Abstract
The KIR genes and their HLA class I ligands have thus far not been investigated in pemphigus foliaceus (PF) and related autoimmune diseases, such as pemphigus vulgaris. We genotyped 233 patients and 204 controls for KIR by PCR-SSP. HLA typing was performed by LABType SSO reagent kits. We estimated the odds ratio, 95% confidence interval and performed logistic regression analyses to test the hypothesis that KIR genes and their known ligands influence susceptibility to PF. We found significant negative association between activating genes and PF. The activating KIR genes may have an overlapping effect in the PF susceptibility and the presence of more than three activating genes was protective (OR = 0.49, p = 0.003). A strong protective association was found for higher ratios activating/inhibitory KIR (OR = 0.44, p = 0.001). KIR3DS1 and HLA-Bw4 were negatively associated to PF either isolated or combined, but higher significance was found for the presence of both together (OR = 0.34, p<10−3) suggesting that the activating function is the major factor to interfere in the PF pathogenesis. HLA-Bw4 (80I and 80T) was decreased in patients. There is evidence that HLA-Bw4(80T) may also be important as KIR3DS1 ligand, being the association of this pair (OR = 0.07, p = 0.001) stronger than KIR3DS1-Bw4(80I) (OR = 0.31, p = 0.002). Higher levels of activating KIR signals appeared protective to PF. The activating KIR genes have been commonly reported to increase the risk for autoimmunity, but particularities of endemic PF, like the well documented influence the environmental exposure in the pathogenesis of this disease, may be the reason why activated NK cells probably protect against pemphigus foliaceus.
Introduction
Pemphigus is a group of blistering autoimmune diseases of the skin that can be subdivided in two major clinical types: pemphigus foliaceus (PF) and pemphigus vulgaris (PV). The target of the autoantibodies differs between these two subtypes: while in PF the antibodies are directed to desmoglein 1 (dsg1), in PV they are direct to desmoglein 3 or to both desmoglein 1 and desmoglein 3 [1]. Endemic in Brazil, PF was first reported in this country in 1903, when it was thought to be a variant of a superficial mycosis [2]. PF represents a health problem in Brazil with an incidence of 25–35 million/cases/year. The highest prevalence of 3.4% was reported for the Terena Amerindian population that sums a total of about 1,200 individuals, in southern Mato Grosso do Sul State [3]. This disease manifests usually in rural workers and other low-wage earners who live near rivers and are exposed to poor living conditions [4]. The anti-dsg1 antibodies in PF (mainly of the IgG4 subclass) recognize epitopes of the amino-terminal domains of the extracellular portion of desmoglein 1 [5], affecting the intercellular junction. The epidermal cell detachment (acantholysis) produces blistering lesions that may appear as localized or generalized forms, being a suffering and painful disease.
The etiology of PF is still unclear. As a multifactorial disease, there are environmental and genetic factors. The first genetic association with PF was reported in 1989 by our group [6] and was corroborated by further studies [7] that showed the importance of the HLA-DR and HLA-DQ genes in the susceptibility to PF. The polymorphism of other genes, as those coding for cytokines [8], desmoglein 1 [9], CTLA4 [10], [11] CD40L, CD40, BLYS and CD19 [12] has also been analyzed.
NK cells are important for the immune response, and are controlled by activating and inhibitory receptors. The killer-cell immunoglobulin-like receptors (KIR) and their cognate HLA class I ligands are crucial for NK regulation [13]. The ligands of several inhibitory (2DL and 3DL) and activating (2DS and 3DS) KIR have been described. The KIR2DL1 recognizes HLA-C2 while KIR2DL2 and 2DL3 recognize the allotypes of HLA-C named C1 and a few C2 allotypes [14]. KIR2DS1 exhibits weak recognition for HLA-C2 and KIR2DS2 appears not to recognize any HLA epitope [15], [16]. KIR3DL1 binds the epitope Bw4, that comprises about 40% of the HLA-B allotypes and some of the HLA-A molecules (A*23, A*24, A*25 and A*32) [17]. The Bw4 positive HLA-A molecules and also a subset of the HLA-B Bw4 molecules that have isoleucine at position 80 (Bw4-80I) showed stronger interaction with KIR receptors than Bw4-80T (threonine) [18], [19]. Despite HLA-B epitopes are currently regarded as the most important KIR3DL1 ligands, we recently suggested that both HLA-A and HLA-B allotypes may be equally important for NK cell function [20]. Moreover, it has been suggested that KIR3DS1 also interacts with Bw4 molecules [21], even with no direct evidence.
Natural killer (NK) cells may play an important role in autoimmune diseases of the skin [22]. Takahashi et al. [23] showed that a higher percentage and number of NK cells are present in the peripheral blood of PV patients. Moreover, the NK cell of patients exhibited lower levels of perforin and granzime. Evidences that NK may play a role in the pathobiology of PV were also reported [24]. KIR polymorphism and combinations KIR/HLA have been associated with several diseases, including autoimmune [25], but the importance of NK and KIR in pemphigus foliaceus has not yet been explored. The aim of this work was to verify if KIR polymorphism and their known HLA ligands influence the susceptibility or resistance to PF.
Results
The frequencies of all the KIR genes analyzed in patients and controls are listed in Table S1. The absence/presence polymorphism of KIR2DS1, KIR2DS3, KIR3DS1 and KIR2DL5 was found negatively associated with PF, while KIR3DL1 was positively associated to the disease in Euro-descendants (Table 1). As extensively known, KIR haplotypes are subdivided in two major haplogroups (A and B), being the A haplogroup characterized by the presence of only KIR2DS4 as classic activating gene [26]. We found that the A/A genotype is associated to a higher susceptibility to PF and that the presence of B haplotypes is protective (Table 2).
Table 1. Genetic association between pemphigus foliaceus and five KIR genes.
| Gene | Patients | Controls | ||||||
| n | F (%) | n | F(%) | P | OR | CI | ||
| 2DS1 | Euro | 45 (143) | 31.5 | 65 (131) | 49.6 | 0.002 | 0.46 | 0.28–0.76 |
| Afro | 35 (90) | 38.9 | 31 (72) | 43.1 | 0.590 | 0.84 | 0.44–1.58 | |
| 2DS3 | Euro | 27 (144) | 18.8 | 44 (130) | 33.8 | 0.004 | 0.45 | 0.26–0.78 |
| Afro | 30 (89) | 33.7 | 20 (73) | 27.4 | 0.386 | 1.34 | 0.68–2.65 | |
| 3DS1 | Euro | 42 (142) | 29.6 | 58 (131) | 44.3 | 0.011 | 0.54 | 0.33–0.89 |
| Afro | 36 (89) | 40.4 | 28 (73) | 38.4 | 0.791 | 1.09 | 0.58–2.06 | |
| 2DL5 | Euro | 65 (144) | 45.1 | 81 (130) | 62.3 | 0.004 | 0.49 | 0.30–0.87 |
| Afro | 55 (89) | 61.8 | 41 (75) | 54.7 | 0.356 | 1.34 | 0.72–2.50 | |
| 3DL1* | Euro | 135 (144) | 93.8 | 116 (131) | 88.6 | 0.126 | 1.93 | 0.81–4.60 |
| Afro | 84 (89) | 94.4 | 64 (73) | 87.7 | 0.130 | 2.35 | 0.75–7.39 | |
In parentheses, the total number of individuals in the sample. Only genes which presented significant (P<0.05) association in at least one population sample are shown. The frequencies of all KIR genes are presented in Table S1.
Euro: predominantly European background; Afro: mixed subsample, with predominantly African background.
For the total sample (combining the Euro and Afro populations) we found a significant association for KIR3DL1: OR = 2.1, 95% CI 1.05–4.14, p = 0.033.
Table 2. The A/A genotype is associated to increased susceptibility to pemphigus foliaceus.
| Genotypes | Patients | Controls | ||||||
| n | F (%) | n | F(%) | P | OR | 95% CI | ||
| A/A | Euro | 50 (145) | 34.5 | 27 (134) | 20.1 | 0.007 | 2.08 | 1.21–3.60 |
| Afro | 25 (90) | 27.8 | 18 (72) | 25.0 | 0.689 | 1.15 | 0.57–2.34 | |
| B/x | Euro | 95 (145) | 65.5 | 107 (134) | 79.9 | 0.007 | 0.48 | 0.28–0.83 |
| Afro | 65 (90) | 72.2 | 54 (72) | 75.0 | 0.689 | 0.87 | 0.43–1.75 | |
In parentheses, the total number of individuals in the sample. B/x indicates A/B or B/B genotypes. Euro: predominantly European background; Afro: mixed subsample, with predominantly African background; Total: total sample.
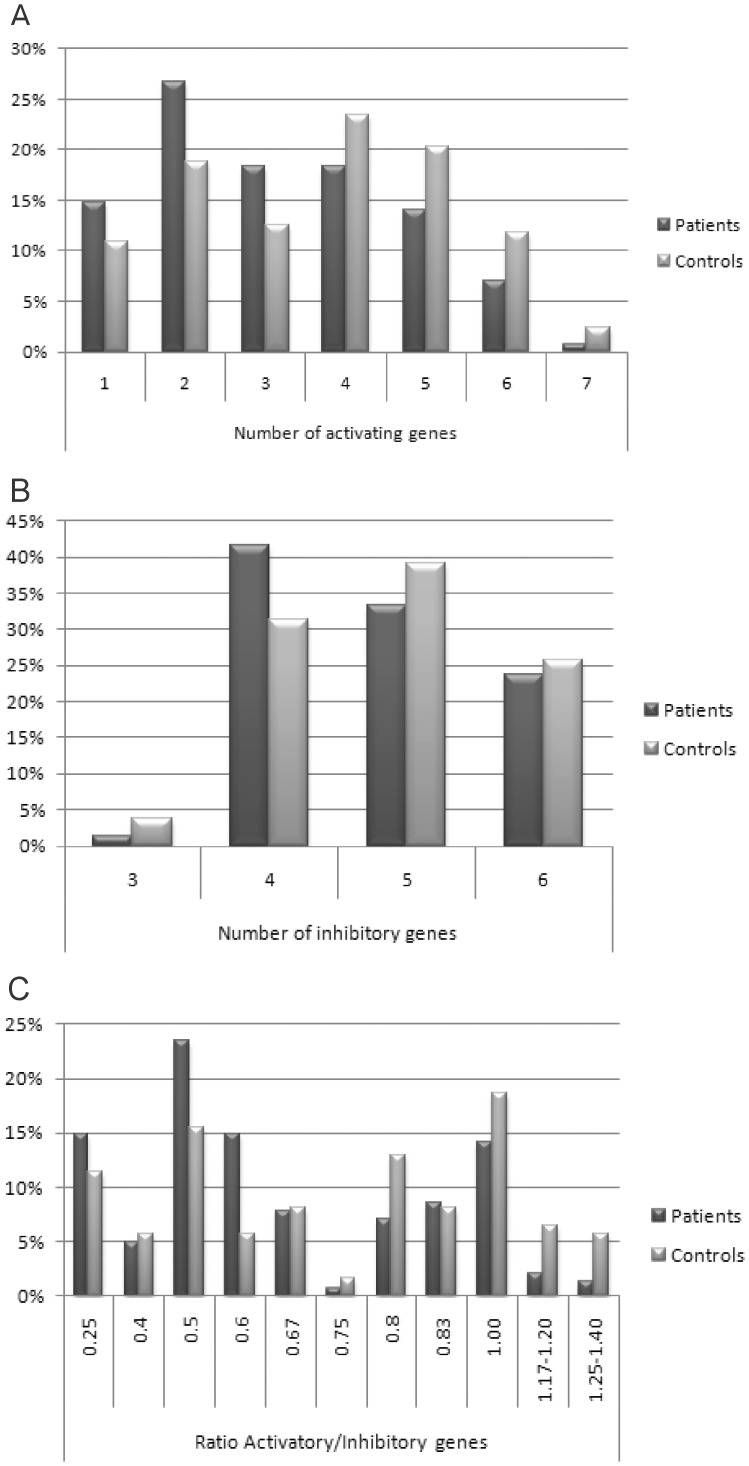
The number of inhibitory and of activating genes was compared between patients and controls. The function of KIR2DL4 is controversial, since it has an inhibitory motif (ITIM) in the intracellular domain [27] and an arginine in the transmembrane domain, putatively responsible for activating function [28]. It has been reported that the predominant function of KIR2DL4 is activating [28], [29] and for that reason, this gene was considered as activating in our analyses. No protein product or ligand was reported for the KIR3DL3 gene thus far [30]. Therefore, this gene was removed from our analyses, considering that its function is still obscure. The deleted form of KIR2DS4 was counted neither as activating nor as inhibitory. We found a significant association for the presence of more than three activating genes (OR = 0.49, 95% CI = 0.30–0.79, p = 0.003), which also means an odds ratio of 2.08 (95% CI = 1.25–3.32) for the presence of three or less activating genes (Figure 1A). For inhibitory genes, no statistical differences were found (Figure 1B). Further, we analyzed the ratio activating/inhibitory genes; the frequencies are shown in Figure 1C. A strong negative association was found for the ratio equal or higher than 0.75 (OR = 0.44; 95% CI = 0.27–0.73; p = 0.001).
Figure 1. Frequency of the number of activating and inhibitory KIR genes in patients and controls.
Only the Euro population was considered. (A) KIR2DL4 was counted as activating gene and the deleted KIR2DS4 was not counted as activating or inhibitory. Significant negative association was found for the presence of more than 3 activating genes (OR = 0.49; 95% CI = 0.30–0.79; p = 0.003). (B) KIR3DL3 was not included as inhibitory. (C) Significant negative association was found for the ratio higher than 0.75 (OR = 0.44; 95% CI = 0.27–0.73; p = 0.001).
The model tested in regression analyzes was performed only in the Euro descendants sample and it included the follow components: Presence of each of the negatively associated genes (KIR2DS1, KIR2DS3, KIR3DS1 and KIR2DL5), presence of HLA ligands (Bw4, C1 and C2), the presence of A/A or B/x haplotypes, the presence of more than three activating genes and the occurrence of activating/inhibitory genes ratio equal or higher than 0.75. The logistic regression analysis showed that the presence of more than three activating genes alone explained the result (OR = 0.25, 95% CI = 0.13–0.49, p<10−3, log likelihood = 195.502).
The frequent deletion of 22 pb in a group of KIR2DS4 alleles reported by Hsu et al. [31] generates a truncated protein with no transmembrane and cytoplasmic domains not expressed on the cell surface. We investigated if this deletion, and consequently the loss of the KIR2DS4 activating function, interferes in PF susceptibility, and no significant result was found (p>0.05).
We analyzed all known HLA class I KIR ligands (Table 3). The presence of one or more Bw4 epitopes (Bw4 80T and Bw4 80I from the HLA-A or HLA-B loci) was negatively associated (OR = 0.45, p = 0.005). Bw4 80T was also associated to protection against the disease (OR = 0.37, p = 0.001) but we did not find any association for Bw4 80I individually. We observed a strong and positive association for the presence of Bw6 (OR = 4.28, p<10−3). We also analyzed the association between individuals who present only Bw4 epitopes, individuals carrying both Bw4 and Bw6 and individuals carrying only Bw6. For these analyses, we considered both HLA-A and HLA-B loci. The effect was compatible with the absence of dominance and epistasis (only Bw4: OR = 0.24, p<10−3; both Bw4 and Bw6: OR = 0.61, p = 0.05; only Bw6: OR = 1.92, p = 0.03). Next, we tested if the Bw4 epitopes were in linkage disequilibrium (LD) with HLA class II alleles associated to PF reported by Pavoni et al. (2003). In that study, the authors separated the HLA-DRB1 alleles in three groups: susceptibility, neutral and protective. We found that LD between the susceptibility group of HLA-DRB1 alleles and Bw4(80T) is negative (D’ = −1.00, p = 0.001). Bw4(80I) was also in negative LD with this group of HLA alleles (D’ = −0.47, p = 0.001) while LD with Bw6 was positive (D’ = 0.59, r2 = 0.13 and p<10−3).
Table 3. Association analyzes between pemphigus foliaceus and HLA ligands of KIR.
| KIR Ligands | Patients | Controls | ||||||
| n | f (%) | n | F(%) | P | OR | 95% CI | ||
| Bw4* | Euro | 64 (99) | 64.6 | 56 (71) | 78.9 | 0.045 | 0.49 | 0.24–0.99 |
| Afro | 42 (64) | 65.6 | 35 (42) | 83.3 | 0.045 | 0.38 | 0.15–0.99 | |
| Total | 106 (163) | 65.0 | 91 (113) | 80.5 | 0.005 | 0.45 | 0.26–0.79 | |
| Bw4 80I | Euro | 57 (99) | 57.6 | 47 (72) | 65.3 | 0.307 | 0.72 | 0.38–1.35 |
| Afro | 38 (64) | 59.4 | 30 (42) | 71.4 | 0.210 | 0.58 | 0.25–1.34 | |
| Total | 95 (163) | 58.3 | 77 (114) | 67.5 | 0.110 | 0.45 | 0.41–1.11 | |
| Bw4 80T | Euro | 15(98) | 15.3 | 16 (66) | 24.2 | 0.144 | 0.56 | 0.25–1.22 |
| Afro | 5 (61) | 8.2 | 14 (41) | 34.2 | 0.001 | 0.17 | 0.06–0.53 | |
| Total | 20 (159) | 12.6 | 30 (107) | 28.0 | 0.001 | 0.37 | 0.20–0.69 | |
| Bw6 | Euro | 91 (96) | 94.8 | 57 (66) | 86.4 | 0.060 | 2.87 | 0.92–9.01 |
| Afro | 58 (61) | 95.1 | 30 (41) | 73.2 | 0.001 | 7.09 | 1.84–27.04 | |
| Total | 149 (157) | 94.9 | 87 (107) | 81.3 | <10−3 | 4.28 | 1.81–10.13 | |
In parentheses, the total number of individual in the sample. Euro: predominantly European background; Afro: mixed subsample, with predominantly African background; Total: total sample. *at least one Bw4 epitope (80I from HLA-A or HLA-B loci or 80T).
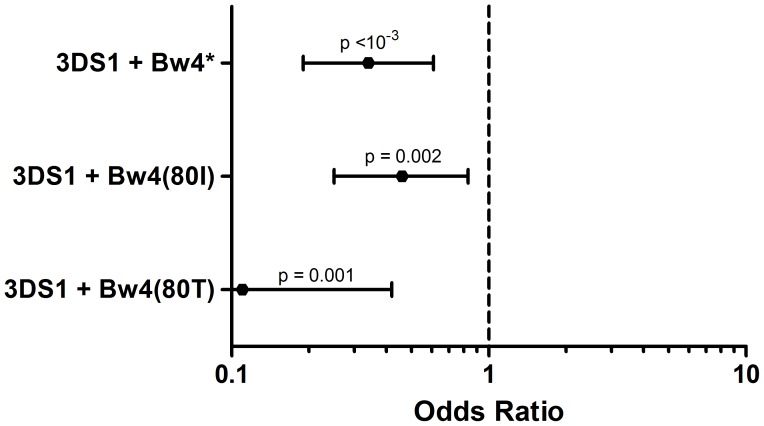
The KIR x HLA combinations were analyzed and are shown in Figure 2 and Table S2. We found negative associations for combinations including KIR3DL1/S1 and Bw4 ligands. We did not find any association for KIR2DL1, KIR2DL2/3 and KIR2DS1 combined with their HLA-C ligands (data not shown).
Figure 2. Association analyzes for the combinations KIR x HLA ligands.
The bars indicate the 95% confidence interval and the black spot indicates the OR. The results for the Euro sample are shown. The results for the two population strata and the complete information about KIR and HLA combinations are found in Table S2. *at least one Bw4 epitope (80I from HLA-A or HLA-B loci or 80T).
Discussion
In this study we describe the KIR gene frequencies in an urban population living in Western Brazil (Table S1) and, for the first time, we show a genetic association between susceptibility to pemphigus foliaceus and KIR genes and their HLA ligands.
All negatively associated genes (KIR2DS1, KIR2DS3, KIR3DS1 and KIR2DL5– Table 1) are not present in haplotype A, what could indicate, at first moment, that the presence of the B haplotype and not of each isolated gene, may protect against PF. In fact, we found a genetic association for the presence of B haplotypes (Table 2). In addition, we analyzed all pairs of associated genes for the presence of both, presence of one or the other and the absence of both (data not shown). Based on this, we hypothesized that the activating KIR could exhibit an additive effect for PF protection. Considering that combinations of several activating genes are typically found in B haplotypes [26], we hypothesized that the KIR activating function, rather than the isolated receptors, could be the cause of the differential susceptibility. Counting the number of activating genes in each individual, it became clear that the presence of a higher number of activating genes has a protective effect (Figure 1A). As expected by this hypothesis, the number of inhibitory genes did not differ significantly between patients and controls (Figure 1B). It is consensus that the balance of activating and inhibitory signals has a major effect on NK cell function [32]. Bearing that in mind, we evaluated the ratio activating/inhibitory genes (Figure 1C) and a stronger negative association was found (OR = 0.44 p = 0.001).
These results point to an at least partially redundant function among the activating genes and they may be all equally involved in PF protection. This hypothesis was corroborated by the regression analyzes, which showed the presence of more than three activating genes as the major factor of differential susceptibility. A similar conclusion was also reported by van der Slik et al., [33] who extensively tested many regression models in type 1 diabetes, showing that activating KIR genotypes influence the overall hierarchy of protection or susceptibility. Although the regression analyzes point the presence of more than three genes as the major factor for susceptibility to PF, it is important to remember that this analyze does not consider other KIR receptors. Considering the balance of signals as the possible major responsible for modulating the NK response [32], the ratio activating/inhibitory genes may be the most important factor if all NK receptors could be analyzed together.
A summary of case/control studies of KIR and KIR+HLA combinations in autoimmune diseases are shown in Table 4. In general, the activating KIR genes seem to increase the autoimmune response, being regarded as susceptibility factors in several studies. However, the interactions among NK cell receptors and HLA ligands may play a different role in the different diseases.
Table 4. Summary of KIR association studies regarding autoimmune diseases.
| Present study | aKimoto et al.2010 [44] bPellett et al.2007 [45] | aTajik et al. 2001 [46] bJiao et al.2008 [47] | Garcia-Leonet al. 2011 [48] | a Williams et al. 2005 [49] bMartin et al.2002 [50] | Pellett et al. 2007 [45] | Nikitina-Zakeet al.2004[51] | Yen, et al.,2001 [52] | aLuszczek et al. 2004 [53] bSuzuki et al.2004 [54] | Karlsen et al., 2007 [55] | |
| Pemphigus foliaceus | Systemic lupus erythematosus | Ankylosing spondylitis | Multiple sclerosis | Psoriatic arthritis | Scleroderma | Diabetes mellitus | Rheumatoid vasculitis | Psoriasis | Sclerosing cholangitis | |
| 2DL2 | Susceptibility | |||||||||
| 2DL5 | Protection | Protectiona | Susceptibilityab | Susceptibility | Susceptibility | Susceptibilityb | ||||
| 2DS1 | Protection | Susceptibilitya | Susceptibility | Susceptibilityab | Susceptibility | Susceptibility | Susceptibilityab | |||
| 2DS1+2DS2− * | Susceptibilityb | Susceptibility | ||||||||
| 2DS1+C2 | Susceptibilitya | |||||||||
| 2DS2 | Susceptibilityb | Susceptibility | Susceptibility | Susceptibility | ||||||
| 2DS3 | Protection | Susceptibility | ||||||||
| 3DL1 | Susceptibility | |||||||||
| 3DL1+Bw4 | Protectiona | Protection | ||||||||
| 3DS1 | Protection | Susceptibilityab | Susceptibility | |||||||
| 3DS1+Bw4 | Protection | Susceptibility | ||||||||
| Bw4 | Protection | Protectiona | Protection | |||||||
| Bw4(80I) | Protectiona | |||||||||
| Bw4(80T) | Protection | |||||||||
| Bw6 | Susceptibility | |||||||||
| Haplotype B | Protection | Susceptibilityb | ||||||||
| HLA-C2 | Protection | |||||||||
| HLA-Cw03 | Susceptibility | |||||||||
| HLA-Cw06 | Susceptibilitya | |||||||||
| More than threeactivating genes | Protection |
The importance of NK cell in autoimmunity mediated by B cell has been studied, [34] showing that NK activation can result in altered antibody responses, including those against autoantigens. Many autoimmune diseases are suspected to be related with infectious etiology, what brings the NK cell, responsible for the first line of defense, to an important role in the protection against autoimmunity. Particularities of PF could explain why the activating KIR function is protecting against this disease. Different from other autoimmune diseases, PF presents extremely high incidence in some endemic geographical areas in Brazil, in sharp contrast to its very low incidence elsewhere in South America and other continents. This unusual characteristic and also the gradual replacement of the endemic area in Brazil beyond years show that one (or more) environmental factor of limited geographic distribution is triggering the disease. Although the exact nature of the environmental contribution is still obscure, there are evidences that a black fly from the Simuliidae family could be involved in the infection with a protein, a virus or a microorganism which could initiate the autoimmune response in genetic susceptible individuals [35]. This agent does not need to result in an overt disease in the exposed individuals but it is related to the onset of pemphigus foliaceus. Moreover, other factors are also associated to PF, such as the low quality of life, the vicinity to rivers, some food, among others [4]; and the patients are usually exposed to this environmental context during their entire life. Many association studies have demonstrated that activating KIR genes protect against infectious disease (reviewed by Kulkarni et al., 2008 [25]). Therefore, carrying more activating KIR genes could represent a better protection against viruses, microorganisms or other environmental factors that trigger PF. Increasing the number of activating genes means a larger repertory of NK receptors recognizing antigens and activating the NK to defense.
Schleinitz et al. [36] presented other hypotheses on the reasons why NK cells could protect against autoimmunity. They could control the presentation of autontigens by killing the immature dendritic cells or control the activation of macrophages, which are responsible for tissue injury in chronic inflammation. However, in this particular disease, the activation of NK cells that control the release of autoantigens, mainly by regulating the environmental factors, such virus or other pathogens, could explain why the activating KIR function may be protective.
Only two inhibitory genes differed between patients and controls. KIR3DL1 was associated to increased susceptibility (OR = 2.1; p = 0.03) in the total sample. Because the odds ratios indicated increased susceptibility in both strata (Euro, OR = 1.93; Afro, OR = 2.35) we merged the two samples for KIR3DL1 association analysis This association could be explained by the absence of KIR3DS1, once 3DL1 and 3DS1 are alleles of the same locus. The presence of KIR3DL1 (and consequently the absence of KIR3DS1), may generate an increase of inhibitory signals, with reduction of the ratio activating/inhibitory receptors, what could explain this association. On the other hand, the haplotype B gene KIR2DL5 was negatively associated to PF in Euro-descendants only. We conclude that this association results from the linkage disequilibrium with several activating genes that are associated to PF only in this population stratum. High positive LD values for the pairs KIR2DL5 and other haplotype B activating genes (2DS3, 2DS5) are well recognized and have been reported in many studies, including a Southern Brazilian population of similar ancestry as our study population [20].
The different association patterns between Euro-descendant and Afro-descendant samples could be consequences of the complexity of the disease, allied to differences of other interacting susceptibility factors, whose frequencies may differ between the two population strata. The functional effects of the gene products depend on complex pathways. Other unknown genetic and environmental factors related to PF susceptibility may be involved in complex interactions with the genes here analyzed, and may depend partially on the ethnic context. Among all factors involved in resistance or susceptibility to PF, the activating KIR relative effect in Euro-descendants is possibly higher, which means that other unknown factors may predominate in this disease in Afro-descendants.
The frequency of HLA-C1/C2 did not differ between patients and controls, but significant associations were found regarding the Bw4 ligands (Table 3). It is noteworthy that, besides the differential susceptibility to PF being strongly associated with HLA class II genes [6], [7], this is the first study reporting significant association between this disease and the class I genes. The Bw4 epitope was found negatively associated with PF, in agreement with the negative association observed for its cognate activating KIR3DS1 receptor. Therefore, the susceptibility conferred by the presence of Bw6 could be interpreted, in this context, as simple consequence of the absence of Bw4. It is also corroborated by the effect of absence of dominance found between Bw4 and Bw6 epitopes. We cannot discard, however, the possibility of Bw6 acting in a direct susceptibility role. Due its weak binding to its receptor, [18], [19] the stronger association of Bw4(80T) than Bw4(80I) could be an evidence that the KIR binding is not the only reason for this epitope being associated. Although we do not know if HLA-A and B have other implications in PF pathogeneses beyond KIR recognition, a careful search for allelic association of these loci should be performed.
To verify if the association between PF and the HLA Bw4 and Bw6 epitopes could result from LD with the previously associated HLA-DRB1 alleles, we analyzed the linkage disequilibrium between these class I and class II genes. Interestingly, Bw4(80T), which is significantly less frequent among patients than among controls, was found in negative LD with the protective group of HLA-DRB1 alleles. The opposite was found for Bw6, which is associated to higher susceptibility to PF and occurs in positive LD with the susceptibility group of HLA-DRB1 alleles. We cannot affirm that the association found for Bw4 and Bw6 is simply a consequence of the LD with HLA class II alleles or vice versa, but this is possible. The associations of pemphigus foliaceus with Bw4 and with HLA-DRB1 might both result from their functions. Differential epitope selection by the HLA-DRB1 molecules encoded by different alleles is a likely mechanism, but we still do not know if this is the cause of the associations, because of the lack of functional studies. Similarly, it is plausible to hypothesize that the Bw4 epitope has a direct influence on pemphigus foliaceus disease susceptibility, because of its importance for recognition by KIR; however, this remains to be formally demonstrated by functional studies. Notwithstanding, the fact that the association between PF and the combination of KIR genes with their cognate HLA ligands is stronger that the associations with the receptors and the ligands individually is an compelling evidence that the interaction of Bw4 by its KIR receptor is directly involved in PF pathogenesis.
Combinations KIR+HLA also exhibited significant differences between patients and controls (Figure 2). The presence both KIR3DS1 and Bw4 showed stronger association than each one separately, which may corroborate that the activating function may be, in fact, the major factor interfering in the PF pathogenesis. Interestingly, the strongest negative association between KIR+HLA combinations was found for KIR3DS1+HLA-Bw4(80T). There are clear evidences that HLA-Bw4(80I) is powerfully recognized by KIR3DL1, and only indirect evidences that its homologue KIR3DS1 interacts with the same ligand [18], [19], [21]. Nevertheless, the isoleucine at position 80 is not essential for this interaction [37] and the studies performed have regarded mainly KIR3DL1 activity, being the comparisons between the KIR3DS1 ability of distinguishing both Bw4 ligands not well explored. These findings show that Bw4(80T) may also be important as a KIR3DS1 ligand, even if they are less strongly recognized.
KIR activating genes, their HLA ligands and combinations of both appear to be protective in the context of PF etiology. This association may be related with the contribution of infectious agents to the pathogenesis of this disease. NK cells have been demonstrated as important for autoreactivity and also have been implicated in several autoimmune diseases. To understand how those cells and their receptors influence the immune response could be a key for the comprehension of one face of the autoimmune complexity.
Materials and Methods
We analyzed 233 patients and 204 controls without history of the disease. The majority of the patients were contacted at Hospital Adventista do Pênfigo, Campo Grande, MS, Brazil. All individuals voluntarily agreed to participate and provided written informed consent. This study was approved by the Human Research Ethics Committee of the Federal University of Parana, in accordance with the Brazilian Federal laws. The individuals were separated in two groups, according to their predominant ancestry: Euro-descendants (Euro) and Afro-descendants (Afro). The reason of this subdivision is because different populations may differ in allele frequencies. Although this subdivision is arbitrary, this approach was validated by population genetic studies, which showed that the distribution of alleles known to be restricted to populations autochthonous from one continent follows a gradient among the population strata, as expected if the classification discriminated the strata according to relative contributions of the ancestral population [38], [39]. When the frequencies between Euro-descendants and Afro-descendants did not differ statistically, they were analyzed as a single population sample, because the total sample may give us a better representation of the whole population.
The KIR genotyping was performed by the PCR-SSP multiplex method [40] using two pairs of primers for each gene. For KIR2DL2 and 2DL3, an additional pair of primers for each gene [41] was included to confirm the results. A subset (163 patients and 114 controls) was genotyped for HLA class I applying the LABType® SSO reagent kits (One Lambda, USA).
Frequencies of KIR genes and haplogroups were obtained by direct counting. Tests of population differentiation were performed by analyzes of 2×2 contingency tables, calculating the exact p value by the metropolis algorithm. The p value of 0.05 was adopted as the significance limit. The Mantel-Haenszel method [42] applied for calculating the odds ratio (OR) and the 95% confidence intervals. Logistic regression analyzes were carried out by IBM® SPSS® Statistics software. HLA haplotypic frequencies were obtained using the ELB algorithm and the estimates of the LD parameters D′, r2, and p value were performed using the Arlequin software package [43].
Supporting Information
KIR frequencies in patients and controls. f = carrier frequency; fG = gene frequency.
(DOC)
Association analyzes for the combinations KIR x HLA ligands. The significant negative association between pairs KIR3DL1 and Bw4 epitopes may result from the high frequency of KIR3DL1 in the population, which means that the association values are explained by the Bw4 epitope frequencies only.
(DOC)
Acknowledgments
We are thankful to the staff of Hospital Adventista do Penfigo, in special to Marley Cafaro, for the friendly collaboration and hospitality. Thanks to all patients and controls who voluntarily participated of this work, and to all the staff of Laboratory of Human Molecular Genetics at the Federal University of Paraná for their assistance and support. Also thanks to Liana A. de Oliveira, Eleidi A. Chautard Freire-Maia and Marina Isabel M. de Almeida for their helpful advice and to Maria Dias da Silva for reading this manuscript.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: This project received financial support from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), PRONEX, Institutos do Milênio, Fundação Araucária, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Chidgey M. Desmosomes and disease: an update. Histol Histopathol. 2002;17:1179–1192. doi: 10.14670/HH-17.1179. [DOI] [PubMed] [Google Scholar]
- 2.Sampaio SA, Rivitti EA, Aoki V, Diaz LA. Brazilian pemphigus foliaceus, endemic pemphigus foliaceus, or fogo selvagem (wild fire). Dermatol Clin. 1994;12:765–776. [PubMed] [Google Scholar]
- 3.Hans-Filho G, dos Santos V, Katayama JH, Aoki V, Rivitti EA, et al. An active focus of high prevalence of fogo selvagem on an Amerindian reservation in Brazil. Cooperative Group on Fogo Selvagem Research. J Invest Dermatol. 1996;107:68–75. doi: 10.1111/1523-1747.ep12298213. [DOI] [PubMed] [Google Scholar]
- 4.Aoki V, Millikan RC, Rivitti EA, Hans-Filho G, Eaton DP, et al. Environmental risk factors in endemic pemphigus foliaceus (fogo selvagem). J Investig Dermatol Symp Proc. 2004;9:34–40. doi: 10.1111/j.1087-0024.2004.00833.x. [DOI] [PubMed] [Google Scholar]
- 5.Sekiguchi M, Futei Y, Fujii Y, Iwasaki T, Nishikawa T, et al. Dominant autoimmune epitopes recognized by pemphigus antibodies map to the N-terminal adhesive region of desmogleins. J Immunol. 2001;167:5439–5448. doi: 10.4049/jimmunol.167.9.5439. [DOI] [PubMed] [Google Scholar]
- 6.Petzl-Erler ML, Santamaria J. Are HLA class II genes controlling susceptibility and resistance to Brazilian pemphigus foliaceus (fogo selvagem)? Tissue Antigens. 1989;33:408–414. doi: 10.1111/j.1399-0039.1989.tb01684.x. [DOI] [PubMed] [Google Scholar]
- 7.Pavoni DP, Roxo VM, Marquart Filho A, Petzl-Erler ML. Dissecting the associations of endemic pemphigus foliaceus (Fogo Selvagem) with HLA-DRB1 alleles and genotypes. Genes Immun. 2003;4:110–116. doi: 10.1038/sj.gene.6363939. [DOI] [PubMed] [Google Scholar]
- 8.Pereira NF, Hansen JA, Lin MT, Roxo VM, Braun K, et al. Cytokine gene polymorphisms in endemic pemphigus foliaceus: a possible role for IL6 variants. Cytokine. 2004;28:233–241. doi: 10.1016/j.cyto.2004.08.006. [DOI] [PubMed] [Google Scholar]
- 9.Petzl-Erler ML, Malheiros D. Pemphigus foliaceus and desmoglein 1 gene polymorphism: is there any relationship? J Autoimmun. 2005;25:121–125. doi: 10.1016/j.jaut.2005.08.001. [DOI] [PubMed] [Google Scholar]
- 10.Pincerati MR, Dalla-Costa R, Petzl-Erler ML. CTLA4CT60 gene polymorphism is not associated with differential susceptibility to pemphigus foliaceus. Genet Mol Biol. 2010;33:442–444. doi: 10.1590/S1415-47572010005000073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pavoni DP, Cerqueira LB, Roxo VM, Petzl-Erler ML. Polymorphism of the promoter region and exon 1 of the CTLA4 gene in endemic pemphigus foliaceus (fogo selvagem). Braz J Med Biol Res. 2006;39:1227–1232. doi: 10.1590/s0100-879x2006000900010. [DOI] [PubMed] [Google Scholar]
- 12.Malheiros D, Petzl-Erler ML. Individual and epistatic effects of genetic polymorphisms of B-cell co-stimulatory molecules on susceptibility to pemphigus foliaceus. Genes Immun. 2009;10:547–558. doi: 10.1038/gene.2009.36. [DOI] [PubMed] [Google Scholar]
- 13.Parham P. Immunogenetics of killer cell immunoglobulin-like receptors. Mol Immunol. 2005;42:459–462. doi: 10.1016/j.molimm.2004.07.027. [DOI] [PubMed] [Google Scholar]
- 14.Winter CC, Gumperz JE, Parham P, Long EO, Wagtmann N. Direct binding and functional transfer of NK cell inhibitory receptors reveal novel patterns of HLA-C allotype recognition. J Immunol. 1998;161:571–577. [PubMed] [Google Scholar]
- 15.Foley B, De Santis D, Lathbury L, Christiansen F, Witt C. KIR2DS1-mediated activation overrides NKG2A-mediated inhibition in HLA-C C2-negative individuals. Int Immunol. 2008;20:555–563. doi: 10.1093/intimm/dxn013. [DOI] [PubMed] [Google Scholar]
- 16.Moesta AK, Graef T, Abi-Rached L, Older Aguilar AM, Guethlein LA, Parham P. Humans differ from other hominids in lacking an activating NK cell receptor that recognizes the C1 epitope of MHC class I. J Immunol. 2010;185:4233–4237. doi: 10.4049/jimmunol.1001951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thananchai H, Gillespie G, Martin MP, Bashirova A, Yawata N, et al. Cutting Edge: Allele-specific and peptide-dependent interactions between KIR3DL1 and HLA-A and HLA-B. J Immunol. 2007;178:33–37. doi: 10.4049/jimmunol.178.1.33. [DOI] [PubMed] [Google Scholar]
- 18.Carr WH, Pando MJ, Parham P. KIR3DL1 polymorphisms that affect NK cell inhibition by HLA-Bw4 ligand. J Immunol. 2005;175:5222–5229. doi: 10.4049/jimmunol.175.8.5222. [DOI] [PubMed] [Google Scholar]
- 19.Cella M, Longo A, Ferrara GB, Strominger JL, Colonna M. NK3-specific natural killer cells are selectively inhibited by Bw4-positive HLA alleles with isoleucine 80. J Exp Med. 1994;180:1235–1242. doi: 10.1084/jem.180.4.1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Augusto DG, Zehnder-Alves L, Pincerati MR, Martin MP, Carrington M, et al. Diversity of the KIR gene cluster in an urban Brazilian population. Immunogenetics. 2012;64:143–152. doi: 10.1007/s00251-011-0565-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Alter G, Martin MP, Teigen N, Carr WH, Suscovich TJ, et al. Differential natural killer cell-mediated inhibition of HIV-1 replication based on distinct KIR/HLA subtypes. J Exp Med. 2007;204:3027–3036. doi: 10.1084/jem.20070695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.von Bubnoff D, Andres E, Hentges F, Bieber T, Michel T, et al. Natural killer cells in atopic and autoimmune diseases of the skin. J Allergy Clin Immunol. 2010;125:60–68. doi: 10.1016/j.jaci.2009.11.020. [DOI] [PubMed] [Google Scholar]
- 23.Takahashi H, Amagai M, Tanikawa A, Suzuki S, Ikeda Y, et al. T helper type 2-biased natural killer cell phenotype in patients with pemphigus vulgaris. J Invest Dermatol. 2007;127:324–330. doi: 10.1038/sj.jid.5700527. [DOI] [PubMed] [Google Scholar]
- 24.Stern JN, Keskin DB, Barteneva N, Zuniga J, Yunis EJ, et al. Possible role of natural killer cells in pemphigus vulgaris - preliminary observations. Clin Exp Immunol. 2008;152:472–481. doi: 10.1111/j.1365-2249.2008.03638.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kulkarni S, Martin MP, Carrington M. The Yin and Yang of HLA and KIR in human disease. Semin Immunol. 2008;20:343–352. doi: 10.1016/j.smim.2008.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Uhrberg M, Parham P, Wernet P. Definition of gene content for nine common group B haplotypes of the Caucasoid population: KIR haplotypes contain between seven and eleven KIR genes. Immunogenetics. 2002;54:221–229. doi: 10.1007/s00251-002-0463-7. [DOI] [PubMed] [Google Scholar]
- 27.Yusa S, Catina TL, Campbell KS. SHP-1- and phosphotyrosine-independent inhibitory signaling by a killer cell Ig-like receptor cytoplasmic domain in human NK cells. J Immunol. 2002;168:5047–5057. doi: 10.4049/jimmunol.168.10.5047. [DOI] [PubMed] [Google Scholar]
- 28.Faure M, Long EO. KIR2DL4 (CD158d), an NK cell-activating receptor with inhibitory potential. J Immunol. 2002;168:6208–6214. doi: 10.4049/jimmunol.168.12.6208. [DOI] [PubMed] [Google Scholar]
- 29.Kikuchi-Maki A, Yusa S, Catina TL, Campbell KS. KIR2DL4 is an IL-2-regulated NK cell receptor that exhibits limited expression in humans but triggers strong IFN-gamma production. J Immunol. 2003;171:3415–3425. doi: 10.4049/jimmunol.171.7.3415. [DOI] [PubMed] [Google Scholar]
- 30.Trundley AE, Hiby SE, Chang C, Sharkey AM, Santourlidis S, et al. Molecular characterization of KIR3DL3. Immunogenetics. 2006;57:904–916. doi: 10.1007/s00251-005-0060-7. [DOI] [PubMed] [Google Scholar]
- 31.Hsu KC, Liu XR, Selvakumar A, Mickelson E, O’Reilly RJ, et al. Killer Ig-like receptor haplotype analysis by gene content: evidence for genomic diversity with a minimum of six basic framework haplotypes, each with multiple subsets. J Immunol. 2002;169:5118–5129. doi: 10.4049/jimmunol.169.9.5118. [DOI] [PubMed] [Google Scholar]
- 32.Parham P. Killer cell immunoglobulin-like receptor diversity: balancing signals in the natural killer cell response. Immunol Lett. 2004;92:11–13. doi: 10.1016/j.imlet.2003.11.016. [DOI] [PubMed] [Google Scholar]
- 33.van der Slik AR, Alizadeh BZ, Koeleman BP, Roep BO, Giphart MJ. Modelling KIR-HLA genotype disparities in type 1 diabetes. Tissue Antigens. 2007;69:101–105. doi: 10.1111/j.1399-0039.2006.762_5.x. [DOI] [PubMed] [Google Scholar]
- 34.Shi FD, Wang HB, Li H, Hong S, Taniguchi M, et al. Natural killer cells determine the outcome of B cell-mediated autoimmunity. Nat Immunol. 2000;1:245–251. doi: 10.1038/79792. [DOI] [PubMed] [Google Scholar]
- 35.Eaton DP, Diaz LA, Hans-Filho G, Santos VD, Aoki V, et al. Comparison of black fly species (Diptera: Simuliidae) on an Amerindian reservation with a high prevalence of fogo selvagem to neighboring disease-free sites in the State of Mato Grosso do Sul, Brazil. The Cooperative Group on Fogo Selvagem Research. J Med Entomol. 1998;35:120–131. doi: 10.1093/jmedent/35.2.120. [DOI] [PubMed] [Google Scholar]
- 36.Schleinitz N, Vely F, Harle JR, Vivier E. Natural killer cells in human autoimmune diseases. Immunology. 2010;131:451–458. doi: 10.1111/j.1365-2567.2010.03360.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gumperz JE, Barber LD, Valiante NM, Percival L, Phillips JH, et al. Conserved and variable residues within the Bw4 motif of HLA-B make separable contributions to recognition by the NKB1 killer cell-inhibitory receptor. J Immunol. 1997;158:5237–5241. [PubMed] [Google Scholar]
- 38.Braun-Prado K, Vieira Mion AL, Farah Pereira N, Culpi L, Petzl-Erler ML. HLA class I polymorphism, as characterised by PCR-SSOP, in a Brazilian exogamic population. Tissue Antigens. 2000;56:417–427. doi: 10.1034/j.1399-0039.2000.560504.x. [DOI] [PubMed] [Google Scholar]
- 39.Probst CM, Bompeixe EP, Pereira NF, de ODMM, Visentainer JE, et al. HLA polymorphism and evaluation of European, African, and Amerindian contribution to the white and mulatto populations from Parana, Brazil. Hum Biol. 2000;72:597–617. [PubMed] [Google Scholar]
- 40.Kulkarni S, Martin MP, Carrington M. KIR genotyping by multiplex PCR-SSP. Methods Mol Biol. 2010;612:365–375. doi: 10.1007/978-1-60761-362-6_25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vilches C, Castano J, Gomez-Lozano N, Estefania E. Facilitation of KIR genotyping by a PCR-SSP method that amplifies short DNA fragments. Tissue Antigens. 2007;70:415–422. doi: 10.1111/j.1399-0039.2007.00923.x. [DOI] [PubMed] [Google Scholar]
- 42.Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959;22:719–748. [PubMed] [Google Scholar]
- 43.Excoffier L, Laval G, Schneider S. Arlequin (version 3.0): an integrated software package for population genetics data analysis. Evol Bioinform. 2005;1:47–50. [PMC free article] [PubMed] [Google Scholar]
- 44.Kimoto Y, Horiuchi T, Tsukamoto H, Kiyohara C, Mitoma H, et al. Association of killer cell immunoglobulin-like receptor 2DL5 with systemic lupus erythematosus and accompanying infections. Rheumatology (Oxford) 2010;49:1346–1353. doi: 10.1093/rheumatology/keq050. [DOI] [PubMed] [Google Scholar]
- 45.Pellett F, Siannis F, Vukin I, Lee P, Urowitz MB, et al. KIRs and autoimmune disease: studies in systemic lupus erythematosus and scleroderma. Tissue Antigens. 2007;69:106–108. doi: 10.1111/j.1399-0039.2006.762_6.x. [DOI] [PubMed] [Google Scholar]
- 46.Tajik N, Shahsavar F, Poormoghim H, Radjabzadeh MF, Mousavi T, et al. KIR3DL1+HLA-B Bw4Ile80 and KIR2DS1+HLA-C2 combinations are both associated with ankylosing spondylitis in the Iranian population. Int J Immunogenet. 2011;38:403–409. doi: 10.1111/j.1744-313X.2011.01024.x. [DOI] [PubMed] [Google Scholar]
- 47.Jiao YL, Ma CY, Wang LC, Cui B, Zhang J, et al. Polymorphisms of KIRs gene and HLA-C alleles in patients with ankylosing spondylitis: possible association with susceptibility to the disease. J Clin Immunol. 2008;28:343–349. doi: 10.1007/s10875-008-9183-6. [DOI] [PubMed] [Google Scholar]
- 48.Garcia-Leon JA, Pinto-Medel MJ, Garcia-Trujillo L, Lopez-Gomez C, Oliver-Martos B, et al. Killer cell immunoglobulin-like receptor genes in Spanish multiple sclerosis patients. Mol Immunol. 2011;48:1896–1902. doi: 10.1016/j.molimm.2011.05.018. [DOI] [PubMed] [Google Scholar]
- 49.Williams F, Meenagh A, Sleator C, Cook D, Fernandez-Vina M, et al. Activating killer cell immunoglobulin-like receptor gene KIR2DS1 is associated with psoriatic arthritis. Hum Immunol. 2005;66:836–841. doi: 10.1016/j.humimm.2005.04.005. [DOI] [PubMed] [Google Scholar]
- 50.Martin MP, Nelson G, Lee JH, Pellett F, Gao X, et al. Cutting edge: susceptibility to psoriatic arthritis: influence of activating killer Ig-like receptor genes in the absence of specific HLA-C alleles. J Immunol. 2002;169:2818–2822. doi: 10.4049/jimmunol.169.6.2818. [DOI] [PubMed] [Google Scholar]
- 51.Nikitina-Zake L, Rajalingham R, Rumba I, Sanjeevi CB. Killer cell immunoglobulin-like receptor genes in Latvian patients with type 1 diabetes mellitus and healthy controls. Ann N Y Acad Sci. 2004;1037:161–169. doi: 10.1196/annals.1337.027. [DOI] [PubMed] [Google Scholar]
- 52.Yen JH, Moore BE, Nakajima T, Scholl D, Schaid DJ, et al. Major histocompatibility complex class I-recognizing receptors are disease risk genes in rheumatoid arthritis. J Exp Med. 2001;193:1159–1167. doi: 10.1084/jem.193.10.1159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Luszczek W, Manczak M, Cislo M, Nockowski P, Wisniewski A, et al. Gene for the activating natural killer cell receptor, KIR2DS1, is associated with susceptibility to psoriasis vulgaris. Hum Immunol. 2004;65:758–766. doi: 10.1016/j.humimm.2004.05.008. [DOI] [PubMed] [Google Scholar]
- 54.Suzuki Y, Hamamoto Y, Ogasawara Y, Ishikawa K, Yoshikawa Y, et al. Genetic polymorphisms of killer cell immunoglobulin-like receptors are associated with susceptibility to psoriasis vulgaris. J Invest Dermatol. 2004;122:1133–1136. doi: 10.1111/j.0022-202X.2004.22517.x. [DOI] [PubMed] [Google Scholar]
- 55.Karlsen TH, Boberg KM, Olsson M, Sun JY, Senitzer D, et al. Particular genetic variants of ligands for natural killer cell receptors may contribute to the HLA associated risk of primary sclerosing cholangitis. J Hepatol. 2007;46:899–906. doi: 10.1016/j.jhep.2007.01.032. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
KIR frequencies in patients and controls. f = carrier frequency; fG = gene frequency.
(DOC)
Association analyzes for the combinations KIR x HLA ligands. The significant negative association between pairs KIR3DL1 and Bw4 epitopes may result from the high frequency of KIR3DL1 in the population, which means that the association values are explained by the Bw4 epitope frequencies only.
(DOC)