Abstract
Background
Energy metabolism is emerging as a driving force for cellular events underlying cognitive processing. The hypothalamus integrates metabolic signals with the function of centers related to cognitive processing such as the hippocampus.
Objective/Hypothesis
Hypothalamic activity can influence molecular systems important for processing synaptic plasticity underlying cognition in the hippocampus. The neurotrophin BDNF may act as a mediator for the effects of energy metabolism on synaptic plasticity and cognitive function.
Methods
The hypothalamus of rats confined to a respiratory chamber was electrically stimulated, and energy expenditure (EE) was assessed via indirect calorimetry. MRNA levels for BDNF and molecules related to synaptic plasticity and control of cellular energy metabolism were assessed in the hippocampus.
Results
Electrical stimulation of the rat hypothalamus elevates mRNA levels of hippocampal BDNF. BDNF mRNA levels increased according to the metabolic rate of the animals, and in proportion to the mRNA of molecules involved in control of cellular energy metabolism such as ubiquitous mitochondrial creatine kinase (uMtCK).
Conclusions
Results show a potential mechanism by which cellular energy metabolism impacts the substrates of cognitive processing, and may provide molecular basis for therapeutic treatments based on stimulation of deep brain structures.
Keywords: Energy metabolism, cognition, diabetes, hippocampus, trophic factors
Introduction
One of the most fundamental needs of all living organisms and body organs is to make an efficient use of energy. This is a particularly challenging task for the brain as it consumes a relatively large portion of the amount of energy consumed by the body in spite of its small size. Thus, it is likely that metabolic energy exerts a strong influence on the regulation of critical brain functions, including cognitive abilities. In particular, behaviors intrinsically related to energy metabolism such as diet and exercise are emerging as important promoters of synaptic plasticity and cognitive function1, 2. Accordingly, it is crucial to understand the mechanisms by which metabolic events can impinge on the molecular substrates of cognitive processing. The hypothalamus functions as the master center for the integration of metabolic signals with the function of centers related to cognitive processing such as the hippocampus. In turn, BDNF, originally brought to light for its neurotrophic action, has been later recognized as a mediator for the effects of energy metabolism on synaptic plasticity and cognitive function2. This makes BDNF an excellent candidate to evaluate how energy metabolism influences synaptic plasticity involving the hypothalamus and hippocampus.
We have conducted studies to determine whether hypothalamic stimulation can regulate elements associated with control of energy metabolism and synaptic plasticity in the hippocampus. Based on the particular position of BDNF as an intermediate molecule between energy metabolism, synaptic plasticity, and cognition, these studies have been focused on the potential action of BDNF. The results of these studies have clinical significance based on findings that electrical stimulation of the hypothalamus in humans helps alleviate neurological disorders3. In fact, many patients worldwide receive deep brain stimulation (DBS) to treat movement disorders, and mental illnesses such as depression 4. The limited knowledge about the mechanisms through which DBS exerts its therapeutic effects has restricted major developments. Our results showing that hypothalamic stimulation modulates hippocampal BDNF may shed light on a potential mechanism by which hypothalamic DBS can influence cognitive abilities in humans3.
Material and Methods
Adult Sprague-Dawley male rats (n=34), weighing 295g to 340g were housed individually in standard polyethylene cages in a 12/12 h light/dark cycle at 22-24 °C, with food and water ad libitum. The experiments were performed in accordance with the NIH guidelines for the care and use of laboratory animals, and approved by the University of California Los Angeles Chancellor's Animal Research Committee.
Hypothalamic Stimulation
Animals were confined to an air-tight respiratory chamber in which energy expenditure (EE) was assessed via indirect calorimetry5. Experimental rats were implanted with 40μm bipolar electrodes into the left dorsomedial portion of the ventromedial hypothalamic nucleus (dmVMH; Fig. 1B). In a bipolar electrode, the current flows between the anode and the cathode such that it is focused between the two microtips. Animals were randomly assigned to groups stimulated with a specific frequency: sham control (n = 5); 25 Hz (n = 6); 50 Hz (n = 6); 100 Hz (n = 6); 250 Hz (n = 6); 7 kHz (n = 5). The stimulator remained off in the sham control group. Stimulation consisted of a 30s ON/30s OFF train, and each animal was stimulated at the intensity level (20-30 μA) needed to obtain a change in stereotypic behavior, i.e., pause in ongoing activity.
Figure 1.

(A) The effects of the frequency of hypothalamic stimulation on mRNA levels for molecules associated with synaptic plasticity. BDNF mRNA levels were significantly increased to 161% at 50 HZ compared to sham control group (no stimulation, 100%), *p<0.05, **p<0.01. (B) Coronal section showing the electrode position in the hypothalamus. Brain regions demarcations are “outlined in white” according to Paxinos atlas 26. Third ventricle (3V), dorsomedial part of the ventromedial hypothalamic nucleus (dmVMH).
Determination of Energy Expenditure
Each animal remained inside the chamber for 160 min during the hypothalamic stimulation: 30-min baseline period, 90-min stimulation period, and 40-min resting period. Metabolic data were analyzed in data sets corresponding to “Before” (baseline period) and “After” (stimulation and resting periods). The Energy expenditure (EE) is the total amount of energy used by the animal, which includes both the mechanical and non-mechanical energy expenditure (MEE, nMEE respectively). Energy used for mechanical work is known as mechanical energy expenditure (MEE) while the type of energy expended to support basal cellular activities including thermogenesis, neuronal function, etc. is known as non-mechanical energy expenditure (nMEE). EE was assessed via indirect calorimetry (the set-up was an air-tight chamber with strict control of the air flow; O2 consumption and CO2 production were constantly monitored). By constantly measuring the force exerted by the animal on the chamber floor we were able to continuously monitor the energy that the animal devoted to physical activity (i.e. MEE). Floor of the chamber was mounted on four (one on each corner) tri-axial force transducers, which were used to compute the force exerted by the animal. MEE was continuously computed via four tri-axial force transducers attached to the chamber5. 10-minute averages of both EE and MEE were obtained and the differences of this averages (EE-MEE) were used to compute nMEE5. We separated the EE and MEE data into two parts, “Before (EEB and MEEB)”, and “After (EEA and MEEA)” stimulation. All the data were normalized by “Before” values such that: , and ; where the “x” is one of B, or A respectively referring to “Before”, and “After”; “n” indicates normalized data.
To control for intrinsic variables such as stress, data were normalized (n) by their “Before” values. Energy expenditure was further expressed as a percentage of the sham control group average. O'Brien's and Shapiro-Wilk tests were respectively used to examine homoscedasticity and normality. A natural logarithmic transformation was used to obtain a normal distribution to allow the use of standard statistical methods.
Assessments of mRNAs
After determinations of the energy expenditure, animals were quickly killed by decapitation and their hippocampi were rapidly dissected out, immediately placed on dry ice, and stored at -70 °C for mRNA measurements. Real-time quantitative reverse transcription-polymerase chain reaction was used to measure mRNA levels in the ipsilateral hippocampal region. Levels of mRNAs for BDNF, neurotrophic factor 3 (NT-3), ubiquitous mitochondrial creatine kinase (uMtCK), AMP activated protein kinase (AMPK), and uncoupling protein 2 (UCP2) were measured according to published protocols 2. We used a 7700 sequence detection instrument, and TaqMan EZ RT-PCR core reagents (Applied Biosystems, Branchburg, NJ). The glyceraldyehyde 3 phosphate dehydrogenase (GAPDH) gene was used as a standard control.
Statistical analysis
One-way ANOVA (SPSS 16.0 software) was performed to determine group effect; and a multiple comparisons analysis was performed to identify the effect of specific frequencies vs. the sham control group using post hoc Bonferroni test. A regression analyses was performed between means of stimulated groups to assess associations. Significance was determined at p<0.05, data are presented as mean ± SEM.
Results
Molecular systems related to the action of BDNF in control of energy metabolism and synaptic plasticity underlying cognitive abilities were measured in the hippocampal formation. The value of the stimulation frequency was a key factor for eliciting changes in specific molecular systems (Fig.1 and 2). For levels of BDNF mRNA, one-way ANOVA showed a significant group effect (F(5, 28)=10.60, p<0.001). Group comparisons showed that BDNF mRNA level was increased to 161% in 50 HZ compared to sham control group (p<0.01). BDNF mRNA levels decreased as the stimulation frequency went higher after 50 Hz such as at 100 Hz, 250 Hz and 7K Hz, BDNF mRNA levels were 87%, 73% and 69% respectively (Fig. 1A). One-way ANOVA analysis showed a significant group effect for uMtCK mRNA (F(5, 28)=3.25, p<0.05) and AMPK mRNA (F(5, 28)=2.53, p<0.05). Group comparisons followed post hoc Bonferroni test showed that uMtCK mRNA levels were increased in all stimulation groups but only the change in 50 Hz group was significant (p<0.05, Fig. 2A). Interestingly, effects of stimulation on AMPK mRNA levels were just opposite of the uMtCK mRNA levels. AMPK mRNA levels were decreased with all stimulation frequencies. Specifically, the levels of AMPK mRNA in 25 Hz, 50 Hz, 100 Hz, 250 Hz and 7k Hz were 90%, 86%, 76%, 83% and 88%, respectively, the change in 100 Hz was significant (p<0.05, Fig. 2B). UCP2 mRNA levels had a similar effects as AMPK mRNA levels: one-way ANOVA showed a significant group effect (F(5, 28)=2.803, p<0.05), and a post hoc Bonferroni test demonstrated a decrease in all experimental groups compared to sham control (100%) (Fig. 2C): 25 Hz: 86%, 50 Hz: 76%; 100 Hz: 66%; 250 Hz: 72%; 7k Hz: 75%, and the changes in 100 Hz were significant (p<0.05). Levels of AMPK and UCP2 were reduced with stimulation, which could be a reflection of an increase in local energy availability6, 7. This possibility is in line with the observed up-regulation of uMtCK and the proposed role of uMtCK in ATP production 8. The fact that the cognate member of the neurotrophin family, namely NT-3, was not affected by any stimulation frequency (data not shown) argues in favor for a specific role of BDNF. Furthermore, there was an association between levels of hippocampal BDNF mRNA and those of select molecular systems important for the control of cellular energy metabolism. For example, regression analysis showed that hippocampal BDNF mRNA levels (group mean) was correlated with uMtCK mRNA levels (Fig. 2D, R=0.93, p<0.05), but not with AMPK mRNA or with UCP2 mRNA (data not shown).
Figure 2.

The effects of the frequency of hypothalamic stimulation on mRNA levels for molecules associated with energy metabolism. (A) uMtCK mRNA levels were significantly increased to 276% at 50 HZ as compared to sham control animals (*p<0.05). AMPK mRNA levels (B) and UCP2 mRNA levels (C) were significantly decreased to 67% and 79% at 100HZ as compared to sham control animals (*p<0.05). (D) Regression analysis showed an association between uMtCK and BDNF mRNA levels across the several frequencies (R=0.92, *p<0.05). Individual points correspond to group mean values. 5-6 animals per group used to determine the effects of the stimulation frequency. SEMs values are displayed for uMtCK mRNA (vertical line) and BDNF mRNA (horizontal line).
Results showed that unilateral low-level electrical stimulation of the dmVMH elicits a frequency-dependent elevation in the overall metabolic activity (Fig. 3A). According to our statistic analysis, nMEE (F(5, 28)=3.99, p<0.01) but not MEE (F(5, 28)=0.84, p>0.05) was affected by the stimulation frequency suggesting that the increase in overall metabolism was not due to spontaneous physical activity. In addition, both nMME (Fig. 3A) and BDNF (Fig.1A) showed significant increases at 50 Hz, which seems to harmonize with other studies showing the physiological significance of 50 Hz stimulation (see below). Our findings also indicate that hypothalamic stimulation upregulates hippocampal BDNF in association with nMEE in a frequency-dependent manner (R=0.89, p<0.05, Fig. 3B). In addition, uMtCK mRNA levels were linearly correlated with levels of nMEE (R=0.94, p<0.05, Fig. 3C). Group means were used for regression analysis.
Figure 3.
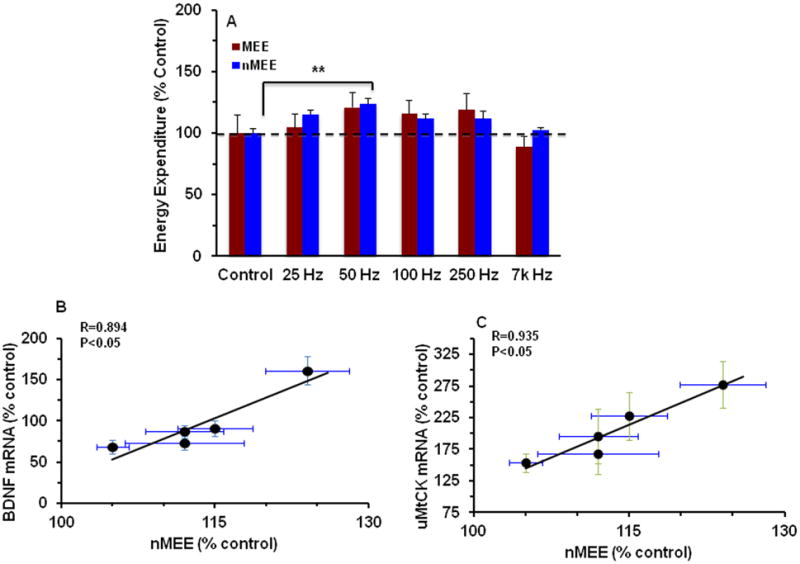
(A) The effects of hypothalamic stimulation on energy expenditure values. nMEE showed a significant increase at 50 HZ stimulation (**p<0.01). MEE: Mechanical Energy Expenditure, nMEE: non-Mechanical Energy Expenditure. (B-C) Regression analysis was carried out for nMEE Vs BDNF mRNA (R=0.94, p<0.05) (B) and nMEE Vs uMtCK mRNA (R=0.86, p<0.05) (C). SEMs values are displayed for BDNF mRNA (vertical line in B), uMtCK mRNA (vertical line in C), nMEE (horizontal line in B and C).
Discussion
Our results show that hypothalamic stimulation influences the hippocampus by affecting expression of molecules important for the control of cellular energy metabolism and synaptic plasticity. Hypothalamic stimulation elevated levels of hippocampal BDNF mRNA, in conjunction with the metabolic rate of the animals. These results are in agreement with the recently described actions of BDNF in metabolism and synaptic plasticity2. In addition, a growing body of evidence emphasizes the potential of energy metabolism to modulate synaptic plasticity underlying cognitive function9. Accordingly, these results seem to illustrate a point of confluence between metabolism and cognition. The hypothalamus plays a fundamental role in the control of overall metabolic rate10, and hippocampal BDNF has a well-established function in synaptic plasticity underlying cognition11. Therefore, the overall results are important to understand how the hypothalamic-hippocampal interaction can serve to modulate cognitive processing.
Energy expenditure Vs Brain Plasticity
According to our results the stimulation frequency is a key factor for the regulation of hippocampal BDNF and uMtCk, in conjunction with nMEE. uMtCK is associated with transduction of energy8, and appears to work in concert with BDNF to modulate hippocampal learning and memory2. Authors in several studies have suggested that specific neuronal elements respond preferentially to select frequencies. For example, while cell bodies and axons are able to respond and follow a 50 Hz train of stimulating pulses, mostly axons, and not cells can respond and follow a 100 Hz train of stimulating pulse.12, 13,14. In addition, stimulation at frequencies in the 50 Hz range has been shown to stimulate vesicle release in hypothalamic presynaptic terminals15. However, frequencies around 100 Hz range have shown to have the most consistent effects on behavior in animal and human studies. For example, Shirvalkar et al. showed that stimulation of the thalamus at 100 Hz facilitates untrained goal-directed rat behavior involving memory recognition16. In turn, Hamani et al. showed memory enhancement in humans using 130 Hz DBS for the first six months and using 50 Hz DBS for the following six months3.
Results also showed that mRNA BDNF levels changed in proportion to levels of nMEE, which are in harmony with the respective functions of nMEE and BDNF. nMEE is likely required for supporting basal physiological functions involved in the conscious state. Indeed, recent positron emission studies in humans have shown that high levels of energy are necessary for maintaining the conscious state17. Therefore, our results showing the influences of metabolic energy on BDNF regulation agree with described roles of BDNF on neuronal plasticity and cognitive function.
Involvement of BDNF in Energy metabolism
The interdependency between BDNF and energy metabolism has recently justified BDNF's designation of “metabotrophin”. These actions of BDNF can be executed in the regulation of synaptic plasticity and cognitive function by engaging molecular systems important for ATP synthesis such as uMtcK and AMPK2, 18. The current results indicate that uMtCK mRNA levels were upregulated in conjunction with BDNF mRNA, which may be agreement with a potential role of uMtCK in neuronal ATP production 8. Along this line of thought, levels of AMPK and UCP2 in our results were reduced with the hypothalamic stimulation, which could be a reflection of an increase in local energy availability. Indeed, it has been shown that AMPK can act as a gauge to detect reductions in levels of cellular energy6, 7.
Physiological implications for the involvement of BDNF on metabolism
The physiological functions of BDNF in metabolism can be observed in the control of appetite, obesity, glucose metabolism, and insulin sensitivity19. It is becoming apparent that the function of the hypothalamus is within the range of functions described for BDNF. For example, it has been found that damage to the hypothalamic VMH results in a glucose-intolerance diabetic-like response20. The overall information seems to emphasize the involvement of the functions of the hypothalamus and BDNF in metabolic homeostasis, and that a failure may result in metabolic disorders such as diabetes. There are an increasing number of reports involving metabolic disorders with cognitive disabilities. For example, obesity and type II diabetes are considered risk factors for the development of cognitive disabilities and mood disorders21, 22, up to the point to increase the risk for neurocognitive disorders such as Alzheimer's disease. Therefore, our results relating hypothalamic stimulation and hippocampal BDNF may shed light on understanding of the molecular basis for the impact of metabolism on cognitive processing.
Based on the demonstrated roles of BDNF supporting cognition11 and reducing depression23, hippocampal levels of mRNA BDNF production by hypothalamic stimulation may have therapeutic implications for the treatment of cognitive disabilities. Elevated hippocampal neuronal activity driven by hypothalamic DBS has been suggested to be responsible for the cognitive enhancement observed in human studies3. Our results appear to provide mechanistic support for these findings as they show that dmVMH stimulation can increase hippocampal BDNF in a frequency-dependent manner. In line with our findings, it has recently been reported that transcranial direct-current stimulation results in a BDNF-dependent augmentation of synaptic plasticity and improvement in motor-skill learning 24.
Conclusions and implications
Energy homeostasis is crucial for maintaining cognitive function such that dysfunctional energy homeostasis, such as in diabetes and obesity, can compromise cognitive abilities25. Our results showing that the metabolic rate of the animals and hippocampal BDNF mRNA levels change as a function of hypothalamic stimulation reveal a potential mechanism for how energy metabolism influences brain function and plasticity. These results can also shed light on potential applications of hypothalamic DBS for the control of metabolic disorders that impose a risk for cognitive function such as diabetes and obesity. The relationship between metabolism and brain function is poorly understood, and further research in this area can provide clues to solve several neurological disorders.
Acknowledgments
This work was supported by National Institutes of Health Grants NS50465 and NS56413, and the UCLA Brain Injury Research Center
Abbreviations
- MEE
mechanical energy expenditure
- nmEE
non-mechanical energy expenditure
- DBS
deep brain stimulation
- VMH
ventromedial hypothalamic nucleus
- uMtCK
ubiquitous mitochondrial creatine kinase
- AMPK
AMP activated protein kinase
- UCP2
uncoupling protein 2
Footnotes
Disclosure/Conflict of Interest: The authors declare no conflict of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Gomez-Pinilla F, Ying Z. Differential effects of exercise and dietary docosahexaenoic acid on molecular systems associated with control of allostasis in the hypothalamus and hippocampus. Neuroscience. 2010;168:130–137. doi: 10.1016/j.neuroscience.2010.02.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gomez-Pinilla F, Vaynman S, Ying Z. Brain-derived neurotrophic factor functions as a metabotrophin to mediate the effects of exercise on cognition. Eur J Neurosci. 2008;28:2278–2287. doi: 10.1111/j.1460-9568.2008.06524.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hamani C, et al. Memory enhancement induced by hypothalamic/fornix deep brain stimulation. Ann Neurol. 2008;63:119–123. doi: 10.1002/ana.21295. [DOI] [PubMed] [Google Scholar]
- 4.Larson PS. Deep brain stimulation for psychiatric disorders. Neurotherapeutics. 2008;5:50–58. doi: 10.1016/j.nurt.2007.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Covalin A, Feshali A, Judy J. 2nd Int IEEE EMBS Conference on Neural Engineering. IEEE EMBS; Arlington, VA: 2005. Deep Brain Stimulation for Obesity Control: Analyzing Stimulation Parameters to Modulate Energy Expenditure; pp. v–viii. [Google Scholar]
- 6.Hardie DG, Carling D. The AMP-activated protein kinase--fuel gauge of the mammalian cell? Eur J Biochem. 1997;246:259–273. doi: 10.1111/j.1432-1033.1997.00259.x. [DOI] [PubMed] [Google Scholar]
- 7.Kim-Han JS, Dugan LL. Mitochondrial uncoupling proteins in the central nervous system. Antioxid Redox Signal. 2005;7:1173–1181. doi: 10.1089/ars.2005.7.1173. [DOI] [PubMed] [Google Scholar]
- 8.Boero J, et al. Restricted neuronal expression of ubiquitous mitochondrial creatine kinase: changing patterns in development and with increased activity. Mol Cell Biochem. 2003;244:69–76. [PubMed] [Google Scholar]
- 9.Gomez-Pinilla F. Brain foods: the effects of nutrients on brain function. Nat Rev Neurosci. 2008;9:568–578. doi: 10.1038/nrn2421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sadava D, O Gordon H, Heller H Craig, Hillis David M, Purves William K. Life: The Science of Biology. W.H. Freeman; 2006. [Google Scholar]
- 11.Vaynman S, Ying Z, Gomez-Pinilla F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur J Neurosci. 2004;20:2580–2590. doi: 10.1111/j.1460-9568.2004.03720.x. [DOI] [PubMed] [Google Scholar]
- 12.Garcia L, D'Alessandro G, Bioulac B, Hammond C. High-frequency stimulation in Parkinson's disease: more or less? Trends Neurosci. 2005;28:209–216. doi: 10.1016/j.tins.2005.02.005. [DOI] [PubMed] [Google Scholar]
- 13.McIntyre CC, Savasta M, Walter BL, Vitek JL. How does deep brain stimulation work? Present understanding and future questions. J Clin Neurophysiol. 2004;21:40–50. doi: 10.1097/00004691-200401000-00006. [DOI] [PubMed] [Google Scholar]
- 14.McIntyre CC, Grill WM. Extracellular stimulation of central neurons: influence of stimulus waveform and frequency on neuronal output. J Neurophysiol. 2002;88:1592–1604. doi: 10.1152/jn.2002.88.4.1592. [DOI] [PubMed] [Google Scholar]
- 15.Hanse E, Gustafsson B. Quantal variability at glutamatergic synapses in area CA1 of the rat neonatal hippocampus. J Physiol. 2001;531:467–480. doi: 10.1111/j.1469-7793.2001.0467i.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shirvalkar P, Seth M, Schiff ND, Herrera DG. Cognitive enhancement with central thalamic electrical stimulation. Proc Natl Acad Sci U S A. 2006;103:17007–17012. doi: 10.1073/pnas.0604811103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shulman RG, Hyder F, Rothman DL. Baseline brain energy supports the state of consciousness. Proc Natl Acad Sci U S A. 2009;106:11096–11101. doi: 10.1073/pnas.0903941106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vaynman S, Ying Z, Wu A, Gomez-Pinilla F. Coupling energy metabolism with a mechanism to support brain-derived neurotrophic factor-mediated synaptic plasticity. Neuroscience. 2006;139:1221–1234. doi: 10.1016/j.neuroscience.2006.01.062. [DOI] [PubMed] [Google Scholar]
- 19.Tsuchida A, et al. Brain-derived neurotrophic factor ameliorates lipid metabolism in diabetic mice. Diabetes Obes Metab. 2002;4:262–269. doi: 10.1046/j.1463-1326.2002.00206.x. [DOI] [PubMed] [Google Scholar]
- 20.Valensi P, et al. Cardiovascular vagosympathetic activity in rats with ventromedial hypothalamic obesity. Obes Res. 2003;11:54–64. doi: 10.1038/oby.2003.10. [DOI] [PubMed] [Google Scholar]
- 21.Kanoski SE, Davidson TL. Western diet consumption and cognitive impairment: links to hippocampal dysfunction and obesity. Physiol Behav. 2011;103:59–68. doi: 10.1016/j.physbeh.2010.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Soczynska JK, et al. Mood disorders and obesity: understanding inflammation as a pathophysiological nexus. Neuromolecular Med. 2011;13:93–116. doi: 10.1007/s12017-010-8140-8. [DOI] [PubMed] [Google Scholar]
- 23.Taliaz D, et al. Resilience to chronic stress is mediated by hippocampal brain-derived neurotrophic factor. J Neurosci. 2011;31:4475–4483. doi: 10.1523/JNEUROSCI.5725-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fritsch B, et al. Direct current stimulation promotes BDNF-dependent synaptic plasticity: potential implications for motor learning. Neuron. 2010;66:198–204. doi: 10.1016/j.neuron.2010.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kapogiannis D, Mattson MP. Disrupted energy metabolism and neuronal circuit dysfunction in cognitive impairment and Alzheimer's disease. Lancet Neurol. 2011;10:187–198. doi: 10.1016/S1474-4422(10)70277-5. [DOI] [PMC free article] [PubMed] [Google Scholar]


