Abstract
Ticks are mostly obligatory blood feeding ectoparasites that have an impact on human and animal health. In addition to direct damage due to feeding, some tick species serve as the vectors for the causative agents of several diseases, such as the spirochetes of the genus Borrelia causing Lyme disease, the virus of tick-borne encephalitis, various Rickettsial pathogens or even protozoan parasites like Babesia spp. Hard ticks are unique among bloodfeeders because of their prolonged feeding period that may last up to two weeks. During such a long period of blood uptake, the host develops a wide range of mechanisms to prevent blood loss. The arthropod ectoparasite, in turn, secretes saliva in the sites of bite that assists blood feeding. Indeed, tick saliva represents a rich source of proteins with potent pharmacologic action that target different mechanisms of coagulation, platelet aggregation and vasoconstriction. Tick adaptation to their vertebrate hosts led to the inclusion of a powerful protein armamentarium in their salivary secretion that has been investigated by high throughput methods. The resulting knowledge can be exploited for the isolation of novel antihemostatic agents. Here we review the tick salivary antihemostatics and their characterized functions at the molecular and cellular levels.
Keywords: tick, salivary gland, hemostasis, coagulation, platelet aggregation, serine proteases, thrombin, kunitz-domain protein, disintegrin, serpin
1. Introduction
Ticks are arthropods of ancient origin, tracing their existence to more than 65 million years ago. Continuous adaptation to their hosts led to the development of unique tick physiological mechanisms to assist blood feeding which include a wide range of molecular effectors that interfere with host defense. Indeed, ticks need to overcome these defensive mechanisms to stay attached in the host for the entire feeding period and to obtain a blood meal successfully. The defenses raised against tick feeding comprise of: i) hemostasis, aimed to heal the bite injury and to prevent blood loss, ii) innate immunity that consists of the inflammatory response and the complement cascade that have an antimicrobial effect and help to remodel damaged tissue, and finally, iii) because of the long duration of attachment to the host and because of the repeated exposure of the same animal to ticks, antigen-specific acquired immunity is raised against a feeding tick, as well. Accordingly, tick saliva contains hundreds of proteins and low molecular weight effectors that target specifically distinct host defensive mechanisms. This is important, because host defenses are often redundant, and ticks must be able to simultaneously interfere with (or inhibit) the measures that are taken by the host to reject the parasite. Thus, it is not surprising that the rich protein armamentarium of tick saliva offers a great opportunity to discover effectors that specifically target various pathways of mammalian physiology. Due to their high target specificity, the pharmacologic action of these effectors is not expected to be accompanied by many side effects, which is not the case for many classical drugs. Medicine is becoming increasingly sophisticated, as our understanding of human physiology and cell biology grows by dissecting the various vertebrate homeostatic mechanisms at the molecular level. The allopathic treatment of diseases needs to be more precise and aimed at specific molecular targets, because many general drugs may cause unwanted side effects due to their wide spectrum of targets in the human body. The same is true for treating hemostatic disorders or for sepsis and traumatology, where there is a demand for more specific antihemostatics. Herein we present the progress in the identification of tick salivary antihemostatic compounds.
2. Vertebrate hemostasis
Among the host's first line of defense against a tick bite is hemostasis. It provides an immediate response to the vascular injury caused by the intrusion of the tick mouthparts in host vessels, thus preventing the extensive loss of host blood. Hemostasis comprises of three conceptually but not physiologically distinct mechanisms: i) coagulation that leads to the production of fibrin clots, ii) thrombus formation and wound healing mediated by platelet aggregation, and iii) vasoconstriction that leads to restricted influx of blood to the injured site. Each of these three mechanisms is activated by several pathways, in response to different exogenous and endogenous stimuli.
The main task of the coagulation cascade is to produce fibrin that supports platelets in a thrombus formation. Thrombus is formed by the cooperation of coagulation and platelet aggregation, as platelets are held together by binding fibrin with their αIIbβ3 integrin; however there is an additional tight connection between coagulation and inflammatory response, as some products of the coagulation cascade, such as thrombin or C3a and C5a complement products, play a role as immune cell activators and inflammation mediators. The coagulation process consists of an enzymatic cascade with two ways of activation, the exogenous and the endogenous, where several amplification points and regulatory mechanisms are known. The exogenous or extrinsic pathway of coagulation begins with the injury of a blood vessel and the formation of the complex between extravascular tissue factor (TF) and factor VIIa (FVIIa/ ‘a’ stands for activated). TF/FVIIa activates factor IX (FIX), and the resulting activated FIX (FIXa) in turn activates factor X (FX) to FXa (Figure 2). FXa activates thrombin that cleaves fibrinogen into fibrin, the primary component of the clot and a binding substrate for platelets; however only a small amount of thrombin is generated with this mechanism, and rather than fibrinogen cleavage, the main function of thrombin during this initiation phase is to activate factor V (FV) and factor VIII (FVIII). The activated FVIII (FVIIIa) binds to activated FIX (FIXa) and together they form the so-called intrinsic Xase (reads "tenase") complex that activates FX at least one million times more effectively than FIXa alone and approximately 50 times more effectively than FVIIa/TF complex [1]. FXa in turn creates the prothrombinase complex with FVa on the platelet surface with a 300,000-times-higher thrombin activation rate than FXa alone [1]. This latter activation phase amplifies the coagulation process extensively. The extrinsic pathway is strictly regulated by the dynamics of the vertebrate host enzymes and their physiological vertebrate host inhibitors— which are called tissue factor pathway inhibitor and antithrombin III. The intrinsic pathway, on the other hand, begins with auto-activation of FXII by a contact with polyanionic surfaces, usually of extrinsic origin; therefore, this pathway is regarded as a contact phase. Intrinsic activators of this coagulation pathway were not known for a long time, but it is suggested an involvement of extracellular RNA that originates from damaged cells [2]. FXIIa activates factor XI (FXI), producing FXIa, which in turn activates FIX to FIXa that—together with FVIIIa—activates FX. Another important crosstalk of this pathway is the activation of the kinin-kallikrein system, leading to the production of the pain-inducing factor bradykinin and also to complement system activation.
Figure 2.
Schematic overview of hemostatic mechanisms targeted by tick salivary protein effectors. Tick proteins are shown in red rectangles, sorted by their respective targets in host hemostasis. Roman numerals in the coagulation cascade refer to coagulation enzymes and factors. TXA2, thromboxane A2; TF, tissue factor. The acronyms for the different tick proteins are explained in the corresponding paragraphs in the text.
Platelets play a pivotal role not only in hemostasis (where they help to prevent blood loss after an injury), but there is increasing evidence that they are crucial for vertebrate immunity because they release many inflammatory mediators such as chemokines or biogenic amines [3]. Platelets can be activated by several effectors such as collagen, thrombin, cathepsin G, or ADP released from damaged cells. Platelet activation leads to the shift of fibrinogen receptor (integrin αIIbβ3) from low-affinity to high-affinity state resulting in the formation of a fibrin/platelet thrombus. The third mechanism of hemostatic response to an injury is vasoconstriction which is mediated by several compounds released from activated platelets (serotonin, thromboxane), the endothelium (endothelins), tissue-resident mast cells (leukotrienes) or some compounds which are present in the bloodstream and are activated by enzymes (angiotensin).
3. Antihemostatic and antiinflammatory properties of tick saliva
Because hemostasis is the first line of defense employed by the vertebrate host against the feeding tick, many evasion mechanisms targeting hemostasis were developed by ticks. In addition to hemostasis, a wide range of host homeostatic mechanisms prevent tick feeding, including innate immune responses—such as acute inflammation, complement cascade activation—or antigen-specific acquired immunity. The detailed and extensive analysis of the molecular mechanisms employed by ticks to evade vertebrate immunity is beyond the scope of this review, although vertebrate immunity and hemostasis crosstalk and they are clearly interconnected, mainly by the actions of serine proteases [4].
Unlike short term feeding ticks from the family Argasidae, ixodid ticks with up to two weeks of feeding duration are exposed to all defensive mechanisms of the host, including antigen specific acquired immunity. As a result, they are under more complex evolutionary pressure to develop efficient countermeasures to be able to complete their blood meal. After three decades of research, it is now known that ticks are not simple "crawling syringes" but they actively contribute to changes in the sites of their attachment, which are necessary for feeding completion. Many effects of tick saliva were discovered in the 1980s by the work of Ribeiro and his colleagues. More specifically, the saliva of the American tick Ixodes dammini (former name of Ixodes scapularis), similarly to that of mosquitoes or tsetse flies, inhibited platelet aggregation induced by ATP, ADP, and platelet aggregation factor (PAF). This activity can be attributed to an enzyme called apyrase that degrades active ATP and ADP into non-active AMP [5]. In addition, antiplatelet activity was associated with tick prostaglandins of E group (e.g., PGE2) and prostacyclins that were discovered in tick saliva a few years later [6]. Connected to tick salivary secretion of PGE2 was the inhibition of IL-2 production by T cells [5]. In 1994, Urioste et al. showed that this T cell activation-blocking mechanism is PGE2-independent and it is mediated by a protein [7]. Collectively these results propose the possibility that both systems (PGE2 and protein dependent) might synergistically contribute to the inhibition of T cell activation. PGs and prostacyclins—which act also as short-term vasodilatators—were found not only in I. dammini, but also in Amblyomma americanum, Boophilus microplus, and Haemaphysalis longicornis [8–9], pointing at the conserved role and importance of PGs in ticks. More information on the synthesis and the role of tick salivary PGs can be found in a review by Bowman and Sauer [10].
The saliva of I. dammini also displayed anti-complement features. More specifically, in the presence of tick saliva, the alternative pathway of complement activation was disrupted, i.e., the deposition of C3b and the release of C3a component. The salivary protein fraction of molecular weight 49 kDa was found responsible for this effect [11]. In addition, rat peritoneal-derived neutrophil aggregation (induced by anaphylotoxin), granule enzyme secretion (induced by fMLP), superoxide secretion (induced by zymosan) and phagocytosis of Borrelia burgdorferi spirochetes were reduced by 40% to 80% in the presence of tick saliva [12]. Coagulation inhibitory activity and anti-complement activity are also present in the saliva of the tick Dermacentor andersoni [13–14].
4. Recent developments in the study of tick salivary secretome
Tick saliva is a mixture of numerous bioactive molecules including a large portion of proteins. In this review, we focus on the protein content of tick saliva. The current work hypothesis predicts more or less a specific function for each tick salivary protein although more than one bioactive protein might mediate the same activity of tick saliva. To date, more than 50 tick salivary proteins have been identified and at least partially characterized at the molecular and biochemical levels. Despite this progress, many salivary proteins still await their characterization. Among the already characterized proteins, there is a large portion of effectors that affect host hemostatic processes. Some of them will be described separately and extensively in the following paragraphs.
Salivary protein identification began in the early 1990s using primarily biochemical methods for the purification and the molecular identification of an activity detected in crude saliva or salivary glands (SGs). The experimental design usually followed a path from discovering a salivary activity in crude salivary gland extracts (SGE) to the identification of the protein(s) that accounts for this activity through a series of chromatographic steps and Edman sequencing or mass spectrometry analysis. It is necessary to mention that there are significant differences in protein composition between saliva and salivary gland extracts; SGE were often used as a substitution for saliva, because it is much easier to obtain SGE than saliva and also because the content of proteins is much higher in SGE. However the problem is that SGE contain large proportion of proteins that are not present in the saliva (e.g. intracellular enzymes). Therefore many activities, observed with SGE might differ from those observed with saliva. Apparently this is the reason that, whenever possible, researchers stopped using SGE and they prioritized the use of saliva in their experimental design.
With the technological developments in the field of molecular biology, a more effective reverse approach (from gene to protein and then to function) was feasible and began to be employed more often for the description of pharmacoactive molecules originating from tick saliva. The methodology includes the identification of coding transcript sequences expressed in tick salivary glands, followed by the production and the characterization of recombinant proteins. This pipeline produced valuable information about the function of tick salivary proteins. More specifically, at the beginning of the 21st century, a high-throughput approach, called sialotranscriptomics (sialo = saliva in Greek), was employed for the massive identification of salivary transcripts and proteins; the end-result was a list of genes from which any researcher could pick a gene of particular interest and focus on its functional characterization [15]. According to this reverse approach, everything starts with cDNA library construction and sequencing of randomly selected clones, which are bioinformatically analyzed and their possible function is predicted in silico. The resulting sequences are automatically compared with millions of transcripts and proteins from several available databases such as the National Centre for Biotechnology Information (NCBI) GenBank, protein families (Pfam) database, Simple Modular Architecture Research Tool (SMART) or Gene Ontology (GO). The results of this bioinformatic analysis are collectively presented in a single table or Excel spreadsheet. The libraries can be subtracted to find differences in gene expression between two different tick developmental stages or different tick-feeding conditions (in the presence or absence of pathogens or for different tick-feeding time points). Furthermore, the development of next generation sequencing led this high-throughput approach to another level; much higher coverage of the sequenced transcripts is now possible at a lower cost as well as better understanding of the transcriptome dynamics. Figure 1 summarizes the workflow of this “gene-to-function” approach in the exploration of tick salivary secretion.
Figure 1.
A simplified workflow description of the “gene-to-function” approach in the discovery of tick salivary secreted proteins. More detailed description is in the text.
The employment of high-throughput approaches brought several surprises in the field. First, it revealed that tick saliva is a much more complex mixture than anticipated; tick SGs produce hundreds of different proteins, many of which are novel and unique among all known organisms in the sense that no similar proteins or genes can be found in public gene databases, such as the non-redundant (NR) database of NCBI. The second notable finding was that the most abundant tick salivary proteins form the so-called multigenic families with sometimes 20 or even 30 members. These proteins might have redundancy in function and this redundancy might, in turn, contribute to the evasion of host defensive mechanisms such as the immune recognition of tick salivary antigens. Indeed, it was shown that there is an alternation in the expression of different members of the same multigenic family as tick feeding progresses [16]. The third surprise—and actually challenge—is that we cannot even predict the possible function of a big proportion of salivary proteins identified by sialotranscriptomics [17].
5. Antihemostatic proteins from ticks
The following sections provide an insight into the functional role of tick salivary proteins as far as it concerns their interference with vertebrate hemostasis. Tick saliva contains many proteins from various protein families that display antihemostatic activities. For presentation purposes we have sorted these proteins by the host hemostatic process they target and their action is abstracted in a simplified schematic overview in Figure 2. Thus far, the largest group comprises of serine protease inhibitors that target thrombin, FXa, and other proteases from the coagulation cascade. Platelet aggregation is inhibited by tick proteins that either bind to platelet integrins or scavenge (bind to) platelet activators (such as ADP, thromboxane, or serotonin) or they inhibit the activation of protease-activated receptors (PARs). The names, main features and species of origin of the tick effectors described in this review, together with the corresponding literature reference, are summarized in Table 1.
Table 1.
Names, main features, and species of origin of tick antihemostatics
| Coagulation inhibitors | |||||
|---|---|---|---|---|---|
| Name | Source | Description | Size | Organism | Ref. |
| Thrombin inhibitors | |||||
| Omithodorin | ? | Kunitz, binding to both exosites and active site | ~14 kDa | Omithodoros moubata | [27] |
| Savignin | SG | Kunitz, binding to both exosites and active site | ~14 kDa | Omithodoros savignyi | [28] |
| Monobin | SG | Kunitz, binding to both exosites and active site | ~14 kDa | Argas monolakensis | [29] |
| Amblin | H | Kunitz, binding to both exosites and active site | ~14 kDa | Amblyomma hebraeum | [31] |
| Boophilin | M | Kunitz, binding to both exosites and active site | ~14 kDa | Rhipicephalus (Boophilus) microplus | [30] |
| Hemalin | M | Kunitz, binding to both exosites and active site | ~14 kDa | Haemaphysalis longicomis | [32] |
| Madanin 1 and 2 | SG | No cysteine, binds to exosite | ~7 kDa | Haemaphysalis longicomis | [34] |
| Chimadanin | SG | No cysteine, inhibition of active site | ~7 kDa | Haemaphysalis longicomis | [35] |
| NTI-1 | WT | Non-competitive inhibition of active site | ~3 kDa | Hyalomma dromedari | [36] |
| Microphilin | S | Exosite binding only | ~1.7 kDa | Rhipicephalus (Boophilus) microplus | [37] |
| Rhipilin | SG | Kunitz, mode of action not determined | ~14 kDa | Rhippicephalus haemaphysaloides | [33] |
| BmAP | S | Inhibition of active site, binding to exosite | ~60 kDa | Rhipicephalus (Boophilus) microplus | [38] |
| BmGTI | M | Binding to exosite only | 26 kDa | Rhipicephalus (Boophilus) microplus | [39] |
| Variegin | SG | C-terminus similar to hirudin binds exosite | 3–4 kDa | Amblyomma variegatum | [40] |
| Americanin | SG | Competitive inhibitor | 12–16 kDa | Amblyoma americanum | [41] |
| IRIS | S | Serpin, inhibits both thrombin and FXa | 41 kDa | Ixodes ricinus | [42] |
| IRS-2 | SG | Serpin, slow binding classical inhibitor | 42 kDa | Ixodes ricinus | [21] |
| Factor Xa inhibitors | |||||
| TAP | WT | Kunitz, binding to both exosites and active site | ~7 kDa | Omithodoros moubata | [43] |
| FXal | SG | Kunitz, binding to both exosites and active site | ~7 kDa | Omithodoras savignyi | [44] |
| Salp 14 | S | BTSP family, inhibits active site | ~14 kDa | Ixodes scapularis | [45] |
| Unnamed | SG | Uncompetitive inhibitors of active site | ~15–17 kDa | Hyalomma truncatum | [46] |
| Unnamed | WT | Uncompetitive inhibitors of active site | ~15–17 kDa | Hyalomma dromedari | [47] |
| FVIIa/TF complex inhibitors | |||||
| Ixolaris | SG | TFPI homologue, Kunitz, inhibits FVIIa/TF complex | ~14 kDa | Ixodes scapularis | [48] |
| Penthalaris | SG | TFPI homologue, Kunitz, inhibits FVIIa/TF complex | ~35 kDa | Ixodes scapularis | [49] |
| Contact phase inhibitors | |||||
| IR-CPI | SG | Kunitz, binds to FXIIa, FXIa and kallikrein | ~7 kDa | Ixodes ricinus | [50] |
| Haemaphysalin | SG | Kunitz, binds to FXIIa | ~7 kDa | Haemaphysalis longicomis | [51] |
| BmTI-A | WT | Kunitz, inhibits kallikrein | ~14 kDa | Rhipicephalus (Boophilus) microplus | [52] |
| Platelet aggregation inhibitors | |||||
| Name | Source | Description | Size | Organism | Ref. |
| ADP hydrolysis | |||||
| Apyrase | SG | Hydrolyses ATP and ADP to inactive AMP | ~62 kDa | Omithodoros savignyi, Rhipicephalus (Boophilus) microplus | [53–54] |
| TXA binding | |||||
| Moubatin | SG | Lipocalin, Binds TXA2 | 17 kDa | Omithodoros moubata | [56] |
| TSGP3 | SG | Binds TXA2 | 16 kDa | Omithodoros savignyi | [57] |
| Longicomin | SG | Most likely binds TXA2 | 16 kDa | Haemaphysalis longicomis | [69] |
| serotonin binding | |||||
| Monotonin | SG | Binds serotonin | 20 kDa | Argas monolakensis | [60] |
| SHBP | SG | Binds serotonin and histamine | 22 kDa | Dermacentor reticularis | [59] |
| Alpha2Beta1 binding | |||||
| TAI | SG | Inhibits adhesion to soluble collagen | 5 kDa | Omithodoros moubata | [68] |
| Thrombin inhibitors | Listed in anticoagulants | ||||
| AlphaIIbBeta3 binding | |||||
| Disagregin | SG | Binds to fibrin receptor on platelets, Kunitz | 6 kDa | Omithodoros moubata | [65] |
| Savignygrin | SG | Binds to fibrin receptor on platelets with RGD motif, Kunitz | 7 kDa | Omithodoros savignyi | [63] |
| Variabilin | SG | Binds to fibrin receptor on platelets with RGD motif | 5 kDa | Dermacentor variabilis | [62] |
| Ixodegrin | SG | Binds to fibrin receptor on platelets with RGD motif | 7 kDa | Ixodes sp. | [66] |
| Savignygrin-like | SG | Binds to fibrin receptor on platelets with RGD motif, Kunitz | 7 kDa | Omithodoros coriaceus | [64] |
| Monogrin | SG | Binds to fibrin receptor on platelets with RGD motif, Kunitz | 10 kDa | Argas monolakensis | [29] |
| Fibrinolytic enzymes | |||||
| Metalloprotease | SG | Degrades fibrinogen and fibrin | 37 kDa | Ixodes scapularis | [55] |
| Cathepsin G inhibitors | |||||
| IRS-2 | SG | Serpin, inhibits cathepsin G and thrombin induced aggregation | 42 kDa | Ixodes ricinus | [21] |
| Vasoactive proteins | |||||
| Histamine function related proteins | |||||
| tHRF | S | Binds to basophils, induces histamine release | ~20 kDa | Ixodes scapularis | [61] |
| HBP | SG | lipocalins, histamine binding, anti-inflammatory | ~20 kDa | Rhipicephalus appendiculatus | [58] |
S - experimentaly found in the saliva, SG -found in salivary glands (putatively secreted), WT - isolated from whole tick, H - isolated from haemolymph, M - isolated from midgut, ? - origin is not stated
5.1 Serine protease inhibitors
Serine protease inhibitors play an important role during tick feeding because many vertebrate physiological processes—including coagulation, platelet aggregation, and complement activation—are driven by proteolysis and proteolytic enzymes. Under physiological conditions, proteolysis regulation is based on the activity and concentration of vertebrate protease inhibitors that interact with their target vertebrate proteases. Disruption of this balance, for example by the presence of an exogenous inhibitor, might lead to the development of pathological conditions. These exogenous inhibitors can be of parasitic origin and ticks apparently secrete salivary protease inhibitors to modulate the proteolytic balance in the sites of their feeding, thus facilitating tick attachment and the subsequent hematophagy.
The family of Kunitz domain-containing proteins is highly represented in the saliva of both soft and hard ticks. Kunitz domain inhibitors belong to the MEROPS inhibitor family I2, clan IB (http://merops.sanger.ac.uk/); their usual targets are serine proteases of the S1 family [18]. The structure of a Kunitz domain typically consists of two β-strands and one α-helix. This structure is stabilized by three disulphide bridges that are created between six conserved cysteines. The molecular weight of one typical Kunitz domain is about 7 kDa; however, many members of this family consist of several Kunitz domains. Salivary transcripts encoding for one, two and five Kunitz domain-containing inhibitors have been found in ticks thus far. The majority of these salivary transcripts are single domain putative inhibitors that vary significantly in their amino acid sequences. Many of these inhibitors possess structural modifications that are the basis for their specific activities [19–20].
The second large group of serine protease inhibitors found in ticks is the serpin superfamily, denoted as I4 family in the MEROPS database. This ubiquitously distributed protein family is interesting primarily because its members are natural regulators of many physiological processes in vertebrates, including hemostasis. For example, α-III antithrombin is a vertebrate serpin that functions as an endogenous inhibitor of thrombin, FIX, and FX, which makes it the major regulatory protein of vertebrate coagulation under physiological conditions. Therefore, it is not so surprising that ticks possess serpins in their salivary armamentarium—and some of them resemble in overall structure to α-III antithrombin [21]—because these proteins might contribute to the modulation of vertebrate hemostasis upon tick feeding. Interestingly, although it is known that serpins are expressed in the SGs of various tick species, most of the serpins were not revealed by the high-throughput sialotranscriptomic projects. Two putative serpins have been described from H. longicornis [22], seventeen from Rhiphicephalus appendiculatus [23], seventeen from Am. americanum [24], and five from Ixodes ricinus [21, 25]. More than 40 serpins were identified in the genome of I. scapularis [26]. Only two of these serpins have been functionally characterized and they are described with some more details in the next paragraphs because of their antihemostatic activities.
The third group of candidate protease inhibitors found in tick SGs comprises mostly of novel proteins or peptides of various size and characteristics, with the majority of them to be characterized as thrombin inhibitors.
We next present all these tick inhibitors according to the vertebrate serine protease they target.
5.1.1 Thrombin inhibitors
Targeting thrombin is a very efficient way to suppress the hemostatic reaction, because of the pivotal role of this enzyme in coagulation. Thrombin is also responsible for platelet activation and for the activation of several other enzymes in the coagulation cascade. Moreover, thrombin is involved in additional vertebrate physiological processes, including complement activation or inflammation. Accordingly, there are several thrombin inhibitors, described from different tick species.
A two Kunitz-domain inhibitor from the saliva of the African soft tick Ornithodoros moubata, called ornithodorin, was described at the structural level and in complex with thrombin [27]. This study showed that ornithodorin does not follow the classical Kunitz inhibitory mechanism, because its mechanism of action differs from that of the bovine pancreatic trypsin inhibitor (BPTI), a thrombin inhibitor used in medicine to inhibit fibrinolysis. Whereas BPTI inserts its reactive site loop (RSL) into the thrombin reactive center, neither of the two ornithodorin RSLs gets in contact with the active center of thrombin. In the case of ornithodorin, the inhibition of thrombin is done by the N-terminus of the protein, while the C-terminus binds to the fibrinogen recognition exosite I of thrombin [27]. A similar mode of action explains the activities of other thrombin inhibitors from related tick species, such as that of savignin from Ornithodoros savignyi [28] and monobin from Argas monolakensis [29]. A related two Kunitz-domain inhibitor from Rhiphicephalus (Boophilus) microplus, named boophilin, is not as specific to thrombin (as the previous three inhibitors) because it also blocks plasmin, kallikrein, and FVIIa [30].
Another two Kunitz-domain inhibitor of thrombin, named amblin, was found in the hemolymph of the tick Ammblyoma hebraeum. In contrast to ornithodorin-like inhibitors, amblin possesses a different N-terminal sequence that makes it unlikely to bind to the thrombin reactive site [31]; however, because amblin was found in the hemolymph and not in the saliva, its relevance as an antihemostatic agent is questionable. The same holds true for hemalin from the midgut of the tick H. longicornis that displays similar activities to those of boophilin and it is important for successful tick feeding, although its role is most likely to inhibit proteases in the midgut and not at the sites of tick feeding [32]. The most recently revealed two Kunitz-domain tick inhibitor of coagulation is rhipilin from the tick Rhipicephalus haemaphysaloides [33] that prolonged both prothrombin time (PT) and activated partial thromboplastin time (aPTT). PT is used as a test for detecting deficiencies in extrinsic coagulation pathways, while aPTT is used for testing the functionality of the intrinsic pathway. The specificity of rhipilin is not determined yet; however, as it inhibits both pathways, rhipilin might be an inhibitor of either FXa or thrombin or both.
Novel thrombin inhibitors, named madanins, were identified in a cDNA library from rapid feeding stages of the tick H. longicornis [34]. Madanin 1 and 2 are homologous proteins with molecular weight about 7 kDa that are classified as the family of protease inhibitors I53 in the MEROPS database. Both proteins prolonged aPTT and PT and inhibited the conversion of fibrinogen to fibrin without impairing thrombin amidolytic activity, which suggests binding of these inhibitors to the exosite I of thrombin and thus a competitive inhibition. Interestingly this family seems to be unique for the tick H. longicornis. Chimadanin (MEROPS family I72), found in the same species, has only slight similarity to proteins from other tick species. Chimadanin prolonged both aPTT and PT but, unlike madanins, it also inhibited thrombin amidolytic activity, suggesting binding into the thrombin active site [35].
Two thrombin inhibitors with anticoagulant activity, named NTI-1 and NTI-2, were isolated by biochemical methods from crude nymphal extracts of the camel tick Hyalomma dromedarii [36]. There is no information on the primary structure of these inhibitors, apart from their molecular masses, which are 3.2 and 14.9 kDa, respectively. The latter mass might imply a two Kunitz-domain inhibitor. Microphilin is a small salivary peptide with a molecular mass of 1.8 kDa isolated from the saliva of the cattle tick R. microplus [37]. Microphilin inhibited the formation of fibrin clot and thrombin-induced platelet aggregation; however, it did not impair thrombin amidolytic activity, indicating that microphilin binds to thrombin exosite I or II. In addition to microphilin, a 60-kDa thrombin inhibitor named BmAP was isolated from the saliva of the same species [38]. BmAP inhibits thrombin by binding to both thrombin active site and exosite. BmGTI is 26kDa big thrombin inhibitor isolated from the gut extract of R. microplus that delays bovine plasma clotting time and inhibits both thrombin induced fibrinogen clotting and thrombin induced platelet aggregation without blocking thrombin active site. Therefore it acts through interfering with one of the thrombin exosites. Moreover BmGTI enhanced activated protein C, an activity never described before among R. microplus molecules [39].
The highly specific and tight thrombin inhibitor named variegin was described in the tick Amblyomma variegatum. Variegin is a peptide with only 32 residues that binds to both thrombin active site and exosite and it was shown to be even more efficient than the rationally designed thrombin inhibitor hirulog that derives from hirudin; hirulog is similar to variegin both functionally and structurally [40]. Americanin is another one reversible, thrombin-specific inhibitor isolated by chromatography from the SG extract of the Lone Star tick, A. americanum. Its molecular mass was estimated to be 12–16 kDa and apart from inhibiting thrombin it did not display any inhibitory activity against any other of the eight tested proteases [41].
Among the identified thrombin inhibitors of tick origin, there are also two serpins, namely I.ricinus immunosuppressant (IRIS) (Prevot et al., 2006) and I. ricinus serpin-2 (IRS-2) [21]. Both serpins originate from the castor bean tick I. ricinus, the main European vector of Lyme disease and tick-borne encephalitis. Both serpins inhibit thrombin although it is not their main enzymatic target. Moreover, IRS-2 inhibited thrombin-induced platelet aggregation [21]. IRIS prolonged the time of fibrinolysis, platelet adhesion, and the contact phase-activated pathway of coagulation; this action cannot be attributed solely to the direct impact of IRIS on thrombin activity, since the protein inhibited additional proteases involved in coagulation, including FXa and tissue plasminogen activator (to the same extent as thrombin). Coagulation inhibition was also achieved in relatively high concentrations of IRIS, where non-specific inhibition of proteases might occur [42].
5.1.2 Factor Xa inhibitors
FXa is the activator of thrombin, and, as such, it plays a central role in the coagulation cascade; however, unlike thrombin, factor Xa is not involved in other host physiological processes apart from coagulation. Although the list of identified tick salivary inhibitors of FXa is shorter than that of thrombin (see Table 1), the first salivary protease inhibitor characterized from ticks was an inhibitor of FXa. More specifically, Tick Anticoagulant Protein (TAP) was the first identified tick salivary protein that initiated further interest in tick salivary biochemistry. It is a ~ 60 amino acid long, 7-kDa peptide purified from the soft tick O. moubata, which belongs to the family of Kunitz inhibitors [43]. TAP is a slow and tight-binding inhibitor that displayed very specific inhibitory activity against FXa while it did not display any activity against other enzymes from the coagulation cascade such as plasmin, kallikrein, or FVII. A related FXa inhibitor that shares 66% identity with TAP was isolated from the tick O. savignyi and displayed very similar inhibitory features [44]. Both inhibitors bind to both the active site and the exosite of FXa.
An interesting multigenic family that comprises of tens of homologs was found mainly in three Ixodes species, and although its members were found in metastriate ticks, it seems that this family is more expanded among prostriate ticks. This family is denoted as basic tail-secreted proteins (BTSP) because its C-terminus consists of mainly lysine residues. Only one member of this family has been functionally characterized thus far. It is an anti-coagulant from the tick I. scapularis named salivary protein 14 (Salp14). Salp14 displayed inhibitory activity against FXa while it did not inhibit other proteases including trypsin, chymotrypsin, thrombin, kallikrein, or FXIIa [45]. Despite the fact that only one member from the BTSP family is functionally characterized to date, there is high probability that Salp14 homologs also possess anticoagulant activity, perhaps with specificities against different serine proteases. Two FXa inhibitors with unidentified primary structure were isolated from the related tick species H. dromedarii and Hyalomma truncatum [46–47]. They have similar molecular masses, about 15–17 kDa, and they inhibit both intrinsic and extrinsic coagulation pathways.
Finally, one I. scapularis transcript encodes for a double Kunitz-domain homolog of tissue factor pathway inhibitor (TFPI) called ixolaris. Ixolaris binds to TF/FVIIa complex and inhibits the initiation of coagulation [48]. Ixolaris acts as a fast- and tight-binding ligand of the heparin-binding exosite of FXa. Moreover, it binds to non-activated FX, thus inhibiting FX activation; however, binding of ixolaris to FXa did not impair—but increased—the amidolytic activity of FXa. This is opposite to what is observed upon the interaction of FXa with TFPI which is the physiological vertebrate regulator of this pathway; TFPI is a slow-binding inhibitor of FXa active site [48]. An ixolaris-like mechanism of coagulation inhibition was observed for a five Kunitz-domain protein called penthalaris that is expressed in the SGs of the same tick species [49]. To conclude, both ixolaris and penthalaris inhibit TF activation which is possibly among the first molecular events in the coagulation cascade after an injury. The mechanism of action of both tick proteins (at least partially) mimics that of a physiological vertebrate inhibitor of TF-mediated coagulation called TFPI. TFPI is a three Kunitz-domain protein that interacts with TF/FVIIa complex with one domain and with FXa using the second Kunitz domain, thus creating a quaternary complex of TF/FVIIa/TFPI/FXa.
5.1.3 Inhibitors of contact phase
To date, four tick-derived inhibitors of the contact phase have been described, and all four belong to the Kunitz family. A single Kunitz-domain inhibitor of FXIIa, FXIa, and kallikrein was identified in the tick I. ricinus and it was named Ir-CPI [50]. Ir-CPI increased aPTT and prevented thrombus formation in rats and mice. Moreover, when mice were injected with Ir-CPI they were also protected against collagen- and epinephrine-induced thromboembolism without increased bleeding. Haemaphysalin, another one single Kunitz-domain inhibitor from the tick H. longicornis, interferes with the reciprocal activation of FXII and prekallikrein. It has been suggested that haemaphysalin recognizes the zinc-induced conformational change of the FXII/prekallikrein-high molecular weight kininogen complex and blocks its cell binding domains [51]. BmTI-A is a two Kunitz-domain inhibitor of kallikrein from the tick R. microplus that prolonged aPTT but not PT and thrombin time (TT). TT is used to detect deficiencies in the direct conversion of fibrinogen to fibrin. Overall these data show that BmTI-A is an inhibitor of the contact-phase coagulation pathway [52]. Interestingly, in addition to kallikrein, BmTI-A also inhibited elastase, suggesting that this protein targets simultaneously another one vertebrate physiological process at the tick feeding site, i.e. inflammation.
5.2 Platelet aggregation inhibitors
Platelet function seems to be detrimental for tick feeding success since there are many tick proteinaceous molecules that affect different aspects of platelet biology. Accordingly, all the tick proteins with antiplatelet function are described in the next paragraphs.
5.2.1 Apyrases and metalloproteases
The most widespread antiplatelet mechanism among blood-feeding arthropods is the degradation of ATP and ADP—two potent platelet aggregation agonists—into inactive AMP. This is achieved by a salivary enzyme called apyrase (adenosine diphosphatase). Tick apyrases belong to the family of 5’-nucleotidases. The first member of the family was identified from the cattle tick R. microplus. This tick apyrase has about 40% sequence identity to a vertebrate 5’ nucleotidase [53]. Several apyrase isoforms were found in the saliva of O. savignyi as 62-kDa proteins belonging to the same family of 5’-nucleotidases [54].
A second group of enzymes that impact platelet aggregation and coagulation contains metalloproteases with fibrinolytic and gelatinase activity. These enzymes were isolated from I. scapularis and they are similar to proteases from snake venoms [55].
5.2.2 Inhibitors of collagen-induced platelet aggregation
A lipocalin from the tick O. moubata named moubatin was shown to inhibit collagen-induced platelet aggregation [56]. Moreover, in higher doses, it also inhibited binding of thromboxane A2 (TXA2) to its receptor on platelets. TXA2 is an agonist of platelet aggregation that binds to the specific TXA2 receptor on platelets upon their activation. Two proteins with similar activities to moubatin—named TSGP2 and 3—were identified in the tick O. savignyi [57].
5.2.3 Biogenic amine-binding proteins
A large group of tick salivary proteins belongs to the lipocalin superfamily. Members of the family inhibit platelet aggregation by binding TXA2, while they also inhibit collagen-induced platelet aggregation. Lipocalins are also responsible for scavenging biogenic amines such as serotonin and histamine. Both amines play a role in hemostasis by modulating platelet aggregation and vasoconstriction. Therefore, biogenic amine scavenging by tick lipocalins clearly favors tick feeding because it modulates platelet and vascular biology; but in addition biogenic amines interfere with vertebrate inflammation/immunity, suggesting a dual role for tick salivary lipocalins. Members of the lipocalin family are found in both soft and hard ticks, and all the related studies thus far point at a conserved and important role for this family among ticks [57–60].
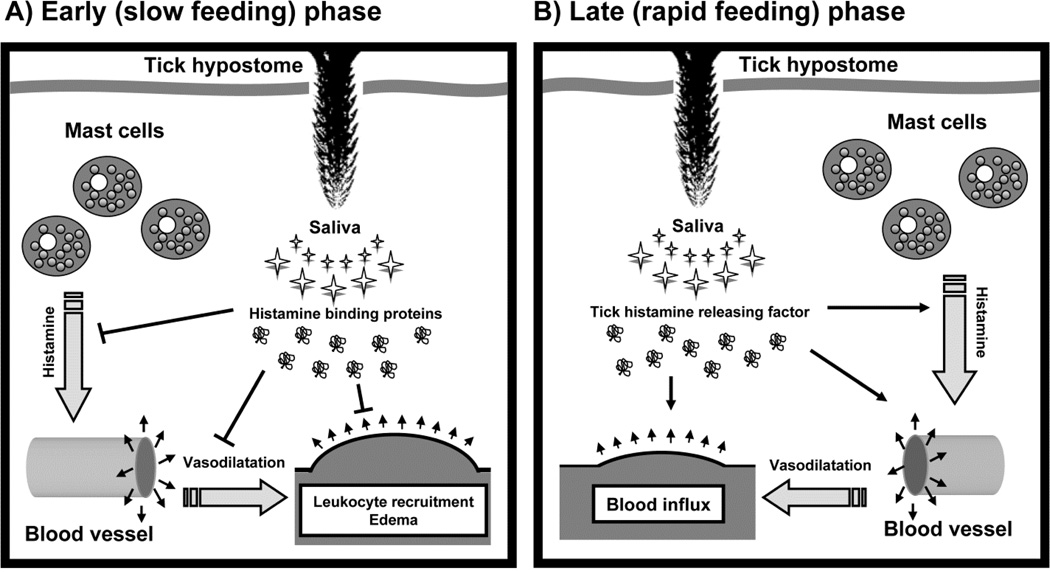
On the other hand, a tick homolog of vertebrate histamine-releasing factor (HRF) was characterized from the tick I. scapularis [61]. It seems contradictory to find various histamine binding and histamine-releasing proteins in tick saliva at the same time, but it most likely reflects the fact that biogenic amines should be regulated in a dynamic way during tick feeding (Figure 3). Upon an inflammatory reaction from the host, biogenic amines are expected to be secreted in high amounts in the tick feeding cavity by the host; biogenic amine scavenging by tick saliva presumably dominates in such an instance. Indeed, most histamine-binding proteins are released during the slow feeding phase of a tick, when establishment of the tick feeding cavity, e.g. by reducing edema that would lead to tick rejection, is the most important issue. HRF is released during the rapid phase of tick feeding, when most blood is imbibed (Figure 3). During this feeding phase, tick sensitivity to histamine is also decreased and there might be already numerous anti-inflammatory substances present in the tick feeding cavity so that the danger of edema formation is low [61]. Histamine is an important vasodilatant; during the late, rapid feeding phase of a tick it is probable that vasodilatation is more crucial for feeding success than the inhibition of platelet aggregation and inflammation.
Figure 3.
Modulation of histamine dynamics in the site of tick bite during the feeding course. In the early, slow feeding phase, large amount of histamine-binding proteins (HBP) from the lipocalin superfamily are secreted into the site of attachment. Histamine, released from tissue resident mast cells, is scavenged and neutralized by tick HBPs, which inhibit vasodilatation and vascular permeability as well as leukocyte recruitment and edema formation. During the late, rapid feeding phase of a tick, the tick needs more blood influx to the feeding cavity, so it secretes histamine releasing factor (tHRP), which enhances vasodilatation and the tick has better access to blood. The edema formation might be prevented by other salivary effectors that may be present simultaneously at the feeding site.
5.2.4 Disintegrin-like proteins
A relatively large portion of tick salivary transcripts encodes for peptides that bear an argininglycine-aspartate (RGD) motif which is involved in binding to αIIbβ3 integrin on platelets. Under physiological conditions, the RGD motif is present in extracellular matrix proteins; however, if the platelets bind to an RGD motif present on a soluble protein—such as a disintegrin from viper venom or tick saliva—then platelet apoptosis will be induced and platelet aggregation will be inhibited.
RGD-containing salivary proteins were found in all tick families, and some were proved to be antiplatelet agents, such as the first-described RGD motif-containing tick protein, named variabilin. Variabilin is a 5-kDa protein from the tick Dermacentor variabilis that serves as an αIIbβ3 integrin antagonist [62]. Members of the same family are savignygrin and monogrin from the ticks O. savignyi and A. monolakensis, respectively, that bear RGD motifs on a Kunitz domain scaffold. Unlike other Kunitz-domain proteins, both polypeptides lack inhibitory activity against serine proteases and thus they mainly function as antiplatelet proteins [29, 63]. Savignygrin-like proteins were found also in the armamentarium of Ornithodoros coriaceus [64].
Disagregin is a Kunitz domain-containing protein from the tick O. moubata that binds to αIIbβ3 integrin, although it lacks an RGD domain [65]. Transcripts encoding for proteins with an RGD motif that are related to variabilin and mamba dendroaspin were found in the transcriptomes of Ixodes species [16, 66–67]. These proteins are named ixodegrins and they contain an intact or slightly modified RGD motif; it is thus possible that they are involved in platelet aggregation inhibition, but their functional characterization and activity are not disclosed thus far.
A protein that inhibits adhesion of platelets to collagen was isolated from the tick O. moubata [68] and it was named as tick adhesion inhibitor (TAI). TAI is a 5-kDa peptide that competes with vertebrate collagen for binding to platelet integrin a2β1 (GPIa/IIa). Under physiological conditions, integrin a2β1 is the collagen receptor on platelets and mediates platelet adhesion to tissue collagen. Thus, it is not surprising that TAI inhibits the binding of platelets to collagen, through its interaction with integrin a2β1. Another inhibitor of collagen-induced platelet aggregation is a 16-kDa protein named longicornin from the tick H. longicornis [69]. Both TAI and longicornin were studied only as purified isolates from the tick salivary content and they were not cloned as recombinant proteins.
5.2.5 Antiplatelet activities of tick serine protease inhibitors
Because platelets are activated via protease activated receptors (PARs), namely PAR-1, -3 and -4 [70], we must iterate the role of protease inhibitors as antiplatelet agents. PAR-1 and -3 are activated by thrombin, so all thrombin inhibitors mentioned in regard to coagulation may also be mentioned here. In addition to thrombin, platelets are activated by cathepsin G via PAR-4 [71]. The only tick inhibitor of cathepsin G is IRS-2 and it was described from the tick I. ricinus as a member of the serpin superfamily [21]. In addition to thrombin-induced platelet aggregation, IRS-2 inhibited cathepsin G-induced platelet aggregation, while it did not interfere with other, non-proteolytic stimuli of platelet aggregation such as the addition of collagen or of arachidonic acid derivatives in platelets.
6. Tick vasoactive proteins
To our knowledge, there are not tick salivary proteins that would be solely considered as specific vasodilatators; however, there is an active crosstalk among all the components of hemostasis and innate immunity. Therefore we may assume that, for example, histamine scavenging proteins from the lipocalin superfamily can affect both inflammation and vasoconstriction, since the scavenged histamine has multiple effects in vertebrate physiology. As mentioned above, I. scapularis secretes a salivary protein that displays an activity similar to mammalian histamine release factor [61] and thus it may be considered as a vasodilator.
Moreover, chymase and cathepsin G, which are activated and released upon tissue injury, proteolytically activate endothelins into potent vasoconstrictors. IRS-2 is an inhibitor of chymase and therefore, IRS-2 can hypothetically act also as a specific vasoconstriction inhibitor by inhibiting chymase-mediated endothelin processing [21]. Interestingly, the endothelin cleavage site for chymase differs only in one amino acid residue from the binding site of IRS-2 to chymase. This suggests a unique inhibition mechanism of chymase-mediated endothelin processing in the presence of IRS-2.
7. Conclusion
Tick saliva is a very potent pharmacological mixture that successfully interferes with a wide range of host defensive mechanisms. In this chapter, we focused only on the mechanisms related to hemostasis. Ticks and vertebrate hosts have co-evolved through adaptation events—primarily from the tick side—for a long time, so ticks have developed unique strategies to deal with the complicated homeostatic mechanisms of the vertebrate host. The database of transcripts that are being expressed in the SGs of ticks may serve as a solid foundation for drug discovery and the development of novel pharmaceutical applications. The advantage of tick proteins is their divergent primary structures that account for their stringent specificity, thus providing the opportunity to target certain host physiological processes with minimal side effects; however, only a small portion of salivary proteins from different ticks is currently functionally characterized. We look forward to a time when we can describe the complexity of interaction between ticks and hosts at the pharmacological level. The research on purified tick salivary proteins began a couple of decades ago and many proteins were identified and characterized to different extent; however, no tick salivary protein has been studied yet for potential therapeutic applications at the level of clinical trials. We anticipate that the main use of tick antihemostatics will be most likely rather in traumatology and sepsis prevention than in therapy.
Highlights.
-
➢
We explain why it is important for ticks to control host hemostasis.
-
➢
The hemostatic processes affected by tick salivary secretion are presented.
-
➢
The developments in the discovery of tick salivary proteins are highlighted.
-
➢
The different tick proteins that have antihemostatic function are reviewed.
-
➢
We abstract the mechanism of action of tick antihemostatics.
Acknowledgments
E.C. and I.M.B.F. received support from the Intramural Research Program of the Division of Intramural Research, National Institute of Allergy and Infectious Diseases, National Institutes of Health. J.C. and M.K. received support from grant number Z60220518 of the National Academy of Sciences of the Czech Republic and from grant number P502/12/2409 of the Grant Agency of the Czech Republic. M.K. received support from a Jan Evangelista Purkyne fellowship of the National Academy of Sciences of the Czech Republic and from a Marie Curie Reintegration grant of the EU-FP7 (PIRG07-GA-2010-268177). JHFP and MK received support from the National Institutes of Health (R01AI093653) and JHFP from the University of California Research Initiation Funds. We thank NIAID intramural editor Brenda Rae Marshall and Dr James Valdes for assistance and comments on the manuscript.
Because E.C. and I.M.B.F., are government employees and this is a government work, the work is in the public domain in the United States. Notwithstanding any other agreements, the NIH reserves the right to provide the work to PubMedCentral for display and use by the public, and PubMedCentral may tag or modify the work consistent with its customary practices. You can establish rights outside of the U.S. subject to a government use license.
Abbreviations used (alphabetically):1
- aPTT
activated partial thromboplastin time
- BmGTI
Boophilus microplus gut thrombin inhibitor
- BmTI-A
Boophilus microplus trypsin inhibitor-A
- BPTI
bovine pancreatic trypsin inhibitor
- BTSP
basic tail secreted proteins
- FIX
coagulation factor IX
- fMLP
formyl-methionyl-leucyl-proline
- FV
coagulation factor V
- FVII
coagulation factor VII
- FVIIa
activated coagulation factor VII
- FVIII
coagulation factor VIII
- FX
coagulation factor X
- FXa
activated coagulation factor X
- FXI
coagulation factor XI
- FXII
coagulation factor XII
- HBP
histamine binding protein
- HRF
histamine releasing factor
- Ir-CPI
Ixodes ricinus contact phase inhibitor
- IRIS
Ixodes ricinus immunosuppressant
- IRS-2
Ixodes ricinus serpin-2
- NR
non reduntant
- NTI
nymphal thrombin inhibitor
- PAF
platelet aggregation factor
- PAR
protease-activated receptor
- PGE2
prostaglandin E2
- PGs
prostaglandins G
- PT
prothrombin time
- RGD motif
arginine-glycine-aspartate motif
- RSL
reactive site loop
- Salp14
salivary protein 14
- SGE
salivary gland extract
- SGs
salivary glands
- TAI
tick adhesion inhibitor
- TAP
tick anticoagulant protein
- TF
tissue factor
- TFPI
tissue factor pathway inhibitor
- TSGP
tick salivary gland protein
- TT
thrombin time
- TXA2
thromboxane A2
- Xase
tenase complex
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Mann KG, Butenas S, Brummel K. The dynamics of thrombin formation. Arterioscler Thromb Vasc Biol. 2003;23:17–25. doi: 10.1161/01.atv.0000046238.23903.fc. [DOI] [PubMed] [Google Scholar]
- 2.Kannemeier C, Shibamiya A, Nakazawa F, Trusheim H, Ruppert C, Markart P, et al. Extracellular RNA constitutes a natural procoagulant cofactor in blood coagulation. Proc Natl Acad Sci U S A. 2007;104:6388–6393. doi: 10.1073/pnas.0608647104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Semple JW, Italiano JE, Jr, Freedman J. Platelets and the immune continuum. Nat Rev Immunol. 2011;11:264–274. doi: 10.1038/nri2956. [DOI] [PubMed] [Google Scholar]
- 4.Massberg S, Grahl L, von Bruehl ML, Manukyan D, Pfeiler S, Goosmann C, et al. Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat Med. 2010;16:887–896. doi: 10.1038/nm.2184. [DOI] [PubMed] [Google Scholar]
- 5.Ribeiro JM, Makoul GT, Levine J, Robinson DR, Spielman A. Antihemostatic, antiinflammatory, and immunosuppressive properties of the saliva of a tick, Ixodes dammini. J Exp Med. 1985;161:332–344. doi: 10.1084/jem.161.2.332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ribeiro JM, Makoul GT, Robinson DR. Ixodes dammini: evidence for salivary prostacyclin secretion. J Parasitol. 1988;74:1068–1069. [PubMed] [Google Scholar]
- 7.Urioste S, Hall LR, Telford SR, 3rd, Titus RG. Saliva of the Lyme disease vector, Ixodes dammini, blocks cell activation by a nonprostaglandin E2-dependent mechanism. J Exp Med. 1994;180:1077–1085. doi: 10.1084/jem.180.3.1077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dickinson RG, O'Hagan JE, Schotz M, Binnington KC, Hegarty MP. Prostaglandin in the saliva of the cattle tick Boophilus microplus. Aust J Exp Biol Med Sci. 1976;54:475–486. doi: 10.1038/icb.1976.48. [DOI] [PubMed] [Google Scholar]
- 9.Inokuma H, Kemp DH, Willadsen P. Comparison of prostaglandin E2 (PGE2) in salivary gland of Boophilus microplus, Haemaphysalis longicornis and Ixodes holocyclus, and quantification of PGE2 in saliva, hemolymph, ovary and gut of B. microplus. J Vet Med Sci. 1994;56:1217–1218. doi: 10.1292/jvms.56.1217. [DOI] [PubMed] [Google Scholar]
- 10.Bowman AS, Sauer JR. Tick salivary glands: function, physiology and future. Parasitology. 2004;129(Suppl):S67–S81. doi: 10.1017/s0031182004006468. [DOI] [PubMed] [Google Scholar]
- 11.Ribeiro JM. Ixodes dammini: salivary anti-complement activity. Exp Parasitol. 1987;64:347–353. doi: 10.1016/0014-4894(87)90046-4. [DOI] [PubMed] [Google Scholar]
- 12.Ribeiro JM, Weis JJ, Telford SR., 3rd Saliva of the tick Ixodes dammini inhibits neutrophil function. Exp Parasitol. 1990;70:382–388. doi: 10.1016/0014-4894(90)90121-r. [DOI] [PubMed] [Google Scholar]
- 13.Gordon JR, Allen JR. Factors V and VII anticoagulant activities in the salivary glands of feeding Dermacentor andersoni ticks. J Parasitol. 1991;77:167–170. [PubMed] [Google Scholar]
- 14.Gordon JR, Allen JR. Nonspecific activation of complement factor 5 by isolated Dermacentor andersoni salivary antigens. J Parasitol. 1991;77:296–301. [PubMed] [Google Scholar]
- 15.Valenzuela JG. High-throughput approaches to study salivary proteins and genes from vectors of disease. Insect Biochem Mol Biol. 2002;32:1199–1209. doi: 10.1016/s0965-1748(02)00083-8. [DOI] [PubMed] [Google Scholar]
- 16.Chmelar J, Anderson JM, Mu J, Jochim RC, Valenzuela JG, Kopecky J. Insight into the sialome of the castor bean tick, Ixodes ricinus. BMC Genomics. 2008;9:233. doi: 10.1186/1471-2164-9-233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Francischetti IM, Sa-Nunes A, Mans BJ, Santos IM, Ribeiro JM. The role of saliva in tick feeding. Front Biosci. 2009;14:2051–2088. doi: 10.2741/3363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rawlings ND, Tolle DP, Barrett AJ. Evolutionary families of peptidase inhibitors. Biochem J. 2004;378:705–716. doi: 10.1042/BJ20031825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Paesen GC, Siebold C, Dallas ML, Peers C, Harlos K, Nuttall PA, et al. An ion-channel modulator from the saliva of the brown ear tick has a highly modified Kunitz/BPTI structure. J Mol Biol. 2009;389:734–747. doi: 10.1016/j.jmb.2009.04.045. [DOI] [PubMed] [Google Scholar]
- 20.Paesen GC, Siebold C, Harlos K, Peacey MF, Nuttall PA, Stuart DI. A tick protein with a modified Kunitz fold inhibits human tryptase. J Mol Biol. 2007;368:1172–1186. doi: 10.1016/j.jmb.2007.03.011. [DOI] [PubMed] [Google Scholar]
- 21.Chmelar J, Oliveira CJ, Rezacova P, Francischetti IM, Kovarova Z, Pejler G, et al. A tick salivary protein targets cathepsin G and chymase and inhibits host inflammation and platelet aggregation. Blood. 2011;117:736–744. doi: 10.1182/blood-2010-06-293241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sugino M, Imamura S, Mulenga A, Nakajima M, Tsuda A, Ohashi K, et al. A serine proteinase inhibitor (serpin) from ixodid tick Haemaphysalis longicornis; cloning and preliminary assessment of its suitability as a candidate for a tick vaccine. Vaccine. 2003;21:2844–2851. doi: 10.1016/s0264-410x(03)00167-1. [DOI] [PubMed] [Google Scholar]
- 23.Mulenga A, Tsuda A, Onuma M, Sugimoto C. Four serine proteinase inhibitors (serpin) from the brown ear tick, Rhiphicephalus appendiculatus; cDNA cloning and preliminary characterization. Insect Biochemistry and Molecular Biology. 2003;33:267–276. doi: 10.1016/s0965-1748(02)00240-0. [DOI] [PubMed] [Google Scholar]
- 24.Mulenga A, Khumthong R, Blandon MA. Molecular and expression analysis of a family of the Amblyomma americanum tick Lospins. Journal of Experimental Biology. 2007;219:3188–3198. doi: 10.1242/jeb.006494. [DOI] [PubMed] [Google Scholar]
- 25.Leboulle G, Crippa M, Decrem Y, Mejri N, Brossard M, Bollen A, et al. Characterization of a novel salivary immunosuppressive protein from Ixodes ricinus ticks. J Biol Chem. 2002;277:10083–10089. doi: 10.1074/jbc.M111391200. [DOI] [PubMed] [Google Scholar]
- 26.Mulenga A, Khumthong R, Chalaire KC. Ixodes scapularis tick serine proteinase inhibitor (serpin) gene family; annotation and transcriptional analysis. BMC Genomics. 2009;10:217. doi: 10.1186/1471-2164-10-217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.van de Locht A, Stubbs MT, Bode W, Friedrich T, Bollschweiler C, Hoffken W, et al. The ornithodorin-thrombin crystal structure, a key to the TAP enigma? EMBO J. 1996;15:6011–6017. [PMC free article] [PubMed] [Google Scholar]
- 28.Nienaber J, Gaspar AR, Neitz AW. Savignin, a potent thrombin inhibitor isolated from the salivary glands of the tick Ornithodoros savignyi (Acari: Argasidae) Exp Parasitol. 1999;93:82–91. doi: 10.1006/expr.1999.4448. [DOI] [PubMed] [Google Scholar]
- 29.Mans BJ, Andersen JF, Schwan TG, Ribeiro JM. Characterization of anti-hemostatic factors in the argasid, Argas monolakensis: implications for the evolution of blood-feeding in the soft tick family. Insect Biochem Mol Biol. 2008;38:22–41. doi: 10.1016/j.ibmb.2007.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Macedo-Ribeiro S, Almeida C, Calisto BM, Friedrich T, Mentele R, Sturzebecher J, et al. Isolation, cloning and structural characterisation of boophilin, a multifunctional Kunitz-type proteinase inhibitor from the cattle tick. PLoS One. 2008;3:e1624. doi: 10.1371/journal.pone.0001624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lai R, Takeuchi H, Jonczy J, Rees HH, Turner PC. A thrombin inhibitor from the ixodid tick, Amblyomma hebraeum. Gene. 2004;342:243–249. doi: 10.1016/j.gene.2004.07.012. [DOI] [PubMed] [Google Scholar]
- 32.Liao M, Zhou J, Gong H, Boldbaatar D, Shirafuji R, Battur B, et al. Hemalin, a thrombin inhibitor isolated from a midgut cDNA library from the hard tick Haemaphysalis longicornis. J Insect Physiol. 2009;55:164–173. doi: 10.1016/j.jinsphys.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 33.Gao X, Shi L, Zhou Y, Cao J, Zhang H, Zhou J. Characterization of the anticoagulant protein Rhipilin-1 from the Rhipicephalus haemaphysaloides tick. J Insect Physiol. 2011;57:339–343. doi: 10.1016/j.jinsphys.2010.12.001. [DOI] [PubMed] [Google Scholar]
- 34.Iwanaga S, Okada M, Isawa H, Morita A, Yuda M, Chinzei Y. Identification and characterization of novel salivary thrombin inhibitors from the ixodidae tick, Haemaphysalis longicornis. Eur J Biochem. 2003;270:1926–1934. doi: 10.1046/j.1432-1033.2003.03560.x. [DOI] [PubMed] [Google Scholar]
- 35.Nakajima C, Imamura S, Konnai S, Yamada S, Nishikado H, Ohashi K, et al. A novel gene encoding a thrombin inhibitory protein in a cDNA library from Haemaphysalis longicornis salivary gland. J Vet Med Sci. 2006;68:447–452. doi: 10.1292/jvms.68.447. [DOI] [PubMed] [Google Scholar]
- 36.Ibrahim MA, Ghazy AH, Maharem T, Khalil M. Isolation and properties of two forms of thrombin inhibitor from the nymphs of the camel tick Hyalomma dromedarii (Acari: Ixodidae) Exp Appl Acarol. 2001;25:675–698. doi: 10.1023/a:1016136207308. [DOI] [PubMed] [Google Scholar]
- 37.Ciprandi A, de Oliveira SK, Masuda A, Horn F, Termignoni C. Boophilus microplus: its saliva contains microphilin, a small thrombin inhibitor. Exp Parasitol. 2006;114:40–46. doi: 10.1016/j.exppara.2006.02.010. [DOI] [PubMed] [Google Scholar]
- 38.Horn F, dos Santos PC, Termignoni C. Boophilus microplus anticoagulant protein: an antithrombin inhibitor isolated from the cattle tick saliva. Arch Biochem Biophys. 2000;384:68–73. doi: 10.1006/abbi.2000.2076. [DOI] [PubMed] [Google Scholar]
- 39.Ricci CG, Pinto AF, Berger M, Termignoni C. A thrombin inhibitor from the gut of Boophilus microplus ticks. Exp Appl Acarol. 2007;42:291–300. doi: 10.1007/s10493-007-9097-7. [DOI] [PubMed] [Google Scholar]
- 40.Koh CY, Kazimirova M, Trimnell A, Takac P, Labuda M, Nuttall PA, et al. Variegin, a novel fast and tight binding thrombin inhibitor from the tropical bont tick. J Biol Chem. 2007;282:29101–29113. doi: 10.1074/jbc.M705600200. [DOI] [PubMed] [Google Scholar]
- 41.Zhu K, Bowman AS, Brigham DL, Essenberg RC, Dillwith JW, Sauer JR. Isolation and characterization of americanin, a specific inhibitor of thrombin, from the salivary glands of the lone star tick Amblyomma americanum (L.) Exp Parasitol. 1997;87:30–38. doi: 10.1006/expr.1997.4175. [DOI] [PubMed] [Google Scholar]
- 42.Prevot PP, Adam B, Boudjeltia KZ, Brossard M, Lins L, Cauchie P, et al. Anti-hemostatic effects of a serpin from the saliva of the tick Ixodes ricinus. J Biol Chem. 2006;281:26361–26369. doi: 10.1074/jbc.M604197200. [DOI] [PubMed] [Google Scholar]
- 43.Waxman L, Smith DE, Arcuri KE, Vlasuk GP. Tick anticoagulant peptide (TAP) is a novel inhibitor of blood coagulation factor Xa. Science. 1990;248:593–596. doi: 10.1126/science.2333510. [DOI] [PubMed] [Google Scholar]
- 44.Gaspar AR, Joubert AM, Crause JC, Neitz AW. Isolation and characterization of an anticoagulant from the salivary glands of the tick, Ornithodoros savignyi (Acari: Argasidae) Exp Appl Acarol. 1996;20:583–598. doi: 10.1007/BF00052809. [DOI] [PubMed] [Google Scholar]
- 45.Narasimhan S, Koski RA, Beaulieu B, Anderson JF, Ramamoorthi N, Kantor F, et al. A novel family of anticoagulants from the saliva of Ixodes scapularis. Insect Mol Biol. 2002;11:641–650. doi: 10.1046/j.1365-2583.2002.00375.x. [DOI] [PubMed] [Google Scholar]
- 46.Joubert AM, Crause JC, Gaspar AR, Clarke FC, Spickett AM, Neitz AW. Isolation and characterization of an anticoagulant present in the salivary glands of the bont-legged tick, Hyalomma truncatum. Exp Appl Acarol. 1995;19:79–92. doi: 10.1007/BF00052548. [DOI] [PubMed] [Google Scholar]
- 47.Ibrahim MA, Ghazy AH, Maharem TM, Khalil MI. Factor Xa (FXa) inhibitor from the nymphs of the camel tick Hyalomma dromedarii. Comp Biochem Physiol B Biochem Mol Biol. 2001;130:501–512. doi: 10.1016/s1096-4959(01)00459-6. [DOI] [PubMed] [Google Scholar]
- 48.Francischetti IM, Valenzuela JG, Andersen JF, Mather TN, Ribeiro JM. Ixolaris, a novel recombinant tissue factor pathway inhibitor (TFPI) from the salivary gland of the tick, Ixodes scapularis: identification of factor X and factor Xa as scaffolds for the inhibition of factor VIIa/tissue factor complex. Blood. 2002;99:3602–3612. doi: 10.1182/blood-2001-12-0237. [DOI] [PubMed] [Google Scholar]
- 49.Francischetti IM, Mather TN, Ribeiro JM. Penthalaris, a novel recombinant five-Kunitz tissue factor pathway inhibitor (TFPI) from the salivary gland of the tick vector of Lyme disease, Ixodes scapularis. Thromb Haemost. 2004;91:886–898. doi: 10.1160/TH03-11-0715. [DOI] [PubMed] [Google Scholar]
- 50.Decrem Y, Rath G, Blasioli V, Cauchie P, Robert S, Beaufays J, et al. Ir-CPI, a coagulation contact phase inhibitor from the tick Ixodes ricinus, inhibits thrombus formation without impairing hemostasis. J Exp Med. 2009;206:2381–2395. doi: 10.1084/jem.20091007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kato N, Iwanaga S, Okayama T, Isawa H, Yuda M, Chinzei Y. Identification and characterization of the plasma kallikrein-kinin system inhibitor, haemaphysalin, from hard tick, Haemaphysalis longicornis. Thromb Haemost. 2005;93:359–367. doi: 10.1160/TH04-05-0319. [DOI] [PubMed] [Google Scholar]
- 52.Tanaka AS, Andreotti R, Gomes A, Torquato RJ, Sampaio MU, Sampaio CA. A double headed serine proteinase inhibitor--human plasma kallikrein and elastase inhibitor--from Boophilus microplus larvae. Immunopharmacology. 1999;45:171–177. doi: 10.1016/s0162-3109(99)00074-0. [DOI] [PubMed] [Google Scholar]
- 53.Liyou N, Hamilton S, Elvin C, Willadsen P. Cloning and expression of ecto 5-nucleotidase from the cattle tick Boophilus microplus. Insect Mol Biol. 1999;8:257–266. doi: 10.1046/j.1365-2583.1999.820257.x. [DOI] [PubMed] [Google Scholar]
- 54.Stutzer C, Mans BJ, Gaspar AR, Neitz AW, Maritz-Olivier C. Ornithodoros savignyi: soft tick apyrase belongs to the 5'-nucleotidase family. Exp Parasitol. 2009;122:318–327. doi: 10.1016/j.exppara.2009.04.007. [DOI] [PubMed] [Google Scholar]
- 55.Francischetti IM, Mather TN, Ribeiro JM. Cloning of a salivary gland metalloprotease and characterization of gelatinase and fibrin(ogen)lytic activities in the saliva of the Lyme disease tick vector Ixodes scapularis. Biochem Biophys Res Commun. 2003;305:869–875. doi: 10.1016/s0006-291x(03)00857-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Keller PM, Waxman L, Arnold BA, Schultz LD, Condra C, Connolly TM. Cloning of the cDNA and expression of moubatin, an inhibitor of platelet aggregation. J Biol Chem. 1993;268:5450–5456. [PubMed] [Google Scholar]
- 57.Mans BJ, Ribeiro JM. Function, mechanism and evolution of the moubatin-clade of soft tick lipocalins. Insect Biochem Mol Biol. 2008;38:841–852. doi: 10.1016/j.ibmb.2008.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Paesen GC, Adams PL, Harlos K, Nuttall PA, Stuart DI. Tick histamine-binding proteins: isolation, cloning, and three-dimensional structure. Mol Cell. 1999;3:661–671. doi: 10.1016/s1097-2765(00)80359-7. [DOI] [PubMed] [Google Scholar]
- 59.Sangamnatdej S, Paesen GC, Slovak M, Nuttall PA. A high affinity serotonin- and histamine-binding lipocalin from tick saliva. Insect Mol Biol. 2002;11:79–86. doi: 10.1046/j.0962-1075.2001.00311.x. [DOI] [PubMed] [Google Scholar]
- 60.Mans BJ, Ribeiro JM, Andersen JF. Structure, function, and evolution of biogenic amine-binding proteins in soft ticks. J Biol Chem. 2008;283:18721–18733. doi: 10.1074/jbc.M800188200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Dai J, Narasimhan S, Zhang L, Liu L, Wang P, Fikrig E. Tick histamine release factor is critical for Ixodes scapularis engorgement and transmission of the lyme disease agent. PLoS Pathog. 2010;6:e1001205. doi: 10.1371/journal.ppat.1001205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang X, Coons LB, Taylor DB, Stevens SE, Jr, Gartner TK. Variabilin, a novel RGD-containing antagonist of glycoprotein IIb-IIIa and platelet aggregation inhibitor from the hard tick Dermacentor variabilis. J Biol Chem. 1996;271:17785–17790. doi: 10.1074/jbc.271.30.17785. [DOI] [PubMed] [Google Scholar]
- 63.Mans BJ, Louw AI, Neitz AW. Savignygrin, a platelet aggregation inhibitor from the soft tick Ornithodoros savignyi, presents the RGD integrin recognition motif on the Kunitz-BPTI fold. J Biol Chem. 2002;277:21371–21378. doi: 10.1074/jbc.M112060200. [DOI] [PubMed] [Google Scholar]
- 64.Francischetti IM, Meng Z, Mans BJ, Gudderra N, Hall M, Veenstra TD, et al. An insight into the salivary transcriptome and proteome of the soft tick and vector of epizootic bovine abortion, Ornithodoros coriaceus. J Proteomics. 2008;71:493–512. doi: 10.1016/j.jprot.2008.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Karczewski J, Endris R, Connolly TM. Disagregin is a fibrinogen receptor antagonist lacking the Arg-Gly-Asp sequence from the tick, Ornithodoros moubata. J Biol Chem. 1994;269:6702–6708. [PubMed] [Google Scholar]
- 66.Francischetti IM, My Pham V, Mans BJ, Andersen JF, Mather TN, Lane RS, et al. The transcriptome of the salivary glands of the female western black-legged tick Ixodes pacificus (Acari: Ixodidae) Insect Biochem Mol Biol. 2005;35:1142–1161. doi: 10.1016/j.ibmb.2005.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ribeiro JM, Alarcon-Chaidez F, Francischetti IM, Mans BJ, Mather TN, Valenzuela JG, et al. An annotated catalog of salivary gland transcripts from Ixodes scapularis ticks. Insect Biochem Mol Biol. 2006;36:111–129. doi: 10.1016/j.ibmb.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 68.Karczewski J, Waxman L, Endris RG, Connolly TM. An inhibitor from the argasid tick Ornithodoros moubata of cell adhesion to collagen. Biochem Biophys Res Commun. 1995;208:532–541. doi: 10.1006/bbrc.1995.1371. [DOI] [PubMed] [Google Scholar]
- 69.Cheng Y, Wu H, Li D. An inhibitor selective for collagen-stimulated platelet aggregation from the salivary glands of hard tickHaemaphysalis longicornis and its mechanism of action. Sci China C Life Sci. 1999;42:457–464. doi: 10.1007/BF02881768. [DOI] [PubMed] [Google Scholar]
- 70.Steinhoff M, Buddenkotte J, Shpacovitch V, Rattenholl A, Moormann C, Vergnolle N, et al. Proteinase-activated receptors: transducers of proteinase-mediated signaling in inflammation and immune response. Endocr Rev. 2005;26:1–43. doi: 10.1210/er.2003-0025. [DOI] [PubMed] [Google Scholar]
- 71.Sambrano GR, Huang W, Faruqi T, Mahrus S, Craik C, Coughlin SR. Cathepsin G activates protease-activated receptor-4 in human platelets. J Biol Chem. 2000;275:6819–6823. doi: 10.1074/jbc.275.10.6819. [DOI] [PubMed] [Google Scholar]