Abstract
The concise and enantioselective total syntheses of (−)-trigonoliimines A, B, and C are described. Our unified strategy to all three natural products is based on asymmetric oxidation and reorganization of a single bistryptamine, a sequence of transformations with possible biogenetic relevance. We revise the absolute stereochemistry of (−)-trigonoliimines A, B, and C.
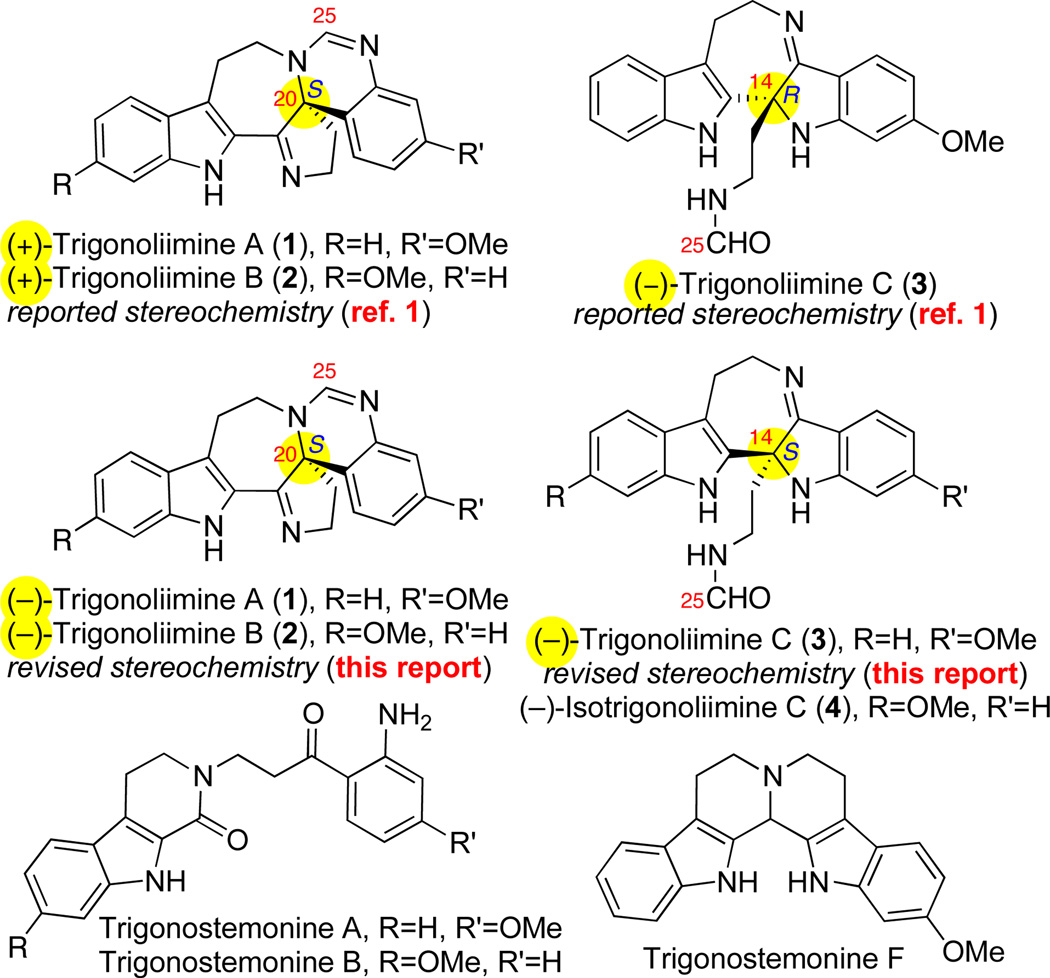
In 2010, Hao and co-workers reported the isolation of structurally fascinating (+)-trigonoliimines A (1), and B (2) along with (−)-trigonoliimine C (3) from the leaves of Trigonostemon lii Y. T. Chang collected in Yunnan province of China.1 They also examined trigonoliimines A (1) and C (3) in an anti-HIV assay where 1 was found to exhibit modest activity (EC50 = 0.95 µg/mL, TI = 7.9).1 Intrigued by their unique molecular structure and inspired by a plausible hypothesis for their biogenesis, we set out to develop a unified synthetic strategy based on oxidation and reorganization of a single heterodimeric bistryptamine to access all trigonoliimine alkaloids. Herein, we report the first total synthesis of (−)-trigonoliimine A–C (1–3) and the related derivate (−)-isotrigonoliimine C (4), enabling our revision of the absolute stereochemistry of alkaloids (−)-1–3 (Figure 1).
Figure 1.
Representative trigonostemon alkaloids including the revised absolute stereochemistry of trigonoliimines A–C (1–3).
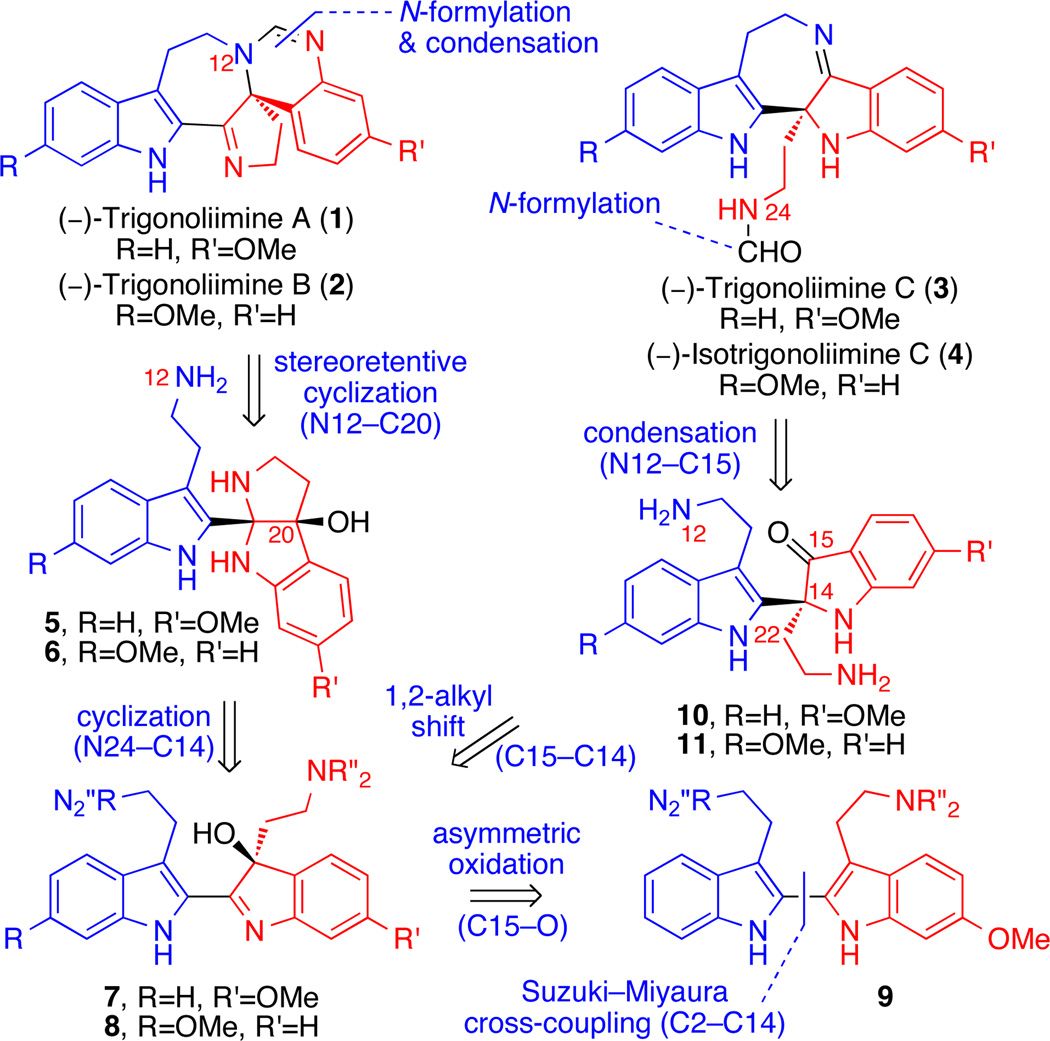
Our unified strategy for the enantioselective total synthesis of all known trigonoliimines was based on the hypothesis that bistryptamine heterodimer 9 (Scheme 1) could serve as a common biosynthetic precursor to these alkaloids. While the chemoselectivity of the oxidation of bisindole 9 was envisioned to determine the ratio of regioisomeric hydroxyindolenines 7 and 8, the stereoselectivity of the transformation was thought to provide a platform for the asymmetric synthesis of the trigonoliimines. We postulated that hydroxyindolenines 7 and 8 would serve as the branching point for divergent synthesis of the two distinct structural motifs found in trigonoliimine alkaloids (Scheme 1). Trigonoliimines A (1) and B (2) were expected to be accessed via a stereoretentive cyclization of N12 onto the C20 carbinol function of precursors 5 and 6, respectively, followed by N-formylation and condensation (Scheme 1). The requisite cis-fused aminals 5 and 6 could result from intramolecular cyclization of hydroxyindolenines 7 and 8, respectively. Alternatively, a stereospecific Wagner–Meerwein type rearrangement2,3 of intermediates 7 and 8 was envisioned to provide the indoxyls 10 and 11, respectively.4,5 Intramolecular condensation of the N12 amine and the C15 ketone of indoxyls 10 and 11 in addition to N24 formylation was expected to provide trigonoliimine C (3) and isotrigonoliimine C (4). Thus, the enantioselective synthesis6,7 of both regioisomeric hydroxyindolenines 7 and 8 was sought to address the asymmetric synthesis of alkaloids 1−4.
Scheme 1.
Retrosynthetic Analysis of (−)-Trigonoliimines A–C (1–3) and Isotrigonoliimine C (4).
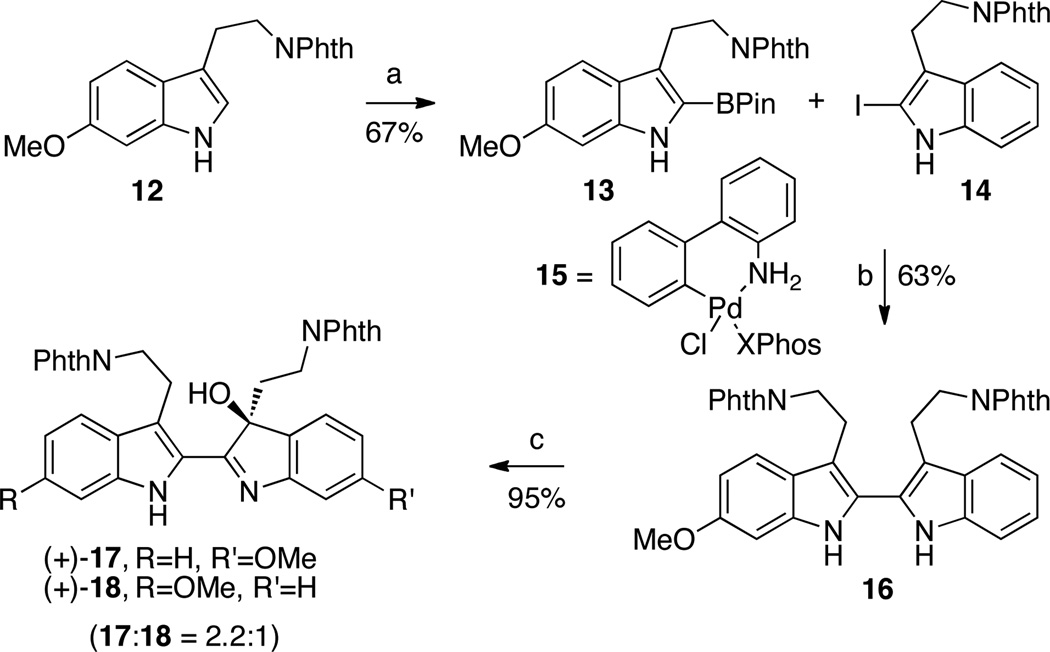
Our synthesis of the (−)-trigonoliimine alkaloids commenced with an iridium catalyzed8 C2-borylation of the 6-methoxy-tryptamine derivative 12.9 We observed that using dichloromethane as solvent at 23 °C minimized the undesired borylation of the phthalimide substructure (Scheme 2). Access to bisindole 16 was possible via a Suzuki–Miyaura cross-coupling10 of boronate 13 and 2-iodo-tryptamine 147,11 using a variety of palladium sources in the presence of XPhos12 and potassium phosphate at elevated temperatures, albeit in low and variable yields (7–44%). Alternatively, the use of Buchwald’s aminobiphenyl precatalyst 1513 enabled a robust cross-coupling of pinacol boronate 13 and iodide 14 at 23 °C to give 16 in 31% yield. After an extensive screening of bases and additives, we noticed that the presence of both a halophile14 and proper base was critical for the overall efficiency of this transformation. We discovered that the use of silver phosphate (2.0 equiv) and the precatalyst 15 optimally promoted this cross-coupling reaction, affording the desired bistryptamine 16 in 63% yield (Scheme 2).
Scheme 2.
Synthesis of Hydroxyindolenines (+)-17 and (+)-18.a
a Conditions: (a) HBPin, [Ir(OMe)(cod)]2 (5 mol%), 4,4'-di-tBu-2,2'-bipyridine, CH2Cl2, 23 °C. (b) Ag3PO4, 15 (20 mol%), PhCH3, H2O, 23 °C. (c) (+)-((8,8-dichlorocamphoryl)sulfonyl)oxaziridine, CH2Cl2, −35→23 °C.
The bistryptamine 16 was found to be sensitive to oxidation under a variety of conditions. In fact, simple exposure of bistryptamine 16 to air over 12 days resulted in autoxidation to (±)-hydroxyindolenines 17 and 18 (17:18 = 1.5:1) in 27% yield along with recovered 16 (65%). Interestingly, the presence of regioisomeric pairs is commonly observed in the trigonostemon alkaloids family15 (Figure 1) and the major autoxidation product (oxidation of 6-methoxy-indole substructure) is consistent with the major isolated trigonoliimines A (1) and C (3).1 Given the rapid oxidation of bistryptamine 16, and based on observations on stereoselective oxidation of related derivatives,4,5,7,16 we focused our attention on the use of oxaziridines. Under optimal conditions, treatment of bistryptamine 16 with readily available (+)-((8,8-dichlorocamphoryl)sulfonyl)oxaziridine (Davis’ oxaziridine)17 provided hydroxyindolenines (+)-17 and (+)-18 (17:18 = 2.2:1, Scheme 2) in 95% yield and with an outstanding level of enantioselection for both isomers (96% ee, vide infra).7,16 This solution provided efficient access to precursors for the enantioselective synthesis of alkaloids 1−4. While the isomeric hydroxyindolenines (+)-17 and (+)-18 were separated for complete characterization and independent derivatization, separation of more advanced intermediates en route to alkaloids 1–4 proved most practical.
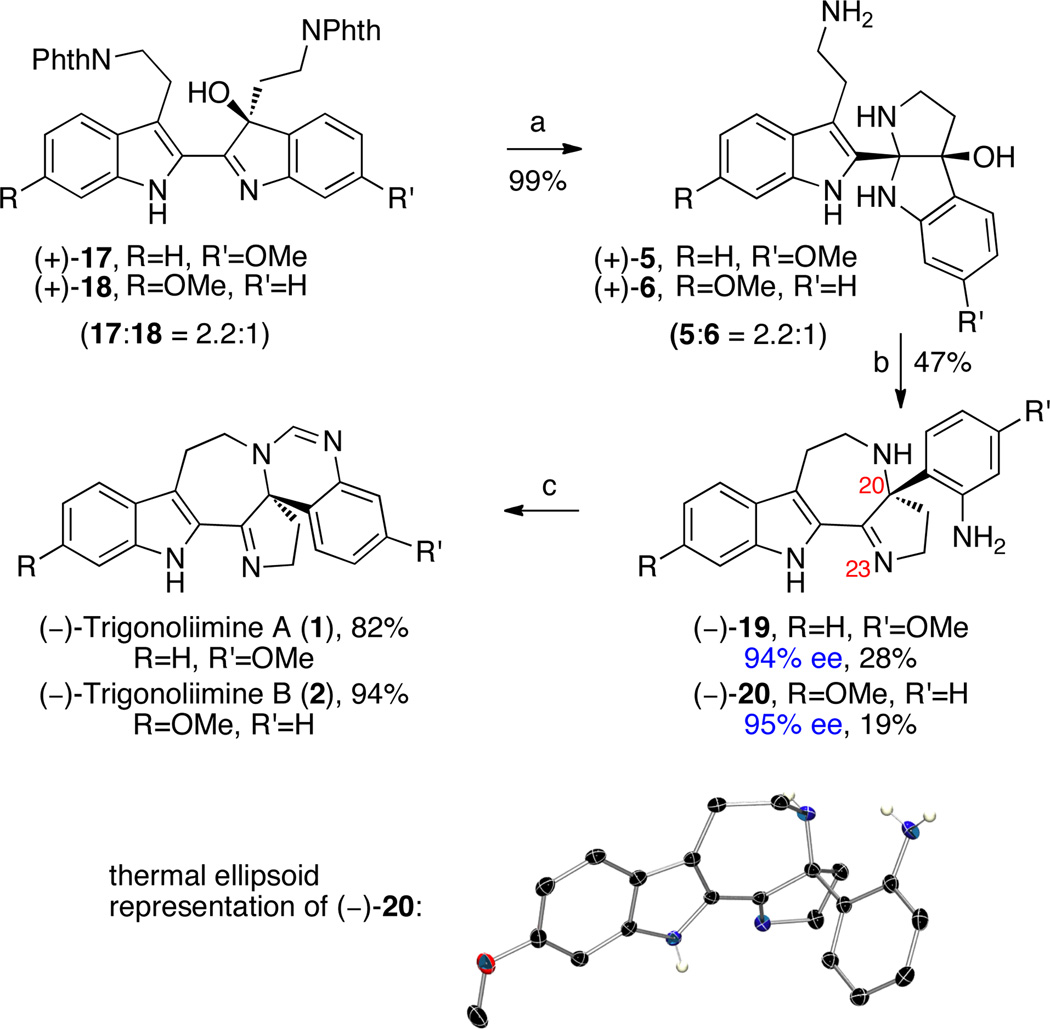
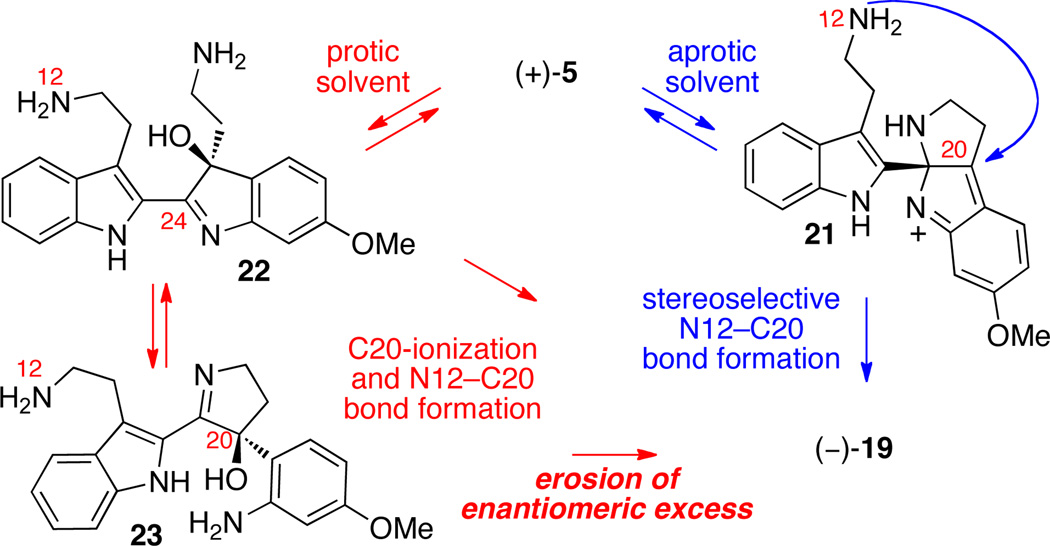
Unveiling the two amino groups of hydroxyindolenines (+)-17 and (+)-18 spontaneously provided the desired cis-fused aminals (+)-5 and (+)-6 (5:6 = 2.2:1, Scheme 3), our proposed precursors for trigonoliimines A (1) and B (2), respectively, in 99% yield. Aminals (+)-5 and (+)-6 were separable at this stage, allowing their independent chemical examination and characterization. Interestingly, heating a solution of aminal (+)-5 in trifluoroethanol (TFE) at 105 °C provided the desired azepane (−)-19 in 34% yield with significant drop in enantiomeric excess (15% ee). On the other hand, aminal (+)-6 led to almost complete decomposition under identical reaction conditions, highlighting the different chemical reactivity of the regioisomeric series of intermediates in our studies.
Scheme 3.
Total Synthesis of (−)-Trigonoliimines A (1) and B (2).a
a Conditions: (a) NH2NH2•H2O, MeOH, 80 °C. (b) Martin Sulfurane, CH2Cl2, −78°C. (c) CH(OiPr)3, PPTS, CH2Cl2, 23 °C.
While 1H NMR analysis of aminals (+)-5 and (+)-6 in deuterated chloroform were consistent with cis-fused pentacycles depicted in Scheme 3, the analysis of the same compounds in deuterated methanol revealed the presence of multiple species consistent with reversible formation of aminal and imine isomers (Scheme 4). We reasoned that the transmutation of (+)-5 to (−)-19, as described above, likely affords the product with greatly diminished optical activity due to a low level of stereoselection in N12–C20 bond construction upon ionization of carbinol 23 at C20 (Scheme 4) or upon formation of a solvent/amine adduct of imine 22. Gratifyingly, treatment of a solution of aminals (+)-5 and (+)-6 (5:6 = 2.2:1) in dichloromethane with the Martin sulfurane reagent18 at −78 °C provided the desired azepanes (−)-19 and (−)-20 in 47% combined yield (28% and 19% yield, respectively, after chromatographic separation). Importantly, azepanes (−)-19 and (−)-20 were obtained with minimal erosion of enantiomeric excess (94% ee and 95% ee, respectively). The X-ray crystal structure analysis of pentacycle (−)-20 (Scheme 3), the direct precursor for (−)-trigonoliimine B (2), unambiguously confirmed the molecular structure and coherently (vide infra) assigned the S-configuration at C20. Using optimal conditions, sequential treatment of pentacycle (−)-19 with pyridinium p-toluenesulfonate (PPTS) and triisopropyl orthoformate in dichloromethane afforded (−)-trigonoliimine A (1) in 82% yield {[α]D24 = −294 (c 0.24, CHCl3)} (Scheme 3). Under identical reaction conditions, the pentacycle (−)-20 was converted to (−)-trigonoliimine B (2) in 94% yield {[α]D24 = −352 (c 0.32, CHCl3)}. All 1H and 13C NMR data for our synthetic (−)-trigonoliimines A (1) and B (2) matched those provided in the isolation report,1 confirming the molecular structure of these alkaloids.
Scheme 4.
Possible competing pathways in conversion of amino alcohol (−)-5 to pentacycle (−)-19.
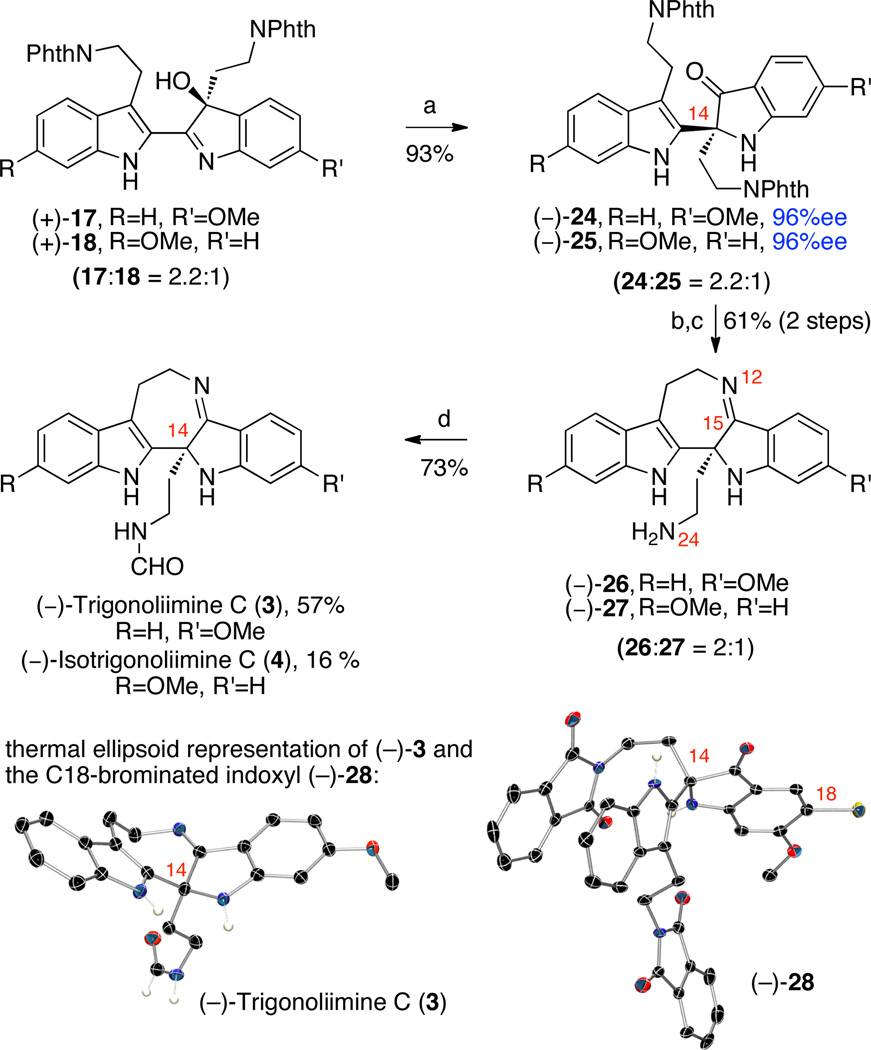
We next aimed to access (−)-trigonoliimine C (3) and (−)- isotrigonoliimine C (4) from the same versatile hydroxyindolenines described above via a divergent synthetic path employing a Wagner–Meerwein type 1,2-alkyl rearrangement.2,3 Oxidation and rearrangement of 2,3-disubstituted indole has served as an efficient strategy to access indoxyl substructures and has been applied in elegant total syntheses of various alkaloids.4 Our group has also utilized oxidation and rearrangement of 2,3-disubstituted indoles as a highly stereoselective entry to oxindole intermediates.5 We observed that exposure of hydroxyindolenines (+)-17 and (+)-18 to various Lewis acids gave the desired indoxyls (−)-24 and (−)-25 along with undesired oxindole byproducts. For example, in the presence of lanthanum trifluoromethane-sulfonate in toluene at 80 °C, hydroxyindolenines (+)-17 and (+)-18 (17:18 = 2.2:1) afforded the undesired oxindoles in 34% yield along with the desired indoxyls in 56% yield.19 The choice of solvent with this rearrangement strongly influenced the ratio of indoxyl to oxindole.5a After significant experimentation, we discovered that heating a solution of hydroxyindolenines (+)-17 and (+)-18 (17:18 = 2.2:1) in trifluoroethanol at 102 °C for 24.5 h resulted in selective formation of the corresponding indoxyls (−)-24 and (−)-25 (24:25 = 2.2:1) in 93% combined yield (Scheme 5). The masking of the two amino groups in the form of phthalimides during this rearrangement was critical in the overall efficiency and selectivity for the formation of the desired products. Separation and independent analysis of indoxyls (−)-24 and (−)-25 revealed a high level of enantioselection (96% ee) in the synthesis of the corresponding hydroxylindolenines (+)-17 and (+)-18. The absolute stereochemistry of indoxyl (−)-24 was secured by X-ray crystal structure analysis of the corresponding C18-bromide (−)-2811 (Scheme 5). The high enantiomeric excess of bromide (−)-28 (96% ee) in conjunction with its X-ray crystal data allowed for unequivocal assignment of the S-configuration at C14. While intermediates en route to (−)-trigonoliimine C (3) and (−)-isotrigonoliimine C (4) were separated for characterization and independent derivatization, delayed separation of isomers proved most practical similar to the case of (−)-trigonoliimines A (1) and B (2). Unraveling the two amino groups of indoxyls (−)-24 and (−)-25, followed by condensative cyclization promoted by titanium ethoxide20 as a one-pot two-step procedure provided the cyclic imine (−)-26 and (−)-27 (26:27 = 2:1) in 61% yield. Notably, we did not observe any of the undesired five-membered ring imines corresponding to condensation of the N24 with C15 carbonyl. Treatment of pentacyclic amines (−)-26 and (−)-27 with N-formyl imidazole followed by silica gel chromatographic separation provided (−)-trigonoliimine C (3) {[α]D24 = −147 (c 0.12, CHCl3)} and (−)-isotrigonoliimine C (4) ([α]D24 = −220 (c 0.10, CHCl3)} in 57% and 16% yield, respectively. All 1H and 13C NMR data for our synthetic (−)-trigonoliimines C (3) matched those provided in the isolation report,1 and analysis of the X-ray crystal structure of our synthetic (−)-3 further confirmed the S-configuration at C14. Interestingly, while isotrigonoliimine C (4) has not been isolated from nature at this time, we have recognized the pentacyclic amine (−)-27 as the most solvolytically sensitive compound amongst those discussed in this report.
Scheme 5.
Total Synthesis of (−)-Trigonoliimine C (3) and (−)-Isotrigonoliimine C (4).a
aConditions: (a) TFE, 102 °C. (b) NH2NH2•H2O, MeOH, 80 °C. (c) Ti(OEt)4, THF, 42 °C. (d) N-formyl imidazole, THF, 23 °C.
The magnitude and sign of specific rotation of our synthetic trigonoliimines in conjunction with our X-ray crystal structure data provide valuable information regarding the stereochemistry of these alkaloids. Interestingly, all of our synthetic (−)-trigonoliimines A–C (1–3) showed a significantly larger magnitude of specific rotations compared to those reported for the naturally isolated samples (Table 1). Importantly, the enantiomeric excess21 of our samples has been quantified by HPLC analysis of enantiomerically enriched samples of several intermediates against readily available racemic samples from our exploratory studies in this area. Additionally, our synthetic trigonoliimines A–C (1–3) derived from hydroxyindolenines (+)-17 and (+)-18 exhibit a negative sign in their specific rotation. However, naturally occurring trigonoliimines A (1) and B (2) were reported to have a positive sign in their specific rotations whereas trigonoliimine C (3) was reported to have a negative sign in its specific rotation. Furthermore, our three X-ray structures of highly enantiomerically enriched compounds (Schemes 3 and 5) provide support for the need to revise the absolute stereochemical assignment of all trigonoliimines (Figure 1). While the absolute stereochemistry of our synthetic (−)-trigonoliimines A–C (1–3) are unequivocally assigned through our studies, given the reported optical rotation values for the natural samples of 1–3, we raise the possibility that either natural trigonoliimines A–C (1–3) were isolated with a low level of enantiomeric excess or the optical rotation values for the natural samples need to be revised.
Table 1.
Specific Rotation Values of Natural1 and Synthetic Trigonoliimines A–C (1–3).
| Entry | Alkaloids | Natural ([α]10D) | Synthetic ([α]24D)a |
|---|---|---|---|
| 1 | Trigonoliimine A | +13.3 (c 0.3, CHCl3) | −294 (c 0.24, CHCl3, 94% ee) |
| 2 | Trigonoliimine B | +5.0 (c 0.5, CHCl3) | −352 (c 0.32, CHCl3, 95% ee) |
| 3 | Trigonoliimine C | −4.8 (c 0.45, CHCl3) | −147 (c 0.12, CHCl3, 96% ee) |
% ee of the key precursor for the corresponding synthetic trigonoliimine.
We have developed the first total syntheses of all trigonoliimine alkaloids inspired by a unified biosynthetic hypothesis22 for oxidation and reorganization of a single bistryptamine precursor (Scheme 1). Our concise enantioselective syntheses of (−)-trigonoliimines A (1) and B (2) are seven steps from commercially available material and employ a critical stereoretentive condensative cyclization of hydroxyindolenines (+)-17 and (+)-18, respectively. Our succinct enantioselective syntheses of (−)-trigonoliimines C (3) and (−)-isotrigonoliimine C (4) are eight steps from commercially available material and draw on the application of the venerable Wagner–Meerwein rearrangement of the hydroxyindolenines (+)-17 and (+)-18, respectively. These divergent and stereocontrolled transformations of hydroxyindolenines (+)-17 and (+)-18 were guided by retrobiosynthetic23 analysis of trigonoliimine alkaloids. Rapid access to the key intermediates is enabled by a Suzuki–Miyaura cross-coupling reaction using Buchwald’s precatalyst in conjunction with silver phosphate followed by a highly enantioselective oxidation at the enantiodetermining and branching point of our syntheses. Additionally, our studies allow us to revise the absolute stereochemistry of alkaloids (−)-1–3.
Acknowledgement
We are grateful for generous financial support by NIH-NIGMS (GM074825). M.M. is a Camille Dreyfus Teacher-Scholar. We thank Mr. Justin Kim and Dr. Peter Müller for X-ray crystal structure analysis of (−)-3, (−)-18 and (−)-24. The X-ray laboratory of MIT Department of Chemistry is supported by NSF CHE-0946721. We thank Amgen and DuPont for additional financial support.
Footnotes
Supporting Information. Experimental procedures, spectroscopic data, copies of 1H and 13C NMR spectra and crystal structure of (−)-3, (−)-20 and (−)-28. This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Tan C-J, Di Y-T, Wang Y-H, Zhang Y, Si Y-K, Zhang Q, Gao S, Hu X-J, Fang X, Li S-F, Hao X-J. Org. Lett. 2010;12:2370–2373. doi: 10.1021/ol100715x. [DOI] [PubMed] [Google Scholar]
- 2.(a) Wagner G, Brickner W. Ber. 1899;32:2302–2325. [Google Scholar]; (b) Meerwein H, Van Emster K, Joussen J. Ber. 1922;55:2500–2528. [Google Scholar]
- 3.(a) Witkop B, Patrick JB. J. Am. Chem. Soc. 1951;73:2188–2195. [Google Scholar]; (b) Zhang X, Foote CS. J. Am. Chem. Soc. 1993;115:8867–8868. [Google Scholar]
- 4.For elegant examples of the application of oxidation and rearrangement of 2,3-disubstituted indoles in complex synthesis see: Finch N, Taylor WI. J. Am. Chem. Soc. 1962;84:3871. Hutchison AJ, Kishi Y. J. Am. Chem. Soc. 1979;101:6786–6788. Williams RM, Glinka T, Kwast E, Coffman H, Stille JK. J. Am. Chem. Soc. 1990;112:808–821. Gueller R, Borschberg H. Helv. Chim. Acta. 1993;76:1847–1862. Stoermer D, Heathcock CH. J. Org. Chem. 1993;58:564–568. Cushing TD, Sanz-Cervera JF, Williams RM. J. Am. Chem. Soc. 1996;118:557–579. Ito M, Clark CW, Mortimore M, Goh JB, Martin SF. J. Am. Chem. Soc. 2001;123:8003. doi: 10.1021/ja010935v. Baran PS, Corey EJ. J. Am. Chem. Soc. 2002;124:7904–7905. doi: 10.1021/ja026663t. Williams RM, Cox RJ. Acc. Chem. Res. 2003;36:127. doi: 10.1021/ar020229e. and references cited therein. Liu Y, McWhorter WW, Jr, Hadden CE. Org. Lett. 2003;5:333–335. doi: 10.1021/ol0202417. Baran PS, Richter JM. J. Am. Chem. Soc. 2005;127:15394–15396. doi: 10.1021/ja056171r. Poriel C, Lachia M, Wilson C, Davies JR, Moody CJ. J. Org. Chem. 2007;72:2978–2987. doi: 10.1021/jo062627r. Miller KA, Tsukamoto S, Williams RM. Nature Chem. 2009;1:63–68. doi: 10.1038/nchem.110.
- 5. Movassaghi M, Schmidt MA, Ashenhurst JA. Org. Lett. 2008;10:4009–4012. doi: 10.1021/ol8015176. (b) For a discussion of a related hypothesis on oxidation and rearrangement of a 2,2'-bistryptamine, see Schmidt MA, Movassaghi M. Synlett. 2008:313–324.
- 6.(a) Hentges SG, Sharpless KB. J. Am. Chem. Soc. 1980;102:4263–4264. [Google Scholar]; (b) Katsuki TK, Sharpless KB. J. Am. Chem. Soc. 1980;102:5974–5976. [Google Scholar]; (c) Zhang W, Loebach JL, Wilson SR, Jacobsen EN. J. Am Chem. Soc. 1990;112:2801–2803. [Google Scholar]; (d) Irie R, Noda K, Ito Y, Katsuki T. Tetrahedron Lett. 1991;32:1055–1058. [Google Scholar]; (e) Tu Y, Wang ZX, Shi Y. J. Am. Chem. Soc. 1996;118:9806–9807. [Google Scholar]; (f) Peris G, Jakobsche CE, Miller SJ. J. Am. Chem. Soc. 2007;129:8710–8711. doi: 10.1021/ja073055a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kolundzic F, Noshi MN, Tjandra M, Movassaghi M, Miller SJ. J. Am. Chem. Soc. 2011;133:9104–9111. doi: 10.1021/ja202706g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.(a) Ishiyama T, Takagi J, Ishida K, Miyaura N, Anastasi NR, Hartwig JF. J. Am. Chem. Soc. 2002;124:390–391. doi: 10.1021/ja0173019. [DOI] [PubMed] [Google Scholar]; (b) Ishiyama T, Takagi J, Hartwig JF, Miyaura N. Angew. Chem. Int. Ed. 2002;41:3056–3058. doi: 10.1002/1521-3773(20020816)41:16<3056::AID-ANIE3056>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]; (c) Boller TM, Murphy JM, Hapke M, Ishiyama T, Miyaura N, Hartwig JF. J. Am. Chem. Soc. 2005;127:14263–14278. doi: 10.1021/ja053433g. [DOI] [PubMed] [Google Scholar]
- 9.Tryptamine 12 could be prepared in one step from commercially available 6-methoxy tryptamine (S2). Alternatively, for the synthesis of S2, see: Woodward RB, Bader FE, Bickel H, Frey AJ, Kierstead RW. Tetrahedron. 1958;2:1–57.
- 10.Miyaura N, Suzuki A. J. Chem. Soc. Chem. Commun. 1979:866–867. [Google Scholar]
- 11.See the Supporting Information for details.
- 12.Huang X, Anderson KW, Zim D, Jiang L, Klapars A, Buchwald SL. J. Am. Chem. Soc. 2003;125:6653–6655. doi: 10.1021/ja035483w. [DOI] [PubMed] [Google Scholar]
- 13.Kinzel T, Zhang Y, Buchwald SL. J. Am. Chem. Soc. 2010;132:14073–14075. doi: 10.1021/ja1073799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Karabelas Kostas, Hallberg A. J. Org. Chem. 1986;51:5286–5290. [Google Scholar]
- 15.Hu X-J, Di Y-T, Wang Y-H, Kong S-Y, Gao S, Li C-S, Liu H-Y, He H-P, Ding J, Xie H, Hao X-J. Planta Med. 2009;75:1157–1161. doi: 10.1055/s-0029-1185505. [DOI] [PubMed] [Google Scholar]
- 16.Pettersson M, Knueppel D, Martin SF. Org. Lett. 2007;9:4623–4626. doi: 10.1021/ol702132v. [DOI] [PubMed] [Google Scholar]
- 17.(a) Davis FA, Weismiller MC. J. Org. Chem. 1990;55:3715–3717. [Google Scholar]; (b) Davis FA. J. Org. Chem. 2006;71:8993–9003. doi: 10.1021/jo061027p. [DOI] [PubMed] [Google Scholar]
- 18.Martin JC, Arhart RJ. J. Am. Chem. Soc. 1971;93:4327–4329. [Google Scholar]
- 19.Upon treatment with europium trifluoromethanesulfonate in acetonitrile at 72 °C, hydroxyindolenines (+)-17 and (+)-18 (17:18 = 2:2:1) afforded the undesired oxindoles in 50% yield along with the desired indoxyls in 48% yield.
- 20.Liu G, Cogan DA, Owens TD, Tang TP, Ellman JA. J. Org. Chem. 1999;64:1278–1284. [Google Scholar]
- 21.(a) Morrison JD, Mosher HS. Asymmetric Organic Reactions. United Kingdom and Eire: Prentice-Hall International, Inc.; Canada: Prentice-Hall of Canada, Ltd.; 1971. Illustrative Examples of Asymmetric Synthesis; pp. 6–14. [Google Scholar]; (b) Gawley RE. J. Org. Chem. 2006;71:2411–2416. doi: 10.1021/jo052554w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim J, Movassaghi M. Chem. Soc. Rev. 2009;38:3035–3051. doi: 10.1039/b819925f. [DOI] [PubMed] [Google Scholar]
- 23.Movassaghi M, Siegel DS, Han S. Chem. Sci. 2010;1:561–566. doi: 10.1039/c0sc00351d. [DOI] [PMC free article] [PubMed] [Google Scholar]