Abstract
Baclofen, a GABAB receptor agonist, is currently under investigation as a potential treatment to prevent relapse to drinking in alcohol dependent persons. In the current study, two groups of baboons were trained under a chained schedule of reinforcement (CSR), with 3 linked components, which were each correlated with different response requirements and cues. Fulfilling the requirement in the 2nd link initiated the 3rd link where either alcohol (n=4) or a preferred non-alcoholic beverage (Tang, n=5) was available for self-administration; failure to complete the response requirement in Link 2 ended the session (no access to alcohol or Tang). Seeking responses in Link 2 were used as indices of the motivational processes thought to be involved in relapse. The effects of baclofen (0.1 – 2.4 mg/kg) were examined under conditions with alcohol or Tang access and under extinction. Under the CSR baclofen (1.8 and 2.4 mg/kg) significantly decreased (p<0.05) alcohol self-administration responses and total g/kg alcohol intake. In contrast, only the highest dose of baclofen (2.4 mg/kg) reduced Tang self-administration and consumption. Under within-session extinction conditions, baclofen (1.8 and 2.4 mg/kg) facilitated extinction of responding for both alcohol and Tang, particularly during the first 10 min of extinction. Baclofen may be effective in reducing craving and alcohol drinking, although the facilitation of extinction and suppression of both alcohol and Tang self-administration by baclofen suggests these effects may be related to a more general suppression of consummatory and conditioned behaviors.
Keywords: alcohol, baclofen, drug-seeking, extinction, nonhuman primate, reinforcement
INTRODUCTION
It has been estimated that 30% of American adults meet DSM-IV criteria for lifetime alcohol use disorder (Hasin et al., 2007). Currently, three medications, disulfiram, naltrexone, and acamprosate, which have been shown to be effective in reducing alcohol craving, relapse and heavy drinking, are approved to treat alcohol dependence (Soyka and Rosner, 2010). However, since these medications exhibit a small to moderate treatment efficacy, with variable individual response, development of new medications is critical.
In contrast to other drugs of abuse, alcohol has no specific target or receptor in the brain, but exerts non-specific widespread effects on multiple systems. Gamma-aminobutyric acid (GABA) is the major inhibitory neurotransmitter in the brain. It has been hypothesized that the rewarding effects of alcohol may be partly due to an increased activation of the mesolimbic dopamine pathway (Swift, 2010). Alcohol increases the release of endogenous opioids, which inhibit GABAergic neurons (Doyon et al., 2006). Inhibition of GABAergic neurons increases the extracellular levels of dopamine in the ventral tegmental area (VTA) and the nucleus accumbens (NAc), and such dopamine release is implicated in the reinforcing effects of alcohol (Heilig and Egli, 2006; Engleman et al., 2011). GABAB receptors are co-localized with dopamine receptors in mesolimbic regions and directly and indirectly modulate dopaminergic neurons. Baclofen, a GABAB receptor agonist, increases GABAergic neurotransmission and attenuates alcohol-induced dopamine release in the VTA (Theile et al., 2008). Given that baclofen may compensate for alcohol-induced alterations in dopamine transmission (Theile et al., 2008), is safe and well tolerated in heavy drinkers (Evans and Bisaga, 2009), and has a low abuse liability profile (Griffiths et al., 1991), it is a good candidate pharmacotherapy for alcohol abuse and dependence.
The preclinical data examining baclofen effects on alcohol drinking have been conducted exclusively in rodent drinking models. When alcohol was freely available, baclofen reduced alcohol intake (Daoust et al., 1987; Colombo et al., 2000; Czachowski et al., 2006) and blocked the acquisition of alcohol drinking in rats (Colombo et al., 2002; but cf. Smith et al., 1992). Baclofen reduced alcohol intake under operant alcohol self-administration procedures (Anstrom et al., 2003; Maccioni et al., 2005; 2008; Liang et al., 2006, Walker and Koob, 2007). Baclofen also attenuated the conditioned effects of alcohol as evidenced by decreased cue-induced reinstatement of responding previously maintained by alcohol seeking (Murschall and Hauber, 2006; Maccioni et al., 2008) and alcohol-induced conditioned place preference (Bechtholt and Cunningham, 2005).
More recently, baclofen has been tested in human laboratory studies and clinical trials (see Leggio et al., 2010). In a laboratory study assessing the effects of baclofen in combination with intoxicating doses of alcohol (>80 mg/dL breath alcohol concentrations), baclofen failed to attenuate behavioral effects (e.g., increased ratings of feeling high, drug liking, good effects) often associated with the reinforcing properties of alcohol (Evans and Bisaga, 2009). Baclofen has been shown to reduce alcohol craving and drinking levels in small, open-label trials in alcoholics (Addolorato et al., 2000; Flannery et al., 2004). However, three randomized double-blind placebo controlled laboratory studies have reported conflicting results. Addolorato and colleagues (2002) reported that 14 of 17 patients treated with 30 mg/day baclofen maintained alcohol abstinence for the duration of the 4-week study and reduced alcohol craving compared to 4 of 11 placebo treated volunteers. In alcohol-dependent patients with liver cirrhosis, baclofen increased the average number of days abstinent compared to placebo (62 and 30 days, respectively; Addolorato et al., 2007). Garbutt and colleagues (2010) reported that no differences were found between 30 mg/day baclofen and placebo treated volunteers (n=80) in percentage of days abstinent, time to first drink, or time to relapse. These authors concluded that given the inconsistency of the data, it is important to further investigate the effect of baclofen on alcohol drinking and relapse and to determine what factors are predictive of response.
The current study extends previous preclinical work by examining the efficacy of baclofen on alcohol-maintained behaviors and consumption in large nonhuman primates (baboons) that self-administer alcohol under a chained schedule of reinforcement (CSR) operant procedure. A CSR consists of a sequence of separate behavioral contingencies (i.e., schedule requirements), each of which is correlated with unique stimuli (‘‘cues’’) that represent links or “components” of a chain of responses that ultimately result in alcohol reinforcement. Fulfilling the schedule requirement in each successive component is necessary to progress to the next component with alcohol available only in the final component of the chain. The extent to which cues come to control behaviors directed towards obtaining alcohol (i.e., seeking) in the earlier links of the chain as well as behaviors related to alcohol intake (self-administration) can be used as indices of the incentive-motivational processes involved in the maintenance of drinking and relapse. Previously we have shown that alcohol-seeking responses under the CSR were maintained by alcohol cues, and seeking and alcohol intake were augmented by alcohol abstinence (Weerts et al., 2006; Kaminski et al., 2008). Examination of baclofen effects on alcohol seeking and self-administration using the CSR allowed us to examine how baclofen altered each of these behaviors and the relationship between seeking and consumption within a single experimental session. In addition, the non-specific effects of baclofen were examined in a second group of baboons that self-administered a preferred non-alcoholic beverage (Tang) under the CSR.
While the efficacy of baclofen in reducing alcohol intake in rodent models has been characterized (see Leggio et al., 2010 for review), there are no data in nonhuman primates. Nonhuman primates are closely related to humans in terms of phylogeny, physiology, neurochemistry, and behavior. In addition, alcohol absorption and metabolism in nonhuman primates is similar to what is observed in humans. The addition of baclofen data in nonhuman primates bridges the translational research gap between rodents and humans. Moreover, the observation of similar effects across multiple species provides greater confidence in the generalization of knowledge from animal models to the human condition.
METHODS AND MATERIALS
Subjects
Nine adult male baboons (Papio anubis; Southwest Foundation for Biomedical Research, San Antonio, TX) weighting 27.8 kg (± 5.3 SD) served as subjects. Baboons were housed singly in customized baboon cages (see below) in two rooms that were maintained under a 12-hour light/dark cycle (lights on at 6:00 AM). Windows in the laboratory provided natural light. All baboons had extensive histories of self-administration under the chained schedule of reinforcement (CSR), but the reinforcer delivered was 4% alcohol w/v for one group (N=4) and 25% or 50% orange-flavored sugar-free Tang for the second group (N=5; Weerts et al., 2006, Kaminski et al., 2008, Weerts et al., submitted). The baboons assigned to the alcohol group only received alcohol and the baboons assigned to the Tang group only received Tang throughout the entire study. Baboons received standard primate chow (e.g., 50–73 kcals/kg) to maintain sufficient caloric intake for normal baboons of their size, age, and weight. They also received daily food supplements [2 pieces of fresh fruit or vegetables (70–120g each) and a children’s chewable multivitamin]. Using these procedures, baboons maintain relatively stable weights but do slowly gain weight as they age. Drinking water was available ad libitum except during experimental sessions. The facilities were maintained in accordance with the USDA and AAALAC standards. The protocol was approved by the JHU Animal Care and Use Committee and followed the Guide for the Care and Use of Laboratory Animals (1996).
Apparatus
During the experiments, baboons were housed in modified standard primate cages as described in detail previously (Weerts et al., 2006, Kaminski et al., 2008). Briefly, the intelligence panel contained 2 vertically operated levers (Med Associates, Georgia, VT), 2 different colored “jewel” lights mounted above each lever, and a “drinkometer” (Kandota Instruments, Sauk Center, MN) with 2 white and 2 green lights which surrounded a protruding drink spout. Contact with the drink spout operated a solenoid valve that delivered fluid for up to 5 seconds (i.e., 1 drink ~30 mL). All solutions were delivered from a calibrated 1000-ml bottle connected to the drinkometer with tygon tubing. A separate panel on the back of the cage contained three colored “cue” lights (red, yellow and blue). A speaker was mounted above the cages for presentation of auditory stimuli (e.g., tones). Experimental conditions and data collection were controlled remotely using computers interfaced with MED Associates hardware and software.
Drugs
All solutions for oral consumption were mixed using reverse osmosis (RO) purified drinking water. Ethyl alcohol (190 Proof, Pharmco-AAPER, Brookville CT) was diluted with RO water to 4% w/v alcohol. Orange-flavored, sugar-free, Tang powder (Kraft Foods) was dissolved in RO water following package instructions and then diluted from full strength to concentrations of 25% or 50%. Baclofen (Sigma-Aldrich) was dissolved in physiological saline. Baclofen doses (0.1–2.4 mg/kg) or vehicle were administered via intramuscular (IM) route (2 ml volume/injection).
Chained Schedule of Reinforcement (CSR) Procedure
The drinking water spout was disabled immediately before the session. Fluids were available only via the drinkometer for the duration of the session, then the drinking water spout was reactivated and water was again freely available for the rest of the day. The CSR procedures used in the current study were the same as those described by Kaminski and colleagues (2008) and were identical for the alcohol and Tang groups. The CSR consisted of 3 separate sequential components, each of which was associated with distinct stimuli and behavioral contingencies. Fulfilling the schedule requirement(s) in each component was necessary to proceed to the next. Briefly, daily sessions for all baboons were initiated at the same time (0830 hr) and were signaled by a 3-sec tone. Component 1 began with the illumination of a red cue light for 20-min. All instrumental responses (i.e., lever presses and drinkometer spout contacts) were recorded but had no consequence. At the onset of Component 2 the yellow cue light was illuminated and a 2-link chained fixed interval (FI 10-min) fixed ratio (FR 10) schedule was in effect. During the first link, the yellow jewel light over the left lever was continuously illuminated, and a concurrent fixed interval 10 min, fixed time 20 min (FI 10-min FT 20-min) schedule was in effect. The second link of Component 2 was initiated after either a) the first response on the left lever after 10 min elapsed or b) 20 min, whichever occurred first. In the second link of Component 2, the jewel light over the lever flashed and a fixed ratio (FR 10) schedule was in effect on the left lever. Completion of the FR10 response requirement ended Component 2; the yellow cue light and the yellow jewel light over the left lever were turned off and Component 3 was initiated. If the FR10 requirement was not completed the session terminated without transitioning to Component 3 (i.e., no alcohol or no Tang access for the day). Component 3 began with the illumination of the blue cue light. During Component 3, a blue jewel light over the right lever was illuminated, and the opportunity to orally self-administer alcohol or Tang was available under a fixed ratio (FR10) schedule on the right lever. Completion of the response requirement on the right lever turned off the jewel light and illuminated the white stimulus lights on the drinkometer faceplate indicating the availability of a ‘drink.’ A drink was operationally defined as fluid delivery that was contingent on contact with the drinkometer spout; contact with the drinkometer spout turned off the white lights, illuminated the green lights on the drinkometer faceplate, and fluid was delivered for the duration of spout contact or for a programmed duration (5 sec), whichever came first. After 120 min, Component 3 ended and all programmed stimuli (e.g., the blue cue light, jewel light) were terminated. The CSR sessions were conducted daily (7d/week) for both alcohol and Tang groups.
Before baclofen tests were initiated, the maximum amount of work to gain access to alcohol or Tang was determined using a progressive ratio procedure during Component 2, Link 2 of the CSR as described previously (Kaminski et al., 2008). Specifically, breaking points (BP) to obtain alcohol or Tang were determined in the alcohol and Tang groups, respectively. The concentration of Tang was titrated so that the BPs for Tang in the Tang group (mean ± SEM: 665.6 ± 91.9) were similar to BPs obtained with 4% w/v alcohol (mean ± SEM: 636 ± 171.6) in the Alcohol group of baboons (i.e., functioned as equivalent reinforcers). The concentration of Tang maintained for all baclofen tests was 50% Tang for three baboons and 25% Tang for the other two baboons.
Baclofen Test Procedures
Acute doses of baclofen (0.1–2.4 mg/kg) or vehicle were administered 30 min before CSR tests and 60 min prior to within-session extinction tests to target peak baclofen effects during the different components of the CSR. The two pretreatment times allowed evaluation of Component 2 (seeking) and Component 3 (self-administration) responding with a similar dosing time frame [i.e., 80 min before the onset of Component 2 (60 min pretreatment) and onset of Component 3 (30 min pretreatment)]. Doses and pretreatment times were based on our previous studies with baclofen (Weerts et al., 2005; 2007). Before baclofen tests were conducted, the CSR baseline criterion for alcohol or Tang was met. The CSR baseline criterion was defined as stable self-administration of alcohol or Tang (i.e., ± 20%) for three consecutive CSR sessions. The CSR was re-established and CSR baseline criterion met before the next dose of baclofen or its vehicle was administered.
Within-Session Extinction Procedure
A within-session extinction procedure was used to examine the extinction of seeking behavior within a single daily session. The stimuli and behavioral contingencies in effect during the within-session extinction procedure were the same as the baseline CSR sessions for Component 1 and for Link 1 of Component 2. During Link 2 of within-session extinction, the FR10 schedule was not in effect and responses did not initiate Component 3; responses were recorded until no left lever responses occurred for 30 consecutive minutes (criterion for extinction), and then the session terminated (i.e., no access to alcohol or Tang for the day). Following each within-session extinction test, the CSR was reinstated and baseline criterion was met. Next, a modified progressive ratio schedule, based on the PR procedures described by Kaminski and colleagues (2008), was implemented, in which the starting ratio was 160 and progressively increased by a factor of 2 until a BP was reached. The purpose of the abbreviated PR sessions was to reduce habituation to repeated extinction testing (Bullock 1960, Bullock and Smith 1953, Clark and Taylor 1960, Zarcone and Ator 2000), and thus maintain a more stable extinction baseline for testing baclofen. Baclofen testing was not done in the context of the abbreviated PR. After a BP was reached under the abbreviated progressive ratio procedure, the CSR baseline was established. The next within-session extinction test with baclofen was only conducted after the CSR baseline criterion was again met. That is, the sequence of conditions was 1) criterion performance under the CSR baseline, 2) abbreviated progressive ratio until BP, 3) criterion performance under CSR baseline, and 4) within-session extinction test. Conditions 1–4 were repeated until all baclofen doses plus vehicle were tested.
Data Analysis
For the CSR, variables of interest were left and right lever presses, drink contacts for Components 2 and 3, as well as the latency to complete the FI requirement in Component 2 and total volume (mL) of fluid (water, alcohol, or Tang) consumed in Component 3. In addition, total g/kg of alcohol was calculated based on individual body weights and total volume of alcohol consumed. For the within-session extinction tests, the primary variables of interest were number of seeking responses on the left lever and time (min) to reach extinction criterion. For each baboon, the mean of the 3 days that preceded each test condition was used as the baseline for comparison with administration of vehicle and doses of baclofen.
Data were analyzed (SigmaPlot 11.0 or Prism 5.0) with separate statistical analysis of variance (ANOVA) for each group (alcohol or Tang) with baclofen dose (0–2.4 mg/kg) as a repeated measure. The 1.8 mg/kg baclofen dose was tested twice in each alcohol-trained animal and the average was used in all analyses. Bonferroni tests were used for pairwise comparisons (i.e., doses vs. vehicle). Patterning of drinks under the CSR and seeking responses during within-session extinction tests were analyzed using a two-way repeated measures ANOVA (Time × Dose) for each group. Pairwise comparisons using Bonferroni tests examined statistical differences between vehicle and doses of baclofen. The alpha level was set at p < 0.05 for all analyses. Due to adverse effects of baclofen observed at 1.8 mg/kg baclofen, one baboon in the Tang group and one baboon in the alcohol group did not receive the 2.4 mg/kg dose (also see results), and data were treated as missing values in the analysis.
RESULTS
Effects of Baclofen on Operant Responses and Intake of Alcohol and Tang Under the CSR
Group means (± SEM) and significant effects (F ratio, df, and Bonferroni pairwise comparisons) of baclofen on operant responses during Components 2 and 3 of the CSR for a) alcohol and b) Tang groups are shown in Table 1. Under the CSR, alcohol and Tang maintained similar levels of responding on the left lever during both Link 1 (FI schedule) and Link 2 (FR schedule) of Component 2. The minimum latency to complete the FI was 600 seconds and the mean (±SEM) latency to complete the FI schedule was 611 (±3.8) seconds for alcohol and 613 (±2.4) for Tang under baseline conditions. Under baseline and vehicle test conditions, baboons always completed the Link 2 FR10 schedule on the left lever with little or no responding on the inactive operanda (right lever and drinkometer) during Component 2 and gained access to alcohol or Tang in Component 3. Baseline self-administration responses [t(7)=8.1, p<0.0001] were significantly greater for Tang than for alcohol resulting in different intake volumes for alcohol and Tang in Component 3 [t(7)=8.6, p<0.0001].
Table 1.
Effects of baclofen on alcohol and tang seeking and self-administration under the CSR.
| A. Alcohol | Component 2 Link 1 (seeking) | Component 2 Link 2 (seeking) | Component 3 (self-administration) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Left Lever FI Resp | FI Resp Latency (s) | Left Lever FR Resp | Right Lever Resp | Drink contacts | Right Lever Resp | Drinks | Volume (ml) | Alcohol (g/kg) | |
| Baclofen (mg/kg) | |||||||||
| V | 93.3 (58.2) | 607.7 (3.0) | 10.0 (0.0) | 0.1 (0.1) | 0.0 (0.0) | 131.3 (12.5) | 15.3 (1.0) | 587.5 (44.3) | 1.0 (0.1) |
| 0.1 | 207.8 (136.9) | 609.4 (3.9) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 132.3 (26.0) | 14.8 (1.5) | 583.8 (74.5) | 1.0 (0.1) |
| 0.3 | 103.8 (54.9) | 607.5 (4.7) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 163.8 (31.1) | 18.8 (2.1) | 693.8 (44.0) | 1.2 (0.1) |
| 0.56 | 141.0 (54.9) | 611.3 (4.0) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 166.3 (27.6) | 18.8 (1.5) | 722.5 (81.9) | 1.2 (0.1) |
| 1.0 | 103.0 (37.2) | 607.1 (4.4) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 127.8 (13.6) | 15.3 (1.9) | 597.5 (78.6) | 1.1 (0.2) |
| 1.8 | 185.0 (93.8) | 611.1 (4.8) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 73.5 (7.8) | 8.5 (1.3) | 330.6 (38.1) | 0.6 (0.1) |
| 2.4 | 292.8 (149.6) | 614.1 (5.5) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 65.0 (19.4) | 8.3 (2.7) | 330.0 (97.7) | 0.6 (0.2) |
| F1 | 0.7 | 0.4 | nc | nc | nc | 6.5 | 6.7 | 8.3 | 8.8 |
| B. Tang | Component 2 Link 1 (seeking) | Component 2 Link 2 (seeking) | Component 3 (self-administration) | |||||
|---|---|---|---|---|---|---|---|---|
| Left Lever FI Resp | FI Resp Latency (s) | Left Lever FR Resp | Right Lever Resp | Drink contacts | Right Lever Resp | Drink Contacts | Volume (ml) | |
| Baclofen (mg/kg) | ||||||||
| V | 137.6 (32.0) | 628.3 (8.0) | 10.0 (0.0) | 0.1 (0.1) | 0.0 (0.0) | 370.6 (28.1) | 37.0 (2.8) | 973.3 (19.4) |
| 0.1 | 120.8 (71.1) | 608.7 (3.9) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 374.4 (17.9) | 37.2 (1.8) | 990.0 (10.0) |
| 0.3 | 35.0 (31.5) | 731.3 (117.2) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 414.6 (26.5) | 41.4 (2.6) | 990.0 (10.0) |
| 0.56 | 18.8 (14.5) | 616.1 (2.5) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 382.8 (32.6) | 38.2 (3.3) | 990.0 (10.0) |
| 1.0 | 210.4 (58.8) | 619.3 (8.9) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 417.8 (42.8) | 41.6 (4.2) | 990.0 (10.0) |
| 1.8 | 215.8 (111.5) | 662.5 (50.2) | 10.0 (0.0) | 0.0 (0.0) | 0.0 (0.0) | 371.2 (57.6) | 37.0 (5.7) | 905.0 (65.4) |
| 2.4 | 144.4 (126.1) | 729.5 (117.7) | 8.0 (2.0) | 0.0 (0.0) | 0.0 (0.0) | 298.5 (79.5) | 29.8 (8.0) | 743.8 (148.0) |
| F1 | 1.7 | 0.6 | nc | nc | nc | 2.6 | 2.2 | 3.2 |
Data shown are group means (±SEM) for vehicle (V) and each baclofen dose (mg/kg). FI, Fixed Interval; Resp, Response. Degrees of freedom were 6, 18 (Alcohol group) and 6, 23 (Tang group).
An underlined F ratio indicates a significant (p<0.05) ANOVA, nc=not calculated. Bolded numbers indicate a significant (p<0.05) Bonferroni pairwise comparison to vehicle.
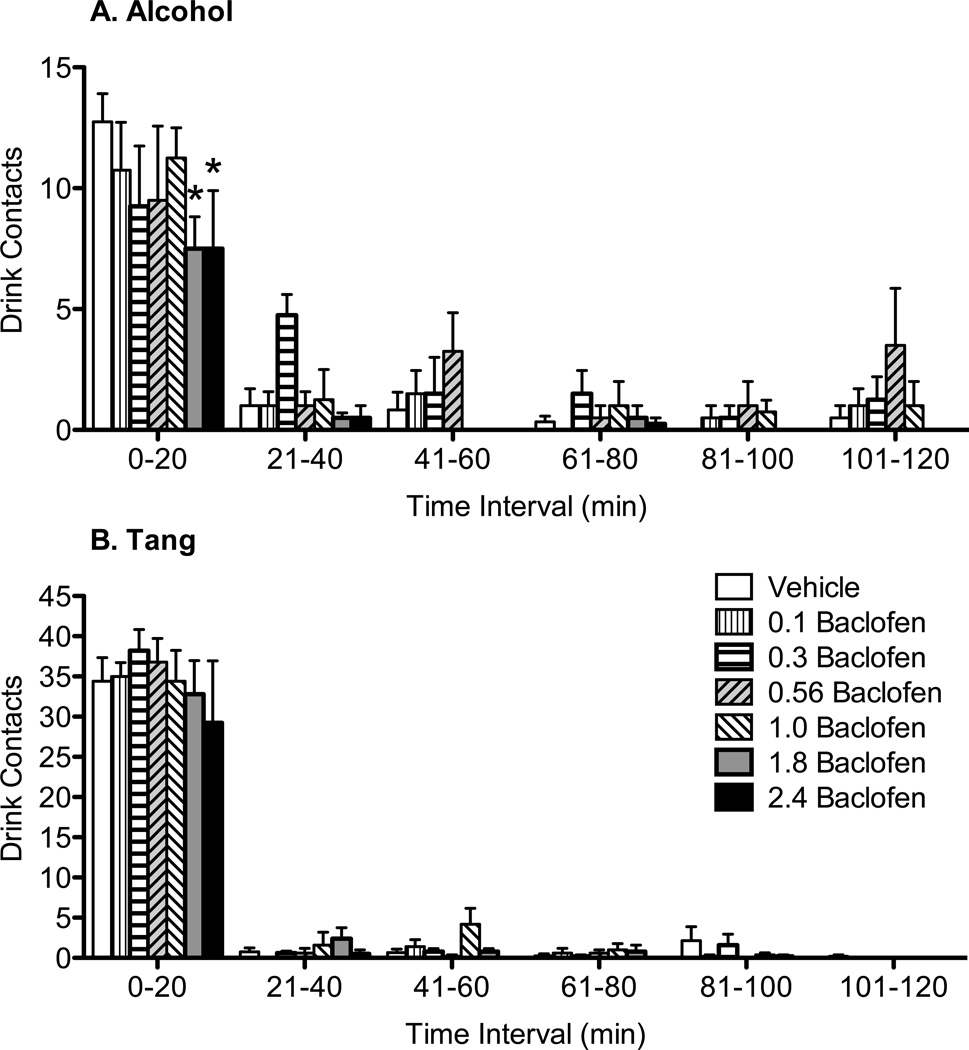
Baclofen did not significantly alter response latency or the number of left lever seeking responses during the FI Link1 in Component 2 directed towards obtaining access to alcohol (Table 1A) or Tang (Table 1B), although one baboon in the Tang group did not complete the FR10 in Link 2 of Component 2 following the 2.4 mg/kg dose of baclofen and thus did not transition to Component 3 (i.e., no Tang self-administration). Both reinforcers maintained self-administration responses (right lever responses and drink contacts) and high volumes of consumption in Component 3 of the CSR. Each completion of 10 right lever responses produced an opportunity to drink, and the number of drink contacts was directly proportional to the number of right lever responses (i.e., all drinks earned were consumed). As shown in Table 1, there were significant main effects of baclofen on the self-administration of a) alcohol and b) Tang, and these effects were dose-related. Bonferroni pairwise comparisons indicated that the number of drinks and total volume of alcohol intake (ml) were significantly decreased at 1.8 and 2.4 mg/kg baclofen for the alcohol group when compared to vehicle. Only the 2.4 mg/kg dose significantly decreased volume of Tang intake (ml) in the Tang group. Both groups showed an overall significant decrease in self-administration responses (right lever responses, Table 1). Bonferroni pairwise comparisons indicated significance at 2.4 mg/kg baclofen only in the alcohol group. The observed increase in alcohol intake and number of drink contacts at the intermediate doses of baclofen was not significant when compared to vehicle. As shown in Figure 1, at least 80% of drink contacts occurred in the first 20 minutes of alcohol or Tang availability; then a low number of drinks was distributed over the remainder of the 120-min drinking period. Baclofen did not alter this general pattern of drinking behavior. Instead, doses of baclofen (1.8–2.4 mg/kg) significantly decreased the number of drink contacts during the initial drinking bout (i.e., first 20 min of alcohol self-administration, Figure 1A). Only the highest dose of baclofen (2.4 mg/kg) significantly decreased drink contacts during Tang self-administration, and this effect also occurred in the first drinking bout (Figure 1B). During baseline conditions, baboons consumed a mean of 1.0 g/kg (± 0.1 SEM) of alcohol under the CSR. Consistent with the decrease in alcohol volume consumed (Table 1), doses of 1.8 and 2.4 mg/kg baclofen produced a significant decrease in total g/kg alcohol intake. When compared to baseline intake, baclofen doses of 1.8 and 2.4 m/kg reduced alcohol intake by an average of 0.4 g/kg.
Figure 1.
The effects of baclofen on the number of drink contacts (Component 3) during alcohol (A) and Tang (B) self-administration. Data shown are mean drink contacts (+ SEM) recorded in 20 min bins during the 120-min period of alcohol or Tang availability (Component 3). *p<0.05 pairwise comparison for each dose vs. vehicle within each time bin.
Effects of Baclofen on Within-Session Extinction
During within-session extinction tests, the same stimuli were presented as during the CSR, but lever presses during Link 2 of Component 2 were only counted and had no consequence. In both groups, responses on the right lever and drinkometer in Component 2 emerged during within-session extinction tests (Table 2), even though responses on these operandum were previously never reinforced under the CSR. As shown in Table 1, responses on the right lever and drinkometer (Component 2 Link 2) were virtually non-existent under baseline conditions for the CSR. Total seeking responses (sum of left lever and right lever responses, and drink contacts) are thus also shown in Table 2. One animal in the alcohol group and one animal in the Tang group did not receive the 2.4 mg/kg baclofen dose due to adverse effects at the 1.8 mg/kg dose; adverse effects included marked suppressed food intake, retching and vomiting, and lying on the cage floor.
Table 2.
Effects of baclofen on within-session extinction of alcohol and tang seeking responses in Component 2.
| A. Alcohol | Component 2 Link 1 (seeking) | Component 2 Link 2 (seeking) | |||||
|---|---|---|---|---|---|---|---|
| Left Lever FI Resp | FI Resp Latency (s) | Left Lever FR Resp | Right Lever Resp | Drink contacts | Total Resp | Time to Extinction2 | |
| Baclofen (mg/kg) | |||||||
| V | 373.9 (127.9) | 604.2 (2.5) | 2397.0 (755.0) | 169.8 (87.8) | 1.8 (0.4) | 2568.5 (834.4) | 121.7 (46.2) |
| 0.3 | 527.8 (244.2) | 601.6 (1.5) | 1928.3 (853.2) | 102.5 (67.3) | 12.5 (10.6) | 2043.3 (910.5) | 101.1 (34.6) |
| 0.56 | 411.5 (135.6) | 600.5 (0.1) | 2527.8 (961.3) | 251.8 (182.5) | 1.5 (1.2) | 2781.0 (1138.7) | 113.7 (25.7) |
| 1.0 | 215.8 (78.6) | 607.1 (3.6) | 2563.0 (1118.0) | 135.5 (60.2) | 1.0 (0.7) | 2699.5 (1167.1) | 127.2 (42.1) |
| 1.8 | 121.3 (67.4) | 661.6 (53.5) | 633.5 (303.5) | 53.3 (47.6) | 0.3 (0.3) | 687.0 (350.6) | 62.3 (10.2) |
| 2.4 | 221.7 (199.2) | 610.0 (69.2) | 625.3 (198.8) | 25.0 (19.3) | 0.0 (0.0) | 650.3 (142.7) | 61.0 (14.9) |
| F1 | 2.1 | 1.1 | 3.7 | 1.8 | 1.2 | 3.7 | 2.4 |
| B. Tang | Component 2 Link 1 (seeking) | Component 2 Link 2 (seeking) | |||||
|---|---|---|---|---|---|---|---|
| Left Lever FI Resp | FI Resp Latency (s) | Left Lever FR Resp | Right Lever Resp | Drink contacts | Total Resp | Time to Extinction2 | |
| Baclofen (mg/kg) | |||||||
| V | 176.2 (30.2) | 639.9 (12.3) | 558.0 (84.2) | 29.0 (19.6) | 5.1 (3.0) | 595.1 (101.0) | 85.4 (6.3) |
| 0.3 | 90.2 (30.9) | 618.7 (6.1) | 746.2 (203.8) | 20.8 (14.4) | 2.8 (1.7) | 769.8 (203.0) | 82.6 (18.1) |
| 0.56 | 201.6 (50.9) | 605.3 (1.9) | 743.2 (99.5) | 18.8 (8.1) | 0.8 (0.8) | 762.8 (101.1) | 68.4 (6.6) |
| 1.0 | 139.0 (66.6) | 724.2 (119.0) | 595.8 (312.8) | 26.6 (23.9) | 7.6 (5.5) | 630.0 (335.9) | 133.7 (49.6) |
| 1.8 | 58.0 (19.7) | 722.3 (86.9) | 265.6 (127.7) | 1.2 (1.2) | 0.0 (0.0) | 266.8 (127.9) | 68.0 (9.9) |
| 2.4 | 84.3 (62.6) | 757.8 (141.6) | 297.0 (109.5) | 2.3 (1.9) | 1.3 (0.9) | 300.5 (110.0) | 65.1 (23.0) |
| F1 | 1.7 | 1.1 | 2.1 | 0.8 | 1.1 | 2.1 | 1.1 |
Data shown are group means (±SEM) for vehicle (V) and each baclofen dose (mg/kg). FI, Fixed Interval; Resp, Response. Degrees of freedom were 5, 14 (Alcohol group) and 5, 19 (Tang group).
Total Responses=sum of the Left Lever Resp, Right Lever Resp, and Drink Contacts for each baboon.
An underlined F ratio indicates a significant (p<0.05) ANOVA, nc=not calculated.
Time to extinction in minutes.
In the alcohol group, the overall ANOVA revealed a significant main effect of baclofen on left lever responses and total seeking responses emitted during Link 2 of Component 2; although pairwise comparisons did not isolate a significant difference for specific doses when compared to vehicle (Bonferroni test; Table 2A). In the alcohol group, baclofen (1.8 mg/kg) significantly decreased overall response rate when compared to vehicle (mean ± SEM: 0.17 ± 0.06, 0.47 ± 0.14, respectively), but did not affect response latency in Link 2 of Component 2. These same doses of baclofen were also examined during within-session extinction tests in the Tang group. Baclofen did not significantly affect seeking responses, response rate, or response latency when doses were compared to vehicle (Table 2B).
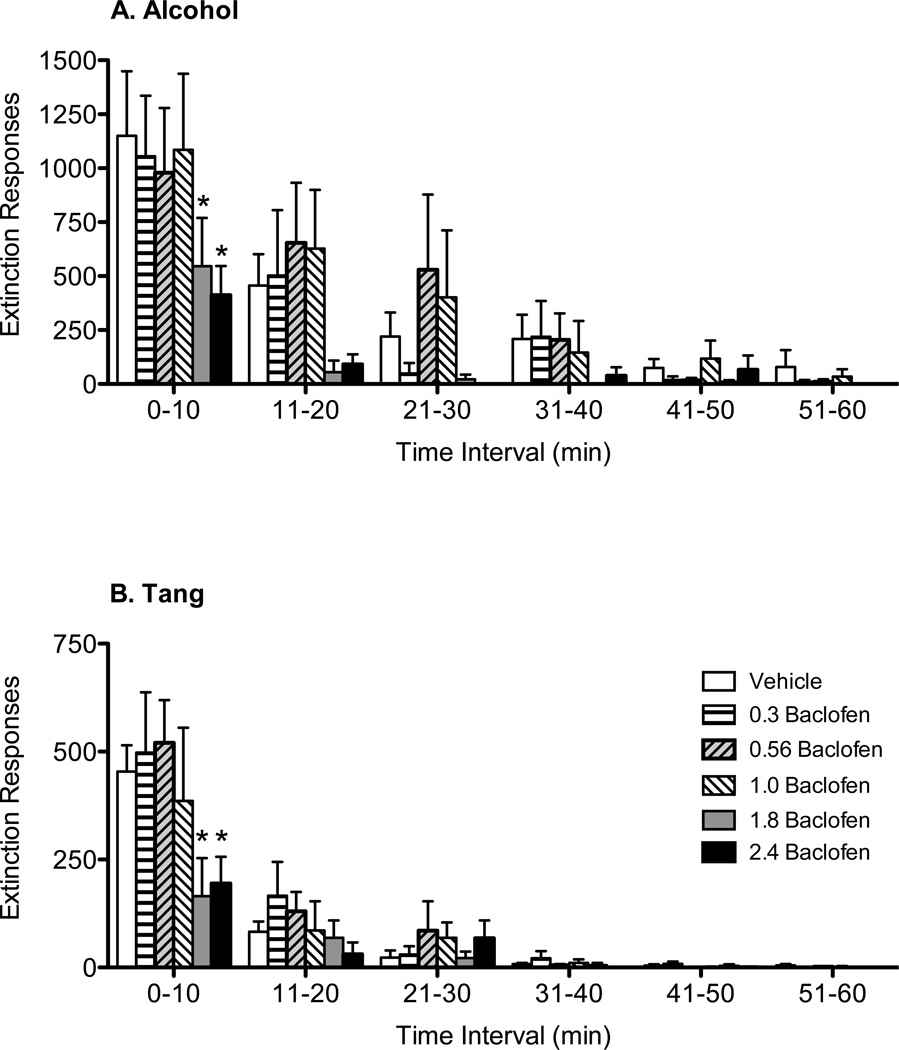
Baclofen reduced the total number of left lever responses during the within-session extinction tests in both alcohol and Tang groups, but this effect was not significant in the Tang group (Table 2). Similarly, the time to meet extinction criterion was variable in both groups, and baclofen did not significantly reduce time to meet extinction criterion (Table 2). However, since extinction responding is not constant over time, but progressively declines, we examined whether baclofen altered the pattern of responses. The number of responses was greatest in the first 10 min of extinction and progressively decreased over successive 10-min bins. As shown in Figure 2, baclofen (1.8–2.4 mg/kg) significantly decreased the number of extinction responses emitted on the left lever during the first 10 min bin in the alcohol and Tang groups compared to vehicle.
Figure 2.
Effects of baclofen on left lever responses emitted during Component 2, Link 2 of the within-session extinction tests for alcohol (A) and Tang (B). Data shown are mean (+ SEM) left lever responses in 10 min bins for the first 60 min of the within-session extinction test session. *p<0.05 for pairwise comparison for each dose vs. vehicle within each time bin.
DISCUSSION
Previous studies in our laboratory demonstrated that alcohol functioned as a reliable reinforcer (i.e., self-administered alcohol greater than its vehicle) under the CSR (Weerts et al., 2006; Kaminski et al., 2008). The current findings showed that baclofen produced biphasic effects on alcohol intake in baboons in the context of ongoing alcohol self-administration. Low baclofen doses (0.3–0.56 mg/kg) engendered moderate non-significant increases, while high dose (1.8–2.4 mg/kg) significantly decreased alcohol intake, lever responses, and drink contacts when compared to vehicle, suggesting an attenuation of the reinforcing effects of alcohol. While the majority of rodent studies have focused on baclofen-induced decreases in alcohol intake, the biphasic effects of baclofen on total alcohol consumed are consistent with some studies in rodents. Baclofen can both increase and decrease alcohol intake, depending on the dose of baclofen administered (Daoust et al., 1987; Smith et al., 1992; 1999; Colombo et al., 2000; Czachowski et al., 2006). The current study further augments rodent data with new data on the effects of baclofen on the pattern of alcohol self-administration across the session and on the patterning of extinction responses. Baclofen reduced total alcohol intake by decreasing the magnitude of the initial drinking bout. The highest dose of baclofen (2.4 mg/kg) tested also decreased Tang intake (ml). Similar to the alcohol group, these decreases occurred in the first drinking bout of the CSR session. These data suggest baclofen alters alcohol intake by reducing the first bout of drinking. With regard to total intake, baclofen may decrease alcohol and Tang similarly, but targeted doses of baclofen may be efficacious in suppressing alcohol consumption without affecting other appetitive behaviors (i.e., a narrow therapeutic window).
The current study has a number of strengths. First, we examined the effects of baclofen on a range of alcohol-related behaviors using a CSR in large nonhuman primates (Baboon, Papio anubis). Alcohol maintained extended sequences of behaviors to gain access to the daily supply of alcohol. These baboons were long-term heavy alcohol drinkers that consumed alcohol daily at levels we have shown sufficient to produce intoxication (i.e., BALs exceeding 0.08%; Kaminski et al., 2008). Second, the specificity of baclofen effects on alcohol behaviors were compared by testing the same doses of baclofen in a separate group of baboons with a similar history of drinking a preferred non-alcoholic beverage (Tang) under the CSR. Third, alcohol and Tang were established as reinforcers prior to baclofen testing. Fourth, alcohol and Tang self-administration stability criterion was reestablished before each test dose of baclofen.
The current study also assessed the effects of baclofen on extinction of conditioned seeking responses associated with alcohol or Tang consumption. Seeking responses in the CSR are strongly linked to alcohol drinking behaviors and the drinking environment, and are thus quite resistant to extinction. Baclofen facilitated extinction of seeking responses; however, decreases in responses observed in the early phase of extinction (first 10 min) at higher doses of baclofen were similar for both alcohol and Tang groups. The effects of baclofen on conditioned behaviors associated with alcohol have been examined in some rodent models. In the context of abstinence, baclofen attenuated cue-induced reinstatement of alcohol seeking behavior in alcohol-preferring rats (Maccioni et al., 2008), and modestly decreased alcohol seeking in an appetitive-consummatory operant procedure (Czachowski et al., 2006). Baclofen also produced a dose-dependent decrease in breaking points for alcohol (Walker and Koob, 2007), although alcohol-dependent rats showed greater sensitivity to baclofen (leftward shift in the dose effect curve), when compared to non-dependent rats. It has been suggested that baclofen may be most effective for blocking conditioned effects of alcohol. When injected into the VTA, baclofen blocked alcohol-induced conditioned place preference (Bechtholt and Cunningham, 2005) and cue-induced reinstatement of operant responding (Murschall and Hauber, 2006).
Studies examining the specificity of baclofen effects on alcohol self-administration and consumption when compared with those of a non-alcohol reinforcer or with water intake and have reported different results in various rodent models. For example, baclofen-induced changes in alcohol self-administration and consumption have been associated with increased total fluid intake (Smith et al., 1992), decreased sucrose or food intake (Petry, 1997; Smith et al., 1999; Anstrom et al., 2003; Janak and Gill, 2003; Maccioni et al., 2005; Czachowski et al., 2006; Tanchuck et al., 2011), and no change in total fluid intake (Daoust et al., 1987). In the present study, baclofen decreased alcohol seeking during within-session extinction tests, although the effects were not specific to alcohol. Similarly, studies have consistently reported that naltrexone, an FDA approved treatment to promote alcohol abstinence, reduces alcohol intake in animals and this reduction is frequently non-selective (Egli 2005).
Baclofen is safe and well tolerated in heavy drinkers and alcohol dependent subjects with liver damage (Addolorato et al., 2007; Evans and Bisaga, 2009; but see Addolorato et al., 2002; 2007 for side effects including headache, vertigo, and sedation), but treatment efficacy in clinical trials have been mixed. In two double blind placebo controlled clinical trials in Europe, baclofen was more effective than placebo for maintenance of total alcohol abstinence and reduction of craving during treatment (Addolorato et al., 2002; 2007). A third double blind placebo controlled trial in the US failed to find differences between baclofen (30 mg/d) and placebo-treated groups (Garbutt et al., 2010). The source of discrepancy between these studies in unclear, but the differences between the patient populations with regard to severity of alcohol dependence, treatment goals and concurrent behavioral therapy have been suggested.
In summary, this initial study in nonhuman primates showed that baclofen reduced both alcohol and Tang seeking and consumption, although baclofen effects on the pattern of drinking at lower doses differed for the two reinforcers. When examining drug effects, typically higher rate behavior is suppressed more readily than low rate behavior, so one would anticipate that the effects of baclofen on self-administration would be greater in the Tang group. This, however, was not the case, as baclofen reduced alcohol self-administration to an equal or greater extent when compared to Tang. Baclofen decreased alcohol intake (g/kg) at one dose that did not significantly affect Tang consumption, suggesting targeted doses of baclofen may be an efficacious treatment, but with a narrow therapeutic window. Baclofen also appeared to facilitate extinction of seeking responses, regardless of the reinforcer that first established the behavior. These initial results suggest that targeted doses of baclofen may be a useful treatment to promote abstinence and reduce heavy drinking if it occurs, but effects may not be specific to alcohol related behaviors.
ACKNOWLEDGEMENTS
This research was supported by NIH/NIAAA R01AA15971. The authors would like to thank Samuel Womack, Susan James and Emily Taylor for excellent technical assistance.
Footnotes
AUTHOR CONTRIBUTIONS
AND, BJK, and EMW contributed to the experimental design, data collection, analysis, and manuscript preparation. EMW secured grant funding for the data represented in the current manuscript. All authors critically reviewed content and approved the final version for publication.
DISCLOSURE/CONFLICT OF INTEREST
Dr. Weerts was a co-investigator on a contract in human subjects (A Phase 2 Study of LY2196044 Compared with Naltrexone and Placebo in the Treatment of Alcohol Dependence) funded by Lilly Research Laboratories. Drs. Duke and Kaminski have no conflicts to disclose.
REFERENCES
- Addolorato G, Caputo F, Capristo E, Colombo G, Gessa GL, Gasbarrini G. Ability of baclofen in reducing alcohol craving and intake: II-preliminary clinical evidence. Alcohol Clin Exp Res. 2000;24:67–71. [PubMed] [Google Scholar]
- Addolorato G, Caputo F, Capristo E, Domenicali M, Bernardi M, Janiri L, Agabio R, Colombo G, Gessa GL, Gasbarrini G. Baclofen efficacy in reducing alcohol craving and intake: a preliminary double-blind randomized controlled study. Alcohol Alcohol. 2002;37:504–508. doi: 10.1093/alcalc/37.5.504. [DOI] [PubMed] [Google Scholar]
- Addolorato G, Leggio L, Ferrulli A, Cardone S, Vonghia L, Mirjello A, Abenavoli L, D’Angelo C, Caputo F, Zambon A, Haber PS, Gasbarrini G. Effectiveness and safety of baclofen for maintenance of alcohol abstinence in alcohol-dependent patients with liver cirrhosis: randomized, double-blind controlled study. Lancet. 2007;370:1915–1922. doi: 10.1016/S0140-6736(07)61814-5. [DOI] [PubMed] [Google Scholar]
- Anstrom KK, Cromwell HC, Markowski T, Woodward DJ. Effect of baclofen on alcohol and sucrose self-administration in rats. Alcohol Clin Exp Res. 2003;27:900–908. doi: 10.1097/01.ALC.0000071744.78580.78. [DOI] [PubMed] [Google Scholar]
- Bechtholt AJ, Cunningham CL. Ethanol-induced conditioned place preference is expressed through a ventral tegmental area dependent mechanism. Beh Neurosci. 2005;119:213–223. doi: 10.1037/0735-7044.119.1.213. [DOI] [PubMed] [Google Scholar]
- Bullock DH. Repeated conditioning-extinction sessions as a function of the reinforcement schedule. J Exp Analysis Beh. 1960;3:241–243. doi: 10.1901/jeab.1960.3-241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bullock DH, Smith WC. An effect of repeated conditioning-extinction upon operant strength. J Exp Psych. 1953;46:349–352. doi: 10.1037/h0054544. [DOI] [PubMed] [Google Scholar]
- Clark GC, Taylor BW. Effects of repeated extinction of an operant on characteristics of extinction curves. Psych Rep. 1960;6:226. [Google Scholar]
- Colombo G, Agabio R, Carai MA, Lobina C, Pani M, Reali R, Addolorato G, Gessa GL. Ability of baclofen in reducing alcohol intake and withdrawal severity: I—Preclinical evidence. Alcohol Clin Exp Res. 2000;24:56–66. [PubMed] [Google Scholar]
- Colombo G, Serra S, Brunetti G, Atzori G, Pani M, Vacca G, Addolorato G, Froestl W, Carai MA, Gessa GL. The GABA(B) receptor agonists baclofen and CGP 44532 prevents acquisition of alcohol drinking behavior in alcohol-preferring rats. Alcohol Alcohol. 2002;37:499–503. doi: 10.1093/alcalc/37.5.499. [DOI] [PubMed] [Google Scholar]
- Czachowski CL, Legg BH, Stansfield KH. Ethanol and sucrose seeking and consumption following repeated administration of the GABA(B) agonist baclofen in rats. Alcohol Clin Exp Res. 2006;30:812–818. doi: 10.1111/j.1530-0277.2006.00094.x. [DOI] [PubMed] [Google Scholar]
- Daoust M, Saligaut C, Lhuintre JP, Moore N, Flipo JL, Boismare F. GABA transmission, but not benzodiazepine receptor stimulation, modulates ethanol intake by rats. Alcohol. 1987;4:469–472. doi: 10.1016/0741-8329(87)90087-5. [DOI] [PubMed] [Google Scholar]
- Doyon WM, Howard EC, Shippenberg TS, Gonzales RA. k-Opioid receptor modulation of accumbal dopamine concentration during operant ethanol self-administration. Neuropharmacology. 2006;51:487–496. doi: 10.1016/j.neuropharm.2006.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Egli M. Can experimental paradigms and animal models be used to discover clinically effective medications for alcoholism? Addict Biol. 2005;10:309–319. doi: 10.1080/13556210500314550. [DOI] [PubMed] [Google Scholar]
- Engleman EA, Keen EJ, Tilford SS, Thielen RJ, Morzorati SL. Ethanol drinking reduces extracellular dopamine levels in the posterior ventral tegmental erea of nondependent alcohol-preferring rats. Alcohol. 2011;45:549–557. doi: 10.1016/j.alcohol.2011.02.304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans SM, Bisaga A. Acute interaction of baclofen in combination with alcohol in heavy social drinkers. Alcohol Clin Exp Res. 2009;33:19–30. doi: 10.1111/j.1530-0277.2008.00805.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flannery BA, Garbutt JC, Cody MW, Renn W, Grace K, Osborne M, Crosby K, Morreale M, Trivette A. Baclofen for alcohol dependence: a preliminary open-label study. Alcohol Clin Exp Res. 2004;28:1517–1523. doi: 10.1097/01.alc.0000141640.48924.14. [DOI] [PubMed] [Google Scholar]
- Garbutt JC, Kampov-Polevoy AB, Gallop R, Kalka-Juhl L, Flannery BA. Efficacy and safety of baclofen for alcohol dependence: a randomized, double-blind, placebo-controlled trial. Alcohol Clin Exp Res. 2010;34:1–9. doi: 10.1111/j.1530-0277.2010.01273.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths RR, Lamb RJ, Sannerud CA, Ator NA, Brady JV. Self-injection of barbiturates, benzodiazepines and other sedative-anxiolytics in baboons. Psychopharmacology. 1991;103:154–161. doi: 10.1007/BF02244196. [DOI] [PubMed] [Google Scholar]
- Hasin DS, Stinson FS, Ogburn E, Grant BF. Prevalence, correlates, disability, and comorbidity of DSM-IV alcohol abuse and dependence in the United States. Arch Gen Psychiatry. 2007;64:830–842. doi: 10.1001/archpsyc.64.7.830. [DOI] [PubMed] [Google Scholar]
- Heilig M, Egli M. Pharmacological treatment of alcohol dependence: target symptoms and target mechanisms. Pharmacol Ther. 2006;111:855–876. doi: 10.1016/j.pharmthera.2006.02.001. [DOI] [PubMed] [Google Scholar]
- Janak PH, Gill TM. Comparison of the effects of allopregnanolone with direct GABAergic agonists on ethanol self-administration with and without concurrently available sucrose. Alcohol. 2003;30:1–7. doi: 10.1016/s0741-8329(03)00068-5. [DOI] [PubMed] [Google Scholar]
- Kaminski BJ, Goodwin AK, Wand G, Weerts EM. Dissociation of alcohol-seeking and consumption under a chained schedule of oral alcohol reinforcement in baboons. Alcohol Clin Exp Res. 2008;32:1014–1022. doi: 10.1111/j.1530-0277.2008.00662.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang JH, Ghen F, Krstew E, Cowen MS, Carroll FY, Crawford D, Beart PM, Laurence AJ. The GABA(B) receptor aoolosteric modulator CGP7930, like baclofen, reduces operant self-administration of ethanol in alcohol-perferring rats. Neuropharmacology. 2006;50:632–639. doi: 10.1016/j.neuropharm.2005.11.011. [DOI] [PubMed] [Google Scholar]
- Leggio L, Garbutt JC, Addolorato G. Effectiveness and safety of baclofen in the treatment of alcohol dependent patients. CNS Neurol Disord Drug Targets. 2010;9:33–44. doi: 10.2174/187152710790966614. [DOI] [PubMed] [Google Scholar]
- Maccioni P, Serra S, Vacca G, Orru A, Pes D, Agabio R, Addolorato G, Carai MA, Gessa G, Colombo G. Baclofen-induced reduction of alcohol reinforcement in alcohol-preferring rats. Alcohol. 2005;36:161–168. doi: 10.1016/j.alcohol.2005.08.003. [DOI] [PubMed] [Google Scholar]
- Maccioni P, Bienkowski P, Carai MA, Gessa GL, Colombo G. Baclofen attenuates cue-induced reinstatement of alcohol-seeking behavior in Sardinian alcohol-preferring (sP) rats. Drug Alcohol Depend. 2008;95:284–287. doi: 10.1016/j.drugalcdep.2008.02.006. [DOI] [PubMed] [Google Scholar]
- Murschall A, Hauber W. Inactivation of the ventral tegmental area abolished the general excitatory influence of Pavlovian cues on instrumental performance. Learn Mem. 2006;13:123–126. doi: 10.1101/lm.127106. [DOI] [PubMed] [Google Scholar]
- Petry NM. Benzodiazepine-GABA modulation of concurrent ethanol and sucrose reinforcement in the rat. Exp Clin Psychopharmacol. 1997;5:183–194. [PubMed] [Google Scholar]
- Smith BR, Robidoux J, Amit Z. GABAergic involvement in the acquisition of voluntary ethanol intake in laboratory rats. Alcohol Alcohol. 1992;27:227–231. [PubMed] [Google Scholar]
- Smith BR, Boyle AE, Amit Z. The effects of the GABA(B) agonist baclofen on the temporal and structural characteristics of ethanol intake. Alcohol. 1999;17:231–240. doi: 10.1016/s0741-8329(98)00053-6. [DOI] [PubMed] [Google Scholar]
- Soyka M, Rosner S. Emerging drugs to treat alcoholism. Expert Opin Emerg Drugs. 2010;15:695–711. doi: 10.1517/14728214.2010.500811. [DOI] [PubMed] [Google Scholar]
- Swift R. Medications acting on the dopaminergic system in the treatment of alcoholic patients. Curr Pharm Des. 2010;16(19):2136–2140. doi: 10.2174/138161210791516323. [DOI] [PubMed] [Google Scholar]
- Tanchuck MA, Yoneyama N, Ford MM, Fretwell AM, Finn DA. Assessment of GABA-B, metabotropic glutamate, and opioid receptor involvement in an animal model of binge drinking. Alcohol. 2011;45:33–44. doi: 10.1016/j.alcohol.2010.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Theile JW, Morikawa H, Gonzales RA, Morrisett RA. Ethanol enhances GABAergic transmission onto dopamine neurons in the ventral tegmental area of the rat. Alcohol Clin Exp Res. 2008;32(6):1040–1048. doi: 10.1111/j.1530-0277.2008.00665.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker BM, Koob GF. The gamma-aminobutyric acid-B receptor agonist baclofen attenuates responding for ethanol in ethanol-dependent rats. Alcohol Clin Exp Res. 2007;31:11–18. doi: 10.1111/j.1530-0277.2006.00259.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weerts EM, Froestl W, Griffiths RR. Effects of GABAergic modulators on food and cocaine self-administration in baboons. Drug Alcohol Depend. 2005;80:369–376. doi: 10.1016/j.drugalcdep.2005.05.006. [DOI] [PubMed] [Google Scholar]
- Weerts EM, Goodwin AK, Kaminski BJ, Hienz RD. Environmental cues, alcohol seeking, and consumption in baboons: effects of response requirement and duration of alcohol abstinence. Alcohol Clin Exp Res. 2006;30:2026–2036. doi: 10.1111/j.1530-0277.2006.00249.x. [DOI] [PubMed] [Google Scholar]
- Weerts EM, Froestl W, Kaminski BJ, Griffiths RR. Attenuation of cocaine-seeking by GABA B receptor agonists baclofen and CGP44532 but not the GABA reuptake inhibitor tiagabine in baboons. Drug Alcohol Depend. 2007;89:203–213. doi: 10.1016/j.drugalcdep.2006.12.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weerts EM, Duke AN, Kaminski BJ. Pattern of drinking and resistance to extinction of conditioned responses associated with alcohol vs. a flavored non-alcoholic drink. submitted. [Google Scholar]
- Zarcone TJ, Ator NA. Drug discrimination: Stimulus control during repeated testing in extinction. J Exp Analysis Beh. 2000;74:283–294. doi: 10.1901/jeab.2000.74-283. [DOI] [PMC free article] [PubMed] [Google Scholar]