Summary
Three clones of E. coli, one of which was harbouring a tetracycline resistance plasmid, were inoculated together into the stomach of axenic mice. Without antibiotic selective pressure, the R-Plasmid bearing strain became dominant in the faeces of mice, while the R-plasmid free strain was eliminated. When the R-plasmid bearing strain was given to mice 4 days after the inoculation with the R-plasmid free strain, it was repressed and remained at the stable level of 104.5 organisms per g of faeces. But a rapid spread of the R-plasmid was observed, tetracycline resistant bacteria become dominant within one day, and replace the tetracycline sensitive E. coli.
The tetracycline resistance plasmid did not disadvantage the mediating strain in the gut, even in the absence of antibiotic pressure. In contrast Lebek and Egger (1983), studying the same strains in vitro, found that in a chemostat the plasmid bearing strain was overgrown by the plasmid free strain. These results strongly suggest that in vitro interactions between E. coli strains cannot be directly extrapolated to in vivo conditions. For the determination of the no-effect level of antibiotic residue on the selection of R-factor in the gut, studies should be made in vivo.
Keywords: Animals; Drug Resistance, Microbial; genetics; Escherichia coli; drug effects; genetics; growth & development; Feces; microbiology; Female; Germ-Free Life; Humans; Kinetics; Mice; Models, Biological; Nalidixic Acid; pharmacology; R Factors; Tetracycline; pharmacology
Keywords: antibiorésistance, plasmide, écologie microbienne, intestin, modèle animal, axénique: gnotoxénique, souris, antibiotique, acide nalidixique, rifampicine, compétition, tétracycline, facteur R
Introduction
Antibiotics are used in food animal production for the treatment and prevention of disease. This could result in the occurrence of residues in meat or milk if witholding times are not adhered to. The question of whether or not such residues select for intestinal bacteria bearing R-plasmids therefore arises.
Lebek and Egger (7) investigated the minimum R-factor selecting tetracycline concentration in an in vitro model. Three clones of Escherichia coli, one of which was harbouring a tetracycline resistance plasmid, were grown together in a chemostat, in the presence of various tetracycline concentrations. In this simple model, 0.25 μg tetracycline/ml of medium was enough to favour the selection of the tetracycline resistant strain. Since in vitro methods may not reflect conditions in vivo, an attempt was made to validate this model, by inoculating axenic mice per os with the three strains.
Materials and Methods
The strains, provided by Professor Lebek, were three E. coli K12 921 lac−, chromosomally resistant to streptomycin or nalidixic acid or rifampicin. The streptomycin resistant strain was plasmid free, the nalidixic acid resistant strain harboured a tetracycline resistance plasmid R 270 (Tet, Suf) IncFII rep, and the rifampicin resistant strain a plasmid R- F’ lac+ IncFI derep (7). Each strain was kept at −25°C in a broth containing 45% glycerol, and for the tetracycline resistant strain 10 μg/ml of tetracycline. It was then cultured overnight at 37°C in a nutrient broth, and diluted in phosphate buffer (KH2PO4 0.75 mg/ml, K2HPO4 3 mg/ml) on the basis of its optical density (400 nm, Spectrophotometer Uvikon 820, Kontron), to a concentration of 2 × 104 E. coli per ml of inoculum. The inoculum was buffered in order to avoid destruction of viable bacteria by the stomachal acidity (5). Number of viable E. coli and resistance pattern were checked a posteriori in the inoculum. The transfer frequency of plasmid R 270 was measured in vitro to confirm the repression of fertility. In standardized conditions of transfer (10) a transfer frequency of 3 × 10−4 was found, which is normal for a repressed plasmid.
The axenic mice, adult female OF1 (Iffa Credo, F 69210 St Germain) were maintained in plastic isolators(La Calhene, F 95870 Bezons). A commercial diet sterilized by a 4 Mrad irradiation (Diet C04, UAR, F 91360 Villemoisson), and autoclaved acidified water were both available ad libitum. The water was brought to pH 2.5 with HCI in order to avoid bacterial multiplication and tetracycline degradation in the drinking bottle. Each experiment was conducted in three mice, which were maintained in individual cages in one isolator. After confirmation of their axenic status, the mice were given 0.5 ml of the inoculum, which was administered by gavage directly into the stomach.
Freshly passed faeces were collected directly at the anus, and after decimal dilution in sterile saline, were plated within 30 minutes onto desoxycholate agar (Difco) which contained either no antibiotic or the following: streptomycin (1 mg/ml), nalidixic acid (50 μg/ml), rifampicin (100 μg/ml), tetracycline (20 μg/ml) or a combination of tetracycline with streptomycin, nalidixic acid or rifampicin (all antibiotics came from Sigma). A preliminary experiment had shown that there was no toxic synergy against the resistant strains between this selective medium and these antibiotic concentrations.
Results
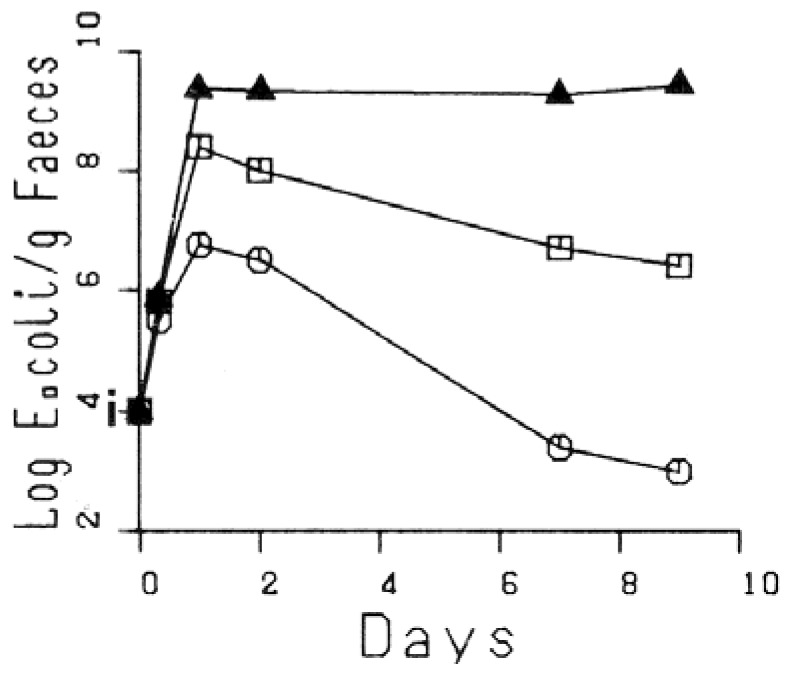
Axenic mice were inoculated with the three strains simultaneously, which became established and were non-pathogenic. On two sequential experiments, the nalidixic acid-tetracycline resistant strain became dominant and the streptomycin resistant strain was eliminated (Fig. 1). The in vivo interactions between the three strains were very different from the results obtained in vitro, where the plasmid-bearing strains were repressed by the plasmid-free strain (7).
Fig. 1.
Competition between three isogenic strains of E. coli in gnotobiotic mice. Each point represents the mean log number of CFU/g faeces from three mice.
Black triangles: nalidixic acid-tetracycline-R, Empty squares: rifampicin-R, Empty circles: streptomycin-R, i: inoculation.
When the nalidixic acid-tetracycline resistant strain was given to mice four days after inoculation with the rifampicin resistant strain and the streptomycin resistant strain, it was repressed. This antagonistic effect was not observed when mice colonised by the streptomycin resistant strain alone were inoculated with the nalidixic acid-tetracycline resistant strain. The latter became dominant five days after introduction into the monoxenic mice. In contrast, the rifampicin resistant strain, already established alone in the mice, was able to antagonize subsequent infection with the nalidixic acid-tetracycline resistant strain. On each of four experiments, the same equilibrium was reached of 109.5 rifampicin resistant and 104.5 nalidixic acid-tetracycline resistant organisms per g of faeces. This system was stable and reproducible and seemed to be a suitable model in which to determine the minimum tetracycline concentration in the drinking water which would select the tetracycline resistance plasmid.
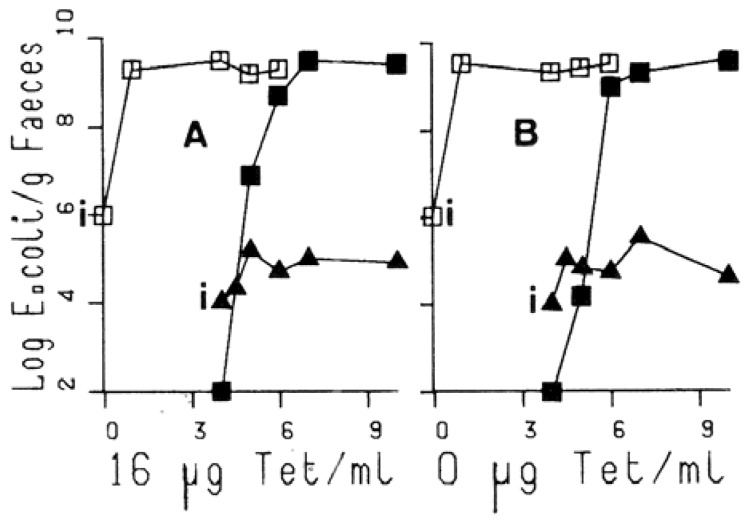
Mice containing only the rifampicin resistant strain were given tetracycline continuously in the drinking water at concentrations of 1, 4, and 16 μg/ml and then challenged orally with the nalidixic acid-tetracycline resistant strain. The administration of tetracycline did not influence the number of nalidixic acid-tetracycline resistant bacteria in faeces (Fig. 2A). When faecal dilutions were plated on agar containing tetracycline and rifampicin, it was found that a rifampicin resistant strain that carried the tetracycline resistance plasmid had evolved after the introduction of the nalidixic acid-tetracycline resistant strain into the ecosystem. This strain became dominant, and replaced the tetracycline-sensitive rifampicin resistant strain. This rapid spread of the R-plasmid occurred independently of the presence of tetracycline in the drinking water (Fig. 2B). These transconjugants had not been observed in the in vitro model (7).
Fig. 2.
Effect of tetracycline on the interaction between two E. coli strains in gnotobiotic mice.
2A: tetracycline (16 μg/ml) was added into the drinking water at day 3.
2B: No tetracycline in the water.
See Fig. 1 for symbols, except
Solid black squares: rifampicin-tetracycline-R.
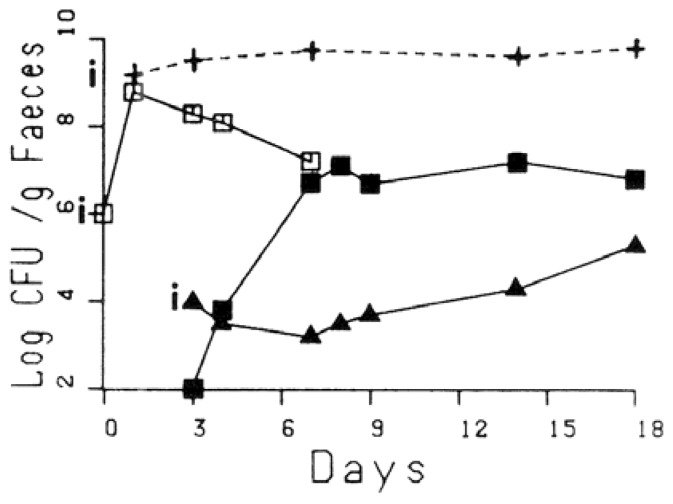
In the presence of a more complex flora, obtained from the anaerobic 10−6 dilution of human faeces (chosen because it harboured less than 106 coliforms per g), given together with the rifampicin resistant strain to axenic mice on two consecutive days, the number of rifampicin resistant E. coli decreased from 109.5 to 107 as shown in Fig. 3. Again, the tetracycline resistance plasmid spread to the rifampicin resistant strain after the introduction of the nalidixic acid-tetracycline resistant strain, in the absence of tetracycline selective pressure. This occurred less quickly, however, than in the previous experiment (see Fig. 3).
Fig. 3.
Competition between two E. coli strains in gnotobiotic mice, in the presence of a human faecal flora. See. Fig. 2 for symbols +---+: total counts (anaerobic).
Control experiments were realized, in which the plasmid bearing tetracycline resistant strain was replaced by a cured strain, obtained from the action of acridine orange on the nalidixic acid-tetracycline resistant strain. The inoculation of the three strains together led to the domination of the nalidixic acid resistant strain (tetracycline susceptible) over the other strains, the kinetics of the three populations being exactly the same as in Fig. 1. The experiment which is shown in Fig. 2B, i.e. inoculation of the nalidixic acid resistant strain three days after the rifampicin resistant strain, was repeated with the cured strain. Obviously, no tetracycline resistant transconjugants appeared, and an equilibrium was reached of 109.5 rifampicin resistant and 107.5 nalidixic acid resistant organisms per g of faeces.
Discussion
There is a general agreement that in vitro results cannot be simply extrapolated to the in vivo situation. The results presented in this paper confirm this in a well control-led system: (i) the chromosomal resistance markers seemed to have no effect on the in vitro behaviour of the three E. coli strains (7) but played a major role in the in vivo interactions between the same strains (the nalidixic acid resistant strain overcame by far the streptomycin resistant strain), and (ii) the R-plasmid appeared to confer a disadvantage to the mediating strain in vitro, but gave no clear ecological disadvantage in the digestive tract (the nalidixic acid resistant strain reached the same level if it was plasmid free or harbouring a R-plasmid, in the presence of the two other strains, cf. Fig. 1).
An explanation for this discrepancy between in vitro and in vivo results comes from the differences in the ecological conditions: In the rich oxygenated broth of the chemostat, the shorter generation time (32 min) of the plasmid free strain allows it to overgrow the plasmid bearing strains (generation time of 42 min) (7). In contrast, in the distal part of digestive tract, where little oxygen and nutrients are available, generation times are usually estimated to range between 4 and 12 h, and other factors may have greater importance, such as attachment to the gut wall or metabolic efficiency in a limiting substrate (5). Although plasmids are important determinants of the bacterial ecology, little is known about the colonisation factors they encode. According to Richmond (9), most plasmid bearing strains are disadvantaged against their plasmid free counterparts, likely because of a reduction in the in vivo growth rate. The factor which may explain the lower in vivo growth rate of plasmid bearing bacteria, is a higher saturation constant for limiting substrate (6), itself likely due to the cost of expressing the plasmidic genes in proteins, which is much more expensive than the energetic cost of duplicating the plasmidic DNA. The presence of a plasmid may sometimes have the opposite effect as well, i.e. to increase the in vivo fitness of a bacterium, likely because of adhesion to the gut wall, the proteic adhesins being often encoded by plasmid. For example, Duval-Iflah and others (3, 4) showed that in gnotobiotic mice, gnotobiotic piglets, and in human neonates, plasmid free strains generally inhibited the establishment of plasmid bearing strains. They found on one occasion, however, a tetracycline resistance plasmid which gave an ecological advantage to the strains bearing it. Therefore, whilst it has already been reported, the type of interaction presented here seems to be a rare occurrence.
We are now going to discuss the design of the experimental model: Numerous authors (e.g. 6, 10) have studied R-factor transfer in gnotobiotic animals. Their results have also shown that plasmid transfer can take place in vivo without antibiotic pressure. It is important to consider this phenomenon, which was not observed in vitro by Lebek and Egger (7) at their detection level (105 CFU/ml), in the development of any model designed to assess antimicrobial safety in vivo. A possible explanation for the lack of R-plasmid transfer in vitro is that the coculture was made for seven hours in the chemostat (7), whereas experiments in mice were conducted for several days. It was not possible in our experimental model to study the early evolution of faecal flora, since the first positive sample arrived at the anus of the mice about eight hours after inoculation in the stomach. To our opinion, long term experiments simulate much better the normal human gut bacterial interactions than short term ones. Another explanation for the difference in the R-plasmid transfer between the continuous flow culture and the gut is that the total number of E. coli in the gut is much higher (109.5) than in the chemostat (107). When the experimental data from in vitro and in vivo models were gathered together in a mathematical model of the plasmid transfer (2, 6), and simulated numerically on a computer, it was found that this difference in the number of potential recipient bacteria was alone sufficient to explain the differences observed in the transfer rates.
The competition between isogenic E. coli strains in vivo, differing in their chromosomal pattern of resistance, had also been studied. Onderdonk and others (8) showed that chromosomal resistance are detrimental to E. coli. It was clear in the results obtained here from the use of the cured nalidixic acid resistant strain, that the chromosomal resistance to streptomycin was much more detrimental in vivo to the bacteria than the resistance to nalidixic acid. The presence of both types of resistance (namely plasmidic and chromosomal) together in the same experiment was found to be rather confusing, and, in the author’s opinion, this kind of marker should be avoided in further studies.
The faecal human microflora given to the mice, has an antagonistic effect against the E. coli population (see Fig. 3), and seems to simulate the bacterial interactions of the human intestine. This kind of ecological situation must be a better model of the digestive tract than the mice harbouring only E. coli strains. It should nevertheless be explored more deeply, particularly in order to obtain a well defined and reproducible anaerobic flora, as Freter and others (5, 6) did with the mouse microflora.
From the point of view of the determination of the no-effect level of an antibiotic residue on the selection of R-factor in the gut, studies should be made in vivo. Axenic mice, inoculated with bacteria of human origin may be a suitable test system (1). Experiments in human volunteers may also to be useful, though the natural variation in the components of the gut flora, due to contaminations or diet modifications, may hide the effect of low levels of antibiotics.
Acknowledgments
The author is grateful to Professor G. Lebek for the kind donation of the strains, and to Mrs. S. Lumeau for her skilful technical assistance.
References
- 1.Corpet DE. The effect of bambermycin, carbadox, chlortetracycline and olaquindox on antibiotic resistance in intestinal coliforms: a new animal model. Ann Microbiol (Inst Pasteur) 1984;135 A:329–339. doi: 10.1016/s0769-2609(84)80015-0. [DOI] [PubMed] [Google Scholar]
- 2.Corpet DE. Ecological factors influencing the plasmid transfer in vivo and in vitro. J Antimicrob Chemother. 1986;18(Suppl C):127–32. doi: 10.1093/jac/18.supplement_c.127. [DOI] [PubMed] [Google Scholar]
- 3.Duval-Iflah Y, Raibaud P, Rousseau M. Antagonisms among isogenic strains of Escherichia coli in the digestive tracts of gnotobiotic mice. Infect Immun. 1981;34:957–969. doi: 10.1128/iai.34.3.957-969.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Duval-Iflah Y, Chappuis JP, Ducluzeau R, Raibaud P. Intraspecific interactions between Escherichia coli-strains in human newborns and in gnotobiotic mice and piglets. Progr Fd Nutr Sci. 1983;7:107–116. [PubMed] [Google Scholar]
- 5.Freter R, Brickner H, Fekete J, Vickerman MM, Carey KE. Survival and implantation of Escherichia coli in the intestinal tract. Infect Immun. 1983;39:686–703. doi: 10.1128/iai.39.2.686-703.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Freter R, Freter RR, Brickner H. Experimental and mathematical models of Escherichia coli plasmid transfer in vitro and in vivo. Infect Immun. 1983;39:60–84. doi: 10.1128/iai.39.1.60-84.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lebek G, Egger R. R-selection of subbacteriostatic tetracyclin-concentrations. Zbl Bakt Hyg, I Abt-Orig A. 1983;255:340–345. [PubMed] [Google Scholar]
- 8.Onderdonk A, Marshall B, Cisneros R, Levy SB. Competition between congenic Escherichia coli K-12 strains in vivo. Infect Immun. 1981;32:74–79. doi: 10.1128/iai.32.1.74-79.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Richmond MH. The survival of R-plasmids in the absence of antibiotic selection pressure. In: Drews J, Hogenauer G, editors. R-Factors: Their Properties and Possible Control. Springer-Verlang; Vienna: 1977. pp. 61–70. [Google Scholar]
- 10.Sansonetti P, Lafont JP, Jaffe-Brachet A, Guillot JF, Chaslus-Dancla E. Parameters controlling interbacterial plasmid spreading in a gnotoxenic chicken gut system: influence of plasmid and bacterial mutations. Antimicrob Agents Chemother. 1980;17:327–333. doi: 10.1128/aac.17.3.327. [DOI] [PMC free article] [PubMed] [Google Scholar]