Summary
Pulmonary emphysema, together with chronic bronchitis is a part of chronic obstructive pulmonary disease (COPD), which is one of the leading causes of death in the United States and worldwide. There are many methods to diagnose emphysema. Unfortunately many of them, for example pulmonary function tests (PFTs), clinical signs and conventional radiology are able to detect emphysema usually in its late stages when a great portion of lung parenchyma has been already destroyed by the disease. Computed tomography (CT) allows for early detection of emphysema. CT also makes it possible to quantify the total amount of emphysema in the lungs which is important in order to precisely estimate the severity of the disease. Those abilities of CT are important in monitoring the course of the disease and in attempts to prevent its further progression. In this review we discuss currently available methods for imaging emphysema with emphasis on the quantitative assessment of emphysema. To date, quantitative methods have not been widely used clinically, however, the initial results of several research studies regarding this subject are very encouraging.
Keywords: emphysema, COPD, HRCT, spiral CT, densitometry, quantitative assessment
Background
According to the National Heart, Lung, and Blood Institute, emphysema is a “condition of the lung characterized by abnormal, permanent enlargement of the airspaces distal to the terminal bronchiole, accompanied by destruction of their walls” [1].
Pulmonary emphysema, together with chronic bronchitis is a part of COPD – chronic obstructive pulmonary disease. A US Centers for Disease Control and Prevention (CDC) Morbidity Mortality Weekly Report study of the National Vital Statistics System reported an age-standardized death rate from COPD in the United States for adults older than 25 years of 64.3 deaths per 100,000 population [2]. COPD remains one of the leading causes of mortality in the USA and worldwide. The incidence of emphysema seems to have a strong correlation with the cigarette smoking. In one of the studies on this subject Remy-Jardin et al. showed that in smokers who underwent pulmonary lobe resection because of the presence of nodules there was a high incidence of emphysema seen in pathologic specimen, even with no apparent clinical symptoms and with no obstruction of the airways [3]. The National Health Interview Survey reports the prevalence of emphysema at 1.8% [4].
There are two main types of emphysema, depending on morphologic appearance and areas of lung parenchyma destruction in regards to a secondary pulmonary lobule. The centriacinar (centrilobular) type is usually associated with destruction of alveolar walls in the central portion of acinus, it is more often found in upper lung zones (upper lobes – posterior and apical segments or superior segments of lower lobes) and is commonly associated with cigarette smoking [5]. The panacinar (panlobular) type is recognized as a destruction involving all portions of the lobule out to the periphery. This form is often found in alfa-1-antitrypsin deficiency and has a marked predilection to lower lobes [5]. In more severe disease these two subtypes can appear side by side but usually one of them predominates and the distinction between these two can be sometimes very difficult or even impossible. Additionally two more subtypes of emphysema have been identified. Paraseptal form which can appear as an isolated finding but is usually found together with severe centriacinar or panacinar emphysema. This type occurs in the periphery of the lung, close to the pleura or interlobular septa. Its most characteristic feature is presence of bullae – single or multiple, well-demarcated, air-filled cysts of 1 cm or more in diameter and smooth, thin (less than 1mm) walls. Another subtype is called paracicatricial and this form is associated with areas of scarring in the lung.
Unfortunately, early emphysema is usually asymptomatic for a long time and more than 30% of the total lung parenchyma has to be destroyed in order for the clinical symptoms to be present or changes in pulmonary function tests (PFTs) to appear [6]. That is why in order to understand the presence and severity of emphysema all clinical features, laboratory data and imaging studies should be gathered and assessed together.
In this review we discuss currently available and used methods of visualization of emphysema with emphasis on newer and promising approaches to quantitative analysis of pulmonary emphysema in vivo, especially by using widely available Computed Tomography (CT).
Chest Radiography
Chest X-ray (CXR) has been the most routinely used method for diagnostic imaging of the lungs in patients with COPD.
The most specific feature of emphysema on CXR is the presence of bullae. The more severe the emphysema, the more likely radiological diagnosis will be made based just on the CXR. The characteristic features of emphysema which can be seen on the routine CXR include: lowering and flattening of diaphragm, increased antero-posterior diameter of the chest (retrosternal space of more than 2.5 cm in front of the ascending aorta on the lateral view), presence of bullae/blebs, “saber-sheath trachea”, “increased lung markings” (probably due to bronchial wall thickening associated with inflammatory changes) or the paucity of pulmonary vessels in the peripheral lungs. Emphysema with increased markings is found especially in patients with cor pulmonale. The presence of peripheral arterial deficiency has a low sensitivity but high specificity [5,7]. In the study conducted by Lohela et al. [8] the researchers compared the signs of emphysema on a routine chest radiograph to the post-mortem evidence of the disease. The main conclusion from the study was that if two or more of the signs of emphysema (according to criteria established by the authors) were present on CXR, a total accuracy of 77% in detecting the disease was achieved. The mean number of radiologic signs increased with the severity of morphologic emphysema. Overall, the sensitivity of chest radiography is low, especially in mild to moderate disease.
Computed Tomography
With the shortcomings of conventional radiography, CT has become the more reliable and widely used and investigated imaging tool for the assessment of pulmonary emphysema. Currently, CT, and specifically high-resolution CT (HRCT) are the methods of choice to diagnose emphysema in vivo. Because of higher spatial resolution HRCT is a preferred tool to visually detect especially small areas of emphysema [9].
Centrilobular emphysema which more often involves upper parts of the lungs, is usually described as the irregular, small, round or confluent areas of low attenuation interspersed with a normal lung. The localization near the center of a secondary pulmonary lobule is also typical. Panacinar emphysema is found mainly in the lower zones of the lungs. It is usually seen as the diffuse areas of low attenuation with little normal lung tissue between the lesions. In severe panacinar emphysema diffuse/ widespread areas of lung parenchyma with low attenuation and significantly decreased vascular markings might be seen. This picture is also known as a “simplification of lung parenchyma” and when present helps to separate normal lung parenchyma from emphysema. Mild or moderate panacinar emphysema might be often very difficult, or even impossible to distinguish from adjacent normal lung parenchyma on CT scans [7].
Quantitative Assessment by CT (QCT)
CT proved to be not just a great method to detect emphysema but also a tool to estimate the extent and severity of this disease. The qualitative subjective evaluation of emphysema performed by a radiologist can be augmented with the objective quantitative assessment by CT, by density measurements or more recently, by postprocessing of CT data and texture analysis. For subjective assessment of emphysema HRCT is of great use, but when it comes to objective densitometric studies, the combination of thin sections and the high spatial frequency algorithm (also known as the edge-enhancing, sharp algorithm) results in poor density resolution, and therefore should be avoided [9].
Initially the diagnosis of pulmonary emphysema on CT scans was greatly dependent on subjective, visual assessment of a radiologist.
Multiple studies, for example by Bergin et al. [10], Hruban et al. [11], Miller et al. [12] and Kuwano et al. [13], were conducted to compare the presence and the degree of emphysema on CT images with corresponding pathological data. They showed that there was a good correlation between radiological visual assessment and pathological report at the same/comparable lung fragment. With using various section thickness (1–10 mm), depending on the study, the correlation coefficients ranged from 0.6 to 0.8. However, Miller et al. also showed that it could be difficult to detect the earliest forms of pulmonary emphysema on CT scans because of the small sizes of those lesions, often less than 5 mm in diameter. Therefore, they were usually missed on CT scans. This study also reported that qualitative evaluation of CT scans consistently underestimated the extent of the disease. Intra- and interobserver variations in visual assessment were significant and limited the reliability of this method. The involvement of “the human factor” is frequently responsible for over- or under- estimation of the total amount of emphysema in the lungs. It is also very difficult, based on information from CT, to estimate the total exact amount of emphysematous lung parenchyma and thus to assess visually the progression of the disease.
This spurred the research looking for more objective methods for quantifying the total percentage of the lungs affected by emphysematous destruction. Since, the visual assessment of emphysema is based on the presence of localized or diffuse areas of perceived low attenuation, which differ considerably from the remaining parenchyma, this difference in lung attenuation can be reliably quantitated by QCT.
Initially quantitative densitometry of the lung was dependent on the measuring the mean value of pixel density in a particular area drawn in the region of interest (ROI) on each scan [5]. The normal lung density during full inspiration is between −750 to −850 Hounsfield units (HU) (on 10 mm slices) and emphysema was considered to be present for values falling more than 2 SD below normal average density.
Based on the analysis of the frequency of attenuation values in the lung, the histogram can be created which displays the data as a distribution curve. The histogram represents the frequency distribution of pixels/voxels at different attenuation intervals. In the normal lung (Figure 1), the attenuation values are most frequently distributed between −750 and −850 HU, with the peak attenuation centered at −789 HU. The abnormal accumulation of air in the non-functioning emphysematous lung contributes to decreased lung density with shift of the lung attenuation values towards lower HU levels, below −900 (Figure 2). That makes the shift of the curve from the normal attenuation interval to the left which provides a visual index of the degree of emphysema. At that point other useful densitometric parameters can be calculated: mean lung attenuation (MLA) which is the average density of all pixels in the image representing the entire lung; the relative area of emphysema (RA)/emphysema index that is the area of the lung under the curve of the histogram occupied by attenuation values lower than predetermined threshold [14,15]. In different studies, different thresholds were employed and validated; the so called ‘percentile point’/’percentile density’ was described as a cutoff density value in HU, for which a predetermined percentage of all pixels has a lower value [16,17].
Figure 1.

Histogram in normal lung. The attenuation values are most frequently distributed between −750 and −850 HU.
Figure 2.

Histogram in emphysema. Emphysematous lung contributes to decreased lung density with the shift of the lung attenuation values towards lower HU levels.
One of the first studies investigating the correlation between CT imaging (using the attenuation density values) and pathologic data in pulmonary emphysema was the study published in 1984 by Hayhurst et al. [7,18]. In a group of patients the author compared the CT images of patients’ lungs with measurements of the density of lung parenchyma with the resected lung specimens. The final conclusion of this study, after assessing the range of attenuation distribution values on CT scans, was that in patients with emphysema, there were more attenuation values between −900 HU and −1000 HU than in normal individuals, which means that if a histogram plot was made of frequency against pixel density (HU), the distribution curve of these densities in emphysema was shifted toward lower attenuation values comparing to people without the disease (Figures 1, 2).
The histogram provides a means for interpreting pulmonary densitometry when the predicted normal and observed curves are compared. The total lung volume can be assessed with the thresholds −1024 and −200 HU. Within the total lung volume, the functional lung parenchyma has the attenuation values in range −700 to −900 HU (Figure 3) and the non-functional lung parenchyma between −1024 and −900 HU represents emphysema. The lung with attenuation values greater than −700 HU has reduced participation in gas exchange. The lung volumes within these three attenuation intervals can be calculated to determine the contribution of each volume to the total lung function. The greater the proportion of lung attenuation values below −900 HU, the more extensive the emphysema.
Figure 3.

Emphysema. The total lung volume is assessed with the thresholds –1024 and –200 HU. Within this total lung volume, the functional lung parenchyma has the attenuation in range –700 to –900 HU. The non-functional lung parenchyma between –1024 and –900 HU represents emphysema.
The next step was taken in the study by Muller et al. [14] where the investigators used a CT scanner with a standard software program called “density mask”. That program highlights pixels with densities within a desired range, automatically calculates the area of highlighted pixels and then is able to express them as a percentage of the total pixels in a given area. Because the density of the emphysematous lung parenchyma is abnormally low, by defining these areas of abnormally low attenuation values Muller et al. were able to quantify the extent and severity of emphysema. In that study the authors compared “density masks” and mean lung attenuation with subjective visual assessment and pathological score of resected lung specimen in order to determine the suitability of this program for assessment of emphysema. The best correlations were observed by highlighting all pixels with attenuation lesser or equal to −910 HU. The correlations between mask density score and the mask density percent area at the value lesser or equal to −910 HU with pathological score were 0.94 and 0.89, respectively. In that particular study −910 HU appeared to be the most reliable threshold and the best discrimination point between normal and emphysematous lung parenchyma. However, this value may vary for different scanners and techniques but once estimated for particular settings it seems to remain stable. The study showed that the density mask allows objective quantification of the total lung volume showing emphysema on CT scans and the percentage of lung involved with emphysema.
Subsequent studies added even more information and enabled even better use of those methods.
According to work undertaken by Gevenois et al. [15,19] −950 HU is the cut-off value for emphysematous lung parenchyma on CT scans which corresponds closely with macroscopic pathological measurements. Thresholds lower than −950 HU underestimated the presence of emphysema, whereas greater than −950 HU overestimated the disease. The researchers reported that based on their studies the relative area of the lung with attenuation values lower than −950HU on HRCT thin – sections scans taken at a full inspiration is very reliable way to objectively quantify macroscopic emphysema in vivo.
Currently, even more elaborated methods in quantifying emphysema are being investigated. These computer-assisted methods rely on the advanced mathematic approaches called fractal dimensions. They help to better characterize examined tissue and thus allow easier differentiation between normal and emphysematous lung parenchyma. Studies conducted by Uppaluri et al. [20] and Mishima et al. [21] showed that fractal analysis is very promising, however this method still requires more research.
Comparison between CT quantification and PFTs
Pulmonary function tests (PFTs) are very important in the diagnosis and monitoring of emphysema. Their low costs, well-established correlation with clinical profile, reproducibility of results, the ability to predict prognosis and non-utilization of ionization radiation give PFTs an advantage over the other methods [22]. However, there are several studies that show that PFTs have limitations especially in patients with less severe emphysema. The insensitivity of PFTs in detection early stages of emphysema was confirmed in the study conducted by Sanders et al. [23]. In this study the signs of emphysema on CT scans were observed not only in about 96% of patients who met the functional criteria of emphysema (decreased diffusing capacity of the lung for carbon monoxide (DLCO) and the presence of obstructive lung disease based on decreased forced expiratory volume in 1 second (FEV1) and/or increased residual volume (RV)) but also in 69% of smokers with completely normal DLCO. On the basis of these results authors concluded that CT seems to be more precise in the diagnosis of the mild forms of emphysema. The associated insensitivity of PFTs probably can be explained by the lack of obstructive deficits (limitation of air-flow) in these patients, although the reduction of lung elastic recoil and increase in the lung size is present. Several other studies were conducted in order to compare the quantitative CT analysis in emphysema with PFTs [24–28]. The evident correlation between PFTs and the total amount of emphysematous parenchyma has been shown as well as between PFTs and the distribution of these changes in the lungs. Emphysema in the lower lung zones seems to have higher correlations with DLCO comparing to lesions located in the upper parts. It is true even if the upper parts of the lungs are much more severely affected by the disease. Gurney et al. tried to explain this phenomenon stating that probably upper lung zones are relatively silent region where great emphysematous destruction can be made before affecting functional tests whereas the presence and degree of emphysema in the lower lung zones has a stronger influence on the PFTs, because of loss of perfusion to the lower lobes. It also has been noticed that centrally located emphysema has higher correlations with PFTs than changes on the periphery of the lungs. Emerging 3D reconstructions of entire image data sets from spiral CT scans allowed more robust data processing with elimination of “regional sampling problems” created by investigation of limited number of slices. Through the comprehensive evaluation of the extent of emphysema, the correlation between CT analysis and PFTs could become even more precise [29].
As it was mentioned before the histogram position in respect to the attenuation intervals, its shape and symmetry, as well as its peak provides important visual clues to the extent of emphysema.
The normal lung HRCT of a 19-year-old female is shown on Figure 4A. The histogram of this patient’s lung at −700 HU threshold (Figure 4B) demonstrates that in the normal lung the attenuation values are most frequently distributed between −750 and −850 HU. Only minimal volume of 10.18 cc (mostly representing the trachea) was detected below −900 HU threshold. The emphysema index of this patient’s lung was estimated to be 0.2%.
Figure 4.
Normal HRCT (A) and histogram (B) of a 19 year old female.
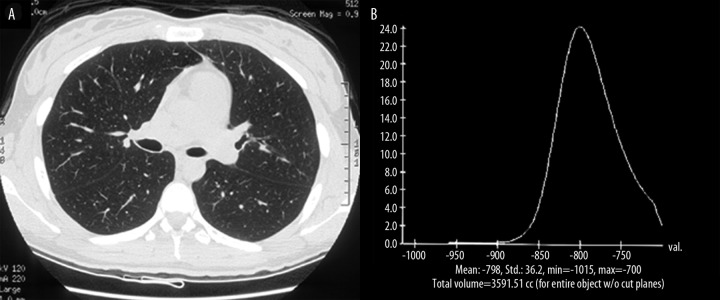
On the other hand in the lungs with emphysema, the histogram is shifted to the left, broad-based, frequently asymmetric, and the peak is located below −850 HU. Figure 5A shows the HRCT image of a 62-year-old male with history of COPD and centrilobular and bullous emphysema.The histogram at −700 HU threshold (Figure 5B) presents a moderate shift of the histogram peak to the left with significant increase in frequency of voxels with attenuation below −900 HU, representing a total volume of 1453.68 cc. The emphysema index of this patient’s lung is 22.5% and his FEV1/FVC demonstrated 53% of the predicted value.
Figure 5.
HRCT(A) and histogram (B) of a 62 year old male with COPD and centrilobular and bullous emphysema.
Figure 6A,B reveal the HRCT and histogram of a 64-year-old male with COPD and history of the left upper lobectomy. The histogram of this patient’s lung at −700 HU threshold (Figure 6B) shows an extreme shift of the curve to the left, with the peak frequency distribution of lung voxels at attenuation level of −950 HU. The volume of the lung represented by voxels with attenuation below −900 HU is 2269.72 cc. His emphysema index was 41.9% and PFTs showed FEV1/FVC=31% of the predicted value.
Figure 6.
HRCT (A) and histogram (B) of a 64 year old male with COPD, after left upper lobectomy.
These cases illustrate that there is a correlation between QCT of emphysema (e.g. histogram shape and emphysema index) and PFTs changes.
Comparison Between Subjective and Objective Methods of QCT
Both the subjective and objective methods of quantitative assessment of emphysema by CT have their pros and cons. Without doubt the major advantage of objective quantification based on computer-assisted methods is the reproducibility of the technique between different institutions and between different readers regardless of their experience. On the other hand, subjective methods are much easier to perform and do not require such an expensive software [30]. The comparison between subjective and objective methods of assessment of emphysema was conducted by Bankier et al. [31]. He compared subjective visual grading of pulmonary emphysema with macroscopic morphometry and objective CT quantification. The study concluded that there was a weaker relationship between subjective visual assessment and the macroscopic data in comparison to the correlation between objective computed analysis and the morphological extent of emphysema. That is why it was recommended that subjective visual grading should be supplemented with objective methods to achieve more precise, reader-independent quantification of emphysema.
Technical Considerations in QCT
The density of the lung parenchyma within a given voxel is determined by the amount of air, tissue, interstitial fluid and blood [32].
There are multiple factors which might influence CT densitometry (30):
Respiratory status of the patient – inspiration vs. expiration scans; some studies have investigated the possibility of respiratory gating while performing CT scanning in order to avoid possible errors due to differences in the lung volumes when breathing. Using spirometrically triggered CT scanning, it was demonstrated [33] that in patients with emphysema the level of inspiration does not significantly influence the lung densitometry. However in healthy persons, the mean change in densitometric parameters between 90% and 10% vital capacity is significant. Expiratory technique plays an important role in differentiating normal subjects from patients with chronic bronchitis in which air trapping can be detected only at 10% vital capacity. Inspiratory CT was found to be equal to expiratory CT in the ability to quantify abnormally low attenuation of the lung with pulmonary emphysema [34].
Patient age – a study conducted by Soeijima et al. [35] showed that the percentage of RA960 (the relative area of lung occupied by attenuation values less than −960 HU) can increase with age, at least in the middle and lower lung zones.
CT acquisition parameters – for example collimation, the amount of kilovoltage, reconstruction algorithm. In one study Mishima et al. [36] reported that three 2-mm thick CT sections, acquired with 200 or more mA tube current, are the most appropriate parameters to assess pulmonary emphysema.
Slice thickness, number and location of sections – because of the heterogeneous distribution of the emphysema in the lungs, precise quantitative evaluation of the total lung emphysema cannot be obtained from the analysis of just a single or selected CT slices. However, concerns about radiation exposure make less slices more desirable than performing measurements in the whole lungs. Yet there are no established recommendations in regards to the minimal number of CT slices needed for accurate assessment of the severity of emphysema and the modern multidetector CT (MDCT) scanners rapidly acquire the entire lung volume.
In spiral CT and multidetector CT (MDCT), the images are acquired during concurrent transport of the patient through the CT gantry at the constant speed when data is being collected continuously over multiple gantry rotations/multiple detectors. The greatest advantages of spiral CT and MDCT is their ability to image the entire thorax during one single breath-hold. Since the acquisition of the entire volume of tissue is performed, density of three-dimensional voxels (volume elements) rather than two-dimensional pixels can be calculated. Using data collected with contemporary CT various 3D reconstructions can be created, lung volumes measured and also, by interpreting the frequency distribution curve of attenuation values within these lung volumes, the quantification of different lung disorders can be made. Specific protocols can be set-up with various attenuation thresholds to post-process the acquired data in an automated fashion to calculate specific volumes of interest and to modify the object selection parameters for additional threshold range adjustments or subtractions. MDCT and 3D techniques can be particularly useful thanks to their ability to analyze the total lung in order to detect and assess the severity of the disease with heterogenous distribution among the lungs/lobes.
Intravenous contrast administration – even though intravenous contrast increases the attenuation of normal lung parenchyma, it does not have a substantial effect on attenuation of the lung with emphysema. Emphysematous destruction results in hypovascular lung regions, therefore the influence of IV contrast administration is negligible in those areas. Thin collimation and contrast may affect the absolute attenuation numbers when a single slice is analyzed. However, with densitometry applied to the total lung volume, the quantitative assessment of emphysema is not affected [22,32].
CT quantification vs. Nuclear Medicine Studies
Scintigraphic studies have been useful in acquiring the information about the most severely affected by emphysema areas in the lung, as documented by some research conducted on this subject [37–39]. Studies showed that functional imaging with ventilation and perfusion lung scintigraphy (VQ scan) can be used to assess regional perfusion, ventilation and possible gas trapping [40]. Quantification is generally performed by dividing the lung into 3 zones: upper, middle and lower and then by calculating the percentage of ventilation and perfusion for each particular zone. A high-quality perfusion study is easily obtained with 99mTc-MAA. For more accurate estimate of the true ventilation the agent krypton-81m (81mKr) and Technegas can be used, however easier available Xenon-133 (133Xe) is used more frequently. In the future single photon emission computed tomography (SPECT) ventilation scans might be used instead of planar (2-dimensional) 133Xe imaging. Preliminary studies have shown that SPECT scanning after ventilation with Technegas and perfusion with 99mTc-MAA can provide detailed data about the most affected by the emphysema lung segments [41].
To date, CT still remains superior to scintigraphy in assessing the extent of the lung parenchyma destruction. However, scintigraphy might complement the CT data, especially regarding some additional physiologic information, for example on airway obstruction and regional gas trapping.
MRI in Emphysema
Magnetic Resonance (MR) is another modality that is being investigated for imaging and quantitation of emphysema. Until recently the lungs were not the most favorable organ to be examined using the magnetic field, but the use of hyper-polarized noble gases as MR contrast agents has enabled better visualization of lung parenchyma and has given researchers more detailed understanding of the structure and function of the lung. Hyperpolarized MR Diffusion Weighted Imaging gives information not only regarding static ventilation of the lungs, but also provides data on ventilation dynamics, regional oxygen partial pressure and the apparent diffusion coefficient (ADC) [42]. Among these parameters, ADC is the simplest quantitative marker to interpret.
The principle of DWI MRI with hyperpolarized gases is that diffusion of 3He atoms in the lung is determined by the size of the alveolar space and some studies have shown that in regions of emphysematous lung destruction, the ADC significantly increases in comparison to healthy lungs [43–47]. Because of the high costs and limited supply of 3He, lung morphometry with hyperpolarized 129Xe was taken into consideration as an attractive alternative for clinical use [42,48,49]. Despite some differences between these two gases (lower gyromagnetic ratio, polarization and free diffusion of 129Xe relative to 3He) hyperpolarized 129Xe ADC maps still show a meaningful correlation with PFTs, similar to what was shown using 3He [42]. Diffusion-weighted hyperpolarized MRI is a very sensitive method. It has been shown to be able to detect the early signs of emphysema, even when clinical signs are still absent [47,50]. The quantitative assessment of emphysema with hyperpolarized MR is possible by creating ADC map, that allows assessment of the size of the affected lung airspaces and also allows analysis of the location and severity of emphysema.
Therefore diffusion-weighted hyperpolarized MR could also become a useful imaging option in evaluation of this disease.
Conclusions
To date, quantitative assessment of emphysema using CT has been studied quite thoroughly. Unfortunately, this method is still not routinely used in the clinical practice, mostly because of its high costs, sophisticated image data processing, and radiation exposure. CT quantification appears to be a robust tool to detect and monitor the severity of emphysema, especially in its early stages, with no evident clinical signs of the disease present. Also, precise quantitative evaluation of the extent of emphysema has been employed for treatment purposes, for example to assess the lung parenchyma during planning for lung volume reduction (LVR) procedures. LVR, by either surgical resection or bronchoscopic occlusion, is one of the most effective ways of therapy for patients with severe emphysema [37]. The good results of this treatment are obtained by resection or occlusion of the most damaged and hyper-inflated areas of the lungs. LVR significantly improves ventilation/perfusion ratio in the remaining lung. In order for LVR to work properly it is crucial to precisely identify the most damaged fragments of the lung parenchyma (i.e. areas of gas trapping) [40]. Quantitative analysis of emphysema is not intended to replace direct observation and visual grading of CT scans by radiologists, but rather to complement the diagnostic information obtained by standard visual approach. 3D CT densitometry, a widely available technique for post-processing of CT imaging data, of the lungs may provide a useful, objective, and reproducible tool for the longitudinal follow-up of patients with emphysema and might become important in further defining the natural history of this disease and the rate of the lung destruction. The initial results from the conducted studies are very encouraging. However, the quantitative techniques have not yet been standardized and general guidelines for QCT are yet to be determined before these methods could be routinely used in clinical practice. The Radiologic Society of North America (RSNA) sponsored the Quantitative Imaging Biomarker Alliance (QIBA) that is currently working on validation and standardization of quantitative methods for the assessment of COPD. QIBA's Chronic Obstructive Pulmonary Disease and Asthma Committee is defining the requirements for quantitative metrics for the evaluation of emphysema with CT, to include assessment of the precision and accuracy of phenotyping, and progression measurements in emphysema and asthma, to classify phenotype and assess longitudinal changes as medically meaningful surrogates for health status, and to compare the sensitivity of CT measurements to spirometry and other accepted measures. This will be necessary in order to determine if progressive disease can be detected significantly sooner with quantitative imaging techniques than with currently accepted methods. Once the quantitative metrics are validated, further studies are needed to correlate CT parameters with functional and histopathologic data and patients outcomes..
References:
- 1.Snider GL, Kleinerman JL, Thurlbeck WM, et al. The definition of emphysema. Report of a National Heart, Lung, and Blood Institute, Division of Lung Diseases workshop. Am Rev Respir Dis. 1985;132(1):182–85. doi: 10.1164/arrd.1985.132.1.182. [DOI] [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention Deaths from chronic obstructive pulmonary disease-United States, 2000–2005. MMWR Morb Mortal Wkly Rep. 2008;57(45):1229–32. [PubMed] [Google Scholar]
- 3.Remy-Jardin M, Remy J, Gosselin B, et al. Lung parenchymal changes secondary to cigarette smoking: pathologic-CT correlations. Radiology. 1993;186:643–51. doi: 10.1148/radiology.186.3.8430168. [DOI] [PubMed] [Google Scholar]
- 4.Adams PF, Barnes PM, Vickerie JL. Summary health statistics for the U.S. population: National Health Interview Survey, 2007. Vital Health Stat. 2008 Nov 10;:1–104. [PubMed] [Google Scholar]
- 5.Friedman PJ. Imaging Studies in Emphysema. Proc Am Thorac Soc. 2008;5:494–500. doi: 10.1513/pats.200708-128ET. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gurney JW, Jones KK, Robbins RA, et al. Regional distribution of emphysema: correlation of high-resolution CT with pulmonary function tests in unselected smokers. Radiology. 1992;183:457–63. doi: 10.1148/radiology.183.2.1561350. [DOI] [PubMed] [Google Scholar]
- 7.Thurlbeck W, Muller N. Emphysema: Definition, Imaging, Quantification. AJR. 1994;163:1017–25. doi: 10.2214/ajr.163.5.7976869. [DOI] [PubMed] [Google Scholar]
- 8.Lohela P, Sutinen S, Paakko P, et al. Diagnosis of emphysema on chest radiographs. Rofo. 1984;141(4):395–402. doi: 10.1055/s-2008-1053157. [DOI] [PubMed] [Google Scholar]
- 9.Saher Burhan Shaker. Imaging in chronic obstructive pulmonary disease. Hot Topics in Respiratory Medicine. 2007;3:19–27. [Google Scholar]
- 10.Bergin C, Muller N, Nichols DM, et al. The diagnosis of emphysema: a computed tomographic-pathologic correlation. Am Rev Respir Dis. 1986;133:541–46. doi: 10.1164/arrd.1986.133.4.541. [DOI] [PubMed] [Google Scholar]
- 11.Hruban RH, Meziane MA, Zerhouni EA, et al. High resolution computed tomography of inflation-fixed lungs: pathologic-radiologic correlation of centrilobular emphysema. Am Rev Respir Dis. 1987;136:935–40. doi: 10.1164/ajrccm/136.4.935. [DOI] [PubMed] [Google Scholar]
- 12.Miller RR, Muller NL, Vedal S, et al. Limitations of computed tomography in the assessment of emphysema. Am Rev Respir Dis. 1989;139:980–83. doi: 10.1164/ajrccm/139.4.980. [DOI] [PubMed] [Google Scholar]
- 13.Kuwano K, Matsuba K, Ikeda T, et al. The diagnosis of mild emphysema: correlation of computed tomography and pathology scores. Am Rev Respir Dis. 1990;141:169–78. doi: 10.1164/ajrccm/141.1.169. [DOI] [PubMed] [Google Scholar]
- 14.Müller NL, Staples CA, Miller RR, et al. „Density mask”. An objective method to quantitate emphysema using computed tomography. Chest. 1988;94(4):782–87. doi: 10.1378/chest.94.4.782. [DOI] [PubMed] [Google Scholar]
- 15.Gevenois PA, de Maertelaer V, De Vuyst P, et al. Comparison of computed density and macroscopic morphometry in pulmonary emphysema. Am J Respir Crit Care Med. 1995;152:653–57. doi: 10.1164/ajrccm.152.2.7633722. [DOI] [PubMed] [Google Scholar]
- 16.Gould GA, Macnee W, McLean A, et al. CT measurements of lung density in life can quantitate distal airspace enlargement: an essential defining feature of human emphysema. Am Rev Respir Dis. 1988;137:380–92. doi: 10.1164/ajrccm/137.2.380. [DOI] [PubMed] [Google Scholar]
- 17.Dirksen A, Dijkman JH, Madsen F, et al. A Randomized clinical trial of alpha(1)-antitrypsin augmentation therapy. Am J Respir Crit Care Med. 1999;160:1468–72. doi: 10.1164/ajrccm.160.5.9901055. [DOI] [PubMed] [Google Scholar]
- 18.Hayhurst MD, MacNee W, Flenly DC, et al. Diagnosis of pulmonary emphysema by computerised tomography. Lancet. 1984;2:320–22. doi: 10.1016/s0140-6736(84)92689-8. [DOI] [PubMed] [Google Scholar]
- 19.Gevenois PA, Koob MC, Jacobovitz D, et al. Whole lung sections for computed tomographic-pathologic correlations. Modified Gough-Wentworth technique. Invest Radiol. 1993;28:242–46. doi: 10.1097/00004424-199303000-00012. [DOI] [PubMed] [Google Scholar]
- 20.Uppaluri R, Mitsa T, Sonka M, et al. Quantification of pulmonary emphysema from lung computed tomography images. Am J Respir Crit Care Med. 1997;156:248–54. doi: 10.1164/ajrccm.156.1.9606093. [DOI] [PubMed] [Google Scholar]
- 21.Mishima M, Hirai T, Itoh H, et al. Complexity of terminal airspace geometry assessed by lung computed tomography in normal subjects and patients with chronic obstructive pulmonary disease. Proc Natl Acad Sci USA. 1999;96:8829–34. doi: 10.1073/pnas.96.16.8829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Irion KL, Marchiori E, Hochhegger B. Tomographic diagnosis of pulmonary emphysema. J Bras Pneumol. 2009;35(9):821–23. doi: 10.1590/s1806-37132009000900001. [DOI] [PubMed] [Google Scholar]
- 23.Sanders C, Nath PH, Bailey WC. Detection of emphysema with computed tomography: correlation with pulmonary function tests and chest radiography. Invest Radiol. 1988;23:262–66. doi: 10.1097/00004424-198804000-00004. [DOI] [PubMed] [Google Scholar]
- 24.Gurney JW, Jones KK, Robbins RA, et al. Regional distribution of emphysema: correlation of high-resolution CT with pulmonary function tests in unselected smokers. Radiology. 1992;183:457–63. doi: 10.1148/radiology.183.2.1561350. [DOI] [PubMed] [Google Scholar]
- 25.Haragushi M, Shimura S, Hida W, et al. Pulmonary function and regional distribution of emphysema as determined by high-resolution computed tomography. Respiration. 1998;65:125–29. doi: 10.1159/000029243. [DOI] [PubMed] [Google Scholar]
- 26.Saitoh T, Koba H, Shijubo N, et al. Lobar distribution of emphysema in computed tomographic densitometric analysis. Invest Radiol. 2000;35:235–43. doi: 10.1097/00004424-200004000-00004. [DOI] [PubMed] [Google Scholar]
- 27.Nakano Y, Sakai H, Hirai T, et al. Comparison of low attenuation areas on computed tomographic scans between inner and outer segments of the lung in patients with chronic obstructive pulmonary disease: incidence and contribution to lung function. Thorax. 1999;54:384–89. doi: 10.1136/thx.54.5.384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Stern EJ, Song JK, Frank MS. CT of the lungs in patients with pulmonary emphysema. nSemin Ultrasound CT MR. 1995;16(5):345–52. doi: 10.1016/0887-2171(95)90023-3. [DOI] [PubMed] [Google Scholar]
- 29.Park KJ, Bergin CJ, Clausen JL. Quantification of emphysema with three-dimensional CT densitometry: comparison with two-dimensional analysis, visual emphysema scores, and pulmonary function test results. Radiology. 1999;211:541–47. doi: 10.1148/radiology.211.2.r99ma52541. [DOI] [PubMed] [Google Scholar]
- 30.A Madani C, Keyzer PA. Gevenois: Quantitative computed tomography assessment of lung structure and function in pulmonary emphysema. Eur Respir J. 2001;18:720–30. doi: 10.1183/09031936.01.00255701. [DOI] [PubMed] [Google Scholar]
- 31.Bankier AA, de Maertelaer V, Keyzer C, et al. CT of pulmonary emphysema: subjective assessment and objective quantification by densitometry and macroscopic morphometry. Radiology. 1999;211:851–58. doi: 10.1148/radiology.211.3.r99jn05851. [DOI] [PubMed] [Google Scholar]
- 32.Adams H, Bernard MS, McConnochie K. An appraisal of CT pulmonary density mapping in normal subjects. Clin Radiol. 1991;43:238–42. doi: 10.1016/s0009-9260(05)80245-4. [DOI] [PubMed] [Google Scholar]
- 33.Lamers RJ, Thelissen GR, Kessels AG, et al. Chronic Obstructive Pulmonary Disease: Evaluation with Spirometrically Controlled CT Lung Densitometry. Radiology. 1994;193:109–13. doi: 10.1148/radiology.193.1.8090878. [DOI] [PubMed] [Google Scholar]
- 34.Mergo PJ, Williams WF, Gonzalez-Rothi R, et al. Three-Dimensional Volumetric Assessment of Abnormally Low Attenuation of the Lung from Routine Helical CT: Inspiratory and Expiratory Quantification. AJR. 1998;170:1355–60. doi: 10.2214/ajr.170.5.9574615. [DOI] [PubMed] [Google Scholar]
- 35.Soeijima K, Yamaguchi K, Kohda E, et al. Longitudinal follow-up study of smoking-induced lung density changes by high resolution computed tomography. Am J Respir Crit Care Med. 2000;161:1264–73. doi: 10.1164/ajrccm.161.4.9905040. [DOI] [PubMed] [Google Scholar]
- 36.Mishima M, Itoh H, Sakai H, et al. Optimized scanning conditions of high resolution CT in the follow-up of pulmonary emphysema. J Comput Assist Tomogr. 1999;23:380–84. doi: 10.1097/00004728-199905000-00011. [DOI] [PubMed] [Google Scholar]
- 37.Mathews JJ, Maurer AH, Steiner RM, et al. New 133Xe gas trapping index for quantifying severe emphysema before partial lung volume reduction. J Nucl Med. 2008;49(5):771. doi: 10.2967/jnumed.107.048637. [DOI] [PubMed] [Google Scholar]
- 38.Thurnheer R, Engel H, Weder W, et al. Role of lung perfusion scintigraphy in relation to chest computed tomography and pulmonary function in the evaluation of candidates for lung volume reduction surgery. Am J Respir Crit Care Med. 1999;159:301–10. doi: 10.1164/ajrccm.159.1.9711030. [DOI] [PubMed] [Google Scholar]
- 39.Wang SC, Fischer KC, Slone RM, et al. Perfusion scintigraphy in the evaluation of lung volume reduction surgery: correlation with clinical outcome. Radiology. 1997;205:243–48. doi: 10.1148/radiology.205.1.9314992. [DOI] [PubMed] [Google Scholar]
- 40.Austin J. Pulmonary emphysema: imaging assessment of lung volume reduction surgery. Radiology. 1999;212:1–3. doi: 10.1148/radiology.212.1.r99jl521. [DOI] [PubMed] [Google Scholar]
- 41.Ell PJ, Gambhir SS. Non-embolic disease of the lung, Section 4. Nuclear Medicine in Clinical Diagnosis and Treatment. (3rd edition) 2004;1:900–3. [Google Scholar]
- 42.Kaushik SS, Cleveland ZI, Cofer GP, et al. Diffusion-weighted hyperpolarized (129)Xe MRI in healthy volunteers and subjects with chronic obstructive pulmonary disease. Magn Reson Med. 2011;65:1155–65. doi: 10.1002/mrm.22697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kauczor HU, Hofmann D, Kreitnef KF, et al. Normal and abnormal pulmonary ventilation: visualization at hyperpolarized He-3 MR imaging. Radiology. 1996;201:564–68. doi: 10.1148/radiology.201.2.8888259. [DOI] [PubMed] [Google Scholar]
- 44.Saam BT, Yablonskiy DA, Kodibagkar VD, et al. MR imaging of diffusion of (3)He in healthy and diseased lungs. Magn Reson Med. 2000;44:174–79. doi: 10.1002/1522-2594(200008)44:2<174::aid-mrm2>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 45.Salerno M, de Lange EE, Altes TA, et al. Emphysema: hyperpolarized helium 3 diffusion MR imaging of the lungs compared with spirometric indexes – initial experience. Radiology. 2002;222:252–60. doi: 10.1148/radiol.2221001834. [DOI] [PubMed] [Google Scholar]
- 46.Yablonskiy DA, Sukstanskii AL, Leawoods JC, et al. Quantitative in vivo assessment of lung microstructure at the alveolar level with hyperpolarized 3He diffusion MRI. Proc Natl Acad Sci USA. 2002;99:3111–16. doi: 10.1073/pnas.052594699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Stavngaard T, Søgaard LV, Batz M, et al. Progression of emphysema evaluated by MRI using hyperpolarized (3)He (HP (3)He) measurements in patients with alpha-1-antitrypsin (A1AT) deficiency compared with CT and lung function tests. Acta Radiol. 2009;50(9):1019–26. doi: 10.3109/02841850903213822. [DOI] [PubMed] [Google Scholar]
- 48.Mata JF, Altes TA, Cai J, et al. Evaluation of emphysema severity and progression in a rabbit model: comparison of hyperpolarized 3He and 129Xe diffusion MRI with lung morphometry. J Appl Physiol. 2007;102:1273–80. doi: 10.1152/japplphysiol.00418.2006. [DOI] [PubMed] [Google Scholar]
- 49.Sukstanskii AL, Yablonskiy DA. Lung morphometry with hyperpolarized (129) Xe: Theoretical background. Magn Reson Med. 2011 doi: 10.1002/mrm.23056. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fain SB, Panth SR, Evans MD, et al. Early emphysematous changes in asymptomatic smokers: detection with 3He MR imaging. Radiology. 2006;239:875–83. doi: 10.1148/radiol.2393050111. [DOI] [PubMed] [Google Scholar]