Abstract
Objective
To assess the incidence and mortality impact of upper and lower gastrointestinal (GI) events in rheumatoid arthritis (RA) compared to non-RA subjects.
Methods
We identified incident upper and lower GI events and estimated their incidence rates using person-year methods in a population-based incident RA cohort of residents of Olmsted County, Minnesota, USA (1987 American College of Rheumatology criteria first fulfilled between January 1, 1980, and January 1, 2008) and non-RA subjects from the same population.
Results
The study included 813 patients with RA and 813 non-RA subjects (mean followup 10.3 and 10.8 yrs, respectively); 68% women; mean age 55.9 yrs in both cohorts. The rate of upper GI events/100 person-years was 2.9 in RA versus 1.7 in the non-RA cohort (rate ratio 1.7, 95% CI 1.4, 2.2); for lower GI events, the rates were 2.1 in RA versus 1.4 in the non-RA cohort (rate ratio 1.5, 95% CI 1.1, 1.9). The incidence of upper GI bleed, perforation, ulcer, obstruction, and any upper GI event in RA declined over calendar time; the incidence of lower GI events remained unchanged. Exposure to glucocorticoids, prior upper GI disease, abdominal surgery, and smoking were associated with lower GI events in RA. Both upper and lower GI events were associated with increased mortality risk in RA.
Conclusion
There is increased risk of serious upper and lower GI events in RA compared to non-RA subjects, and increased GI-related mortality in RA. Prominent declines in incidence of upper, but not lower GI events in RA highlight the need for studies investigating lower GI disease in patients with RA.
Key Indexing Terms: RHEUMATOID ARTHRITIS, GASTROINTESTINAL DISEASE, INCIDENCE
Gastrointestinal (GI) disease is one of the most common comorbidities that can adversely affect the quality of life and the lifespan of patients with rheumatoid arthritis (RA)1,2,3,4,5,6,7. There is extensive research showing increased risk of upper GI disease, particularly, drug-related upper GI disease, in RA8,9,10,11,12,13. Following major therapeutic advances in prevention of severe upper GI complications, a decline in nonsteroidal antiinflammatory drug (NSAID)-associated upper GI events in RA has been reported in the United States and Europe in recent decades14,15.
Until recently, and in contrast to widely studied upper GI disease, lower GI disease in RA remained underappreciated. Awareness of increased risk of lower GI disease associated with the use of glucocorticoids, NSAID, and biologic response modifiers has grown3,8,16,17,18,19,20,21. However, the incidence and risk factors for lower GI disease in RA remain uncertain and trends in occurrence of lower GI events in RA are unknown. We conducted a longitudinal population-based study of occurrence of serious upper and lower GI events in patients with incident RA compared to non-RA subjects. To better understand GI disease in RA, we studied potential determinants of lower GI events in patients with RA and the influence of GI events on mortality in RA.
MATERIALS AND METHODS
Study setting
This retrospective population-based study was performed using resources of the Rochester Epidemiology Project (REP) medical record linkage system22,23. This system allows ready access to complete inpatient and outpatient medical records from all community medical providers including Mayo Clinic and Olmsted Medical Center and their affiliated hospitals, local nursing homes, and the few private practitioners. The REP ensures virtually complete ascertainment of all clinically recognized cases of RA among residents of Olmsted County, Minnesota, USA. The unique features of the REP and its potential for population-based studies in rheumatology have been described in detail22,23,24.
Study subjects and data collection
The study population included a retrospectively identified incidence cohort, Olmsted County, MN, residents, aged ≥ 18 years who first fulfilled 1987 American College of Rheumatology criteria for RA25 between January 1, 1980, and January 1, 2008. The earliest date at which the patient fulfilled ≥ 4 criteria was defined as the RA incidence date. The reference cohort comprised non-RA subjects randomly selected from the same underlying population with similar age, sex, and calendar year. The population of Olmsted County was ~95,000 in 1980 and ~138,000 in 2008. The index date for each non-RA subject was defined as the RA incidence date of the corresponding RA patient. All subjects were followed until death, migration from Olmsted County, or December 31, 2009.
Detailed medical record review was performed for all study subjects. Information on GI events (including diagnoses, surgeries, and hospitalizations) was collected throughout the lifetime of each subject, without regard to whether the GI event was before or after RA incidence/index date. The following serious upper and/or lower GI events were ascertained: ulcer, bleed, perforation, bowel obstruction, esophagitis, diverticulitis, appendicitis, and various types of colitis (i.e., ischemic colitis, infectious colitis, drug-induced colitis, and other colitis for which cause could not be determined), as well as Crohn’s disease and ulcerative colitis. Lower GI events were defined as GI lesions beyond the duodenojejunal junction (below the ligament of Treitz). Cases were reviewed to identify whether bleeds were due to perforations. Bleeding perforations were included in both categories (i.e., perforations and bleeds). The following were excluded from our aggregate definitions of any GI event, any lower GI event and any upper GI event: functional GI disorders (i.e., functional dyspepsia, gastroesophageal reflux disease, irritable bowel syndrome), diverticular disease without diverticulitis, GI varices without bleeding, hemorrhoid bleeding, and appendicitis. GI tumors (malignant and benign) as a category of events were excluded. However, tumor-associated complications (e.g., bleed, perforation, obstruction) were included in the respective categories of GI events.
Data regarding demographics (age and sex), smoking, and alcohol abuse (based on physician diagnosis of chronic alcoholism) were gathered. Information regarding RA disease was collected, including results of all clinically performed tests for rheumatoid factor and erythrocyte sedimentation rate (ESR), persistently high ESR (≥ 3 ESR values ≥ 60 mm/h with ≥ 30 days between measurements), large joint swelling, joint erosions/destructive changes on radiographs, joint surgery (i.e., synovectomy and arthroplasty), and severe extraarticular manifestations of RA defined according to the Malmö criteria26. The use of antirheumatic medications including disease-modifying antirheumatic drugs (DMARD; i.e., methotrexate, hydroxychloroquine and other DMARD, biologic response modifiers), systemic glucocorticoids, NSAID, coxibs, and acetylsalicylic acid (ASA; if > 1950 mg/day for ≥ 3 months) was recorded. In addition, data on use of low-dose ASA (75–81 mg/day) were collected. The study protocol was approved by Institutional Review Boards from Mayo Clinic and Olmsted Medical Center.
Statistical methods
Descriptive statistics were used to examine the characteristics of RA and non-RA subjects at the incidence/index date. Among subjects who did not have the GI event prior to incidence/index date, the rates of development of each GI event during followup were calculated using person-years methods. Rate ratios (RR) were used to compare GI event rates in RA and non-RA cohorts. CI for RR were calculated using an F approximation27. Poisson regression models were used to assess time trends in the occurrence of GI events28. Cox models were used to examine the effect of GI events on mortality in RA. Dichotomous time-dependent covariates were used to model GI events that developed during followup. Specifically, over the followup period, a patient’s status was changed from “unexposed” to “exposed” at the time of first occurrence of a particular GI event.
Cox proportional hazard models were used to examine risk factors for incident lower GI events adjusting for age, sex, and calendar year of RA diagnosis. The risk factors included RA characteristics and medications, history of upper GI disease, prior GI surgery, smoking, and alcohol abuse.
Cumulative inflammatory burden was estimated by applying the most recent ESR value to each day of followup and summing these ESR values. All clinically performed ESR measures during the patient’s followup were used for this analysis. Inflammatory burden was analyzed continuously using smoothing splines to allow for nonlinear effects and was also categorized into tertiles. Similarly, cumulative glucocorticoid exposure was estimated by summing the daily dose of glucocorticoids throughout the followup and classified into the tertiles.
RESULTS
Patients’ characteristics
The study included 813 patients with RA and 813 non-RA subjects. Age, sex, and length of followup were similar in both cohorts (Table 1). Patients with RA were more likely to be current smokers (p = 0.04) and ever-smokers (p < 0.001) than non-RA subjects. Except for the higher likelihood of any GI diagnoses (32% vs 27%, respectively; p = 0.03), and upper GI diagnoses (21% vs 17%, respectively; p = 0.04) before the incidence/index date in the RA versus non-RA cohort, there were no statistically significant differences in the occurrence of GI events before the incidence/index date (Table 1).
Table 1.
Demographic data and distribution of gastrointestinal (GI) events before the rheumatoid arthritis (RA) incidence/index date in RA and non-RA cohorts.
| Variable | RA, n = 813 | Non-RA, n = 813 | p* |
|---|---|---|---|
| Mean age, yrs (SD) | 55.9 (15.7) | 55.9 (15.7) | 0.99 |
| Female, n (%) | 556 (68.4) | 556 (68.4) | 1.00 |
| Smoking at incidence/index date, n (%) | |||
| Current | 178 (22) | 144 (18) | 0.04 |
| Ever | 449 (55) | 377 (47) | < 0.001 |
| Alcohol abuse, n (%) | 56 (7) | 55 (7) | 0.92 |
| Mean length of followup, yrs (SD) | 10.2 (6.8) | 10.8 (7.02) | 0.11 |
| Diagnoses before incidence/index date, n (%) | |||
| Any GI event | 261 (32) | 222 (27) | 0.03 |
| Upper GI event | 169 (21) | 137 (17) | 0.04 |
| Lower GI event | 135 (17) | 131 (16) | 0.79 |
| Abdominal surgery before incidence/index date**, n (%) | |||
| Upper GI surgery | 26 (3) | 30 (4) | 0.59 |
| Lower GI surgery | 57 (7) | 52 (6) | 0.62 |
| GI-related hospitalizations before incidence/index date, n (%) | 102 (12) | 111 (14) | 0.51 |
Indicates the differences between RA and non-RA cohorts.
Any upper or lower abdominal surgery, excluding appendectomy.
Incidence of upper and lower GI events
During the followup, patients with RA developed 154 upper and 129 lower GI events, corresponding to event rates of 2.85 and 2.10 per 100 person-years, for upper and lower GI events, respectively (Table 2). Non-RA subjects developed 110 upper and 99 lower GI events corresponding to event rates of 1.66 and 1.43 per 100 person-years, for upper and lower GI events, respectively. This resulted in a 1.7-fold increased risk of upper GI events (95% CI 1.4, 2.2) and 1.5-fold increased risk of lower GI events in RA versus non-RA subjects (95% CI 1.1, 1.9). The risk of any GI event was 1.7-fold higher in RA versus non-RA subjects (95% CI 1.3, 2.2). The ratio of upper to lower GI events was 1.35 in RA versus 1.16 in the non-RA cohort.
Table 2.
Incidence and risks of upper and lower gastrointestinal (GI) events in patients with rheumatoid arthritis (RA) and non-RA subjects.
| GI Events | No. Events | Incidence Rates (95% CI)* | RR (95% CI) | ||
|---|---|---|---|---|---|
| RA | Non-RA | RA | Non-RA | ||
| Any GI event | 162 | 124 | 3.64 (3.10, 4.25) | 2.19 (1.82, 2.62) | 1.66 (1.32, 2.16) |
| Any upper GI event | 154 | 110 | 2.85 (2.41, 3.33) | 1.66 (1.36, 2.00) | 1.72 (1.35, 2.20) |
| Any lower GI event | 129 | 99 | 2.10 (1.76, 2.50) | 1.43 (1.16, 1.74) | 1.47 (1.13, 1.91) |
| Upper GI diagnoses | |||||
| Ulcer | 74 | 47 | 1.07 (0.84, 1.35) | 0.60 (0.44, 0.79) | 1.79 (1.25, 2.6) |
| Bleed | 52 | 333 | 0.68 (0.51, 0.90) | 0.40 (0.06, 0.21) | 1.71 (1.12, 2.67) |
| Perforation | 10 | 10 | 0.12 (0.06, 0.22) | 0.12 (0.06, 0.21) | 1.05 (0.44, 2.54) |
| Obstruction | 46 | 35 | 0.60 (0.44, 0.80) | 0.42 (0.29, 0.59) | 1.41 (0.91, 2.2) |
| Esophagitis | 73 | 46 | 1.01 (0.79, 1.26) | 0.57 (0.41, 0.57) | 1.77 (1.23, 2.58) |
| Lower GI diagnoses | |||||
| Ulcer | 16 | 14 | 0.20 (0.11, 0.32) | 0.16 (0.09, 0.28) | 1.21 (0.59, 2.5) |
| Bleed | 40 | 33 | 0.54 (0.38, 0.73) | 0.40 (0.28, 0.56) | 1.33 (0.85, 2.12) |
| Perforation | 11 | 5 | 0.13 (0.07, 0.24) | 0.06 (0.02, 0.13) | 2.23 (0.85, 7.1) |
| Obstruction | 41 | 31 | 0.53 (0.38, 0.72) | 0.38 (0.26, 0.53) | 1.40 (0.89, 2.26) |
| Diverticulitis | 35 | 25 | 0.45 (0.31, 0.62) | 0.29 (0.19, 0.43) | 1.51 (0.91, 2.56) |
| Ischemic colitis | 9 | 11 | 0.11 (0.05, 0.21) | 0.13 (0.06, 0.22) | 0.88 (0.36, 2.09) |
| Infectious colitis | 32 | 19 | 0.41 (0.28, 0.58) | 0.23 (0.14, 0.35) | 1.81 (1.05, 3.26) |
| Drug-induced colitis | 34 | 18 | 0.43 (0.29, 0.59) | 0.21 (0.12, 0.34) | 1.98 (1.15, 3.60) |
| Other colitis** | 9 | 12 | 0.11 (0.05, 0.21) | 0.14 (0.07, 0.24) | 0.81 (0.33, 1.87) |
| Crohn’s disease/ulcerative colitis | 5 | 3 | 0.06 (0.02, 0.14) | 0.04 (0.01, 0.10) | 1.68 (0.45, 7.89) |
Rates are given per 100 person-years.
Cases of colitis for which the cause could not be determined. Statistically significant differences (p < 0.05) are shown in bold type. RR: relative risk.
Among upper GI events, ulcers [rate ratio (RR) 1.8], bleeds (RR 1.7), and esophagitis (RR 1.8) were significantly more common in RA versus non-RA subjects (Table 2). Among lower GI events, the risks of infectious colitis (RR 1.8) and drug-induced colitis (RR 2.0) were significantly increased in RA versus non-RA. When drug-induced colitis was separated by type of drug (i.e., NSAID, DMARD, antibiotics, other drugs), patients with RA experienced more colitis due to NSAID (RR 16.0, 95% CI 0.02, 99.1) and DMARD (RR 18.1, 95% CI 0.02, 109.3) than non-RA subjects. Patients with RA also appeared to experience more colitis due to antibiotics than non-RA subjects (RR 1.6, 95% CI 0.7, 4.0), but this comparison did not reach statistical significance. However, due to the paucity of events in some subgroups of drug-induced colitis, statistical power was lacking to draw firm conclusions regarding these associations. There were no statistically significant differences in other lower GI events, although the numbers of some lower GI events (particularly lower GI perforations) were small in both cohorts.
Hospitalizations for any GI event (RR 1.6, 95% CI 1.2, 2.2), particularly esophagitis (RR 3.4, 95% CI 1.3, 12.3), diverticulitis (RR 2.0, 95% CI 1.01, 4.4), bleeds and/or perforations (RR 1.6, 95% CI 1.1, 2.2), bowel obstruction (RR 1.5, 95% CI 1.04, 2.3), and infectious colitis (RR 2.3, 95% CI 1.1, 5.1), but not for other GI events, were significantly increased in RA versus the non-RA cohort. The risk of hospitalization for drug-induced colitis in RA versus non-RA approached statistical significance (RR 2.7, 95% CI 0.99, 9.8). Among GI surgeries, only surgery for upper bowel obstruction was significantly more likely in RA versus non-RA subjects (RR 2.2, 95% CI 1.04, 5.1).
Trends in incidence of upper and lower GI events in RA and non-RA cohorts
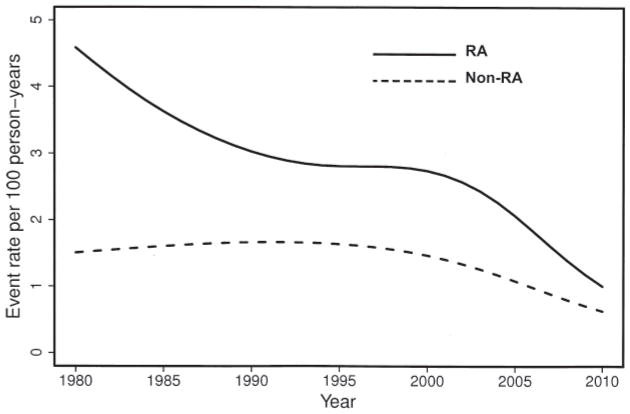
To determine the dynamics of occurrence of GI events over time, we analyzed trends in incidence of upper and lower GI events in RA and non-RA subjects since 1980. There was a significant decline in incidence of any upper GI event in RA (p = 0.006) and in non-RA subjects (p = 0.046) over calendar time (Figure 1). A similar declining trend was noted for upper GI ulcers in RA (p < 0.001), but not in the non-RA cohort (p = 0.13). Significant declines in the incidence of upper GI bleeds (p < 0.001) and upper GI obstructions (p = 0.024) were found in RA, but not in the non-RA cohort (p = 0.08 and p = 0.96, respectively).
Figure 1.
Trends in incidence of any upper gastrointestinal event in patients with rheumatoid arthritis (RA; solid line) and non-RA subjects (broken line) during the study period 1980–2009.
No significant trends in any lower GI event, lower GI bleeds, perforations, or obstructions were found. A significant increase in incidence of lower GI ulcers was observed in the non-RA cohort (p = 0.019), but not in RA (p = 0.39). However, these trends in lower GI ulcers were based on small numbers of events (16 in RA and 14 in non-RA). There was no evidence of a difference in calendar year trends of lower GI events for different age groups in the cohorts. As expected, the occurrence of individual GI events, any upper and any lower GI event, increased with age in both the RA and non-RA cohorts. No significant trends in upper and/or lower GI events occurred over the RA disease course.
Risk factors associated with lower GI events in RA
On univariate analysis, the use of glucocorticoids, history of upper GI disease, and prior abdominal surgery were significantly associated with any lower GI event, with adjustment for age, sex, and calendar year of RA diagnosis (Table 3). A history of upper GI disease was significantly associated with lower GI bleed. Use of low-dose ASA approached statistical significance with any lower GI event and lower GI bleed (p = 0.05 for both associations). A history of ever smoking increased the risk of lower GI events [hazard ratio (HR) 1.4, 95% CI 1.0, 2.1; p = 0.049], but no significant association was found with alcohol abuse (p = 0.23). Multivariable modeling revealed that glucocorticoids (HR 1.7, 95% CI 1.2, 2.6) and history of upper GI disease (HR 1.9, 95% CI 1.3, 2.8) were independent predictors, but smoking (HR 1.4, 95% CI 0.98, 2.1) and prior abdominal surgery (HR 1.3, 95% CI 0.8, 2.2) were no longer significant.
Table 3.
Effects of RA characteristics and medications, history of upper gastrointestinal (GI) disease, and history of abdominal surgery on the development of lower GI events and lower GI bleed in RA. Data are given as number (%) unless indicated otherwise.
| Characteristic | Value | Lower GI Events, HR (95% CI) | Lower GI Bleed* HR (95% CI) |
|---|---|---|---|
| At RA incidence | |||
| Rheumatoid factor-positive | 537 (66) | 1.28 (0.87, 1.88) | 0.99 (0.52, 1.90) |
| Mean ESR, mm/h (SD) | 24.8 ± 20.5 | 1.00 (0.99, 1.01) | 1.00 (0.99, 1.02) |
| At any time during followup | |||
| ESR ≥ 3 values ≥= 60 mm/h | 105 (13) | 1.08 (0.61, 1.90) | 1.10 (0.38, 3.16) |
| Large joint swelling | 639 (79) | 1.13 (0.75, 1.72) | 1.31 (0.61, 2.80) |
| Joint erosions/destructive changes on radiographs | 433 (53) | 0.84 (0.59, 1.20) | 1.41 (0.75, 2.67) |
| % radiographed | 95 | — | — |
| Joint surgery | |||
| Synovectomy | 85 (10) | 0.99 (0.54, 1.82) | 0.79 (0.24, 2.59) |
| Arthroplasty | 141 (17) | 0.87 (0.52, 1.46) | 1.09 (0.44, 2.68) |
| Severe extraarticular manifestations of RA | 90 (11) | 1.29 (0.72, 2.31) | 1.40 (0.49, 4.00) |
| Antirheumatic drug use | |||
| Methotrexate | 469 (58) | 1.05 (0.72, 1.53) | 1.16 (0.58, 2.29) |
| Hydroxychloroquine | 480 (59) | 1.18 (0.82, 1.70) | 1.24 (0.65, 2.38) |
| Other DMARD | 258 (32) | 1.41 (0.94, 2.10) | 1.83 (0.93, 3.61) |
| Biologic response modifiers | 137 (17) | 1.14 (0.57, 2.29) | 1.74 (0.58, 5.16) |
| Glucocorticoids | 627 (77) | 1.77 (1.17, 2.68) | 1.71 (0.85, 3.46) |
| NSAID | 737 (91) | 1.66 (0.86, 3.18) | 1.20 (0.42, 3.41) |
| Coxibs | 390 (48) | 1.12 (0.74, 1.68) | 0.77 (0.35, 1.69) |
| ASA for arthritis** | 337 (41) | 1.21 (0.81, 1.79) | 1.18 (0.57, 2.44) |
| Low-dose ASA for cardioprotection | 349 (43) | 1.50 (0.99, 2.26) | 2.09 (0.99, 4.38) |
| Upper GI disease | 329 (40) | 2.07 (1.44, 2.97) | 2.77 (1.45, 5.32) |
| Abdominal surgery*** | 150 (18) | 1.80 (1.10, 2.93) | 1.07 (0.44, 2.61) |
Excluding perforations.
Use of > 6 tablets of ASA per day (> 1950 mg/day) for ≥ 3 months.
Any upper or lower abdominal surgery, excluding appendectomy. Statistically significant associations (p < 0.05) are shown in bold type. RA: rheumatoid arthritis; HR: hazard ratio; ESR: erythrocyte sedimentation rate; DMARD: disease-modifying antirheumatic drugs; ASA: acetylsalicylic acid; NSAID: nonsteroidal antiinflammatory drugs.
We estimated risks of lower GI events associated with cumulative ESR measures and cumulative glucocorticoid exposure (Table 4). Cumulative inflammatory burden was not associated with the risk of any lower GI event or lower GI bleed. Cumulative glucocorticoid exposure was significantly associated with incident lower GI events, but there was no significant difference in risk between the tertiles of glucocorticoid exposure (p = 0.55).
Table 4.
Effects of inflammatory burden and exposure to glucocorticoids on development of lower gastrointestinal (GI) events and lower GI bleed in rheumatoid arthritis.
| Characteristic | Lower GI Events, HR (95% CI) | Lower GI Bleed*, HR (95% CI) |
|---|---|---|
| Cumulative inflammatory burden of ESR** | ||
| Low tertile | 1.00 (reference) | 1.00 (reference) |
| Middle tertile | 1.00 (0.59, 1.70) | 0.58 (0.20, 1.71) |
| High tertile | 1.14 (0.61, 2.12) | 1.12 (0.37, 3.43) |
| p = 0.87 | p = 0.36 | |
| Cumulative glucocorticoid exposure*** | ||
| Never exposed | 1.00 (reference) | 1.00 (reference) |
| Low tertile | 2.63 (1.46, 4.73) | 1.59 (0.68, 3.68) |
| Middle tertile | 3.40 (1.99, 5.79) | 1.18 (0.45, 3.09) |
| High tertile | 2.70 (1.42, 5.13) | 2.00 (0.82, 4.86) |
| p < 0.001 | p = 0.42 | |
Excluding perforations.
For cumulative inflammatory burden of ESR, low tertile: ≤ 15,000 mm/h, mid tertile: > 15,000 to ≤ 45,000 mm/h, high tertile: > 45,000 mm/h.
For cumulative glucocorticoid exposure, low tertile: ≤ 1500 mg, mid tertile: > 1500 to ≤ 7000 mg, high tertile: > 7000 mg. Statistically significant associations (p < 0.05) are shown in bold type. HR: hazard ratio.
Mortality impact of upper and lower GI events in RA
During followup, 229 patients with RA died. Univariate analysis revealed that the following events were significantly associated with mortality after adjustment for age, sex, and calendar year of RA diagnosis: any upper GI event (HR 1.4, 95% CI 1.06, 1.8), upper GI bleed and/or perforation (HR 2.1, 95% CI 1.5, 2.8), lower GI bleed and/or perforation (HR 1.6, 95% CI 1.1, 2.4), upper GI obstruction (HR 1.8, 95% CI 1.2, 2.5), lower GI obstruction (HR 1.7, 95% CI 1.2, 2.4), and lower GI ulcer (HR 2.2, 95% CI 1.3, 3.6). The association of upper GI ulcer with mortality approached statistical significance (HR 1.3, 95% CI 0.98, 1.7; p = 0.07). In multivariable models, after adjustment for other known predictors of mortality in RA, i.e., the presence of severe extraarticular manifestations of RA, comorbidity (including cardiovascular disease, renal disease, liver disease, dementia, cancer), history of alcohol abuse, and glucocorticoid use29, only upper GI bleed and/or perforation remained significantly associated with mortality (HR 2.1, 95% CI 1.5, 3.0).
DISCUSSION
The available evidence suggests a substantial burden of GI disease in RA. We report the results of a longitudinal population-based study of incidence of upper and lower GI events in patients with incident RA compared with non-RA subjects from the same underlying population. Concordant with findings from previous studies, the risk of incident upper GI events (particularly ulcers, bleeds, esophagitis, and perhaps upper bowel obstruction) remains increased in RA versus non-RA subjects in recent decades8,9,12,13.
Evidence of increased risk of lower GI events (primarily ulcers, bleeds, and more recently perforations) in RA is mounting. This risk is predominantly described within the scope of adverse effects of NSAID, glucocorticoids, and biologic response modifiers8,9,10,17,19,20,30,31,32,33,34,35. We found a significantly increased risk of any incident lower GI event, particularly infectious colitis and drug-induced colitis, and a trend to increased risk of lower GI bleeding, lower GI perforation, bowel obstruction, and diverticulitis, in RA versus non-RA subjects.
There was a significantly increased risk of hospitalizations for most of the “high-risk” upper and lower GI events examined (including any GI event, esophagitis, bleeds, perforations, bowel obstructions, diverticulitis, infectious colitis, and perhaps drug-induced colitis) in RA versus non-RA subjects. Supporting the existing evidence of increased mortality associated with GI bleeds in RA, upper GI bleeds and/or perforations were found to be an independent determinant of mortality in RA in this study14,36.
Declines in incidence of upper GI disease were previously reported in the general population37,38. Some authors have reported a decrease in hospitalization rates for NSAID-induced upper GI gastropathy in RA in the United States14. Decreasing incidence of NSAID-gastropathy in RA in recent years was suggested in a large survey in Amsterdam15. Concordantly, we found a significant decline in any upper GI event in both the RA and non-RA cohorts and prominent declines in upper GI bleeds, ulcers, and obstructions in patients with RA over time. These declines may be related in part to the implementation of guidelines for prevention of NSAID-gastropathy in the late 1990s14,15,39. Other reasons, including early diagnosis and increased awareness of upper GI disease, could also contribute to the decline in serious upper GI complications (e.g., bleeds, perforations, and obstructions).
Despite favorable trends in upper GI events, the incidence of lower GI events does not appear to be declining over recent decades. In contrast, some increase in occurrence of lower GI ulcers was noted in the non-RA cohort. This is in agreement with recent data from Europe, where a slight increase in lower GI events has been reported in the general population38. That there has been no significant improvement in incidence of lower GI events, particularly in RA, suggests the lack of effective strategies and algorithms for primary and secondary prevention of lower GI disease and the need for improved ascertainment of associated risk factors.
The association of systemic glucocorticoid use with risk of serious lower GI events in our study corroborates the findings of others10,13,17,31. There was no apparent cumulative dose-dependent effect of glucocorticoids on lower GI events in our study. There were no statistically significant associations of other antirheumatic medications or RA characteristics (including cumulative inflammatory burden) with lower GI events in RA. However, statistical power may be limited in these analyses, suggesting that some associations could be missed. Although not statistically significant, there was a trend toward decreased risk of lower GI bleeds in patients with RA who used coxibs compared to nonusers, which corroborates the emerging evidence of better GI safety in coxib users compared to NSAID users37.
Increased risk of lower GI events in patients with RA and a history of prior abdominal surgery, and in those with prior upper GI disease, could be due to changes in GI motility following upper GI disorders and surgical distress, resulting in the compromised postoperative local immune response40. Increased risk of any GI event and upper GI events before RA may be due to more medication use (NSAID use in particular) in patients with evolving RA versus non-RA subjects, which suggests a preexisting unfavorable background for GI disease in RA. Increased risk of lower GI events in smokers with RA supports the evidence of the adverse effects of smoking on upper and lower GI tract suggested by others41,42. Exposure to these and other factors (e.g., decreasing adherence to healthy lifestyle, and particularly worsening dietary habits) could contribute to the lack of decrease in occurrence of lower GI events in RA43. In contrast, timely treatment of upper GI disease, minimizing exposure to glucocorticoids, avoiding smoking, and vigilant screening for lower GI disease may be beneficial for decreasing the incidence of lower GI disease in RA. These issues require further investigation.
Our study has particular strengths; it is the first to report longterm trends in incidence of both upper and lower GI events with specification of GI diagnoses, GI surgeries, and GI-related hospitalizations in RA versus non-RA subjects. The longitudinal population-based design with extensive followup data obtained through the unique medical record linkage system (the REP) and the use of complete inpatient and outpatient medical records strengthen our work by providing complete ascertainment of study outcomes for all study subjects.
As in any retrospective study, only those events that came to medical attention were included. However, the vast majority of clinically important GI events are likely to come to medical attention. While there may be a chance of misclassification of GI events, the detailed review of each questionable case by our multidisciplinary team including clinicians specializing in rheumatology (ELM, EM) and gastroenterology (NJT) makes this unlikely. The definition of alcohol abuse in our study was based on physician’s diagnosis, and thus may not reflect the true extent of alcohol abuse, as some patients may never be diagnosed. Because of the long followup and completeness of the medical records, we believe the majority of cases of alcohol abuse, including cases of alcohol abuse complicated by serious GI events, were recorded reliably.
Because of the retrospective longitudinal design of the study and the use of medical records, information required for calculation of RA disease activity scores was not available. To minimize this shortcoming the results of all clinically performed ESR tests were used to assess the effect of inflammatory burden on serious GI events in RA over time.
Information on the use of over-the-counter medications (particularly NSAID and ASA) was not available for non-RA subjects; therefore, we were unable to analyze the effects of these medications on GI events in the non-RA cohort. Statistical power may be limited in some analyses, particularly assessing some rare events (e.g., lower GI perforations, subcategories of drug-induced colitis). These results should be interpreted with caution.
Our study shows increased risk of incident upper and lower GI events in RA versus non-RA subjects and increased GI-related mortality in those with RA. While the incidence of upper GI events decreased significantly, particularly in patients with RA, there was no apparent decrease in lower GI events in RA over time. Patients with RA who were exposed to systemic glucocorticoids, those with prior abdominal surgery or underlying upper GI disease, and smokers appear to be at increased risk of lower GI events. Favorable trends in upper GI disease should trigger more research into lower GI disease in RA. Studies to develop effective approaches for prevention and treatment of lower GI disease in RA are needed.
Acknowledgments
The authors are grateful to Ruchi G. Sharma, MS, for data analysis, to Cynthia L. Nosek, RN, Lorna Stevens, RN, and Denise M. Herman, RN, for performing data abstraction, and to Konnie B. Bicknese for study coordination.
Supported by Roche Pharmaceuticals; and by grants from the National Institutes of Health, NIAMS (R01 AR46849) and the Rochester Epidemiology Project (R01 AG034676 from the National Institute on Aging).
References
- 1.Gabriel SE, Crowson CS, O’Fallon WM. Comorbidity in arthritis. J Rheumatol. 1999;26:2475–9. [PubMed] [Google Scholar]
- 2.Koivuniemi R, Paimela L, Suomalainen R, Piirainen H, Karesoja M, Helve T, et al. Causes of death in patients with rheumatoid arthritis autopsied during a 40-year period. Rheumatol Int. 2008;28:1245–52. doi: 10.1007/s00296-008-0685-6. [DOI] [PubMed] [Google Scholar]
- 3.Michaud K, Wolfe F. Comorbidities in rheumatoid arthritis. Best Pract Res Clin Rheumatol. 2007;21:885–906. doi: 10.1016/j.berh.2007.06.002. [DOI] [PubMed] [Google Scholar]
- 4.Mikuls TR. Co-morbidity in rheumatoid arthritis. Best Pract Res Clin Rheumatol. 2003;17:729–52. doi: 10.1016/s1521-6942(03)00041-x. [DOI] [PubMed] [Google Scholar]
- 5.Schneider A, Merikhi A, Frank BB. Autoimmune disorders: Gastrointestinal manifestations and endoscopic findings. Gastrointest Endosc Clin N Am. 2006;16:133–51. doi: 10.1016/j.giec.2006.01.013. [DOI] [PubMed] [Google Scholar]
- 6.Wolfe F, Mitchell DM, Sibley JT, Fries JF, Bloch DA, Williams CA, et al. The mortality of rheumatoid arthritis. Arthritis Rheum. 1994;37:481–94. doi: 10.1002/art.1780370408. [DOI] [PubMed] [Google Scholar]
- 7.Lanas A, Perez-Aisa MA, Feu F, Ponce J, Saperas E, Santolaria S, et al. A nationwide study of mortality associated with hospital admission due to severe gastrointestinal events and those associated with nonsteroidal antiinflammatory drug use. Am J Gastroenterol. 2005;100:1685–93. doi: 10.1111/j.1572-0241.2005.41833.x. [DOI] [PubMed] [Google Scholar]
- 8.Bannwarth B. Safety of the nonselective NSAID nabumetone: Focus on gastrointestinal tolerability. Drug Saf. 2008;31:485–503. doi: 10.2165/00002018-200831060-00004. [DOI] [PubMed] [Google Scholar]
- 9.Dijkmans BA, Janssen M, Vandenbroucke JP, Lamers CB. NSAID-associated upper gastrointestinal damage in patients with rheumatoid arthritis. Scand J Gastroenterol Suppl. 1995;212:105–8. doi: 10.3109/00365529509090308. [DOI] [PubMed] [Google Scholar]
- 10.Fries JF, Bruce B. Rates of serious gastrointestinal events from low dose use of acetylsalicylic acid, acetaminophen, and ibuprofen in patients with osteoarthritis and rheumatoid arthritis. J Rheumatol. 2003;30:2226–33. [PubMed] [Google Scholar]
- 11.Goldstein JL, Eisen GM, Agrawal N, Stenson WF, Kent JD, Verburg KM. Reduced incidence of upper gastrointestinal ulcer complications with the COX-2 selective inhibitor, valdecoxib. Aliment Pharmacol Ther. 2004;20:527–38. doi: 10.1111/j.1365-2036.2004.02118.x. [DOI] [PubMed] [Google Scholar]
- 12.Janssen M, Dijkmans BA, van der Sluys FA, van der Wielen JG, Havenga K, Vandenbroucke JP, et al. Upper gastrointestinal complaints and complications in chronic rheumatic patients in comparison with other chronic diseases. Br J Rheumatol. 1992;31:747–52. doi: 10.1093/rheumatology/31.11.747. [DOI] [PubMed] [Google Scholar]
- 13.Wolfe F, Hawley DJ. The comparative risk and predictors of adverse gastrointestinal events in rheumatoid arthritis and osteoarthritis: A prospective 13 year study of 2131 patients. J Rheumatol. 2000;27:1668–73. [PubMed] [Google Scholar]
- 14.Fries JF, Murtagh KN, Bennett M, Zatarain E, Lingala B, Bruce B. The rise and decline of nonsteroidal antiinflammatory drug-associated gastropathy in rheumatoid arthritis. Arthritis Rheum. 2004;50:2433–40. doi: 10.1002/art.20440. [DOI] [PubMed] [Google Scholar]
- 15.Steen KS, Nurmohamed MT, Visman I, Heijerman M, Boers M, Dijkmans BA, et al. Decreasing incidence of symptomatic gastrointestinal ulcers and ulcer complications in patients with rheumatoid arthritis. Ann Rheum Dis. 2008;67:256–9. doi: 10.1136/ard.2006.068015. [DOI] [PubMed] [Google Scholar]
- 16.Combe B, Swergold G, McLay J, McCarthy T, Zerbini C, Emery P, et al. Cardiovascular safety and gastrointestinal tolerability of etoricoxib vs diclofenac in a randomized controlled clinical trial (The MEDAL study) Rheumatology. 2009;48:425–32. doi: 10.1093/rheumatology/kep005. [DOI] [PubMed] [Google Scholar]
- 17.Curtis JR, Xie F, Chen L, Spettell C, McMahan RM, Fernandes J, et al. The incidence of gastrointestinal perforations among rheumatoid arthritis patients. Arthritis Rheum. 2011;63:346–51. doi: 10.1002/art.30107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Emery P, Keystone E, Tony HP, Cantagrel A, van Vollenhoven R, Sanchez A, et al. IL-6 receptor inhibition with tocilizumab improves treatment outcomes in patients with rheumatoid arthritis refractory to anti-tumour necrosis factor biologicals: Results from a 24-week multicentre randomised placebo-controlled trial. Ann Rheum Dis. 2008;67:1516–23. doi: 10.1136/ard.2008.092932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gout T, Ostor AJ, Nisar MK. Lower gastrointestinal perforation in rheumatoid arthritis patients treated with conventional DMARDs or tocilizumab: A systematic literature review. Clin Rheumatol. 2011;30:1471–4. doi: 10.1007/s10067-011-1827-x. [DOI] [PubMed] [Google Scholar]
- 20.Laine L, Curtis SP, Langman M, Jensen DM, Cryer B, Kaur A, et al. Lower gastrointestinal events in a double-blind trial of the cyclo-oxygenase-2 selective inhibitor etoricoxib and the traditional nonsteroidal anti-inflammatory drug diclofenac. Gastroenterology. 2008;135:1517–25. doi: 10.1053/j.gastro.2008.07.067. [DOI] [PubMed] [Google Scholar]
- 21.Laine L, Smith R, Min K, Chen C, Dubois RW. Systematic review: The lower gastrointestinal adverse effects of non-steroidal anti-inflammatory drugs. Aliment Pharmacol Ther. 2006;24:751–67. doi: 10.1111/j.1365-2036.2006.03043.x. [DOI] [PubMed] [Google Scholar]
- 22.Maradit Kremers H, Crowson CS, Gabriel SE. Rochester Epidemiology Project: A unique resource for research in the rheumatic diseases. Rheum Dis Clin North Am. 2004;30:819–34. vii. doi: 10.1016/j.rdc.2004.07.010. [DOI] [PubMed] [Google Scholar]
- 23.Melton LJ., 3rd History of the Rochester Epidemiology Project. Mayo Clin Proc. 1996;71:266–74. doi: 10.4065/71.3.266. [DOI] [PubMed] [Google Scholar]
- 24.Maradit Kremers H, Myasoedova E, Crowson CS, Savova G, Gabriel SE, Matteson EL. The Rochester Epidemiology Project: Exploiting the capabilities for population-based research in rheumatic diseases. Rheumatology. 2011;50:6–15. doi: 10.1093/rheumatology/keq199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31:315–24. doi: 10.1002/art.1780310302. [DOI] [PubMed] [Google Scholar]
- 26.Turesson C, Jacobsson L, Bergstrom U. Extra-articular rheumatoid arthritis: Prevalence and mortality. Rheumatology. 1999;38:668–74. doi: 10.1093/rheumatology/38.7.668. [DOI] [PubMed] [Google Scholar]
- 27.Cox D. Some simple approximations for Poisson variates. Biometrika. 1953;40:354–60. [Google Scholar]
- 28.McCullagh P, Nelder JA. Generalized linear models. New York: Chapman and Hall; 1989. [Google Scholar]
- 29.Gabriel SE, Crowson CS, Kremers HM, Doran MF, Turesson C, O’Fallon WM, et al. Survival in rheumatoid arthritis: A population-based analysis of trends over 40 years. Arthritis Rheum. 2003;48:54–8. doi: 10.1002/art.10705. [DOI] [PubMed] [Google Scholar]
- 30.Chan FK, Lanas A, Scheiman J, Berger MF, Nguyen H, Goldstein JL. Celecoxib versus omeprazole and diclofenac in patients with osteoarthritis and rheumatoid arthritis (CONDOR): A randomised trial. Lancet. 2010;376:173–9. doi: 10.1016/S0140-6736(10)60673-3. [DOI] [PubMed] [Google Scholar]
- 31.Hoes JN, Jacobs JW, Verstappen SM, Bijlsma JW, Van der Heijden GJ. Adverse events of low- to medium-dose oral glucocorticoids in inflammatory diseases: A meta-analysis. Ann Rheum Dis. 2009;68:1833–8. doi: 10.1136/ard.2008.100008. [DOI] [PubMed] [Google Scholar]
- 32.Hutchinson D, Lynch M. Sigmoid diverticular abscess perforation in 2 patients with rheumatoid arthritis treated with high dose corticosteroids. A cautionary tale. J Rheumatol. 2001;28:1935–6. [PubMed] [Google Scholar]
- 33.Laine L, Connors LG, Reicin A, Hawkey CJ, Burgos-Vargas R, Schnitzer TJ, et al. Serious lower gastrointestinal clinical events with nonselective NSAID or coxib use. Gastroenterology. 2003;124:288–92. doi: 10.1053/gast.2003.50054. [DOI] [PubMed] [Google Scholar]
- 34.Mpofu S, Mpofu CM, Hutchinson D, Maier AE, Dodd SR, Moots RJ. Steroids, non-steroidal anti-inflammatory drugs, and sigmoid diverticular abscess perforation in rheumatic conditions. Ann Rheum Dis. 2004;63:588–90. doi: 10.1136/ard.2003.010355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Myllykangas-Luosujarvi R. Diverticulosis — a primary cause of life-threatening complications in rheumatoid arthritis. Clin Exp Rheumatol. 1995;13:79–82. [PubMed] [Google Scholar]
- 36.Matteson EL, Yachyshyn V, Yachyshyn J, O’Fallon WM. Trends in hospitalizations for gastrointestinal bleeding among patients with rheumatoid arthritis in Rochester, Minnesota, 1950–1991. J Rheumatol. 1995;22:1471–7. [PubMed] [Google Scholar]
- 37.Lanas A. A review of the gastrointestinal safety data — A gastroenterologist’s perspective. Rheumatology. 2010;49(Suppl 2):ii3–10. doi: 10.1093/rheumatology/keq058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lanas A, Garcia-Rodriguez LA, Polo-Tomas M, Ponce M, Alonso-Abreu I, Perez-Aisa MA, et al. Time trends and impact of upper and lower gastrointestinal bleeding and perforation in clinical practice. Am J Gastroenterol. 2009;104:1633–41. doi: 10.1038/ajg.2009.164. [DOI] [PubMed] [Google Scholar]
- 39.Lanza FL. A guideline for the treatment and prevention of NSAID-induced ulcers. Members of the Ad Hoc Committee on Practice Parameters of the American College of Gastroenterology. Am J Gastroenterol. 1998;93:2037–46. doi: 10.1111/j.1572-0241.1998.00588.x. [DOI] [PubMed] [Google Scholar]
- 40.Decker D, Tolba R, Springer W, Lauschke H, Hirner A, von Ruecker A. Abdominal surgical interventions: Local and systemic consequences for the immune system — a prospective study on elective gastrointestinal surgery. J Surg Res. 2005;126:12–8. doi: 10.1016/j.jss.2005.01.006. [DOI] [PubMed] [Google Scholar]
- 41.Russell RI. Defining patients at risk of non-steroidal anti-inflammatory drug gastropathy. Ital J Gastroenterol Hepatol. 1999;31 (Suppl 1):S14–8. [PubMed] [Google Scholar]
- 42.Carlens C, Hergens MP, Grunewald J, Ekbom A, Eklund A, Hoglund CO, et al. Smoking, use of moist snuff, and risk of chronic inflammatory diseases. Am J Respir Crit Care Med. 2010;181:1217–22. doi: 10.1164/rccm.200909-1338OC. [DOI] [PubMed] [Google Scholar]
- 43.King DE, Mainous AG, 3rd, Carnemolla M, Everett CJ. Adherence to healthy lifestyle habits in US adults, 1988–2006. Am J Med. 2009;122:528–34. doi: 10.1016/j.amjmed.2008.11.013. [DOI] [PubMed] [Google Scholar]