Review of immunity to influenza virus infection in humans, noting evidence from murine studies and suggesting directions in which future research is most needed.
Keywords: influenza, human, immune response
Abstract
A clear understanding of immunity in individuals infected with influenza virus is critical for the design of effective vaccination and treatment strategies. Whereas myriad studies have teased apart innate and adaptive immune responses to influenza infection in murine models, much less is known about human immunity as a result of the ethical and technical constraints of human research. Still, these murine studies have provided important insights into the critical correlates of protection and pathogenicity in human infection and helped direct the human studies that have been conducted. Here, we examine and review the current literature on immunity in humans infected with influenza virus, noting evidence offered by select murine studies and suggesting directions in which future research is most warranted.
INFLUENZA VIRUS DISEASE
Influenza infection in humans can cause a wide range of disease symptoms, from asymptomatic to serious illness, characterized by sudden onset of fever, myalgia, and respiratory indications, including nonproductive cough, sore throat, and rhinitis. In addition, children infected with influenza commonly present with otitis media, nausea, and vomiting and more frequently, complain of rhinorrhea [1, 2]. Historically, the pediatric and elderly populations are considered vulnerable during influenza epidemics [3–6], and although still debated, studies suggest that children have a unique role in the spread of influenza during epidemics [7–9]. Children under 2 years old are more susceptible to secondary complications [10], and infants with severe respiratory tract viral infection are often at greater danger for pulmonary dysfunction, including wheezing, allergy, and asthma later in life [11–13]. In contrast, elderly individuals are at higher risk for developing hemorrhagic bronchitis, pneumonia, and death when infected with influenza [14], and this population generally has a less-robust immune response following influenza immunization.
Although RSV infections are the leading cause of hospitalizations of children in the United States, influenza accounts for the highest incidence of infection in children under 2 years of age [15]. Influenza epidemics occur annually and can cause significant morbidity and mortality worldwide. During 1976–2007, the CDC estimated that in the United States alone, annually, 8.5% (6309) of all “deaths with underlying pneumonia and influenza causes” (74,363) were influenza-associated, and 2.1% (23,607) of “deaths with underlying respiratory and circulatory causes” (1,132,319) were influenza-associated [16]. Furthermore, individuals with underlying respiratory and cardiac disease or diabetes mellitis, infected with influenza, are often at higher risk for developing hemorrhagic bronchitis or pneumonia and associated comorbidities. In 2008, it was estimated that there were 28,000–111,500 deaths attributable to influenza in children <5 years of age globally, with developing countries carrying the largest burden [17]. In the United States, annual influenza epidemics account for an estimated $10 billion (12% of the total economic burden) in direct medical costs [18] and an estimated $44–$163 million in hospitalization costs for children [19].
Introduction of a swine-origin influenza into the human population in 2009 quickly disseminated globally, causing the first pandemic of the 21st century. This novel H1N1 virus, hereafter referred to as A(H1N1)pdm09 [20], disproportionately affected children and young adults [21, 22]. Observed mortality increased between four and 10 times in the pediatric population during the initial wave of A(H1N1)pdm09 infection in 2009 compared with previous influenza seasons [23], whereas those individuals born prior to 1950 had pre-existing, cross-reactive antibodies against A(H1N1)pdm09 [24]. As a result, within the population of those with laboratory-confirmed A(H1N1)pdm09, 2% were older individuals (>60 years), whereas 79% were <30 years old. Risk factors for severe A(H1N1)pdm09 infection are similar to those for seasonal influenza but also include obesity. The majority of the severe or fatal cases have accompanying chronic illnesses, such as cardiac disease, chronic respiratory diseases, and diabetes [25]. Several studies suggest that pregnant and postpartum women are at increased risk for complications from infections with A(H1N1)pdm09 [26–28]. Furthermore, severity of A(H1N1)pdm09 infection was related to the length of time between symptom onset and the initiation of antiviral treatment with NA inhibitors [29].
Influenza infections are associated with secondary bacterial infections, or superinfections, most notably, Streptococcus pneumoniae and Staphylococcus aureus (reviewed in ref. [30]). In children, the most common secondary bacterial infections lead to acute otitis media, which is found in 50% of patients with symptomatic influenza infection, with five to six cases observed annually for every 100 children <2 years of age [10]. Since the emergence of A(H1N1)pdm09, approximately one-third of fatal infections are associated with bacterial coinfection [31, 32], but this number includes cases where respiratory support is needed, adding to the risk of secondary infections. In fatal cases of those infected by A(H1N1)pdm09, diffuse alveolar damage alone, diffuse alveolar damage associated with necrotizing bronchiolitis, and diffuse alveolar damage with hemorrhage were found, and in most of the cases where necrotizing bronchiolitis was found, the individuals had bacterial coinfections [33].
INFLUENZA IMMUNIZATION
Vaccination remains an effective and primary preventive strategy to avert influenza infection. The World Health Organization and CDC recommend that children and adults over the age of 6 months receive an influenza vaccination annually [34]. Currently, there are three licensed seasonal vaccines administered in the United States: 1) a TIV, administered by i.m. injection; 2) a LAIV, delivered intranasally; and 3) an i.d.-administered TIV preparation [35]. Each includes three circulating strains of influenza grown in eggs, reflecting annual surveillance data that predict which strains (A/H1, A/H3, and B) are most likely to be circulating the following season in northern and southern hemispheres. The i.m. TIV is approved for use in children and adults over 6 months of age, whereas the newer i.d. TIV is approved for adults aged 18–64 years. LAIV is approved for use in healthy children and adults between 2 and 49 years of age. The current influenza vaccines work to induce memory recall responses, primarily via humoral immune responses against the HA and NA surface glycoproteins, and HA inhibition antibody responses following immunization correlates with protection against infection [36, 37].
The main component standardized in influenza vaccines is the HA protein, and it is well-characterized in safety trials and challenge studies that the amount of HA within the vaccine correlates with level of protection elicited by immunization [38, 39]. Beginning during the northern hemisphere's 2010–2011 influenza season, a vaccine containing 60 μg HA/vaccine strain, as opposed to 15 μg in other preparations, was approved as an alternative TIV for elderly individuals (over age 65 years) [35]. Following vaccination, ASCs rapidly proliferate upon antigen exposure, and cell numbers peak 1 week postimmunization in healthy adults and children [40, 41]. IgG and IgA ASCs decline to low levels by 4–6 weeks postimmunization [42, 43]. This increase in the total numbers of ASCs corresponds to nAb levels postvaccination, peaking at 4 weeks in adults and children [43]. LAIV elicits a strong serum and mucosal influenza-specific antibody response [44], and studies in young children (6–59 months of age) showed that those receiving LAIV had significantly reduced incidence of influenza infection [45]. Although limited data exist, influenza-specific ASCs and nAb titers are increased to a greater degree following TIV immunization compared with LAIV vaccination [43, 46, 47], but the role of neutralization of LAIV occurring in antigen-experienced individuals is not well understood.
Influenza immunization is effective, and studies in healthy children <15 years of age have shown TIV efficacies ranging from 31% to 90% (reviewed in ref. [48]). Since 2010, universal influenza vaccination is recommended, i.e., for all individuals at least 6 months of age, but traditional efforts remain focused on those individuals at greatest risk of serious disease: young children, elderly, individuals with pulmonary or cardiovascular disorders, and those who are immunocompromised or pregnant [35]. Vaccination programs also elicit indirect benefits, including herd immunity, by which immunized individuals protect those who are immunocompromised or other nonvaccinated individuals [49, 50]. This effect was observed in a retrospective epidemiological study in Japan and the United States examining mortality and influenza vaccination rates between 1949 and 1998 [51]. As children have long been considered to have an important role in the spread of influenza during epidemics [7–9], Japanese officials legislated compulsory vaccination of school-aged children in 1977, and as the policy changed to optional immunization in 1987, it became clear that mortality rates were inversely correlated with overall vaccination coverage. However, recent reviews of vaccine studies in various populations have concluded that overestimation of the benefits of vaccination is common, particularly within the elderly population [52, 53], but studies that take into account selection biases do suggest that vaccination is associated with lower risk of hospitalization for pneumonia or influenza, as well as all-cause mortality [52, 54, 55]. Furthermore, in October 2009, the United States commenced a national influenza H1N1/2009 vaccination campaign, and it is estimated that ∼41.2% and 43% of the U.S. population received the 2009–2010 and 2010–2011 influenza vaccines, respectively [56]. Following implementation of this campaign, the frequency of positive influenza cultures reported to the CDC declined quickly. Immunization and herd immunity resulting from prior exposure of A(H1N1)pdm09 likely resulted in decreased cases of influenza during the 2010–2011 influenza season.
Undoubtedly, influenza vaccines can be improved, particularly for the most vulnerable populations. Continuous epidemiological surveillance of circulating influenza strains is required for keeping pace with antigenic drift and shift (reviewed in ref. [57]). Antigenic drift is an endless battle, whereby viral variants emerge as a result of point mutations within the viral genome, escaping antibody-mediated viral neutralization. Whereas many mutations occur throughout the viral genome, the point mutations within the HA or NA proteins can change antibody-binding domains, such that individuals no longer have adequate, protective B cell memory responses. In contrast, major qualitative changes in antigenicity as a result of antigenic shift occur as a result of genetic reassortment and replacement of one or more influenza gene segments. This, in turn, creates a new influenza virus subtype, to which little or no immunity may exist within the population. Antigenic shift remains a chief concern, in that highly pathogenic influenza viruses could potentially gain efficient human-to-human transmission. As influenza viruses frequently undergo antigenic drift and less frequently, shift, a desirable vaccine candidate would be able to induce highly specific, cross-reactive antibodies and T cell responses.
One strategy used in vaccine improvement includes the use of adjuvant to increase antigen uptake by APCs and enhance cytokine production. Neither of the U.S.-licensed TIVs nor LAIV include adjuvant, but there are several licensed vaccines available outside of the United States that use oil and water emulsions for seasonal influenza as well as for A/H5N1 (reviewed in ref. [58]). For instance, Novartis Vaccines and Diagnostics (Switzerland) manufactures Fluad using MF59 adjuvant, and GlaxoSmithKline Biologicals (Belgium) produces Pandemrix, which includes AS03. Fluad has been used in Europe since 1997 and was recently approved for use in Canada in adults 65 years and older. MF59 and AS03 are both squalene-based with surfactants included to stabilize the emulsions. Clinical trials of MF59-adjuvanted vaccines are well-tolerated and elicit higher antibody responses in adults [59–61] and in children [62–64] compared with unadjuvanted vaccine preparations.
The concept of a universal influenza vaccine, where one vaccine elicits protection against multiple influenza subtypes, is becoming a reality. The influenza M2 ion channel is expressed at high levels on the surface of influenza-infected cells and has a conserved ectodomain (M2e) that is 24 aa in length [65]. Animal studies have demonstrated improved viral clearance and cross-protection upon lethal challenge in mice following passive M2 mAb transfer or M2-immunized mice (reviewed in ref. [66]). Phase I clinical safety trials evaluating VAX102, manufactured by VaxInnate, have reported increased M2e antibody responses [67]. Creation of a universal influenza vaccine that targets conserved regions of the HA stem region is particularly appealing, and several groups have demonstrated encouraging results using broadly neutralizing mAb to regions of HA that are cross-reactive between Group 1 and Group 2 influenza strains [68–70]. Structural analyses indicate that these antibodies bind to conserved pockets of the A helix of the HA stalk, located below the trimeric head region [71–73]. It is therefore within the realm of possibility to focus immunization strategies to these conserved regions in the HA and M2 proteins to elicit effective antibody, and presumably T cell, responses in the case of emergent influenza viruses. Still, it remains a mystery as to why these conserved epitopes have not generated a high level of cross-protection in populations repeatedly immunized or infected with diverse viral strains.
IMMUNE RESPONSE TO INFLUENZA VIRUS INFECTION
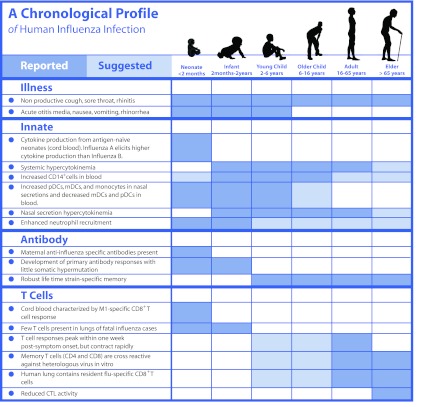
Factors that limit influenza vaccine efficacy in humans include age, immunocompetence, and pre-existing antibody levels from previous infections or immunizations. Whereas key lessons have been learned using animal models of influenza infection, studies done using human participants are invaluable for describing the unique and often complex characteristics of the human immune response to infection and immunization. Here, we describe central findings obtained from human influenza challenge studies, natural infections, and ex vivo characterization of human immune cells (Table 1). Figure 1 indicates where questions remain for the human immune response to infection.
Table 1. Important Findings from Human Studies of Influenza Virus Infection.
| Major findings | References |
|---|---|
| Human challenge studies | |
| Main correlate of protection is neutralizing antibodies against HA. | [37, 47, 74–79], reviewed in ref. [80] |
| Evidence for heterosubtypic immunity | |
| Pre-existing immunity from previous influenza exposure elicits protection against heterologous infections. | [81–88] |
| Broadly neutralizing antibodies to influenza | [68–70, 73, 89, 90] |
| Influenza-specific T cells are cross-reactive against heterologous strains. | [91–94] |
| Dysregulated immune responses during influenza infection | |
| Increased cytokine levels during infection | [95–102] |
| Infiltration of, or impaired chemotaxis of, leukocytes into the lung following infection | [33, 96, 103–106] |
| Low-avidity antibodies and immune complexes in lungs of patients with severe disease | [107] |
| Age-dependent defects in adaptive immune responses | |
| Reduced T cell infiltration or function in children | [100, 108] |
| Subsequent influenza exposures are necessary for maturation of CTL responses in influenza-naïve individuals. | [109] |
| Reduced CTL activity in elderly | [110–115] |
| Genetic associations | |
| Indigenous people have more severe illness during A/H1N1 infections. | [29, 116–118] |
| SNPs within FCGR2A, C1QBP, and MBL2 are associated with severe disease. | [119, 120] |
| Cellular immunity | |
| Class I HLA presents peptides from internal and external viral proteins. | [121, 122] |
Figure 1. A chronological profile of human influenza infection.
Reported and suggested findings are highlighted in dark blue and light blue, respectively. Outstanding questions remain in white.
INNATE IMMUNITY
Effective control and clearance of influenza infection can involve most components of the innate and adaptive immune responses. In mammals, the cellular innate immune system acts as a first-line defense consisting of multipotent hematopoietic stem cells that differentiate into NK cells, mast cells, eosinophils, and basophils, as well as macrophages, neutrophils, and DCs, collectively known as phagocytic cells. Phagocytes function as APCs, engulfing and digesting opsonized pathogens and apoptotic cells. These cells essentially act as scavengers to uptake toxic metabolic byproducts and produce myriad inflammatory mediators, which ideally, result in the killing of viruses, bacteria, and parasites. However, abundant inflammation often has a role in creating immunopathology and can contribute to diverse inflammatory diseases, including asthma [123–125].
In the absence of protective immune memory, the innate response limits initial viral replication, while the adaptive immune response develops. Influenza primarily infects epithelial cells lining the respiratory tract but can infect directly or be phagoctyosed by “professional” innate immune cells, DCs, and macrophages, which mature in response to viral stimuli or signals from other infected cells. After activation, mature DCs migrate to draining LNs, where they have a major role in antigen presentation to naïve T cells. Influenza infection of human monocyte-derived DCs can result in decreased DC maturation from NS1 suppression of type I IFN production [126]. Seasonal H1N1 viruses, including A(H1N1)pdm09, elicit weak, proinflammatory cytokine gene expression in human DCs [127].
We know from murine models that influenza contains ligands that activate several PRR signaling pathways, including TLR3 and retinoic acid-inducible gene I (RIG-I), which recognize dsRNA, and TLR7, which recognizes ssRNA [128–130]. Type I (IFN-α/β) and type III (IFN-λ1–λ3) IFNs mediate expression of more than 300 ISGs via the JAK/STAT pathway and are important in the induction of antiviral responses against influenza viruses [131]. Stimulation of these pathways in DCs and macrophages leads to the induction of a potent antiviral response, including production of proinflammatory cytokines IL-6 and IFN-α within the respiratory tract by Day 2 following infection, paralleling the peak of viral replication, mucus production, and disease symptoms. TNF-α and IL-8 levels peak between Day 3 and 6 after infection as symptoms begin to subside [95].
Antimicrobial peptides, such as collectins, defensins, and cathelicidins, are important innate effectors in the human defense against influenza infection. Collectins, or collagenous lectins, are found primarily in mucosal secretions and include SP-A and SP-D; SP-D has been shown to inhibit HA activity by binding to N-linked, high-mannose oligosaccharides in a calcium-dependent manner, while enhancing activation of neutrophils in in vitro assays [132, 133]. In murine models, SP-A and SP-D have roles in promoting viral clearance from lungs and reducing inflammatory cytokines [134, 135], but their role during human influenza infection is less well-defined. Influenza virus impairs calcium metabolism and subsequent calcium deactivation in neutrophils [136], and interestingly, SP-D can protect neutrophils from calcium deactivation if preincubated with virus, presumably by inhibiting HA activity [132]. In humans, the only cathelicidin, human cathelicidin 18, the active form of which is LL-37, is found in diverse cell types, including neutrophils, NK cells, monocytes, B cells, γδ T cells, mast cells, as well as epithelial cells lining the lungs [137, 138]. Like surfactants, LL-37 shows antiviral activity against influenza and can reduce viral titers in in vitro studies [139].
Defensins have a key role in host defense and act primarily to disrupt lipid membranes leading to lysis and subsequent death of bacteria, fungi, and enveloped viruses [140]. There are two classes of defensins, α-defensin and β-defensin, which were first described in models of host defense against bacterial pathogens. Both types are found in the respiratory and gastrointestinal tracts. The α-defensins in humans include HNP-1, -2, -3, and -4, all found in high concentrations within neutrophil granules and released within the lungs in response to proinflammatory signals during infection and inflammation [141]. In vitro, HNP-1 and HNP-2 act as chemokines for human monocytes [142], and HNP-1, -2, and -3 can induce neutrophil internalization of influenza in vitro [143]. Moreover, the α-defensins may have a role in directly inactivating influenza virus as A/WSN preincubated with HNP-1 showed decreased infection in vitro in a dose-dependent manner [144]. HNP-1 can also inhibit influenza virus replication via modulation of cellular pathways, including the PKC pathway [145].
Heterogeneous populations of monocytes comprise ∼10% of blood leukocytes in humans (4% in the mouse), and their roles in controlling pathogens and inflammation can be characterized by functional cellular subsets. Human blood monocytes can be divided into three populations based primarily on cell surface expression of CD14 (LPS receptor) and CD16 (FcγRIII). The first subgroup consists of “classical monocytes”, characterized by CD14 expression and lack of CD16 expression (CD14+CD16−). The CD14+CD16− monocytes comprise 80–90% of blood monocytes and resemble murine inflammatory Ly6C+ (Gr1+) monocytes [146]. The second subgroup is defined by high expression of CD14 and CD16 (CD14+CD16+) and release TNF following LPS stimulation [147]. The third monocytic subgroup is characterized by low expression of CD14 and high expression of CD16 (CD14dimCD16+), and at least one study has shown increased numbers in the blood of sepsis patients [148]. CD14dim blood monocytes function as efficient producers of proinflammatory cytokines in response to viruses and nucleic acids [149, 150]. Few studies have examined the role of monocytes during influenza infection in humans, particularly regarding the specific subsets mentioned above, but comparison of IFN-γ production from T cells cocultured with CD64+CD16− and CD64−CD16+ monocytes showed that CD64−CD16+ monocytes (equivalent to the CD14+CD16+ monocytes) induced a greater response to influenza A antigen [151]. In vitro, human DCs and macrophages treated with type I or type III IFN prior to influenza infection result in potent antiviral activity and decreased viral replication [127]. Among DC populations, CD11c−CD14−HLA-DR+CD123+ pDCs were found to produce more IFN-α in response to a high dose of A/PR8/8/34 (H1N1) [152]. Whereas increased numbers of total CD14+ cells are observed in the blood of influenza-infected individuals, decreased blood myeloid DCs and pDCs were observed (defined in this study as HLA-DR+CD14−CD16−CD11c+ and HLA-DR+CD14−CD16−CD123+, respectively) [153]. However, increased numbers of pDCs were observed in nasal secretions compared with healthy controls within this cohort.
The NS1 protein is a well-characterized antagonist of the host's antiviral response, acting primarily to circumvent IFN-α/β responses (reviewed in ref. [154]), and in vivo models show that its deletion results in virus attenuation and higher levels of IFN [155]. Type I IFN antagonism can result in inhibition of adaptive immunity by interfering with DC maturation and thus, the ability of DCs to activate T cells [126]. The NS1 protein from influenza B virus inhibits ISGylation, an important host antiviral effector mechanism, by interacting with the N-terminal domain of ISG15 [156].
Murine models have provided several clues regarding the actual role of monocytes during the innate immune response to influenza infection. In vivo, CCR2-dependent recruitment of inflammatory monocytes is critical for host defense and contributes to the innate immune response, as well as downstream adaptive responses [157]. During influenza infection, CCR2-deficient mice have reduced monocyte and increased neutrophil recruitment, but, despite higher viral lung titers, in one model of infection these mice were protected from severe pneumonitis [158, 159]. In the lungs, CCR2-mediated recruitment of TNF and iNOS-producing DCs augments influenza-specific T cell responses [160]. At least one murine study suggests that pregnant mice have increased infiltration of neutrophils (CD11bhighLy6GhighMHCII−) and macrophages (CD11bhighLy6G−MHCIIhigh) and elevated, proinflammatory cytokine levels, despite no change in viral titers in the lungs compared with nonpregnant animals [161]. In vitro, influenza infection of murine alveolar epithelial cells resulted in monocyte production of MCP-1 (CCL2, the ligand for CCR2) and RANTES, and monocyte migration was dependent on CCR2 expression [162]. Although some murine models suggest that neutrophils may have little role in mediating viral clearance [163], type I interferon (IFN-α/β)-dependent generation of Ly6Chi monocytes following influenza infection may be necessary to limit excess neutrophil infiltration, preventing tissue damage caused by uncontrolled inflammation [164]. In addition, the newly described innate lymphoid cells have been implicated in having a role in airway integrity following influenza infection [165]. Thus, innate immune cells, particularly monocytes, have at least two roles during influenza infection: to boost the innate immune response by producing proinflammatory cytokines and to enhance influenza-specific T cell responses. However, the lack of clear correspondence between murine and human monocytic subsets has made the application of these data to the human situation difficult and highlights the need for specific studies focused on human innate immune responses.
During RSV and influenza infection of infants, neutrophils are recruited to the upper and lower airways in high numbers [103, 104], and fatal cases during the A(H1N1)pdm09 pandemic with necrotizing bronchiolitis had more neutrophils within the lung infiltrate [33]. The neutrophil chemoattractant IL-8, produced by bronchial epithelial cells, is elevated early during experimental human influenza infection [95] and is increased in the serum of A/H5N1-infected individuals [96]. Increased IL-8 levels in BALs of humans are associated with the infiltration of neutrophils and may have a role in neutrophil-mediated lung injury and development of acute respiratory distress syndrome [97]. Ex vivo influenza infection of PBMCs has been shown to induce apoptosis in neutrophils [166] and peripheral blood monocytes [167]. Influenza infection of neutrophils resulted in increased hydrogen peroxide production, enhanced surface expression of Fas antigen and FasL, and secretion of FasL into the supernatant [166]. Interestingly, this study showed that influenza infection of neutrophils resulted in increased internalization of Escherichia coli, suggesting a role in controlling bacterial secondary infections in humans. Moreover, standard chemotaxis assays during experimental influenza infection [168–170] or chemokine receptor expression during ex vivo influenza infection [105] demonstrated impaired chemotaxis of monocytes and granulocytes. Still, although all of these studies suggest that neutrophils can be regulated and recruited during human influenza infection, the specific protective and pathological roles that they play in vivo remain unclear.
Nasopharygeal aspirates of infants infected with influenza have increased levels of proinflammatory cytokines such as IL-6, TNF-α, IL-10, or IFN-γ compared with RSV- or human metapneumovirus-infected infants [98–100], and MIP-1α levels are higher in infants with more severe influenza infections characterized by hypoxic bronchiolitis [101]. Variability between these studies is likely a result of seasonal differences observed year-to-year. Individuals infected with A/H5N1 have high viral loads detected within respiratory secretions and peripheral hypercytokinemia [96, 102]. Severe disease is associated with macrophage infiltration into the lungs, and chemoattractants of monocytes and macrophages, such as IFN-inducible protein 10 (IP-10), monokine induced by IFN-γ (MIG), and MCP-1, were found to be elevated in the serum of influenza-infected individuals and were highest in those infected with A/H5N1 [96]. Therefore, peripheral (or local) hypercytokinemia likely contributes to enhanced immunopathogenesis during influenza disease. Although there is no difference in the duration of viral shedding in children hospitalized for A/H1N1 or A/H3N2 infections compared with those infected with influenza B virus, children with influenza A exhibited different serum cytokine profiles. Serum IFN-γ levels were similar during acute-phase infection [171], but levels were increased 3 weeks following influenza B infection compared with influenza A infection. Furthermore, serum IL-4 levels were significantly higher during the acute and recovery phases of influenza A infection. Influenza strain differences were also observed in umbilical cord blood lymphocytes treated with UV-inactivated viruses. UV-inactivated influenza B virus induced less IFN-γ, IL-4, or IL-10 relative to UV-inactivated A/H1N1, A/H3N2, and A/H2N2 [172].
Together, these data strongly suggest that innate cells and their associated responses have an important role in influenza disease manifestation, by providing protection and by contributing to pathological outcomes. The murine studies cited here clearly demonstrate that the local inflammatory environment can be dramatically different from the blood profile. The vast majority of experimental data in humans is restricted to blood leukocytes, with minimal emphasis placed on sites of infection. Further, we lack good, functional analysis of these cells in humans, and most studies provide correlational, cross-sectional observations. As a result, there is a significant gap in our knowledge of innate immune cells in affected tissues.
ROLE OF ANTIBODY-MEDIATED IMMUNITY IN HUMANS
The history of respiratory virus challenge studies dates back to the 1918 influenza pandemic [173], and early studies in humans infected with influenza identified the principal correlate of protection against influenza infection as strain-specific, virus-neutralizing antibodies directed against the HA. These studies (reviewed in ref. [80]) suggested that the presence of serum antibody is associated with protection, as illness is less frequently observed in those people with pre-existing nAb [74]. Subsequent studies showed that protection is also correlated with the development of serum and mucosal anti-NA or anti-HA IgG and IgA [37, 47, 75–78], but this protection can be incomplete, as individuals with detectable nAb can still be infected [79, 174]. Antibodies specific for the NA have been shown to reduce disease severity by restricting virus release from infected cells and enhancing viral clearance (reviewed in ref. [175]). In addition to HA and NA, antibodies are produced to NP, matrix M2 protein, and PB1-F2 [176], although the significance of these responses remains unclear. Of course, antigenic shift and antigenic drift can both thwart the efficacy of serological memory.
The recent circulation of A(H1N1)pdm09 has provided a modern “experiment of nature”, demonstrating the importance of pre-existing antibody from prior infection or vaccination [81–83]. Even prior to 2009, it was well-recognized that heterosubtypic immunity has a large role in protection against antigenic variants of influenza [84, 85], and vaccine studies support the idea that cross-protective, adaptive immune responses occur [86]. Likewise, neutralizing polyclonal antibodies cross-reactive against H5 can also be produced in individuals vaccinated against seasonal influenza, as well as those naturally infected with A(H1N1)pdm09 [87, 88]. Compared with seasonal influenza infections prior to 2009, ∼31% of B cell epitopes are conserved in A(H1N1)pdm09, with 17% of those conserved within the surface viral proteins [177]. The HA of A(H1N1)pdm09 contains sequences homologous with that of A/H1N1 (1918) demonstrated by the A/H1N1 (1918) mAb 2D1, which binds to the Sa antigenic site of the HA [89]. Furthermore, this particular antibody can neutralize HA activity in vitro, and mAb-treated mice show reduced viral titers when challenged with A(H1N1)pdm09 [90]. Most neutralizing antibodies bind to the exposed regions of the HA that surround the receptor-binding site and interfere with attachment to sialic acids on the cell surface. Therefore, generation of cross-protective neutralizing antibodies is elusive, as the external viral proteins are most exposed to immune pressure, but universal immunization against conserved HA stalk and M2e domains is promising [71–73].
CELL-MEDIATED IMMUNITY IN HUMANS
Human PBMCs include at least three major subsets of DCs: CD141+ mDCs, CD1c+ mDCs, and pDCs (reviewed in ref. [178]). DCs process and present antigen to T cells and therefore have an important role in regulating the adaptive immune response. Depending on the local cytokine milieu, mDCs can differentiate into macrophages or tissue-specific DCs (i.e., epithelial Langerhans cells or interstitial DCs). As influenza primarily infects epithelial cells lining the respiratory tract, lung-resident DCs and macrophages are particularly important for efficient development of an adaptive immune response. Once activated, mDCs function as APCs to activate T cells; on the other hand, pDCs are major producers of type I IFN upon TLR activation. In response to influenza infection in vitro, human DCs and macrophages undergo maturation, secreting type I IFN and inducing T cell proliferation and IFN-γ production [179–181].
Cellular immune responses (CD8+ CTLs and CD4+ Th cells) are important for virus clearance in murine models of influenza infection, with CD4+ and CD8+ T cell-depleted mice displaying delayed viral clearance. The relevance of these observations remains somewhat controversial in humans. Regardless, as in mice, the human lung contains resident respiratory virus-specific CD8+ memory T cells [182], which can effectively kill virus-infected cells. Human CD8+ T cells recognize antigen in the context of surface HLA molecules and generally recognize a broad range of influenza epitopes, including those from structural and nonstructural influenza proteins [121]. Approximately 41% of CD4+ and 69% of CD8+ T cell epitopes are conserved in A(H1N1)pdm09 from the prior circulating seasonal H1N1 [177]. Furthermore, CD8+ intraepithelial T cells can mount fast and efficient recall responses upon viral infection [183], and immunization studies in humans have shown that T cell responses peak within 1 week after symptom onset [174]. In terms of pandemic viruses, individuals infected with A(H1N1)pdm09 were shown to have rapid, virus-specific CD8+ T cell recall responses early during infection, but this CTL response wanes over time [184]. Likewise, vaccinated individuals, who were then naturally infected with A(H1N1)pdm09, had T cell responses increase by twofold early after infection [174]. Memory CD4+ or CD8+ T cells isolated from PBMCs of healthy individuals are also cross-reactive against proteins of heterologous viruses, including those from A(H1N1)pdm09 and A/H5N1 [91–94], but immunocompromised patients do not have lasting antibody or CD4+/CD8+ memory responses following natural A(H1N1)pdm09 infection [185]. Furthermore, CTLs, isolated from A(H1N1)pdm09- or A/H5N1-inexperienced individuals with a memory CD45RAmedCD62 ligandloCCR7− phenotype, will respond robustly to peptide pools and influenza-infected cells [34, 177, 186, 187], suggesting that T cells may provide some level of protection against conserved epitopes [110]. Although nonconserved regions of the virus may effectively escape recognition by T cells [188], the majority of T cell responses is elicited against conserved, internal viral proteins, generating much interest in vaccine or therapeutic strategies that involve T cell-mediated immunity.
A frequently neglected population of lymphocytes, γδ T cells comprise 1–5% of T cells in the blood of humans, the majority of which are Vγ9Vδ2 T cells. These γδ T cells are considered to be a major innate-like T cell subset (reviewed in ref. [189]), and in vitro studies suggest that activated human Vγ9Vδ2 T cells may have a role in the antiviral response by killing influenza-infected, monocyte-derived macrophages and producing high levels of IFN-γ [190, 191].
CELLULAR RESPONSES IN AT-RISK POPULATIONS
In particular, neonates are known to have compromised cellular immune responses and represent an important target population for influenza immunization. Despite being such a critical target population, there are relatively few neonatal models of respiratory infections, and for ethical reasons, it is appropriately difficult to acquire data from human neonates. In the case of RSV infection, murine neonate models suggest that the age of primary viral infection can determine the immunopathology of a secondary viral infection [192–194]. During RSV infection, younger murine neonates (≤7 days) will develop an immune response characterized by increased eosinophil and T cell recruitment to the lungs, enhanced airway hyper-responsiveness, and higher IL-13 levels upon reinfection 12 weeks later, whereas older mice (≥4 weeks) develop an immune response characterized by decreased T cell and eosinophil recruitment to the lungs upon reinfection [192, 193]. Likewise, a neonatal mouse model of influenza A virus showed that early infection was associated with decreased CD8+ T cell recruitment and function and enhanced airway inflammation [195]. These studies highlight an important concept, whereby infection too early results not in protective immunity but immunopathogenesis. Consistent with an age-dependent defect in adaptive immunity, during RSV and influenza infection of infants, few CD4+ or CD8+ T cells or CD56+ NK cells are found in the upper and lower airways [100, 103, 104], in contrast to the observed infiltration of lymphocytes in adult A/H5N1 influenza [106] and SARS CoV infections [196, 197]. Moreover, fatal cases of A(H1N1)pdm09 had increased numbers of CD8+ T cells and granzyme B+ cells in their lungs [33]. This observation may represent a correlation with higher levels of viral antigen or may indicate a pathogenic role of exuberant CD8+ T cell responses. On the other hand, the elderly have been shown to have reduced CTL activity compared with younger individuals, and this can have a negative impact on immunization efficacy [111–113]. However, prevaccination T cell responses do not correlate with postvaccination antibody titers [114], and as the elderly also have decreased antibody responses following vaccination, T cell responses may provide a better correlate of vaccine efficacy in the elderly population [115]. Interestingly, unstimulated memory T cells from older individuals are in a higher state of differentiation than those of younger individuals, and it was shown that fewer senescent, influenza-specific T cells, characterized as killer cell lectin-like receptor subfamily G member 1 (KLRG1)hiCD57hi, are associated with a more robust antibody response following TIV delivery [110].
The immunodominance hierarchy in HLA-A*02 individuals is often reported as directed primarily against the conserved influenza A M158–66 (GILGFVFTL) [198, 199]. However, studies suggest that although this CD8+ T cell response to M158–66 is conserved in HLA-A*02 individuals, it may not be strictly immunodominant, as greater responses were observed against internal NP and polymerase PB1 subunit proteins in some individuals [122, 200]. In terms of HLA presentation of influenza peptides, the HLA-A*02:01-restricted M158–66 peptide-specific CTL response is characterized by expression of Vβ17 TCR use [201], and in HLA-A*02:01 individuals, Vβ17+ CTLs were the dominant cells responding to in vitro influenza exposure. In contrast, cord blood from HLA-A*02:01 infants, thus having no prior influenza exposure, was characterized by a M158–66-specific CD8+ T cell response that was less dependent on the presence of the CD8+Vβ17+ T cell population [109]. Therefore, maturation of the CTL response upon subsequent influenza infections is likely related to effective clearance of viral infections and has important implications for therapeutic interventions. Although children beyond infancy who are considered high-risk for influenza infection, i.e., children with underlying diseases, such as asthma, sickle cell disease, and those receiving solid organ transplantation, have significant increases in serum nAb and T cell responses, children with systemic lupus erythematosus are unable to mount significant T cell responses following TIV immunization [108]. These findings suggest that the pathogenesis of respiratory viral infections in infants may be associated with the absence of immunological memory or a failure to develop an appropriate, well-regulated T cell response.
GENETIC ASSOCIATIONS WITH SEVERE INFLUENZA INFECTIONS
Undoubtedly, host genetics have a role in the outcome of many infectious diseases, including influenza. Mouse models have provided some clues regarding the role of the host genetic component on the inflammatory response in the respiratory tract. Briefly, these studies found that influenza-induced pathology is altered depending on the inbred mouse strain used for infection. The genetic background of the host also controls cumulative and maximal viral titer, underscoring the importance of limiting viral replication early during influenza infection [111, 202].
In humans, certain populations worldwide have been found to be more prone to severe disease during influenza infection. For instance, Aboriginal peoples have more severe illness and increased mortality during the A/H1N1 pandemics of 1918 and 2009 compared with those of non-Aboriginal descent [29, 116–118]. It is important to note, however, that poor socioeconomic conditions and metabolic diseases, such as diabetes, are more common in indigenous populations [203], and this may contribute to influenza-related disease. Moreover, HLA alleles are proposed to have some role during influenza infection, as differential CTL responses exist in individuals with varying HLA alleles [204].
Gene polymorphisms located in human chromosomes 1 and 17 were also determined to be associated with severe pneumonia during A(H1N1)pdm09 infection [120]. Interestingly, two of these SNPs are mapped to genes FCGR2A and C1QBP. The protein encoded by FCGR2A is involved in phagocytosis and clearance of immune complexes and expressed on the surface of phagocytic cells. The protein encoded by C1QBP is a critical factor involved in complement activation. Notably, presence of low-avidity antibodies and subsequent formation of immune complex-mediated complement activation in the lungs of patients with severe influenza infection are associated with severe disease following influenza infection [107]. Recently, “systems biology” strategies have emerged to improve vaccine efficacy, measuring expression patterns to expose protective molecular signatures following immunization or natural infection [46, 205].
Confirming in vitro mouse studies, which suggest that MBL can neutralize influenza [206, 207], human polymorphisms identified within MBL2 are associated with poor antibody responses to influenza vaccination [119]. Although MBL deficiency has been observed to influence the outcome in several respiratory diseases, including pneumococcus infection, tuberculosis, and SARS CoV (reviewed in ref. [208]), it did not have an obvious role in A(H1N1)pdm09 infection of naïve individuals [209]. These studies highlight the involvement of host genetics in development of enhanced disease, although they likely represent a small fraction of the potential regulatory host genetic elements.
CONCLUSION
Studies of innate and adaptive immune responses to influenza virus infection have been restricted primarily to animal models of disease and inflammation. As our immunological tools become more sophisticated, more emphasis should be placed on human immunology as opposed to relying solely on animal infection models. Moreover, the vast majority of experimental data in humans is restricted to blood leukocytes, with minimal emphasis placed on the innate immune response, particularly at sites of infection. As a result, there is limited scientific knowledge regarding the specific roles of immune cells in affected tissues. Understanding the innate response in particular is critical to appreciate early determinants of pathogenicity and disease outcome during influenza infection.
ACKNOWLEDGMENTS
The authors are funded in part by U.S. National Institutes of Health/National Institute of Allergy and Infectious Diseases contracts HHSN266200700005C (St. Jude Center of Excellence for Influenza Research and Surveillance) and HHSN272200800058C (Systems Influenza) and the American Lebanese Syrian Associated Charities (ALSAC).
Footnotes
- AS03
- Adjuvant System 03
- ASC
- antibody-secreting cell
- CDC
- Centers for Disease Control and Prevention
- FasL
- Fas ligand
- HNP
- human neutrophil peptide
- ISG
- IFN-stimulated gene
- LAIV
- live attenuated influenza vaccine
- MBL
- mannose-binding lectin
- mDC
- myeloid DC
- NA
- neuraminidase
- nAb
- neutralizing antibody
- NP
- nucleoprotein
- pDC
- plasmacytoid DC
- RSV
- respiratory syncytial virus
- SARS CoV
- severe acute respiratory syndrome coronavirus
- SNP
- single nucleotide polymorphism
- SP
- surfactant protein
- TIV
- trivalent-inactivated vaccine
REFERENCES
- 1. U.S. Centers for Disease Control and Prevention (2009) 2008–09 Influenza Prevention and Control Recommendations: Recommendations of the Advisory Committee on Immunization Practices (ACIP). CDC, Atlanta, GA, USA [Google Scholar]
- 2. Peltola V., Ziegler T., Ruuskanen O. (2003) Influenza A and B virus infections in children. Clin. Infect. Dis. 36, 299–305 [DOI] [PubMed] [Google Scholar]
- 3. Heikkinen T., Silvennoinen H., Peltola V., Ziegler T., Vainionpaa R., Vuorinen T., Kainulainen L., Puhakka T., Jartti T., Toikka P., Lehtinen P., Routi T., Juven T. (2004) Burden of influenza in children in the community. J. Infect. Dis. 190, 1369–1373 [DOI] [PubMed] [Google Scholar]
- 4. Poehling K. A., Edwards K. M., Weinberg G. A., Szilagyi P., Staat M. A., Iwane M. K., Bridges C. B., Grijalva C. G., Zhu Y., Bernstein D. I., Herrera G., Erdman D., Hall C. B., Seither R., Griffin M. R. (2006) The underrecognized burden of influenza in young children. N. Engl. J. Med. 355, 31–40 [DOI] [PubMed] [Google Scholar]
- 5. Neuzil K. M., Mellen B. G., Wright P. F., Mitchel E. F., Jr., Griffin M. R. (2000) The effect of influenza on hospitalizations, outpatient visits, and courses of antibiotics in children. N. Engl. J. Med. 342, 225–231 [DOI] [PubMed] [Google Scholar]
- 6. Louie J.K., Gavali S., Acosta M., Samuel M.C., Winter K., Jean C., Glaser C.A., Matyas B.T., Schechter R. (2010) Children hospitalized with 2009 novel influenza A(H1N1) in California. Arch. Pediatr. Adolesc. Med. 164, 1023–1031 [DOI] [PubMed] [Google Scholar]
- 7. Schanzer D., Vachon J., Pelletier L. (2011) Age-specific differences in influenza A epidemic curves: do children drive the spread of influenza epidemics? Am. J. Epidemiol. 174, 109–117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Longini I. M., Jr., Koopman J. S., Monto A. S., Fox J. P. (1982) Estimating household and community transmission parameters for influenza. Am. J. Epidemiol. 115, 736–751 [DOI] [PubMed] [Google Scholar]
- 9. Viboud C., Boelle P. Y., Cauchemez S., Lavenu A., Valleron A. J., Flahault A., Carrat F. (2004) Risk factors of influenza transmission in households. Br. J. Gen. Pract. 54, 684–689 [PMC free article] [PubMed] [Google Scholar]
- 10. Neuzil K. M., Zhu Y., Griffin M. R., Edwards K. M., Thompson J. M., Tollefson S. J., Wright P. F. (2002) Burden of interpandemic influenza in children younger than 5 years: a 25-year prospective study. J. Infect. Dis. 185, 147–152 [DOI] [PubMed] [Google Scholar]
- 11. Pullan C. R., Hey E. N. (1982) Wheezing, asthma, and pulmonary dysfunction 10 years after infection with respiratory syncytial virus in infancy. Br. Med. J. (Clin. Res. Ed). 284, 1665–1669 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Sigurs N., Bjarnason R., Sigurbergsson F., Kjellman B. (2000) Respiratory syncytial virus bronchiolitis in infancy is an important risk factor for asthma and allergy at age 7. Am. J. Respir. Crit. Care Med. 161, 1501–1507 [DOI] [PubMed] [Google Scholar]
- 13. Sigurs N., Gustafsson P. M., Bjarnason R., Lundberg F., Schmidt S., Sigurbergsson F., Kjellman B. (2005) Severe respiratory syncytial virus bronchiolitis in infancy and asthma and allergy at age 13. Am. J. Respir. Crit. Care Med. 171, 137–141 [DOI] [PubMed] [Google Scholar]
- 14. Thompson W. W., Shay D. K., Weintraub E., Brammer L., Bridges C. B., Cox N. J., Fukuda K. (2004) Influenza-associated hospitalizations in the United States. JAMA 292, 1333–1340 [DOI] [PubMed] [Google Scholar]
- 15. Monto A. S. (2002) Epidemiology of viral respiratory infections. Am. J. Med. 112 (Suppl. 6A), 4S–12S [DOI] [PubMed] [Google Scholar]
- 16. U.S. Centers for Disease Control and Prevention (2010) Estimates of deaths associated with seasonal influenza—United States, 1976–2007. MMWR 59, 1057–1062 [PubMed] [Google Scholar]
- 17. Nair H., Brooks W. A., Katz M., Roca A., Berkley J. A., Madhi S. A., Simmerman J. M., Gordon A., Sato M., Howie S., Krishnan A., Ope M., Lindblade K. A., Carosone-Link P., Lucero M., Ochieng W., Kamimoto L., Dueger E., Bhat N., Vong S., Theodoratou E., Chittaganpitch M., Chimah O., Balmaseda A., Buchy P., Harris E., Evans V., Katayose M., Gaur B., O'Callaghan-Gordo C., Goswami D., Arvelo W., Venter M., Briese T., Tokarz R., Widdowson M. A., Mounts A. W., Breiman R. F., Feikin D. R., Klugman K. P., Olsen S. J., Gessner B. D., Wright P. F., Rudan I., Broor S., Simoes E. A., Campbell H. (2011) Global burden of respiratory infections due to seasonal influenza in young children: a systematic review and meta-analysis. Lancet 378, 1917–1930 [DOI] [PubMed] [Google Scholar]
- 18. Molinari N. A., Ortega-Sanchez I. R., Messonnier M. L., Thompson W. W., Wortley P. M., Weintraub E., Bridges C. B. (2007) The annual impact of seasonal influenza in the US: measuring disease burden and costs. Vaccine 25, 5086–5096 [DOI] [PubMed] [Google Scholar]
- 19. Fairbrother G., Cassedy A., Ortega-Sanchez I. R., Szilagyi P. G., Edwards K. M., Molinari N. A., Donauer S., Henderson D., Ambrose S., Kent D., Poehling K., Weinberg G. A., Griffin M. R., Hall C. B., Finelli L., Bridges C., Staat M. A. (2010) High costs of influenza: direct medical costs of influenza disease in young children. Vaccine 28, 4913–4919 [DOI] [PubMed] [Google Scholar]
- 20. U.S. Centers for Disease Control and Prevention (2011) Standardization of terminology of the pandemic A(H1N1) 2009 virus. Wkly. Epidemiol. Rec. 86, 480. [PubMed] [Google Scholar]
- 21. Chowell G., Bertozzi S. M., Colchero M. A., Lopez-Gatell H., Alpuche-Aranda C., Hernandez M., Miller M. A. (2009) Severe respiratory disease concurrent with the circulation of H1N1 influenza. N. Engl. J. Med. 361, 674–679 [DOI] [PubMed] [Google Scholar]
- 22. Perez-Padilla R., de la Rosa-Zamboni D., Ponce de Leon S., Hernandez M., Quinones-Falconi F., Bautista E., Ramirez-Venegas A., Rojas-Serrano J., Ormsby C. E., Corrales A., Higuera A., Mondragon E., Cordova-Villalobos J. A. (2009) Pneumonia and respiratory failure from swine-origin influenza A (H1N1) in Mexico. N. Engl. J. Med. 361, 680–689 [DOI] [PubMed] [Google Scholar]
- 23. Libster R., Bugna J., Coviello S., Hijano D. R., Dunaiewsky M., Reynoso N., Cavalieri M. L., Guglielmo M. C., Areso M. S., Gilligan T., Santucho F., Cabral G., Gregorio G. L., Moreno R., Lutz M. I., Panigasi A. L., Saligari L., Caballero M. T., Egues Almeida R. M., Gutierrez Meyer M. E., Neder M. D., Davenport M. C., Del Valle M. P., Santidrian V. S., Mosca G., Garcia Dominguez M., Alvarez L., Landa P., Pota A., Bolonati N., Dalamon R., Sanchez Mercol V. I., Espinoza M., Peuchot J. C., Karolinski A., Bruno M., Borsa A., Ferrero F., Bonina A., Ramonet M., Albano L. C., Luedicke N., Alterman E., Savy V., Baumeister E., Chappell J. D., Edwards K. M., Melendi G. A., Polack F. P. (2010) Pediatric hospitalizations associated with 2009 pandemic influenza A (H1N1) in Argentina. N. Engl. J. Med. 362, 45–55 [DOI] [PubMed] [Google Scholar]
- 24. Hancock K., Veguilla V., Lu X., Zhong W., Butler E. N., Sun H., Liu F., Dong L., DeVos J. R., Gargiullo P. M., Brammer T. L., Cox N. J., Tumpey T. M., Katz J. M. (2009) Cross-reactive antibody responses to the 2009 pandemic H1N1 influenza virus. N. Engl. J. Med. 361, 1945–1952 [DOI] [PubMed] [Google Scholar]
- 25. Van Kerkhove M. D., Vandemaele K. A., Shinde V., Jaramillo-Gutierrez G., Koukounari A., Donnelly C. A., Carlino L. O., Owen R., Paterson B., Pelletier L., Vachon J., Gonzalez C., Hongjie Y., Zijian F., Chuang S. K., Au A., Buda S., Krause G., Haas W., Bonmarin I., Taniguichi K., Nakajima K., Shobayashi T., Takayama Y., Sunagawa T., Heraud J. M., Orelle A., Palacios E., van der Sande M. A., Wielders C. C., Hunt D., Cutter J., Lee V. J., Thomas J., Santa-Olalla P., Sierra-Moros M. J., Hanshaoworakul W., Ungchusak K., Pebody R., Jain S., Mounts A. W. (2011) Risk factors for severe outcomes following 2009 influenza A (H1N1) infection: a global pooled analysis. PLoS Med. 8, e1001053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. U.S. Centers for Disease Control and Prevention (2009) Novel influenza A (H1N1) virus infections in three pregnant women—United States, April–May 2009. MMWR 58, 497–500 [PubMed] [Google Scholar]
- 27. Louie J. K., Acosta M., Jamieson D. J., Honein M. A. (2010) Severe 2009 H1N1 influenza in pregnant and postpartum women in California. N. Engl. J. Med. 362, 27–35 [DOI] [PubMed] [Google Scholar]
- 28. Jamieson D. J., Honein M. A., Rasmussen S. A., Williams J. L., Swerdlow D. L., Biggerstaff M. S., Lindstrom S., Louie J. K., Christ C. M., Bohm S. R., Fonseca V. P., Ritger K. A., Kuhles D. J., Eggers P., Bruce H., Davidson H. A., Lutterloh E., Harris M. L., Burke C., Cocoros N., Finelli L., MacFarlane K. F., Shu B., Olsen S. J. (2009) H1N1 2009 influenza virus infection during pregnancy in the U.S.A. Lancet 374, 451–458 [DOI] [PubMed] [Google Scholar]
- 29. Zarychanski R., Stuart T.L., Kumar A., Doucette S., Elliott L., Kettner J., Plummer F. (2010) Correlates of severe disease in patients with 2009 pandemic influenza (H1N1) virus infection. CMAJ 182, 257–264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. McCullers J. A. (2006) Insights into the interaction between influenza virus and pneumococcus. Clin. Microbiol. Rev. 19, 571–582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Murray R. J., Robinson J. O., White J. N., Hughes F., Coombs G. W., Pearson J. C., Tan H. L., Chidlow G., Williams S., Christiansen K. J., Smith D. W. (2010) Community-acquired pneumonia due to pandemic A(H1N1)2009 influenzavirus and methicillin resistant Staphylococcus aureus co-infection. PLoS One 5, e8705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. U.S. Centers for Disease Control and Prevention (2009) Bacterial coinfections in lung tissue specimens from fatal cases of 2009 pandemic influenza A (H1N1)—United States, May–August 2009. MMWR 58, 1071–1074 [PubMed] [Google Scholar]
- 33. Mauad T., Hajjar L. A., Callegari G. D., da Silva L. F., Schout D., Galas F. R., Alves V. A., Malheiros D. M., Auler J. O., Jr., Ferreira A. F., Borsato M. R., Bezerra S. M., Gutierrez P. S., Caldini E. T., Pasqualucci C. A., Dolhnikoff M., Saldiva P. H. (2010) Lung pathology in fatal novel human influenza A (H1N1) infection. Am. J. Respir. Crit. Care Med. 181, 72–79 [DOI] [PubMed] [Google Scholar]
- 34. Kreijtz J. H., de Mutsert G., van Baalen C. A., Fouchier R. A., Osterhaus A. D., Rimmelzwaan G. F. (2008) Cross-recognition of avian H5N1 influenza virus by human cytotoxic T-lymphocyte populations directed to human influenza A virus. J. Virol. 82, 5161–5166 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. U.S. Centers for Disease Control and Prevention (2011) Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP), 2011. MMWR 60, 1128–1132 [PubMed] [Google Scholar]
- 36. Couch R. B. (1975) Assessment of immunity to influenza using artifical challenge of normal volunteers with influenza virus. Dev. Biol. Stand. 28, 295–306 [PubMed] [Google Scholar]
- 37. Clements M. L., Betts R. F., Tierney E. L., Murphy B. R. (1986) Serum and nasal wash antibodies associated with resistance to experimental challenge with influenza A wild-type virus. J. Clin. Microbiol. 24, 157–160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Keitel W. A., Atmar R. L., Cate T. R., Petersen N. J., Greenberg S. B., Ruben F., Couch R. B. (2006) Safety of high doses of influenza vaccine and effect on antibody responses in elderly persons. Arch. Intern. Med. 166, 1121–1127 [DOI] [PubMed] [Google Scholar]
- 39. Couch R. B., Winokur P., Brady R., Belshe R., Chen W. H., Cate T. R., Sigurdardottir B., Hoeper A., Graham I. L., Edelman R., He F., Nino D., Capellan J., Ruben F. L. (2007) Safety and immunogenicity of a high dosage trivalent influenza vaccine among elderly subjects. Vaccine 25, 7656–7663 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Brokstad K. A., Eriksson J. C., Cox R. J., Tynning T., Olofsson J., Jonsson R., Davidsson A. (2002) Parenteral vaccination against influenza does not induce a local antigen-specific immune response in the nasal mucosa. J. Infect. Dis. 185, 878–884 [DOI] [PubMed] [Google Scholar]
- 41. Cox R. J., Brokstad K. A., Zuckerman M. A., Wood J. M., Haaheim L. R., Oxford J. S. (1994) An early humoral immune response in peripheral blood following parenteral inactivated influenza vaccination. Vaccine 12, 993–999 [DOI] [PubMed] [Google Scholar]
- 42. El-Madhun A. S., Cox R. J., Soreide A., Olofsson J., Haaheim L. R. (1998) Systemic and mucosal immune responses in young children and adults after parenteral influenza vaccination. J. Infect. Dis. 178, 933–939 [DOI] [PubMed] [Google Scholar]
- 43. Sasaki S., Jaimes M. C., Holmes T. H., Dekker C. L., Mahmood K., Kemble G. W., Arvin A. M., Greenberg H. B. (2007) Comparison of the influenza virus-specific effector and memory B-cell responses to immunization of children and adults with live attenuated or inactivated influenza virus vaccines. J. Virol. 81, 215–228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Belshe R. B., Gruber W. C., Mendelman P. M., Mehta H. B., Mahmood K., Reisinger K., Treanor J., Zangwill K., Hayden F. G., Bernstein D. I., Kotloff K., King J., Piedra P. A., Block S. L., Yan L., Wolff M. (2000) Correlates of immune protection induced by live, attenuated, cold-adapted, trivalent, intranasal influenza virus vaccine. J. Infect. Dis. 181, 1133–1137 [DOI] [PubMed] [Google Scholar]
- 45. Belshe R. B., Edwards K. M., Vesikari T., Black S. V., Walker R. E., Hultquist M., Kemble G., Connor E. M. (2007) Live attenuated versus inactivated influenza vaccine in infants and young children. N. Engl. J. Med. 356, 685–696 [DOI] [PubMed] [Google Scholar]
- 46. Nakaya H. I., Wrammert J., Lee E. K., Racioppi L., Marie-Kunze S., Haining W. N., Means A. R., Kasturi S. P., Khan N., Li G. M., McCausland M., Kanchan V., Kokko K. E., Li S., Elbein R., Mehta A. K., Aderem A., Subbarao K., Ahmed R., Pulendran B. (2011) Systems biology of vaccination for seasonal influenza in humans. Nat. Immunol. 12, 786–795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Clements M. L., Murphy B. R. (1986) Development and persistence of local and systemic antibody responses in adults given live attenuated or inactivated influenza A virus vaccine. J. Clin. Microbiol. 23, 66–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Ruben F. L. (2004) Inactivated influenza virus vaccines in children. Clin. Infect. Dis. 38, 678–688 [DOI] [PubMed] [Google Scholar]
- 49. Piedra P. A., Gaglani M. J., Kozinetz C. A., Herschler G., Riggs M., Griffith M., Fewlass C., Watts M., Hessel C., Cordova J., Glezen W. P. (2005) Herd immunity in adults against influenza-related illnesses with use of the trivalent-live attenuated influenza vaccine (CAIV-T) in children. Vaccine 23, 1540–1548 [DOI] [PubMed] [Google Scholar]
- 50. Esposito S., Marchisio P., Cavagna R., Gironi S., Bosis S., Lambertini L., Droghetti R., Principi N. (2003) Effectiveness of influenza vaccination of children with recurrent respiratory tract infections in reducing respiratory-related morbidity within the households. Vaccine 21, 3162–3168 [DOI] [PubMed] [Google Scholar]
- 51. Reichert T. A., Sugaya N., Fedson D. S., Glezen W. P., Simonsen L., Tashiro M. (2001) The Japanese experience with vaccinating schoolchildren against influenza. N. Engl. J. Med. 344, 889–896 [DOI] [PubMed] [Google Scholar]
- 52. Osterholm M. T., Kelley N. S., Sommer A., Belongia E. A. (2012) Efficacy and effectiveness of influenza vaccines: a systematic review and meta-analysis. Lancet Infect Dis. 12, 36–44 [DOI] [PubMed] [Google Scholar]
- 53. Simonsen L., Taylor R. J., Viboud C., Miller M. A., Jackson L. A. (2007) Mortality benefits of influenza vaccination in elderly people: an ongoing controversy. Lancet Infect. Dis. 7, 658–666 [DOI] [PubMed] [Google Scholar]
- 54. Fireman B., Lee J., Lewis N., Bembom O., van der Laan M., Baxter R. (2009) Influenza vaccination and mortality: differentiating vaccine effects from bias. Am. J. Epidemiol. 170, 650–656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Baxter R., Ray G. T., Fireman B. H. (2010) Effect of influenza vaccination on hospitalizations in persons aged 50 years and older. Vaccine 28, 7267–7272 [DOI] [PubMed] [Google Scholar]
- 56. U.S. Centers for Disease Control and Prevention (2011) FluVaxView: Influenza Vaccination Coverage. CDC, Atlanta, GA, USA [Google Scholar]
- 57. Carrat F., Flahault A. (2007) Influenza vaccine: the challenge of antigenic drift. Vaccine 25, 6852–6862 [DOI] [PubMed] [Google Scholar]
- 58. O'Hagan D.T., Tsai T., Reed S. (2011) Emulsion-based adjuvants for improved influenza vaccines. In Influenza Vaccines for the Future, 2nd edition (Del Giudice G., Rappuoli R., eds.), Springer Basel AG, Switzerland, 327–357 [Google Scholar]
- 59. Podda A. (2001) The adjuvanted influenza vaccines with novel adjuvants: experience with the MF59-adjuvanted vaccine. Vaccine 19, 2673–2680 [DOI] [PubMed] [Google Scholar]
- 60. Clark T. W., Pareek M., Hoschler K., Dillon H., Nicholson K. G., Groth N., Stephenson I. (2009) Trial of 2009 influenza A (H1N1) monovalent MF59-adjuvanted vaccine. N. Engl. J. Med. 361, 2424–2435 [DOI] [PubMed] [Google Scholar]
- 61. O'Hagan D. T. (2007) MF59 is a safe and potent vaccine adjuvant that enhances protection against influenza virus infection. Exp. Rev. Vaccines 6, 699–710 [DOI] [PubMed] [Google Scholar]
- 62. Vesikari T., Groth N., Karvonen A., Borkowski A., Pellegrini M. (2009) MF59-adjuvanted influenza vaccine (FLUAD) in children: safety and immunogenicity following a second year seasonal vaccination. Vaccine 27, 6291–6295 [DOI] [PubMed] [Google Scholar]
- 63. Vesikari T., Pellegrini M., Karvonen A., Groth N., Borkowski A., O'Hagan D.T., Podda A. (2009) Enhanced immunogenicity of seasonal influenza vaccines in young children using MF59 adjuvant. Pediatr. Infect. Dis. J. 28, 563–571 [DOI] [PubMed] [Google Scholar]
- 64. Vesikari T., Knuf M., Wutzler P., Karvonen A., Kieninger-Baum D., Schmitt H.J., Baehner F., Borkowski A., Tsai T.F., Clemens R. (2011) Oil-in-water emulsion adjuvant with influenza vaccine in young children. N. Engl. J. Med. 365, 1406–1416 [DOI] [PubMed] [Google Scholar]
- 65. Huleatt J. W., Nakaar V., Desai P., Huang Y., Hewitt D., Jacobs A., Tang J., McDonald W., Song L., Evans R. K., Umlauf S., Tussey L., Powell T. J. (2008) Potent immunogenicity and efficacy of a universal influenza vaccine candidate comprising a recombinant fusion protein linking influenza M2e to the TLR5 ligand flagellin. Vaccine 26, 201–214 [DOI] [PubMed] [Google Scholar]
- 66. Schotsaert M., De Filette M., Fiers W., Saelens X. (2009) Universal M2 ectodomain-based influenza A vaccines: preclinical and clinical developments. Expert Rev. Vaccines 8, 499–508 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Turley C. B., Rupp R. E., Johnson C., Taylor D. N., Wolfson J., Tussey L., Kavita U., Stanberry L., Shaw A. (2011) Safety and immunogenicity of a recombinant M2e-flagellin influenza vaccine (STF2.4xM2e) in healthy adults. Vaccine 29, 5145–5152 [DOI] [PubMed] [Google Scholar]
- 68. Ekiert D. C., Friesen R. H., Bhabha G., Kwaks T., Jongeneelen M., Yu W., Ophorst C., Cox F., Korse H. J., Brandenburg B., Vogels R., Brakenhoff J. P., Kompier R., Koldijk M. H., Cornelissen L. A., Poon L. L., Peiris M., Koudstaal W., Wilson I. A., Goudsmit J. (2011) A highly conserved neutralizing epitope on group 2 influenza A viruses. Science 333, 843–850 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Corti D., Voss J., Gamblin S. J., Codoni G., Macagno A., Jarrossay D., Vachieri S. G., Pinna D., Minola A., Vanzetta F., Silacci C., Fernandez-Rodriguez B. M., Agatic G., Bianchi S., Giacchetto-Sasselli I., Calder L., Sallusto F., Collins P., Haire L. F., Temperton N., Langedijk J. P., Skehel J. J., Lanzavecchia A. (2011) A neutralizing antibody selected from plasma cells that binds to group 1 and group 2 influenza A hemagglutinins. Science 333, 850–856 [DOI] [PubMed] [Google Scholar]
- 70. Whittle J. R., Zhang R., Khurana S., King L. R., Manischewitz J., Golding H., Dormitzer P. R., Haynes B. F., Walter E. B., Moody M. A., Kepler T. B., Liao H. X., Harrison S. C. (2011) Broadly neutralizing human antibody that recognizes the receptor-binding pocket of influenza virus hemagglutinin. Proc. Natl. Acad. Sci. USA 108, 14216–14221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Ekiert D. C., Bhabha G., Elsliger M. A., Friesen R. H., Jongeneelen M., Throsby M., Goudsmit J., Wilson I. A. (2009) Antibody recognition of a highly conserved influenza virus epitope. Science 324, 246–251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Sui J., Hwang W. C., Perez S., Wei G., Aird D., Chen L. M., Santelli E., Stec B., Cadwell G., Ali M., Wan H., Murakami A., Yammanuru A., Han T., Cox N. J., Bankston L. A., Donis R. O., Liddington R. C., Marasco W. A. (2009) Structural and functional bases for broad-spectrum neutralization of avian and human influenza A viruses. Nat. Struct. Mol. Biol. 16, 265–273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Krause J. C., Tsibane T., Tumpey T. M., Huffman C. J., Basler C. F., Crowe J. E., Jr. (2011) A broadly neutralizing human monoclonal antibody that recognizes a conserved, novel epitope on the globular head of the influenza H1N1 virus hemagglutinin. J. Virol. 85, 10905–10908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Smorodintseff A. A., Tushinsky M. D., Drobyshevskaya I., Korovin A. A., Osetroff A. I. (1937) Investigation of volunteers infected with the influenza virus. Am. J. Med. Sci. 194, 159–170 [Google Scholar]
- 75. Johnson P. R., Jr., Feldman S., Thompson J. M., Mahoney J. D., Wright P. F. (1985) Comparison of long-term systemic and secretory antibody responses in children given live, attenuated, or inactivated influenza A vaccine. J. Med. Virol. 17, 325–335 [DOI] [PubMed] [Google Scholar]
- 76. Johnson P. R., Feldman S., Thompson J. M., Mahoney J. D., Wright P. F. (1986) Immunity to influenza A virus infection in young children: a comparison of natural infection, live cold-adapted vaccine, and inactivated vaccine. J. Infect. Dis. 154, 121–127 [DOI] [PubMed] [Google Scholar]
- 77. Treanor J. J., Roth F. K., Betts R. F. (1990) Use of live cold-adapted influenza A H1N1 and H3N2 virus vaccines in seropositive adults. J. Clin. Microbiol. 28, 596–599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Knight V., Kasel J. A., Alford R. H., Loda F., Morris J. A., Davenport F. M., Robinson R. Q., Buescher E. L. (1965) New research on influenza: studies with normal volunteers. Combined Clinical Staff Conference at the National Institutes of Health. Ann. Intern. Med. 62, 1307–1325 [DOI] [PubMed] [Google Scholar]
- 79. Treanor J., Wright P. F. (2003) Immune correlates of protection against influenza in the human challenge model. Dev. Biol. (Basel) 115, 97–104 [PubMed] [Google Scholar]
- 80. Killingley B., Enstone J., Booy R., Hayward A., Oxford J., Ferguson N., Van-Tam J. N. (2011) Potential role of human challenge studies for investigation of influenza transmission. Lancet Infect. Dis. 11, 879–886 [DOI] [PubMed] [Google Scholar]
- 81. Garcia-Garcia L., Valdespino-Gomez J. L., Lazcano-Ponce E., Jimenez-Corona A., Higuera-Iglesias A., Cruz-Hervert P., Cano-Arellano B., Garcia-Anaya A., Ferreira-Guerrero E., Baez-Saldana R., Ferreyra-Reyes L., Ponce-de-Leon-Rosales S., Alpuche-Aranda C., Rodriguez-Lopez M. H., Perez-Padilla R., Hernandez-Avila M. (2009) Partial protection of seasonal trivalent inactivated vaccine against novel pandemic influenza A/H1N1 2009: case-control study in Mexico City. BMJ 339, b3928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Johns M. C., Eick A. A., Blazes D. L., Lee S. E., Perdue C. L., Lipnick R., Vest K. G., Russell K. L., DeFraites R. F., Sanchez J. L. (2010) Seasonal influenza vaccine and protection against pandemic (H1N1) 2009-associated illness among US military personnel. PLoS One 5, e10722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Cowling B. J., Ng S., Ma E. S., Cheng C. K., Wai W., Fang V. J., Chan K. H., Ip D. K., Chiu S. S., Peiris J. S., Leung G. M. (2010) Protective efficacy of seasonal influenza vaccination against seasonal and pandemic influenza virus infection during 2009 in Hong Kong. Clin. Infect. Dis. 51, 1370–1379 [DOI] [PubMed] [Google Scholar]
- 84. Epstein S. L. (2006) Prior H1N1 influenza infection and susceptibility of Cleveland Family Study participants during the H2N2 pandemic of 1957: an experiment of nature. J. Infect. Dis. 193, 49–53 [DOI] [PubMed] [Google Scholar]
- 85. Sonoguchi T., Naito H., Hara M., Takeuchi Y., Fukumi H. (1985) Cross-subtype protection in humans during sequential, overlapping, and/or concurrent epidemics caused by H3N2 and H1N1 influenza viruses. J. Infect. Dis. 151, 81–88 [DOI] [PubMed] [Google Scholar]
- 86. Hammitt L. L., Bartlett J. P., Li S., Rahkola J., Lang N., Janoff E. N., Levin M. J., Weinberg A. (2009) Kinetics of viral shedding and immune responses in adults following administration of cold-adapted influenza vaccine. Vaccine 27, 7359–7366 [DOI] [PubMed] [Google Scholar]
- 87. Corti D., Suguitan A. L., Jr., Pinna D., Silacci C., Fernandez-Rodriguez B. M., Vanzetta F., Santos C., Luke C. J., Torres-Velez F. J., Temperton N. J., Weiss R. A., Sallusto F., Subbarao K., Lanzavecchia A. (2010) Heterosubtypic neutralizing antibodies are produced by individuals immunized with a seasonal influenza vaccine. J. Clin. Invest. 120, 1663–1673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Wrammert J., Koutsonanos D., Li G. M., Edupuganti S., Sui J., Morrissey M., McCausland M., Skountzou I., Hornig M., Lipkin W. I., Mehta A., Razavi B., Del Rio C., Zheng N. Y., Lee J. H., Huang M., Ali Z., Kaur K., Andrews S., Amara R. R., Wang Y., Das S. R., O'Donnell C. D., Yewdell J. W., Subbarao K., Marasco W. A., Mulligan M. J., Compans R., Ahmed R., Wilson P. C. (2011) Broadly cross-reactive antibodies dominate the human B cell response against 2009 pandemic H1N1 influenza virus infection. J. Exp. Med. 208, 181–193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Yu X., Tsibane T., McGraw P. A., House F. S., Keefer C. J., Hicar M. D., Tumpey T. M., Pappas C., Perrone L. A., Martinez O., Stevens J., Wilson I. A., Aguilar P. V., Altschuler E. L., Basler C. F., Crowe J. E., Jr. (2008) Neutralizing antibodies derived from the B cells of 1918 influenza pandemic survivors. Nature 455, 532–536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Krause J. C., Tumpey T. M., Huffman C. J., McGraw P. A., Pearce M. B., Tsibane T., Hai R., Basler C. F., Crowe J. E., Jr. (2010) Naturally occurring human monoclonal antibodies neutralize both 1918 and 2009 pandemic influenza A (H1N1) viruses. J. Virol. 84, 3127–3130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Lee L. Y., Ha do L. A., Simmons C., de Jong M. D., Chau N. V., Schumacher R., Peng Y. C., McMichael A. J., Farrar J. J., Smith G. L., Townsend A. R., Askonas B. A., Rowland-Jones S., Dong T. (2008) Memory T cells established by seasonal human influenza A infection cross-react with avian influenza A (H5N1) in healthy individuals. J. Clin. Invest. 118, 3478–3490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Richards K. A., Topham D., Chaves F. A., Sant A. J. (2010) Cutting edge: CD4 T cells generated from encounter with seasonal influenza viruses and vaccines have broad protein specificity and can directly recognize naturally generated epitopes derived from the live pandemic H1N1 virus. J. Immunol. 185, 4998–5002 [DOI] [PubMed] [Google Scholar]
- 93. Cusick M. F., Wang S., Eckels D. D. (2009) In vitro responses to avian influenza H5 by human CD4 T cells. J. Immunol. 183, 6432–6441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Ge X., Tan V., Bollyky P. L., Standifer N. E., James E. A., Kwok W. W. (2010) Assessment of seasonal influenza A virus-specific CD4 T-cell responses to 2009 pandemic H1N1 swine-origin influenza A virus. J. Virol. 84, 3312–3319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Hayden F. G., Fritz R., Lobo M. C., Alvord W., Strober W., Straus S. E. (1998) Local and systemic cytokine responses during experimental human influenza A virus infection. Relation to symptom formation and host defense. J. Clin. Invest. 101, 643–649 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Peiris J. S., Yu W. C., Leung C. W., Cheung C. Y., Ng W. F., Nicholls J. M., Ng T. K., Chan K. H., Lai S. T., Lim W. L., Yuen K. Y., Guan Y. (2004) Re-emergence of fatal human influenza A subtype H5N1 disease. Lancet 363, 617–619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Jorens P. G., Van Damme J., De Backer W., Bossaert L., De Jongh R. F., Herman A. G., Rampart M. (1992) Interleukin 8 (IL-8) in the bronchoalveolar lavage fluid from patients with the adult respiratory distress syndrome (ARDS) and patients at risk for ARDS. Cytokine 4, 592–597 [DOI] [PubMed] [Google Scholar]
- 98. Sung R. Y., Hui S. H., Wong C. K., Lam C. W., Yin J. (2001) A comparison of cytokine responses in respiratory syncytial virus and influenza A infections in infants. Eur. J. Pediatr. 160, 117–122 [DOI] [PubMed] [Google Scholar]
- 99. Laham F. R., Israele V., Casellas J. M., Garcia A. M., Lac Prugent C. M., Hoffman S. J., Hauer D., Thumar B., Name M. I., Pascual A., Taratutto N., Ishida M. T., Balduzzi M., Maccarone M., Jackli S., Passarino R., Gaivironsky R. A., Karron R. A., Polack N. R., Polack F. P. (2004) Differential production of inflammatory cytokines in primary infection with human metapneumovirus and with other common respiratory viruses of infancy. J. Infect. Dis. 189, 2047–2056 [DOI] [PubMed] [Google Scholar]
- 100. Welliver T. P., Garofalo R. P., Hosakote Y., Hintz K. H., Avendano L., Sanchez K., Velozo L., Jafri H., Chavez-Bueno S., Ogra P. L., McKinney L., Reed J. L., Welliver R. C., Sr. (2007) Severe human lower respiratory tract illness caused by respiratory syncytial virus and influenza virus is characterized by the absence of pulmonary cytotoxic lymphocyte responses. J. Infect. Dis. 195, 1126–1136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Garofalo R. P., Hintz K. H., Hill V., Patti J., Ogra P. L., Welliver R. C., Sr. (2005) A comparison of epidemiologic and immunologic features of bronchiolitis caused by influenza virus and respiratory syncytial virus. J. Med. Virol. 75, 282–289 [DOI] [PubMed] [Google Scholar]
- 102. De Jong M. D., Simmons C. P., Thanh T. T., Hien V. M., Smith G. J., Chau T. N., Hoang D. M., Chau N. V., Khanh T. H., Dong V. C., Qui P. T., Cam B. V., Ha do Q., Guan Y., Peiris J. S., Chinh N. T., Hien T. T., Farrar J. (2006) Fatal outcome of human influenza A (H5N1) is associated with high viral load and hypercytokinemia. Nat. Med. 12, 1203–1207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Everard M. L., Swarbrick A., Wrightham M., McIntyre J., Dunkley C., James P. D., Sewell H. F., Milner A. D. (1994) Analysis of cells obtained by bronchial lavage of infants with respiratory syncytial virus infection. Arch. Dis. Child 71, 428–432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Smith P. K., Wang S. Z., Dowling K. D., Forsyth K. D. (2001) Leucocyte populations in respiratory syncytial virus-induced bronchiolitis. J. Paediatr. Child Health 37, 146–151 [DOI] [PubMed] [Google Scholar]
- 105. Salentin R., Gemsa D., Sprenger H., Kaufmann A. (2003) Chemokine receptor expression and chemotactic responsiveness of human monocytes after influenza A virus infection. J. Leukoc. Biol. 74, 252–259 [DOI] [PubMed] [Google Scholar]
- 106. Beigel J. H., Farrar J., Han A. M., Hayden F. G., Hyer R., de Jong M. D., Lochindarat S., Nguyen T. K., Nguyen T. H., Tran T. H., Nicoll A., Touch S., Yuen K. Y. (2005) Avian influenza A (H5N1) infection in humans. N. Engl. J. Med. 353, 1374–1385 [DOI] [PubMed] [Google Scholar]
- 107. Monsalvo A. C., Batalle J. P., Lopez M. F., Krause J. C., Klemenc J., Hernandez J. Z., Maskin B., Bugna J., Rubinstein C., Aguilar L., Dalurzo L., Libster R., Savy V., Baumeister E., Cabral G., Font J., Solari L., Weller K. P., Johnson J., Echavarria M., Edwards K. M., Chappell J. D., Crowe J. E., Jr., Williams J. V., Melendi G. A., Polack F. P. (2011) Severe pandemic 2009 H1N1 influenza disease due to pathogenic immune complexes. Nat. Med. 17, 195–199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Long C. B., Ramos I., Rastogi D., Manwani D., Janow G., Del Rio M., Mayers M., Herold B. C., Fernandez-Sesma A., Madan R. P. (2012) Humoral and cell-mediated immune responses to monovalent 2009 influenza A/H1N1 and seasonal trivalent influenza vaccines in high-risk children. J. Pediatr. 160, 74–81 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Lawson T. M., Man S., Williams S., Boon A. C., Zambon M., Borysiewicz L. K. (2001) Influenza A antigen exposure selects dominant Vβ17+ TCR in human CD8+ cytotoxic T cell responses. Int. Immunol. 13, 1373–1381 [DOI] [PubMed] [Google Scholar]
- 110. Wagar L. E., Gentleman B., Pircher H., McElhaney J. E., Watts T. H. (2011) Influenza-specific T cells from older people are enriched in the late effector subset and their presence inversely correlates with vaccine response. PLoS One 6, e23698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Boon A. C., Fringuelli E., Graus Y. M., Fouchier R. A., Sintnicolaas K., Iorio A. M., Rimmelzwaan G. F., Osterhaus A. D. (2002) Influenza A virus specific T cell immunity in humans during aging. Virology 299, 100–108 [DOI] [PubMed] [Google Scholar]
- 112. McElhaney J. E. (2011) Influenza vaccine responses in older adults. Ageing Res. Rev. 10, 379–388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Murasko D. M., Bernstein E. D., Gardner E. M., Gross P., Munk G., Dran S., Abrutyn E. (2002) Role of humoral and cell-mediated immunity in protection from influenza disease after immunization of healthy elderly. Exp. Gerontol. 37, 427–439 [DOI] [PubMed] [Google Scholar]
- 114. Co M. D., Orphin L., Cruz J., Pazoles P., Rothman A. L., Ennis F. A., Terajima M. (2008) Discordance between antibody and T cell responses in recipients of trivalent inactivated influenza vaccine. Vaccine 26, 1990–1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. McElhaney J. E., Xie D., Hager W. D., Barry M. B., Wang Y., Kleppinger A., Ewen C., Kane K. P., Bleackley R. C. (2006) T cell responses are better correlates of vaccine protection in the elderly. J. Immunol. 176, 6333–6339 [DOI] [PubMed] [Google Scholar]
- 116. La Ruche G., Tarantola A., Barboza P., Vaillant L., Gueguen J., Gastellu-Etchegorry M. (2009) The 2009 pandemic H1N1 influenza and indigenous populations of the Americas and the Pacific. Euro Surveill 14, pii: 19366 [DOI] [PubMed] [Google Scholar]
- 117. Webb S. A., Pettila V., Seppelt I., Bellomo R., Bailey M., Cooper D. J., Cretikos M., Davies A. R., Finfer S., Harrigan P. W., Hart G. K., Howe B., Iredell J. R., McArthur C., Mitchell I., Morrison S., Nichol A. D., Paterson D. L., Peake S., Richards B., Stephens D., Turner A., Yung M. (2009) Critical care services and 2009 H1N1 influenza in Australia and New Zealand. N. Engl. J. Med. 361, 1925–1934 [DOI] [PubMed] [Google Scholar]
- 118. U.S. Centers for Disease Control and Prevention (1919) Public Health Weekly Reports for October 17, 1919. Public Health Rep. 34, 2289–333 [PMC free article] [PubMed] [Google Scholar]
- 119. Tang Y. W., Li H., Wu H., Shyr Y., Edwards K. M. (2007) Host single-nucleotide polymorphisms and altered responses to inactivated influenza vaccine. J. Infect. Dis. 196, 1021–1025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Zuniga J., Buendia I., Zhao Y., Jimenez L., Torres D., Romo J., Ramirez G., Cruz A., Vargas-Alarcon G., Sheu C. C., Chen F., Su L., Tager A. M., Pardo A., Selman M., Christiani D. C. (2011) Genetic variants associated with severe pneumonia in A/H1N1 influenza infection. Eur. Respir. J., Epub ahead of print. doi: 10.1183/09031936.00020611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Gianfrani C., Oseroff C., Sidney J., Chesnut R. W., Sette A. (2000) Human memory CTL response specific for influenza A virus is broad and multispecific. Hum. Immunol. 61, 438–452 [DOI] [PubMed] [Google Scholar]
- 122. Wahl A., Schafer F., Bardet W., Buchli R., Air G. M., Hildebrand W. H. (2009) HLA class I molecules consistently present internal influenza epitopes. Proc. Natl. Acad. Sci. USA 106, 540–545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Rouse B. T., Sehrawat S. (2010) Immunity and immunopathology to viruses: what decides the outcome? Nat. Rev. Immunol. 10, 514–526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Suzuki T., Chow C. W., Downey G. P. (2008) Role of innate immune cells and their products in lung immunopathology. Int. J. Biochem. Cell Biol. 40, 1348–1361 [DOI] [PubMed] [Google Scholar]
- 125. Subrata L. S., Bizzintino J., Mamessier E., Bosco A., McKenna K. L., Wikstrom M. E., Goldblatt J., Sly P. D., Hales B. J., Thomas W. R., Laing I. A., LeSouef P. N., Holt P. G. (2009) Interactions between innate antiviral and atopic immunoinflammatory pathways precipitate and sustain asthma exacerbations in children. J. Immunol. 183, 2793–2800 [DOI] [PubMed] [Google Scholar]
- 126. Fernandez-Sesma A., Marukian S., Ebersole B. J., Kaminski D., Park M. S., Yuen T., Sealfon S. C., Garcia-Sastre A., Moran T. M. (2006) Influenza virus evades innate and adaptive immunity via the NS1 protein. J. Virol. 80, 6295–6304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Osterlund P., Pirhonen J., Ikonen N., R̈onkkö E., Strengell M., Makela S. M., Broman M., Hamming O. J., Hartmann R., Ziegler T., Julkunen I. (2010) Pandemic H1N1 2009 influenza A virus induces weak cytokine responses in human macrophages and dendritic cells and is highly sensitive to the antiviral actions of interferons. J. Virol. 84, 1414–1422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Guillot L., Le Goffic R., Bloch S., Escriou N., Akira S., Chignard M., Si-Tahar M. (2005) Involvement of Toll-like receptor 3 in the immune response of lung epithelial cells to double-stranded RNA and influenza A virus. J. Biol. Chem. 280, 5571–5580 [DOI] [PubMed] [Google Scholar]
- 129. Barchet W., Krug A., Cella M., Newby C., Fischer J. A., Dzionek A., Pekosz A., Colonna M. (2005) Dendritic cells respond to influenza virus through TLR7- and PKR-independent pathways. Eur. J. Immunol. 35, 236–242 [DOI] [PubMed] [Google Scholar]
- 130. Lund J. M., Alexopoulou L., Sato A., Karow M., Adams N. C., Gale N. W., Iwasaki A., Flavell R. A. (2004) Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc. Natl. Acad. Sci. USA 101, 5598–5603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Mordstein M., Neugebauer E., Ditt V., Jessen B., Rieger T., Falcone V., Sorgeloos F., Ehl S., Mayer D., Kochs G., Schwemmle M., Gunther S., Drosten C., Michiels T., Staeheli P. (2010) λ Interferon renders epithelial cells of the respiratory and gastrointestinal tracts resistant to viral infections. J. Virol. 84, 5670–5677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Hartshorn K. L., Crouch E. C., White M. R., Eggleton P., Tauber A. I., Chang D., Sastry K. (1994) Evidence for a protective role of pulmonary surfactant protein D (SP-D) against influenza A viruses. J. Clin. Invest. 94, 311–319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Hartshorn K. L., White M. R., Voelker D. R., Coburn J., Zaner K., Crouch E. C. (2000) Mechanism of binding of surfactant protein D to influenza A viruses: importance of binding to haemagglutinin to antiviral activity. Biochem. J. 351, 449–458 [PMC free article] [PubMed] [Google Scholar]
- 134. LeVine A. M., Whitsett J. A., Hartshorn K. L., Crouch E. C., Korfhagen T. R. (2001) Surfactant protein D enhances clearance of influenza A virus from the lung in vivo. J. Immunol. 167, 5868–5873 [DOI] [PubMed] [Google Scholar]
- 135. LeVine A. M., Hartshorn K., Elliott J., Whitsett J., Korfhagen T. (2002) Absence of SP-A modulates innate and adaptive defense responses to pulmonary influenza infection. Am. J. Physiol. Lung Cell. Mol. Physiol. 282, L563–L572 [DOI] [PubMed] [Google Scholar]
- 136. Hartshorn K. L., Collamer M., Auerbach M., Myers J. B., Pavlotsky N., Tauber A. I. (1988) Effects of influenza A virus on human neutrophil calcium metabolism. J. Immunol. 141, 1295–1301 [PubMed] [Google Scholar]
- 137. Bals R., Wang X., Zasloff M., Wilson J. M. (1998) The peptide antibiotic LL-37/hCAP-18 is expressed in epithelia of the human lung where it has broad antimicrobial activity at the airway surface. Proc. Natl. Acad. Sci. USA 95, 9541–9546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Agerberth B., Charo J., Werr J., Olsson B., Idali F., Lindbom L., Kiessling R., Jornvall H., Wigzell H., Gudmundsson G. H. (2000) The human antimicrobial and chemotactic peptides LL-37 and α-defensins are expressed by specific lymphocyte and monocyte populations. Blood 96, 3086–3093 [PubMed] [Google Scholar]
- 139. Barlow P. G., Svoboda P., Mackellar A., Nash A. A., York I. A., Pohl J., Davidson D. J., Donis R. O. (2011) Antiviral activity and increased host defense against influenza infection elicited by the human cathelicidin LL-37. PLoS One 6, e25333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Lehrer R. I., Barton A., Daher K. A., Harwig S. S., Ganz T., Selsted M. E. (1989) Interaction of human defensins with Escherichia coli. Mechanism of bactericidal activity. J. Clin. Invest. 84, 553–561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Ashitani J., Mukae H., Hiratsuka T., Nakazato M., Kumamoto K., Matsukura S. (2002) Elevated levels of α-defensins in plasma and BAL fluid of patients with active pulmonary tuberculosis. Chest 121, 519–526 [DOI] [PubMed] [Google Scholar]
- 142. Territo M. C., Ganz T., Selsted M. E., Lehrer R. (1989) Monocyte-chemotactic activity of defensins from human neutrophils. J. Clin. Invest. 84, 2017–2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Hartshorn K. L., White M. R., Shepherd V., Reid K., Jensenius J. C., Crouch E. C. (1997) Mechanisms of anti-influenza activity of surfactant proteins A and D: comparison with serum collectins. Am. J. Physiol. 273, L1156–L1166 [DOI] [PubMed] [Google Scholar]
- 144. Daher K. A., Selsted M. E., Lehrer R. I. (1986) Direct inactivation of viruses by human granulocyte defensins. J. Virol. 60, 1068–1074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Salvatore M., Garcia-Sastre A., Ruchala P., Lehrer R.I., Chang T., Klotman M. E. (2007) α-Defensin inhibits influenza virus replication by cell-mediated mechanism(s). J. Infect. Dis. 196, 835–843 [DOI] [PubMed] [Google Scholar]
- 146. Geissmann F., Jung S., Littman D. R. (2003) Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19, 71–82 [DOI] [PubMed] [Google Scholar]
- 147. Belge K. U., Dayyani F., Horelt A., Siedlar M., Frankenberger M., Frankenberger B., Espevik T., Ziegler-Heitbrock L. (2002) The proinflammatory CD14+CD16+DR++ monocytes are a major source of TNF. J. Immunol. 168, 3536–3542 [DOI] [PubMed] [Google Scholar]
- 148. Fingerle G., Pforte A., Passlick B., Blumenstein M., Strobel M., Ziegler-Heitbrock H. W. (1993) The novel subset of CD14+/CD16+ blood monocytes is expanded in sepsis patients. Blood 82, 3170–3176 [PubMed] [Google Scholar]
- 149. Cros J., Cagnard N., Woollard K., Patey N., Zhang S. Y., Senechal B., Puel A., Biswas S. K., Moshous D., Picard C., Jais J. P., D'Cruz D., Casanova J. L., Trouillet C., Geissmann F. (2010) Human CD14dim monocytes patrol and sense nucleic acids and viruses via TLR7 and TLR8 receptors. Immunity 33, 375–386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Azeredo E. L., Neves-Souza P. C., Alvarenga A. R., Reis S. R., Torrentes-Carvalho A., Zagne S. M., Nogueira R. M., Oliveira-Pinto L. M., Kubelka C. F. (2010) Differential regulation of Toll-like receptor-2, Toll-like receptor-4, CD16 and human leucocyte antigen-DR on peripheral blood monocytes during mild and severe dengue fever. Immunology 130, 202–216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Grage-Griebenow E., Zawatzky R., Kahlert H., Brade L., Flad H., Ernst M. (2001) Identification of a novel dendritic cell-like subset of CD64(+)/CD16(+) blood monocytes. Eur. J. Immunol. 31, 48–56 [DOI] [PubMed] [Google Scholar]
- 152. Masten B. J., Olson G. K., Tarleton C. A., Rund C., Schuyler M., Mehran R., Archibeque T., Lipscomb M. F. (2006) Characterization of myeloid and plasmacytoid dendritic cells in human lung. J. Immunol. 177, 7784–7793 [DOI] [PubMed] [Google Scholar]
- 153. Gill M. A., Long K., Kwon T., Muniz L., Mejias A., Connolly J., Roy L., Banchereau J., Ramilo O. (2008) Differential recruitment of dendritic cells and monocytes to respiratory mucosal sites in children with influenza virus or respiratory syncytial virus infection. J. Infect. Dis. 198, 1667–1676 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Hale B. G., Randall R. E., Ortin J., Jackson D. (2008) The multifunctional NS1 protein of influenza A viruses. J. Gen. Virol. 89, 2359–2376 [DOI] [PubMed] [Google Scholar]
- 155. Garcia-Sastre A., Egorov A., Matassov D., Brandt S., Levy D. E., Durbin J. E., Palese P., Muster T. (1998) Influenza A virus lacking the NS1 gene replicates in interferon-deficient systems. Virology 252, 324–330 [DOI] [PubMed] [Google Scholar]
- 156. Versteeg G. A., Hale B. G., van Boheemen S., Wolff T., Lenschow D. J., Garcia-Sastre A. (2010) Species-specific antagonism of host ISGylation by the influenza B virus NS1 protein. J. Virol. 84, 5423–5430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Serbina N. V., Jia T., Hohl T. M., Pamer E. G. (2008) Monocyte-mediated defense against microbial pathogens. Annu. Rev. Immunol. 26, 421–452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. Dawson T. C., Beck M. A., Kuziel W. A., Henderson F., Maeda N. (2000) Contrasting effects of CCR5 and CCR2 deficiency in the pulmonary inflammatory response to influenza A virus. Am. J. Pathol. 156, 1951–1959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Wareing M. D., Lyon A., Inglis C., Giannoni F., Charo I., Sarawar S. R. (2007) Chemokine regulation of the inflammatory response to a low-dose influenza infection in CCR2−/− mice. J. Leukoc. Biol. 81, 793–801 [DOI] [PubMed] [Google Scholar]
- 160. Aldridge J. R., Jr., Moseley C. E., Boltz D. A., Negovetich N. J., Reynolds C., Franks J., Brown S. A., Doherty P. C., Webster R. G., Thomas P. G. (2009) TNF/iNOS-producing dendritic cells are the necessary evil of lethal influenza virus infection. Proc. Natl. Acad. Sci. USA 106, 5306–5311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Marcelin G., Aldridge J. R., Duan S., Ghoneim H. E., Rehg J., Marjuki H., Boon A. C., McCullers J. A., Webby R. J. (2011) Fatal outcome of pandemic H1N1 2009 influenza virus infection is associated with immunopathology and impaired lung repair, not enhanced viral burden in pregnant mice. J. Virol. 85, 11208–11219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162. Herold S., von Wulffen W., Steinmueller M., Pleschka S., Kuziel W. A., Mack M., Srivastava M., Seeger W., Maus U. A., Lohmeyer J. (2006) Alveolar epithelial cells direct monocyte transepithelial migration upon influenza virus infection: impact of chemokines and adhesion molecules. J. Immunol. 177, 1817–1824 [DOI] [PubMed] [Google Scholar]
- 163. Wareing M. D., Shea A. L., Inglis C. A., Dias P. B., Sarawar S. R. (2007) CXCR2 is required for neutrophil recruitment to the lung during influenza virus infection, but is not essential for viral clearance. Viral Immunol. 20, 369–378 [DOI] [PubMed] [Google Scholar]
- 164. Seo S. U., Kwon H. J., Ko H. J., Byun Y. H., Seong B. L., Uematsu S., Akira S., Kweon M. N. (2011) Type I interferon signaling regulates Ly6C monocytes and neutrophils during acute viral pneumonia in mice. PLoS Pathog. 7, e1001304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165. Monticelli L. A., Sonnenberg G. F., Abt M. C., Alenghat T., Ziegler C. G., Doering T. A., Angelosanto J. M., Laidlaw B. J., Yang C. Y., Sathaliyawala T., Kubota M., Turner D., Diamond J. M., Goldrath A. W., Farber D. L., Collman R. G., Wherry E. J., Artis D. (2011) Innate lymphoid cells promote lung-tissue homeostasis after infection with influenza virus. Nat. Immunol. 12, 1045–1054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166. Colamussi M. L., White M. R., Crouch E., Hartshorn K. L. (1999) Influenza A virus accelerates neutrophil apoptosis and markedly potentiates apoptotic effects of bacteria. Blood 93, 2395–2403 [PubMed] [Google Scholar]
- 167. Fesq H., Bacher M., Nain M., Gemsa D. (1994) Programmed cell death (apoptosis) in human monocytes infected by influenza A virus. Immunobiology 190, 175–182 [DOI] [PubMed] [Google Scholar]
- 168. Craft A. W., Reid M. M., Low W. T. (1976) Effect of virus infections on polymorph function in children. Br. Med. J. 1, 1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169. Kleinerman E. S., Snyderman R., Daniels C. A. (1975) Depressed monocyte chemotaxis during acute influenza infection. Lancet 2, 1063–1066 [DOI] [PubMed] [Google Scholar]
- 170. Martin R. R., Couch R. B., Greenberg S. B., Cate T. R., Warr G. A. (1981) Effects of infection with influenza virus on the function of polymorphonuclear leukocytes. J. Infect. Dis. 144, 279–280 [DOI] [PubMed] [Google Scholar]
- 171. Sato M., Hosoya M., Wright P. F. (2009) Differences in serum cytokine levels between influenza virus A and B infections in children. Cytokine 47, 65–68 [DOI] [PubMed] [Google Scholar]
- 172. Divanovic S., Lai A. C. (2004) Cytokine induction in human cord blood lymphocytes after pulsing with UV-inactivated influenza viruses. Immunol. Lett. 94, 201–207 [DOI] [PubMed] [Google Scholar]
- 173. Rosenau J. M. (1919) Experiments to determine mode of spread of influenza. JAMA 73, 311–313 [Google Scholar]
- 174. Liu W., de Vlas S. J., Tang F., Ma M. J., Wei M. T., Liu L. J., Li Z. D., Zhang L., Xin Z. T., Tong Y. G., Jiang T., Zhang X. A., He C., Li C., Xu X. N., Yang H., Richardus J. H., Cao W. C. (2010) Clinical and immunological characteristics of patients with 2009 pandemic influenza A (H1N1) virus infection after vaccination. Clin. Infect. Dis. 51, 1028–1032 [DOI] [PubMed] [Google Scholar]
- 175. Gerhard W. (2001) The role of the antibody response in influenza virus infection. Curr. Top. Microbiol. Immunol. 260, 171–190 [DOI] [PubMed] [Google Scholar]
- 176. Khurana S., Suguitan A. L., Jr., Rivera Y., Simmons C. P., Lanzavecchia A., Sallusto F., Manischewitz J., King L. R., Subbarao K., Golding H. (2009) Antigenic fingerprinting of H5N1 avian influenza using convalescent sera and monoclonal antibodies reveals potential vaccine and diagnostic targets. PLoS Med. 6, e1000049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177. Greenbaum J. A., Kotturi M. F., Kim Y., Oseroff C., Vaughan K., Salimi N., Vita R., Ponomarenko J., Scheuermann R. H., Sette A., Peters B. (2009) Pre-existing immunity against swine-origin H1N1 influenza viruses in the general human population. Proc. Natl. Acad. Sci. USA 106, 20365–20370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178. Collin M., Bigley V., Haniffa M., Hambleton S. (2011) Human dendritic cell deficiency: the missing ID? Nat. Rev. Immunol 11, 575–583 [DOI] [PubMed] [Google Scholar]
- 179. Bhardwaj N., Bender A., Gonzalez N., Bui L. K., Garrett M. C., Steinman R. M. (1994) Influenza virus-infected dendritic cells stimulate strong proliferative and cytolytic responses from human CD8+ T cells. J. Clin. Invest. 94, 797–807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180. Cella M., Facchetti F., Lanzavecchia A., Colonna M. (2000) Plasmacytoid dendritic cells activated by influenza virus and CD40L drive a potent TH1 polarization. Nat. Immunol. 1, 305–310 [DOI] [PubMed] [Google Scholar]
- 181. Sareneva T., Matikainen S., Kurimoto M., Julkunen I. (1998) Influenza A virus-induced IFN-α/β and IL-18 synergistically enhance IFN-γ gene expression in human T cells. J. Immunol. 160, 6032–6038 [PubMed] [Google Scholar]
- 182. De Bree G. J., van Leeuwen E. M., Out T. A., Jansen H. M., Jonkers R. E., van Lier R. A. (2005) Selective accumulation of differentiated CD8+ T cells specific for respiratory viruses in the human lung. J. Exp. Med. 202, 1433–1442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183. Piet B., de Bree G. J., Smids-Dierdorp B. S., van der Loos C. M., Remmerswaal E. B., von der Thusen J. H., van Haarst J. M., Eerenberg J. P., ten Brinke A., van der Bij W., Timens W., van Lier R. A., Jonkers R. E. (2011) CD8 T cells with an intraepithelial phenotype upregulate cytotoxic function upon influenza infection in human lung. J. Clin. Invest. 121, 2254–2263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184. Hillaire M. L., van Trierum S. E., Bodewes R., van Baalen C. A., van Binnendijk R. S., Koopmans M. P., Fouchier R. A., Osterhaus A. D., Rimmelzwaan G. F. (2011) Characterization of the human CD8+ T cell response following infection with 2009 pandemic influenza H1N1 virus. J. Virol. 85, 12057–12061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185. Baluch A., Humar A., Egli A., Gubbay J., Lisboa L., Wilson L., Kumar D. (2011) Long term immune responses to pandemic influenza A/H1N1 infection in solid organ transplant recipients. PLoS One 6, e28627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186. Scheible K., Zhang G., Baer J., Azadniv M., Lambert K., Pryhuber G., Treanor J. J., Topham D. J. (2011) CD8+ T cell immunity to 2009 pandemic and seasonal H1N1 influenza viruses. Vaccine 29, 2159–2168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187. Tu W., Mao H., Zheng J., Liu Y., Chiu S. S., Qin G., Chan P. L., Lam K. T., Guan J., Zhang L., Guan Y., Yuen K. Y., Peiris J. S., Lau Y. L. (2010) Cytotoxic T lymphocytes established by seasonal human influenza cross-react against 2009 pandemic H1N1 influenza virus. J. Virol. 84, 6527–6535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188. Gras S., Kedzierski L., Valkenburg S. A., Laurie K., Liu Y. C., Denholm J. T., Richards M. J., Rimmelzwaan G. F., Kelso A., Doherty P. C., Turner S. J., Rossjohn J., Kedzierska K. (2010) Cross-reactive CD8+ T-cell immunity between the pandemic H1N1-2009 and H1N1-1918 influenza A viruses. Proc. Natl. Acad. Sci. USA 107, 12599–12604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189. Bonneville M., O'Brien R. L., Born W. K. (2010) γδ T cell effector functions: a blend of innate programming and acquired plasticity. Nat. Rev. Immunol 10, 467–478 [DOI] [PubMed] [Google Scholar]
- 190. Qin G., Mao H., Zheng J., Sia S. F., Liu Y., Chan P. L., Lam K. T., Peiris J. S., Lau Y. L., Tu W. (2009) Phosphoantigen-expanded human γδ T cells display potent cytotoxicity against monocyte-derived macrophages infected with human and avian influenza viruses. J. Infect. Dis. 200, 858–865 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191. Qin G., Liu Y., Zheng J., Ng I. H., Xiang Z., Lam K. T., Mao H., Li H., Peiris J. S., Lau Y. L., Tu W. (2011) Type 1 responses of human Vγ9Vδ2 T cells to influenza A viruses. J. Virol. 85, 10109–10116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192. Culley F. J., Pollott J., Openshaw P. J. (2002) Age at first viral infection determines the pattern of T cell-mediated disease during reinfection in adulthood. J. Exp. Med. 196, 1381–1386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193. Dakhama A., Park J. W., Taube C., Joetham A., Balhorn A., Miyahara N., Takeda K., Gelfand E. W. (2005) The enhancement or prevention of airway hyperresponsiveness during reinfection with respiratory syncytial virus is critically dependent on the age at first infection and IL-13 production. J. Immunol. 175, 1876–1883 [DOI] [PubMed] [Google Scholar]
- 194. You D., Becnel D., Wang K., Ripple M., Daly M., Cormier S. A. (2006) Exposure of neonates to respiratory syncytial virus is critical in determining subsequent airway response in adults. Respir. Res. 7, 107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195. You D., Ripple M., Balakrishna S., Troxclair D., Sandquist D., Ding L., Ahlert T. A., Cormier S. A. (2008) Inchoate CD8+ T cell responses in neonatal mice permit influenza-induced persistent pulmonary dysfunction. J. Immunol. 181, 3486–3494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196. Sihoe A. D., Wong R. H., Lee A. T., Lau L. S., Leung N. Y., Law K. I., Yim A. P. (2004) Severe acute respiratory syndrome complicated by spontaneous pneumothorax. Chest 125, 2345–2351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197. Lee N., Hui D., Wu A., Chan P., Cameron P., Joynt G. M., Ahuja A., Yung M. Y., Leung C. B., To K. F., Lui S. F., Szeto C. C., Chung S., Sung J. J. (2003) A major outbreak of severe acute respiratory syndrome in Hong Kong. N. Engl. J. Med. 348, 1986–1994 [DOI] [PubMed] [Google Scholar]
- 198. Boon A. C., de Mutsert G., Graus Y. M., Fouchier R. A., Sintnicolaas K., Osterhaus A. D., Rimmelzwaan G. F. (2002) The magnitude and specificity of influenza A virus-specific cytotoxic T-lymphocyte responses in humans is related to HLA-A and -B phenotype. J. Virol. 76, 582–590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199. Bednarek M. A., Sauma S. Y., Gammon M. C., Porter G., Tamhankar S., Williamson A. R., Zweerink H. J. (1991) The minimum peptide epitope from the influenza virus matrix protein. Extra and intracellular loading of HLA-A2. J. Immunol. 147, 4047–4053 [PubMed] [Google Scholar]
- 200. Wu C., Zanker D., Valkenburg S., Tan B., Kedzierska K., Zou Q. M., Doherty P. C., Chen W. (2011) Systematic identification of immunodominant CD8+ T-cell responses to influenza A virus in HLA-A2 individuals. Proc. Natl. Acad. Sci. USA 108, 9178–9183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201. Lehner P. J., Wang E. C., Moss P. A., Williams S., Platt K., Friedman S. M., Bell J. I., Borysiewicz L. K. (1995) Human HLA-A0201-restricted cytotoxic T lymphocyte recognition of influenza A is dominated by T cells bearing the V beta 17 gene segment. J. Exp. Med. 181, 79–91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202. Boon A. C., deBeauchamp J., Hollmann A., Luke J., Kotb M., Rowe S., Finkelstein D., Neale G., Lu L., Williams R. W., Webby R. J. (2009) Host genetic variation affects resistance to infection with a highly pathogenic H5N1 influenza A virus in mice. J. Virol. 83, 10417–10426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203. U.S. Centers for Disease Control and Prevention (2003) Diabetes prevalence among American Indians and Alaska Natives and the overall population—United States, 1994–2002. MMWR 52, 702–704 [PubMed] [Google Scholar]
- 204. Boon A. C., De Mutsert G., Fouchier R. A., Sintnicolaas K., Osterhaus A. D., Rimmelzwaan G. F. (2004) Preferential HLA usage in the influenza virus-specific CTL response. J. Immunol. 172, 4435–4443 [DOI] [PubMed] [Google Scholar]
- 205. Nakaya H. I., Li S., Pulendran B. (2011) Systems vaccinology: learning to compute the behavior of vaccine induced immunity. Wiley Interdiscip. Rev. Syst. Biol. Med. 4, 193–205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206. Anders E. M., Hartley C. A., Reading P. C., Ezekowitz R. A. (1994) Complement-dependent neutralization of influenza virus by a serum mannose-binding lectin. J. Gen. Virol. 75, 615–622 [DOI] [PubMed] [Google Scholar]
- 207. Kase T., Suzuki Y., Kawai T., Sakamoto T., Ohtani K., Eda S., Maeda A., Okuno Y., Kurimura T., Wakamiya N. (1999) Human mannan-binding lectin inhibits the infection of influenza A virus without complement. Immunology 97, 385–392 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208. Eisen D. P. (2010) Mannose-binding lectin deficiency and respiratory tract infection. J. Innate Immun. 2, 114–122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209. Eisen D. P., Marshall C., Dean M. M., Sasadeusz J., Richards M., Buising K., Cheng A., Johnson P. D., Barr I. G., McBryde E. S. (2011) No association between mannose-binding lectin deficiency and H1N1 2009 infection observed during the first season of this novel pandemic influenza virus. Hum. Immunol. 72, 1091–1094 [DOI] [PMC free article] [PubMed] [Google Scholar]