Abstract
Background
Pre-operative risk stratification is commonly performed by assessing end organ function (such as cardiac and pulmonary) to define post-operative risk. Little is known about impaired pre-operative cognition and outcomes. The purpose of this study was to evaluate the impact of baseline impaired cognition on post-operative outcomes in geriatric surgery patients.
Study Design
Prospectively, subjects 65 years and older undergoing a planned elective operation requiring post-operative ICU admission were recruited. Pre-operative baseline cognition was assessed using the validated Mini-Cog test. Impaired cognition was defined as a Mini-Cog score of ≤3. Delirium was assessed using the Confusion Assessment Method-ICU by a trained research team. Adverse outcomes were defined using the Veterans Affairs Surgical Quality Improvement Program definitions.
Results
186 subjects were included with an average age of 73±6 years. Eighty-two subjects (44%) had baseline impaired cognition. The impaired cognition group had the unadjusted outcomes: increased incidence of ≥1 post-operative complication (41% versus 24%; p=.011), higher incidence of delirium (78% versus 37%; p<.001), longer hospital stays (15±14 versus 9±9 days; p=.008), higher rate of discharge institutionalization (42% versus 18%; p=.001) and higher six-month mortality (13% versus 5%; p=.040). Adjusting for potential confounders determined by univariate analysis, logistic regression found impaired cognition was still associated to the occurrence of one or more post-operative complications (odds ratio 2.401; 95% confidence interval 1.185, 4.865; p=0.015). Kaplan-Meier survival analysis revealed higher mortality in the impaired cognition group (Log-Rank p =.008).
Conclusions
Baseline cognitive impairment in older adults undergoing major elective operations is related to adverse post-operative outcomes including increased complications, length of stay and long-term mortality. Improved understanding of baseline cognition and surgical outcomes may aid surgical decision-making in older adults.
INTRODUCTION
With a growing population of older adults undergoing complex surgical care, pre-operative risk assessment is becoming increasingly important. Pre-operative risk stratification allows the clinical team to forecast post-operative outcomes. The most common strategy to identify high risk patients prior to surgery is assessment of single end-organ function.1 This tactic is most widely recognized by the American Heart Association’s guideline for cardiac evaluation,2 but is also well described for pulmonary,3 hepatic4 and renal5 organ systems. Little is known about the effects of pre-existing cognitive (or brain) function (perhaps the most vital human organ) on post-operative outcomes.
Cognition refers to an individual’s mental function. Impaired cognition, commonly referred to as dementia (but also includes mild cognitive impairment), is an acquired dysfunction in at least two domains of intellectual capacity including memory, language, visuospatial, executive functioning and calculation that impacts functional abilities.6 Dementia in older adults is closely related to an increased incidence of adverse healthcare events including morbidity (pneumonia, febrile episodes and feeding problems) as well as high six-month mortality.7 The relationship between impaired cognition and post-operative outcomes is not known.
The purpose of this study was to evaluate the impact of baseline impaired cognition on post-operative outcomes in older adults undergoing major elective operations. The specific aims were to compare post-operative outcomes in older adults with and without baseline impaired cognition including: the incidence of adverse events, the incidence and duration of delirium, and long-term survival.
METHODS
This prospective cohort study was performed at the Denver Veteran Affairs Medical Center. Regulatory approval was obtained through the Colorado Multiple Institutional Review Board (COMIRB #05-0281) and written informed consent was obtained from all study participants. Participants were enrolled between October 2005 and November 2008.
Inclusion criteria were persons 65 years and older undergoing an elective operation with a planned post-operative admission to the intensive care unit (ICU) on the general, cardiac, non-cardiac thoracic, urologic and vascular surgical services. Exclusion criteria were: (1) patients with vision or hearing impairment who could not visualize pictures or hear instructions associated with the delirium assessments, (2) non-English speaking patients (so language barrier confusion was not mistaken for delirium), and (3) patients who could not undergo informed consent.
Cognitive function was assessed at baseline prior to the elective operation in all subjects using the Mini-Cog test by a member of a trained research team. The Mini-Cog is a simple, brief screening tool designed to detect cognitive impairment (or dementia) that has been validated in community based and veteran patient populations.8–9 The Mini-Cog combines an uncued three-item recall test with a clock drawing task and typically takes less than two minutes to complete. The Mini-Cog has 99% sensitivity and 93% specificity when used as a screening tool for impaired cognition.10 Scoring of the Mini-Cog ranges from 0 (severe cognitive impairment) to 5 (normal cognition). This study defined impaired cognition as a Mini-Cog score of 3 or less; a definition previously used by our group11–12 and others.9
Routine pre-, intra- and post-operative variables were recorded prospectively. Pre-operative variables recorded included four cardiovascular disease risk factors including a history of hypertension, diabetes mellitus, coronary artery disease and hypercholesterolemia. Because cardiovascular disease is a major cause of cognitive impairment and might confound the relationship of impaired cognition and the occurrence one or more complications, cardiovascular risk factors were summed in patients to reflect total burden of cardiovascular disease. Subjects were grouped if they had two or more cardiovascular risk factors and three or more cardiovascular risk factors. Post-operative complications were defined using Veterans Affairs Surgery Quality Improvement Program (VASQIP)13 definitions and were recorded posptectively by the research team: cardiac (cardiac arrest requiring cardiopulmonary resuscitation [CDARREST] or myocardial infarction [CDMI]); respiratory (pneumonia [OUPNEUMO], pulmonary embolism [PULEMBOL] or reintubation for respiratory/cardiac failure [REINTUB]); renal insufficiency [RENAINSF]; neurologic (cerebral vascular accident/stroke [CNSCVA] or coma>24 hours [CNSCOMA]); post-operative infection (deep wound surgical site infection [WNDINFD], superficial surgical site infection [SUPINFEC] or urinary tract infection [URNINFEC]); sepsis [OTHSYSEP]; deep vein thrombosis [OTHDVT]; and re-operation (Return to OR [RETURNOR]). The sum of complications was totaled for each subject. Institutionalization was defined as discharge to an institutional care facility (e.g., a nursing home, skilled nursing facility or rehabilitation center). Long-term survival was determined by chart review in which the last clinic encounter was used as the last known date alive or the date of death was recorded.
Delirium was prospectively measured daily by a trained research team using the Confusion Assessment Method-ICU (CAM-ICU).14–15 The research team was trained to assess for delirium by reading and reviewing in a group meeting the publically available online “Confusion Assessment Method for the ICU – A Training Manual”.16 This validated assessment tool evaluates delirium according to four features: (1) fluctuation in mental status, (2) inattention, (3) disorganized thinking, and (4) an altered level of consciousness. Because delirium has a fluctuating course and can be missed by a single daily assessment, delirium researchers have a validated chart review method, looking for key words to indicate its presence (i.e. confusion; agitation; disorientation).17 To establish inter-rater reliability for delirium assessment within the research team, a pilot study of 100 encounters was performed. The research team had a high inter-rater reliability with a concordance rate ≥ 98% and a kappa statistic ≥ 0.96 (95% confidence interval: 0.91 to 1.00). Subjects in the pilot group were excluded from further analysis. This study used the same methodology and subjects as two previously reported studies.18–19
Statistical analysis was performed using bivariate comparisons for cognition (the primary predictor variable). Pre-, intra- and post-operative variables were analyzed using Chi-square tests for categorical variables and Wilcoxon rank-sum tests for non-parametric continuous variables. Multivariate logistic regression was performed with variables that were associated with the occurrence of one or more complication (defined as p<0.1 on univariate analysis) which included age, prior history of stroke, hypertension and American Society of Anesthesiology score. Odds ratios and 95% confidence intervals were determined for each independent variable in the multivariate logistic regression model. A Kaplan-Meier curve estimated the distribution of overall survival and a log-rank test compared time to event distributions. To test for an interaction, or effect modification, between delirium and the association between pre-operative cognition and post-operative death, the data was stratified on delirium and separate models were analyzed. All analyses were performed using SAS version 9.2 (Cary, NC).
RESULTS
A total of 186 patients were studied. The mean age was 73±6 years and 96% (178) were male. The overall incidence of impaired cognition at baseline was 44% (82). The incidence of impaired cognition by decade of age was: 60’s: 17% (11/63), 70’s: 52% (47/90), 80’s: 63% (19/30) and 90’s: 100% (3/3). Baseline characteristics of the impaired and normal cognition groups were compared. The incidence of hypertension, chronic obstructive pulmonary disease, diabetes mellitus and serum creatinine levels were similar in the two groups. The impaired cognition group was older (p<0.001) and had a higher incidence of prior strokes (p=0.001) in comparison to normal cognition group. (see Table 1)
Table 1.
Baseline Characteristics of Patients and Operations
| COGNITION | ||||
|---|---|---|---|---|
| Total Group (n= 186) | Impaired | Normal (n=104) | p Value | |
| Age, y, mean ± SD | 73±6 | 76±6 | 71±5 | <.001 |
| COPD | 25% (47) | 28% (23) | 23% (24) | 0.461 |
| Creatinine, mg/dL | 1.2±0.4 | 1.2±0.4 | 1.2±0.4 | 0.474 |
| Stroke | 8% (14) | 15% (12) | 2% (2) | 0.001 |
| ASA Score | 2.9±0.4 | 3.0±0.4 | 2.9±0.4 | 0.092 |
| Baseline CV Disease Risks | ||||
| Hypertension | 82% (153) | 88% (72) | 78% (81) | 0.086 |
| Diabetes mellitus | 30% (56) | 35% (29) | 26% (27) | 0.198 |
| Coronary artery disease | 39% (73) | 36% (37) | 44% (36) | 0.290 |
| Hypercholesterolemia | 70% (130) | 72% (59) | 68% (71) | 0.651 |
| Burden of CV Disease | ||||
| Two or more* | 72% (134) | 78% (64) | 67% (70) | 0.128 |
| Three or more† | 44% (82) | 50% (41) | 39% (41) | 0.181 |
| Operations | 0.747 | |||
| Abdominal | 43% (80) | 46% (38) | 40% (42) | |
| Cardiac | 39% (72) | 38% (31) | 39% (41) | |
| Non-Cardiac Thoracic | 6% (11) | 6% (5) | 6% (6) | |
| Vascular | 12% (23) | 10% (8) | 14% (15) | |
| OR Time, min | 281±125 | 277±138 | 284±114 | 0.401 |
| Blood loss, mL | 556±912‡ | 617±962 | 508±876 | 0.497 |
p Values compare groups based on cognition (present / absent impaired cognition).
Two or more cardiovascular disease risks (hypertension, diabetes mellitus, coronary artery disease, high cholesterol) present in the same patient.
Three or more cardiovascular disease risks present in the same patient.
Seventy-three operations were cardiac and had no recordable blood loss.
ASA, American Society Anesthesiology; CV, cardiovascular. .
Operative characteristics of the normal and impaired cognition groups were compared. Types of operations, time in the operating room and blood loss were similar in the two groups. (see Table 1) All of the cardiac operations used cardiopulmonary bypass.
Post-operative characteristics were compared between the normal cognition and impaired cognition groups. Cognitive impairment was associated with a higher incidence of one or more post-operative complications (p=0.011), longer hospital stay (p=0.001), higher rate of discharge to an institutional care facility (p=0.001), higher 30-day readmission rate (p=0.033) and a higher six-month mortality (p=0.040). (See Table 2) Post-operative mechanical ventilation was required in 12% (22/186) (mean 2.4 days, median 1 day, rage 1 to 8 days) of the total population, 10% (10/104) in the normal cognition group and 15% (12/82) in the impaired cognition group (p=0.362).
Table 2.
Postoperative Outcomes
| Impaired cognition | ||||
|---|---|---|---|---|
| Complications | Total group (n= 186) | Present (n=82) | Absent (n=104) | p Value |
| One or more complication | 32% (59) | 41% (34) | 24% (25) | 0.011 |
| Cardiac | 3% (5) | 6% (5) | 0% (0) | |
| Respiratory | 6% (12) | 13% (11) | 1% (1) | |
| Renal | 4% (7) | 7% (6) | 1% (1) | |
| Neurologic | 1% (2) | 1% (1) | 1% (1) | |
| Infection | 26% (48) | 33% (27) | 20% (21) | |
| Sepsis | 10% (19) | 17% (14) | 5% (5) | |
| DVT | 2% (3) | 4% (3) | 0% (0) | |
| Reoperation | 7% (13) | 11% (9) | 4% (4) | |
| Delirium | 55% (102) | 78% (64) | 37% (38) | <0.001 |
| Days of delirium | 2±4 | 4±6 | 1±2 | 0.012 |
| ICU Stay, d | 7±9.1 | 9±12.1 | 6±5.5 | 0.008 |
| Hospital stay, d | 12±12 | 15±14 | 9±9 | 0.001 |
| Institutionalization | 29% (52) | 42% (33) | 18% (19) | 0.001 |
| 30-Day readmission | 15% (27) | 21% (17) | 10% (10) | 0.033 |
| 30-Day mortality | 2% (4) | 4% (3) | 1% (1) | 0.322 |
| 6-Month mortality | 9% (16) | 13% (11) | 5% (5) | 0.040 |
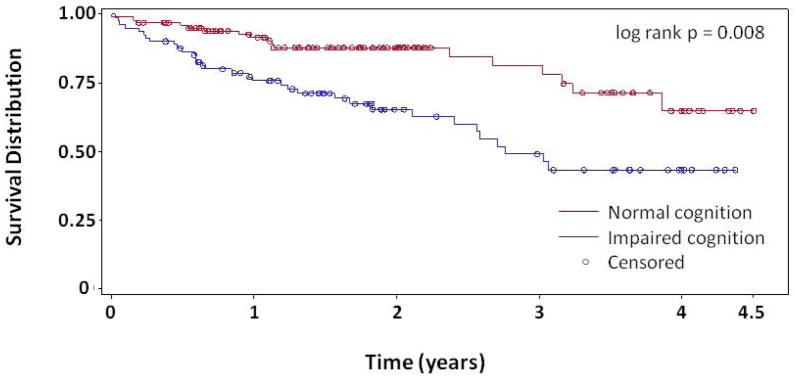
Long-term survival was recorded. The mean follow-up of alive patients was 759 days (range 71 to 1,687; median 667). The mean follow-up of deceased patients was 479 days (range 1 to 1,446; median 362). All patients had follow-up longer than 30 days post-operatively (excluding those with mortality within 30 days). Long-term overall survival was decreased in the impaired cognition group (Log Rank p=0.008). (See Figure 1)
Figure 1.
Kaplan-Meier Survival Curve.
Logistic regression fit to the occurrence of one or more complication was used to adjust for all independent variables associated with the occurrence of one or more complication (defined by a univariate p<0.1). Independent variables (in addition to cognition) included were age, prior history of stroke, hypertension and American Society of Anesthesiology score. After adjusting for age, prior history of stroke, hypertension and American Society of Anesthesiology score, impaired cognition was still significantly associated to the occurrence of one or more complications (odds ratio 2.401; 95% confidence interval 1.185, 4.865; p=0.015).
Delirium occurred in 55% (102). The impaired cognition group had a higher incidence (p<0.001) and longer duration (p=0.012) of delirium in comparison to the normal cognition group. (see Table 2) The data was stratified on the presence/absence of delirium to determine if delirium acted as an effect modifier or a confounder on the relationship of baseline cognition and time to death. When stratified by delirium, the association between cognition and death were different. In the delirium group, the hazard ratio for death was 2.77 (95% CI: 1.14, 6.70) times greater in the impaired cognition group. In the no delirium group, the hazard ratio for death was 1.86 (95% CI: 0.64, 5.46) times greater in the impaired cognition group.
DISCUSSION
A variety of end-organ compromise has been suggested for pre-operative risk stratification (e.g., heart, lung); but correlation of perhaps the most vital organ, the brain, with post-operative outcomes is not well established. The current study found increased postoperative complications in older adults with baseline impaired cognition in comparison to those with normal cognition; a finding that was independent of increasing age, a higher cardiovascular disease burden, prior stoke, history of hypertension and higher ASA score. In addition, longer post-operative hospital stays, higher rates of discharge to an institutional care facility and increased incidence of 30-day readmissions were all related to pre-operative impaired cognitive function. From the standpoint of post-operative delirium, the impaired cognition group not only had a higher incidence of postoperative delirium, but the duration of post-operative delirium was also longer. Finally, patients with baseline impaired cognition had higher overall long-term mortality following major operation in comparison to individuals with normal cognition. These findings occurred in a study population who underwent operations which uniformly created substantial surgical stress (large enough to require a post-operative intensive care unit stay), a fact that is likely more important than the individual organ system involved in the operation.
Impaired cognition, or dementia, is a clinical condition uniquely relevant to older adults. Dementia is uncommon in individuals less than 70 years old, occurs in more than 10% of individuals older than 70 years and increases exponentially with age.9, 20–21 Common causes include neurodegenerative (e.g., Alzheimer’s) and vascular disorders. Impaired cognition is often unrecognized; a fact highlighted by a recent screening study that found 26% of community dwelling individuals 70 years or older without the diagnosis of dementia had a some amount of impaired cognition when screened for the disorder.9 One reason for the under-diagnosis of cognitive impairment may be that the precursor condition of dementia, mild cognitive impairment, or “MCI”, is not apparent in day-to-day interactions. The critical relevance of both mild cognitive impairment and dementia is their association with increased mortality in community dwelling older adults.7, 22
A practical approach that a surgeon can use to assess cognition is the Mini-Cog test.10, 23 The clinician first instructs the patient to remember three unrelated words (e.g., apple, table, penny). The patient is requested to repeat the words back to the clinician to ensure they heard the words. Next, the patient is instructed to draw the face of a clock with the hands on a specific time (the time 11:10 has been endorsed previously23–25). Finally, the patient is instructed to recall the three words. The Mini-Cog is scored by giving one point for each word recalled and a normal clock is scored with 2 points whereas an abnormal clock receives 0 points. Mini-Cog scores can range from 0 (worst score) to 5 (best score). While a more complex scoring method was initially described,10 defining a person as having some cognitive impairment if they score 3 or less on the Mini-Cog (methodology used in this study) is a simplified, accurate scoring method.9
An important finding of this study is that delirium was found to be an effect modifier in the relationship between impaired cognition and adverse post-operative outcomes. Stratifying for delirium when examining the relationship of cognitive function and time to death, the hazards ratios across stratum were different (2.77 versus 1.66). This suggests that delirium acts as an effect modifier, and not a confounder, in the relationship between cognition and time to death. This concept is vitally important because it suggests that pre-existing impaired cognition may be a contributing mechanism explaining why patients develop delirium in the hospital have higher long-term mortality.18, 26
There are two additional reasons this study is important. First, understanding clinical conditions unique to older adults that affect operative outcomes is important. To our knowledge, this is the first study that relates impaired cognition to outcomes following major elective operations. Cognitive impairment is currently not part of any routinely performed pre-operative assessment strategy.1 With an increasing number of complex surgical procedures being performed in older adults, the clinician’s awareness of geriatric specialty topics (e.g., cognitive impairment) that are specifically relevant to older adults’ outcomes will aid both pre-operative counseling and decision-making. The current study reports the relationship between baseline, pre-operative impaired cognition and post-operative outcomes; this study does not account for incorporating logical surgical decision-making responses based on pre-operative cognitive assessment. Second, implementing cognitive assessment into a surgical decision-making process is easily achieved. Assessing cognition may initially seem intimidating to unfamiliar clinicians, a fact that likely explains why an organ as relevant as the brain is not recognized in pre-operative assessment strategies whereas the kidneys, an organ evaluated by a simple/routine blood test, are commonly assessed. Tests such as the Mini-Cog make evaluating cognition feasible to incorporate into a routine pre-operative assessment.
There are two main limitations of this study. First, the majority of study participants were male which does not allow for gender differences of cognition and post-operative outcomes to be detected by this study. However, the study population is demographically similar to a patient population at a Veterans Affairs medical center and does not represent a selection bias. And second, a heterogeneity of operations were included in this study. Including procedures which ranged from cardiopulmonary bypass to pelvic exenterations does not provide a uniform baseline surgical stress. This fact leads to large variability in measurements such as blood loss, operating room time and length of stay. While some of the standard deviations of the outcomes were large, our baseline demographics did find that similar types and number of operations were performed in the impaired and normal cognition groups.
In the future, pre-operative risk stratification will likely not be based on physiologic compromise of a single organ system alone. Instead, the sum of compromise across multiple health-related domains (e.g., cognition, function, nutrition, disease burden) will likely be used to forecast post-operative outcomes.11–12 Future directions of this work include synthesizing information about baseline cognition and post-operative outcomes with other clinical domains relevant to older adults (e.g., function, nutrition, chronic disease burden). The aim of such work will be to construct a measure of vulnerability (or frailty) to accurately predict outcomes following major operations in older adults.
Acknowledgments
Financial Support: Paul B Beeson Award – NIA K23AG034632 (TNR); Dennis W Jahnigen Award - American Geriatrics Society (TNR); NIH K24-HL-089223 (MM).
Footnotes
Disclosure Information: Nothing to disclose.
Presented at the Western Surgical Association 119th Scientific Session, Tucson, AZ, November 2011.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Laine C, Williams SV, Wilson JF. In the clinic. Preoperative evaluation. Ann Intern Med. 2009;151:ITC1-15. doi: 10.7326/0003-4819-151-1-200907070-01006. quiz ITC16. [DOI] [PubMed] [Google Scholar]
- 2.Fleisher LA, Beckman JA, Brown KA, et al. 2009 ACCF/AHA focused update on perioperative beta blockade incorporated into the ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery. J Am Coll Cardiol. 2009;54:e13–e118. doi: 10.1016/j.jacc.2009.07.010. [DOI] [PubMed] [Google Scholar]
- 3.Calderini E, Adrario E, Petrini F, et al. Indications to chest radiograph in preoperative adult assessment: recommendations of the SIAARTI-SIRM commission. Minerva Anestesiol. 2004;70:443–451. [PubMed] [Google Scholar]
- 4.Schneider PD. Preoperative assessment of liver function. Surg Clin North Am. 2004;84:355–373. doi: 10.1016/S0039-6109(03)00224-X. [DOI] [PubMed] [Google Scholar]
- 5.Michota FA., Jr The preoperative evaluation and use of laboratory testing. Cleve Clin J Med. 2006;73 (Suppl 1):S4–7. doi: 10.3949/ccjm.73.suppl_1.s4. [DOI] [PubMed] [Google Scholar]
- 6.Duthie EH, Katz PR, Malone ML. Practice of Geriatrics. 4. Philadelphia: W.B. Saunders Elsevier; 2007. [Google Scholar]
- 7.Mitchell SL, Teno JM, Kiely DK, et al. The clinical course of advanced dementia. N Engl J Med. 2009;361:1529–1538. doi: 10.1056/NEJMoa0902234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Borson S, Scanlan JM, Chen P, Ganguli M. The Mini-Cog as a screen for dementia: validation in a population-based sample. J Am Geriatr Soc. 2003;51:1451–1454. doi: 10.1046/j.1532-5415.2003.51465.x. [DOI] [PubMed] [Google Scholar]
- 9.McCarten JR, Anderson P, Kuskowski MA, et al. Screening for cognitive impairment in an elderly veteran population: acceptability and results using different versions of the Mini-Cog. J Am Geriatr Soc. 2011;59:309–313. doi: 10.1111/j.1532-5415.2010.03249.x. [DOI] [PubMed] [Google Scholar]
- 10.Borson S, Scanlan J, Brush M, et al. The mini-cog: a cognitive 'vital signs' measure for dementia screening in multi-lingual elderly. Int J Geriatr Psychiatry. 2000;15:1021–1027. doi: 10.1002/1099-1166(200011)15:11<1021::aid-gps234>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 11.Robinson TN, Eiseman B, Wallace JI, et al. Redefining geriatric preoperative assessment using frailty, disability and co-morbidity. Ann Surg. 2009;250:449–455. doi: 10.1097/SLA.0b013e3181b45598. [DOI] [PubMed] [Google Scholar]
- 12.Robinson TN, Wallace JI, Wu DS, et al. Accumulated frailty characteristics predict postoperative discharge institutionalization in the geriatric patient. J Am Coll Surg. 2011;213:37–42. doi: 10.1016/j.jamcollsurg.2011.01.056. discussion 42–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Khuri SF, Daley J, Henderson W, et al. The Department of Veterans Affairs' NSQIP: the first national, validated, outcome-based, risk-adjusted, and peer-controlled program for the measurement and enhancement of the quality of surgical care. National VA Surgical Quality Improvement Program. Ann Surg. 1998;228:491–507. doi: 10.1097/00000658-199810000-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ely EW, Inouye SK, Bernard GR, et al. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU) Jama. 2001;286:2703–2710. doi: 10.1001/jama.286.21.2703. [DOI] [PubMed] [Google Scholar]
- 15.Ely EW, Margolin R, Francis J, et al. Evaluation of delirium in critically ill patients: validation of the Confusion Assessment Method for the Intensive Care Unit (CAM-ICU) Crit Care Med. 2001;29:1370–1379. doi: 10.1097/00003246-200107000-00012. [DOI] [PubMed] [Google Scholar]
- 16.Ely EW. The Confusion Assessment Method for the ICU - Training Manual. 2002 http://www.mc.vanderbilt.edu/icudelirium/assessment.html.
- 17.Pisani MA, Araujo KL, Van Ness PH, et al. A research algorithm to improve detection of delirium in the intensive care unit. Crit Care. 2006;10:R121. doi: 10.1186/cc5027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Robinson TN, Raeburn CD, Tran ZV, et al. Postoperative delirium in the elderly: risk factors and outcomes. Ann Surg. 2009;249:173–178. doi: 10.1097/SLA.0b013e31818e4776. [DOI] [PubMed] [Google Scholar]
- 19.Robinson TN, Raeburn CD, Tran ZV, et al. Motor subtypes of postoperative delirium in older adults. Arch Surg. 2011;146:295–300. doi: 10.1001/archsurg.2011.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Plassman BL, Langa KM, Fisher GG, et al. Prevalence of dementia in the United States: the aging, demographics, and memory study. Neuroepidemiology. 2007;29:125–132. doi: 10.1159/000109998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hebert LE, Scherr PA, Bienias JL, et al. Alzheimer disease in the US population: prevalence estimates using the 2000 census. Arch Neurol. 2003;60:1119–1122. doi: 10.1001/archneur.60.8.1119. [DOI] [PubMed] [Google Scholar]
- 22.Bennett DA, Wilson RS, Schneider JA, et al. Natural history of mild cognitive impairment in older persons. Neurology. 2002;59:198–205. doi: 10.1212/wnl.59.2.198. [DOI] [PubMed] [Google Scholar]
- 23.Carolan Doerflinger DM. How To Try This: The Mini Cog. American Journal of Nursing. 2007;107:6–71. doi: 10.1097/01.NAJ.0000301030.81651.66. [DOI] [PubMed] [Google Scholar]
- 24.Adams JH, Gruber NP, Hays JR. Interrater reliability of scores derived from two methods for scoring the clock drawing test. Psychol Rep. 2006;98:39–42. doi: 10.2466/pr0.98.1.39-42. [DOI] [PubMed] [Google Scholar]
- 25.Juby A, Tench S, Baker V. The value of clock drawing in identifying executive cognitive dysfunction in people with a normal Mini-Mental State Examination score. CMAJ. 2002;167:859–864. [PMC free article] [PubMed] [Google Scholar]
- 26.Ely EW, Shintani A, Truman B, et al. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA. 2004;291:1753–1762. doi: 10.1001/jama.291.14.1753. [DOI] [PubMed] [Google Scholar]