Background: Activation of Atf1 is regulated by the Sty1 MAPK under multiple stresses, and the Pmk1 MAPK is also suggested to regulate the activity of Atf1.
Results: We obtained a measurable activation of Atf1 in Δsty1Δpmk1 cells.
Conclusion: Activation of Atf1 is not fully dependent on Sty1 and Pmk1.
Significance: The Renilla reporter assay identified a Sty1-independent activation of Atf1.
Keywords: Cell Signaling, MAP Kinases (MAPKs), Reactive Oxygen Species (ROS), Transcription Factors, Yeast, Schizosaccharomyces pombe, Atf1, Pmk1, Stress-activated MAPK, Sty1
Abstract
In Schizosaccharomyces pombe, the stress-activated Sty1 MAPK pathway is essential for cell survival under stress conditions. The Sty1 MAPK regulates Atf1 transcription factor to elicit stress responses in extreme conditions of osmolarity and reactive oxygen species-generating agents such as hydrogen peroxide, heat, low glucose, and heavy metal. Herein, using a newly developed Renilla luciferase reporter assay with enhanced detection sensitivity and accuracy, we show that distinct signaling pathways respond to cadmium and other reactive oxygen species-generating agents for the activation of Atf1. Also, surprisingly, a measurable activation of Atf1 transcription factor was still observed devoid of Sty1 MAPK activity. Further genetic and biological analyses revealed that the residual activation is caused by the activation of the cell wall integrity Pmk1 MAPK pathway and a redox-mediated activation of Atf1.
Introduction
The mitogen-activated protein kinase (MAPK) signaling pathways are critical for the response of cells to changes in their external environment. Such responses involve changes in the gene expression pattern of the cell, leading to increased levels and activities of proteins that have stress-protective functions (1). In mammalian cells, these stress-activated MAP kinases (SAPKs) fall into two distinct classes based on sequence and are termed the c-Jun N-terminal kinase (JNK) and p38 kinase (2). Both of these two SAPKs phosphorylate the activating transcription factor 2 (ATF2), which plays an important role in the cellular stress response (3, 4).
In Schizosaccharomyces pombe, the Sty1 MAPK (also known as Spc1 or Phh1), homologous to the mammalian JNK and p38 (5), plays a major role in response to ROS2-generating agents and many other stress factors (6–9). Activated Sty1 accumulates in the nucleus (10, 11), where it activates transcription factors, such as Atf1. As a bZIP transcription factor with strong homology to the mammalian factor ATF2, Atf1 regulates the expression of many genes that are relevant to the stress adaptation. Thus, the Sty1/Atf1-dependent core environmental stress response has been identified as being important for protection against different stress conditions, including osmolarity and some ROS-generating agents (12).
Our previous results of the firefly luciferase reporter assay led us to the conclusion that KCl stimulates the Sty1 MAPK pathway through MAPKKKs, whereas CdCl2 and LG stimulate the pathway through the inhibition of Pyp1 independently of MAPKKKs (13). However, the firefly luciferase requires a relatively abundant and continual supply of ATP that might lead to inaccurate or misleading conclusions in investigating the cellular responses that may consume ATP. Therefore, we newly developed a Renilla luciferase reporter system that is barely influenced by the level of ATP in living cells to avoid artifacts due to ATP consumption in response to stimuli such as H2O2. Here we show the results of S. pombe haploid strains obtained by the new Renilla luciferase reporter system. We also improved the assay by using a new type of luminometer (AB-2350; ATTO Co., Tokyo, Japan) with enhanced detection accuracy and sensitivity. The results show that distinct signaling pathways respond to cadmium and other ROS-generating agents for the activation of Atf1 and, surprisingly, that Atf1 transcription factor can still be activated by different extracellular stress agents independently of the activation of the Sty1 MAPK pathway. Further genetic and biological analyses revealed that the cell wall integrity Pmk1 MAPK pathway also activates Atf1 in a parallel manner and that there exists a redox-mediated activation of Atf1 in cells devoid of Sty1 and Pmk1 MAPK activities.
EXPERIMENTAL PROCEDURES
Strains, Media, and Genetic and Molecular Biology Methods
S. pombe haploid strains used in this study are listed in Table 1. The medium, notation, and genetic methods have been described previously (14, 15).
TABLE 1.
S. pombe haploid strains used in this study
| Strain | Genotype | Reference |
|---|---|---|
| HM123 | h− leu1-32 | Our stock |
| KP208 | h− leu1-32 ura4-D18 pmk1::ura4+ | Our stock |
| KP226 | h− leu1-32 his1-102 wis1::his1+ | 29 |
| KP471 | h− leu1-32 ura4-D18 sty1::ura4+ | 29 |
| KP495 | h− leu1-32 ura4-D18 atf1::ura4+ | 30 |
| KP2729 | h− leu1-32 mcs4::KanMX6 | This study |
| KP2730 | h− leu1-32 ura4-D18 wis4::ura4+ | 31 |
| KP2731 | h− leu1-32 ura4-D18 win1::ura4+ | 32 |
| KP2773 | h− leu1-32 ura4-D18 wis4::ura4+ win1::ura4+ | This study |
| KP2858 | h− leu1-32 ura4-D18 pyp1::ura4+ | 33 |
| KP2941 | h− leu1-32 ura4-D18 wis4::ura4+ mcs4::KanMX6 | This study |
| KP2943 | h− leu1-32 ura4-D18 win1::ura4+ mcs4::KanMX6 | This study |
| KP4330 | h− leu1-32 lys3-1 ura4-D18 sty1::lys3+ pmk1::ura4+ | This study |
Construction of cAMP-response Element (CRE) Reporter Plasmids
The 3xCRE::luc (R2.2) reporter vector (pKB5760 for the firefly luciferase reporter assay) was constructed as described previously (13). The 3xCRE::Renilla reporter vector (pKB5878 for the Renilla luciferase reporter assay) was constructed as follows. A 1176-bp fragment (86–1226 bases) containing Renilla-type luciferase gene was cut with HindIII and XbaI from a Rapid Response reporter vector phRG(R2.2)-basic (Promega) and subcloned into the HindIII/XbaI sites of pBluescript II SK(+) (pKB1019) to give pKB5836. Again, a Renilla-type luciferase gene-containing fragment was cut with NcoI and NotI from pKB5836 and subcloned into three tandem repeats of CRE-like sequence containing pKB5760 to displace the firefly-type luciferase gene.
Real-time Monitoring Assays of Atf1-mediated Transcriptional Activity
The method of real-time monitoring of Atf1-mediated transcriptional activity by using the firefly luciferase reporter assay has been described previously (13). The method of the 3xCRE::Renilla reporter assay is similar to that of the firefly luciferase reporter assay except that the substrate luciferin was displaced by coelenterazine. The experiments with CdCl2 and LG were performed as described previously (13). For the experiments with heat shock, cells were cultured at 27 °C in EMM containing 2% glucose overnight to mid-log phase and incubated in 42 or 34 °C for 10 min just before detection. N-Acetyl-l-cysteine (NAC; NACALAI TESQUE, Inc.) was added to the cells and incubated for 2 min before the addition of CdCl2 and H2O2. Emission of light was detected at 1-min intervals and reported as relative light units using a new type of luminometer (AB-2350; ATTO Co.).
Real-time Reverse Transcription-PCR (RT-PCR)
The total RNA was isolated by QIAzol (Qiagen) extraction. DNA was removed from RNA preparations using recombinant DNase I (RNase-free; TaKaRa). RNA quantitation assays were performed by a two-step method. In the first step, cDNA synthesis was performed using High Capacity cDNA reverse transcription kits (Applied Biosystems). Hsp9 primers for RT-PCR were 5′-ACC CTC GAC AAG GCC AAG GAA-3′ and 5′-TGT CAT CAA CAA AGC GTT GAG CCT-3′. Arp3 was used as an internal control, and Arp3 primers for RT-PCR were 5′-TTT CAG CTA TGC TGG GAA CGA TGC-3′ and 5′-GCT TGA GAG ATG CCC ACT GCC-3′. In the second step, SYBR® Green PCR master mix (Applied Biosystems) was used, and real-time PCR was run on Applied Biosystems 7500.
RESULTS AND DISCUSSION
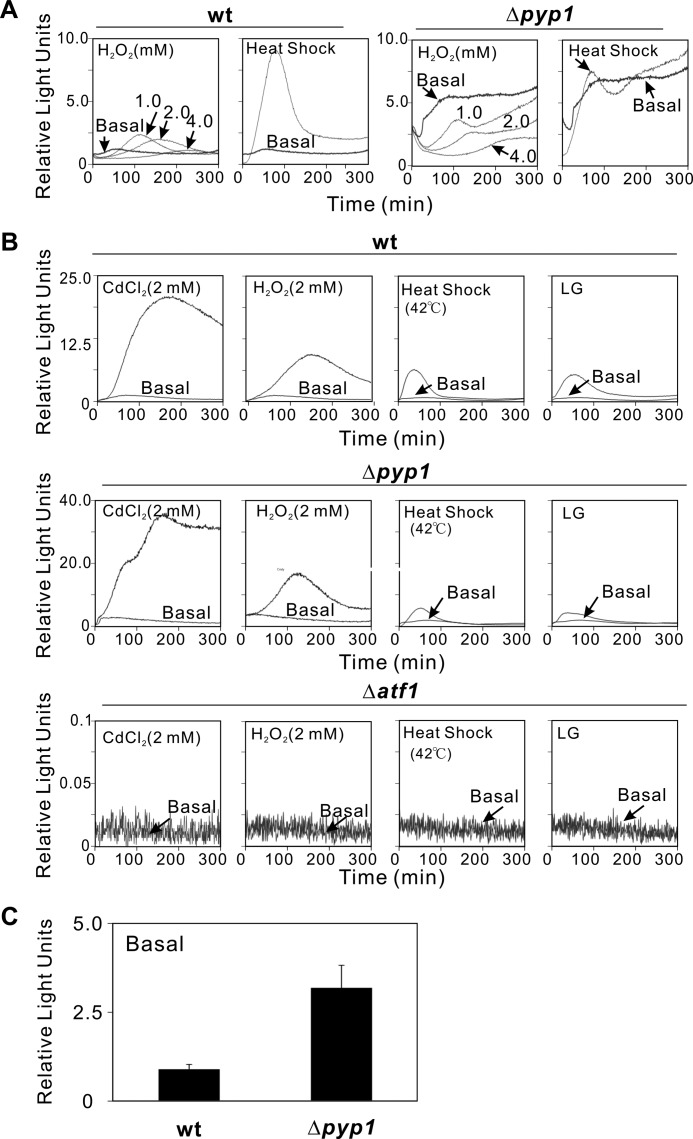
Our previous data obtained by using the firefly luciferase reporter assay suggest that the protein tyrosine phosphatase Pyp1 is responsible for sensing and transmitting the signals to Sty1 MAPK upon treatment with CdCl2 or LG (13). These results led us to look into the Atf1 transcriptional activities induced by other ROS-generating agents such as heat shock and H2O2. Upon heat shock (42 °C, 10 min), in wild-type cells, the Atf1 transcriptional activity showed a steady increase, with a peak rise at about 80 min; in contrast, in Δpyp1 cells, the Atf1 activity was barely observed and even lower than the basal level after the peak rise (Fig. 1A). Upon the elevated extracellular H2O2 (1.0–4.0 mm H2O2), in wild-type cells, the Atf1 transcriptional activity showed the steady increases with the dose-dependent delay of the peak. On the other hand, in Δpyp1 cells, unexpectedly, a series of three doses of H2O2 caused dose-dependent suppression of the peak (Fig. 1A). These results suggest that ATP consumption in cells treated by H2O2 (16) may lead to an insufficient ATP supply to ATP-dependent d-luciferin oxidation of the firefly luciferase reporter assay, thereby resulting in apparently low Atf1 transcriptional activity. It should be noted that although we use the same firefly luciferase reporter vector, the levels of the Atf1 transcriptional activities detected by the new type of luminometer (AB-2350) were much higher (Fig. 1A) than those detected by the previous type (AB-2300) (13).
FIGURE 1.
Real-time monitoring of Atf1 activity in living cells. A, live cell monitoring of Atf1 activity in wild-type (wt) and Δpyp1 cells in response to H2O2 and heat shock by using the firefly reporter assay. Wild-type cells and Δpyp1 cells transformed with the firefly reporter plasmid (3xCRE::luc(R2.2)) were incubated with d-luciferin and treated with H2O2 (1–4 mm) or incubated in 42 °C for 10 min before detection. Relative light units are expressed as the ratio of light emission of each sample to the basal (without stimulation) light emission of wild-type cells in EMM at 120 min. The data shown are representative of multiple experiments. B, live cell monitoring of Atf1 activity in wild-type, Δpyp1, and Δatf1 cells in response to ROS-generating agents by using the Renilla reporter assay. The cells as indicated were transformed with the 3xCRE::Renilla reporter plasmid, incubated with coelenterazine, and treated with CdCl2 (2 mm), H2O2 (2 mm), heat shock (42 °C, 10 min), or LG (0.1% glucose), as described under “Experimental Procedures.” C, the Δpyp1 cells showed a higher basal reporter activity than wild-type cells. Values are from at least three independent experiments. Error bars indicate means ± S.D.
In view of the facts that the transcriptional activity of Atf1 upon H2O2 treatment in living cells is not capable of being monitored accurately by the firefly luciferase reporter assay, we constructed the 3xCRE::Renilla reporter vector for the ATP-independent Renilla luciferase reporter system. The wild-type, Δatf1, and Δpyp1 cells were subjected to heavy metal stress (2 mm CdCl2), oxidative stress (2 mm H2O2), heat shock (42 °C, 10 min), and LG (0.1% glucose), respectively. In wild-type cells, various kinds of stresses caused various patterns of increase in response, and in Δatf1 cells, there was no response upon any treatment (Fig. 1B), indicating that the 3xCRE::Renilla reporter assay also reflects the Atf1 activity.
Similar to the results obtained with the firefly luciferase reporter assay (Fig. 1A, basal), Δpyp1 cells still showed a higher basal reporter activity than wild-type cells with the Renilla reporter (Fig. 1C), suggesting that a higher level of Atf1 transcriptional activity in the absence of extracellular stresses was induced by the relief of phosphatase Pyp1 inhibition of the Sty1 MAPK. However, in Δpyp1 cells treated with ROS-generating agents, the results obtained with the Renilla reporter were completely different from those obtained with the firefly luciferase reporter assay (Fig. 1B). Upon treatment with CdCl2, H2O2, heat shock, or LG, in Δpyp1 cells, an increase in Atf1 transcriptional activity was observed, similarly to wild-type cells (Fig. 1B), suggesting that Pyp1 does not function as a sensor for these ROS-generating agents in the Sty1 MAPK pathway. This is quite in contrast with our previous hypothesis obtained according to the results obtained with the firefly luciferase reporter assay (13). We reasoned that changes in ATP consumption upon treatment with these ROS-generating agents (16–20) and high basal transcriptional activity of Atf1 in Δpyp1 cells might lead to incomplete response of the firefly luciferase that is ATP-dependent.
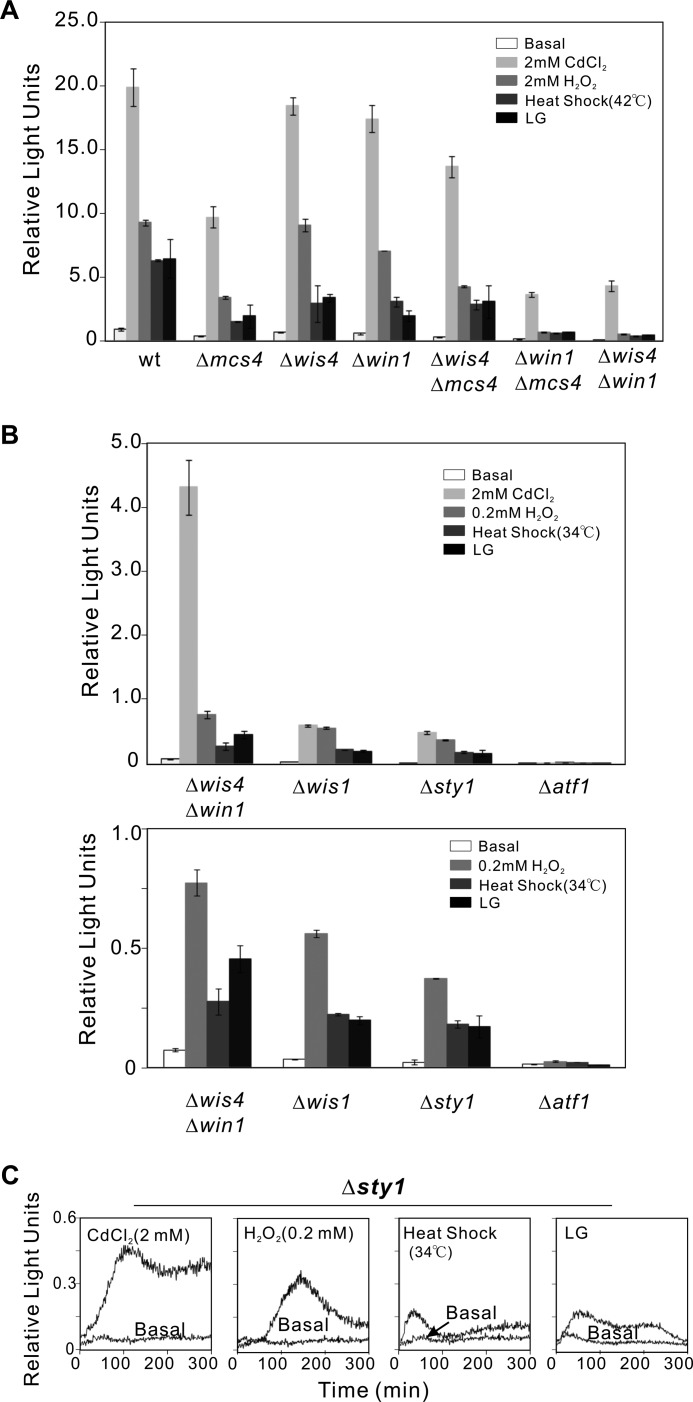
The Sty1/Atf1-dependent core environmental stress response is directly regulated by a MAPKK, Wis1, which is phosphorylated by two redundant MAPKKKs, Wis4 and Win1 (21). Also, a response regulator protein, Mcs4, associates with Wis4, and probably with Win1, to regulate MAPKKK activity in response to several forms of stress (8, 22). Then, we monitored Atf1 transcriptional activity using the Renilla reporter in the following cells: Δmcs4 (response regulator), Δwis4 and Δwin1 (MAPKKK), Δwis4Δmcs4, Δwin1Δmcs4 or Δwis4Δwin1 double mutants, and Δwis1 (MAPKK) and Δsty1 (MAPK).
These cells showed varying degrees of the Renilla reporter response upon treatment with the ROS-generating agents (Fig. 2, A and B). It should be noted that the Atf1 transcriptional activities in Δwis4 and Δwin1 cells induced by the ROS-generating agents are similar, whereas the reporter activities in Δwin1Δmcs4 double mutants were markedly lower than that in Δwis4Δmcs4 double mutants and were almost at the same level as the reporter activities in Δwis4Δwin1 double mutants (Fig. 2A). These results suggest that the interaction of MAPKKK Win1 with either the response regulator Mcs4 or MAPKKK Wis4 is important for the signal transduction induced by the ROS-generating agents.
FIGURE 2.
Detection of Atf1 transcriptional activity in various cells of the Sty1 MAPK pathway. A, interaction of MAPKKK Win1 with either the response regulator Mcs4 or MAPKKK Wis4 plays an important role for the activation of Atf1. Wild-type, Δmcs4, Δwis4, Δwin1, Δwis4Δmcs4, Δwin1Δmcs4, or Δwis4Δwin1 cells transformed with the Renilla reporter plasmid were treated as indicated. In each case, the peak height of each response was compared with the basal (without stimulation) light emission of wild-type cells in EMM at 120 min. Data were analyzed and plotted as in Fig. 1. B, distinct signaling pathways exist in response to cadmium and other ROS-generating agents upstream of the Wis1 MAPKK for the activation of Atf1. The Δwis4Δwin1, Δwis1, or Δsty1 cells transformed with the Renilla reporter plasmid were treated as indicated. Data were analyzed and plotted as in Fig. 1. Error bars in A and B indicate means ± S.D. C, activation of Atf1 in response to ROS-generating agents is not fully dependent on Sty1. The Δsty1 cells were transformed with the 3xCRE::Renilla reporter plasmid, incubated with coelenterazine, and treated with CdCl2 (2 mm), H2O2 (0.2 mm), heat shock (34 °C, 10 min), or LG (0.1% glucose), as described under “Experimental Procedures.”
Our previous results obtained with the firefly luciferase reporter assay showed that a measurable Atf1 activation was observed in the MAPKKK double mutant Δwis4Δwin1 by stimulation with ROS-generating agents, whereas it was abolished in Δsty1 or Δwis1 (13). These results led us to conclude that CdCl2 and LG stimulate the Sty1 pathway independently of MAPKKKs. Using the Renilla luciferase reporter assay, under treatment with the ROS-generating agents, a measurable Atf1 activation was observed not only in Δwis4Δwin1, but also in Δwis1 or Δsty1 cells, although it was extremely low (Fig. 2, B and C), suggesting that activation of Atf1 in response to these ROS-generating agents is not fully dependent on Sty1 MAPK. Moreover, the Atf1 transcriptional activities in Δwis4Δwin1 double mutants induced by H2O2, heat shock, or LG are not much different from those in Δwis1 or Δsty1 cells, whereas CdCl2-induced reporter activity in Δwis4Δwin1 double mutants is much higher than that in Δwis1 or Δsty1 cells (Fig. 2B). These results suggest that there exist distinct signaling pathways in response to cadmium and other ROS-generating agents upstream of the Wis1 MAPKK for the activation of Atf1.
It should be noted that Δwis1 and Δsty1 cells were subjected to a lower concentration of H2O2 (0.2 mm) (Fig. 2C) and a lower temperature (34 °C, 10 min) because the response curves of these cells quickly decreased to almost zero under H2O2 treatment (2 mm) and heat shock (42 °C, 10 min) (data not shown). Presumably, the transcriptional activity of Atf1 in these vulnerable cells may be turned off upon severe oxidative stress (2 mm H2O2) or heat shock (42 °C, 10 min).
To validate the results obtained by our reporter assay, Δsty1 and Δatf1 cells treated or untreated with 2 mm CdCl2 and 0.2 mm H2O2 were isolated, and mRNA levels of Atf1-dependent transcripts hsp9+ (23) were quantified by real-time RT-PCR. There was almost no increase of hsp9+ mRNA in Δatf1 cells upon stress, and under the same conditions, significant increases were observed in Δsty1 cells (supplemental Fig. S1). These results again proved the existence of some activators of Atf1 other than Sty1 MAPK.
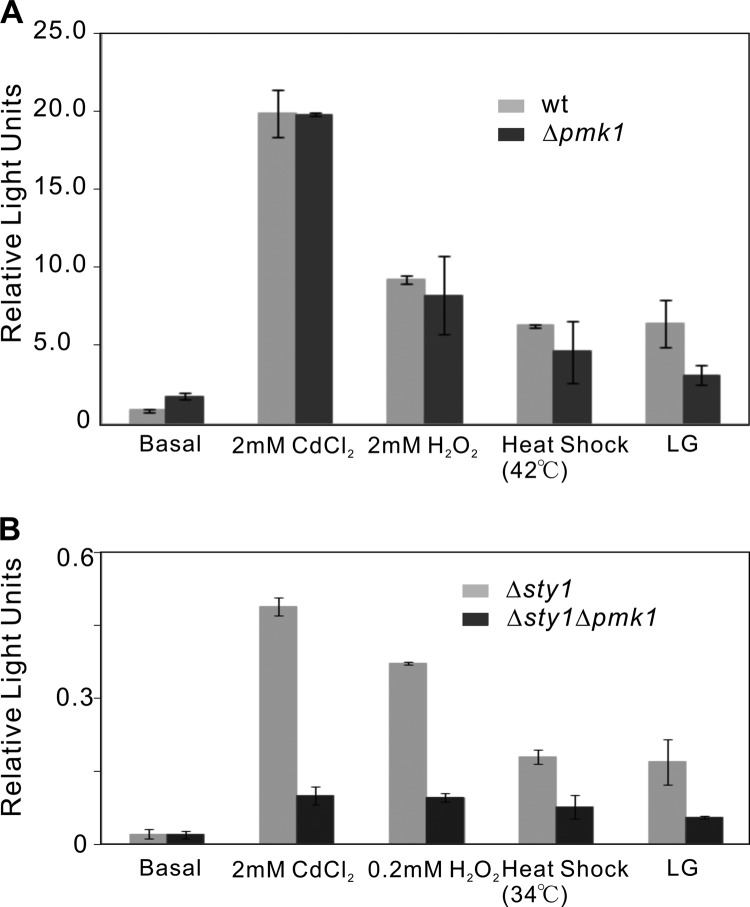
In fission yeast, the Pmk1 MAPK signaling pathway is also activated under multiple stresses, including heat shock, cell wall damage, and low glucose (15, 24, 25), and recent studies have reported that Pmk1 MAPK pathway plays a key role in cell wall integrity by regulating Atf1 (26). These results led us to investigate the Renilla reporter response upon treatment with the ROS-generating agents in Δpmk1 single and Δsty1Δpmk1 double mutants. The single mutant Δpmk1 cells showed a similar reporter activity with wild-type cells (Fig. 3A), suggesting that the absence of Pmk1 MAPK barely affects the Atf1 transcriptional activity in the presence of the Sty1 MAPK. However, in Δsty1Δpmk1 double mutants, significantly lower but still measurable reporter activities were observed than those observed in Δsty1 cells (Fig. 3B). Also, the Δsty1Δpmk1 cells showed synergism in the sensitivities to osmotic and oxidative stress when compared with parental cells (supplemental Fig. S2A). We further examined the effect of the overexpression of pmk1+ in Δsty1 under these stress conditions (supplemental Fig. S2B) and found that Pmk1 cannot rescue the phenotypes of Δsty1. These results indicate that Sty1 and Pmk1 MAPK regulate Atf1 activity in a parallel manner.
FIGURE 3.
Detection of Atf1 transcriptional activity in Δpmk1 and Δsty1Δpmk1 cells. A, activation of Atf1 in response to ROS-generating agents is not dependent on Pmk1 MAPK in the presence of Sty1 MAPK. Wild-type and Δpmk1 cells transformed with the Renilla reporter plasmid were treated with CdCl2 (2 mm), H2O2 (2 mm), heat shock (42 °C, 10 min), or LG (0.1% glucose) as indicated. B, Pmk1 MAPK turns into one of the regulatory factors of Atf1 in the absence of Sty1 MAPK. The Δsty1 and Δsty1Δpmk1 cells transformed with the Renilla reporter plasmid were treated with CdCl2 (2 mm), H2O2 (0.2 mm), heat shock (34 °C, 10 min), or LG (0.1% glucose) as indicated. Data were analyzed and plotted as in Fig. 1. Error bars in A and B indicate means ± S.D.
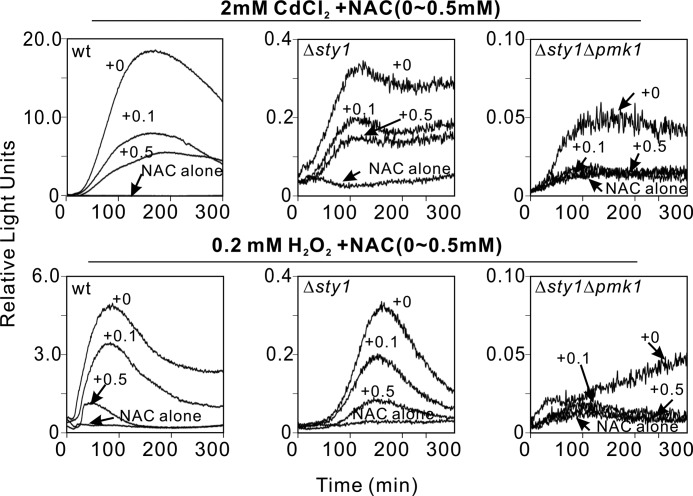
NAC, a well known thiol antioxidant, could increase intracellular cysteine pools to provide a source of sulfhydryl groups as an acetylated precursor of reduced GSH and also directly interact with ROS. NAC can block ROS-generating agent-induced changes in signal transduction and suppress activation of MAPKs (27, 28). Upon CdCl2 (2 mm) and H2O2 (0.2 mm) addition, NAC caused dose-dependent decrease of reporter activation in wild-type and Δsty1 cells (Fig. 4, left and middle panels), and these responses still remained higher than the activity in the presence of 0.5 mm NAC alone. However, in Δsty1Δpmk1 cells, the addition of 0.1 mm NAC abolished CdCl2- and H2O2-induced Atf1 reporter activity (Fig. 4, right panel), suggesting that the residual activity in Δsty1Δpmk1 cells may be mediated by redox reactions.
FIGURE 4.
The measurable Atf1 activity in Δsty1Δpmk1 cells is scavenged by NAC. Wild-type, Δsty1, and Δsty1Δpmk1 cells transformed with the Renilla reporter plasmid were treated with or without NAC (0.1 or 0.5 mm) before CdCl2 (2 mm) and H2O2 (2 mm) were added as described under “Experimental Procedures.” The reporter activities in cells were compared with the activity in the presence of 0.5 mm NAC alone.
We also investigated the Renilla reporter response under treatment with NAC alone. By adding different doses of NAC, Atf1 activity showed dose-dependent decrease in various cells (data not shown), suggesting that even in the absence of CdCl2 and H2O2, cellular redox products may cause the increase of reporter activity.
In conclusion, here we have presented data that establish the Renilla luciferase reporter assay as a novel semiquantification method for monitoring the Atf1 transcriptional activity in living cells upon treatment with ROS-generating agents including CdCl2, H2O2, heat shock, and LG. By using the new reporter assay and the luminometer with enhanced detection sensitivity and accuracy, a measurable activation of Atf1 transcription factor induced by ROS-generating agents was clearly observed in Δwis1, Δsty1, as well as in Δsty1Δpmk1 cells. This measurable activity was abolished by the addition of a thiol antioxidant NAC.
We also obtained similar increased reporter activities in Δwis4Δwin1, Δwis1, and Δsty1 cells induced by osmotic stress (0.3 m KCl) by using both the firefly and the Renilla luciferase reporter assays with the new type of luminometer (data not shown). These unexpected results not only evidenced that the Pmk1 MAPK also takes part in regulating Atf1 activity in the absence of the Sty1 MAPK, but also suggested that the redox state of cells may directly regulate Atf1 upon exposure to many environmental stresses, and future work will be continued to test this hypothesis.
This work was supported by research grants from the Japan Society for the Promotion of Science.

This article contains supplemental Figs. 1 and 2.
- ROS
- reactive oxygen species
- LG
- low glucose
- EMM
- Edinburgh minimal medium
- NAC
- N-acetyl-l-cysteine.
REFERENCES
- 1. Marshall C. J. (1994) MAP kinase kinase kinase, MAP kinase kinase, and MAP kinase. Curr. Opin. Genet. Dev. 4, 82–89 [DOI] [PubMed] [Google Scholar]
- 2. Davis R. J. (1994) MAPKs: new JNK expands the group. Trends Biochem. Sci. 19, 470–473 [DOI] [PubMed] [Google Scholar]
- 3. Livingstone C., Patel G., Jones N. (1995) ATF-2 contains a phosphorylation-dependent transcriptional activation domain. EMBO J. 14, 1785–1797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. van Dam H., Wilhelm D., Herr I., Steffen A., Herrlich P., Angel P. (1995) ATF-2 is preferentially activated by stress-activated protein kinases to mediate c-jun induction in response to genotoxic agents. EMBO J. 14, 1798–1811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Toone W. M., Morgan B. A., Jones N. (2001) Redox control of AP-1-like factors in yeast and beyond. Oncogene 20, 2336–2346 [DOI] [PubMed] [Google Scholar]
- 6. Shiozaki K., Russell P. (1995) Cell cycle control linked to extracellular environment by MAP kinase pathway in fission yeast. Nature 378, 739–743 [DOI] [PubMed] [Google Scholar]
- 7. Millar J. B., Buck V., Wilkinson M. G. (1995) Pyp1 and Pyp2 PTPases dephosphorylate an osmosensing MAP kinase controlling cell size at division in fission yeast. Genes Dev. 9, 2117–2130 [DOI] [PubMed] [Google Scholar]
- 8. Degols G., Shiozaki K., Russell P. (1996) Activation and regulation of the Spc1 stress-activated protein kinase in Schizosaccharomyces pombe. Mol. Cell Biol. 16, 2870–2877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Degols G., Russell P. (1997) Discrete roles of the Spc1 kinase and the Atf1 transcription factor in the UV response of Schizosaccharomyces pombe. Mol. Cell Biol. 17, 3356–3363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Gaits F., Degols G., Shiozaki K., Russell P. (1998) Phosphorylation and association with the transcription factor Atf1 regulate localization of Spc1/Sty1 stress-activated kinase in fission yeast. Genes Dev. 12, 1464–1473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Gaits F., Russell P. (1999) Active nucleocytoplasmic shuttling required for function and regulation of stress-activated kinase Spc1/StyI in fission yeast. Mol. Biol. Cell 10, 1395–1407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Chen D., Toone W. M., Mata J., Lyne R., Burns G., Kivinen K., Brazma A., Jones N., Bähler J. (2003) Global transcriptional responses of fission yeast to environmental stress. Mol. Biol. Cell 14, 214–229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Zhou X., Ma Y., Sugiura R., Kobayashi D., Suzuki M., Deng L., Kuno T. (2010) MAP kinase kinase kinase (MAPKKK)-dependent and -independent activation of Sty1 stress MAPK in fission yeast. J. Biol. Chem. 285, 32818–32823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Moreno S., Klar A., Nurse P. (1991) Molecular genetic analysis of fission yeast Schizosaccharomyces pombe. Methods Enzymol. 194, 795–823 [DOI] [PubMed] [Google Scholar]
- 15. Toda T., Dhut S., Superti-Furga G., Gotoh Y., Nishida E., Sugiura R., Kuno T. (1996) The fission yeast pmk1+ gene encodes a novel mitogen-activated protein kinase homolog which regulates cell integrity and functions coordinately with the protein kinase C pathway. Mol. Cell Biol. 16, 6752–6764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Holmsen H., Robkin L. (1977) Hydrogen peroxide lowers ATP levels in platelets without altering adenyalte energy charge and platelet function. J. Biol. Chem. 252, 1752–1757 [PubMed] [Google Scholar]
- 17. Martens G. A., Cai Y., Hinke S., Stangé G., Van de Casteele M., Pipeleers D. (2005) Glucose suppresses superoxide generation in metabolically responsive pancreatic β cells. J. Biol. Chem. 280, 20389–20396 [DOI] [PubMed] [Google Scholar]
- 18. Osorio H., Carvalho E., del Valle M., Günther Sillero M. A., Moradas-Ferreira P., Sillero A. (2003) H2O2, but not menadione, provokes a decrease in the ATP and an increase in the inosine levels in Saccharomyces cerevisiae: an experimental and theoretical approach. Eur. J. Biochem. 270, 1578–1589 [DOI] [PubMed] [Google Scholar]
- 19. Schrader M., Fahimi H. D. (2006) Peroxisomes and oxidative stress. Biochim. Biophys. Acta 1763, 1755–1766 [DOI] [PubMed] [Google Scholar]
- 20. Nicholls D. G. (1992) Neurotransmission: a retrograde step forward. Nature 360, 106–107 [DOI] [PubMed] [Google Scholar]
- 21. Price M. A., Cruzalegui F. H., Treisman R. (1996) The p38 and ERK MAP kinase pathways cooperate to activate ternary complex factors and c-fos transcription in response to UV light. EMBO J. 15, 6552–6563 [PMC free article] [PubMed] [Google Scholar]
- 22. Buck V., Quinn J., Soto Pino T., Martin H., Saldanha J., Makino K., Morgan B. A., Millar J. B. (2001) Peroxide sensors for the fission yeast stress-activated mitogen-activated protein kinase pathway. Mol. Biol. Cell 12, 407–419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Ors A., Grimaldi M., Kimata Y., Wilkinson C. R., Jones N., Yamano H. (2009) The transcription factor Atf1 binds and activates the APC/C ubiquitin ligase in fission yeast. J. Biol. Chem. 284, 23989–23994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Sugiura R., Toda T., Dhut S., Shuntoh H., Kuno T. (1999) The MAPK kinase Pek1 acts as a phosphorylation-dependent molecular switch. Nature 399, 479–483 [DOI] [PubMed] [Google Scholar]
- 25. Madrid M., Soto T., Khong H. K., Franco A., Vicente J., Pérez P., Gacto M., Cansado J. (2006) Stress-induced response, localization, and regulation of the Pmk1 cell integrity pathway in Schizosaccharomyces pombe. J. Biol. Chem. 281, 2033–2043 [DOI] [PubMed] [Google Scholar]
- 26. Takada H., Nishida A., Domae M., Kita A., Yamano Y., Uchida A., Ishiwata S., Fang Y., Zhou X., Masuko T., Kinoshita M., Kakehi K., Sugiura R. (2010) The cell surface protein gene ecm33+ is a target of the two transcription factors Atf1 and Mbx1 and negatively regulates Pmk1 MAPK cell integrity signaling in fission yeast. Mol. Biol. Cell 21, 674–685 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Lee Y. J., Galoforo S. S., Berns C. M., Chen J. C., Davis B. H., Sim J. E., Corry P. M., Spitz D. R. (1998) Glucose deprivation-induced cytotoxicity and alterations in mitogen-activated protein kinase activation are mediated by oxidative stress in multidrug-resistant human breast carcinoma cells. J. Biol. Chem. 273, 5294–5299 [DOI] [PubMed] [Google Scholar]
- 28. Zafarullah M., Li W. Q., Sylvester J., Ahmad M. (2003) Molecular mechanisms of N-acetylcysteine actions. Cell Mol. Life Sci. 60, 6–20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Kato T., Jr., Okazaki K., Murakami H., Stettler S., Fantes P. A., Okayama H. (1996) Stress signal, mediated by a Hog1-like MAP kinase, controls sexual development in fission yeast. FEBS Lett. 378, 207–212 [DOI] [PubMed] [Google Scholar]
- 30. Takeda T., Toda T., Kominami K., Kohnosu A., Yanagida M., Jones N. (1995) Schizosaccharomyces pombe atf1+ encodes a transcription factor required for sexual development and entry into stationary phase. EMBO J. 14, 6193–6208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Samejima I., Mackie S., Fantes P. A. (1997) Multiple modes of activation of the stress-responsive MAP kinase pathway in fission yeast. EMBO J. 16, 6162–6170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Samejima I., Mackie S., Warbrick E., Weisman R., Fantes P. A. (1998) The fission yeast mitotic regulator win1+ encodes an MAP kinase kinase kinase that phosphorylates and activates Wis1 MAP kinase kinase in response to high osmolarity. Mol. Biol. Cell 9, 2325–2335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Ottilie S., Chernoff J., Hannig G., Hoffman C. S., Erikson R. L. (1992) The fission yeast genes pyp1+ and pyp2+ encode protein tyrosine phosphatases that negatively regulate mitosis. Mol. Cell Biol. 12, 5571–5580 [DOI] [PMC free article] [PubMed] [Google Scholar]