Abstract
AIM
To investigate the difference of retinal nerve fiber layer (RNFL) thickness between Alzheimer's disease patients and normal people, so as to provide clue for the early diagnosis of Alzheimer's disease.
METHODS
The articles on the association of RNFL thickness and Alzheimer's disease were retrieved by searching international and national databases. The qualified articles were assessed by meta analysis with Stata11.0 software. The results were pooled using weighted mean difference (WMD) with a corresponding 95% confidence interval (CI).
RESULTS
Totally 7 studies enrolled 324 eyes were included in the meta-analysis. The results of meta analysis showed that in AD patients, there was a significant average RNFL thickness reduction compared with the control group [WMD=-17.561, 95%CI: (-23.971, -11.151)]. There were significant differences in superior, inferior, nasal and temporal RNFL thickness between the two groups. WMD with a 95%CI were [-18.829, 95%CI:(-25.915, -11.743); P<0.05], [-25.775, 95%CI:(-34.304, -17.247); P<0.05], [-16.877, 95%CI: (-29.141, -4.613); P<0.001] and [-14.565, 95%CI:(-28.002, -1.128); P<0.001] respectively. Begg's test and Egger's test did not show significant difference, funnel plot was basically symmetrical, indicating that there was no publication bias existed.
CONCLUSION
There are significant differences in the RNFL thickness in all quadrants between the two groups. RNFL thickness is reduced in AD patients compared with the control group.
Keywords: retinal nerve fiber layer thickness, optical coherence tomography, Alzheimer's disease, meta analysis
INTRODUCTION
Optical coherence tomography (OCT) is a relatively new non-invasive, non-contact, transpupillary imaging technology that provides high-resolution cross-sectional images of the retinal nerve fiber layer (RNFL) and macular volume. It has been extensively applied to ocular pathologies and is now being studied as a biomarker in various neurologic conditions[1],[2]. Alzheimer's disease (AD) is the most common degenerative dementia and causes a progressive decline in cognitive function[3]. Mild cognitive impairment (MCI) is defined as impairment in cognitive functions with otherwise normal performance of activities of daily living. MCI lies between and overlaps normal aging and AD and is now recognized to be a risk factor for AD[4]. In addition, visual symptoms are frequent early complaints in patients suffering from AD and visual acuity, visual field and contrast sensitivity seems to be also altered[5]. Recent studies investigated the differences between RNFL thickness in both healthy individuals and patients with MCI or AD with OCT. However, inconsistent conclusions were obtained[2], [6].
To date there are no published system reviews investigating the relationship between RNFL thickness and AD. To determine whether RNFL thickness is associated reduced in AD patients, we therefore performed a meta-analysis of cross-sectional studies that examined the difference of RNFL thickness between AD patients and healthy individuals.
METERIALS AND METHODS
Search strategy
We made a literature search of PubMed, the Cochrane Collaboration's Database of Systematic Review, and the Web of Science to identify potentially relevant studies by combinations of Medical Subject Headings and key words such as “Alzheimer's disease”, “retinal nerve fiber layer thickness”, “Optical coherence tomography”, “OCT”, “retinal thickness” and “RNFL”. The reference lists of obtained articles were also manually searched in International Journal of Ophthalmology, Chinese Journal of Ophthalmology, Chinese Journal of Neurology, National Medical Journal of China, Chinese Journal of Epidemiology.
Studies were included when they met the following criteria: (1) cross-sectional studies; (2) outcome of cases was defined as diagnosis of AD or MCI; (3) studies should provide the information of RNFL thickness (mean±SD) to estimate weighted mean difference (WMD) and corresponding 95% confidence intervals (CIs); (4) care was taken not to include any study population more than once. In case of duplicate reports of the same study population, the first publication should prevail; (5) there is no limit to sample size and language.
Study selection and data extraction
The diagnosis of AD was made according to the National Institute of Neurologic and Communicative Disorders and Stroke-Alzheimer's Disease and Related Disorders Association(NINCDS-ADRDA)[7] and the Diagnostic and Statistical Manual of Mental Disorders (DSM IV) criteria[8]. The criteria used for the inclusion of MCI patients were DSM-IV criteria[8] and Petersen criteria[9].
The RNFL thickness was measured using time domain OCT (TD-OCT) technology based on the Stratus OCT (Zeiss, software version 3.0 and 4.0). The parapapillary RNFL thickness parameters evaluated in these studies were in the areas of temporal quadrant (45°-316°), superior quadrant (226°-315°), inferior quadrant (46°-135°) and nasal quadrant (136°-225°). All controls and patients tested with OCT had no diseases, rather than AD, affecting the eye or optic nerve, like glaucoma or ischemic optic neuropathy or any pathological retinal finding in the ophthalmic examination. Only studies where age, sex and intraocular pressure were similar between the two groups were included in the meta-analysis. Studies were excluded if they were reviews, case report or duplicated report.
Data were extracted and quality was assessed independently by two reviewers (Xue-Fei He, Yi-Ting Liu) using guidelines published by the Cochrane Collaboration (The Cochrane Collaboration). Disagreement was resolved by consulting the third reviewer (Cheng Peng).
Statistical Analysis
Meta-analysis was conducted using STATA 11.0 for Windows (Stata, College Station, TX, USA). RNFL thickness in each study were extracted as mean±SD. Weighted mean difference (WMD) and 95%CIs were calculated for all eligible studies in the meta-analysis. Heterogeneity between the results of different studies was examined using χ2 tests for significance (P<0.05 was considered to be statistically significant) and I2 tests (I2>50%: significant heterogeneity; I2<25%: insignificant heterogeneity). According to the criteria, fixed or random-effects models were used appropriately[10]. Publication bias was assessed by Begg's and Egger's tests[11],[12]. Visual inspection of publication bias using a funnel plot of the data was not employed because bias may be incorrectly inferred if studies are heterogeneous.
RESULT
Studies and data included in the meta-analysis
We initially identified the 45 reports by using our key search terms. First, we excluded 33 that did not provide detailed information of RNFL thickness (mean±SD). Second, three of these studies were not in a format allowing inclusion into the meta-analysis because they are reviews and case report. Third, when similar data were published twice, we included the articles with the most definitive and extractable information.
Finally, seven cross-sectional studies remained and were included in the meta-analysis[2],[6],[13]-[17], containing a total of 336 eyes tested with OCT, and Table 1 showed average RNFL thickness(mean±SD) in patients and healthy individuals. Six of them provided RNFL thickness (mean±SD) in superior, inferior, nasal and temporal quadrants respectively[2],[6],[14]-[17], and the characteristics of these studies are showed in Table 2. Five of them compared AD patients with healthy individuals[2], [6],[14]-[16]. One of them compared AD and MCI patients with healthy individuals respectively[17]. One study that compared severe AD and mild AD with healthy individuals were excluded for it failed providing clues for the early diagnosis of AD in our meta-analysis. Taken together, we considered mild AD and MCI as dementia patients and healthy individuals as control in our meta-analysis.
Table 1. Characteristics of studies of Average RNFL thickness between patients and normal control subjects.
| First author | Year | Sample size(eyes) |
Average RNFL thicknessmean ± SD (µm) |
||
| patients | control | patients | control | ||
| Parisi et al[6] | 2001 | 17 | 14 | 59.5±16.7 | 99.9±8.95 |
| Iseri et al[2] | 2006 | 28 | 30 | 87.46±23.78 | 113.16± 6.72 |
| Berisha et al[14] | 2007 | 9 | 8 | 85.5±7.4 | 93.8±10.4 |
| Paquet et al[13] | 2007 | 14 | 15 | 89.2±2.9 | 102.2±1.8 |
| Deng et al[15] | 2009 | 15 | 15 | 82.92±12.93 | 100.54±9.98 |
| Chi et al[16] | 2010 | 12 | 17 | 93.18±11.36 | 99.44±8.88 |
| Kesler et al[17] | 2011 | 92 | 38 | 85.2±10.83 | 94.3±11.3 |
Table 2. RNFL thickness in each quadrant between patients and healthy controls.
| First author | Superior |
Inferior |
Nasal |
Temporal |
||||
| patients | control | patients | control | patients | control | patients | control | |
| Parisi et al[6] | 72.1±21.4 | 104.6±12.1 | 77.9±26.4 | 116.2±9.87 | 50.4±23.2 | 93.4±13.7 | 37.9±17.6 | 85.6±8.21 |
| Iseri et al[2] | 112.64±35.32 | 137.16±16.48 | 103.10±33.64 | 141.56± 19.09 | 63.57±19.09 | 96.00±34.39 | 64.92±17.7 | 72.30±16.42 |
| Berisha et al[14] | 92.2±21.6 | 113.6±10.8 | 117.0±15.3 | 128.1±11.4 | 65.7±15.1 | 64.1±7.3 | 67.0±15.0 | 69.5±11.1 |
| Deng et al[15] | 87.35±12.36 | 106.33±12.10 | 90.23±12.40 | 117.12±8.60 | 70.34±10.23 | 92.13±11.40 | 64.76±14.20 | 86.60±8.22 |
| Chi et al[16] | 115.09±14.05 | 127.94±12.29 | 120.64±17.99 | 129.56±15.17 | 65.81±13.02 | 65.31±8.99 | 72.36±17.85 | 74.69±11.72 |
| Kesler et al[17] | 100.0±15.17 | 110.0±16.7 | 110.8±16.14 | 141.6±19.1 | 66.41±15.07 | 76.4±21.8 | 64.79±10.19 | 67.8±15.1 |
Analysis of average RNFL thickness between patients and normal control subjects
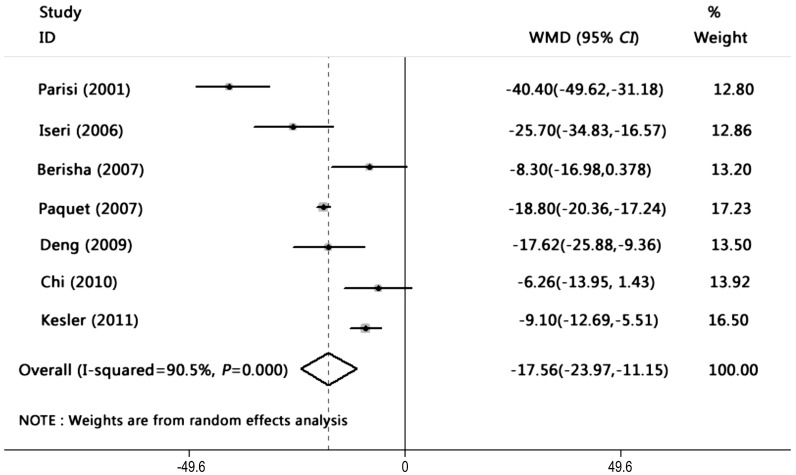
Heterogeneity between the results of different studies was conducted using STATA 11.0 (χ2=62.96, P<0.0001) and random-effects models were used. The results of meta analysis showed that the RNFL thickness in AD and MCI was reduced significantly compared with healthy controls [WMD= -17.561, 95%CI: (-23.971, -11.151), P<0.0001, Figure 1].
Figure 1. Meta-analysis of average RNFL thickness between patients and healthy controls.
Analysis of RNFL thickness in each quadrant between patients and normal control subjects
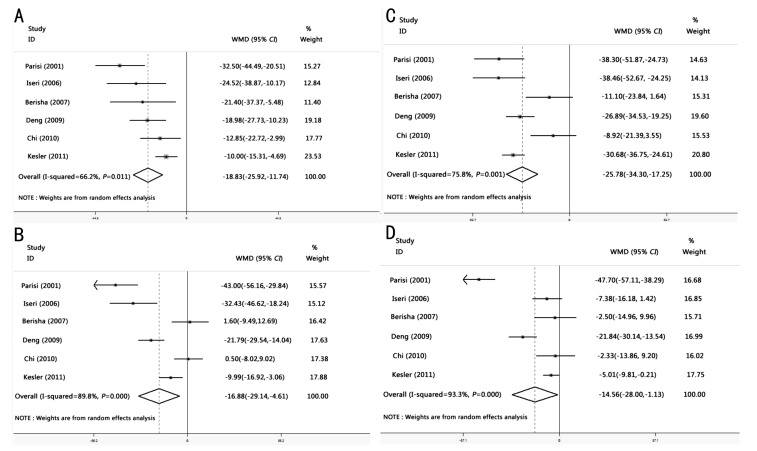
Heterogeneity between the results of different studies was conducted using STATA 11.0 and random-effects models were used. The results of meta analysis showed that there was a significant difference of RNFL thickness between the two groups in superior, inferior, nasal and temporal quadrants. Superior quadrant [WMD= -18.829, 95%CI:(-25.915, -11.743); P<0.05]; inferior quadrant [WMD= -25.775, 95%CI:(-34.304, -17.247); P<0.05]; nasal quadrant [WMD= -16.877, 95%CI:(-29.141, -4.613); P<0.001]; temporal quadrant [WMD= -14.565, 95%CI:(-28.002, -1.128); P<0.001]. In summary, the results of meta analysis showed that there was a significant RNFL thickness reduction in all quadrants in AD patients compared with the control group (Figure 2).
Figure 2. Meta-analysis of RNFL thickness between patients and healthy controls.
A: Superior RNFL thickness; B: Nasal RNFL thickness; C: Inferior RNFL thickness; D: Temporal RNFL thickness.
Neither the Egger's test (P=0.886) nor the Begg's (P=0.230) indicated significant publication bias. We performed a sensitivity analysis excluding the study of Berisba et al[14], which has a small sample size. The exclusion of that study did not significantly modify the results.
Subgroup meta-analysis
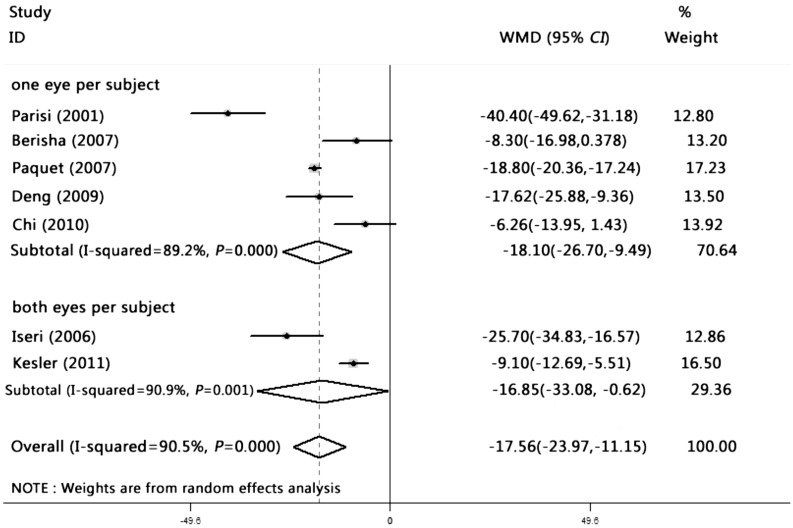
The above seven studies are divided into two groups: subjects have only one eye per subject and subjects have both eyes per subject. Two studies enrolled one eye per subject and five enrolled both eyes per subject. Subgroup analysis was performed, and subgroup results show that in monocular group, there was a significant difference of RNFL thickness between the patients and healthy controls [WMD= -18.096, 95%CI:( -26.699, -9.493); P<0.05]. In group that enrolled both eyes per subject, there was also a significant difference between the two groups [WMD= -16.848, 95%CI:(-33.079, -0.616); P<0.05]. All variables contributing to heterogeneity reached statistic, indicating that whether only one eye or both eyes per subject did not affect the final result (Figure 3).
Figure 3. Meta-analysis of subgroup between patients with only one eye and both eyes per subject.
DISCUSSION
With the rapidly aging population, AD represents one of the most frequent, major public health problems. To date, there are no effective ways to reverse the disease. The most effective strategy is early diagnose and early intervention[18]. Visual function is affected in most patients with AD, and causing reduced quality of life in AD patient. It has been thought to be due to neurodegeneration in the CNS, especially in the visual cortex. The defects in the nerve fiber layer may be the earliest sign of AD, even prior to damage to the hippocampal region that impacts memory.
The published data suggest an association between RNFL thinning and AD, several recent studies have compared the RNFL thickness in MCI and AD patients with healthy controls, however, there are various conclusions[2],[6], Berisha et al[14], Chi et al[16] and Kesler et al[17] showed significant differences in the superior quadrant thickness of RNFL between the groups. Parisi et al[6] showed an RNFL thickness reduction in each quadrant, and statistically significant differences were identified; some study indicated no relationship between them[19]. Our results of meta analysis showed that the RNFL thickness reduced in dementia patients compared with normal age-matched control in all the quadrants.
The results of meta analysis showed that there was a significant difference of RNFL thickness between the two groups in superior, inferior, nasal and temporal quadrants. This demonstrated that in AD patients RNFL thickness reduced in all quadrants compared with the control group, and the defects identified with OCT may be unique to AD.
Optical coherence tomography (OCT) is a reliable noninvasive technique that enables quantitative cross-sectional imaging of the RNFL. As a measure of neuronal degeneration, longitudinal OCT measures of RNFL change can act as a surrogate marker of axonal health. OCT could become an invaluable tool for measuring the efficacy of potential neuroprotective agents.
Confounding may partially account for the current findings. Our eligibility criteria required all studies to report results adjusted for, at minimum, age and sex. As the current review is based on published work, we cannot exclude the possibility that publication bias influences our findings; however, no evidence of publication bias was present with standard tests.
Ultimately, the results of Meta analysis showed that in AD patients, there was a significant RNFL thickness reduction in all quadrants compared with the healthy control group. In the light of our analysis, we suggest that OCT may become a standard part of drug trials and a standard part of the evaluation of patients in early AD. However, because of a limited number of current studies, additional prospective randomized studies are necessary to evaluate these observations.
REFERENCES
- 1.Ratchford JN, Quigg ME, Conger A, Frohman T, Frohman E, Balcer LJ, Calabresi PA, Kerr DA. Optical coherence tomography helps differentiate neuromyelitis optica and MS optic neuropathies. Neurology. 2009;73(4):302–308. doi: 10.1212/WNL.0b013e3181af78b8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Iseri PK, Altinas O, Tokay T, Yuksel N. Relationship between cognitive impairment and retinal morphological and visual functional abnormalities in Alzheimer disease. J Neuroophthalmol. 2006;26(1):18–24. doi: 10.1097/01.wno.0000204645.56873.26. [DOI] [PubMed] [Google Scholar]
- 3.Blennow K, de Leon MJ, Zetterberg H. Alzheimer's disease. Lancet. 2006;368(9533):387–403. doi: 10.1016/S0140-6736(06)69113-7. [DOI] [PubMed] [Google Scholar]
- 4.Petersen RC. Mild cognitive impairment. Lancet. 2006;367(9527):1979. doi: 10.1016/S0140-6736(06)68881-8. [DOI] [PubMed] [Google Scholar]
- 5.Trick GL, Trick LR, Morris P, Wolf M. Visual field loss in senile dementia of the Alzheimer's type. Neurology. 1995;45(1):68–74. doi: 10.1212/wnl.45.1.68. [DOI] [PubMed] [Google Scholar]
- 6.Parisi V, Restuccia R, Fattapposta F, Mina C, Bucci MG, Pierelli F. Morphological and functional retinal impairment in Alzheimer's disease patients. Clin Neurophysiol. 2001;112(10):1860–1867. doi: 10.1016/s1388-2457(01)00620-4. [DOI] [PubMed] [Google Scholar]
- 7.McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM. Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease. Neurology. 1984;34(7):939–944. doi: 10.1212/wnl.34.7.939. [DOI] [PubMed] [Google Scholar]
- 8.4th ed. Washington, DC: American Psychiatric Association; 1994. Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) [Google Scholar]
- 9.Petersen RC, Stevens JC, Ganguli M, Tangalos EG, Cummings JL, DeKosky ST. Practice parameter: early detection of dementia: mild cognitive impairment (an evidence-based review). Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2001;56(9):1133–1142. doi: 10.1212/wnl.56.9.1133. [DOI] [PubMed] [Google Scholar]
- 10.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50(4):1088–1101. [PubMed] [Google Scholar]
- 12.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Paquet C, Boissonnot M, Roger F, Dighiero P, Gil R, Hugon J. Abnormal retinal thickness in patients with mild cognitive impairment and Alzheimer's disease. Neurosci Lett. 2007;420(2):97–99. doi: 10.1016/j.neulet.2007.02.090. [DOI] [PubMed] [Google Scholar]
- 14.Berisha F, Feke GT, Trempe CL, McMeel JW, Schepens CL. Retinal abnormalities in early Alzheimer's disease. Invest Ophthalmol Vis Sci. 2007;48(5):2285–2289. doi: 10.1167/iovs.06-1029. [DOI] [PubMed] [Google Scholar]
- 15.Deng YL, Chen Y, Xie LQ, Wang G, Xu Wei, Tang YD, Wang Y, Chen SD. Morphological change of retina in mild Alzheimer's disease. Zhenduanxue Lilun Yu Shijian. 2009;8(4):397–400. [Google Scholar]
- 16.Chi Y, Wang YH, Yang L. The investigation of retinal nerve fiber loss in Alzheimer's disease. Zhonghua Yanke Zazhi. 2010;46(2):134–139. [PubMed] [Google Scholar]
- 17.Kesler A, Vakhapova V, Korczyn AD, Naftaliev E, Neudorfer M. Retinal thickness in patients with mild cognitive impairment and Alzheimer's disease. Clin Neurol Neurosurg. 2011;113(7):523–526. doi: 10.1016/j.clineuro.2011.02.014. [DOI] [PubMed] [Google Scholar]
- 18.Korczyn AD, Aharonson V. Computerized methods in the assessment and prediction of dementia. Curr Alzheimer Res. 2007;4(4):364–369. doi: 10.2174/156720507781788954. [DOI] [PubMed] [Google Scholar]
- 19.Kergoat H, Kergoat MJ, Justino L, Chertkow H, Robillard A, Bergman H. An evaluation of the retinal nerve fiber layer thickness by scanning laser polarimetry in individuals with dementia of the Alzheimer type. Acta Ophthalmol Scand. 2001;79(2):187–191. doi: 10.1034/j.1600-0420.2001.079002187.x. [DOI] [PubMed] [Google Scholar]