Abstract
The spread of multiple myeloma (MM) involves (re)circulation into the peripheral blood and (re)entrance or homing of MM cells into new sites of the BM. Hypoxia in solid tumors was shown to promote metastasis through activation of proteins involved in the epithelial-mesenchymal transition (EMT) process. We hypothesized that MM-associated hypoxic conditions activate EMT-related proteins and promote metastasis of MM cells. In the present study, we have shown that hypoxia activates EMT-related machinery in MM cells, decreases the expression of E-cadherin, and, consequently, decreases the adhesion of MM cells to the BM and enhances egress of MM cells to the circulation. In parallel, hypoxia increased the expression of CXCR4, consequently increasing the migration and homing of circulating MM cells to new BM niches. Further studies to manipulate hypoxia to regulate tumor dissemination as a therapeutic strategy are warranted.
Introduction
Multiple myeloma (MM) involves a continuous spread of MM cells in and out of the BM,1,2 and progression of MM occurs through the continuous interaction between the BM and the MM cells, controlling the ability of MM cells to egress out of the BM to the bloodstream and home into new BM niches. The interaction of MM cells with the BM microenvironment plays a crucial role in MM progression and dissemination.3–5 We showed previously that disrupting the interaction between MM cells and the BM microenvironment through inhibition of CXCR4 or its downstream signaling altered MM cell trafficking, including inhibition of MM cell homing to the BM, decreased MM proliferation, tumor progression, and drug resistance.6–8 However, the driving force that leads MM cells to egress from one site of the BM to disseminate and home to the next BM site is still not fully understood.
Regions of low oxygenation (hypoxia) arise in tumors because of rapid cell division and aberrant blood vessel formation.9 Several studies have indicated that the hypoxic microenvironment contributes to cancer progression by activating adaptive transcriptional programs, thereby promoting tumor-cell survival, motility, and metastasis.10,11 The BM of MM patients was shown recently to be hypoxic, so targeting hypoxia niches was suggested as a novel approach for the treatment of MM.12–14 Intratumoral hypoxia, followed by stabilization of hypoxia-inducible factor 1-alpha (HIF1α), promotes metastasis through acquisition of epithelial-mesenchymal transition (EMT) features in several models of solid tumors.15–17 EMT is fundamental for embryonic development and involves changes that include the loss of cell-cell adhesion and the acquisition of migratory and invasive properties.18 The acquisition of EMT features in cancer cells has been associated with metastasis and is involved in carcinogenesis, stem cell features, invasion, and tumor recurrence.19–21 EMT, however, is a phenomenon that occurs in solid tumors. In the present study, we sought to investigate whether hypoxia regulates egress and dissemination in MM and, if so, whether this occurs through the acquisition of EMT-like features. Our results reveal unprecedented features of MM cells acquiring metastatic potential because of hypoxia in the BM microenvironment, leading to de-adhesion and increased cell migration and homing to new BM niches.
Methods
Cells
The human MM cell lines MM1s, H929, U266, RPMI8226, and LP1 were purchased from ATCC. Stromal cells were obtained from BM samples from MM patients as described previously.22 Informed consent was obtained from all patients and donors in accordance with the Declaration of Helsinki. Approval for these studies was obtained by the Dana-Farber Cancer Institute Institutional Review Board.
Animals models for MM in vivo
Male SCID mice (7-9 weeks of age) were obtained from Charles River Laboratories and injected with human MM1s cells. Approval for these studies was obtained from the Dana-Farber Cancer Institute and Massachusetts General Hospital Institutional Animal Care and Use Committees.
The 5T33 MM originated spontaneously in aging C57BL/KaLwRij mice and has since been propagated in vivo by IV transfer of the diseased BM in young syngeneic mice.23 C57BL/KaLwRijHsd mice were purchased from Harlan CPB. Mice were 6-10 weeks of age when used and were housed and treated following the conditions approved by the Ethical Committee for Animal Experiments, Vrije Universiteit Brussel (license LA1230281).
Effect of hypoxia on dissemination of MM in vivo
For evaluation of the kinetics of tumor burden and hypoxia in the SCID-MM1s model, MM1s were genetically engineered to express green fluorescent protein (GFP) and luciferase, as described previously (MM1s-GFP+/Luc+).6 MM1s-GFP+/Luc+ cells were injected into 12 SCID mice and tumor progression was monitored weekly through IP injection of luciferin and whole-body imaging using bioluminescent imaging (BLI). Mice with different stages of tumor development based on tumor size detected by BLI were treated with the hypoxia marker pimonidazole hydrochloride (PIM; 100 mg/kg by IP injection; Hypoxyprobe Store). After 4 hours, peripheral blood (PB) was drawn and BM was isolated from one femur by flushing with cold PBS; the other femur was used for immuno-histochemistry (IHC). RBCs in the PB and BM sample were lysed; mononuclear cells (MNCs) were obtained by centrifugation, followed by washing with cold PBS. MNCs from the PB and BM were used for flow cytometric analysis.
For evaluation of the kinetics of tumor burden and the hypoxia in the BM of the 5T33MM murine model, 18 C57BL/KaLwRij mice were injected IV with 0.5 × 106 5T33MM cells (n = 6); 1 group of 6 untreated mice was included as a negative control (day 0). At different time points (days 0, 7, 14, and 24) during the development of disease, the mice were killed to: (1) monitor the number of circulating MM cells in the PB by flow cytometry, and (2) isolate the femur and tibiae for IHC staining of the hypoxia markers HIF1α and PIM. Mice were administered 60 mg/kg of PIM IV 4 hours before being biopsied.
Flow cytometric analysis of GFP, PIM, E-cadherin, and CXCR4 on MM cells and stromal cells
MNCs obtained from the PB and BM of mice at different stages of MM were fixed and permeabilized with 90% cold ethanol in PBS, washed with cold PBS, and blocked with 5% FBS in PBS. Cells were stained with mouse anti–human cadherin mAb (clone CH-19; Abcam), followed by a secondary Alexa Fluor 594–conjugated goat anti–mouse Ab (Invitrogen; excitement, 532 nm; filter, 610/20nm), washed twice, and stained with allophycocyanin-conjugated anti-PIM (Hypoxyprobe Store; excitement, 635 nm; filter, 660/20nm), PE-conjugated mouse anti–human CXCR4 (clone 12G5; BD Biosciences; excitement, 488 nm; filter, 575/26nm), and samples were analyzed by flow cytometry using a FACSAria (BD Biosciences). MM cells in the BM and PB were identified by gating cells with a high GFP signal (excitement, 488 nm; filter, 510/20nm); cells negative for GFP were considered to be stroma.
In some cases, MM cells (MM1s, H929, U266, and LP1) were cultured under normoxic or hypoxic (1% or 6% O2) conditions for 24 hours. In some cases, hypoxic MM cells were exposed to normal oxygen levels for 1 hour or incubated in medium from normoxic or hypoxic stroma for 6 hours and then analyzed by flow cytometry. For flow cytometric analysis, cells were then washed with cold PBS and stained with PE-conjugated mouse anti–human CXCR4 (clone 12G5; BD Biosciences; excitement, 488 nm; filter, 575/26nm) and the expression of CXCR4 was determined by flow cytometry on a FACSCanto II (BD Biosciences). HIF1α was down-regulated in MM cells using transfection with siRNA.
To detect the MM cells in the PB during the development of the 5T33MM model, PB was drawn from each mouse, followed by washing and RBC lysis. MNCs from the PB were then used for flow cytometric analysis. PE-conjugated rat anti–mouse CD138 (561070; BD Pharmingen) was used to stain MM cells for this purpose, PE-labeled rat IgG2a (κ) was used as a negative control. Biotin-labeled 5T33MMvv-specific idiotype and streptavidin-FITC were also used to identify 5T33vv MM in the PB.24,25
Detection of CD138, HIF1α, PIM, and E-cadherin by IHC
Femurs from mice in the disseminated xenograft MM model were isolated from mice at different stages of MM. After injection of MM1s-GFP-Luc, cells were rinsed with cold PBS, fixed with 4% formaldehyde in PBS, dehydrated with ethanol, embedded in paraffin blocks, and sequentially sectioned. Sections were stained with mAbs for CD138 (clone B-A38; Abcam), HIF1α (clone H1α 67; Santa Cruz Biotechnology), and E-cadherin (clone 24E10; Cell Signaling Technology).
In the 5T33MM model, tibias and femurs were fixed in zinc fixative for 48 hours, decalcified for 48 hours, and embedded in paraffin. Rabbit polyclonal Ab (PAb2627; Hypoxyprobe Store), secondary Ab (goat anti–rabbit IgG-polymerized HRP; 3051-1; Epitomics), Universal Negative Control NC498AA, Ag retrieval reagent Pronase Reagent M31, and the Betazoid DAB Chromogen Kit BDB2004H (Biocare Medical) were used for the detection of Hypoxyprobe. For the detection of endogenous hypoxia marker, HIF1α Ab (NB100-479) was purchased from Novus Biologicals.
Gene-expression analysis
To determine the gene expression of HIF1α (probe ID 200989_at), HIF1β (probe ID 218221_at), HIF2β (probe ID 202986_at), CREBBP (probe ID 211808_s_at), HYOU1 (probe ID 200825_s_at), VEGF1 (probe ID 211527_x_at), HIF1α-inhibitor (probe ID 218525_s_at), E-cadherin (Probe ID 201131_s_at), SNAIL (probe ID 219480_at), FOXC2 (probe ID 214520_at), TGFb1 (probe ID 203085_s_at) in plasma cells isolated from normal subjects or from newly diagnosed MM patients, we used published datasets from the Gene Expression Omnibus by Chng et al (series number GSE 6477).26 Data points that were out of the range of mean ± 1.5 SD were considered “outliers” and were excluded.
Quantitative real-time PCR
RNA extraction was performed using the RNeasy kit (QIAGEN). RNA was analyzed using an Agilent 2100 bioanalyzer. Total RNA was reverse transcribed using the Verso cDNA synthesis kit (Thermo Scientific) according to the manufacturer's instructions. Real-time PCR was performed with Maxima SYBR Green/ROX qPCR Master Mix (Fermantas) on an ABI Prism 7900 Fast instrument using gene-specific primers. The primer sets used were: human CXCR4: forward CCACGCCACCAACAGTCAGAG; reverse GAAGTCGGGAATAGTCAGCA GGAG; mouse CXCR4: forward ACCTCTACAGCAGCGTTCTCATC; reverse CA CCACCATCCACAGGCTATCG; human β-actin: Forward ATCGTGCGTGACATT AAGGAGAAG; reverse AGGAAGGAAGGCTGGAAGAGTG; mouse β-actin: forward GCGACAGCAGTTGGTTGGAG; reverse TTTGGGAGG-GTGAGGGACTTC. The thermal cycling conditions included 2 minutes at 50°C and 10 minutes at 95°C, followed by 40 cycles of 95°C for 0.15 minutes and 60°C for 1 minute. Ct values were collected for β-actin and the genes of interest during the log phase of the cycle. Quantification of given genes expressed as mRNA level was normalized to β-actin RNA using the ΔΔCt method.
Down-regulation of HIF1α in MM cells using siRNA
MM1s cells were transiently transfected with HIF1α siRNA SmartPool (Dharmacon) using the Cell Line Nucleofectot Kit V according to the manufacturer's instructions (Amaxa Biosystems). Scrambled siRNA was used as a control. After transfection, MM1s cells were subjected to Western blotting and the expression of CXCR4 was examined using flow cytometry and chemotaxis assays. In some experiments, HIF1α was down-regulated in MM cells using transfection with siRNA.
Immunoblotting
MM cells (MM1s and H929) and stromal cells isolated from the BM of 3 different MM patients were cultured under normoxic or hypoxic (1% O2) conditions for 24 hours. In some cases hypoxic MM cells were incubated in media from hypoxic or normoxic stroma. In some experiments, HIF1α was down-regulated in MM cells using transfection with siRNA. The expression of HIF1α, HIF2α, pGSK3β, SNAIL, and E-cadherins was detected by immunoblotting using mAbs for HIF1α (clone H1α 67; Santa Cruz Biotechnology), HIF2α (clone A-5; Santa Cruz Biotechnology), pGSK3b (clone 5B3; Cell Signaling Technology), SNAIL (clone C15D3; Cell Signaling Technology), and E-cadherin (clone 24E10; Cell Signaling Technology), as described previously.7 β-actin was used as a loading control and was detected using a mAb for β-actin (clone 13E5; Cell Signaling Technology).
Adhesion assay
A confluent monolayer of BM stromal cells (BMSCs; passages 2-5) was generated by plating 1 × 104 cells/well in 96-well plates for 24 hours. In some cases, BMSCs were then cultured under hypoxic (1% or 6% O2) or normoxic conditions for 24 hours. MM1s and H929 cells were cultured under hypoxic or normoxic conditions for 24 hours. MM cells were fluorescently labeled by calcein-AM (1 μg/mL for 1 hour), washed, and a suspension of 0.5 × 106 cells/mL was prepared. In some experiments, MM cells were incubated in medium from normoxic or hypoxic stromal cells for 6 hours and then analyzed by flow cytometry. In other experiments, MM cells or stroma were treated separately with E-cadherin–blocking Ab for 1 hour, and then washed before being used in the adhesion assay. BMSCs were washed, 100 μL of a MM cell suspension was added to each well, and then cells were incubated for 1 hour. For cells cultured under hypoxic conditions, all procedures were performed in the hypoxic chamber. Nonadherent cells were washed and adhesion was detected by measuring the fluorescence intensity in the wells using a plate-reader-fluorometer (excitement/emission, 485/520 nm).
Chemotaxis assay
Chemotaxis was determined using 8-μ-pore filters for the Transwell migration assay (Costar; Corning), according to the manufacturer's instructions. MM cells (MM1s and H929) were cultured under normoxic or hypoxic (1% or 6% O2) conditions for 24 hours. In some experiments, HIF1α was down-regulated in MM cells using transfection with siRNA. In other cases, MM cells were treated with a 50μM concentration of the CXCR4 inhibitor AMD3100 (Sigma-Aldrich) for 2 hours before the migration assay. Then cells were placed in the upper migration chambers in the presence of medium containing 0 or 30nM recombinant SDF1α (R&D Systems) in the lower chamber. In some cases, conditioned medium from normoxic and hypoxic stromal cells was placed in the lower chamber. After 5 hours of incubation, cells that migrated to the lower chambers were counted using flow cytometry.
Effect of hypoxia on the secretion of SDF1α from stromal cells
A confluent monolayer of stromal cells isolated from 5 MM patients was generated by plating 5 × 104 cells/well in 24-well plates for 24 hours. Cells were then cultured under hypoxic (1% O2) or normoxic conditions for 24 hours. The conditioned medium was then filtered using a 0.2-μm filter and the levels of SDF1α were detected with the human CXCL12/SDF1α immunoassay according to the manufacturer's instructions (CXCL12/SDF1α ELISA kit; R&D Systems). Briefly, assay diluent was mixed with control standard or sample and then incubated for 2 hours at room temperature on a horizontal shaker. Samples were then aspirated and wells were washed 3 times. Anti-SDF1α Ab conjugated to HRP solution was added, incubated for 2 hours at room temperature, and then washed 3 times. Substrate solution was added for 30 minutes, followed by stop solution; optical density was measure at 450 nm, with subtraction of the reading at 570 nm.
Actin polymerization assay
MM1s cells were cultured under normoxic or hypoxic (1% O2) conditions for 24 hours and then treated with 0 or 30nM SDF1α for 1.5 minutes. Cells were immediately fixed in 2% formaldehyde for 15 minutes at room temperature, permeabilized with 0.2% saponin, and stained with (5 μg/mL) phalloidin tagged with Alexa Fluor 488. Cells were analyzed by flow cytometry (excitement, 488 nm; filter, 510/20nm).
Extravasation of MM cells in vivo
In vivo flow cytometry is a new technology that allows real-time continuous monitoring of fluorescent cells in the circulation of live animals without the need to draw blood samples. MM1s cells were cultured under normoxic or hypoxic (1% O2) conditions for 24 hours. In some cases, MM cells were treated with 50μM AMD3100 for 2 hours before the extravasation assay (3 mice were used for this condition). Hypoxic cells were labeled with calcein-AM (Invitrogen), and normoxic cells were labeled with calcein-red/orange (Invitrogen) in 2 mice and the colors were inverted in a third mouse. Equal numbers (2 × 106 cells) of each cell population were mixed and injected into the tail veins of SCID mice. In some experiments, HIF1α was down-regulated in MM cells using transfection with siRNA. Detection of circulating cells was as described previously.7
In vivo confocal imaging
MM1s cell homing to BM vasculature of the skull was analyzed using fluorescence confocal microscopy, as described previously.7 MM1s cells (1 × 106 cells) were cultured under normoxic or hypoxic (1% O2) conditions for 24 hours, labeled with calcein AM (Invitrogen), and injected via the tail vein into anesthetized SCID mice with a skin flap in their scalp to expose the underlying calvaria. Retro-orbital injection of 20 μL of Evans blue (2 mg/mL; Sigma-Aldrich) was done immediately before imaging. Four images of the BM niches (1 image/niche) were captured between 2 and 10 minutes after cell injection, and a map of the BM niches was consolidated.
Statistical analysis
Statistical differences between experimental groups were analyzed using Microsoft Office Excel 2003 software using the Student t test or single-factor ANOVA to assess the P values in correlations. P < .05 was considered statistically significant.
Results
Tumor progression increases hypoxia in MM cells in vivo
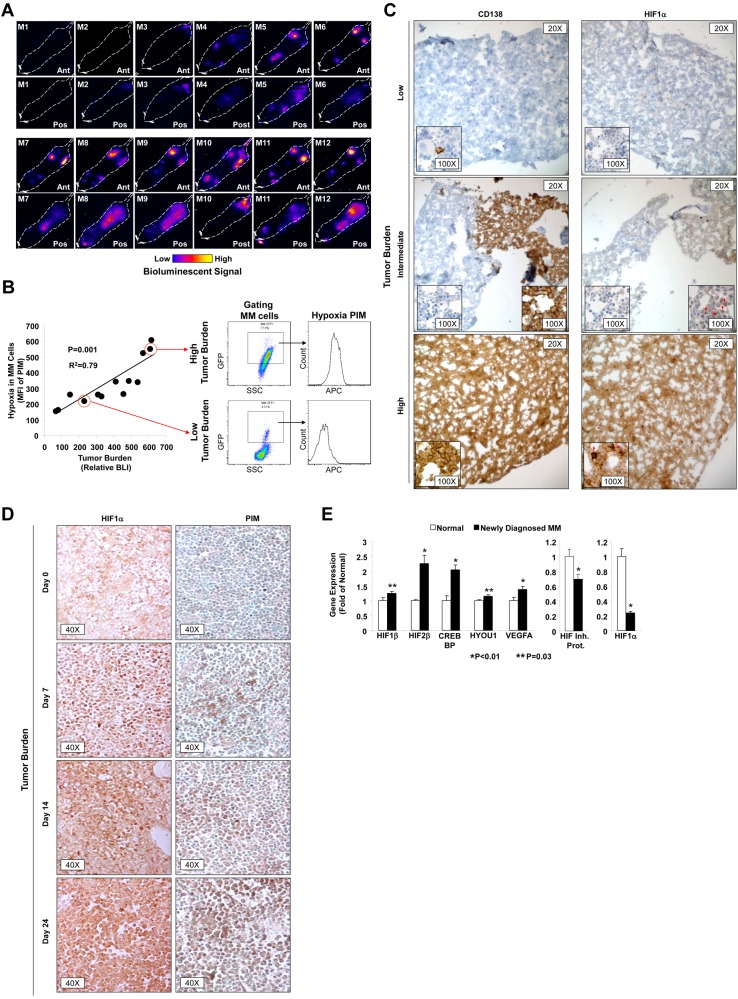
We used 2 animal models to examine the role of hypoxia in the progression of MM. In the first, we injected MM1s-GFP-Luc cells into 12 SCID mice via the tail vein and followed the progression of MM tumors in a disseminated xenograft model using luciferin and BLI. Figure 1A shows representative images of 12 mice with different stages of MM progression, which varied from minimal tumors detectable by BLI to advanced tumors. We compared tumor size with the mean fluorescence intensity (MFI) of the hypoxia marker PIM in the MM cells (GFP+ cells). The hypoxic state of the MM cells in the BM (MFI of PIM) was increased with tumor progression as detected by BLI (P = .001; Figure 1B). These results were confirmed by IHC. Specimens from the femurs of mice injected with MM1s-GFP-Luc with different levels of BLI were stained with Abs for CD138 and HIF1α; Figure 1C shows that specimens with higher CD138+ cell involvement had increased expression of HIF1α protein.
Figure 1.
Tumor progression increases hypoxia in MM cells in vivo. (A) Representative BLIs of 12 SCID mice with different levels of tumor burden after injection of MM1s-GFP-Luc cells. (B) Correlation between tumor burden as detected by BLI and hypoxia in MM cells shown as the MFI of allophycocyanin-PIM in the MM cells (GFP+). (C) IHC images of specimens taken from the femurs of mice injected with MM1s-GFP-Luc with different levels of BLI stained with Abs for CD138 and HIF1α. Red arrows show nuclear staining with HIF1α. (D) IHC images of specimens taken from mice at different time points after the injection of 5T33MM mouse cells stained with Abs for PIM and HIF1α showing that binding of PIM and expression of HIF1α were directly correlated with tumor burden. (E) Gene-expression analysis of hypoxia-induced genes (including HIF1β, HIF2β, CREBBP, HYOU1, and VEGF1) in plasma cells isolated from normal subjects and MM patients using published datasets from the Gene Expression Omnibus by Chng et al (series number GSE 647726).
These results were confirmed in our second model, the 5T33MM mouse model (Figure 1C). Specimens from mice with progression of murine MM in the 5T33MM model with different levels of tumor involvement were stained with Abs for PIM and HIF1α. Figure 1D shows that binding of PIM and the expression of HIF1α increased with tumor progression. To test the level of transcription regulation of hypoxia-induced genes in plasma cells isolated from the BM of normal subjects and MM patients, we analyzed the published gene-expression datasets by Chng et al (series number GSE 6477).26 Figure 1E shows an up-regulation of hypoxia-induced genes (including HIF1β, HIF2β, CREBBP, HYOU1, and VEGFA) and down-regulation of the ΗIF1α-inhibitory protein known to facilitate HIF1α protein degradation, demonstrating the induction of hypoxic response due to tumor progression. In contrast to the increase in the protein level, expression of HIF1α at the mRNA level was lower in MM patients compared with normal subjects. The low level of HIF1α α mRNA is consistent with prior data indicating a discrepancy between the protein and mRNA levels of ΗIF1α, which is associated with negative-feedback loops regulating the mRNA levels of ΗIF1α in chronic hypoxia.27–29
Hypoxia in the BM is correlated with the egress of MM to the PB
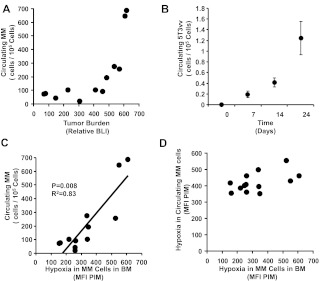
To test the role of hypoxia in MM-cell egress during tumor progression, we analyzed the number of circulating cells in mice with different levels of MM in our 2 animal models. The increased numbers of circulating cells associated with tumor progression was confirmed in both the SCID-MM1s model and the 5T33MM model (Figure 2A-B, respectively). However, the increase in the number of circulating cells was observed mainly in the late stages of tumor progression and not in the early stages. A better direct linear correlation was obtained between the number of circulating cells and the level of hypoxia in the cells in the BM (P = .008), possibly indicating that the egress of MM cells to the circulation could be regulated by hypoxia (Figure 2C). To further confirm the role of hypoxia in egress, we tested the hypoxia level using PIM in the circulating MM cells. Figure 2D shows that circulating MM cells sustained a steady level of hypoxia independently of the tumor burden in the BM, possibly indicating that hypoxic cells that egress from the BM sustain a certain level of hypoxia through the circulation to avoid anoikis until they home to new BM niches. This is consistent with prior observations in solid tumors showing that hypoxia induces resistance to anoikis in tumor cells that are traveling in the circulation,30,31 the time of most vulnerability to cell death during the stages of cell metastasis.
Figure 2.
Correlation of hypoxia in MM cells with their egress into the circulation. (A-B) Correlation between the number of circulating MM cells detected by flow cytometry and tumor progression in SCID-MM1s model detected by BLI in the disseminated xenograft model (A) and in the 5T33MM model (B). Results show increased numbers of circulating MM cells mainly in the late stages of tumor progression. (C) Correlation between the number of circulating cells and the level of hypoxia in MM cells in the BM indicated as MFI of PIM, which shows a direct linear correlation. (D) Correlation between hypoxia levels in circulating MM cells and hypoxia in MM cells in the BM from mice with different tumor burdens, which shows that circulating MM cells were hypoxic, with no correlation with the hypoxia in the BM.
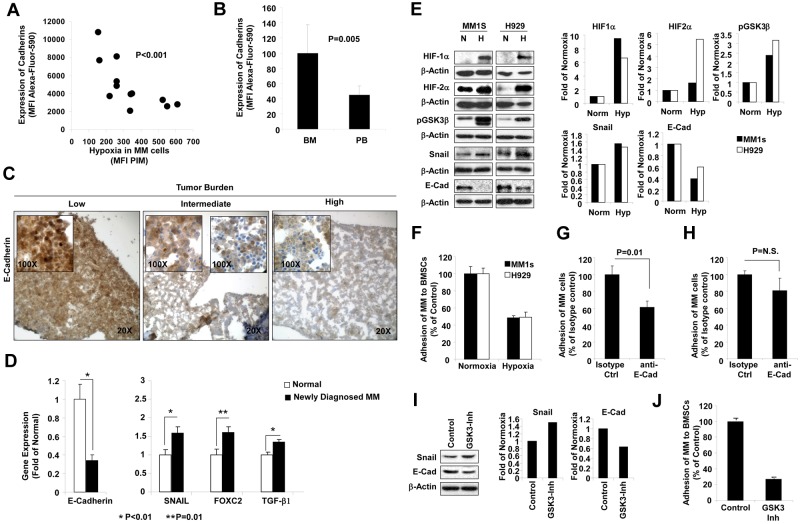
Hypoxia reduces adhesion of MM cells to the BM stroma via decreased expression of E-cadherin and increased EMT-regulating proteins
To investigate the mechanisms by which hypoxia induces MM-cell egress, we tested the expression of adhesion molecules during tumor progression. Cells isolated from the BM of mice at different stages of MM progression were analyzed for the expression of cadherins by flow cytometry and compared with the hypoxic state in these cells. Figure 3A shows that the expression of cadherins was decreased significantly with the increase in hypoxia in the MM cells (P < .001). Figure 3B shows that the expression of E-cadherin was lower in circulating MM cells than in MM cells present in the BM (P = .005). These results were confirmed by IHC; Figure 3C shows that the expression of E-cadherin was decreased with tumor progression in specimens isolated from mice in the disseminated xenograft model. These results were in agreement with the decreased expression of E-cadherin at the mRNA level in plasma cells isolated from the BM of MM patients compared with normal subjects (Figure 3D). The decrease of E-cadherin expression was accompanied by an increased expression of genes known to down-regulate the expression of E-cadherin in the EMT process, including SNAIL, FOXC2, and TGFβ1. Therefore, we tested the role of hypoxia in inducing EMT-related proteins and E-cadherin in vitro by immunoblotting. Incubation of MM cells (MM1s and H929) in hypoxic conditions in vitro expectedly increased the expression of HIF1α and HIF2α, in agreement with the findings in both the SCID-MM1s and the 5T33-MM model (Figure 3E). In addition, hypoxia inhibited (ie, increased phosphorylation) of GSK3β, increased the expression of SNAIL, and decreased the expression of E-cadherin. Consequently, incubation of MM cells (MM1s and H929) in hypoxia reduced their adhesion to a monolayer of BMSCs isolated from MM patients (Figure 3F). To further confirm that the effect was E-cadherin dependent, MM cells were treated with E-cadherin–blocking Ab before the adhesion assay, which caused a significant reduction in adhesion of normoxic MM cells (Figure 3G), but not hypoxic MM cells (Figure 3H). To better understand the role of hypoxia in decreasing adhesion through the SNAIL/E-cadherin axis, we mimicked the hypoxic response in MM cells by inhibition of GSK3β using a small-molecule inhibitor (GSK-3β Inhibitor XII; EMD Chemicals). Similar to the MM response to hypoxia, inhibition of GSK3β increased the expression of SNAIL, decreased the expression E-cadherin (Figure 3I), and consequently decreased the adhesion of MM cells to a monolayer of BMSCs (Figure 3J).
Figure 3.
Effect of hypoxia on expression of E-cadherin and adhesion to BMSCs. (A) MM cells isolated from the BM of mice injected with MM1s cells at different tumor burdens were analyzed for the expression of cadherins by flow cytometry and compared with the hypoxic state in these cells indicated as the MFI of PIM in the MM cells. The expression of cadherins was decreased significantly with the increase in hypoxia in the MM cells. (B) The expression of E-cadherin in circulating MM cells compared with MM cells in the BM, showing that circulating MM cells maintained a low expression of E-cadherin. (C) IHC images of specimens taken from the femurs of mice with different tumor burdens stained with Ab for E-cadherin, which shows that the expression of E-cadherin decreased with tumor progression. (D) Gene-expression analysis of E-cadherin and EMT-related genes (including SNAIL, FOXC2, and TGFβ1) in plasma cells isolated from normal subjects and MM patients using published datasets from the Gene Expression Omnibus by Chng et al (series number GSE 647726). This shows decreased E-cadherin expression and increased expression of SNAIL, FOXC2, and TGFβ1 in MM. (E) The effect of incubation of MM cells (MM1s and H929) under hypoxic conditions for 24 hours in vitro on expression of HIF1α and HIF2α GSK3β, SNAIL, and E-cadherin in MM cells detected by immunoblotting. (F) The effect of incubation of MM cells (MM1s and H929) under hypoxic conditions for 24 hours in vitro on adhesion to a monolayer of BMSCs isolated from MM patients. This shows decreased adhesion of hypoxic MM cells. The effect of treating MM cells with E-cadherin–blocking Ab on adhesion of MM cells to stromal cells under normoxic (G) or hypoxic (H) conditions. This shows that the blocking Ab significantly decreased adhesion in normoxic conditions but not in hypoxic conditions. Effect of inhibition of GSK3β by a small-molecule inhibitor under normoxic conditions on expression of SNAIL and E-cadherin detected by immunoblotting (I) and on adhesion of MM cells to a monolayer of BMSCs (J). This shows increased expression of SNAIL, decreased expression of E-cadherin, and decreased adhesion to BMSCs.
Progression induces hypoxia and decreased E-cadherin in the BM microenvironment
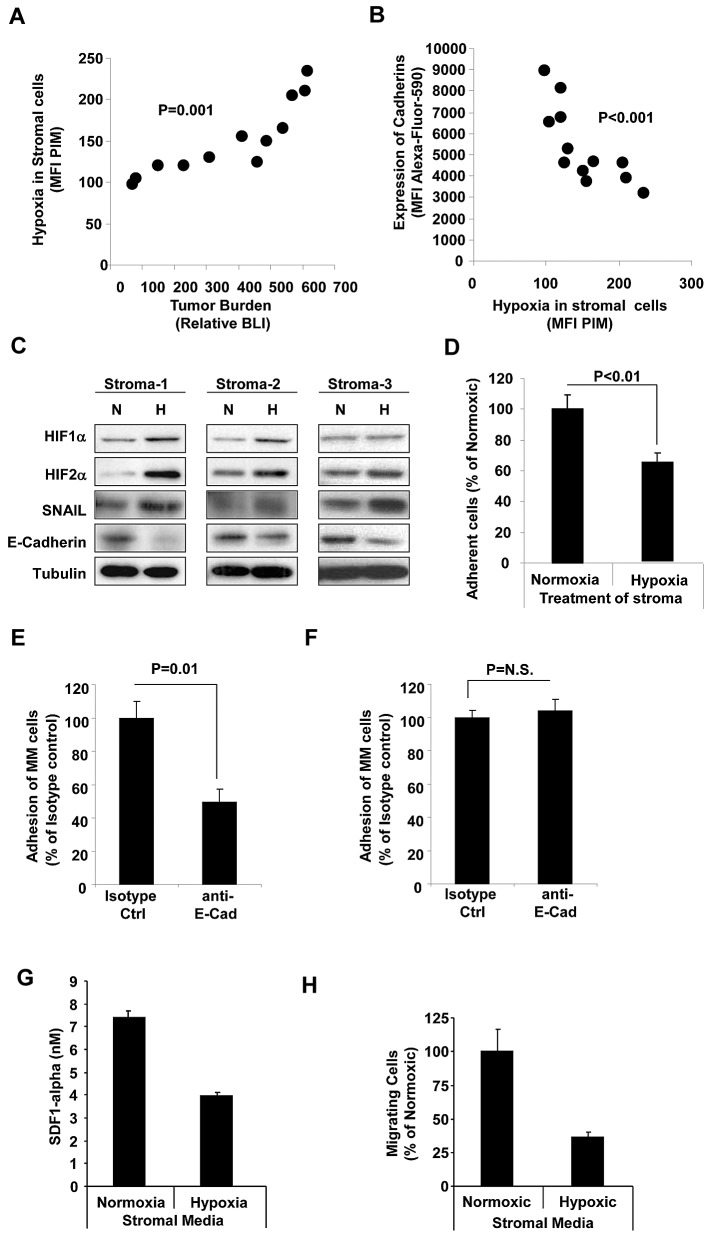
In parallel to the role of hypoxia on the clonal tumor cells, we investigated the effect of tumor progression on hypoxia in the BM microenvironment. In the SCID-MM1s model, MNCs isolated from the BM of mice at different stages of MM were analyzed for their hypoxic state (as detected by flow cytometry revealing the MFI of PIM in GFP− cells—MM cells are GFP+). Figure 4A shows that, similar to increased hypoxia in the MM cells, we found a direct correlation between hypoxia in BM stromal cells and tumor progression (as detected by BLI; P = .001). In addition, increased hypoxia was correlated with decreased expression of E-cadherin (P < .001). These results were confirmed in vitro by incubating BMSCs isolated from 3 MM patients under hypoxic conditions, which revealed increased expression of HIF1α, HIF2α, and SNAIL and decreased expression of E-cadherin (Figure 4C). Accordingly, the adhesion of nontreated MM cells to hypoxic BMSCs was decreased compared with normoxic BMSCs (Figure 4D). These results indicate that hypoxia in the BM microenvironment also plays a factor in inducing de-adhesion and egress of MM cells out of the BM and their dissemination to new, less hypoxic BM niches. To confirm that the effect was E-cadherin dependent, stromal cells were treated with E-cadherin–blocking Ab before the adhesion assay, which resulted in a significant reduction in the adhesion of MM cells to normoxic stroma (Figure 4E), but not to hypoxic stroma (Figure 4F).
Figure 4.
Effect of tumor progression on hypoxia and expression of E-cadherin in the BM microenvironment. (A) MNCs isolated from the BM of mice injected with MM1s. Different stages were analyzed and the hypoxic state of BM microenvironment was determined by flow cytometry as the MFI of PIM in GFP− cells. The correlation between hypoxia in the BM microenvironment and tumor progression (detected by BLI) demonstrates increased hypoxia in the microenvironment with tumor progression. (B) Correlation between expression of E-cadherin and level of hypoxia in BM microenvironment showing decreased expression of E-cadherin with hypoxia. (C) Effect of incubation of BMSCs (isolated from 3 different MM patients) under hypoxic conditions for 24 hours in vitro on expression of HIF1α, HIF2α, SNAIL, and E-cadherin in MM cells detected by immunoblotting. This shows increased expression of HIF1α, HIF2α, and SNAIL and decreased expression of E-cadherin in hypoxia. (D) The effect of incubation of BMSCs (isolated from 3 different MM patients) under hypoxic conditions for 24 hours in vitro on adhesion of MM cells to a monolayer of BMSCs. This shows decreased adhesion of MM cells to hypoxic BMSCs. The effect of treating stromal cells with E-cadherin–blocking Ab on adhesion of MM cells to stroma under normoxic (E) or hypoxic (F) conditions is shown. The blocking Ab decreased adhesion to normoxic but not to hypoxic stroma. (G) Effect of hypoxia on the secretion of SDF1α from MM stroma showing decreased secretion of SDF1α under hypoxic conditions. (H) The migration of MM cells to medium from normoxic and hypoxic stroma showing decreased migration of MM cells toward the medium from hypoxic stroma.
We next examined whether the hypoxic BM microenvironment regulates the secretion of SDF1α from the stroma. Figure 4G shows that there was a significant decrease in SDF1α secretion by hypoxic stroma compared with normoxic stroma. Consequently, migration of MM cells toward medium from hypoxic stroma was significantly lower compared with chemotaxis of MM cells toward medium from normoxic stroma (Figure 4H).
Hypoxia increases the expression of CXCR4 and chemotaxis of MM cells
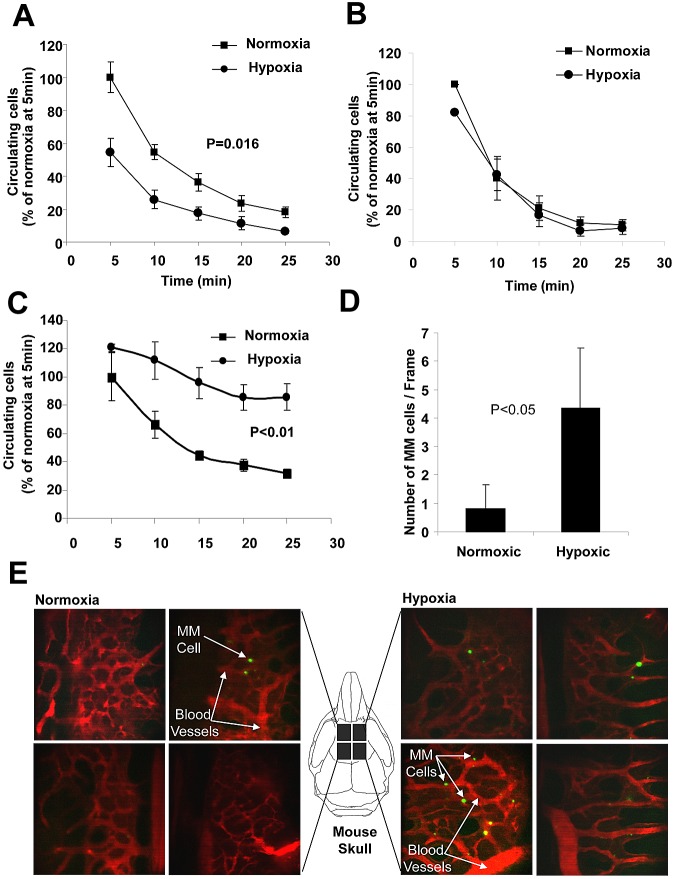
We also investigated whether hypoxia plays a role in facilitating MM chemotaxis to promote cell homing and dissemination, in addition to the decreased adhesion triggered by hypoxia. We showed previously that CXCR4 plays a major role in the homing of MM cells to the BM6; therefore, we tested the effect of hypoxia on the expression of CXCR4 in MM cells. Analysis of the expression of CXCR4 in MM cells isolated from BM of mice with different tumor burdens in the SCID-MM1s model showed a direct correlation between the level of hypoxia and the expression of CXCR4 (P < .001; Figure 5A). Moreover, testing the level of the expression of CXCR4 in cells isolated from the BM and PB revealed that MM cells in the PB had a higher expression of CXCR4 compared with MM in BM (Figure 5B). This is consistent with our previous data in patient samples showing that CXCR4 levels are higher in circulating tumor cells compared with those present in the BM in MM,1 and with our hypothesized model that hypoxia induces CXCR4 progression that leads to egress of these cells into the circulation and homing to BM. To confirm the effect of hypoxia on the expression of CXCR4, MM cells (MM1s, H929, LP1, and 5T33MMvt) were incubated under hypoxic or normoxic conditions for 24 hours and the expression of CXCR4 was analyzed by flow cytometry. Figure 5C shows that the expression of CXCR4 was increased in MM cells after hypoxia. Similarly, mRNA levels of CXCR4 increased after incubation of MM cells (RPMI, LP1, and 5T33) under hypoxic conditions (Figure 5D). Consequently, the chemotaxis of MM cells (MM1s and H929) toward SDF1α was increased significantly in hypoxic MM cells (Figure 5E). Mechanistically, SDF1α induced higher actin polymerization in hypoxic MM1s cells compared with normoxic cells (Figure 5F). To further confirm that the chemotactic effect was mediated through CXCR4, we used the CXCR4 inhibitor AMD3100 to abolish the increased chemotactic effect that hypoxia induced in MM cells (Figure 5G). In addition, we tested the effect of knockdown of HIF1α on the expression of CXCR4 and the chemotaxis of MM cells under hypoxic conditions. Figure 5H shows successful knockdown of HIF1α in MM1s cells: the expression of HIF1α in hypoxic MM cells was abrogated in cells with HIF1α knockdown. Functionally, down-regulation of HIF1α prevented the increase of CXCR4 expression due to hypoxia (Figure 5I) and consequently reversed the hypoxia-induced increase of MM1s chemotaxis to SDF1α (Figure 5J).
Figure 5.
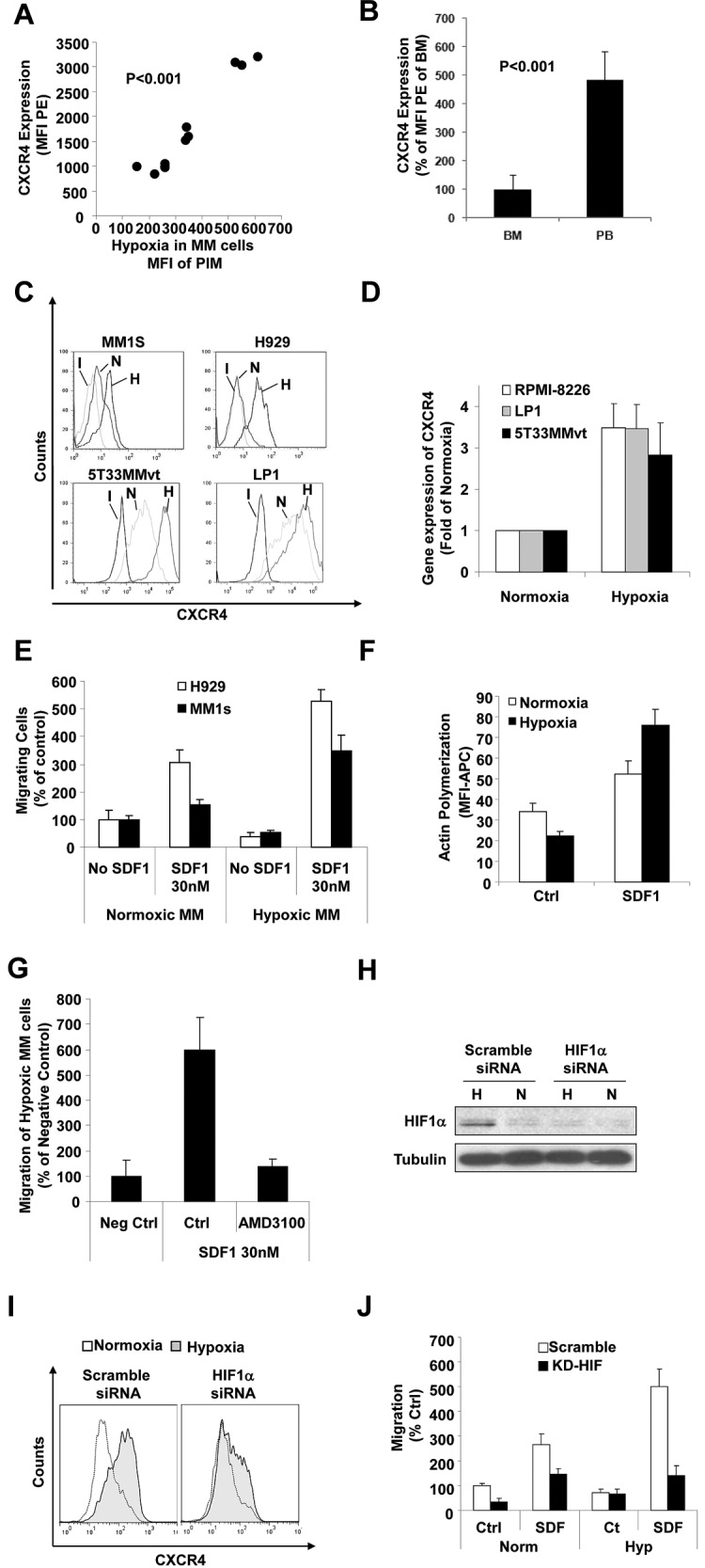
Effect of hypoxia on the expression of CXCR4 and chemotaxis of MM cells. (A) MM cells isolated from the BM of mice injected with MM1s cells at different tumor burden were analyzed for the expression of CXCR4 by flow cytometry and compared with the hypoxic state in these cells as shown by the MFI of PIM in the MM cells. This shows that the expression of CXCR4 increased significantly with the increase in hypoxia in MM cells. (B) Expression of CXCR4 in MM cells isolated from the PB and BM of mice injected with MM1s cells, which shows a higher expression of CXCR4 in circulating MM cells compared with MM cells residing in the BM. Shown is the effect of incubation of MM cells (MM1s and H929) under hypoxic conditions for 24 hours in vitro on the expression of CXCR4 in MM cells (MM1s, H929, U266, and 5T33MMvt) detected at the protein level by flow cytometry (C) or detected mRNA level by quantitative RT-PCR (D). This shows that hypoxia increased the expression of CXCR4 in MM cells. (E) Effect of incubation of MM cells (MM1s and H929) under hypoxic conditions for 24 hours in vitro on chemotaxis toward SDF1α and (F) actin polymerization showing a significant increase of chemotaxis and actin polymerization in hypoxic MM cells. (G) Effect of the CXCR4 inhibitor AMD1300 on chemotaxis of hypoxic MM cells. It can be seen that chemotaxis was abolished. (H) Knockdown of HIF1α in MM1s cells showing decreased expression of HIF1α under hypoxic conditions in cells transfected with HIF1α siRNA. (I) Effect of knockdown of HIF1α on the expression of CXCR4 induced by hypoxia. This shows that knockdown of HIF1α decreased the expression of CXCR4 induced by hypoxia. (J) The effect of knockdown of HIF1α on the increased chemotaxis induced by hypoxia. This shows that knockdown of HIF1α reversed the increase of chemotaxis in response to hypoxia.
Hypoxia enhances MM cell homing to the BM in vivo
The determine the effect of the enhanced chemotactic properties induced by hypoxia on the homing of MM cells to the BM, a critical part of the dissemination process, we incubated MM cells under hypoxic or normoxic conditions for 24 hours and then cells were IV injected into mice. We followed the time needed for the cells to extravasate using live in vivo flow cytometry. Figure 6A shows that whereas 90% of the normoxic MM cells needed 25 minutes to extravasate (ie, to home to the BM), 90% of the hypoxic cells homed within the first 10 minutes after injection. To confirm that the accelerated homing was hypoxia dependent, we knocked down HIF1α and tested the extravasation of these cells after incubation under hypoxic and normoxic conditions. Figure 6B shows that knocking down HIF1α resulted in similar behavior of normoxic and hypoxic MM. To further confirm that this effect was driven through CXCR4, MM cells were pretreated with AMD3100 before injection. AMD3100 reversed the homing of the normoxic and hypoxic MM cells, whereas the effect on prevention of hypoxic cells was more than that of normoxic cells, with 60% of the cells still present in the circulation at 25 minutes after injection (Figure 6C). These results were confirmed by visualization of the homing of MM cells to the BM using live in vivo confocal microscopy. MM cells were incubated under hypoxic or normoxic conditions for 24 hours, injected into mice, and the homing of MM cells in the BM of the mouse skull was imaged 5-7 minutes after the injection using live in vivo confocal microscopy. We found that hypoxia accelerated homing of MM to the BM. A higher number of hypoxic cells homed to the BM, shown as the mean of number of MM cells in each frame in Figure 6D and in the representative images of BM niches compared with normoxic cells in Figure 6E.
Figure 6.
Effect of hypoxia on MM cell homing to the BM in vivo. (A) The effect of incubation of MM1s cells under hypoxic conditions for 24 hours in vitro on homing to the BM of mice after IV injection detected by in vivo flow cytometry. This showed that hypoxia accelerated the homing of MM cells to the BM. (B) Results of homing of MM1s cells with HIF1α knockdown incubated in hypoxic or normoxic conditions for 24 hours before injection into the tail veins of mice. Hypoxia did not accelerate homing of MM cells with knockdown of HIF1α. (C) Pretreatment of hypoxic and normoxic cells with the CXCR4 inhibitor reversed their homing. MM cells were incubated under hypoxic or normoxic conditions for 24 hours and injected into mice. (D) Homing of MM cells to the BM of mouse skulls was imaged 5-7 minutes after the injection using live in vivo confocal microscopy. This shows that hypoxia increased the number of MM cells that homed to the BM, shown as the mean number of MM cells in each frame or as representative images showing the increase in the number of hypoxic cells in BM niches compared with normoxic cells (E). Green indicates MM cells; red, blood vessels.
Recovery of hypoxic MM cells in the normoxic environment
We have tested the expression of CXCR4 in hypoxic MM cells after exposure to normoxic levels of oxygen, and found that the increased expression of CXCR4 in hypoxic cells was retained after exposure to 1 hour of normal levels of oxygen (Figure 7A). However, exposure to conditioned medium from normoxic stroma decreased the expression of CXCR4 (Figure 7B), increased the adhesion properties of the MM cells to stroma (Figure 7C), and increased the expression of E-cadherin (Figure 7D).
Figure 7.
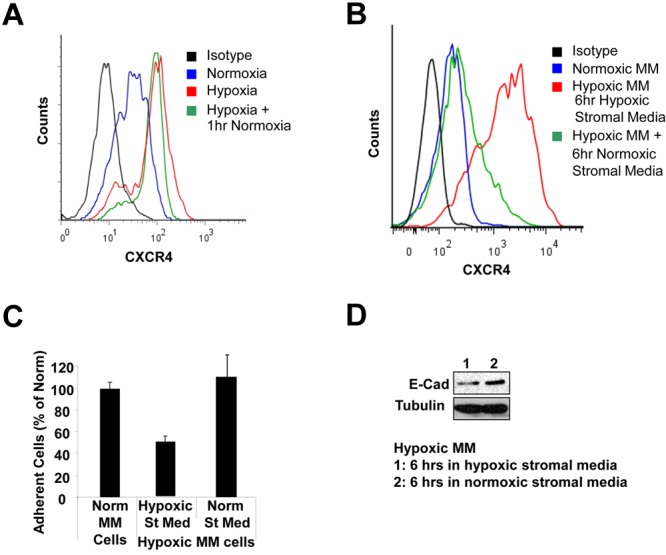
Recovery of hypoxic MM cells in a normoxic environment. (A) Hypoxic MM cells retained a high level of CXCR4 expression after exposure to 1 hour of normoxic levels of oxygen. (B) The decreased expression of CXCR4 in hypoxic MM cells after incubation for 6 hours in medium from normoxic stroma. (C) Increased adhesion properties of hypoxic MM cells after incubation for 6 hours in medium from normoxic stroma. (D) Increased E-cadherin expression in hypoxic MM cells after incubation for 6 hours in medium from normoxic stroma.
Discussion
MM is characterized by widespread dissemination of the BM at diagnosis, with multiple focal lesions in the BM, (re)circulation into the PB, and (re)entrance or homing of MM cells into new sites of the BM.17,18 However, the driving force for MM cells to leave the BM, egress, and home to new BM niches is still not well understood. Metastasis is a multistep process responsible for most cancer deaths and is a complex process involving different mechanisms and factors. Hypoxia (low oxygen) has been associated with metastasis in solid tumors and with poor patient outcome.32,33 Hypoxia in solid-tumor cancers was shown to promote metastasis through activation of proteins involved in the EMT process.16 In the present study, we hypothesized that MM tumor progression induces hypoxic conditions, which activate EMT-related proteins and promote metastasis of MM cells.
To test this hypothesis, we examined levels of hypoxia in MM cells at different tumor burdens in vivo, and found that tumor progression induced hypoxia in both the MM cells and the tumor microenvironment. In the 2 in vivo animal models used, we found an increased expression of HIF1α that was correlated with tumor progression. Similarly, hypoxia-induced genes were increased in MM patients compared with plasma cells from healthy donors. These results are in agreement with previous studies showing induction of hypoxia due to tumor progression in solid tumors34–36 and in MM.12–14 In agreement with previous findings,6,37 we showed in the present study that the number of circulating cells increased with the progression of MM in mice. The correlation was observed mainly in late stages of the progression. However, when we compared the number of circulating MM cells with the hypoxic state of the MM cells in the BM, we found a direct correlation with a stronger association than tumor burden. It is possible that these cells were stained with PIM while in the hypoxic primary tumor in the BM, and then migrated to the circulation during the 4-hour period after PIM injection. These PIM+ cells could still be detected in the circulation because of the covalent binding of PIM under hypoxic conditions, which was no longer sensitive to changes in oxygen when the cells migrated to the oxygenated environment in the circulation. These data suggest that hypoxia may play a role in cell egress, which is not just a passive passage of tumor cells into the circulation due to increased tumor burden in the BM.
To examine the mechanisms by which hypoxia could induce egress, we tested the effect of hypoxia on adhesion and EMT-regulating proteins. Hypoxia in solid tumors has been shown to promote tumor cell motility and acquisition of EMT features, which has been associated with carcinogenesis, invasion, and metastasis.10,11,16,19–21 A critical EMT event is the down-regulation or silencing of E-cadherin through up-regulation of SNAIL,38 resulting in an impairment of cell-cell adhesion, which allows detachment of cells, favoring tumor invasion.39 In the present study, MM cells isolated from MM patients showed an increased expression of EMT-related proteins, including SNAIL, FOXC3, and TGFb, compared with normal plasmal cells, in parallel with decreased expression of E-cadherin. In addition in our in vivo mouse model, we found down-regulation of E-cadherin expression in correlation with the increase of hypoxia in MM cell and cells in the BM microenvironment. These results are in agreement with recently published studies showing that hypoxia abrogated cell-cell contact between fibroblasts in rheumatoid arthritis due to a decrease in the expression of cadherins.40 In addition, circulating MM cells maintained the hypoxic behavior and had low expression of E-cadherin compared with MM cells in the BM. Mechanistically, incubation of MM cells under hypoxic conditions in vitro induced the expression of the hypoxia-related proteins HIF1α and HIF1β; in addition, it induced EMT-related features including down-regulation of E-cadherin. SNAIL is known to be an important player in the EMT machinery and is known to down-regulate the transcription of E-cadherin during EMT.41 We found that hypoxia increased the expression of SNAIL in MM cells. We confirmed that the effect was through E-cadherin by testing the effect of E-cadherin–blocking Ab on adhesion of MM and stromal cells under hypoxic and normoxic conditions. We found that the E-cadherin Ab decreased the adhesion under normoxic conditions but not under hypoxic conditions. These results are consistent with our data showing that E-cadherin is inhibited in hypoxic conditions, and therefore further inhibition by an E-cadherin Ab was not effective. Furthermore, we found that GSK3β, which serves as an endogenous inhibitor of SNAIL in the control of EMT,42 was inhibited (phosphorylated) by hypoxia. We further tested the effect of hypoxia-induced down-regulation of E-cadherin on adhesion of MM cells to BMSCs, and found that hypoxia decreased adhesion in vitro. Inhibition of GSK3β increased the expression of SNAIL, decreased the expression of E-cadherin, and, consequently, decreased the adhesion of MM cells to stromal cells. These results are in agreement with previous studies showing that inhibition of GSK3β by pathways such as PI3K and Wnt or by an exogenous inhibitor decreased cell adhesion and increased metastasis.42,43
We also investigated the effect of hypoxia on the secretion of SDF1α from stromal cells isolated from 5 different MM patients and found that the secretion of SDF1α was reduced with hypoxia, which consequently decreased the migration of MM cells toward conditioned medium from hypoxic stroma. This may provide an additional explanation for the increased egress of MM cells due to hypoxia.
To complete the metastatic process after egress, MM cells need to home to new sites in the BM. We have shown previously that CXCR4 plays a major role in the migration and homing of MM to the BM.6 Therefore, in the present study, we investigated the effect of hypoxia on expression of CXCR4, chemotaxis, and homing of MM cells to the BM. We found that the expression of CXCR4 in MM cells in vivo was directly correlated with hypoxia in the MM cells in vivo. Hypoxia induced CXCR4 expression and enhanced chemotaxis to SDF1α in vitro and homing in vivo. These effects were abrogated by a CXCR4 inhibitor such as AMD3100, potentially indicating that the process of cell dissemination and “metastasis” in MM can be abrogated by pharmacologic inhibitors such as CXCR4 inhibitors in future clinical trials. We further confirmed that the chemotactic effect was hypoxia dependent by knocking down HIF1α and showing that this reversed the increased expression of CXCR4 and chemotaxis of MM cells in vitro, as well as abrogation of the enhanced homing of hypoxic cells in vivo. These results are in agreement with previous studies showing that hypoxia induces the expression of CXCR4 in solid tumors and increases cell migration.44–46 Similarly, CXCR4 was reported to be up-regulated in EMT. Induction of EMT in squamous cell carcinoma increased the CXCR4 expression and cell migration in response to SDF1α.43,47 Similarly, activation of downstream effectors of CXCR4, including Rho-GTPases, was observed during EMT and promoted cell movement and migration.48–50 We have shown previously that downstream effectors of CXCR4, Rho-GTPases, were up-regulated in MM compared with normal plasma cells and play key roles in the movement, migration, and homing of MM cells to the BM.7
In addition to the findings that circulating MM cells displayed hypoxic MM features, such as decreased expression of E-cadherin and increased expression of CXCR4, we investigated the role of exposure to a normoxic environment on the behavior of hypoxic cells. We have shown previously that cells extravasate in less than 30 minutes in vivo, so we tested the effect of exposure of hypoxic cells to normal oxygen levels on their hypoxic features and found that they retained their high expression of CXCR4. However, when we tested the effect of prolonged exposure to medium from normoxic stroma on the hypoxic features, mimicking the conditions that MM cells will be exposed to once they home to new BM niches, we found that the hypoxic MM cells recovered from the hypoxic behavior and displayed normoxic MM-cell features, including decreased CXCR4 expression, increased adhesion properties, and increased E-cadherin expression.
To mimic the physiologic hypoxia level in the BM, we performed some key experiments at mild hypoxic conditions (6% oxygen) and found that doing so did not induce a hypoxic response in MM cells, did not decrease adhesion of MM cells to stroma, and did not decrease the expression of E-cadherin in MM cells or stromal cells. Hypoxia also did not increase the migration of MM cells to SDF1α or increase the expression of CXCR4 in MM cells (supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article).
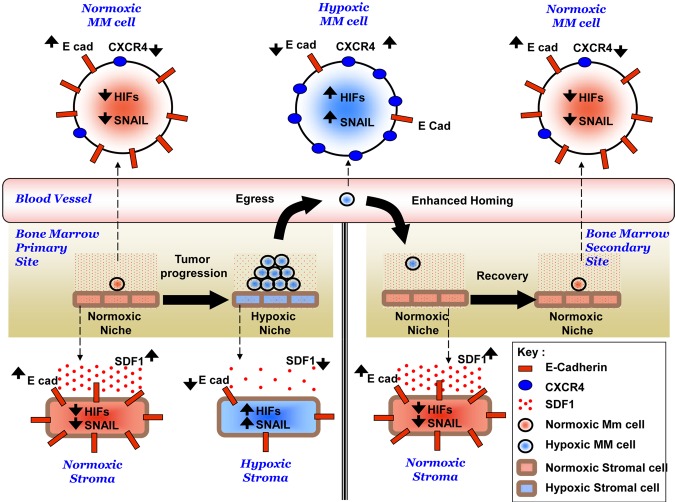
Figure 8 summarizes the hypothesized mechanism for the role of hypoxia and EMT in the dissemination of MM. In the present study, we have demonstrated that tumor progression induces hypoxia in MM cells and other cells in the BM microenvironment. Hypoxia activates EMT-related machinery in MM cells, decreases expression of E-cadherin, decreases adhesion of MM cells to the BM, decreases the secretion of SDF1α from stromal cells, and enhances the egress of MM cells to the circulation. In parallel, hypoxia increases the expression of CXCR4, thus increasing the migration and homing of circulating MM cells to new BM niches, where they recover and display normoxic behavior. Further studies to manipulate hypoxia to regulate tumor dissemination as a therapeutic strategy are warranted.
Figure 8.
Hypothesized mechanism of role of hypoxia in the dissemination of MM. Tumor progression induces hypoxia in the MM cells and other cells in the BM microenvironment. Hypoxia activates EMT-related machinery in MM cells and stromal cells, including activation of HIFs, activation of SNAIL, and decreased expression of E-cadherin, which leads to decreased adhesion of MM cells to the BM, decreased SDF1α secretion from stroma, and enhanced egress of MM cells to the circulation. In parallel, hypoxia increases the expression of CXCR4, and consequently increases the migration and homing of MM cells in to the BM to form metastasis to new BM niches.
Supplementary Material
Acknowledgments
This study was supported in part by the National Institutes of Health (R01CA125690 and R01CA152607), an American Association for Cancer Research–Amgen fellowship, and Fonds voor Wetenschappelijk Onderzoek (National Fund for Scientific Research).
Footnotes
There is an Inside Blood commentary on this article in this issue.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: A.K.A. designed and performed the research, analyzed the data, and wrote the manuscript; J.H. performed the research, analyzed the data, and wrote the manuscript; P.Q., F.A., C.P., R.A., B.T., P.M., J.D.S., C.P.H., and R.D.C. performed the research and analyzed the data; A.M.R., A.S., H.T.N., C.P.L., and A.L.K. analyzed the data; and K.V. and I.M.G. designed the research and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Irene M. Ghobrial, MD, Medical Oncology, Dana-Farber Cancer Institute, 44 Binney St, Mayer 548A, Boston, MA 02115; e-mail: irene_ghobrial@dfci.harvard.edu.
References
- 1.Alsayed Y, Ngo H, Runnels J, et al. Mechanisms of regulation of CXCR4/SDF-1 (CXCL12)-dependent migration and homing in multiple myeloma. Blood. 2007;109(7):2708–2717. doi: 10.1182/blood-2006-07-035857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lapidot T, Dar A, Kollet O. How do stem cells find their way home? Blood. 2005;106(6):1901–1910. doi: 10.1182/blood-2005-04-1417. [DOI] [PubMed] [Google Scholar]
- 3.Damiano JS, Cress AE, Hazlehurst LA, Shtil AA, Dalton WS. Cell adhesion mediated drug resistance (CAM-DR): role of integrins and resistance to apoptosis in human myeloma cell lines. Blood. 1999;93(5):1658–1667. [PMC free article] [PubMed] [Google Scholar]
- 4.Hideshima T, Chauhan D, Hayashi T, et al. The biological sequelae of stromal cell-derived factor-1alpha in multiple myeloma. Mol Cancer Ther. 2002;1(7):539–544. [PubMed] [Google Scholar]
- 5.Pagnucco G, Cardinale G, Gervasi F. Targeting multiple myeloma cells and their bone marrow microenvironment. Ann N Y Acad Sci. 2004;1028:390–399. doi: 10.1196/annals.1322.047. [DOI] [PubMed] [Google Scholar]
- 6.Azab AK, Runnels JM, Pitsillides C, et al. CXCR4 inhibitor AMD3100 disrupts the interaction of multiple myeloma cells with the bone marrow microenvironment and enhances their sensitivity to therapy. Blood. 2009;113(18):4341–4351. doi: 10.1182/blood-2008-10-186668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Azab AK, Azab F, Blotta S, et al. RhoA and Rac1 GTPases play major and differential roles in stromal cell-derived factor-1-induced cell adhesion and chemotaxis in multiple myeloma. Blood. 2009;114(3):619–629. doi: 10.1182/blood-2009-01-199281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Menu E, Asosingh K, Indraccolo S, et al. The involvement of stromal derived factor 1alpha in homing and progression of multiple myeloma in the 5TMM model. Haematologica. 2006;91(5):605–612. [PubMed] [Google Scholar]
- 9.Keith B, Simon MC. Hypoxia-inducible factors, stem cells, and cancer. Cell. 2007;129(3):465–472. doi: 10.1016/j.cell.2007.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rohwer N, Lobitz S, Daskalow K, et al. HIF-1alpha determines the metastatic potential of gastric cancer cells. Br J Cancer. 2009;100(5):772–781. doi: 10.1038/sj.bjc.6604919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang Y, Li M, Yao Q, Chen C. Recent advances in tumor hypoxia: tumor progression, molecular mechanisms, and therapeutic implications. Med Sci Monit. 2007;13(10):RA175–RA180. [PubMed] [Google Scholar]
- 12.Hu J, Handisides DR, Van Valckenborgh E, et al. Targeting the multiple myeloma hypoxic niche with TH-302, a hypoxia-activated prodrug. Blood. 2010;116(9):1524–1527. doi: 10.1182/blood-2010-02-269126. [DOI] [PubMed] [Google Scholar]
- 13.Martin SK, Diamond P, Gronthos S, Peet DJ, Zannettino AC. The emerging role of hypoxia, HIF-1 and HIF-2 in multiple myeloma. Leukemia. 2011;25(10):1533–1542. doi: 10.1038/leu.2011.122. [DOI] [PubMed] [Google Scholar]
- 14.Martin SK, Diamond P, Williams SA, et al. Hypoxia-inducible factor-2 is a novel regulator of aberrant CXCL12 expression in multiple myeloma plasma cells. Haematologica. 2010;95(5):776–784. doi: 10.3324/haematol.2009.015628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cannito S, Novo E, di Bonzo LV, Busletta C, Colombatto S, Parola M. Epithelial-mesenchymal transition: from molecular mechanisms, redox regulation to implications in human health and disease. Antioxid Redox Signal. 2010;12(12):1383–1430. doi: 10.1089/ars.2009.2737. [DOI] [PubMed] [Google Scholar]
- 16.Hung JJ, Yang MH, Hsu HS, Hsu WH, Liu JS, Wu KJ. Prognostic significance of hypoxia-inducible factor-1alpha, TWIST1 and Snail expression in resectable non-small cell lung cancer. Thorax. 2009;64(12):1082–1089. doi: 10.1136/thx.2009.115691. [DOI] [PubMed] [Google Scholar]
- 17.Liang X, Zheng M, Jiang J, Zhu G, Yang J, Tang Y. Hypoxia-inducible factor-1 alpha, in association with TWIST2 and SNIP1, is a critical prognostic factor in patients with tongue squamous cell carcinoma. Oral Oncol. 2011;47(2):92–97. doi: 10.1016/j.oraloncology.2010.11.014. [DOI] [PubMed] [Google Scholar]
- 18.Thiery JP, Acloque H, Huang RY, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139(5):871–890. doi: 10.1016/j.cell.2009.11.007. [DOI] [PubMed] [Google Scholar]
- 19.Iwatsuki M, Mimori K, Yokobori T, et al. Epithelial-mesenchymal transition in cancer development and its clinical significance. Cancer Sci. 2010;101(2):293–299. doi: 10.1111/j.1349-7006.2009.01419.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guarino M, Rubino B, Ballabio G. The role of epithelial-mesenchymal transition in cancer pathology. Pathology. 2007;39(3):305–318. doi: 10.1080/00313020701329914. [DOI] [PubMed] [Google Scholar]
- 21.Guarino M. Epithelial-mesenchymal transition and tumour invasion. Int J Biochem Cell Biol. 2007;39(12):2153–2160. doi: 10.1016/j.biocel.2007.07.011. [DOI] [PubMed] [Google Scholar]
- 22.Leleu X, Jia X, Runnels J, et al. The Akt pathway regulates survival and homing in Waldenstrom macroglobulinemia. Blood. 2007;110(13):4417–4426. doi: 10.1182/blood-2007-05-092098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vanderkerken K, Asosingh K, Croucher P, Van Camp B. Multiple myeloma biology: lessons from the 5TMM models. Immunol Rev. 2003;194:196–206. doi: 10.1034/j.1600-065x.2003.00035.x. [DOI] [PubMed] [Google Scholar]
- 24.Vanderkerken K, De Greef C, Asosingh K, et al. Selective initial in vivo homing pattern of 5T2 multiple myeloma cells in the C57BL/KalwRij mouse. Br J Cancer. 2000;82(4):953–959. doi: 10.1054/bjoc.1999.1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vanderkerken K, De Raeve H, Goes E, et al. Organ involvement and phenotypic adhesion profile of 5T2 and 5T33 myeloma cells in the C57BL/KaLwRij mouse. Br J Cancer. 1997;76(4):451–460. doi: 10.1038/bjc.1997.409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chng WJ, Kumar S, Vanwier S, et al. Molecular dissection of hyperdiploid multiple myeloma by gene expression profiling. Cancer Res. 2007;67(7):2982–2989. doi: 10.1158/0008-5472.CAN-06-4046. [DOI] [PubMed] [Google Scholar]
- 27.Henze AT, Acker T. Feedback regulators of hypoxia-inducible factors and their role in cancer biology. Cell Cycle. 2010;9(14):2749–2763. doi: 10.4161/cc.9.14.12591. [DOI] [PubMed] [Google Scholar]
- 28.Ng PK, Yu RM, Kwong TF, Wong MM, Kong RY. Transcriptional regulation and functional implication of the grass carp CITED1 (gcCITED1) in the negative regulation of HIF-1. Int J Biochem Cell Biol. 2010;42(9):1544–1552. doi: 10.1016/j.biocel.2010.06.007. [DOI] [PubMed] [Google Scholar]
- 29.Horak P, Crawford AR, Vadysirisack DD, et al. Negative feedback control of HIF-1 through REDD1-regulated ROS suppresses tumorigenesis. Proc Natl Acad Sci U S A. 2010;107(10):4675–4680. doi: 10.1073/pnas.0907705107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rohwer N, Welzel M, Daskalow K, et al. Hypoxia-inducible factor 1alpha mediates anoikis resistance via suppression of alpha5 integrin. Cancer Res. 2008;68(24):10113–10120. doi: 10.1158/0008-5472.CAN-08-1839. [DOI] [PubMed] [Google Scholar]
- 31.Whelan KA, Caldwell SA, Shahriari KS, et al. Hypoxia suppression of Bim and Bmf blocks anoikis and luminal clearing during mammary morphogenesis. Mol Biol Cell. 2010;21(22):3829–3837. doi: 10.1091/mbc.E10-04-0353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Erler JT, Bennewith KL, Cox TR, et al. Hypoxia-induced lysyl oxidase is a critical mediator of bone marrow cell recruitment to form the premetastatic niche. Cancer Cell. 2009;15(1):35–44. doi: 10.1016/j.ccr.2008.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Erler JT, Bennewith KL, Nicolau M, et al. Lysyl oxidase is essential for hypoxia-induced metastasis. Nature. 2006;440(7088):1222–1226. doi: 10.1038/nature04695. [DOI] [PubMed] [Google Scholar]
- 34.Coquelle A, Toledo F, Stern S, Bieth A, Debatisse M. A new role for hypoxia in tumor progression: induction of fragile site triggering genomic rearrangements and formation of complex DMs and HSRs. Mol Cell. 1998;2(2):259–265. doi: 10.1016/s1097-2765(00)80137-9. [DOI] [PubMed] [Google Scholar]
- 35.Simiantonaki N, Taxeidis M, Jayasinghe C, Kurzik-Dumke U, Kirkpatrick CJ. Hypoxia-inducible factor 1 alpha expression increases during colorectal carcinogenesis and tumor progression. BMC Cancer. 2008;8:320. doi: 10.1186/1471-2407-8-320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vaupel P. The role of hypoxia-induced factors in tumor progression. Oncologist. 2004;9(Suppl 5):10–17. doi: 10.1634/theoncologist.9-90005-10. [DOI] [PubMed] [Google Scholar]
- 37.Nowakowski GS, Witzig TE, Dingli D, et al. Circulating plasma cells detected by flow cytometry as a predictor of survival in 302 patients with newly diagnosed multiple myeloma. Blood. 2005;106(7):2276–2279. doi: 10.1182/blood-2005-05-1858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wells A, Yates C, Shepard CR. E-cadherin as an indicator of mesenchymal to epithelial reverting transitions during the metastatic seeding of disseminated carcinomas. Clin Exp Metastasis. 2008;25(6):621–628. doi: 10.1007/s10585-008-9167-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schmalhofer O, Brabletz S, Brabletz T. E-cadherin, beta-catenin, and ZEB1 in malignant progression of cancer. Cancer Metastasis Rev. 2009;28(1-2):151–166. doi: 10.1007/s10555-008-9179-y. [DOI] [PubMed] [Google Scholar]
- 40.Nonomura Y, Mizoguchi F, Suzuki A, et al. Hypoxia-induced abrogation of contact-dependent inhibition of rheumatoid arthritis synovial fibroblast proliferation. J Rheumatol. 2009;36(4):698–705. doi: 10.3899/jrheum.080188. [DOI] [PubMed] [Google Scholar]
- 41.de Herreros AG, Peiro S, Nassour M, Savagner P. Snail family regulation and epithelial mesenchymal transitions in breast cancer progression. J Mammary Gland Biol Neoplasia. 2010;15(2):135–147. doi: 10.1007/s10911-010-9179-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhou BP, Deng J, Xia W, et al. Dual regulation of Snail by GSK-3beta-mediated phosphorylation in control of epithelial-mesenchymal transition. Nat Cell Biol. 2004;6(10):931–940. doi: 10.1038/ncb1173. [DOI] [PubMed] [Google Scholar]
- 43.Larue L, Bellacosa A. Epithelial-mesenchymal transition in development and cancer: role of phosphatidylinositol 3′ kinase/AKT pathways. Oncogene. 2005;24(50):7443–7454. doi: 10.1038/sj.onc.1209091. [DOI] [PubMed] [Google Scholar]
- 44.Chaudary N, Hill RP. Increased expression of metastasis-related genes in hypoxic cells sorted from cervical and lymph nodal xenograft tumors. Lab Invest. 2009;89(5):587–596. doi: 10.1038/labinvest.2009.16. [DOI] [PubMed] [Google Scholar]
- 45.Ishikawa T, Nakashiro K, Klosek SK, et al. Hypoxia enhances CXCR4 expression by activating HIF-1 in oral squamous cell carcinoma. Oncol Rep. 2009;21(3):707–712. [PubMed] [Google Scholar]
- 46.Kubo M, Li TS, Kamota T, Ohshima M, Qin SL, Hamano K. Increased expression of CXCR4 and integrin alphaM in hypoxia-preconditioned cells contributes to improved cell retention and angiogenic potency. J Cell Physiol. 2009;220(2):508–514. doi: 10.1002/jcp.21803. [DOI] [PubMed] [Google Scholar]
- 47.Onoue T, Uchida D, Begum NM, Tomizuka Y, Yoshida H, Sato M. Epithelial-mesenchymal transition induced by the stromal cell-derived factor-1/CXCR4 system in oral squamous cell carcinoma cells. Int J Oncol. 2006;29(5):1133–1138. [PubMed] [Google Scholar]
- 48.Gulhati P, Bowen KA, Liu J, et al. mTORC1 and mTORC2 regulate EMT, motility, and metastasis of colorectal cancer via RhoA and Rac1 signaling pathways. Cancer Res. 2011;71(9):3246–3256. doi: 10.1158/0008-5472.CAN-10-4058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Broders-Bondon F, Chesneau A, Romero-Oliva F, Mazabraud A, Mayor R, Thiery JP. Regulation of XSnail2 expression by Rho GTPases. Dev Dyn. 2007;236(9):2555–2566. doi: 10.1002/dvdy.21273. [DOI] [PubMed] [Google Scholar]
- 50.Zhang H, Liu X, Liu Y, Yi B, Yu X. Epithelial-mesenchymal transition of rat peritoneal mesothelial cells via Rhoa/Rock pathway. In Vitro Cell Dev Biol Anim. 2011;47(2):165–172. doi: 10.1007/s11626-010-9369-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.