Abstract
Protein phosphorylation and dephosphorylation form a major post-translation mechanism that enables a given cell to respond to ever-changing internal and external environments. Neurons, similarly to any other cells, use protein phosphorylation/dephosphorylation to maintain an internal homeostasis, but they also use it for updating the state of synaptic and intrinsic properties, following activation by neurotransmitters and growth factors. In the present review we focus on the roles of several families of kinases, phosphatases, and other synaptic-plasticity-related proteins, which activate membrane receptors and various intracellular signals to promote transcription, translation and protein degradation, and to regulate the appropriate cellular proteomes required for taste memory acquisition, consolidation and maintenance. Attention is especially focused on the protein phosphorylation state in two forebrain areas that are necessary for taste-memory learning and retrieval: the insular cortex and the amygdala. The various temporal phases of taste learning require the activation of appropriate waves of biochemical signals. These include: extracellular signal regulated kinase I and II (ERKI/II) signal transduction pathways; Ca2+-dependent pathways; tyrosine kinase/phosphatase-dependent pathways; brain-derived neurotrophicfactor (BDNF)-dependent pathways; cAMP-responsive element bindingprotein (CREB); and translation-regulation factors, such as initiation and elongation factors, and the mammalian target of rapamycin (mTOR). Interestingly, coding of hedonic and aversive taste information in the forebrain requires activation of different signal transduction pathways.
Keywords: taste, insular cortex, amygdala, phosphorylation, translation regulation, ERK-MAPK
INTRODUCTION
Taste recognition and memory are linked to hedonic values such as "attractive", "palatable" or "repulsive", and also to degree of familiarity, and nutritive or toxic characteristics. The most commonly used taste-memory models are taste preference, conditioned taste aversion (CTA) and latent inhibition of conditioned taste aversion (LI-CTA). The consumption of a novel taste that is not associated with negative visceral consequences leads to the formation of a non-associative form of sensory memory, known as incidental taste memory. Habituation followed by exposure to a novel taste, choice preference, and attenuation of neophobia are the models that are most widely used to decipher incidental taste learning and memory. CTA is an associative learning paradigm; by means of CTA, organisms learn to avoid a novel food substance (conditioned stimulus, CS) associated with delayed poisoning (unconditioned stimulus, US) [1-3]. CTA can be acquired in a single trial and it differs from other associative paradigms in the long delay between CS and US. In LI-CTA, a novel taste (CS) is learned days before a CTA trial, and it results in retarded performance of a CTA task; a positive tag of the learned taste is established and, consequently, weaker aversion is expressed [4, 5]. Experimentally, following 3 days of restricted access to water, animals sample novel taste solution. Since animals learn novel taste without any negative visceral consequence, they both remember the taste as incidental memory (i.e. no UCS) but also learn that the taste is safe. Whereas in CTA, lithium chloride (0.05~0.15 M) is injected 40 minutes after the drinking session (UCS), which elicits gastric pain and incomfort and therefore animals learn the given taste as an aversive one. The most commonly used novel taste is saccharin (0.1~0.5% w/v in plain tap water) and sodium chloride (0.1~0.5% w/v also in plain tap water) [4, 5].
There is extensive overlapping between the gustatory neural circuits associated with the acquisition and expression of novel-taste and aversive-taste memories. The neural circuitry of taste learning includes the nucleus of the solitary tract (NST, also known as nucleus of tractus solitarius - NTS), the parabrachial nucleus (PBN), the parvocellular part of the ventralis postmedial thalamic nucleus of the thalamus (pVPMpc), the nucleus basalis magnocellularis (NBM), the amygdala (AMY) and the insular cortex (IC) [5-7]. Recent findings indicate that the nucleus accumbens (NAcc), prefrontal cortex (PFC) and perirhinal cortex (PC) also participate in taste-memory formation [8-16]. Although the hippocampus (HIP) has been shown to be involved in several forms of learning, its involvement in taste learning is unclear. Hippocampal lesion studies in the past decade have yielded inconclusive results regarding the role of the HIP in taste learning and CTA [17-23]. Recent studies demonstrated that the HIP is necessary for the consolidation of attenuation of neophobia [16], and inhibition of protein synthesis in the HIP impairs consolidation of CTA extinction [24]. Interestingly, another study indicated thatvarious molecular pathways were activated in the HIP and in the IC, during different temporal windows following novel taste learning [25]. Clearly, further studies are required to identify and define the role of the HIP in taste learning. Many studies have shown that the IC and AMY are highly involved in taste-memory formation, consolidation and retrieval, therefore, in this review we will focus on delineating the prominent role of protein phosphorylation in taste learning in these two temporal lobe structures.
GENE EXPRESSION AND PROTEIN SYNTHESIS
Memory consolidation, a progressive process of post-acquisition stabilization that facilitates the permanent storage of memory, is dependent on the gene expression and synthesis of new proteins within the relevant brain structures. Recent studies elucidated the mechanisms of gene expression and the regulation of translation that underlies taste memory consolidation.
Phosphorylation of the transcription factor, cAMP-response element-binding protein (CREB) on serine 133 causes it to bind the cAMP response element (CRE), which is then bound to a CREB-binding protein (CBP). This complex, in turn, leads the CREB to turn the transcription of certain geneson oroff [26], and CREB has been shown to be phosphorylated directly by cAMP-dependent protein kinase (PKA) and Ca2+/calmodulin-dependent protein kinase IV, and indirectly by the extracellular signal-regulated kinase (ERKI/II) cascade through activation of RSK2, which is a member of the pp90rsk family of S6 kinases [27, 28]. Novel-taste learning increased CREB phosphorylation in the HIP but, interestingly, not in the IC [25], whereas intraperitoneal (i.p.) injection of LiCl (as a US) increased CREB activation in the IC [29]. Moreover, CTA conditioning increased CREB activation in the lateral septum, lateral amygdala (LA) and IC [29, 30]. Phosphorylated CREB activated the transcription of many genes, including c-Fos, brain-derived neurotropic factor (BDNF), and CCAAT/enhancer binding protein (C/EBPβ) [31, 32]. BDNF stimulated CREB phosphorylation, and this CREB phosphorylation played a central role in mediating the BDNF response [32]. CTA conditioning was correlated with increased expression of BDNF protein from 4 to 12 h after conditioning in the central nucleus of the amygdala (CeA), and from 8 to 12 h after conditioning in the IC [33]. Ma et al. [33] also have shown that association of novel taste and malaise was essential for BDNF signalling to be involved in taste learning. This is consistent with previous studies, which found no changes in CREB phosphorylation in the IC following novel-taste learning [25], and activation of CREB in the LA only following CTA [29]. Moreover, novel-taste consumption and association of novel taste with malaise in CTA learning elicited increased expression levels of c-Fos - an immediate early gene whose expression is mediated by CREB - in the CeA and IC [34-36]. Furthermore, activation of c-Fos in the IC and AMY has been found to be required for CTA memory formation [15, 37]. These particular examples clearly suggest that some proteins, e.g., BDNF, that are implicated in taste memory consolidation are mainly related to the consolidation of CS-US association with CTA and not to CS or US exposure alone.
Antisense oligonucleotide-mediated inhibition of CREB in the AMY, particularly in the CeA, has been shown to impair CTA memory [38], and CREB mutant mice have also been found to be impaired in CTA memory [39], which suggests that the activity of CREB is vital for CTA memory formation.
Another transcription factor that was induced in correlation with taste learning is C/EBPβ, which also has been associated with HIP-dependent learning processes [40-42]. Yefet et al. [25] have found increased expression levels of C/EBPβ protein 18 h following novel-taste consumption; furthermore, it is interesting thatfurther study demonstrated that this correlative induction of C/EBPβ could be deleted if another novel taste was consumed 4 h after the first one, i.e. retrograde amnesia [43], which suggests that a long process of biochemical changes leads to the increased expression of the proteins required for taste-memory consolidation.
Modification of the chromatin structure can facilitate functioning of transcription factors. Lysine acetylation of histones alters the higher-order chromatin structure and exposes DNA to transcription processes. Novel-taste consumption has been shown to be associated with increased activity of histone acetyl transferase in the IC [44]. Moreover, this increased lysine acetylation of p42Ack and p55Ack proteins appears to be at least partly downstream of activation of ERKI/II in the IC [44], which suggests that novel-taste consumption enhances the chromatin structural reorganization and thereby leads to increased transcription.
Memory consolidation is generally considered to be a progressive stabilization process that requires the synthesis of new proteins. Indeed, pharmacological studies based on the protein-synthesis inhibitor, anisomycin demonstrated the general requirement for protein synthesis in novel-taste learning and CTA memory consolidation [4, 45-47].
Translation is the mechanism by which the relevant neurons fine-tune the levels of proteins in both time and space. The translation process includes three distinct phases: initiation, elongation, and termination. The primary target for translation regulation is the initiation phase, therefore, key roles in controlling translation are played by translation initiation factors through phosphorylation of the various initiation factors and ribosomal proteins [48, 49]. Novel-taste learning enhances the phosphorylation of ribosomal protein S6 kinase1 (S6K1) on threonine 389 [50], whereas this phosphorylation was positively correlated with enhanced initiation of translation [51]. Interestingly, within the same temporal window, there was an increase in the phosphorylation of a eukaryotic elongation factor (eEF2) [50] - whose phosphorylation correlates with a general reduction of the elongation process in translation - on threonine 56 [52-54]. These results indicate that although novel-taste consumption increases the general initiation of translation, it decreases the elongation rate. On the assumption that the processes take place in the same cell, it is reasonable to suggest that during taste-memory consolidation, in spite of the general increase in the initiation phase of translation, elongation becomes the rate-limiting step allowingonly of poorly initiated mRNAs for further translation [50]. This distinct population of mRNAs consists of plasticity-related mRNAs, such as C/EBPβ [25].
Another hub molecule in translation control is mammalian target of rapamycin-sensitive kinase (mTOR). Phosphorylated mTOR on serine 2448 can regulate several downstream elements of the translation machinery, including S6K1 and eukaryotic elongation factors 1A and 2 (eEF1A and eEF2) [55]. Novel-taste learning induced a correlative activation of mTOR in a biphasic pattern, at 15 min and 3h following novel-taste learning [56]. Moreover, phosphorylation of S6K1 has also been shown to be increased within the same temporal window [56]. Biochemical fractionation analysis revealed that phosphorylation of S6K1 was increased in a synaptoneurosomal preparation at 15 min but not at 3 h following novel-taste learning, indicating the importance of spatial segregation of signaling molecules during taste-memory formation. Unlike general protein synthesis inhibitors such as anisomycin and cycloheximide, rapamycin selectively inhibits the translation of certain populations of mRNAsthat contain an oligo-pyrimidine tract at their transcriptional start (5'TOP), most notably mRNAs encoding ribosomal proteins and elongation factors [57, 58]. Inhibition of mTOR by microinjection of rapamycin locally into the IC reduced expression of eEF1A and post-synaptic density protein 95 (PSD-95), reduced S6K1 phosphorylation, and increased eEF2 phosphorylation; at the same time, this inhibition of mTOR in the IC attenuated long-term taste memory as analyzed by LI-CTA paradigm [56]. These results support the notion that there is a selective control of translation of proteins during taste-memory consolidation.
Behavioral analyses of transgenic mice with specific modifications in the translation machinery revealed clear attenuation of taste memory (Table 1): S6K1 knock-out mice showed clear attenuation of taste memory [59]; the mutant mice that lacked the translation repressor, eukaryotic initiation factor 4E-binding protein (4E-BP2) exhibited enhanced formation of the eIF4F complex, and analysis of taste learning in these mice revealed no change in taste recognition, but enhanced CTA learning [60]. Similarly, mice with reduced phosphorylation of eukaryotic initiation factor 2 alpha (eIF2α) exhibited enhanced taste learning, but no effect on taste recognition [61].
Table 1.
Taste learning and memory in transgenic mice
Protein degradation in parallel with protein synthesis regulates the proper turnover of proteins and the quality of the cellular proteome. The ubiquitin- proteasome system (UPS), and lysosome- and proteases-mediated proteolysis are the main routes of protein degradation. Whereas the lysosome accounts for protein turnover of ~20% in cells [62], the UPS is highly conserved and serves as a principal means of targeting proteins for degradation [63, 64]. Pharmacological and genetic studies that demonstrated the relationship between the UPS and several forms of learning and memory have been reviewed extensively [63-65]. Briefly, pharmacological inhibition of the UPS in the HIP blocked long-term memory formation in a one-trial inhibitory avoidance task and in a Morris water-maze task, and prevented extinction of contextual fear memory [66-68]. In addition, increased protein ubiquitination and 26S proteasome tryptic-like activity have been observed 4 h following a one-trial inhibitory avoidance task in the HIP [66]. Similarly, polyubiquitinated postsynaptic proteins such as shank and GKAP were increased in the HIP 1 h after retrieval of contextual fear memory [68]. Taken together, these findings indicate that, in addition to gene expression and protein synthesis, the degradation of proteins may be equally important for long-term memory formation. Bilateral microinjection of the UPS inhibitor, lactacystin into both the IC and the AMY appeared to impair the CTA memory; however, the cellular events and mechanisms, and the temporal activity of the lactacystin underlying this behavioral phenotype have not been clearly described [69].
PROTEIN KINASES AND PHOSPHATASES
Important kinases that are well documented in the field of learning and memory processes are: ERKI/II, Ca2+/phospholipid-dependent protein kinase (PKC), PKA, and Ca2+/calmodul-independent protein kinase II (CaMKII). Within the scope of the present review, we will focus on the rolesof these fourpathways in taste learning and memory.
Activation of ERKI/II - a serine/threonine kinase with the two isoforms ERKI/II (p42/44) belonging to the mitogen-activated protein kinase (MAPK) family of signal cascades - is necessary for several forms of learning and memory. Several lines of evidence showed activation of ERKI/II by phosphorylation on threonine 202/ tyrosine 204, following novel taste learning [44, 50, 70, 71]. However, the temporal window of the activation is not clear; for example, several studies have shown activation of ERKI/II in the IC a few minutes after novel-taste consumption, but found it undetectable after more than 1 h [25, 50, 70, 71]. However, Swank and Sweatt [44] have found sustained activation of ERKI/II lasting between 2 and 6 h, accompanied by a second wave of activation in the IC at 24 and 48 h following novel-taste learning in mice. Interestingly, when differences in the activation of ERKI/II are observed in different studies, there is a tendency to attribute them to differences among the experimental setups. The studies that demonstrated immediate and transient activation of ERKI/II have all used the rat model with water as a control [25, 50, 70, 71], whereas Swank and Sweatt [44] used the mouse model with a time point of zero as a control. It is also important to note that most of the studies, including those that observed transient activation of ERKI/II, used novel taste in the form of solutions such as saccharin, NaCl, etc., but Swank and Sweatt [44] used blueberry bar, a solid taste source, thus indicating that the texture and the odor of the food, as well as its taste, also plays a key role in taste learning and memory. Further examination revealed phosphorylation of ELK-1 - a substrate of activated ERKI/II - concomitant with muscarinic and N-methy-D-aspartate receptor (NMDAR) activation in the IC, following novel-taste learning [70]. As shown in Table 2, novel-taste learning also increased the phosphorylation and activation of several members of the MAPK pathway, such as Raf on serine 259, MEK on serine 217/221, p90RSK on serine 381, and Jun N-terminal kinase 1/2 (JNK1/2) [44, 70]. It has also been shown that consumption of a given novel taste for the second time, but not for the fourth time increased ERKI/II activation. These results indicate that ERKI/II activity in the IC is no longer significant once a taste has become familiar [44, 70].
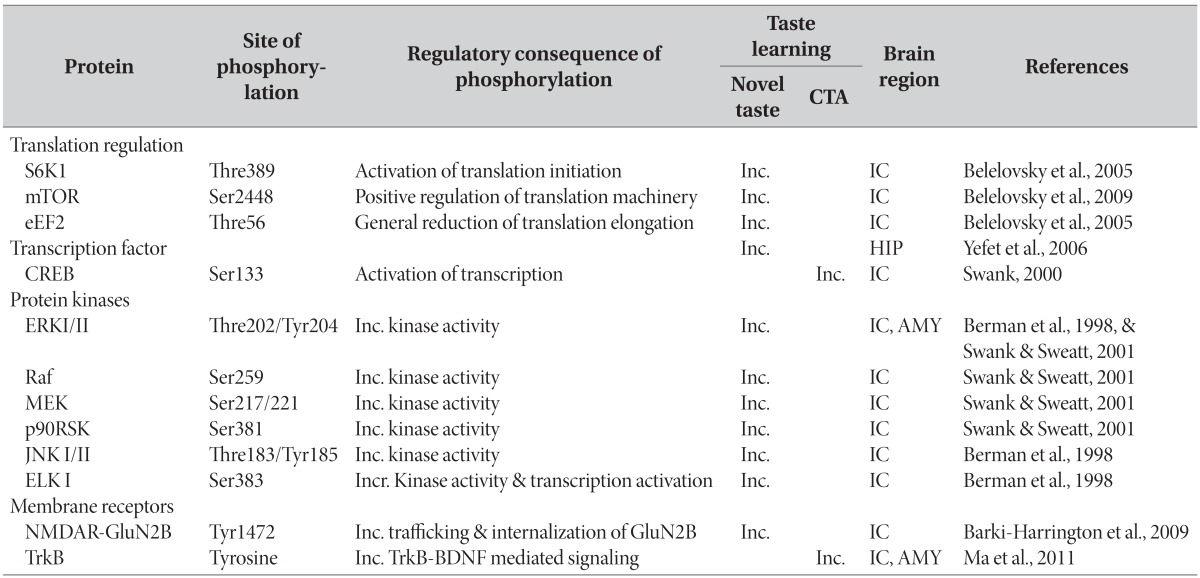
Table 2.
Phosphorylation of proteins and their regulatory consequences for taste learning (Inc.-Increase)
As in the IC, novel taste learning activates ERKI/II in the LA also [29]. Interestingly, i.p. injection of LiCl increased the activation of ERKI/II in the CeA [29,72]. CTA conditioning was found to increase activation of ERKI/II in the LA and IC [29,72]. Pharmacological inhibition of MEK, an upstream kinase of ERKI/II, in the IC or AMY attenuated CTA memory [70, 72]. Interestingly, the expression of long-term potentiation (LTP), a neural model of learning and memory, in the IC was found to be ERKI/II-dependent [73]. As in other behavioral paradigms and brain regions, ERKI/II activation is highly correlated with taste learning, probably mainly during the consolidation phase of learning. As mentioned elsewhere [3, 5], the upstream mechanism of ERKI/II activation has been extensively studied, but the downstream targets of activated ERKI/II during taste learning, and especially during consolidation, remain elusive. One of the major roles of activated ERKI/II is its involvement in translation regulation [74]. As shown in Fig. 1, activated ERKI/II enhances the transcription via CREB and translation of synaptic-plasticity-related proteins directly through its downstream kinases during taste learning and memory.
Fig. 1.

Schematic diagram depicting major signaling pathways underlying novel taste learning: (A) versus conditioned taste aversion, and (B) in an imaginary neuron within the GC as described in detail in the text. (A) ERK/MAPK, a signaling cascade that is critical for taste-memory formation, enhances the activity of synaptic-plasticity-related proteins, and transcription through ELK-1. The ERK/MAPK and PI3K-AKT-mTOR pathways regulate general protein synthesis. Following novel-taste learning, phosphorylation of mTOR increases and eEF2 phosphorylation decreases, providing a negative-feedback signaling loop to enable the translation of poorly initiated and, presumably, the learning-related mRNAs. (B) Upon binding with the active BDNF, TrkB regulates multiple signaling pathways through ERK/MAPK, PI3K and PLCγ. Several kinases, including PKA, CaMKIV and ERK/MAPK, were implicated as CREB kinases; however, ERK/MAPK-and CREB-mediated transcription is illustrated according to taste-learning publications. The activation of CREB leads to the expression of target genes, including BDNF and a transcription factor C/EBPβ which, in turn, presumably, regulates the transcription of late-responding genes. Like novel-taste learning, CTA learning enhances translation mediated by the ERK/MAPK and PI3K-AKT-mTOR pathways. CaMKIIα is associated with the endoplasmic reticulum (ER), which facilitates the synaptic trafficking and transport of postsynaptic proteins such as GluN2B and PSD95 via Golgi. The question mark indicates that the signaling molecules involved are not known. This simplified and schematic representation assumes that the depicted molecular processes take place within a single neuron; however, this remains to be explored by means of new tools and methods.
PKC comprises a family of serine/threonine kinases that has been reported to be necessary in the IC, AMY and PBN for formation of a CTA memory [75-77]. Injection of US elicited PKC enzyme activity in the membrane-bound fraction in the PBN and, furthermore, CTA conditioning increased the activity of PKC persistently for 48 h in the PBN [78, 79]. However, despite the identification and characterization of 11 previously known PKC isoforms, the functional role of each isoform in taste learning is not clear, and the temporal activities of PKC in the IC and AMY remain to be identified. A constitutively active PKC isoform M zeta (PKMζ) has been shown to be important for LTP maintenance and HIP dependent long-term memory (Sacktor, 2011). Pharmacological inhibition or lentivirally mediated overexpression of PKMζ led, respectively, to erasure or enhancement of long-term CTA memory [80-83]. PKMζ expression was increased in the IC 2 weeks after CTA conditioning - a finding that correlated with the long-term CTA memory [82]. Interestingly, application of the PKMζ inhibitor, ZIP into the AMY 15 min before US association, but not 30 min after CTA conditioning, attenuated CTA memory [84]. Taken together, these results reveal the importance of PKMζ in the early and late stages of consolidation of CTA memory in the IC; its role in the AMY may be related to the acquisition or early consolidation phase of CTA. Pharmacological studies showed that inhibition of PKC in the IC, AMY or PBN impaired long-term CTA memory formation, without affecting short-term CTA memory or attenuation of neophobia in the IC [34, 76, 77], which suggests that PKC plays a prominent role in formation of aversive-taste memory, but not of novel-taste memory.
PKA, a serine/threonine kinase composed of four regulatory and two catalytic subunit isoforms has been shown to be activated by cAMP. Although it has not been investigated whether novel taste and/or CTA influence the expression and activation of PKA, behavioral analyses that involved the targeted disruption of RIIβ subunits of PKA in mice, and pharmacological studies using PKA inhibitors have shown that the blockage of PKA activity in the IC or the AMY impaired long-term but not short-term CTA memory [85-88]. Further pharmacological study that microinjected cAMP analog, 8-bromo-adenosine 3'-5' cAMP, into the IC 30 minutes after novel taste consumption and immediately before US, has shown that increasing cAMP enhanced CTA memory [89]. These results demonstrate the functional importance of PKA during taste learning and memory consolidation.
CaMKII is a serine/threonine kinase that is a highly abundant protein in the post-synaptic density (PSD); it has a broad range of postsynaptic, membrane-bound and cytoplasmic substrates. Activation of CaMKIIα, an isoform of CaMKII, by Ca2+/CaM enables intra-molecular auto-phosphorylation of several sites, including threonine 286, 305 and 306. Auto-phosphorylation of CaMKIIα on threonine 286 enables the subsequent dissociation of bound Ca2+/CaM, thereby prolonging CaMKIIα activation, which enables partial activity even after full dissociation of Ca2+/CaM. Thus, CaMKIIα can translate a transient event of elevation of intracellular Ca2+ into a prolonged change that is independent of that transient change [90, 91]. The roles of CaMKIIα in synaptic plasticity and in various learning tasks have been intensively investigated [90, 92-96], but very little is known regarding the role of CaMKIIα in taste learning. Biochemically, novel-taste consumption was shown to increase expression of CaMKIIα protein in the IC [50]. It is important to note that in vitro studies of the enzyme activity that followed CTAhave shown that it attenuated the activity of CaMKIIα in the PBN [97]. However, a pharmacological study with a classical CaMKIIα inhibitor, KN-62 has shown that inhibition of CaMKIIα in the PBN following noveltaste and/or CTA elicited CTA [98]. Clearly, many more studies are needed to determine whether the activation of CaMKIIα is necessary for taste learning and memory and, if so, what is the nature of its role(s) in these processes.
Although the functions of protein phosphorylation by protein kinases in learning and memory have been studied extensively, recent studies also highlighted the importance of dephosphorylation by protein phosphatases. Protein phosphatases (PP) can be grouped into three main classes, according to sequence, structure, and catalytic function [99]. Phosphoprotein phosphatases constitute the largest family, comprising PP1, PP2A, PP2B (also known as PP3), PP4, PP5, PP6, and PP7 and the Mg2+-/Mn2+-dependent (PPM) family, designated PP2C [99]. The superfamiliesof protein Tyr phosphatases and the aspartate-based protein phosphatases, respectively, form the second and third classes [100]. The importance and the functional role of PP1/2A in several forms of learning and memory have been documented and, in addition, recent findings also showed the importance of PP1/2A in LTP and LTD [101-108]. Decreased activity of calcineurin (PP3) was observed selectively in the AMY 40 min after second CTA extinction, whereas PP1 remained unchanged [109]. Conditional transgenic mice with genetically inhibited calcineurin showed improved CTA memory and enhanced resistance to CTA extinction [109]. Conversely, conditional transgenic mice with increased calcineurin activity showed impaired memory and substantially faster CTA extinction. However, the inhibition or over-expression of calcineurin that followed the CTA training appeared not to affect the CTA memory, which suggests that calcineurin plays a major role during the CTA acquisition [109]. In support of these transgenic animal findings, a recent pharmacological study using okadaic acid - a potent inhibitor of PP1 and PP2A phosphatases - showed that inhibition of PP1 and PP2A enhanced the CTA memory if injected locally into the AMY 5 min before, but not after CTA acquisition [110].
MEMBRANE RECEPTORS
NMDA receptors are composed of GluN1, GluN2 (GluN2A-D), and GluN3 (GluN3A-B) subunits. Although the interpretation is not yet definitive, a large body of evidence supports a tetrameric arrangement of NMDA receptors, incorporating two GluN1 and two GluN2 subunits of the same or different subtypes [111-114]. GluN3 co-assembles with GluN1 and GluN2 to form ternary GluN1/ GluN2/ GluN3 tetrameric complexes, and GluN3 has also been hypothesized to serve as an alternative for one of the GluN2 subunits [114-117]. The available evidence indicates that NMDAR modulation is mediated through phosphorylation of intracellular loops of its subunits. Activation of PKA, PKC, Fyn (of the Src family) kinase, and CaMKIIα has been shown to phosphorylate NMDAR subunits. The first indication that the NMDAR undergoes phosphorylation following taste learning arose from the finding that the net tyrosine phosphorylation was enhanced in several proteins in the rat IC following novel-taste and CTA learning [118]. A subsequent study demonstrated that consumption of a novel taste led to increased tyrosine phosphorylation of the GluN2B subunit of the NMDA receptor in the IC, and that it returned to baseline levels once the taste became familiar [119]. An important finding was that this tyrosine phosphorylation wasindependent of NMDAR activity, as indicated by the observation that local application of the NMDAR antagonist,2-amino-5-phosphonovaleric acid (APV), did not affect the tyrosine phosphorylation [119]. However, local injection of the muscarinic acetylcholine receptors (AChR) agonist, carbachol into the IC mimicked this tyrosine phosphorylation [120], and release of ACh in the IC following novel-taste consumption has also been observed [121, 122]. Nevertheless, it remains to be determined whether the prolonged endogenous release of ACh is the physiological reason for tyrosine phosphorylation of GluN2B. It is noteworthy that the release of dopamine was observed following both novel-taste learningand CTA, whereas glutamate release was observed following only CTA, but not after novel-taste consumption [88]. More specifically, recent findings have shown a correlation between Fyn kinase-dependent increase in tyrosine phosphorylation on residue 1472 of GluN2B and novel-taste learning [123]. Moreover, this increased tyrosine 1472 phosphorylation of GluN2B reinforced the association of this NMDAR subunit with PSD95, a highly abundant PSD scaffolding protein that has crucial importance for learning and memory processes. In addition, local injection of the tyrosine kinase inhibitor, genistein impaired novel-taste memory in the LI-CTA paradigm [123], and a correlative increase in PSD-95 expression was observed after novel-taste learning [124].
As was observed in connection with the enhanced general tyrosine phosphorylation of GluN2B that followed novel-taste learning and CTA [118, 119], novel-taste learning also has been shown to increase the general serine phosphorylation of GluN2B. However, this phosphorylation diminished once the taste became familiar [125], which was not observed in connection with the tyrosine phosphorylation of this protein. Although PKC and CaMKIIα were shown to phosphorylate GluN2B at different sites on serine residues [126, 127], it is not clear whether PKC/CaMKIIα mediates this GluN2B serine phosphorylation. Therefore, it will be intriguing to check which of the serine/threonine protein kinases phosphorylates this observed GluN2B phosphorylation that followsnovel-taste learning.
The activity of NMDAR can also be mediated by its spatial localization. Studies have shown that relocalization of NMDAR followed LTP and inhibitory-avoidance learning [128, 129]. Novel-taste learning increased GluN2A and GluN2B in the detergent-insoluble fraction (130), and another study showed that novel-taste learning decreased the expressions of GluN1, GluN2A, and the GluN2B subunits of NMDAR in the synaptoneurosomal preparation [123]. These differences in NMDAR subunits expression from different studies could be attributed to the differences in the fractionation protocol and the sampling time following novel taste learning. The binding of phosphorylated CaMKIIα to the overexpressed cytoplasmic (C-) tail of the GluN2B in mutant mice that overexpressed C-tail affected its biding to the endogenous GluN2B receptor complex. This CaMKIIα-GluN2B complex formation also was associated with changes in the phosphorylation of the GluA1 subunit of α-amino-3-hydroxy-5-methyl-isoxazole-4-propionic acid receptor (AMPAR), and the spatial performance of these mutant mice was impaired, according to Morris water maze analysis, which suggests that localization of GluN2B is essential for mediating the downstream signaling cascade that leads to activation of learning-related protein changes [95]. Thus, it is very important to determine precisely the movement/localization of NMDAR, and whether phosphorylation impacts the movement/localization process.
AMPAR comprises hetero-tetrameric complexes composed of at least two of four subunits designated glutamate receptor GluA1-4 [131, 132]. The cytoplasmic tail of the GluA1 subunit undergoes distinct phosphorylation on serine 831 and dephosphorylation on serine 845 following LTP and long-term depression (LTD), respectively [133]. Functionally, phosphorylation on serine 831 and serine 845 is involved in regulation of AMPAR channel properties such as modulation of single-channel conductance and total and surface expressions of GluA1 [134]. Following inhibitory avoidance training - an associative learning paradigm wherein animals learn to associate the dark side of a chamber with a foot-shock and therefore avoid the dark side in successive trials - the increased phosphorylation of serine 831 of GluA1 has been observed in the dorsal HIP, in addition to increased expression levels of total GluA1 and GluA2 proteins [135]. Moreover, spatial learning is also associated with the phosphorylation of GluA1, as demonstrated with serine 831/Serine 845 double mutant mice in the water maze, an HIP-dependent spatial-learning paradigm [136]. Taken together, these findings suggest that phosphorylation of AMPAR and its synaptic incorporation are involved in these aspects of learned behaviors. The application of the AMPAR antagonist, NBQX directly into the IC impaired both acquisition and retrieval of taste memory [71], which suggests that the AMPA receptor plays a major role in taste recognition and learning. As discussed above, PKMζ is essential for CTA memory [80-82] and, interestingly, another independent study based on a fear conditioning paradigm has shown that PKMζ maintains memories by regulating GluA2-dependent AMPA receptor trafficking [137]. Further studies are required, to elucidate whether PKMζ also maintains CTA memory by regulating the GluA2 subunit. Currently, very little is known about the regulation of AMPAR in the context of taste learning and memory, therefore, further studies are needed to understand the regulation of AMPAR with respect to taste learning.
Tropomyosin receptor kinase B (TrkB) is a membrane receptor tyrosine kinase activated by BDNF and neurotrophin 4. There are two tyrosine phosphorylation residues outside the kinase activation domain of TrkB: tyrosine 515 and tyrosine 785. The binding of BDNF to its high-affinity receptor TrkB leads to the dimerization and autophosphorylation of TrkB on tyrosine residues in the intracellular domains, which then triggers one of three signaling cascades, depending on the site of phosphorylation; they are MAPK, phosphatidylinositol 3-kinase (PI3K), and phospholipase Cγ (PLCγ) (Yoshii and Constantine-Paton [138]). Ma et al. [33] used immunoprecipitation followed by immunoblotting with anti-tyrosine antibody to show that CTA conditioning increased the tyrosine phosphorylation of TrkB, and that the increase in TrkB phosphorylation was prolonged; it was detected 1 and 8 h following conditioning in both the IC and the CeA. In causality experiments. Ma et al. [33] have shown that inhibition of Trk receptors impaired both short-term and long-term CTA memory. BDNF-TrkB signaling is involved in transcription, translation and trafficking of proteins during various phases of synaptic development, and has been associated with several forms of synaptic plasticity and memory [138, 139]. As discussed above, CTA conditioning has been shown to increase the secretion and synthesis of BDNF in CeA and IC; and Ma et al. [33] have observed increased phosphorylation of the BDNF receptor, TrkB at the time points before and after the increase of BDNF synthesis in the CeA and IC, which suggests that CTA learning first induced the rapid release of pre-existing BDNF, and then increased BDNF synthesis.
Pharmacological analyses that used anti-BDNF antibodies and a BDNF antisense oligonucleotide, which, respectively, neutralize the endogenously released BDNF and block de novo protein synthesis, have found that inhibition of BDNF in the CeA significantly impaired CTA [33]. Moreover, exogenous application of BDNF into the IC has been shown to reverse the CTA memory deficits caused by the anisomycin-mediated inhibition of protein synthesis [140]. It is consistent with the previous findings that a recent study has also shown that the injection of BDNF immediately after anisomycin was applied 5 and 7 h following CTA conditioning reversed the anisomycin-mediated CTA impairment [47]; micro-infusion of BDNF into the IC was also shown to enhance CTA retention [141]. These results are clear evidence that BDNF signaling is necessary for the early and late consolidation stages of CTA. Importantly, recent findings have shown this BDNF-mediated enhanced CTA retention to depend on the activation of MAPK-ERKI/II and phatidylinositol-3-kinase (PI3K) [142], which suggests that TrkB-BDNF signaling following CTA acts via both the MAPK-ERKI/II cascade and the PI3K cascade.
FUTURE DIRECTIONS
In the mammalian CNS cells that perform a given function or share common functional properties are oft en clustered together [6]. Recent studies based on sophisticated methods of optical imaging of intrinsic signals indicated the presence within the IC of discrete clusters of neurons, which responded selectively to different taste modalities [143-145]. Moreover, a functional magnetic resonance imaging (fMRI) study has shown that in humans different taste modalities elicited differing specific patterns, with some overlap [146]. However, neither of these imaging techniques currently has the power to provide high-precision resolutionboth in time and at the cellular spatialscale in intact mammalian neural tissue. Nevertheless, it is reasonable to assume that clusters of neurons responding to single taste modalities might contain more narrowly tuned neurons. Moreover, as shown in Fig. 1, novel-taste consumption and CTA activate many intracellular signaling cascades in the IC and the AMY, but it is not known whether these cascades occur within a single cell, within narrowly tuned clusters of cells, or in different populations of cells. Therefore, techniques with higher cellular and spatial resolution of single neurons need to be developed, to investigate the molecular signaling cascades that underlie taste learning and memory. The recent development of optogenetic methods has rendered feasible investigations of specific cell types in the nervous system, with unprecedented precision and control [147, 148]. Thus, combinations of optogenetics with laser capture microdissection and genetic manipulation, followed by use of mRNA/protein arrays, liquid chromatography-mass spectroscopy, and bioinformatics tools or with new imaging techniques might provide better tools for such as yet unanswered questions.
CONCLUDING REMARKS
For several decades it was believed that consolidation of memory, a progressive process of changes and stabilization, was a unitary process. However, findings from the recent decade demonstrated that memories restabilize over time [149-151] and thatconsolidation of memory evolves over time, which suggests that memories change with time [151]. Interestingly, it was shown that, in the absence of negative visceral consequences, the strength of novel-taste memory stabilizes over time [152]. Despite the differing taste-learning models, available literature on taste learning appears to support the concept that the post-acquisition phase involves activation of waves of several biochemical pathways, and that the reactivation of particular pathways is required for consolidation. As discussed in the present review, several biochemical changes take place within the first hour after taste learning, and these are followed by other waves of activity during the subsequent few hours, which suggests a temporal segregation of signaling cascades that subservetaste-memory consolidation. Novel taste and associations of novel taste with malaise differentially release the aforementioned neurotransmitters in the IC and the AMY and, accordingly, waves of kinase activation also have been observed. Furthermore, novel-taste learning induces two waves of activation of the mTOR and S6K, respectively, which suggests that independent rounds of translation activation that lead to increased expression of proteins are required for taste-memory consolidation. Ample evidence indicates that the activation of NMDAR, TrkB, protein kinases, transcription factors, and translation regulators, all of which play roles in synaptic plasticity are also involved in taste learning and in memory formation. The available information regarding the modulation of neurotransmitter receptor response, pluripotent kinase signaling cascades, gene expression, translation regulation and translation by phosphorylation processes has presented opportunities for understanding taste learning and memory formation.
References
- 1.Garcia J, Kimeldorf DJ, Koelling RA. Conditioned aversion to saccharin resulting from exposure to gamma radiation. Science. 1955;122:157–158. [PubMed] [Google Scholar]
- 2.Bures J, Bermúdez-Rattoni F, Yamamoto Y. Conditioned taste aversion: memory of a special kind. Oxford, New York: Oxford University Press; 1998. [Google Scholar]
- 3.Gal-Ben-Ari S, Rosenblum K. Molecular mechanisms underlying memory consolidation of taste information in the cortex. Front Behav Neurosci. 2011;5:87. doi: 10.3389/fnbeh.2011.00087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rosenblum K, Meiri N, Dudai Y. Taste memory: the role of protein synthesis in gustatory cortex. Behav Neural Biol. 1993;59:49–56. doi: 10.1016/0163-1047(93)91145-d. [DOI] [PubMed] [Google Scholar]
- 5.Rosenblum K. Conditioned taste aversion and taste learning: molecular mechanisms. In: Byrne JH, editor. Learning and memory: a comprehensive reference. Amsterdam: Elsevier; 2008. pp. 217–234. [Google Scholar]
- 6.Carleton A, Accolla R, Simon SA. Coding in the mammalian gustatory system. Trends Neurosci. 2010;33:326–334. doi: 10.1016/j.tins.2010.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Núñez-Jaramillo L, Ramírez-Lugo L, Herrera-Morales W, Miranda MI. Taste memory formation: latest advances and challenges. Behav Brain Res. 2010;207:232–248. doi: 10.1016/j.bbr.2009.10.040. [DOI] [PubMed] [Google Scholar]
- 8.Mark GP, Blander DS, Hoebel BG. A conditioned stimulus decreases extracellular dopamine in the nucleus accumbens after the development of a learned taste aversion. Brain Res. 1991;551:308–310. doi: 10.1016/0006-8993(91)90946-s. [DOI] [PubMed] [Google Scholar]
- 9.Tassoni G, Lorenzini CA, Baldi E, Sacchetti B, Bucherelli C. Role of the perirhinal cortex in rats' conditioned taste aversion response memorization. Behav Neurosci. 2000;114:875–881. [PubMed] [Google Scholar]
- 10.Hernádi I, Karádi Z, Vigh J, Petykó Z, Egyed R, Berta B, Lénárd L. Alterations of conditioned taste aversion after microiontophoretically applied neurotoxins in the medial prefrontal cortex of the rat. Brain Res Bull. 2000;53:751–758. doi: 10.1016/s0361-9230(00)00361-0. [DOI] [PubMed] [Google Scholar]
- 11.Mickley GA, Kenmuir CL, Yocom AM, Wellman JA, Biada JM. A role for prefrontal cortex in the extinction of a conditioned taste aversion. Brain Res. 2005;1051:176–182. doi: 10.1016/j.brainres.2005.05.033. [DOI] [PubMed] [Google Scholar]
- 12.Akirav I, Khatsrinov V, Vouimba RM, Merhav M, Ferreira G, Rosenblum K, Maroun M. Extinction of conditioned taste aversion depends on functional protein synthesis but not on NMDA receptor activation in the ventromedial prefrontal cortex. Learn Mem. 2006;13:254–258. doi: 10.1101/lm.191706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ferreira G, Ferry B, Meurisse M, Lévy F. Forebrain structures specifically activated by conditioned taste aversion. Behav Neurosci. 2006;120:952–962. doi: 10.1037/0735-7044.120.4.952. [DOI] [PubMed] [Google Scholar]
- 14.Ramírez-Lugo L, Zavala-Vega S, Bermúdez-Rattoni F. NMDA and muscarinic receptors of the nucleus accumbens have differential effects on taste memory formation. Learn Mem. 2006;13:45–51. doi: 10.1101/lm.103206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yasoshima Y, Scott TR, Yamamoto T. Memory-dependent c-Fos expression in the nucleus accumbens and extended amygdala following the expression of a conditioned taste aversive in the rat. Neuroscience. 2006;141:35–45. doi: 10.1016/j.neuroscience.2006.03.019. [DOI] [PubMed] [Google Scholar]
- 16.De la Cruz V, Rodriguez-Ortiz CJ, Balderas I, Bermudez-Rattoni F. Medial temporal lobe structures participate differentially in consolidation of safe and aversive taste memories. Eur J Neurosci. 2008;28:1377–1381. doi: 10.1111/j.1460-9568.2008.06432.x. [DOI] [PubMed] [Google Scholar]
- 17.Solomon PR, Moore JW. Latent inhibition and stimulus generalization of the classically conditioned nictitating membrane response in rabbits (Oryctolagus cuniculus) following dorsal hippocampal ablation. J Comp Physiol Psychol. 1975;89:1192–1203. doi: 10.1037/h0077183. [DOI] [PubMed] [Google Scholar]
- 18.Krane RV, Sinnamon HM, Thomas GJ. Conditioned taste aversions and neophobia in rats with hippocampal lesions. J Comp Physiol Psychol. 1976;90:680–693. doi: 10.1037/h0077236. [DOI] [PubMed] [Google Scholar]
- 19.Miller JS, Nonneman AJ, Kelly KS, Neisewander JL, Isaac WL. Disruption of neophobia, conditioned odor aversion, and conditioned taste aversion in rats with hippocampal lesions. Behav Neural Biol. 1986;45:240–253. doi: 10.1016/s0163-1047(86)90816-2. [DOI] [PubMed] [Google Scholar]
- 20.Reilly S, Harley C, Revusky S. Ibotenate lesions of the hippocampus enhance latent inhibition in conditioned taste aversion and increase resistance to extinction in conditioned taste preference. Behav Neurosci. 1993;107:996–1004. doi: 10.1037//0735-7044.107.6.996. [DOI] [PubMed] [Google Scholar]
- 21.Gallo M, Cándido A. Dorsal hippocampal lesions impair blocking but not latent inhibition of taste aversion learning in rats. Behav Neurosci. 1995;109:413–425. doi: 10.1037//0735-7044.109.3.413. [DOI] [PubMed] [Google Scholar]
- 22.Yamamoto T, Fujimoto Y, Shimura T, Sakai N. Conditioned taste aversion in rats with excitotoxic brain lesions. Neurosci Res. 1995;22:31–49. doi: 10.1016/0168-0102(95)00875-t. [DOI] [PubMed] [Google Scholar]
- 23.Stone ME, Grimes BS, Katz DB. Hippocampal inactivation enhances taste learning. Learn Mem. 2005;12:579–586. doi: 10.1101/lm.32305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Garcia-Delatorre P, Rodríguez-Ortiz CJ, Balderas I, Bermúdez-Rattoni F. Differential participation of temporal structures in the consolidation and reconsolidation of taste aversion extinction. Eur J Neurosci. 2010;32:1018–1023. doi: 10.1111/j.1460-9568.2010.07365.x. [DOI] [PubMed] [Google Scholar]
- 25.Yefet K, Merhav M, Kuulmann-Vander S, Elkobi A, Belelovsky K, Jacobson-Pick S, Meiri N, Rosenblum K. Different signal transduction cascades are activated simultaneously in the rat insular cortex and hippocampus following novel taste learning. Eur J Neurosci. 2006;24:1434–1442. doi: 10.1111/j.1460-9568.2006.05009.x. [DOI] [PubMed] [Google Scholar]
- 26.Sheng M, Thompson MA, Greenberg ME. CREB: a Ca2+-regulated transcription factor phosphorylated by calmodulin-dependent kinases. Science. 1991;252:1427–1430. doi: 10.1126/science.1646483. [DOI] [PubMed] [Google Scholar]
- 27.Impey S, Obrietan K, Wong ST, Poser S, Yano S, Wayman G, Deloulme JC, Chan G, Storm DR. Cross talk between ERK and PKA is required for Ca2+ stimulation of CREB-dependent transcription and ERK nuclear translocation. Neuron. 1998;21:869–883. doi: 10.1016/s0896-6273(00)80602-9. [DOI] [PubMed] [Google Scholar]
- 28.Alberini CM. Transcription factors in long-term memory and synaptic plasticity. Physiol Rev. 2009;89:121–145. doi: 10.1152/physrev.00017.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Swank MW. Phosphorylation of MAP kinase and CREB in mouse cortex and amygdala during taste aversion learning. Neuroreport. 2000;11:1625–1630. doi: 10.1097/00001756-200006050-00006. [DOI] [PubMed] [Google Scholar]
- 30.Desmedt A, Hazvi S, Dudai Y. Differential pattern of cAMP response element-binding protein activation in the rat brain after conditioned aversion as a function of the associative process engaged: taste versus context association. J Neurosci. 2003;23:6102–6110. doi: 10.1523/JNEUROSCI.23-14-06102.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sheng M, McFadden G, Greenberg ME. Membrane depolarization and calcium induce c-fos transcription via phosphorylation of transcription factor CREB. Neuron. 1990;4:571–582. doi: 10.1016/0896-6273(90)90115-v. [DOI] [PubMed] [Google Scholar]
- 32.Finkbeiner S, Tavazoie SF, Maloratsky A, Jacobs KM, Harris KM, Greenberg ME. CREB: a major mediator of neuronal neurotrophin responses. Neuron. 1997;19:1031–1047. doi: 10.1016/s0896-6273(00)80395-5. [DOI] [PubMed] [Google Scholar]
- 33.Ma L, Wang DD, Zhang TY, Yu H, Wang Y, Huang SH, Lee FS, Chen ZY. Region-specific involvement of BDNF secretion and synthesis in conditioned taste aversion memory formation. J Neurosci. 2011;31:2079–2090. doi: 10.1523/JNEUROSCI.5348-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yamamoto T, Sako N, Sakai N, Iwafune A. Gustatory and visceral inputs to the amygdala of the rat: conditioned taste aversion and induction of c-fos-like immunoreactivity. Neurosci Lett. 1997;226:127–130. doi: 10.1016/s0304-3940(97)00265-6. [DOI] [PubMed] [Google Scholar]
- 35.Wilkins EE, Bernstein IL. Conditioning method determines patterns of c-fos expression following novel taste-illness pairing. Behav Brain Res. 2006;169:93–97. doi: 10.1016/j.bbr.2005.12.006. [DOI] [PubMed] [Google Scholar]
- 36.Doron G, Rosenblum K. c-Fos expression is elevated in GABAergic interneurons of the gustatory cortex following novel taste learning. Neurobiol Learn Mem. 2010;94:21–29. doi: 10.1016/j.nlm.2010.03.003. [DOI] [PubMed] [Google Scholar]
- 37.Lamprecht R, Dudai Y. Transient expression of c-Fos in rat amygdala during training is required for encoding conditioned taste aversion memory. Learn Mem. 1996;3:31–41. doi: 10.1101/lm.3.1.31. [DOI] [PubMed] [Google Scholar]
- 38.Lamprecht R, Hazvi S, Dudai Y. cAMP response element-binding protein in the amygdala is required for long- but not short-term conditioned taste aversion memory. J Neurosci. 1997;17:8443–8450. doi: 10.1523/JNEUROSCI.17-21-08443.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Josselyn SA, Kida S, Silva AJ. Inducible repression of CREB function disrupts amygdala-dependent memory. Neurobiol Learn Mem. 2004;82:159–163. doi: 10.1016/j.nlm.2004.05.008. [DOI] [PubMed] [Google Scholar]
- 40.Taubenfeld SM, Milekic MH, Monti B, Alberini CM. The consolidation of new but not reactivated memory requires hippocampal C/EBPbeta. Nat Neurosci. 2001;4:813–818. doi: 10.1038/90520. [DOI] [PubMed] [Google Scholar]
- 41.Taubenfeld SM, Wiig KA, Monti B, Dolan B, Pollonini G, Alberini CM. Fornix-dependent induction of hippocampal CCAAT enhancer-binding protein [beta] and [delta] Co-localizes with phosphorylated cAMP response element-binding protein and accompanies long-term memory consolidation. J Neurosci. 2001;21:84–91. doi: 10.1523/JNEUROSCI.21-01-00084.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Milekic MH, Pollonini G, Alberini CM. Temporal requirement of C/EBPbeta in the amygdala following reactivation but not acquisition of inhibitory avoidance. Learn Mem. 2007;14:504–511. doi: 10.1101/lm.598307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Merhav M, Kuulmann-Vander S, Elkobi A, Jacobson-Pick S, Karni A, Rosenblum K. Behavioral interference and C/EBPbeta expression in the insular-cortex reveal a prolonged time period for taste memory consolidation. Learn Mem. 2006;13:571–574. doi: 10.1101/lm.282406. [DOI] [PubMed] [Google Scholar]
- 44.Swank MW, Sweatt JD. Increased histone acetyltransferase and lysine acetyltransferase activity and biphasic activation of the ERK/RSK cascade in insular cortex during novel taste learning. J Neurosci. 2001;21:3383–3391. doi: 10.1523/JNEUROSCI.21-10-03383.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Berman DE, Dudai Y. Memory extinction, learning anew, and learning the new: dissociations in the molecular machinery of learning in cortex. Science. 2001;291:2417–2419. doi: 10.1126/science.1058165. [DOI] [PubMed] [Google Scholar]
- 46.Merhav M, Rosenblum K. Facilitation of taste memory acquisition by experiencing previous novel taste is protein-synthesis dependent. Learn Mem. 2008;15:501–507. doi: 10.1101/lm.986008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Martínez-Moreno A, Rodríguez-Durán LF, Escobar ML. Late protein synthesis-dependent phases in CTA long-term memory: BDNF requirement. Front Behav Neurosci. 2011;5:61. doi: 10.3389/fnbeh.2011.00061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Proud C. Control of the elongation phase of protein synthesis. In: Sonenberg N, Hershey J, Mathews M, editors. Translational control of gene expression. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory Press; 2000. pp. 719–739. [Google Scholar]
- 49.Sonenberg N, Hinnebusch AG. Regulation of translation initiation in eukaryotes: mechanisms and biological targets. Cell. 2009;136:731–745. doi: 10.1016/j.cell.2009.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Belelovsky K, Elkobi A, Kaphzan H, Nairn AC, Rosenblum K. A molecular switch for translational control in taste memory consolidation. Eur J Neurosci. 2005;22:2560–2568. doi: 10.1111/j.1460-9568.2005.04428.x. [DOI] [PubMed] [Google Scholar]
- 51.Holz MK, Ballif BA, Gygi SP, Blenis J. mTOR and S6K1 mediate assembly of the translation preinitiation complex through dynamic protein interchange and ordered phosphorylation events. Cell. 2005;123:569–580. doi: 10.1016/j.cell.2005.10.024. [DOI] [PubMed] [Google Scholar]
- 52.Nairn AC, Palfrey HC. Identification of the major Mr 100,000 substrate for calmodulin-dependent protein kinase III in mammalian cells as elongation factor-2. J Biol Chem. 1987;262:17299–17303. [PubMed] [Google Scholar]
- 53.Redpath NT, Proud CG. The tumour promoter okadaic acid inhibits reticulocyte-lysate protein synthesis by increasing the net phosphorylation of elongation factor 2. Biochem J. 1989;262:69–75. doi: 10.1042/bj2620069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Marin P, Nastiuk KL, Daniel N, Girault JA, Czernik AJ, Glowinski J, Nairn AC, Prémont J. Glutamate-dependent phosphorylation of elongation factor-2 and inhibition of protein synthesis in neurons. J Neurosci. 1997;17:3445–3454. doi: 10.1523/JNEUROSCI.17-10-03445.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hay N, Sonenberg N. Upstream and downstream of mTOR. Genes Dev. 2004;18:1926–1945. doi: 10.1101/gad.1212704. [DOI] [PubMed] [Google Scholar]
- 56.Belelovsky K, Kaphzan H, Elkobi A, Rosenblum K. Biphasic activation of the mTOR pathway in the gustatory cortex is correlated with and necessary for taste learning. J Neurosci. 2009;29:7424–7431. doi: 10.1523/JNEUROSCI.3809-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Terada N, Patel HR, Takase K, Kohno K, Nairn AC, Gelfand EW. Rapamycin selectively inhibits translation of mRNAs encoding elongation factors and ribosomal proteins. Proc Natl Acad Sci U S A. 1994;91:11477–11481. doi: 10.1073/pnas.91.24.11477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Jefferies HB, Fumagalli S, Dennis PB, Reinhard C, Pearson RB, Thomas G. Rapamycin suppresses 5'TOP mRNA translation through inhibition of p70s6k. EMBO J. 1997;16:3693–3704. doi: 10.1093/emboj/16.12.3693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Antion MD, Merhav M, Hoeffer CA, Reis G, Kozma SC, Thomas G, Schuman EM, Rosenblum K, Klann E. Removal of S6K1 and S6K2 leads to divergent alterations in learning, memory, and synaptic plasticity. Learn Mem. 2008;15:29–38. doi: 10.1101/lm.661908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Banko JL, Merhav M, Stern E, Sonenberg N, Rosenblum K, Klann E. Behavioral alterations in mice lacking the translation repressor 4E-BP2. Neurobiol Learn Mem. 2007;87:248–256. doi: 10.1016/j.nlm.2006.08.012. [DOI] [PubMed] [Google Scholar]
- 61.Costa-Mattioli M, Gobert D, Stern E, Gamache K, Colina R, Cuello C, Sossin W, Kaufman R, Pelletier J, Rosenblum K, Krnjević K, Lacaille JC, Nader K, Sonenberg N. eIF2alpha phosphorylation bidirectionally regulates the switch from short- to long-term synaptic plasticity and memory. Cell. 2007;129:195–206. doi: 10.1016/j.cell.2007.01.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ciechanover A. Intracellular protein degradation: from a vague idea thru the lysosome and the ubiquitin-proteasome system and onto human diseases and drug targeting. Exp Biol Med (Maywood) 2006;231:1197–1211. doi: 10.1177/153537020623100705. [DOI] [PubMed] [Google Scholar]
- 63.Bingol B, Sheng M. Deconstruction for reconstruction: the role of proteolysis in neural plasticity and disease. Neuron. 2011;69:22–32. doi: 10.1016/j.neuron.2010.11.006. [DOI] [PubMed] [Google Scholar]
- 64.Fioravante D, Byrne JH. Protein degradation and memory formation. Brain Res Bull. 2011;85:14–20. doi: 10.1016/j.brainresbull.2010.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hegde AN. The ubiquitin-proteasome pathway and synaptic plasticity. Learn Mem. 2010;17:314–327. doi: 10.1101/lm.1504010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lopez-Salon M, Alonso M, Vianna MR, Viola H, Mello e Souza T, Izquierdo I, Pasquini JM, Medina JH. The ubiquitin-proteasome cascade is required for mammalian long-term memory formation. Eur J Neurosci. 2001;14:1820–1826. doi: 10.1046/j.0953-816x.2001.01806.x. [DOI] [PubMed] [Google Scholar]
- 67.Artinian J, McGauran AM, De Jaeger X, Mouledous L, Frances B, Roullet P. Protein degradation, as with protein synthesis, is required during not only long-term spatial memory consolidation but also reconsolidation. Eur J Neurosci. 2008;27:3009–3019. doi: 10.1111/j.1460-9568.2008.06262.x. [DOI] [PubMed] [Google Scholar]
- 68.Lee SH, Choi JH, Lee N, Lee HR, Kim JI, Yu NK, Choi SL, Kim H, Kaang BK. Synaptic protein degradation underlies destabilization of retrieved fear memory. Science. 2008;319:1253–1256. doi: 10.1126/science.1150541. [DOI] [PubMed] [Google Scholar]
- 69.Rodriguez-Ortiz CJ, Balderas I, Saucedo-Alquicira F, Cruz-Castañeda P, Bermudez-Rattoni F. Long-term aversive taste memory requires insular and amygdala protein degradation. Neurobiol Learn Mem. 2011;95:311–315. doi: 10.1016/j.nlm.2010.12.010. [DOI] [PubMed] [Google Scholar]
- 70.Berman DE, Hazvi S, Rosenblum K, Seger R, Dudai Y. Specific and differential activation of mitogen-activated protein kinase cascades by unfamiliar taste in the insular cortex of the behaving rat. J Neurosci. 1998;18:10037–10044. doi: 10.1523/JNEUROSCI.18-23-10037.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Berman DE, Hazvi S, Neduva V, Dudai Y. The role of identified neurotransmitter systems in the response of insular cortex to unfamiliar taste: activation of ERK1-2 and formation of a memory trace. J Neurosci. 2000;20:7017–7023. doi: 10.1523/JNEUROSCI.20-18-07017.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kwon B, Houpt TA. Mitogen-activated protein kinase in the amygdala plays a critical role in lithium chloride-induced taste aversion learning. Neurobiol Learn Mem. 2012;97:132–139. doi: 10.1016/j.nlm.2011.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Jones MW, French PJ, Bliss TV, Rosenblum K. Molecular mechanisms of long-term potentiation in the insular cortex in vivo. J Neurosci. 1999;19:RC36. doi: 10.1523/JNEUROSCI.19-21-j0002.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Govindarajan A, Kelleher RJ, Tonegawa S. A clustered plasticity model of long-term memory engrams. Nat Rev Neurosci. 2006;7:575–583. doi: 10.1038/nrn1937. [DOI] [PubMed] [Google Scholar]
- 75.Yasoshima Y, Yamamoto T. Rat gustatory memory requires protein kinase C activity in the amygdala and cortical gustatory area. Neuroreport. 1997;8:1363–1367. doi: 10.1097/00001756-199704140-00009. [DOI] [PubMed] [Google Scholar]
- 76.Sacchetti B, Bielavska E. Chelerythrine, a specific PKC inhibitor, blocks acquisition but not consolidation and retrieval of conditioned taste aversion in rat. Brain Res. 1998;799:84–90. doi: 10.1016/s0006-8993(98)00460-0. [DOI] [PubMed] [Google Scholar]
- 77.Núñez-Jaramillo L, Delint-Ramirez I, Bermúdez-Rattoni F. PKC blockade differentially affects aversive but not appetitive gustatory memories. Brain Res. 2007;1148:177–182. doi: 10.1016/j.brainres.2007.02.032. [DOI] [PubMed] [Google Scholar]
- 78.Krivanek J. Conditioned taste aversion and protein kinase C in the parabrachial nucleus of rats. Neurobiol Learn Mem. 1996;65:154–162. doi: 10.1006/nlme.1996.0017. [DOI] [PubMed] [Google Scholar]
- 79.Krivanek J. Protein kinase C in the parabrachial nucleus of rats during conditioned taste aversion induced by amphetamine. Neurosci Lett. 1997;236:17–20. doi: 10.1016/s0304-3940(97)00737-4. [DOI] [PubMed] [Google Scholar]
- 80.Shema R, Sacktor TC, Dudai Y. Rapid erasure of long-term memory associations in the cortex by an inhibitor of PKM zeta. Science. 2007;317:951–953. doi: 10.1126/science.1144334. [DOI] [PubMed] [Google Scholar]
- 81.Shema R, Hazvi S, Sacktor TC, Dudai Y. Boundary conditions for the maintenance of memory by PKMzeta in neocortex. Learn Mem. 2009;16:122–128. doi: 10.1101/lm.1183309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Shema R, Haramati S, Ron S, Hazvi S, Chen A, Sacktor TC, Dudai Y. Enhancement of consolidated long-term memory by overexpression of protein kinase Mzeta in the neocortex. Science. 2011;331:1207–1210. doi: 10.1126/science.1200215. [DOI] [PubMed] [Google Scholar]
- 83.Edry E, Lamprecht R, Wagner S, Rosenblum K. Virally mediated gene manipulation in the adult CNS. Front Mol Neurosci. 2011;4:57. doi: 10.3389/fnmol.2011.00057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Gámiz F, Gallo M. Intra-amygdala ZIP injections impair the memory of learned active avoidance responses and attenuate conditioned taste-aversion acquisition in rats. Learn Mem. 2011;18:529–533. doi: 10.1101/lm.2253311. [DOI] [PubMed] [Google Scholar]
- 85.Koh MT, Thiele TE, Bernstein IL. Inhibition of protein kinase A activity interferes with long-term, but not short-term, memory of conditioned taste aversions. Behav Neurosci. 2002;116:1070–1074. [PubMed] [Google Scholar]
- 86.Koh MT, Bernstein IL. Inhibition of protein kinase A activity during conditioned taste aversion retrieval: interference with extinction or reconsolidation of a memory? Neuroreport. 2003;14:405–407. doi: 10.1097/00001756-200303030-00021. [DOI] [PubMed] [Google Scholar]
- 87.Koh MT, Clarke SN, Spray KJ, Thiele TE, Bernstein IL. Conditioned taste aversion memory and c-Fos induction are disrupted in RIIbeta-protein kinase A mutant mice. Behav Brain Res. 2003;143:57–63. doi: 10.1016/s0166-4328(03)00024-x. [DOI] [PubMed] [Google Scholar]
- 88.Guzmán-Ramos K, Osorio-Gómez D, Moreno-Castilla P, Bermúdez-Rattoni F. Off-line concomitant release of dopamine and glutamate involvement in taste memory consolidation. J Neurochem. 2010;114:226–236. doi: 10.1111/j.1471-4159.2010.06758.x. [DOI] [PubMed] [Google Scholar]
- 89.Miranda MI, McGaugh JL. Enhancement of inhibitory avoidance and conditioned taste aversion memory with insular cortex infusions of 8-Br-cAMP: involvement of the basolateral amygdala. Learn Mem. 2004;11:312–317. doi: 10.1101/lm.72804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Lisman J, Schulman H, Cline H. The molecular basis of CaMKII function in synaptic and behavioural memory. Nat Rev Neurosci. 2002;3:175–190. doi: 10.1038/nrn753. [DOI] [PubMed] [Google Scholar]
- 91.Wayman GA, Lee YS, Tokumitsu H, Silva AJ, Soderling TR. Calmodulin-kinases: modulators of neuronal development and plasticity. Neuron. 2008;59:914–931. doi: 10.1016/j.neuron.2008.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Rodrigues SM, Farb CR, Bauer EP, LeDoux JE, Schafe GE. Pavlovian fear conditioning regulates Thr286 autophosphorylation of Ca2+/calmodulin-dependent protein kinase II at lateral amygdala synapses. J Neurosci. 2004;24:3281–3288. doi: 10.1523/JNEUROSCI.5303-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Irvine EE, Vernon J, Giese KP. AlphaCaMKII autophosphorylation contributes to rapid learning but is not necessary for memory. Nat Neurosci. 2005;8:411–412. doi: 10.1038/nn1431. [DOI] [PubMed] [Google Scholar]
- 94.Irvine EE, Danhiez A, Radwanska K, Nassim C, Lucchesi W, Godaux E, Ris L, Giese KP. Properties of contextual memory formed in the absence of alphaCaMKII autophosphorylation. Mol Brain. 2011;4:8. doi: 10.1186/1756-6606-4-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Zhou Y, Takahashi E, Li W, Halt A, Wiltgen B, Ehninger D, Li GD, Hell JW, Kennedy MB, Silva AJ. Interactions between the NR2B receptor and CaMKII modulate synaptic plasticity and spatial learning. J Neurosci. 2007;27:13843–13853. doi: 10.1523/JNEUROSCI.4486-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Buard I, Coultrap SJ, Freund RK, Lee YS, Dell'Acqua ML, Silva AJ, Bayer KU. CaMKII "autonomy" is required for initiating but not for maintaining neuronal long-term information storage. J Neurosci. 2010;30:8214–8220. doi: 10.1523/JNEUROSCI.1469-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Krivanek J. Conditioned taste aversion and Ca/calmodulin-dependent kinase II in the parabrachial nucleus of rats. Neurobiol Learn Mem. 2001;76:46–56. doi: 10.1006/nlme.2000.3987. [DOI] [PubMed] [Google Scholar]
- 98.Sacchetti B, Baldi E, Tassoni G, Bielavska E. CAMKII inhibition in the parabrachial nuclei elicits conditioned taste aversion in rats. Neurobiol Learn Mem. 2001;75:253–261. doi: 10.1006/nlme.2000.3978. [DOI] [PubMed] [Google Scholar]
- 99.Tweedie-Cullen RY, Park CS, Mansuy IM. Protein phosphatases in the brain: regulation, function and disease. In: Vidal CJ, editor. Protein reviews: post-translational modifications in health and disease. Vol. 13. New York: Springer; 2011. pp. 233–257. [Google Scholar]
- 100.Moorhead GB, Trinkle-Mulcahy L, Ulke-Lemée A. Emerging roles of nuclear protein phosphatases. Nat Rev Mol Cell Biol. 2007;8:234–244. doi: 10.1038/nrm2126. [DOI] [PubMed] [Google Scholar]
- 101.Mulkey RM, Herron CE, Malenka RC. An essential role for protein phosphatases in hippocampal long-term depression. Science. 1993;261:1051–1055. doi: 10.1126/science.8394601. [DOI] [PubMed] [Google Scholar]
- 102.Blitzer RD, Wong T, Nouranifar R, Iyengar R, Landau EM. Postsynaptic cAMP pathway gates early LTP in hippocampal CA1 region. Neuron. 1995;15:1403–1414. doi: 10.1016/0896-6273(95)90018-7. [DOI] [PubMed] [Google Scholar]
- 103.Bennett PC, Zhao W, Ng KT. Concentration-dependent effects of protein phosphatase (PP) inhibitors implicate PP1 and PP2A in different stages of memory formation. Neurobiol Learn Mem. 2001;75:91–110. doi: 10.1006/nlme.1999.3959. [DOI] [PubMed] [Google Scholar]
- 104.He J, Yamada K, Zou LB, Nabeshima T. Spatial memory deficit and neurodegeneration induced by the direct injection of okadaic acid into the hippocampus in rats. J Neural Transm. 2001;108:1435–1443. doi: 10.1007/s007020100018. [DOI] [PubMed] [Google Scholar]
- 105.He J, Yang Y, Xu H, Zhang X, Li XM. Olanzapine attenuates the okadaic acid-induced spatial memory impairment and hippocampal cell death in rats. Neuropsychopharmacology. 2005;30:1511–1520. doi: 10.1038/sj.npp.1300757. [DOI] [PubMed] [Google Scholar]
- 106.Genoux D, Haditsch U, Knobloch M, Michalon A, Storm D, Mansuy IM. Protein phosphatase 1 is a molecular constraint on learning and memory. Nature. 2002;418:970–975. doi: 10.1038/nature00928. [DOI] [PubMed] [Google Scholar]
- 107.Peters M, Bletsch M, Catapano R, Zhang X, Tully T, Bourtchouladze R. RNA interference in hippocampus demonstrates opposing roles for CREB and PP1alpha in contextual and temporal long-term memory. Genes Brain Behav. 2009;8:320–329. doi: 10.1111/j.1601-183X.2009.00474.x. [DOI] [PubMed] [Google Scholar]
- 108.Koshibu K, Gräff J, Beullens M, Heitz FD, Berchtold D, Russig H, Farinelli M, Bollen M, Mansuy IM. Protein phosphatase 1 regulates the histone code for long-term memory. J Neurosci. 2009;29:13079–13089. doi: 10.1523/JNEUROSCI.3610-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Baumgärtel K, Genoux D, Welzl H, Tweedie-Cullen RY, Koshibu K, Livingstone-Zatchej M, Mamie C, Mansuy IM. Control of the establishment of aversive memory by calcineurin and Zif268. Nat Neurosci. 2008;11:572–578. doi: 10.1038/nn.2113. [DOI] [PubMed] [Google Scholar]
- 110.Oberbeck DL, McCormack S, Houpt TA. Intra-amygdalar okadaic acid enhances conditioned taste aversion learning and CREB phosphorylation in rats. Brain Res. 2010;1348:84–94. doi: 10.1016/j.brainres.2010.06.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Luo J, Wang Y, Yasuda RP, Dunah AW, Wolfe BB. The majority of N-methyl-D-aspartate receptor complexes in adult rat cerebral cortex contain at least three different subunits (NR1/NR2A/NR2B) Mol Pharmacol. 1997;51:79–86. doi: 10.1124/mol.51.1.79. [DOI] [PubMed] [Google Scholar]
- 112.Laube B, Kuhse J, Betz H. Evidence for a tetrameric structure of recombinant NMDA receptors. J Neurosci. 1998;18:2954–2961. doi: 10.1523/JNEUROSCI.18-08-02954.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Dingledine R, Borges K, Bowie D, Traynelis SF. The glutamate receptor ion channels. Pharmacol Rev. 1999;51:7–61. [PubMed] [Google Scholar]
- 114.Schüler T, Mesic I, Madry C, Bartholomäus I, Laube B. Formation of NR1/NR2 and NR1/NR3 heterodimers constitutes the initial step in N-methyl-D-aspartate receptor assembly. J Biol Chem. 2008;283:37–46. doi: 10.1074/jbc.M703539200. [DOI] [PubMed] [Google Scholar]
- 115.Perez-Otano I, Schulteis CT, Contractor A, Lipton SA, Trimmer JS, Sucher NJ, Heinemann SF. Assembly with the NR1 subunit is required for surface expression of NR3A-containing NMDA receptors. J Neurosci. 2001;21:1228–1237. doi: 10.1523/JNEUROSCI.21-04-01228.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Sasaki YF, Rothe T, Premkumar LS, Das S, Cui J, Talantova MV, Wong HK, Gong X, Chan SF, Zhang D, Nakanishi N, Sucher NJ, Lipton SA. Characterization and comparison of the NR3A subunit of the NMDA receptor in recombinant systems and primary cortical neurons. J Neurophysiol. 2002;87:2052–2063. doi: 10.1152/jn.00531.2001. [DOI] [PubMed] [Google Scholar]
- 117.Kew JN, Kemp JA. Ionotropic and metabotropic glutamate receptor structure and pharmacology. Psychopharmacology (Berl) 2005;179:4–29. doi: 10.1007/s00213-005-2200-z. [DOI] [PubMed] [Google Scholar]
- 118.Rosenblum K, Schul R, Meiri N, Hadari YR, Zick Y, Dudai Y. Modulation of protein tyrosine phosphorylation in rat insular cortex after conditioned taste aversion training. Proc Natl Acad Sci U S A. 1995;92:1157–1161. doi: 10.1073/pnas.92.4.1157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Rosenblum K, Berman DE, Hazvi S, Lamprecht R, Dudai Y. NMDA receptor and the tyrosine phosphorylation of its 2B subunit in taste learning in the rat insular cortex. J Neurosci. 1997;17:5129–5135. doi: 10.1523/JNEUROSCI.17-13-05129.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Rosenblum K, Berman DE, Hazvi S, Dudai Y. Carbachol mimics effects of sensory input on tyrosine phosphorylation in cortex. Neuroreport. 1996;7:1401–1404. doi: 10.1097/00001756-199605310-00015. [DOI] [PubMed] [Google Scholar]
- 121.Shimura T, Suzuki M, Yamamoto T. Aversive taste stimuli facilitate extracellular acetylcholine release in the insular gustatory cortex of the rat: a microdialysis study. Brain Res. 1995;679:221–226. doi: 10.1016/0006-8993(95)00225-f. [DOI] [PubMed] [Google Scholar]
- 122.Miranda MI, Ramírez-Lugo L, Bermúdez-Rattoni F. Cortical cholinergic activity is related to the novelty of the stimulus. Brain Res. 2000;882:230–235. doi: 10.1016/s0926-6410(00)00050-1. [DOI] [PubMed] [Google Scholar]
- 123.Barki-Harrington L, Elkobi A, Tzabary T, Rosenblum K. Tyrosine phosphorylation of the 2B subunit of the NMDA receptor is necessary for taste memory formation. J Neurosci. 2009;29:9219–9226. doi: 10.1523/JNEUROSCI.5667-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Elkobi A, Ehrlich I, Belelovsky K, Barki-Harrington L, Rosenblum K. ERK-dependent PSD-95 induction in the gustatory cortex is necessary for taste learning, but not retrieval. Nat Neurosci. 2008;11:1149–1151. doi: 10.1038/nn.2190. [DOI] [PubMed] [Google Scholar]
- 125.Jiménez B, Tapia R. Biochemical modulation of NMDA receptors: role in conditioned taste aversion. Neurochem Res. 2004;29:161–168. doi: 10.1023/b:nere.0000010445.27905.aa. [DOI] [PubMed] [Google Scholar]
- 126.Leonard AS, Hell JW. Cyclic AMP-dependent protein kinase and protein kinase C phosphorylate N-methyl-D-aspartate receptors at different sites. J Biol Chem. 1997;272:12107–12115. doi: 10.1074/jbc.272.18.12107. [DOI] [PubMed] [Google Scholar]
- 127.Bayer KU, De Koninck P, Leonard AS, Hell JW, Schulman H. Interaction with the NMDA receptor locks CaMKII in an active conformation. Nature. 2001;411:801–805. doi: 10.1038/35081080. [DOI] [PubMed] [Google Scholar]
- 128.Cammarota M, de Stein ML, Paratcha G, Bevilaqua LR, Izquierdo I, Medina JH. Rapid and transient learning-associated increase in NMDA NR1 subunit in the rat hippocampus. Neurochem Res. 2000;25:567–572. doi: 10.1023/a:1007590415556. [DOI] [PubMed] [Google Scholar]
- 129.Grosshans DR, Clayton DA, Coultrap SJ, Browning MD. LTP leads to rapid surface expression of NMDA but not AMPA receptors in adult rat CA1. Nat Neurosci. 2002;5:27–33. doi: 10.1038/nn779. [DOI] [PubMed] [Google Scholar]
- 130.Núñez-Jaramillo L, Jimenez B, Ramirez-Munguía N, Delint-Ramírez I, Luna-Illades C, Tapia R, Bermúdez-Rattoni F. Taste novelty induces intracellular redistribution of NR2A and NR2B subunits of NMDA receptor in the insular cortex. Brain Res. 2008;1215:116–122. doi: 10.1016/j.brainres.2008.03.032. [DOI] [PubMed] [Google Scholar]
- 131.Collingridge GL, Olsen RW, Peters J, Spedding M. A nomenclature for ligand-gated ion channels. Neuropharmacology. 2009;56:2–5. doi: 10.1016/j.neuropharm.2008.06.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Keifer J, Zheng Z. AMPA receptor trafficking and learning. Eur J Neurosci. 2010;32:269–277. doi: 10.1111/j.1460-9568.2010.07339.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Lee HK, Barbarosie M, Kameyama K, Bear MF, Huganir RL. Regulation of distinct AMPA receptor phosphorylation sites during bidirectional synaptic plasticity. Nature. 2000;405:955–959. doi: 10.1038/35016089. [DOI] [PubMed] [Google Scholar]
- 134.Newpher TM, Ehlers MD. Glutamate receptor dynamics in dendritic microdomains. Neuron. 2008;58:472–497. doi: 10.1016/j.neuron.2008.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Whitlock JR, Heynen AJ, Shuler MG, Bear MF. Learning induces long-term potentiation in the hippocampus. Science. 2006;313:1093–1097. doi: 10.1126/science.1128134. [DOI] [PubMed] [Google Scholar]
- 136.Lee HK, Takamiya K, Han JS, Man H, Kim CH, Rumbaugh G, Yu S, Ding L, He C, Petralia RS, Wenthold RJ, Gallagher M, Huganir RL. Phosphorylation of the AMPA receptor GluR1 subunit is required for synaptic plasticity and retention of spatial memory. Cell. 2003;112:631–643. doi: 10.1016/s0092-8674(03)00122-3. [DOI] [PubMed] [Google Scholar]
- 137.Migues PV, Hardt O, Wu DC, Gamache K, Sacktor TC, Wang YT, Nader K. PKMzeta maintains memories by regulating GluR2-dependent AMPA receptor trafficking. Nat Neurosci. 2010;13:630–634. doi: 10.1038/nn.2531. [DOI] [PubMed] [Google Scholar]
- 138.Yoshii A, Constantine-Paton M. Postsynaptic BDNF-TrkB signaling in synapse maturation, plasticity, and disease. Dev Neurobiol. 2010;70:304–322. doi: 10.1002/dneu.20765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Musumeci G, Minichiello L. BDNF-TrkB signalling in fear learning: from genetics to neural networks. Rev Neurosci. 2011;22:303–315. doi: 10.1515/RNS.2011.031. [DOI] [PubMed] [Google Scholar]
- 140.Moguel-González M, Gómez-Palacio-Schjetnan A, Escobar ML. BDNF reverses the CTA memory deficits produced by inhibition of protein synthesis. Neurobiol Learn Mem. 2008;90:584–587. doi: 10.1016/j.nlm.2008.06.003. [DOI] [PubMed] [Google Scholar]
- 141.Castillo DV, Figueroa-Guzmán Y, Escobar ML. Brain-derived neurotrophic factor enhances conditioned taste aversion retention. Brain Res. 2006;1067:250–255. doi: 10.1016/j.brainres.2005.10.085. [DOI] [PubMed] [Google Scholar]
- 142.Castillo DV, Escobar ML. A role for MAPK and PI-3K signaling pathways in brain-derived neurotrophic factor modification of conditioned taste aversion retention. Behav Brain Res. 2011;217:248–252. doi: 10.1016/j.bbr.2010.10.013. [DOI] [PubMed] [Google Scholar]
- 143.Accolla R, Bathellier B, Petersen CC, Carleton A. Differential spatial representation of taste modalities in the rat gustatory cortex. J Neurosci. 2007;27:1396–1404. doi: 10.1523/JNEUROSCI.5188-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Accolla R, Carleton A. Internal body state influences topographical plasticity of sensory representations in the rat gustatory cortex. Proc Natl Acad Sci U S A. 2008;105:4010–4015. doi: 10.1073/pnas.0708927105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Chen X, Gabitto M, Peng Y, Ryba NJ, Zuker CS. A gustotopic map of taste qualities in the mammalian brain. Science. 2011;333:1262–1266. doi: 10.1126/science.1204076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Schoenfeld MA, Neuer G, Tempelmann C, Schussler K, Noesselt T, Hopf JM, Heinze HJ. Functional magnetic resonance tomography correlates of taste perception in the human primary taste cortex. Neuroscience. 2004;127:347–353. doi: 10.1016/j.neuroscience.2004.05.024. [DOI] [PubMed] [Google Scholar]
- 147.Fenno L, Yizhar O, Deisseroth K. The development and application of optogenetics. Annu Rev Neurosci. 2011;34:389–412. doi: 10.1146/annurev-neuro-061010-113817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Kravitz AV, Kreitzer AC. Optogenetic manipulation of neural circuitry in vivo. Curr Opin Neurobiol. 2011;21:433–439. doi: 10.1016/j.conb.2011.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.McGaugh JL. Memory--a century of consolidation. Science. 2000;287:248–251. doi: 10.1126/science.287.5451.248. [DOI] [PubMed] [Google Scholar]
- 150.Dudai Y. The neurobiology of consolidations, or, how stable is the engram? Annu Rev Psychol. 2004;55:51–86. doi: 10.1146/annurev.psych.55.090902.142050. [DOI] [PubMed] [Google Scholar]
- 151.Alberini CM. The role of reconsolidation and the dynamic process of long-term memory formation and storage. Front Behav Neurosci. 2011;5:12. doi: 10.3389/fnbeh.2011.00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Gutiérrez R, Rodriguez-Ortiz CJ, De La Cruz V, Núñez-Jaramillo L, Bermudez-Rattoni F. Cholinergic dependence of taste memory formation: evidence of two distinct processes. Neurobiol Learn Mem. 2003;80:323–331. doi: 10.1016/s1074-7427(03)00066-2. [DOI] [PubMed] [Google Scholar]