Abstract
We investigated diet supplementation with shiitake mushroom fruiting bodies on biochemical and histological changes in hypercholesterolemic rats. Six-wk old female Sprague-Dawley albino rats were divided into three groups of 10 rats each. A diet containing 5% Lentinus edodes fruiting bodies given to hypercholesterolemic rats reduced plasma total cholesterol, triglyceride, low-density lipoprotein (LDL), total lipid, phospholipids, and the LDL/high-density lipoprotein ratio by 34.33, 53.21, 75.00, 34.66, 25.73, and 71.43%, respectively. Feeding mushroom also significantly reduced body weight in hypercholesterolemic rats. However, it had no detrimental effects on plasma albumin, total bilirubin, direct bilirubin, creatinine, blood urea nitrogen, uric acid, glucose, total protein, calcium, sodium, potassium, chloride, inorganic phosphate, magnesium, or enzyme profiles. Feeding mushroom increased total lipid and cholesterol excretion in feces. The plasma lipoprotein fraction, separated by agarose gel electrophoresis, indicated that L. edodes significantly reduced plasma β and pre-β-lipoprotein but increased α-lipoprotein. A histological study of hepatic cells by conventional hematoxylin-eosin and oil red-O staining showed normal findings for mushroom-fed hypercholesterolemic rats. These results suggest that shiitake mushrooms could be recommended as a natural cholesterol lowering substance in the diet.
Keywords: Agarose gel electrophoresis, Atherogenic lipid profile, Histopathology, Hypercholesterolemic rats, Lentinus edodes
Lentinus edodes, widely known as the shiitake mushroom, has an established history of use in time-honored oriental therapies. Modern clinical practice in Korea, Japan, China, and other Asian countries continues to rely on mushroom-derived preparations. These practices still form the basis of modern scientific studies of mushroom medicinal activities [1]. Eritadenine is an adenosine analogue alkaloid and lentinacin is a purine alkaloid that reduces cholesterol levels in rats by 25% after 7 days of oral administration at doses as low as 0.005% of feed intake [2]. The main cause of the hypocholesterolemic action of eritadenine seems to be associated with a modification in hepatic phospholipid (PL) metabolism by inducing phosphatylethanolamine N-methyltransferase deficiency [3]. Dietary eritadenine also alters the fatty acid and molecular profile of the liver and plasma by suppressing the metabolic conversion of linoleic acid to arachidonic acid and by decreasing the Δ6-desaturase activity, which can be affected through transcriptional regulation.
The microsomal enzyme 3-hydroxy-3-methylglutarylcoenzymeA (HMG-CoA) reductase is the major rate-limiting enzyme in cholesterol biosynthesis, which converts HMG-CoA to mevalonate. Therefore, inhibiting HMGCoA reductase decreases intracellular cholesterol biosynthesis [4]. Mevinolin is a known pharmacological HMGCoA reductase inhibitor. A high quantity of this inhibitor has been found in mushroom fruiting bodies, particularly in the pileus and sporocarps of Pleurotus ostreatus. Mushrooms also contain various biologically active compounds such as gallic acid, protocatechuic acid, chlorogenic acid, naringenin, hesperetin, and biochanin-A [5, 6].
The fruiting bodies of L. edodes lessen blood platelet effectiveness during coagulation and consequently those who bleed easily or who take anticoagulants should be cautious when chronically consuming L. edodes extracts [7]. Nevertheless, the exact mechanisms of action remain to be understood before considering a human treatment to prevent or cure cardiovascular diseases. Various studies have shown that shiitake mushrooms lower both blood pressure and free cholesterol in plasma and accelerate lipid accumulation in the liver by removing them from circulation [8]. The present study was designed to evaluate the dietary effectiveness of L. edodes on plasma, feces, and hepatic tissue function in hypercholesterolemic rats.
Materials and Methods
Mushroom
Fresh fruiting bodies of L. edodes were purchased from E-mart at Incheon in Korea. A pure culture was deposited in the Culture Collection and DNA Bank of Mushroom (CCDBM), Division of Life Sciences, University of Incheon, Korea and acquired accession number, IUM-4681. Fresh fruiting bodies were dried with hot air at 40℃ for 48 hr and pulverized.
Animals
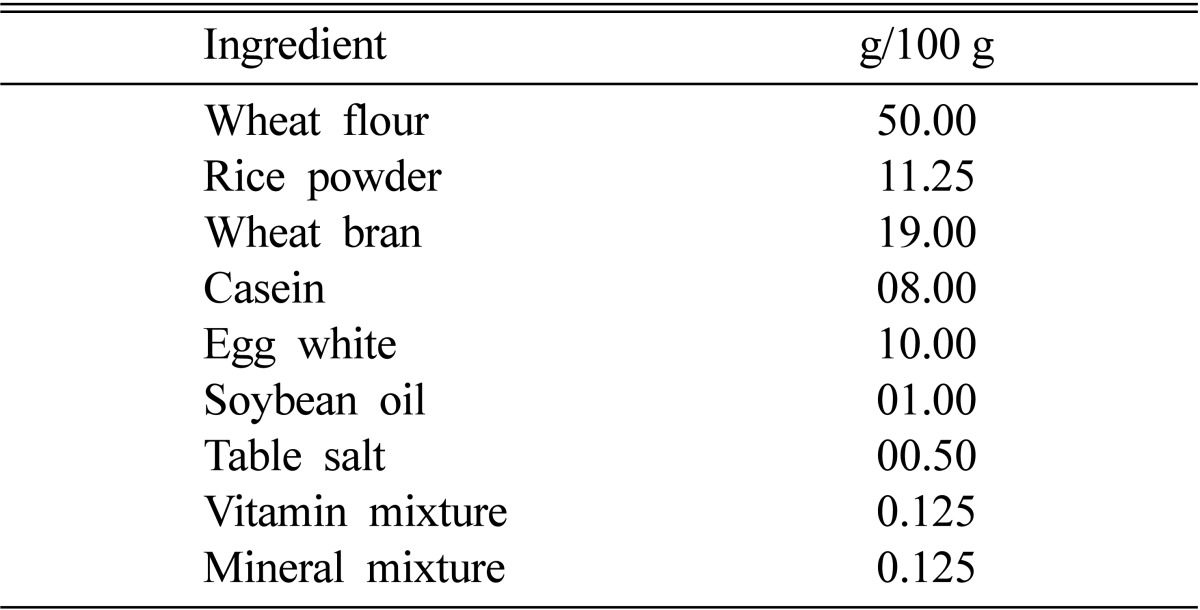
Thirty female Sprague-Dawley albino rats (101 ± 4.2 g, 6 wk old, purchased from Central Lab. Animal Inc., Seoul, Korea) were used. All animals were acclimated to the animal room for 1 wk. The rats were housed in an animal room at 23 ± 2℃ under a 12 hr dark-light cycle (17:00~05:00) and a relative humidity of 50~60%. Rats were divided into three feed groups: basal diet (normocholesterolemic control rats; NC), basal diet with 1% cholesterol (hypercholesterolemic rats; HC), and basal diet with 1% cholesterol and 5% L. edodes powder (mushroom-fed hypercholesterolemic rats; HC + LE). The basal diet compositions are presented in Table 1, and the rats were fed for 42 days.
Table 1.
Basal diet composition
The composition of the vitamin mixture in the diet was as follows (g/100 g vitamin mixture): retinyl acetate 9.5 × 10-4, cholecalciferol 1.2 × 10-3, α-tocopherol acetate 0.05, thiamine hydrochloride 2.4, nicotinic acid 12, riboflavin 2.4, D-calcium pantothenate 9.6, pyridoxine hydrochloride 1.2, folic acid 9.5 × 10-2, vitamin K 0.25, cyanocobalamine 9.5 × 10-3, inositol 47.95, and ascorbic acid 24.0. The composition of the mineral mixture added to the diet was as follows (g/100 g of mineral): calcium gluconate 28.5, K2HPO4 17.3, CaCO3 26, MgSO4 12.6, KCl 12.6, CuSO4 0.06, FeSO4 0.3, MnSO4 0.55, NaF 2.5 × 10-4, KI 9× 10-4, sodium molybdate 3 × 10-4, SeO2 3 × 10-4, and CrSO2 1.5 × 10-3.
Plasma chemical analysis
At the end of the experimental period, overnight-fasted animals were sacrificed under injectable anesthetic (Zoletil 50; Virbac Laboratories, Carros, France). Blood samples were collected with a disposable plastic syringe into heparinized tubes. Plasma was obtained by centrifugation at 2,493 ×g for 10 min. Plasma triglyceride (TG) concentration was measured enzymatically using the glycerophosphate oxidase assay. Plasma total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), very low-density lipoprotein cholesterol (VLDL-C), total lipid (TL), and PL levels were measured enzymatically by the cholesterol oxidase assay [9] using commercially available assay kits (Sekisui Medical Co., Ltd., Tokyo, Japan). Plasma albumin, total bilirubin, direct bilirubin, creatinine, blood urea nitrogen, uric acid, glucose, total protein, and electrolytes, including calcium, sodium, potassium, chloride, inorganic phosphate, and magnesium were measured by standard methods using an auto analyzer (Hitachi 7600-210; Hitachi, Tokyo, Japan).
VLDL-C was calculated as follows:
VLDL-C = [TC - (HDL-C + LDL-C)]
Plasma enzyme analysis
Plasma transaminases, glutamate pyruvate transaminase (GPT), and glutamate oxaloacetate transaminase (GOT) activities were determined using the kinetic method [9]. Plasma alkaline phosphatase (ALP) activity was determined using 4-nitrophenyl phosphate. ALP catalyzes the hydrolysis of 4-nitrophenyl phosphate, forming phosphate and free 4-nitrophenol, which is colorless in dilute acid solutions. But, under alkaline conditions, 4-nitrophenol is converted to the 4-nitrophenoxide ion, which is an intense yellow color. The absorbance of this color compound was measured spectrophotometrically at 420 nm to determine plasma ALP activity.
Fecal TL and TC analysis
Feces were collected for 7 days before and at the end of 42 days, lyophilized, and then milled into powder. TLs were extracted with chloroform/methanol (2 : 1, v/v), according to the method of Folch et al. [10]. One gram of fecal powder was mixed with 10 mL of chloroform and 5 mL of methanol solution and stirred at 150 rpm for 3 days at room temperature. The suspension was filtered through Whatman No. 2 filter paper (Whatman, Maidstone, UK), the methanol was aspirated, and the chloroform was evaporated. The extracted lipids were then weighed. Two mL of H2O was added, and a suspension was created using a bath sonicator. This suspension was used to estimate fecal cholesterol content by an enzymatic method using the cholesterol oxidase assay.
Plasma lipoprotein separation
Plasma lipoprotein fractions were determined by agarose gel electrophoresis [11]. Three lipoprotein fractions were detected by electrophoresis, which will henceforth be referred to as β-lipoprotein (LDL), pre-β-lipoprotein (VLDL), and α-lipoprotein (HDL). Sample application (2 µL), electrophoresis (80 V, 30 min), staining (Fat Red 7B), drying, and densitometric scanning (525 nm) were performed automatically by the Helena TITAN GEL lipoprotein electrophoresis system (Helena Laboratories, Beaumont, TX, USA). After electrophoresis, lipoprotein fractions were visualized with enzymatic staining reagents. The visualized gel plate was scanned on a densitometer, and the lipoprotein scanning patterns were identified using analytical software (electrophoresis data bank, K.K. Helena Laboratories, Saitama, Japan). The scanned patterns were divided into lipoprotein fractions using the nadirs of the lipoprotein sequential curve. Lipoprotein levels were estimated from the area percentages and total concentrations.
Histological analysis of liver
Liver tissues were rapidly dissected, fixed in liquid nitrogen and 10% formalin solution, and stored until use at -80℃. A representative part of the frozen tissues was processed with a cryo microtome (Cryotome FSE Cryostat; Thermo Electron Corp., Cambridge, MA, USA) using 5-µm thick section and stained with oil red-O [12]. A representative part of the formalin fixed liver tissues was processed for 4-µm thick paraffin embedded sections using a microtome (Microtome HM 450; Thermo Electron Crop.) and then stained with hematoxylin and eosin. Both stained tissue samples were then examined and photographed under a light microscope to assess the presence of lipid. Digital images were obtained using an Olympus BX51 microscope equipped with a Camedia C3040ZOOM digital camera (Olympus America Inc., Melville, NY, USA). All images were taken under ×40 magnification.
Statistical analysis
Results are expressed as means ± SDs. Intergroup differences were analyzed by a one-way analysis of variance followed by post-hoc tests. We used SPSS ver. 11.5 (SPSS Inc., Chicago, IL, USA). A p ≤ 0.05 was considered statistically significant.
Results and Discussion
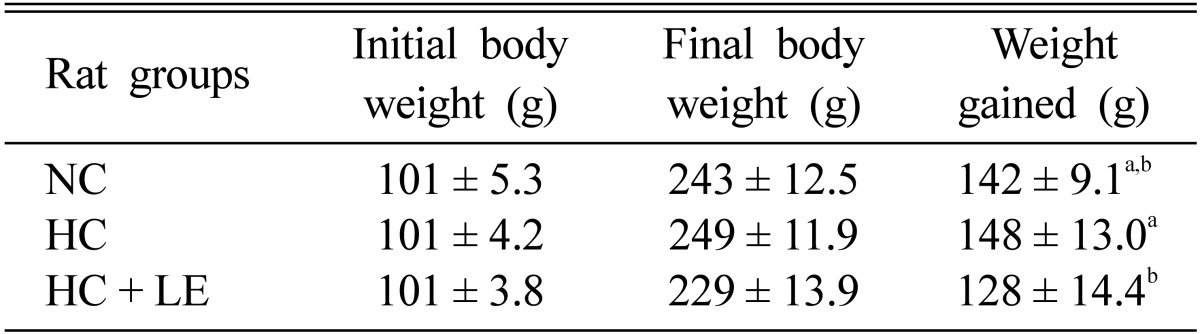
Effects of L. edodes on body weight
Feeding of L. edodes reduced body weight significantly in hypercholesterolemic and normocholesterolemic rats by 13.51% and 9.86%, respectively (Table 2), showing that shiitake mushrooms reduced body weight in both hyper and normocholesterolemic rats. This finding is of special significance because obesity is associated with numerous diseases including diabetes, atherosclerosis, coronary heart disease, and others [13].
Table 2.
Effect of Lentinus edodes on the body weight of hypercholesterolemic rats
Results are means ± SD. Data were analyzed by one-way analysis of variance and then subjected to the LSD post hoc test. Values in the fourth column with different superscripts are significantly different at p ≤ 0.05.
LSD, least significantly different; NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats.
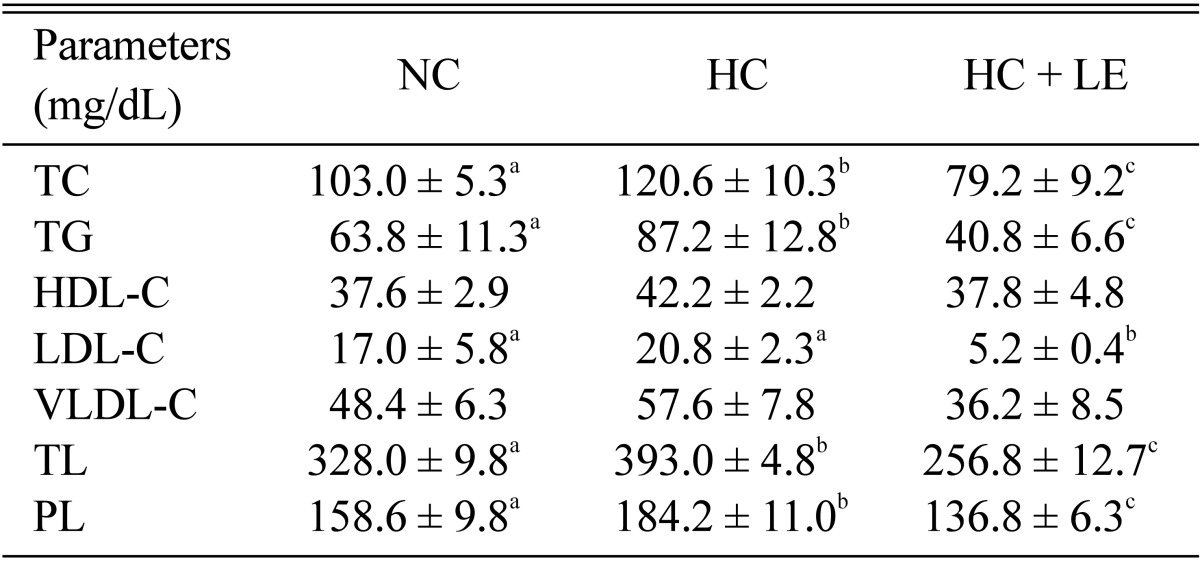
Effects of L. edodes on plasma lipid profiles
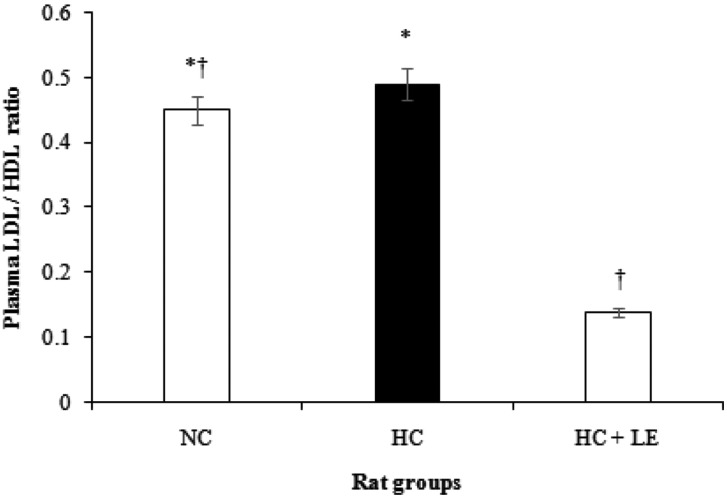
Plasma lipid profile concentrations in NC, HC, and HC + LE rats after feeding L. edodes for 6 wk are presented in Table 3. Plasma TC, TG, HDL-C, LDL-C, VLDL-C, TL, and PL in HC rats increased by 17.09, 36.68, 12.23, 22.35, 19.01, 19.82, and 16.14%, respectively compared with levels in NC rats, whereas these parameters decreased significantly by 34.33, 53.21, 10.43, 75.00, 37.15, 34.66, and 25.73%, respectively in HC + LE rats compared with those in HC rats. The ratio of plasma LDL and HDL is shown in Fig. 1. In HC rats, this ratio increased by 8.89%, compared with that in NC rats, whereas this ratio was reduced significantly by 71.43% in HC + LE compared with that in HC rats. The results show that feeding 5% L. edodes to rats significantly ameliorated the plasma atherogenic lipid profiles in experimentally induced hypercholesterolemic rats.
Table 3.
Effect of Lentinus edodes on plasma lipid profiles in hypercholesterolemic rats
Results are means ± SD. Values in the same row that do not share a common superscript are significantly different at p ≤ 0.05 (one-way analysis of variance followed by an LSD post-hoc comparison).
LSD, least significantly different; NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats; TC, total cholesterol; TG, triglycerides; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; VLDL-C, very low-density lipoprotein cholesterol; TL, total lipid; PL, phospholipids.
Fig. 1.
Effects of Lentinus edodes on the plasma low density lipoprotein (LDL)/high density lipoprotein (HDL) ratio in hypercholesterolemic rats. Results are means ± SD. Different symbols indicate significant differences at p ≤ 0.05. NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats.
Rats are particularly resistant to developing hypercholesterolemia and atherosclerosis [14] and have a strong ability to maintain their plasma cholesterol levels [15, 16]. Therefore, to induce hypercholesterolemia or atherosclerosis in rats, cholesterol feeding is used with other additives, including bile acids and propylthiouracil (an antithyroid drug), which increase intestinal absorption of cholesterol [17]. However, in the present study, adding 1% cholesterol to the basal diet without bile acids and/or antithyroid drugs produced hypercholesterolemia in the rats, because cholesterol feeding itself increases bile acid secretion by approximately three to four-fold in rats [18]. The 34.33% increase in plasma cholesterol in the hypercholesterolemic rats in the present study was comparable with that reported by Bobek et al. [19], who fed rats cholesterol (0.3%) diet with added bile acids (0.5%) and showed a 1.7-fold higher cholesterolemia in cholesterol-fed rats than that in normal rats. In this experiment, feeding 5% L. edodes to hypercholesterolemic rats significantly repressed the increase in plasma cholesterol. The mechanism by which mushrooms reduce plasma lipoprotein levels in hypercholesterolemic rats is not clearly understood. Mushrooms contain the hypocholesterolemic agent mevinolin [20], which may be involved in decreasing HMG-CoA activity [19], the rate-limiting enzyme of cholesterol biosynthesis. Thus, feeding mushrooms may involve suppressing endogenous cholesterol biosynthesis by inhibiting HMG-CoA reductase activity.
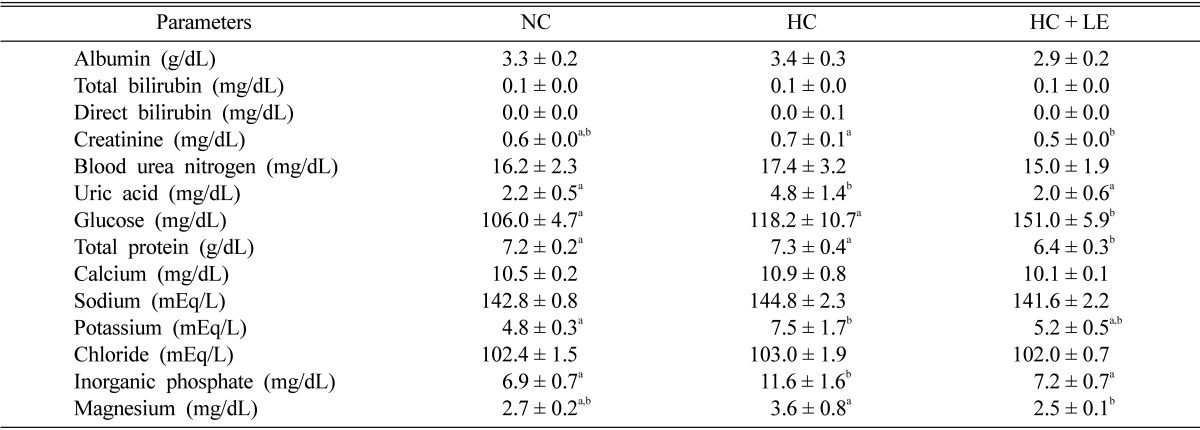
Effects of L. edodes on plasma biochemical and electrolyte function
The results of the plasma biochemical and electrolyte concentrations indicated that creatinine, uric acid, glucose, total protein, potassium, inorganic phosphate, and magnesium decreased significantly in hypercholesterolemic rats by 28.57, 58.33, 18.36, 12.33, 30.66, 37.93, and 30.55%, respectively compared with levels in mushroom-fed rats. Conversely, no significant difference was found for plasma albumin, total bilirubin, direct bilirubin, blood urea nitrogen, calcium, sodium, or chloride levels among the normocholesterolemic, hypercholesterolemic, and mushroom-fed hypercholesterolemic rats (Table 4).
Table 4.
Effect of Lentinus edodes on biochemical and electrolyte function in hypercholesterolemic rats
Results are means ± SD. Values in the same row that do not share a common superscript are significantly different at p ≤ 0.05 (one-way analysis of variance followed by an LSD post-hoc comparison).
LSD, least significantly different; NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats.
The glucose-lowering effect of propionate is associated with gluconeogenesis and the regulation of serum lipid levels [21]. A reduction in plasma potassium, sodium, and chloride concentration is one of the mechanisms of action of antihypertensive drugs, particularly diuretics [22]. Many diuretics act by diminishing sodium chloride reabsorption at different sites in the nephrons, thereby increasing urinary sodium chloride and water losses, and consequently leading to decreased plasma levels of these electrolytes. Antonov et al. [23] reported that plasma electrolyte contents increase significantly in hypertensive rats. Impaired function of Na, K-ATPase, Na-H antiport, which is typical of arterial hypertension, may promote an increase in plasma electrolytes.
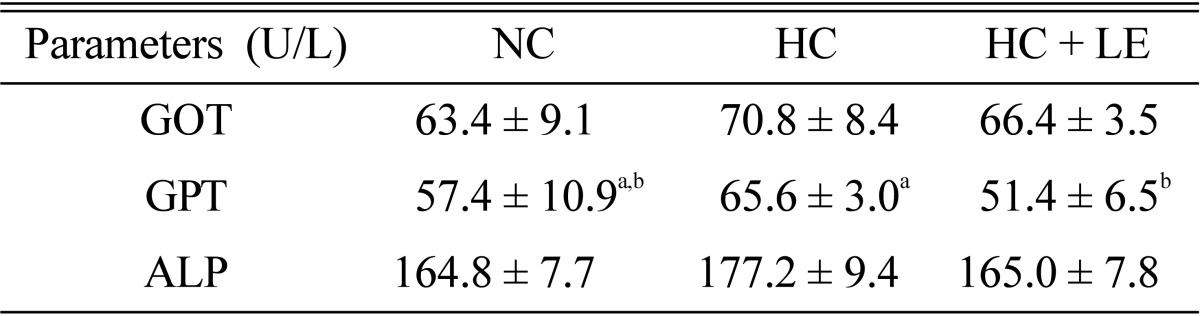
Effects of L. edodes on plasma enzyme profiles
Lower plasma GOT, GPT, and ALP concentrations were observed in shiitake mushroom-fed hypercholesterolemic rats than those in normocholesterolemic rats (Table 5). No significant difference was observed in the activities of plasma GOT and ALP in the NC, HC, or HC + LE rats groups, whereas plasma GPT activity was significantly higher in HC than that in NC rats. Nevertheless, 5% shiitake mushroom-fed hypercholesterolemic rats revealed decreased plasma GOT, GPT, and ALP activity by 6.21, 21.64, and 6.84%, respectively.
Table 5.
Effect of Lentinus edodes on plasma enzyme profiles related to liver and kidney function in hypercholesterolemic rats
Results are means ± SD. Values in the same row that do not share a common superscript are significantly different at p ≤ 0.05 (one-way analysis of variance followed by an LSD post-hoc comparison).
LSD, least significantly different; NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats; GOT, glutamate oxaloacetate transaminase; GPT, glutamate pyruvate transaminase; ALP, alkaline phosphatase.
Due to the increasing frequency of antihyperlipidemic drug use and their common side effects, there is a need to identify natural products with few or no side effects. Thus, investigation continues to identify highly effective natural ingredients from foods, such as mushrooms, which can decrease hyperlipidemia [19, 24]. Previous studies have shown that GOT and GPT are typically elevated following cellular damage as a result of enzyme leakage from the cells into blood [25]. Therefore, the increased enzyme activities resulting from mushroom treatment may prevent oxidative damage by detoxifying reactive oxygen species; thus, reducing hyperlipidemia.
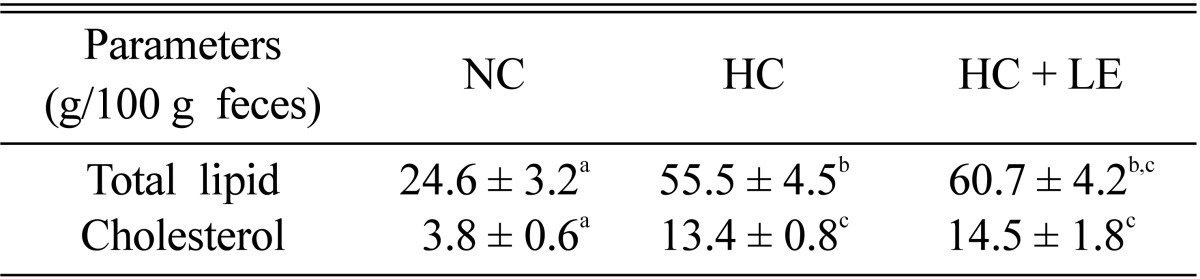
Effects of L. edodes on fecal TLs and cholesterol
Fecal TLs and cholesterol of the 5% L. edodes-fed hypercholesterolemic rats increased significantly by 2.5 and 3.8-fold, respectively, compared with those in NC rats (Table 6). Thus, decreased plasma cholesterol may have attributed to such a mechanism.
Table 6.
Effects of Lentinus edodes on fecal total lipids and cholesterol
Results are means ± SD. Values in the same row that do not share a common superscript are significantly different at p ≤ 0.05 (one-way analysis of variance followed by an LSD post-hoc comparison). LSD, least significantly different; NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats.
The higher level of plasma HDL-C indicates that more cholesterol from peripheral tissues was returning to the liver for catabolism and subsequent excretion. Plasma VLDL-C and TG contents in HC + LE rats were lower compared with those in hypercholesterolemic rats. VLDLC is the major transport vehicle for TG from the liver to extrahepatic tissues, whereas LDL-C is not secreted as such in the liver but seems to be formed from VLDL-C after partial removal of TG by lipoprotein lipase [26]. LDL-C became the prime carrier for cholesterol after feeding cholesterol to the rats, leading to decreased VLDL-C and HDL-C content in HC + LE rats.
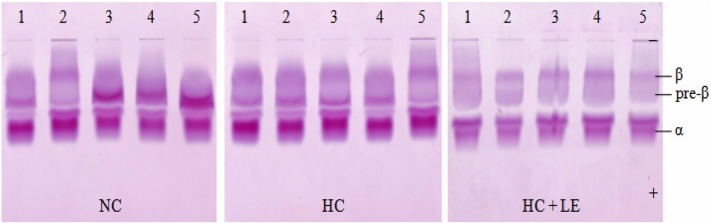
Effects of L. edodes on the plasma lipoprotein fraction by agarose gel electrophoresis
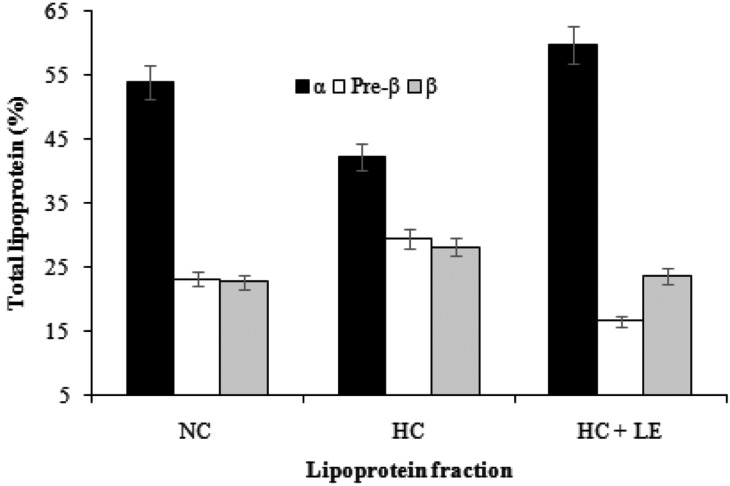
The α-lipoprotein band was the fast-moving fraction and was located nearest the anode. The β-lipoprotein band was usually the most prominent fraction and was near the origin, migrating only slightly toward the anode from the point of application. The pre-β lipoprotein band migrated between α- and β-lipoprotein (Fig. 2). The effects of feeding L. edodes on the plasma lipoprotein fraction are presented in Fig. 3. The results indicated no significant difference in the lipoprotein fractions between normocholesterolemic and mushroom-fed hypercholesterolemic rats compared to those in hypercholesterolemic rats. The results revealed that feeding 5% shiitake mushrooms significantly reduced plasma β and pre-β lipoprotein, whereas it increased α-lipoprotein.
Fig. 2.
Separation of plasma lipoproteins by agarose gel electrophoresis. Lanes 1~5 represent the plasma lipoprotein fraction of five different rats from each group. NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats; β, β-lipoprotein; pre-β, pre-β-lipoprotein; α, α-lipoprotein.
Fig. 3.
Effects of Lentinus edodes on the plasma lipoprotein fraction following agarose gel electrophoresis. Results are means ± SD. NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats; α, α-lipoprotein; pre-β, pre-β-lipoprotein; β, β-lipoprotein.
The hypocholesterolemic effect of mushrooms is mediated by the interplay of a complex mixture of substances [27]. Water-soluble gel-forming components of the fiber substance β-1,3-D-glucan with a low degree of polymerization and forming 15~20% of dry matter interact with bile acids and affect micelle formation. Such substances might be interfering with the absorption of cholesterol in this manner.
Effects of L. edodes on rat liver histopathology
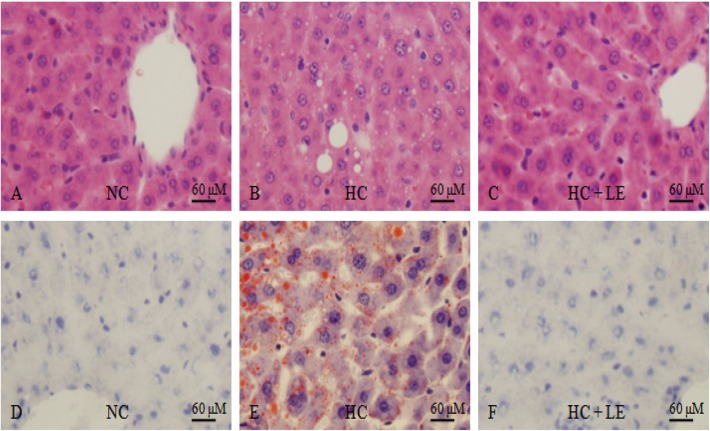
The effect of L. edodes on hepatocyte cells of HC rats is presented in Fig. 4. Liver tissues were stained with hematoxylin-eosin and oil red-O. The hepatic cords were typically arranged and located in liver tissue near the central vein in the NC, HC, and HC + LE groups. Lipid droplets were observed only in the liver tissue of HC rats, which was attributed to lipid accumulation in the hepatocyte cell cytoplasm.
Fig. 4.
Effects of feeding Lentinus edodes on hepatocyte cells in hypercholesterolemic rats. A~C, Hematoxylin-eosin stained photomicrographs (×40); D~F, photomicrographs of oil red-O stain (×40); NC, normocholesterolemic control rats; HC, hypercholesterolemic rats; HC + LE, L. edodes-fed hypercholesterolemic rats.
Oxidized LDL induces the expression of scavenger receptors on the macrophage surface. These scavenger receptors promote the accumulation of modified lipoproteins, forming an early atheroma. The histological results indicated that the liver tissues of the 5% HC + LE rats were almost similar to those of NC rats and that the hepatic biosynthesis of cholesterol was suppressed, which may have been due to a reduction in HMG-CoA activity [28]. Hyperlipidemia is the leading risk factor for atherosclerosis, but the atherosclerotic pathological process could be slowed or reversed by reducing serum LDL, TGs, and PLs and increasing serum HDL. Several studies have demonstrated a protective effect of HDL in atherosclerosis and cardiovascular disease, whereas high levels of LDL constitute a risk factor. Excess LDL in the blood is deposited on the blood vessel walls and becomes a major component of atherosclerotic plaque lesions, whereas HDL facilitates translocation of cholesterol from peripheral tissues, such as arterial walls, to the liver for catabolism [29]. Bobek and Galbavý [30] observed that oyster mushrooms prevent the formation of atheromatous plaques and reduce the incidence and extent of atherosclerotic lesions in the aorta and coronary arteries as well as focal fibrosis in the myocardium of rabbits.
Acknowledgements
This work was supported by research grant from the Korea National Research Resource Center Program (2011-0000525) through National Research Foundation of Korea (NRF) for Culture Collection and DNA Bank of Mushroom (CCDBM), University of Incheon.
References
- 1.Wasser SP, Weis AL. Medicinal properties of substances occurring in higher Basidiomycetes mushrooms: current perspectives. Int J Med Mushrooms. 1999;1:31–62. [PubMed] [Google Scholar]
- 2.Chibata I, Okumura K, Takeyama S, Kotera K. Lentinacin: a new hypocholesterolemic substance in Lentinus edodes. Experientia. 1969;25:1237–1238. doi: 10.1007/BF01897467. [DOI] [PubMed] [Google Scholar]
- 3.Sugiyama K, Akachi T, Yamakawa A. Eritadenine-induced alteration of hepatic phospholipid metabolism in relation to its hypocholesterolemic action in rats. J Nutr Biochem. 1995;6:80–87. doi: 10.1093/jn/125.8.2134. [DOI] [PubMed] [Google Scholar]
- 4.Steinberg D, Parthasarathy S, Carew TE, Khoo JC, Witztum JL. Beyond cholesterol: modifications of low-density lipoprotein that increase its atherogenicity. N Engl J Med. 1989;320:915–924. doi: 10.1056/NEJM198904063201407. [DOI] [PubMed] [Google Scholar]
- 5.Alam N, Amin R, Khan A, Ara I, Shim MJ, Lee MW, Lee TS. Nutritional analysis of cultivated mushrooms in Bangladesh: Pleurotus ostreatus, Pleurotus sajor-caju, Pleurotus florida and Calocybe indica. Mycobiology. 2008;36:228–232. doi: 10.4489/MYCO.2008.36.4.228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Alam N, Yoon KN, Lee KR, Shin PG, Cheong JC, Yoo YB, Shim MJ, Lee MW, Lee UY, Lee TS. Antioxidant activities and tyrosinase inhibitory effects of different extracts from Pleurotus ostreatus fruiting bodies. Mycobiology. 2010;38:295–301. doi: 10.4489/MYCO.2010.38.4.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang QY, Jong SC. Medicinal mushrooms in China. Mushroom Sci. 1989;1:631–643. [Google Scholar]
- 8.Kabir Y, Kimura S. Dietary mushrooms reduce blood pressure in spontaneously hypertensive rats (SHR) J Nutr Sci Vitaminol (Tokyo) 1989;35:91–94. doi: 10.3177/jnsv.35.91. [DOI] [PubMed] [Google Scholar]
- 9.Burtis CA, Ashwood ER. Tietz fundamentals of clinical chemistry. New Delhi: Reed Elsevier India Private Ltd.; 2006. [Google Scholar]
- 10.Folch J, Lees M, Sloane Stanley GH. A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem. 1957;226:497–509. [PubMed] [Google Scholar]
- 11.Kido T, Kurata H, Matsumoto A, Tobiyama R, Musha T, Hayashi K, Tamai S, Utsunomiya K, Tajima N, Fidge N, et al. Lipoprotein analysis using agarose gel electrophoresis and differential staining of lipids. J Atheroscler Thromb. 2001;8:7–13. doi: 10.5551/jat1994.8.7. [DOI] [PubMed] [Google Scholar]
- 12.Bayliss HO. Lipids. In: Bancroft JD, Stevens A, editors. Theory and practice of histological techniques. Edinburgh: Churchill Livingstone; 1990. pp. 215–244. [Google Scholar]
- 13.Alam N, Amin R, Khan A, Ara I, Shim MJ, Lee MW, Lee UY, Lee TS. Comparative effects of oyster mushrooms on lipid profile, liver and kidney function in hypercholesterolemic rats. Mycobiology. 2009;37:37–42. doi: 10.4489/MYCO.2009.37.1.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Andrus SB, Fillios LC, Mann GV, Stare FJ. Experimental production of gross atherosclerosis in the rat. J Exp Med. 1956;104:539–554. doi: 10.1084/jem.104.4.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fujioka T, Nara F, Tsujita Y, Fukushige J, Fukami M, Kuroda M. The mechanism of lack of hypocholesterolemic effects of pravastatin sodium, a 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitor, in rats. Biochim Biophys Acta. 1995;1254:7–12. doi: 10.1016/0005-2760(94)00154-q. [DOI] [PubMed] [Google Scholar]
- 16.Roach PD, Balasubramaniam S, Hirata F, Abbey M, Szanto A, Simons LA, Nestel PJ. The low-density lipoprotein receptor and cholesterol synthesis are affected differently by dietary cholesterol in the rat. Biochim Biophys Acta. 1993;1170:165–172. doi: 10.1016/0005-2760(93)90067-j. [DOI] [PubMed] [Google Scholar]
- 17.Dolphin PJ, Forsyth SJ. Nascent hepatic lipoproteins in hypothyroid rats. J Lipid Res. 1983;24:541–551. [PubMed] [Google Scholar]
- 18.Uchida K, Satoh T, Chikai T, Takase H, Nomura Y, Nakao H, Takeuchi N. Influence of cholesterol feeding on bile acid metabolism in young and aged germ-free rats. Jpn J Pharmacol. 1996;71:113–118. doi: 10.1254/jjp.71.113. [DOI] [PubMed] [Google Scholar]
- 19.Bobek P, Hromadová M, Ozdín L. Oyster mushroom (Pleurotus ostreatus) reduces the activity of 3-hydroxy-3-methylglutaryl CoA reductase in rat liver microsomes. Experientia. 1995;51:589–591. doi: 10.1007/BF02128749. [DOI] [PubMed] [Google Scholar]
- 20.Gunde-Cimerman N, Plemenitas A, Cimerman A. Pleurotus fungi produce mevinolin an inhibitor of HMG CoA reductase. FEMS Microbiol Lett. 1993;113:333–337. doi: 10.1111/j.1574-6968.1993.tb06536.x. [DOI] [PubMed] [Google Scholar]
- 21.Yang BK, Jung YS, Song CH. Hypoglycemic effects of Ganoderma applanatum and Collybia confluens exo-polymers in streptozotocin-induced diabetic rats. Phytother Res. 2007;21:1066–1069. doi: 10.1002/ptr.2214. [DOI] [PubMed] [Google Scholar]
- 22.Jude IC, Catherine IC, Frank OC. Effect of aqueous extract of Tridax procumbens Linn on plasma electrolytes of saltloaded rats. Pak J Nutr. 2010;9:103–105. [Google Scholar]
- 23.Antonov AR, Efremov AV, Letyagina VV, Nacharov YV, Markel AL, Yakobson GS. Plasma and lymph electrolyte and endocrine parameters in rats with genetically-determined arterial hypertension. Bull Exp Biol Med. 1997;124:652–654. [Google Scholar]
- 24.Alarcón J, Águila S, Arancibia-Avila P, Fuentes O, Zamorano-Ponce E, Hernández M. Production and purification of statins from Pleurotus ostreatus (Basidiomycetes) strains. Z Naturforsch C. 2003;58:62–64. doi: 10.1515/znc-2003-1-211. [DOI] [PubMed] [Google Scholar]
- 25.Noori S, Zafar H, Mahboob T. Biochemical effectiveness of cocoa powder on electrolytes homeostasis, liver and cardiac specific enzymes and renal function. Pak J Nutr. 2009;8:882–886. [Google Scholar]
- 26.Mayes PA. Metabolism of lipids. In: Harper HA, Rodwell VW, Mayes PA, editors. Reviews of physiological chemistry. Los Altos: Lange Medical Publications; 1997. pp. 280–321. [Google Scholar]
- 27.Bobek P, Ozdín L, Kuniak L. Effect of oyster mushroom (Pleurotus ostreatus) and its ethanolic extract in diet on absorption and turnover of cholesterol in hypercholesterolemic rat. Nahrung. 1996;40:222–224. doi: 10.1002/food.19960400413. [DOI] [PubMed] [Google Scholar]
- 28.Keim NL, Marlett JA, Amundson CH, Hagemann LD. Comparison of rat hepatic cholesterol biosynthesis during skim milk versus whey permeate ingestion. J Dairy Sci. 1982;65:2274–2280. doi: 10.3168/jds.S0022-0302(82)82497-1. [DOI] [PubMed] [Google Scholar]
- 29.Li H, Zhang M, Ma G. Hypolipidemic effect of the polysaccharide from Pholiota nameko. Nutrition. 2010;26:556–562. doi: 10.1016/j.nut.2009.06.009. [DOI] [PubMed] [Google Scholar]
- 30.Bobek P, Galbavý S. Hypocholesterolemic and antiatherogenic effect of oyster mushroom (Pleurotus ostreatus) in rabbits. Nahrung. 1999;43:339–342. doi: 10.1002/(SICI)1521-3803(19991001)43:5<339::AID-FOOD339>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]