Abstract
Mechanisms of drug resistance in Plasmodium vivax have been difficult to study partially because of the difficulties in culturing the parasite in vitro. This hampers monitoring drug resistance and research to develop or evaluate new drugs. There is an urgent need for a novel method to study mechanisms of P. vivax drug resistance. In this paper we report the development and application of the first Plasmodium falciparum expression system to stably express P. vivax dhfr-ts alleles. We used the piggyBac transposition system for the rapid integration of wild-type, single mutant (117N) and quadruple mutant (57L/58R/61M/117T) pvdhfr-ts alleles into the P. falciparum genome. The majority (81%) of the integrations occurred in non-coding regions of the genome; however, the levels of pvdhfr transcription driven by the P. falciparum dhfr promoter were not different between integrants of non-coding and coding regions. The integrated quadruple pvdhfr mutant allele was much less susceptible to antifolates than the wild-type and single mutant pvdhfr alleles. The resistance phenotype was stable without drug pressure. All the integrated clones were susceptible to the novel antifolate JPC-2067. Therefore, the piggyBac expression system provides a novel and important tool to investigate drug resistance mechanisms and gene functions in P. vivax.
Introduction
Plasmodium vivax is the most widely distributed of the five known human malaria causing parasites and accounts for 80–391 million cases of malaria annually [1], [2], [3]. P. vivax infections are responsible for considerable morbidity and economic loss in endemic countries and have previously been thought to be rarely lethal. However, recent reports revealed that severe complications and death caused by P. vivax are not uncommon [2], [4], [5], [6]. Appropriate and timely treatment is the key to prevent morbidity and severe complications.
P. vivax parasites are susceptible to most antimalarials, particularly to chloroquine (CQ), which is used as the first line treatment for P. vivax infections in most areas of the world today. However, over the last twenty years there have been many reports that highlight the significant increase of resistance of P. vivax to CQ [7], [8], [9], [10], [11], [12], [13], [14], [15], [16], [17], [18], [19], [20] and to the sulfa/antifolate combination sulfadoxine/pyrimethamine (SP) [7], [21], [22], [23], [24], [25]. The spread of drug resistance in P. vivax makes the control and elimination of this species much more difficult.
A better understanding of mechanisms of drug resistance in P. vivax will facilitate the development of molecular methods for monitoring the spread and extent of resistance, which will guide national treatment policy and help the development of new drugs. However, investigations into genetic mechanisms of resistance in P. vivax have been severely restricted, due largely to difficulties to culture this species of parasites in vitro. Innovative methods are urgently needed to study drug resistance mechanism in P. vivax.
The genetic basis of P. vivax resistance to antifolates has been determined to be polymorphisms within the parasite’s dihydrofolate reductase (DHFR), a key enzyme in the folate biosynthetic pathway that is targeted by antifolates [26], [27], [28], [29], [30]. A particular set of mutations (encoding F57L + S58R+ T61M + S117T) within the P. vivax dhfr gene (pvdhfr) was shown to correlate with SP treatment failures [30], [31]. The role of mutant pvdhfr alleles to antifolate resistance was initially demonstrated in various heterologous expression systems, such as E. coli [32] and yeast [33], [34] because of the difficulties in culturing P. vivax.
More recently, we used a homologous P. falciparum episomal (transient) expression system [35], [36] to directly assess the effect of wild-type and various mutant pvdhfr alleles to parasites’ drug susceptibility. The study demonstrated that this episomal expression system has the potential to provide a rapid screening system for drug development and for studying the mechanism of resistance. However, transient P. falciparum expression systems tend to have copy number variability between parasite generations and rely on a constant drug selection pressure to ensure that the episomes are not lost over time. This constant drug pressure may also lead to other unforeseen effects on the parasite.
In recent years a piggyBac mediated genome integration method has been developed enabling stable transgene expression in P. falciparum [37], [38], [39]. The piggyBac transposition system for P. falciparum involves a transiently expressed helper plasmid (transposase) that is used to activate the insertion of the transposon plasmid. This transposon contains the positive selectable marker and the gene of interest expression cassette flanked by the Inverted Terminal Repeat (ITR) sequences. Upon expression in the parasite, the piggyBac transposase identifies a TTAA target in the P. falciparum genome, cuts into this position within the genome and then inserts the expression cassette from within the transposon into the P. falciparum genome. Due to the orientation of the ITRs in the transposon only the expression cassette is integrated and the remainder of the plasmid is left outside [40], [41]. The piggyBac transposition system has been shown previously [41], [42] to rapidly and efficiently create stable genomic integrations within a few weeks as opposed to 6–12 months compared to homologous recombination.
To date, there are limited reports of the stable transfection of P. vivax genes into the P. falciparum genome [43], [44], [45]. In this article we report the development of the piggyBac transposition system for the integration of pvdhfr into the P. falciparum genome, the transcription of the pvdhfr gene, and the response of the pvdhfr alleles to conventional antifolate drugs as a proof of concept for the utility of the system. We have also compared the phenotype stability between piggyBac pvdhfr integrants and the transiently expressed pvdhfr [35]. To demonstrate the usefulness of the expression system for screening and evaluating new drugs against P. vivax we also used this expression system to test the inhibitory effect of a new generation antifolate, JPC-2067, which is an active dihydrotriazine metabolite of JPC-2056. JPC-2056 is a third generation phenoxypropoxybiguanide prodrug that was synthesized [46] to overcome the poor gastrointestinal tolerability associated with WR99210, as well as the safety and regulatory restrictions associated with the WR99210 precursor PS-15 [47], [48]. The JPC-2067 metabolite has been found to be as potent as WR99210 in vitro against P. falciparum pyrimethamine sensitive and resistance strains and has also been selected as the lead candidate for pre-clinical development based on the equivalent efficacy to PS-15 and comparable oral tolerability in mice and monkeys to proguanil [46], [49]. The findings will help the further development of this compound.
Results
Transfection and Insertion Site Analysis
Three alleles of the pvdhfr gene, wild-type, single mutant (117N) and quadruple mutant (57L, 58R, 61M and 117T) alleles were cloned into the piggyBac transposition vector and transfected into the P. falciparum line NF54. The transfected parasites were selected under blasticidin selection pressure. Blasticidin resistant populations were selected rapidly within 2–3 weeks and the total number of piggyBac insertions obtained per transfected population varied between 1 and 3. Through 6 independent transfections, blasticidin selection pressure and limiting dilution procedures, 16 unique clones of P. falciparum with piggyBac insertions in their genomes were generated. Southern blot hybridization analysis performed on these clones, revealed that a single piggyBac insertion occurred in each clone (Table 1) and that none of the clones retained the piggyBac plasmid as episomes indicating highly efficient transposition events (data not shown). Importantly, none of the transfected P. falciparum clones displayed any growth defects as judged by parasite morphology and development.
Table 1. pvdhfr alleles inserted and insertion sites in the integrated clones.
| Plasmid Name | Pvdhfr Amino Acid Mutations | Clone | Insertion Site | ||||||||
| 33 | 50 | 57 | 58 | 61 | 117 | 173 | Inter/intragenic | Genes | Chromosome | ||
| TruncatedPv bsd | B8 | Intergenic | GTP binding protein - tRNA binding protein | 14 | |||||||
| G3 | Intergenic | MAL8P1.156 - MAL8P1.154 | 8 | ||||||||
| pvdhfrwild-type allele | P | N | F | S | T | S | I | I | Intragenic | PFD0885 | 4 |
| K | Intragenic | PFL0190w | 9 | ||||||||
| D4 | Intergenic | PF10_0114 - PF10_0015 | 10 | ||||||||
| pvdhfr single mutant allele | P | N | F | S | T | N | I | E | Intergenic | PF11_0047 - PF11_0048 | 11 |
| H7 | Intergenic | PF10_0182 - PF10_0183 | 10 | ||||||||
| A4 | Intergenic | PFF0825c - PFF0830w | 6 | ||||||||
| C5 | Intergenic | PF14_0729 - etramp14.2 | 14 | ||||||||
| B7 | Intergenic | PF13_0061 - PF13_0062 | 13 | ||||||||
| D12 | Intergenic | PF11_0151 - PF11_0153 | 11 | ||||||||
| E1 | Intergenic | PF14_0071 - PF14_0072 | 14 | ||||||||
| pvdhfr quadruple mutant allele | P | N | L | R | M | T | I | C3 | Intragenic | 813289 | 9 |
| B3 | Intergenic | MAL13P1.114 - MAL13P1.115 | 13 | ||||||||
| D1 | Intergenic | pHISTa-like - plasmodium exported protein(hyp12) | 13 | ||||||||
| H8 | Intergenic | phosphomannomutase putative - a conserved plasmodium protein | 10 | ||||||||
All of the piggyBac insertions could be mapped unambiguously on the P. falciparum genome by performing BLAST searches using the NCBI http://www.ncbi.nlm.nih.gov/genome/seq/BlastGen/BlastGen.cgi?taxid=5833 database. The sites of integration in the transformed populations were identified by either Thermal asymmetric interlaced (TAIL) PCR or vectorette PCR. The vectorette PCR was more superior to TAIL PCR in determining the integration sites in terms of sensitivity and specificity (data not shown). All piggyBac insertions identified were at the expected TTAA target sequence sites and their insertion sites distributed throughout the P. falciparum genome with no bias for any particular chromosome. Of the 16 piggyBac insertions 13 (81.25%) were identified in intergenic regions while the 3 remaining piggyBac insertions occurred within coding regions. Table 1 summarizes the pvdhfr allele inserted and insertion sites of these clones.
Transcription and Expression of the pvdhfr Wild-type and Mutant Alleles in P. falciparum Erythrocyte Stage Parasites
To demonstrate the transcription and expression levels of the integrated pvdhfr gene, we selected a minimum of two clones from each pvdhfr allele that have different insertion sites to firstly determine the transcription of the endogenous pfdhfr, and secondly determine the transcription and expression of the pvdhfr alleles in these clones by conducting both real-time RT-PCR and Western blots.
1) Transcription of Endogenous pfdhfr Over the Erythrocytic Life Cycle
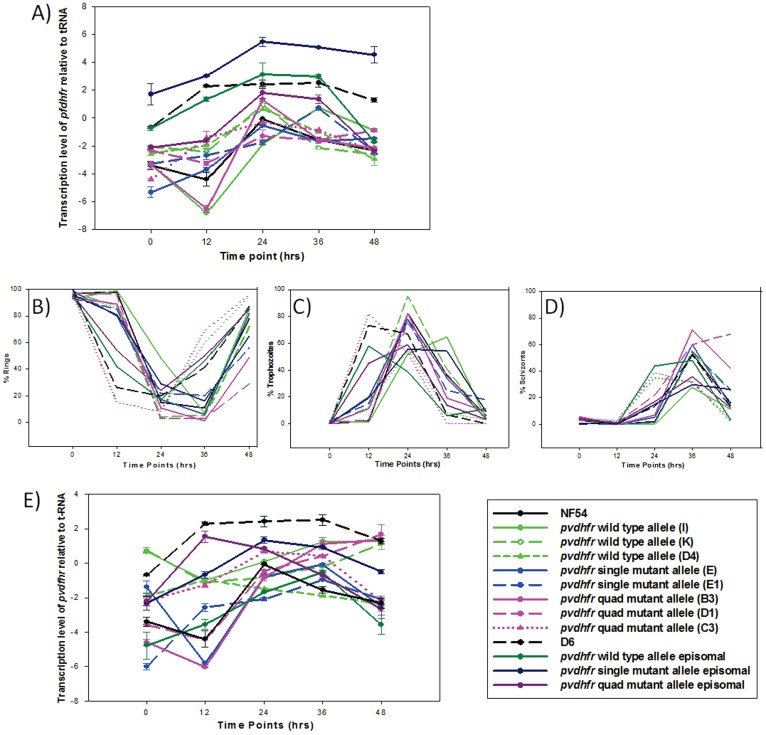
The transcription levels of the pfdfhr gene relative to that of the seryl-tRNA synthetase (seryl-tRNA) gene (PR07-0073) was determined every 12 hrs, starting with rings (time 0 hrs), through trophozoites (12–24 hrs), schizonts (24–36 hrs) and ending at rings (48 hrs) (Figure 1A). The proportions of ring, trophozoite and schizont at each time point are shown in Figure 1B, C and D. In the parental NF54 line, the transcription level of pfdhfr increased as the parasite developed from ring stage to trophozoite stage and peaked at late trophozoite stage (at 24 hrs). All integrated clones showed a similar pfdhfr transcription profiles as the parental NF54 strain except clone I (expressing a wild-type pvdhfr) and clone E1 (expressing a single mutant), which had a transcription peak at 36 hrs. The overall transcription levels of pfdhfr in all integrated clones were lower than that of the seryl-tRNA (Figure 1A). In contrast, the overall transcription levels of pfdhfr in D6 and episomally transfected D6 lines (containing pvdhfr as episomes) were generally higher than or comparable to that of seryl-tRNA (Figure 1A).
Figure 1. Transcription profile and parasite stages of dhfr mRNA expression over the parasite life cycle.
A) Transcription levels of the pfdhfr relative to that of seryl-tRNA gene. B) % rings over the parasite life cycle. C) % trophozoites over the parasite life cycle. D) % schizonts over the parasite life cycle. E) Transcription levels of pvdhfr relative to that of seryl-tRNA. Note: NF54 and D6 in panel E represent level of pfdhfr transcription as a comparison.
2) Transcription of Transfected pvdhfr Over the Erythocytic Life Cycle
To determine if the transcription and expression of pvdhfr driven by a P. falciparum dhfr promoter coincides with the expression of the endogenous pfdhfr, the pvdhfr transcription profile was compared to that of pfdhfr within one erythrocytic cycle. The pvdhfr transcription profiles of the integrated clones were less homogenous compared to the transcription of the endogenous pfdhfr peaking between 24 and 48 hrs, with the overall level of transcription lower than that of seryl-tRNA (Figure 1E). Episomally expressed pvdhfr alleles also peaked at trophozoite and schizont stages (Figure 1E). A one way ANOVA analysis showed that there was no statistically significant difference (P = 0.123) in pvdhfr transcription level between the differently transfected parasites.
3) Transcription of pvdhfr in Different Clones
To determine if the location of the transgene integration affects expression of the parasites pvdhfr, we compared clones that had integrated within the coding region to those integrants that had integrated into non-coding regions. No statistical significance (P = 0.432) of the pvdhfr transcription was found.
4) Protein Expression of Transfected pvdhfr Gene
The protein expression was investigated using both anti-cmyc and anti-bsd antibodies. Neither antibody produced a positive signal on Western blot, while a Rex-1 antibody (kindly supplied by Dr Don Gardiner, QIMR) recognized parasite proteins on the same blot (data not shown).
Susceptibility to Conventional Antifolates
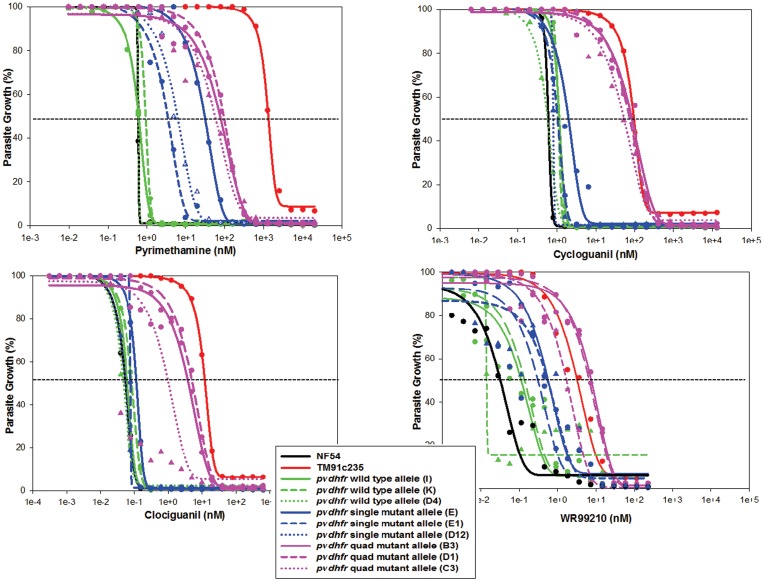
Three different clones of each pvdhfr allele transfectants were assayed using the in vitro susceptibility assay to determine firstly, if the susceptibility profile was the same between the different clones with the same pvdhfr allele and secondly, to assess the direct effect the different pvdhfr alleles had on the parasites susceptibility to pyrimethamine, cycloguanil, clociguanil and WR99210 (Table 2 and Figure 2). The three clones transfected with wild-type pvdhfr exhibited similar susceptibility to all drugs (P>0.05). The three clones expressing single mutant pvdhfr allele showed comparable susceptibility to cycloguanil, clociguanil and WR99210 with a maximum 2.4, 2.2 and 1.8 fold difference in IC50 values between the clones (P>0.05) for cycloguanil, clociguanil and WR99210, respectively. However, one single mutant clone (E) exhibited a 10 fold higher IC50 value to pyrimethamine compared to two other clones expressing the same allele. The pvdhfr quadruple mutant clones, B3 and D1 had a similar susceptibility profiles for all the antifolates (P>0.05), however, clone C3 was significantly different compared to the other two quadruple mutant clones (P<0.05).
Table 2. Susceptibility of parasites and transfected parasite lines to pyrimethamine, cycloguanil, clociguanil, WR99210 and JPC-2067.
| Parasite/Transfected Clone | Pyrimethamine | Cycloguanil | Clociguanil | WR99210 | JPC-2067 | |||||
| IC50 (± SD) | RR | IC50 (± SD) | RR | IC50 (± SD) | RR | IC50 (± SD) | RR | IC50 (± SD) | RR | |
| Native Parasite Strain | ||||||||||
| NF54 | 0.61 (±0.00) | 1 | 0.61 (±0.09) | 1 | 0.05 (±0.02) | 1 | 0.03 (±0.00) | 1 | 0.05 (±0.00) | 1 |
| TM91c235 | 1276.60 (±2.32) | 2103 | 85.58 (±15.20) | 74 | 11.19 (±2.10) | 211 | 5.70 (±0.26) | 190 | 0.06 (±0.26) | 1 |
| Clones of NF54 integrated with different pvdhfr alleles | ||||||||||
| Wild-type (I) | 0.56 (±0.18) | 1 | 1.16 (±0.29) | 2 | 0.06 (±0.02) | 1 | 0.10 (±0.01) | 4 | 0.14 (±0.01) | 3 |
| Wild-type (K) | 0.94 (±0.01) | 2 | 1.10 (±0.12) | 2 | 0.08 (±0.06) | 2 | 0.16 (±0.00) | 5 | 0.12 (±0.00) | 3 |
| Wild-type (D4) | 0.61 (±0.01) | 1 | 0.57 (±0.12) | 1 | 0.05 (±0.02) | 1 | 0.01 (±0.02) | 0 | 0.09 (±0.02) | 2 |
| Single mutant (E) | 30.20 (±7.32) | 50 | 1.92 (±0.37) | 3 | 0.11 (±0.04) | 2 | 0.53 (±0.09) | 18 | 0.12 (±0.09) | 3 |
| Single mutant (E1) | 3.31 (±1.51) | 5 | 1.03 (±0.11) | 2 | 0.08 (±0.01) | 1 | 0.32 (±0.25) | 11 | 0.07 (±0.25) | 2 |
| Single mutant (D12) | 5.85 (±0.09) | 10 | 0.81 (±0.02) | 1 | 0.05 (±0.02) | 1 | 0.57 (±0.27) | 19 | 0.11 (±0.27) | 2 |
| Quadruple mutant (B3) | 75.06 (±37.00) | 134 | 87.51 (±24.67) | 144 | 4.04 (±1.92) | 76 | 7.23 (±0.09) | 241 | 0.03 (±0.02) | 1 |
| Quadruple mutant (D10) | 93.02 (±30.05) | 166 | 73.21 (±20.23) | 121 | 5.14 (±1.86) | 97 | 6.68 (±0.38) | 223 | 0.06 (±0.04) | 1 |
| Quadruple mutant (C3) | 57.11 (±16.97) | 94 | 119.00 (±3.90) | 196 | 1.02 (±0.10) | 19 | 1.57 (±0.01) | 52 | 0.05 (±0.01) | 1 |
| Clones of D6 episomally expressing different pvdhfr alleles | ||||||||||
| Wild-type | 0.17 (±0.01) | 4 | ||||||||
| Single mutant | 0.19 (±0.02) | 4 | ||||||||
| Quadruple mutant | 1.03 (±0.30) | 22 | ||||||||
IC50s are means of triplicate data points and are expressed as mean nM ± SD. The relative resistance index (RR) was determined in comparison to NF54.
Figure 2. In vitro susceptibility of P. falciparum NF54 and integrated parasite clones stably expressing various pvdhfr mutants to DHFR inhibitors: pyrimethamine (top left panel), cycloguanil (top right panel), clociguanil (bottom left panel), and WR99210 (bottom right panel).
Each color represents a pvdhfr allele and lines of the same color represent different clones of the same expressed pvdhfr allele. The symbols represent the means of triplicate data points.
The clones of NF54 transfected with the wild-type pvdhfr-ts (WT) had a similar susceptibility profile to that of the parental NF54 strain. This demonstrated that the WT pvdhfr allele was equally susceptible to antifolates compared to the wild-type pfdhfr-ts (P = 0.477). In contrast, the NF54 transfected with the mutant pvdhfr alleles were less susceptible to antifolates compared to the WT. NF54 clones transfected with the single mutant pvdhfr 117N were 5 to 50 fold less susceptible to pyrimethamine (P = <0.001) and 11 to 19 fold less susceptible to WR99210 (P = 0.013) compared to the clones with wild-type pvdhfr allele. However, their susceptibilities to cycloguanil and clociguanil were not different to the clones with the wild-type pvdhfr allele (P = 0.469 and P = 0.440).
The pvdhfr quadruple mutants (57L/58R/61M/117T) that were integrated into NF54 parasites were relatively more resistant to all the antifolates tested than the WT and single mutant pvdhfr 117N allele transfected clones. The quadruple mutant pvdhfr allele transfectants were 94–166, 121–196, 19–97 and 52–241 fold more resistant to pyrimethamine, cycloguanil, clociguanil and WR99210 respectively, compared to the NF54 parental line (P = <0.005), but were 13–22 and 2–11 fold less resistant to pyrimethamine and clociguanil, respectively, than the native P. falciparum quadruple mutant, TM91c235 (P = <0.001 and P = 0.003, respectively) (Table 2). Each clone was assayed three times on three different occasions and the results were comparable in terms of the rank order and differences between alleles (data not shown).
To confirm that the above described changes in susceptibility to antifolate drugs were not due to mutations or amplification that had occurred in the endogenous pfdhfr in response to the selection pressure with blasticidin, we sequenced the pfdhfr and measured the copy number of pfdhfr for all the transfected clones. No mutations within the open reading frame of pfdhfr and no copy number changes were identified in the integrated parasites (data not shown). Furthermore, to determine if any plasmid components other than pvdhfr had any effect on the susceptibility to antifolate drugs, we compared the NF54 transfected with a truncated bsd pvdhfr to NF54 transfected with the P. vivax wild-type dhfr-ts (WT) and found no difference (data not shown).
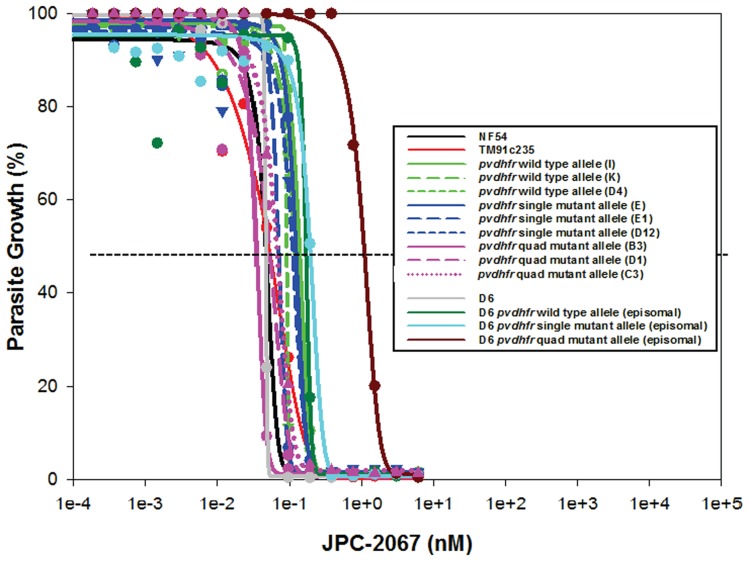
Susceptibility to a Novel Antifolate, JPC-2067
The purpose of being able to express P. vivax drug resistance genes in P. falciparum is so that we can better understand the effect that the various mutant alleles will have against new compounds. Table 2 and Figure 3 show the effect of JPC-2067 against the wild-type and various mutant pvdhfr integrated alleles. The P. falciparum lines of NF54 (carrying wild-type pfdhfr) and TM91c235 (carrying quadruple mutant pfdhfr) were equally susceptible to JPC-2067(P = >0.05). All of the integrated clones with different pvdhfr alleles had a similar susceptibility profile as NF54 and TM91c235, with a maximum difference of 4 fold in IC50 values between them (P>0.05).
Figure 3. In vitro susceptibility profiles of P. falciparum NF54, NF54 stably transfected parasites, D6, D6 episomally transfected parasites and TM91c235 to JPC-2067.
The symbols represent the means of triplicate data points.
Stability of Copy Number (CN) and the Susceptibility to Conventional Antifolates
To determine if the removal of drug selection pressure on the transfected parasites resulted in a change in CN and/or the susceptibility of transfected parasites to antifolates, six integrated clones (two wild-type, two single and two quadruple mutant clones) were monitored for 32–53 erythrocytic cycles (64–106 days) after selection pressure was withdrawn. Sample were taken at 4–5 time points and used to assess CN and susceptibility to antifolates. D6 parasites expressing various pvdhfr alleles as episomes [35], [36] were also monitored in parallel for CN and susceptibility with and without drug pressure.
For the six integrated NF54 clones, the CN of pvdhfr remained at approximately one throughout the 32 cycles with no significant changes in parasite susceptibility to pyrimethamine. For the episomal transfected lines, CN did not reduce with time when cultured under drug pressure. However, CN reduced over time in single and quadruple mutant transfectants when cultured without drug pressure. It was observed that approximately one episome was lost every 9 cycles for the single mutant and every 4 erythrocytic cycles for quadruple mutant transfected parasites on average (Table 3). As the CN of the quadruple mutant pvdhfr episomes decreased, the susceptibility of the parasite to pyrimethamine increased significantly (Spearman test, P = 0.0167). The same trend was seen in the single mutant although Spearman test was just above significance (P = 0.0833). No significant relationship was observed between the CN of the episomes and the susceptibility to pyrimethamine for the wild-type (P = 0.7707). This trend was also observed for cycloguanil, clociguanil and WR99210 (data not shown). It was also observed that there was a significant difference between the IC50 values of the integrated quadruple pvdhfr allele and the episomally expressed quadruple pvdhfr allele (P = 0.001).
Table 3. Copy number of pvdhfr (CN) and IC50 to pyrimethamine of transfected parasites at five time points over 53 parasite life cycles.
| Parasite strain/transfected clones | Number of days since the start of experiment | |||||||||||
| 0 | 24 | 46 | 64 | 106 | ||||||||
| CN | IC50 (± SD) | CN | IC50 (± SD) | CN | IC50 (± SD) | CN | IC50 (± SD) | CN | IC50 (± SD) | |||
| Native parasite strains | ||||||||||||
| D6 | 0 | 1.41 (±0.42) | 0 | 0.95 (±0.32) | 0 | 1.19 (±0.17) | 0 | 1.19 (±0.52) | 0 | 0.81 (±ND) | ||
| NF54 | 0 | 0.78 (±0.05) | 0 | 0.94 (±0.03) | 0 | 0.74 (±0.15) | 0 | 0.64 (±0.16) | 0 | ND | ||
| TM91c235 | 0 | 2067.00 (±162.00) | 0 | 2526.00 (±420.00) | 0 | 2760.00 (±72.00) | 0 | 1126.00 (±27.30) | 0 | 2252.00 (± ND) | ||
| D6 parasites episomally expressing various pvdhfr alleles cultured without and with drug selection | ||||||||||||
| Wild-type | 1.65 | 0.78 (±0.07) | 2.03 | 0.54 (±0.26) | 2.59 | 0.59 (±0.11) | 2.61 | 0.59 (±0.16) | 2.39 | 0.56 (±0.04) | ||
| Wild-type + blasticidin pressure | 1.65 | 0.78 (±0.07) | 2.17 | 0.69 (±0.13) | 1.98 | 0.73 (±0.12) | 1.92 | 0.72 (±0.17) | 2.16 | 0.35 (±0.01) | ||
| Single mutant | 11.94 | 1.57 (±0.50) | 8.10 | 0.20 (±0.06) | 8.90 | 0.87 (±0.07) | 7.00 | 0.32 (±0.10) | 5.70 | 0.13 (±0.02) | ||
| Single mutant + blasticidin pressure | 11.94 | 1.57 (±0.50) | 13.83 | 0.45 (±0.09) | 16.95 | 1.08 (±0.08) | 15.75 | 0.99 (±0.13) | 19.00 | 0.41 (±0.12) | ||
| Quad mutant | 15.99 | 1698.00 (±547.00) | 6.94 | 725.00 (±128.00) | 4.71 | 636 (±81.00) | 4.52 | 507.00 (±63.00) | 1.24 | 94.71 (±4.40) | ||
| Quad mutant +pyr pressure | 15.99 | 1698.00 (±547.00) | 11.09 | 1179.00 (±74.00) | 10.42 | 1393.00 (±224.00) | 11.35 | 1156 (±186.00) | 33.00 | 1011.18 (±110.00) | ||
| NF54 parasite clones integrated with different pvdhfr alleles cultured without drug selection | ||||||||||||
| Wild-type (I) | 0.99 | 0.93 (±0.39) | 1.18 | 0.54 (±0.47) | 0.74 | 2.82 (±0.93) | 1.20 | 0.46± (0.93) | ND | ND | ||
| Wild-type (D4) | 0.63 | 0.71 (±0.34) | 1.19 | 0.56 (±0.24) | 1.19 | 1.50 (±0.34) | 1.19 | 0.53 (±0.07) | ND | ND | ||
| Single mutant (E) | 0.50 | 79.40 (±8.66) | 0.80 | 43.2 (±7.32) | 0.72 | 65.58 (±3.84) | ND | 42.3 (±6.35) | ND | ND | ||
| Single mutant (E1) | 0.60 | 8.31 (±1.51) | ND | 7.89 (±0.83) | 0.54 | 4.10 (±0.16) | ND | 1.41 (±0.30) | ND | ND | ||
| Quad mutant (B3) | 1.15 | 68.42 (±6.78) | 0.70 | 137.08 (±0.93) | 0.92 | 84.93 (±6.78) | 0.83 | 201 (±51.99) | ND | ND | ||
| Quad mutant (C3) | 0.84 | 69.38 (±2.62) | 0.86 | 53.64 (±2.62) | 1.20 | 94.6 (±59.86) | 0.82 | 183.00 (±22.13) | ND | ND | ||
Discussion
This study demonstrates the integration and stable expression of pvdhfr alleles in a heterologous system of P. falciparum. It provides evidence that the transposon-mediated genomic integration method, piggyBac, is a useful method to create stable transformed clones for studying P. vivax drug resistance mechanisms and gene functions in P. falciparum. This new system will enable studies into the biology of P. vivax parasites which had been significantly lacking due, partially to, our inability to culture this pathogenic parasite species.
The piggyBac transfections inserted a single copy of pvdhfr into one of the P. falciparum chromosomes. The insertion sites appear to be random with the majority of sites identified in non-coding regions of different chromosomes as previously described [41], [42]. The insertion of pvdhfr did not affect the timing and level of endogenous pfdhfr as the transcription timing and levels of endogenous pfdhfr in all clones were comparable to those of the parental untransfected parasites. The location of the insertions, which were identified in this study, also did not appear to affect the transcription level of transfected pvdhfr because the transcription levels were not significantly different between clones that were inserted in different locations on different chromosomes. Interestingly, the peak transcription time of pvdhfr (∼36 hrs in the experiment) appears to lag behind that of the endogenous pfdhfr (∼24 hrs in the experiment). Since the promoter used to transcribe pvdhfr is the pfdhfr promoter, it is not clear why this shift of timing has occurred. Differences in developmental stages of parasites should not have resulted in this shift as the transcription of pfdhfr and pvdhfr were measured in the same sample. One possibility may be due to the epigenetic control at the pfdhfr locus.
The protein expression was also investigated using both anti-cmyc and anti-bsd antibodies. Unfortunately, neither antibody produced a positive signal on Western blot. There are several possible explanations for negative Western blot. Firstly, the amount of protein expressed was too small for these antibodies to bind and generate a positive signal. Earlier reports [50], [51], [52] also described failure to obtain positive signals on Western blot and demonstrated that this was due to DHFR-TS being a low abundance protein. Secondly, the expressed PvDHFR maybe conformationally folded such that the cmyc and bsd were not accessible to the antibodies. Nevertheless, the transfected parasites had a susceptibility profile corresponding to the transfected pvdhfr allele, indicating the successful expression of functional PvDHFR in the transfected P. falciparum parasites.
As was seen previously in the episomal expression of the pvdhfr alleles [35], [36], the same in vitro susceptibility phenotypic profile was observed for the integrated wild-type and mutant pvdhfr alleles. That is, the wild-type pvdhfr transfected clones were as susceptible to antifolates as the susceptible parent strain while the mutant pvdhfr alleles conferred significantly reduced susceptibility to the antifolate drugs; with the quadruple mutant pvdhfr allele conferring a higher resistance than the single mutant pvdhfr allele tested. This increase in resistance to the antifolates was not due to mutations in the endogenous pfdhfr, nor was it due to amplification of the endogenous pfdhfr gene or changes in its expression as all clones had a comparable timing and level of pfdhfr transcription.
Three different clones of the same pvdhfr allele (wild-type, single and quadruple mutant) were used to determine the susceptibility phenotype of the transfected parasites to rule out the possible effect of other unintended mutations or unintended positional effects caused by the integration of the transfected allele into the parasite genome [53]. Indeed, while consistent susceptibility phenotype profiles were observed between the three wild-type clones (I, K and D4), between two single mutant clones (D12 and E1) and between two quadruple mutant clones (B3 and D10), significantly different phenotypes were observed in one of the three single mutant clones (E) and one of the three quadruple mutant clones (C3). This highlights the importance of using multiple clones with different insertion sites to determine the phenotype profile of transfected parasites.
Interestingly, the level of resistance transferred by two systems was not identical, particularly in susceptibilities to antifolates between episomal and integrated quadruple mutant pvdhfr allele (57L/58R/61M/117T) transfected parasites. The integrated parasite clones showed a lower level of resistance to all antifolate drugs compared to episomal transfected parasites. The IC50 values to pyrimethamine was 75.06±37.00 nM, 57.11±16.97 nM and 93.02±30.05 nM for integrated clones and 1698±547 nM for the episomal transfected parasite line. The quadruple mutant pvdhfr integrated clones also showed a lower level of resistance to the new antifolate compound JPC-2067 (resistance index 0.72–1.26) than the episomal transfected parasites (resistance index 22) [49]. The difference in susceptibility to these drugs is likely resulted from difference in pvdhfr copy number between the episomal and integrated quadruple mutant parasites. When the copy number of pvdhfr episomes reduced to 1.24 after culturing 106 days without drug selection, the IC50 value of the episomal quadruple mutant parasite decreased to 94.71±4.4 nM, comparable to that of integrated quadruple mutant parasite.
The relationship between copy number and susceptibility to antifolate drugs has been studied in the past without a clear conclusion. Results of our previous study [35], suggested that the susceptibility of the antifolates was not affected by the copy number of the episomes carrying the wild-type and mutant pvdhfr alleles. In the current study, we conducted a test over 106 days on the same parasite line and identified a good correlation over time between episome copy number and IC50 values to pyrimethamine for both the single and quadruple mutant pvdhfr transfected parasites, while no such a correlation was identified for the wild-type pvdhfr transfected parasites. The results demonstrate that variation in copy number within the same transfectant parasite line will cause changes in parasite susceptibility to antifolates.
The development of a homologous system to stably express P. vivax genes removes the variability and uncertainty in parasite susceptibility caused by episomal copy number and allows us to determine more accurately the susceptibility of antifolates against parasites with mutant pvdhfr alleles. This system has a better potential to provide a rapid screening system for drug development and for studying drug resistance mechanisms in P. vivax than the episomal expression system. To demonstrate this potential we tested a novel antifolate compound JPC-2067 against parasite clones transfected with various pvdhfr alleles. This compound when given to monkeys as a pro-drug (JPC-2056) has been shown to be highly potent against P. falciparum parasite carrying wild-type and mutant pfdhfr alleles [49]. However, its effect against P. vivax carrying different pvdhfr was unknown causing concerns on the further development of this compound. Using the stably transfected parasites we demonstrated that the compound was highly effective in killing parasites integrated with pvdhfr quadruple mutant allele with the same potency as against parasites integrated with wild-type pvdhfr. These results provide evidence and support for the future development of JPC-2056 for the treatment of both P. falciparum and P. vivax infections. In addition to assisting drug development, this stable P. falciparum expression system has the potential to identify and confirm other P. vivax drug targets, as well as elucidate the biological function of other P. vivax genes.
Materials and Methods
Parasites
P. falciparum lines NF54 and TM91c235 were used in this study as antifolate susceptible and resistant controls, respectively. Parasite lines were cultured continuously in a 4% hematocrit blood-LPLF RPMI 1640 as previously reported [54]. Parasite cultures were synchronized at the ring stage by repeated D-Sorbitol treatment [55] and were allowed to grow for one cycle after the last treatment and then used for transfection or in vitro susceptibility testing. P. falciparum D6 and D6 episomally expressing various alleles of pvdhfr [35] were used in copy number, transcription and susceptibility tests. For CN and transcription analyses, aliquots of parasite cultures were taken every 12 hrs, starting from early ring stage, over a period of 48 hrs.
Plasmid Construction
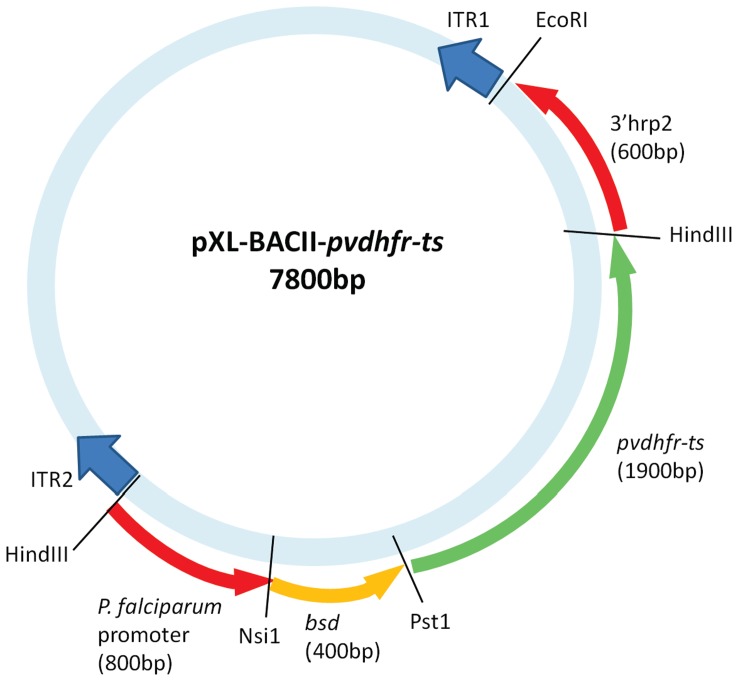
The open reading frames (ORFs) of wild-type, single mutant (117N) and quadruple (57L/58R/61M/117T) mutant alleles of pvdhfr-ts were amplified from genomic DNA of P. vivax isolates [56] by PCR using primers and conditions described previously [30], [56]. The plasmid construction was performed as previously described [35], [36] with minor modifications. The pvdhfr PCR product was inserted between the P. falciparum dhfr-ts promoter and P. vivax thymidylate synthase (pvts); the pvdhfr cassette had the 0.4-kb Saccharomyces cereviseae bsd ORF inserted in frame between the promoter and dhfr-ts ORF as described previously [36]. The pvdhfr cassette was then cloned into the pXL-BacII vector [42] at the HindIII sites between ITR2 and 3′histidine-rich protein-2 (Figure 4).
Figure 4. pXL-BACII-pvdhfr plasmid design for piggyBac transformation of P. falciparum.
The pXL-BacII-DHFR vector [42] was cut with HindIII to remove the 5′ calmodulin and hdhfr fragments from the pXL-BacII vector. The pfdhfr promoter-bsd-cmyc-pvdhfr-ts cassette was excised from the pRSET-C vector as a 3.1 kb HindIII fragment and cloned into pXL-BacII, such that it is flanked by the piggyBac ITR2 and 3′ histidine-rich protein-2 (hrp2). The sizes of the boxes are not proportional to the lengths of the genes. The pfdhfr promoter drives the expression of bsd-cmyc-pvdhfr-ts fused proteins.
The pHTH and pDCTH transposases were constructed as previously described in [41], [42], respectively. The plasmids for the parasites with transient expression of the pvdhfr were developed and transfected as described previously [35].
Transfections
Transfection of P. falciparum NF54 was achieved by parasite invasion of plasmid DNA-loaded RBCs as described earlier [41], [42] with minor modifications. Briefly, mature blood-stage parasites were purified on a MACS magnetic column (Miltentyi Biotec) and 1 million purified parasites were added to erythrocytes loaded with either 300 µg of the transposon plasmid containing either wild-type or various mutant pvdhfr and 150 µg of the transposase plasmid pHTH [42] or 300 µg of the transposon plasmid and 300 µg of the transposase plasmid pDCTH [41] to start a 5 ml parasite culture. Individual clones were obtained by limiting dilution of parasites post-drug selection and parasite clones were detected as previously described [57].
DNA and RNA Extractions
Genomic DNA was extracted from parasites using QIAamp DNA Blood Mini Kits (QIAGEN, USA) and used for: Southern blot hybridisation to confirm the piggyBac integration in the P. falciparum genome [42]; inverse PCR [42], [58] or vectorette PCR reactions to identify the piggyBac insertion sites; quantitative real-time PCR to determine copy number of the pvdhfr gene [35].
Total RNA was extracted from parasites using Nucleospin RNA II kits (Macherey-Nagel, Germany) with an additional 1 u/µl DNase I (New England Biolabs, USA) treatment at 37°C for ∼3 hrs, followed by a second round of RNA purification using the same kit. Total RNA was used in quantitative real-time reverse transcription PCR (RT-PCR) to determine the transcription of both pfdhfr and pvdhfr genes [59].
Determining CN of pfdhfr and pvdhfr
The CN of the pfdhfr and pvdhfr was determined by a quantitative real-time PCR in an Mx4000 multiplex quantitative PCR system (Stratagene) using a SYBR green-based assay and pfdhfr and pvdhfr primers. The single copy gene eba175 gene was used as a reference (normaliser) gene for estimating the CN for pfdhfr and pvdhfr, respectively (the target). Primers used to amplify fragments of pvdhfr and the eba175 genes were published previously [35]. The CNs of the plasmids were determined based on the threshold cycle (CT) values of pvdhfr using the ΔΔCT method [59]. The difference in the CN between the different time points was determined and tested (P value) by using a nonparametric comparison (Spearman-rank test).
Identification of piggyBac Insertion Sites
piggyBac insertion sites in transfected parasites were identified either by inverse PCR [42], [58] or vectorette PCR reactions [60](see Supporting Information S1). The insertion sites were identified as described previously [41].
Sequencing of pfdhfr
To exclude the possibility that altered drug phenotypes could be due to the mutations or an increase in the copy number of the endogenous pfdhfr gene, the pfdhfr gene was amplified and sequenced using the primers and PCR conditions described previously [61].
Analysis of pvdhfr and pfdhfr Transcription
To determine the transcription levels of endogenous pfdhfr and transfected pvdhfr, quantitative real-time reverse transcription PCR (RT-PCR) assays were performed as described previously [59], using primers specific for pvdhfr as described previously [35] and for pfdhfr (PfDHFR Fwd 5′ACCTGCGCAGTTCATACACG3’ and PfDHFR Rev 5′TCTTGGGAATGGATAGGGTATTCTGT3’). The statistical significance of the mRNA expression between the transfected parasite lines and their parental strain was determined by either the paired t-test or one way analysis of variance (ANOVA) provided by Sigma Plot for Windows, version 11 (Systat Software, Inc., San Jose, CA).
In vitro Susceptibility, Bioassay and Time Course Studies
WR99210 and JPC-2067 were kindly supplied by Jacobus Pharmaceutical Company (Princeton, N.J.), Cycloguanil and clociguanil, was purchased from ICI Pharmaceuticals (Macclesfield, United Kingdom) and pyrimethamine, was purchased from Hoffmann-La Roche (Switzerland). All compounds were initially dissolved in dimethyl-sulfoxide (DMSO) and diluted in plasma free LPLF 1640 RPMI. The susceptibility assays were performed as described previously [35], [62].
The bioassays were performed as described previously [49] with minor modifications. Briefly, JPC-2067 working drug solutions (50 µl) were serially diluted by 2-fold on 96 well microtitre plates using complete LPLF 1640 RPMI followed by the addition of 50 µl of infected red blood cells suspended in culture medium. The final cell suspension (100 µl) had a haematocrit of 2%, of which 0.5% were infected erythrocytes (>95% rings).
A solution of 10 µl of 1∶100 dilution of 14.0 Ci/mmol [3H] hypoxanthine (Perkin-Elmer): PRPMI (final concentration of 1 µCi/well) was added after 48 hrs when parasites had already reinvaded the red blood cells and were at ring stages, and the parasites were harvested after 96 hrs. Incorporation of 3H by intra-erythrocytic malaria parasites (measured in cpm) was recorded for each well, and the concentrations of drugs that inhibited 50% of parasite growth (IC50s) when compared with drug-free serum samples (controls) were determined [49]. That is, reduction = 100 x [(mean counts per minute no drug-control samples – mean counts per minute test samples)/mean counts per minute no-drug control samples]. The percentage of growth inhibition was plotted for each drug concentration. The IC50 was determined using nonlinear regression analysis provided by Sigma Plot for Windows, version 11. All assays were performed in duplicate on at least three different occasions. The time point susceptibility assays were conducted as described previously [35]. However, the assays were only performed once in triplicate at each time point.
Statistical tests were performed to compare resistance profiles (IC50) between the transfected parasite lines and between them and their parental strain as well as to TM91c235 using either the paired t-test or one way analysis of variance (ANOVA) provided by Sigma Plot for Windows, version 11 (Systat Software, Inc., San Jose, CA).
Supporting Information
Materials and Methods for inverse PCR or vectorette PCR reactions to identify the piggyBac insertion sites.
(DOC)
Acknowledgments
The authors are most thankful to the Australian Red Cross Blood Service (Brisbane) for providing human erythrocytes and plasma for the in vitro cultivation of parasites; Dr David Jacobus for providing the WR99210 and JPC-2067 and; Dr Don Gardiner for providing the Rex1 antibody.
The opinions expressed herein are those of the authors and do not necessarily reflect those of the Australian Defence Force (ADF), any ADF extant policy, or the U.S. Army.
Footnotes
Competing Interests: While Jacobus Pharmaceutical Company (Princeton, New Jersey) kindly supplied the antimalarial compounds WR99210 and JPC-2067 and Dr. Don Gardiner, QIMR (Brisbane, Australia) kindly supplied Rex 1 antibody, the authors do not have commercial or other association that might pose a conflict. This does not alter the authors’ adherance to all the PLoS ONE policies on sharing data and materials.
Funding: The work was supported in part by National Institutes of Health grants R21AI070888 and R01AI033656 (JHA). AMA received travel awards from the Australian American Fulbright Scholarship (2009), Chief of Army Scholarship (2009) and ARC/NHMRC Parasitology Network travel scholarship (2006). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. No additonal external funding received for this study.
References
- 1.Mendis K, Sina BJ, Marchesini P, Carter R. The neglected burden of Plasmodium vivax malaria. Am J Trop Med Hyg. 2001;64:97–106. doi: 10.4269/ajtmh.2001.64.97. [DOI] [PubMed] [Google Scholar]
- 2.Price RN, Tjitra E, Guerra CA, Yeung S, White NJ, et al. Vivax malaria: neglected and not benign. Am J Trop Med Hyg. 2007;77:79–87. [PMC free article] [PubMed] [Google Scholar]
- 3.Hay SI, Guerra CA, Tatem AJ, Noor AM, Snow RW. The global distribution and population at risk of malaria: past, present, and future. Lancet Infect Dis. 2004;4:327–336. doi: 10.1016/S1473-3099(04)01043-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Poespoprodjo JR, Fobia W, Kenangalem E, Lampah DA, Hasanuddin A, et al. Vivax malaria: a major cause of morbidity in early infancy. Clin Infect Dis. 2009;48:1704–1712. doi: 10.1086/599041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mueller I, Galinski MR, Baird JK, Carlton JM, Kochar DK, et al. Key gaps in the knowledge of Plasmodium vivax, a neglected human malaria parasite. The Lancet Infectious Diseases. 2009;9:555–566. doi: 10.1016/S1473-3099(09)70177-X. [DOI] [PubMed] [Google Scholar]
- 6.Tjitra E, Anstey NM, Sugiarto P, Warikar N, Kenangalem E, et al. Multidrug-resistant Plasmodium vivax associated with severe and fatal malaria: a prospective study in Papua, Indonesia. PLoS Med. 2008;5:e128. doi: 10.1371/journal.pmed.0050128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ratcliff A, Siswantoro H, Kenangalem E, Wuwung M, Brockman A, et al. Therapeutic response of multidrug-resistant Plasmodium falciparum and P. vivax to chloroquine and sulfadoxine-pyrimethamine in southern Papua, Indonesia. Trans R Soc Trop Med Hyg. 2007;101:351–359. doi: 10.1016/j.trstmh.2006.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Coatney GR. Pitfalls in a discovery: the chronicle of chloroquine. Am J Trop Med Hyg. 1963;12:121–128. doi: 10.4269/ajtmh.1963.12.121. [DOI] [PubMed] [Google Scholar]
- 9.Peters W. Drug resistance in malaria parasites of animals and man. Adv Parasitol. 1998;41:1–61. doi: 10.1016/s0065-308x(08)60421-2. [DOI] [PubMed] [Google Scholar]
- 10.Pukrittayakamee S, Chantra A, Simpson JA, Vanijanonta S, Clemens R, et al. Therapeutic responses to different antimalarial drugs in vivax malaria. Antimicrob Agents Chemother. 2000;44:1680–1685. doi: 10.1128/aac.44.6.1680-1685.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wellems TE, Plowe CV. Chloroquine-resistant malaria. J Infect Dis. 2001;184:770–776. doi: 10.1086/322858. [DOI] [PubMed] [Google Scholar]
- 12.Rieckmann K, Davis DR, C HD. Plasmodium vivax resistance to chloroquine? Lancet. 1989;ii:1183–1884. doi: 10.1016/s0140-6736(89)91792-3. [DOI] [PubMed] [Google Scholar]
- 13.Fryauff DJ, Tuti S, Mardi A, Masbar S, Patipelohi R, et al. Chloroquine-resistant Plasmodium vivax in transmigration settlements of West Kalimantan, Indonesia. Am J Trop Med Hyg. 1998;59:513–518. doi: 10.4269/ajtmh.1998.59.513. [DOI] [PubMed] [Google Scholar]
- 14.Dua VK, Kar PK, Sharma VP. Chloroquine resistant Plasmodium vivax malaria in India. Trop Med Int Health. 1996;1:816–819. doi: 10.1111/j.1365-3156.1996.tb00116.x. [DOI] [PubMed] [Google Scholar]
- 15.Marlar T, Myat PK, Aye YS, Khaing KG, Ma S, et al. Development of resistance to chloroquine by Plasmodium vivax in Myanmar. Trans R Soc Trop Med Hyg. 1995;89:307–308. doi: 10.1016/0035-9203(95)90556-1. [DOI] [PubMed] [Google Scholar]
- 16.Soto J, Toledo J, Gutierrez P, Luzz M, Llinas N, et al. Plasmodium vivax clinically resistant to chloroquine in Colombia. Am J Trop Med Hyg. 2001;65:90–93. doi: 10.4269/ajtmh.2001.65.90. [DOI] [PubMed] [Google Scholar]
- 17.Phillips EJ, Keystone JS, Kain KC. Failure of combined chloroquine and high-dose primaquine therapy for Plasmodium vivax malaria acquired in Guyana. South America. Clin Infect Dis. 1996;23:1171–1173. doi: 10.1093/clinids/23.5.1171. [DOI] [PubMed] [Google Scholar]
- 18.Alecrim MG, Alecrim W, Macedo V. Plasmodium vivax resistance to chloroquine (R2) and mefloquine (R3) in Brazilian Amazon region. Rev Soc BrasMed Trop. 1999;32:67–68. doi: 10.1590/s0037-86821999000100013. [DOI] [PubMed] [Google Scholar]
- 19.Kurcer MA, Simsek Z, Zeyrek FY, Atay S, Celik H, et al. Efficacy of chloroquine in the treatment of Plasmodium vivax malaria in Turkey. Ann Trop Med Parasitol. 2004;98:447–451. doi: 10.1179/000349804225021343. [DOI] [PubMed] [Google Scholar]
- 20.Phan GT, de Vries PJ, Tran BQ, Le HQ, Nguyen NV, et al. Artemisinin or chloroquine for blood stage Plasmodium vivax malaria in Vietnam. Trop Med Int Health. 2002;7:858–864. doi: 10.1046/j.1365-3156.2002.00948.x. [DOI] [PubMed] [Google Scholar]
- 21.Darlow B, Vrbova H, Gibney S, Jolley D, Stace J, et al. Sulfadoxine-pyrimethamine for the treatment of acute malaria in children of Papua New Guinea. II. Plasmodium vivax. . Am J Trop Med and Hyg. 1982;31:10–13. doi: 10.4269/ajtmh.1982.31.10. [DOI] [PubMed] [Google Scholar]
- 22.Alam MT, Bora H, Bharti PK, Saifi MA, Das MK, et al. Similar trends of pyrimethamine resistance-associated mutations in Plasmodium vivax and P. falciparum. Antimicrob Agents Chemother. 2007;51:857–863. doi: 10.1128/AAC.01200-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Doberstyn EB, Teerakiartkamjorn C, Andre RG, Phintuyothin P, Noeypatimanondh S. Treatment of vivax malaria with sulfadoxine-pyrimethamine and with pyrimethamine alone. Trans R Soc Trop Med Hyg. 1979;73:15–17. doi: 10.1016/0035-9203(79)90121-4. [DOI] [PubMed] [Google Scholar]
- 24.Baird JK. Resistance to therapies for infection by Plasmodium vivax. Clin Microbiol Rev. 2009;22:508–534. doi: 10.1128/CMR.00008-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Young MD, Burgess RW. Pyrimethamine resistance in Plasmodium vivax malaria. Bull World Health Organ. 1959;20:27–36. [PMC free article] [PubMed] [Google Scholar]
- 26.de Pecoulas PE, Basco LK, Tahar R, Ouatas T, Mazabraud A. Analysis of the Plasmodium vivax dihydrofolate reductase-thymidylate synthase gene sequence. gene. 1998;211:177–185. doi: 10.1016/s0378-1119(98)00118-8. [DOI] [PubMed] [Google Scholar]
- 27.de Pecoulas PE, Tahar R, Ouatas T, Mazabraud A, Basco LK. Sequence variations in the Plasmodium vivax dihydrofolate reductase-thymidylate synthase gene and their relationship with pyrimethamine resistance. Mol Biochem Parasitol. 1998;92:267–273. doi: 10.1016/s0166-6851(97)00247-8. [DOI] [PubMed] [Google Scholar]
- 28.Imwong M, Pukrittakayamee S, Looareesuwan S, Pasvol G, Poirreiz J, et al. Association of genetic mutations in Plasmodium vivax dhfr with resistance to sulfadoxine-pyrimethamine: geographical and clinical correlates. Antimicrob Agents Chemother. 2001;45:3122–3127. doi: 10.1128/AAC.45.11.3122-3127.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Imwong M, Pukrittayakamee S, Renia L, Letourneur F, Charlieu JP, et al. Novel point mutations in the dihydrofolate reductase gene of Plasmodium vivax: evidence for sequential selection by drug pressure. Antimicrob Agents Chemother. 2003;47:1514–1520. doi: 10.1128/AAC.47.5.1514-1521.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tjitra E, Baker J, Suprianto S, Cheng Q, Anstey NM. Therapeutic efficacies of artesunate-sulfadoxine-pyrimethamine and chloroquine-sulfadoxine-pyrimethamine in vivax malaria pilot studies: relationship to Plasmodium vivax dhfr mutations. Antimicrob Agents Chemother. 2002;26:3947–3953. doi: 10.1128/AAC.46.12.3947-3953.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hastings MD, Porter KM, Maguire JD, Susanti I, Kania W, et al. Dihydrofolate reductase mutations in Plasmodium vivax from Indonesia and therapeutic response to sulfadoxine plus pyrimethamine. J Infect Dis. 2004;189:744–750. doi: 10.1086/381397. [DOI] [PubMed] [Google Scholar]
- 32.Leartsakulpanich U, Imwong M, Pukrittayakamee S, White NJ, Snounou G, et al. Molecular characterization of dihydrofolate reductase in relation to antifolate resistance in Plasmodium vivax. Mol Biochem Parasitol. 2002;119:63–73. doi: 10.1016/s0166-6851(01)00402-9. [DOI] [PubMed] [Google Scholar]
- 33.Hastings MD, Sibley CH. Pyrimethamine and WR99210 exert opposing selection on dihydrofolate reductase from Plasmodium vivax. Proc Natl Acad Sci U S A. 2002;99:13137–13141. doi: 10.1073/pnas.182295999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hastings MD, Maguire JD, Bangs MJ, Zimmerman PA, Reeder JC, et al. Novel Plasmodium vivax dhfr alleles from the Indonesian Archipelago and Papua New Guinea: association with pyrimethamine resistance determined by a Saccharomyces cerevisiae expression system. Antimicrob Agents Chemother. 2005;49:733–740. doi: 10.1128/AAC.49.2.733-740.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Auliff AM, Adams JH, O’Neil MT, Cheng Q. Defining the role of mutations in Plasmodium vivax dihydrofolate reductase-thymidylate synthase gene using an episomal Plasmodium falciparum transfection system. Antimicrob Agents Chemother. 2010;54:3927–3932. doi: 10.1128/AAC.00628-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.O’Neil MT, Korsinczky ML, Gresty KJ, Auliff A, Cheng Q. A novel Plasmodium falciparum expression system for assessing antifolate resistance caused by mutant P. vivax dihydrofolate reductase-thymidylate synthase. J Infect Dis. 2007;196:467–474. doi: 10.1086/519286. [DOI] [PubMed] [Google Scholar]
- 37.Bhattacharjee S, van Ooij C, Balu B, Adams JH, Haldar K. Maurer’s clefts of Plasmodium falciparum are secretory organelles that concentrate virulence protein reporters for delivery to the host erythrocyte. Blood. 2008;111:2418–2426. doi: 10.1182/blood-2007-09-115279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.van Ooij C, Tamez P, Bhattacharjee S, Hiller NL, Harrison T, et al. The malaria secretome: from algorithms to essential function in blood stage infection. PLoS Pathog. 2008;4:e1000084. doi: 10.1371/journal.ppat.1000084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tamez PA, Bhattacharjee S, van Ooij C, Hiller NL, Llinas M, et al. An erythrocyte vesicle protein exported by the malaria parasite promotes tubovesicular lipid import from the host cell surface. PLoS Pathog. 2008;4:e1000118. doi: 10.1371/journal.ppat.1000118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Balu B, Adams JH. Advancements in transfection technologies for Plasmodium. International Journal for Parasitology. 2007;37:1–10. doi: 10.1016/j.ijpara.2006.10.001. [DOI] [PubMed] [Google Scholar]
- 41.Balu B, Chauhan C, Maher SP, Shoue DA, Kissinger JC, et al. piggyBac is an effective tool for functional analysis of the Plasmodium falciparum genome. BMC Microbiol. 2009;9:83. doi: 10.1186/1471-2180-9-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Balu B, Shoue DA, Fraser MJJ, Adams JH. High-efficiency transformation of Plasmodium falciparum by the lepidopteran transposable element piggyBac. . Proc Natl Acad Sci U S A. 2005;102:16391–16396. doi: 10.1073/pnas.0504679102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pfahler JM, Galinski MR, Barnwell JW, Lanzer M. Transient transfection of Plasmodium vivax blood stage parasites. Molecular and Biochemical Parasitology. 2006;149:99–101. doi: 10.1016/j.molbiopara.2006.03.018. [DOI] [PubMed] [Google Scholar]
- 44.Sa JM, Yamamoto MM, Fernandez-Becerra C, de Azevedo MF, Papakrivos J, et al. Expression and function of pvcrt-o, a Plasmodium vivax ortholog of pfcrt, in Plasmodium falciparum and Dictyostelium discoideum. Mol Biochem Parasitol. 2006;150:219–228. doi: 10.1016/j.molbiopara.2006.08.006. [DOI] [PubMed] [Google Scholar]
- 45.Somsak V, Uthaipibull C, Prommana P, Srichairatanakool S, Yuthavong Y, et al. Transgenic Plasmodium parasites stably expressing Plasmodium vivax dihydrofolate reductase-thymidylate synthase as in vitro and in vivo models for antifolate screening. Malar J. 2011;10:291. doi: 10.1186/1475-2875-10-291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Shearer TW, Kozar MP, O’Neil MT, Smith PL, Schiehser GA, et al. In vitro metabolism of phenoxypropoxybiguanide analogues in human liver microsomes to potent antimalarial dihydrotriazines. J Med Chem. 2005;48:2805–2813. doi: 10.1021/jm049683+. [DOI] [PubMed] [Google Scholar]
- 47.Canfield CJ. Geneva: World Health Organisation Monograph Series; Bruce-Chwatt L.J e, editor. 99–100 p. 1986.
- 48.Canfield CJ, Milhous WK, Ager AL, Rossan RN, Sweeney TR, et al. PS-15: a potent, orally active antimalarial from a new class of folic acid antagonists. Am J Trop Med Hyg. 1993;49:121–126. doi: 10.4269/ajtmh.1993.49.121. [DOI] [PubMed] [Google Scholar]
- 49.Edstein MD, Kotecka BM, Ager AL, Smith KS, DiTusa CA, et al. Antimalarial pharmacodynamics and pharmacokinetics of a third-generation antifolate–JPC2056–in cynomolgus monkeys using an in vivo in vitro model. J Antimicrob Chemother. 2007;60:811–818. doi: 10.1093/jac/dkm280. [DOI] [PubMed] [Google Scholar]
- 50.Zhang K, Rathod PK. Divergent regulation of dihydrofolate reductase between malaria parasite and human host. Science. 2002;296:545–547. doi: 10.1126/science.1068274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Nirmalan N, Wang P, Sims PF, Hyde JE. Transcriptional analysis of genes encoding enzymes of the folate pathway in the human malaria parasite Plasmodium falciparum. Mol Microbiol. 2002;46:179–190. doi: 10.1046/j.1365-2958.2002.03148.x. [DOI] [PubMed] [Google Scholar]
- 52.Nirmalan N, Sims PF, Hyde JE. Translational up-regulation of antifolate drug targets in the human malaria parasite Plasmodium falciparum upon challenge with inhibitors. Mol Biochem Parasitol. 2004;136:63–70. doi: 10.1016/j.molbiopara.2004.02.013. [DOI] [PubMed] [Google Scholar]
- 53.Goldberg DE, Janse CJ, Cowman AF, Waters AP. Has the time come for us to complement our malaria parasites? Trends Parasitol. 2010. [DOI] [PubMed]
- 54.Trager W, Jensen JB. Human malaria parasites in continuous culture. 1976. J Parasitol. 1976;91:484–486. doi: 10.1645/0022-3395(2005)091[0484:HMPICC]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 55.Lambros C, Vanderberg JP. Synchronization of Plasmodium falciparum erythrocytic stages in culture. J Parasitol. 1979;65:418–420. [PubMed] [Google Scholar]
- 56.Auliff A, Wilson DW, Russell B, Gao Q, Chen N, et al. Amino acid mutations in Plasmodium vivax DHFR and DHPS from several geographical regions and susceptibility to antifolate drugs. Am J Trop Med Hyg. 2006;75:617–621. [PubMed] [Google Scholar]
- 57.Maher SP, Balu B, Shoue DA, Weissenbach ME, Adams JH. A highly sensitive, PCR-based method for the detection of Plasmodium falciparum clones in microtiter plates. Malar J. 2008;7:222. doi: 10.1186/1475-2875-7-222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Liu YG, Huang N. Efficient Amplification of Insert End Sequences from Bacterial Artificial Chromosome Clones by Thermal Asymmetric Interlaced PCR. Plant Molecular Biology Reporter. 1998;16:175–181. [Google Scholar]
- 59.Chavchich M, Gerena L, Peters J, Chen N, Cheng Q, et al. Role of pfmdr1 amplification and expression in induction of resistance to artemisinin derivatives in Plasmodium falciparum. Antimicrob Agents Chemother. 2010;54:2455–2464. doi: 10.1128/AAC.00947-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Arnold C, Hodgson IJ. Vectorette PCR: a novel approach to genomic walking. PCR Methods Appl. 1991;1:39–42. doi: 10.1101/gr.1.1.39. [DOI] [PubMed] [Google Scholar]
- 61.Duraisingh MT, Curtis J, Warhurst DC. Plasmodium falciparum: detection of polymorphisms in the dihydrofolate reductase and dihydropteroate synthetase genes by PCR and restriction digestion. Exp Parasitol. 1998;89:1–8. doi: 10.1006/expr.1998.4274. [DOI] [PubMed] [Google Scholar]
- 62.Edstein MD, Bahr S, Kotecka B, Shanks GD, Rieckmann KH. In vitro activities of the biguanide PS-15 and its metabolite, WR99210, against cycloguanil-resistant Plasmodium falciparum isolates from Thailand. Antimicrob Agents Chemother. 1997;41:2300–2301. doi: 10.1128/aac.41.10.2300. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Materials and Methods for inverse PCR or vectorette PCR reactions to identify the piggyBac insertion sites.
(DOC)