Abstract
Background
Obesity/diabetes adversely affects post-ischemic heart remodeling via incompletely understood underlying mechanisms. C1q/TNF-related protein-3 (CTRP3) is a newly identified adipokine exerting beneficial metabolic regulation, similar to adiponectin. The current study determined whether CTRP3 may regulate post-ischemic cardiac remodeling and cardiac dysfunction, and, if so, sought to elucidate the involved underlying mechanisms.
Methods and Results
Male adult mice were subjected to myocardial infarction (MI) via left anterior descending (LAD) coronary artery occlusion. Both the effect of MI upon endogenous CTRP3 expression/production and the effect of exogenous CTRP3 (adenovirus or recombinant CTRP3) replenishment upon MI injury were investigated. MI significantly inhibited adipocyte CTRP3 expression and reduced plasma CTRP3 level, reaching nadir 3 days post-MI. CTRP3 replenishment improved survival rate (P<0.05), restored cardiac function, attenuated cardiomyocyte apoptosis, increased revascularization, and dramatically reduced interstitial fibrosis (P values all <0.01). CTRP3 replenishment had no significant effect upon cardiac AMP-activated protein kinase (AMPK) phosphorylation, but significantly increased Akt phosphorylation and expression of hypoxia inducing factor-1α (HIF-1α) and vascular endothelial growth factor (VEGF). Surprisingly, treatment of human umbilical vascular endothelial cells (HUVECs) with CTRP3 did not directly affect NO production or tube formation. However, pre-conditioned medium from CTRP3-treated cardiomyocytes significantly enhanced HUVEC tube formation, an effect blocked by either pre-treatment of cardiomyocytes with a PI3K inhibitor, or pre-treatment of HUVECs with a VEGF inhibitor. Finally, pre-conditioned medium from CTRP3-knockdown 3T3 cells significantly enhanced hypoxia-induced cardiomyocyte injury.
Conclusions
CTRP3 is a novel anti-apoptotic, pro-angiogenic, and cardioprotective adipokine, whose expression is significantly inhibited following MI.
Keywords: CTRP3, revascularization, myocardial ischemia, signal transduction
Cardiovascular disease remains a leading cause of mortality worldwide. Although improved reperfusion strategies have led to declined death rates after acute MI, both incidence and prevalence of post-MI heart failure have continually increased in recent years1. Despite pharmacological advances (eg, β-blockers, renin–angiotensin–aldosterone system inhibitors), mortality post-MI remains very high, with 5-year rates being 30–70%. Left ventricular remodeling, which includes ventricular dilatation and increased interstitial fibrosis, is the critical process underlying progression to heart failure. A novel approach preventing left ventricular remodeling after MI is therefore in great need.
Due to increased incidence of diabetes and its association with heart failure, there is now great interest in elucidating the underlying molecular mechanisms linking these pathologies. Since their original discovery, the important regulatory role of adipose-derived hormones and cytokines (adipokines) in myocardial function has gained recognition. Considerable evidence exists that the majority of adipokines (including tumor necrosis factor-α, leptin, plasminogen activator inhibitor type 1, transforming growth factor β, and resistin) adversely regulate myocardial metabolism, cardiomyocyte hypertrophy, extracellular matrix structure and composition, and cell death2. Despite having been consistently shown to increase cardiomyocyte glucose uptake, stimulate fatty acid oxidation, and protect against acute myocardial ischemia/reperfusion injury3, adiponectin’s effect upon chronic cardiac remodeling and heart failure development remains controversial4. Whether there exists any adipokine exerting beneficial regulatory effect upon post-ischemic cardiac remodeling, improving cardiac function, remains unknown.
Recently, a highly conserved family of adiponectin paralogs, designated C1q tumor necrosis factor (TNF) related proteins (CTRPs), has been discovered. Each of the known ten members (CTRP1 through 10) consist of four distinct domains, including an N-terminal signal peptide, a short variable domain, a collagen-like domain, and a C-terminal C1q-like globular domain5, 6. Both CTRPs and adiponectin belong to the C1q/TNF protein superfamily, which continues to grow as more C1q domain proteins are discovered7. Investigated for its structural similarity to adiponectin, the CTRP family members exhibit broadly diverse functions. CTRP3 (also known as cartducin) is a growth plate secretory protein of primarily cartilaginous origin, but has also been reported in monocytic and osteosarcoma cells8. Initially identified during a search for genes responsible for chondrocyte differentiation induction, CTRP3 also stimulates adiponectin and resistin release, and therefore serves as a regulator of adiponectin secretion from adipocytes9, 10. Most importantly, CTRP3 is the first and only CTRP whose in vivo biological function (as a metabolic regulator of glucose homeostasis) has been recently established11.
Interestingly, CTRP3 has been reported to stimulate in vitro endothelial cell proliferation and migration12. However, whether CTRP3 might promote in vivo revascularization in the infarct border zones after MI is unknown. Moreover, whether CTRP3, a key member of the newest adipokine family, may function as a mediator or inhibitor of post-MI remodeling has never been previously investigated. Therefore, the aims of this study were: 1) to determine the effect of MI on CTRP3 expression/production; 2) to investigate the effect of CTRP3 replenishment upon post-ischemic cardiac remodeling and dysfunction; 3) to determine whether CTRP3 administration might promote revascularization after MI; and 4) to elucidate the mechanisms responsible for CTRP3’s cardiac biological actions.
Methods
All experiments were performed in adherence with the National Institutes of Health Guidelines on the Use of Laboratory Animals, and were approved by the Thomas Jefferson University Committee on Animal Care. All authors had full access to, and take full responsibility for data integrity. All authors have read and agree to the manuscript as written.
Experimental protocols
Adult male C57/BL6 mice were anesthetized with 2% isoflurane and MI was induced via LAD ligation as previously described13. Sham-operated control mice (Sham) underwent the same surgical procedures except that the suture placed under the LAD was not tied. After full recovery from cardiac surgery (4 hours after MI), animals were randomized to one of the following groups: Sham+Vehicle, Sham+CTRP3, MI+Vehicle, MI+CTRP3 (0.25 µg/g/day intraperitoneal infusion via mini-osmotic pump, ALZET® DURECT Corporation, Cupertino, CA). After 2 weeks, mice were sacrificed. Immunohistological, biochemical, and Western blot assays were performed as we previously reported14, and described in detail in online supplementation.
Construct and expression of globular CTRP3
Globular domain of mouse CTRP3 gene was generated by PCR, and cloned into the prokaryotic protein expression vector pET45b (Novagen, Merck, USA). The construct was verified by DNA sequencing. Globular CTRP3 prokaryotic expression vector was transferred into BL21 (DE3) bacterium protein expression host, grown in LB medium and shaken overnight at 37°C. Protein expression inducer IPTG (isopropyl-β-D-thio-galactoside) was added to medium (final concentration 1mM). The solution was shaken for 4–5 hours, and subject to 5,000 RPM centrifugation. Proteins were purified in native condition by Ni-NTA resin per manufacturer’s instructions (Novagen, #70666-3, Merck, USA). Endotoxin was removed by endotoxin-removing column (Sterogene ActiCleanEtoc resin, Carlsbad, CA). Proteins were desalted, and concentrated by centrifugation (Millipore Centricon, Plus-20). Purified proteins were examined by Western blot analysis (online Figure 1A,B), and the endotoxin activity was determined using the LAL assay kit (BioWhittaker, Walkersville, MD) according to the manufacturer’s recommendation (27.6±3.9 EU/mg protein).
Figure 1. CTRP3 improves survival and restores left ventricular cardiac function after MI.
The effects of CTRP3 treatment upon survival and LV function were determined 14 days post operation. WT mice were subjected to MI or sham operation (Sham), and treated with CTRP3 or saline as vehicle (Veh. abbreviation is used across all figures). (A) Survival curve; (B) Echocardiographic analysis; (C–D) Hemodynamic analysis. *P<0.05, **P<0.01.
Adenoviral vectors production and transfection
Adenovirus expressing human full length CTRP3 was constructed by SinoGenoMax Co., Ltd (Beijing, China). Briefly, pShuttle-GFP-CTRP3 was constructed by cloning the target gene CTRP3 into pShuttle-GFP-CMV. After sequence confirmation, pShuttle-GFP-CTRP3 was transferred into the recombinant adenovirus frame vector of pAdxsi. The pADxsi-GFP-CTRP3 adenovirus vector was then amplified in 293 cells and viral titer was measured. Control vectors (Ad.Null) not expressing CTRP3 but β-galactosidase (β-gal) were constructed and produced concomitantly. 2×108 plaque-forming units (p.f.u.) of Ad.CTRP3 or Ad.Null were injected into the jugular vein of mice 3 days prior to MI.
Statistical analysis
Data were analyzed with GraphPad Prism-5 statistic software (La Jolla, CA). All values in the text and figures are presented as the mean±SEM of n independent experiments. ANOVA was conducted across all investigated groups first. Post hoc tests were then performed with Bonferroni correction and all two-group comparisons were made. Time and group differences (Figure 5A) were determined by two-way analysis of variance for repeated measures, followed by post-hoc tests with Holm adjustment. Survival data were analyzed via Kaplan-Meier method followed by logrank test. Western blot densities were analyzed by the Kruskal-Wallis test followed by the Dunn post hoc test. P values less than or equal to 0.05 were considered statistically significant.
Figure 5. CTRP3 adenovirus improves survival, restores cardiac function, and attenuates left ventricular cardiac remodeling.
Ad.CTRP3 or Ad.Null were injected into the jugular vein of mice 3 days prior to MI. Effects of adenoviral CTRP3 supplement upon plasma CTRP3 level by Western analysis (A), survival rate by survival curve analysis (B), LV function by echocardiographic analysis (C), and cardiac remodeling by interstitial fibrosis (D), angiogenesis (E), and cardiomyocyte apoptosis (F) were determined 14 days post-MI. *P<0.05, **P<0.01.
Results
MI inhibits adipocyte CTRP mRNA expression and decreases plasma CTRP3 level
Clinical and experimental studies have demonstrated that plasma adiponectin levels are significantly reduced post-MI. However, the effect of MI upon CTRP expression has never been previously determined. Adipocyte CTRP3 mRNA expression significantly decreased 1 day after MI, and gradually recovered thereafter (online Figure 2A). Consequently, plasma CTRP3 levels (determined by Western blot densitometry analysis) are significantly decreased after MI, reaching nadir 3 days post-MI, and gradually recovered thereafter (online Figure 2B, solid line). These results demonstrate that CTRP3 expression/production is regulated during post-MI cardiac remodeling.
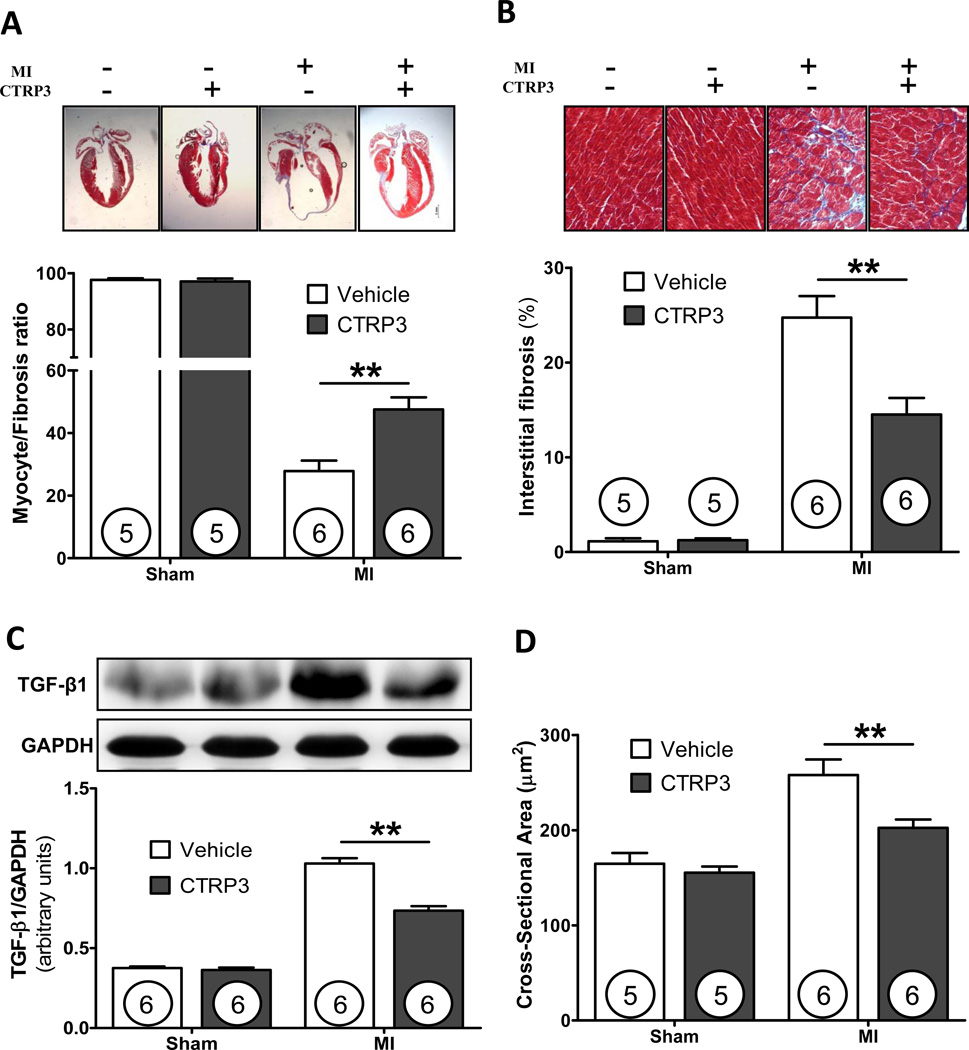
Figure 2. CTRP3 prevents left ventricular cardiac remodeling after MI.
(A) Gross observation; (B) Heart weight to tibia length ratio; (C and D) LVEDD and LVESD, from echocardiographic analysis. *P<0.05, **P<0.01.
Replenishment of CTRP3 improves survival and restores left ventricular cardiac function after MI
Having demonstrated that adipocyte CTRP3 expression is inhibited and plasma CTRP3 levels are reduced in MI animals, we attempted to determine 1) whether post-MI CTRP3 reduction is pathologically relevant and 2) whether supplementation of exogenous CTRP3 may protect heart against post-MI remodeling. As illustrated in online Figure 2B, administration of recombinant CTRP3 via intraperitoneal osmotic pump (dose 0.25 µg/g/day) caused approximately 1.5-fold CTRP3 increase in sham-operated mice (open triangles connected with dashed lines) and prevented MI-induced plasma CTRP3 decline (open circles connected with dashed lines). Administration of CTRP3 in sham operated mice had no effect upon cardiac function assessed by echocardiography or ventricular catheterization. However, CTRP3 administration significantly improved post-MI survival rate (Figure 1A), augmented left ventricular ejection fraction (LVEF, Figure 1B), increased dP/dtmax (Figure 1C), and decreased LV end diastolic pressure (LVEDP, Figure 1D). These data demonstrate that pharmacological restoration of plasma CTRP3 to physiological levels improved both LV systolic and diastolic function in MI animals, and increased survival rate.
CTRP3 prevents left ventricular cardiac remodeling after MI
Since pathological remodeling plays a critical role in post-ischemic cardiac dysfunction, we determined indices of remodeling by gross anatomy, echocardiography, and Masson’s trichrome staining 14 days after MI. Compared to MI+Vehicle, CTRP3 administration reduced heart size and mass (Figure 2, A and B), and preserved LV end-diastolic dimension (LVEDD) and LV end-systolic dimension (LVESD) (Figure 2, C and D). Most noticeably, CTRP3 treatment dramatically increased myocyte/fibrotic cell ratio in the ischemic zone (Figure 3A), significantly attenuated interstitial fibrosis (Figure 3B), reduced TGF-β1 expression, and decreased cardiomyocyte size in remote non-ischemic zone (Figure 3C and 3D). Together, these results suggest that endogenous CTRP3 may play a significant role in regulating cardiac remodeling in post-MI hearts, and that recombinant CTRP3 may have therapeutic potential against MI injury.
Figure 3. CTRP3 attenuates left ventricular cardiac fibrosis after MI.
(A) Cardiac Masson trichrome staining; (B). Interstitial fibrosis; (C) Western blot analysis for TGF-β1 expression; (D) Relative cross-sectional area of myocytes in the remote area. Cell size of sham-operated mice receiving vehicle was used as a control, and set as 100%. **P<0.01.
CTRP3 promotes angiogenesis and activates the Akt-HIF1α-VEGF axis in infarct border zone
Angiogenesis is a critical step initiating cardiac repair post-MI. Having demonstrated that CTRP3 is a novel adipokine potently inhibiting fibrosis and strongly augmenting cardiomyocyte survival, we assessed the angiogenic effects of CTRP3 as a potential underlying mechanism. As illustrated in Figure 4A, CTRP3 treatment markedly increased the number of CD31-positive capillary vessels and increased α-SMA-positive arterial density (Figure 4B) in the border zone 14 days post-MI, suggesting that CTRP3 induces mature vessel formation after MI.
Figure 4. CTRP3 promotes angiogenesis and activates the Akt-HIF1α-VEGF axis in infarct border zone.
(A) Capillary density measured by immunohistochemical staining for CD31 (brown) in the border region (MI group) or LV free wall (sham group); (B) Immunostaining for α-SMA (green) and actinin (red) shown together with DAPI (blue) staining in the border region (MI group) or LV free wall (sham group) 14 days post operation. Quantity of α-SMA-positive vessels within the infarct border zone is summarized by bar graph. **P<0.01; Western blot analysis for pAMPK(Thr172)/AMPK (C), pAkt(Ser473)/Akt (D), HIF-1α (E), and VEGF-A (F) from tissue within the border region (MI group) or LV free wall (sham group). *P<0.05, **P<0.01.
To further determine the molecular signaling mechanisms responsible for increased angiogenesis after CTRP3 treatment, several critical mediators and cytokines requisite for angiogenesis were assessed. CTRP3 had no significant effect upon AMPK phosphorylation, but significantly enhanced Akt phosphorylation, and increased HIF1-α and VEGF expression (Figure 4C–F). These results suggest that CTRP3 promotes angiogenesis after MI possibly through the Akt-HIF1α-VEGF axis.
Adenoviral supplementation of CTRP3 significantly attenuates left ventricular cardiac remodeling
Recognizing that the globular domain of CTRP3 induced angiogenesis and prevented left ventricular cardiac remodeling, we further determined the effect of adenoviral production of full length CTRP3 upon adverse left ventricular cardiac remodeling. CTRP3-expressing adenovirus infection resulted in approximately 1.9-fold increase in plasma CTRP3 before MI, and maintained plasma CTRP3 at significantly higher levels than that seen in Ad.Null-treated animals throughout the observation period (Figure 5A). CTRP3 overproduction markedly improved survival rate (Figure 5B), and restored left cardiac function (Figure 5C). Moreover, CTRP3-expressing adenovirus significantly attenuated interstitial fibrosis in the remote area (Figure 5D), increased CD31 positive capillary density (Figure 5E), and ameliorated TUNEL and α-actinin double positive apoptotic cardiomyocyte death in the border zone (Figure 5F). Finally, transfection of neonatal cardiomyocytes with adenoviral CTRP3 activated Akt and upregulated HIF1α expression in vitro (online supplementation 3).
CTRP3 does not significantly promote angiogenesis in cultured HUVECs
Endothelial cells are essential in angiogenesis and post-ischemic cardiac repair. To directly investigate the effect of CTRP3 upon endothelial cells, we determined the effect of CTRP3 upon tube formation in cultured HUVECs, with vascular endothelial growth factor (VEGF) as a positive control. Somewhat to our surprise, although in vivo administration of CTRP3 significantly stimulated angiogenesis, direct HUVEC treatment with CTRP3 (both gCTRP3 and full length CTRP3) failed to significantly promote tube formation (Figure 6A) or stimulate nitric oxide (NO, determined as previously described14) production (Figure 6B). Moreover, although in vivo CTRP3 treatment significantly increased Akt phosphorylation and increased HIF1-α and VEGF expression in the ischemic heart, in vitro treatment of HUVECs with CTRP3 neither increased Akt phosphorylation, nor HIF1-α or VEGF expression (Figure 6, C–G). These results indicate CTRP3’s pro-angiogenic effect observed in vivo cannot be attributed to its direct effect upon endothelial cells.
Figure 6. CTRP3 does not significantly promote angiogenesis in cultured HUVECs.
(A) Tube formation assay. HUVECs were treated with gCTRP3 (3 µg/ml), fCTRP3 (30 µg/ml), vehicle, or VEGF (positive control) for 6 hours. Representative experiment data shown (n=6 per condition). **P<0.01 (versus vehicle); (B) NO formation assay. HUVECs were treated with CTRP3 (3 µg/ml) or vehicle for 12 hours. NO formation was analyzed after 0.5, 1, 3, 6, and 12 hours as described in our previous studies; (C–G) Western blot analysis for pAkt(Ser473)/Akt, pAMPK (Thr172)/AMPK, HIF-1α, and VEGF from cultured HUVECs treated with CTRP3 directly.
CTRP3 promotes angiogenesis via a cardiomyocyte-initiated, Akt-dependent pathway
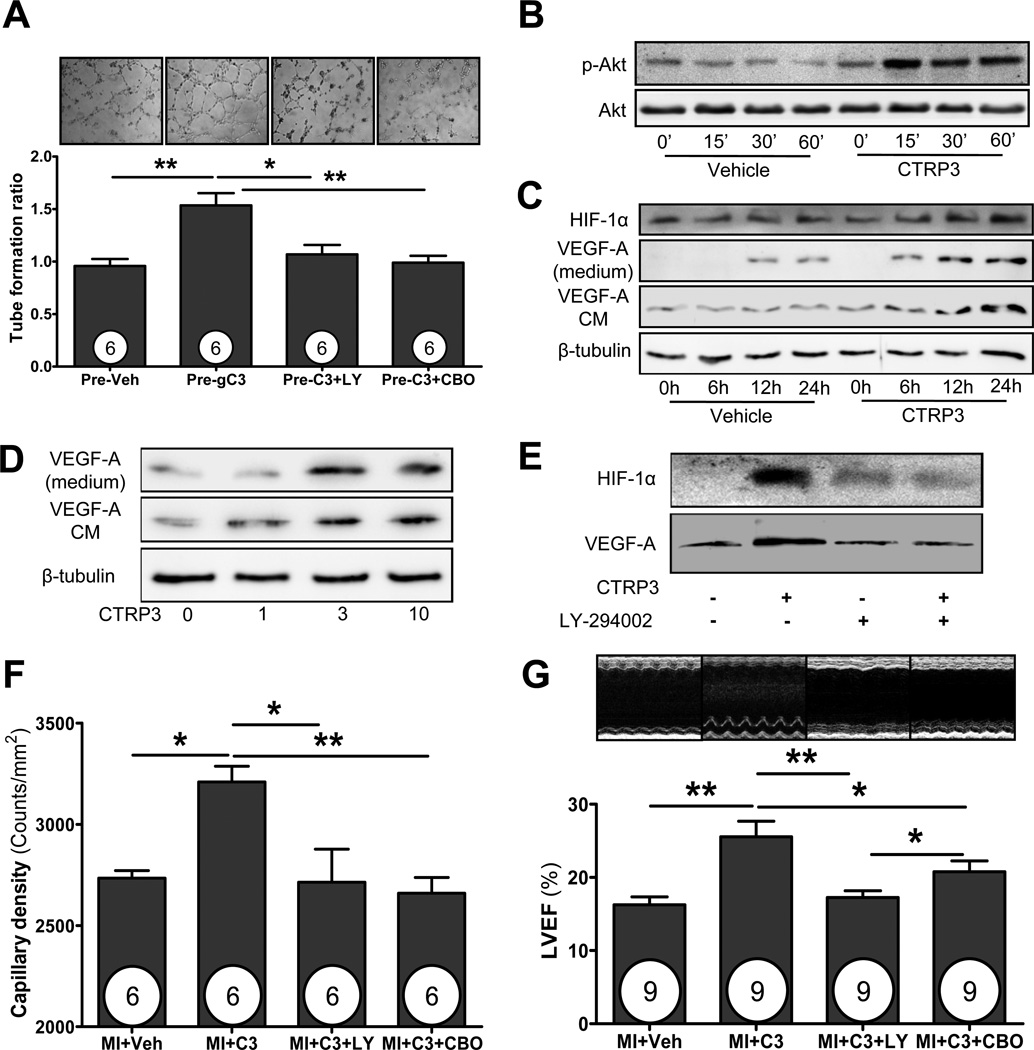
Recognizing that CTRP3 induced angiogenesis in vivo, but not in endothelial cells in vitro, we considered the hypothesis that cardiomyocytes, in response to CTRP3 stimulation, might secrete factors promoting angiogenesis in endothelial cells (cardiomyocyte-endothelial cell communication). To test this hypothesis, adult mouse cardiomyocytes were isolated as described in our previous study15, and treated with CTRP3 in vitro for varying periods (6, 12, and 24 hours). The conditioned medium from CTRP3-treated cardiomyocytes was then administered to cultured HUVECs for 6 hours. As illustrated in Figure 7A, HUVEC treatment with conditioned medium significantly enhanced HUVEC tube formation, suggesting that CTRP3 invokes an angiogenic response by inducing cardiomyocyte-secreted paracrine factors. Furthermore, CTRP3 up-regulated levels of pAkt, HIF-1α, and VEGF in cultured cardiomyocytes in both time-(Figure 7B and C) and dose-dependent manner (Figure 7D). These effects were not inhibited by pre-treatment with polymixin B (online supplementation Figure 4). Additionally, blocking cardiomyocyte Akt activation with a PI3-kinase inhibitor (LY294002) abolished CTRP3-induced HIF-1α and VEGF up-regulation (Figure 7E), and eliminated HUVEC tube formation induced by the conditioned medium (Figure 7A, third bar). Finally, HUVEC tube formation induced by the conditioned medium from CTRP3-treated cardiomyocytes was significantly suppressed by a VEGF receptor antagonist CBO-P11 (12 µM, Calbiochem) administered directly to HUVECs (Figure 7A, last bar).
Figure 7. CTRP3 promotes angiogenesis via a cardiomyocyte-mediated, Akt-dependent signaling mechanism in vitro.
(A) Tube formation assay. Isolated adult cardiomyocytes were pretreated with CTRP3 (3 µg/ml) or saline as vehicle for 12 hours. HUVECs were treated with CTRP3-pretreated conditioned medium (Pre-C3) or saline-pretreated medium (Pre-veh). Specific inhibitors of PI3-K (LY294002) and VEGF (CBO-P11) were added to culture medium of cardiomyocytes or HUVECs, respectively. Western blot analysis showing time dependent activation of cardiac Akt (B), cardiac expression of HIF-1α and VEGF in both cardiomyocytes (CM) and culture medium (C) after treatment with CTRP3 or vehicle; (D) Western blot analysis showing dose-dependent increasing of VEGF in both cardiomyocyte (CM) and culture medium (n=3); (E) Western blot analysis showing CTRP3-induced cardiomyocyte HIF-1α and VEGF expression was blocked by an Akt inhibitor; (F) Capillary density measured by CD31 immunohistochemical staining in the border region. Results show that CTRP3-induced capillary formation in vivo is abolished by co-administration of an Akt inhibitor (LY294002) or a VEGF receptor blocker (CBO-P11); (G) Echocardiographic analysis showing cardiac functional improvement by CTRP3 is completely blocked by Akt inhibition and significantly inhibited by VEGF inhibition. *P<0.05, **P<0.01
In vitro experimental results presented above demonstrate that CTRP3 enhances HUVEC tube formation by promoting cardiomyocyte/endothelial cell communication involving Akt-HIF-1α-VEGF signaling. To determine whether this signaling pathway is also responsible for CTRP3 cardioprotection observed in vivo, LY294002 (to inhibit Akt) or CBO-P11 (to inhibit VEGF) was administered together with CTRP3 via mini-osmotic pump, and cardiac function and capillary density were determined 14 days post-MI. As summarized in Figure 7F, CTRP3-induced capillary formation was completely abolished when either Akt or VEGF inhibited. However, the cardiac protective effect of CTRP3 (determined by LVEF) was completely blocked by LY294002 and partially blocked by CBO-P11 (Figure 7G). These results suggest that although the cardioprotective effect of CTRP3 is largely mediated by Akt-HIF-1α-VEGF signaling-induced angiogenesis, other mechanisms exist that also contribute to CTRP3’s cardioprotection against MI.
Endogenous CTRP3 produced under physiological conditions is cardioprotective
In a final attempt to obtain more evidence that CTRP3 is an endogenous cardioprotective adipokine, an additional in vitro experiment was performed as recently reported16. 3T3-L1 fibroblasts (ATCC, Manassas, VA, USA) were maintained per manufacturer’s protocol, and transfected with CTRP3 siRNA or scramble siRNA. 48 hours after transfection, cells were washed and cultured for an additional 6 hours. Conditioned medium from 3T3-L1 cells was then collected. Adult cardiomyocytes were isolated as described previously15, incubated with regular control culture medium or conditioned medium from control (no siRNA), scramble or CTRP3 siRNA pre-treated 3T3-L1 cells, and exposed to normoxia (20% oxygen) or hypoxia (2% oxygen) conditions. LDH concentration in culture medium and caspase-3 activity in cardiomyocytes were determined 12 hours after normoxia/hypoxia incubation. Compared to regular control medium, hypoxia-induced LDH release (Figure 8A) and caspase-3 activation (Figure 8B) were significantly attenuated in cardiomyocytes cultured with conditioned medium from 3T3-L1 cells (second vs. fourth bars). Pre-treatment of 3T3-L1 cells with scramble siRNA did not alter the protective effect offered by adipocyte conditioned medium (fourth vs. fifth bars in Figure 8A and 8B). However, the protective effect conferred by adipocyte conditioned medium was significantly blunted (last bars in Figure 8A and 8B) when cardiomyocytes were cultured with conditioned medium from CTRP3-knockdown 3T3-L1 cells (72±2.2% reduced CTRP3 protein expression, online supplementation 1C). These results indicate that CTRP3 deficiency tilts the balance between cytoprotective and cytotoxic molecules produced by 3T3-L1 cells, indicating that CTRP3 is an endogenous cardioprotective adipokine.
Figure 8. Endogenous CTRP3 produced under physiological conditions is cardioprotective.
LDH release (A) and caspase-3 activity (B) from adult cardiomyocytes (cultured with regular culture medium, adipocyte conditioned medium with or without CTRP3 knockdown) exposed to normoxia or hypoxia (2% O2 for 12 hours); *P<0.05, **P<0.01. Con-M=Conditioned medium. (C) Proposed mechanism responsible for the cardioprotective effects of CTRP3 post-MI.
Discussion
Our study presents several important observations: 1) To the best of our knowledge, we demonstrate for the first time that expression and production of CTRP3, a key member of a newly identified adipokine family, are significantly reduced post-MI; 2) replenishment of CTRP3 improves survival and restores cardiac function after MI; 3) CTRP3 attenuates post-ischemic pathologic remodeling, as evidenced by reduced heart size, preserved left ventricular chamber dimensions, decreased remote area interstitial fibrosis, increased ischemic regional cardiomyocyte survival/regeneration, and enhanced infarct border zone revascularization and reduced apoptosis; and 4) CTRP3 exerts its angiogenic effect by promoting cardiomyocyte-endothelial cell communication involving Akt-HIF1α-VEGF signaling.
The extent of tissue loss in the acute phase following MI is a major determinant of resultant cardiac dysfunction degree. However, chronic processes such as extracellular matrix turnover, fibrosis, and inflammation stimulate adverse remodeling, and are crucial determinants in the transition from compensatory cardiac hypertrophy to decompensatory heart failure17. Many cytokines have been reported to either mediate post-MI adverse remodeling or prevent cardiac remodeling and dysfunction. It is thus widely accepted that the degree of post-MI cardiac remodeling is critically dependent upon the balance between detrimental and protective cytokines18. Due to increased diabetes incidence, and its association with heart failure, the role of adipocyte-derived cytokines (adipokines) in the development of post-ischemic remodeling has attracted great attention2. The effects of leptin and adiponectin, among many adipokines examined, upon cardiac remodeling have been extensively investigated in recent years. The majority of published studies demonstrate the opposing effects of these adipokines upon post-ischemic remodeling, and the balance between detrimental leptin and protective adiponectin is critically influential upon post-MI heart failure development19, 20. In patients with type-2 diabetes, the balance is disturbed, with markedly increased plasma leptin levels and significantly reduced plasma adiponectin levels, favoring adverse cardiac remodeling2. Therefore, identifying cardioprotective adipokines, and augmenting their production, is scientifically and clinically imperative. In the current study, we have provided the first direct evidence that CTRP3 is an adipokine possessing strong anti-remodeling and cardioprotective properties, suggesting that CTRP3 might be a novel therapeutic target attenuating post-ischemic cardiac remodeling, improving cardiac function.
Angiogenesis represents an excellent therapeutic modality for ischemic heart disease treatment. Many preclinical studies have been performed with gene-, cell- and protein-based therapies17. Gene therapy, once regarded as a potential advanced treatment of choice for cardiovascular disease, remains impractical for regular clinical application due to many serious, unsolved problems. Cell-based pro-angiogenic therapies linger in early research stages, hindered by numerous unresolved questions regarding best employable cell types and dosages. The failure of gene- or cell-based therapeutics to deliver, as of yet, a suitable treatment choice for diseases stemming from poor blood flow, has led to resurgence of interest returning to protein-based therapies stimulating angiogenesis.
Among many different growth factors implicated in angiogenesis, VEGF and fibroblast growth factor (FGF) are the two most intensively investigated growth factors. Numerous animal model experimental studies have demonstrated that VEGF or FGF treatment stimulates angiogenesis with great efficacy, increasing capillary quantity, and alleviating post-ischemic remodeling21. Reproducible and credible successes in these early animal studies garnered enthusiastic expectations such new therapeutic approaches could be rapidly translated for clinical benefit in millions of MI patients. Unfortunately, in spite of many pioneering clinical trials’ optimistic early data, recent larger and better-designed clinical trials have reported one disappointment after another22–24. These failures suggest: 1) incorrectly selected molecular targets inducing neovascularization; 2) improper therapeutic agent formulation or administration; or 3) ignorance of the overall cellular micro-environmental context, affecting therapeutic utility and efficacy. Presentation of therapeutic proteins in a manner mimicking natural signaling events, accounting for concentration, spatial and temporal profiles, and their simultaneous or sequential presentation with other appropriate factors may be requisite for salutary effects.
Our current study demonstrates that CTRP3 effectively promotes post-ischemic angiogenesis and attenuates adverse remodeling. More interestingly, we have demonstrated that CTRP3 stimulates angiogenesis by promoting cardiomyocyte-endothelial cell communication, a process closely mimicking natural cardiac signaling events. Considerable evidence supports that communication between endothelial cells and cardiomyocytes regulates not only early cardiac development, but also adult cardiomyocyte function, including the contractile state25. Cardiomyocytes depend upon endothelial cells, not only for oxygenated blood supply, but also for locally protective signals promoting cardiomyocyte organization and survival. While endothelial cells direct cardiomyocytes, cardiomyocytes reciprocally secrete factors impacting endothelial cell function. Specifically, cardiomyocyte-specific deletion of VEGF-A results in vasculogenic/angiogenic defects, and a thinned ventricular wall26. Additionally, erythropoietin, a cardioprotective cytokine, has been recently demonstrated to promote angiogenesis by increasing cardiomyocyte VEGF expression27. It is thus highly likely that interventions stimulating the ongoing molecular conversation between endothelial cells and cardiomyocytes may more closely mimic natural cardiac development events, and thus exert superior cardioprotection compared to direct growth factor administration. Presently, whether CTRP3 may stimulate other mediators required for different angiogenesis stages remains unknown. However, the fact that CTRP3 is known to directly promote endothelial cell proliferation and migration, but not enhance tube formation (a process requiring complexity beyond proliferation and migration), suggests that pro-angiogenic factors other than VEGF might also be released by cardiomyocytes after CTRP3 treatment. This intriguing possibility will be investigated in our future studies. Moreover, although our in vitro experiments demonstrated that conditioned medium from CTRP3-treated cardiomyocytes promotes angiogenesis in cultured endothelial cells, and that CTRP3 significantly promoted angiogenesis in MI heart, administration of CTRP3 in the sham-operated control heart only slightly increased capillary density (Figure 4A). These results suggest that under normal physiologic conditions (in vivo normal heart), angiogenesis is tightly controlled by a balance between pro- and anti-angiogenesis factors, and that the pro-angiogenic effect of CTRP3 cannot be observed under this condition. The important underlying mechanisms responsible for the differences between in vitro and in vivo experimental results, and between normal and MI heart, will be directly investigated in our future experiments.
It is worth noting that the CTRP3-treated group manifested not only significantly increased angiogenesis and dramatically reduced fibrosis, but also marked preservation of intact myocytes in the infarct area at risk (Figure 3A). The precise mechanisms responsible for this protection are likely complex, and cannot be answered in a single experiment. Multiple possibilities exist. Firstly, the potent anti-apoptotic effect of CTRP3 observed in the border zone may block or attenuate infarct expansion. Secondly, the significant angiogenic effect of CTRP3 observed in the border zone may facilitate blood flow restoration, thus salvaging dying cardiomyocytes in infarcted regions, and/or promote endothelial cell trans-differentiation into cardiomyocytes28. Finally, CTRP3 may promote homing of cardiac stem cells from bone marrow and/or stimulate differentiation of resident cardiac stem cells. Such intriguing possibilities all warrant direct investigation.
Although CTRP3 is a paralog of adiponectin, our current study demonstrates distinctly different cardioprotective signaling between CTRP3 and adiponectin. It is well accepted that adiponectin promotes angiogenesis via AMPK signaling activation, and augmented NO production29. However, CTRP3 had no significant effect upon AMPK or eNOS phosphorylation, and failed to stimulate NO production. In contrast, CTRP3 induced significant Akt phosphorylation, both in vivo and in cultured cardiomyocytes. More importantly, inhibition of Akt phosphorylation abolished CTRP3-induced HIF-1α and VEGF expression, and blocked the angiogenic effect of CTRP3. These results support an Akt-dependent mechanism for CTRP3’s angiogenic effects.
In conclusion, we have demonstrated that CTRP3, a key member of a newly identified adipokine family, directly prevented apoptotic death of cardiomyocytes, increased cardiomyocyte survival/regeneration, attenuated post-infarct fibrosis, and enhanced cardiomyocyte expression of angiogenic cytokines inductive of robust angiogenesis, thus attenuating post-MI remodeling and augmenting post-MI contractile function (Figure 8E). Preventing post-MI CTRP3 inhibition or CTRP3 supplementation may be a promising therapeutic avenue creating stable and functional vessels in post-infarcted myocardium, restoring cardiac function, and mitigating the heart failure phenotype.
Supplementary Material
Clinical Perspective.
Cardiovascular disease remains a leading cause of mortality worldwide. Although improved reperfusion strategies have led to declined death rates after acute myocardial infarction (MI), both incidence and prevalence of post-MI heart failure have continually increased in recent years. Due to increased incidence of diabetes and its association with heart failure, there is now great interest in elucidating the underlying molecular mechanisms linking these pathologies. Considerable evidence exists that the majority of adipokines (including tumor necrosis factor-α, leptin, plasminogen activator inhibitor type 1, transforming growth factor β, and resistin) adversely regulate myocardial metabolism, cardiomyocyte hypertrophy, extracellular matrix structure and composition, and cell death. Our current study demonstrated that CTRP3, a key member of a newly identified adipokine family, exerts opposing cardiac actions as the majority of other adipokines, and is a novel and potent cardioprotective molecule. Expression and production of CTRP3 are significantly reduced post-MI, and replenishment of CTRP3 protects the heart by attenuating post-ischemic pathologic remodeling and promoting cardiomyocyte-endothelial cell communication. These results suggest the balance between cardioprotective and cardio-harmful cytokines tilts during pathological conditions such as heart failure. Interventions restoring the balance may represent a novel therapeutic strategy in the treatment of cardiovascular diseases, particularly in those patients with diabetes.
Acknowledgments
Funding Sources:
This research was supported by the following grants: NIH HL-63828, HL-096686, American Diabetes Association 7-11-BS-93 (XLM), and American Diabetes Association 1-11-JF56 (YJW).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosures: None
Reference
- 1.Roger VL, Go AS, Lloyd-Jones DM, Adams RJ, Berry JD, Brown TM, Carnethon MR, Dai S, de SG, Ford ES, Fox CS, Fullerton HJ, Gillespie C, Greenlund KJ, Hailpern SM, Heit JA, Ho PM, Howard VJ, Kissela BM, Kittner SJ, Lackland DT, Lichtman JH, Lisabeth LD, Makuc DM, Marcus GM, Marelli A, Matchar DB, McDermott MM, Meigs JB, Moy CS, Mozaffarian D, Mussolino ME, Nichol G, Paynter NP, Rosamond WD, Sorlie PD, Stafford RS, Turan TN, Turner MB, Wong ND, Wylie-Rosett J. Heart disease and stroke statistics--2011 update: a report from the American Heart Association. Circulation. 2011;123:e18–e209. doi: 10.1161/CIR.0b013e3182009701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schram K, Sweeney G. Implications of myocardial matrix remodeling by adipokines in obesity-related heart failure. Trends Cardiovasc Med. 2008;18:199–205. doi: 10.1016/j.tcm.2008.10.001. [DOI] [PubMed] [Google Scholar]
- 3.Ouchi N, Shibata R, Walsh K. Cardioprotection by adiponectin. Trends Cardiovasc Med. 2006;16:141–146. doi: 10.1016/j.tcm.2006.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.O'Shea KM, Chess DJ, Khairallah RJ, Rastogi S, Hecker PA, Sabbah HN, Walsh K, Stanley WC. Effects of adiponectin deficiency on structural and metabolic remodeling in mice subjected to pressure overload. Am J Physiol Heart Circ Physiol. 2010;298:H1639–H1645. doi: 10.1152/ajpheart.00957.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kishore U, Gaboriaud C, Waters P, Shrive AK, Greenhough TJ, Reid KB, Sim RB, Arlaud GJ. C1q and tumor necrosis factor superfamily: modularity and versatility. Trends Immunol. 2004;25:551–561. doi: 10.1016/j.it.2004.08.006. [DOI] [PubMed] [Google Scholar]
- 6.Wong GW, Wang J, Hug C, Tsao TS, Lodish HF. A family of Acrp30/adiponectin structural and functional paralogs. Proc Natl Acad Sci U S A. 2004;101:10302–10307. doi: 10.1073/pnas.0403760101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kopp A, Bala M, Weigert J, Buchler C, Neumeier M, Aslanidis C, Scholmerich J, Schaffler A. Effects of the new adiponectin paralogous protein CTRP-3 and of LPS on cytokine release from monocytes of patients with type 2 diabetes mellitus. Cytokine. 2010;49:51–57. doi: 10.1016/j.cyto.2009.10.001. [DOI] [PubMed] [Google Scholar]
- 8.Weigert J, Neumeier M, Schaffler A. The adiponectin paralog CORS-26 has anti-inflammatory properties and is produced by human monocytic cells. FEBS Lett. 2005;579:5565–5570. doi: 10.1016/j.febslet.2005.09.022. [DOI] [PubMed] [Google Scholar]
- 9.Akiyama H, Furukawa S, Wakisaka S, Maeda T. CTRP3/cartducin promotes proliferation and migration of endothelial cells. Mol Cell Biochem. 2007;304:243–248. doi: 10.1007/s11010-007-9506-6. [DOI] [PubMed] [Google Scholar]
- 10.Wolfing B, Buechler C, Weigert J, Neumeier M, Aslanidis C, Schoelmerich J, Schaffler A. Effects of the New C1q/TNF-related Protein (CTRP-3) [ldquo]Cartonectin[rdquo] on the Adipocytic Secretion of Adipokines. Obesity. 2008;16:1481–1486. doi: 10.1038/oby.2008.206. [DOI] [PubMed] [Google Scholar]
- 11.Peterson JM, Wei Z, Wong GW. C1q/TNF-related Protein-3 (CTRP3), a Novel Adipokine That Regulates Hepatic Glucose Output. J Biol Chem. 2010;285:39691–39701. doi: 10.1074/jbc.M110.180695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kopp A, Bala M, Buechler C, Falk W, Gross P, Neumeier M, Scholmerich J, Schaffler A. C1q/TNF-Related Protein-3 Represents a Novel and Endogenous Lipopolysaccharide Antagonist of the Adipose Tissue. Endocrinology. 2010;151:5267–5278. doi: 10.1210/en.2010-0571. [DOI] [PubMed] [Google Scholar]
- 13.Gao E, Lei YH, Shang X, Huang ZM, Zuo L, Boucher M, Fan Q, Chuprun JK, Ma XL, Koch WJ. A novel and efficient model of coronary artery ligation and myocardial infarction in the mouse. Circ Res. 2010;107:1445–1453. doi: 10.1161/CIRCRESAHA.110.223925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tao L, Gao E, Jiao X, Yuan Y, Li S, Christopher TA, Lopez BL, Koch W, Chan L, Goldstein BJ, Ma XL. Adiponectin cardioprotection after myocardial ischemia/reperfusion involves the reduction of oxidative/nitrative stress. Circulation. 2007;115:1408–1416. doi: 10.1161/CIRCULATIONAHA.106.666941. [DOI] [PubMed] [Google Scholar]
- 15.Wang Y, Gao E, Tao L, Lau WB, Yuan Y, Goldstein BJ, Lopez BL, Christopher TA, Tian R, Koch W, Ma XL. AMP-activated protein kinase deficiency enhances myocardial ischemia/reperfusion injury but has minimal effect on the antioxidant/antinitrative protection of adiponectin. Circulation. 2009;119:835–844. doi: 10.1161/CIRCULATIONAHA.108.815043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Palanivel R, Vu V, Park M, Fang X, Sweeney G. Differential impact of adipokines derived from primary adipocytes of wild-type versus streptozotocin-induced diabetic rats on glucose and fatty acid metabolism in cardiomyocytes. J Endocrinol. 2008;199:389–397. doi: 10.1677/JOE-08-0336. [DOI] [PubMed] [Google Scholar]
- 17.Eapen Z, Rogers JG. Strategies to attenuate pathological remodeling in heart failure. Curr Opin Cardiol. 2009;24:223–229. doi: 10.1097/HCO.0b013e32832a11ff. [DOI] [PubMed] [Google Scholar]
- 18.Nian M, Lee P, Khaper N, Liu P. Inflammatory cytokines and postmyocardial infarction remodeling. Circ Res. 2004;94:1543–1553. doi: 10.1161/01.RES.0000130526.20854.fa. [DOI] [PubMed] [Google Scholar]
- 19.Purdham DM, Rajapurohitam V, Zeidan A, Huang C, Gross GJ, Karmazyn M. A neutralizing leptin receptor antibody mitigates hypertrophy and hemodynamic dysfunction in the postinfarcted rat heart. Am J Physiol Heart Circ Physiol. 2008;295:H441–H446. doi: 10.1152/ajpheart.91537.2007. [DOI] [PubMed] [Google Scholar]
- 20.Sam F, Duhaney TA, Sato K, Wilson RM, Ohashi K, Sono-Romanelli S, Higuchi A, De Silva DS, Qin F, Walsh K, Ouchi N. Adiponectin deficiency, diastolic dysfunction, and diastolic heart failure. Endocrinology. 2010;151:322–331. doi: 10.1210/en.2009-0806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lavu M, Gundewar S, Lefer DJ. Gene therapy for ischemic heart disease. J Mol Cell Cardiol. 2010;50:742–750. doi: 10.1016/j.yjmcc.2010.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Henry TD, Annex BH, McKendall GR, Azrin MA, Lopez JJ, Giordano FJ, Shah PK, Willerson JT, Benza RL, Berman DS, Gibson CM, Bajamonde A, Rundle AC, Fine J, McCluskey ER. The VIVA trial: Vascular endothelial growth factor in Ischemia for Vascular Angiogenesis. Circulation. 2003;107:1359–1365. doi: 10.1161/01.cir.0000061911.47710.8a. [DOI] [PubMed] [Google Scholar]
- 23.Simons M, Annex BH, Laham RJ, Kleiman N, Henry T, Dauerman H, Udelson JE, Gervino EV, Pike M, Whitehouse MJ, Moon T, Chronos NA. Pharmacological treatment of coronary artery disease with recombinant fibroblast growth factor-2: double-blind, randomized, controlled clinical trial. Circulation. 2002;105:788–793. doi: 10.1161/hc0802.104407. [DOI] [PubMed] [Google Scholar]
- 24.Grines CL, Watkins MW, Helmer G, Penny W, Brinker J, Marmur JD, West A, Rade JJ, Marrott P, Hammond HK, Engler RL. Angiogenic Gene Therapy (AGENT) trial in patients with stable angina pectoris. Circulation. 2002;105:1291–1297. doi: 10.1161/hc1102.105595. [DOI] [PubMed] [Google Scholar]
- 25.Hsieh PC, Davis ME, Lisowski LK, Lee RT. Endothelial-cardiomyocyte interactions in cardiac development and repair. Annu Rev Physiol. 2006;68:51–66. doi: 10.1146/annurev.physiol.68.040104.124629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Giordano FJ, Gerber HP, Williams SP, VanBruggen N, Bunting S, Ruiz-Lozano P, Gu Y, Nath AK, Huang Y, Hickey R, Dalton N, Peterson KL, Ross J, Jr, Chien KR, Ferrara N. A cardiac myocyte vascular endothelial growth factor paracrine pathway is required to maintain cardiac function. PNAS. 2001 doi: 10.1073/pnas.091415198. 091415198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ueda K, Takano H, Niitsuma Y, Hasegawa H, Uchiyama R, Oka T, Miyazaki M, Nakaya H, Komuro I. Sonic hedgehog is a critical mediator of erythropoietin-induced cardiac protection in mice. J Clin Invest. 2010;120:2016–2029. doi: 10.1172/JCI39896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Condorelli G, Borello U, De AL, Latronico M, Sirabella D, Coletta M, Galli R, Balconi G, Follenzi A, Frati G, Cusella De Angelis MG, Gioglio L, Amuchastegui S, Adorini L, Naldini L, Vescovi A, Dejana E, Cossu G. Cardiomyocytes induce endothelial cells to trans-differentiate into cardiac muscle: implications for myocardium regeneration. Proc Natl Acad Sci U S A. 2001;98:10733–10738. doi: 10.1073/pnas.191217898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shimano M, Ouchi N, Shibata R, Ohashi K, Pimentel DR, Murohara T, Walsh K. Adiponectin deficiency exacerbates cardiac dysfunction following pressure overload through disruption of an AMPK-dependent angiogenic response. J Mol Cell Cardiol. 2010;49:210–220. doi: 10.1016/j.yjmcc.2010.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.