mont MA, Maar DC. Limited femoral endoprosthesis for avascular necrosis of the femoral head. Orthop Rev. 1993;22(4):457–463. [PubMed] [Google Scholar]
- 12.Schmalzried TP. Metal-on-metal resurfacing arthroplasty: no way under the sun!--in opposition. J Arthroplasty. 2005;20(Suppl 2):70–71. doi: 10.1016/j.arth.2005.03.007. [DOI] [PubMed] [Google Scholar]
- 13.Dorr LD, Wan Z, Longjohn DB, Dubois B, Murken R. Total hip arthroplasty with use of the Metasul metal-on-metal articulation. J Bone Joint Surg Am. 2000;82(6):789–798. doi: 10.2106/00004623-200006000-00005. Four to seven-year results. [DOI] [PubMed] [Google Scholar]
- 14.Schmalzried TP, Peters PC, Maurer BT, Bragdon CR, Harris WH. Long-duration metal-on-metal total hip arthroplasties with low wear of the articulating surfaces. J Arthroplasty. 1996;11(3):322–331. doi: 10.1016/s0883-5403(96)80085-4. [DOI] [PubMed] [Google Scholar]
- 15.Northmore-Ball MD, Bannister GC, Mears DC, Durbhakular S. Clinical challenges in orthopedics: the hip. London: Martin Dunitz Ltd. 2002.
- 16.Berry DJ, von Knoch M, Schleck CD, Harmsen WS. Effect of femoral head diameter and operative approach on risk of dislocation after primary total hip arthroplasty. J Bone Joint Surg Am. 2005;87(11):2456–2463. doi: 10.2106/JBJS.D.02860. [DOI] [PubMed] [Google Scholar]
- 17.Huo MH, Muller MS. What’s new in hip arthroplasty. J Bone Joint Surg Am. 2004;86-A(10):2341–2353. doi: 10.2106/00004623-200410000-00033. [DOI] [PubMed] [Google Scholar]
- 18.Little CP, Ruiz AL, Harding IJ, McLardy-Smith P, Gundle R, Murray DW, et al. Osteonecrosis in retrieved femoral heads after failed resurfacing arthroplasty of the hip. J Bone Joint Surg Br. 2005;87(3):320–323. doi: 10.1302/0301-620x.87b3.15330. [DOI] [PubMed] [Google Scholar]
- 19.Steffen RT, Smith SR, Urban JP, McLardy-Smith P, Beard DJ, Gill HS, et al. The effect of hip resurfacing on oxygen concentration in the femoral head. J Bone Joint Surg Br. 2005;87(11):1468–1474. doi: 10.1302/0301-620X.87B11.16780. [DOI] [PubMed] [Google Scholar]
- 20.Amstutz HC, Campbell PA, Le Duff MJ. Fracture of the neck of the femur after surface arthroplasty of the hip. J Bone Joint Surg Am. 2004;86(9):1874–1877. doi: 10.2106/00004623-200409000-00003. [DOI] [PubMed] [Google Scholar]
- 21.MacDonald SJ. Metal-on-metal total hip arthroplasty: the concerns. Clin Orthop Relat Res. 2004;429:86–93. doi: 10.1097/01.blo.0000150309.48474.8b. [DOI] [PubMed] [Google Scholar]
- 22.MacDonald SJ, Brodner W, Jacobs JJ. A consensus paper on metal ions in metal-on-metal hip arthroplasties. J Arthroplasty. 2004;19(8 SUPPL):12–16. doi: 10.1016/j.arth.2004.09.009. [DOI] [PubMed] [Google Scholar]
- 23.MacDonald SJ. Can a safe level for metal ions in patients with metal-on-metal total hip arthroplasties be determined? J Arthroplasty. 2004;8(Suppl 3):71–77. doi: 10.1016/j.arth.2004.09.008. [DOI] [PubMed] [Google Scholar]
- 24.Brodner W, Bitzan P, Meisinger V, Kaider A, GottsaunerWolf F, Kotz R. Serum cobalt levels after metal-on-metal total hip arthroplasty. J Bone Joint Surg Am. 2003;85(11):2168–2173. doi: 10.2106/00004623-200311000-00017. [DOI] [PubMed] [Google Scholar]
- 25.Silva M, Heisel C, Schmalzried TP. Metal-on-metal total hip replacement. Clin Orthop Relat Res. 2005;430:53–61. doi: 10.1097/01.blo.0000149995.84350.d7. [DOI] [PubMed] [Google Scholar]
- 26.Willert HG, Buchhorn GH, Fayyazi A, Flury R, Windler M, Koster G, et al. Metal-on-metal bearings and hypersensitivity in patients with artificial hip joints. [A clinical and histomorphological study]. J Bone Joint Surg Am. 2005;87(1):28–36. doi: 10.2106/JBJS.A.02039pp. [DOI] [PubMed] [Google Scholar]
- 27.Hallab NJ, Anderson S, Caicedo M, Skipor A, Campbell P, Jacobs JJ. Immune responses correlate with serum-metal in metal-on-metal hip arthroplasty. J Arthroplasty. 2004;19(8 SUPPL):88–93. doi: 10.1016/j.arth.2004.09.012. [DOI] [PubMed] [Google Scholar]
- 28.Lhotka C, Szekeres T, Steffan I, Zhuber K, Zweymuller K. Four-year study of cobalt and chromium blood levels in patients managed with two different metal-on-metal total hip replacements. J Orthop Res. 2003;21(2):189–195. doi: 10.1016/S0736-0266(02)00152-3. [DOI] [PubMed] [Google Scholar]
- 29.Goldberg VM. Surface replacement solutions for the arthritic hip. Orthopedics. 2005;28(9):943–4. doi: 10.3928/0147-7447-20050901-18. [DOI] [PubMed] [Google Scholar]
- 30.Amstutz HC, Grigoris P, Dorey FJ. Evolution and future of surface replacement of the hip. J Orthop Sci. 1998;3(3):169–186. doi: 10.1007/s007760050038. [DOI] [PubMed] [Google Scholar]
- 31.Charnley J. Arthroplasty of the hip. Lancet. 1961;1(7187):1129–1132. doi: 10.1016/s0140-6736(61)92063-3. A new operation. [DOI] [PubMed] [Google Scholar]
- 32.Rieker CB, Schon R, Konrad R, Liebentritt G, Gnepf P, Shen M, et al. Influence of the clearance on in-vitro tribology of large diameter metal-on-metal articulations pertaining to resurfacing hip implants. Orthop Clin North Am. 2005;36(2):135–142, vii. doi: 10.1016/j.ocl.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 33.Howie DW, McGee MA, Costi K, Graves SE. Metal-on-metal resurfacing versus total hip replacement - The value of a randomized clinical trial. Orthop Clin North Am. 2005;36(2):195–201. doi: 10.1016/j.ocl.2004.12.001. [DOI] [PubMed] [Google Scholar]
- 34.Amstutz HC, Beaule PE, Dorey FJ, Le Duff MJ, Campbell PA, Gruen TA. Metal-on-metal hybrid surface arthroplasty: two to six-year follow-up study. J Bone Joint Surg Am. 2004;86-A(1):28–39. [PubMed] [Google Scholar]
- 35.Daniel J, Pynsent PB, McMinn DJ. Metal-on-metal resurfacing of the hip in patients under the age of 55 years with osteoarthritis. J Bone Joint Surg Br. 2004;86(2):177–184. doi: 10.1302/0301-620x.86b2.14600. [DOI] [PubMed] [Google Scholar]
- 36.DeSmet KA. Belgium experience with metal-on-metal surface arthroplasty. Orthop Clin North Am. 2005;36(2):203–213. doi: 10.1016/j.ocl.2005.01.004. [DOI] [PubMed] [Google Scholar]
- 37.Back DL, Dalziel R, Young D, Shimmin A. Early results of primary Birmingham hip resurfacings. J Bone Joint Surg Br. 2005;87(3):324–329. doi: 10.1302/0301-620x.87b3.15556. An independent prospective study of the first 230 hips. [DOI] [PubMed] [Google Scholar]
- 38.Treacy RB, McBryde CW, Pynsent PB. Birmingham hip resurfacing arthroplasty. [A minimum follow-up of five years]. J Bone Joint Surg Br. 2005;87(2):167–170. doi: 10.1302/0301-620x.87b2.15030. [DOI] [PubMed] [Google Scholar]
- 39.Lilikakis AK, Vowler SL, Villar RN. Hydroxyapatite-coated femoral implant in metal-on-metal resurfacing hip arthroplasty: minimum of two years follow-up. Orthop Clin North Am. 2005;36(2):215–222. doi: 10.1016/j.ocl.2004.12.003. [DOI] [PubMed] [Google Scholar]
- 40.Beaule PE, Dorey FJ, LeDuff M, Gruen T, Amstutz HC. Risk factors affecting outcome of metal-on-metal surface arthroplasty of the hip. Clin Orthop Relat Res. 2004;418:87–93. [PubMed] [Google Scholar]
- 41.Beaule PE, LeDuff M, Campbell P, Dorey FJ, Park SH, Amstutz HC. Metal-on-metal surface arthroplasty with a cemented femoral component: a 7-10 year follow-up study. J Arthroplasty. 2004;19(8 Suppl):17–22. doi: 10.1016/j.arth.2004.09.004. [DOI] [PubMed] [Google Scholar]
- 42.Harris WH. Traumatic arthritis of the hip after dislocation and acetabular fractures: treatment by mold arthroplasty. [An end-result study using a new method of result evaluation]. J Bone Joint Surg Am. 1969;51(4):737–755. [PubMed] [Google Scholar]
- 43.Dawson J, Fitzpatrick R, Carr A, Murray D. Questionnaire on the perceptions of patients about total hip replacement. J Bone Joint Surg Br. 1996;78(2):185–190. [PubMed] [Google Scholar]
- 44.Chandler HP, Reineck FT, Wixson RL, McCarthy JC. Total hip replacement in patients younger than 30 years old. J Bone Joint surg. 1981;63 A:1426–1434. [PubMed] [Google Scholar]
- 45.Mai MT, Schmalzried TP, Dorey FJ, Campbell PA, Amstutz HC. The contribution of frictional torque to loosening at the cement-bone interface in Tharies hip replacements. J Bone Joint Surg Am. 1996;78(4):505–511. doi: 10.2106/00004623-199604000-00004. [DOI] [PubMed] [Google Scholar]
- 46.Dunstan E, Sanghrajka AP, Tilley S, Unwin P, Blunn G, Cannon SR, et al. Metal ion levels after metal-on-metal proximal femoral replacements: a 30-year follow-up. J Bone Joint Surg Br. 2005;87(5):628–631. doi: 10.1302/0301-620X.87B5.15384. [DOI] [PubMed] [Google Scholar]
- 47.Milosev I, Pisot V, Campbell P. Serum levels of cobalt and chromium in patients with Sikomet metal-metal total hip replacements. J Orthop Res. 2005;23(3):526–535. doi: 10.1016/j.orthres.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 48.Brodner W, Grubl A, Jankovsky R, Meisinger V, Lehr S, GottsaunerWolf F. Cup inclination and serum concentration of cobalt and chromium after metal-on-metal total hip arthroplasty. J Arthroplasty. 2004;19(8 SUPPL):66–70. doi: 10.1016/j.arth.2004.09.003. [DOI] [PubMed] [Google Scholar]
- 49.Ladon D, Doherty A, Newson R, Turner J, Bhamra M, Case CP. Changes in metal levels and chromosome aberrations in the peripheral blood of patients after metal-on-metal hip arthroplasty. J Arthroplasty. 2004;19(8 SUPPL):78–83. doi: 10.1016/j.arth.2004.09.010. [DOI] [PubMed] [Google Scholar]
- 50.MacDonald SJ, McCalden RW, Chess DG, Bourne RB, Rorabeck CH, Cleland D, et al. Metal-on-metal versus polyethylene in hip arthroplasty: a randomized clinical trial. Clin Orthop Relat Res. 2003. pp. 282–296. [DOI] [PubMed]
- 51.Adami G, Smarrelli D, Martinelli B, Acquavita A, Reisenhofer E. Cobalt blood levels after total hip replacement (THR): a new follow-up study in Trieste (Italy) Ann Chim. 2003;93(1-2):1–10. [PubMed] [Google Scholar]
- 52.Masse A, Bosetti M, Buratti C, Visentin O, Bergadano D, Cannas M. Ion release and chromosomal damage from total hip prostheses with metal-on-metal articulation. J Biomed Mater Res B Appl Biomater. 2003;67(2):750–757. doi: 10.1002/jbm.b.10070. [DOI] [PubMed] [Google Scholar]
- 53.Savarino L, Granchi D, Ciapetti G, Cenni E, Greco M, Rotini R, et al. Ion release in stable hip arthroplasties using metal-on-metal articulating surfaces: a comparison between short- and medium-term results. J Biomed Mater Res A. 2003;66(3):450–456. doi: 10.1002/jbm.a.10595. [DOI] [PubMed] [Google Scholar]
- 54.Maezawa K, Nozawa M, Hirose T, Matsuda K, Yasuma M, Shitoto K, et al. Cobalt and chromium concentrations in patients with metal-on-metal and other cementless total hip arthroplasty. Arch Orthop Trauma Surg. 2002;122(5):283–287. doi: 10.1007/s00402-001-0382-3. [DOI] [PubMed] [Google Scholar]
- 55.Savarino L, Granchi D, Ciapetti G, Cenni E, Nardi PA, Rotini R, et al. Ion release in patients with metal-on-metal hip bearings in total joint replacement: a comparison with metal-on-polyethylene bearings. J Biomed Mater Res. 2002;63(5):467–474. doi: 10.1002/jbm.10299. [DOI] [PubMed] [Google Scholar]
- 56.Schaffer AW, Pilger A, Engelhardt C, Zweymueller K, Ruediger HW. Increased blood cobalt and chromium after total hip replacement. J Toxicol Clin Toxicol. 1999;37(7):839–844. doi: 10.1081/clt-100102463. [DOI] [PubMed] [Google Scholar]
- 57.Jacobs JJ, Skipor AK, Patterson LM, Hallab NJ, Paprosky WG, Black J, et al. Metal release in patients who have had a primary total hip arthroplasty. [A prospective, controlled, longitudinal study]. J Bone Joint Surg Am. 1998;80(10):1447–1458. doi: 10.2106/00004623-199810000-00006. [DOI] [PubMed] [Google Scholar]
- 58.Brodner W. Elevated serum cobalt with metal-on-metal articulating surfaces [published erratum appears in J Bone Joint Surg Br 1997 Sep;79(5):885] J Bone Joint Surg Br. 1997;79(2):316–321. doi: 10.1302/0301-620x.79b2.7326. [DOI] [PubMed] [Google Scholar]
- 59.Clarke MT, Lee PT, Arora A, Villar RN. Levels of metal ions after small- and large-diameter metal-on-metal hip arthroplasty. J Bone Joint Surg Br. 2003;85(6):913–917. [PubMed] [Google Scholar]
- 60.Skipor AK, Campbell PA, Patterson LM, Anstutz HC, Schmalzried TP, Jacobs JJ. Serum and urine metal levels in patients with metal-on-metal surface arthroplasty. J Mater Sci Mater Med. 2002;13(12):1227–1234. doi: 10.1023/a:1021179029640. [DOI] [PubMed] [Google Scholar]
- 61.Horowitz SF, Matza D, Machac J. Cardiotoxic effects of chemicals. Mt Sinai J Med. 1985;52(8):650–655. [PubMed] [Google Scholar]
- 62.Kurppa K, Hietanen E, Klockars M, Partinen M, Rantanen J, Ronnemaa T, et al. Chemical exposures at work and cardiovascular morbidity. [Atherosclerosis, ischemic heart disease, hypertension, cardiomyopathy and arrhythmias]. Scand J Work Environ Health. 1984;10(6 Spec No):381–388. doi: 10.5271/sjweh.2316. [DOI] [PubMed] [Google Scholar]
- 63.International Agency for Research on Cancer (IARC) [[cited 2006 June 30]];Chromium, nickel, and welding. Summary of data reported and evaluation. [report on the Internet] IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. 1997 Nov;49 Available at: http://monographs.iarc.fr/ENG/Monographs/vol49/volume49.pdf . [Google Scholar]
- 64.International Agency for Research on Cancer (IARC) Surgical implants and other foreign bodies [Web page] 1999. [[cited 2006 May 15]]. Available at: http://www.inchem.org/documents/iarc/vol74/implants.html .
- 65.Visuri T, Pukkala E, Paavolainen P, Pulkkinen P, Riska EB. Cancer risk after metal on metal and polyethylene on metal total hip arthroplasty. Clin Orthop Relat Res. 1996. pp. S280–S289. [DOI] [PubMed]
- 66.Mathiesen EB, Ahlbom A, Bermann G, Lindgren JU. Total hip replacement and cancer. [A cohort study]. J Bone Joint Surg Br. 1995;77(3):345–350. [PubMed] [Google Scholar]
- 67.Morin YL, Foley AR, Martineau G, Roussel J. Quebec beer-drinkers’ cardiomyopathy: forty-eight cases. Can Med Assoc J. 1967;97(15):881–883. [PMC free article] [PubMed] [Google Scholar]
- 68.Morin Y. Quebec beer-drinkers’ cardiomyopathy: hemodynamic alterations. Can Med Assoc J. 1967;97(15):901–904. [PMC free article] [PubMed] [Google Scholar]
- 69.Morin Y, Daniel P. Quebec beer-drinkers’ cardiomyopathy: etiological considerations. Can Med Assoc J. 1967;97(15):926–928. [PMC free article] [PubMed] [Google Scholar]
- 70.Mercier G, Patry G. Quebec beer-drinkers’ cardiomyopathy: clinical signs and symptoms. Can Med Assoc J. 1967;97(15):884–888. [PMC free article] [PubMed] [Google Scholar]
- 71.Nadeau G, Milon H. Quebec beer-drinkers’ cardiomyopathy: biochemical studies. Can Med Assoc J. 1967;97(15):889–893. [PMC free article] [PubMed] [Google Scholar]
- 72.Tetu A, Samson M. Quebec beer-drinkers’ cardiomyopathy: electrocardiographic study. Can Med Assoc J. 1967;97(15):893–900. [PMC free article] [PubMed] [Google Scholar]
- 73.Bonenfant JL, Miller G, Roy PE. Quebec beer-drinkers’ cardiomyopathy: pathological studies. Can Med Assoc J. 1967;97(15):910–916. [PMC free article] [PubMed] [Google Scholar]
- 74.Sargent AU, Rose B. Quebec beer-drinkers’ cardiomyopathy: immunochemical studies. Can Med Assoc J. 1967;97(15):922–923. [PMC free article] [PubMed] [Google Scholar]
- 75.Lapointe A. Quebec beer-drinkers’ cardiomyopathy: radiological aspects. Can Med Assoc J. 1967;97(15):905–909. [PMC free article] [PubMed] [Google Scholar]
- 76.Taskar PK, Senecal N. Quebec beer-drinkers’ cardiomyopathy: dietary assessment of patients. Can Med Assoc J. 1967;97(15):924–925. [PMC free article] [PubMed] [Google Scholar]
- 77.The mystery of the Quebec beer-drinkers’ cardiomyopathy. Can Med Assoc J. 1967;97(15):930–931. [PMC free article] [PubMed] [Google Scholar]
- 78.Roy PE, Bonenfant JL, Turcot L. Thyroid changes in cases of Quebec beer drinkers myocardosis. Am J Clin Pathol. 1968;50(2):234–239. doi: 10.1093/ajcp/50.2.234. [DOI] [PubMed] [Google Scholar]
- 79.Morin Y, Tetu A, Mercier G. Quebec beer-drinkers’ cardiomyopathy: clinical and hemodynamic aspects. Ann N Y Acad Sci. 1969;156(1):566–576. doi: 10.1111/j.1749-6632.1969.tb16751.x. [DOI] [PubMed] [Google Scholar]
- 80.Bonenfant JL, Auger C, Miller G, Chenard J, Roy PE. Quebec beer-drinkers’ myocardosis: pathological aspects. Ann N Y Acad Sci. 1969;156(1):577–582. doi: 10.1111/j.1749-6632.1969.tb16752.x. [DOI] [PubMed] [Google Scholar]
- 81.Sullivan JF, Egan JD, George RP. A distinctive myocardiopathy occurring in Omaha, Nebraska: clinical aspects. Ann N Y Acad Sci. 1969;156(1):526–543. doi: 10.1111/j.1749-6632.1969.tb16749.x. [DOI] [PubMed] [Google Scholar]
- 82.Sullivan JF, George R, Bluvas R, Egan JD. Myocardiopathy of beer drinkers: subsequent course. Ann Intern Med. 1969;70(2):277–282. doi: 10.7326/0003-4819-70-2-277. [DOI] [PubMed] [Google Scholar]
- 83.McDermott PH, Delaney RL, Egan JD, Sullivan JF. Myocardosis and cardiac failure in men. JAMA. 1966;198(3):253–256. [PubMed] [Google Scholar]
- 84.Sullivan J, Parker M, Carson SB. Tissue cobalt content in “beer drinkers’ myocardiopathy”. J Lab Clin Med. 1968;71(6):893–911. [PubMed] [Google Scholar]
- 85.Tipton IH, Cook MJ. Trace elements in human tissue. Health Phys. 2006;1963;9(103) doi: 10.1097/00004032-196302000-00002. [DOI] [PubMed] [Google Scholar]
- 86.Alexander CS. Cobalt-beer cardiomyopathy. [A clinical and pathologic study of twenty-eight cases]. Am J Med. 1972;53(4):395–417. doi: 10.1016/0002-9343(72)90136-2. [DOI] [PubMed] [Google Scholar]
- 87.Alexander CS. Nutritional heart disease. Cardiovasc Clin. 1972;4(1):221–244. [PubMed] [Google Scholar]
- 88.Kesteloot H, Terryn R, Bosmans P, Joossens JV. Alcoholic perimyocardiopathy. Acta Cardiol. 1966;21(3):341–357. [PubMed] [Google Scholar]
- 89.Morin Y, Cote G. Toxic agents and cardiomyopathies. Cardiovasc Clin. 1972;4(1):245–267. [PubMed] [Google Scholar]
- 90.Mohiuddin SM, Taskar PK, Rheault M, Roy PE, Chenard J, Morin Y. Experimental cobalt cardiomyopathy. Am Heart J. 1970;80(4):532–543. doi: 10.1016/0002-8703(70)90202-4. [DOI] [PubMed] [Google Scholar]
- 91.Jarvis JQ, Hammond E, Meier R, Robinson C Cobalt cardiomyopathy. A report of two cases from mineral assay laboratories and a review of the literature. J Occup Med. 1992;34(6):620–626. [PubMed] [Google Scholar]
- 92.Barborik M, Dusek J. Cardiomyopathy accompaning industrial cobalt exposure. Br Heart J. 1972;34(1):113–116. doi: 10.1136/hrt.34.1.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kennedy A, Dornan JD, King R. Fatal myocardial disease associated with industrial exposure to cobalt. Lancet. 1981;1(8217):412–414. doi: 10.1016/s0140-6736(81)91793-1. [DOI] [PubMed] [Google Scholar]
- 94.Vermel’ AE, Nikitina LS, Barabanov AA, Dasaeva LA, Druzhinin VI, Ivanova LA, et al. Cobalt-induced cardiomyopathy in workers engaged in the manufacture of hard alloys. Ter Arkh. 1991;63(4):101–104. [PubMed] [Google Scholar]
- 95.McKenzie L, Vale L, Stearns S, McCormack K. Metal on metal hip resurfacing arthroplasty. [An economic analysis]. Eur J Health Econ. 2003;4(2):122–129. doi: 10.1007/s10198-002-0158-x. [DOI] [PubMed] [Google Scholar]
- 96.National Institute for Clinical Excellence. Guidance on the use of metal on metal hip resurfacing arthroplasty [report on the Internet] No. 44. London: National Institute for Clinical Excellence (NICE); Jun, 2002. [[cited 2006 June 4]]. Available at: http://www.nice.org.uk/page.aspx?o=TA044guidance . [Google Scholar]
- 97.GRADE Working Group. Grading quality of evidence and strength of recommendations. BMJ. 2004;328:1–8. doi: 10.1136/bmj.328.7454.1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Guyatt G GDBM. Grading strength of recommendations and quality of evidence in clinical guidelines. Report from an American College of Chest Physicians Task Force. 2006;129:174–181. doi: 10.1378/chest.129.1.174. Chest. [DOI] [PubMed] [Google Scholar]


