Abstract
Purpose
Recent investigations have revealed that populations of macrophages and dendritic cells (DCs) are present in the stroma and epithelium of the cornea, although the precise phenotype and distribution are still controversial. CX3CR1, the sole receptor for the chemokine fractalkine, is expressed by these monocyte-derived cells. Transgenic CX3CR1GFP mice, in which either one (heterozygous) or both (homozygous) copies of the CX3CR1 gene were replaced by enhanced green fluorescent protein (eGFP), were used to characterize monocyte-derived cells in the mouse cornea and to determine whether the expression of this receptor influences the recruitment of these cells into the normal cornea.
Methods
Wholemount corneas were immunostained with anti-leukocyte antibodies to the phenotypic markers major histocompatibility complex (MHC) class II, CD169, CD68, CD11b, and CD45 and analyzed by epifluorescence and confocal microscopy. The density of intraepithelial MHC class II+ cells was quantified in wild-type, CX3CR1+/GFP heterozygous, CX3CR1GFP/GFP homozygous, and CX3CR1-knockout mice.
Results
There was a significant reduction in the number of MHC class II+ cells (putative DCs) in the corneal epithelium of CX3CR1-deficient mice (P < 0.009) compared with wild-type mice, and the few cells that were present did not possess classic dendriform morphology. No GFP+ MHC class II– cells were noted in the epithelium. Dual immunostaining of corneas in both heterozygous and homozygous (CX3CR1-deficient) mice revealed GFP+ cells with a more pleomorphic morphology throughout the entire corneal stroma that were CD11b+ CD169+, and had variable degrees of expression of CD68 and MHC class II. The immunophenotype and morphology of these intrastromal cells is strongly indicative of a macrophage phenotype.
Conclusions
This study has identified a role for CX3CR1 in the normal recruitment of MHC class II+ putative DCs into the corneal epithelium and establishes a model for investigating monocyte-derived cells and fractalkine/CX3CR1 interactions during corneal disease.
The topographical distribution and functional heterogeneity of cells of the mononuclear phagocyte system1,2 in the cornea are most likely crucial in the course of innate immune responses, such as bacterial keratitis. Furthermore, the role of dendritic cells (DCs) and macrophages in the afferent and efferent arms of cell-mediated corneal immune responses has significant implications for the outcome of several clinical conditions such as corneal transplant rejection.3 The central region of the mammalian cornea was traditionally thought to be largely devoid of DCs or resident tissue macrophages4 - 6; however, it was well recognized that the peripheral corneal and limbal epithelia contain a resident population of major histocompatibility complex (MHC) class II+ intraepithelial DCs, sometimes referred to as Langerhans cells (LCs), which decline sharply in density centripetally.7 This was the generally held view until the recent discovery of heterogeneous populations of CD11c+ MHC class II– DC in the normal murine central corneal epithelium and stroma.3,8-10 In addition, there was an independent discovery of populations of CD11b+ resident macrophages within the murine corneal stroma,11 an observation subsequently confirmed by Sosnova et al.12 Activation of resident corneal and limbal DC and macrophages during keratoplasty or keratitis8,13-15 or after exposure to proinflammatory stimuli, such as LPS or TNF-α in vitro, has been clearly demonstrated.16
In addition to subsets of tissue-specific, monocyte-derived macrophage populations (e.g., Kupffer cells in liver, alveolar macrophages, and microglia in neural parenchyma), it is now increasingly recognized that resident macrophages can be further divided into distinct subsets on the basis of their cytokine secretion and expression of chemokine receptors. Chemokines comprise a family of structurally related proteins that play a pivotal role in leukocyte emigration from blood vessels and can be classified as belonging to either the C, CC, CXC, or CX3C subfamily according to the arrangement of their (N)-terminal cysteine motifs.17 The sole member of the CX3C, or δ-chemokine, subfamily is the novel chemokine CX3CL1, also known as fractalkine or neurotactin.18–20 Fractalkine is a membrane-bound glycoprotein that sits atop an extended mucin-like stalk. It can assume a soluble form after proteolytic cleavage at an extracellular site near the plasma membrane.18,21 Through interaction with its unique receptor, CX3CR1, fractalkine is able to mediate cell–cell adhesion when membrane bound, and in its soluble form it acts to mediate cell migration of CX3CR1-bearing cells such as monocytes, NK cells, T-cells, DCs, and macrophages including microglia.19,22,23 Constitutive expression of fractalkine has been demonstrated in vitro on epithelial cells in the gut, skin, and tonsils24 and lamina propria of the small intestine.25,26 Numerous studies on the biological role of the fractalkine/CX3CR1 dyad demonstrate contributions to the development of several inflammatory diseases including atherosclerosis, psoriasis, rheumatoid arthritis, and experimental autoimmune myositis.27–30 Recently, the ability to investigate the in vivo fate of blood monocytes experimentally has been greatly enhanced by the development of mice in which a green fluorescent protein (eGFP)–encoding gene is inserted in one or both copies of the CX3CR1 locus.26,31 This model and the adoptive transfer of labeled monocytes from these CX3CR1GFP mice into wild-type (WT) recipients has begun to shed light on monocyte heterogeneity and factors regulating their differentiation within tissues, and in particular the role of the chemokine receptor CX3CR1 in the accumulation of normal resident monocyte-derived DC and macrophages in a diverse range of tissues.32 By investigating heterozygous (CX3CR1+/GFP) and homozygous (CX3CR1GFP/GFP) transgenic mice, Niess et al.26 demonstrated a role for CX3CR1 in the formation of transepithelial dendrites by intestinal DCs. The deficiency of CX3CR1 in homozygous mice led to impaired sampling of intestinal lumen antigens and impaired defense during bacterial challenge.
In the present study, we took advantage of CX3CR1GFP transgenic mice31 to confirm the presence of monocyte-derived cells in the normal murine cornea. Furthermore, we investigated whether CX3CR1 expression plays a role in DC and macrophage recruitment in the normal corneal epithelium and stroma. The present study confirmed previous findings in several laboratories that the normal murine corneal stroma contains extensive populations of resident monocyte-derived macrophages. Our data revealed that CX3CR1-deficient mice lack MHC class II+ putative DCs in the corneal epithelium and indicate that the absence of fractalkine signaling hinders the ability of these cells to form the characteristic dendriform processes of DCs. These findings provide evidence that fractalkine/CX3CR1 interactions play a role in the normal homeo-static recruitment of bone-marrow derived monocytic cells into the corneal epithelium.
Materials and Methods
Animals
Transgenic heterozygous CX3CR1+/GFP mice, which retain a WT copy of the CX3CR1 receptor, and homozygous CX3CR1GFP/GFP mice, which are CX3CR1 deficient, were used in the present study. These transgenic mice were on a BALB/c background, with WTs of this strain used as control animals. In addition, conventional CX3CR1 knockout (KO) mice on a C57Bl/6 background (CX3CR1–/–) were obtained from Jackson Laboratory (Bar Harbor, ME). All experiments used female mice aged between 6 and 12 weeks, and all procedures conformed to the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research.
Tissue Collection and Immunostaining
Eyes were enucleated and fixed in 4% paraformaldehyde and stored at 4°C until further processing. Corneas were dissected from the eye, and radial incisions were made to produce eight pie-shaped wedges consisting of limbus and conjunctiva peripherally and central cornea apically, as previously documented.33 Tissue pieces were washed in PBS, incubated in 20 mM prewarmed EDTA tetrasodium for 30 minutes at 37°C, and then incubated with a 0.2% solution of Triton-X in PBS plus 1% bovine serum albumin for 5 minutes at room temperature (RT), to increase antibody penetration. In the CX3CR1GFP mice (heterozygotes, and homozygotes), corneas were double stained with anti-GFP and a range of primary monoclonal antibodies. After overnight incubation with rabbit anti-GFP (1:200; Chemicon, Temecula, CA) at 4°C, the tissues were washed and incubated with Alexa Fluor 488–conjugated goat anti-rabbit (1:100; Invitrogen-Molecular Probes, Eugene, OR) plus 10% mouse serum for 60 minutes at RT. After further washes with PBS, the tissues were incubated overnight at 4°C with anti-MHC class II (M5/114; 1:200; BD-Pharmingen, San Diego, CA) anti-CD169 (Ser4; 1:100; Serotec, Raleigh, NC), anti-CD68 (1:100; Serotec), CD11b (1: 100, Pharmingen), or CD45 (1:100 Serotec). The following day, the tissues were incubated with biotinylated goat anti-rat IgG (1:100; GE Healthcare, Piscataway, NJ) for 60 minutes at RT, washed three times, and incubated in streptavidin-Cy3 (1:100; Jackson ImmunoResearch Laboratories, West Chester, PA) for 60 minutes at RT. Tissues were incubated in DAPI (1:50 in PBS; Roche Molecular Biochemicals, Indianapolis, IN) for 10 minutes at RT. Stained tissue wholemounts were placed in aqueous mounting medium (Thermo Fisher Scientific, Pittsburgh, PA) onto glass slides and coverslipped. For C57Bl/6 CX3CR1+/+ (WT) and CX3CR1–/– (KO) mouse tissue, corneas were prepared in an identical manner except the anti-GFP primary and secondary antibodies were omitted, and MHC class II was visualized with Alexa Fluor 488–conjugated rabbit anti-rat IgG (Invitrogen-Molecular Probes). As negative control samples, either FITC-conjugated isotype rat IgG was substituted for primary antibodies or the step was omitted completely.
Examination of Corneal Mounts and Quantitative Analysis of Intraepithelial DCs
Corneal wholemounts were examined using both conventional epifluorescence microscopy (Olympus, Tokyo, Japan; DMRBE, Leica, Hawthorn East, VIC, Australia) and confocal microscopy (TCSSP2; Leica). To perform quantitative evaluation of bone-marrow–derived cells each wedge-shaped portion of the corneal wholemount was divided into three different regions or zones. The central region was defined as within 0.5 mm from the center of the cornea, the paracentral region was the area between 0.5 and 1.0 mm from the center, and the peripheral cornea was defined as the area between 1.0 mm and the limbal border.10 Intraepithelial DCs were counted in each region and converted to density/mm2 based on the area of the region evaluated (Metamorph 6.2 software; Molecular Devices, Downingtown, PA). At least three mice were used for each strain. Confocal microscopy was used to characterize the eGFP+ cells or immunopositive cells in the corneal stroma and epithelium. Images of corneal wholemounts were generated by a series of z-stacks from the internal aspect moving through to the external aspect in 0.5-μm increments in various regions of the cornea from the peripheral to the central cornea. Final image processing was performed with commercial image management software (Photoshop, ver 7.0; Adobe Systems, San Jose, CA).
Statistics
Statistical significance was determined with an unpaired t-test (Prism; GraphPad Software, San Diego, CA). P < 0.05 was considered to be significant.
Results
Effect of CX3CR1 Expression on Homing of MHC class II+ Putative DCs to the Corneal Epithelium
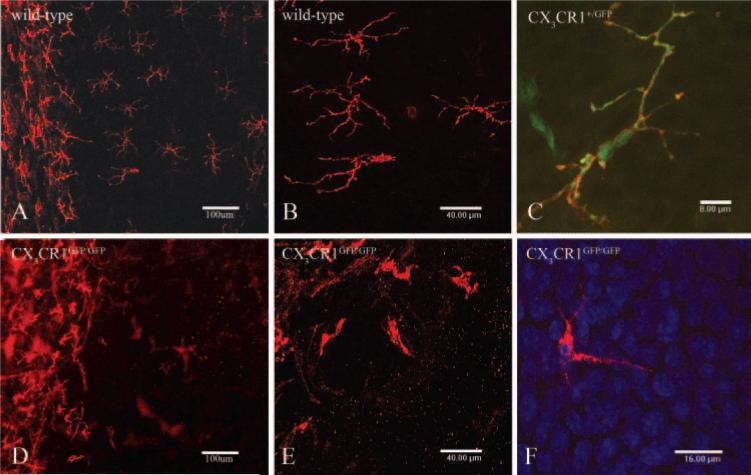
Immunofluorescence was used to investigate the distribution of MHC class II+ putative DC in the corneal epithelium in WT and heterozygous mice (Figs. 1A-C) and CX3CR1-deficient mice (Figs. 1D-F). In WT mice we observed a progressive decrease in density from the periphery to the central cornea (Figs. 1A, 1B). By contrast, CX3CR1GFP/GFP mice exhibited a dramatic reduction in the number of MHC class II+ cells in all regions of the corneal epithelium (Figs. 1D, 1E); however, the limbal/conjunctival populations appeared normal. In CX3CR1+/GFP mice, all intraepithelial MHC class II+ cells were CX3CR1+ (Fig. 1C). The intraepithelial MHC class II+ cells displayed extensive dendritic, ramified cell processes that interdigitated between the corneal epithelial cells (Figs. 1B, 1C). The MHC class II+ cells in the peripheral corneal epithelium in CX3CR1GFP/GFP mice (Figs. 1D–F), failed to form the classic network of cells seen in WT mice, and furthermore, those few cells present displayed a pleomorphic form lacking extensive ramified dendritic processes.
Figure 1.
Epifluorescence and confocal images of naïve WT (A, B), CX3CR1+/GFP (C), and CX3CR1GFP/GFP (D-F) mouse corneas stained with anti-MHC class II antibody. Low-power epifluorescence image of a WT mouse cornea (A) demonstrates the numerous resident MHC class II+ cells in the peripheral and limbal epithelium (left) in contrast with the low number of cells in the CX3CR1GFP/GFP mouse cornea (D). Intermediate magnification of intraepithelial MHC class II+ cells in the peripheral cornea of WT (B) and CX3CR1GFP/GFP mice (E) demonstrating the differing morphologic appearances of the putative DCs. MHC class II+ eGFP+ cell in the corneal epithelium of an CX3CR1+/GFP mouse (C) displaying typical dendriform morphology compared with the stuntedlike appearance of intraepithelial MHC class II+ cells of CX3CR1GFP/GFP mice (F). DAPI (blue)-stained nuclei (F) to confirm the anatomic position of the MHC class II+ cells in the corneal epithelium.
Quantitative analysis of MHC class II+ cell populations in the corneal epithelium confirm previous observations of a decline in density from the peripheral toward the central zone.4,7
In light of previous descriptions of MHC class II– CD11c+ intraepithelial DCs, we were particularly interested to determine whether there were any MHC class II– GFP+ cells. None were noted in either homozygous or heterozygous CX3CR1 mice, suggesting that the MHC class II+ cells described represents the entire population of intraepithelial putative DCs. Anti-CD11c staining was performed on cor-neal wholemounts, but results were variable and do not allow us to state unequivocally whether the putative DCs were CD11c positive or negative. This question is the subject of ongoing research.
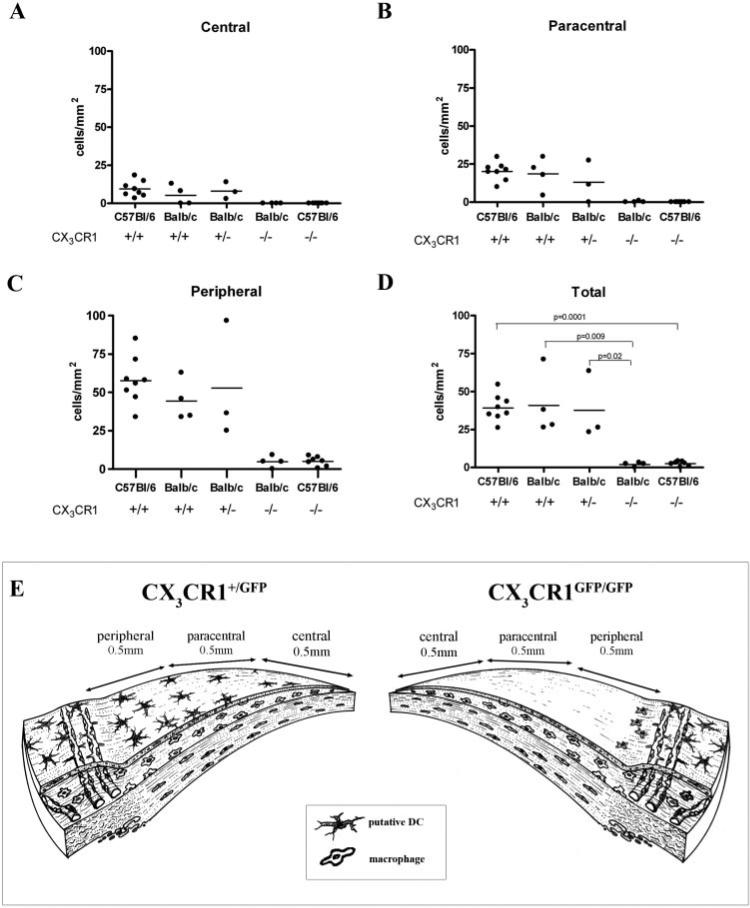
In addition to CX3CR1GFP transgenic mice on a BALB/c background our observations were further confirmed using CX3CR1 knockout mice on a C57Bl/6 genetic background. In CX3CR1-deficient mice on both genetic backgrounds the number of intraepithelial MHC class II+ cells in the central, para-central, and peripheral cornea was quantified by using epifluorescence microscopy. The distribution and density (per mm2) of these cells did not differ between WT C57Bl/6, BALB/c (both CX3CR1+/+) and CX3CR1+/GFP mice (Fig. 2B-D). However, there were statistically significant differences in the density of MHC class II+ cells in the corneal epithelium of CX3CR1-deficient mice compared with their WT counterparts in both BALB/c and C57Bl/6 genetic backgrounds (BALB/c; P < 0.0002; C57Bl/6; P < 0.009; Figs. 2B-D). In summary, the present study clearly revealed the importance of CX3CR1 expression in the homing of MHC class II+ putative DCs to the corneal epithelium and in the establishment of typical dendri-form morphology and network patterns (Fig. 2E).
Figure 2.
Quantitation of MHC class II+ cells in the corneal epithelium of CX3CR1GFP transgenic and CX3CR1 knockout mice and their WT counterparts (A–D). There were significantly fewer MHC class II+ cells in the central, paracentral, and peripheral corneal epithelium of CX3CR1GFP/GFP and CX3CR1–/– mice. Note the centripetal decline in the density of intraepithelial MHC class II+ cells (per mm2). (E) Summary diagram showing the zones of the cornea used in quantitative studies and the difference in the distribution of intraepithelial putative DCs between heterozygous (CX3CR1+/GFP) and homozygous (CX3CR1GFP/GFP) mice.
Phenotype of the eGFP/CX3CR1+ Cells in the Naïve Mouse Corneal Stroma
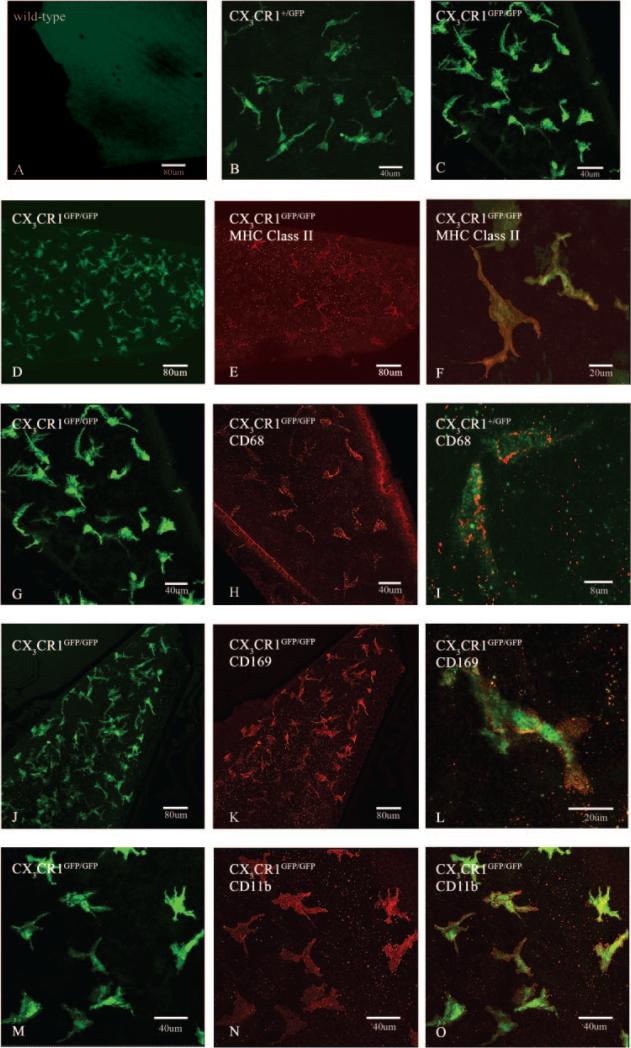
Corneal wholemounts from naïve WT, CX CR1+/GFP, and CX3CR1GFP/GFP mice were incubated with anti-eGFP to enhance the green signal and analyzed using confocal microscopy. In WT mouse corneas, there was no eGFP staining (Fig. 3A), whereas in heterozygous and homozygous mice, eGFP+ cells were evenly distributed in all regions and depths of the corneal stroma (Figs. 3B, 3C). This confirms that bone marrow monocytic-derived cells are a constitutive resident cell population of the corneal stroma and highlights the value of these transgenic animals in visualizing the normal resident monocytic-derived cells in the mouse cornea.
Figure 3.
Confocal microscopic analysis of corneal wholemounts from naïve WT (A), CX3CR1+/GFP (B, I), and CX3CR1GFP/GFP (C, D-H, J-O) mice. The epithelium was deliberately omitted from the z-stacked confocal images to better enable visualization of stromal eGFP+ cells. In WT mice (A) there were no eGFP+ cells in the cornea, whereas a large number of pleomorphic eGFP+ cells were seen in the corneas of CX3CR1+/GFP (B) and CX3CR1GFP/GFP mice (C). Immunophenotypic characterization using anti-leukocyte antibodies revealed a proportion of eGFP+ cells were MHC class II+/low (D-F) and CD68+/low (G-I) cells whereas all were CD169+ (sialoadhesin marker; J-L) and CD11b+ (M-O). All images were taken from the central or paracentral corneal stroma using either a ×20 or ×40 objective lens.
Immunophenotypic analysis using dual staining of corneal wholemounts was performed to characterize the eGFP+ cells in the corneal epithelium and stroma of CX3CR1GFP mice. In the corneal stroma of both CX3CR1+/GFP and CX3CR1GFP/GFP mice, a subpopulation of the large number of eGFP+ cells were MHC class II+; however, the level of expression was lower than in the intraepithelial population (Figs. 3D-F). There were no overt differences observed in the stromal MHC class II+ cells in either CX3CR1+/GFP or CX3CR1GFP/GFP mice. This result contrasts with the altered intraepithelial putative DC network observed in homozygous mice lacking this receptor, as described above.
In CX3CR1+/GFP and CX3CR1GFP/GFP mice, eGFP+ cells in the corneal stroma had a more pleomorphic morphology than intraepithelial putative DC or LC-like cells. Immunophenotypic analysis of these populations revealed that they coexpressed the recognized macrophages markers CD68 (Figs. 3G-I), CD169 (Figs. 3J-L), and CD11b (Figs. 3M-O). They were also all CD45+ (not illustrated).
There were no qualitative differences between the CX3CR1+/GFP and CX3CR1GFP/GFP mice in the density or morphology of eGFP+CD68+ or eGFP+CD169+ cells. The morphologic appearance, distribution, and phenotype are strong evidence that most of the bone marrow-derived cells in the corneal stroma are resident tissue macrophages.
Discussion
Until recently, a central tenet of corneal immunology was that the central mammalian cornea was largely devoid of DCs or resident tissue macrophages.34 Many observers had described intraepithelial DCs or LCs with characteristic dendriform morphology and immunophenotype (CD45+ MHC class II+) in the conjunctiva and limbal region, but noted a sharp centripetal decline in density.4,7 This paradigm has come under scrutiny with the recent discovery of heterogeneous populations of CD45+ CD11c+ MHC class II– DCs in the normal murine central corneal epithelium and stroma,3,8,10 as well as an independent discovery of populations of CD45+ CD11b+ resident macrophages throughout the murine corneal stroma.11 Subsequently, others have confirmed the presence of stromal macrophages in the mouse, a proportion of which are MHC II+ and also express CD34 (hemopoietic stem cell marker).12 In light of these more recent observations of monocyte-derived cell populations in the avascular cornea it is imperative that the signals involved in mediating the normal homeostatic turnover of these cells be determined. The chemokine receptor CX3CR1 has recently been shown to be critically involved in the normal homeostatic recruitment of monocyte-derived cells in a variety of tissues.26,32 Investigations of these dynamic events have been greatly aided by the development of CX3CR1GFP transgenic mice. In the present investigation, we sought to examine the role of CX3CR1 on homing of monocyte-derived cells in the murine cornea. Studying the corneas from CX3CR1 transgenic mice allowed the visualization of CX3CR1+ cells in both heterozygous (CX3CR1+/GFP) and homozygous (CX3CR1GFP/GFP) animals, in which the function of CX3CR1 is normal or deficient, respectively. With the aid of the combined approach of immunostaining techniques on wholemount corneas, in a variety of transgenic and knockout mice, we were able to identify a role for CX3CR1 in the normal homeostatic recruitment of MHC class II+ cells into the corneal epithelium.
Our observations of intraepithelial MHC class II+ cells (sometimes referred to as LCs) in WT and CX3CR1+/GFP corneas complement several previous descriptions (see review in Ref. 7) and confirmed that these cells declined sharply in density from the limbus toward the center of the cornea. We were unable to demonstrate CD11c reactivity in these cells because of the lack of consistent staining patterns. However, the failure to identify CX3CR1+/GFP cells (i.e., all monocyte-derived cells) in the corneal epithelium that were completely MHC class II– seems to suggest that the CD11c+ MHC class II– DCs described by Hamrah et al.8,10 and Dana3 were not present in our murine corneas.
Our data revealed a previously unknown CX3CR1 dependency on recruitment of putative DCs in the normal resting corneal epithelium. A similar CX3CR1-dependency of DC recruitment to an epithelial barrier under normal and inflamed conditions has been demonstrated in the gut.26 That study showed evidence of impaired bacterial sampling of the gut by intraepithelial DC in CX3CR1GFP/GFP mice, which the investigators postulated was the result of the altered morphology of the intraepithelial DC population, which appeared suboptimal for the trapping and sampling of luminal antigen. The statistically significant diminution of MHC class II+ cells in the cor-neal epithelium of CX3CR1GFP/GFP mice in the present study compared to WT BALB/c mice and a parallel observation in independent groups of CX3CR1–/– and WT mice on a C57Bl/6 background demonstrates that the presence of these putative DCs in the naive corneal epithelium is dependent on the expression of CX3CR1. Whether this is due to the expression of the ligand CX3CL1/fractalkine by the corneal epithelium is currently unknown, but is the focus of ongoing investigations.
Besides their postulated sentinel role in Ag capture and mediation of the adaptive immune responses in the cornea, it is emerging that APCs may be critical in innate immune responses.35 For example, it has been suggested that epithelial responses to pathogens, including primary production of proinflammatory cytokines and chemokines, are in fact due to pathogen recognition by resident DCs and macrophages via the activation of Toll-like receptors.36 We hypothesize that CX3CR1-deficient mice may have an impaired function in the homeostatic surveillance of the corneal epithelium due to their absence of intraepithelial MHC class II+ cells and studies to test this hypothesis are being performed presently.
Corneal epithelial DC networks may act as pivotal sentinels in the detection of invading pathogens. It is now becoming clear, however, that another population of monocyte-derived cells, resident tissue macrophages, and possibly DCs, within the corneal stroma, may constitute a secondary frontier line of immunoreactive cells that participate in the detection and destruction of pathogens, should they breach the epithelial barrier. In the present study, we did not observe any significant qualitative difference in the presence of stromal macrophages as defined by CX3CR1 expression and immunophenotypic analysis in naïve heterozygous and homozygous CX3CR1GFP animals. The observation of extensive networks of eGFP+ cells in the corneal stroma is of significance in two respects. First, it served to confirm previous descriptions of monocyte-derived cells in the normal mouse cornea.8,11,12 Second, it indicates that the homeostatic recruitment of mononuclear cells in the corneal stroma is not mediated by CX3CR1. The phenotypic characterization of corneal stromal monocyte-derived cells revealed that these cells in CX3CR1GFP mice were almost universally CD45+ CD11b+ CD169+ and CD68+/low, indicative of a macrophage phenotype. To the authors’ knowledge, this is the first time CD169, a sialic acid–binding receptor found on the surface of macrophages,37 has been identified in situ in the naïve mouse cornea. The lysosomal marker CD68, present in resident tissue macrophages and in small amounts in DCs,38 has recently been shown to be upregulated in primary keratocyte cultures.39 This finding may indicate either that activated keratocytes perform a phagocytic or endocytic role, a previously recognized property, or that cultured keratocytes contain remnants of stromal macrophage populations. Our data support the latter explanation. A subpopulation of corneal stromal CX3CR1GFP cells expressed low levels of MHC class II indicating either that there is a subpopulation of resident stromal macrophages that coexpress this activation molecule or that a population of DCs analogous to dermal DCs in the skin exist in the stroma, a suggestion previously proposed by Hamrah et al.10 Unfortunately, we were unable to characterize these cells fully with a pan-DC marker, such as CD11c, because of inconsistent immunohistochemical staining. The non-CX3CR1-dependency of monocyte populations in the corneal stroma was also noted in other ocular tissues (iris, ciliary body, choroid, and retina, data not shown). Similar observations in our laboratory suggest that the same phenomena may also be true of monocyte-derived cells in the connective tissues of the respiratory mucosa (Ruitenberg M, unpublished data, 2006).
In conclusion our data indicate a role for CX3CR1/fractalkine signaling in the recruitment of DCs to the corneal epithelium but not in the recruitment of monocyte-derived cells to the corneal stroma. In addition, the use of CX3CR1GFP as a marker of monocyte-derived cells has confirmed the findings emerging from other laboratories that the mouse cornea contains extensive populations of BM-derived cells in the normal steady state. This transgenic mouse model by enabling visualization of monocyte-derived cells offers a unique tool to investigate the role of these cells in vivo and ex vivo in a variety of models of ocular disease in which DC and macrophages have been implicated.40 - 42
Acknowledgments
The authors thank the Centre for Microscopy and Microanalysis for assistance.
Supported by National Health and Medical Research Council Research Program Grants 353617 and 9935975, a grant from the University of Western Australia, National Eye Institute Grant EY14362, and the Neurotrauma Research Program. GP was supported by an R.D. Wright Fellowship (303265). MR was supported by Western Australian Institute for Medical Research Fellowship 4713.
Footnotes
Disclosure: H.R. Chinnery, None; M.J. Ruitenberg, None; G.W. Plant, None; E. Pearlman, None; S. Jung, None; P.G. McMenamin, None
References
- 1.van Furth R, Cohn ZA. The origin and kinetics of mononuclear phagocytes. J Exp Med. 1968;128:415–435. doi: 10.1084/jem.128.3.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wiktor-Jedrzejczak W, Gordon S. Cytokine regulation of the macrophage (M phi) system studied using the colony stimulating factor-1-deficient op/op mouse. Physiol Rev. 1996;76:927–947. doi: 10.1152/physrev.1996.76.4.927. [DOI] [PubMed] [Google Scholar]
- 3.Dana MR. Corneal antigen-presenting cells: diversity, plasticity, and disguise: the Cogan lecture. Invest Ophthalmol Vis Sci. 2004;45:722–727. doi: 10.1167/iovs.03-0803. [DOI] [PubMed] [Google Scholar]
- 4.Gillette TE, Chandler JW, Greiner JV. Langerhans cells of the ocular surface. Ophthalmology. 1982;89:700–711. doi: 10.1016/s0161-6420(82)34737-5. [DOI] [PubMed] [Google Scholar]
- 5.Niederkorn JY, Peeler JS, Mellon J. Phagocytosis of particulate antigens by corneal epithelial cells stimulates interleukin-1 secretion and migration of Langerhans cells into the central cornea. Reg Immunol. 1989;2:83–90. [PubMed] [Google Scholar]
- 6.Williamson JS, DiMarco S, Streilein JW. Immunobiology of Langer-hans cells on the ocular surface. I. Langerhans cells within the central cornea interfere with induction of anterior chamber associated immune deviation. Invest Ophthalmol Vis Sci. 1987;28:1527–1532. [PubMed] [Google Scholar]
- 7.Streilein JW. Immunobiology and immunopathology of corneal transplantation (review). Chem Immunol. 1999;73:186–206. doi: 10.1159/000058746. [DOI] [PubMed] [Google Scholar]
- 8.Hamrah P, Huq SO, Liu Y, Zhang Q, Dana MR. Corneal immunity is mediated by heterogeneous population of antigen-presenting cells. J Leukoc Biol. 2003;74:172–178. doi: 10.1189/jlb.1102544. [DOI] [PubMed] [Google Scholar]
- 9.Hamrah P, Liu Y, Zhang Q, Dana MR. The corneal stroma is endowed with a significant number of resident dendritic cells. Invest Ophthalmol Vis Sci. 2003;44:581–589. doi: 10.1167/iovs.02-0838. [DOI] [PubMed] [Google Scholar]
- 10.Hamrah P, Zhang Q, Liu Y, Dana MR. Novel characterization of MHC class II-negative population of resident corneal Langerhans cell-type dendritic cells. Invest Ophthalmol Vis Sci. 2002;43:639–646. [PubMed] [Google Scholar]
- 11.Brissette-Storkus CS, Reynolds SM, Lepisto AJ, Hendricks RL. Identification of a novel macrophage population in the normal mouse corneal stroma. Invest Ophthalmol Vis Sci. 2002;43:2264–2271. [PMC free article] [PubMed] [Google Scholar]
- 12.Sosnova M, Bradl M, Forrester JV. CD34+ corneal stromal cells are bone marrow-derived and express hemopoietic stem cell markers. Stem Cells. 2005;23:507–515. doi: 10.1634/stemcells.2004-0291. [DOI] [PubMed] [Google Scholar]
- 13.Chen H, Hendricks RL. B7 costimulatory requirements of T cells at an inflammatory site. J Immunol. 1998;160:5045–5052. [PubMed] [Google Scholar]
- 14.Dekaris I, Zhu SN, Dana MR. TNF-alpha regulates corneal Langer-hans cell migration. J Immunol. 1999;162:4235–4239. [PubMed] [Google Scholar]
- 15.Hamrah P, Liu Y, Zhang Q, Dana MR. Alterations in corneal stromal dendritic cell phenotype and distribution in inflammation. Arch Ophthalmol. 2003;121:1132–1140. doi: 10.1001/archopht.121.8.1132. [DOI] [PubMed] [Google Scholar]
- 16.Yamagami S, Yokoo S, Usui T, et al. Distinct populations of dendritic cells in the normal human donor corneal epithelium. Invest Ophthalmol Vis Sci. 2005;46:4489–4494. doi: 10.1167/iovs.05-0054. [DOI] [PubMed] [Google Scholar]
- 17.Rot A, von Andrian UH. Chemokines in innate and adaptive host defense: basic chemokinese grammar for immune cells. Annu Rev Immunol. 2004;22:891–928. doi: 10.1146/annurev.immunol.22.012703.104543. [DOI] [PubMed] [Google Scholar]
- 18.Bazan JF, Bacon KB, Hardiman G, et al. A new class of membrane-bound chemokine with a CX3C motif. Nature. 1997;385:640–644. doi: 10.1038/385640a0. [DOI] [PubMed] [Google Scholar]
- 19.Imai T, Hieshima K, Haskell C, et al. Identification and molecular characterization of fractalkine receptor CX3CR1, which mediates both leukocyte migration and adhesion. Cell. 1997;91:521–530. doi: 10.1016/s0092-8674(00)80438-9. [DOI] [PubMed] [Google Scholar]
- 20.Pan Y, Lloyd C, Zhou H, et al. Neurotactin, a membrane-anchored chemokine upregulated in brain inflammation. Nature. 1997;387:611–617. doi: 10.1038/42491. [DOI] [PubMed] [Google Scholar]
- 21.Garton KJ, Gough PJ, Blobel CP, et al. Tumor necrosis factor-alpha-converting enzyme (ADAM17) mediates the cleavage and shedding of fractalkine (CX3CL1). J Biol Chem. 2001;276:37993–38001. doi: 10.1074/jbc.M106434200. [DOI] [PubMed] [Google Scholar]
- 22.Nishiyori A, Minami M, Ohtani Y, et al. Localization of fractalkine and CX3CR1 mRNAs in rat brain: does fractalkine play a role in signaling from neuron to microglia? FEBS Lett. 1998;429:167–172. doi: 10.1016/s0014-5793(98)00583-3. [DOI] [PubMed] [Google Scholar]
- 23.Papadopoulos EJ, Sassetti C, Saeki H, et al. Fractalkine, a CX3C chemokine, is expressed by dendritic cells and is up-regulated upon dendritic cell maturation. Eur J Immunol. 1999;29:2551–2559. doi: 10.1002/(SICI)1521-4141(199908)29:08<2551::AID-IMMU2551>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- 24.Lucas AD, Chadwick N, Warren BF, et al. The transmembrane form of the CX3CL1 chemokine fractalkine is expressed predominantly by epithelial cells in vivo. Am J Pathol. 2001;158:855–866. doi: 10.1016/S0002-9440(10)64034-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Muehlhoefer A, Saubermann LJ, Gu X, et al. Fractalkine is an epithelial and endothelial cell-derived chemoattractant for intraepithelial lymphocytes in the small intestinal mucosa. J Immunol. 2000;164:3368–3376. doi: 10.4049/jimmunol.164.6.3368. [DOI] [PubMed] [Google Scholar]
- 26.Niess JH, Brand S, Gu X, et al. CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science. 2005;307:254–258. doi: 10.1126/science.1102901. [DOI] [PubMed] [Google Scholar]
- 27.Combadiere C, Potteaux S, Gao JL, et al. Decreased atherosclerotic lesion formation in CX3CR1/apolipoprotein E double knockout mice. Circulation. 2003;107:1009–1016. doi: 10.1161/01.cir.0000057548.68243.42. [DOI] [PubMed] [Google Scholar]
- 28.Raychaudhuri SP, Jiang WY, Farber EM. Cellular localization of fractalkine at sites of inflammation: antigen-presenting cells in psoriasis express high levels of fractalkine. Br J Dermatol. 2001;144:1105–1113. doi: 10.1046/j.1365-2133.2001.04219.x. [DOI] [PubMed] [Google Scholar]
- 29.Ruth JH, Volin MV, Haines GK, 3rd, et al. Fractalkine, a novel chemokine in rheumatoid arthritis and in rat adjuvant-induced arthritis. Arthritis Rheum. 2001;44:1568–1581. doi: 10.1002/1529-0131(200107)44:7<1568::AID-ART280>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 30.Suzuki F, Nanki T, Imai T, et al. Inhibition of CX3CL1 (fractalkine) improves experimental autoimmune myositis in SJL/J mice. J Immunol. 2005;175:6987–6996. doi: 10.4049/jimmunol.175.10.6987. [DOI] [PubMed] [Google Scholar]
- 31.Jung S, Aliberti J, Graemmel P, et al. Analysis of fractalkine receptor CXCR1 function by targeted deletion and green fluorescent protein reporter gene insertion. Mol Cell Biol. 2000;20:4106–4114. doi: 10.1128/mcb.20.11.4106-4114.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Geissmann F, Jung S, Littman DR. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity. 2003;19:71–82. doi: 10.1016/s1074-7613(03)00174-2. [DOI] [PubMed] [Google Scholar]
- 33.McMenamin PG. Optimal methods for preparation and immuno-staining of iris, ciliary body, and choroidal wholemounts. Invest Ophthalmol Vis Sci. 2000;41:3043–3048. [PubMed] [Google Scholar]
- 34.Niederkorn JY. The immune privilege of corneal allografts. Transplantation. 1999;67:1503–1508. doi: 10.1097/00007890-199906270-00001. [DOI] [PubMed] [Google Scholar]
- 35.Pasare C, Medzhitov R. Toll-like receptors: linking innate and adaptive immunity. Microbes Infect. 2004;6:1382–1387. doi: 10.1016/j.micinf.2004.08.018. [DOI] [PubMed] [Google Scholar]
- 36.Liu L, Roberts AA, Ganz T. By IL-1 signaling, monocyte-derived cells dramatically enhance the epidermal antimicrobial response to lipopolysaccharide. J Immunol. 2003;170:575–580. doi: 10.4049/jimmunol.170.1.575. [DOI] [PubMed] [Google Scholar]
- 37.Crocker PR, Gordon S. Mouse macrophage hemagglutinin (sheep erythrocyte receptor) with specificity for sialylated glycoconju-gates characterized by a monoclonal antibody. J Exp Med. 1989;169:1333–1346. doi: 10.1084/jem.169.4.1333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Strobl H, Scheinecker C, Riedl E, et al. Identification of CD68+lin-peripheral blood cells with dendritic precursor characteristics. J Immunol. 1998;161:740–748. [PubMed] [Google Scholar]
- 39.Chakravarti S, Wu F, Vij N, Roberts L, Joyce S. Microarray studies reveal macrophage-like function of stromal keratocytes in the cornea. Invest Ophthalmol Vis Sci. 2004;45:3475–3484. doi: 10.1167/iovs.04-0343. [DOI] [PubMed] [Google Scholar]
- 40.Carlson EC, Drazba J, Yang X, Perez VL. Visualization and characterization of inflammatory cell recruitment and migration through the corneal stroma in endotoxin-induced keratitis. Invest Ophthalmol Vis Sci. 2006;47:241–248. doi: 10.1167/iovs.04-0741. [DOI] [PubMed] [Google Scholar]
- 41.Rosenbaum JT, Planck SR, Martin TM, et al. Imaging ocular immune responses by intravital microscopy. Int Rev Immunol. 2002;21:255–272. doi: 10.1080/08830180212065. [DOI] [PubMed] [Google Scholar]
- 42.Xu H, Forrester JV, Liversidge J, Crane IJ. Leukocyte trafficking in experimental autoimmune uveitis: breakdown of blood-retinal barrier and upregulation of cellular adhesion molecules. Invest Ophthalmol Vis Sci. 2003;44:226–234. doi: 10.1167/iovs.01-1202. [DOI] [PubMed] [Google Scholar]