Abstract
The type III secretion system (T3SS) is an important virulence factor of pathogenic bacteria, but the natural occurrence of variants of bacterial plant pathogens with deficiencies in their T3SS raises questions about the significance of the T3SS for fitness. Previous work on T3SS-deficient plant pathogenic bacteria has focused on strains from plants or plant debris. Here we have characterized T3SS-deficient strains of Pseudomonas syringae from plant and nonplant substrates in pristine nonagricultural contexts, many of which represent recently described clades not yet found associated with crop plants. Strains incapable of inducing a hypersensitive reaction (HR−) in tobacco were detected in 65% of 126 samples from headwaters of rivers (mountain creeks and lakes), snowpack, epilithic biofilms, wild plants and leaf litter and constituted 2 to 100% of the P. syringae population associated with each sample. All HR− strains lacked at least one gene in the canonical hrp/hrc locus or the associated conserved effector locus, but most lacked all six of the genes tested (hrcC, hrpL, hrpK1, avrE1 and hrpW1) and represented several disparate phylogenetic clades. Although most HR− strains were incapable of causing symptoms on cantaloupe seedlings as expected, strains in the recently described TA-002 clade caused severe symptoms in spite of the absence of any of the six conserved genes of the canonical T3SS according to PCR and Southern blot assays. The phylogenetic context of the T3SS variants we observed provides insight into the evolutionary history of P. syringae as a pathogen and as an environmental saprophyte.
Keywords: hypersensitive reaction, aquatic microbiology, environmental microbiology, evolutionary ecology, fitness, adaptation
Introduction
The type III secretion system (T3SS) is believed to confer a major fitness advantage to a wide range of Gram-negative bacteria that are pathogens of plants or animals. By delivering effectors that suppress the defense mechanisms of hosts (Hueck, 1998; Cornelis and Van Gijsegem, 2000; Block and Alfano, 2011), it contributes to creating more favorable conditions for bacterial multiplication in the host. The functions and evolutionary origins of this secretion system have been intensively explored because of its critical role in pathogenicity (Troisfontaines and Cornelis, 2005). Three complementary analytical approaches are needed to attain a full understanding of the significance of the T3SS for bacterial fitness, pathogenicity and evolution. These approaches are as follows: mutational analyses of model strains and population genetics and comparative genomics of strains representative of pathogen diversity. For plant pathogenic bacteria, the latter two have been hindered by the research focus on highly virulent crop pathogens and a concomitant neglect of the pathogen diversity present on wild plants and in the environment in general. This neglect of pathogen diversity is partly due to the narrow species descriptions of plant pathogenic bacteria, which include the ability to induce a hypersensitive response (HR) on tobacco (see for example the LOPAT test used to identify Pseudomonas syringae, (Lelliot et al., 1966)). This narrow species description automatically excludes plant pathogen species members that might be missing a T3SS as induction of an HR is an indicator of a functional T3SS (He, 1996). Strains deficient in their capacity to induce HR (HR−) have recently been described for Erwinia pyrifoliae (Jock et al., 2003), Pectobacterium sp. (Kim et al., 2009) and P. syringae (Mohr et al., 2008; Clarke et al., 2010; Kniskern et al., 2011). All strains examined in these studies were isolated from plants (wild or cultivated) or from leaf litter in cultivated fields. The HR− strains of Pectobacterium represent two species, P. carotovorum and P. wasabiae. But the P. syringae strains are remarkably homogenous. All HR− strains of P. syringae characterized to date represent a single phylogenetic group and consistently lack the canonical hrp/hrc cluster (Mohr et al., 2008; Clarke et al., 2010; Kniskern et al., 2011). Furthermore, these strains harbor a novel hrp/hrc cluster with limited effectiveness in expressing and/or delivering effectors to plant cells. HR− strains of E. pyrifoliae and P. syringae were reported to be incapable of causing disease on the hosts tested, whereas the HR− strains of Pectobacterium spp. maintained pathogenicity on the original host from which they were isolated. The considerable role of pectolytic enzymes in the pathogenicity of Pectobacterium spp. might explain why there was no loss in pathogenicity for this group of bacteria.
Recent work on the ecology of P. syringae in nonagricultural habitats revealed the existence of nine new clades of this bacterium that had not been previously observed (Morris et al., 2010). All phylogenetic descriptions of this species before the study of nonagricultural habitats focused on strains from plants. The extensive phylogenetic diversity observed in nonagricultural habitats could also be a source of genetic diversity of the T3SS and of pathotypes. Strains in these clades are clearly apt at surviving in a wide range of habitats, such as snowpack, water and epilithic biofilms. The key traits for adaptation to these specific habitats, and for enabling many strains of P. syringae to be fit in multiple habitats including plant tissues, have not been identified. In particular, the role that the T3SS has in this adaptation has not been explored. Hence, we have explored the frequency of occurrence and the genetic diversity of strains of P. syringae, from nonagricultural habitats, that are HR− in tobacco. This work reveals that HR− strains are frequent in nonagricultural habitats and have diverse phylogenetic origins. Furthermore—and surprisingly—their incapacity to induce hypersensitivity is not necessarily indicative of their incapacity to cause disease on plants. This work provides a unique comparative foundation for identifying and studying the genetic factors that underlie evolutionary adaptation and ecological specialization of P. syringae.
Materials and methods
Origin of strains
A total of 63 strains of P. syringae were deployed in this study, including 40 strains unable to incite a hypersensitive reaction (HR−) and 23 strains capable of inciting a hypersensitive reaction (HR+) obtained from 21 samples of water, snowpack, epilithic biofilms and wild plants as described in the Supplementary Table 1. Six reference strains were also included. Samples were part of a survey for hypersensitive incompetent strains that included a range of environmental substrates collected from 2004 to 2010, mostly in alpine regions and outside of agricultural fields (Table 1) and processed as described previously (Morris et al., 2007, 2008, 2010) to determine the density of P. syringae populations. Phenotypic traits that confirm identity as P. syringae were determined as described previously (absence of arginine dihydrolase and cytochrome oxidase). The 40 strains of P. syringae unable to incite a hypersensitive reaction used for this study were selected from that survey to represent the range of sample sites, substrates and phylogenetic diversity. Reference strains included the HR− strains Psy508 and Psy642 (Mohr et al., 2008; Clarke et al., 2010) and the HR+ strains B728A (from the stock culture of S Hirano, University of Wisconsin, Madison, WI, USA), DC3000 and 1448A from J Greenberg (The University of Chicago), and Cit7 from SE Lindow (University of California, Berkeley, CA, USA). All strains were stored at −80 °C in 20% glycerol, and all phenotypic and molecular tests were conducted on cultures prepared from a single transfer of the storage cultures.
Table 1. Characteristics of environmental samples, from alpine regions and outside of cropped fields, used in survey for populations of Pseudomonas syringae incapable of inducing a hypersensitive reaction in tobacco (HR−).
| Substrate Region | n° of samplesa | n° of HR− samplesb | Size range of the total P. syringae population (log c.f.u. g−1and log c.f.u. l−1)c | Previous description of sample or sampling site | |
|---|---|---|---|---|---|
| Epilithic biofilm | |||||
| France, Alps | 1 | 1 | 4.9 | (Morris et al., 2007) | |
| France, southeast | 3 | 0 | 1.70–2.43 | (Morris et al., 2007) | |
| USA, Western Rocky mountains | 6 | 1 | 1.56–3.76 | (Morris et al., 2007) | |
| Headwater | |||||
| France, Alps | 37 | 34 | 1.47–5.8 | (Morris et al., 2008; Morris et al., 2010) | |
| France, southwest | 2 | 1 | 2.65–3.03 | (Morris et al., 2010) | |
| New Zealand | 1 | 1 | 3.45 | (Morris et al., 2010) | |
| USA, Western Rocky mountains | 3 | 1 | 2.69 | (Morris et al., 2010) | |
| Wild plants | |||||
| France–Italy border, Alps | 10 | 6 | 2.68–9.00 | (Morris et al., 2008) | |
| France, southwest | 2 | 1 | 3.85–5.73 | (Morris et al., 2008) | |
| Alpine leaf litter | |||||
| France, Alps | 11 | 8 | 2.89–6.45 | ||
| Raind | |||||
| France, Alps | 1 | 1 | 4.23 | ||
| France, southeast | 5 | 1 | 1.00–3.05 | (Morris et al., 2008) | |
| Snow pack | |||||
| Austrian Alps | 1 | 0 | 2 | ||
| France, Alps | 35 | 25 | 1.20–6.95 | (Morris et al., 2008) | |
| Morocco, Atlas mountains | 1 | 1 | 4.72 | ||
| Snow fall | |||||
| France, Alps | 6 | 0 | 2.18–5.11 | (Morris et al., 2008) | |
| USA, Western Rocky mountains | 1 | 0 | 1.83 | ||
Number of total samples of each substrate that were analyzed and in which P. syringae was detected.
Number of samples of each substrate in which HR− strains of P. syringae were detected. When fewer than 20 strains of P. syringae were isolated from the sample, all were tested for capacity to induce hypersensitivity. Otherwise, a random set of about 30 strains was tested per sample.
The range of sizes of populations of P. syringae, expressed as c.f.u. g−1 (plants, leaf litter and epilithic biofilms) or c.f.u. l−1 (rain, headwaters and meltwater from snow) observed in the different substrates.
Some rain samples were collected in peri-urban zones, but outside of cropped fields.
Hypersensitivity, pathogenicity and growth dynamics of bacteria in plants
For hypersensitivity tests, aqueous bacterial suspensions at 108 colony-forming units (c.f.u.) ml−1 from 48 h cultures on King's medium B (King et al., 1954) were infiltrated with a needleless syringe into the leaves of tobacco (Nicotiana tabacum L. cv. Xanthi). Hypersensitivity was scored as negative if necrotic patches did not develop in the infiltrated zone within 48 h after infiltration. Assays were repeated three times and included an HR+ strain as a control.
The capacity of strains to induce disease symptoms on cantaloupe seedlings was determined for strains, regardless of their capacity to induce hypersensitivity on tobacco. Aggressiveness on cantaloupe plants (Cucumis melo var. cantalupensis Naud. cv. Védrantais) was quantified by injecting 50 μl of bacterial suspensions (prepared as for hypersensitivity tests) into the junction of the cotyledons of 12 plants at the cotyledon stage (ca. 10 days after sowing) as described previously (Morris et al., 2010). Plants were incubated for 7 days at 25 °C during the daylight period (16 h) and 18 °C at night (8 h), and symptoms were observed daily after inoculation. Final disease severity was noted at 7 days after inoculation on a scale of 0–4: 0 (no symptoms), 1 (1 cotyledon wilted or discolored), 2 (both cotyledons wilted or discolored), 3 (both cotyledons completely wilted and the upper stem of the plant collapsed) and 4 (the plant collapsed at the base and the entire plant had fallen on the soil surface).
To determine population dynamics of strains in cantaloupe plants at the cotyledon stage, plants were inoculated and incubated in the same way as for pathogenicity tests. At each sampling time for up to 7 days after inoculation, 3–6 plants were randomly sampled from the 12 plants inoculated with each strain, weighed, and individually macerated in 10 ml of 0.1 M phosphate buffer. Aliquots of the macerate were diluted and plated on KBC medium (Mohan and Schaad, 1987), the same medium used to isolate these strains from the environment. Colonies were counted after 4 days of incubation at 22–25 °C in the dark. All colonies were fluorescent under ultraviolet light as expected and there were no indications of contamination of the plants with other bacteria.
PCR detection of genes in the hrp/hrc cluster
PCR detection was conducted on DNA purified from 48 h cultures on KB medium from strains stored at −80 °C as described above. Bacterial suspensions (1012 c.f.u. ml−1) prepared in sterile distilled water were used for DNA extraction with a Puregene DNA purification system cell and tissue kit (Gentra Systems, Minneapolis, MN, USA) following the manufacturer's instructions.
For detection of hrp/hrc cluster genes (hrpK1, hrpL, hrcC), CEL effectors (avrE1, hrpW1) and hopI1, primers described by Mohr et al. (2008) were used for amplification, whereas for phage detection we designed forward primer PhF (5′-TCAGGCCGCTTTCAACTTGGC-3′) and reverse primer PhR (5′-CCATAYGGCGCAGTGGTGG- 3′) based on the sequence of a putative transcriptional regulator gene (locus tag ABX64468) located within the prophage at the hrp/hrc locus of Psy508 (Mohr et al., 2008). To detect the presence of the hrcC ortholog of Psy642, colony PCR was performed as previously described (Clarke et al., 2010). Amplification conditions for detection of these eight genes are described in detail in the Supplementary Information section.
Southern blotting
To validate the results of PCR reactions, Southern blot analyses were performed for a set of strains representing the different profiles of absence and presence of the six conserved genes as described previously (Vinatzer et al., 2006; Mohr et al., 2008). Genomic DNA of P. syringae pv. syringae B728a, P. syringae 508, and hypersensitive incompetent P. syringae UB15, UB185, UB186, UB190, UB192, UB193, UB195, UB246, UB313, SZ30, SZ37, SZ45, SZ124, SZ131 and TA043 was digested with HindIII and EcoRI (New England Biolabs, Frankfurt, Germany). Digested DNA was electrophoresed on a 1% Tris-borate-EDTA agarose gel. Southern blotting, hybridization and detection was performed following the manufacturer's instructions (Roche Diagnostics, Indianapolis, IN, USA) using a mixture of digoxigenin-labeled PCR products of the six genes amplified from strain B728a and the digoxigenin-labeled PCR product of the amplification of a housekeeping gene (gapA) from strain B728a as probes.
Gene sequencing and phylogenetic analysis
From previous studies (Mohr et al., 2008; Clarke et al., 2010; Morris et al., 2010), the clade identity was known for 19 strains used here relative to the phylogenetic tree reported by Morris et al. (2010), and based on four housekeeping genes (rpoD, gyrB, cts and gapA). For the remaining strains, assignment to clades was based on our previous results that strains could be accurately assigned to clades based on DNA similarity in the cts sequence using a threshold dissimilarity of 1.8% (Morris et al., 2010). The sequence of the cts gene was determined as previously described (Morris et al., 2010). On the basis of the aligned and truncated (ca. 400 bp) cts sequences (DAMBE version 5.1.1 (Xia and Xie, 2001)), the genetic distance (dissimilarity) of each strain from each of the other 121 strains of P. syringae that constitute our previously reported phylogenetic tree (Morris et al., 2010) was determined using MEGA, version 3.1 (Kumar et al., 1994). There were no cases in which strains had ambiguous similarities (that is, there were no cases in which the similarity criterion was met for more than one clade).
If the similarity criterion was not met, the other three housekeeping genes (rpoD, gyrB and gapA) were sequenced. Details of PCR amplifications are described in the Supplementary Information section. A phylogenetic tree was constructed with the concatenated four housekeeping gene sequences from these strains and those from the ensemble of P. syringae, P. viridiflava and P. fluorescens used in a previous work (Morris et al., 2010). All strains fell into previously described clades.
Sequences of the PCR-amplified bacteriophage gene fragment were aligned with DAMBE version 5.1.1 and truncated to 302 bp.
Results
HR− strains are abundant in nonagricultural habitats and can dominate the P. syringae population in some cases
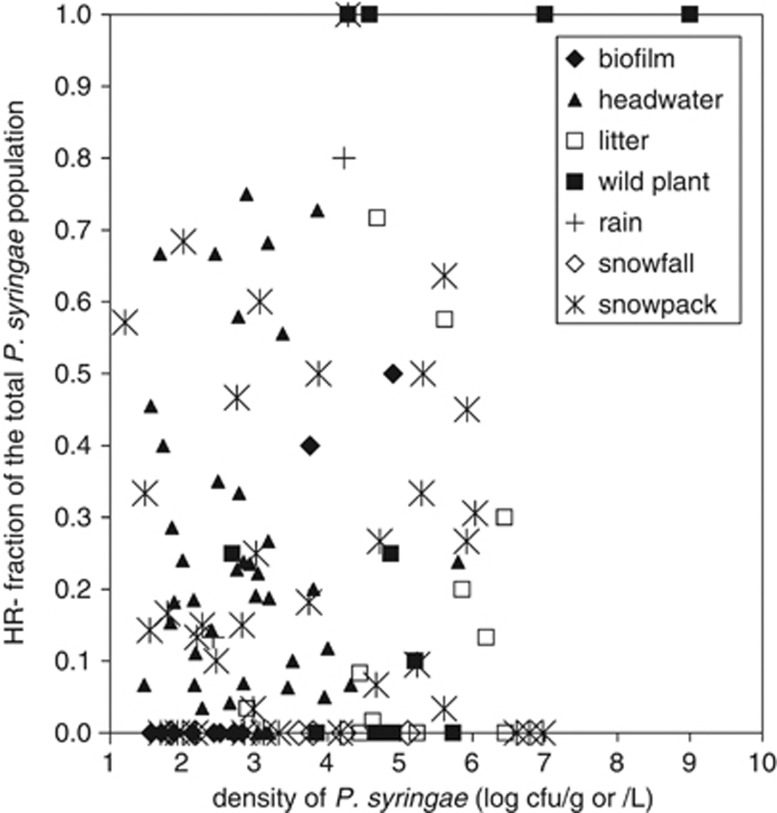
HR− strains were detected in 65% of the 126 environmental samples analyzed (Table 1) and were found in headwaters of rivers, snowpack, epilithic biofilms, wild plants and leaf litter. HR− strains were also found in some samples of freshly fallen rain but not in any of the samples of fresh snowfall. When detected, the HR− fraction of the total P. syringae population varied greatly, from 2 to 100%, and HR− strains attained population densities of up to 105 c.f.u. l−1 of river water or 109 c.f.u. g−1 of plant tissue. In 20% of the samples, HR− strains constituted 50% or more of the total P. syringae population (Figure 1). The frequency of HR− strains in the total P. syringae population was not related to the P. syringae population density (Figure 1). For the samples analyzed here, P. syringae population density did not exceed 107 c.f.u. g−1 or c.f.u. l−1, except in one case. Strains from some of these samples were further characterized for the presence of components of the T3SS and for their pathogenicity to cantaloupe seedlings (Table 2).
Figure 1.
Fraction of HR− strains in the total P. syringae populations in environmental samples in terms of the density of the P. syringae population in the sample. Population densities are expressed as log c.f.u. g−1 for biofilms, plant and leaf litter or log c.f.u. l−1 for water, rain and snow (as snowmelt). The data constitute values from 100 of the 126 samples described in Table 1. For these 100 samples, they were processed to permit quantification of the density of the HR− component population as well as that of the total P. syringae population.
Table 2. Genetic and pathogenic diversity of strains of Pseudomonas syringae according to the phylogenetic context of the strains. The presence or absence of genes was based on PCR detection.
| Cladea | Strainb | HRc |
Conserved genes in the canonical T3SS |
Psy642 hrcCf | Phage | gapA | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Disease on cantaloupe seedlings |
hopl1 |
CEL effectors |
hrp/hrc Cluster |
||||||||||
| Severityd | Incidencee | avrE | hrpW | hrpK | hrpL | hrcC | |||||||
| SZ-030 | Psy508g | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + |
| SZ-030 | − | 0.08 | 0.08 | − | − | − | − | − | − | + | + | + | |
| SZ-037 | − | 0.08 | 0.08 | − | − | − | − | − | − | + | + | + | |
| SZ-045 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| SZ-051 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| SZ-124 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| SZ-142 | − | 0.17 | 0.17 | − | − | − | − | − | − | + | + | + | |
| SZ-144 | − | 0.08 | 0.08 | − | − | − | − | − | − | + | + | + | |
| UB-015 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| UB-185 | − | 0.08 | 0.08 | − | − | − | − | − | − | + | + | + | |
| UB-186 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| UB-190 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| UB-192 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| UB-193 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| UB-194 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| UB-195 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| UB-313 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | + | + | |
| UB-330 | − | ndh | nd | − | − | − | − | − | − | + | + | + | |
| Psy-642g | − | 0.00 | 0.00 | − | − | − | − | − | − | + | − | + | |
| SZ-093 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | − | + | |
| UB-184 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | − | + | |
| UB-218 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | − | + | |
| CC1504 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | − | + | |
| USA-052 | − | 0.00 | 0.00 | − | + | + | + | − | − | + | − | + | |
| TA-002 | TA-043 | − | 1.67 | 1.00 | − | − | − | − | − | − | − | + | + |
| CCE-284 | − | 2.17 | 0.83 | − | − | − | − | − | − | − | − | + | |
| CCE-286 | − | 1.75 | 0.75 | − | − | − | − | − | − | − | − | + | |
| CCE-291 | − | 1.33 | 0.75 | − | − | − | − | − | − | − | − | + | |
| CCE-321 | − | 0.17 | 0.42 | − | − | − | − | − | − | − | − | + | |
| CCE-324 | − | 0.50 | 0.50 | − | − | − | − | − | − | − | − | + | |
| CCV-0050 | − | 1.17 | 1.00 | − | − | − | − | − | − | − | − | + | |
| CCV-0172 | − | 0.17 | 0.17 | − | − | − | − | − | − | − | − | + | |
| CCV-0178 | − | 2.91 | 1.00 | − | − | − | − | − | − | − | − | + | |
| CCV-0180 | − | 2.50 | 1.00 | − | − | − | − | − | − | − | − | + | |
| CSZ-0284 | − | 1.25 | 0.67 | − | − | − | − | − | − | − | − | + | |
| CSZ-0297 | − | 2.08 | 0.67 | − | − | − | − | − | − | − | − | + | |
| CC-1582 | − | 2.58 | 1.00 | − | − | − | − | − | + | − | − | + | |
| CC-0655 | + | 0.50 | 0.42 | − | + | − | − | − | + | − | − | + | |
| CC-0657 | + | 0.00 | 0.00 | − | + | − | + | + | + | − | − | + | |
| UB-246 | SZ-131 | − | 0.08 | 0.08 | − | − | − | − | − | − | − | + | + |
| UB-246 | − | 0.00 | 0.00 | − | − | − | − | − | − | − | + | + | |
| CC-1524 | CC-1419 | − | 0.00 | 0.00 | − | + | − | − | + | + | − | − | + |
| CC1418 | + | 0.00 | 0.00 | − | + | + | − | + | + | − | − | + | |
| CC1422 | + | 0.00 | 0.00 | − | + | + | − | + | + | − | − | + | |
| UB-169 | + | 0.00 | 0.00 | − | − | − | + | − | + | − | − | + | |
| UB-172 | + | 0.00 | 0.00 | − | − | − | + | − | + | − | − | + | |
| Cit7 | Cit7g | + | No known hostsi | + | + | + | + | + | + | − | − | + | |
| Group 1 | SZ-122 | − | 0.00 | 0.00 | − | − | + | + | + | − | − | − | + |
| SZ-026 | + | 0.25 | 0.25 | + | + | + | + | + | + | − | − | + | |
| SZ-046 | + | 0.25 | 0.25 | + | + | + | + | + | + | − | − | + | |
| SZ-049 | + | 0.33 | 0.25 | + | + | + | + | + | + | − | − | + | |
| SZ-145 | + | 0.00 | 0.00 | + | + | + | + | + | + | − | − | + | |
| CC-1569 | + | nd | nd | + | + | + | + | + | + | − | − | + | |
| DC3000g | + | 0.00 | 0.00 | + | + | + | + | + | + | − | − | + | |
| Group 2a | UB-419 | + | 2.83 | 1.00 | + | + | + | + | + | + | − | − | + |
| SZ-035 | + | 0.46 | 0.30 | + | + | + | + | + | + | − | − | + | |
| SZ-048 | + | 0.50 | 0.42 | + | + | + | + | + | + | − | − | + | |
| SZ-089 | + | 2.17 | 1.00 | + | + | + | + | + | + | − | − | + | |
| UB-197 | + | 0.00 | 0.00 | + | + | + | − | + | + | − | − | + | |
| UB-210 | + | 0.33 | 0.08 | + | + | + | − | + | + | − | − | + | |
| UB-421 | + | 2.83 | 1.00 | + | + | + | + | + | + | − | − | + | |
| Group 2b | USA-050 | − | 0.00 | 0.00 | − | − | − | − | − | − | + | − | + |
| CC0654 | + | 1.40 | 0.50 | + | + | + | + | + | + | − | − | + | |
| B728Ag | + | 0.00 | 0.00 | + | + | + | + | + | + | − | − | + | |
| Group 3 | 1448Ag | + | 0.00 | 0.00 | + | + | + | + | + | + | − | − | + |
| UB-370 | UB-370 | + | 0.00 | 0.00 | − | + | + | − | + | + | − | − | + |
| USA-102 | USA-046 | + | 0.00 | 0.00 | − | + | + | − | + | − | − | − | + |
| USA-106 | + | 0.00 | 0.00 | − | + | + | + | + | + | − | − | + | |
| USA-032 | USA-032 | + | 3.25 | 1.00 | − | + | + | − | + | + | − | − | + |
Abbreviations: CEL, conserved effector loci; HR, hypersensitive reaction; nd, not determined.
Clades are those described by Morris et al. (2010).
The origins of strains from non agricultural habitats used in this study are described in the supplementary table.
Hypersensitive reaction on tobacco (cv. Xanthi).
Mean intensity, on a scale of 0 to 4, of disease symptoms on 12 cantaloupe seedlings at 7 days after inoculation.
The proportion of 12 cantaloupe seedlings showing disease symptoms at 7 days after inoculation.
Detection of the Psy642 variant of the hrcC gene was based on primers different from those for detecting the canonical hrcC.
Reference strains described previously as follows: Psy508 (Mohr et al., 2008), Psy-642 (Clarke et al., 2010), Cit7 (Hirano and Upper, 1990), DC3000, B728A and 1448A (P. syringae Genome Resources: http://pseudomonas-syringae.org/home.html).
Not determined.
The pathogenicity of Cit7 has been previously well characterized. There are no known hosts and it is considered to be nonpathogenic (Hirano and Upper, 1990).
HR− strains lack at least one conserved effector gene
The conserved type III effector gene hopI1 (Jelenska et al., 2007) was absent from all HR− strains. In most of these HR− strains, a varying number (or even all) of the canonical hrp/hrc locus genes hrpK1, hrpL and hrcC and the conserved effector locus effector genes avrE1 and hrpW1 were also absent based on PCR detection and subsequent Southern blotting (Table 2). Interestingly, for about a dozen HR+ strains we were also unable to detect one or more of the six tested genes (Table 2). For the other 16 HR+ strains, all six genes were detected with PCR. Among the HR− strains, the most frequent molecular profile was similar to that previously reported for strain Psy508 (Mohr et al., 2008) and Psy642 (Clarke et al., 2010) and for HR− strains from Arabidopsis thaliana (Kniskern et al., 2011), viz. strains lacked all six conserved genes tested (Table 2). An example of the results observed with PCR detection and subsequent Southern blotting are presented in the Supplementary Figure S1.
HR− strains lacking all six tested hrp/hrc genes and conserved effector genes are found in multiple distinct clades
In all previous descriptions of HR− P. syringae lacking the six tested genes, all strains fell into a single phylogenetic group, clade 2C (Mohr et al., 2008; Clarke et al., 2010; Kniskern et al., 2011). Clade 2C corresponds to clade SZ-030 in our previous work (Morris et al., 2010)) and we will thus continue calling this clade SZ-030 here. In this present work, HR− strains lacking all six conserved genes of the canonical hrp/hrc locus were found in clade SZ-030 and in three additional phylogenetic groups, clades TA-002, UB-246 and Group 2b (Table 2). In addition to the previously reported strains from plants (cultivated and wild plants, weeds, debris), strains lacking the canonical hrp/hrc locus were isolated from snow pack and headwaters of rivers.
Previous descriptions of strains in the SZ-030 clade indicated that they possess a distantly related variant of the hrcC gene (Clarke et al., 2010), called hrcCPsy642 from here on. The presence of hrcCPsy642 was observed only in HR− strains lacking all the six conserved genes in the canonical T3SS. This included all strains in the SZ-030 clade tested here and one strain outside of this clade, strain USA-050. The sequence of a 560-bp long fragment of the USA-050 hrcC allele was found to be 97% similar to that of strain Psy642.
Some but not all P. syringae strains from French sources that lack the canonical hrp/hrc locus island carry identical sequences originating from a bacteriophage
Previous work reported that strain Psy508 lacking the hrp/hrc locus harbors a prophage downstream of the associated tRNAleu (Mohr et al., 2008). We sought to determine whether all strains lacking the canonical hrp/hrc locus island had this same sequence because its placement in the genome is suggestive of its potential role in the absence of this island. PCR with primers for one of the prophage genes of Psy508, a putative transcriptional regulator, revealed the presence of the prophage sequence in 21 strains lacking the hrp/hrc locus, including the reference strain Psy508. Strains containing the phage sequence were found in all but one of the clades that contained strains without the hrp/hrc locus, viz. clades SZ-030, TA-002 and UB-254 (Table 2). However, the phage sequence was not present in all strains lacking the hrp/hrc locus; it was found in 18 of 23 strains in clade SZ-030, in 1 of 12 strains in clade TA-002, in 2 of 2 strains in clade UB-246 and not in the strain from the Group 2b clade. It was not detected in any strain for which at least one gene in the canonical hrp/hrc cluster was detected. The nucleotide sequence of the prophage was determined. The 380-bp sequence was identical among all strains collected in France and showed 91% similarity with the prophage sequence reported for strain Psy508 isolated from New York (USA; Mohr et al., 2008).
Incapacity to induce HR is not necessarily indicative of the incapacity to cause disease or to persist in plant tissue
For many species of plant pathogenic bacteria, and in particular for P. syringae, the ability to induce hypersensitivity has been considered to be an indicator of the capacity to cause disease in susceptible host plants (He, 1996). Similarly, the inability to induce hypersensitivity is considered indicative of the absence or incapacitation of a functional T3SS and hence of the incapacity to cause disease. Therefore, it is not surprising that most HR− strains were not pathogenic in an assay on cantaloupe seedlings. In all clades, with the exception of clade TA-002, HR− strains caused at most weak disease symptoms (score=1) in 1 of the 12 inoculated seedlings after 7 days of incubation. However, HR− strains in clade TA-002 were among the most aggressive on cantaloupe of the strains tested in this study. In particular, of 15 strains characterized in the TA-002 clade, 13 were HR− and 12 lacked all six genes in the canonical hrp/hrc locus; 8 of these strains caused compatible disease reactions on 75% or more of the cantaloupe seedlings that were inoculated (Table 2; Figure 3). The range of pathogenic capacity of these strains, indicated by their aggressiveness on cantaloupe, was similar to that observed among HR+ strains in the broad host range clades Groups 2a and 2b (Morris et al., 2010).
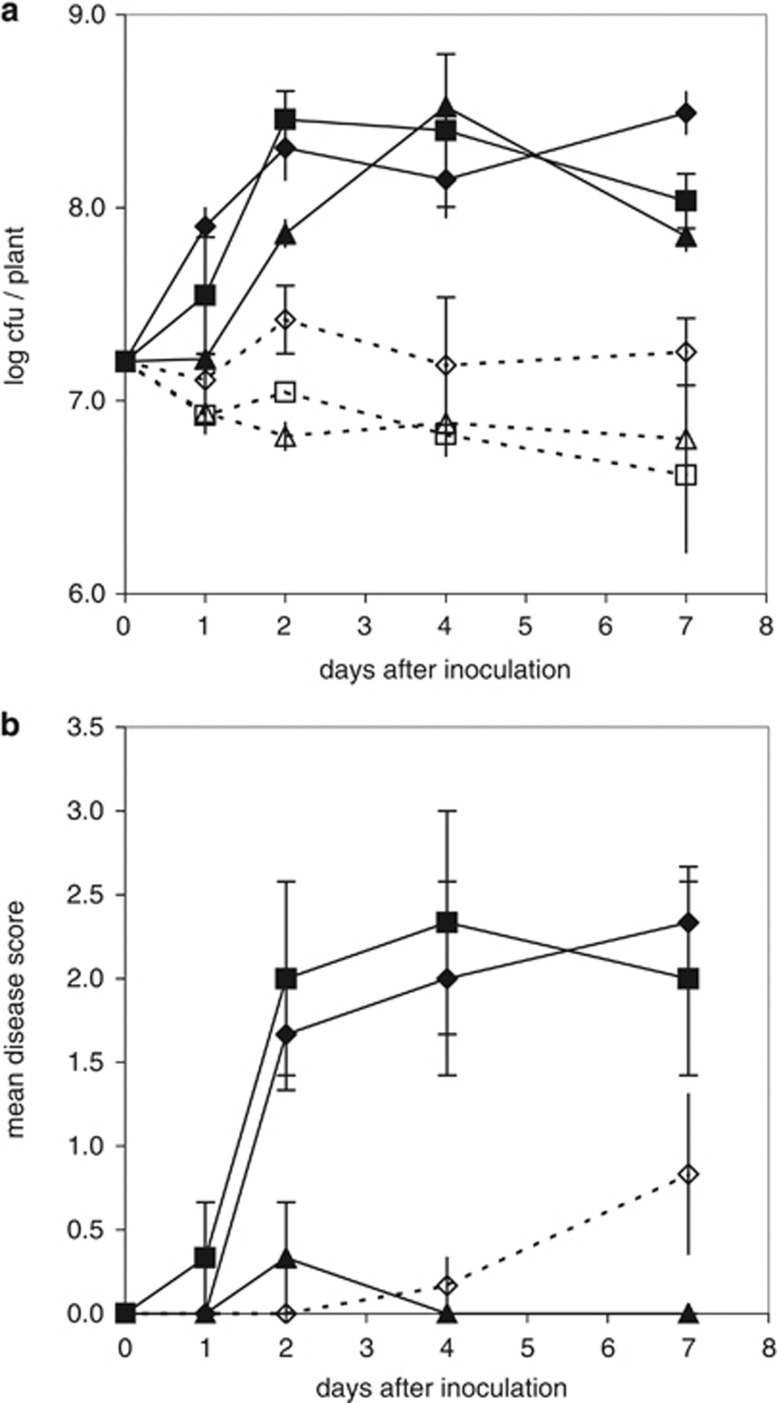
When inoculated into cantaloupe seedlings, HR+ strains with complete canonical T3SS (CC0654, DC3000) or with a partial set of the canonical alleles (USA-032) increased their mean population size by a factor of 10–30 within 4 days after inoculation (Figure 2a). For CC0654 and USA-032, this corresponded to marked disease symptoms, but for strain DC3000 only one plant manifested slight symptoms (disease score=1) during the incubation period (Figure 2b). HR− strain TA-043 showed a slight population increase at 2 days after inoculation and afterwards stabilized around the initial population size although disease symptoms were expressed. For the HR− strains SZ-030 and CC1504, population size decreased slightly and stabilized at around 107 c.f.u. per plant. As expected, no disease symptoms were observed for these strains. For all strains considered together, plants with no symptoms harbored population sizes up to 3 × 108 c.f.u. per plant. Populations in plants with a disease score of 1 ranged in size from 2 × 107 to 1.5 × 108 c.f.u. per plant; those with a disease score of 2 or more had populations of 5 × 107 to 6 × 108 c.f.u. per plant.
Figure 2.
Population dynamics of P. syringae (a) and disease (b) in cantaloupe seedlings after inoculation with either HR+ or HR− strains. HR+ strains are represented by solid lines and symbols (♦ CC0654; ▴ DC3000; ▪ USA-032) and HR− strains by dashed lines and open symbols (⋄ TA-043; ▵ SZ-030; □ CC1504). At each sampling time, 3–6 seedlings were cut off at the soil level and individually macerated to determine population densities. Disease was rated on each of the 3–6 plants sampled. Plants inoculated with strains SZ-030 and CC1504 did not show any disease symptoms. Only one plant inoculated with strain DC3000 showed disease symptoms at 2 days after inoculation. For strain TA-043, half of the inoculated plants showed symptoms at 7 days after inoculation. Error bars indicate the s.e.m.
Figure 3.
Reaction of cantaloupe plants at the cotyledon stage to inoculation with HR+ and HR− strains of P. syringae after 7 days of incubation: plants were inoculated with (a) sterile distilled water, (b) HR− strain CC1524 in the clade of the same name (c) HR+ strain CC0094 in the Group 2b clade, or (d) HR− strain TA043 in the TA002 clade. Plants shown here were chosen at random among the 12 plants inoculated for each strain.
Discussion
The natural occurrence of variants of bacterial plant pathogens with apparent deficiencies in their T3SS has raised questions about the significance of the T3SS for the fitness of these bacterial species. For P. syringae, it has been proposed strains deficient in their overall pathogenicity have adapted to reduce the fitness costs of host-specific virulence in an environment where there is a series of ephemeral host plants (Kniskern et al., 2011). Absence of an effective T3SS could widen the range of plant species on which strains can grow without triggering an HR. In fact, many of the HR− strains have the same level of reproductive fitness in plants as pathogenic strains—in absence of symptoms (Clarke et al., 2010; Kniskern et al., 2011). For the strains tested here, induction of disease symptoms in cantaloupe was associated with increases of population densities of about 1 order of magnitude. However, HR− strains were able to persist in plants at densities above 106 c.f.u. per plant (or 106 c.f.u. g−1, data converted to a per g basis not shown here), which is on the high range of P. syringae population densities naturally observed in wild plants and symptomless crops (Morris et al., 2008). Ephemeral weeds and wild plants might be part of the selective pressures leading to the successful survival of strains of P. syringae deficient in their T3SS. However, P. syringae is clearly apt at inhabiting a range of nonplant and nonliving substrates, thereby suggesting that this species has considerable fitness in these substrates. The proliferation of strains deficient in the T3SS might result from selection pressures specifically related to these substrates. Here we have shown that T3SS-deficient P. syringae can be found not only in wild plants but also in epilithic biofilms, snow pack and river and lake waters in relatively pristine environments and can compose the dominant fraction of the P. syringae population in these substrates. These observations strongly suggest that deficiency in the canonical T3SS does not confer a marked loss of fitness of P. syringae in the context of the ensemble of habitats it exploits under natural conditions. T3SSs are considered to be all-purpose tools for undermining eukaryotic biology, thereby assuring an advantage for the bacterium (Pallen et al., 2005) not only during pathogenesis but also during symbiosis (Downie, 2010; Marchetti et al., 2010) and to foster mutualism or commensalism in the rhizosphere (Lugtenberg and Kamilova, 2009) or the mycosphere (Nazir et al., 2010). Co-existence of HR+ and HR− strains would suggest that in certain environmental habitats, the subversion of eukaryotic cell biology—that of the wild plant host or of the algal or fungal co-inhabitants of the same biofilm, for example, has no decisive role in the fitness of P. syringae. This might be because the population densities that they attain under natural conditions are not large enough to trigger detrimental host responses. However, we cannot rule out that T3SSs in some HR− strains, for example, the T3SS in clade SZ-030 (Clarke et al., 2010; O'Brien et al., 2010), have a role in the interaction with eukaryotes and are equally effective as the canonical T3SS in suppressing host responses.
We have also observed that pathogenicity does not necessarily depend on the canonical T3SS. The surprising capacity of HR− strains to cause disease symptoms in cantaloupe was confined to one clade (TA-002), but the symptoms induced were of the same range of severity as those caused by HR+ strains across diverse clades. The type III injectisome is usually one of many different munitions in the whole arsenal used to attack plant cells, and is often complimented by toxins and cell wall-degrading enzymes. Pathogenicity of strains in the TA-002 clade might involve a range of arms independent of a type III injectisome. However, no syringomycin-like broad host range toxins have been detected in these strains according to bioassays (data not shown). Alternatively (or in addition), pathogenicity of strains in the TA-002 clade might be the result of an unknown T3SS that does not elicit hypersensitivity in tobacco but that is capable of delivering effectors. Genome sequencing has led to an important broadening of the known diversity of T3SSs (Pallen et al., 2005). Current efforts to analyze fully sequenced genomes of P. syringae strains from nonagricultural habitats, including HR− strains, will eventually reveal the full range of potential pathogenicity factors of P. syringae. Nevertheless, this result is troubling because it weakens the utility of the hypersensitivity test in disease diagnostics. This test has been a key criterion to separate pathogens from saprophytes. Furthermore, all papers reporting HR− strains of P. syringae have referred to them as ‘nonpathogenic'. If strains in the TA-002 clade have not been reported as the causal agent of epidemics of crop disease it might be due to their incapacity to do so under field conditions, but more likely it is due to the fact that a positive hypersensitive reaction is generally used early in the diagnostic process as a criterion for targeting candidate causal agents.
The absence of the canonical T3SS in multiple disparate clades raises interesting questions about the processes leading to the acquisition or the loss of the canonical T3SS in P. syringae. There is strong evidence that the canonical T3SS was acquired by the most recent common ancestor of the P. syringae species complex (Sawada et al., 1999) and has evolved via diversifying selection involving mostly mutations (Guttman et al., 2006). However, those phylogenetic studies ignored the existence of nine clades of P. syringae, five of which are more deeply rooted in the phylogenetic tree than the other previously described clades (Morris et al., 2010), the most deeply rooted being clade UB-246. We speculate that the canonical T3SS was acquired at the time of diversification of UB-246 from all of the other clades. The most parsimonious explication for T3SS deficiency in the other clades is subsequent loss by the ancestor of the clades in which it is currently absent. However, clade TA-002 contains several strains that effectively induce hypersensitivity and for which some, but not all, of the six conserved genes were detected. This suggests a diversification of the genes beyond what could be detected with the primers used here, designed based on only three completely sequenced P. syringae genomes (Mohr et al., 2008). For all the strains in the CC1524 clade, closely related to TA-002, we could only detect some of the six conserved genes in the canonical T3SS. Similarly, for all strains in clades UB-370, USA-102 and USA-032, which are together in another branch of the phylogenetic tree, some, but not all, of the six conserved genes were detected in these otherwise HR+ strains. This suggests that divergence of the alleles in the canonical T3SS is more rapid in some branches than elsewhere in the phylogenetic tree of P. syringae. Our observations highlight that natural habitats likely harbor a greater diversity of alleles of the genes in the T3SS of P. syringae than currently observed in strains from crops thereby raising exciting questions about the nature of the drivers of this diversification.
The presence of a bacteriophage sequence in strain Psy508 near the site of the (missing) canonical hrp/hrc cluster (Mohr et al., 2008) raised questions about the link between the presence of this phage and the absence of the canonical T3SS. Here we show that, although this prophage sequence is present only in strains lacking the canonical T3SS, it is not universally present in all strains with this deficiency. Furthermore, acquisition of the phage seems to have occurred multiple times and perhaps at distinctly different dates as indicated by sequence divergence between the French and US strains. This suggests that there were multiple and very recent acquisitions of the same phage in clades TA-002, UB-246 and SZ-030 in France, and that strain Psy508 acquired a variant of the same phage or had acquired it at a much earlier date. We must also consider that some strains in which we did not detect the phage may have at one time acquired it and then lost it again.
For the two functional T3SSs described to date in P. syringae (Clarke et al., 2010; Tampakaki et al., 2010), our observations suggest an exclusivity of the type of T3SS harbored in the genomes of P. syringae. Similarly in P. viridiflava, strains harbor only one of the two types of T3SS identified by Bergelson and colleagues (Araki et al., 2006). However, Pallen et al. (2005) reported that certain bacteria (for example, Burkholderia spp.,) can accumulate different T3SSs. The secrets of T3SS polymorphism in P. syringae will be disclosed by genomic analyses that incorporate the novel strains described here.
Our results also have implications for understanding which habitats have been important in the evolutionary history of P. syringae. Compared with other members of the genus such as P. fluorescens, P. syringae has a much reduced capacity to use carbon sources (Sands et al., 1970). It has been suggested that the ancestor of P. syringae lost a range of metabolic functions, as an adaptation to the nutritionally constrained environment characteristic of the plants compared with the more nutritionally diverse and copious environments encountered by P. fluorescens as a saprophyte (Mithani et al., 2011). This analysis was based on genomes of P. syringae in the Groups 1, 2 and 3 clades. In light of the new clades of P. syringae that our work has revealed, it would be important to consider the metabolic adaptations of the strains in the UB-246 clade, the most deeply rooted of the known P. syringae clades and represented solely by HR− strains. If they resemble those of P. syringae in general, then this would suggest that adaptation to a nutritionally constrained environment would be most probably for success as a resident of pristine freshwater habitats. We propose that loss of metabolic capacities in P. syringae might have originally been an adaptation for the oligotrophic conditions of certain freshwater habitats. Dissolved organic carbon sources in freshwater generally originate from plants (leaching and degradation). The ability to use sparse quantities of plant-derived carbon sources could have prepared P. syringae for a more intimate association with plants, but the intimate association with plants itself might not have been the initial selection pressure for adaptation to oligotrophic conditions.
The persistence of nonpathogenic variants of plant and animal pathogens in the environment has been a springboard for the development of biological control for plant pathogens (van Alfen, 1982; Kerr, 1987; Larkin et al., 1996) and of diseases of human and animal intestinal and urinary tracts (Kruis et al., 2004; Schneitz, 2005). The diversity, abundance and ubiquity of nonpathogenic strains of P. syringae suggest that competitive exclusion of pathogen strains by nonpathogenic conspecifics as a means to control disease might be worth exploring. Competitive exclusion between ice nucleation active and inactive variants of P. syringae has successfully limited frost damage induced by this bacterium under experimental conditions (Lindow, 1987). Inundative biological control is the most obvious of the potential practical applications of this work, but strategies for plant breeding or crop management that foster proliferation of nonpathogenic strains of P. syringae might also have potential to mitigate disease development.
Acknowledgments
We are grateful to B Moury, O Berge, A Roig, V Decognet, A Bouffière, C Leyronas, V Rittner (INRA-PACA), to P Amato (CNRS-University Blaise Pascal), and to David Sands (MSU, Bozeman, MT, USA) for fruitful discussion relative to this work. We thank the American Philosophical Society for a grant to CEM for fieldwork in the United States and the numerous colleagues who helped with field sampling. Samples were collected from the Grand Teton National Park in accordance with permit number GRTE-2007-SCI-0023. This work was supported by INRA's Department of Plant Health and Environment.
Footnotes
Supplementary Information accompanies the paper on The ISME Journal website (http://www.nature.com/ismej)
Supplementary Material
References
- Araki H, Tian D, Goss EM, Jakob K, Halldorsdottir SS, Kreitman M, et al. Presence/absence polymorphism for alternative pathogenicity islands in Pseudomonas viridiflava, a pathogen of Arabidopsis. Proc Natl Acad Sci USA. 2006;103:5887–5892. doi: 10.1073/pnas.0601431103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Block A, Alfano JR. Plant targets for Pseudomonas syringae type III effectors: virulence targets or guarded decoys. Curr Opin Microbiol. 2011;14:39–46. doi: 10.1016/j.mib.2010.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke CR, Cai R, Studholme DJ, Guttman DS, Vinatzer BA. Pseudomonas syringae strains naturally lacking the classical P. syringae hrp/hrc locus are common leaf colonizers equipped with an atypical type III secretion system. Mol Plant Microb Interact. 2010;23:198–210. doi: 10.1094/MPMI-23-2-0198. [DOI] [PubMed] [Google Scholar]
- Cornelis GR, Van Gijsegem F. Assembly and function of type III secretory systems. Annu Rev Microbiol. 2000;54:735–774. doi: 10.1146/annurev.micro.54.1.735. [DOI] [PubMed] [Google Scholar]
- Downie JA. The roles of extracellular proteins, polysaccharides and signals in the interactions of rhizobia with legume roots. FEMS Microbiol Rev. 2010;34:150–170. doi: 10.1111/j.1574-6976.2009.00205.x. [DOI] [PubMed] [Google Scholar]
- Guttman DS, Gropp SJ, Morgan RL, Wang PW. Diversifying selection drives the evolution of the type III secretion system pilus of Pseudomonas syringae. Mol Biol Evol. 2006;23:2342–2354. doi: 10.1093/molbev/msl103. [DOI] [PubMed] [Google Scholar]
- He SY. Elicitation of plant hypersensitive response by bacteria. Plant Physiol. 1996;112:865–869. doi: 10.1104/pp.112.3.865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirano SS, Upper CD. Population biology and epidemiology of Pseudomonas syringae. Annu Rev Phytopathol. 1990;28:155–177. [Google Scholar]
- Hueck CJ. Type III protein secretion systems in bacterial pathogens of animals and plants. Microbiol Mol Biol Rev. 1998;62:379–433. doi: 10.1128/mmbr.62.2.379-433.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jelenska J, Yao N, Vinatzer BA, Wright CM, Brodsky JL, Greenberg JT. A J-domain virulence effector of Pseudomonas syringae remodels host chloroplasts and suppresses defenses. Curr Biol. 2007;17:499–508. doi: 10.1016/j.cub.2007.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jock S, Kim W-S, Barny M-A, Geider K. Molecular characterization of natural Erwinia pyrifoliae strains deficient in hypersensitive response. Appl Environ Microbiol. 2003;69:679–682. doi: 10.1128/AEM.69.1.679-682.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerr A. Daniel Mcalpine Memorial Lecture. Agrobacterium: pathogen, genetic engineer and biological control agent. Australasian Plant Pathol. 1987;16:45–47. [Google Scholar]
- Kim H-S, Ma B, Perna NT, Charkowski AO. Phylogeny and virulence of naturally occurring type III secretion system-deficient Pectobacterium strains. Appl Environ Microbiol. 2009;75:4539–4549. doi: 10.1128/AEM.01336-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King EO, Ward MK, Raney DE. Two simple media for the demonstration of pyocyanin and fluorescein. J Lab Clin Med. 1954;44:301–307. [PubMed] [Google Scholar]
- Kniskern JM, Barrett LG, Bergelson J. Maladaption in wild populations of the generalist plant pathogen Pseudomonas syringae. Evolution. 2011;65:818–830. doi: 10.1111/j.1558-5646.2010.01157.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruis W, Frič P, Pokrotnieks J, Lukáš M, Fixa B, Kascák M, et al. Maintaining remission of ulcerative colitis with the probiotic Escherichia coli Nissle 1917 is as effective as with standard mesalazine. Gut. 2004;53:1617–1623. doi: 10.1136/gut.2003.037747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar S, Tamura K, Nei M. MEGA—molecular evolutionary genetics analysis software for microcomputers. Comput Appl Biosci. 1994;10:189–191. doi: 10.1093/bioinformatics/10.2.189. [DOI] [PubMed] [Google Scholar]
- Larkin RP, Hopkins DL, Martin FN. Suppression of Fusarium wilt of watermelon by non pathogenic Fusarium oxysporum and other microorganisms recovered from a disease-supressive soil. Phytopathology. 1996;86:812–819. [Google Scholar]
- Lelliot RA, Billing E, Hayward AC. A determinative scheme for the fluorescent plant pathogenic Pseudomonads. J Appl Bacteriol. 1966;29:470–489. doi: 10.1111/j.1365-2672.1966.tb03499.x. [DOI] [PubMed] [Google Scholar]
- Lindow SE. Competitive exclusion of epiphytic bacteria by ice- Pseudomonas syringae mutants. Appl Environ Microbiol. 1987;53:2520–2527. doi: 10.1128/aem.53.10.2520-2527.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lugtenberg B, Kamilova F. Plant-growth-promoting rhizobacteria. Ann Rev Microbiol. 2009;63:541–556. doi: 10.1146/annurev.micro.62.081307.162918. [DOI] [PubMed] [Google Scholar]
- Marchetti M, Capela D, Glew M, Cruveiller S, Chane-Woon-Ming B, Gris C, et al. Experimental evolution of a plant pathogen into a legume symbiont. PLoS Biol. 2010;8:e1000280. doi: 10.1371/journal.pbio.1000280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mithani A, Hein J, Preston GM. Comparative analysis of metabolic networks provides insight into the evolution of plant pathogenic and nonpathogenic lifestyles in Pseudomonas. Mol Biol Evol. 2011;28:483–499. doi: 10.1093/molbev/msq213. [DOI] [PubMed] [Google Scholar]
- Mohan SK, Schaad NW. An improved agar plating assay for detecting Pseudomonas syringae pv. syringae and P. s. pv. phaseolicola in contaminated bean seed. Phytopathology. 1987;77:1390–1395. [Google Scholar]
- Mohr TJ, Liu H, Yan S, Morris CE, Castillo JA, Jelenska J, et al. Naturally occurring non-pathogenic isolates of the plant pathogen species Pseudomonas syringae lack a Type III secretion system and effector gene orthologues. J Bacteriol. 2008;190:2858–2870. doi: 10.1128/JB.01757-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris CE, Kinkel LL, Kun X, Prior P, Sands DC. A surprising niche for the plant pathogen Pseudomonas syringae. Infect Genet Evol. 2007;7:84–92. doi: 10.1016/j.meegid.2006.05.002. [DOI] [PubMed] [Google Scholar]
- Morris CE, Sands DC, Vanneste JL, Montarry J, Oakley B, Guilbaud C, et al. Inferring the evolutionary history of the plant pathogen Pseudomonas syringae from its biogeography in headwaters of rivers in North America, Europe and New Zealand. MBio. 2010;1 (3:e00107–10-e00107-20. doi: 10.1128/mBio.00107-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morris CE, Sands DC, Vinatzer BA, Glaux C, Guilbaud C, Buffière A, et al. The life history of the plant pathogen Pseudomonas syringae is linked to the water cycle. ISME J. 2008;2:321–334. doi: 10.1038/ismej.2007.113. [DOI] [PubMed] [Google Scholar]
- Nazir R, Warmink JA, Boersma H, van Elsas JD. Mechanisms that promote bacterial fitness in fungal-affected soil microhabitats. FEMS Microbiol Ecol. 2010;71:169–185. doi: 10.1111/j.1574-6941.2009.00807.x. [DOI] [PubMed] [Google Scholar]
- O'Brien HE, Desveaux D, Guttman DS. Next-generation genomics of Pseudomonas syringae. Curr Opin Microbiol. 2010;14:24–30. doi: 10.1016/j.mib.2010.12.007. [DOI] [PubMed] [Google Scholar]
- Pallen MJ, Beatson SA, Bailey CM. Bioinformatics, genomics and evolution of non-flagellar type-III secretion systems: a Darwinian perpective. FEMS Microbiol Rev. 2005;29:201–229. doi: 10.1016/j.femsre.2005.01.001. [DOI] [PubMed] [Google Scholar]
- Sands DC, Schroth MN, Hildebrand DC. Taxonomy of phytopathogenic pseudomonads. J Bacteriol. 1970;101:9–23. doi: 10.1128/jb.101.1.9-23.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawada H, Suzuki F, Matsuda I, Saitou N. Phylogenetic analysis of Pseudomonas syringae pathovars suggests the horizontal gene transfer of argK and the evolutionary stability of hrp gene cluster. J Mol Evol. 1999;49:627–644. doi: 10.1007/pl00006584. [DOI] [PubMed] [Google Scholar]
- Schneitz C. Competitive exclusion in poultry––30 years of research. Food Control. 2005;16:657–667. [Google Scholar]
- Tampakaki AP, Skandalis N, Gazi AD, Bastaki MN, Sarris PF, Charova SN, et al. Playing the ‘harp': evolution of our understanding of hrp/hrc genes. Annu Rev Phytopathol. 2010;48:347–370. doi: 10.1146/annurev-phyto-073009-114407. [DOI] [PubMed] [Google Scholar]
- Troisfontaines P, Cornelis GR. Type III secretion: more systems than you think. Physiology. 2005;20:326–339. doi: 10.1152/physiol.00011.2005. [DOI] [PubMed] [Google Scholar]
- van Alfen NK. Biology and potential for disease control of hypovirulence of Endothia parasitica. Annu Rev Phytopathol. 1982;20:349–362. [Google Scholar]
- Vinatzer BA, Teitzel GM, Lee M-W, Jelenska J, Hotton S, Fairfax K, et al. The type III effector repertoire of Pseudomonas syringae pv. syringae B728a and its role in survival and disease on host and non-host plants. Mol Microbiol. 2006;62:26–44. doi: 10.1111/j.1365-2958.2006.05350.x. [DOI] [PubMed] [Google Scholar]
- Xia X, Xie Z. DAMBE: data analysis in molecular biology and evolution. J Heredity. 2001;92:371–373. doi: 10.1093/jhered/92.4.371. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.