Abstract
PURPOSE
This study was designed to evaluate potential preventive effects of meditation or exercise on incidence, duration, and severity of acute respiratory infection (ARI) illness.
METHODS
Community-recruited adults aged 50 years and older were randomized to 1 of 3 study groups: 8-week training in mindfulness meditation, matched 8-week training in moderate-intensity sustained exercise, or observational control. The primary outcome was area-under-the-curve global illness severity during a single cold and influenza season, using the Wisconsin Upper Respiratory Symptom Survey (WURSS-24) to assess severity. Health care visits and days of missed work were counted. Nasal wash collected during ARI illness was assayed for neutrophils, interleukin-8, and viral nucleic acid.
RESULTS
Of 154 adults randomized into the study, 149 completed the trial (82% female, 94% white, mean age 59.3 ± 6.6 years). There were 27 ARI episodes and 257 days of ARI illness in the meditation group (n = 51), 26 episodes and 241 illness days in the exercise group (n = 47), and 40 episodes and 453 days in the control group (n = 51). Mean global severity was 144 for meditation, 248 for exercise, and 358 for control. Compared with control, global severity was significantly lower for meditation (P = .004). Both global severity and total days of illness (duration) trended toward being lower for the exercise group (P=.16 and P=.032, respectively), as did illness duration for the meditation group (P=.034). Adjusting for covariates using zero-inflated multivariate regression models gave similar results. There were 67 ARI-related days of-work missed in the control group, 32 in the exercise group (P = .041), and 16 in the meditation group (P <.001). Health care visits did not differ significantly. Viruses were identified in 54% of samples from meditation, 42% from exercise, and 54% from control groups. Neutrophil count and interleukin-8 levels were similar among intervention groups.
CONCLUSIONS
Training in meditation or exercise may be effective in reducing ARI illness burden.
Keywords: respiratory tract infections, common cold, exercise, influenza, meditation, perceived stress, psychological
INTRODUCTION
Acute respiratory infection (ARI) is extremely common, often debilitating, and among the most costly of human illnesses. Influenza, the most serious of the viral ARIs, is associated with approximately 36,000 deaths and more than 500,000 hospitalizations in the United States yearly.1 Nevertheless, symptoms of influenza infection are usually indistinguishable from those produced by other viruses.2,3 In the United States each year, noninfluenza ARI accounts for more than 20 million doctor visits and 40 million lost school and work days, with an economic impact of more than $40 billion, making noninfluenza ARI rank first in the top 10 most expensive illnesses.4 Reducing this burden even modestly could lead to substantial economic and quality-of-life benefits.
Available treatments are not very effective. If started early enough, antiviral medications have limited efficacy for influenza,5 but not for other viral ARIs. Symptomatic treatments may reduce severity slightly but have never been shown to reduce illness duration or overall severity. Influenza vaccination is accepted as effective for prevention,6 but it is imperfect, with sero-protection rates ranging from 60% to 80% in healthy younger adults and 40% to 60% in the elderly.7 For noninfluenza ARI, immunization strategies are impractical, and preventive strategies are limited to not smoking,8 hand washing,9 and avoiding sick contacts.
There is some evidence that enhancing general physical and mental health may reduce ARI burden. In a series of observational and viral inoculation studies, perceived stress,10 negative emotion,11 and lack of social support12 predicted not only self-reported illness, but also such biomarkers as viral shedding and inflammatory cytokine activity. Evidence suggests that mindfulness meditation can reduce experienced stress and negative emotions.13,14 Similarly, both epidemiological and experimental studies have suggested that regular exercise may protect people from ARI illness.15,16 A recent observational cohort study (n = 1,002 adults) reported 32% to 46% lower incidence, duration, and severity of ARI illness among the most active vs least active participants.17 Thus, sufficient evidence exists to justify testing the hypothesis that training in meditation or exercise can reduce susceptibility to ARI illness.
METHODS
Design
The experimental design used balanced, randomized allocation to 3 parallel groups: (1) mindfulness meditation, (2) moderate-intensity exercise, or (3) observational control. Allocation was directed using computer-generated randomization codes (balanced blocks of 3) in sequentially numbered envelopes.
The primary aim was to determine whether training in either meditation or exercise could reduce ARI illness burden compared with no intervention in the control group. Secondary aims included assessment of whether self-reported psychosocial states would respond to intervention, and whether these responses could help explain potential effects on ARI outcomes. These measures included perceived stress, positive and negative emotion, perceived social support, anxiety, optimism, sleep quality, and general physical and mental health. Secondary aims also included assessment of antibody response to influenza vaccination, which will be reported elsewhere. All participants received trivalent inactivated influenza vaccination during week 6 of the interventions.
Mindfulness Meditation
The meditation intervention was derived from work by Jon Kabat-Zinn and others at the University of Massachusetts Medical Center, where mindfulness-based stress reduction (MBSR) was developed.18 The University of Wisconsin (UW) program has been in existence since 1993. All UW instructors have advanced degrees, and all were trained in Massachusetts by the Kabat-Zinn’s group. The standardized 8-week MBSR course includes weekly 2½-hour group sessions and 45 minutes of daily at-home practice. This intervention, originally designed for stress reduction, is based on the idea that an increased awareness of physical, emotional, and cognitive manifestations of stress may lead to a healthier mind-body response to stress. The concept of mindfulness refers to a state of nonjudgmental awareness, a heightened sensitivity to bodily sensation, and attention to one’s own thoughts and emotions. The goal for learners is to continue lifelong meditation practice.
Exercise
The exercise program was designed and led by senior exercise physiology staff at the UW Health Sports Medicine Center and was matched to the mindfulness meditation program in terms of duration (8 weeks), contact time (weekly 2½ hour group sessions), home practice (45 minutes per day), and location. Two of the 3 UW exercise instructors had master’s degrees in clinical exercise physiology. The third had a bachelor’s in sports management. All had many years of experience, and all carried licensed athletic trainer and certified strength and conditioning coach certifications. Borg’s Rating of Perceived Exertion was used to guide participants toward moderate-intensity sustained exercise, with a target rating of 12 to 16 points on the 6 to 20 point scale.19 Weekly group sessions were divided into didactic instruction (cognitive, logistic, and behavioral) and practice (moderately intensive exercise using stationary bicycles, treadmills, and other equipment). For most participants, home exercise consisted of brisk walking or jogging.
Control
Because this trial was preliminary, we opted for observational control as the comparison study group, hoping to minimize risk of type II, as well as type I, error.20 Control participants were monitored in the same manner as those receiving interventions and were eligible to receive meditation or exercise training, or monetary equivalent, at the conclusion of the trial.
Randomization
SAS software (SAS Institute Inc) was used to generate 165 unique identification numbers in balanced blocks of 3. Codes were concealed in consecutively numbered sealed envelopes, which were opened after consent to indicate allocation.
Setting
This community-based trial was coordinated by the UW Department of Family Medicine. Behavioral training interventions were conducted at UW Research Park, a multipurpose outpatient clinic with exercise facilities and space suitable for meditation training.
Participants
Inclusion criteria were aged 50 years or older, willingness to undertake any of the 3 randomization outcomes, and reporting either 2 or more colds in the last 12 months or an average of 1 or more cold per year. Exclusion criteria were previous training or current practice of meditation, moderate exercise at least 2 times a week or vigorous exercise at least 1 time a week, and a score of less than 24 points on the Folstein Mini-Mental State Examination21 or more than 14 points on the 9-item Patient Health Questionnaire (PHQ-9) depression screen22; immunodeficiency, autoimmune, or malignant disease; or prior allergic reaction to influenza vaccine or egg allergy.
Human Subjects and Safety
The protocol was approved by the UW Health Sciences Institutional Review Board. The trial was conducted in accordance with the protocol approved by the National Institutes of Health (NIH) and was monitored by a data-and safety-monitoring committee. The American Heart Association guidelines23 for safety when beginning an exercise program were followed. There were no specific adverse outcomes designated for monitoring.
Recruitment and Monitoring
Community-targeted recruitment methods included advertising in local media. Prospective participants were screened by telephone and then met in person for entry into the run-in trial, which consisted of 2 in-person appointments, 2 telephone contacts, and 1 set of homework questionnaires. Those completing the run-in trial were eligible for consent and entry into the main trial. For logistical reasons related to NIH funding under the American Recovery and Reinvestment Act, the study had to be done in 1 year, and 2 cohorts were needed. The first cohort began in September, and the second cohort began in January. Participants were monitored biweekly by telephone beginning postintervention and continuing until study exit at the end of May.
Primary Outcome Measures
The primary outcome was defined as area-under-the-curve global severity for all ARI illness days throughout observation, from consent to study exit. During each ARI illness episode, severity was assessed once daily using a 24-item version of the Wisconsin Upper Respiratory Symptom Survey (WURSS).24–26 The WURSS-24 adds items assessing headache, body aches, and fever to the well-validated WURSS-21.27 Daily severity scores come from summing items 2 through 23; the first and last items are analyzed separately. The beginning of each ARI illness episode was defined as the time a participant first answered yes to either: “Do you think you are coming down with a cold?” or “Do you think that you have a cold?” The participant also had to score 2 or more points on the Jackson scale,28 with at least 1 point from the following cold symptoms (or synonyms): (1) nasal discharge (runny nose), (2) nasal obstruction (plugged nose, stopped up nose, stuffiness), (3) sneezing, or (4) sore throat (scratchy throat). To be classified as an ARI illness episode, these criteria had to be met at least 2 days in a row. The last day of the ARI illness episode was the last day the participant rated illness severity higher than 0 using the first item of the WURSS instrument, “How sick do you feel today?” Times of self-report were recorded, and illness duration was assessed in hours and minutes and then converted to decimalized days. Participants were provided Jackson and WURSS questionnaires at enrollment and were reminded with each study contact to begin documenting ARI symptoms as soon as they believed they might be getting a cold.
Biomarkers
With each ARI illness episode, a nasal wash was collected within 3 days of symptom onset and analyzed for interleukin-8 (IL-8), neutrophil count, and viral nucleic acid. Elevated neutrophil count and IL-8 levels are indicators of inflammation and correlate with symptom severity and viral shedding.29,30 Multiplex polymerase chain reaction (PCR) methods developed and validated at UW were used to identify respiratory viruses.31
Psychosocial Self-Report Measures
Several validated self-report questionnaires were used to explore potential explanatory pathways linking behavioral interventions to ARI outcomes. The SF-12 is a 12-item version of the Medical Outcomes Study Short Form (SF-36) that measures overall health and provides algorithm-weighted physical and mental health scores.32 The PSS-10 is a 10-item version of Cohen’s Perceived Stress Scale that has been linked to ARI outcomes, including influenza.10 The Positive and Negative Affect Schedule (PANAS)33 assesses positive and negative emotion, also known to be linked to ARI outcomes. Spielberger’s State Trait Anxiety Inventory (STAI) assesses anxiety either as trait (baseline) or state (secondary outcome, explanatory factor).34 The 6-item Life Orientation Test (LOT) assesses cognitive aspects of optimism.35 To assess perceived social support, we used Ryff’s 9-item Positive Relationships with Others (PR) scale.36 The Pittsburgh Sleep Quality Index (PSQI)37 served as our measure of sleep quality. The International Physical Activity Questionnaire (IPAQ)38 assessed exercise, and the Mindful Attention Awareness Scale (MAAS)39 assessed mindfulness. The SF-12, PSS-10, PANAS, IPAQ, and MAAS were administered at baseline, 1 week postintervention, and monthly thereafter. The STAI, LOT, PR, and PSQI indices were scored at baseline, 1 week postintervention, and then again 3 months later.
Health Care Utilization and Days of Work or School Missed (Sick Days)
During each biweekly telephone call, we asked about visits to health care facilities (clinic, hospital, or urgent care), the reason for the visit, and about missed work or school activities. Health care visits and missed work days were then classified as either related or unrelated to ARI illness, both by asking the participant’s opinion and by cross-checking against self-report WURSS-24 data. In questionable cases, classification as ARI-related or not was done by the senior author (B.B.), guided by relevant data but blinded to allocation.
Statistical Methods
The sample size of 150 was based on power estimates contrasting (1) meditation vs control and (2) exercise vs control. To control for multiple testing, we chose a P ≤.025 cutoff for null hypothesis rejection. One-sided testing was justified by previously published research, all in the direction of positive results. To arrive at the primary outcome of area-under-the-curve global severity, we first computed daily WURSS-24 scores by simple summation and then applied trapezoidal approximation over illness episodes. For the less than 2% of missing WURSS-24 data, we assessed patterns of missing data, which satisfied missing-at-random criteria, then applied a multiple imputation strategy, using an expectancy maximization algorithm,40 which was undertaken before unblinding. Unadjusted between-group contrasts were calculated using 1-sided t tests for continuous variables and proportional difference testing for binomials. Because most participants did not experience ARI illness, zero-inflated regression models were used to control for potential confounders. These models take into account both logistic (incidence) and linear (days of illness or global severity) data. Covariates used in these models were age, sex, education, smoking status, body mass index, baseline physical and mental health (SF-12), and cohort. Because global severity was skewed, Box-Cox transformation was used for this outcome in these models. To explore potential causal pathways, we assessed the relationship of secondary outcomes measured just after interventions to the main outcomes. The statistical framework for potential mediation was based on the Baron and Kenny model,41 modified by Krull and MacKinnon,42 and by coauthor Brown.43
Additional detail on the results of the zero-inflated regression models and potential confounders is available in Supplemental Table 1 and Table 2. See Supplemental Table 3 for information on model-data fit (available at http://annfammed.org/content/10/4/337/suppl/DC1).
RESULTS
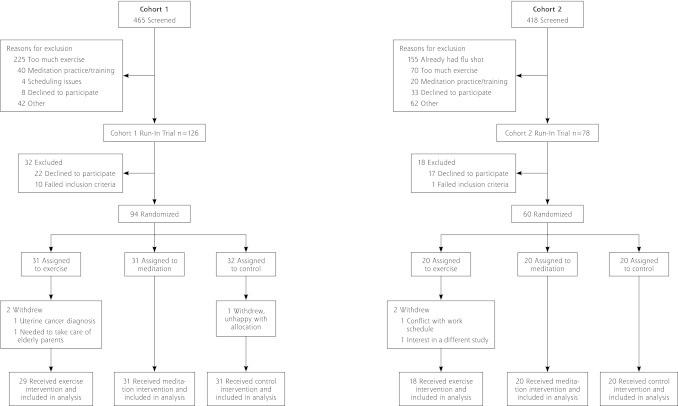
Of 883 adults screened, 204 were entered in the run-in trial; 154 consented and were enrolled and randomized into the main trial, with 94 in cohort 1 and 60 in cohort 2 (Figure 1). Interventions began in September 2009 for cohort 1 and in January 2010 for cohort 2. Baseline measures were similar across the 3 groups (Table 1). Both cohorts were monitored through May 2010. Retention was high, with 149 (96.7%) providing primary outcome data; 82% were female, 94% were white, and their mean age was 59.3 ± 6.6 years.
Figure 1.
CONSORT diagram.
Table 1.
Demographic and Psychosocial Characteristics of Study Population
| Characteristic | Exercise | Meditationa | Control | P Value |
|---|---|---|---|---|
| Sample, n | 47 | 51 | 51 | |
| Age, mean y (SD) | 59.0 (6.6) | 60.0 (6.5) | 58.8 (6.8) | 0.63 |
| Female, n (%) | 39 (83.0) | 42 (82.4) | 41 (80.4) | 0.94 |
| Nonsmokers, n (%) | 43 (91.5) | 48 (94.1) | 48 (94.1) | 0.84 |
| Race,b n (%) | 3 (6.4) | 1(1.9) | 2 (3.9) | 0.52 |
| Black | ||||
| White, n (%) | 43 (91.5) | 49 (92.5) | 48 (94.1) | 0.88 |
| Other, n (%) | 1 (2.13) | 3(5.7) | 1 (2.0) | 0.50 |
| Ethnicity non-Hispanic, n (%) | 47 (100) | 51 (100) | 49 (96.1) | 0.14 |
| BMI, mean (SD) | 29.0 (6.9) | 29.0 (6.0) | 29.8 (6.8) | 0.77 |
| SF-12 physical, mean score (SD) | 50.9 (9.3) | 50.7 (9.4) | 50.0 (9.3) | 0.89 |
| SF-12 mental, mean score (SD) | 52.3 (6.6) | 50.9 (8.6) | 51.1 (7.8) | 0.62 |
| College graduate or higher, n (%) | 27 (57.4) | 36 (70.6) | 35 (68.6) | 0.34 |
| Income >$50,000, n (%) | 25 (53.2) | 31 (60.8) | 29 (56.9) | 0.75 |
| Mean scores at baseline, n (SD) | ||||
| Physical health (SF-12) | 50.9 (9.3) | 50.7 (9.4) | 50.0 (9.3) | 0.89 |
| Mental health (SF-12) | 52.3 (6.6) | 50.9 (8.6) | 51.1 (7.8) | 0.62 |
| Negative emotion (PANAS) | 15.2 (5.1) | 15.8 (4.0) | 14.8 (3.7) | 0.45 |
| Positive emotion (PANAS) | 36.7 (6.2) | 36.2 (6.5) | 36.3 (6.6) | 0.91 |
| Optimism (LOT) | 27.1 (3.4) | 27.1 (3.9) | 28.3 (3.6) | 0.17 |
| Social support (Ryff PR) | 44.9 (7.2) | 45.4 (6.6) | 45.9 (6.3) | 0.73 |
| Perceived stress (PSS-10) | 11.4 (6.0) | 13.0 (4.7) | 11.2 (5.4) | 0.19 |
| Anxiety (current state) (STAI) | 30.7 (9.1) | 32.2 (8.1) | 29.8 (7.3) | 0.31 |
| Sleep quality (PSQI) | 4.6 (3.1) | 5.1 (2.6) | 4.7 (2.5) | 0.46 |
| Mindfulness (MAAS) | 4.6 (0.7) | 4.5 (0.7) | 4.6 (0.7) | 0.37 |
| Exercise, MET min/wk (IPAQ) | 1,214 (1,526) | 644 (664) | 925 (1,225) | 0.06 |
BMI = body mass index (weight/height2); IPAQ = International Physical Activity Questionnaire; LOT = Life Orientation Test; MAAS = Mindful Attention Awareness Scale; PANAS = Positive and Negative Affect Schedule; PSS-10 = 10-item Perceived Stress Scale; PSQI = Pittsburgh Sleep Quality Index; Ryff PR = Ryff’s 9-item Positive Relationships with Others scale; SF-12 = Medical Outcomes Study Short Form-12; STAI = State Trait Anxiety Inventory.
Note: P values were calculated by analysis of variance using NCSS 2007, LLC (http://www.ncss.com).
Missing information on income from meditation group (n = 2).
One person in the meditation group reported 3 racial categories.
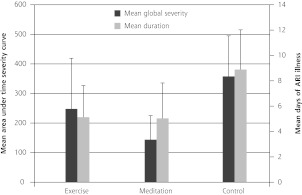
There were 27 ARI episodes and 257 days of illness in the meditation group, 26 episodes and 241 ARI illness days for the exercise group, and 40 episodes and 453 ARI illness days for the control group (Table 2). Mean global severity across all randomized participants was 144 for meditation, 248 for exercise, and 358 for control (Figure 2). Comparing meditation with control groups, 1-sided t tests yielded P = .034 for illness days and P = .004 for global severity. Comparing exercise with control groups, corresponding P values were .032 for illness days and .16 for global severity. Adjusting for covariates using zero-inflated multivariate regression models, both total days of illness (P = .033) and global severity (P = .010) appeared to be lower for meditation, but not for exercise (P = 0.47 and 0.31, respectively).
Table 2.
Main Outcomes
| Between-Group Differences | |||||
|---|---|---|---|---|---|
|
|
|||||
| Outcome | Exercise (n = 47) | Meditation (n = 51) | Control (n = 51) | Exercise vs Control | Meditation vs Control |
| Participants with ARI illness, n (%) or (95% CI) | 17 (36) | 21 (41) | 28 (55) | 0.19 (−0.01 to 0.37) P=.032 |
0.14 (−0.06 to 0.32) P=.083 |
| Number of ARI episodes | 26 | 27 | 40 | ||
| Mean global severity score, n (95% CI)a | 248 (77 to 419) | 144 (62 to 225) | 358 (221 to 495) | 110 (−105 to 324) P=.16 |
214 (56 to 372) P=.0042 |
| Total days of ARI illness | 241 | 257 | 453 | ||
| Mean ARI illness days, n (95% CI) | 5.13 (2.64 to 7.62) | 5.04 (2.25 to 7.83) | 8.89 (5.76 to 12.02) | 3.76 (−0.24 to 7.75) P=.032 |
3.85 (−0.29 to 7.99) P=.034 |
| Health care visits and work days lost to illness | |||||
| Episodes of missed work, n | 21 | 25 | 33 | ||
| Total days missed, n | 91.5 | 99 | 144.5 | ||
| Mean missed days, n (95% CI) | 1.9 (0.8 to 3.1) | 1.9 (0.7 to 3.2) | 2.8 (1.5 to 4.2) | 0.9 (−0.9 to 2.7) P=.16 |
0.9 (−0.9 to 2.7) P=.17 |
| ARI-related missed days, n (95% CI) | 32 | 16 | 67 | 0.11 (−0.01 to 0.24) P=.041 |
0.30 (0.19 to 0.40) P <.001 |
| Mean ARI-related missed days, n (95% CI) | 0.68 (0.1 to 1.2) | 0.31 (0.1 to 0.5) | 1.31 (0.5 to 2.1) | 0.63 (−0.4 to 1.6) P=.10 |
1.0 (0.2 to 1.8) P=.011 |
| Total number of health care visits | 116 | 116 | 121 | ||
| Mean health care visits, n (95% CI) | 2.5 (1.7 to 3.2) | 2.3 (1.6 to 2.9) | 2.4 (1.8 to 2.9) | −0.1 (−1.0 to 0.8) P=.42 |
0.1 (−0.7 to 0.9) P=.41 |
| ARI-related health care visits, n (95% CI) | 15 | 10 | 16 | 0.003 (−0.09 to 0.09) P=.47 |
0.05 (−0.04 to 0.13) P=.13 |
| Mean ARI-related health care visits, n (95% CI) | 0.32 (0.14 to 0.49) | 0.20 (0.07 to 0.32) | 0.31 (0.12 to 0.50) | −0.01 (−0.26 to 0.25) P=.48 |
0.11 (−0.11 to 0.34) P=.15 |
| Mean non-ARI-related health care visits, n (95% CI) | 2.15 (1.5 to 2.8) | 2.08 (1.5 to 2.7) | 2.06 (1.5 to 2.6) | −0.09 (−0.89 to 0.71) P=.41 |
−0.02 (−0.80 to 0.76) P=.48 |
| Biomarker and viral identification data from nasal secretions | |||||
| IL-8, mean pg/mL (95% CI)b | 694 (484 to 904) | 910 (696 to 1,124) | 658 (511 to 806) | −36 (−282 to 211) | −252 (−497 to −6) |
| Neutrophils, mean n/HPF (95% CI) | 103.7 (−46.1 to 253.5) | 107.9 (23.0 to 192.8) | 110.4 (22.9 to 197.9) | 6.7 (−150.8 to 164.2) | 2.5 (−119.6 to 124.6) |
| Positive for virus, n (95% CI) | 8 | 14 | 19 | 0.12 (−0.15 to 0.38) | 0.004 (−0.24 to 0.25) |
| Specific viruses (n)c | HRV (7) | CoV (2) | Adv C (1) | ||
| MPV (1) | HRV (10) | CoV (3) | |||
| MPV (1) | Flu A (2) | ||||
| PIV (2) | HRV (11) | ||||
| RSV (1) | MPV (1) | ||||
| PIV (1) | |||||
| RSV (1) | |||||
| Negative for virus, n | 11 | 12 | 16 | ||
AdV=adenoviruses; ARI=acute respiratory infection; CoV=coronaviruses; flu A=influenza A/H1N1/California 2009; HPF = high-power field; HRV=human rhinoviruses; IL-8=interleukin 8; MPV=metapneumovirus; PIV=parainfluenza viruses; RSV = respiratory syncytial virus. WURSS-24 = 24-item Wisconsin Upper Respiratory Symptom Survey.
Notes: P values come from unadjusted intervention-to-control contrasts, using 2 sample t test for continuous means (SAS software) and proportional difference for binomials (StatXact-5, Cytel Statistical Software & Services). P values represent unadjusted contrasts of interventions to control. Significance was set at P ≤.025.
Global severity calculated as the area under the curve using WURSS-24 scores for y-axis and duration of ARI illness as x-axis.
IL-8: interleukin-8 (pg/mL) assessed by ELISA (Human IL-8 BD OptEIA Set, BD Biosciences Pharmingen).
Two nasal wash samples from the meditation group yielded both CoV and HRV. One ARI in the control group yielded both AdV and HRV.
Figure 2.
Primary outcomes: days of acute respiratory infection illness and area-under-curve global severity.
ARI = acute respiratory infection; global severity = area under curve time severity curve, calculated as trapezoidal approximation using daily scores on Wisconsin Upper Respiratory Infection Symptom Severity (WURSS-24) for y-axis and duration of ARI illness as x-axis.
Note: Error bars indicate 95% confi dence intervals.
Specific viruses were identified for 53.8% of nasal wash samples tested from the meditation group, 42.1% of samples from the exercise group, and 54.3% of control samples (Table 2). Viruses identified included adenovirus (1), coronavirus (5), influenza (2), metapneumovirus (3), parainfluenza (3), respiratory syncytial virus (2), and rhinovirus (28). Both cases of influenza (A/2009/California/H1N1) were in the control group. Mean neutrophil counts (per high-power field [HPF]) and IL-8 concentration in the 3 groups were 108/HPF and 910 pg/mL for the meditation group, 104/HPF and 694 pg/mL for the exercise group, and 110/HPF and 658 pg/mL for the control group. Between-group differences were not statistically significant for these biomarkers, except for slightly higher IL-8 levels in nasal wash collected during ARIs from participants in the meditation group (P = .022).
In the meditation group, there were 99 days of missed work (16 were ARI-related) and 116 health care visits (10 were ARI-related). Among the exercise group there were 91.5 days of missed work (32 ARI-related) and 116 health care visits (15 ARI-related). In the control group there were 144.5 missed days of work (67 ARI-related) and 121 health care visits (16 ARI-related). Total number of health care visits and total and ARI-related missed work days were not statistically distinguishable among the groups. ARI-related absenteeism, however, was significantly lower for the meditation group (P <.001) and marginally so for the exercise group (P = .041) when compared with the control group.
Although most self-reported psychosocial health indicators trended in expected directions, only self-reported exercise and the mental health portion of the SF-12 displayed statistically significant differences when compared with the control group (Table 3). Compared with control participants, those assigned to exercise training increased their IPAQ scores at 9 weeks, a change which persisted at 3 months. Statistically significant improvements in SF-12 mental health in both groups were more modest, and were seen at 9 weeks in the exercise group and at both 9 weeks and 3 months among those assigned meditation training. Statistical analyses directed at assessing potential mediation failed to show clear and convincing pathways from interventions to outcomes.
Table 3.
Secondary Outcomes at 9 Weeks and 3 Months
| Exercise, Mean (95% CI) | Meditation, Mean (95% CI) | Control, Mean (95% CI) | ||||
|---|---|---|---|---|---|---|
|
|
||||||
| Outcome | 9 wk | 3 mo | 9 wk | 3 mo | 9 wk | 3 mo |
| Exercise | ||||||
| MET min/wk (IPAQ) | 2,222 (1,815 to 2,628)a | 1,805 (1,356 to 2,253)a | 1,037 (694 to 1,381) | 1,122 (804 to 1,440) | 1,224 (810 to 1,638) | 1,050 (688 to 1,412) |
| Mindfulness score (MAAS) | 4.59 (4.36 to 4.82) | 4.82 (4.59 to 5.05) | 4.55 (4.37 to 4.73) | 4.73 (4.54 to 4.91) | 4.60 (4.40 to 4.81) | 4.59 (4.37 to 4.82) |
| Indicators of good health (positive change indicates improvement) | ||||||
| Physical health score (SF-12) | 51.8 (49.3 to 54.2) | 52.0 (49.4 to 54.6) | 49.8 (47.2 to 52.3) | 50.5 (48.0 to 53.1) | 51.1 (48.5 to 53.6) | 50.6 (47.8 to 53.6) |
| Mental health score (SF-12) | 53.0 (50.9 to 55.1)b | 49.7 (46.7 to 52.7) | 52.6 (50.5 to 54.7)b | 50.5 (48.1 to 53.0)b | 49.0 (46.4 to 51.5) | 46.3 (43.5 to 49.0) |
| Positive emotion score (PANAS) | 36.8 (34.6 to 39.0) | 36.2 (33.9 to 38.5) | 36.6 (34.8 to 38.4) | 36.3 (34.3 to 38.3) | 35.3 (33.0 to 37.5) | 34.9 (32.7 to 37.0) |
| Optimism (LOT) score | 29.0 (27.9 to 30.1) | 28.5 (27.3 to 29.6) | 27.9 (26.7 to 29.0) | 28.0 (26.6 to 29.3) | 28.6 (27.4 to 29.7) | 29.0 (27.9 to 30.1) |
| Social support score (Ryff PR) | 43.4 (41.1 to 45.7) | 43.6 (41.3 to 45.9) | 42.4 (39.8 to 44.9) | 44.5 (42.4 to 46.5) | 44.0 (42.1 to 45.9) | 44.0 (42.1 to 46.0) |
| Sleep quality score (PSQI) | 4.61 (3.49 to 5.73) | 4.54 (3.47 to 5.61) | 5.02 (4.06 to 5.98) | 5.02 (4.20 to 5.84) | 5.22 (4.33 to 6.12) | 5.10 (4.22 to 5.99) |
| Indicators of poor health (negative change indicates improvement) | ||||||
| Perceived stress score (PSS-10) | 9.5 (7.8 to 11.2) | 10.0 (8.2 to 11.7) | 11.2 (9.7 to 12.8) | 11.4 (9.5 to 13.4) | 10.5 (8.6 to 12.3) | 11.4 (9.5 to 13.2) |
| Negative emotion score (PANAS) | 14.0 (12.8 to 15.2) | 14.4 (13.1 to 15.7) | 15.0 (13.7 to 16.2) | 15.0 (13.3 to 16.7) | 14.6 (13.3 to 15.9) | 14.9 (13.8 to 16.0) |
| Anxiety (STAI) score | 30.2 (27.6 to 32.8) | 29.1 (26.6 to 31.7) | 30.7 (28.0 to 33.4) | 29.7 (26.9 to 32.5) | 31.2 (28.4 to 33.9) | 30.4 (27.9 to 32.9) |
IPAQ = International Physical Activity Questionnaire; LOT = Life Orientation Test; MAAS = Mindful Attention Awareness Scale; MET = metabolic equivalent task; Ryff PR = Ryff’s Positive Relationships with Other scale; PANAS = Positive and Negative Affect Schedule; PSQI = Pittsburgh Sleep Quality Index; PSS-10 = 10-item Perceived Stress Scale; SF-12 = 12-item Medical Outcomes Study Short Form; STAI = State Trait Anxiety Inventory.
Notes: Data shown represent mean at 9 weeks (1 week after 8-week intervention), then approximately 3 months later. Two-sided P values calculated by simple 2-sample contrast using t test, SAS software. Statistical significance for secondary outcomes should be interpreted as exploratory and hypothesis-generating rather than confirmatory and hypothesis-testing.
P <.01 for comparison of intervention means vs control means at each period.
P <.05 for comparison of intervention means vs control means at each period.
DISCUSSION
We observed substantive reductions in ARI illness among those randomized to exercise training, and even greater benefits among those receiving mindfulness meditation training. Incidence, duration, and global severity of ARI illness were 29%, 43%, and 31% lower in the exercise group and 33%, 43%, and 60% lower in the mindfulness group, respectfully, compared with control. Although not all of these observed benefits were statistically significant, the magnitude of the observed reductions in ARI illness is surely clinically significant. That our findings are corroborated both by laboratory-measured biomarkers and by reductions in work absenteeism further supports a positive interpretation. Implications for the workplace may be especially important. Compared with the control group, all-cause absenteeism was 31% lower in both intervention groups. Looking at ARI-related absenteeism, there were 48% fewer days missed in the exercise group, and 76% fewer in the meditation group. For meditation especially, these findings are unlikely be due to chance. Given that apart from hand washing, no ARI prevention strategies have before been proven, these findings are especially noteworthy.
This clinical trial has both strengths and limitations. In terms of strengths, this randomized trial is the first to assess effects of mindfulness meditation on ARI illness and the first to use a validated outcome measure to assess effects of exercise on ARI illness. It is also the first to compare 2 behavioral interventions with a valid control condition, allowing head-to-head comparative effectiveness assessment. The use of multiplex PCR to identify viruses and of IL-8 and neutrophil assays to assess inflammation is also a strength. Limitations include the inescapable fact that participants in such a trial cannot be blinded to behavioral training interventions, thus allowing for the possibility of self-report bias. Also, even with the large effect sizes observed, our sample size was only marginal for statistical significance for several outcomes. Although it is not likely that our findings are entirely due to chance, the a priori selection of P ≤.025 as a cutoff for null hypothesis rejection does leave that statistical possibility open for some outcomes. Finally, interpretation of results is limited in that the first cohort was followed for a full cold and influenza season, whereas the second was followed for fewer months during the winter-spring ARI season.
The data do not suggest generalized self-report bias. If participants in the control group were overreporting ARI illness, we would expect them to also report a similar magnitude of improved scores on other self-report health measures, which they did not do. Similar thinking applies to the biomarkers. If participants in the intervention groups were under-reporting ARI illness, one might expect that the episodes they did report would be relatively more severe, with higher levels of inflammatory markers and higher viral identification rates. These trends were not seen, except for marginally higher IL-8 levels in the meditation group.
We were surprised to find that, apart from marginally reduced stress and increased optimism, the interventions did not appear to have much influence on self-reported psychosocial health measures. One partial explanation may be that this was a relatively healthy population. National norms for similarly aged adults using the SF-12 are 46.3 for physical health and 50.1 for mental health.44 At baseline, participants in our study had SF-12 scores of 50.5 and 51.4, respectively. Our participants also reported less stress than is usual, with mean PSS-10 scores of 11.9, well below national averages of 13.4 reported in 2006 and 14.5 in 2009.45 Interpreting these findings, one might hypothesize that sicker and more stressed people would have more to gain, and that similar interventions might yield greater benefits in those populations. That hypothesis deserves further testing.
In conclusion, this ground-breaking randomized trial of meditation and exercise vs wait-list control among adults aged 50 years and older found significant reductions in ARI illness. Corresponding reductions in time lost to work point toward socioeconomic as well as personal health benefits. If these results are confirmed in future studies, there will be important implications for public and private health-related policy and practice, as well as for scientific research regarding mechanisms of health maintenance and disease prevention.
Acknowledgments
The authors would also like to thank the University of Wisconsin, School of Medicine and Public Health, and especially the UW Department of Family Medicine, for providing an institutional base and collegial support. They would also like to thank the UW Health Mindfulness Program and the UW Exercise Physiology staff at the UW Health Sports Medicine Center for conducting the meditation and exercise interventions.
Footnotes
Conflicts of interest: Bruce Barrett was previously supported by career development grants from the National Center for Complementary and Alternative Medicine (K23 AT00051) and from the Robert Wood Johnson Foundation, both of which were essential for developing the methodology and infrastructure for recruiting and monitoring research subjects, and for assessing their acute respiratory infections.
Funding support: This study was supported by a grant from the National Institutes of Health (NIH), National Center for Complementary and Alternative Medicine (1R01AT004313); and by a grant UL1RR025011 from the Clinical and Translational Science Award (CTSA) Program of the National Center for Research Resources, National Institutes of Health. Aleksandra Zgierska is supported by grant K23 AA017508 from National Institute on Alcohol Abuse and Alcoholism at NIH.
Disclaimer: Beyond normal scientific review process, the National Center for Complementary and Alternative Medicine did not contribute to design or conduct of the study, data collection, management, analysis, or interpretation of the data, or preparation, review, or approval of the manuscript.
References
- 1.Fiore AE, Uyeki TM, Broder K, et al. Centers for Disease Control and Prevention (CDC) Prevention and control of influenza with vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP). 2010. MMWR Recomm Rep. 2010;59(RR-8):1–62 [PubMed] [Google Scholar]
- 2.Ebell MH, Alfonso A. A systematic review of clinical prediction rules for the diagnosis of influenza. Ann Fam Med. 2011;9(1):61–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Eccles R. Understanding the symptoms of the common cold and influenza. Lancet Infect Dis. 2005;5(11):718–725 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fendrick AM, Monto AS, Nightengale B, Sarnes M. The economic burden of non-influenza-related viral respiratory tract infection in the United States. Arch Intern Med. 2003;163(4):487–494 [DOI] [PubMed] [Google Scholar]
- 5.Jefferson T, Demicheli V, Rivetti D, Jones M, Di Pietrantonj C, Rivetti A. Antivirals for influenza in healthy adults: systematic review. Lancet. 2006;367(9507):303–313 [DOI] [PubMed] [Google Scholar]
- 6.Centers for Disease Control and Prevention (CDC) Advisory Committee on Immunization Practices. Prevention and control of influenza. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2006;55(1):1–41 [PubMed] [Google Scholar]
- 7.Voordouw AC, Sturkenboom MC, Dieleman JP, et al. Annual revaccination against influenza and mortality risk in community-dwelling elderly persons. JAMA. 2004;292(17):2089–2095 [DOI] [PubMed] [Google Scholar]
- 8.Cohen S, Tyrrell DA, Russell MAH, Jarvis MJ, Smith AP. Smoking, alcohol consumption, and susceptibility to the common cold. Am J Public Health. 1993;83(9):1277–1283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Carter JM. Hand washing decreases risk of colds and flu. J Natl Med Assoc. 2002;94(2):A11. [PMC free article] [PubMed] [Google Scholar]
- 10.Cohen S, Tyrrell DA, Smith AP. Psychological stress and susceptibility to the common cold. N Engl J Med. 1991;325(9):606–612 [DOI] [PubMed] [Google Scholar]
- 11.Doyle WJ, Gentile DA, Cohen S. Emotional style, nasal cytokines, and illness expression after experimental rhinovirus exposure. Brain Behav Immun. 2006;20(2):175–181 [DOI] [PubMed] [Google Scholar]
- 12.Cohen S, Doyle WJ, Skoner DP, Rabin BS, Gwaltney JM., Jr Social ties and susceptibility to the common cold. JAMA. 1997;277(24): 1940–1944 [PubMed] [Google Scholar]
- 13.Carmody J, Baer RA. Relationships between mindfulness practice and levels of mindfulness, medical and psychological symptoms and well-being in a mindfulness-based stress reduction program. J Behav Med. 2008;31(1):23–33 [DOI] [PubMed] [Google Scholar]
- 14.Mars TS, Abbey H. Mindfulness meditation practise as a healthcare intervention: A systematic review. Int J Osteopath Med. 2010;13(2): 56–66 [Google Scholar]
- 15.Chubak J, McTiernan A, Sorensen B, et al. Moderate-intensity exercise reduces the incidence of colds among postmenopausal women. Am J Med. 2006;119(11):937–942 [DOI] [PubMed] [Google Scholar]
- 16.Nieman DC. Exercise, upper respiratory tract infection, and the immune system. Med Sci Sports Exerc. 1994;26(2):128–139 [DOI] [PubMed] [Google Scholar]
- 17.Nieman DC, Henson DA, Austin MD, Sha W. Upper respiratory tract infection is reduced in physically fit and active adults. Br J Sports Med. 2010;45(12):987–992 [DOI] [PubMed] [Google Scholar]
- 18.Kabat-Zinn J. Mindfullness-based interventions in context: Past, present, future. Clin Psychol Sci Pract. 2003;10:144–156 [Google Scholar]
- 19.Borg GV, Linderholm H. Percieved exertion and pulse rate during graded exercise in various age groups. Acta Med Scand. 1974;472: 194–206 [Google Scholar]
- 20.Mohr DC, Spring B, Freedland KE, et al. The selection and design of control conditions for randomized controlled trials of psychological interventions. Psychother Psychosom. 2009;78(5):275–284 [DOI] [PubMed] [Google Scholar]
- 21.Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–198 [DOI] [PubMed] [Google Scholar]
- 22.Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fletcher GF, Balady GJ, Amsterdam EA, et al. Exercise standards for testing and training: a statement for healthcare professionals from the American Heart Association. Circulation. 2001;104(14):1694–1740 [DOI] [PubMed] [Google Scholar]
- 24.Barrett B, Brown R, Mundt M, et al. The Wisconsin Upper Respiratory Symptom Survey is responsive, reliable, and valid. J Clin Epidemiol. 2005;58(6):609–617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Barrett B, Brown R, Voland R, Maberry R, Turner R. Relations among questionnaire and laboratory measures of rhinovirus infection. Eur Respir J. 2006;28(2):358–363 [DOI] [PubMed] [Google Scholar]
- 26.Barrett B, Brown R, Mundt M. Comparison of anchor-based and distributional approaches in estimating important difference in common cold. Qual Life Res. 2008;17(1):75–85 [DOI] [PubMed] [Google Scholar]
- 27.Barrett B, Brown RE, Mundt MP, et al. Validation of a short form Wisconsin Upper Respiratory Symptom Survey (WURSS-21). Health Qual Life Outcomes. 2009;7(1):76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jackson GG, Dowling HF, Muldoon RL. Acute respiratory diseases of viral etiology. VII. Present concepts of the common cold. Am J Public Health Nations Health. 1962;52:940–945 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Turner RB, Weingand KW, Yeh CH, Leedy DW. Association between interleukin-8 concentration in nasal secretions and severity of symptoms of experimental rhinovirus colds. Clin Infect Dis. 1998;26(4): 840–846 [DOI] [PubMed] [Google Scholar]
- 30.Teran LM, Johnston SL, Schröder JM, Church MK, Holgate ST. Role of nasal interleukin-8 in neutrophil recruitment and activation in children with virus-induced asthma. Am J Respir Crit Care Med. 1997; 155(4):1362–1366 [DOI] [PubMed] [Google Scholar]
- 31.Lee WM, Grindle K, Pappas T, et al. High-throughput, sensitive, and accurate multiplex PCR-microsphere flow cytometry system for large-scale comprehensive detection of respiratory viruses. J Clin Microbiol. 2007;45(8):2626–2634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cheak-Zamora NC, Wyrwich KW, McBride TD. Reliability and validity of the SF-12v2 in the medical expenditure panel survey. Qual Life Res. 2009;18(6):727–735 [DOI] [PubMed] [Google Scholar]
- 33.Watson D, Clark LA, Tellegen A. Development and validation of brief measures of positive and negative affect: the PANAS scales. J Pers Soc Psychol. 1988;54(6):1063–1070 [DOI] [PubMed] [Google Scholar]
- 34.Hodgues WF, Spielberger CD. An indicant of trait or state anxiety? J Consult Clin Psychol. 1969;33(4):430–434 [DOI] [PubMed] [Google Scholar]
- 35.Scheier MF, Carver CS, Bridges MW. Distinguishing optimism from neuroticism (and trait anxiety, self-mastery, and self-esteem): A reevaluation of the Life Orientation Test. J Pers Soc Psychol. 1994; 67(6):1063–1078 [DOI] [PubMed] [Google Scholar]
- 36.Ryff CD, Singer B. Psychological well-being: meaning, measurement, and implications for psychotherapy research. Psychother Psychosom. 1996;65(1):14–23 [DOI] [PubMed] [Google Scholar]
- 37.Backhaus J, Junghanns K, Broocks A, Riemann D, Hohagen F. Test-retest reliability and validity of the Pittsburgh Sleep Quality Index in primary insomnia. J Psychosom Res. 2002;53(3):737–740 [DOI] [PubMed] [Google Scholar]
- 38.Washburn RA, Montoye HJ. The assessment of physical activity by questionnaire. Am J Epidemiol. 1986;123(4):563–576 [DOI] [PubMed] [Google Scholar]
- 39.Carlson LE, Brown KW. Validation of the Mindful Attention Awareness Scale in a cancer population. J Psychosom Res. 2005;58(1):29–33 [DOI] [PubMed] [Google Scholar]
- 40.Schafer JL. Analysis of Incomplete Multivariate Data. London: Chapman & Hall, 1997 [Google Scholar]
- 41.Baron RM, Kenny DA. The moderator-mediator variable distinction in social psychological research: conceptual, strategic, and statistical considerations. J Pers Soc Psychol. 1986;51(6):1173–1182 [DOI] [PubMed] [Google Scholar]
- 42.Krull JL, MacKinnon DP. Multilevel modeling of individual and group level mediated effects. Multivariate Behav Res. 2001;36(2):249–277 [DOI] [PubMed] [Google Scholar]
- 43.Brown RL. Assessing specific mediational effects in complex theoretical models. Struct Equ Modeling. 1997;4(2):142–156 [Google Scholar]
- 44.Ware JE, Kosinski M, Turner-Bowker DM, Gandek B. User’s Manual for the SF-12v2 Health Survey. Boston, MA: QualityMetric; 2008 [Google Scholar]
- 45.Cohen S, Janicki-Deverts D. Who’s stressed? Distributions of psychological stress in the United States in probability samples from 1983, 2006 and 2009. J Appl Psychol. In press. [Google Scholar]